Abstract
Obstructive sleep apnoea (OSA) and hypertension commonly coexist. Observational studies indicate that untreated OSA is strongly associated with an increased risk of prevalent hypertension, whereas prospective studies of normotensive cohorts suggest that OSA may increase the risk of incident hypertension. Randomized evaluations of continuous positive airway pressure (CPAP) indicate an overall modest effect on blood pressure (BP). Determining why OSA is so strongly linked to having hypertension in cross-sectional studies, but yet CPAP therapy has limited BP benefit needs further exploration. The CPAP studies do, however, indicate a wide variation in the BP effects of CPAP, with some patients manifesting a large antihypertensive benefit such that a meaningful BP effect can be anticipated in some individuals. OSA is particularly common in patients with resistant hypertension (RHTN). The reason for this high prevalence of OSA is not fully explained, but data suggest that it may be related to the high occurrence of hyperaldosteronism in patients with RHTN. In patients with RHTN, it has been shown that aldosterone levels correlate with severity of OSA and that blockade of aldosterone reduces the severity of OSA. Overall, these findings are consistent with aldosterone excess contributing to worsening of underlying OSA. We hypothesize that aldosterone excess worsens OSA by promoting accumulation of fluid within the neck, which then contributes to increased upper airway resistance.
Keywords: obstructive sleep apnoea, hyperaldosteronism, resistant hypertension, continuous positive airway pressure
Resistant hypertension, sleep apnoea and aldosterone
Obstructive sleep apnoea (OSA) and hypertension are commonly associated. Approximately 50–56% of individuals with OSA are hypertensive, and an estimated 30–40% of hypertensive subjects have OSA.1–4 In younger adults, the presence of OSA is strongly associated with having hypertension, and, in normotensive individuals, having OSA predicts the development of hypertension.
Moreover, recent reports suggested that masked hypertension is frequently underestimated in OSA, suggesting that normotensive patients represent only one-third of the OSA population.
Interestingly, when using 24-h ambulant blood pressure (BP) monitoring in patients with OSA without a history of hypertension, the prevalence of hypertension increased from 42% in the apnoeic patients demonstrating clinical hypertension, either daytime hypertension or night-time hypertension, or both, to 80%.5–7 Cross-sectional studies indicate that individuals with moderate–severe OSA (apnoea–hypopnoea index (AHI) >15 events h−1) are much more likely to be hypertensive than individuals similar in age and body weight but without OSA. These studies generally indicate a positive correlation between severity of OSA and level of BP, suggesting that the more severe the OSA the higher the likelihood of also having severe hypertension. In the Wisconsin Sleep Cohort Study, initial cross-sectional analyses indicated that moderate–severe OSA was associated with an 80% increased risk of having hypertension.8 Moreover, using an observational design, Lavie et al.9 found that both the prevalence and severity of hypertension increased with increasing AHI. Overall, an increasing AHI was positively associated with both higher systolic and diastolic BP, with each 1 event h−1 increase in AHI being associated with a 1% higher risk of having hypertension. In a large cross-sectional population of 1741 subjects, Bixler et al.10 confirmed a strong positive correlation between moderate–severe OSA and risk of prevalent hypertension. Interestingly, in this evaluation, the strength of the relationship between OSA and BP diminished with increasing age, such that the risk of having hypertension associated with OSA was less pronounced in older individuals.
Similarly, longitudinal studies suggest a dichotomous relationship between OSA and risk of developing hypertension that is dependent upon age. In the Wisconsin Sleep Cohort Study, normotensive subjects with moderate–severe OSA had a threefold increase in the risk of developing hypertension over a 4-year period compared with subjects without OSA.11 In contrast, investigators conducting the Sleep Heart Health Study did not find, after adjusting for body mass index, that having OSA predicted development of hypertension in previously normotensive individuals during a 5-year follow-up.12 Although there were several important methodological differences in these two studies, including differences in cohort size and diversity and use of attended in-laboratory versus at-home unattended polysomnography, a particularly important distinction was that participants in the Wisconsin Sleep Cohort Study were, on average, considerably younger than participants in the Sleep Heart Health Study (47 versus 60 years, respectively).13 Consistent with the observational data indicating that the positive relationship between OSA and risk-prevalent hypertension diminishes with age, it is tempting to attribute the contrasting results of the two prospective studies of OSA and risk of incident hypertension also to a blunting of the interaction with increasing age. Such an interpretation suggests that younger individuals are more susceptible to the potential hypertensive effects of untreated OSA.
CPAP treatment and OSA
Treatment studies of OSA with use of continuous positive airway pressure (CPAP) have generally shown a consistent but modest antihypertensive benefit. Importantly, these studies indicate a wide variation in the BP response, with some individuals manifesting a very high BP response and others having no antihypertensive benefit. Persons who are more likely to benefit are patients with more severe OSA, higher baseline BP levels and patients most adherent with CPAP use.
