Abstract
Purpose
We compared the efficacy of human Langerhans cells (LCs) as tumor immunogens in vivo with monocyte-derived DCs (moDCs) and investigated how IL15 supports optimal DC-stimulated antitumor immunity.
Experimental Design
AJCC stage III/IV melanoma patients participated in this first clinical trial comparing melanoma peptide-pulsed LC with moDC vaccines (NCT00700167,www.ClinicalTrials.gov). Correlative studies evaluated mechanisms mediating IL15 support of DC-stimulated antitumor immunity.
Results
Both DC vaccines were safe and immunogenic for melanoma antigens. LC-based vaccines stimulated significantly greater tyrosinase-HLA-A*0201 tetramer reactivity than did moDC-based vaccines. The two DC subtypes were otherwise statistically comparable, in contrast to extensive prior data in vitro demonstrating LC superiority. LCs synthesize much more IL15 than moDCs and stimulate significantly more antigen-specific lymphocytes with a cytolytic IFN-gamma profile even without exogenous IL15. When supplemented by low dose IL15, instead of IL2, moDCs stimulate 5-6 logs more tumor antigen-specific effector memory T-cells (TEMRA) over 3-4 weeks in vitro. IL2 and IL15 can be synergistic in moDC stimulation of cytolytic T-cells. IL15 promotes T-cell expression of the antiapoptotic bcl-2 and inhibits candidate regulatory T-cell (Treg) expansion after DC stimulation, countering two effects of IL2 that do not foster tumor immunity.
Conclusions
MoDC-based vaccines will require exogenous IL15 to achieve clinical efficacy. Alternatively, LCs can couple the endogenous production of IL15 with potent T-cell stimulatory activity. Optimization of full length tumor antigen expression for processing into multiple immunogenic peptides for presentation by both class I and II MHC therefore merits emphasis to support more effective antitumor immunity stimulated by LCs.
INTRODUCTION
Antigen-specific expansion of effector and memory CD8+ T-cells is a central goal of immunotherapy against tumors. For stimulation of MHC-restricted, antigen-specific, CD8+, cytolytic T lymphocytes (CTL) in vitro (1), human Langerhans cells (LCs) derived from CD34+ hematopoietic progenitor cells (HPCs) have demonstrated superiority over other known conventional or myeloid human DC subtypes, e.g., monocyte-derived DCs (moDCs) and dermal-interstitial DCs (DDC-IDCs). This has held true for LCs either presenting peptide for recall responses against viral antigens or cross-presenting dying tumor cells to elicit tumor antigen-specific CTLs (1). Detailed comparisons between resident DC populations isolated from human skin have demonstrated similar potency of LCs (2). Clinical trial data have also suggested greater efficacy of DC vaccines that contain LCs (3); but there have been no direct comparisons in vivo in humans between defined DC subtypes as vaccines. We therefore undertook a phase I clinical trial to determine safety and toxicity of melanoma peptide-pulsed LCs compared with the more commonly used moDCs. Laboratory studies then compared their immunologic efficacy based on T-cell tetramer reactivity and ELISpot assay of IFN-gamma secretion.
LCs achieve robust stimulation of CTLs without producing any IL-12p70 (1), yet they secrete more IL15 than any other conventional DC subtype (1, 4). IL15 is therefore of particular interest because of its role in lymphocyte homeostasis; the expansion of memory T-cells, especially CD8+ CTLs; and its autocrine protection of DCs from apoptosis (2, 5-11). IL15 also has a contrasting role with IL2 in that it counters tolerance and supports autoimmunity (5), rendering IL15 an attractive cytokine to support targeted immune responses against self-differentiation antigens expressed by tumors like melanoma. Most IL15 data derive from mouse rather than human studies, although a recent non-human primate study provides important insight into the safety and cytotoxicity of the IL15 administered in vivo (12).
Monocyte precursors of moDCs have proven malleable in their differentiation, depending on cytokine exposure (13). Provision of IL15 during development leads to moDCs with Langerhans-like DC properties (14, 15). The moDCs and LCs used in the vaccine trial, however, had been generated according to standard protocols without IL15 (1); and we wished to investigate the role of this cytokine on T cell responses in the context of restimulation by the same DC subtype to which the T cells had been exposed in vivo during vaccination. Our findings establish an essential role for IL15 in the generation of cytolytic, IFN-gamma secreting T cells, even when stimulated by potent DCs. MoDCs depend on an exogenous source of IL15, yet LCs remain effective even with limiting or no exogenous IL15. These data have important implications for the design of DC-based immunotherapy trials, which going forward, must also include optimized approaches to provide full length antigens for processing into multiple immunogenic peptides for presentation on both class I and II MHC.
MATERIALS AND METHODS
Human cells, media, and cytokines
Human cell collection and use adhered to protocols approved by the Institutional Review and Privacy Board of Memorial Hospital, Memorial Sloan-Kettering Cancer Center (MSKCC). Healthy volunteers or patients provided peripheral blood mononuclear cells (PBMCs) or G-CSF-elicited CD34+ hematopoietic progenitor cells (HPCs) for the isolation of T-cells and the generation of moDCs and LCs exactly as published (1) (see erratum for correct FLT-3-ligand dose) (16). CD34+ HPCs have greater expansion and differentiation potential than monocytes, so the progeny containing LCs were more heterogeneous, primarily including immature eosinophils (17, 18). Viable LCs and moDCs with large forward scatter by flow cytometry were dosed according to phenotypic expression of HLA-DRbright CD86bright CD83+ CD14neg epitopes. All cells were used either fresh or thawed after cryopreservation without compromising phenotype or activity (19). Viable recovery was consistently ≥ 85% and usually >90%.
Phenotypic analyses by flow cytometry
Fluorescein (FITC), phycoerythrin (PE), PE-cyanine-5 (PE-Cy5), PE-Cy7, peridinin chlorophyll protein-cynine-5.5 (PerCP-Cy5.5), allophycocyanin (APC), APC-Alexa Fluor750, PE-Texas Red (ECD)-conjugated mouse-anti-human monoclonal antibodies (mAbs) included anti-CD3, anti-CD8, anti-CD25, anti-CD27, anti-CD28, anti-CD69 (BD Biosciences, San Jose, CA); anti-CD4, anti-CD45RA, anti-CD45RO (Beckman Coulter, Fullerton, CA); anti-CD127, anti-human Foxp3, anti-CD8, and anti-CD62L (eBioscience, San Diego, CA); and anti-CCR7 (R&D Systems). Isotype controls included the appropriate fluorochrome-conjugated mouse IgG1 or rat IgG2a (Dako, Carpenteria, CA; eBioscience). Foxp3 detection required intracellular staining, using cell fixation and permeabilization buffers provided with the Foxp3 kit (eBioscience).
HLA-A*0201-restricted tyrosinase, gp100, and fluMP-streptavidin-PE-labeled tetramers and negative tetramer controls (Beckman Coulter) detected antigen-specific T-cells. Positive tetramer reactivity required a distinct population at least one log above the mean fluorescent intensity of the negative control.
Flow cytometry analyses used a Cytomics FC500 (Beckman Coulter) or a Cyan-ADP flow cytometer (DAKO). Gates were set for collection and analysis of > 200,000 live events based on propidium iodide exclusion.
Immune responses to tumor antigenic peptide-pulsed dendritic cells in vivo
Clinical trial design
We conducted a phase I clinical trial to test safety and toxicity and to compare immune responses stimulated by tumor peptide-pulsed moDCs versus LCs in HLA-A*0201+ patients with AJCC stage III or IV melanoma (registered as no. NCT00700167 at www.ClinicalTrials.gov; Supplemental Figure 1). There were no significant differences between assignment to either type DC vaccine with regard to demographics, Karnofsky performance score, or disease stage (Table 1A). Phase Ia accrued cohorts of three patients to each of three vaccine doses (3×106, 10×106, or 30×106), using either peptide-loaded (see below) moDCs or LCs for a priming dose, followed by two boosters of the same peptide-loaded DC subtype at approximately 4 wk intervals. Patients received a total of 3 vaccines with no crossover between moDCs or LCs. MoDCs or LCs were freshly made and used for the initial vaccine. Subsequent vaccines used thawed cells from the cryopreserved initial product (19). Table 1B lists release criteria required for each vaccine, and Table 1C reports the viability and phenotype of all administered DC vaccines. Purity of the CD34+-derived LC progeny was lower than that of the moDCs, reflecting the more variable differentiation potential of the starting populations of CD34+ HPCs compared with blood monocytes (1). The major contaminants of the LCs were immature myeloid cells, mostly eosinophils (1). Vaccines were dosed according to the absolute number of CD83+ CD86bright HLA-DRbright CD14neg moDCs or LCs by flow cytometry, so the DC numbers were equivalent.
