Summary
Fibroblast growth factor (FGF) signalling has been implicated during several phases of early embryogenesis, including the patterning of the embryonic axes, the induction and/or maintenance of several cell lineages and the coordination of morphogenetic movements. Here, we summarise our current understanding of the regulation and roles of FGF signalling during early vertebrate development.
Keywords: Fibroblast growth factor, Embryogenesis, Mesoderm, Morphogenesis, Patterning, Stem cells
Introduction
The first fibroblast growth factor (FGF) ligands, FGF1 and FGF2, were initially purified from brain as mitogenic factors of fibroblasts grown in culture (Gospodarowicz and Moran, 1975). Since their discovery, FGF ligands and their receptors have been implicated in numerous biological processes (Table 1), and their dysregulation causes several congenital diseases (such as dwarfism) and some types of cancer (Table 2) (reviewed by Beenken and Mohammadi, 2009). In addition to their mitogenic capacity, FGFs can also modulate cell survival, migration and differentiation in culture (Dailey et al., 2005; Xian et al., 2005).
Table 1. Phenotypes of mutants with disrupted FGF signalling.
| Gene and allele | Phenotype | References |
|---|---|---|
|
Mouse
| ||
| Fgf4 KO | Post-implantation lethality; impaired ICM proliferation; defect in TE and PrE maintenance |
Feldman et al., 1995; Goldin and Papaioannou, 2003 |
| Fgf8 KO | Early embryonic lethal; failure of cell migration during gastrulation |
Meyers et al., 1998; Sun et al., 1999 |
| Fgfr1 KO | Lethal E7.5-9.5; defect in morphogenetic movements; lack of paraxial mesoderm (PxM); inability to migrate away from the primitive streak |
Yamaguchi et al., 1994; Deng et al., 1994; Ciruna et al., 1997 |
| Fgfr2 KO | Early embryonic lethal; defect in visceral endoderm differentiation and in ICM maintenance |
Arman et al., 1998 |
|
| ||
|
Xenopus
| ||
| DN FGFR1 | Defects in mesoderm specification and gastrulation movements; loss of trunk and tail |
Amaya et al., 1991; Amaya et al., 1993 |
| DN FGFR4 | Blocks anterior neural induction; represses posterior neural induction |
Hongo et al., 1999; Hardcastle et al., 2000 |
| MO FGF4 | Inhibits muscle formation; expanded blood | Fisher et al., 2002; Isaacs et al., 2007 |
| MO FGF8 | Disruption of gastrulation; reduction of paraxial mesoderm, hindbrain and spinal cord |
Fletcher et al., 2006 |
| MO FGF8a | Lack of posterior neuronal tissue | Fletcher et al., 2006 |
| SU5402 (general FGF receptor antagonist) |
Open blastopore; decreased expression of mesodermal markers; inhibition of PxM induction; failure in axial mesoderm maintenance |
Sivak et al., 2005; Fletcher and Harland, 2008 |
|
| ||
|
Drosophila
| ||
| heartless | Defect in mesodermal cell migration; lack of visceral mesoderm | Gisselbrecht et al., 1996; Beiman et al., 1996 |
| thisbe | Defect in mesoderm spreading | Kadam et al., 2009 |
| pyramus | Lack of differentiation of dorsal mesoderm | Kadam et al., 2009 |
|
| ||
|
Zebrafish
| ||
| Fgf8 (mutants and MO) | Defects in somitogenesis and in MHB maintenance; defect in L/R axis specification |
Reifers et al., 1998; Albertson and Yelick, 2005 |
|
Fgf8 (mutant) + Fgf24 (MO) |
Lack of posterior mesoderm | Draper et al., 2003 |
| Fgf4 (MO) | Defect in L/R axis specification | Yamauchi et al., 2009 |
| DN Fgfr1 | Loss of trunk and tail; loss of ntl expression | Griffin et al., 1995 |
| DN Fgfr3 | Defect in AP patterning of the neural plate and absence of notochord |
Ota et al., 2009 |
|
| ||
|
Chick
| ||
| SU5402 | Disruption of movements of streak cells; lack of neuronal induction |
Yang et al., 2002; Streit et al., 2000 |
| DN FGFR1 | Disruption of spinal cord elongation | Mathis et al., 2001 |
|
| ||
|
Other organisms
| ||
| Sea urchin MO FGFA |
Failure of PMC migration | Rottinger et al., 2007 |
| Ascidian FGF4/6/9/20 (MO) +FGF8/17/18 (MO) |
Absence of mesenchymal cells No neural induction |
Imai et al., 2002; Bertrand et al., 2003; Yasuo and Hudson, 2007 |
| FGF9/16/20 (MO) | Absence of notochord | |
AP, anterior posterior; DN, dominant negative; ICM, inner cell mass; KO, knockout; L/R, left/right; MHB, midbrain-hindbrain; MO, morpholino oligonucleotide; ntl, no tail; PMC, primary mesenchyme cells; PrE, primitive endoderm; PxM, paraxial mesoderm; TE, trophectoderm.
Table 2. Human congenital and pathological diseases associated with FGF signalling.
| Ligand or receptor | Disease |
|---|---|
| Loss-of-function mutations | |
|
| |
| FGF3 | Deafness |
| FGF8 | Kallman syndrome; cleft palate |
| FGF9 | Colorectal, endometrial and ovarian carcinomas |
| FGF10 | Aplasia of lacrymal and salivary glands; non-syndromic cleft lip and palate; hearing loss |
| FGF14 | Spinocerebellar ataxia |
| FGF23 | Familial tumoural calcinosis (FTC) |
|
| |
| Increased level of expression | |
|
| |
| FGF2/FGF6 | Prostate cancer |
| FGF19 | Liver, colon and lung squamous carcinomas |
| FGF23 | Osteomalacia |
|
| |
| Gain-of-function mutations | |
|
| |
| FGF23 | Hypophosphataemia |
| FGFR1 (germline) | Kallman and Pfeiffer syndromes; osteoglophonic dysplasia |
| FGFR1 (somatic) | Glioblastoma; malignant prostate cells; melanoma (rare) |
| FGFR2 (germline) | Apert syndrome; Crouzon; Pfeiffer; Jackson-Weiss; Antlley-Bixler; Beare-Stevenson syndromes |
| FGFR2 (somatic) | Endometrial cancer (12%) and gastric cancer (rare) |
| FGFR3 (germline) | Muencke syndrome; hypochondroplasia; thanatophoric dysplasia |
| FGFR3 (somatic) | Bladder cancer (50%); cervical cancer (5%); B-cell malignancy; myelanomas |
| FGFR4 (somatic) | Mutation associated with aggressive prostate cancer |
|
| |
| Genomic translocations | |
|
| |
| ZNF198-FGFR1 | Myeloproliferative disease |
| BCR-FGFR1 | Stem cell leukaemia and lymphoma; chronic myelogenous leukaemia (CML, rare) |
| ETV6-FGFR3 | Myelanomas (15%); peripheral T-cell lymphoma (rare) |
|
| |
| Amplification | |
|
| |
| FGFR1 | Breast, ovarian and bladder cancers (fewer than 10% of the cases) |
| FGFR2 | Gastric cancer (10%) and breast cancer (~1%) |
|
| |
| SNPs | |
|
| |
| FGF20 | Parkinson’s disease |
| FGFR2 | Increase incidence of breast cancer |
| FGFR4 | Poor prognosis in breast, colon and lung adenocarcinomas |
Data compiled from published sources (Beenken and Mohammadi, 2009; Krejci et al., 2009; Turner and Grose, 2010; Wilkie, 2005).
BCR, breakpoint cluster region; ETV, ETS variant; SNP, single-nucleotide polymorphism; ZNF, zinc finger, MYM-type.
During embryogenesis, FGF signalling plays an important role in the induction/maintenance of mesoderm and neuroectoderm, the control of morphogenetic movements, anteroposterior (AP) patterning, somitogenesis and the development of various organs (Table 1) (Bottcher and Niehrs, 2005; Itoh, 2007; McIntosh et al., 2000). Here, we briefly describe the FGF signalling pathway and then summarise the main developmental processes in which FGF signalling plays an important role during early vertebrate embryogenesis, including cell fate specification and axis determination.
FGF signalling: an overview
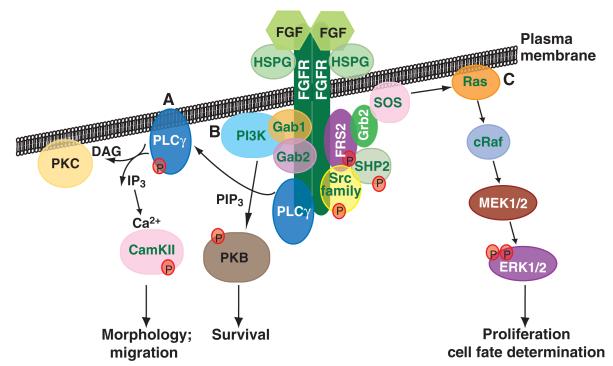
Members of the FGF family of extracellular ligands are characterised by a conserved core of 140 amino acids and their strong affinity for heparin sulphate (HS) (see Glossary, Box 1). In vertebrates, 22 family members have been identified and are grouped into seven subfamilies according to their sequence homology and function (Ornitz, 2000). All FGFs, with the exception of the intracellular FGFs (iFGFs, FGF11-14), signal through a family of tyrosine kinase receptors, the FGF receptors (FGFRs). In vertebrates, the FGFR family consists of four genes, FGFR1-4, which undergo alternative splicing in their extracellular domain to generate a vast variety of receptors with different affinities for their ligands (Zhang et al., 2006). FGF ligands bind the extracellular domain of the FGFRs in combination with heparan sulphate to form a 2:2:2 FGF:FGFR:heparan dimer. The dimerisation of the receptor results in the transphosphorylation of specific intracellular tyrosine residues (Fig. 1). This triggers the activation of cytoplasmic signal transduction pathways, such as the Ras/ERK pathway (which is associated with proliferation and differentiation), the Akt pathway (associated with cell survival) or the protein kinase C (PKC) pathways (involved in cell morphology and migration) (Dailey et al., 2005; Mohammadi et al., 2005; Schlessinger, 2000).
Box 1. Glossary.
- Blastopore.
Site of continuous cell involution during gastrulation. In vertebrates, the blastopore gives rise to the anus of the embryo.
- Bottle cells.
Cells that lead to the initiation of involution during gastrulation, as they adopt a characteristic bottle shape through apical constriction.
- Gastrocoel roof plate.
Ciliated epithelium on the roof of the archenteron in Xenopus embryos; important in establishing the left/right (L/R) axis.
- Heparin sulphate (HS).
A highly sulphated glycosaminoglycan found at the surface of the cells. It considerably increases the affinity of FGF ligands for their receptors.
- Inner cell mass (ICM).
Population of cells in the early mouse embryo that occupies the inside of the preimplantation embryo. These cells give rise to the embryo proper and to some extra-embryonic membranes.
- Kupffer’s vesicle.
Ciliated epithelium in the zebrafish embryo that plays an important role in L/R axis establishment.
- Marginal zone.
The equatorial region in Xenopus and zebrafish embryos at the late blastula stage, which gives rise to the mesoderm.
- Primitive blood.
First wave of blood formation in the embryo. In frogs and fish, the cells fated to differentiate into primitive myeloid cells arise from anterior ventral mesoderm and cells fated to differentiate into primitive erythroid cells arise from posterior ventral mesoderm.
- Primitive endoderm.
Derived from the ICM. It consists of the cell layer facing the blastocyst cavity in the preimplantation mammalian embryo and gives rise to the extra-embryonic endoderm of the yolk sac.
- Primitive mesenchymal cells (PMCs).
Cells in the vegetal plate of the sea urchin embryo, which ingress during gastrulation. The PMCs are fated to become mesoderm and they form the skeletal elements of the embryo.
- Regulative development.
Embryonic development in which cells are specified by their environment, rather than by inheriting cytoplasmic determinants (as occurs during mosaic development).
- Stem zone.
Population of cells in the posterior-most region of the epiblast in the chick and mouse that are undifferentiated and proliferative and give rise to either spinal cord or somitic mesoderm.
- Trophectoderm.
The extra-embryonic ectoderm occupying the outer layer of the mammalian blastocyst. It gives rise to the trophoblast and contributes to the placenta.
- Vascular endothelial growth factor A (VEGF-A).
One of three ligands (together with VEGF-B and VEGF-C) that activate the VEGF receptor (VEGFR), a member of the receptor tyrosine kinase family. The VEGF signalling pathway is primarily involved in vascular development.
Fig. 1. An overview of FGF signalling.