Recently, four meta-analyses of randomized, controlled CPAP trials have been published. In the two studies reporting changes in clinical BP levels, being randomized to CPAP therapy compared with control reduced systolic BP by ~1.5–2.5 mm Hg and diastolic BP by 1.5–2 mm Hg.14,15 In the two metaanalyses reporting changes in BP obtained by ambulatory monitoring, CPAP use was associated with an ~1.0–1.5 mm Hg reduction in both 24-h systolic and diastolic BP.16,17 In subgroup analyses done by the authors, severe OSA (>30 events h−1), higher BP levels and greater CPAP adherence were associated with larger reductions in BP.14–17
Although the antihypertensive benefit of CPAP is certainly not inconsequential, it is perplexing as to why treatment of OSA does not improve lower BP given the strong association between the two disease processes. Although speculative, given the probable long-term presence of OSA before being diagnosed and treated, it might be that CPAP will be better at preventing the progression of OSA-induced increases in BP as opposed to reversing hypertension. Another important consideration is that the benefit of CPAP might be substantially limited by poor adherence. Even in short-term intervention studies of CPAP, adherence averages 4–5 h a night, meaning that most patients are untreated for a large proportion of their normal sleep period.17 Similarly, better results would be observed with overall better CPAP adherence. Lastly, it is important to highlight the fact that, although the mean treatment effect of CPAP is modest, there is a wide range in the response such that some individuals have a substantial antihypertensive benefit. The patients most likely to manifest benefit are more likely to have severe OSA and poorly controlled hypertension, and because of these factors patients are at increased cardiovascular risk.
Mechanisms of OSA-induced hypertension
Multiple mechanisms undoubtedly contribute to OSA-induced increases in BP. Stimulation of sympathetic activity by OSA is well described and likely represents the most important effect by which OSA increases BP. This effect is not limited to the period of apnoea/hypopnoea but instead manifests as a sustained increase in sympathetic activation even during the daytime when affected patients are awake.18 Heightened sympathetic activity would be expected to increase BP through elevation of vascular resistance, greater cardiac output and, possibly, stimulation of the renin–angiotensin–aldosterone system. Effective treatment of the OSA with CPAP suppresses the sympathetic activation.19
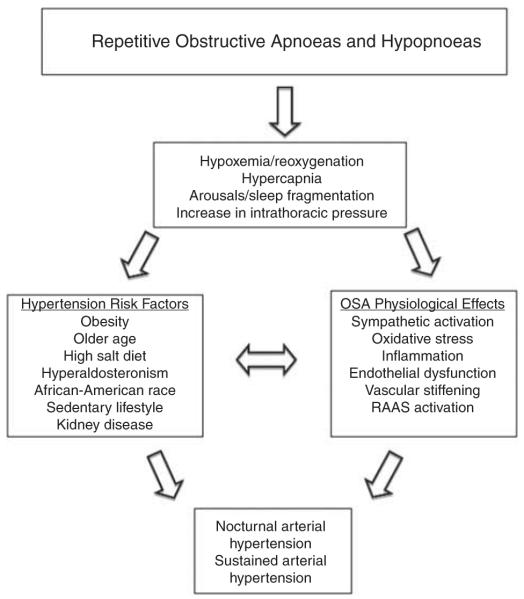
Other pathophysiological effects of OSA that likely contribute to increased risk of developing hypertension include a proinflammatory effect, increased oxidative stress and increased vascular stiffness (Figure 1). Small CPAP intervention trials suggest that each of these effects can be reduced with effective CPAP use, often quite rapidly.20–24
Figure 1.
Pathophysiological mechanisms involved in the aetiology of OSA-induced hypertension. RAAS, renin–angiotensin–aldosterone system.
OSA and resistant hypertension
Resistant hypertension (RHTN) is most simply defined as BP that requires four or more antihypertensive medications.25,26
Although the exact prevalence of RHTN is not known, cross-sectional studies suggest that ~15% of patients being treated in a primary care setting fulfil the above criteria for RHTN.27 As a subgroup, patients with RHTN are distinguished from patients with more easily controlled hypertension by an increased likelihood of comorbidities, such as obesity, diabetes, left ventricular hypertrophy, chronic kidney disease and a higher cardiovascular risk, as evidenced by cross-sectional and prospective assessments.28–32 The prevalence of OSA is also increased in patients with RHTN compared with hypertensive patients whose BP is not resistant to treatment.