Table 1.
Patient Demographics, Vaccine Characterization, and Patient Accrual and Completion on Trial
| Table 1A. Patient Demographics and Disease Characteristics at Protocol Entry | ||||
|---|---|---|---|---|
| All patients, all dose levels |
10 × 106 dose level only |
|||
| Variable | LC (n=18) No. (%) |
moDC (n=18) No. (%) |
LC (n=12) No. (%) |
moDC (n=12) No. (%) |
|
|
|
|||
| Gender | ||||
| Male | 10 (56) | 11 (61) | 8 (66) | 7 (58) |
| Female | 8 (44) | 7 (39) | 4 (34) | 5 (42) |
| Age, years | ||||
| Mean | 61.4 | 57.2 | 63.2 | 57.4 |
| Range | 43-81 | 31-83 | 46-81 | 39-77 |
|
Karnofsky Performance
Score (KPS) |
||||
| Median | 100 | 100 | 100 | 100 |
| Range | 90-100 | 90-100 | 90-100 | 90-100 |
| Disease Stage (AJCC) | ||||
| III, NED | 15 (83) | 16 (89) | 9 (75) | 10 (83) |
| IV, NED | 2 (11) | 1 (6) | 2 (17) | 1 (8) |
| IV, slow progressive | ||||
| disease | 1 (6) | 1 (6) | 1 (8) | 1 (8) |
| Table 1B. Criteria for Release of Dendritic Cell Vaccines for Patient Administration | ||
|---|---|---|
| Variable | Test | Result required for release |
| Bacteria and fungus | Culture in thioglycolate broth and soybean casein digest medium Gram stain |
No growth after 5 days of in- process culture. No growth confirmation of final product after administration. Negative on final product |
| Endotoxin | Limulus amebocyte lysate (BioWhittaker; CBER/FDA biologic license number 709) |
<5 endotoxin units |
| Mycoplasma | PCR | Negative result, in process 48 hrs before end of culture. |
| Viability | Propidium iodide (PI) staining of large forward scatter (FSC) cells on flow cytometry |
<30% PI positive (or ≥ 70% viable) |
|
Phenotype (flow
cytometry) |
Flow cytometry: gated population of large FSC, CD14 neg, class II MHC bright cells |
≥ 50% CD83+ ≥ 50% CD86+ |
| Table 1C. Viability and Phenotype of Dendritic Cell Vaccines Administered to Patients | ||||
|---|---|---|---|---|
| % viability of total (mean ± SEM) |
% viable HLA- DRbrightCD14neg of total (mean ± SEM) |
%CD83 of viable HLA- DRbright CD14neg (mean ± SEM) |
%CD86 of viable HLA- DRbright CD14neg mean ± SEM) |
|
|
Blood monocyte-
derived DCs |
89.6 ± 1.7 | 81.3 ± 2.1 | 89.6 ± 2.1 | 98.0 ± 0.5 |
| CD34+ derived LCs | 93.1 ± 0.7 | 56.8 ± 1.91 | 65.6 ± 2.11 | 98.2 ± 0.4 |
| Table 1D. Patient accrual to and completion of vaccine trial, including numbers evaluable for response assessments | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Total accrued (n) |
Off study (n) |
Reason for withdrawal from study |
Completed all three vaccines (n) |
Evaluable for response assessments (adequate cell yields) |
||||||
| Tetramer | ELISpot | |||||||||
|
|
||||||||||
| Phase Ia | gp100 | tyr | fluMP | gp100 | tyr | fluMP | ||||
| MoDCs | ||||||||||
| 3 × 106 moDCs/vaccine | 3 | 0 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | |
| 10 × 106 moDCs/vaccine | 3 | 0 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | |
| 30 × 106 moDCs/vaccine | 4 | 1 |
|
3 | 3 | 3 | 3 | 3 | 3 | 3 |
|
| ||||||||||
| LCs | ||||||||||
| 3 × 106 LCs/vaccine | 4 | 0 | 41 | 3 | 3 | 3 | 3 | 3 | 3 | |
| 10 × 106 LCs/vaccine | 3 | 0 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | |
| 30 × 106 LCs/vaccine | 3 | 0 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | |
|
| ||||||||||
|
Phase Ib
2 | ||||||||||
| MoDCs | ||||||||||
| 10 × 106 moDCs/vaccine | 10 | 1 |
|
9 | 9 | 9 | 7 | 7 | 7 | 7 |
|
| ||||||||||
| LCs | ||||||||||
| 10 × 106 LCs/vaccine | 13 | 4 |
|
9 | 8 | 8 | 8 | 7 | 8 | 8 |
|
| ||||||||||
| Phase Ia + Ib, all at | ||||||||||
|
10 × 106 cells/vaccine
| ||||||||||
| MoDCs | 12 | 12 | 11 | 12 | 10 | 10 | 10 | 10 | ||
| LCs | 12 | 11 | 11 | 10 | 11 | 11 | 39 | |||
Purity of the CD34+ -derived LC progeny was lower than that of the moDCs, reflecting the more variable differentiation potential of the starting populations of CD34+ HPCs compared with blood monocytes (1). The major contaminants of the LCs were immature myeloid cells, mostly eosinophils (1). Regardless, vaccines were dosed according to the absolute number of CD83+ CD86+ HLA-DRbright CD14neg moDCs or LCs.
Three evaluable patients were required at each dose level in phase Ia in order to select the optimal biologic dose (defined in Methods) to use in phase Ib. Hence one additional patient was accrued to replace one who had inadequate cell yields for response assessments despite a normal complete blood count.
Response assessments for the trial overall were based on the evaluable patients at the 10×106 moDC or LC dose per vaccine in phase Ia, plus all evaluable patients in phase Ib.
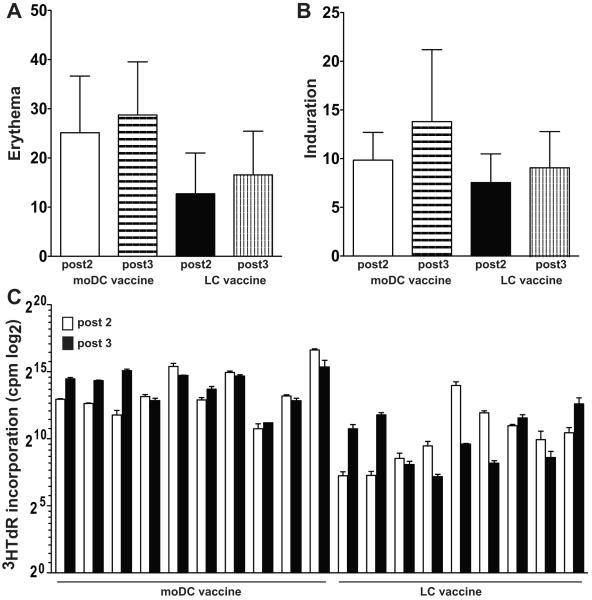
Each moDC or LC vaccine, regardless of total dose, was administered as ten 0.1 ml deep intradermal injections divided equally between two sites in the proximal arms or thighs, excluding areas adjacent to prior lymph node resections. Surrounding erythema and induration were measured for delayed type hypersensitivity reactions 48 hours after the second and third vaccines.
The dose that yielded a positive response by tetramer reactivity or ELISpot (see below), defined as >2 SD above the prevaccine baseline response in at least two of the three initial phase Ia patients at each dose level, determined the optimal biologic dose. Hence the 10×106 dose level, which proved most feasible in terms of starting cell numbers, cytokine usage, costs, and labor, was also the optimal biologic dose for each DC subtype. Phase Ib of the trial then accrued nine additional patients to each of the moDC or LC arm, resulting in a total of 12 patients who received either moDC or LC vaccines at the 10×106 dose level in phases Ia and Ib combined.
