Abstract
The cellulose binding elicitor lectin (CBEL) of the genus Phytophthora induces necrosis and immune responses in several plant species, including Arabidopsis thaliana. However, the role of CBEL-induced responses in the outcome of the interaction is still unclear. This study shows that some of CBEL-induced defence responses, but not necrosis, required the receptor-like kinase BAK1, a general regulator of basal immunity in Arabidopsis, and the production of a reactive oxygen burst mediated by respiratory burst oxidases homologues (RBOH). Screening of a core collection of 48 Arabidopsis ecotypes using CBEL uncovered a large variability in CBEL-induced necrotic responses. Analysis of non-responsive CBEL lines Ws-4, Oy-0, and Bla-1 revealed that Ws-4 and Oy-0 were also impaired in the production of the oxidative burst and expression of defence genes, whereas Bla-1 was partially affected in these responses. Infection tests using two Phytophthora parasitica strains, Pp310 and Ppn0, virulent and avirulent, respectively, on the Col-0 line showed that BAK1 and RBOH mutants were susceptible to Ppn0, suggesting that some immune responses controlled by these genes, but not CBEL-induced cell death, are required for Phytophthora parasitica resistance. However, Ws-4, Oy-0, and Bla-1 lines were not affected in Ppn0 resistance, showing that natural variability in CBEL responsiveness is not correlated to Phytophthora susceptibility. Overall, the results uncover a BAK1- and RBOH-dependent CBEL-triggered immunity essential for Phytophthora resistance and suggest that natural quantitative variation of basal immunity triggered by conserved general elicitors such as CBEL does not correlate to Phytophthora susceptibility.
Key words: Arabidopsis, cell death, elicitor, immunity, natural variability, necrosis, oomycete, Phytophthora.
Introduction
Plant immunity is based on multilayered defence mechanisms (Jones and Dangl, 2006). The first layer and probably the most ancestral mechanism of plant immunity, relies on the recognition of molecular patterns that are found in microbial molecules (pathogen-associated molecular patterns, PAMPs) or plant endogenous molecular patterns exposed by self-products released during the interaction (damage-associated molecular patterns, DAMPs; Boller and Felix, 2009). This recognition is mediated by specific pattern recognition receptors that trigger the so-called PAMP-triggered immunity. Both PAMPs and DAMPs are found in molecules previously designed as ‘general elicitors’. PAMP-triggered immunity is generally characterized by the induction of various defence responses, including induction of a reactive oxygen species (ROS) burst, callose deposition, and expression of defence genes without any signs of programmed cell death (Boller and Felix, 2009). Adapted pathogens have evolved mechanisms to avoid the PAMP-triggered immunity by secreting apoplastic and translocated effectors (Bozkurt et al., 2012) leading to the so-called effector-triggered susceptibility. A second layer of immunity, named effector-triggered immunity, is based on the direct or indirect recognition of microbial effectors by specific plant receptors encoded by resistance genes. Effector-triggered immunity, but not PAMP-triggered immunity, has been generally associated with the development of the hypersensitive response, corresponding to a localized programmed cell death, and with systemic acquired resistance (Jones and Dangl, 2006). However, several general necrosis-inducing elicitors have been characterized in plant pathogens and particularly in pathogens belonging to the oomycete genus Phytophthora (Hein et al., 2009). This class of elicitors includes elicitins (Ponchet et al., 1999), the cellulose binding elicitor lectin (CBEL glycoprotein; Mateos et al., 1997), Nep1-like proteins (Fellbrich et al., 2002), and small cysteine-rich proteins such as Phytophthora infestans Scr74 and Phytophthora cactorum PcF (Orsomando et al., 2001; Liu et al., 2005). While cell death induced by Nep1-like proteins could be due to the phytotoxic nature of the protein (Qutob et al., 2006; Ottmann et al., 2009), little is known about the role of the hypersensitive-like response induced by necrosis-inducing elicitors. It has been suggested that responses to these proteins can be very similar to effector-triggered immunity and play a role in the outcome of Phytophthora-plant interactions (Thomma et al., 2011). Accordingly, recognition of elicitins, such as INF1 from Phytophthora infestans, and the inability of the parasite to suppress this recognition could limit the host range of the pathogen (Kamoun et al., 1998). Indeed, the Nicotiana benthamiana receptor-like kinase gene BAK1/SERK3 contributes to INF1-mediated necrosis, and BAK1/SERK3-silenced mutants displayed enhanced susceptibility to Phytophthora infestans (Chaparro-Garcia et al., 2011). Furthermore, INF1- and CBEL-induced cell death could be suppressed by the host-translocated Phytophthora infestans Avr3a effector through interaction and stabilization of the E3-ubiquitin ligase CMPG1 (Bos et al., 2006; Gilroy et al., 2011). These results demonstrate that oomycetes have evolved mechanisms to counteract the host-cell death triggered by necrosis-inducing elicitors.
The Phytophthora parasitica CBEL glycoprotein exhibits a modular structure with two repeated domain combinations separated by a threonine-proline-rich region. Each combination is constituted by a carbohydrate-binding module family 1 domain, which mediates cellulose binding, linked to a PAN/APPLE domain, involved in protein–polysaccharide or protein–protein interactions (Tordai et al., 1999; Boraston et al., 2004; Larroque et al., 2013). When infiltrated into tobacco leaf tissues, CBEL induces strong defence responses (Mateos et al., 1997). CBEL is also active in Arabidopsis ecotype Columbia (Col-0), inducing accumulation of hydroxyproline-rich glycoprotein accumulation, ethylene synthesis, defence-gene expression and an hypersensitive-like response (Khatib et al., 2004). Such responses require the binding of CBEL to the cell wall which is mediated by carbohydrate-binding module family 1 domains (Gaulin et al., 2006). Point mutations of carbohydrate-binding module family 1 domains on key aromatic residues involved in cellulose binding lead to a decrease of cell death inducing activity and defence gene expression in tobacco (Gaulin et al., 2006).
The main objective of this study was to investigate the role of CBEL-triggered immunity in the Phytophthora parasitica–Arabidopsis interaction by identifying mutants and natural lines affected in CBEL responses. Overall, the results show that CBEL-triggered immunity required BAK1 and RBOH (respiratory burst oxidase homologue), which also control resistance to the non-adapted Phytophthora parasitica Ppn0 strain. However, natural variability in CBEL responses is not correlated to the outcome of the interaction, suggesting that other mechanisms are involved in Phytophthora resistance.
Materials and methods
Plant material and Phytophthora strains
Arabidopsis thaliana lines were obtained from the Natural Variation of Arabidopsis thaliana collection (VNAT; INRA Versailles; McKhann et al., 2004). Seeds were sown on pots (Jiffy, Lyon, France), incubated for 2 days at 4 °C in darkness, and transferred for 4 weeks to a light growth chamber with a 8/16 light/dark cycle (270 μmol m–2 s–1), 22/20 °C light/dark, and 70% relative humidity. Phytophthora parasitica Dastur isolate 310 (Attard et al., 2010) and Phytophthora parasitica Dastsur var. nicotianae race 0 (Mateos et al., 1997) were grown at 25 °C and maintained at 15 °C in the dark on V-8 agar medium (5 % V-8 juice and 20g l–1 agar, pH 5.0).