FGF signalling is initiated by ligand-dependent dimerisation of the FGFR, which leads to the cross-phosphorylation (P) of tyrosine residues in the intracellular domain of the receptor tyrosine kinase (not shown). These phosphorylated residues are then bound specifically by several intracellular signal transduction proteins, including PLCγ, FRS2 and Src family members. These initiate several intracellular signalling pathways, including the (A) PLCγ pathway, (B) PI3K/PKB pathway and (C) the Ras/ERK pathway. The cell responses to these different pathways are shown. CamKII, calcium/calmodulin-dependent protein kinase II; DAG, diacylglycerol; ERK, extracellular-signal related kinase; FGF, fibroblast growth factor; FGFR, fibroblast growth factor receptor; FRS2, fibroblast growth factor receptor substrate 2; Gab, Grb2-associated protein; Grb2, growth factor receptor-bound protein 2; HSPG, heparan sulphate proteoglycan; IP3, inositol (1,4,5)-trisphosphate; MEK, mitogen-activated protein kinase kinase (also known as MAP2K); PI3K, phosphoinositide 3-kinase; PIP3, phosphatidylinositol (3,4,5)-trisphosphate; PKB, protein kinase B; PKC, protein kinase C; PLCγ, phospholipase C γ; cRaf, v-raf-leukemia viral oncogene homologue 1 (also known as RAF1); Ras, rat sarcoma (also known as Harvey rat sarcoma virus oncogene homologue); SHP2, SH2 domain-containing tyrosine phosphatase 2 (also known as PTPN11); SOS, son of sevenless; Src, sarcoma proto-oncogene tyrosine kinase.
The role of FGF signalling during mesoderm formation
FGF and mesoderm specification
Although the general principle of induction was established by Hans Spemann and colleagues in the early part of the 20th century (for a review, see Hamburger, 1988), it was not until the late 1980s that the molecular nature of the inducing signals began to be elucidated. Indeed, the discovery that FGF1 and FGF2 could induce mesoderm from naïve prospective ectodermal cells in Xenopus was a turning point in experimental embryology, propelling it into the modern molecular age (Kimelman and Kirschner, 1987; Slack et al., 1987). Since this discovery, an impressive amount of work has been done to try to elucidate the various roles that FGF has during mesoderm formation.
Early experiments carried out primarily in Xenopus and zebrafish showed that FGF signalling is required for the formation of axial mesoderm (which forms the notochord) and paraxial mesoderm (which gives rise to the axial skeleton, skeletal muscles and dermis) (Amaya et al., 1991; Amaya et al., 1993; Griffin et al., 1995). Whether the role for FGF during axial and paraxial mesoderm formation is in the initial induction or in the maintenance of these mesodermal subtypes has remained a contentious issue for some time. Recently, Fletcher and Harland addressed this question by performing a careful analysis of the initiation of expression of several early mesodermal markers, when FGF signalling was inhibited with the FGFR inhibitor SU5402 (Fletcher and Harland, 2008). They found that if FGF signalling is inhibited before mesoderm induction, then the early paraxial mesodermal markers myogenin D (myoD) and myogenic regulatory factor 5 (myf5) are never expressed (Fletcher and Harland, 2008). By contrast, the inhibition of FGF signalling with SU5402 before mesoderm induction left axial mesoderm induction largely unaffected initially, although several axial mesoderm markers are subsequently lost due to a requirement for FGF signalling for the maintenance of axial mesoderm.
In summary, these data suggest that the induction of paraxial mesoderm requires FGF signalling, whereas axial mesoderm requires FGF signalling primarily for its maintenance but not for its induction. It is now clear that FGF signalling is not essential for mesoderm formation per se. For example, a pan-mesodermal marker, eomes, is not affected by inhibiting FGF signalling in Xenopus (Fletcher and Harland, 2008; Kumano et al., 2001). In addition, some mesodermal subtypes, such as primitive blood (see Glossary, Box 1), are inhibited by FGF (Isaacs et al., 2007; Kumano and Smith, 2000; Walmsley et al., 2008; Xu, R. H. et al., 1999). See Box 2 for a further discussion of the role of FGF signalling during neural induction.
Box 2. The role of FGF signalling in neural induction: a controversy.
Neural tissue forms from embryonic ectoderm via the activation and inhibition of several signalling pathways (reviewed by Levine and Brivanlou, 2007; Stern, 2005). FGF signalling is one pathway implicated in neural induction, based on evidence obtained from several model organisms, including ascidians, Xenopus, zebrafish and chick (Alvarez et al., 1998; Hudson and Lemaire, 2001; Inazawa et al., 1998; Kengaku and Okamoto, 1995; Kudoh et al., 2004; Lamb and Harland, 1995; Rodriguez-Gallardo et al., 1997; Storey et al., 1998). Several controversies, however, remain as to the exact role of FGF signalling during this process [for a review, see Stern (Stern, 2005); see also Linker et al. (Linker et al., 2009) versus Wills et al. (Wills et al., 2010)]. Indeed, whether FGF signalling is absolutely necessary for neural induction remains a matter of debate. Experiments in Xenopus have shown that FGF signalling is both dispensable (Amaya et al., 1991; Holowacz and Sokol, 1999; Kroll and Amaya, 1996; Ribisi et al., 2000; Wills et al., 2010) and indispensable (Delaune et al., 2005; Launay et al., 1996; Linker and Stern, 2004; Sasai et al., 1996) for neural induction, possibly owing to slight differences in the experimental approaches used or to differences in the competence of the ectoderm between experimental regimes (which can vary even more between different model organisms, such as between frog and chick embryos). Indeed, evidence from chick suggests that FGF signalling provides the initiating neuralising signal that prepares the prospective neural plate for further neural-inducing signals (Sheng et al., 2003; Streit et al., 2000; Wilson et al., 2000). As such, FGF signalling appears to act very early as a competence factor for neural induction. Alternatively, perhaps a very early, but transient, requirement for FGF signalling is required to push primitive ectoderm (epiblast) cells away from pluripotency and toward differentiation, as suggested from mouse ES cell studies (Stavridis et al., 2010; Stavridis et al., 2007). Yet other studies suggest that FGF signalling leads to neural induction through the attenuation of bone morphogenetic protein (BMP) signalling (for a review, see De Robertis and Kuroda, 2004). Although the exact function (or functions) that FGF signalling performs during neural induction remains unclear, a consensus is emerging that FGF signalling does play an important role, in particular during the induction of the posterior nervous system (Holowacz and Sokol, 1999; Rentzsch et al., 2004; Wills et al., 2010).
Several key questions remain regarding the role of FGF signalling during the induction and maintenance of axial and paraxial mesoderm. For example, what regulates the expression of the different FGF ligands and their receptors during development, especially given that their expression is highly dynamic during early embryogenesis (Lea et al., 2009)? What are the distinct roles versus the redundant roles that each ligand and receptor has during early development? Although some progress toward resolving these questions has been made over recent years (Beenken and Mohammadi, 2009; Itoh and Ornitz, 2008; Ota et al., 2009), a comprehensive study of the function of each FGF ligand and receptor, alone or in combination, during early embryonic development is still a priority for the future, although such an analysis has been undertaken for limb formation in the mouse (Mariani et al., 2008).
FGF and morphogenetic movements
In addition to its role in the formation of axial and paraxial mesoderm, there is compelling evidence that FGF signalling also has an essential role in the coordination of cell movements during gastrulation. A potential role for FGF signalling in gastrulation movements was first suggested by the phenotype observed in Xenopus embryos that overexpress a dominant-negative FGFR (Amaya et al., 1991). Although bottle cell and blastopore lip formation was undisturbed in these embryos, gastrulation movements failed soon after, leaving the embryos with an open blastopore (see Glossary, Box 1) (Amaya et al., 1991). Given that FGF signalling is required for the formation of axial and paraxial mesoderm, the two mesodermal cell types that are primarily responsible for orchestrating the movements of gastrulation, it could not easily be discerned whether the defect in cell movements was a direct or indirect effect, mediated by a failure of proper mesoderm specification. However, the identification of Sprouty and Spred proteins, two modulators of FGFR signalling, facilitated the uncoupling of the two distinct roles of FGF signalling during mesoderm specification and morphogenesis (Nutt et al., 2001; Sivak et al., 2005). Rather than blocking FGF signalling completely, these two proteins inhibit different intracellular signalling pathways downstream of the FGFR and thereby modulate mesoderm specification versus morphogenesis distinctly. More specifically, Sprouty proteins inhibit the phospholipase C (PLC) γ/PKCδ/Ca2+ pathway, but leave the Ras/ERK pathway downstream of FGFR intact, allowing the specification of the mesoderm during the early gastrula stages (Nutt et al., 2001; Sivak et al., 2005). During the mid- to late gastrula stages, the expression of the Sprouty genes decreases, while the expression of the Spred genes increases (Sivak et al., 2005). In contrast to the Sprouty proteins, Spred proteins inhibit the Ras/ERK pathway while leaving the PLCγ/PKCδ/Ca2+ pathway unaffected. This switches the intracellular pathways activated by FGF from the Ras/ERK pathway to the PLCγ/PKCδ/Ca2+ pathway. Thus, cells specified as mesoderm can now be instructed to undergo morphogenetic movements, using the same primary signal, FGF. As such, the role of FGF signalling during gastrulation can be divided in two distinct elements: (1) an early, ERK-dependent transcriptional role that specifies and/or maintains axial and paraxial mesoderm; and (2) a later morphogenetic role, which is not ERK-dependent and which coordinates cell movements during gastrulation and neurulation (see Fig. 1).
How the switch from Sprouty to Spred gene expression occurs at the transcriptional level is still unknown, but resolving this question should provide us with important clues as to how cells control the way they interpret growth factor signals appropriately during development. The molecular mechanism by which Sprouty proteins inhibit the PLCγ/PKCδ/Ca2+ pathway is also unclear, although cell culture experiments have shown that Sprouty4 can prevent PKCδ phosphorylation and phosphatidylinositol (4,5)-bisphosphate (PIP2) breakdown downstream of vascular endothelial growth factor A (VEGF-A) signalling (see Glossary, Box 1) (Ayada et al., 2009). Whether the same mechanism applies downstream of FGFR signalling is not yet known.
The ability of FGF signalling to control morphogenetic movements seems to be conserved throughout evolution because it has been shown that, in the sea urchin, the ligand FGFA and its receptor FGFR2 are necessary for the migration of the primary mesenchyme cells (see Glossary, Box 1) (Rottinger et al., 2007). Similarly, in Drosophila, a mutation in the fgfr2 gene heartless results in the failure of mesodermal cells to migrate away from the midline during gastrulation (Beiman et al., 1996; Gisselbrecht et al., 1996). In addition, thisbe and pyramus, two fgf8-like genes in Drosophila, are important for mesoderm migration during gastrulation (Gryzik and Muller, 2004; Kadam et al., 2009; Klingseisen et al., 2009). Also Fgfr1-null and Fgf8-null mouse embryos display severe defects in cell migration during gastrulation (Deng et al., 1994; Sun et al., 1999; Yamaguchi et al., 1994). An analysis of chimeric mice that contain Fgfr1−/− cells has shown that the primary defect in these cells is their inability to traverse and migrate away from the primitive streak (Ciruna et al., 1997). Finally, studies using FGF4- and FGF8b-coated beads implanted into chick embryos have shown that these ligands have striking, but opposite, effects on the migration of primitive streak cells: primitive streak cells move towards an FGF4 source but away from an FGF8b source (Yang et al., 2002).
Although these studies highlight the importance of FGF signalling in the coordination of cell movements during gastrulation, it is still not understood how the different ligands can induce different cellular responses. Furthermore, it is not known which intracellular signalling pathways are responsible for mediating these different migratory behaviours. It is notable that cells migrating out of the primitive streak in the mouse do not appear to stain with antibodies specific for activated ERK (Corson et al., 2003), suggesting that the pathway responsible for migration is not dependent on the Ras/ERK pathway. However, it is not yet known in the mouse whether the PLCγ/PKCδ/Ca2+ pathway or the PI3K pathway, which has been shown in the chick to be crucial for directed cell migration (Leslie et al., 2007), mediates the migratory behaviour of the mesodermal cells through the primitive streak. Furthermore, it is not known how FGF signalling interacts with other signalling pathways, such as the Wnt planar cell polarity (PCP) pathway, to control the cellular movements of gastrulation. However, there are at least two possible mechanisms by which the non-canonical Wnt pathway and the FGF pathway could interact. One is transcriptional, as wnt11, which encodes a key ligand in the control of convergent extension movements in zebrafish and Xenopus, depends on brachyury and FGF signalling for its expression (Amaya et al., 1993; Heisenberg et al., 2000; Tada and Smith, 2000). The other mechanism involves the shared common regulator of both pathways in the form of PKCδ (Kinoshita et al., 2003; Sivak et al., 2005).
