Abstract
A small subset of mammary tumor initiating cells (also known as breast cancer stem cells; bCSC), characterized by expression of different markers [CD44high/CD24low/epithelial specific antigen (ESA)+], aldehyde dehydrogenase-1 (ALDH1) activity, and ability to form mammospheres under ultra-low attachment culture conditions, are suspected to evade conventional therapies leading to disease recurrence. Elimination of both therapy-sensitive epithelial tumor cells and therapy-resistant bCSC is therefore necessary for prevention of breast cancer. We have shown previously that a nontoxic small-molecule constituent of edible cruciferous vegetables (benzyl isothiocyanate; BITC) inhibits mammary cancer development in mouse mammary tumor virus-neu (MMTV-neu) transgenic mice by causing epithelial tumor cell apoptosis. The present study demonstrates efficacy of BITC against bCSC in vitro and in vivo. Mammosphere formation frequency and CD44high/CD24low/ESA+ and/or ALDH1+ populations in cultured MCF-7 (estrogen-receptor positive) and SUM159 (triple-negative) human breast cancer cells were decreased significantly in the presence of plasma achievable concentrations of BITC. BITC administration in the diet (3 μmol BITC/g diet for 29 weeks) resulted in a marked decrease in bCSC in the MMTV-neu mice tumors in vivo. Overexpression of full-length Ron as well as its truncated form (sfRon), but not urokinase-type plasminogen activator receptor, conferred near complete protection against BITC-mediated inhibition of bCSC in MCF-7 cells. The BITC treatment downregulated protein levels of Ron and sfRon in cultured breast cancer cells and in tumor xenografts. Ron overexpression resulted in up-regulation of bCSC-associated genes Oct-4, SOX-2, and Nanog. In conclusion, the present study indicates that BITC treatment eliminates bCSC in vitro and in vivo.
Keywords: Benzyl isothiocyanate, Breast cancer stem cells, Ron, Chemoprevention
Introduction
Breast cancer remains a major public health concern for women worldwide accounting for over 40,000 deaths each year in the United States alone despite tremendous advances toward targeted therapies (1,2). Preventive strategies, as exemplified by selective estrogen-receptor (ER) modulators (e.g., tamoxifen and raloxifene) and aromatase inhibitors (e.g., exemestane) for breast cancer risk reduction (3–5), are necessary to alleviate disease-related cost, morbidity, and mortality associated with this disease. Unfortunately, the currently available preventive options are ineffective against ER-negative breast cancers as well as a subset of women with ER-positive cancer (3–5). Unwanted side effects associated with the long-term use of selective ER modulators and aromatase inhibitors further underscores the need for novel cancer preventive options that are not only safe but can also target ER-positive as well as ER-negative breast cancers.
Bioactive small-molecules from edible plants remain attractive for prevention of breast and other cancers due to their favorable safety profile (6,7). Benzyl isothiocyanate (BITC) is one such naturally-occurring phytochemical in cruciferous vegetables (e.g., garden cress) with in vivo preventive efficacy in experimental animals (8–10). For example, BITC treatment prior to the carcinogen challenge inhibited polycyclic aromatic hydrocarbon-induced mammary cancer in rats (8). Previous work from our own laboratory has revealed that BITC administration in the diet confers significant protection against ER-negative breast cancer development in mouse mammary tumor virus-neu (MMTV-neu) transgenic mice without any signs of overt toxicity (9). The growth of human breast cancer cells (MDA-MB-231) implanted in athymic mice is retarded markedly after treatment with BITC (10).
BITC is structurally quite simple but elicits a complex set of signaling events leading to apoptotic as well as autophagic cell death in breast cancer cells (11–14). The molecular circuitry of BITC-induced cell death involves inhibition of complex III of the mitochondrial respiration and ensuing production of reactive oxygen species (11–13). Remarkably, a normal human mammary epithelial cell line (MCF-10A) is significantly more resistant to apoptotic and autophagic cell death as well as production of reactive oxygen species resulting from BITC exposure compared with cancerous mammary cells potentially contributing to its safety profile (11–13). The BITC-induced apoptosis and autophagy are not confined to cellular models as these effects are observed in epithelial mammary tumor cells in vivo (9,13).
The current paradigm insists on elimination of both therapy-sensitive epithelial tumor cells as well as therapy-insensitive breast cancer stem cells (bCSC) for prevention and treatment of this disease (15,16). The bCSC are not only intrinsically resistant to therapies but also have the ability to self-renew and enrich after treatment leading to tumor recurrence (15–17). Evidence continues to accumulate to indicate that bCSC share biochemical features of epithelial-mesenchymal transition (EMT), a process implicated in development of aggressive breast cancer (18–20). We have shown previously that the BITC-mediated inhibition of breast cancer xenograft growth in vivo is associated with biochemical features of EMT inhibition characterized by induction of adherens junction protein E-cadherin and suppression of mesenchymal marker vimentin (21,22). Because of coalition between bCSC and EMT, it was only logical to test whether BITC inhibits bCSC. The present study addresses this question using relevant in vitro and in vivo models of breast cancer.
Material and methods
Ethical issues concerning use and care of laboratory animals
The use of mice and their care for the studies described herein were in accordance with the University of Pittsburgh Institutional Animal Care and Use Committee guidelines (protocol #12080818).
Reagents and cell lines
BITC (purity >98%) was purchased from LKT laboratories. Reagents for cell culture including medium, fetal bovine serum, and antibiotics were purchased from Invitrogen-Life Technologies. Sources of the antibodies were as follows: anti-Ron, which recognizes both full-length Ron (pro-Ron), Ronβ (active form), and short form (sfRon)- Santa Cruz Biotechnology, anti-N-cadherin- Cell Signaling Technology; antiE-cadherin- BD Biosciences, and anti-vimentin and anti-actin- Sigma-Aldrich. The MCF-7 and MDA-MB-231 cells were purchased from the American Type Culture Collection (Manassas, VA) and authenticated prior to the work. The cells were last authenticated in February of 2012. The SUM159 cell line was purchased from Asterand and authenticated by the provider. The MCF-7 and MDA-MB-468 cells stably transfected with a plasmid encoding for urokinase-type plasminogen activator receptor (uPAR) were generously provided by Dr. Steven L Gonias (University of California, San Diego, CA) (23). The wild-type, and Ron and sfRon overexpressing MCF-7 cells were generously provided by Dr. Alana L. Welm (University of Utah, Salt Lake City, UT) (24). Each cell line was maintained at 37°C in an atmosphere of 95% air and 5% CO2 according the recommendations of the providers.