Infection tests
Infection tests were performed using a protocol derived from (Elfstrand et al., 2002; Attard et al., 2010). Briefly, Arabidopsis seeds were sterilized and deposited in 6-well multiplates on a 17g m–2 sterile polypropylene mesh (cut from a garden protection film; Nortène SA, Lille, France) placed on top of 4ml of Murashige and Skoog medium (MS medium; Sigma) containing 30g l–1sucrose. Plates were incubated at 25 °C with 16h daily illumination at 30 μmol photons m–2 s–1. After 2 weeks, the mesh with the seedlings was transferred onto a new plate containing 4ml of Phytophthora zoospores (1000 cells ml–1), freshly prepared in distilled water. Plates were incubated in the same conditions as above and symptoms were scored after 10 days. The surface of green area in each well was quantified upon image acquisition using ImageJ software (http://rsbweb.nih.gov/ij/).
Elicitor preparations
CBEL was purified from the mycelium of Phytophthora parasitica as described previously (Séjalon-Delmas et al., 1997). Protein concentration was determined using a protein reagent (Bio-Rad, Ivry sur Seine, France), according to the supplier’s instructions and using BSA as standard protein. The elicitor P2 was purified from the mycelium as described previously (Roux et al., 1994) and dissolved in water at 50 μg ml–1.
Leaf infiltration assays
Four-week-old Arabidopsis plants were transferred to a growth chamber with a 16/8 22/20 °C light/dark cycle 24h before infiltration. CBEL (5 μg ml–1) and P2 (50 μg ml–1) or water were infiltrated into the mesophyll of fully expanded leaves with a needleless syringe. One leaf per plant and per elicitor was infiltrated, three replicate plants were tested for each Arabidopsis lines and four independent biological replicates were performed. Cell-death development was monitored at 5, 7, 10, and 12 days after infiltration and scored according to a symptom-index (Supplementary Fig. S1, available at JXB online).
Measurement of ROS production
The production of ROS was measured using an assay adapted from Gomez-Gomez et al. (1999). Briefly, 4-mm-diameter leaf discs of 4-week-old plants were equilibrated in water overnight then transferred to assay tubes (five discs per assay) containing 100 μl reaction mix with 20 μM luminol and 1 unit of horseradish peroxidase (Sigma-Aldrich, Saint-Quentin Fallavier, France). Luminescence was measured using a digital luminometer (BioOrbit 1250, Berthold) for 40 minutes after addition of the native CBEL (5 μg ml–1).
Quantification of gene expression
Total RNA was extracted using SV Total RNA Isolation System kit (Promega, Charbonnières, France). For each sample, 1 μg of extracted RNA was reverse transcribed using the High-Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Courtaboeuf, France). High-throughput quantitative PCR was performed using the BioMarkTM HD System (Fluidigm, Issy les Moulineaux, France). Briefly, cDNA was diluted to ~50ng μl–1 prior to specific target amplification by 14 cycles of 95 °C for 15sec and 60 °C for 4min in a reaction mix containing the 96 primer pairs (50nM) and TaqMan PreAmp Master Mix (1:2, Applied Biosystems). The primer sequences are presented in Supplementary Table S1. Pre-amplified cDNA was diluted with TE buffer (1:5) and used for quantitative PCR array analysis in a reaction mix containing TaqMan Gene Expression Master Mix (Applied Biosystems), DNA Binding Dye Sample Loading Reagent (Fluidigm, Issy les Moulineaux, France), and EvaGreen (Interchim, Montluçon, France). Data were analysed by the use of BioMark Real-Time PCR Analysis software version 2.0 (Fluidigm). The relative expression was calculated using AT2G28390 as standard. Each data point is based on three independent biological replicates.
Detection of phosphorylated mitogen-activated protein kinases
Infiltrated plant material was harvested and used for total protein extraction in 25mM Tris-HCl pH 7.8, 75mM NaCl, 15mM EGTA, 15mM glycerophosphate, 15mM 4-nitrophenylpyrophosphate, 10mM MgCl2, 1mM DTT, 1mM NaF, 0.5mM Na3VO4, 0.5mM PMSF, 10mg ml-1 leupeptin, 10mg ml-1 aprotinin, and 0.1% Tween 20. Proteins were collected by centrifugation (20 000 g, 10min, 4 °C) and subjected to SDS-PAGE (10 μg protein/lane). After migration, the proteins were blotted to a nitrocellulose membrane by using a Biorad semi-dry apparatus at a constant current (1.8 mA cm–2). The membrane was soaked for 30min in a TRIS-buffered saline solution (TRIS-HCl pH 7.5, 150mM NaCl) containing 3% non-fat milk before being incubated overnight at 4 °C in the same buffer containing 1% non-fat milk and a 1:1000 dilution of phospho-p44/42 mitogen-activated protein (MAP) kinase antibody (Cell Signaling Technology). The antigen– antibody complexes were visualized by colorimetric detection using alkaline phosphatase-conjugated goat anti-rabbit immunoglobulin G.
Results
BAK1 and NADPH oxidase genes are required for CBEL-induced oxidative burst and defence responses but not for necrosis
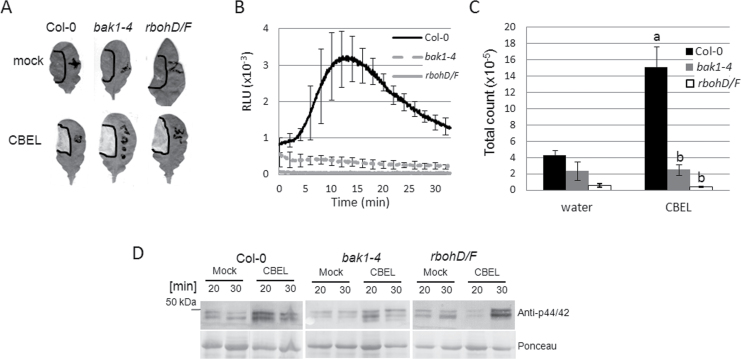
In Arabidopsis, the receptor-like kinase BAK1 is a positive regulator of immune responses triggered by bacterial PAMPs and DAMPs (Chinchilla et al., 2007; Heese et al., 2007; Shan et al., 2008; Krol et al., 2010) and controls plant cell-death in response to necrotrophic pathogens (Kemmerling et al., 2007). A common response to PAMPs is the generation of an oxidative burst mediated by the NADPH oxidases RBOH D and F. To investigate the role of BAK1 and NADPH oxidases in CBEL responses, leaves of bak1-4 (Kemmerling et al., 2007) and the double mutant rbohD/F (Torres et al., 2002) were infiltrated with a solution containing CBEL at 5 μg ml–1. Both mutants induced a necrotic response 5 days after infiltration that was identical to the response observed in Col-0 plants (Fig. 1A).
Fig. 1.