Antigen-loading of moDCs or LCs
The clinical trial used two synthetic, HLA-A*0201-restricted, heteroclitic melanoma peptides: tyrosinase (TYR368-376) YMDGTMSQV and gp100 (gp100209-217) IMDQVPFSV (Research Genetics, Invitrogen, Carlsbad, CA). Synthetic influenza matrix peptide (fluMP58-66 GILGFVFTL; Research Genetics, Invitrogen) served as a positive control for HLA-A*0201-restricted responses. KLH (high purity, endotoxin-free; Calbiochem) served as a positive control neoantigen for class II MHC-restricted responses.
Terminally maturing moDCs or LCs for the clinical trial were cultured at 1×106 cells/3ml in 6 well plates, to which peptides (1 μM) or KLH (10 μ/mL) were added overnight at 37°C, but in separate cultures to avoid MHC binding competition. Cells were washed and evaluated for release criteria (Table 1B) before combining and resuspending to the appropriate concentration for vaccination.
Response assessments in vitro and correlative laboratory studies
Response assessments for the clinical trial used peripheral blood mononuclear cells (PBMCs) collected at pre-vaccine baseline and then approximately three weeks after each vaccine. CD8+ T-cell reactivity with HLA-A*0201-peptide tetramers over time and ELISpot assay of IFN-gamma secretion by total T-cells compared over time as fold increase over baseline. These followed a single 6-7 day restimulation of the PBMC responders in vitro, using the same peptide-loaded autologous moDCs or LCs used for a patient’s vaccines, but without exogenous cytokines. Ten to 12 patients, out of 12 total in each DC subtype vaccine group at the optimal biologic dose of 10 × 106 cells/vaccine, were evaluable. Zero to two patients in each group became inevaluable due to inadequate cell yields or insufficient replicates (Table 1D). For assessment of KLH-specific responses, PBMCs from moDC and LC vaccinees were harvested at baseline prevaccine and approximately three weeks after each of three vaccines and cultured with KLH (1μg/ml). KLH-specific immunity was determined based on the incorporation of 3HTdR (1 μCi/well) by proliferating responder T cells in the last 8 h of a 5-day culture.
All patients consented to the use of any residual cryopreserved cells for subsequent thaw and assessment in additional correlative laboratory studies. These used responder PBMCs, total T-cells, or T-cell subsets selected using immunomagnetic columns (Miltenyi Biotec, Germany) as indicated. Responder PBMCs or T-cells were resuspended in 10% autologous serum v/v in complete RPMI-1640 and plated at 2 × 106 cells/ml final per well of a 24-well plate (Costar, Corning, NY) or at 1 × 105/100ul final per well of a 96-round bottomed well plate (Costar). Mature autologous moDCs or LCs were resuspended at 106/mL in cytokine and serum-free RPMI-1640, then loaded with 10 uM concentration of a single 9-mer peptide for 1h at RT before restimulation. TargeT-cells for ELISpot or CTL assays were similarly loaded with single peptides. Peptides were always separately loaded onto the respective DCs to avoid MHC binding competition. Peptide-loaded moDCs or LCs were added in graded doses to fixed numbers of responders. Where experiments required repeated stimulation, responder cells were harvested after 6-7d initial stimulation and restimulated by the same DC subtype and exogenous cytokine combination each round.
The above cultures were supplemented or not with either recombinant human IL2 (10 IU/ml; Chiron, Emeryville, CA) and/or IL15 (10 ng/ml; R&D Systems) (see also Supplemental Figure 1). Although IL2 10 IU/ml is less than what DC stimulated allogeneic T-cells produce (20), we avoided supraphysiologic doses of IL2 in these autologous combinations that could activate NK cells among bulk PBMCs or T-cells via their IL15Rs, which share common beta and gamma chains with the IL2R (21).
Inhibition of IL15 binding to IL15R-alpha on dendritic cells
LCs or moDCs were opsonized for 2 hrs with a polyclonal goat anti-human IL15R-alpha (5ug/mL; R&D Systems) or its isotype control (5ug/mL; R&D Systems). These were then separately cocultured with responder PBMCs in functional assays at the indicated ratios. Anti-human IL15R-alpha or its isotype control was replenished at the same concentration in culture every 48h. When cells underwent a second round of stimulation after one week, responders were harvested, washed, and restimulated for 7d by anti-human IL15R-alpha-opsonized gp100-loaded moDCs or LCs from the same population.
IFN-gamma enzyme-linked immunosorbent spot (ELISpot) assay
After antigen-specific stimulation, responder lymphocytes were tested for IFN-gamma production by ELISpot assay (1-DIK ELISpot for human IFN-gamma; Mabtech, DiaPharma Group, West Chester, OH; Vector ABC kit, Vector Laboratories, Burlingame, CA; Automated ELISpot Reader System and KS 4.3 software, Carl Zeiss Vision, Göttingen, Germany) according to manufacturer’s instructions. Targets for lymphocyte restimulation during overnight rechallenge in the ELISpot assays were either mature peptide-pulsed moDCs or LCs, whichever had been used for stimulations in vitro, plated in triplicates at 30:1 effector:target (E:T) ratio. Control wells contained effectors and unloaded targets. ELISpot reactivity against both moDC and LC targets without antigenic peptide was subtracted from reactivity against the peptide-loaded moDC and LC targets, because unloaded moDCs caused such high background.
Cytolytic T-cell assays
Cytolytic activity exerted by T lymphocytes responding to antigen-specific DC stimulation was assessed in standard 51Cr release assays against the same target cells used in ELISpot assays. 5×103 51Cr-labeled target cells were added at the indicated T-cell effector:target ratios. Supernatants were collected after 4-6hrs from replicate microwells for calculating specific 51Cr release.
Assessment of early and late CD3+CD8+ T-cell apoptosis
Early and late apoptosis were evaluated using a FITC-conjugated Annexin V probe (BD Biosciences), together with propidium iodide (PI; Sigma-Aldrich,S. Louis, MO). Fluorochrome-conjugated MAbs costained the CD3+CD8+ T-cell subset of interest. Early apoptotic cells were Annexin V+, PIneg, whereas late apoptotic and necrotic cells were Annexin V+, PI+.
Protein extraction and Western Blotting
Positive immunomagnetic selection (Miltenyi Biotec) yielded CD3+ CD8+ T-cells with 97-99% purity. Protein extraction from 2-5 × 106 cells used RIPA lysis buffer (Pierce, Rockford, IL) with protease inhibitors (Roche Applied Science, Mannheim, Germany) for 20 min on ice. 20ugs of cell lysate were applied to a 12% Bis-Tris gel (Invitrogen). Proteins were blotted onto a PVDF membrane (Biorad, Hercules, CA), blocked with 5% non-fat dry milk in PBS/0.1% Tween20, and then probed with mouse-anti-human Bcl-2 monoclonal antibody (BD Biosciences). Mouse-anti-human GAPDH (Ambion, Austin, TX) served as the loading control. Secondary antibody was HRP-conjugated goat anti-mouse IgG (PerkinElmer Life Sciences, Boston, MA). Immunoreactive protein bands were detected using an ECL detection kit (Amersham, Buckinghamshire, UK).
Statistics
Responses to the moDC or LC vaccines in the clinical trial were tested by a stratified permutation (Wilcoxon) rank sum statistic. This compared moDCs with LCs where the outcomes were reactivity against gp100, tyrosinase, or control fluMP. The test was stratified by vaccine number over time.
For the correlative laboratory studies in vitro, replicate means from >3 independent experiments were averaged and SEM calculated as the measure of variability. Otherwise, SD was calculated for the replicate mean of a single representative experiment of at least three. A stratified t-test (stratified by the effector:target or responder:stimulator ratio) was used for the functional assays. The t-test was used for each comparison within a given time point or condition for all other analyses.
