Abstract
Little is known about the pathophysiology of acute exacerbation (AE) of idiopathic pulmonary fibrosis (IPF). Heat shock protein 47 (HSP47), a collagen-specific molecular chaperone, is essential for biosynthesis and secretion of collagen molecules. Previous studies in experimental animal fibrosis models have shown that downregulation of HSP47 expression reduces collagen production and diminishes fibrosis progression. In this study, serum HSP47 levels were evaluated to elucidate pathogenic differences involving HSP47 between AE-IPF and stable (S)-IPF. Subjects comprised 20 AE-IPF and 33 S-IPF patients. Serum levels of HSP47, Krebs von den Lungen-6 (KL-6), surfactant protein (SP)-A, SP-D, and lactate dehydrogenase (LDH) were measured. Immunohistochemical analysis of lung HSP47 expression was determined in biopsy and autopsy tissues diagnosed as diffuse alveolar damage (DAD) and usual interstitial pneumonia (UIP). Serum levels of HSP47 were significantly higher in AE-IPF than in S-IPF patients, whereas serum levels of KL-6, SP-A, and SP-D did not differ significantly. Receiver operating characteristic curves revealed that HSP47 was superior for discriminating AE-IPF and S-IPF. The cutoff for HSP47 resulting in the highest diagnostic accuracy was 559.4 pg/mL; sensitivity, specificity, and diagnostic accuracy were 100.0 %, 93.9 %, and 96.2 %, respectively. Immunohistochemical analysis revealed that pulmonary HSP47 expression was greater in DAD than UIP tissues. Serum HSP47 was significantly higher in AE-IPF than in S-IPF patients, suggesting that underlying fibrogenic mechanisms involving HSP47 differ in the two conditions.
Keywords: Acute exacerbation of idiopathic pulmonary fibrosis, Heat shock protein 47, Krebs von den Lungen-6, Surfactant protein A, Surfactant protein D
Background
The natural history of idiopathic pulmonary fibrosis (IPF) is complex, and many patients have an unpredictable disease course with periods of relative stability punctuated by episodes of acute and often fatal decline (Hyzy et al. 2007; Collard et al. 2007; Song et al. 2010; Raghu et al. 2011). This acute worsening of the disease is sometimes attributed to identifiable conditions such as pneumonia or heart failure. However, many of these events occur without an identifiable cause, and therefore are termed acute exacerbations of (AE)-IPF. AE-IPF histologically manifests as acute or organizing diffuse alveolar damage (DAD) (Raghu et al. 2011). During the exudative (acute) phase of DAD, there is interstitial edema, acute and chronic inflammation, type II cell hyperplasia, and hyaline membrane formation. Despite extensive research conducted in the past, little is known about the pathophysiology of AE-IPF.
Heat shock protein 47 (HSP47), a collagen-specific molecular chaperone, is essential for the biosynthesis and secretion of collagen molecules (Nagata 1996; Nagata et al. 1986, 1988; Ishida et al. 2006; Nagai et al. 2000; Sauk et al. 1994). Previous studies of various experimental diseases affecting the lungs have shown that there is a close association between increased expression of HSP47 and excessive accumulation of collagen (Ishii et al. 2003; Kakugawa et al. 2004, 2010). Other studies have demonstrated that HSP47 expression is highly tissue- and cell-specific, mainly restricted to phenotypically altered collagen producing cells and is well correlated with expression of collagen (Ishii et al. 2003; Iwashita et al. 2000; Kakugawa et al. 2004, 2005, 2010; Abe et al. 2000; Shioshita et al. 2000). It was reported that HSP47 expression was higher in the lungs of patients with idiopathic usual interstitial pneumonia (UIP) than in those with collagen vascular disease-associated UIP and idiopathic nonspecific interstitial pneumonia (NSIP) (Kakugawa et al. 2005); idiopathic fibrotic NSIP patients with higher pulmonary HSP47 expression had poorer prognosis than patients with lower levels (Amenomori et al. 2010). These findings suggest that HSP47 plays an important role in the fibrotic process and correlates with fibrotic disease activity.
Recent data have suggested that AE-IPF may be due to an acceleration of the underlying fibroproliferative disease process (Konishi et al. 2009; Collard et al. 2007); it is therefore assumed that HSP47 is also involved in its pathogenesis. Previous studies in several experimental animal fibrosis models have shown that downregulation of HSP47 expression reduces production of collagen and subsequently diminishes progression of fibrosis (Sunamoto et al. 1998; Nishino et al. 2003; Hagiwara et al. 2007a, b, c; Obata et al. 2012). Therefore, HSP47 may be a novel therapeutic target for patients with AE-IPF. Accordingly, the interesting clinical question of whether HSP47 is upregulated in patients with AE-IPF warrants investigation.
The purpose of this study was to compare serum HSP47 concentrations in patients with AE-IPF and stable (S)-IPF, as well as to assess and compare pulmonary expression levels in biopsy and autopsy tissues diagnosed as DAD and UIP.
Materials and methods
Study population
Study subjects consisted of patients who were admitted to Nagasaki University Hospital from April 1996 to April 2012—20 with AE-IPF and 33 with S-IPF. Diagnoses were made according to the official ATS/ERS/JRS/ALAT statement (Raghu et al. 2011): 19 of 33 (57.6 %) S-IPF diagnoses were confirmed pathologically in multiple lobes by open lung biopsy or video-assisted thoracoscopic surgery. In addition, seven of the 33 S-IPF patients were identical to seven of the 20 with AE-IPF; these patients were first diagnosed with IPF, and later experienced an acute exacerbation. None of the IPF patients had any signs or positive serological, or other, markers of collagen vascular disease. S-IPF patients were defined as outpatients with IPF who were not experiencing a rapid decline in respiratory function (Collard et al. 2010). Patients with AE-IPF met the following criteria: (1) unexplained worsening of dyspnea within 1 month; (2) evidence of hypoxemia, as defined by worsened or severely impaired gas exchange; (3) new radiographic alveolar infiltrates; and (4) absence of an alternative explanation such as infection, pulmonary embolism, pneumothorax, or heart failure (Raghu et al. 2011). Serological and urinary studies were performed and were negative in all patients diagnosed with AE-IPF for the following pathogens and pathogen components: endotoxin, Mycoplasma pneumoniae, Chlamydophila pneumoniae, Chlamydophila psittaci, cytomegalovirus antigen, β-D glucan, Legionella spp., and Streptococcus pneumoniae. Blood, sputum, and urine cultures were also negative. An echocardiography demonstrated no evidence of heart failure in any of the patients. Sera were obtained at the time of diagnosis and stored at −80 °C. Patient characteristics were collected from clinical notes recorded at the time of diagnosis and included age, sex, PaO2/fraction of inspired oxygen ratio (P/F ratio), and alveolar–arterial difference of oxygen (A-a DO2). Serum concentrations of Krebs von den Lungen-6 (KL-6), surfactant protein (SP)-A, SP-D, and lactate dehydrogenase (LDH) were also collected from the clinical notes recorded at the time of diagnosis. For records lacking data for these markers, measurements were done using the preserved serum samples. The 30- and 90-day mortality rates were determined for all disease groups.