Mammosphere formation assay
Mammosphere formation assay was performed as described by Li et al. (25). Single cells were plated in ultra-low attachment plates (Corning) at a density 500–5,000 cells/well in serum-free mammary epithelium basal medium (Lonza) supplemented with 1% penicillin/streptomycin, B27 (1:50, Invitrogen-Life Technologies), 5 μg/mL Insulin, 1 μg/mL hydrocortisone (Sigma), 20 ng/mL epidermal growth factor (R&D Systems), 20 ng/mL basic fibroblast growth factor (Stem Cell) and 2-mercaptoethanol. Indicated concentrations of BITC were added to the media for primary mammosphere assay. Stock solution of BITC was prepared in dimethyl sulfoxide (DMSO), and an equal volume of DMSO (final concentration <0.15%) was added to controls. After 5 days, the primary mammospheres were harvested, dissociated with trypsin, and then passed through a 40 μm strainer. Single cells were then re-plated in ultra-low attachment plates for second (5 days) and third generation (7 days) mammosphere formation assays without further treatment with BITC or DMSO. The mammospheres were counted under an inverted microscope.
Flow cytometric analysis of aldehyde dehydrogenase 1-positive (ALDH1+) cells
The flow cytometric quantitation of ALDH1+ cells was carried out by following the manufacturer’s instructions (Stem cells). Briefly, cells were suspended in assay buffer containing an ALDH1 substrate (bodipy-aminoacetaldehyde) and incubated at 37°C for 30 min. As a positive control, half of the sample was transferred to a tube containing the ALDH1 inhibitor diethylaminobenzaldehyde (DEAB). Prior to flow cytometric analysis, cells were re-suspended in assay buffer and mixed with 1 μg/mL propidium iodide.
Analysis of CD44high/CD24low/epithelial specific antigen (ESA)+ population
Cells were trypsinized, washed with phosphate-buffered saline (PBS) twice, and stained with 20 μL of anti-ESA (FITC-conjugated, BD Biosciences), anti-CD24 (PE-conjugated, BD Biosciences) and anti-CD44 (APC-conjugated, BD biosciences) antibodies. Cells were incubated in dark for 30 min at room temperature followed by washing with PBS. The cells were analyzed using CyAn ADP Analyzer from Beckman Coulter.
In vivo experiment
Three-five week old female MMTV-neu transgenic mice [homozygous; strain FVB/N-Tg(MMTV-neu) 202Mul/J] were purchased from Jackson Laboratories and acclimated for 1 week prior to start of the experiment. The mice were randomized into 2 groups with 10 mice in each group. The control mice were placed on basal AIN-76A diet, whereas the experimental group of mice were fed AIN-76A diet supplemented with 3 μmol BITC/g diet (447.6 mg BITC/kg diet). The diet was replaced every 3–4 days at the time of cage change. Diet consumption and body weights were recorded once weekly. Each mouse in every group was also monitored on alternate days for any other signs of distress such as impaired movement, unusual posture, indigestion, and areas of redness or swelling. Mice were sacrificed after 29 weeks of treatment. Tumor tissue was dissected and washed with PBS and digested in DMEM supplemented with 300 units/mL collagenase and 100 units/mL hyaluronidase for 3–4 h at 37°C. Cells were re-suspended in Hank’s balanced salt solution (HBSS) supplemented with 2% fetal bovine serum and ammonium chloride. The resultant suspension was re-suspended in 0.25% Trypsin-EDTA and 5 mg/mL dispase, 0.1 mg/mL DNase1 in HBSS and filtered through 40 μm strainer. The cells were used for ALDH1 assay or mammosphere formation assay. Mammosphere formation from tumor samples was performed as described by Liao et al. (26). Single cells were plated in ultra-low attachment plate in DMEM/F12 supplemented with 2% fetal bovine serum, 10 mmol/L HEPES, 5% bovine serum albumin, 1% penicillin/streptomycin, 10 μg/mL insulin, 20 ng/mL epidermal growth factor, 20 ng/mL basic fibroblast growth factor, B27, 10 μg/mL heparin and 2-mercaptoethanol for 7 days.
Western blotting
Cell lysates and xenograft supernatants were prepared as described previously (27,28). Western blotting was performed as described previously (27,28).
Real-time quantitative polymerase chain reaction (qPCR)
Total RNA from DMSO-treated control and BITC-treated cells was isolated using RNeasy kit (Qiagen). First-strand cDNA was synthesized using Superscript Reverse Transcriptase (Invitrogen-Life Technologies) with oligo (dT)20 primer. Primers were as follow: Ron: forward 5′-AGCCCACGCTCAGTGTCTAT-3′, reverse 5′-GGGCACTAGGATCATCTGTCA-3′; Oct-4: forward 5′-GTGGAGAGCAACTCCGATG-3′, reverse 5′-TGCTCCAGCTTCTCCTTCTC-3′; Nanog, forward 5′-ATTCAGGACAGCCCTGATTCTTC-3′, reverse 5′-TTTTTGCGACACTCTTCTCTGC-3′; SOX-2, forward 5′-CGAGTGGAAACTTTTGTCGGA-3′, reverse 5′-TGTGCAGCGCTCGCAG-3′.The qPCR was done using 2 × SYBR Green master mix (Applied Biosystems-Life Technologies) with 95°C (60 seconds), 55°C annealing (60 seconds for Ron), 60°C annealing (60 second for Oct-4, Nanog and SOX-2) and 72°C (60 seconds) for 40 cycles. Relative gene expression was calculated using the method described by Livak and Schmittgen (29).
Quantitation of macrophage stimulating protein (MSP)
A commercially available kit from R&D Systems was used for quantitative measurement of MSP in cell lysates. Manufacturer’s instructions were followed for quantitation of the MSP levels.
Statistical analysis
Two-tailed unpaired Student’s t-test with Welch’s correction or one-way analysis of variance (ANOVA) followed by Dunnett’s adjustment (for dose-response effects) or Bonferroni’s test (for multiple comparisons) were used to determine statistical significance of difference in measured variables. Results are expressed as mean ± SD. Difference was considered significant at P <0.05.