RESULTS
Peptide-loaded moDCs and LCs are safe in patients with advanced melanoma and generate measurable tumor Ag-specific immune responses
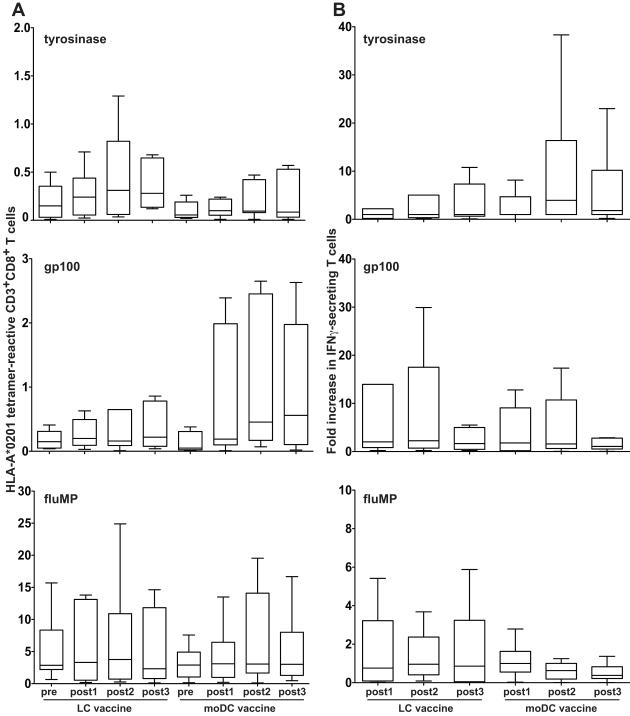
Two groups of twelve patients each received three vaccines of either 10×106 peptide-pulsed moDCs or CD34+ derived LCs, which was the optimal biologic dose as explained in Materials and Methods. No patient experienced more than grade 2 toxicity according to Common Terminology Criteria for Adverse Events (CTCAE), version 3.0, possibly or probably related to these vaccines. All patients developed some degree of erythema (Figure 1A), induration (Figure 1B), and mild pruritus at the injection sites. Two patients experienced grade I vitiligo, possibly related to the vaccine. Grade 2 toxicities were due to combinations of grade 1 cutaneous toxicities. Pre-vaccine baseline and post-vaccine responses were assessed simultaneously in vitro to avoid interassay variability. Proliferative responses to rechallenge with KLH (Figure 1C) were greater in the moDC than LC vaccinees (post 2, P = 0.002; post 3, P < 0.001; both relative to prevaccine baseline). Figure 2A depicts the absolute tetramer reactivity over time, including the prevaccine baseline. Figure 2B shows IFN-gamma secretion measured by ELISpot as the fold-increase over 2 standard deviations above the prevaccine baseline mean. Background reactivity against targets without peptide was subtracted from each ELISpot condition, and resulting negative or zero values were normalized to 1 for calculation of fold-increase.
Figure 1. Peptide-pulsed moDC and LC vaccines stimulate delayed type hypersensitivity reactions and KLH-specific responses after the second and third vaccines.
Erythema and induration were measured at the greatest diameter around each of ten injection sites to calculate the mean for each parameter for each patient after vaccine #2 and again after vaccine #3. Shown are the averages +/− SD of the individual patient means (n = 12 patients) for each subtype DC vaccine. P = NS comparing moDCs with LCs. PBMCs from moDC and LC vaccinees were harvested at baseline prevaccine and approximately three weeks after each of three vaccines and cultured with KLH (1μg/ml). KLH-specific immunity was determined based on the incorporation of 3HTdR by proliferating responder T cells (y-axis, log2 scale) in the last 8 h of a 5-day culture.
Figure 2. Peptide-loaded moDCs and LCs generate immune responses in patients with advanced melanoma, but neither is clearly superior in this setting.
Responses against peptide-loaded moDCs or LCs were measured at baseline prevaccine and again approximately three weeks after each of three vaccines given at nearly four week intervals. Responses were based on a single 6-7 day restimulation in vitro, using the same moDCs or LCs used to vaccinate a given patient, pulsed with the indicated peptides. No exogenous cytokines were added. Box plots show the medians and interquartile ranges (25th to 75th percentiles) with whiskers approximating +/− 2 SD or 95% of the data (Tukey method). (A) Absolute numbers of CD3+ CD8+ T cells reactive with tyrosinase-, gp100-, or fluMP-HLA-A*0201 tetramers are shown over time, where positive events exhibited at least one log higher fluorescent intensity than the negative controls. (B) The fold increases in IFN-gamma secretion over time by total T cells in ELISpot assays, relative to the prevaccine baseline average + 2 SDs, are depicted. Empty moDC targets without peptides were associated with high background IFN-gamma secretion in the ELISpot assay, so the background values were subtracted from all conditions at all timepoints to calculate fold increases. LCs were compared with moDCs where the outcomes were reactivity against tyrosinase, gp100, or control fluMP. (A) and (B) The test was stratified by vaccine number over time, and a permutation test generated P values. Only tyrosinase-HLA-A*0201 tetramer reactivity achieved significance in favor of LCs over moDCs (P = 0.04). There was a trend in favor of moDCs in the ELISpot assays for tyrosinase-specific responses (P = 0.08). P = NS for all other comparisons between LCs and moDCs.
Data comparing moDCs with LCs in vitro (1) led to the hypothesis that LCs would be superior immunogens. Only CD8+ T-cell reactivity with tyrosinase peptide-HLA-A*0201 tetramers reached statistical significance, however, slightly favoring LCs over moDCs (P = 0.04). There was a non-significant trend supporting moDCs for stimulating CD8+ T-cell reactivity with gp100 peptide-HLA-A*0201 tetramers (P = 0.11), but LCs and moDCs were equivalent in stimulating fluMP tetramer reactivity (P = 0.93). Limited cell numbers precluded isolation of CD8+ T-cells for ELISpot assays. Total T-cell production of IFN-gamma showed a trend without reaching statistical significance in favor of responses stimulated by tyrosinase peptide-pulsed moDCs (P = 0.08) and by fluMP-loaded LCs (P = 0.14), whereas the two subtypes showed no difference in stimulating gp100-specific responses (P = 0.30).
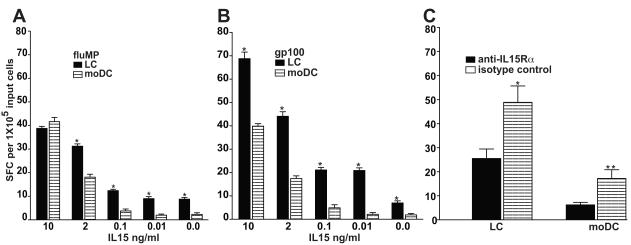
LCs maintain superior potency over moDCs in inducing IFN-gamma secreting lymphocytes at limiting doses of exogenous IL15
Pertinent to the generation of Ag-specific CTLs was our previous finding that LCs secreted significantly more IL15 than moDCs (1, 4). By titrating doses of exogenous IL15 down to zero, we evaluated the biologic dependence of LCs compared with moDC on exogenous IL15. T-cells remaining after completion of the above response assessments underwent three weekly rounds of stimulation in vitro by the same gp100 peptide- or fluMP-pulsed DC subtype used for primary immunization in vivo. LCs stimulated significantly greater reactivity than did moDCs, especially at limiting doses of exogenous IL15 (Figures 3A, 3B). Even in the absence of any exogenous IL15, LCs stimulated significantly greater T-cell responses than did comparable numbers of moDCs.
Figure 3. LCs are more potent than moDCs in inducing IFN-gamma-secreting CTL at limiting doses of exogenous IL15.
PBMCs from melanoma patients previously vaccinated with peptide-pulsed, mature, autologous, dendritic cells (LCs or moDCs) were restimulated in vitro by the same type of autologous dendritic cell pulsed with (A) fluMP or (B) gp100 peptide. Following three weekly restimulations, ELISpot assays measured IFN-gamma secretion after overnight exposure to the respective peptide-pulsed dendritic cell targets. Limited cell numbers precluded isolation of CD3+CD8+ T cells for the ELISpot assays. IL15 was supplemented at the indicated concentrations during the restimulations in vitro. Shown are the averaged triplicate means +/− SEM (n=3 independent experiments) for the number of IFN-gamma spot forming cells (SFC) per triplicate of 105 input cells. By Student’s t test, P < 0.05 LCs vs moDCs at each IL15 concentration tested. Only LCs were still stimulatory in the absence of exogenous IL15. (See also Figure 4C, which validates the concordance between IFN-gamma secretion and actual target lysis by CTLs). (C) PBMCs from melanoma patients, previously vaccinated with either peptide-pulsed, mature, autologous LCs or moDCs, were restimulated in vitro by the same type of autologous dendritic cells pulsed with gp100 peptide over each of two weeks (stimulator:responder = 1:30). No exogenous cytokines, including IL15, were added to any condition. The peptide-pulsed LCs and moDCs were opsonized at the outset and upon each restimulation with anti-IL15R-alpha or isotype control. IFN-gamma secretion by PBMCs was analyzed by ELISpot after overnight exposure to the same peptide-pulsed LC or moDC targets. Shown are the averaged triplicate means +/− SEM (n=3 independent experiments) for the number of IFN-gamma spot forming cells (SFC) per triplicate of 105 input cells. By Student’s t test, *P < 0.03 for LCs and **P < 0.0001 for moDCs, with vs without blockade of IL15R-alpha.