Necrotic and early defence responses induced by cellulose binding elicitor lectin (CBEL) in BAK1 and NADPH oxidase mutants. (A) bak1-4 and rbohD/F leaves were infiltrated with a CBEL solution at a concentration of 5 μg ml–1; necrotic symptoms were observed 5 days later. (B) Kinetics of ROS production monitored over 40min on leaf discs of 4-week-old bak1-4, rbohD/F, and Col-0 plants; data are means of three biological replicates and bars represent standard deviation. (C) Total photon counts from the results shown on (B); bars with different letters indicate significantly different results (P < 0.05). (D) Detection of MAP kinase phosphorylation; total protein extracts from leaves infiltrated with CBEL (5 μg ml–1) were analysed by Western blot using a p44/42 antibody and the membranes were stained by Ponceau red to control the equal loading of protein amount.
CBEL-induced ROS burst was analysed in Col-0 and mutant plants using the luminol-based assay (Gomez-Gomez et al., 1999). The ROS response observed in Col-0 was characterized by a transient rise of luminescence starting 5min after elicitation with an emphasis at 15min, followed by a strong decrease to the basal level 25min after the treatment (Fig. 1B). In bak1-4 and rbohD/F plants, no production of ROS was observed after addition of a CBEL solution (Fig. 1B). Total photon counts after a prolonged exposure (40min) to CBEL confirmed the absence of ROS burst in the mutants (Fig. 1C).
The Arabidopsis MAP kinases are induced by various PAMPs including flg22 and elf18 (Zipfel et al., 2006). The impact of CBEL treatment on MAP kinase phosphorylation was then tested using anti-p42/44 MAP kinase antibodies on Col-0 and mutant lines. MAP kinase activation was clearly detectable 20min upon treatment with CBEL but reduced in bak1-4 and delayed in rbohD/F (Fig 1D).
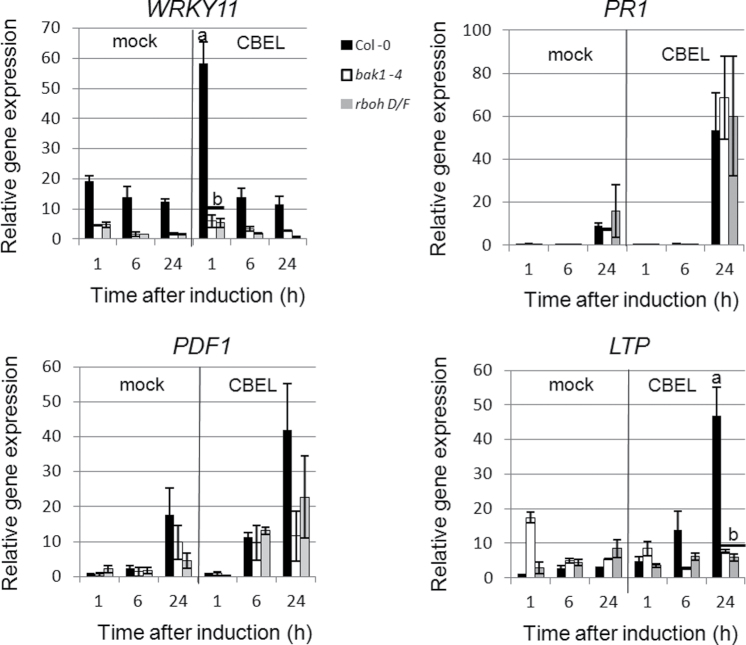
Expression of defence genes was tested in the rbohD/F and bak1-4 lines (Fig. 2). Tested genes included an early-responsive (1 hour post infection) gene encoding a WRKY transcription factor involved in the regulation of jasmonic acid–dependent responses (WRKY11; Journot-Catalino et al., 2006), two late-induced (24 and 48 hour post infection) defence genes previously shown to respond to CBEL treatment (PDF1.2 and PR1; Khatib et al., 2004), and a gene encoding a protease inhibitor/seed storage/lipid transfer protein (LTP) induced by a Nep1-like protein from Phytophthora parasitica (Qutob et al., 2006). Mutation in BAK1 and RBOHD/F led to a decrease in WRKY11, PDF1.2, and LTP expression but not PR1.
Fig. 2.
Cellulose binding elicitor lectin (CBEL)-induced defence gene expression in bak1-4 and rbohD/F lines. Expression of WRKY11 (AT4G31550), PR1 (AT2G14610), PDF1.2 (AT5G44420), and LTP (AT3G22600) was measured by quantitative reverse-transcription PCR in bak1-4 and rboh D/F leaves upon infiltration of a CBEL solution at 5 μg ml–1. Results were standardized using a stably expressed gene encoding a chloroplastic housekeeping protein (SAND family protein; AT2G28390). Data are means of three independent biological repetitions and bars represent the standard deviation. Bars with different letters indicate significantly different results (P < 0.05).
Overall, these results showed that some CBEL-induced defence responses but not necrosis CBEL, are regulated through a signalling pathway involving BAK1 and RBOHD/F proteins.
Identification of CBEL-insensitive Arabidopsis lines
Natural variability in CBEL-responses was explored by mining a collection of 48 Arabidopsis accessions that maximizes the genetic variability observed among 265 accessions collected worldwide (McKhann et al., 2004). An additional line, Ws-4, was included in the collection. One leaf per plant was infiltrated with a CBEL solution at 5 μg ml–1 and the development of the necrotic reaction was scored at 5, 7, and 10 days after infiltration. Among the 49 lines tested, only 37 lines were adapted to the screening assay since some lines showed a reduced leaf development which hampered infiltration or gave a necrotic symptom when infiltrated with water. A quantitative variability in CBEL-induced necrotic response was observed among the accessions ranging from no symptoms to necrosis of the infiltrated area (Supplementary Fig. S1). Necrosis appeared 5 days after infiltration in the case of well-responsive lines (Col-0, N6, Jea, Ct-1, Ta-0) whereas no symptoms were observed for four lines (Ran, Bl-1, Ws-4, Oy-0) 10 days after infiltration. Intermediate phenotypes ranged from yellowing (Alc-0, Sp-0, Jm-0, Yo-0, Blh-1, Cvi-0, Tsu-0, N-14, Pa-1) to partial necrosis of the infiltrated tissues (Mt-0, Ri-0, Bla-1). For further studies, two non-responsive CBEL lines (Oy-0 and Ws-4) as well as a line giving an intermediate phenotype (Bla-1) were selected. These lines are quite similar in terms of development with Col-0, facilitating comparative studies.