Results
BITC inhibited self-renewal of bCSC
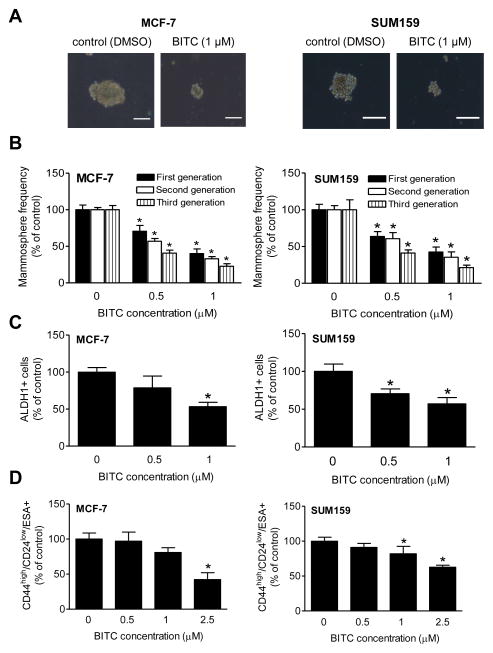
Initially, we used MCF-7 (ER-positive) and SUM159 (triple-negative) human breast cancer cells to determine the effect of BITC on bCSC. The BITC concentrations used in these experiments were (0.5–1 μmol/L) lower than pro-apoptotic doses (11,12). Fig. 1A exemplifies mammospheres resulting after 5 days of treatment of MCF-7 and SUM159 cells with DMSO (control) or the indicated concentration of BITC. First generation mammosphere frequency was decreased significantly and dose-dependently in the presence of BITC in both cell lines when compared with DMSO-treated control (Fig. 1B). The mammospheres from the first generation were disaggregated and the resulting single cells were re-plated without further treatment with DMSO (control) or BITC. Second (after 5 days) and third generation (after 7 days) mammosphere frequency was significantly lower in BITC-treated MCF-7/SUM159 cells compared with control (Fig. 1B). Inhibitory effect of BITC on bCSC was confirmed by flow cytometric analysis of ALDH1 activity (Fig. 1C). As can be seen in Supplementary Fig. S1, the ALDH1 activity was reduced in the presence of BITC in a dose-dependent manner. For example, the BITC treatment (1 μmol/L, 72 h) caused an approximate 46% and 42% decrease in ALDH1 activity in MCF-7 and SUM159 cells, respectively, compared with DMSO-treated control (Fig. 1C). Consistent with these observations, percentage of CD44high/CD24low/ESA+ fraction was lower in BITC-treated MCF-7 and SUM159 cell cultures compared with control (Fig. 1D). Collectively, these results indicated in vitro inhibition of bCSC in the presence of BITC.
Figure 1.
A, Representative images depicting MCF-7 and SUM159 primary (first generation) mammospheres after 5 days of treatment with DMSO or the indicated concentration of BITC (100 magnification; scale bar- 100 μm). B, Quantitation of first (after 5days), second generation (after 5 days), and third generation (after 7 days) mammospheres. The bar graph shows the frequency of mammosphere formation normalized to DMSO-treated control (mean ± SD, n=3). C, Effect of BITC treatment on ALDH1+ population. The bar graph shows the percentage of ALDH1+ population relative to DMSO-treated control in MCF-7 and SUM159 cells (mean ± SD, n=3). D, Effect of BITC treatment on CD44high/CD24low/ESA+ population relative to DMSO-treated control (mean ± SD, n=3). *Significantly different (P < 0.05) compared with DMSO-treated control by one-way ANOVA with Dunnett’s adjustment. Comparable results were observed in two independent experiments. Representative data from one such experiment are shown.
Dietary BITC administration decreased bCSC in MMTV-neu mice tumors in vivo
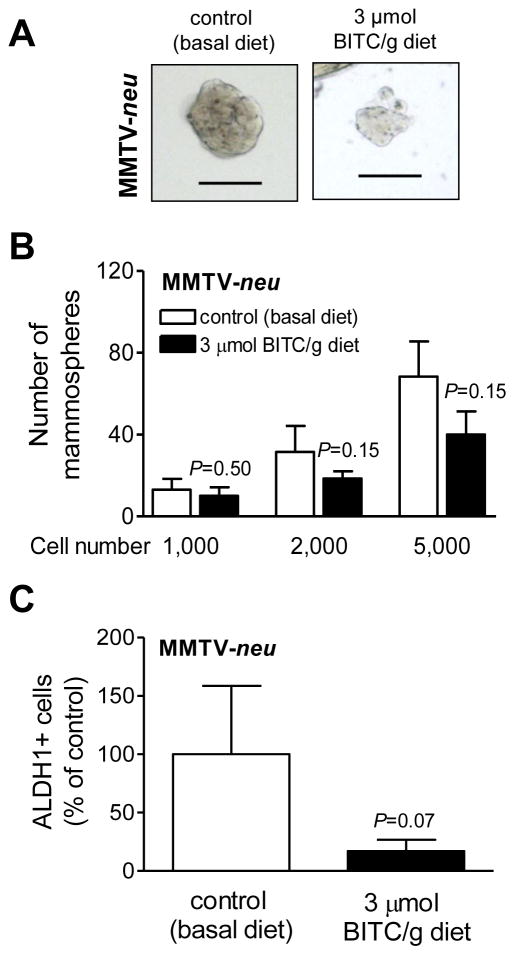
We next proceeded to determine the in vivo efficacy of BITC against bCSC using MMTV-neu mouse model. Rationale for the use of this model in the present study was based on the following considerations: (a) we have shown previously that dietary administration of BITC (3 μmol/g diet) prevents mammary cancer development in MMTV-neu mouse model (9), and (b) increased self-renewal and replicative potential of bCSC has been reported in this model (30). We used the same protocol of BITC treatment (3 μmol BITC/g diet for 29 weeks) to determine the effect on bCSC. After 29 weeks of BITC administration, the tumor incidence in the control and BITC treatment groups was 40% and 20%, respectively. Tumors from the control mice (n=4) and those treated with BITC (n=2) were disaggregated and single cell suspensions were used for mammosphere formation assay (Fig. 2A). At each cell seeding density, the number of mammospheres from BITC-treated tumors was markedly lower compared with those from control tumors with the difference did not reach statistical significance due to an outlier in the control group and small sample size in the BITC treatment arm (Fig. 2B). Nevertheless, consistent with these results, the ALDH1 activity was about 83% lower in the tumors from BITC-treated mice compared with control mice (P=0.07 by two-tailed unpaired Student’s t-test with Welch’s correction; Fig. 2C). These results provided in vivo evidence for BITC-mediated inhibition of bCSC.
Figure 2.