The study protocol was approved by the institutional review board of Nagasaki University Hospital and the ethics committee, Nagasaki University Graduate School of Biomedical Sciences. A written informed consent was obtained from all subjects.
Sandwich enzyme-linked immunosorbent assay for determining HSP47 concentration
Sandwich enzyme-linked immunosorbent assay for determining HSP47 concentration was carried out as described previously (Yokota et al. 2000, 2003). Serum levels of HSP47 were measured in all enrolled patients.
Measurement of serum KL-6, SP-A, SP-D, and LDH levels
Serum levels of KL-6, SP-A, SP-D, and LDH were measured using specific kits according to the manufacturers’ protocols. KL-6 concentrations were measured using a sandwich-type electrochemiluminescence immunoassay kit (Picolumi KL-6; Sanko Junyaku Co., Tokyo, Japan). SP-A and SP-D levels were measured using sandwich-type enzyme immunoassay kits (SP-A test-F: Kokusai Shiyaku Co., Hyogo, Japan; SP-D kit: Yamasa; Yamasa Shoyu Co., Tokyo, Japan). LDH levels were measured using an ultraviolet method with an L-type WAKO LDH kit (Wako Pure Chemical Industries, Ltd., Osaka, Japan). All assays were performed in duplicate. Data regarding these markers were not obtained from all enrolled patients because some preserved serum samples were not large enough. Serum levels of SP-A were measured in 17 of 20, and of KL-6, SP-D, and LDH in all, patients with AE-IPF. Of the 33 S-IPF patients, the numbers in whom these markers were measured are as follows: KL-6, 29; SP-A, 29; SP-D, 28; and LDH, 29.
Immunohistochemistry
Immunohistochemistry was performed as described previously (Kakugawa et al. 2005).
Statistical analysis
The values of continuous variables were expressed as median (range). The differences among the groups were determined by the Kruskal–Wallis test for continuous variables and the chi-squared test for categorical variables, as appropriate. The upper left corner coordinate point of the receiver operating characteristic (ROC) curve was used to determine the optimum cutoff level for discriminating between AE-IPF and S-IPF. Changes in serum levels were analyzed by the Wilcoxon’s signed rank test. A statistical analysis was performed using the statistical software package (SAS 9.1.3, SAS Institute, Cary, NC, USA). A P value <0.05 was considered statistically significant.
Results
Patient characteristics
Table 1 lists characteristics of enrolled patients. The P/F ratio of the AE-IPF group was significantly lower and the A-a DO2 significantly higher, as compared with the S-IPF group.
Table 1.
Patient characteristics
| Stable IPF (N = 33) | Acute exacerbation of IPF (N = 20) | P value | |||
|---|---|---|---|---|---|
| Age (years) | 65.0 | (34–79) | 69.0 | (41–82) | 0.13 |
| Sex (male/female) | 25/8 | 14/6 | 0.64 | ||
| Smoking (s/ex/n) | 8/15/10 | 1/14/5 | 0.12 | ||
| P/F ratio | 384.3 | (283.3–491.9) | 138.7 | (55.4–276.3) | <0.001 |
| A-a DO2 | 19.4 | (−3.5 to 52.7) | 185.8 | (80.8–553.2) | <0.001 |
Data presented as median (range). P/F ratio is PaO2/fraction of inspired oxygen ratio
IPF idiopathic pulmonary fibrosis, s current smoker, ex ex-smoker, n nonsmoker
Disease outcomes
In the AE-IPF group, the 30- and 90-day mortality rates were 25.0 % (five of 20) and 65.0 % (13 of 20), respectively.
Serum levels of HSP47, KL-6, SP-A, SP-D, and LDH
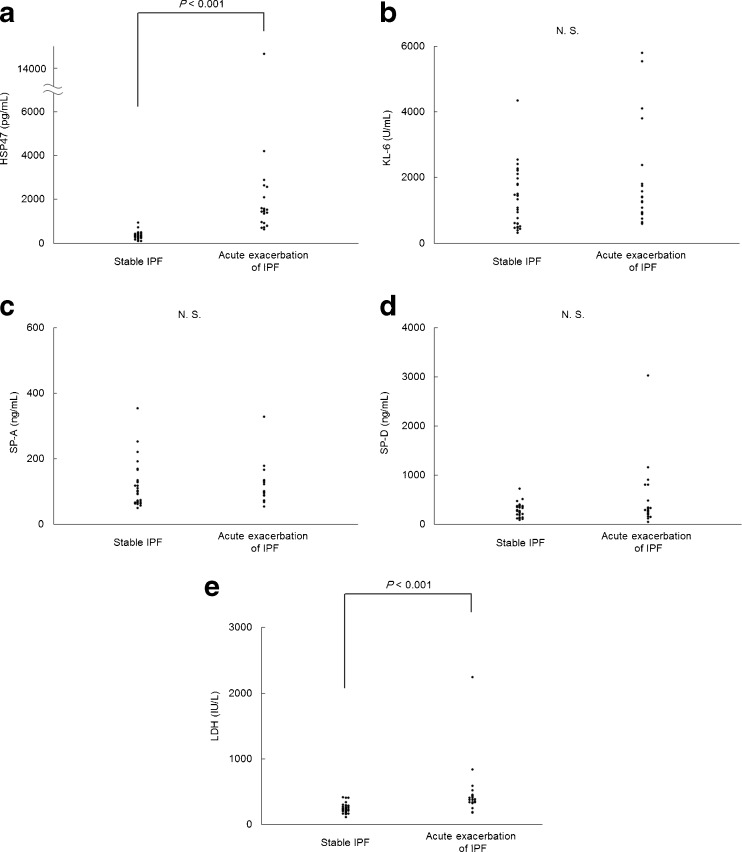
Serological data are presented in Table 2 and Fig. 1. Serum levels of HSP47 and LDH were significantly higher in the acute exacerbation, compared with the stable, IPF group (Fig. 1a, e), whereas levels of KL-6, SP-A, and SP-D did not differ significantly (Fig. 1b, c, d).
Table 2.