FGF signalling in axes specification
The dorsoventral axis
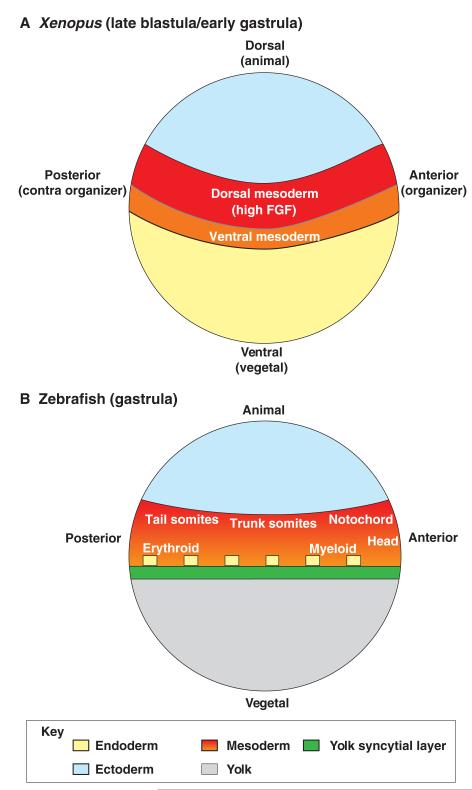
As mentioned previously, FGF signalling is essential for the specification and/or maintenance of axial and paraxial mesoderm (Amaya et al., 1993; Fletcher and Harland, 2008). In addition, FGF inhibits blood development (Isaacs et al., 2007; Kumano and Smith, 2000; Walmsley et al., 2008; Xu, R. H. et al., 1999). As such, FGF signalling promotes dorsal mesoderm specification and inhibits ventral mesoderm specification. In Xenopus, FGF appears to perform this patterning function by specifying the animal-vegetal axis of the embryo (Kumano et al., 2001; Kumano and Smith, 2000; Kumano and Smith, 2002b). In this organism, the dorsoventral (DV) axis aligns with the animal-vegetal axis of the embryo (Fig. 2A) (Kumano and Smith, 2002a; Lane and Sheets, 2002; Lane and Smith, 1999). Cells occupying the animal sector of the marginal zone are fated to give rise to dorsal mesoderm (Kumano and Smith, 2000; Kumano and Smith, 2002a) (see Glossary, Box 1; Fig. 2A), whereas those occupying the more vegetal sector around the entire marginal zone (the first cells to involute during gastrulation) are fated to give rise to ventral mesoderm. In the anterior (organizer) region, those cells that involute first give rise to anterior head mesoderm and to anterior ventral blood islands, which are the precursors of primitive myeloid blood cells (Chen et al., 2009; Kumano and Smith, 2002a; Lane and Sheets, 2002; Lane and Smith, 1999). In the contra organizer sector (i.e. in the posterior region) of the marginal zone, the first involuting cells give rise to the posterior ventral blood islands, which are the precursors of primitive erythroid cells (Kumano and Smith, 2002a; Lane and Sheets, 2002; Lane and Smith, 1999). Although it is not known whether this is the case in organisms other than Xenopus, the fate map of the zebrafish embryo makes it a distinct possibility (see Fig. 2B) (Kimelman, 2006; Lieschke et al., 2002).
Fig. 2. FGF signalling is necessary for the specification and maintenance of dorsal mesoderm.
Fate map of different germ layers at the late blastula/early gastrula stage, along the dorsal-ventral (animal-vegetal) and anterior-posterior (organizer-contra organizer) axes in (A) Xenopus and (B) zebrafish embryos. FGF signalling is high in the animal sector of the marginal zone (red), which is fated to become dorsal axial and paraxial mesoderm, whereas it is low or absent in the vegetal sector of the marginal zone (green in B), which is fated to give rise to ventral mesoderm, such as blood.
In summary, FGF signalling plays a crucial role in specifying the animal-vegetal axis of the Xenopus embryo and promotes dorsal fates in the animal sector of the marginal zone (Kumano et al., 2001; Kumano and Smith, 2000; Kumano and Smith, 2002b). Consistent with this model, expression of FGF4, FGF8 and FGF20 is contained within the animal (dorsal) sector of the marginal zone and is absent from the vegetal (ventral) sector of the marginal zone (Christen and Slack, 1997; Isaacs et al., 1992; Lea et al., 2009). Furthermore, FGF-dependent activation of ERK is found only in the animal (dorsal) sector of the marginal zone at the gastrula stages (Christen and Slack, 1999; Curran and Grainger, 2000; Kumano et al., 2001).
FGF signalling, however, does not pattern the DV axis alone. In fact, this axis is defined primarily by the maternal Wnt/β-catenin pathway and the zygotic bone morphogenetic protein (BMP) pathway (reviewed by De Robertis, 2009; Little and Mullins, 2006; Schier and Talbot, 2005; Weaver and Kimelman, 2004). Several lines of evidence in Xenopus and zebrafish suggest that FGF signalling promotes dorsal fates and inhibits ventral fates by restricting the expression and activity of BMPs. For example, FGF signalling in zebrafish inhibits the expression of BMPs in dorsal mesoderm, thus limiting their expression to ventral mesoderm (Furthauer et al., 1997; Furthauer et al., 2004). Furthermore, FGF signalling in Xenopus is required for the continued expression of the BMP antagonists chordin and noggin in the anterior dorsal mesoderm (Branney et al., 2009; Fletcher and Harland, 2008). Finally, FGF signalling has been shown to inhibit BMP signalling via ERK-dependent phosphorylation of the linker domain of Smad1, a crucial intracellular mediator of BMP signalling (Eivers et al., 2008; Pera et al., 2003). Indeed cross-inhibitory effects between FGF and BMP signalling are found throughout embryogenesis, constituting a common module in development (Koshida et al., 2002; Minina et al., 2002; Niswander and Martin, 1993; Wilson et al., 2000; Xu, R. H. et al., 1999).
The anteroposterior axis
In addition to its role in promoting dorsal fates and inhibiting ventral fates, FGF signalling has also been implicated in the establishment of the AP axis of the early embryo. A role for FGF in AP patterning has been implicated following both gain- and loss-of-function experiments in Xenopus, zebrafish, chick and mouse (Amaya et al., 1991; Christen and Slack, 1997; Davidson et al., 2000; Draper et al., 2003; Griffin et al., 1995; Isaacs et al., 1994; Isaacs et al., 1992; Kudoh et al., 2002; Ota et al., 2009; Partanen et al., 1998; Storey et al., 1998; Xu, X. et al., 1999). In particular, FGF has a strong posteriorising effect on neuroectoderm, suggesting that it might provide at least part of the transforming/caudalising signal first postulated by Pieter Nieuwkoop and Lauri Saxen in the 1950s (Cox and Hemmati-Brivanlou, 1995; Doniach, 1995; Kengaku and Okamoto, 1995; Lamb and Harland, 1995; Nieuwkoop, 1952; Saxen and Toivonen, 1961). FGFs perform this posteriorising function, at least in part, through their regulation of the ParaHox and Hox genes (Bel-Vialar et al., 2002; Cho and De Robertis, 1990; Haremaki et al., 2003; Isaacs et al., 1998; Keenan et al., 2006; Northrop and Kimelman, 1994; Partanen et al., 1998; Pownall et al., 1996; Shiotsugu et al., 2004). Furthermore, in all vertebrate species tested, FGF4 and/or FGF8 are expressed in posterior mesoderm, and therefore FGFs are present at the right time and place to act as endogenous posteriorising factors (Christen and Slack, 1997; Crossley and Martin, 1995; Dubrulle and Pourquie, 2004; Isaacs et al., 1995; Isaacs et al., 1992; Ohuchi et al., 1994; Shamim and Mason, 1999). Interestingly, FGF signalling patterns the AP axis in all germ layers (neurectoderm, mesoderm and endoderm) (Cox and Hemmati-Brivanlou, 1995; Dessimoz et al., 2006; Lamb and Harland, 1995; Partanen et al., 1998; Pownall et al., 1996; Wells and Melton, 2000; Xu, X. et al., 1999).
As with DV patterning, FGF signalling does not regulate AP patterning on its own. Indeed, the embryonic AP axis is established through the coordinated action of several signalling molecules, including FGFs, retinoic acid (RA) and Wnts (Bayha et al., 2009; Blumberg et al., 1997; Doniach, 1995; Durston et al., 1989; Kiecker and Niehrs, 2001; McGrew et al., 1997; McGrew et al., 1995; Sive et al., 1990; Takada et al., 1994) (Fig. 3). In recent years, how these signalling pathways interact to pattern the embryo has begun to emerge. One clue appears to be their shared ability to regulate the expression of the caudal transcription factor (Cdx) genes (Bel-Vialar et al., 2002; Haremaki et al., 2003; Houle et al., 2000; Houle et al., 2003; Ikeya and Takada, 2001; Isaacs et al., 1998; Pilon et al., 2006; Pownall et al., 1996; Shiotsugu et al., 2004). However, there are important differences in how these pathways regulate the ParaHox and Hox gene clusters. For example, FGF signalling appears to preferentially regulate the 5′ (more-posterior) Hox genes, whereas RA preferentially regulates the 3′ (more-anterior) Hox genes (Bel-Vialar et al., 2002).
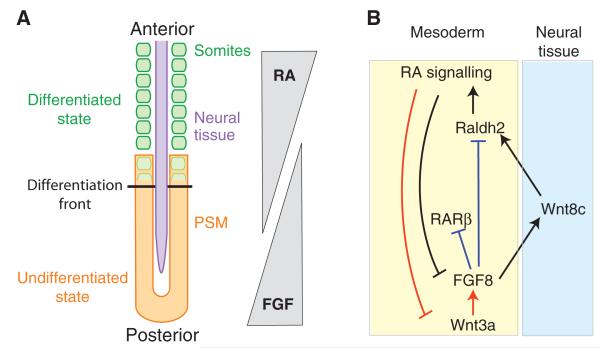
Fig. 3. FGF signalling during posterior body axis extension.
(A) A schematic of an extending body axis in mouse and chick embryos. In the extreme posterior, FGF signalling is high, maintaining the stem zone at the posterior end of the axis in an undifferentiated state. Retinoic acid (RA) promotes differentiation of neural ectoderm and somitic mesoderm. A gradient of FGF signalling is established, which is antagonised by an inverse gradient of RA signalling. The differentiation front is the position at which RA signalling wins over FGF signalling, resulting in the overt differentiation of neural ectoderm and somitic mesoderm, starting at the transition zone. Neural plate is in purple; undifferentiated presomitic mesoderm (PSM) in orange; differentiated somitic mesoderm in green; and somitic mesoderm in the process of differentiation is shown in overlapping orange and green. (B) These two inverse gradients of FGF and RA signalling are themselves established by Wnt signalling. Note that Wnt8c is induced in the neural plate, whereas FGF8 and Raldh2 are expressed in the mesoderm. Thus, these molecules signal across germ layers. Black lines depict interactions shown in both chick and mouse, red lines are interactions shown in mouse only, and blue are interactions shown in chick only. RARβ, retinoic acid receptor β; Raldh2, retinaldehyde dehydrogenase 2.
In general, FGF and RA primarily appear to interact antagonistically during posterior development (Diez del Corral et al., 2003) (Fig. 3). This is most clearly seen in chick and mouse embryos during posterior axial elongation, in which FGF signalling maintains the stem zone (see Glossary, Box 1), whereas RA promotes exit from the stem zone, thereby driving neural and somitic differentiation (Diez del Corral and Storey, 2004; Wilson et al., 2009). During this stage of development, the patterns of expression of Raldh2 (also known as Aldh1a2; which encodes retinaldehyde dehydrogenase 2, the enzyme that synthesizes RA) and Fgf8 are mutually exclusive (Diez del Corral and Storey, 2004; Wilson et al., 2009). In particular, Fgf8 is expressed in the stem zone, whereas Raldh2 is expressed in the presomitic mesoderm and somitic mesoderm, anterior to the differentiation front (see Fig. 3). In the chick, activation of RA signalling inhibits Fgf8 expression in the stem zone (Diez del Corral et al., 2003). Furthermore, FGF signalling inhibits the onset of Raldh2 expression in the paraxial mesoderm (Diez del Corral et al., 2003). Interestingly, FGF signalling modulates Raldh2 expression through Wnt8c (Olivera-Martinez and Storey, 2007). Furthermore, in the mouse, Wnt3a maintains the expression of Fgf8 in the stem zone (Aulehla et al., 2003). A similar signal relay, resulting in the establishment of inverse gradients of RA and FGF signalling in the posterior of the embryo, has also been reported in the mouse (Ribes et al., 2009; Zhao and Duester, 2009). An antagonistic relationship between RA and FGFs is also largely conserved in Xenopus, in which inhibition of RA signalling leads to an expansion in the fgf8 domain of expression (Shiotsugu et al., 2004) and activation of RA induces the expression of mkp3, a dual mitogen-activated protein kinase (MAPK) phosphatase and potent inhibitor of FGF signalling (Moreno and Kintner, 2004). Thus, the establishment of two inverted gradients of FGF activity (from posterior to anterior) and RA activity (from anterior to posterior) is a conserved mechanism that regulates the patterning and timing of differentiation in the posterior embryo during vertebrate body axis extension (Diez del Corral and Storey, 2004; Wilson et al., 2009) (Fig. 3).