We then assessed the effect of blocking IL15R-alpha (Figure 3C). After two weekly rounds of restimulation in vitro without addition of exogenous cytokine, ELISpot assays of IFN-gamma secretion by responder PBMCs showed that LCs were again more potent APCs than moDCs. Blocking anti-IL15R-alpha significantly reduced the stimulatory capacity of each DC subtype (Figure 3C). Near complete inhibition of moDC activity indicated that the blocking antibody was effective. Although the inhibition of LC activity by anti-IL15R-alpha achieved statistical significance (P<0.03), it was less complete than with moDCs (P <0.0001).
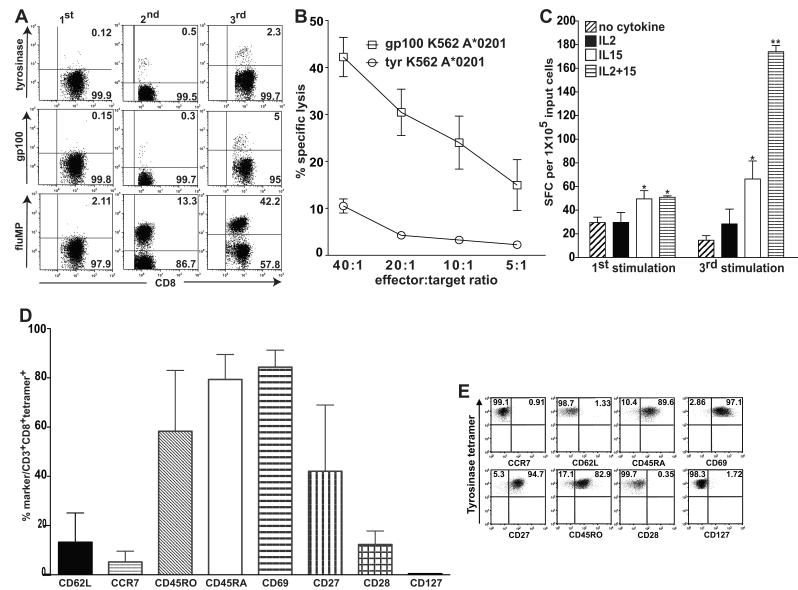
IL15 and IL2 synergize in supporting development of antigen specific CD3+CD8+ T-cells with an IFN-gamma-secreting, cytolytic profile and effector memory phenotype
We used moDCs to distinguish the effects of IL15 from IL2, the receptors for which share common beta and gamma chains. MoDCs depend on exogenous IL15, unlike LCs, so endogenous IL15 would not confound the results. Pilot studies established that IL2 at 10 IU/ml was only minimally additive to IL15 at 10ng/ml in supporting proliferation of antigen-specific T-cells (Supplemental Figures 2 and 3). Representative dot plots from the combined IL2 + IL15 condition in a single set of experiments illustrate the expansion of tetramer-reactive T-cells in vitro against moDC-presented tyrosinase, gp100, and influenza matrix peptide antigens (Figure 4A). These CD3+CD8+ tetramer-reactive T-cells attained a cytotoxic IFN-gamma secreting profile (Figure 4B). In contrast to cell proliferation (Supplemental Figure 3), IL2 and IL15 synergized to support CTL development (P < 0.05 for IL15 or IL2+IL15 vs IL2 or no cytokine; and P < 0.005 by the third round of stimulation for the combination of IL2+IL15 vs IL15 alone).
Figure 4. IL15 and IL2 synergize in supporting development of moDC-stimulated, antigen specific CD3+CD8+ T cells with an IFN-gamma-secreting, cytolytic profile and effector memory phenotype.
(A) CD3+CD8+ T cells were gated from PBMCs obtained from melanoma patients previously vaccinated with tyrosinase, gp100, and flu matrix peptide-pulsed moDCs in vivo. CD3+CD8+ T cells (X axis) that reacted with HLA-A*0201 tetramers bearing the respective peptides (Y axis) were measured at the outset of each restimulation by autologous peptide-pulsed moDCs in vitro. Dot plots show only the combined IL2 (10 IU/ml) + IL15 (10 ng/ml) condition. Numbers in the right upper quadrants represent the percentages of tetramer-reactive cells in the CD3+CD8+ gate. Quadrants are the same for each round of stimulation in a single column but different across rows, because staining and analyses occurred at different time points after each round of stimulation. These data represent one of three independent experiments. (B) ELISpot assays measuring IFN-gamma secretion by responder PBMCs tested as effectors against gp100 peptide, tyrosinase peptide, and fluMP-pulsed moDC targets were performed after the first and third stimulations. Shown are the results for the gp100 target antigen, but the other two antigens stimulated similar response patterns (not shown). The averaged triplicate means ± SEM of IFN-gamma spot forming cells (SFC) per 105 input T cells combined from three independent experiments are shown. **P < 0.005 for the combination of IL2 + IL15 relative to all other conditions after the third round of stimulation. *P < 0.05 relative to the IL2 or no added exogenous cytokine conditions for IL15 either alone at both time points or in combination with IL2 after the first stimulation. (C) gp100-specific CD8+ T cells that developed in the presence of IL2 (10 IU/ml) and IL15 (10ng/ml) killed HLA-A*0201-transfected K562 targets (gift of Dr. Thomas Wolfel, Univ. Mainz, Germany (37)) pulsed with gp100 but not with tyrosinase peptide in a 51Cr release assay. The triplicate means for specific lysis {({sample release - spontaneous release}/{total release – spontaneous release}) × 100%} were averaged from three independent experiments and plotted +/− SEM, P <0.001. (D) The percentages of tyrosinase tetramer-reactive cells that also expressed each of the epitopes listed along the X-axis were pooled from three independent experiments (mean +/− SD). (E) Dot plots from one of the three experiments summarized in (D). Numbers in the upper quadrants indicate the percentages of tetramer reactive cells that did or did not express the respective epitope indicated on the X-axis. While the proportion expressing CD27 was quite variable over the three separate experiments, as shown in (D), the tetramer-reactive T cells from this experiment displayed a very clear population of CD27+ cells.
Because limited cell numbers precluded isolation of CD8+ T-cells for most ELISpot assays, we confirmed cytolytic activity in standard 51Cr release assays (Figure 4C). Bulk responder T-cells included CTLs capable of dose-dependent, MHC-restricted, tumor antigen-specific killing (Figure 4C; P < 0.001). Although IL15 was more effective than IL2 alone in promoting robust expansion of tetramer-reactive, CD3+CD8+ T-cells, IFN-gamma secretion and cytolytic function benefited from IL15 and IL2 synergy when stimulated by peptide-loaded moDCs.
After three 7d rounds of stimulation in vitro in the presence of IL2 and IL15, we assessed activation and lymphoid homing markers on T-cells from the previously vaccinated melanoma patients (Figures 4D, 4E). CD3+CD8+ T-cell responders expressed neither of the lymphoid homing receptors, CCR7 or L-selectin (CD62L); nor did these T-cells express CD28, all of which are usually found on central memory (TCM) or naïve/precursor memory T-cells (22). These tetramer-reactive T-cells instead expressed the activation markers CD45RO, CD69, and variable amounts of CD27, as well as CD45RA (Figure 4D). Data from three independent experiments gated on tyrosinase-reactive tetramers were pooled to illustrate the aggregate phenotype, as well as variability, characteristic of these CD45RA+ effector memory T-cells (TEMRA: CD62Lneg, CCR7neg, CD28neg, CD27variable, CD45RO+, CD69+, and CD45RA+; Figure 5D) (22-24). Figure 4E shows the dot plots from the one experiment of three with the highest number of tetramer-reactive cells among the gated CD3+CD8+ T-cells.
