Abstract
Background
Previous studies reported an epidemiological association between CagA-positive H. pylori strains and pre-eclampsia. As antibodies anti-CagA cross-react with endothelial cells and trophoblast cells show an endothelial phenotypic profile, we hypothesized that anti-CagA antibodies may recognize antigens of cytotrophoblast cells, thus impairing their function.
Materials and Methods
Placenta samples were obtained from healthy women. Cytotrophoblast cells were cultured in a medium containing increasing concentration of polyclonal anti-CagA antibodies. Binding of anti-CagA antibodies to cytotrophoblast cells was evaluated by cell ELISA and immunofluorescence assay. Invasive potential of those cells was assessed by an invasion culture system and by measuring of MMP-2. Protein sequencing was performed on antigens precipitated by anti-CagA antibodies. Measurement of phosphorylated ERK expression and NF-kB DNA-binding activity in trophoblast cells incubated with anti-CagA or irrelevant antibodies was also performed.
Results
Anti-CagA antibodies recognized β-actin of cytotrophoblast cells, showing a dose-dependent binding. Incubation of cytotrophoblast cells with increasing doses of anti-CagA antibodies significantly reduced their invasiveness and determined a significant decrease in phosphorylated ERK expression and a reduced NF-kB translocation activity.
Conclusions
This study shows that anti-CagA antibodies recognize β-actin of cytotrophoblast cells, reducing their invasiveness ability, possibly giving a biological explanation for the epidemiological association.
Keywords: CagA+, Helicobacter pylori, pregnancy
Pre-eclampsia (PE) is defined as a hypertensive and coagulative disorder affecting about 2–7% of all pregnancies and is one of the main causes of either maternal or fetal mortality and morbidity [1]. Despite the great amount of studies carried out on this field, little is known about the precise pathogenic mechanisms behind PE [2]. While endothelial and trophoblast dys-function has been shown to play a key role in the occurrence of PE, together with the presence of an exaggerated inflammatory response and hypercoagulative state, the primary trigger is still unknown [3,4]. One of the hypothesis is that some infectious agents may cause PE [5,6].
Helicobacter pylori is a Gram-negative bacterium with a specific tropism for the gastric mucosa [7]; it is main cause of chronic gastritis and peptic ulcer as well as a risk factor for MALT-lymphoma and gastric cancer [8]. While all H. pylori possess some determinants of virulence, only some strains possess also determinants of pathogenicity, able to modulate the local and systemic inflammatory response [9]. A well-recognized determinant of pathogenicity is represented by the cytotoxin-associated gene-A (CagA), which encodes for a hydrophilic, surface-exposed protein [10]. CagA-positive strains have been shown to induce an inflammatory response in the gastric mucosa greater than that induced by CagA negative and are associated with a more severe gastric mucosa damage [11]. Owing to its capability to stimulate the immune system, H. pylori has also been proposed to play a role in some extragastric diseases; in fact, several studies have been aimed at testing the epidemiological association between H. pylori infection and vascular diseases, including ischemic heart diseases (IHD), primary Raynaud’ phenomenon and migraine, all conditions characterized by endothelial dysfunction [12,13]. Studies from our group have clearly shown that anti-CagA antibodies recognize antigens localized on the surface of endothelial cells in either normal or atherosclerotic arteries, thus providing a possible explanation for this association [14,15].
Recent studies have also demonstrated the presence of an epidemiological link between H. pylori infection and PE [16–19]. Ustun et al. [18] reported a significantly higher positivity for IgA anti-H. pylori in patients with PE compared with controls (p = .034). Moreover, Ponzetto et al. [17] clearly showed that H. pylori seropositivity frequency is higher in mothers with PE (51.1%) compared with women with uneventful pregnancy (31.9%) and the difference is even greater when considering positivity for CagA (80.9 and 14.9%, respectively). On the other hand, it is known that women who experienced PE in the course of their life have a higher probability to develop IHD as well as an increased mortality for cardiovascular diseases [20]. Interestingly, patients with PE were shown to have a higher prevalence of CagA-positive strains of H. pylori, which modulate the release of IL-18 and increase C reactive protein and TNF-alpha and malondialdehyde levels, all known to be associated with PE [19,21].
Considering that anti-CagA antibodies are able to cross-react with antigens of endothelial cells and that cytotrophoblast cells show an endothelial origin, we have hypothesized that a similar mechanism may occur with trophoblast cells, thus impairing their function. Therefore, we have designed a study able to test this hypothesis.
Materials and Methods
Cell Cultures
Placentas were obtained from healthy women immediately after uncomplicated vaginal delivery. Cytotrophoblast cells were isolated as detailed elsewhere [22]. Their viability was 90% by trypan blue dye exclusion. The purity of the cell preparation was evaluated by immunohistochemical staining for markers of (1) macrophages (3%, determined using a polyclonal anti-α 1-chymotrypsin antibody; Dako, Santa Barbara, CA, USA); (2) fibroblasts (2%, determined using a polyclonal anti-vimentin antibody; Labsystems, Helsinki, Finland); and (3) syncytiotrophoblast (1% determined using an mAb against low molecular weight cytokeratins; Labsystems, Chicago, IL, USA). The enriched (95%) trophoblast cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM, Sigma-Aldrich, St. Louis, MI, USA) with 10% fetal calf serum (FCS, Sigma) at 37 °C in 5% CO2/95% air.