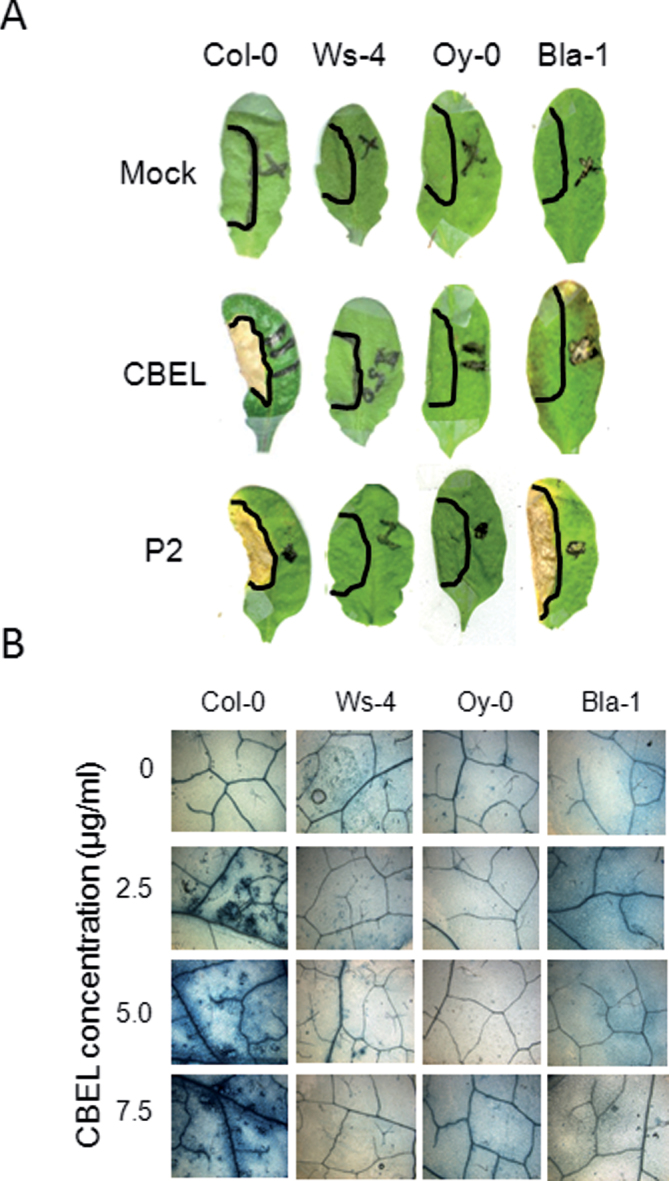
The selected lines did not show any sign of necrosis even when an upper concentration of CBEL was used (up to 7.5 μg ml–1; Fig. 3A) and after a prolonged observation (up to 10 days). Trypan blue staining did not reveal any microlesions in these lines, in contrast to the cell-death observed in Col-0 leaves at a minimal CBEL concentration of 2.5 μg ml–1 (Fig. 3B).
Fig. 3.
Identification of Arabidopsis lines insensitive to cellulose binding elicitor lectin (CBEL). (A) Arabidopsis leaves (Col-0, Ws-4, Oy-0, Bla-1) treated with mock, CBEL (5 μg ml–1), and P2 (50 μg ml–1) 10 days later after infiltration. (B) Trypan blue staining of the CBEL-infiltrated area 48h after treatment.
To determine whether non-responsive lines were also affected in the response to other necrotic Phytophthora parasitica elicitors, this study tested a crude extract of mycelium, named P2 at 50 μg ml–1 (Roux et al., 1994). Col-0 and Bla-1 plants developed necrotic symptoms 5 days after P2 infiltration, whereas Oy-0 and Ws-4 did not show any symptoms even at 10 days after treatment (Fig. 3). These results suggest that a common step involved in CBEL- and P2-induced cell death is affected in Oy-0 and Ws-4, while Bla-1 is specifically deficient in CBEL-induced cell death.
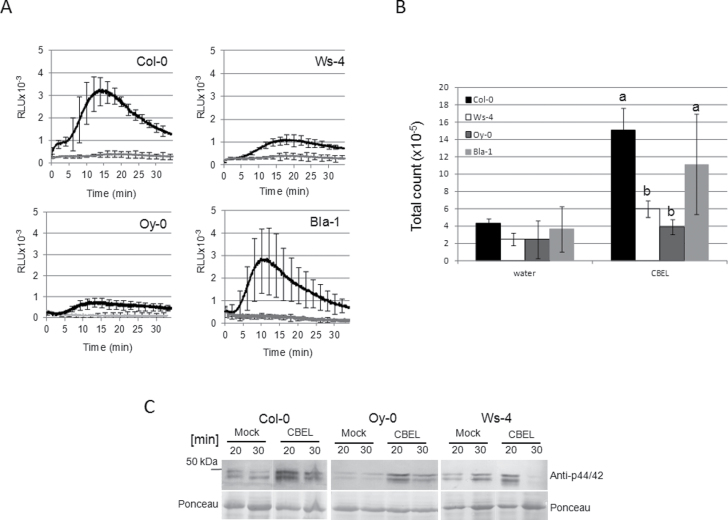
This study next investigated the correlation between the natural variation observed in the cell death response and other defence responses including ROS production, MAP kinase activation, and expression of defence genes. No significant difference in ROS production was observed in CBEL-treated Col-0 and Bla-1 (Fig. 4A and B). However, Ws-4 and Oy-0 plants exhibited a significantly reduced ROS production (Fig. 4A and B). MAP kinase activation assay was conducted in Col-0, Oy-0, and Ws-4 lines. A fainter but detectable activation of MAP kinases 20min after CBEL treatments was detected in Oy-0 and Ws-4 as compared to Col-0 (Fig. 4C). Interestingly, the signal was not recovered in Ws-4 30min after treatment, suggesting a rapid inactivation of MAP kinases in this line.
Fig. 4.
ROS production and MAP kinase activation in response to cellulose binding elicitor lectin (CBEL). (A) Kinetics of ROS production monitored over 35min on leaf discs of 4-week-old A. thaliana plants. ROS production in response to 5 μg ml–1 CBEL was detected on Col-0, Ws-4, Oy-0, and Bla-1 accessions as indicated and compared to water treatments (grey lines); data presented are means of kinetics measured over three–five biological repetitions and bars represent standard deviation. (B) Total photon counts of the kinetics shown on (A); Col-0 data are from Fig. 1 and were included for comparison; bars represent standard deviation and different letters different letters indicate significantly different results (P < 0.05). (C) Detection of MAP kinases phosphorylation. Total protein extracts from leaves infiltrated with CBEL (5 μg ml–1) were analysed by Western blots using a p44/42 antibody. The membranes were stained by Ponceau red to control the equal loading of proteins. Note that Col-0 data are duplicated from Fig. 1 to facilitate direct comparison between lines.
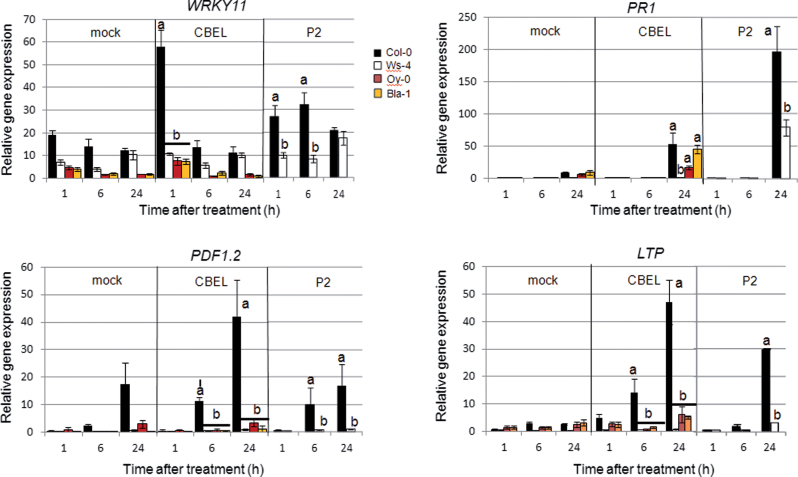
Defence gene expression was monitored in Col-0 and natural variant leaves infiltrated with CBEL (5 μg ml–1) or water at 1, 6, and 24h after treatment. Expression of an early responsive gene (WRKY11) and late responsive genes (LTP, PDF1.2) was not significantly induced after CBEL treatment in the three lines Ws-4, Oy-0, and Bla-1 (Fig. 5). Only Bla-1 plants showed an induction of the late-responsive gene PR-1. In P2-treated Ws-4 plants, WRK11, LTP, and PDF1.2 did not exhibit transcriptional induction as compared to Col-0, though PR-1 did at 24h.