A, Representative mammosphere images from tumors of MMTV-neu mice placed on basal (control) diet or 3 μmol BITC/g diet (100 magnification; scale bar- 100 μm). B, The bar graph shows the number of mammosphere from tumors of control and BITC-treated MMTV-neu mice. Results shown are mean ± SD (n=4 for control and n=2 for BITC-treated). C, The bar graph shows the percentage of ALDH1+ population in tumors from control and BITC-treated MMTV-neu mice. Results shown are mean ± SD (n=4 for control group and n=2 for BITC treated group). Statistical significance was determined by two-tailed unpaired Student’s t-test with Welch’s correction.
Urokinase-type plasminogen activator receptor (uPAR) was dispensable for BITC-mediated inhibition of bCSC
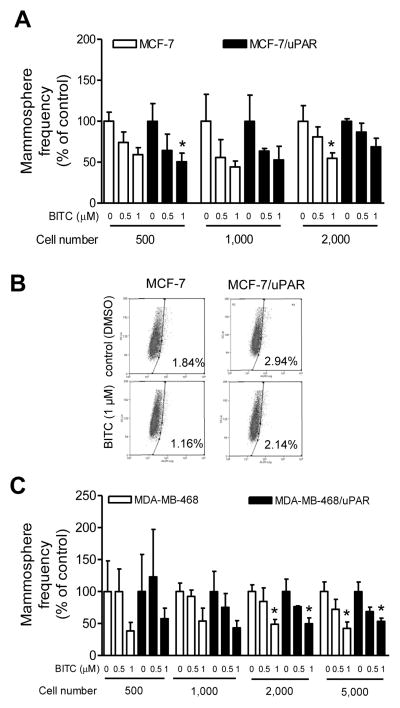
Studies have shown that overexpression of uPAR alone is sufficient to drive both EMT and stemness in human breast cancer cells (23,31). Recent work from our own laboratory has revealed transcriptional repression of uPAR in BITC-treated breast cancer cells (22). We therefore raised the question of whether inhibition of bCSC upon treatment with BITC was due to uPAR suppression. Overexpression of uPAR (overexpression of uPAR was confirmed, results not shown) increased mammosphere formation frequency in MCF-7 cells by about 1.5-fold. Mammosphere formation frequency was decreased in a dose-dependent manner upon treatment with BITC in MCF-7 cells stably transfected with the empty vector as well as uPAR plasmid. However, when the results were normalized against corresponding DMSO-treated control, the percentage of inhibition in mammosphere formation frequency upon treatment with BITC was comparable for MCF-7 cells and MCF-7/uPAR cells (Fig. 3A). These results were confirmed by analysis of ALDH1 activity (Fig. 3B). Consistent with results in MCF-7 cells (Fig. 3A), overexpression of uPAR failed to confer any protection against BITC-mediated inhibition of mammosphere formation (Fig. 3C) or ALDH1 activity (results not shown) in MDA-MB-468 cells. Collectively, these results indicated that uPAR suppression was dispensable for BITC-mediated inhibition of bCSC at least in breast cancer cells used in the present study.
Figure 3.
A, Frequency of mammosphere formation (normalized to respective DMSO-treated control) from MCF-7 cells stably transfected with empty vector (MCF-7) or the same vector encoding for uPAR (MCF-7/uPAR). Results shown are mean ± SD (n=3). B, Representative flow histograms showing ALDH1+ cell population in MCF-7 cells stably transfected with empty vector or the same vector encoding for uPAR after 72 h treatment with DMSO or 1 μM BITC. C, The bar graph shows the frequency of mammosphere formation (normalized to respective DMSO-treated control) from MDA-MB-468 cells stably transfected with empty vector (MDA-MB-468) or the same vector encoding for uPAR (MDA-MB-468/uPAR). Results shown are mean ± SD (n=3). Each experiment was repeated at least twice and the results were similar. *Significantly different compared with corresponding DMSO-treated control by one-way ANOVA with Bonferroni’s adjustment. There was no difference between empty vector transfected cells and uPAR overexpressing cells at any dose.
BITC down-regulated Ron in breast cancer cells
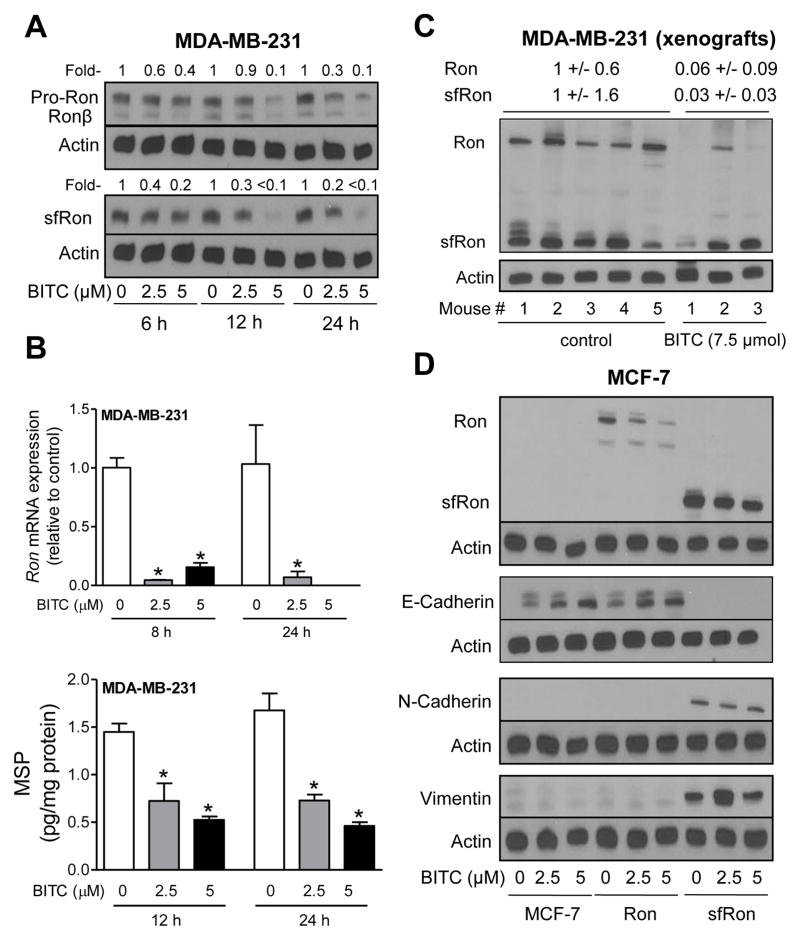
The Ron tyrosine receptor kinase and/or its 55-kDa N-terminally truncated form (sfRon) has been implicated in EMT induction as well as metastasis of breast cancer (32–34). Initially, we designed experiments to determine if BITC treatment affected Ron and/or sfRon protein levels. Exposure of MDA-MB-231 cells, which express substantial amount of Ron and sfRon proteins, to BITC resulted in a dose- and time-dependent decline in protein levels of pro-Ron and its active form (Ronβ) and truncated form (sfRon) (Fig. 4A). The BITC-mediated suppression of Ron protein was accompanied by down-regulation of its mRNA (Fig. 4B; upper panel). BITC treatment also caused a significant decrease in protein levels of Ron ligand MSP (Fig. 4B; lower panel). Moreover, the BITC-mediated inhibition of MDA-MB-231 xenograft growth in vivo (10) was associated with downregulation of Ron and sfRon protein levels (Fig. 4C).