Binding Assay
A cell-based ELISA was carried out to determine whether the anti-CagA Ab bound to cytotrophoblast cells. For all experiment, homemade polyclonal anti-CagA IgG antibodies were used. Cells were cultured in standard medium for 72 hours, washed three times with HBSS, and incubated with serial concentrations (from 6 to 200 μg/mL) of home-made mouse polyclonal anti-CagA Ab or normal mouse IgG as control, in complete medium at a final volume of 100 μL. After a 2 hours incubation followed by three washes, the plates were incubated with alkaline phosphatase conjugated goat anti-mouse (Sigma) for 90 minutes. After two further washes, p-nitrophenylphosphate (1 mg/mL) in 10% diethanolamine buffer, ph 9.8, was added to each well and incubated for 30 minutes. Optical density (O.D.) was read at 405 nm by a microplate photometer (Platereader, Bio-Rad Laboratories, Milan, Italy).
Immunofluorescence Staining
Cytotrophoblast cells were rinsed twice in PBS and then fixed with 4% paraformaldehyde, PFA (5 minutes). After rinsing with PBS, the cells were incubated with anti-CagA Ab (200 μg/mL) or normal mouse IgG as control for 1 hour at room temperature. Then, the secondary antibody conjugated to FITC (Novus Biologicals, Inc, Cambridge, UK, dilution 1:500) was added for 1 hour at 37 °C. The cells were then rinsed twice in PBS, mounted under microscopic glass coverslips in a Gel/Mount permanent fluorescence-preserving aqueous mounting medium (Biomeda Corporation, Foster City, CA, USA), and evaluated under an inverted phase fluorescent microscope (Zeiss, 200×). Images were acquired with a digital camera (Nikon, Tokyo, Japan).
Invasion Assay
To assess the invasive potential of cytotrophoblast, cells were plated on transwell inserts (Cell Invasion assay Kit, Chemicon International, Millipore, Billerica, MA, USA) containing polycarbonate membrane with 8-μm pores over which a thin layer of ECMatrix is dried. After dehydration, 3 × 105 cells in 300 μL DMEM serum free were plated in the insert with mouse polyclonal anti-CagA Ab (12–50–200 μg/mL) or normal mouse IgG as control. Then, 500 μL of DMEM supplemented with 10% FCS, as a chemo attractant, was added to the lower wells. After 72 hours of culture, using a cotton-tipped swab, noninvading cells as well as the ECMatrix gel from the interior of the insert were gently removed. Stain of invasive cells on lower surface of the membrane was performed by dipping inserts in the staining solution for 20 minutes. Then, stained cells were dissolved in 10% acetic acid, and 100 μL were transferred to a 96-well plate for colorimetric reading at 560 nm.
Gelatin Zymography
MMP-2 gelatinolytic activity in the supernatant of trophoblast cell culture pretreated with anti-CagA (12–50–200 ugr/mL) or normal mouse IgG was measured by gelatine zymography. MMP-2 is synthesized and secreted from the cells as an inactive precursor (proMMP-2) of 72 KDa. Thus, activation of proMMP-2 is a crucial step for the expression of its enzymic activity. ProMMP-2 is readily activated in vitro by 4-aminophenylmercuric acetate (APMA, Sigma-Aldrich). Samples were electrophoresed on SDS polyacrylamide gel containing 1% gelatin. Following electrophoresis, gels were washed three times for 10 minutes at room temperature in 2,5% Triton X-100, to remove SDS. After over-night incubation at 37 °C in 50 mmol/L Tris-HCl (pH 7.4, containing 5 mmol/L CaCl2, 0,15 mol/L NaCl and 0,02% NaN3), gels were stained with 0,5% Coomassie Brilliant Blue for 30 minutes and destained in 20% methanol and 10% acetic acid. Gelatinolytic activity was observed as a clear band of digested gelatine on a blue background. Images were acquired with a digital camera (Nikon,Tokyo, Japan), and bands were analyzed on the image analysis system Gel Doc 200 System (Bio-Rad) using the Quantity One Quantitation Software (Bio-Rad).
Western Blot Analysis of Total and Phosphorylated ERK (pERK) Expression
Total and phosphorylated ERK (pERK) expression was investigated by Western blot analysis. Cytotrophoblast cells treated with anti-CagA Ab (50 μg/mL) or irrelevant control antibody (50 μg/mL) for 4 hours were collected and lysed with cold lysis buffer (1 mmol/L MgCl2, 350 mmol/L NaCl, 20 mmol/L HEPES, 0.5 mmol/L EDTA, 0.1 mmol/L EGTA, 1 mmol/L DTT, 1 mmol/L Na4P2O7, 1 mmol/L PMSF, 1 mmol/L aprotinin, 1.5 mmol/L leupeptin, 20% glycerol, 1% NP-40). Total cell proteins (80 μg) were subjected to electrophoresis on 10% polyacrylamide gel and after electroblotting onto PVDF membrane incubated with 5% nonfat dry milk in TBST 1× and then exposed overnight at 4 °C to TBST containing 0.2–0.4 μg/mL of primary antibody to total ERK or pERK (anti-ERK polyclonal and anti-pERK monoclonal IgG antibodies, Santa Cruz laboratories). Following incubation with secondary antibody, the immunocomplexes were visualized with ECL-Plus detection System (Amersham, GE Healthcare Bio-Sciences Corp, NJ, US) according to the instruction of the manufacturer. Bands were analyzed on the image analysis system Gel Doc 200 System (Bio-Rad Laboratories) and quantified with the Quantity One quantitation Software (Bio-Rad). The levels of total or pERK were estimated versus the constant level of a 42-kD protein present in the cytosolic extract (β-actin; mouse monoclonal, Sigma-Aldrich; data not shown).