The left-right axis
The left-right (L/R) axis is the third axis to be established in the embryo, and its specification is interlinked with the other two axes. Therefore, disruption of the DV or AP axis may disrupt the establishment of the L/R axis as well (Danos and Yost, 1995). The dorsal midline and notochord are also important for the specification of the embryonic L/R axis (Danos and Yost, 1996). Given that FGF signalling is essential for proper DV and AP axis specification, it is not surprising that disrupted FGF signalling also affects the L/R axis. In recent years, however, a more direct role for FGF signalling, particularly for FGF8, in L/R axis determination has emerged from studies in the mouse, chick, rabbit and zebrafish (Albertson and Yelick, 2005; Boettger et al., 1999; Fischer et al., 2002; Meyers and Martin, 1999). The specific role that FGF8 plays during this process appears to depend on the geometry of the embryo (Fischer et al., 2002). In the cylindrical-shaped mouse embryo, FGF8 induces on the left side the expression of Nodal, a TGFβ superfamily member and conserved signal in the determination of the L/R axis (Meyers and Martin, 1999), whereas in the disc-shaped chick and rabbit embryos (and presumably in human embryos, given that they are similarly shaped), FGF8 inhibits Nodal expression on the right-hand side (Boettger et al., 1999; Fischer et al., 2002).
In all vertebrates, the initial symmetry-breaking event that establishes the L/R axis is mediated by the extracellular flow of signals mediated by polarised monocilia in the node (Essner et al., 2002; Hamada et al., 2002). FGF signalling seems to play several roles at this early step of L/R axis determination. For example, FGF signalling is required for the release of vesicular nodal parcels (VNPs) into the node in the mouse (Tanaka et al., 2005). The VNPs contain cargos of Shh and RA, which are secreted and transported towards the left side of the node via the extracellular flow generated by the polarised monocilia (Tanaka et al., 2005). In zebrafish, FGF signalling also has a crucial role in the initial formation of the Kupffer’s vesicle (KV; see Glossary, Box 1) (Albertson and Yelick, 2005). In addition, in zebrafish and Xenopus, FGF signalling has a key role in the formation of the monocilia within the KV and gastrocoel roof plate (see Glossary, Box 1), respectively (Hong and Dawid, 2009; Neugebauer et al., 2009; Yamauchi et al., 2009). Indeed, Neugebauer and colleagues reported that FGF signalling plays a essential role in the regulation of cilia length in several epithelial structures in zebrafish and Xenopus embryos, including the inner ear, pronephros kidney and external mucociliary epidermis, suggesting that FGF signalling is generally required to control the length of cilia (Neugebauer et al., 2009). Furthermore, they and others showed that FGF signalling controls the length of cilia by regulating the expression of several genes responsible for ciliogenesis, including foxj1, rfx2, ift88, ier2 and fibp1 (Hong and Dawid, 2009; Neugebauer et al., 2009). Whether the role of FGF signalling in ciliogenesis is at least partly responsible for the defect in the release of VNPs in the mouse node is an intriguing question that remains to be addressed.
In summary, FGF signalling plays a crucial role in several steps during L/R axis determination, from the initial formation of the node, to the formation and function of nodal cilia, to the eventual induction or repression of Nodal expression on the right or left side, respectively.
FGF signalling in early mouse lineage specification
As discussed in the previous two sections, FGF signalling plays a crucial role in the specification of the mesoderm and neuroectoderm (see Box 2) and in patterning the three axes of the embryo. In early mammalian embryos, however, FGF signalling also plays an essential role during the first cell fate decisions, namely the specification of the trophectoderm and primitive endoderm (see Glossary, Box 1).
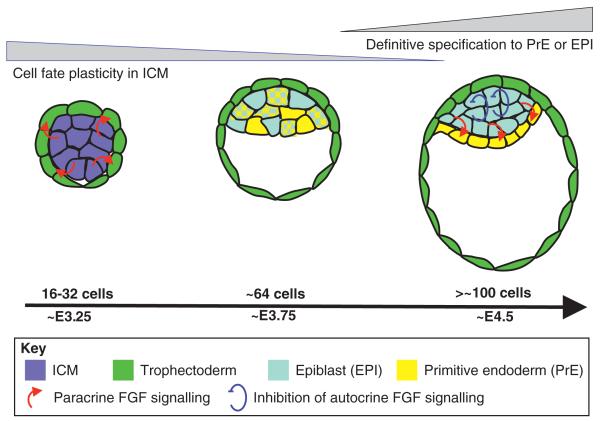
Early mammalian embryonic development is highly regulative (see Glossary, Box 1), and all cells are totipotent. Just before implantation, however, cells begin to restrict their developmental potential, such that the outer cells of the embryo, the trophectoderm, will give rise exclusively to the foetal contribution of the placenta, whereas the inner cell mass (ICM) (see Glossary, Box 1) will give rise to the primitive endoderm and the epiblast (see Glossary, Box 1) (reviewed by Yamanaka et al., 2006). FGF signalling plays an essential role in specification of two of the earliest lineages that are established in the mouse: the trophectoderm and primitive endoderm (Table 1; Fig. 4). Remarkably, it is possible to freeze development in the mouse at this point by deriving stem cells from the three earliest cell lineages and maintaining them long-term in culture (reviewed by Rossant, 2008) (Table 3). Indeed, FGF signalling plays an essential role in the maintenance and/or differentiation of trophectoderm stem (TS) cells. For example, TS cells can be derived and maintained long-term in culture by FGF4, and FGF4 removal from the cultured medium drives TS cells to terminally differentiate in vitro (Tanaka et al., 1998). TS cells give rise exclusively to trophectodermal lineages in chimaeras in vivo (Tanaka et al., 1998), consistent with data showing that FGF4 is required for the maintenance of trophectoderm in vivo (Goldin and Papaioannou, 2003) (Fig. 4). Fgf4 is expressed primarily in the ICM, whereas Fgfr2 is expressed primarily in the trophectoderm, which suggests that FGF4 acts as a paracrine maintenance factor for the trophectoderm in vivo (Arman et al., 1998; Goldin and Papaioannou, 2003; Rappolee et al., 1994). Strikingly, Fgf4−/− and Fgfr2−/− mouse embryos display very similar postimplantation lethal phenotypes, which further suggests that FGF4 acts through FGFR2 in the early mouse embryo (Arman et al., 1998; Feldman et al., 1995). Thus, FGF4 is essential for the maintenance of the first cell lineage specified in the mouse embryo – the trophectoderm.
Fig. 4. FGF signalling in early cell lineage specification in the mouse embryo.
Schematics of mouse embryos at embryonic day (E) 3.25, E3.75 and E4.5. FGF signalling specifies the two first lineages of the mammalian embryo: the trophectoderm, at around the 16- to 32-cell stage; and the primitive endoderm (PrE), at E3.5-4.5. This specification occurs primarily through paracrine induction from the inner cell mass (ICM) and epiblast (EPI), respectively. Meanwhile, FGF signalling must be inhibited within the epiblast to maintain its pluripotency. After implantation, FGF then promotes the overt differentiation of cell types within the epiblast.
Table 3. Origin and behaviour of mammalian embryo-derived stem cells.
| Stem cell line |
Origin | In vitro/teratoma potency |
Differentiation in chimaeras |
Ground state pluripotency |
Self-renewal conditions |
Differentiation conditions |
|---|---|---|---|---|---|---|
| Mouse ES cells |
Preimplantation epiblast |
Pluripotent | Pluripotent, including germline |
Yes | LIF + BMP4; no FGF/ERK, no GSK3 |
High FGF, activin, RA, Wnts, no LIF |
| XEN | Preimplantation primitive endoderm |
Primitive endoderm only |
Primitive endoderm only |
No | Not defined | Not defined |
| TS | Preimplantation trophoblast |
Trophoblast only |
Trophoblast only | No | FGF4 | No FGF4 |
| Mouse EpiSC |
Postimplantation epiblast |
Pluripotent | None | No | FGF2 + activin | BMP4 alone (extra- embryonic); FGF2 alone (neural); BMP4, FGF2 and activin (mesendoderm) |
| Rat ES cell |
Postimplantation epiblast |
Pluripotent | Pluripotent, including germline |
Yes | LIF + BMP4; no FGF/ERK, no GSK3 |
High FGF, activin, RA, Wnts, no LIF |
| Human ES cell |
Peri-implantation epiblast |
Pluripotent | Not known | Not known | FGF2 + activin | BMP4 alone (extra- embryonic); FGF2 alone (neural); BMP4, FGF2 and activin (mesendoderm) |
Data compiled from published sources (Buehr et al., 2008; Kunath et al., 2005; Li et al., 2008; Li et al., 2009; Nichols et al., 2009a; Rossant, 2008; Silva and Smith, 2008; Tanaka et al., 1998; Yamanaka et al., 2006; Ying et al., 2008).
BMP, bone morphogenetic protein; EpiSC, epiblast stem cell; ERK, extracellular signal-regulated kinase; ES, embryonic stem; FGF, fibroblast growth factor; GSK3, glycogen synthase kinase 3; LIF, leukemia inhibitory factor; RA, retinoic acid.
The second cell fate decision in the mouse ICM results in the specification of the epiblast lineage, which gives rise to the embryo proper, and the primitive endoderm lineage, which gives rise to the extra-embryonic endoderm and yolk sac (Yamanaka et al., 2006). It is possible to derive stem cells for both of these lineages (Rossant, 2008). Primitive endoderm stem cell lines, called XEN cells, have been derived from preimplantation mouse embryos using FGF4 in the culture medium, although FGF4 is not required for the long-term maintenance of XEN cells in vitro (Kunath et al., 2005). Importantly, the specification of the primitive endoderm lineage in the embryo requires the Grb2/Ras/ERK pathway, which acts downstream of FGF signalling (Chazaud et al., 2006; Yamanaka et al., 2010). ICM cells give rise to all the lineages of the embryo, including the germ cells, and thus the stem cells generated from them – mouse ES cells – are pluripotent (Bradley et al., 1984; Evans and Kaufman, 1981; Martin, 1981; Rossant, 2008). Unlike in TS and XEN cells, the FGF-mediated activation of Ras/ERK signalling in mouse ES cells promotes the transition from self-renewal to differentiation (Kunath et al., 2007; Rossant, 2008), possibly because pluripotency in the preimplantation epiblast requires inhibition of the FGF/ERK pathway (Lanner and Rossant, 2010; Nichols et al., 2009b). Indeed, a transient period of FGF/ERK activation has been shown to play a crucial role in driving the initial stages of differentiation in mouse ES cells (Stavridis et al., 2010; Stavridis et al., 2007). Interestingly, the epiblast (as do ES cells) expresses FGF4, which acts in a paracrine fashion to maintain trophectoderm and to specify primitive endoderm (Goldin and Papaioannou, 2003; Yamanaka et al., 2010). Within the epiblast, however, FGF4 acts in an autocrine fashion to drive cells away from pluripotency and toward cell fate specification, thus ensuring that embryogenesis progresses toward differentiation. Thus, to freeze ES cells in a pluripotent state, FGF/ERK signalling needs to be continually inhibited (Silva and Smith, 2008; Ying et al., 2008). Importantly, inhibition of the FGF/ERK pathway [in combination with inhibition of glycogen synthase kinase (GSK) 3] has facilitated the derivation and maintenance of pluripotent ES cells from recalcitrant mouse strains, from which the derivation of ES cells has previously proven to be difficult, and from rat embryos (Buehr et al., 2008; Li et al., 2008; Nichols et al., 2009a; Ying et al., 2008). A similar approach has also been used to drive induced pluripotent stem (iPS) cells from rat and human toward a more pluripotent state (Li et al., 2009).
In summary, the activation and/or inhibition of FGF/ERK signalling is crucial during the maintenance and/or differentiation of each of the first three lineages that are established in the preimplantation mouse embryo. This knowledge has facilitated the isolation of stem cell lines for each of the lineages, including pluripotent ES cells. Moreover, these findings are highly significant, as they might facilitate the isolation of pluripotent stem cells from other mammalian species, including humans.
Conclusions
Remarkably, FGF signalling plays an essential role in virtually every cell fate decision, patterning event and coordinated cell movement in the early embryo. What is not known is how FGF signalling can play such diverse roles. How do the cells interpret FGF signalling appropriately during each event? The answers to these questions are likely to include changes in the competence of the cells through time, the presence or absence of other synergistic or antagonist signals, and an intricate modulation of intracellular signalling pathways, through positive- or negative-feedback regulation. Given the complexity of the system, it will be essential to generate informative and testable models of the various roles that FGF plays during early development. Only then will the ultimate aim of gaining a complete understanding of the various roles that FGF signalling plays during early development become a reality.