Figure 4.
A, Western blotting for pro-Ron, Ronβ, and sfRon proteins using lysates from MDA-MB-231 cells after 6, 12, and 24 h treatment with DMSO (control) or the indicated concentrations of BITC. B, Quantitation of Ron mRNA level (upper panel) by qPCR in MDA-MB-231 cells after 8 or 24 h treatment with DMSO (control) or BITC (2.5 or 5 μM). Quantitation of MSP (lower panel) in lysates from MDA-MB-231 cells after 12 or 24 h treatment with DMSO (control) or the indicated concentrations of BITC. Results shown are mean ± SD (n=3). *Significantly different (P < 0.05) compared with DMSO-treated control by one-way ANOVA with Dunnett’s test. C, Western blotting for Ron and sfRon using MDA-MB-231 xenograft supernatants from control and BITC-treated athymic mice (n=5 for control, and n=3 for BITC-treated). D, Immunoblotting for Ron, sfRon, E-cadherin, N-cadherin, and vimentin using lysates from parental MCF-7 cells or those transduced to overexpress full-length Ron or sfRon after 24 h treatment with DMSO or the indicated concentrations of BITC. Similar results were obtained from replicate experiments.
We used the MCF-7 cells for functional studies to determine the role of Ron and sfRon in BITC-mediated inhibition of EMT and bCSC. Protein levels of exogenously expressed Ron and sfRon were decreased in the presence of BITC (Fig. 4D). Overexpression of sfRon, but not full-length Ron, triggered EMT as evidenced by suppression of E-cadherin and induction of mesenchymal markers N-cadherin and vimentin (Fig. 4D). The BITC treatment caused induction of E-cadherin in control and Ron overexpressing MCF-7 cells, but not in cells with stable overexpression of sfRon (Fig. 4D). These results indicated that overexpression of sfRon attenuated BITC-mediated induction of epithelial marker E-cadherin.
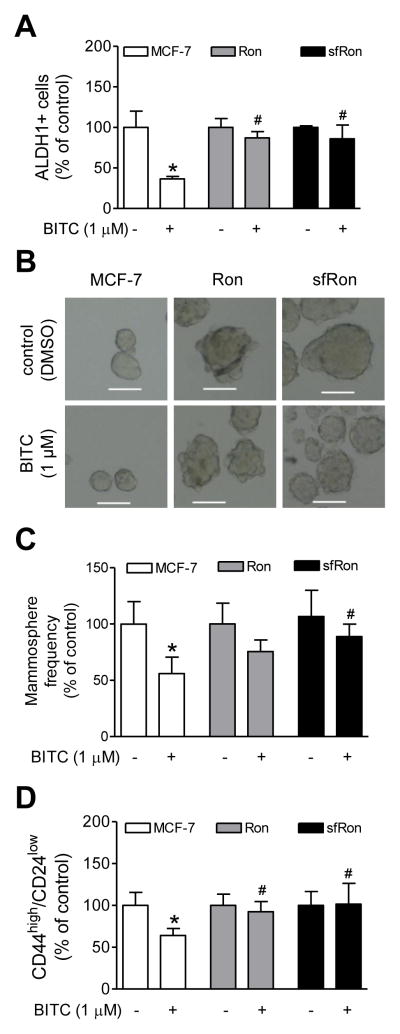
Overexpression of Ron and sfRon conferred protection against BITC-mediated inhibition of bCSC
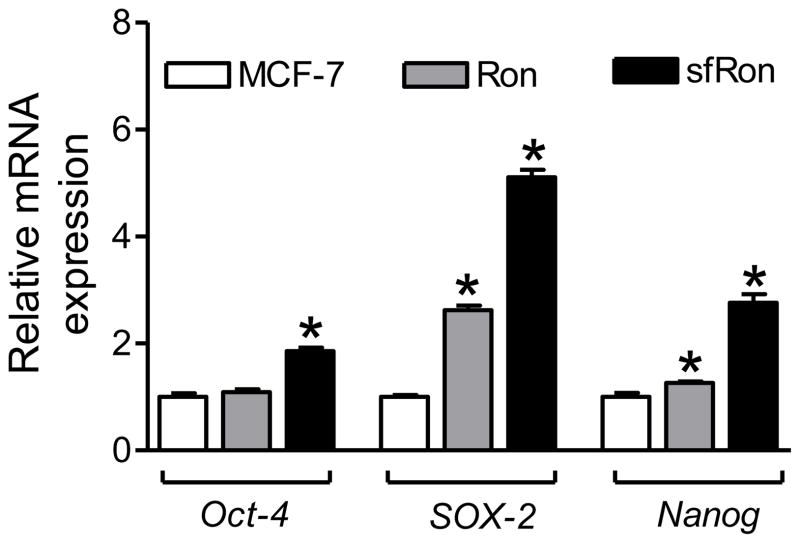
The inhibition of ALDH1+ fraction resulting from BITC exposure was nearly completely abolished by overexpression of Ron as well as sfRon (Fig. 5A). The mammospheres from Ron and sfRon overexpressing MCF-7 cells were relatively larger in size than those observed from parental MCF-7 cells (Fig. 5B). In agreement with ALDH1 activity data, the BITC-mediated inhibition of mammosphere formation (Fig. 5C) and CD44high/CD24low population (Fig. 5D) was significantly attenuated by overexpression of Ron and sfRon. Moreover, the Ron and/or sfRon overexpressing MCF-7 cells exhibited a significant increase in mRNA levels of bCSC-associated genes Oct-4, SOX-2, and Nanog (Fig. 6). These results indicated that BITC-mediated inhibition of bCSC was attenuated by overexpression of Ron and sfRon at least in MCF-7 cells.
