Abstract
We present a challenging case in which the dermatomal pain associated with herpes zoster ophthalmicus preceded the cutaneous rash by several days. It thus highlights the need to consider this diagnosis among the differentials for severe unilateral headache in the elderly. The patient unfortunately progressed to develop encephalitis, an uncommon but serious complication of zoster reactivation and a reminder that this remains an important cause of morbidity and mortality in the elderly population.
Background
Herpes zoster reactivation is more common in the elderly population, resulting from a decline in cell-mediated immunity. The incidence in those >65 years of age is thought to be in the range of 3.9–11.8 cases/1000 person years.1
We present a challenging case in which the dermatomal pain associated with herpes zoster ophthalmicus (HZO) preceded the cutaneous rash by several days. It highlights a serious complication of this condition and is an important reminder that HZO should be considered among the differentials for severe unilateral headache in the elderly. It also highlights a potential pitfall of attributing confusion in the elderly to more ‘common’ infections, such as urinary tract infection (UTI), in the absence of supporting symptomatology.
Case presentation
An 80-year-old female patient was referred to the medical assessment unit (MAU) by her general practitioner. She presented with a 1-day history of transient frontal headache of insidious onset, associated with vomiting and photophobia.
There was a history of hypothyroidism, type 2 diabetes mellitus and rheumatoid arthritis, the latter treated with weekly methotrexate therapy. The patient had chronic obstructive airways disease and also a prior history of a pneumonectomy during childhood. Persistent atrial fibrillation was rate-controlled with digoxin, with aspirin for stroke prevention.
Upon assessment on MAU, the patient was reportedly ‘confused’, though a mental test score was not obtained. There was evidence of subtle right-sided facial nerve weakness and slurred speech. She was afebrile and haemodynamically stable. There were no features of meningism. She had symptoms of frontal headache, with right-sided preponderance.
Initial investigations undertaken revealed a normocytic anaemia (haemoglobin 10.8 g/dL (11.5–16.50)) without leucocytosis or change in inflammatory markers (C reactive protein <5 mg/L (0–10)). Biochemistry revealed a normal renal function with hyponatraemia (sodium 128 mmol/L (134–145)). Chest X-ray showed no acute changes. ECG confirmed atrial fibrillation.
Collateral history subsequently revealed that the patient had sustained a fall at home 3 days prior to admission. A CT head was performed, but did not identify any acute intracranial haemorrhage or territorial infarct.
The patient was treated for a presumed UTI in the absence of associated symptoms. Her urine dipstick was positive for protein, leucocytes and nitrites. Following transfer to an acute geriatric ward, a single febrile episode occurred (38.3°C) with associated subjective deterioration. Oral antibiotic therapy was changed to intravenous co-amoxiclav under the assumption of developing sepsis.
Seventy-two hours into her admission the patient began to report severe right eye pain, radiating to the right forehead. Examination confirmed a tender globe with minimal conjunctival injection. Both temporal arteries were readily palpable. An urgent ophthalmic opinion was sought, which excluded acute angle closure glaucoma. MRI of the head was performed, including diffusion-weighted imaging and MR angiography. This identified an acute small right-sided parietal subcortical infarct in addition to moderate small vessel ischaemic change (figure 1). There was no evidence of arterial dissection. Appropriate analgesia was prescribed and her aspirin and antibiotics continued.
Figure 1.
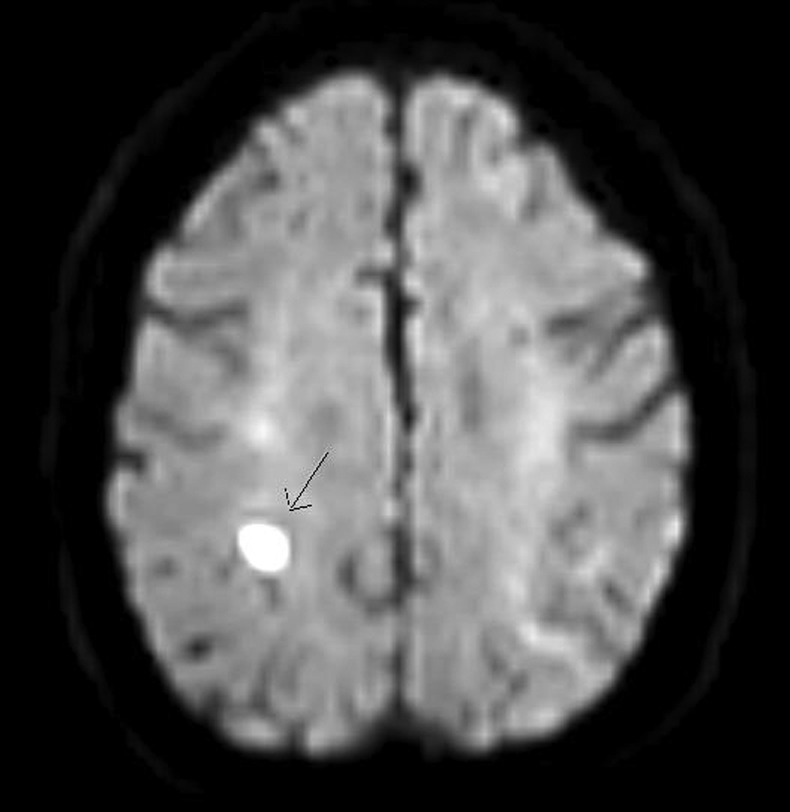
MRI (DWI) showing an acute small right parietal subcortical infarct.