Measurement of Nuclear Factor-kB DNA-binding Activity
DNA-binding activity of Nuclear Factor-kB (NF-kB) was measured with a sensitive multi-well colorimetric assay (Transcription Factor Assay kit; Millipore, Temecula, CA, USA). Briefly, cytotrophoblast cells were treated with anti-CagA Ab (50 μg/mL) or irrelevant control antibody (50 μg/mL) for 4 hours. Subsequently cells were scraped and centrifuged for 10 minutes at 400 g. The pellet was re-suspended in 100 μL of lysis buffer, and the lysate was centrifuged for 20 minutes at 20,000 g. Supernatant represented the total protein extract, and the remaining pellet contained the nuclear portion of cell lysate. The nuclear pellet was resuspended in ice-cold nuclear extraction buffer for 30–60 minutes at 4 °C and then centrifuged at 22,000 g for 5 minutes. The nuclear extracts in the supernatant (5 μg/5μL) from each sample were incubated in 96-well plates coated with NF-kB consensus double-stranded oligonucleotide sequence (5′-AGTTGAGGGGACTTTCCCAGGC-3′) for 1 hour, then with primary NF-kB antibody (1:500) for 1 hour, then with peroxidase-conjugated secondary antibody (1:500) for 1 hour, and subsequently with peroxidase- conjugated secondary antibody (1:1000) for 1 hour at room temperature. After the colorimetric reaction, optical density was read at 450 nm. For competition assays, cell extracts were incubated with 22-bp double-stranded DNA, either wild-type or mutated (5′-AGTTGAGCTCACTTTCCCAGGC-3′; underline denotes the substitution).
Immunoprecipitation
A cytotrophoblast cell line, placental, myometrial and ovarian tissues were lysed in ice-cold PBS buffer containing 1% nonylphenol polyoxyethylene 40 (NP-40), 100 mmol/L Na3VO4, 5 mmol/L EGTA, 10 mmol/L Ma4P2O7, 100 mmol/L phenylmethylsulfonyl fluoride (PMSF), and 2 μgr/mL each of leupeptin, pepstatin, and aprotinin. Samples were then incubated on ice for 30 minutes, cleared of insoluble debris by centrifugation at 13,000 × g for 20 minutes at 4 °C, and diluted with PBS to obtain a final concentration NP-40 of 0.5%. Immunoprecipitation was performed by incubating the lysates overnight at 4 °C with mouse polyclonal anti-CagA Ab or normal mouse IgG as control (Bio-Rad, Hercules, CA, USA), followed by incubation with 100 μL protein A/G Agarose (Santa Cruz Biotechnology, Inc, Santa Cruz, CA, USA) for 1 hour at 4 °C. After three washes with PBS buffer containing protease inhibitors, precipitated proteins were separated by SDS-PAGE and revealed with Coomassie Brilliant Blue staining.
Protein Sequencing
ESI LTQ-OT MS was performed on a LTQ Orbitrap XL from Thermo Electron (San Jose, CA, USA) equipped with a NanoAcquity system from Waters. Peptides were trapped on a home-made 5-μm 200 Å Magic C18 AQ (Michrom) 0.1 × 20 mm pre-column and separated on a home-made 5-μm 100 Å Magic C18 AQ (Michrom) 0.75 × 150 mm column with a gravity-pulled emitter. The analytical separation was run for 65 minutes using a gradient of H2O/FA 99.9%/0.1% (solvent A) and CH3CN/FA 99.9%/0.1% (solvent B). The gradient was run as follows: 0–1 minutes 95% A and 5% B, then to 65% A and 35% B at 55 minutes, and 20% A and 80% B at 65 minutes at a flow rate of 220 nL/min. For MS survey scans, the OT resolution was set to 60,000 and the ion population was set to 5E5 with an m/z window from 400 to 2000. Five precursor ions were selected for collision-induced dissociation (CID) in the LTQ. For this, the ion population was set to 1E4 (isolation width of 2 m/z). The normalized collision energies were set to 35% for CID.
Protein identification peak lists were generated from raw data using the embedded software from the instrument vendor (extract_MSN.exe). The monoisotopic masses of the selected precursor ions were corrected using an in-house written Perl script [23]. The corrected mgf file was searched against the UniProt database (v15.4 of 16-June-2009, selected for Homo sapiens, containing 87,630 entries) using the software Mascot (http://www.matrixscience.com). The parent ion tolerance was set to 10 ppm, fragment to 0.6 Daltons. Variable amino acid modifications were oxidized methionine. Trypsin was selected as the enzyme, with one potential missed cleavage, and the normal cleavage mode was used. The criteria for protein identification were the following: Scaffold (version 3.00.03, from Proteome Software Inc., Portland, OR) was used to validate MS/MS-based peptide and protein identifications. Peptide identifications were accepted if they could be established at >95.0% probability as specified by the Peptide Prophet algorithm [24]. Protein identifications were accepted if they could be established at >95.0% probability and contained at least two identified peptides. Protein probabilities were assigned by the Protein Prophet algorithm [25]. Proteins that contained similar peptides and could not be differentiated based on MS/MS analysis alone were grouped to satisfy the principles of parsimony.