Figure 5.
A, The percentage of ALDH1+ population normalized to respective DMSO-treated control in MCF-7 cells or those overexpressing Ron or sfRon after 72 h treatment with DMSO or BITC. Results shown are mean ± SD (n=6). B, Representative mammosphere images (100 magnification; scale bar- 100 μm) for MCF-7 cells and those transduced to overexpress Ron and sfRon. C, The bar graph shows the frequency of mammosphere formation normalized to respective DMSO-treated control in MCF-7 cells or those overexpressing Ron or sfRon after 4 days of treatment with DMSO or BITC. Results shown are mean ± SD (n=6). D, The bar graph shows the percentage of CD44high/CD24low population normalized to respective DMSO-treated control in parental MCF-7 cells or those overexpressing Ron or sfRon after 3 days of treatment with DMSO or BITC. Results shown are mean ± SD (n=6). Significantly different (P < 0.05) compared with *respective DMSO-treated control and #between BITC treated MCF-7 cells and BITC-treated Ron or sfRon overexpressing cells by one-way ANOVA followed by Bonferroni’s multiple comparison test. Each experiment was performed at least twice in triplicate and combined data from both experiments are shown (A, C and D).
Figure 6.
Quantitation of Oct-4, SOX-2 and Nanog mRNA levels by qPCR in MCF-7 cells or those stably overexpressing Ron or sfRon. Results shown are mean ± SD (n=3).
Discussion
We have shown previously that mammary cancer prevention by BITC in MMTV-neu mice is associated with tumor cell apoptosis. The present study building upon these observations shows, for the first time, that BITC treatment inhibits self-renewal of bCSC as evidenced by mammosphere formation, ALDH1 activity, and cell-surface expression of cancer stem cell markers CD44 and ESA. Of note, the BITC-mediated in vitro inhibition of bCSC is observed at concentrations (0.5–1 μmol/L) that are not very cytotoxic but within the plasma achievable levels (35). We also provide evidence that BITC administration eliminates bCSC in vivo. Based on these observations, it is reasonable to propose that elimination of both epithelial tumor cells via apoptosis and autophagy induction and bCSC likely contributes to breast cancer prevention by BITC.
Overexpression of uPAR alone is sufficient to drive EMT (MDA-MB-468) and/or stemness (MCF-7 and MDA-MB-468) in breast cancer cells (23,31). The uPAR and its ligand uPA constitute an integral component of the extracellular matrix proteolysis, cell-extracellular matrix interaction, as well as cell signaling involving receptor tyrosine kinases (36,37). High uPA/uPAR level has been shown to be an adverse prognostic indicator for different cancers including breast tumors (38). Down-regulation of constitutive uPA in cultured HT29 human colon cancer cells and suppression of hepatocyte growth factor-stimulated secretion of uPA in MDA-MB-231 cells upon treatment with BITC has been reported previously (39,40). We have also shown recently that BITC treatment causes transcriptional repression of uPAR protein in several breast cancer cells, but this system is dispensable for BITC-mediated inhibition of EMT (22). The present study reveals that overexpression of uPAR fails to confer any protection against BITC-mediated inhibition of bCSC in MCF-7 or MDA-MB-468 cells. However, it is possible that suppression of uPA/uPAR system BITC contributes to its inhibitory effect on breast cancer cell proliferation and possibly metastasis. Additional work is needed to experimentally test this possibility.
Ron (recepteur d’origine nantais) tyrosine kinase expression is very low in normal human breast epithelium but its overexpression has been observed in about 50% of primary breast cancers (41). Moreover, increased expression of the Ron receptor correlates with more aggressive phenotype in node-negative breast tumors (42). Mammary-specific overexpression of wild-type Ron as well as constitutively active Ron induces highly metastatic breast tumors in mice (34). Studies have also indicated that Ron receptor tyrosine kinase can promote cell spreading and survival in breast cancer cells independent of its only known ligand MSP (43). Activation of Ron by hepatocyte growth factor-like protein has been shown to confer resistance to tamoxifen in breast cancer cell lines (44). The present study demonstrates, for the first time, that overexpression of full-length Ron and sfRon induces stemness in MCF-7 cells that is accompanied by increased expression of stem cell-associated genes Oct-4, SOX-2, and Nanog. We also found that BITC treatment inhibits protein and/or mRNA levels of full-length Ron receptor tyrosine kinase and its 55-kDa N-terminally truncated form (sfRon). Moreover, overexpression of full-length Ron and sfRon confers near complete protection against BITC-mediated inhibition of bCSC in MCF-7 cells. On the other hand, overexpression of sfRon, but not the full-length Ron, is sufficient to attenuate BITC-mediated induction of E-cadherin.
The novel findings of the present study are: (a) overexpression of full-length Ron as well as its truncated form (sfRon) is sufficient to drive stemness in MCF-7 cells; (b) BITC treatment inhibits bCSC in vitro and in vivo; and (c) the BITC-mediated inhibition of bCSC is nearly fully abolished by overexpression of Ron and sfRon at least in MCF-7 cells. In conclusion, the present study identified Ron receptor tyrosine kinase as a critical target of bCSC inhibition by BITC.
Supplementary Material
Acknowledgments
The authors thank Julie A. Arlotti with the animal studies.
Grant Support
This work was supported by the grant 2 RO1 CA129347-06 awarded by the National Cancer Institute. This research used the animal facility and the flow cytometry facility supported in part by a grant from the National Cancer Institute at the National Institutes of Health (P30 CA047904).
