P53 mutations occur in more than half of all human cancers. Mutant p53 proteins are mostly full-length with a single residue change, remarkably stable and abundantly present in tumor cells. Mutant p53 proteins do not represent only the mere loss of wt-p53 activity, but also the gain of oncogenic functions that strongly contribute to the establishment, the maintenance and the spreading of a given tumor. The last decade has witnessed remarkable experimental efforts to elucidate the molecular mechanisms underlying the oncogenic role of mutant p53 proteins. To date, two main molecular scenarios are intensively investigated: (1) mutant p53 protein can function as an oncogenic transcription factor as a component of large transcriptional competent protein complexes that also include transcription factors, acetylases, deacetylases and scaffold proteins.1,2 This leads to the aberrant expression of distinct sets of genes; (2) mutant p53 protein physically interacts with and inactivates tumor suppressor gene products, which are exemplified by the p53 family members p73 and p63.3 Previous work from the Haupt’s group showed that PML interacts with and enhances mutant p53 transcriptional activity (Fig. 1A).4 PML was required for aberrant proliferation and colony-forming ability of mutant p53 cancer cells.4 This, intriguingly, highlighted that, unlike the case of p73 and p63 proteins, which are bound and inactivated by mutant p53, binding to PML allows mutant p53 to parasitize PML activities and consequently enhance its pro-tumorigenic effects (Fig. 1A). Thus, PML nuclear bodies might endow mutant p53 proteins with gain of function activity. In this issue of Cell Cycle, Sue Haupt and colleagues provide novel in vivo evidence, which further strengthens the concept of a close relationship existing between gain of function mutant p53 protein and the tumor suppressor protein PML.5 They elegantly investigated the contribution of PML to mutant p53-driven tumorigenesis in a mouse model harboring a p53 mutation (p53wild-type/R172H) that recapitulates a frequent p53 mutation (p53R175H) in human sporadic and Li-Fraumeni cancers. First, loss of PML expression unleashed a pro-tumorigenic response, as manifested by increased protein levels of p19ARF, c-MYC and mutant p53 (Fig. 1B). Second, loss of PML expression dictated tumor manifestation of p53wild-type/R172H mice reducing the incidence of lymphomas and increasing the frequency of sarcomas (Fig. 1B). Third, PML loss in male mutant p53 mice led to reduced survival when compared with that of females (Fig. 1B). These findings have several implications. Previous work has shown that mutant p53 protein is unstable in untransformed tissues.6 The transformed cellular context plays a pivotal role in the accumulation of mutant p53 proteins. Haupt’s findings indicate that loss of PML expression might represent a major oncogenic event, leading to accumulation of mutant p53 protein and thereby enhancing its oncogenic effects.5 The fine deciphering of mutant p53 accumulation upon loss of PML might unveil molecular targets whose aberrant activity could be pharmacologically tackled either to attenuate its oncogenic effects, through the reduction of its half-life, or to promote anti-tumoral effects, by switching its conformation to that of wt-p53 protein. Mutant p53 has been shown to interact with diverse transcription factors, ranging from NF-Y and VDR to ETS1, SP1, ZEB-1 and E2F-1.1,2,7,8 It’s reasonable to speculate that each of these protein complexes might drive the aberrant expression of selected genes whose protein products drive oncogenesis of specific subsets of human tumors. This may explain why loss of PML through the re-wiring of the network of mutant p53 protein interactions determined a switch in the tumor manifestation of p53 mutant mice. Gender appears to be an additional layer of complexity that can have a profound impact in the development of specific subsets of human tumors. Although further work is required to corroborate Sue Haupt’s findings, loss of PML expression emerges to be a candidate factor for determining gender-related cancer disease. Mutant p53 mice with PML exhibited perturbation of the hematopoietic compartment manifested as lymphomas or extramedullary hematopoiesis (EMH). EMH was associated with leucocytosis and macrocytic anemia indicative of myeloprolferative/myelodisplastic overlap. Thus, EMH underlines a molecular lesion that may provide an early prediction for the development of hematopoietic tumors in the context of mutant p53 mice.
