Abstract
Many of the synthetic biological devices, pathways and systems that can be engineered are multi-use, in the sense that they could be used both for commercially-important applications and to help meet global health needs. The on-going development of models and simulation tools for assembling component parts into functionally-complex devices and systems will enable successful engineering with much less trial-and-error experimentation and laboratory infrastructure. As illustrations, I draw upon recent examples from my own work and the broader Keasling research group at the University of California Berkeley and the Joint BioEnergy Institute, of which I was formerly a part. By combining multi-use synthetic biology research agendas with advanced computer-aided design tool creation, it may be possible to more rapidly engineer safe and effective synthetic biology technologies that help address a wide range of global health problems.
Keywords: Model-driven design and engineering, Computer-aided design
Applied synthetic biology
Synthetic biological systems constructed through genetic engineering (Carothers et al. 2009) may help meet global health needs by providing routes to discover and produce low cost nutrients, drugs, vaccines and medical diagnostic materials (Gostin 2007; Keasling 2010). Creating approaches that minimize the time and resources required to engineer novel systems will be essential, particularly for applications targeting diseases and conditions of poverty in low and middle-income countries (Towse and Kettler 2005). With regard to safety, security and bioterrorism, ‘multi-use’ synthetic biology is often seen as a pejorative (Koblentz 2009)—something to guard against. But, in terms of the potential for using synthetic biology for global health, multi-use may be the ‘feature, not the bug’.1 Here, I argue that research agendas advancing design-driven, multi-use synthetic biology may lead to efficient platforms and tools for addressing a variety of global health challenges.
4,000-year-old Egyptian depictions of fermentation to make beer show that humans have a long history of harnessing the biotransformation potential of microorganisms (Rosicrucian Egyptian Museum 2012). In the modern era, microbial biotransformations have played crucial roles in producing materials for health and medicine; the successful development of penicillin between the years of 1928 and 1942, is one of the earliest, and best-known, examples. Remarkably, following the initial clinical trials, large-scale penicillin production was obtained in only 4 years, with yields increasing from 1 % in bench-top, 1 L cultures to 80–90 % in 38,000 L tanks (American Chemical Society 2012).
The development of recombinant DNA technologies in the 1970s meant that microbes could be purposefully constructed to produce targeted compounds, as compared to being restricted to the relatively small set of molecules that could be produced in large-scale cultures from naturally-occurring (or randomly mutagenized) organisms (Carothers et al. 2009). The successful production of human insulin from an engineered strain of the common gut bacterium Eschericia coli by Genentech, Inc. in 1982 was a landmark accomplishment. Roughly speaking, the challenge was to engineer a genetic cassette to overexpress a single gene product that was subsequently purified from the E. coli cell. The result was an insulin supply free of dangerous porcine-related antigens and a vivid demonstration of the potential for using genetic engineering to address health needs (Johnson 1983).
Synthetic biological systems are comprised of large numbers of components with activities that must be precisely coordinated to obtain the desired functions (Holtz and Keasling 2010). As a consequence, the challenges involved in engineering synthetic biological systems are fundamentally different from those associated with constructing microbial platforms to overproduce a single therapeutic protein like insulin. To obtain high levels of production, much of the engineering effort must be aimed at solving the embedded pathway and system control problems, such as preventing the buildup of toxic intermediates or the wasteful overexpression of pathway enzymes (Carothers et al. 2009). For example, engineering E. coli and yeast platforms to produce the antimalarial compound, artemisinin (Ro et al. 2006) (Westfall et al. 2012), required carefully balancing the expression levels and activities of 8 to 11 different gene products. In practice, thousands of iterations of laboratory testing and analysis (and dozens of person-years) were necessary to assemble more than 30 unique genetic elements and give yields high enough to support economically-viable production (Westfall et al. 2012).
There are four take-home messages: (1) humans have a long history of using microbes for specific applications (2) there is a 30+ year track record of safely engineering microorganisms for health and medicine (3) the challenges involved in engineering synthetic biological systems are fundamentally different from those involved in engineering microbial systems to overproduce individual therapeutic proteins and (4) many resources, in the forms of people, laboratory infrastructure and money have been essential for engineering synthetic biological systems for novel applications. Next, I will examine how the material and human resources necessary for synthetic biology applications in global health can be reduced by emphasizing the development of multi-use biological devices, platforms, and systems.