Footnotes
Disclosure of Potential Conflicts of Interest:
None
Authors’ Contributions
Conception and design: S.H. Kim, A. Sehrawat, S.V. Singh
Development of methodology: S.H. Kim
Acquisition of data (provided animals, acquired and managed patients, provided facilities, etc.): S.H. Kim, A. Sehrawat, S.V. Singh
Analysis and interpretation of data (e.g., statistical analysis, biostatistics, computational analysis): S.H. Kim, A. Sehrawat, S.V. Singh
Writing, review and/or revision of the manuscript: S.H. Kim, A. Sehrawat, S.V. Singh
Study supervision: S.V. Singh
References
- 1.Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA Cancer J Clin. 2012;62:10–29. doi: 10.3322/caac.20138. [DOI] [PubMed] [Google Scholar]
- 2.Higgins MJ, Baselga J. Breast cancer in 2010: Novel targets and therapies for a personalized approach. Nat Rev Clin Oncol. 2011;8:65–6. doi: 10.1038/nrclinonc.2010.217. [DOI] [PubMed] [Google Scholar]
- 3.Fisher B, Costantino JP, Wickerham DL, Redmond CK, Kavanah M, Cronin WM, et al. Tamoxifen for prevention of breast cancer: report of the National Surgical Adjuvant Breast and Bowel project P-1 Study. J Natl Cancer Inst. 1998;90:1371–88. doi: 10.1093/jnci/90.18.1371. [DOI] [PubMed] [Google Scholar]
- 4.Cauley JA, Norton L, Lippman ME, Eckert S, Krueger KA, Purdie DW, et al. Continued breast cancer risk reduction in postmenopausal women treated with raloxifene: 4-year results from the MORE trial: Multiple outcomes of raloxifene evaluation. Breast Cancer Res Treat. 2001;65:125–34. doi: 10.1023/a:1006478317173. [DOI] [PubMed] [Google Scholar]
- 5.Goss PE, Ingle JN, Ales-Martinez JE, Cheung AM, Chlebowski RT, Wactawski-Wende J, et al. Exemestane for breast-cancer prevention in postmenopausal women. N Eng J Med. 2011;364:2381–91. doi: 10.1056/NEJMoa1103507. [DOI] [PubMed] [Google Scholar]
- 6.Surh YJ. Cancer chemoprevention with dietary phytochemicals. Nat Rev Cancer. 2003;3:768–80. doi: 10.1038/nrc1189. [DOI] [PubMed] [Google Scholar]
- 7.Stan SD, Kar S, Stoner GD, Singh SV. Bioactive food components and cancer risk reduction. J Cell Biochem. 2008;104:339–56. doi: 10.1002/jcb.21623. [DOI] [PubMed] [Google Scholar]
- 8.Wattenberg LW. Inhibition of carcinogenic effects of polycyclic hydrocarbons by benzyl isothiocyanate and related compounds. J Natl Cancer Inst. 1977;58:395–8. doi: 10.1093/jnci/58.2.395. [DOI] [PubMed] [Google Scholar]
- 9.Warin R, Chambers WH, Potter DM, Singh SV. Prevention of mammary carcinogenesis in MMTV-neu mice by cruciferous vegetable constituent benzyl isothiocyanate. Cancer Res. 2009;69:9473–80. doi: 10.1158/0008-5472.CAN-09-2960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Warin R, Xiao D, Arlotti JA, Bommareddy A, Singh SV. Inhibition of human breast cancer xenograft growth by cruciferous vegetable constituent benzyl isothiocyanate. Mol Carcinogenesis. 2010;49:500–7. doi: 10.1002/mc.20600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Xiao D, Vogel V, Singh SV. Benzyl isothiocyanate-induced apoptosis in human breast cancer cells is initiated by reactive oxygen species and regulated by Bax and Bak. Mol Cancer Ther. 2006;5:2931–45. doi: 10.1158/1535-7163.MCT-06-0396. [DOI] [PubMed] [Google Scholar]
- 12.Xiao D, Powolny AA, Singh SV. Benzyl isothiocyanate targets mitochondrial respiratory chain to trigger reactive oxygen species-dependent apoptosis in human breast cancer cells. J Biol Chem. 2008;283:30151–63. doi: 10.1074/jbc.M802529200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Xiao D, Bommareddy A, Kim SH, Sehrawat A, Hahm ER, Singh SV. Benzyl isothiocyanate causes FoxO1-mediated autophagic death in human breast cancer cells. PLoS ONE. 2012;7:e32597. doi: 10.1371/journal.pone.0032597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Antony ML, Kim SH, Singh SV. Critical role of p53 upregulated modulator of apoptosis in benzyl isothiocyanate-induced apoptotic cell death. PLoS ONE. 2012;7:e32267. doi: 10.1371/journal.pone.0032267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Oliveira LR, Jeffrey SS, Rebeiro-Silva A. Stem cells in human breast cancer. Histol Histopathol. 2010;25:371–85. doi: 10.14670/HH-25.371. [DOI] [PubMed] [Google Scholar]
- 16.Velasco-Velázquez MA, Homsi N, De La Fuente M, Pestell RG. Breast cancer stem cells. Int J Biochem Cell Biol. 2012;44:573–7. doi: 10.1016/j.biocel.2011.12.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.O’Brien CS, Farnie G, Howell SJ, Clarke RB. Breast cancer stem cells and their role in resistance to endocrine therapy. Horm Cancer. 2011;2:91–103. doi: 10.1007/s12672-011-0066-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Raimondi C, Gradilone A, Naso G, Vincenzi B, Petracca A, Nicolazzo C, et al. Epithelial-mesenchymal transition and stemness features in circulating tumor cells from breast cancer patients. Breast Cancer Res Treat. 2011;130:449–55. doi: 10.1007/s10549-011-1373-x. [DOI] [PubMed] [Google Scholar]
- 19.Dave B, Mittal V, Tan NM, Chang JC. Epithelial-mesenchymal transition, cancer stem cells, and treatment resistance. Breast Cancer Res. 2012;14:202. doi: 10.1186/bcr2938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Velasco-Velázquez MA, Popov VM, Lisanti MP, Pestell RG. The role of breast cancer stem cells in metastasis and therapeutic implications. Am J Pathol. 201l;179:2–11. doi: 10.1016/j.ajpath.2011.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sehrawat A, Singh SV. Benzyl isothiocyanate inhibits epithelial-mesenchymal transition in cultured and xenografted human breast cancer cells. Cancer Prev Res. 2011;4:1107–17. doi: 10.1158/1940-6207.CAPR-10-0306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sehrawat A, Kim SH, Vogt A, Singh SV. Suppression of FOXQ1 in benzyl isothiocyanate-mediated inhibition of epithelial-mesenchymal transition in human breast cancer cells. Carcinogenesis. 