Review on the fifth day of admission revealed no significant improvement. Both urine and blood cultures were negative and she had experienced no further febrile episodes. However, she was now drowsy (Glasgow Coma Scale (GCS) 13/15; E3 M6 V4) exhibiting marked periorbital oedema of the right eye with emerging evidence of crusting lesions to the upper eyelid and scalp. A diagnosis of HZO was suspected and intravenous aciclovir (10 mg/kg thrice daily) and flucloxacillin were immediately started. A further ophthalmic opinion corroborated a clinical diagnosis of HZO with no associated uveitis.
Outcome and follow-up
Over the next 24 h the patient continued to deteriorate rapidly, with decreasing conscious level (GCS 9/15) and evidence of haemodynamic compromise and hypoxaemia. Examination revealed right basal crepitations and a working diagnosis of aspiration pneumonia, complicating possible viral encephalitis, was made.
An urgent head CT was performed, but this showed no significant change from previous imaging (figure 2). A lumbar puncture was also performed and cerebrospinal fluid (CSF) analysis revealed a raised protein level (617 mg/L (150–450)), normal glucose (3.1 mmol/L (2.8–4.4)), 10 red blood cells and no white blood cells; a sample was also obtained for viral PCR analysis. Unfortunately the patient succumbed to her illness 7 days into her admission despite treatment.
Figure 2.
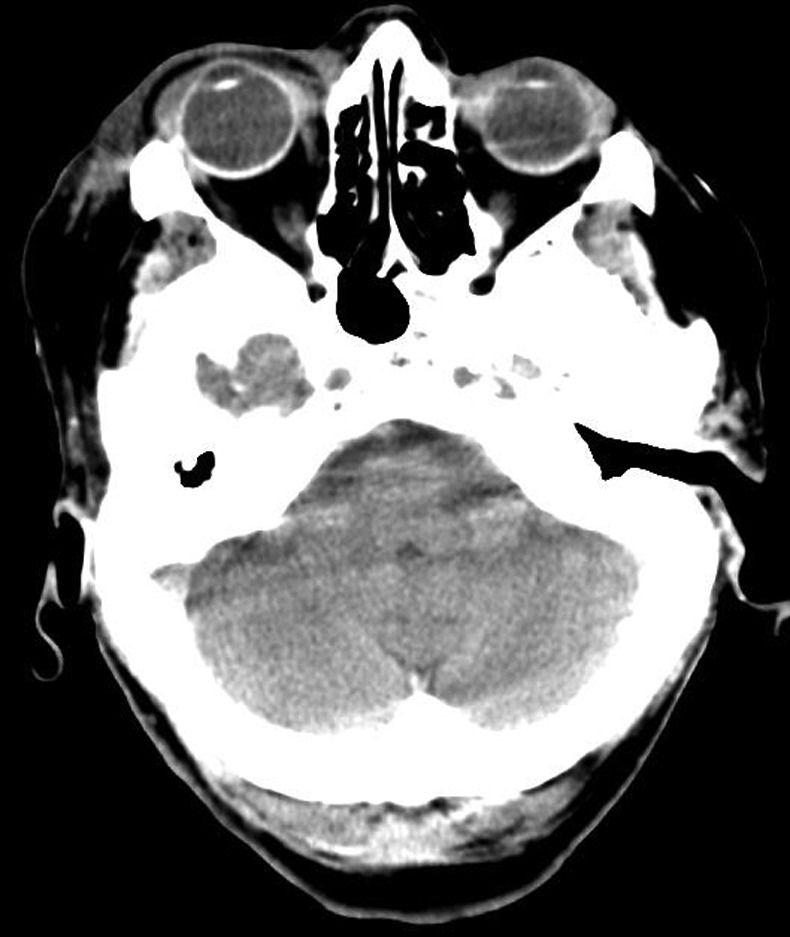
CT head showing significant right periorbital swelling.
Subsequent to the patient's death, the results of the viral PCR revealed the presence of herpes zoster virus within the CSF. The diagnosis of herpes zoster encephalitis (HZE) was therefore confirmed.