Fig. 5.
Defence gene expression upon infiltration of Phytophthora parasitica elicitors. Gene expression of WRKY11 (AT4G31550), PR1 (AT2G14610), PDF1.2 (AT5G44420), and LTP (AT3G22600) was measured by quantitative reverse-transcription PCR and standardized using a stably expressed gene encoding a chloroplastic housekeeping protein (SAND family protein; AT2G28390). Time-course gene expression was determined upon CBEL infiltration (5 μg ml–1) in Col-0, Oy-0, Ws-4 and Bla-1 leaves and P2 infiltration (50 μg ml–1) in Col-0 and Ws-4 leaves. Data are means of three independent biological repetitions and bars represent the standard deviation. Note that Col-0 data are duplicated from Fig. 2 to facilitate direct comparison between lines.
Taken together, these data revealed a large natural diversity in CBEL responses and showed that some lines were deficient in several CBEL responses, including the development of necrotic symptoms and expression of defence markers.
BAK1 and NADPH oxidase genes are required for Arabidopsis root resistance to Phytophthora
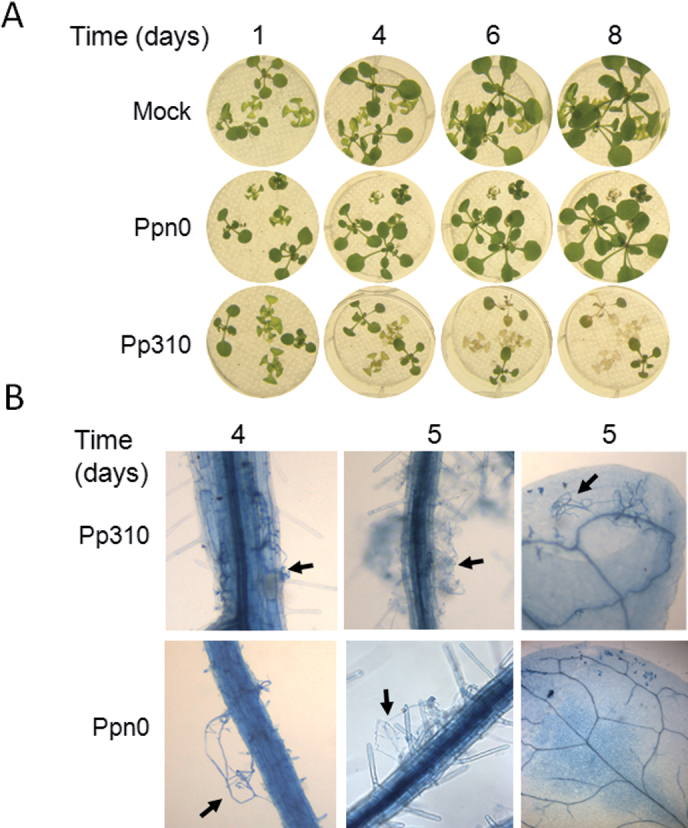
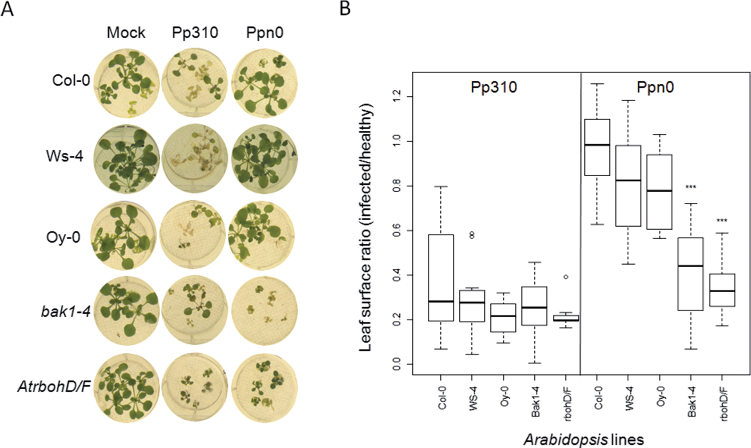
To determine whether the difference in CBEL signalling could affect Phytophthora development in Arabidopis roots, an inoculation method modified from Attard et al. (2010) and Elfstrand et al. (2002) was established. Two Phytophthora parasitica strains initially isolated from tobacco were used. The Pp310 strain is known to cause symptoms on Col-0 (Attard et al., 2010) whereas the Ppn0 strain, from which CBEL was purified (Mateos et al., 1997), was not tested. To quantify the symptoms, digital image analyses were performed by evaluating the mean surface of green tissue per plantlet, 7 days after root treatment with Phytophthora zoospores. As expected, wilting of Col-0 plants inoculated with Pp310 zoospores was observed, whereas no symptom appeared after Ppn0 inoculation (Fig. 6A), indicating that Ppn0 was avirulent on Col-0. Detection of Phytophthora parasitica mycelia by Trypan blue staining revealed that Pp310, but not Ppn0, colonized efficiently root and leaf tissues at 5 days post infection. This pathosystem was used to evaluate the susceptibility of Oy-0 and Ws-4 as well as the bak1-4 and rbohD/F mutants. As shown in Fig. 7, Oy-0 and Ws-4 were equally affected by Pp310, while no symptoms were detected with Ppn0. However, the bak1-4 and rbohD/F mutants appeared to be significantly more susceptible to Ppn0 strain than the wild type, being as susceptible to this strain as to Pp310 strain. Collectively these data show that BAK1 and RBOH genes are required for Phytophthora parasitica resistance in Arabidopsis but that natural variability in CBEL and P2 responses does not have a detectable impact on infection outcome.
Fig. 6.
Disease development on Col-0 plants challenged with Phytophthora parasitica zoospores. Seedlings grown on liquid MS medium were inoculated with 103 zoospores from Phytophthora parasitica Pp310 and Ppn0. (A) Disease progression after inoculation. (B) Detection of Phytophthora parasitica mycelia by Trypan blue staining; arrows indicate mycelia outside or inside plant tissues, 4 and 5 days after inoculation.
Fig. 7.