Serum levels of HSP47, KL-6, SP-A, SP-D, and LDH
| Stable IPF | Number | Acute exacerbation of IPF | Number | P value | |||
|---|---|---|---|---|---|---|---|
| HSP47 (pg/mL) | 330.9 | (102.2–918.9) | 33 | 1,464.4 | (631.2–14,650) | 20 | <0.001 |
| KL-6 (U/mL) | 1,330.0 | (319–4,340) | 29 | 1,395.0 | (594–5,800) | 20 | 0.25 |
| SP-A (ng/mL) | 93.8 | (42.2–355) | 29 | 101.0 | (54–328) | 17 | 0.31 |
| SP-D (ng/mL) | 271.0 | (19.8–721) | 28 | 289.0 | (44.8–3,030) | 20 | 0.33 |
| LDH (IU/L) | 235.0 | (113–416) | 29 | 382.0 | (177–2,250) | 20 | <0.001 |
Data presented as median (range)
Fig. 1.
Scattergram of serum levels of (a) HSP47, (b) KL-6, (c) SP-A, (d) SP-D and (e) LDH in patients with stable IPF and acute exacerbation of IPF. Not significant (N.S.)
Changes in serum levels of HSP47, KL-6, SP-A, SP-D, and LDH
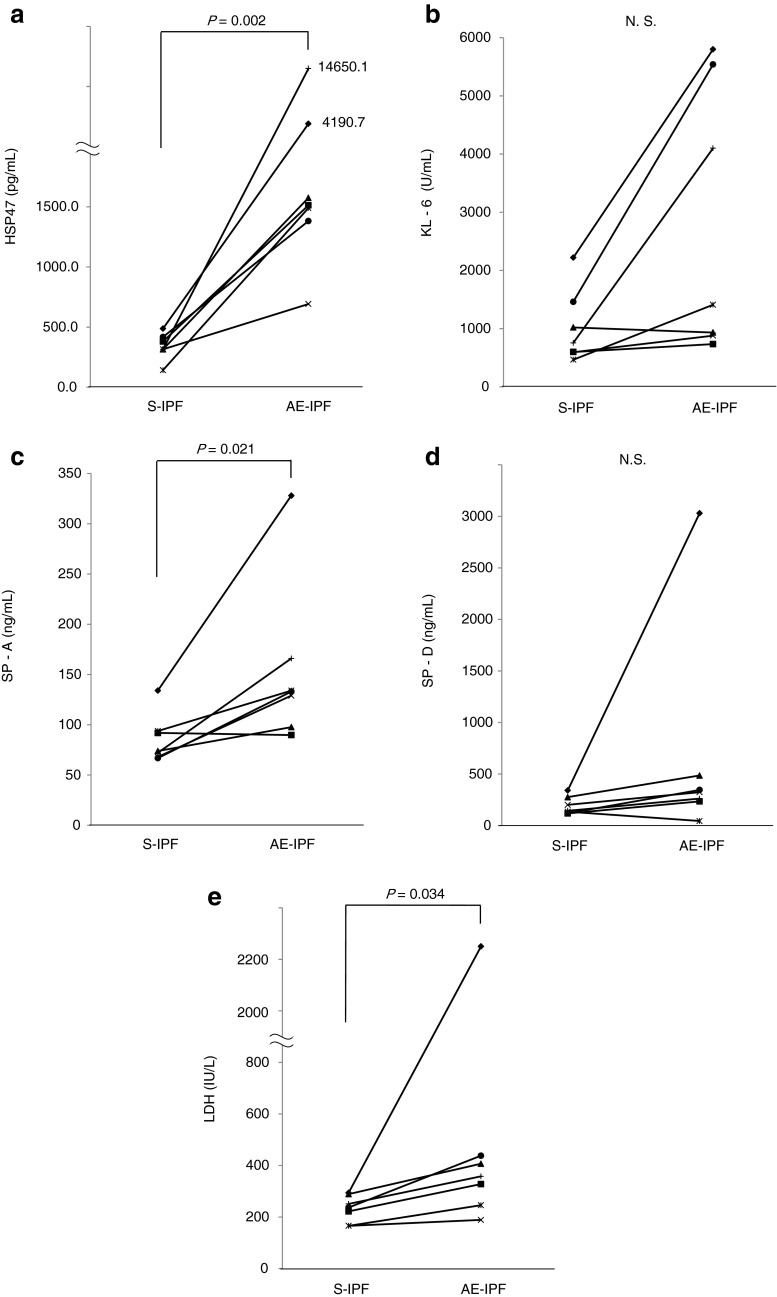
Changes in levels of HSP47, KL-6, SP-A, SP-D, and LDH were compared in seven S-IPF patients who subsequently developed AE-IPF (Table 3; Fig. 2). Sera were obtained at the time of each diagnosis. Levels of HSP47, SP-A, and LDH were significantly higher on diagnosis of the acute exacerbation as compared with levels on diagnosis of S-IPF, but KL-6 and SP-D levels did not change significantly.
Table 3.
Changes in serum levels of HSP47, KL-6, SP-A, SP-D and LDH in seven patients diagnosed first with stable IPF and subsequently with acute exacerbation of IPF
| S-IPF (N = 7) | AE-IPF (N = 7) | P value | |||
|---|---|---|---|---|---|
| HSP47 (pg/mL) | 314.8 | (141.8–487.4) | 1,512.9 | (692.3–14,650) | 0.002 |
| KL-6 (U/mL) | 754.0 | (465–2,220) | 1,410.0 | (734–5,800) | 0.110 |
| SP-A (ng/mL) | 73.9 | (66.8–134) | 133.0 | (89.8–328) | 0.021 |
| SP-D (ng/mL) | 144.0 | (118–341) | 324.0 | (45–3,030) | 0.110 |
| LDH (IU/L) | 237.0 | (166–294) | 358.0 | (189–2,250) | 0.034 |
Sera were obtained at diagnosis. Data presented as median (range)
Fig. 2.
Changes in serum levels of (a) HSP47, (b) KL-6, (c) SP-A, (d) SP-D and (e) LDH in seven patients diagnosed with stable IPF and subsequently with acute exacerbation of IPF. Sera were obtained at diagnosis. Stable IPF (S-IPF), acute exacerbation of IPF (AE-IPF), not significant (N.S.)