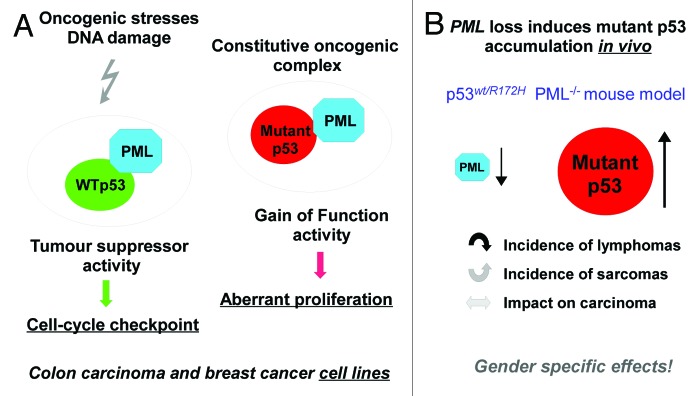
Figure 1. Cross-talk between PML and mutant p53 proteins. (A) Gain-of-function mutant p53 proteins parasite PML tumor suppressor activity and enhance their pro-tumorigenic effects. (B) Loss of PML expression accumulates mutant p53 protein and dictates gender-associated tumor spectrum.
Collectively, a functional cross-talk between gain-of-function mutant p53 proteins and PML exists; the fine molecular dissection might hold great therapeutic potential for the treatment of mutant p53-driven tumors.
Footnotes
Previously published online: www.landesbioscience.com/journals/cc/article/25174
References
- 1.Di Agostino S, Strano S, Emiliozzi V, Zerbini V, Mottolese M, Sacchi A, et al. Gain of function of mutant p53: the mutant p53/NF-Y protein complex reveals an aberrant transcriptional mechanism of cell cycle regulation. Cancer Cell. 2006;10:191–202. doi: 10.1016/j.ccr.2006.08.013. [DOI] [PubMed] [Google Scholar]
- 2.Fontemaggi G, Dell’Orso S, Trisciuoglio D, Shay T, Melucci E, Fazi F, et al. The execution of the transcriptional axis mutant p53, E2F1 and ID4 promotes tumor neo-angiogenesis. Nat Struct Mol Biol. 2009;16:1086–93. doi: 10.1038/nsmb.1669. [DOI] [PubMed] [Google Scholar]
- 3.Di Agostino S, Cortese G, Monti O, Dell’Orso S, Sacchi A, Eisenstein M, et al. The disruption of the protein complex mutantp53/p73 increases selectively the response of tumor cells to anticancer drugs. Cell Cycle. 2008;7:3440–7. doi: 10.4161/cc.7.21.6995. [DOI] [PubMed] [Google Scholar]
- 4.Haupt S, di Agostino S, Mizrahi I, Alsheich-Bartok O, Voorhoeve M, Damalas A, et al. Promyelocytic leukemia protein is required for gain of function by mutant p53. Cancer Res. 2009;69:4818–26. doi: 10.1158/0008-5472.CAN-08-4010. [DOI] [PubMed] [Google Scholar]
- 5.Haupt S, Mitchell C, Corneille V, Shortt J, Fox S, Pandolfi PP, et al. Loss of PML cooperates with mutant p53 to drive more aggressive cancers in a gender-dependent manner. Cell Cycle. 2013;12 doi: 10.4161/cc.24805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lozano G. The oncogenic roles of p53 mutants in mouse models. Curr Opin Genet Dev. 2007;17:66–70. doi: 10.1016/j.gde.2006.12.003. [DOI] [PubMed] [Google Scholar]
- 7.Stambolsky P, Tabach Y, Fontemaggi G, Weisz L, Maor-Aloni R, Siegfried Z, et al. Modulation of the vitamin D3 response by cancer-associated mutant p53. Cancer Cell. 2010;17:273–85. doi: 10.1016/j.ccr.2009.11.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Masciarelli S, Fontemaggi G, Di Agostino S, Donzelli S, Carcarino E, Strano S, et al. Gain-of-function mutant p53 downregulates miR-223 contributing to chemoresistance of cultured tumor cells. Oncogene. 2013 doi: 10.1038/onc.2013.106. [DOI] [PubMed] [Google Scholar]