Acknowledgements
This work was supported by a Wellcome Trust Programme Grant (E.A.) and an RCUK fellowship (K.D.). We thank Berenika Plusa for help with Figure 4 and helpful discussions on early mouse embryogenesis, and Nick Love and Yaoyao Chen for comments on the manuscript.
Footnotes
Competing interests statement The authors declare no competing financial interests.
References
- Albertson RC, Yelick PC. Roles for fgf8 signaling in left-right patterning of the visceral organs and craniofacial skeleton. Dev. Biol. 2005;283:310–321. doi: 10.1016/j.ydbio.2005.04.025. [DOI] [PubMed] [Google Scholar]
- Alvarez IS, Araujo M, Nieto MA. Neural induction in whole chick embryo cultures by FGF. Dev. Biol. 1998;199:42–54. doi: 10.1006/dbio.1998.8903. [DOI] [PubMed] [Google Scholar]
- Amaya E, Musci TJ, Kirschner MW. Expression of a dominant negative mutant of the FGF receptor disrupts mesoderm formation in Xenopus embryos. Cell. 1991;66:257–270. doi: 10.1016/0092-8674(91)90616-7. [DOI] [PubMed] [Google Scholar]
- Amaya E, Stein PA, Musci TJ, Kirschner MW. FGF signalling in the early specification of mesoderm in Xenopus. Development. 1993;118:477–487. doi: 10.1242/dev.118.2.477. [DOI] [PubMed] [Google Scholar]
- Arman E, Haffner-Krausz R, Chen Y, Heath JK, Lonai P. Targeted disruption of fibroblast growth factor (FGF) receptor 2 suggests a role for FGF signaling in pregastrulation mammalian development. Proc. Natl. Acad. Sci. USA. 1998;95:5082–5087. doi: 10.1073/pnas.95.9.5082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aulehla A, Wehrle C, Brand-Saberi B, Kemler R, Gossler A, Kanzler B, Herrmann BG. Wnt3a plays a major role in the segmentation clock controlling somitogenesis. Dev. Cell. 2003;4:395–406. doi: 10.1016/s1534-5807(03)00055-8. [DOI] [PubMed] [Google Scholar]
- Ayada T, Taniguchi K, Okamoto F, Kato R, Komune S, Takaesu G, Yoshimura A. Sprouty4 negatively regulates protein kinase C activation by inhibiting phosphatidylinositol 4,5-biphosphate hydrolysis. Oncogene. 2009;28:1076–1088. doi: 10.1038/onc.2008.464. [DOI] [PubMed] [Google Scholar]
- Bayha E, Jorgensen MC, Serup P, Grapin-Botton A. Retinoic acid signaling organizes endodermal organ specification along the entire anteroposterior axis. PLoS ONE. 2009;4:e5845. doi: 10.1371/journal.pone.0005845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beenken A, Mohammadi M. The FGF family: biology, pathophysiology and therapy. Nat. Rev. Drug Discov. 2009;8:235–253. doi: 10.1038/nrd2792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beiman M, Shilo BZ, Volk T. Heartless, a Drosophila FGF receptor homolog, is essential for cell migration and establishment of several mesodermal lineages. Genes Dev. 1996;10:2993–3002. doi: 10.1101/gad.10.23.2993. [DOI] [PubMed] [Google Scholar]
- Bel-Vialar S, Itasaki N, Krumlauf R. Initiating Hox gene expression: in the early chick neural tube differential sensitivity to FGF and RA signaling subdivides the HoxB genes in two distinct groups. Development. 2002;129:5103–5115. doi: 10.1242/dev.129.22.5103. [DOI] [PubMed] [Google Scholar]
- Bertrand V, Hudson C, Caillol D, Popovici C, Lemaire P. Neural tissue in ascidian embryos is induced by FGF9/16/20, acting via a combination of maternal GATA and Ets transcription factors. Cell. 2003;115:615–627. doi: 10.1016/s0092-8674(03)00928-0. [DOI] [PubMed] [Google Scholar]
- Blumberg B, Bolado J, Jr, Moreno TA, Kintner C, Evans RM, Papalopulu N. An essential role for retinoid signaling in anteroposterior neural patterning. Development. 1997;124:373–379. doi: 10.1242/dev.124.2.373. [DOI] [PubMed] [Google Scholar]
- Boettger T, Wittler L, Kessel M. FGF8 functions in the specification of the right body side of the chick. Curr. Biol. 1999;9:277–280. doi: 10.1016/s0960-9822(99)80119-5. [DOI] [PubMed] [Google Scholar]
- Bottcher RT, Niehrs C. Fibroblast growth factor signaling during early vertebrate development. Endocr. Rev. 2005;26:63–77. doi: 10.1210/er.2003-0040. [DOI] [PubMed] [Google Scholar]
- Bradley A, Evans M, Kaufman MH, Robertson E. Formation of germ-line chimaeras from embryo-derived teratocarcinoma cell lines. Nature. 1984;309:255–256. doi: 10.1038/309255a0. [DOI] [PubMed] [Google Scholar]
- Branney PA, Faas L, Steane SE, Pownall ME, Isaacs HV. Characterisation of the fibroblast growth factor dependent transcriptome in early development. PLoS ONE. 2009;4:e4951. doi: 10.1371/journal.pone.0004951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buehr M, Meek S, Blair K, Yang J, Ure J, Silva J, McLay R, Hall J, Ying QL, Smith A. Capture of authentic embryonic stem cells from rat blastocysts. Cell. 2008;135:1287–1298. doi: 10.1016/j.cell.2008.12.007. [DOI] [PubMed] [Google Scholar]
- Chazaud C, Yamanaka Y, Pawson T, Rossant J. Early lineage segregation between epiblast and primitive endoderm in mouse blastocysts through the Grb2-MAPK pathway. Dev. Cell. 2006;10:615–624. doi: 10.1016/j.devcel.2006.02.020. [DOI] [PubMed] [Google Scholar]
- Chen Y, Costa RM, Love NR, Soto X, Roth M, Paredes R, Amaya E. C/EBPalpha initiates primitive myelopoiesis in pluripotent embryonic cells. Blood. 2009;114:40–48. doi: 10.1182/blood-2008-11-189159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cho KW, De Robertis EM. Differential activation of Xenopus homeo box genes by mesoderm-inducing growth factors and retinoic acid. Genes Dev. 1990;4:1910–1916. doi: 10.1101/gad.4.11.1910. [DOI] [PubMed] [Google Scholar]
- Christen B, Slack JM. FGF-8 is associated with anteroposterior patterning and limb regeneration in Xenopus. Dev. Biol. 1997;192:455–466. doi: 10.1006/dbio.1997.8732. [DOI] [PubMed] [Google Scholar]
- Christen B, Slack JM. Spatial response to fibroblast growth factor signalling in Xenopus embryos. Development. 1999;126:119–125. doi: 10.1242/dev.126.1.119. [DOI] [PubMed] [Google Scholar]
- Ciruna BG, Schwartz L, Harpal K, Yamaguchi TP, Rossant J. Chimeric analysis of fibroblast growth factor receptor-1 (Fgfr1) function: a role for FGFR1 in morphogenetic movement through the primitive streak. Development. 1997;124:2829–2841. doi: 10.1242/dev.124.14.2829. [DOI] [PubMed] [Google Scholar]
- Corson LB, Yamanaka Y, Lai KM, Rossant J. Spatial and temporal patterns of ERK signaling during mouse embryogenesis. Development. 2003;130:4527–4537. doi: 10.1242/dev.00669. [DOI] [PubMed] [Google Scholar]
- Cox WG, Hemmati-Brivanlou A. Caudalization of neural fate by tissue recombination and bFGF. Development. 1995;121:4349–4358. doi: 10.1242/dev.121.12.4349. [DOI] [PubMed] [Google Scholar]
- Crossley PH, Martin GR. The mouse Fgf8 gene encodes a family of polypeptides and is expressed in regions that direct outgrowth and patterning in the developing embryo. Development. 1995;121:439–451. doi: 10.1242/dev.121.2.439. [DOI] [PubMed] [Google Scholar]
- Curran KL, Grainger RM. Expression of activated MAP kinase in Xenopus laevis embryos: evaluating the roles of FGF and other signaling pathways in early induction and patterning. Dev. Biol. 2000;228:41–56. doi: 10.1006/dbio.2000.9917. [DOI] [PubMed] [Google Scholar]
- Dailey L, Ambrosetti D, Mansukhani A, Basilico C. Mechanisms underlying differential responses to FGF signaling. Cytokine Growth Factor Rev. 2005;16:233–247. doi: 10.1016/j.cytogfr.2005.01.007. [DOI] [PubMed] [Google Scholar]
- Danos MC, Yost HJ. Linkage of cardiac left-right asymmetry and dorsal-anterior development in Xenopus. Development. 1995;121:1467–1474. doi: 10.1242/dev.121.5.1467. [DOI] [PubMed] [Google Scholar]
- Danos MC, Yost HJ. Role of notochord in specification of cardiac left-right orientation in zebrafish and Xenopus. Dev. Biol. 1996;177:96–103. doi: 10.1006/dbio.1996.0148. [DOI] [PubMed] [Google Scholar]
- Davidson BP, Cheng L, Kinder SJ, Tam PP. Exogenous FGF-4 can suppress anterior development in the mouse embryo during neurulation and early organogenesis. Dev. Biol. 2000;221:41–52. doi: 10.1006/dbio.2000.9663. [DOI] [PubMed] [Google Scholar]
- De Robertis EM. Spemann’s organizer and the self-regulation of embryonic fields. Mech. Dev. 2009;126:925–941. doi: 10.1016/j.mod.2009.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Robertis EM, Kuroda H. Dorsal-ventral patterning and neural induction in Xenopus embryos. Annu. Rev. Cell Dev. Biol. 2004;20:285–308. doi: 10.1146/annurev.cellbio.20.011403.154124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delaune E, Lemaire P, Kodjabachian L. Neural induction in Xenopus requires early FGF signalling in addition to BMP inhibition. Development. 2005;132:299–310. doi: 10.1242/dev.01582. [DOI] [PubMed] [Google Scholar]
- Deng CX, Wynshaw-Boris A, Shen MM, Daugherty C, Ornitz DM, Leder P. Murine FGFR-1 is required for early postimplantation growth and axial organization. Genes Dev. 1994;8:3045–3057. doi: 10.1101/gad.8.24.3045. [DOI] [PubMed] [Google Scholar]
- Dessimoz J, Opoka R, Kordich JJ, Grapin-Botton A, Wells JM. FGF signaling is necessary for establishing gut tube domains along the anterior-posterior axis in vivo. Mech. Dev. 2006;123:42–55. doi: 10.1016/j.mod.2005.10.001. [DOI] [PubMed] [Google Scholar]
- Diez del Corral R, Storey KG. Opposing FGF and retinoid pathways: a signalling switch that controls differentiation and patterning onset in the extending vertebrate body axis. BioEssays. 2004;26:857–869. doi: 10.1002/bies.20080. [DOI] [PubMed] [Google Scholar]
- Diez del Corral R, Olivera-Martinez I, Goriely A, Gale E, Maden M, Storey K. Opposing FGF and retinoid pathways control ventral neural pattern, neuronal differentiation, and segmentation during body axis extension. Neuron. 2003;40:65–79. doi: 10.1016/s0896-6273(03)00565-8. [DOI] [PubMed] [Google Scholar]
- Doniach T. Basic FGF as an inducer of anteroposterior neural pattern. Cell. 1995;83:1067–1070. doi: 10.1016/0092-8674(95)90133-7. [DOI] [PubMed] [Google Scholar]
- Draper BW, Stock DW, Kimmel CB. Zebrafish fgf24 functions with fgf8 to promote posterior mesodermal development. Development. 2003;130:4639–4654. doi: 10.1242/dev.00671. [DOI] [PubMed] [Google Scholar]
- Dubrulle J, Pourquie O. fgf8 mRNA decay establishes a gradient that couples axial elongation to patterning in the vertebrate embryo. Nature. 2004;427:419–422. doi: 10.1038/nature02216. [DOI] [PubMed] [Google Scholar]
- Durston AJ, Timmermans JP, Hage WJ, Hendriks HF, de Vries NJ, Heideveld M, Nieuwkoop PD. Retinoic acid causes an anteroposterior transformation in the developing central nervous system. Nature. 1989;340:140–144. doi: 10.1038/340140a0. [DOI] [PubMed] [Google Scholar]
- Eivers E, Fuentealba LC, De Robertis EM. Integrating positional information at the level of Smad1/5/8. Curr. Opin. Genet. Dev. 2008;18:304–310. doi: 10.1016/j.gde.2008.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Essner JJ, Vogan KJ, Wagner MK, Tabin CJ, Yost HJ, Brueckner M. Conserved function for embryonic nodal cilia. Nature. 2002;418:37–38. doi: 10.1038/418037a. [DOI] [PubMed] [Google Scholar]