ROC curve
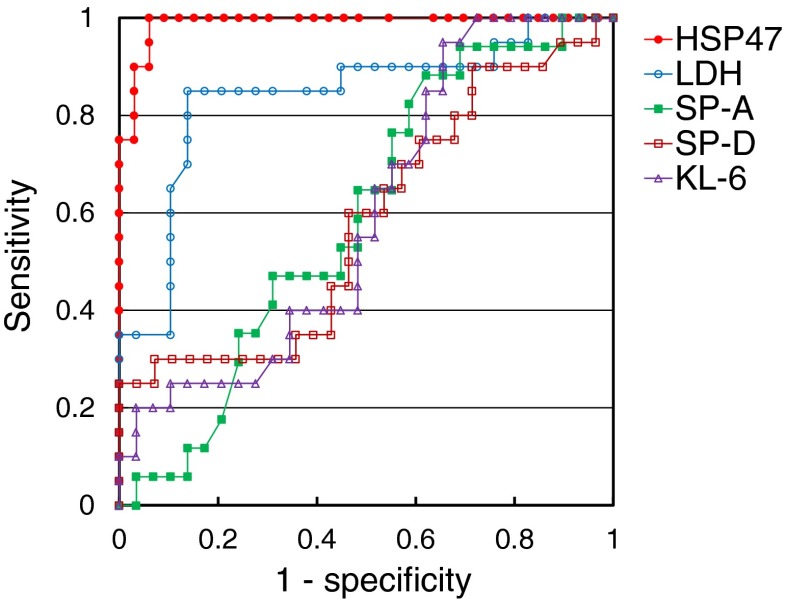
Based on an ROC curve (Fig. 3), the cutoff concentration of HSP47 that resulted in the highest diagnostic accuracy was 559.4 pg/mL. This value discriminated between AE-IPF and S-IPF with 100.0 % sensitivity and 93.9 % specificity. The diagnostic accuracy was 96.2 %. Use of serum HSP47 levels for diagnosis of AE-IPF resulted in the largest area under the curve—0.989—compared to areas of 0.597, 0.591, 0.583, and 0.841 for use of serum levels of KL-6, SP-A, SP-D, and LDH, respectively.
Fig. 3.
Receiver operating characteristic curve for serum levels of HSP47, KL-6, SP-A, SP-D and LDH in patients with S-IPF and AE-IPF. The cut-off level for HSP47 that resulted in the highest diagnostic accuracy was 559.4 pg/mL, which discriminated between AE-IPF and S-IPF with 100.0 % sensitivity and 93.9 % specificity; diagnostic accuracy was 96.2 %. Use of serum HSP47 level for diagnosis of AE-IPF resulted in the largest area under the curve (0.989) compared to use of serum levels of KL-6, SP-A, SP-D and LDH (0.597, 0.591, 0.583 and 0.841, respectively)
Histopathological and immunohistochemical findings
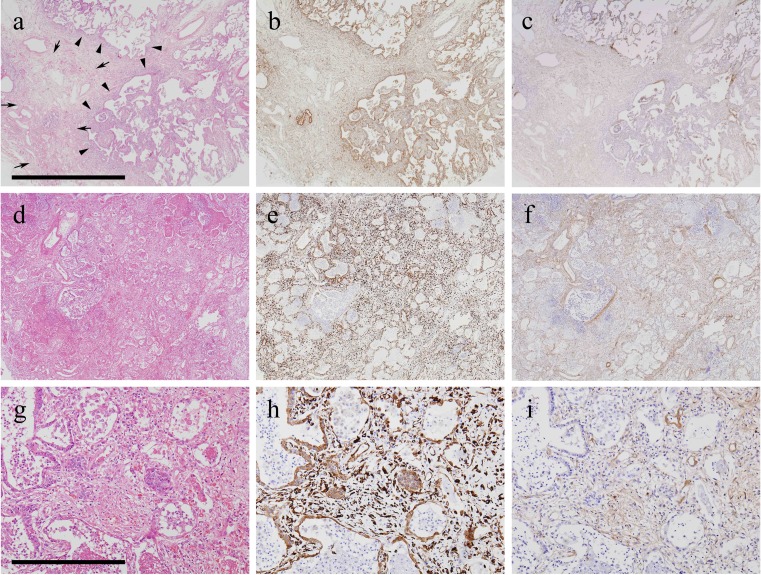
Immunohistochemical analyses of tissue specimens revealed that HSP47 staining intensity and distribution were greater in DAD than in UIP; HSP47 and type I procollagen expression was diffuse in the former but heterogeneous in the latter. Photomicrographs of results in representative UIP surgical lung biopsy specimens and DAD autopsy specimens are shown in Fig. 4: the groupings of a–c, d–f, and g–i depict sequential sections obtained from the same patient, who first underwent surgical lung biopsy (Fig. 4a–c, diagnosed as UIP), and later experienced acute exacerbation and death (Fig. 4d–i, autopsy specimens representing areas of DAD). At low magnification, the surgical biopsy specimens can be seen to have fibrotic zones with temporal heterogeneity as indicated by the presence of dense acellular fibrosis and active fibrotic areas with scattered fibroblastic foci; HSP47 and type I procollagen expression were seen in the active fibrotic areas, while expression was low in areas of dense fibrosis. The serum level of HSP47 was 487.4 pg/mL at the time of biopsy. This patient underwent AE-IPF 5 months after the biopsy was obtained; the serum HSP47 was found to be 4,190.7 pg/mL, and increased to 4,709.3 pg/mL 44 days later, 4 days before death occurred. Autopsy lung specimens revealed a combination of UIP and DAD patterns. Areas of DAD are depicted in Fig. 4g–i. At low magnification, diffuse involvement, including hyaline membranes, interstitial edema and inflammation, was seen. Expression of HSP47 and type I procollagen in DAD areas was diffuse and higher than in the previously biopsied lung specimens. Histopathological examination at high magnification revealed hyaline membranes, interstitial edema, fibrosis, and inflammation in DAD areas; expression of HSP47 was noted predominantly in fibroblasts, epithelial cells, and endothelial cells in DAD areas. Negative control studies using nonspecific immunoglobulin-G revealed no positive cells (data not shown).
Fig. 4.
Photomicrographs of histological and immunohistochemical studies of representative surgical lung biopsy specimens depicting UIP and subsequent autopsy specimens with areas of DAD. a–c, d–f, and g–i depict groups of sequential sections obtained from one patient initially diagnosed with UIP who underwent acute exacerbation of IPF 5 months later, and died 48 days after that. a, d, g Hematoxylin–eosin staining. b, e, h HSP47 expression. c, f, i Type I procollagen. a–c UIP surgical biopsy specimens. d–i Autopsy specimens representing areas of DAD patterns. Area framed by arrows depicts an area of dense fibrosis. Arrowheads indicate active fibrotic areas with scattered fibroblastic foci. Scale bars: a–f 2 mm, g–i 400 μm
Discussion
In the present study, HSP47 serum levels in patients with AE-IPF were found to be markedly higher than in patients with S-IPF. To the best of our knowledge, this is the first study to evaluate HSP47 serum levels of patients with AE-IPF.