Multi-use platforms for applied synthetic biology
Artemisinin is just one compound in a family of more than 100,000 isoprenoid-pathway derived molecules (Chang and Keasling 2006). Many terpenes are encountered in foods (e.g. lycopene is the red color in tomatoes) and smells (e.g. limonene is a component of lemon scent), while others are pharmacologically-active or have chemical properties that make them good candidates for bio-based diesel and jet fuel replacements (Lee et al. 2008). Because isoprenoids are structurally- and functionally-diverse, the artemisinin production systems are very good case studies in multi-use synthetic biology. In this regard, it is noteworthy that Amyris, Inc. is building a business for producing isoprenoid-based chemicals and fuels by reengineering the pathways and yeast chassis, or platform hosts, that were initially engineered to produce artemisinin. That is, engineering systems for high-titer isoprenoid production led not only to a new source of the antimalarial compound, but also enabled the construction of industrial biotechnology platforms with significant commercial value (Dietrich et al. 2011). Separately, efforts to engineer synthetic biological systems to produce isoprenoid-based fuels such as bisabolene (Peralta-Yahya et al. 2011) from renewable lignocellulosic materials (Bokinsky et al. 2011) are now major projects of the US Department of Energy Joint BioEnergy Institute.
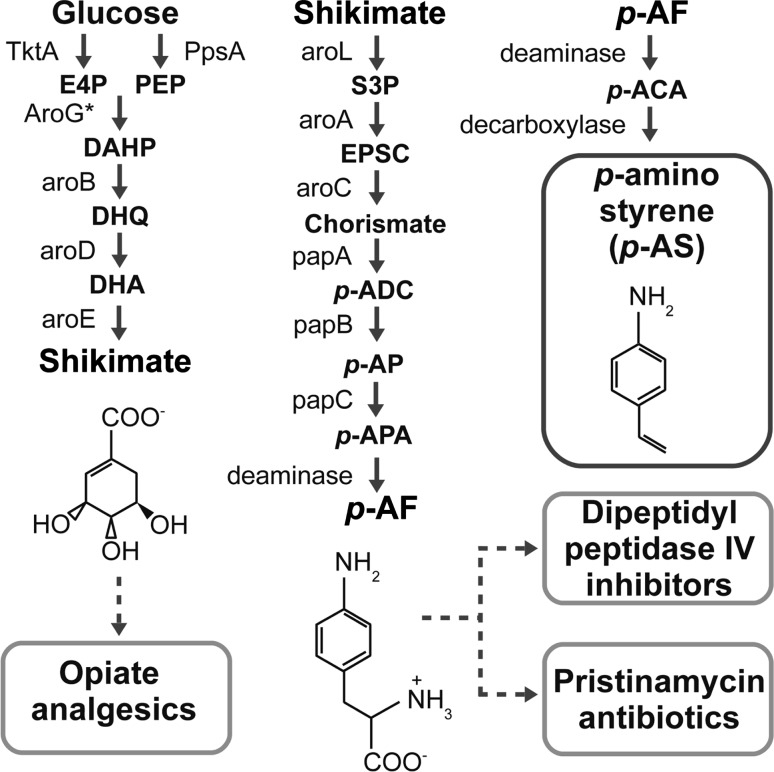
In fact, there are many devices, pathways, and systems with multi-use potential. As a second example, I initiated a project to engineer an E. coli platform for p-aminostyrene (p-AS) production. p-AS is a vinyl aromatic whose co-polymers have optical and mechanical properties that may be useful for a number of advanced applications, including as materials for photonics, photovoltaics and biomedicine (Goikhman et al. 2011). In principle, p-AS can be synthesized in 14 enzymatic steps; 12 steps to convert sugar (glucose) to p-aminophenylalanine (p-AF) and two steps to convert p-AF to p-AS (Fig. 1). p-AF is both the immediate chemical precursor of the pristinamycin family of antibiotics (Blanc et al. 1997; Reid et al. 2010) and novel compounds investigated as diabetes treatments (Duffy et al. 2007). Several of the immediate chemical precursors of p-AF are themselves starting points for synthesizing potent analgesic compounds (Juminaga et al. 2012). Above, I described how the commercial development of isoprenoid-based chemicals and fuels was coupled to the development of the artemisinin production system. Similarly, the development of reengineered platforms for reliable, low cost antibiotic or analgesic production could be supported by coupling those projects to the creation of platforms for producing high-value, industrially-relevant aromatic compounds (Sariaslani 2007).
Fig. 1.