Immunofluorescence Staining
Trophoblast cells were rinsed twice in PBS and fixed with 4% PFA 5 minutes at room temperature. Next, the cells were incubated with both primary antibodies against β-actin (Sigma – Aldrich, 20 ugr/mL in PBS) with or without anti-CagA Ab (200 μgr/mL in PBS), for 1 hour at room temperature. After three washes in PBS, the secondary antibody for anti–CagA conjugated to FITC was added for 1 hour at room temperature (working solution 1:200 in PBS). The cells were then rinsed twice in PBS, mounted under microscopic glass coverslips in a Gel/Mount permanent fluorescence-preserving aqueous mounting medium (Biomeda Corporation), and evaluated under an inverted phase fluorescent microscope (Zeiss, 200×). Images were acquired with a digital camera (Nikon, Tokyo, Japan).
Results
Anti-CagA Ab Binding to Trophoblast Cells
To demonstrate the anti-CagA Ab binding to trophoblast cells, a cell ELISA was performed. The highest binding was seen at 72 hours of culture, when the largest differentiation of cytotrophoblast into syncytiotrophoblast was found [26]. Through cell-based ELISA, we observed that increasing doses of anti-CagA Ab (6–12–50–200 μg/mL) showed a dose-dependent binding activity with protein concentration as low as 12 μg/mL (n = three experiments; Fig. 1A). In contrast, normal mouse IgG, used as control, did not display any significant cell binding in this assay.
Figure 1.

Anti-CagA antibodies bind trophoblast cells in a dose-dependent manner (A). Indirect immunofluorescence assay performed on trophoblast cells using anti-CagA antibodies displayed a significant reactivity that was confined to the plasma cell membrane (B).
To further confirm the anti-CagA Ab binding, an indirect immunofluorescence assay was performed. As shown in the Fig. 1B, cytotrophoblast cells in the presence of anti-CagA Ab IgG displayed a significant reactivity that was confined to the plasma cell membrane.
Anti-CagA Ab Effects on Trophoblast Invasion
Trophoblast differentiation is characterized by the development of extravillous trophoblasts that migrates into maternal myometrium. To explore whether trophoblast invasiveness was affected by the presence of anti-CagA Ab IgG, we examined the invasion activity using a membrane invasion culture system, in presence of increasing concentrations of anti-Cag Ab. Figure 2 shows the results of a 72 hours in vitro Matrigel invasion assay: polyclonal anti-CagA Ab, but not normal mouse IgG, was able to significantly impair trophoblast cell invasiveness in a dose-dependent manner. This effect was detectable at the lowest protein concentration of 12 μgr/mL (p < .05).
Figure 2.
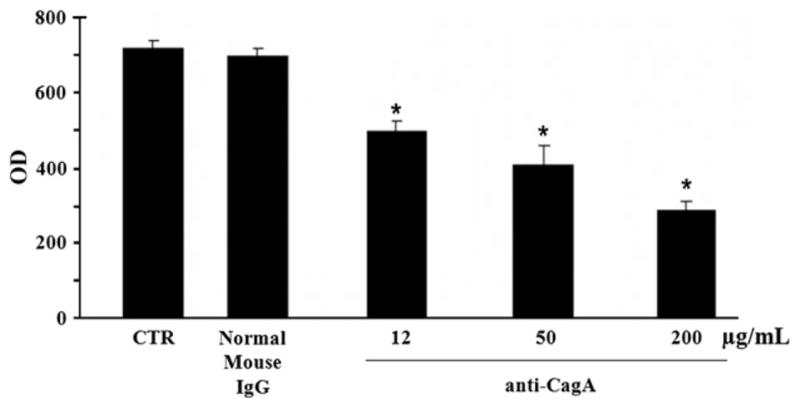
Matrigel invasion assay: polyclonal anti-CagA antibodies, but not normal mouse IgG impair trophoblast cell invasiveness in a dose-dependent manner.
Gelatin Zymography: Anti-CagA Ab Effect on MMP-2 Expression
Matrix metalloproteinases-2 (MMP-2) represents a degradative enzyme involved in the process of trophoblast invasion. To investigate whether anti-Cag Ab are able to impair this process affecting MMP-2 activity, we performed a gelatin zymography assay. Incubation with IgG anti-CagA Ab (12–200 μg/mL) significantly reduced both pro-MMP-2 and active MMP-2 (Fig. 3). MMP-2 in the presence of normal mouse IgG was at the same levels observed in untreated trophoblast cells (data not shown).
Figure 3.

Gelatin zymography assay: incubation of trophoblast cells with anti-CagA antibodies significantly reduced both pro-MMP-2 and active MMP-2.
Effect on ERK 1/2 Activation
To better understand the molecular mechanisms involved in the anti-CagA inhibited cytotrophoblast invasiveness, we investigated the possible role of MAP Kinases (MAPKs). It has been reported that MAPKs activation results in phosphorylation of multifunctional protein kinases, including ERKs. The activation of ERK pathway is required for promoting the proliferation and invasiveness of cytotrophoblast cells [27].