Susceptibility of Arabidopsis mutants and lines to Phytophthora parasitica. Seedlings grown on liquid MS medium were inoculated with 103 zoospores from Phytophthora parasitica Pp310 and Ppn0 strains as indicated. (A) Symptoms obtained 7 days after inoculation. (B) Quantification of disease was achieved by measuring the ratio of green leaf area between inoculated and healthy plants. Analysis of variance was performed to detect potential ‘line’ and ‘strain’ effects on the ratio of inoculated to non-inoculated leaf green area. Following ANOVA, pairwise Student t-test identified those lines which differed significantly from others in terms of response to Phytophthora strains. *** P <0.001. The data presented are 10–15 independent replicates.
Discussion
The main objective of this study was to gain insight on the mechanisms involved in the response to the necrotic oomycetal elicitor CBEL and the role of CBEL-induced responses in Phytophthora resistance.
As a first approach, Arabidopsis lines mutated in general modulators of PAMP-triggered immunity were used. Arabidopsis bak1-4 and rbohD/F mutants were not impaired in CBEL-induced cell death but were clearly affected in the production of ROS. This result demonstrates that a NADPH oxidase-dependent oxidative burst is not required to trigger CBEL-induced cell death. Moreover, ROS production in the ecotype Bla-1 was similar to that observed in Col-0 whereas Bla-1 did not develop a necrotic reaction after CBEL infiltration. These observations provide evidence that the generation of ROS mediated by NADPH oxidase is not needed for CBEL-induced cell death. Similar results were obtained in previous studies showing that cell death triggered by necrosis-inducing elicitors (β-megaspermin, cryptogein, sphingosin) in tobacco was independent of ROS accumulation (Dorey et al., 1999; Sasabe et al., 2000; Lherminier et al., 2009; Lachaud et al., 2011). However, paradoxical results were obtained on N. benthamiana for which silencing of NbRBOHB compromises INF1-mediated cell death (Yoshioka et al., 2003). In Arabidopsis, two situations have been observed by studying the role of RBOH isoforms during biotic stress. While reduced cell death was observed upon inoculation of rboh mutants with Pseudomonas syringae, enhanced cell death was obtained upon inoculation with the oomycete Peronospora parasitica (Torres et al., 2002). Subsequent work revealed that ROS generated by NADPH oxidases can antagonize salicylic acid-dependent pro-death signals (Torres et al., 2005). These results illustrate the complex and sometimes controversial role of NADPH oxidase in biotic stress and that production of an oxidative burst is not necessarily linked to a cell-death response.
Several reports have shown the role of BAK1 in the induction of basal defence and the resistance to pathogens in Arabidopsis (Chinchilla et al., 2007; Kemmerling et al., 2007; Roux et al., 2011) and in tobacco (Heese et al., 2007; Chaparro-Garcia et al., 2011). In Arabidopsis, BAK1 has been also involved in cell-death control upon infection with necrotrophic pathogens (Kemmerling et al., 2007) and the tobacco SERK3/BAK1 homologues have been shown to be involved in the necrotic response to the Phytophthora infestans INF1 elicitor (Chaparro-Garcia et al., 2011). However, the current study did not detect an effect of the bak1-4 allele on the CBEL-induced necrotic reaction. Similarly, no difference in cell death induced by different necrotic elicitors has been observed in Arabidopsis BAK1 mutants (Postel et al., 2010).
A second approach to study the role of CBEL responses was to identify Arabidopsis lines unable to mount a necrotic response after CBEL treatment. By mining a collection of 50 ecotypes, comprising 37 lines from a core collection maximizing the genetic diversity in A. thaliana (McKhann et al., 2004) and the well-characterized line Ws-4, an extensive variability in CBEL response was detected. Most of these lines, including Ws-4, did not show necrosis after CBEL treatment. While Arabidopsis natural variability of resistance to bacterial and fungal pathogens has been documented for some pathogens including Phytophthora (Denby et al., 2004; Birker et al., 2009; Attard et al., 2010; Nemri et al., 2010; Wang et al., 2011), natural variability to necrotic elicitors has not been explored. The current results suggest that necrotic response to CBEL cannot be generalized to the A. thaliana species. Since the lack of response to CBEL could reflect a general inability to respond to other oomycetal PAMPs, this study tested a Phytophthora crude extract (P2) which is a complex mixture of polysaccharides and proteins (Roux et al., 1994). Among the three lines selected, Oy-0 and Ws-4 were deficient in the response to both elicitors, whereas Bla-1 was only affected in CBEL response. This result suggests that different genetic determinants involved in necrosis-inducing elicitor responses are affected in these lines.
To evaluate the behaviour of CBEL-responsive and CBEL non-responsive lines to Phytophthora infection, two strains of the root pathogen Phytophthora parasitica were used. The Pp310 strain has been recently shown to be virulent on several Arabidopsis ecotypes (Attard et al., 2010). Symptom development of infection was quantitatively affected in Arabidopsis mutants impaired in defence responses related to the salicylic acid, jasmonic acid, and ethylene pathways (Attard et al., 2010), suggesting that basal immune responses could be important to limit the infection. By using a different inoculation procedure of infection, the current work also observed the rapid appearance of disease symptoms upon root inoculation with Pp310 zoospores in all the natural accessions and mutants analysed, confirming the compatibility of this strain with Arabidopsis. However, the use of the Ppn0 strain gave an incompatible phenotype on Col-0. Interestingly this strain was virulent on bak1-4 and rbohD/F, showing that successful resistance to Phytophthora parasitica Ppn0 strongly depends on the induction of a BAK1-dependent basal immune response involving and NADPH oxidase activity. This result also suggests that Pp310, but not Ppn0, is able to suppress this response probably through the production of effectors which remain to be identified in oomycetes. A likely candidate could be an orthologue of the Phytophthora infestans translocated effector Avr3a, which has been has been shown recently to suppress the CBEL-induced necrosis through interaction with the E3 ubiquitin ligase CMPG1 in N. benthamiana (Gilroy et al., 2011).
While Ppn0 resistance depends on an efficient induction of basal immune response, this strain is avirulent on Ws-4 and Oy-0 lines indicating that perception and response to CBEL is not absolutely required to give an incompatible phenotype with Ppn0. These lines are also impaired in the response to the P2 extract, a crude preparation of Phytophthora elicitors, but it cannot be excluded that they are able to perceive other Phytophthora PAMPs or effectors.
To conclude, these results demonstrate that Arabidopsis resistance to the root pathogen Phytophthora parasitica is dependent on PAMP-triggered responses requiring the central immune regulator BAK1 and the production of a NADPH oxidase-mediated oxidative burst. However, natural variability in these responses does not necessarily affect Phytophthora parasitica resistance, suggesting that other mechanisms could be involved in resistance.
Supplementary material
Supplementary data are available at JXB online.
Supplementary Table S1. Primers used in quantitative RT-PCR experiments.
Supplementary Fig. S1. Natural variation among Arabidopsis accessions for cell death response to CBEL.
Acknowledgements
This study was funded by the Université Paul Sabatier (Toulouse, France) and the Centre National de la Recherche Scientifique (France). This work has been done in the LRSV part of the ‘Laboratoire d’Excellence’ (LABEX) entitled TULIP (ANR-10-LABX-41). The authors thank Arnaud Bottin and Christian Mazars (LRSV, Toulouse, France) for helpful discussions, Maxime Bonhomme (LRSV, Toulouse, France) for help with statistical analyses, and Franck Panabières, Mathieu Gourgues, and Agnès Attard (Institut Sophia Agrobiotech, France) for the gift of the Pp310 strain and advice on the Arabidopsis–Phytophthora pathosystem. Mathieu Larroque was supported by a PhD grant from the French Ministère de l’Enseignement Supérieur et de la Recherche. We are grateful to the Toulouse Genomics Platform for access to the BioMarkTM HD System.