OSA and lack of BP control
Untreated OSA, particularly severe OSA, is strongly associated with poor BP control. In a comparison of hypertensive and normotensive men referred for a sleep study, Hirshkowitz et al.33 found that treated but uncontrolled hypertension was associated with significantly more severe OSA compared with treated and controlled hypertension. In a cross-sectional evaluation of almost 600 patients referred to a sleep centre in Sweden for suspicion of sleep apnoea, it was observed that increasing severity of OSA was positively correlated with both systolic and diastolic BP.34 In patients <50 years of age, severity of OSA was an independent predictor of uncontrolled hypertension. In a cross-sectional analysis of hypertensive patients who had been evaluated by polysomnography, Lavie et al.35 found that patients with treated but uncontrolled hypertension had a significantly higher AHI compared with patients with controlled hypertension (44±29 versus 33.4±25 events h−1, respectively). The significant difference in mean AHI persisted even after adjusting for age, gender and body mass index. In a prospective assessment of patients selected for having uncontrolled hypertension as opposed to symptoms of OSA, more than 70% of the 49 patients had moderate–severe OSA based on an AHI >10 events h−1.36 In this analysis, the more severe the OSA the more likely both daytime and night-time ambulatory BP remained elevated, in spite of the use of a greater number of antihypertensive agents compared with patients with less severe OSA. Similarly, in a prospective assessment of patients on haemodialysis, it was found that patients with more severe OSA required more antihypertensive medications to achieve the same level of BP control than patients with less severe OSA.37 Overall, patients with OSA were on an average on 3.5 antihypertensive medications compared with only 1.6 medications for patients without OSA. These studies clearly demonstrate that untreated OSA is associated with greater difficulty in the control of hypertension.
The effect is dose dependent in that as the severity of OSA increases BP severity increases, and so does the number antihypertensive medications needed for BP control.
RHTN and prevalence of OSA
The prevalence of OSA is extraordinarily high in patients with RHTN. In studies of general hypertensive cohorts, the prevalence of OSA has been found to be ~30–40%, with higher rates in men. In contrast, in studies of patients with RHTN, the prevalence of OSA has been found to be 70–90%, with rates approaching 100% of the men. The reason for the much higher prevalence of OSA in patients with RHTN has not been fully elucidated, but observations from our laboratory suggest that hyperaldosteronism may contribute importantly to the difference.
In a prospective study, Logan et al.38 found the overall prevalence of OSA, based on an AHI of >10 events h−1, to be 83% among consecutive patients diagnosed with RHTN. The prevalence of OSA in women with RHTN was 65%, whereas 96% in men. In a prospective comparison of 63 patients with RHTN and 63 control patients with treated and controlled hypertension, Gonçalves et al.39 found that 71% of the former versus 38% of the latter had OSA (AHI >10 events h−1). In the RHTN group, 86% of the men and 64% of the women were confirmed to have OSA. Our experience at the University of Alabama at Birmingham has been similar to the above two studies. In a prospective evaluation, we enrolled consecutive patients referred to our clinic for RHTN.40 After confirming the diagnosis, all patients were sent for a full, overnight polysomnogram, irrespective of whether or not they had symptoms of OSA. A total of 71 patients with RHTN were studied. Among these patients, the overall prevalence of OSA based on an AHI >5 events h−1 was 85%. Similar to the above studies, OSA was more common in male subjects with RHTN (90%) compared with women (77%). The findings of these three studies are consistent in demonstrating an especially high prevalence of OSA among patients with RHTN. In the Gonçalves study, which included a control group, OSA was considerably higher in patients with RHTN compared with patients whose BP was controlled.39
Aldosterone and OSA severity
Multiple laboratory studies have found that aldosterone excess is common in patients with RHTN.41–43 It contributes importantly to the development of treatment resistance as evidenced by the large antihypertensive benefit of aldosterone antagonists that is observed in patients who had been resistant to other classes of antihypertensive agents.44,45 Recognizing the fact that both OSA and hyperaldosteronism are common in patients with RHTN, we hypothesized that the two may be mechanistically related, with one contributing to the development of the other. We have conducted a series of studies testing this possibility and on the basis of those results have come to the conclusion that aldosterone excess likely contributes to the severity of OSA. Although speculative, we attribute this worsening of OSA to aldosterone-induced fluid retention, leading to parapharyngeal oedema and increased upper airway resistance.
In our first study exploring the relationship between OSA and aldosterone levels, we prospectively assessed 114 patients with RHTN for risk of having OSA on the basis of their responses to the Berlin Questionnaire.46 We found that patients at high risk of having OSA had significantly greater 24-h urinary aldosterone excretion and were more than twice as likely to be diagnosed with primary aldosteronism compared with patients at low risk of having OSA. In a separate study, we found that plasma aldosterone levels positively correlate with the AHI in patients with RHTN (Figure 2);40 that is, in these patients with RHTN, the higher the aldosterone levels the more severe their OSA. Most recently, in an evaluation of over 109 patients with RHTN, we reported that the positive correlation between plasma and 24-h urinary aldosterone levels and AHI is largely attributable to patients with hyperaldosteronism.47 In patients with normal or low aldosterone levels, the aldosterone levels and AHI were generally unrelated, indicating that the aldosterone-induced exacerbation of OSA severity is limited to patients with demonstrable aldosterone excess.
Figure 2.