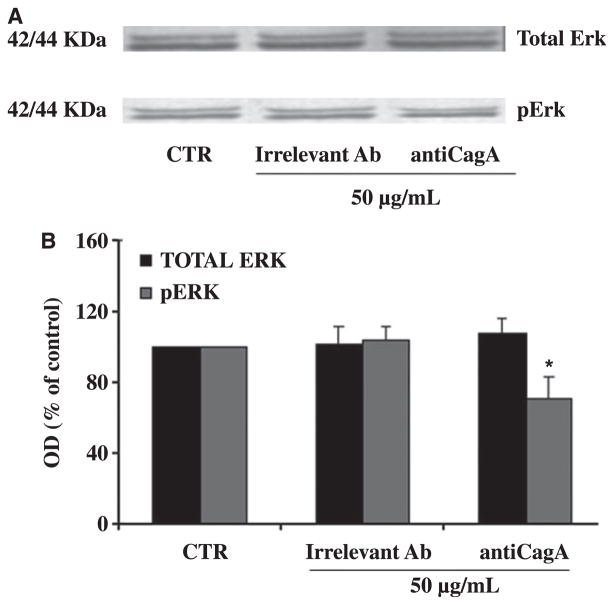
Western blot analysis showed a significant decrease in phosphorylated ERK after treatment with IgG anti-CagA Ab (50 μg/mL), but not after treatment with irrelevant control antibody (Fig. 4A,B). Total form expression was not modified in presence of anti-CagA Ab.
Figure 4.
Western blot analysis of total and phosphorylated ERK expression in cytotrophoblast cells treated with anti-CagA Ab (50 μg/mL). (A) A representative Western Blot analysis for total and phosphorylated ERK in cytotrophoblast cells. (B) Densitometric analysis. The level of total p-ERK protein after anti-CagA Ab treatment was estimated in comparison with the constant level of beta-actin and expressed as percentage of the control (untreated cells = 100%). A significant decrease in phosphorylated ERK-1 and -2, in presence of IgG anti-CagA Ab (50 μg/mL), but not in presence of irrelevant control antibody was observed. Total form expression was not modified in presence of anti-CagA Ab. Results are means ± SE of three independent experiments. Significance versus untreated cells (control): *p < .05.
NF-kB Translocation Activity
NF-κB plays a key role in promoting cell proliferation, survival, and invasion [28]. In this study, to determine the influence of anti-CagA Ab on NFkB activation, we studied the translocation of NFkB and its DNA binding. We observed that polyclonal anti-CagA Ab (Anti-CagA, 50 μg/mL), but not irrelevant control antibody, reduced NF-kB translocation (85% vs control 100%, Fig. 5).
Figure 5.
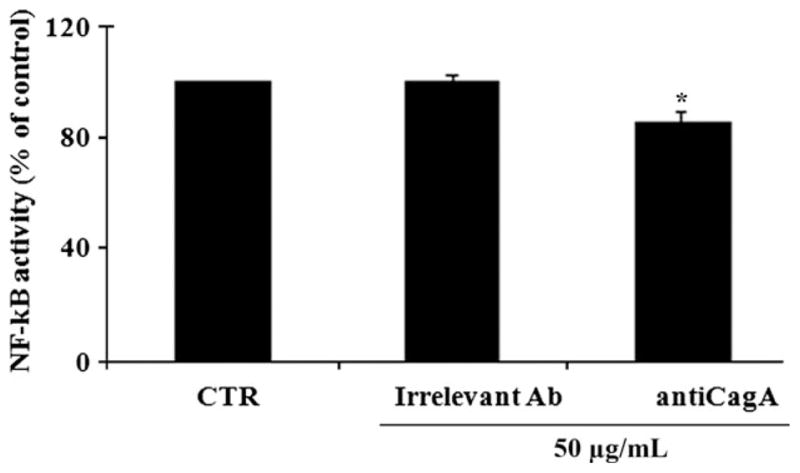
Effects of anti-CagA on nuclear factor NF-kB DNA-binding activity in trophoblast cells. Polyclonal anti-CagA Ab (Anti-CagA, 50 μg/mL), but not irrelevant control antibody, reduced NF-kB translocation.
Competition experiments performed with a 100-fold excess of unlabeled KB oligonucleotide demonstrated the specificity of DNA-binding activity (data not shown).
Immunoprecipitation
To identify the cellular surface component reacting specifically with the anti-CagA Ab, a immunoprecipitation was performed. Anti-CagA Ab immunoreacted with the trophoblast cell line, whereas no immunoreaction was observed with placental, myometrial, and ovarian tissues. The anti-CagA antigen from cellular lysates was identified as a 43-KDa band (Fig. 6). Similarly, bacterial lysates, expressing CagA antigen, consistently showed a band at approximately 130 KDa (data not shown). No antigens were precipitated by normal mouse IgG, confirming the specificity of the reaction.
Figure 6.
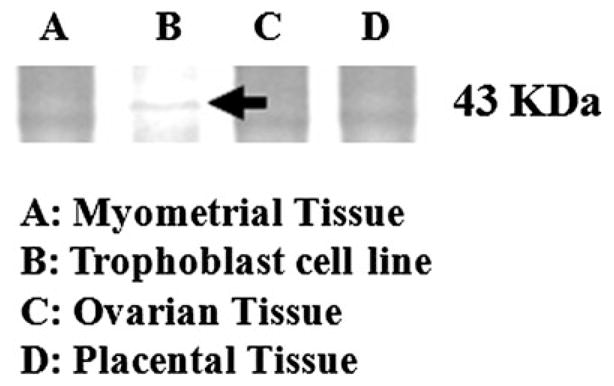
Immunoprecipitation performed on trophoblast, myometrial, ovarian and placental tissue lysates using anti-CagA antibodies revealed the presence of a cross-reactive band only for trophoblast cells.