Eschericia coli platform for p-aminostyrene production. Many synthetic biological devices, pathways and systems have multi-use potential; they can be used to produce both industrially-relevant compounds and low cost materials for global health. As shown, metabolic pathways constructed to produce industrially-relevant aromatics (p-aminostryene, p-AS) could be reengineered to producelow cost drugs, such as painkillers (opiate analgesics), experimental type II diabetes treatments (dipeptidyl peptidase IV inhibitors) and antibiotics (pristinamycin antibiotics). Gene products and molecule names are as described (Carothers et al. 2011)
Progress toward design-driven synthetic biology
Models and simulation tools for designing and assembling components into functionally complex devices and systems are the hallmarks of most engineering fields. Identifying the relevant design parameters and defining the domains over which expected component behaviors are exerted have been key steps in the development of these technologies (Goler et al. 2008). For instance, early progress in the 1970s toward electronic design automation, which can now be used to assemble integrated circuits with millions of transistors, was predicated upon techniques for simulating modular network responses and geometric rules for specifying physical implementations (Mead and Conway 1980). In chemical engineering, otherwise difficult design problems are made tractable by framing them as unit operations such that complex processes can be evaluated with coarse-grained models (Chau 2002). Biological systems exhibit functional complexity due to component interactions at many different scales, from individual protein and RNA subunits to genes, pathways, circuits and cells (Hazen et al. 2007). In the absence of effective models and simulation tools, it remains difficult and resource intensive to engineer complex biological devices and systems (Keasling 2010).
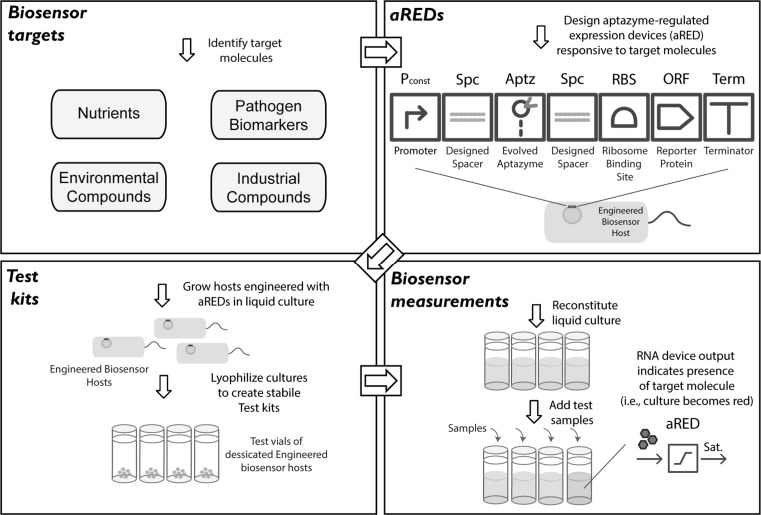
To show that biochemical and biophysical modeling can be used to develop effective biological design methods, we formulated an approach (Fig. 2) for engineering RNA-based genetic control devices with quantitatively-predictable activities (Carothers et al. 2011). We used mechanistic modeling and kinetic RNA folding simulations to design RNA devices that can program gene expression in pathways engineered to produce industrial chemicals, establishing a conceptual and experimental framework for developing computer-aided design (CAD) platforms for complex RNA-based genetic control systems. The excellent quantitative agreement between the predicted and measured gene expression levels (94 % correlation) experimentally validated both the underlying models and the overall design strategy. We have also shown that these devices can be employed as dynamic biosensors and controllers for metabolic pathways and circuits. In principle the same approach can be used to engineer programmable biosensors responsive to almost any targeted molecule (Carothers et al. 2010). The ability to engineer genetic control devices that can couple production to biosensing is expected to increase the sizes and complexities of the systems that can be engineered if individual devices, pathways and circuits can be constructed to dynamically tune expression levels and optimize functions in novel contexts (Holtz and Keasling 2010). Biosensor host cells engineered with RNA-regulated expression devices could function as components of multiplexed, point-of-care diagnostics that detect pathogens or nutritional states and produce genetically-encoded outputs that are easy to observe even in low-resource clinical settings (Yager et al. 2008) (Fig. 3).
Fig. 2.
Design-driven RNA device engineering. We formulated a model-driven approach for engineering RNA-based genetic control devices to program pathway and circuit gene expression, with a 94 % correlation observed between the predicted and measured outputs. This approach could be used to engineer sets of static and dynamic RNA devices and implement novel designs derived from systems-level models (after Carothers et al. 2011). More broadly, this work established generalizable frameworks for integrating biochemical and biophysical modeling and developing biological CAD tools that reduce the time and resources needed to engineer functionally-complex devices, pathways and systems
Fig. 3.
RNA-regulated expression devices as biosensors. Aptazyme-regulated expression devices (aREDs) (Carothers et al. 2011) could be designed for dessicated (Billi et al. 2000) engineered biosensor hosts to function as point-of-care diagnostics that produce genetically-encoded outputs upon detecting target compounds, such as pathogen biosensors or nutritional states
The successful development of our model-driven approach for engineering RNA devices relied upon the application of three generalizable strategies for managing biological complexity and enabling engineering tractability (Hazen et al. 2007). First, a coarse-grained mechanistic model (Chau 2002), based on well-understood biochemistry (Deana et al. 2008), was created to simulate device functions from measurable and tunable component characteristics. Second, global sensitivity analysis (Saltelli et al. 2008) was employed to map the space of feasible designs and focus efforts on design variables that make the largest contributions to device functions. Third, by separating functional design from physical implementation (Mead and Conway 1980) we could investigate biophysical constrains and create a method to enable device assembly from individually-engineered and characterized components.