Despite extensive research in the past, the precise molecular mechanisms leading to AE-IPF are poorly understood. Recent data have suggested that AE-IPF may be due to an acceleration of the underlying fibroproliferative disease process (Konishi et al. 2009; Collard et al. 2007). Finding higher pulmonary expression of HSP47, a collagen-specific molecular chaperone, in DAD than in UIP tissues in this study is consistent with that hypothesis. The current study demonstrated that serum HSP47 levels were markedly elevated in AE-IPF compared to S-IPF patients, indicating that the underlying pathogenic fibrotic molecular mechanisms regarding HSP47 may differ distinctly between these two conditions. The present study also demonstrated, using ROC curve analysis, that HSP47 was superior to KL-6, SP-A, SP-D, and LDH for discriminating between AE-IPF and S-IPF, suggesting that the expression of HSP47 may be an important pathobiological difference between the two conditions.
It was assumed before starting this study that serum levels of KL-6 and SP-D in AE-IPF would be higher than those in S-IPF patients because previous reports have suggested that KL-6 and SP-D might be useful markers for interstitial pneumonias (Ohnishi et al. 2002; Takahashi et al. 2000). However, Ishizaka et al. reported that at the onset of acute lung injury, plasma levels of KL-6 were similar to those of controls, perhaps because of its relatively large molecular size (Ishizaka et al. 2004). In addition, in the current study, serum levels of KL-6 and SP-D levels were elevated even in patients with S-IPF (when compared with normal ranges), consistent with previous reports (Ohnishi et al. 2002; Takahashi et al. 2000). These facts may explain why serum levels of KL-6 and SP-D did not differ significantly between patients with AE-IPF and those with S-IPF in the current study.
Since we previously demonstrated that HSP47 expression was higher in the lungs of patients with UIP than in those of controls, it was assumed that serum levels of HSP47 in S-IPF patients also would be higher than control levels. However, Yokota (one of the authors of the present manuscript) et al. previously reported that the serum levels of HSP47 did not differ significantly between patients with S-IPF and healthy controls (Yokota et al. 2003), and, in fact, that finding was reproduced in the present study (data not shown). Although a precise mechanism for the elevation of serum HSP47 levels only in patients with AE-IPF and not in patients with S-IPF was not elucidated in this study, the following is hypothesized: (1) the present study demonstrated that HSP47 expression was higher in DAD than in UIP lung tissue. The amount of HSP47 in the lung might correlate with serum levels, although the current study was not adequately powered to demonstrate this (2) elevation of serum HSP47 in AE-IPF might be due to distinctive characteristics of DAD, including severe inflammation, tissue destruction, apoptosis and/or cell necrosis, and severe fibrosis within the alveolar walls. These changes may possibly induce leakage of HSP47 protein into the extracellular matrix. (3) The pathobiology of DAD includes alveolar epithelial and endothelial injury with increased vascular permeability (Ware and Matthay 2000). Therefore, high expression of HSP47 in epithelial cells and endothelial cells and increased vascular permeability might lead to the elevation of serum HSP47 in AE-IPF. Abnormalities in type II alveolar epithelial cell function have been proposed as central to the pathogenesis of AE-IPF (Collard et al. 2010). Finding a high expression level of HSP47 in epithelial cells in DAD lungs is consistent with this hypothesis. The current results suggest that HSP47 could play an important role in the pathogenesis of AE-IPF. Further investigations regarding the precise mechanisms involved are needed.
Several studies have demonstrated that elevated plasma procollagen amino-terminal propeptide levels reflect collagen synthesis at the disease site and may be used as a marker of the reparative process, independent of etiology (Horslev-Petersen 1990; Kirk et al. 1984). Meduri et al. also reported that plasma levels of types I and III procollagen amino-terminal propeptide are elevated in early acute respiratory distress syndrome (ARDS) and that these levels could be a useful marker for ARDS (Meduri et al. 1998). Therefore, because AE-IPF shares clinical and histopathological features with ARDS, serum HSP47 might prove to be a useful marker for AE-IPF. In this study, whether serum levels of HSP47 correlated with clinical course, namely survival and clinical data, was also investigated; no statistically significant correlation was found (data not shown). However, the present study was not designed to fully evaluate such a relationship and was therefore underpowered; the focus was on pathogenesis. The interesting clinical question of whether serum HSP47 is a useful marker to reflect disease severity, predict mortality, and monitor disease progression and response to treatment remains to be answered by future investigations.
There is no proven treatment for AE-IPF. Mechanical ventilation is often required but is not usually successful; in one previous study, the median survival was 2.2 months from onset of acute exacerbation, and a half died during hospitalisation (Song et al. 2010). Among surviving patients, recurrence of acute exacerbation is common and usually results in death (Kim et al. 2006). Previous studies in several experimental animal fibrosis models have shown that downregulation of HSP47 expression by antisense oligodeoxynucleotides or small interfering ribonucleic acid (siRNA) reduces collagen production and subsequently diminishes progression of fibrosis (Sunamoto et al. 1998; Nishino et al. 2003; Hagiwara et al. 2007a, b, c; Obata et al. 2012). In addition, in mice with bleomycin-induced pulmonary fibrosis, treatment with pirfenidone, an anti-fibrotic agent, inhibited fibrosis through inhibition of HSP47-positive cells (Kakugawa et al. 2004). There are other reports of studies in experimental animal models indicating that suppression of HSP47 expression with antisense HSP47 oligodeoxynucleotides or HSP47-siRNA not only reduces the accumulation of collagen to delay progression of fibrosis but also abrogates α-smooth muscle actin expression and reduces macrophage infiltration in blood vessels and monocyte chemotactic protein-1 expression (Nishino et al. 2003; Wang et al. 2002; Obata et al. 2012). Considered together, these studies suggest that affecting downregulation of HSP47 could become a novel strategy for treating AE-IPF.
Recently, direct hemoperfusion with a polymyxin B-immobilized fiber cartridge (PMX-DHP) has been reported to have a beneficial impact on the respiratory status and long-term outcome of patients with AE-IPF (Seo et al. 2006; Abe et al. 2010; Enomoto et al. 2008; Hara et al. 2011). Oishi et al. reported that proinflammatory, profibrotic, and proangiogenic cytokines, including TGF-β were eluted from PMX fibers used for the treatment of AE-IPF and suggested that adsorption of such cytokines onto PMX fibers might be one of the mechanisms of action involved. Because TGF-β has been reported to induce the expression of HSP47 (Hisatomi et al. 2012; Nakayama et al. 2008), adsorption of cytokines by PMX-DHP could decrease expression of HSP47 and thereby attenuate the progression of inflammation and fibrosis in the DAD lung. The precise effects of PMX-DHP on HSP47 expression should be investigated in a future study.