Discussion
This case highlights an uncommon yet important cause of encephalitis in the elderly population. Encephalitis is a rare complication of zoster infection occurring in less than 0.1%. Most cases have been reported in patients who are elderly and/or immunocompromised either through concurrent disease or drug therapy.2 The condition usually presents with a subacute delirium, occurring within a few days of the appearance of the typical herpetiform rash. However, it is important to note that the condition can occur in the absence of cutaneous manifestations.1 There may also be absence of fever, or CSF pleocytosis. Conversely, CSF lymphocytic pleocytosis, raised protein and positive PCR for varicella zoster virus (VZV) can accompany uncomplicated acute herpes zoster infection, making diagnosis of HZE even more challenging and necessitating a high index of suspicion. PCR is nonetheless helpful in supporting a diagnosis of HZE, with a high reported sensitivity (80–95%) and specificity (>95%) in immunocompromised patients with central nervous system symptoms.3–5 MRI is used predominantly to exclude other differentials, but can show subcortical non-enhancing spherical lesions that typically represent infarcts or demyelination (figure 1).2 3 6 This itself relates to direct transneuronal and haematogenous spread of VZV to leptomeningeal arteries resulting in a necrotising angiitis.1 7
The effectiveness of intravenous aciclovir in HZE remains unclear; 14 days of this treatment is considered gold standard.3 8 A short course of steroids can also be given in view of the inflammatory aetiology, but again evidence is rather limited.8 In general, outcome is favourable with the majority of cases returning to near normal cognitive function.2 Nonetheless, mortality stands in the order of 10%.1
The apparent predilection of zoster encephalitis among the frail elderly does perhaps support a low threshold for the use of intravenous aciclovir therapy in patients presenting with HZO, especially in those with new confusion and in the presence of ‘immune-modulating’ medication or diseases.9
The use of a live attenuated vaccine for herpes zoster has been shown in observational studies to reduce the incidence of infection and associated hospitalisation in community dwelling immunocompetent adults over the age of 60 (HR 0.45, 95% CI 0.42 to 0.48). Subgroup analysis revealed that the risk reduction was maintained in all age groups including the very elderly (HR 0.48, 95% CI 0.38 to 0.61).10
The Joint Committee on Vaccination and Immunisation in the UK has recommended a universal vaccination programme in the 70–79-year olds.11 Although this is subject to the availability of the vaccine at cost-effective price, it is expected that this will become part of the national vaccination programme in the next few years.
Another important discussion point in this case, is whether a lumbar puncture should have been performed earlier to rule out the possibility of intracerebral infection, looking mainly for herpes simplex virus encephalitis (HSVE) rather than HZE. In terms of the cardinal features of encephalitis, that is, fever and altered mental status, this patient was relatively late to present with fever. In a retrospective review of 184 patients with HSVE, the absence of fever was associated with late initiation of aciclovir (on univariate analysis).12 Fifteen per cent of patients in this series did not present with fever at admission. The presence of severe underlying disease (comorbidity) was also associated with delay to treatment.12 A compounding factor in this case was the interpretation of possible new neurology in the context of a recent fall. An MRI performed within 48 h of hospital admission is abnormal in approximately 90% of cases of HSVE.13 14 The features of a vasculopathy in HZE predominate on MRI over the temporal lobe oedema typically observed in the more common HSVE,1 6 and thereby perhaps gave us false reassurance in this case that we were not dealing with a picture of encephalitis. The presence of acellular CSF is another recognised delay to initiation of aciclovir;12 though this had little bearing in our case as aciclovir had already been started for HZO. It is not uncommon (5–10% of cases) for initial CSF findings to be normal (including negative PCR) in patients with HSVE, only to have abnormal CSF 24–48 h later.7 14 15 This is particularly true in immunocompromised patients.14 15 The current UK guidance is that lumbar puncture must be performed as soon as encephalitis is suspected.8 This is certainly true, but many elderly patients with delirium and fever have an associated septic encephalopathy that closely mirrors this picture.8 16 If we were to lumbar puncture all such patients where the source of infection was initially unclear, the proportion found to have abnormal CSF would undoubtedly be very low. There are also practical challenges of performing lumbar puncture safely in an agitated delirious patient. Nonetheless, encephalitis is a diagnosis that should not be missed and indeed delays in initiation of aciclovir in HSVE by >2 days is independently associated with poorer outcomes at 6 months.14 Perhaps a sensible approach is to first establish whether there is indeed a credible alternative source of infection; if at time of admission this is not forthcoming, aciclovir could be given with the knowledge that CSF PCR for HSVE obtained between day 2 and 10 of illness has a sensitivity of >95% and that patients will still often be PCR positive even after starting antiviral therapy.8 14
Learning points.