References
- Attard A, Gourgues M, Callemeyn-Torre N, Keller H. 2010. The immediate activation of defense responses in Arabidopsis roots is not sufficient to prevent Phytophthora parasitica infection. New Phytologist 187, 449–460. [DOI] [PubMed] [Google Scholar]
- Birker D, Heidrich K, Takahara H, Narusaka M, Deslandes L, Narusaka Y, Reymond M, Parker JE, O’Connell R. 2009. A locus conferring resistance to Colletotrichum higginsianum is shared by four geographically distinct Arabidopsis accessions. The Plant Journal 60, 602–613. [DOI] [PubMed] [Google Scholar]
- Boller T, Felix G. 2009. A renaissance of elicitors: perception of microbe-associated molecular patterns and danger signals by pattern-recognition receptors. Annual Review of Plant Biology 60, 379–406. [DOI] [PubMed] [Google Scholar]
- Boraston AB, Bolam DN, Gilbert HJ, Davies GJ. 2004. Carbohydrate-binding modules: fine-tuning polysaccharide recognition. The Biochemical Journal 382, 769–781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bos JIB, Kanneganti TD, Young C, Cakir C, Huitema E, Win J, Armstrong MR, Birch PRJ, Kamoun S. 2006. The C-terminal half of Phytophthora infestans RXLR effector AVR3a is sufficient to trigger R3a-mediated hypersensitivity and suppress INF1-induced cell death in Nicotiana benthamiana . The Plant Journal 48, 165–176. [DOI] [PubMed] [Google Scholar]
- Bozkurt TO, Schornack S, Banfield MJ, Kamoun S. 2012. Oomycetes, effectors, and all that jazz. Current Opinion in Plant Biology 15, 483–492. [DOI] [PubMed] [Google Scholar]
- Chaparro-Garcia A, Wilkinson RC, Gimenez-Ibanez S, Findlay K, Coffey MD, Zipfel C, Rathjen JP, Kamoun S, Schornack S. 2011. The receptor-like kinase SERK3/BAK1 is required for basal resistance against the late blight pathogen Phytophthora infestans in Nicotiana benthamiana . Plos One 6, e16608 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chinchilla D, Zipfel C, Robatzek S, Kemmerling B, Nürnberger T, Jones JD, Felix G, Boller T. 2007. A flagellin-induced complex of the receptor FLS2 and BAK1 initiates plant defence. Nature 448, 497–500. [DOI] [PubMed] [Google Scholar]
- Denby KJ, Kumar P, Kliebenstein DJ. 2004. Identification of Botrytis cinerea susceptibility loci in Arabidopsis thaliana . The Plant Journal 38, 473–486. [DOI] [PubMed] [Google Scholar]
- Dorey S, Kopp M, Geoffroy P, Fritig B, Kauffmann S. 1999. Hydrogen peroxide from the oxidative burst is neither necessary nor sufficient for hypersensitive cell death induction, phenylalanine ammonia lyase stimulation, salicylic acid accumulation, or scopoletin consumption in cultured tobacco cells treated with elicitin. Plant Physiology 121, 163–171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elfstrand M, Sitbon F, Lapierre C, Bottin A, von Arnold S. 2002. Altered lignin structure and resistance to pathogens in spi 2-expressing tobacco plants. Planta 214, 708–716. [DOI] [PubMed] [Google Scholar]
- Fellbrich G, Romanski A, Varet A, Blume B, Brunner F, Engelhardt S, Felix G, Kemmerling B, Krzymowska M, Nürnberger T. 2002. NPP1, a Phytophthora-associated trigger of plant defense in parsley and Arabidopsis . The Plant Journal 32, 375–390. [DOI] [PubMed] [Google Scholar]
- Gaulin E, Dramé N, Lafitte C, et al. 2006. Cellulose binding domains of a Phytophthora cell wall protein are novel pathogen-associated molecular patterns. The Plant Cell 18, 1766–1777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilroy EM, Taylor RM, Hein I, Boevink P, Sadanandom A, Birch PRJ. 2011. CMPG1-dependent cell death follows perception of diverse pathogen elicitors at the host plasma membrane and is suppressed by Phytophthora infestans RXLR effector AVR3a. New Phytologist 190, 653–666. [DOI] [PubMed] [Google Scholar]
- Gomez-Gomez L, Felix G, Boller T. 1999. A single locus determines sensitivity to bacterial flagellin in Arabidopsis thaliana . The Plant Journal 18, 277–284. [DOI] [PubMed] [Google Scholar]
- Heese A, Hann DR, Gimenez-Ibanez S, Jones AME, He K, Li J, Schroeder JI, Peck SC, Rathjen JP. 2007. The receptor-like kinase SERK3/BAK1 is a central regulator of innate immunity in plants. Proceedings of the National Academy of Sciences, USA 104, 12217–12222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hein I, Gilroy EM, Armstrong MR, Birch PR. 2009. The zig-zag-zig in oomycete–plant interactions. Molecular Plant Pathology 10, 547–562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones JDG, Dangl JL. 2006. The plant immune system. Nature 444, 323–329. [DOI] [PubMed] [Google Scholar]
- Kamoun S, van West P, Vleeshouwers VGAA, de Groot KE, Govers F. 1998. Resistance of Nicotiana benthamiana to Phytophthora infestans is mediated by the recognition of the elicitor protein INF1. The Plant Cell 10, 1413–1425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Journot-Catalino N, Somssich IE, Roby D, Kroj T. 2006. The transcription factors WRKY11 and WRKY17 act as negative regulators of basal resistance in Arabidopsis thaliana . The Plant Cell 18, 3289–3302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kemmerling B, Schwedt A, Rodriguez P, et al. 2007. The BRI1-associated kinase 1, BAK1, has a Brassinoli-independent role in plant cell-death control. Current Biology 17, 1116–1122. [DOI] [PubMed] [Google Scholar]
- Khatib M, Lafitte C, Esquerré-Tugayé MT, Bottin A, Rickauer M. 2004. The CBEL elicitor of Phytophthora parasitica var. nicotianae activates defence in Arabidopsis thaliana via three different signalling pathways. New Phytologist 162, 501–510. [Google Scholar]
- Krol E, Mentzel T, Chinchilla D, et al. 2010. Perception of the Arabidopsis danger signal peptide 1 involves the pattern recognition receptor AtPEPR1 and its close homologue AtPEPR2. Journal of Biological Chemistry 285, 13471–13479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lachaud C, Da, Silva D, Amelot N, Beziat C, Brière C, Cotelle V, Graziana A, Grat S, Mazars C, Thuleau P. 2011. Dihydrosphingosine-induced programmed cell death in tobacco BY-2 cells is independent of H2O2 production. Molecular Plant 4, 310–318. [DOI] [PubMed] [Google Scholar]