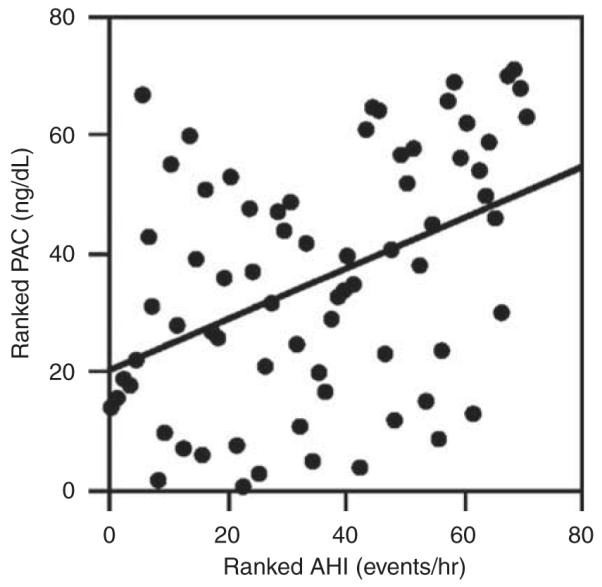
AHI (ρ = 0.44, P = 0.0002) correlates with plasma aldosterone concentration (PAC) in subjects with RHTN. Reprinted with permission from Pratt-Ubunama et al.40
Aldosterone antagonism improves severity of OSA
A positive correlation between OSA severity and aldosterone levels suggests2 opposite possibilities: either untreated OSA is stimulating aldosterone release or aldosterone excess is worsening OSA. The former possibility is supported by experimental data suggesting that intermittent hypoxaemia promotes a generalized stimulation of the renin–angiotensin–aldosterone system.48 Human studies, however, have generally not shown treatment of OSA with CPAP to have a substantive effect on aldosterone levels.
For example, in a recent clinical study, the first night of CPAP did not lower either plasma renin activity or plasma aldosterone levels in normotensive subjects undergoing CPAP titration.49 The lack of a consistent effect of CPAP on aldosterone levels in several studies seems to argue against the belief that OSA causes excess aldosterone release; however, definitive evidence is still needed.
Clinical data, however, do support the opposite effect, that is, aldosterone excess worsens OSA severity by showing that aldosterone blockade with spironolactone decreases the severity of OSA in patients with RHTN. In this study, 12 patients with RHTN and moderate–severe OSA (AHI > 15 events h−1) were treated with spironolactone 50 mg daily, in addition to their normal antihypertensive regimen, which included a thiazide diuretic.50 A full-night polysomnogram was done at baseline and then repeated 8 weeks after beginning treatment with spironolactone. Spironolactone use was associated with an almost 50% reduction in severity of OSA, based on an improvement in the total AHI, as well as on significant reductions in AHI during supine sleeping and during rapid-eye-movement sleep, when OSA tends to be most severe (Figure 3). These results are important in providing confirmatory support of the hypothesized role of aldosterone excess in worsening OSA.
Figure 3.
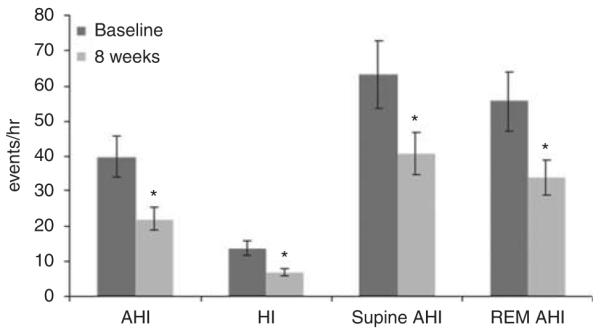
Effects of 8 weeks of treatment with spironolactone on AHI; hypoxic index (HI); supine AHI; and rapid-eye-movement sleep (REM). AHI at 8 weeks (light grey bars) compared with baseline (dark grey bars) in patients with RHTN. *Different compared with baseline, P>0.05. Reprinted with permission from Gaddam et al.50
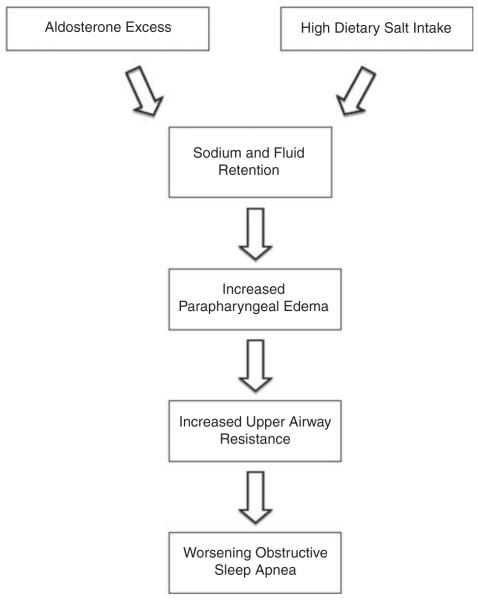
The mechanism by which aldosterone promotes worsening of OSA has not been fully elucidated, but we hypothesize that it is related to aldosterone-induced fluid retention, some of which is localized to the neck, leading to parapharyngeal oedema and associated increases in upper airway resistance (Figure 4). Such an effect is supported by studies of normotensive volunteers demonstrating increases in neck circumference and upper airway resistance after acute increases in central venous return during lower-body positive pressure.51 Greater increases in neck circumference during overnight recumbency have also been observed in patients with RHTN compared with patients with controlled hypertension.52 Such an effect of fluid retention contributing to worsening OSA is also suggested by studies of patients with acute heart failure, in whom diuresis improves the severity of their sleep apnoea.53
Figure 4.