- Evans MJ, Kaufman MH. Establishment in culture of pluripotential cells from mouse embryos. Nature. 1981;292:154–156. doi: 10.1038/292154a0. [DOI] [PubMed] [Google Scholar]
- Feldman B, Poueymirou W, Papaioannou VE, DeChiara TM, Goldfarb M. Requirement of FGF-4 for postimplantation mouse development. Science. 1995;267:246–249. doi: 10.1126/science.7809630. [DOI] [PubMed] [Google Scholar]
- Fischer A, Viebahn C, Blum M. FGF8 acts as a right determinant during establishment of the left-right axis in the rabbit. Curr. Biol. 2002;12:1807–1816. doi: 10.1016/s0960-9822(02)01222-8. [DOI] [PubMed] [Google Scholar]
- Fisher ME, Isaacs HV, Pownall ME. eFGF is required for activation of XmyoD expression in the myogenic cell lineage of Xenopus laevis. Development. 2002;129:1307–1315. doi: 10.1242/dev.129.6.1307. [DOI] [PubMed] [Google Scholar]
- Fletcher RB, Harland RM. The role of FGF signaling in the establishment and maintenance of mesodermal gene expression in Xenopus. Dev. Dyn. 2008;237:1243–1254. doi: 10.1002/dvdy.21517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fletcher RB, Baker JC, Harland RM. FGF8 spliceforms mediate early mesoderm and posterior neural tissue formation in Xenopus. Development. 2006;133:1703–1714. doi: 10.1242/dev.02342. [DOI] [PubMed] [Google Scholar]
- Furthauer M, Thisse C, Thisse B. A role for FGF-8 in the dorsoventral patterning of the zebrafish gastrula. Development. 1997;124:4253–4264. doi: 10.1242/dev.124.21.4253. [DOI] [PubMed] [Google Scholar]
- Furthauer M, Van Celst J, Thisse C, Thisse B. Fgf signalling controls the dorsoventral patterning of the zebrafish embryo. Development. 2004;131:2853–2864. doi: 10.1242/dev.01156. [DOI] [PubMed] [Google Scholar]
- Gisselbrecht S, Skeath JB, Doe CQ, Michelson AM. heartless encodes a fibroblast growth factor receptor (DFR1/DFGF-R2) involved in the directional migration of early mesodermal cells in the Drosophila embryo. Genes Dev. 1996;10:3003–3017. doi: 10.1101/gad.10.23.3003. [DOI] [PubMed] [Google Scholar]
- Goldin SN, Papaioannou VE. Paracrine action of FGF4 during periimplantation development maintains trophectoderm and primitive endoderm. Genesis. 2003;36:40–47. doi: 10.1002/gene.10192. [DOI] [PubMed] [Google Scholar]
- Gospodarowicz D, Moran JS. Mitogenic effect of fibroblast growth factor on early passage cultures of human and murine fibroblasts. J. Cell Biol. 1975;66:451–457. doi: 10.1083/jcb.66.2.451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffin K, Patient R, Holder N. Analysis of FGF function in normal and no tail zebrafish embryos reveals separate mechanisms for formation of the trunk and the tail. Development. 1995;121:2983–2994. doi: 10.1242/dev.121.9.2983. [DOI] [PubMed] [Google Scholar]
- Gryzik T, Muller HA. FGF8-like1 and FGF8-like2 encode putative ligands of the FGF receptor Htl and are required for mesoderm migration in the Drosophila gastrula. Curr. Biol. 2004;14:659–667. doi: 10.1016/j.cub.2004.03.058. [DOI] [PubMed] [Google Scholar]
- Hamada H, Meno C, Watanabe D, Saijoh Y. Establishment of vertebrate left-right asymmetry. Nat. Rev. Genet. 2002;3:103–113. doi: 10.1038/nrg732. [DOI] [PubMed] [Google Scholar]
- Hamburger V. The Heritage of Experimental Embryology: Hans Spemann and the Organizer. Oxford University Press; New York: 1988. [Google Scholar]
- Hardcastle Z, Chalmers AD, Papalopulu N. FGF-8 stimulates neuronal differentiation through FGFR-4a and interferes with mesoderm induction in Xenopus embryos. Curr. Biol. 2000;10:1511–1514. doi: 10.1016/s0960-9822(00)00825-3. [DOI] [PubMed] [Google Scholar]
- Haremaki T, Tanaka Y, Hongo I, Yuge M, Okamoto H. Integration of multiple signal transducing pathways on Fgf response elements of the Xenopus caudal homologue Xcad3. Development. 2003;130:4907–4917. doi: 10.1242/dev.00718. [DOI] [PubMed] [Google Scholar]
- Heisenberg CP, Tada M, Rauch GJ, Saude L, Concha ML, Geisler R, Stemple DL, Smith JC, Wilson SW. Silberblick/Wnt11 mediates convergent extension movements during zebrafish gastrulation. Nature. 2000;405:76–81. doi: 10.1038/35011068. [DOI] [PubMed] [Google Scholar]
- Holowacz T, Sokol S. FGF is required for posterior neural patterning but not for neural induction. Dev. Biol. 1999;205:296–308. doi: 10.1006/dbio.1998.9108. [DOI] [PubMed] [Google Scholar]
- Hong SK, Dawid IB. FGF-dependent left-right asymmetry patterning in zebrafish is mediated by Ier2 and Fibp1. Proc. Natl. Acad. Sci. USA. 2009;106:2230–2235. doi: 10.1073/pnas.0812880106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hongo I, Kengaku M, Okamoto H. FGF signaling and the anterior neural induction in Xenopus. Dev. Biol. 1999;216:561–581. doi: 10.1006/dbio.1999.9515. [DOI] [PubMed] [Google Scholar]
- Houle M, Prinos P, Iulianella A, Bouchard N, Lohnes D. Retinoic acid regulation of Cdx1: an indirect mechanism for retinoids and vertebral specification. Mol. Cell. Biol. 2000;20:6579–6586. doi: 10.1128/mcb.20.17.6579-6586.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Houle M, Sylvestre JR, Lohnes D. Retinoic acid regulates a subset of Cdx1 function in vivo. Development. 2003;130:6555–6567. doi: 10.1242/dev.00889. [DOI] [PubMed] [Google Scholar]
- Hudson C, Lemaire P. Induction of anterior neural fates in the ascidian Ciona intestinalis. Mech. Dev. 2001;100:189–203. doi: 10.1016/s0925-4773(00)00528-1. [DOI] [PubMed] [Google Scholar]
- Ikeya M, Takada S. Wnt-3a is required for somite specification along the anteroposterior axis of the mouse embryo and for regulation of cdx-1 expression. Mech. Dev. 2001;103:27–33. doi: 10.1016/s0925-4773(01)00338-0. [DOI] [PubMed] [Google Scholar]
- Imai KS, Satoh N, Satou Y. Early embryonic expression of FGF4/6/9 gene and its role in the induction of mesenchyme and notochord in Ciona savignyi embryos. Development. 2002;129:1729–1738. doi: 10.1242/dev.129.7.1729. [DOI] [PubMed] [Google Scholar]
- Inazawa T, Okamura Y, Takahashi K. Basic fibroblast growth factor induction of neuronal ion channel expression in ascidian ectodermal blastomeres. J. Physiol. 1998;511:347–359. doi: 10.1111/j.1469-7793.1998.347bh.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Isaacs HV, Tannahill D, Slack JM. Expression of a novel FGF in the Xenopus embryo. A new candidate inducing factor for mesoderm formation and anteroposterior specification. Development. 1992;114:711–720. doi: 10.1242/dev.114.3.711. [DOI] [PubMed] [Google Scholar]
- Isaacs HV, Pownall ME, Slack JM. eFGF regulates Xbra expression during Xenopus gastrulation. EMBO J. 1994;13:4469–4481. doi: 10.1002/j.1460-2075.1994.tb06769.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Isaacs HV, Pownall ME, Slack JM. eFGF is expressed in the dorsal midline of Xenopus laevis. Int. J. Dev. Biol. 1995;39:575–579. [PubMed] [Google Scholar]
- Isaacs HV, Pownall ME, Slack JM. Regulation of Hox gene expression and posterior development by the Xenopus caudal homologue Xcad3. EMBO J. 1998;17:3413–3427. doi: 10.1093/emboj/17.12.3413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Isaacs HV, Deconinck AE, Pownall ME. FGF4 regulates blood and muscle specification in Xenopus laevis. Biol. Cell. 2007;99:165–173. doi: 10.1042/BC20060103. [DOI] [PubMed] [Google Scholar]
- Itoh N. The Fgf families in humans, mice, and zebrafish: their evolutional processes and roles in development, metabolism, and disease. Biol. Pharm. Bull. 2007;30:1819–1825. doi: 10.1248/bpb.30.1819. [DOI] [PubMed] [Google Scholar]
- Itoh N, Ornitz DM. Functional evolutionary history of the mouse Fgf gene family. Dev. Dyn. 2008;237:18–27. doi: 10.1002/dvdy.21388. [DOI] [PubMed] [Google Scholar]
- Kadam S, McMahon A, Tzou P, Stathopoulos A. FGF ligands in Drosophila have distinct activities required to support cell migration and differentiation. Development. 2009;136:739–747. doi: 10.1242/dev.027904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keenan ID, Sharrard RM, Isaacs HV. FGF signal transduction and the regulation of Cdx gene expression. Dev. Biol. 2006;299:478–488. doi: 10.1016/j.ydbio.2006.08.040. [DOI] [PubMed] [Google Scholar]
- Kengaku M, Okamoto H. bFGF as a possible morphogen for the anteroposterior axis of the central nervous system in Xenopus. Development. 1995;121:3121–3130. doi: 10.1242/dev.121.9.3121. [DOI] [PubMed] [Google Scholar]
- Kiecker C, Niehrs C. A morphogen gradient of Wnt/beta-catenin signalling regulates anteroposterior neural patterning in Xenopus. Development. 2001;128:4189–4201. doi: 10.1242/dev.128.21.4189. [DOI] [PubMed] [Google Scholar]
- Kimelman D. Mesoderm induction: from caps to chips. Nat. Rev. Genet. 2006;7:360–372. doi: 10.1038/nrg1837. [DOI] [PubMed] [Google Scholar]
- Kimelman D, Kirschner M. Synergistic induction of mesoderm by FGF and TGF-beta and the identification of an mRNA coding for FGF in the early Xenopus embryo. Cell. 1987;51:869–877. doi: 10.1016/0092-8674(87)90110-3. [DOI] [PubMed] [Google Scholar]
- Kinoshita N, Iioka H, Miyakoshi A, Ueno N. PKC delta is essential for Dishevelled function in a noncanonical Wnt pathway that regulates Xenopus convergent extension movements. Genes Dev. 2003;17:1663–1676. doi: 10.1101/gad.1101303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klingseisen A, Clark IB, Gryzik T, Muller HA. Differential and overlapping functions of two closely related Drosophila FGF8-like growth factors in mesoderm development. Development. 2009;136:2393–2402. doi: 10.1242/dev.035451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koshida S, Shinya M, Nikaido M, Ueno N, Schulte-Merker S, Kuroiwa A, Takeda H. Inhibition of BMP activity by the FGF signal promotes posterior neural development in zebrafish. Dev. Biol. 2002;244:9–20. doi: 10.1006/dbio.2002.0581. [DOI] [PubMed] [Google Scholar]
- Krejci P, Prochazkova J, Bryja V, Kozubik A, Wilcox WR. Molecular pathology of the fibroblast growth factor family. Hum. Mutat. 2009;30:1245–1255. doi: 10.1002/humu.21067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kroll KL, Amaya E. Transgenic Xenopus embryos from sperm nuclear transplantations reveal FGF signaling requirements during gastrulation. Development. 1996;122:3173–3183. doi: 10.1242/dev.122.10.3173. [DOI] [PubMed] [Google Scholar]
- Kudoh T, Wilson SW, Dawid IB. Distinct roles for Fgf, Wnt and retinoic acid in posteriorizing the neural ectoderm. Development. 2002;129:4335–4346. doi: 10.1242/dev.129.18.4335. [DOI] [PubMed] [Google Scholar]
- Kudoh T, Concha ML, Houart C, Dawid IB, Wilson SW. Combinatorial Fgf and Bmp signalling patterns the gastrula ectoderm into prospective neural and epidermal domains. Development. 2004;131:3581–3592. doi: 10.1242/dev.01227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumano G, Smith WC. FGF signaling restricts the primary blood islands to ventral mesoderm. Dev. Biol. 2000;228:304–314. doi: 10.1006/dbio.2000.9937. [DOI] [PubMed] [Google Scholar]