- Larroque M, Barriot R, Bottin A, Barre A, Rougé P, Dumas B, Gaulin E. 2013. The unique architecture and function of cellulose-interacting proteins in oomycetes revealed by genomic and structural analyses. BMC Genomics 13, 605 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lherminier J, Elmayan T, Fromentin J, Elaraqui KT, Vesa S, Morel J, Verrier JL, Cailleteau B, Blein JP, Simon-Plas F. 2009. NADPH oxidase-mediated reactive oxygen species production: subcellular localization and reassessment of its role in plant defense. Molecular Plant–Microbe Interactions 22, 868–881. [DOI] [PubMed] [Google Scholar]
- Liu Z, Bos JI, Armstrong M, Whisson SC, da Cunha L, Torto-Alalibo T, Win J, Avrova AO, Wright F, Birch PR, Kamoun S. 2005. Patterns of diversifying selection in the phytotoxin-like scr74 gene family of Phytophthora infestans . Molecular and Biological Evolution 22, 659–672. [DOI] [PubMed] [Google Scholar]
- Mateos FV, Rickauer M, Esquerré-Tugayé MT. 1997. Cloning and characterization of a cDNA encoding an elicitor of Phytophthora parasitica var. nicotianae that shows cellulose-binding and lectin-like activities. Molecular Plant–Microbe Interactions 10, 1045–1053. [DOI] [PubMed] [Google Scholar]
- McKhann HI, Camilleri C, Berard A, Bataillon T, David JL, Reboud X, Le, Corre V, Caloustian C, Gut IG, Brunel D. 2004. Nested core collections maximizing genetic diversity in Arabidopsis thaliana . The Plant Journal 38, 193–202. [DOI] [PubMed] [Google Scholar]
- Nemri A, Atwell S, Tarone AM, Huang YS, Zhao K, Studholme DJ, Nordborg M, Jones JDG. 2010. Genome-wide survey of Arabidopsis natural variation in downy mildew resistance using combined association and linkage mapping. Proceedings of the National Academy of Sciences, USA 107, 10302–10307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orsomando G, Lorenzi M, Raffaelli N, Dalla, Rizza M, Mezzetti B, Ruggieri S. 2001. Phytotoxic protein PcF, purification, characterization, and cDNA sequencing of a novel hydroxyproline-containing factor secreted by the strawberry pathogen Phytophthora cactorum . The Journal of Biological Chemistry 276, 21578–21584. [DOI] [PubMed] [Google Scholar]
- Ottmann C, Luberacki B, Kufner I, et al. 2009. A common toxin fold mediates microbial attack and plant defense. Proceedings of the National Academy of Sciences, USA 106, 10359–10364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ponchet M, Panabières F, Milat ML, Mikes V, Montillet JL, Suty L, Triantaphylides C, Tirilly Y, Blein JP. 1999. Are elicitins cryptograms in plant–oomycete communications? Cellular and Molecular Life Sciences 56, 1020–1047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Postel S, Kufner I, Beuter C, Mazzotta S, Schwedt A, Borlotti A, Halter T, Kemmerling B, Nürnberger T. 2010. The multifunctional leucine-rich repeat receptor kinase BAK1 is implicated in Arabidopsis development and immunity. European Journal of Cell Biology 89, 169–174. [DOI] [PubMed] [Google Scholar]
- Qutob D, Kemmerling B, Brunner F, et al. 2006. Phytotoxicity and innate immune responses induced by Nep1-like proteins. The Plant Cell 18, 3721–3744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roux C, Mazau D, Rickauer M, Fournier J, Berthalon E, Esquerré-Tugayé MT. 1994. Hydroxyproline-containing fragments in the cell wall of Phytophthora parasitica . Phytochemistry 35, 591–595. [Google Scholar]
- Roux M, Schwessinger B, Albrecht C, Chinchilla D, Jones A, Holton N, Malinovsky FG, Tor M, de Vries S, Zipfel C. 2011. The Arabidopsis leucine-rich repeat receptor-like kinases BAK1/SERK3 and BKK1/SERK4 are required for innate immunity to hemibiotrophic and biotrophic pathogens. The Plant Cell 23, 2440–2455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sasabe M, Takeuchi K, Kamoun S, Ichinose Y, Govers F, Toyoda K, Shiraishi T, Yamada T. 2000. Independent pathways leading to apoptotic cell death, oxidative burst and defense gene expression in response to elicitin in tobacco cell suspension culture. European Journal of Biochemistry 267, 5005–5013. [DOI] [PubMed] [Google Scholar]
- Séjalon-Delmas N, Mateos FV, Bottin A, Rickauer M, Dargent R, Esquerré-Tugayé MT. 1997. Purification, elicitor activity, and cell wall localization of a glycoprotein from Phytophthora parasitica var. nicotianae, a fungal pathogen of tobacco. Phytopathology 87, 899–909. [DOI] [PubMed] [Google Scholar]
- Shan L, He P, Li J, Heese A, Peck SC, Nurnberger T, Martin GB, Sheen J. 2008. Bacterial effectors target the common signaling partner BAK1 to disrupt multiple MAMP receptor-signaling complexes and impede plant immunity. Cell Host and Microbe 4, 17–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomma BPJH, Nürnberger T, Joosten MHAJ. 2011. Of PAMPs and effectors: the blurred PTI-ETI dichotomy. The Plant Cell 23, 4–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tordai H, Banyai L, Patthy L. 1999. The PAN module: the N-terminal domains of plasminogen and hepatocyte growth factor are homologous with the apple domains of the prekallikrein family and with a novel domain found in numerous nematode proteins. FEBS Letters 461, 63–67. [DOI] [PubMed] [Google Scholar]
- Torres MA, Dangl JL, Jones JDG. 2002. Arabidopsis gp91(phox) homologues AtrbohD and AtrbohF are required for accumulation of reactive oxygen intermediates in the plant defense response. Proceedings of the National Academy of Sciences, USA 99, 517–522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torres MA, Jones JDG, Dangl JL. 2005. Pathogen-induced, NADPH oxidase-derived reactive oxygen intermediates suppress spread of cell death in Arabidopsis thaliana . Nature Genetics 37, 1130–1134. [DOI] [PubMed] [Google Scholar]
- Wang Y, Meng YL, Zhang M, Tong XM, Wang QH, Sun YY, Quan JL, Govers F, Shan WX. 2011. Infection of Arabidopsis thaliana by Phytophthora parasitica and identification of variation in host specificity. Molecular Plant Pathology 12, 187–201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshioka H, Numata N, Nakajima K, Katou S, Kawakita K, Rowland O, Jones JD, Doke N. 2003. Nicotiana benthamiana gp91phox homologs NbrbohA and NbrbohB participate in H2O2 accumulation and resistance to Phytophthora infestans . The Plant Cell 15, 706–718. 12615943 [Google Scholar]
- Zipfel C, Kunze G, Chinchilla D, Caniard A, Jones JD, Boller T, Felix G. 2006. Perception of the bacterial PAMP EF-Tu by the receptor EFR restricts Agrobacterium-mediated transformation. Cell 125, 749–760. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.