The current study had several limitations. (1) The number of patients enrolled was small. However, despite the small study population, a clear-cut difference between AE-IPF and S-IPF patients was demonstrated. To confirm these results, a prospective multicenter study of a larger patient cohort is planned. (2) Serum levels of HSP47 in patients with rapid deterioration of IPF with identifiable causes were not evaluated. Patients with IPF may suffer “rapid deterioration” secondary to infections, pulmonary embolism, pneumothorax, or heart failure or “acute exacerbation” without an identifiable cause (Song et al. 2010; Raghu et al. 2011). It is often difficult for clinicians to distinguish these two conditions; the interesting clinical question of whether serum HSP47 could be useful for this purpose remains to be answered.
Conclusions
In conclusion, this study demonstrated that serum HSP47 levels were elevated in patients with AE-IPF. This finding suggests that underlying fibrogenic mechanisms that affect HSP47 levels differ between AE-IPF and S-IPF patients. Further studies involving larger patient cohorts are warranted to determine whether serum HSP47 is a useful disease marker for AE-IPF.
Acknowledgments
The authors thank Dr. M. Kitaichi (Department of Laboratory Medicine and Pathology, NHO Kinki-chuo Chest Medical Center, Sakai, Japan) for the valuable advice regarding pathological diagnosis; Prof. T. Nagayasu (Division of Surgical Oncology, Department of Translational Medical Sciences, Nagasaki University Graduate School of Biomedical Sciences, Nagasaki, Japan) for performing the surgical lung biopsies and providing the tissues that made this work possible; and Mr. A. Yokoyama for the excellent technical support.
This study was supported in part by a research grant from Takeda Science Foundation and The Kato Memorial Trust for Nambyo Research. This study was also partly supported by a grant to the Diffuse Lung Diseases Research Group from the Ministry of Health, Labour and Welfare, in Japan.
Competing interests
T. Kakugawa received a research grant from Takeda Science Foundation and the Kato Memorial Trust for Nambyo Research. T. Kakugawa and S. Yokota have a patent application pending for research related to this manuscript. Y. Ishimatsu and N. Sakamoto received a research grant from the Ministry of Education, Science, Sports, and Culture of Japan.
Contributor Information
Tomoyuki Kakugawa, Phone: +81-95-8197273, FAX: +81-95-8497285, Email: kakugawa@nagasaki-u.ac.jp.
Shin-ichi Yokota, Email: syokota@sapmed.ac.jp.
Yuji Ishimatsu, Email: yuji-i@nagasaki-u.ac.jp.
Tomayoshi Hayashi, Email: toma@nagasaki-u.ac.jp.
Shota Nakashima, Email: shonaka@nagasaki-u.ac.jp.
Shintaro Hara, Email: hara_shin123@me.com.
Noriho Sakamoto, Email: nsakamot@nagasaki-u.ac.jp.
Hiroshi Kubota, Email: hkubota@ipc.akita-u.ac.jp.
Mariko Mine, Email: mmine@nagasaki-u.ac.jp.
Yasuhiro Matsuoka, Email: koto-koto@ny.thn.ne.jp.
Hiroshi Mukae, Email: hmukae@med.uoeh-u.ac.jp.
Kazuhiro Nagata, Email: nagata@cc.kyoto-su.ac.jp.
Shigeru Kohno, Email: s-kohno@nagasaki-u.ac.jp.
References
- Abe K, Ozono Y, Miyazaki M, Koji T, Shioshita K, Furusu A, Tsukasaki S, Matsuya F, Hosokawa N, Harada T, Taguchi T, Nagata K, Kohno S. Interstitial expression of heat shock protein 47 and alpha-smooth muscle actin in renal allograft failure. Nephrol Dial Transplant. 2000;15(4):529–535. doi: 10.1093/ndt/15.4.529. [DOI] [PubMed] [Google Scholar]
- Abe S, Seo Y, Hayashi H, Matsuda K, Usuki J, Azuma A, Kudoh S, Gemma A. Neutrophil adsorption by polymyxin B-immobilized fiber column for acute exacerbation in patients with interstitial pneumonia: a pilot study. Blood Purif. 2010;29(4):321–326. doi: 10.1159/000287232. [DOI] [PubMed] [Google Scholar]
- Amenomori M, Mukae H, Sakamoto N, Kakugawa T, Hayashi T, Hara A, Hara S, Fujita H, Ishimoto H, Ishimatsu Y, Nagayasu T, Kohno S. HSP47 in lung fibroblasts is a predictor of survival in fibrotic nonspecific interstitial pneumonia. Respir Med. 2010;104(6):895–901. doi: 10.1016/j.rmed.2010.01.011. [DOI] [PubMed] [Google Scholar]
- Collard HR, Moore BB, Flaherty KR, Brown KK, Kaner RJ, King TE, Jr, Lasky JA, Loyd JE, Noth I, Olman MA, Raghu G, Roman J, Ryu JH, Zisman DA, Hunninghake GW, Colby TV, Egan JJ, Hansell DM, Johkoh T, Kaminski N, Kim DS, Kondoh Y, Lynch DA, Muller-Quernheim J, Myers JL, Nicholson AG, Selman M, Toews GB, Wells AU, Martinez FJ. Acute exacerbations of idiopathic pulmonary fibrosis. Am J Respir Crit Care Med. 2007;176(7):636–643. doi: 10.1164/rccm.200703-463PP. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collard HR, Calfee CS, Wolters PJ, Song JW, Hong SB, Brady S, Ishizaka A, Jones KD, King TE, Jr, Matthay MA, Kim DS. Plasma biomarker profiles in acute exacerbation of idiopathic pulmonary fibrosis. Am J Physiol Lung Cell Mol Physiol. 2010;299(1):L3–L7. doi: 10.1152/ajplung.90637.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Enomoto N, Suda T, Uto T, Kato M, Kaida Y, Ozawa Y, Miyazaki H, Kuroishi S, Hashimoto D, Naito T, Fujisawa T, Matsui T, Inui N, Nakamura Y, Sato J, Mizuguchi T, Kato A, Chida K. Possible therapeutic effect of direct haemoperfusion with a polymyxin B immobilized fibre column (PMX-DHP) on pulmonary oxygenation in acute exacerbations of interstitial pneumonia. Respirology. 2008;13(3):452–460. doi: 10.1111/j.1440-1843.2008.01290.x. [DOI] [PubMed] [Google Scholar]