- Kumano G, Smith WC. Revisions to the Xenopus gastrula fate map: implications for mesoderm induction and patterning. Dev. Dyn. 2002a;225:409–421. doi: 10.1002/dvdy.10177. [DOI] [PubMed] [Google Scholar]
- Kumano G, Smith WC. The nodal target gene Xmenf is a component of an FGF-independent pathway of ventral mesoderm induction in Xenopus. Mech. Dev. 2002b;118:45–56. doi: 10.1016/s0925-4773(02)00186-7. [DOI] [PubMed] [Google Scholar]
- Kumano G, Ezal C, Smith WC. Boundaries and functional domains in the animal/vegetal axis of Xenopus gastrula mesoderm. Dev. Biol. 2001;236:465–477. doi: 10.1006/dbio.2001.0341. [DOI] [PubMed] [Google Scholar]
- Kunath T, Arnaud D, Uy GD, Okamoto I, Chureau C, Yamanaka Y, Heard E, Gardner RL, Avner P, Rossant J. Imprinted X-inactivation in extra-embryonic endoderm cell lines from mouse blastocysts. Development. 2005;132:1649–1661. doi: 10.1242/dev.01715. [DOI] [PubMed] [Google Scholar]
- Kunath T, Saba-El-Leil MK, Almousailleakh M, Wray J, Meloche S, Smith A. FGF stimulation of the Erk1/2 signalling cascade triggers transition of pluripotent embryonic stem cells from self-renewal to lineage commitment. Development. 2007;134:2895–2902. doi: 10.1242/dev.02880. [DOI] [PubMed] [Google Scholar]
- Lamb TM, Harland RM. Fibroblast growth factor is a direct neural inducer, which combined with noggin generates anterior-posterior neural pattern. Development. 1995;121:3627–3636. doi: 10.1242/dev.121.11.3627. [DOI] [PubMed] [Google Scholar]
- Lane MC, Smith WC. The origins of primitive blood in Xenopus: implications for axial patterning. Development. 1999;126:423–434. doi: 10.1242/dev.126.3.423. [DOI] [PubMed] [Google Scholar]
- Lane MC, Sheets MD. Primitive and definitive blood share a common origin in Xenopus: a comparison of lineage techniques used to construct fate maps. Dev. Biol. 2002;248:52–67. doi: 10.1006/dbio.2002.0717. [DOI] [PubMed] [Google Scholar]
- Lanner F, Rossant J. The role of FGF/Erk signaling in pluripotent cells. Development. 2010;137:3351–3360. doi: 10.1242/dev.050146. [DOI] [PubMed] [Google Scholar]
- Launay C, Fromentoux V, Shi DL, Boucaut JC. A truncated FGF receptor blocks neural induction by endogenous Xenopus inducers. Development. 1996;122:869–880. doi: 10.1242/dev.122.3.869. [DOI] [PubMed] [Google Scholar]
- Lea R, Papalopulu N, Amaya E, Dorey K. Temporal and spatial expression of FGF ligands and receptors during Xenopus development. Dev. Dyn. 2009;238:1467–1479. doi: 10.1002/dvdy.21913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leslie NR, Yang X, Downes CP, Weijer CJ. PtdIns(3,4,5)P(3)-dependent and -independent roles for PTEN in the control of cell migration. Curr. Biol. 2007;17:115–125. doi: 10.1016/j.cub.2006.12.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levine AJ, Brivanlou AH. Proposal of a model of mammalian neural induction. Dev. Biol. 2007;308:247–256. doi: 10.1016/j.ydbio.2007.05.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li P, Tong C, Mehrian-Shai R, Jia L, Wu N, Yan Y, Maxson RE, Schulze EN, Song H, Hsieh CL, et al. Germline competent embryonic stem cells derived from rat blastocysts. Cell. 2008;135:1299–1310. doi: 10.1016/j.cell.2008.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li W, Wei W, Zhu S, Zhu J, Shi Y, Lin T, Hao E, Hayek A, Deng H, Ding S. Generation of rat and human induced pluripotent stem cells by combining genetic reprogramming and chemical inhibitors. Cell Stem Cell. 2009;4:16–19. doi: 10.1016/j.stem.2008.11.014. [DOI] [PubMed] [Google Scholar]
- Lieschke GJ, Oates AC, Paw BH, Thompson MA, Hall NE, Ward AC, Ho RK, Zon LI, Layton JE. Zebrafish SPI-1 (PU.1) marks a site of myeloid development independent of primitive erythropoiesis: implications for axial patterning. Dev. Biol. 2002;246:274–295. doi: 10.1006/dbio.2002.0657. [DOI] [PubMed] [Google Scholar]
- Linker C, Stern CD. Neural induction requires BMP inhibition only as a late step, and involves signals other than FGF and Wnt antagonists. Development. 2004;131:5671–5681. doi: 10.1242/dev.01445. [DOI] [PubMed] [Google Scholar]
- Linker C, De Almeida I, Papanayotou C, Stower M, Sabado V, Ghorani E, Streit A, Mayor R, Stern CD. Cell communication with the neural plate is required for induction of neural markers by BMP inhibition: evidence for homeogenetic induction and implications for Xenopus animal cap and chick explant assays. Dev. Biol. 2009;327:478–486. doi: 10.1016/j.ydbio.2008.12.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Little SC, Mullins MC. Extracellular modulation of BMP activity in patterning the dorsoventral axis. Birth Defects Res. C Embryo Today. 2006;78:224–242. doi: 10.1002/bdrc.20079. [DOI] [PubMed] [Google Scholar]
- Mariani FV, Ahn CP, Martin GR. Genetic evidence that FGFs have an instructive role in limb proximal-distal patterning. Nature. 2008;453:401–405. doi: 10.1038/nature06876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin GR. Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc. Natl. Acad. Sci. USA. 1981;78:7634–7638. doi: 10.1073/pnas.78.12.7634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mathis L, Kulesa PM, Fraser SE. FGF receptor signalling is required to maintain neural progenitors during Hensen’s node progression. Nat. Cell Biol. 2001;3:559–566. doi: 10.1038/35078535. [DOI] [PubMed] [Google Scholar]
- McGrew LL, Lai CJ, Moon RT. Specification of the anteroposterior neural axis through synergistic interaction of the Wnt signaling cascade with noggin and follistatin. Dev. Biol. 1995;172:337–342. doi: 10.1006/dbio.1995.0027. [DOI] [PubMed] [Google Scholar]
- McGrew LL, Hoppler S, Moon RT. Wnt and FGF pathways cooperatively pattern anteroposterior neural ectoderm in Xenopus. Mech. Dev. 1997;69:105–114. doi: 10.1016/s0925-4773(97)00160-3. [DOI] [PubMed] [Google Scholar]
- McIntosh I, Bellus GA, Jab EW. The pleiotropic effects of fibroblast growth factor receptors in mammalian development. Cell Struct. Funct. 2000;25:85–96. doi: 10.1247/csf.25.85. [DOI] [PubMed] [Google Scholar]
- Meyers EN, Martin GR. Differences in left-right axis pathways in mouse and chick: functions of FGF8 and SHH. Science. 1999;285:403–406. doi: 10.1126/science.285.5426.403. [DOI] [PubMed] [Google Scholar]
- Meyers EN, Lewandoski M, Martin GR. An Fgf8 mutant allelic series generated by Cre- and Flp-mediated recombination. Nat. Genet. 1998;18:136–141. doi: 10.1038/ng0298-136. [DOI] [PubMed] [Google Scholar]
- Minina E, Kreschel C, Naski MC, Ornitz DM, Vortkamp A. Interaction of FGF, Ihh/Pthlh, and BMP signaling integrates chondrocyte proliferation and hypertrophic differentiation. Dev. Cell. 2002;3:439–449. doi: 10.1016/s1534-5807(02)00261-7. [DOI] [PubMed] [Google Scholar]
- Mohammadi M, Olsen SK, Ibrahimi OA. Structural basis for fibroblast growth factor receptor activation. Cytokine Growth Factor Rev. 2005;16:107–137. doi: 10.1016/j.cytogfr.2005.01.008. [DOI] [PubMed] [Google Scholar]
- Moreno TA, Kintner C. Regulation of segmental patterning by retinoic acid signaling during Xenopus somitogenesis. Dev. Cell. 2004;6:205–218. doi: 10.1016/s1534-5807(04)00026-7. [DOI] [PubMed] [Google Scholar]
- Neugebauer JM, Amack JD, Peterson AG, Bisgrove BW, Yost HJ. FGF signalling during embryo development regulates cilia length in diverse epithelia. Nature. 2009;458:651–654. doi: 10.1038/nature07753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nichols J, Jones K, Phillips JM, Newland SA, Roode M, Mansfield W, Smith A, Cooke A. Validated germline-competent embryonic stem cell lines from nonobese diabetic mice. Nat. Med. 2009a;15:814–818. doi: 10.1038/nm.1996. [DOI] [PubMed] [Google Scholar]
- Nichols J, Silva J, Roode M, Smith A. Suppression of Erk signalling promotes ground state pluripotency in the mouse embryo. Development. 2009b;136:3215–3222. doi: 10.1242/dev.038893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nieuwkoop PD. Activation and organization of the central nervous system in amphibians. Part III. Synthesis of a new working hypothesis. J. Exp. Zool. 1952;120:83–108. [Google Scholar]
- Niswander L, Martin GR. FGF-4 and BMP-2 have opposite effects on limb growth. Nature. 1993;361:68–71. doi: 10.1038/361068a0. [DOI] [PubMed] [Google Scholar]
- Northrop JL, Kimelman D. Dorsal-ventral differences in Xcad-3 expression in response to FGF-mediated induction in Xenopus. Dev. Biol. 1994;161:490–503. doi: 10.1006/dbio.1994.1047. [DOI] [PubMed] [Google Scholar]
- Nutt SL, Dingwell KS, Holt CE, Amaya E. Xenopus Sprouty2 inhibits FGF-mediated gastrulation movements but does not affect mesoderm induction and patterning. Genes Dev. 2001;15:1152–1166. doi: 10.1101/gad.191301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohuchi H, Yoshioka H, Tanaka A, Kawakami Y, Nohno T, Noji S. Involvement of androgen-induced growth factor (FGF-8) gene in mouse embryogenesis and morphogenesis. Biochem. Biophys. Res. Commun. 1994;204:882–888. doi: 10.1006/bbrc.1994.2542. [DOI] [PubMed] [Google Scholar]
- Olivera-Martinez I, Storey KG. Wnt signals provide a timing mechanism for the FGF-retinoid differentiation switch during vertebrate body axis extension. Development. 2007;134:2125–2135. doi: 10.1242/dev.000216. [DOI] [PubMed] [Google Scholar]
- Ornitz DM. FGFs, heparan sulfate and FGFRs: complex interactions essential for development. BioEssays. 2000;22:108–112. doi: 10.1002/(SICI)1521-1878(200002)22:2<108::AID-BIES2>3.0.CO;2-M. [DOI] [PubMed] [Google Scholar]
- Ota S, Tonou-Fujimori N, Yamasu K. The roles of the FGF signal in zebrafish embryos analyzed using constitutive activation and dominant-negative suppression of different FGF receptors. Mech. Dev. 2009;126:1–17. doi: 10.1016/j.mod.2008.10.008. [DOI] [PubMed] [Google Scholar]
- Partanen J, Schwartz L, Rossant J. Opposite phenotypes of hypomorphic and Y766 phosphorylation site mutations reveal a function for Fgfr1 in anteroposterior patterning of mouse embryos. Genes Dev. 1998;12:2332–2344. doi: 10.1101/gad.12.15.2332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pera EM, Ikeda A, Eivers E, De Robertis EM. Integration of IGF, FGF, and anti-BMP signals via Smad1 phosphorylation in neural induction. Genes Dev. 2003;17:3023–3028. doi: 10.1101/gad.1153603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pilon N, Oh K, Sylvestre JR, Bouchard N, Savory J, Lohnes D. Cdx4 is a direct target of the canonical Wnt pathway. Dev. Biol. 2006;289:55–63. doi: 10.1016/j.ydbio.2005.10.005. [DOI] [PubMed] [Google Scholar]
- Pownall ME, Tucker AS, Slack JM, Isaacs HV. eFGF, Xcad3 and Hox genes form a molecular pathway that establishes the anteroposterior axis in Xenopus. Development. 1996;122:3881–3892. doi: 10.1242/dev.122.12.3881. [DOI] [PubMed] [Google Scholar]
- Rappolee DA, Basilico C, Patel Y, Werb Z. Expression and function of FGF-4 in peri-implantation development in mouse embryos. Development. 1994;120:2259–2269. doi: 10.1242/dev.120.8.2259. [DOI] [PubMed] [Google Scholar]