Naturally-occurring biological systems have evolved control circuitry that couples chemical inputs to genetic outputs in order to minimize the accumulation of toxic intermediates and maintain homeostasis (Carothers et al. 2009). We have demonstrated that models and simulation tools can inform the design and analysis of synthetic control circuitry by engineering E. coli dynamic sensor-regulator systems (DSRS) that produce fatty acid-based chemicals and biodiesel molecules (Zhang et al. 2012). We used mechanistic modeling to interrogate the DSRS design space and identify parameters consistent with targeted biodiesel production performance criteria. Global sensitivity analysis showed that relative improvements in biodiesel production could be obtained across a broad range of parameter values and regulatory topologies. Consistent with the simulation results, DSRS-controlled strains exhibited more genetic stability and produced 3 times more fatty acid ethyl ester biodiesel than strains without the DSRS, with production reaching 28 % of the theoretical maximum, showing that genetic control system engineering can significantly improve production. Protein- or model-driven RNA genetic device engineering that extends DSRS-control to other pathways and systems should be a general procedure for improving the efficiency of biosynthetic production platforms (Zhang and Keasling 2011).
Our results show that it may be possible to use biochemical and biophysical modeling to develop full-fledged CAD platforms for designing complex genetic control systems (Carothers et al. 2011). In turn, by making it easier to implement novel designs, CAD tools can dramatically reduce the number of genetic variants that must be tested, which decreases the amount of large-scale, trial-and-error experimentation and laboratory infrastructure required. By employing algorithms that optimize the process of genetic construction (e.g., identifying ways to reuse genetic material, and otherwise minimize de novo oligonucleotide synthesis), new DNA design software like ‘j5’ (Hillson et al. 2011; Chen et al. 2012) further lessens the volumes and costs of materials and reagents. TeseleGen, Inc., a company developing commercial versions of the j5 DNA design software, has said that their existing design algorithms can reduce the time needed to construct a simple metabolic pathway by 20 % (to approximately 2 weeks), and at half the cost ($700 USD), compared to traditional cloning. They have estimated that a j5-designed combinatorial library of 240 constructs would cost <$30,000 to assemble with an optimized strategy, and take only 1.5 months, compared to the roughly $120,000 cost and 11 months required using traditional cloning. By way of comparison, TeseleGen estimates that direct DNA synthesis of the same 240 constructs would cost $540,000 at current prices. Although peer-reviewed data are not yet available to evaluate the TeseleGen claims, it is clear that these kinds of improvements in model-driven synthetic biology [other recent advances are reviewed in (Koeppl 2011) (Blount et al. 2012), (Medema et al. 2012) and (Shiue and Prather 2012)] will reduce the number of cycles of design, construction and analysis needed to engineer systems that meet performance goals.
Research agendas for global health
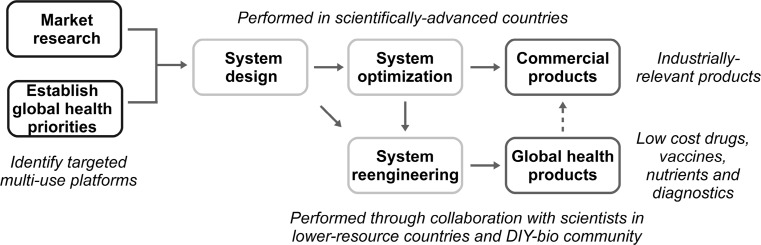
If agreements for licensing and material sharing can be established (Roose-Snyder and Doyle 2009; Oye and Wellhausen 2010), it may be feasible to reengineer devices and systems for a given global health application through collaborations between researchers in scientifically-advanced countries, scientists in lower-resource settings and members of the Do-It-Yourself (DIY)-bio community (Fig. 4). A loose model of distributed system construction and optimization may enable global health priorities to be addressed even when they fall under the radar of researchers (or, more pointedly, their funding sources) in scientifically-advanced countries. Such a strategy would also help ensure equitable access to new technologies by providing vehicles for developing-country scientists to nurture their own technical capabilities (Wagner et al. 2001) while identifying and addressing region-specific issues. A recent analysis of pharmaceutical biotechnology innovation in developing countries (specifically, India, China and Brazil), showed between country differences in targeted disease areas (Rezaie et al. 2012). For instance, of the 165 compounds developed by 41 indigenous companies, Indian firms accounted for 66 % of the drugs for infectious diseases and 85 % of the products for diabetes. Interestingly, 16 of the compounds identified by the study are for diseases primarily affecting the developing world, suggesting that the involvement of indigenous firms may intensify the amount of research focused on global health problems (Rezaie et al. 2012).