Proposed pathophysiological mechanisms of aldosterone-induced worsening of OSA.
CPAP and RHTN
A small number of uncontrolled studies have reported a significant but varying effect of CPAP on BP in patients with RHTN. In 11 patients with RHTN, Logan et al.54 reported that 2 months of CPAP use reduced 24-h systolic BP by 11.0 mm Hg, with the largest benefit being caused overnight (−14.4 mm Hg). CPAP also significantly reduced 24-h diastolic BP (−7.8 mm Hg), again with the largest benefit being caused overnight (−7.9 mm Hg). In a separate study of 33 patients with RHTN, 3 months of CPAP treatment had a considerably smaller effect, reducing 24-h systolic BP by 5.2 mm Hg and having no significant effect on ambulatory diastolic BP.55 In a comparison between patients with RHTN (n = 42) and patients with controlled hypertension (n = 56), 1 year of CPAP treatment reduced mean clinical arterial BP by 5.6 mm Hg in the former group, while having no significant effect in the latter group.26 CPAP use did allow for a reduction in antihypertensive therapy in 71% of the patients with RHTN. Overall, these studies suggest that CPAP therapy does provide antihypertensive benefit to patients with RHTN, but, similar to patients with generalized hypertension, the treatment effect is variable, with some patients manifesting a large benefit and many having little or no benefit. More definitive assessment of benefit would be provided by randomized studies of CPAP use, which, to this point, are lacking in patients with RHTN.
Clinical implications
Ambulatory BP monitoring is a useful diagnostic tool to diagnose truly resistant hypertensive patients and a valid, non-invasive method to describe circadian BP variation.
Non-dipping BP status is particularly common in OSA and often coexists in patients with RHTN. Patients with suspected OSA should undergo a polysomnography, the ‘gold standard’ to diagnose OSA, and if confirmed should be treated with CPAP.
Primary aldosteronism in patients with RHTN needs to be evaluated and, if biochemically confirmed, a workup to rule out a unilateral aldosterone-producing adrenal adenoma needs to be further considered, compared with simply treating them medically. Patients having aldosterone excess without adenoma should be treated with an aldosterone antagonist. We have demonstrated that aldosterone blockade improves severity of OSA in patients with RHTN. Until confirmed by randomized studies, it is premature to specifically recommend such agents for this purpose, but as aldosterone antagonists are routinely used for their antihypertensive benefit in patients with RHTN, the added benefit of reducing the severity of a patient’s OSA might be accomplished when targeting his or her hypertension. To what extent aldosterone antagonists might reduce the severity of OSA in patients with primary aldosteronism or even less severe cases of hypertension awaits separate evaluation.
Acknowledgements
This paper was supported by grants NHLBI SCCOR P50 HL077100, RO1-HL79040 and CCTS 5UL1 RR025777.
Footnotes
Conflict of interest The authors declare no conflict of interest.
References
- 1.Fletcher EC, DeBehnke RD, Lovoi MS, Gorin AB. Undiagnosed sleep apnea in patients with resistant hypertension. Ann Intern Med. 1985;103:190–195376. doi: 10.7326/0003-4819-103-2-190. [DOI] [PubMed] [Google Scholar]
- 2.Lavie P, Ben-Yosef R, Rubin AE. Prevalence of sleep apnea syndrome among patients with essential hypertension. Am Heart J. 1984;108:373–376. doi: 10.1016/0002-8703(84)90628-8. [DOI] [PubMed] [Google Scholar]
- 3.Worsnop CJ, Naughton MT, Barter CE, Morgan TO, Anderson Al, Pierce RJ. The prevalence of obstructive sleep apnea in hypertensives. Am J Respir Crit Care Med. 1998;157:111–115. doi: 10.1164/ajrccm.157.1.9609063. [DOI] [PubMed] [Google Scholar]
- 4.Drager LF, Genta RP, Nerbass FB, Gonzagea CC, Krieger EM, Lorenzi-Filho G. Characteristics and predictors of obstructive sleep apnea in patients with systemic hypertension. Am J Cardiol. 2010;105:1135–1139. doi: 10.1016/j.amjcard.2009.12.017. [DOI] [PubMed] [Google Scholar]