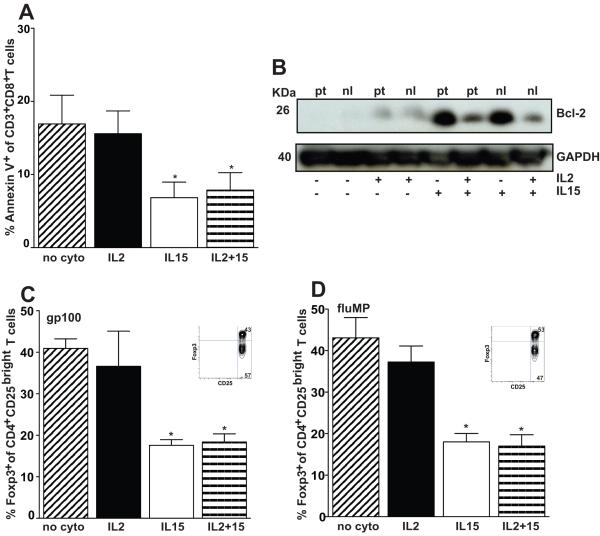
Figure 5. IL15 helps rescue CD3+CD8+ T cells from IL2 induced apoptotic cell death and reduces the frequency of CD4+CD25bright Foxp3+ regulatory T cells after moDC stimulation.
(A) Responder T cells underwent a single round of stimulation by autologous gp100-pulsed moDCs and were assessed for cell death by flow cytometry after gating on the CD3+CD8+ population. The percentages of Annexin-V+ and either PIneg (early apoptotic) or PI+ (late apoptotic) were summed to obtain the total percentage of apoptotic cells. Data represent the averages of duplicate means ± SEM from 6 independent experiments. P < 0.05 for either IL15 condition with or without IL2, compared with IL2 alone or no exogenous cytokine condition. (B) Western blot analysis of Bcl-2 was performed on total cell lysates from CD3+CD8+ T cells purified by positive immunomagnetic selection and stimulated for 7 days by gp100-pulsed autologous moDCs in the presence of different cytokine conditions. CD3+CD8+ T cells originated from a previously vaccinated melanoma patient (pt) or from a healthy donor (nl). Results from one of 3 independent experiments are shown. GAPDH in CD3+CD8+ T cells served as an internal control. (C) T cells were restimulated in vitro twice over 7d each by either autologous (C) gp100- or (D) fluMP-pulsed moDCs, using the indicated cytokine conditions. Bar graphs represent the proportion of phenotypic Tregs based on CD4+CD25bright T cells that coexpressed intracellular Foxp3 (means +/− SD from 3-4 independent experiments). Insets depict the Foxp3+ expression by these gated CD4+CD25bright T cells from a representative condition without cytokine. P < 0.01 for pairwise comparisons between either of the IL15-containing and the IL2 alone or no cytokine conditions.
IL15 prevents contraction of the human immune response by countering apoptosis and expansion of candidate Tregs
We then asked whether IL15 rescued T-cells from IL2 activation-induced cell death or apoptosis. We again focused on moDCs in lieu of LCs, because we could better control the amount of IL15 in the system in vitro. T-cells from the gp100-pulsed moDC-vaccinated melanoma patients underwent a single 7d restimulation with autologous gp100-pulsed moDCs in the presence of IL2 and/or IL15 or neither. CD3+CD8+-gated responder T-cells were analyzed cytofluorographically for early {AnnexinV+ propidium iodide (PI)neg} and late (AnnexinV+ PI+) apoptosis (Figure 5A). IL15 significantly reduced apoptosis, thus prolonging CD3+CD8+ T-cell survival after restimulation (P < 0.05). The control without cytokines was comparable to the condition with exogenous IL2 alone, because such low dose IL2 did not enhance the higher amounts of endogenous IL2 already secreted by DC-stimulated T-cells (20).
CD3+CD8+ responder T-cells increased expression of the antiapoptotic protein, Bcl-2, after restimulation by gp100-pulsed moDCs with exogenous IL15 (Figure 5B). IL15 also mitigated the reduction in Bcl-2 expression caused by IL2. Levels were nearly undetectable with IL2 alone.
Neither mouse nor human studies have addressed in detail whether IL15 could also attenuate Treg expansion by IL2. We therefore determined the percentage of Foxp3+ Tregs among the gated CD3+CD4+CD25bright T-cells responding to gp100 peptide- (Figure 5C) or fluMP- (Figure 5D) pulsed human moDCs. IL15 reduced the proportion of candidate Tregs significantly below the proportions generated by moDCs alone or with only low dose exogenous IL2 (n=3 independent experiments, P < 0.01; Figures 5C and 5D). These data demonstrate that IL15, while expanding total and tetramer-reactive T-cells, reduced the proportion of candidate Tregs that would otherwise respond to IL2 produced in culture or added exogenously. In contrast to the mixed effect of IL15 and IL2 on Bcl-2 expression, IL15 suppression of these phenotypic Tregs overrode any effect of IL2.
DISCUSSION
This clinical trial was the first head-to-head comparison between vaccines using peptide-pulsed LCs or moDCs in humans. Immune responses to tumor peptide and control antigens developed in vivo in patients with advanced melanoma in response to both DC subtypes, which proved safe and well-tolerated in all patients. There was a trend toward greater erythema and induration 48hrs after peptide-pulsed moDC injections, possibly indicating less trafficking away from the local sites to draining nodes; but these reactions were not significantly different from those after LC-based vaccines. The increased proliferative response to KLH rechallenge with moDCs may also account for their trend toward greater DTH responses. We set a high threshold to define a positive response, and LC-based vaccines stimulated significantly greater tyrosinase-HLA-A*0201 tetramer reactivity than did moDC-based vaccines. Beyond that, the results of this particular trial did not yet corroborate extensive data in vitro that had predicted LC superiority across the board (1, 2) or confirm the findings of some investigators suggesting that there is an advantage to including CD34+ HPC-derived LCs in DC vaccines (3).
We conducted additional experiments to establish mechanisms that could account for LC superiority over moDCs in vitro (1, 2), using T-cells primed in vivo by DC vaccination and focusing on the activity of IL15 with a view toward future optimization of tumor Ag presentation. IL15 is a pleiotropic cytokine that affects homeostasis, activation, and homing of lymphocytes in both innate and adaptive immunity (5). LCs secrete more IL15 than do moDCs (1, 4), however; and when exogenous IL15 was either limiting or absent, only LCs could still stimulate a statistically significant enhancement of IFN-gamma production by tumor and viral antigen-specific T-cells. Anti-IL15R-alpha significantly inhibited both LC- and moDC-stimulated IFN-gamma responses, yet blockade was incomplete for LCs. Limited cell numbers precluded isolation of CD8+ T-cell responders, so CD4+ T-cells could have been less sensitive to IL15R-alpha blockade. Lacking a positive control to guide antibody dosing, complete blockade of LCs may also have required a higher MAb concentration. Other unmeasured factors could have played roles downstream of the initial T-cell activation events, especially over two 7d rounds of stimulation.
Somewhat paradoxically, we then concentrated on mature moDCs so that we could control the amount of IL15 in the system in vitro and not confound our assessments by what LCs could produce themselves. Exposure to IL15 during moDC development confers Langerhans-like properties on moDCs (14, 15), but does not provide IL15 during DC stimulation of tumor-specific T-cells, which was the focus of our inquiry.
Our data demonstrated that IL15 supported the long-term expansion by DCs of tumor antigen-specific T-cells with effector memory phenotype (TEMRA) and cytolytic function. There was a modest additive effect on proliferation when IL2 combined with IL15. The two cytokines were synergistic in enhancing CTL activity stimulated by moDCs against melanoma antigens, however, with IL2 being an essential component of CD4+ T-cell help for the long-term expansion of CD8+ memory T-cells (25-30). Low dose exogenous IL2 avoided bystander activation of either IL15R (21) or NK cells in the responder populations, insofar as conventional DCs already stimulate T-cells to secrete comparable or higher amounts of IL2 (20). Hence, IL2 did not add much to the DC-stimulated controls without exogenous cytokines.
More importantly, IL15 countered the apoptosis induced by IL2 activation, which otherwise impeded T-cell expansion by IL2 beyond the first week or two. IL15 achieved this by partially abrogating IL2’s reduction of the antiapoptotic protein, Bcl-2. Mouse data predicted these findings, but there have been no comparable studies with human cells. IL15 also trumped all of the IL2 effect on T reg expansion, which has not been previously reported. These data with human cells confirm that IL2 and IL15 have distinct roles in DC-stimulated T-cell apoptosis and survival.