Fig. 4.
Applied synthetic biology for global health. Research agendas should target biosynthetic and biosensor platforms with multi-use potential, as identified by market research and analysis of global health priorities. Researchers in scientifically-advanced nations could perform initial system design and experimentation, with industrially-relevant materials produced following device, pathway and system optimization focused on commercial development. Device, pathway and system reengineering for applications in global health could then be carried out in collaboration with scientists in lower-resource countries and the DIY-bio community
Even though most developing countries do not have the scientific and industrial capabilities of India, Brazil and China, researchers in lower-resource settings can nonetheless participate in the creation of global health solutions employing synthetic biology. Constructing a functional synthetic biological system is only one part of deploying a new technology and addressing global health problems in contexts with the most need, which will necessarily have enormous operational and governance challenges, many of which are outlined in some detail by Zhang in this issue. Clearly, scientists in developed nations are not privy to those contexts and it will be imperative to work with scientist in low resource settings to establish research agendas, to shape the system design goals to address contextual constraints and, more pragmatically, to establish supply chains for obtaining media and performing the process engineering needed for scale-up. Vaccine trials conducted in low resource settings have shown that interactions with local scientists and health care delivery personnel help build indigenous capacity and identify contextual constraints imposed by limited technical training and fragmented supply chains (Marchetti et al. 2012). Researchers conducting trials for the MenAfriVac™ epidemic meningitis vaccine in West Africa found that the on-site development of ‘must have’ standard operating procedures helped ensure that the sites were prepared to perform the work and resulted in formal mechanisms for sharing best practices. Mocked-up vaccine shipments provided a route for technical training and enabled the sponsors to identify conditions, such as extreme weather, low vehicle access, and poor road quality, that might otherwise prevent cold-chain maintenance (Marchetti et al. 2012). Douglas and Stemerding (this issue) provide further discussion of the ‘real world contexts’ in which synthetic biology might be deployed to meaningfully address global health problems.
The crowd-sourced protein folding and design ‘games’ undertaken by the global Foldit community to identify inhibitors that bind and inhibit the 1918 pandemic influenza virus have shown that distributed problem solving in medicine and health can be very powerful (Fleishman et al. 2011). The global Foldit community is comprised of thousands of people, many of whom lack formal scientific training altogether. Protein folding and design problems are cast in the form of collaborative games, using software written for this purpose. Although it is not clear how many participants in the flu inhibitor game were from developing countries, this example is important because it shows that non-experts can be involved in cutting edge research. Interestingly, several of the protein folding algorithms created by the ‘non-expert’ gamers have been superior to those developed using state-of-the-art computational method (Khatib et al. 2011). Similarly, it is not difficult to imagine that DIY-bio communities, comprised of individuals with broad differences in levels of formal scientific training, could be readily engaged to tackle problems in global health (see Landrain et al. and Betten et al. in this issue for further description of DIY-bio and the role for different stakeholders in developing these technologies, respectively).
Federally-funded researchers in the United States must follow the strictest US National Institutes of Health (NIH) and Centers for Disease Control and Prevention (CDC) biosafety regulations for human safety and to prevent the accidental release of engineered materials into the environment (Gutmann and Wagner 2010). Of course, there is nothing to guarantee that health-oriented bioengineering will be done with the world’s safety in mind, or that ‘technical vectors’ will be sufficient for avoiding adverse outcomes, no matter how small the risks presently appear (Bennett et al. 2009). For this reason, experimental research to assess and mitigate potential risks associated with large-scale biotechnology deployment will be crucial for ensuring that engineering and process controls to safeguard health and the environment advance alongside other developments in synthetic biology (Gutmann and Wagner 2010; Dana et al. 2012).