- 5.Baguet JP, Hammer L, Lévy P, Pierre H, Rossini E, Mouret S, et al. Night-time and diastolic hypertension are common and underestimated conditions in newly diagnosed apnoeic patients. J Hypertens. 2005;23:521–527. doi: 10.1097/01.hjh.0000160207.58781.4e. [DOI] [PubMed] [Google Scholar]
- 6.Drager LF, Diegues-Silva L, Diniz MP, Bortolotto LA, Pedrosa RP, Couto RB, et al. Obstructive sleep apnea, masked hypertension, and arterial stiffness in men. Am J Hypertens. 2010;23:249–254. doi: 10.1038/ajh.2009.246. [DOI] [PubMed] [Google Scholar]
- 7.Baguet JP, Levy P, Baron-Rochette G, Tamisier R, Pierre H, Peeters M, et al. Masked hypertension in obstructive sleep apnea syndrome. J Hypertens. 2008;26:885–892. doi: 10.1097/HJH.0b013e3282f55049. [DOI] [PubMed] [Google Scholar]
- 8.Young T, Peppard P, Palta M, Hla KM, Finn L, Morgan B, et al. Population-based study of sleep-disordered breathing as a risk factor for systemic hypertension. Arch Intern Med. 1997;157:1746–1752. [PubMed] [Google Scholar]
- 9.Lavie P, Herer P, Hoffstein V. Obstructive sleep apnoea syndrome as a risk factor for hypertension: population study. BMJ. 2000;320:479–482. doi: 10.1136/bmj.320.7233.479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bixler EO, Vgontzas AN, Lin BK, Ten Have T, Leiby BE, Vela-Bueno A, et al. Association of hypertension and sleep-disordered breathing. Arch Intern Med. 2000;160:2289–2295. doi: 10.1001/archinte.160.15.2289. [DOI] [PubMed] [Google Scholar]
- 11.Peppard PE, Young T, Palta M, Skatrud J. Prospective study of the association between sleep-disordered breathing and hypertension. N Engl J Med. 2000;342:1378–1384. doi: 10.1056/NEJM200005113421901. [DOI] [PubMed] [Google Scholar]
- 12.O’Connor GT, Caffo B, Newman AB, Quan SF, Rapoport DM, Redline S, et al. Prospective study of sleep-disordered breathing and hypertension: the Sleep Heart Health Study. Am J Respir Crit Care Med. 2009;179:1159–1164. doi: 10.1164/rccm.200712-1809OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Peppard PE. Is obstructive sleep apnea a risk factor for hypertension?—Differences between the Wisconsin Sleep Cohort and the Sleep Heart Health Study. J Clin Sleep Med. 2009;5:404–405. [PMC free article] [PubMed] [Google Scholar]
- 14.Bazzano LA, Khan Z, Reynolds K, He J. Effect of nocturnal nasal continuous positive airway pressure on blood pressure in obstructive sleep apnea. Hypertension. 2007;50:417–423. doi: 10.1161/HYPERTENSIONAHA.106.085175. [DOI] [PubMed] [Google Scholar]
- 15.Alajmi M, Mulgrew AT, Fox J, Davidson W, Schulzer M, Mak E, et al. Impact of continuous positive airway pressure therapy on blood pressure in patients with obstructive sleep apnea hypopnea: a meta-analysis of randomized controlled trials. Lung. 2007;185:67–72. doi: 10.1007/s00408-006-0117-x. [DOI] [PubMed] [Google Scholar]
- 16.Mo L, He QY. Effect of long-term continuous positive airway pressure ventilation on blood pressure in patients with obstructive sleep apnea hypopnea syndrome: a meta-analysis of clinical trials. Zhonghua Yi Xue Za Zhi. 2007;87:1177–1180. [PubMed] [Google Scholar]
- 17.Haentjens P, Van Meerhaeghe A, Moscariello A, De Weerdt S, Poppe K, Dupont A, et al. The impact of continuous positive airway pressure on blood pressure in patients with obstructive sleep apnea syndrome: evidence from a meta-analysis of placebo-controlled randomized trials. Arch Intern Med. 2007;167:757–764. doi: 10.1001/archinte.167.8.757. [DOI] [PubMed] [Google Scholar]
- 18.Somers VK, Dyken ME, Clary MP, Abboud FM. Sympathetic neural mechanisms in obstructive sleep apnea. J Clin Invest. 1995;96:1897–1904. doi: 10.1172/JCI118235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Narkiewicz K, Kato M, Phillips BG, Pesek CA, Davison DE, Somers VK. Nocturnal continuous positive airway pressure decreases daytime sympathetic traffic in obstructive sleep apnea. Circulation. 1999;100:2332–2335. doi: 10.1161/01.cir.100.23.2332. [DOI] [PubMed] [Google Scholar]
- 20.Ishida K, Kato M, Kato Y, Yanagihara K, Kinugasa Y, Kotani K, et al. Appropriate use of nasal continuous positive airway pressure decreases elevated C-reactive protein in patients with obstructive sleep apnea. Chest. 2009;136:125–129. doi: 10.1378/chest.08-1431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.de Lima AM, Franco CM, de Castro CM, Bezerra Ade A, Ataíde L, Jr, Halpern A. Effects of nasal continuous positive airway pressure treatment on oxidative stress and adiponectin levels in obese patients with obstructive sleep apnea. Respiration. 2010;79:370–376. doi: 10.1159/000227800. [DOI] [PubMed] [Google Scholar]