- Hagiwara S, Iwasaka H, Matsumoto S, Noguchi T. Antisense oligonucleotide inhibition of heat shock protein (HSP) 47 improves bleomycin-induced pulmonary fibrosis in rats. Respir Res. 2007;8:37. doi: 10.1186/1465-9921-8-37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagiwara S, Iwasaka H, Matsumoto S, Noguchi T. An antisense oligonucleotide to HSP47 inhibits paraquat-induced pulmonary fibrosis in rats. Toxicology. 2007;236(3):199–207. doi: 10.1016/j.tox.2007.04.013. [DOI] [PubMed] [Google Scholar]
- Hagiwara S, Iwasaka H, Matsumoto S, Noguchi T. Introduction of antisense oligonucleotides to heat shock protein 47 prevents pulmonary fibrosis in lipopolysaccharide-induced pneumopathy of the rat. Eur J Pharmacol. 2007;564(1–3):174–180. doi: 10.1016/j.ejphar.2007.02.057. [DOI] [PubMed] [Google Scholar]
- Hara S, Ishimoto H, Sakamoto N, Mukae H, Kakugawa T, Ishimatsu Y, Mine M, Kohno S. Direct hemoperfusion using immobilized polymyxin B in patients with rapidly progressive interstitial pneumonias: a retrospective study. Respiration. 2011;81(2):107–117. doi: 10.1159/000321958. [DOI] [PubMed] [Google Scholar]
- Hisatomi K, Mukae H, Sakamoto N, Ishimatsu Y, Kakugawa T, Hara S, Fujita H, Nakamichi S, Oku H, Urata Y, Kubota H, Nagata K, Kohno S. Pirfenidone inhibits TGF-beta1-induced over-expression of collagen type I and heat shock protein 47 in A549 cells. BMC Pulm Med. 2012;12:24. doi: 10.1186/1471-2466-12-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horslev-Petersen K. Circulating extracellular matrix components as markers for connective tissue response to inflammation. A clinical and experimental study with special emphasis on serum aminoterminal type III procollagen peptide in rheumatic diseases. Dan Med Bull. 1990;37(4):308–329. [PubMed] [Google Scholar]
- Hyzy R, Huang S, Myers J, Flaherty K, Martinez F. Acute exacerbation of idiopathic pulmonary fibrosis. Chest. 2007;132(5):1652–1658. doi: 10.1378/chest.07-0299. [DOI] [PubMed] [Google Scholar]
- Ishida Y, Kubota H, Yamamoto A, Kitamura A, Bachinger HP, Nagata K. Type I collagen in Hsp47-null cells is aggregated in endoplasmic reticulum and deficient in N-propeptide processing and fibrillogenesis. Mol Biol Cell. 2006;17(5):2346–2355. doi: 10.1091/mbc.E05-11-1065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishii H, Mukae H, Kakugawa T, Iwashita T, Kaida H, Fujii T, Hayashi T, Kadota J, Kohno S. Increased expression of collagen-binding heat shock protein 47 in murine bleomycin-induced pneumopathy. Am J Physiol Lung Cell Mol Physiol. 2003;285(4):L957–L963. doi: 10.1152/ajplung.00305.2002. [DOI] [PubMed] [Google Scholar]
- Ishizaka A, Matsuda T, Albertine KH, Koh H, Tasaka S, Hasegawa N, Kohno N, Kotani T, Morisaki H, Takeda J, Nakamura M, Fang X, Martin TR, Matthay MA, Hashimoto S. Elevation of KL-6, a lung epithelial cell marker, in plasma and epithelial lining fluid in acute respiratory distress syndrome. Am J Physiol Lung Cell Mol Physiol. 2004;286(6):L1088–L1094. doi: 10.1152/ajplung.00420.2002. [DOI] [PubMed] [Google Scholar]
- Iwashita T, Kadota J, Naito S, Kaida H, Ishimatsu Y, Miyazaki M, Ozono Y, Kohno S. Involvement of collagen-binding heat shock protein 47 and procollagen type I synthesis in idiopathic pulmonary fibrosis: contribution of type II pneumocytes to fibrosis. Hum Pathol. 2000;31(12):1498–1505. doi: 10.1053/hupa.2000.20378. [DOI] [PubMed] [Google Scholar]
- Kakugawa T, Mukae H, Hayashi T, Ishii H, Abe K, Fujii T, Oku H, Miyazaki M, Kadota J, Kohno S. Pirfenidone attenuates expression of HSP47 in murine bleomycin-induced pulmonary fibrosis. Eur Respir J. 2004;24(1):57–65. doi: 10.1183/09031936.04.00120803. [DOI] [PubMed] [Google Scholar]
- Kakugawa T, Mukae H, Hayashi T, Ishii H, Nakayama S, Sakamoto N, Yoshioka S, Sugiyama K, Mine M, Mizuta Y, Kohno S. Expression of HSP47 in usual interstitial pneumonia and nonspecific interstitial pneumonia. Respir Res. 2005;6:57. doi: 10.1186/1465-9921-6-57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kakugawa T, Mukae H, Hishikawa Y, Ishii H, Sakamoto N, Ishimatsu Y, Fujii T, Koji T, Kohno S. Localization of HSP47 mRNA in murine bleomycin-induced pulmonary fibrosis. Virchows Arch. 2010;456(3):309–315. doi: 10.1007/s00428-009-0876-x. [DOI] [PubMed] [Google Scholar]
- Kim DS, Park JH, Park BK, Lee JS, Nicholson AG, Colby T. Acute exacerbation of idiopathic pulmonary fibrosis: frequency and clinical features. Eur Respir J. 2006;27(1):143–150. doi: 10.1183/09031936.06.00114004. [DOI] [PubMed] [Google Scholar]
- Kirk JM, Bateman ED, Haslam PL, Laurent GJ, Turner-Warwick M. Serum type III procollagen peptide concentration in cryptogenic fibrosing alveolitis and its clinical relevance. Thorax. 1984;39(10):726–732. doi: 10.1136/thx.39.10.726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konishi K, Gibson KF, Lindell KO, Richards TJ, Zhang Y, Dhir R, Bisceglia M, Gilbert S, Yousem SA, Song JW, Kim DS, Kaminski N. Gene expression profiles of acute exacerbations of idiopathic pulmonary fibrosis. Am J Respir Crit Care Med. 2009;180(2):167–175. doi: 10.1164/rccm.200810-1596OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meduri GU, Tolley EA, Chinn A, Stentz F, Postlethwaite A. Procollagen types I and III aminoterminal propeptide levels during acute respiratory distress syndrome and in response to methylprednisolone treatment. Am J Respir Crit Care Med. 1998;158(5 Pt 1):1432–1441. doi: 10.1164/ajrccm.158.5.9801107. [DOI] [PubMed] [Google Scholar]