Multi-use is the feature
The artemisin and p-AS pathways were presented as illustrations of biosynthetic platforms that can be engineered to produce marketable chemicals and subsequently reengineered to produce low cost global health materials. Among the many other examples are systems for producing fatty-acids (Steen et al. 2010), alkaloids (Dietrich et al. 2011), polyketides (Keasling 2010) and utilizing nitrogen flux (Huo et al. 2011). Notably, the development of efficient, scalable biosynthetic platforms also provides experimental testbeds for creating CAD tools and design-driven approaches (Carothers et al. 2011) that reduce the demands for wet-lab infrastructure and repeated iterations of trial-and-error optimization (Carothers et al. 2009). While the development of generalizable production platforms could offer routes to rapid innovation (Rip and Kemp 1998), CAD tools and design-driven approaches should also be useful for rapidly constructing customized biosensors, pathways, and systems for more niche applications. In either case, it is important to realize that there are no engineering ‘silver bullets’, and that a given synthetic biological device or system will rarely be sufficient for solving any particular global health problem (Hersch 2012). The on-going development of design-driven approaches for synthetic biology is quickly increasing the sizes and complexities of the systems that can be constructed. I am optimistic that collaborations can be formed to exploit and distribute these technologies as parts of broader efforts to solve some of the world’s most pressing problems.
Acknowledgments
Discussions with Jay Keasling, Nathan Hillson, Paul Yager, and the participants from the Global Health workshop held in The Hague in February of 2012 were very helpful for preparing this manuscript. The author’s work in the Keasling research group was supported in part by the Synthetic Biology Engineering Research Center (National Science Foundation Award No. 0540879), by the Joint BioEnergy Institute (US Department of Energy, Office of Science, Office of Biological and Environmental Research, through contract DE-AC02-05CH11231) and, in part, by a Jane Coffin Childs Memorial Fund Postdoctoral Fellowship. J.M.C. is currently a Fellow of the Alfred P. Sloan Foundation.
Abbreviation
- CAD
Computer-aided design
Footnotes
To paraphrase a popular, unattributed, technology witticism, “That’s not a bug, it’s a feature”.
References
- American Chemical Society (2012) The discovery and development of penicillin. http://acswebcontent.acs.org/landmarks/landmarks/penicillin/pharma.html. Accessed 25 Jan 2012
- Bennett G, Gilman N, Stavrianakis A, Rabinow P. From synthetic biology to biohacking: are we prepared? Nat Biotechnol. 2009;27:1109–1111. doi: 10.1038/nbt1209-1109. [DOI] [PubMed] [Google Scholar]
- Billi D, Wright DJ, Helm RF, et al. Engineering desiccation tolerance in Escherichia coli. Appl Environ Microbiol. 2000;66:1680–1684. doi: 10.1128/AEM.66.4.1680-1684.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blanc V, Gil P, Bamas Jacques N, et al. Identification and analysis of genes from Streptomyces pristinaespiralis encoding enzymes involved in the biosynthesis of the 4 dimethylamino l phenylalanine precursor of pristinamycin I. Mol Microbiol. 1997;23:191–202. doi: 10.1046/j.1365-2958.1997.2031574.x. [DOI] [PubMed] [Google Scholar]
- Blount BA, Weenink T, Ellis T (2012) Construction of synthetic regulatory networks in yeast. FEBS Lett. doi:10.1016/j.febslet.2012.01.053 [DOI] [PubMed]
- Bokinsky G, Peralta-Yahya PP, George A, et al. Synthesis of three advanced biofuels from ionic liquid-pretreated switchgrass using engineered Escherichia coli. Proc Natl Acad Sci USA. 2011;108:19949–19954. doi: 10.1073/pnas.1106958108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carothers JM, Goler JA, Keasling JD. Chemical synthesis using synthetic biology. Curr Opin Biotechnol. 2009;20:498–503. doi: 10.1016/j.copbio.2009.08.001. [DOI] [PubMed] [Google Scholar]
- Carothers JM, Goler JA, Kapoor Y, et al. Selecting RNA aptamers for synthetic biology: investigating magnesium dependence and predicting binding affinity. Nucleic Acids Res. 2010;38:2736–2747. doi: 10.1093/nar/gkq082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carothers JM, Goler JA, Juminaga D, Keasling JD. Model-driven engineering of RNA devices to quantitatively program gene expression. Science. 2011;334:1716–1719. doi: 10.1126/science.1212209. [DOI] [PubMed] [Google Scholar]