Protein Sequencing
The protein from trophoblast cell lysate recognized by anti-CagA antibodies through immunoprecipitation was shown to be β-actin. This experiment has been repeated three times to avoid false positive or negative.
Immunofluorescence Staining
As resulting from protein sequencing, we identified the protein β-actin as the possible membrane component able to cross-react with the anti-CagA Ab. Although β-vactin is more commonly known as an intracellular protein, it has also been reported on the surface of endothelial-like cells [29–31]. The detection of anti-Cag Ab binding to trophoblast cell membrane allowed us to investigate whether the anti-CagA Ab binding to trophoblasts was reduced by previous treatment with anti-β actin Ab.
First, the anti-CagA Ab binding, evaluated by indirect immunofluorescence on human trophoblast cells, was confirmed to be confined to the peripheral cellular membranes (Fig. 7A).
Figure 7.
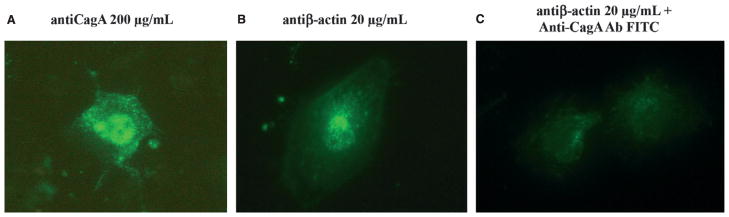
Binding of anti-CagA (A) and anti-β-actin (B) antibodies on human trophoblast cells evaluated by indirect immunofluorescence. In the panel C trophoblast cells were exposed first to anti-β-actin and secondly to anti-CagA antibodies conjugated to FITC. Under such conditions a lower fluorescence was detected on trophoblast cells, suggesting that the antigen was masked by the anti-β-actin antibodies previously bound to cellular membranes.
Then, we examined anti-β actin Ab binding through indirect immunofluorescence: Fig. 7B shows the anti-β actin Ab activity. We found a fluorescent pattern identical to Anti-CagA immunofluorescence staining confined on cellular membranes of nonpermeabilized cells appearing as a microspot-like staining surrounding the cells.
Finally, trophoblast cells were exposed first to anti-β actin and secondly to anti-CagA Ab conjugated to FITC. Under such conditions, a lower fluorescence was detected on trophoblast cells, suggesting that the antigen was masked by the anti-β-actin Ab previously bound to cellular membranes (Fig. 7C).
Overall, these studies suggest that anti-CagA Ab binding might be due to its cross-reaction with β-actin expressed on trophoblast surface.
Discussion
Several studies have shown an epidemiological association between PE and the occurrence of cardiovascular disease later in the life [20]. Interestingly, for both diseases, a possible role of CagA-positive H. pylori infection has been proposed [15,17]. For IHD, one of the most accredited theory is that anti-CagA antibodies may cross-react with endothelial cell antigens as well as with other peptides localized inside atherosclerotic plaque, thus triggering plaque activation and instability [15]. As trophoblast shows an endothelial origin, we tested the hypothesis that anti-CagA antibodies may also recognize antigens localized on the surface of trophoblast cells, possibly representing a link between those pathological conditions.
Data from this study show, for the first time, that anti-CagA antibodies are able to recognize β-actin on the surface of trophoblast cells. Interestingly, anti-CagA antibodies showed a dose-dependent binding activity; it follows that the bioavailability of the cross-reactive protein expressed by trophoblast cells may be high enough to produce a biological effect. This effect, in our study, consisted in the impairment of cytotrophoblast invasiveness in vitro, a crucial point for PE development. To better understand the molecular mechanisms involved in the antibody-mediated effect, we examined the effect of anti-CagA on ERK activation and NF-kB nuclear translocation, two important factors activated during trophoblast proliferation. We observed that the anti- CagA antibodies are able to inhibit the activation of both elements, thus suggesting a possible explanation for the reduced trophoblast invasiveness observed pregnancy disorders related to an impaired placental development. Furthermore, immunofluorescence performed on trophoblast cells using either anti-CagA or antiβ-actin antibodies showed an identical pattern of reaction thus confirming β-actin to be the real cross-reacting protein. Interestingly, actin in either endothelial or trophoblast cells is not only important for maintaining the cell structure but is also crucial for intercellular adhesion. Moreover, it is now well established that actinassociated adhesions contribute to placental anchorage. The phenomenon of implantation anchors the blastocyst into the uterine wall and produces a hemochorial placenta that maintains pregnancy and fetal growth. Implantation and placentation are intimately linked and represent a crucial step; PE, in fact, may be a consequence of a defective implantation [32].
Finally, the fact that anti-CagA antibodies bound β-actin of trophoblast cells in a dose-dependent manner may contribute to explain why not all patients with CagA-positive H. pylori infection may develop PE. For IHD, it has been proposed that the higher is the titer of anti-CagA antibodies in the serum, the higher is the probability to produce a biological effect in coronary arteries [15]. As anti-CagA antibody titer has been shown to vary over the course of the infection [33,34], we may speculate that it may enhance the risk of PE development only when markedly increased during pregnancy.