Antigen-experienced T-cells comprise both effector memory (TEM) and central memory T-cells (TCM), which home respectively to inflamed tissues and lymphoid organs (7, 22). Investigators have ascribed TCM development to IL15 and TEM generation to IL2 (6, 7), but IL15 supports TEM in HIV infection (8). Our data demonstrated that conventional DCs with IL15 expanded both tumor and viral antigen-reactive human TEM cells lacking CCR7, CD62L, CD127, yet coexpressing CD45RO, CD45RA, and CD69. This characterizes a CD45RA+ TEM subpopulation (TEMRA) (22), which has the largest amount of perforin and appears late in the immune response (31-33). Somewhat atypical was the persistent expression of CD27 on these cells, although a subset of CD27+ TEMRA cells with intermediate effector activity exists (34). Resting rather than activated populations like those in this study, however, have mostly established the phenotypes for TCM and TEM cells. Our data also showed that TEMRA cells developed under antigen-bearing DC-driven conditions with IL15, arguing against their development through homeostatic rather than antigen-driven pathways (31, 32).
Despite encouraging findings from the clinical trial establishing safety and efficacy of LC and moDC-based vaccines, a clear winner did not emerge across all antigens tested, although LCs stimulated significantly greater tetramer reactivity at least against HLA-A*0201-restricted tyrosinase peptide. Among the outstanding challenges, foremost is the need to reconcile abundant data in vitro that LCs are in fact superior to moDCs for stimulation of virus and tumor antigen-specific CTLs, with the responses observed in vivo. The clinical trial data do not exclude the possibility that sufficient IL15 may have been present in vivo, as reported in a mouse model of influenza infection (35). Given the trend toward greater DTH reactivity after moDC vaccination and the significant increase in moDC-stimulated KLH specific proliferation on rechallenge, we also cannot exclude induction in vivo of IL15 in moDCs by KLH-specific CD4+ T cells.
Additional limitations when translating studies in vitro to a clinical trial included differences in peptide hydrophobicity that affected solubility and loading onto class I MHC. There was also no reliable way to confirm that sufficient peptide remained bound to MHC long enough to stimulate T-cells in draining lymph nodes. These problems beg for approaches that achieve durable expression of full length tumor antigens, which could in turn be processed into multiple epitopes for simultaneous presentation on both class I and II MHC. Furthermore, because LCs secrete more IL15 (1, 2, 4) than do moDCs, and given that LCs can stimulate significantly higher numbers of virus- and tumor-specific T-cells in the complete absence of exogenous IL15 or IL2, LCs merit further specific investigation as immunogens for the optimal induction of tumor immunity. Alternatively, continued use of moDCs as vaccines would benefit from supplementation with IL15 by transfection (36) or direct administration of the cytokine as a drug (#NCT01021059, ClinicalTrials.gov). Available reagents and approaches have not yet detected IL15R-alpha by flow cytometry on human CD34+ HPC-derived LCs (1, 4), which is a prerequisite for binding and presenting IL15 in trans to responder lymphocytes. Alternative approaches are underway to resolve this conundrum and to ascertain distinctions between the mechanisms used by LCs vs moDCs for early T-cell activation events that depend on IL15.
Supplementary Material
Statement of Translational Relevance.
Dendritic cell (DC) vaccination protocols most often use monocyte-derived DCs (moDCs), although Langerhans-type DCs (LCs) have repeatedly proven superior to moDCs and other conventional DCs for stimulating immunity against viral and tumor antigens in vitro. We report a phase I comparison trial between melanoma peptide-pulsed LCs and moDCs, where LCs proved more potent than moDCs for one parameter but were otherwise comparable in eliciting immunity against melanoma antigens. We nevertheless established a critical role for IL15 in countering IL2-induced apoptosis and phenotypic Treg expansion during CTL generation by DC stimulation. MoDCs require exogenous IL15, whereas LCs do not. LCs providing endogenous IL15 therefore merit optimization as cancer immunogens in vivo, whereas continued use of moDCs will require exogenous IL15. Circumventing the inherent limitations of single peptides will require improved methods for loading full-length antigen for processing and presentation of multiple class I and II MHC-restricted epitopes.
ACKNOWLEDGMENTS
We appreciate the stimulating discussion and intellectual input from other members of the Young Lab and the technical assistance of Jennifer P. Ghith, Nicholas Sakellarios, and Dana E. Pepe. The support and assistance of the MSKCC attending physicians, nurses, and staff of the Melanoma/Sarcoma and Adult Bone Marrow Transplant Services, the Blood Bank Donor Room, and the Cytotherapy Lab in obtaining patient samples are gratefully acknowledged. We also thank the clinical trial participants and healthy volunteers who provided samples for research.
SUPPORTED BY: R01-CA083070 (JWY), R01-CA118974 (JWY), R21-CA119528 (JWY), R21-CA097714 (JWY), P01-CA23766 (GH, JWY), from the National Cancer Institute, National Institutes of Health; Mr. William H. Goodwin and Mrs. Alice Goodwin of the Commonwealth Cancer Foundation for Research and The Experimental Therapeutics Center of Memorial Sloan-Kettering Cancer Center (JWY); Swim Across America (JDW, JWY), and the University of Rome “La Sapienza” (ER).
Footnotes
EXPLANATION OF AUTHORS’ CONTRIBUTIONS: ER, MR, GR, JY, and JWY designed, planned, and performed the experiments. JY, KSP, PBC, and ANH designed the clinical trial. GR, MR, MAdeC, ER, DJC, JY, and JWY conducted the clinical trial. KSP assisted with statistical design of the clinical trial, and GH provided statistical design and analysis of the trial and lab correlative studies. JWY conceived, planned, and reviewed the experiments. ER and JWY wrote the manuscript.
CONFLICTS OF INTEREST: The authors have no conflicts of interest to declare
Online supplemental material Supplemental Figure 1 shows the protocol schema. Supplemental Figure 2 is a dose-finding pilot study for IL2 and IL15 using fluMP-pulsed moDC stimulators and fluMP-HLA-A*0201 tetramer reactive T-cell readouts. Supplemental Figure 3 illustrates the proliferation of total cells and the absolute number of CD3+ CD8+ T-cells reactive with tyrosinase, gp100, and fluMP-HLA-A*0201 tetramers over 3-4 rounds of weekly stimulation with and without IL2, IL15, or both in combination.