- 22.Phillips CL, Yang Q, Williams A, Roth M, Yee BJ, Hedner JA, et al. The effect of short-term withdrawal from continuous positive airway pressure therapy on sympathetic activity and markers of vascular inflammation in subjects with obstructive sleep apnoea. J Sleep Res. 2007;16:217–225. doi: 10.1111/j.1365-2869.2007.00589.x. [DOI] [PubMed] [Google Scholar]
- 23.Drager LF, Bortolotto LA, Figueiredo AC, Krieger EM, Lorenzi GF. Effects of continuous positive airway pressure on early signs of atherosclerosis in obstructive sleep apnea. Am J Respir Crit Care Med. 2007;176:706–712. doi: 10.1164/rccm.200703-500OC. [DOI] [PubMed] [Google Scholar]
- 24.Kitahara Y, Hattori N, Yokoyama A, Nakajima M, Kohno N. Effect of CPAP on brachial-ankle pulse wave velocity in patients with OSAHS: an open-labelled study. Respir Med. 2006;100:2160–2169. doi: 10.1016/j.rmed.2006.03.015. [DOI] [PubMed] [Google Scholar]
- 25.Calhoun DA, Jones D, Textor S, Goff DC, Murphy TP, Toto RD, et al. Resistant hypertension: a scientific statement from the American Heart Association Professional Education Committee of the Council for High Blood Pressure Research. Hypertension. 2008;51:1403–1419. doi: 10.1161/HYPERTENSIONAHA.108.189141. [DOI] [PubMed] [Google Scholar]
- 26.Dernaika TA, Kinasewitz GT, Tawk MM. Effects of nocturnal continuous positive airway pressure therapy in patients with resistant hypertension and obstructive sleep apnea. J Clin Sleep Med. 2009;5:103–107. [PMC free article] [PubMed] [Google Scholar]
- 27.Egan BM, Zhao Y, Rehman SU, Brzezinski WA, Clyburn B, Basile JN, et al. Treatment resistant hypertension in a community-based practice network. J Clin Hypertens. 2009;11(Supplement 1):5. [Google Scholar]
- 28.McAdam-Marx C, Ye X, Sung JC, Brixner DI, Kahler KH. Results of a retrospective, observational pilot study using electronic medical records to assess the prevalence and characteristics of patients with resistant hypertension in an ambulatory care setting. Clin Ther. 2009;3:1116–1123. doi: 10.1016/j.clinthera.2009.05.007. [DOI] [PubMed] [Google Scholar]
- 29.Cuspidi C, Macca G, Sampieri L, Michev I, Salerno M, Fusi V, et al. High prevalence of cardiac and extra-cardiac target organ damage in refractory hypertension. J Hypertens. 2001;19:2063–2070. doi: 10.1097/00004872-200111000-00018. [DOI] [PubMed] [Google Scholar]
- 30.Muxfeldt ES, Bloch KV, Nogueira AR, Salles GF. Twenty-four hour ambulatory blood pressure monitoring pattern of resistant hypertension. Blood Press Monit. 2003;8:181–185. doi: 10.1097/00126097-200310000-00001. [DOI] [PubMed] [Google Scholar]
- 31.Pierdomenico SD, Lapenna D, Bucci A, Di Tommaso R, Di Mascio R, Manente BM, et al. Cardiovascular outcome in treated hypertensive patients with responder, masked, false resistant and true resistant hypertension. Am J Hypertens. 2005;18:1422–1428. doi: 10.1016/j.amjhyper.2005.05.014. [DOI] [PubMed] [Google Scholar]
- 32.Isaksson H, Ostergren J. Prognosis in therapy-resistant hypertension. J Intern Med. 1994;236:643–649. doi: 10.1111/j.1365-2796.1994.tb00857.x. [DOI] [PubMed] [Google Scholar]
- 33.Hirshkowitz M, Karacan I, Gurakar A, Williams RL. Hypertension, erectile dysfunction, and occult sleep apnea. Sleep. 1989;12:223–232. doi: 10.1093/sleep/12.3.223. [DOI] [PubMed] [Google Scholar]
- 34.Grote L, Hedner J, Peter JH. Sleep-related breathing disorder is an independent risk factor for uncontrolled hypertension. J Hypertens. 2000;18:679–685. doi: 10.1097/00004872-200018060-00004. [DOI] [PubMed] [Google Scholar]
- 35.Lavie P, Hoffstein V. Sleep apnea syndrome: a possible contributing factor to resistant hypertension. Sleep. 2001;24:721–725. doi: 10.1093/sleep/24.6.721. [DOI] [PubMed] [Google Scholar]
- 36.Martínez-García MA, Gómez-Aldaraví R, Gil-Martínez T, Soler-Cataluña JJ, Bernácer-Alpera B, Román-Sánchez P. Sleep-disordered breathing in patients with difficult-to-control hypertension. Arch Bronconeumol. 2006;42:14–20. doi: 10.1016/s1579-2129(06)60108-0. [DOI] [PubMed] [Google Scholar]