- Chang MCY, Keasling JD. Production of isoprenoid pharmaceuticals by engineered microbes. Nat Chem Biol. 2006;2:674–681. doi: 10.1038/nchembio836. [DOI] [PubMed] [Google Scholar]
- Chau PC. Process control: a first course with MATLAB. Cambridge: Cambridge University Press; 2002. [Google Scholar]
- Chen J, Densmore D, Ham TS, et al. Device editor visual biological CAD canvas. J Biol Eng. 2012;6:1. doi: 10.1186/1754-1611-6-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dana GV, Kuiken T, Rejeski D, Snow AA. Synthetic biology: four steps to avoid a synthetic-biology disaster. Nature. 2012;483:29. doi: 10.1038/483029a. [DOI] [PubMed] [Google Scholar]
- Deana A, Celesnik H, Belasco JG. The bacterial enzyme RppH triggers messenger RNA degradation by 5-prime pyrophosphate removal. Nature. 2008;451:355–358. doi: 10.1038/nature06475. [DOI] [PubMed] [Google Scholar]
- Dietrich JA, Fortman JL, Juminaga D, Keasling JD (2011) Microbial production of plant-derived pharmaceutical natural products through metabolic engineering: artemisinin and beyond. In: Tao J, Kazlauskas R (eds) Biocatalysis for green chemistry and chemical process development, Wiley, Hoboken, NJ, USA
- Duffy JL, Kirk BA, Wang L, et al. 4-Aminophenylalanine and 4-aminocyclohexylalanine derivatives as potent, selective, and orally bioavailable inhibitors of dipeptidyl peptidase IV. Bioorg Med Chem Lett. 2007;17:2879–2885. doi: 10.1016/j.bmcl.2007.02.066. [DOI] [PubMed] [Google Scholar]
- Fleishman SJ, Whitehead TA, Ekiert DC, et al. Computational design of proteins targeting the conserved stem region of influenza hemagglutinin. Science. 2011;332:816. doi: 10.1126/science.1202617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goikhman MY, Subbotina LI, Martynenkov AA, et al. Synthesis and optical properties of copolymers of 4-aminostyrene with the side styrylquinoline chromophore groups. Russ Chem Bull. 2011;60:295–303. doi: 10.1007/s11172-011-0048-4. [DOI] [Google Scholar]
- Goler JA, Bramlett BW, Peccoud J. Genetic design: rising above the sequence. Trends Biotechnol. 2008;26:538–544. doi: 10.1016/j.tibtech.2008.06.003. [DOI] [PubMed] [Google Scholar]
- Gostin LO. Meeting basic survival needs of the world’s least healthy people: toward a framework convention on global health. Georget Law J. 2007;96:331–392. [Google Scholar]
- Gutmann A, Wagner J. New directions: the ethics of synthetic biology and emerging technologies. Washington, DC: Presidential Commission for the Study of Bioethical Issues; 2010. [Google Scholar]
- Hazen RM, Griffin PL, Carothers JM, Szostak JW. Functional information and the emergence of biocomplexity. Proc Natl Acad Sci. 2007;104:8574–8581. doi: 10.1073/pnas.0701744104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hersch F (2012) Transforming health care: one SMS at a time? Transl Glob Health. http://blogs.plos.org/globalhealth/2012/12/26/transforming-health-care-one-sms-at-a-time/. Accessed 26 Dec 2012
- Hillson NJ, Rosengarten RD, Keasling JD (2011) j5 DNA assembly design automation software. ACS Synth Biol 1:14–21 [DOI] [PubMed]
- Holtz WJ, Keasling JD. Engineering static and dynamic control of synthetic pathways. Cell. 2010;140:19–23. doi: 10.1016/j.cell.2009.12.029. [DOI] [PubMed] [Google Scholar]
- Huo YX, Cho KM, Rivera JG, et al. Conversion of proteins into biofuels by engineering nitrogen flux. Nat Biotechnol. 2011;29:346–351. doi: 10.1038/nbt.1789. [DOI] [PubMed] [Google Scholar]
- Johnson IS. Human insulin from recombinant DNA technology. Science. 1983;219:632–637. doi: 10.1126/science.6337396. [DOI] [PubMed] [Google Scholar]
- Juminaga D, Baidoo EE., Redding-Johanson AM et al. (2012) Modular engineering of l-tyrosine production in Escherichia coli. Appl Environ Microbiol 78(1):89–98. doi:10.1128/AEM.06017-11 [DOI] [PMC free article] [PubMed]
- Keasling JD. Manufacturing molecules through metabolic engineering. Science. 2010;330:1355. doi: 10.1126/science.1193990. [DOI] [PubMed] [Google Scholar]
- Khatib F, Cooper S, Tyka MD, et al. Algorithm discovery by protein folding game players. Proc Natl Acad Sci. 2011;108:18949–18953. doi: 10.1073/pnas.1115898108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koblentz GD. Living weapons: biological warfare and international security. Ithaca, NY: Cornell University Press; 2009. [Google Scholar]
- Koeppl H. Design and analysis of bio-molecular circuits. Berlin: Springer; 2011. [Google Scholar]