In conclusion, this study demonstrated for the first time the presence of a cross-reactivity between anti- CagA antibodies and β-actin of trophoblast cells, thus providing an additional biological explanation for the previously described epidemiological association between CagA positivity and PE. Further studies are now needed to confirm our findings.
Footnotes
Competing interests: the authors have no competing interests.
References
- 1.Shenoy V, Kanasaki K, Kalluri R. Pre-eclampsia: connecting angiogenic and metabolic pathways. Trends Endocrinol Metab. 2010;21:529–36. doi: 10.1016/j.tem.2010.05.002. [DOI] [PubMed] [Google Scholar]
- 2.Steegers EA, von Dadelszen P, Duvekot JJ, et al. Pre-eclampsia. Lancet. 2010;376:631–44. doi: 10.1016/S0140-6736(10)60279-6. [DOI] [PubMed] [Google Scholar]
- 3.James JL, Whitley GS, Cartwright JE. Pre-eclampsia: fitting together the placental, immune and cardiovascular pieces. J Pathol. 2010;221:363–78. doi: 10.1002/path.2719. [DOI] [PubMed] [Google Scholar]
- 4.Macey MG, Bevan S, Alam S, et al. Platelet activation and endogenous thrombin potential in pre-eclampsia. Thromb Res. 2010;125:e76–81. doi: 10.1016/j.thromres.2009.09.013. [DOI] [PubMed] [Google Scholar]
- 5.Horton AL, Boggess KA, Moss KL, et al. Periodontal disease, oxidative stress, and risk for preeclampsia. J Periodontol. 2010;81:199–204. doi: 10.1902/jop.2009.090437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Xie F, Hu Y, Magee LA, et al. An association between cytomegalovirus infection and pre-eclampsia: a case-control study and data synthesis. Acta Obstet Gynecol Scand. 2010;89:1162–7. doi: 10.3109/00016349.2010.499449. [DOI] [PubMed] [Google Scholar]
- 7.Marshall BJ, Barrett LJ, Prakash C, et al. Urea protects Helicobacter (Campylobacter) pylori from the bactericidal effect of acid. Gastroenterology. 1990;99:697–702. doi: 10.1016/0016-5085(90)90957-3. [DOI] [PubMed] [Google Scholar]
- 8.Gasbarrini G, Malfertheiner P, Deltenre M, et al. New concepts concerning management of Helicobacter pylori infection: 2 years after the Maastricht Consensus Report. Ital J Gastroenterol Hepatol. 1998;30:S244–7. [PubMed] [Google Scholar]
- 9.Crabtree JE, Kersulyte D, Li SD, et al. Modulation of Helicobacter pylori induced interleukin-8 synthesis in gastric epithelial cells mediated by cag PAI encoded VirD4 homologue. J Clin Pathol. 1999;52:653–7. doi: 10.1136/jcp.52.9.653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Nguyen LT, Uchida T, Tsukamoto Y, et al. Clinical relevance of cagPAI intactness in Helicobacter pylori isolates from Vietnam. Eur J Clin Microbiol Infect Dis. 2010;29:651–60. doi: 10.1007/s10096-010-0909-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sugimoto M, Ohno T, Graham DY, et al. Gastric mucosal interleukin- 17 and-18 mRNA expression in Helicobacter pyloriinduced Mongolian gerbils. Cancer Sci. 2009;100:2152–9. doi: 10.1111/j.1349-7006.2009.01291.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Pellicano R, Franceschi F, Saracco G, et al. Helicobacters and extragastric diseases. Helicobacter. 2009;14(Suppl 1):58–68. doi: 10.1111/j.1523-5378.2009.00699.x. [DOI] [PubMed] [Google Scholar]
- 13.Franceschi F, Gasbarrini A. Helicobacter pylori and extragastric diseases. Best Pract Res Clin Gastroenterol. 2007;2:325–34. doi: 10.1016/j.bpg.2006.10.003. [DOI] [PubMed] [Google Scholar]
- 14.Franceschi F, Sepulveda AR, Gasbarrini A, et al. Cross-reactivity of anti-CagA antibodies with vascular wall antigens: possible pathogenic link between Helicobacter pylori infection and atherosclerosis. Circulation. 2002;106:430–4. doi: 10.1161/01.cir.0000024100.90140.19. [DOI] [PubMed] [Google Scholar]
- 15.Franceschi F, Niccoli G, Ferrante G, et al. CagA antigen of Helicobacter pylori and coronary instability: insight from a clinicopathological study and a meta-analysis of 4241 cases. Atherosclerosis. 2009;202:535–42. doi: 10.1016/j.atherosclerosis.2008.04.051. [DOI] [PubMed] [Google Scholar]
- 16.Panarelli M, Sattar N. Pre-eclampsia associated with Helicobacter pylori seropositivity. J Hypertens. 2006;24:2353–4. doi: 10.1097/HJH.0b013e3280113638. [DOI] [PubMed] [Google Scholar]
- 17.Ponzetto A, Cardaropoli S, Piccoli E, et al. Pre-eclampsia is associated with Helicobacter pylori seropositivity in Italy. J Hypertens. 2006;24:2445–9. doi: 10.1097/HJH.0b013e3280109e8c. [DOI] [PubMed] [Google Scholar]