Be wary of attributing confusion in the elderly febrile patient to a urinary tract infection; especially in the absence of associated symptomatology.
Ideally, assuming no contraindication, lumbar puncture should be performed as soon as a diagnosis of encephalitis is suspected. However, the window of opportunity to do so and still be meaningful can extend beyond the first 48 h of admission.
Herpes zoster ophthalmicus (HZO) should be considered among the differential diagnoses of unilateral frontal headache in the elderly.
The dermatomal pain from acute zoster infection can precede the development of a rash by several days. On rare occasions, a rash may not develop at all.
Consider herpes zoster encephalitis in older patients with acute zoster infection who subsequently develop delirium/changes in mental status. Be particularly wary in patients with HZO and/or those receiving immunsuppressive agents.
A universal varicella vaccination programme for 70–79 year olds is to be introduced in UK in the next few years.
Footnotes
Contributors: The article is written by NN and PM; edited by AB. Case was identified by AB and originally managed by NN and AB. NN is the guarantor.
Competing interests: None.
Patient consent: Obtained.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1.Dworkin RH, Johnson RW, Breuer J, et al. Recommendations for the management of Herpes zoster. Clin Infect Dis 2007;2013:S1–26 [DOI] [PubMed] [Google Scholar]
- 2.Espiritu R, Rich M. Herpes zoster encephalitis: 2 case reports and review of the literature. Infect Dis Clin Pract 2007;2013:284–8 [Google Scholar]
- 3.Tunkel AR, Carol A, Glaser KC, et al. The management of encephalitis: clinical practice guidelines by the Infectious Diseases Society America. Clin Infect Dis 2008;2013:303–27 [DOI] [PubMed] [Google Scholar]
- 4.DeBiasi RL, Tyler KL. Polymerase chain reaction in the diagnosis and management of central nervous system infections. Arch Neurol 1999;2013:1215–19 [DOI] [PubMed] [Google Scholar]
- 5.DeBiasi RL, Tyler KL. Molecular methods for diagnosis of viral encephalitis. Clin Microbiol Rev 2004;2013:903–25 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Haanpa M, Dastidar P, Weinberg A, et al. CSF and MRI findings in patients with acute herpes zoster. Neurology 1998;2013:1405–11 [DOI] [PubMed] [Google Scholar]
- 7.Aygun N, Finelli DA, Rodgers MS, et al. Multifocal varicella zoster virus leukoencephalitis in a patient with AIDS: MR findings. AJNR Am J Neuroradiol 1998;19:1897–9 [PMC free article] [PubMed] [Google Scholar]
- 8.Solomon T, Michael BD, Smith PE, et al. Management of suspected viral encephalitis in adults—Association of British Neurologists and British Infection Association National Guidelines. J Infect 2012;2013:347–73 [DOI] [PubMed] [Google Scholar]
- 9.Lyon CC, Thompson D. Herpes zoster encephalomyelitis associated with low dose methotrexate for rheumatoid arthritis. J Rhematol 1997;2013:589–91 [PubMed] [Google Scholar]
- 10.Tseng HF, Smith N, Harpaz R. Herpes zoster vaccine in older adults and the risk of subsequent herpes zoster disease. JAMA 2011;2013:160–6 [DOI] [PubMed] [Google Scholar]
- 11.Joint Committee on Vaccination and Immunisation Statement on varicella and herpes zoster vaccines [updated April 2011]. http://www.dh.gov.uk/prod_consum_dh/groups/dh_digitalassets/@dh/@ab/documents/digitalasset/dh_133599.pdf (accessed 25 May 2012).
- 12.Poissy J, Wolff M, Dewilde A, et al. Factors associated with delay to acyclovir administration in 184 patients with herpes simplex virus encephalitis. Clin Microbiol Infect 2009;2013:560–4 [DOI] [PubMed] [Google Scholar]
- 13.Hollinger P, Matter L, Sturzenegger M. Normal MRI findings in herpes simplex virus encephalitis. J Neurol 2000;2013:799–801 [DOI] [PubMed] [Google Scholar]
- 14.Tyler KL. Update on herpes simplex encephalitis. Rev Neurol Dis 2004;2013:169–78 [PubMed] [Google Scholar]
- 15.Raschilas F, Wolff M, Delatour F, et al. Outcome of and prognostic factors for herpes simplex encephalitis in adult patients: results of multicenter study. Clin Infect Dis 2002;2013:254–60 [DOI] [PubMed] [Google Scholar]
- 16.Ringer TM, Axer H, Bernd FM, et al. Neurological sequelae of sepsis: (1) septic encephalopathy. Open Crit Care Med J 2011;2013:2–7 http://www.benthamscience.com/open/toccmj/articles/V004/SI0001TOCCMJ/2TOCCMJ.pdf (accessed 15 Apr 2013). [Google Scholar]