- Lee SK, Chou H, Ham TS, et al. Metabolic engineering of microorganisms for biofuels production: from bugs to synthetic biology to fuels. Curr Opin Biotechnol. 2008;19:556–563. doi: 10.1016/j.copbio.2008.10.014. [DOI] [PubMed] [Google Scholar]
- Marchetti E, Mazarin-Diop V, Chaumont J et al (2012) Conducting vaccine clinical trials in sub-Saharan Africa: operational challenges and lessons learned from the Meningitis Vaccine Project. Vaccine [DOI] [PubMed]
- Mead C, Conway L. Introduction to VLSI systems. MA: Addison-Wesley Reading; 1980. [Google Scholar]
- Medema MH, van Raaphorst R, Takano E, Breitling R. Computational tools for the synthetic design of biochemical pathways. Nat Rev Microbiol. 2012;10:191–202. doi: 10.1038/nrmicro2717. [DOI] [PubMed] [Google Scholar]
- Oye KA, Wellhausen R. Synthetic biology: the technoscience and its societal consequences. Berlin: Springer; 2010. pp. 121–140. [Google Scholar]
- Peralta-Yahya PP, Ouellet M, Chan R, et al. Identification and microbial production of a terpene-based advanced biofuel. Nat Commun. 2011;2:483. doi: 10.1038/ncomms1494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reid AB, Daffy JR, Stanley P, Buising KL. Use of pristinamycin for infections by gram-positive bacteria: clinical experience at an Australian Hospital. Antimicrob Agents Chemother. 2010;54:3949–3952. doi: 10.1128/AAC.00212-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rezaie R, McGahan AM, Daar AS, Singer PA. Innovative drugs and vaccines in China, India and Brazil. Nat Biotechnol. 2012;30:923–926. doi: 10.1038/nbt.2380. [DOI] [PubMed] [Google Scholar]
- Rip A, Kemp RPM (1998) Technological change. In: Rayner S, Malone EL (eds) Human choice and climate change. Resources and Technology, vol II. Battelle Press, Columbus, Ohio, pp 327–399
- Ro D-K, Paradise EM, Ouellet M, et al. Production of the antimalarial drug precursor artemisinic acid in engineered yeast. Nature. 2006;440:940–943. doi: 10.1038/nature04640. [DOI] [PubMed] [Google Scholar]
- Roose-Snyder B, Doyle MK. The global health licensing program: a new model for humanitarian licensing at the university level. Am J Law Med. 2009;35:281–700. doi: 10.1177/009885880903500203. [DOI] [PubMed] [Google Scholar]
- Rosicrucian Egyptian Museum (2012) Rosicrucian Egyptian Museum Virtual Gallery. Brewery Scene Model (2000 B.C.E.). http://www.egyptianmuseum.org/dailylifegallery. Accessed 16 July 2013
- Saltelli A, Ratto M, Andres T, et al. Global sensitivity analysis: the primer. England: Wiley; 2008. [Google Scholar]
- Sariaslani FS. Development of a combined biological and chemical process for production of industrial aromatics from renewable resources. Annu Rev Microbiol. 2007;61:51–69. doi: 10.1146/annurev.micro.61.080706.093248. [DOI] [PubMed] [Google Scholar]
- Shiue E, Prather KLJ (2012) Synthetic biology devices as tools for metabolic engineering. Biochem Eng J. doi:10.1016/j.bej.2012.04.006
- Steen EJ, Kang Y, Bokinsky G, et al. Microbial production of fatty-acid-derived fuels and chemicals from plant biomass. Nature. 2010;463:559–562. doi: 10.1038/nature08721. [DOI] [PubMed] [Google Scholar]
- Towse A, Kettler H. Advance price or purchase commitments to create markets for treatments for diseases of poverty: lessons from three policies. Bull World Health Organ. 2005;83:301–307. [PMC free article] [PubMed] [Google Scholar]
- Wagner CS, Brahmakulam I, Jackson B et al (2001) Science and technology collaboration: building capability in developing countries (No. RAND/MR-1357.0-WB). RAND Corp., Santa Monica, CA
- Westfall PJ, Pitera DJ, Lenihan JR, et al. Production of amorphadiene in yeast, and its conversion to dihydroartemisinic acid, precursor to the antimalarial agent artemisinin. Proc Natl Acad Sci. 2012;109:E111–E118. doi: 10.1073/pnas.1110740109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yager P, Domingo GJ, Gerdes J. Point-of-care diagnostics for global health. Annu Rev Biomed Eng. 2008;10:107–144. doi: 10.1146/annurev.bioeng.10.061807.160524. [DOI] [PubMed] [Google Scholar]
- Zhang F, Keasling J (2011) Biosensors and their applications in microbial metabolic engineering. Trends Microbiol 19:323–329 [DOI] [PubMed]
- Zhang F, Carothers JM, Keasling JD. Design of a dynamic sensor-regulator system for production of chemicals and fuels derived from fatty acids. Nat Biotechnol. 2012;30:354–359. doi: 10.1038/nbt.2149. [DOI] [PubMed] [Google Scholar]