- Reifers F, Bohli H, Walsh EC, Crossley PH, Stainier DY, Brand M. Fgf8 is mutated in zebrafish acerebellar (ace) mutants and is required for maintenance of midbrain-hindbrain boundary development and somitogenesis. Development. 1998;125:2381–2395. doi: 10.1242/dev.125.13.2381. [DOI] [PubMed] [Google Scholar]
- Rentzsch F, Bakkers J, Kramer C, Hammerschmidt M. Fgf signaling induces posterior neuroectoderm independently of Bmp signaling inhibition. Dev. Dyn. 2004;231:750–757. doi: 10.1002/dvdy.20244. [DOI] [PubMed] [Google Scholar]
- Ribes V, Le Roux I, Rhinn M, Schuhbaur B, Dolle P. Early mouse caudal development relies on crosstalk between retinoic acid, Shh and Fgf signalling pathways. Development. 2009;136:665–676. doi: 10.1242/dev.016204. [DOI] [PubMed] [Google Scholar]
- Ribisi S, Jr, Mariani FV, Aamar E, Lamb TM, Frank D, Harland RM. Ras-mediated FGF signaling is required for the formation of posterior but not anterior neural tissue in Xenopus laevis. Dev. Biol. 2000;227:183–196. doi: 10.1006/dbio.2000.9889. [DOI] [PubMed] [Google Scholar]
- Rodriguez-Gallardo L, Climent V, Garcia-Martinez V, Schoenwolf GC, Alvarez IS. Targeted over-expression of FGF in chick embryos induces formation of ectopic neural cells. Int. J. Dev. Biol. 1997;41:715–723. [PubMed] [Google Scholar]
- Rossant J. Stem cells and early lineage development. Cell. 2008;132:527–531. doi: 10.1016/j.cell.2008.01.039. [DOI] [PubMed] [Google Scholar]
- Rottinger E, Saudemont A, Duboc V, Besnardeau L, McClay D, Lepage T. FGF signals guide migration of mesenchymal cells, control skeletal morphogenesis of the skeleton and regulate gastrulation during sea urchin development. Development. 2007;135:353–365. doi: 10.1242/dev.014282. [DOI] [PubMed] [Google Scholar]
- Sasai Y, Lu B, Piccolo S, De Robertis EM. Endoderm induction by the organizer-secreted factors chordin and noggin in Xenopus animal caps. EMBO J. 1996;15:4547–4555. [PMC free article] [PubMed] [Google Scholar]
- Saxen L, Toivonen S. The two-gradient hypothesis in primary induction. The combined effect of two types of inductors mixed in different ratios. J. Embryol. Exp. Morphol. 1961;9:514–533. [PubMed] [Google Scholar]
- Schier AF, Talbot WS. Molecular genetics of axis formation in zebrafish. Annu. Rev. Genet. 2005;39:561–613. doi: 10.1146/annurev.genet.37.110801.143752. [DOI] [PubMed] [Google Scholar]
- Schlessinger J. Cell signaling by receptor tyrosine kinases. Cell. 2000;103:211–225. doi: 10.1016/s0092-8674(00)00114-8. [DOI] [PubMed] [Google Scholar]
- Shamim H, Mason I. Expression of Fgf4 during early development of the chick embryo. Mech. Dev. 1999;85:189–192. doi: 10.1016/s0925-4773(99)00093-3. [DOI] [PubMed] [Google Scholar]
- Sheng G, dos Reis M, Stern CD. Churchill, a zinc finger transcriptional activator, regulates the transition between gastrulation and neurulation. Cell. 2003;115:603–613. doi: 10.1016/s0092-8674(03)00927-9. [DOI] [PubMed] [Google Scholar]
- Shiotsugu J, Katsuyama Y, Arima K, Baxter A, Koide T, Song J, Chandraratna RA, Blumberg B. Multiple points of interaction between retinoic acid and FGF signaling during embryonic axis formation. Development. 2004;131:2653–2667. doi: 10.1242/dev.01129. [DOI] [PubMed] [Google Scholar]
- Silva J, Smith A. Capturing pluripotency. Cell. 2008;132:532–536. doi: 10.1016/j.cell.2008.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sivak JM, Petersen LF, Amaya E. FGF signal interpretation is directed by Sprouty and Spred proteins during mesoderm formation. Dev. Cell. 2005;8:689–701. doi: 10.1016/j.devcel.2005.02.011. [DOI] [PubMed] [Google Scholar]
- Sive HL, Draper BW, Harland RM, Weintraub H. Identification of a retinoic acid-sensitive period during primary axis formation in Xenopus laevis. Genes Dev. 1990;4:932–942. doi: 10.1101/gad.4.6.932. [DOI] [PubMed] [Google Scholar]
- Slack JM, Darlington BG, Heath JK, Godsave SF. Mesoderm induction in early Xenopus embryos by heparin-binding growth factors. Nature. 1987;326:197–200. doi: 10.1038/326197a0. [DOI] [PubMed] [Google Scholar]
- Stavridis MP, Lunn JS, Collins BJ, Storey KG. A discrete period of FGF-induced Erk1/2 signalling is required for vertebrate neural specification. Development. 2007;134:2889–2894. doi: 10.1242/dev.02858. [DOI] [PubMed] [Google Scholar]
- Stavridis MP, Collins BJ, Storey KG. Retinoic acid orchestrates fibroblast growth factor signalling to drive embryonic stem cell differentiation. Development. 2010;137:881–890. doi: 10.1242/dev.043117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stern CD. Neural induction: old problem, new findings, yet more questions. Development. 2005;132:2007–2021. doi: 10.1242/dev.01794. [DOI] [PubMed] [Google Scholar]
- Storey KG, Goriely A, Sargent CM, Brown JM, Burns HD, Abud HM, Heath JK. Early posterior neural tissue is induced by FGF in the chick embryo. Development. 1998;125:473–484. doi: 10.1242/dev.125.3.473. [DOI] [PubMed] [Google Scholar]
- Streit A, Berliner AJ, Papanayotou C, Sirulnik A, Stern CD. Initiation of neural induction by FGF signalling before gastrulation. Nature. 2000;406:74–78. doi: 10.1038/35017617. [DOI] [PubMed] [Google Scholar]
- Sun X, Meyers EN, Lewandoski M, Martin GR. Targeted disruption of Fgf8 causes failure of cell migration in the gastrulating mouse embryo. Genes Dev. 1999;13:1834–1846. doi: 10.1101/gad.13.14.1834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tada M, Smith JC. Xwnt11 is a target of Xenopus Brachyury: regulation of gastrulation movements via Dishevelled, but not through the canonical Wnt pathway. Development. 2000;127:2227–2238. doi: 10.1242/dev.127.10.2227. [DOI] [PubMed] [Google Scholar]
- Takada S, Stark KL, Shea MJ, Vassileva G, McMahon JA, McMahon AP. Wnt-3a regulates somite and tailbud formation in the mouse embryo. Genes Dev. 1994;8:174–189. doi: 10.1101/gad.8.2.174. [DOI] [PubMed] [Google Scholar]
- Tanaka S, Kunath T, Hadjantonakis AK, Nagy A, Rossant J. Promotion of trophoblast stem cell proliferation by FGF4. Science. 1998;282:2072–2075. doi: 10.1126/science.282.5396.2072. [DOI] [PubMed] [Google Scholar]
- Tanaka Y, Okada Y, Hirokawa N. FGF-induced vesicular release of Sonic hedgehog and retinoic acid in leftward nodal flow is critical for left-right determination. Nature. 2005;435:172–177. doi: 10.1038/nature03494. [DOI] [PubMed] [Google Scholar]
- Turner N, Grose R. Fibroblast growth factor signalling: from development to cancer. Nat. Rev. Cancer. 2010;10:116–129. doi: 10.1038/nrc2780. [DOI] [PubMed] [Google Scholar]
- Walmsley M, Cleaver D, Patient R. Fibroblast growth factor controls the timing of Scl, Lmo2, and Runx1 expression during embryonic blood development. Blood. 2008;111:1157–1166. doi: 10.1182/blood-2007-03-081323. [DOI] [PubMed] [Google Scholar]
- Weaver C, Kimelman D. Move it or lose it: axis specification in Xenopus. Development. 2004;131:3491–3499. doi: 10.1242/dev.01284. [DOI] [PubMed] [Google Scholar]
- Wells JM, Melton DA. Early mouse endoderm is patterned by soluble factors from adjacent germ layers. Development. 2000;127:1563–1572. doi: 10.1242/dev.127.8.1563. [DOI] [PubMed] [Google Scholar]
- Wilkie AO. Bad bones, absent smell, selfish testes: the pleiotropic consequences of human FGF receptor mutations. Cytokine Growth Factor Rev. 2005;16:187–203. doi: 10.1016/j.cytogfr.2005.03.001. [DOI] [PubMed] [Google Scholar]
- Wills AE, Choi VM, Bennett MJ, Khokha MK, Harland RM. BMP antagonists and FGF signaling contribute to different domains of the neural plate in Xenopus. Dev. Biol. 2010;337:335–350. doi: 10.1016/j.ydbio.2009.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson SI, Graziano E, Harland R, Jessell TM, Edlund T. An early requirement for FGF signalling in the acquisition of neural cell fate in the chick embryo. Curr. Biol. 2000;10:421–429. doi: 10.1016/s0960-9822(00)00431-0. [DOI] [PubMed] [Google Scholar]
- Wilson V, Olivera-Martinez I, Storey KG. Stem cells, signals and vertebrate body axis extension. Development. 2009;136:1591–1604. doi: 10.1242/dev.021246. [DOI] [PubMed] [Google Scholar]
- Xian W, Schwertfeger KL, Vargo-Gogola T, Rosen JM. Pleiotropic effects of FGFR1 on cell proliferation, survival, and migration in a 3D mammary epithelial cell model. J. Cell Biol. 2005;171:663–673. doi: 10.1083/jcb.200505098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu RH, Ault KT, Kim J, Park MJ, Hwang YS, Peng Y, Sredni D, Kung H. Opposite effects of FGF and BMP-4 on embryonic blood formation: roles of PV.1 and GATA-2. Dev. Biol. 1999;208:352–361. doi: 10.1006/dbio.1999.9205. [DOI] [PubMed] [Google Scholar]
- Xu X, Li C, Takahashi K, Slavkin HC, Shum L, Deng CX. Murine fibroblast growth factor receptor 1alpha isoforms mediate node regression and are essential for posterior mesoderm development. Dev. Biol. 1999;208:293–306. doi: 10.1006/dbio.1999.9227. [DOI] [PubMed] [Google Scholar]
- Yamaguchi TP, Harpal K, Henkemeyer M, Rossant J. fgfr-1 is required for embryonic growth and mesodermal patterning during mouse gastrulation. Genes Dev. 1994;8:3032–3044. doi: 10.1101/gad.8.24.3032. [DOI] [PubMed] [Google Scholar]
- Yamanaka Y, Ralston A, Stephenson RO, Rossant J. Cell and molecular regulation of the mouse blastocyst. Dev. Dyn. 2006;235:2301–2314. doi: 10.1002/dvdy.20844. [DOI] [PubMed] [Google Scholar]
- Yamanaka Y, Lanner F, Rossant J. FGF signal-dependent segregation of primitive endoderm and epiblast in the mouse blastocyst. Development. 2010;137:715–724. doi: 10.1242/dev.043471. [DOI] [PubMed] [Google Scholar]
- Yamauchi H, Miyakawa N, Miyake A, Itoh N. Fgf4 is required for left-right patterning of visceral organs in zebrafish. Dev. Biol. 2009;332:177–185. doi: 10.1016/j.ydbio.2009.05.568. [DOI] [PubMed] [Google Scholar]
- Yang X, Dormann D, Munsterberg AE, Weijer CJ. Cell movement patterns during gastrulation in the chick are controlled by positive and negative chemotaxis mediated by FGF4 and FGF8. Dev. Cell. 2002;3:425–437. doi: 10.1016/s1534-5807(02)00256-3. [DOI] [PubMed] [Google Scholar]
- Yasuo H, Hudson C. FGF8/17/18 functions together with FGF9/16/20 during formation of the notochord in Ciona embryos. Dev. Biol. 2007;302:92–103. doi: 10.1016/j.ydbio.2006.08.075. [DOI] [PubMed] [Google Scholar]
- Ying QL, Wray J, Nichols J, Batlle-Morera L, Doble B, Woodgett J, Cohen P, Smith A. The ground state of embryonic stem cell self-renewal. Nature. 2008;453:519–523. doi: 10.1038/nature06968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang X, Ibrahimi OA, Olsen SK, Umemori H, Mohammadi M, Ornitz DM. Receptor specificity of the fibroblast growth factor family. The complete mammalian FGF family. J. Biol. Chem. 2006;281:15694–15700. doi: 10.1074/jbc.M601252200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao X, Duester G. Effect of retinoic acid signaling on Wnt/beta-catenin and FGF signaling during body axis extension. Gene Expr. Patterns. 2009;9:430–435. doi: 10.1016/j.gep.2009.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]