REFERENCES
- 1.Ratzinger G, Baggers J, de Cos MA, et al. Mature human Langerhans cells derived from CD34+ hematopoietic progenitors stimulate greater cytolytic T lymphocyte activity in the absence of bioactive IL-12p70, by either single peptide presentation or cross-priming, than do dermal-interstitial or monocyte-derived dendritic cells. J Immunol. 2004;173:2780–91. doi: 10.4049/jimmunol.173.4.2780. [DOI] [PubMed] [Google Scholar]; J Immunol. 174:3818. Erratum in. 005. [Google Scholar]
- 2.Klechevsky E, Morita R, Liu M, et al. Functional specializations of human epidermal Langerhans cells and CD14+ dermal dendritic cells. Immun. 2008;29:497–510. doi: 10.1016/j.immuni.2008.07.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Banchereau J, Palucka AK, Dhodapkar M, et al. Immune and clinical responses in patients with metastatic melanoma to CD34(+) progenitor-derived dendritic cell vaccine. Cancer Res. 2001;61:6451–8. [PubMed] [Google Scholar]
- 4.Munz C, Dao T, Ferlazzo G, de Cos MA, Goodman K, Young JW. Mature myeloid dendritic cell subsets have distinct roles for activation and viability of circulating human natural killer cells. Blood. 2005;105:266–73. doi: 10.1182/blood-2004-06-2492. [DOI] [PubMed] [Google Scholar]
- 5.Waldmann TA. The biology of interleukin-2 and interleukin-15: implications for cancer therapy and vaccine design. Nat Rev Immunol. 2006;6:595–601. doi: 10.1038/nri1901. [DOI] [PubMed] [Google Scholar]
- 6.Klebanoff CA, Gattinoni L, Torabi-Parizi P, Kerstann K, Cardones AR, Finkelstein SE, et al. Central memory self/tumor-reactive CD8+ T cells confer superior antitumor immunity compared with effector memory T cells. Proc Natl Acad Sci U S A. 2005;102:9571–6. doi: 10.1073/pnas.0503726102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Weninger W, Crowley MA, Manjunath N, von Andrian UH. Migratory properties of naive, effector, and memory CD8(+) T cells. J Exp Med. 2001;194:953–66. doi: 10.1084/jem.194.7.953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mueller YM, Bojczuk PM, Halstead ES, et al. IL-15 enhances survival and function of HIV-specific CD8+ T cells. Blood. 2003;101:1024–9. doi: 10.1182/blood-2002-07-1957. [DOI] [PubMed] [Google Scholar]
- 9.Oh S, Perera LP, Burke DS, Waldmann TA, Berzofsky JA. IL-15/IL-15Ralpha-mediated avidity maturation of memory CD8+ T cells. Proc Natl Acad Sci U S A. 2004;101:15154–9. doi: 10.1073/pnas.0406649101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Stonier SW, Ma LJ, Castillo EF, Schluns KS. Dendritic cells drive memory CD8 T cell homeostasis via IL-15 trans-presentation. Blood. 2008 doi: 10.1182/blood-2008-05-156307. blood-2008-05-156307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Dubois SP, Waldmann TA, Müller R., Jr Survival adjustment of mature dendritic cells by IL-15. Proceedings of the National Academy of Sciences of the United States of America. 2005;102:8662–7. doi: 10.1073/pnas.0503360102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Berger C, Berger M, Hackman RC, et al. Safety and immunologic effects of IL-15 administration in nonhuman primates. Blood. 2009;114:2417–26. doi: 10.1182/blood-2008-12-189266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Banchereau J, Palucka AK. Dendritic cells as therapeutic vaccines against cancer. Nat Rev Immunol. 2005;5:296–306. doi: 10.1038/nri1592. [DOI] [PubMed] [Google Scholar]
- 14.Mohamadzadeh M, Berard F, Essert G, et al. Interleukin 15 Skews Monocyte Differentiation into Dendritic Cells with Features of Langerhans Cells. J Exp Med. 2001;194:1013–20. doi: 10.1084/jem.194.7.1013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dubsky P, Saito H, Leogier M, et al. IL-15-induced human DC efficiently prime melanoma-specific naive CD8+ T cells to differentiate into CTL. Eur J Immunol. 2007;37:1678–90. doi: 10.1002/eji.200636329. [DOI] [PubMed] [Google Scholar]
- 16.Ratzinger G, Baggers J, de Cos MA, et al. Mature Human Langerhans Cells Derived from CD34+ Hematopoietic Progenitors Stimulate Greater Cytolytic T Lymphocyte Activity in the Absence of Bioactive IL-12p70, by Either Single Peptide Presentation or Cross-Priming, Than Do Dermal-Interstitial or Monocyte-Derived Dendritic Cells. J Immunol. 2005;174:3818. doi: 10.4049/jimmunol.173.4.2780. [DOI] [PubMed] [Google Scholar]
- 17.Szabolcs P, Moore MAS, Young JW. Expansion of immunostimulatory dendritic cells among the myeloid progeny of human CD34+ bone marrow precursors cultured with c-kit ligand, granulocyte-macrophage colony-stimulating factor, and TNF-α. JImmunol. 1995;154:5851–61. [PubMed] [Google Scholar]
- 18.Szabolcs P, Avigan D, Gezelter S, et al. Dendritic cells and macrophages can mature independently from a human bone marrow-derived, post-colony-forming unit intermediate. Blood. 1996;87:4520–30. [PubMed] [Google Scholar]
- 19.Maecker HT, Moon J, Bhatia S, et al. Impact of cryopreservation on tetramer, cytokine flow cytometry, and ELISPOT. BMC Immunol. 2005;6:17. doi: 10.1186/1471-2172-6-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Young JW, Baggers J, Soergel SA. High dose UV-B radiation alters dendritic cell costimulatory activity but does not tolerize T lymphocytes to alloantigen in vitro. Blood. 1993;81:2987–97. [PubMed] [Google Scholar]
- 21.Fehniger TA, Caligiuri MA. Interleukin 15: biology and relevance to human disease. Blood. 2001;97:14–32. doi: 10.1182/blood.v97.1.14. [DOI] [PubMed] [Google Scholar]
- 22.Sallusto F, Geginat J, Lanzavecchia A. Central memory and effector memory T cell subsets: function, generation, and maintenance. Annu Rev Immunol. 2004;22:745–63. doi: 10.1146/annurev.immunol.22.012703.104702. [DOI] [PubMed] [Google Scholar]
- 23.Sallusto F, Lenig D, Forster R, Lipp M, Lanzavecchia A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature. 1999;401:708–12. doi: 10.1038/44385. [DOI] [PubMed] [Google Scholar]
- 24.Romero P, Zippelius A, Kurth I, et al. Four functionally distinct populations of human effector-memory CD8+ T lymphocytes. J Immunol. 2007;178:4112–9. doi: 10.4049/jimmunol.178.7.4112. [DOI] [PubMed] [Google Scholar]
- 25.Williams MA, Tyznik AJ, Bevan MJ. Interleukin-2 signals during priming are required for secondary expansion of CD8+ memory T cells. Nature. 2006;441:890–3. doi: 10.1038/nature04790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Williams MA, Bevan MJ. Effector and Memory CTL Differentiation. Annual Review of Immunology. 2007;25:171–92. doi: 10.1146/annurev.immunol.25.022106.141548. [DOI] [PubMed] [Google Scholar]
- 27.Kamimura D, Bevan MJ. Naive CD8+ T cells differentiate into protective memory-like cells after IL-2 anti IL-2 complex treatment in vivo. J Exp Med. 2007;204:1803–12. doi: 10.1084/jem.20070543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Boeckh M, Leisenring W, Riddell SR, et al. Late cytomegalovirus disease and mortality in recipients of allogeneic hematopoietic stem cell transplants: importance of viral load and T-cell immunity. Blood. 2003;101:407–14. doi: 10.1182/blood-2002-03-0993. [DOI] [PubMed] [Google Scholar]
- 29.Bonini C, Lee SP, Riddell SR, Greenberg PD. Targeting antigen in mature dendritic cells for simultaneous stimulation of CD4+ and CD8+ T cells. J Immunol. 2001;166:5250–7. doi: 10.4049/jimmunol.166.8.5250. [DOI] [PubMed] [Google Scholar]
- 30.Hakki M, Riddell SR, Storek J, et al. Immune reconstitution to cytomegalovirus after allogeneic hematopoietic stem cell transplantation: impact of host factors, drug therapy, and subclinical reactivation. Blood. 2003;102:3060–7. doi: 10.1182/blood-2002-11-3472. [DOI] [PubMed] [Google Scholar]
- 31.Alves NL, Hooibrink B, Arosa FA, van Lier RAW. IL-15 induces antigen-independent expansion and differentiation of human naive CD8+ T cells in vitro. Blood. 2003;102:2541–6. doi: 10.1182/blood-2003-01-0183. [DOI] [PubMed] [Google Scholar]
- 32.Geginat J, Lanzavecchia A, Sallusto F. Proliferation and differentiation potential of human CD8+ memory T-cell subsets in response to antigen or homeostatic cytokines. Blood. 2003;101:4260–6. doi: 10.1182/blood-2002-11-3577. [DOI] [PubMed] [Google Scholar]
- 33.Hislop AD, Annels NE, Gudgeon NH, Leese AM, Rickinson AB. Epitope-specific evolution of human CD8(+) T cell responses from primary to persistent phases of Epstein-Barr virus infection. J Exp Med. 2002;195:893–905. doi: 10.1084/jem.20011692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rufer N, Zippelius A, Batard P, et al. Ex vivo characterization of human CD8+ T subsets with distinct replicative history and partial effector functions. Blood. 2003;102:1779–87. doi: 10.1182/blood-2003-02-0420. [DOI] [PubMed] [Google Scholar]
- 35.McGill J, Van Rooijen N, Legge KL. IL-15 trans-presentation by pulmonary dendritic cells promotes effector CD8 T cell survival during influenza virus infection. J Exp Med. 2010;207:521–34. doi: 10.1084/jem.20091711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Steel JC, Ramlogan CA, Yu P, et al. Interleukin-15 and its receptor augment dendritic cell vaccination against the neu oncogene through the induction of antibodies partially independent of CD4 help. Cancer Res. 2010;70:1072–81. doi: 10.1158/0008-5472.CAN-09-1301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Britten CM, Meyer RG, Kreer T, Drexler I, Wolfel T, Herr W. The use of HLA-A*0201-transfected K562 as standard antigen-presenting cells for CD8(+) T lymphocytes in IFN-gamma ELISPOT assays. J Immunol Methods. 2002;259:95–110. doi: 10.1016/s0022-1759(01)00499-9. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.