- 18.UstUn Y, Engin-UstUn Y, Ozkaplan E, et al. Association of Helicobacter pylori infection with systemic inflammation in pre-eclampsia. J Matern Fetal Neonatal Med. 2010;23:311–4. doi: 10.3109/14767050903121456. [DOI] [PubMed] [Google Scholar]
- 19.Pugliese A, Beltramo T, Todros T, et al. Interleukin-18 and gestosis: correlation with Helicobacter pylori seropositivity. Cell Biochem Funct. 2008;26:817–9. doi: 10.1002/cbf.1503. [DOI] [PubMed] [Google Scholar]
- 20.Bellamy L, Casas JP, Hingorani AD, et al. Pre-eclampsia and risk of cardiovascular disease and cancer in later life: systematic review and meta-analysis. BMJ. 2007;335:974. doi: 10.1136/bmj.39335.385301.BE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Aksoy H, Ozkan A, Aktas F, et al. Helicobacter pylori seropositivity and its relationship with serum malondialdehyde and lipid profile in preeclampsia. J Clin Lab Anal. 2009;23:219–22. doi: 10.1002/jcla.20330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Di Simone N, Caliandro D, Castellani R, et al. Interleukin-3 and human trophoblast: in vitro explanations for the effect of interleukin in patients with antiphospholipid antibody syndrome. Fertil Steril. 2000;73:1194–200. doi: 10.1016/s0015-0282(00)00533-1. [DOI] [PubMed] [Google Scholar]
- 23.Scherl A, Tsai YS, Shaffer SA, et al. Increasing information from shotgun proteomic data by accounting for misassigned precursor ion masses. Proteomics. 2008;8:2791–7. doi: 10.1002/pmic.200800045. [DOI] [PubMed] [Google Scholar]
- 24.Keller A, Nesvizhskii AI, Kolker E, et al. Empirical statistical model to estimate the accuracy of peptide identifications made by MS/MS and database search. Anal Chem. 2002;74:5383–92. doi: 10.1021/ac025747h. [DOI] [PubMed] [Google Scholar]
- 25.Nesvizhskii AI, Keller A, Kolker E, et al. A statistical model for identifying proteins by tandem mass spectrometry. Anal Chem. 2003;75:4646–58. doi: 10.1021/ac0341261. [DOI] [PubMed] [Google Scholar]
- 26.Di Simone N, Marana R, Castellani R, et al. Decreased expression of heparin-binding epidermal growth factor-like growth factor as a newly identified pathogenic mechanism of anti-phospholipid-mediated defective placentation. Arthritis Rheum. 2010;62:1504–12. doi: 10.1002/art.27361. [DOI] [PubMed] [Google Scholar]
- 27.Zhou WH, Du MR, Dong L, et al. Cyclosporin A increases expression of matrix metalloproteinase 9 and 2 and invasive-ness in vitro of the first-trimester human trophoblast cells via the mitogen-activated protein kinase pathway. Hum Reprod. 2007;22:2743–50. doi: 10.1093/humrep/dem097. [DOI] [PubMed] [Google Scholar]
- 28.Torchinsky A, Toder V. To die or not to die: the function of the transcription factor NF-kB in embryos exposed to stress. Am J Reprod Immunol. 2004;51:138–43. doi: 10.1046/j.8755-8920.2003.00134.x. [DOI] [PubMed] [Google Scholar]
- 29.Pardridge WM, Nowlin DM, Choi TB, et al. Brain capillary 46,000 dalton protein is cytoplasmic actin and is localized to endothelial plasma membrane. J Cereb Blood Flow Metab. 1989;9:675–80. doi: 10.1038/jcbfm.1989.95. [DOI] [PubMed] [Google Scholar]
- 30.Aplin JD, Jones CJP, Harris LK. Adhesion molecules in human trophoblast – a review. I. Villous trophoblast. Placenta. 2009;30:293–8. doi: 10.1016/j.placenta.2008.12.001. [DOI] [PubMed] [Google Scholar]
- 31.Aplin JD, Jones CJP, Harris LK. Adhesion molecules in human trophoblast – a review. II. Extravillous trophoblast. Placenta. 2009;30:299–304. doi: 10.1016/j.placenta.2008.12.003. [DOI] [PubMed] [Google Scholar]
- 32.Merviel P, Carbillon L, Challier JC, le Rabreau M, Beaufils M, Uzan S. Pathophysiology of preeclampsia: links with implantation disorders. Eur J Obstet Gynecol Reprod Biol. 2004;115:134–47. doi: 10.1016/j.ejogrb.2003.12.030. [DOI] [PubMed] [Google Scholar]
- 33.Figueiredo C, Quint W, Nouhan N, et al. Assessment of Helicobacter pylori vacA and cagA genotypes and host serological response. J Clin Microbiol. 2001;39:1339–44. doi: 10.1128/JCM.39.4.1339-1344.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Peters TM, Owen RJ, Slater E, et al. Genetic diversity in the Helicobacter pylori cag pathogenicity island and effect on expression of anti-CagA serum antibody in UK patients with dyspepsia. J Clin Pathol. 2001;54:219–23. doi: 10.1136/jcp.54.3.219. [DOI] [PMC free article] [PubMed] [Google Scholar]