- Nagai N, Hosokawa M, Itohara S, Adachi E, Matsushita T, Hosokawa N, Nagata K. Embryonic lethality of molecular chaperone hsp47 knockout mice is associated with defects in collagen biosynthesis. J Cell Biol. 2000;150(6):1499–1506. doi: 10.1083/jcb.150.6.1499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagata K. Hsp47: a collagen-specific molecular chaperone. Trends Biochem Sci. 1996;21(1):22–26. doi: 10.1016/0968-0004(96)80881-4. [DOI] [PubMed] [Google Scholar]
- Nagata K, Saga S, Yamada KM. A major collagen-binding protein of chick embryo fibroblasts is a novel heat shock protein. J Cell Biol. 1986;103(1):223–229. doi: 10.1083/jcb.103.1.223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagata K, Hirayoshi K, Obara M, Saga S, Yamada KM. Biosynthesis of a novel transformation-sensitive heat-shock protein that binds to collagen. Regulation by mRNA levels and in vitro synthesis of a functional precursor. J Biol Chem. 1988;263(17):8344–8349. [PubMed] [Google Scholar]
- Nakayama S, Mukae H, Sakamoto N, Kakugawa T, Yoshioka S, Soda H, Oku H, Urata Y, Kondo T, Kubota H, Nagata K, Kohno S. Pirfenidone inhibits the expression of HSP47 in TGF-beta1-stimulated human lung fibroblasts. Life Sci. 2008;82(3–4):210–217. doi: 10.1016/j.lfs.2007.11.003. [DOI] [PubMed] [Google Scholar]
- Nishino T, Miyazaki M, Abe K, Furusu A, Mishima Y, Harada T, Ozono Y, Koji T, Kohno S. Antisense oligonucleotides against collagen-binding stress protein HSP47 suppress peritoneal fibrosis in rats. Kidney Int. 2003;64(3):887–896. doi: 10.1046/j.1523-1755.2003.00169.x. [DOI] [PubMed] [Google Scholar]
- Obata Y, Nishino T, Kushibiki T, Tomoshige R, Xia Z, Miyazaki M, Abe K, Koji T, Tabata Y, Kohno S. HSP47 siRNA conjugated with cationized gelatin microspheres suppresses peritoneal fibrosis in mice. Acta Biomater. 2012;8(7):2688–2696. doi: 10.1016/j.actbio.2012.03.050. [DOI] [PubMed] [Google Scholar]
- Ohnishi H, Yokoyama A, Kondo K, Hamada H, Abe M, Nishimura K, Hiwada K, Kohno N. Comparative study of KL-6, surfactant protein-A, surfactant protein-D, and monocyte chemoattractant protein-1 as serum markers for interstitial lung diseases. Am J Respir Crit Care Med. 2002;165(3):378–381. doi: 10.1164/ajrccm.165.3.2107134. [DOI] [PubMed] [Google Scholar]
- Raghu G, Collard HR, Egan JJ, Martinez FJ, Behr J, Brown KK, Colby TV, Cordier JF, Flaherty KR, Lasky JA, Lynch DA, Ryu JH, Swigris JJ, Wells AU, Ancochea J, Bouros D, Carvalho C, Costabel U, Ebina M, Hansell DM, Johkoh T, Kim DS, King TE, Jr, Kondoh Y, Myers J, Muller NL, Nicholson AG, Richeldi L, Selman M, Dudden RF, Griss BS, Protzko SL, Schunemann HJ. An official ATS/ERS/JRS/ALAT statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. Am J Respir Crit Care Med. 2011;183(6):788–824. doi: 10.1164/rccm.2009-040GL. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sauk JJ, Smith T, Norris K, Ferreira L. Hsp47 and the translation-translocation machinery cooperate in the production of alpha 1(I) chains of type I procollagen. J Biol Chem. 1994;269(6):3941–3946. [PubMed] [Google Scholar]
- Seo Y, Abe S, Kurahara M, Okada D, Saito Y, Usuki J, Azuma A, Koizumi K, Kudoh S. Beneficial effect of polymyxin B-immobilized fiber column (PMX) hemoperfusion treatment on acute exacerbation of idiopathic pulmonary fibrosis. Intern Med. 2006;45(18):1033–1038. doi: 10.2169/internalmedicine.45.6018. [DOI] [PubMed] [Google Scholar]
- Shioshita K, Miyazaki M, Ozono Y, Abe K, Taura K, Harada T, Koji T, Taguchi T, Kohno S. Expression of heat shock proteins 47 and 70 in the peritoneum of patients on continuous ambulatory peritoneal dialysis. Kidney Int. 2000;57(2):619–631. doi: 10.1046/j.1523-1755.2000.00883.x. [DOI] [PubMed] [Google Scholar]
- Song JW, Hong SB, Lim CM, Koh Y, Kim DS. Acute exacerbation of idiopathic pulmonary fibrosis: incidence, risk factors and outcome. Eur Respir J. 2010;37(2):356–363. doi: 10.1183/09031936.00159709. [DOI] [PubMed] [Google Scholar]
- Sunamoto M, Kuze K, Tsuji H, Ohishi N, Yagi K, Nagata K, Kita T, Doi T. Antisense oligonucleotides against collagen-binding stress protein HSP47 suppress collagen accumulation in experimental glomerulonephritis. Lab Invest. 1998;78(8):967–972. [PubMed] [Google Scholar]
- Takahashi H, Fujishima T, Koba H, Murakami S, Kurokawa K, Shibuya Y, Shiratori M, Kuroki Y, Abe S. Serum surfactant proteins A and D as prognostic factors in idiopathic pulmonary fibrosis and their relationship to disease extent. Am J Respir Crit Care Med. 2000;162(3 Pt 1):1109–1114. doi: 10.1164/ajrccm.162.3.9910080. [DOI] [PubMed] [Google Scholar]
- Wang Y, Li C, Wang X, Zhang J, Chang Z. Heat shock response inhibits IL-18 expression through the JNK pathway in murine peritoneal macrophages. Biochem Biophys Res Commun. 2002;296(3):742–748. doi: 10.1016/S0006-291X(02)00930-0. [DOI] [PubMed] [Google Scholar]
- Ware LB, Matthay MA. The acute respiratory distress syndrome. N Engl J Med. 2000;342(18):1334–1349. doi: 10.1056/NEJM200005043421806. [DOI] [PubMed] [Google Scholar]
- Yokota S, Hirata D, Minota S, Higashiyama T, Kurimoto M, Yanagi H, Yura T, Kubota H. Autoantibodies against chaperonin CCT in human sera with rheumatic autoimmune diseases: comparison with antibodies against other Hsp60 family proteins. Cell Stress Chaperones. 2000;5(4):337–346. doi: 10.1379/1466-1268(2000)005<0337:AACCIH>2.0.CO;2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yokota S, Kubota H, Matsuoka Y, Naitoh M, Hirata D, Minota S, Takahashi H, Fujii N, Nagata K. Prevalence of HSP47 antigen and autoantibodies to HSP47 in the sera of patients with mixed connective tissue disease. Biochem Biophys Res Commun. 2003;303(2):413–418. doi: 10.1016/S0006-291X(03)00352-8. [DOI] [PubMed] [Google Scholar]