2013;34:864–73. doi: 10.1093/carcin/bgs397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Jo M, Eastman BM, Webb DL, Stoletov K, Klemke R, Gonias SL. Cell signaling by urokinase-type plasminogen activator receptor induces stem cell-like properties in breast cancer cells. Cancer Res. 2010;70:8948–58. doi: 10.1158/0008-5472.CAN-10-1936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Liu X, Zhao L, Derose YS, Lin YC, Bieniasz M, Eyob H, et al. Short-form Ron promotes spontaneous breast cancer metastasis through interaction with phosphoinositide 3-kinase. Genes Cancer. 2011;2:753–62. doi: 10.1177/1947601911421924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Li Y, Zhang T, Korkaya H, Liu S, Lee HF, Newman B, et al. Sulforaphane, a dietary component of broccoli/broccoli sprouts, inhibits breast cancer stem cells. Clin Cancer Res. 2010;16:2580–90. doi: 10.1158/1078-0432.CCR-09-2937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Liao MJ, Zhang CC, Zhou B, Zimonjic DB, Mani SA, Kaba M, et al. Enrichment of a population of mammary gland cells that form mammosphere and have in vivo repopulating activity. Cancer Res. 2007;67:8131–8. doi: 10.1158/0008-5472.CAN-06-4493. [DOI] [PubMed] [Google Scholar]
- 27.Xiao D, Srivastava SK, Lew KL, Zeng Y, Hershberger P, Johnson CS, et al. Allyl isothiocyanate, a constituent of cruciferous vegetables, inhibits proliferation of human prostate cancer cells by causing G2/M arrest and inducing apoptosis. Carcinogenesis. 2003;24:891–7. doi: 10.1093/carcin/bgg023. [DOI] [PubMed] [Google Scholar]
- 28.Powolny AA, Bommareddy A, Hahm ER, Normolle DP, Beumer JH, Nelson JB, et al. Chemopreventative potential of the cruciferous vegetable constituent phenethyl isothiocyanate in a mouse model of prostate cancer. J Natl Cancer Inst. 2011;103:571–84. doi: 10.1093/jnci/djr029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods. 2001;25:402–8. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 30.Cicalese A, Bonizzi G, Pasi CE, Faretta M, Ronzoni S, Giulini B, et al. The tumor suppressor p53 regulates polarity of self-renewing divisions in mammary stem cells. Cell. 2009;138:1083–95. doi: 10.1016/j.cell.2009.06.048. [DOI] [PubMed] [Google Scholar]
- 31.Lester RD, Jo M, Montel V, Takimoto S, Gonias SL. uPAR induces epithelial-mesenchymal transition in hypoxic breast cancer cells. J Cell Biol. 2007;178:425–36. doi: 10.1083/jcb.200701092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bardella C, Costa B, Maggiora P, Patane’ S, Olivero M, Ranzani GN, et al. Truncated RON tyrosine kinase drives tumor cell progression and abrogates cell-cell adhesion through E-cadherin transcriptional repression. Cancer Res. 2004;64:5154–61. doi: 10.1158/0008-5472.CAN-04-0600. [DOI] [PubMed] [Google Scholar]
- 33.Kretschmann KL, Eyob H, Buys SS, Welm AL. The macrophage stimulating protein/Ron pathway as a potential therapeutic target to impede multiple mechanisms involved in breast cancer progression. Curr Drug Targets. 2010;11:1157–68. doi: 10.2174/138945010792006825. [DOI] [PubMed] [Google Scholar]
- 34.Zinser GM, Leonis MA, Toney K, Pathrose P, Thobe M, Kader SA, et al. Mammary-specific Ron receptor overexpression induces highly metastatic mammary tumors associated with beta-catenin activation. Cancer Res. 2006;66:11967–74. doi: 10.1158/0008-5472.CAN-06-2473. [DOI] [PubMed] [Google Scholar]
- 35.Boreddy SR, Pramanik KC, Srivastava SK. Pancreatic tumor suppression by benzyl isothiocyanate is associated with inhibition of PI3K/AKT/FOXO pathway. Clin Cancer Res. 2011;17:1784–95. doi: 10.1158/1078-0432.CCR-10-1891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hildenbrad R, Allgayer H, Marx A, Stroebel P. Modulators of the urokinase-type plasminogen activation system for cancer. Expert Opin Investig Drugs. 2010;19:641–52. doi: 10.1517/13543781003767400. [DOI] [PubMed] [Google Scholar]
- 37.Jo M, Thomas KS, O’Donnell DM, Gonias SL. Epidermal growth factor receptor-dependent and - independent cell-signaling pathways originating from the urokinase receptor. J Biol Chem. 2003;278:1642–6. doi: 10.1074/jbc.M210877200. [DOI] [PubMed] [Google Scholar]
- 38.Kwaan HC, McMahon B. The role of plasminogen-plasmin system in cancer. Cancer Treat Res. 2009;148:43–66. doi: 10.1007/978-0-387-79962-9_4. [DOI] [PubMed] [Google Scholar]
- 39.Lai KC, Huang AC, Hsu SC, Kuo CL, Yang JS, Wu SH, et al. Benzyl isothiocyanate (BITC) inhibits migration and invasion of human colon cancer HT29 cells by inhibiting matrix metalloproteinase-2/-9 and urokinase plasminogen (uPA) through PKC and MAPK signaling pathway. J Agric Food Chem. 2010;58:2935–42. doi: 10.1021/jf9036694. [DOI] [PubMed] [Google Scholar]
- 40.Kim EJ, Eom SJ, Hong JE, Lee JY, Choi MS, Park JH. Benzyl isothiocyanate inhibits basal and hepatocyte growth factor-stimulated migration of breast cancer cells. Mol Cell Biochem. 2011;359:431–40. doi: 10.1007/s11010-011-1039-3. [DOI] [PubMed] [Google Scholar]
- 41.Maggiora P, Marchio S, Stella MC, Giai M, Belfiore A, De Bortoli M, et al. Overexpression of the RON gene in human breast carcinoma. Oncogene. 1998;16:2927–33. doi: 10.1038/sj.onc.1201812. [DOI] [PubMed] [Google Scholar]
- 42.Lee WY, Chen HH, Chow NH, Su WC, Lin PW, Guo HR. Prognostic significance of co-expression of RON and MET receptors in node-negative breast cancer patients. Clin Cancer Res. 2005;11:2222–8. doi: 10.1158/1078-0432.CCR-04-1761. [DOI] [PubMed] [Google Scholar]
- 43.Feres KJ, Ischenko I, Hayman MJ. The RON receptor tyrosine kinase promotes MSP-independent cell spreading and survival in breast epithelial cells. Oncogene. 2009;28:279–88. doi: 10.1038/onc.2008.383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.McClaine RJ, Marshall AM, Wagh PK, Waltz SE. Ron receptor tyrosine kinase activation confers resistance to tamoxifen in breast cancer cell lines. Neoplasia. 2010;12:650–8. doi: 10.1593/neo.10476. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.