- 37.Elias MR, Castro MCM, de Queiroz EL, Abensur H, Romão-Junior JE, Lorenzi-Fiho G. Obstructive sleep apnea in patients on conventional and short daily hemodialysis. Am J Nephrol. 2009;29:493–500. doi: 10.1159/000178941. [DOI] [PubMed] [Google Scholar]
- 38.Logan AG, Perlikowski SM, Mente A, Tisler A, Tkacova R, Niroumand M, et al. High prevalence of unrecognized sleep apnea in drug-resistant hypertension. J Hypertens. 2001;19:2271–2277. doi: 10.1097/00004872-200112000-00022. [DOI] [PubMed] [Google Scholar]
- 39.Gonçalves SC, Martinez D, Gus M, Oliveira de Abreu-Silva E, Bertoluci C, Dutra I, et al. Obstructive sleep apnea and resistant hypertension: a case-control study. Chest. 2007;132:1858–1862. doi: 10.1378/chest.07-1170. [DOI] [PubMed] [Google Scholar]
- 40.Pratt-Ubunama MN, Nishizaka MK, Boedefeld RL, Cofield SS, Harding SM, Calhoun DA. Plasma aldosterone is related to severity of obstructive sleep apnea in subjects with resistant hypertension. Chest. 2007;131:453–459. doi: 10.1378/chest.06-1442. [DOI] [PubMed] [Google Scholar]
- 41.Calhoun DA, Nishizaka MK, Zaman MA, Thakkar RB, Weissmann P. Hyperaldosteronism among black and white subjects with resistant hypertension. Hypertension. 2002;40:892–896. doi: 10.1161/01.hyp.0000040261.30455.b6. [DOI] [PubMed] [Google Scholar]
- 42.Gallay BJ, Ahmad S, Xu L, Toivola B, Davidson RC. Screening for primary aldosteronism without discontinuing hypertensive medications: plasma aldosterone-renin ratio. Am J Kidney Dis. 2001;37:699–705. doi: 10.1016/s0272-6386(01)80117-7. [DOI] [PubMed] [Google Scholar]
- 43.Eide IK, Torjesen PA, Drolsum A, Babovic A, Lilledahl NP. Low-renin status in therapy-resistant hypertension: a clue to efficient treatment. J Hypertens. 2004;22:2217–2226. doi: 10.1097/00004872-200411000-00026. [DOI] [PubMed] [Google Scholar]
- 44.Nishizaka MK, Zaman MA, Calhoun DA. Efficacy of low-dose spironolactone in subjects with resistant hypertension. Am J Hypertens. 2003;16:925–930. doi: 10.1016/s0895-7061(03)01032-x. [DOI] [PubMed] [Google Scholar]
- 45.Chapman N, Dobson J, Wilson S, Dahlöf B, Sever PS, Wedel H, et al. Effect of spironolactone on blood pressure in subjects with resistant hypertension. Hypertension. 2007;49:839–845. doi: 10.1161/01.HYP.0000259805.18468.8c. [DOI] [PubMed] [Google Scholar]
- 46.Calhoun DA, Nishizaka MK, Zaman MA, Harding SM. Aldosterone excretion among subjects with resistant hypertension and symptoms of sleep apnea. Chest. 2004;125:112–117. doi: 10.1378/chest.125.1.112. [DOI] [PubMed] [Google Scholar]
- 47.Gonzaga CC, Gaddam KK, Ahmed MI, Pimenta E, Thomas SJ, Harding SM, et al. Severity of obstructive sleep apnea is related to aldosterone in subjects with resistant hypertension. J Clin Sleep Med. 2010;6:363–368. [PMC free article] [PubMed] [Google Scholar]
- 48.Raff H, Roarty TP. Renin, ACTH, and aldosterone during acute hypercapnia and hypoxia in conscious rats. Am J Physiol. 1988;254:R431–R435. doi: 10.1152/ajpregu.1988.254.3.R431. [DOI] [PubMed] [Google Scholar]
- 49.Svatikova A, Olson LJ, Wolk R, Phillips BG, Adachi T, Schwartz GL, et al. Obstructive sleep apnea and aldosterone. Sleep. 2009;32:1589–1592. doi: 10.1093/sleep/32.12.1589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Gaddam K, Pimenta E, Thomas SJ, Oparil S, Harding SM, Calhoun DA. Spironolactone reduces severity of obstructive sleep apnea in patients with resistant hypertension: a preliminary report. J Hum Hypertens. 2010;24:532–537. doi: 10.1038/jhh.2009.96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Chiu KL, Ryan CM, Shiota S, Ruttanaumpawan P, Arzt M, Haight JS, et al. Fluid shift by lower body airway positive pressure increases pharyngeal resistance in healthy subjects. Am J Respir Crit Care Med. 2006;174:1378–1383. doi: 10.1164/rccm.200607-927OC. [DOI] [PubMed] [Google Scholar]
- 52.Friedman O, Bradley TD, Chan CT, Parkes C, Logan AG. Relationship between overnight rostral fluid shift and obstructive sleep apnea in drug-resistant hypertension. Hypertension. 2010;56:1077–1082. doi: 10.1161/HYPERTENSIONAHA.110.154427. [DOI] [PubMed] [Google Scholar]
- 53.Bucca CB, Brussino L, Battisti A, Mutani R, Rolla G, Mangiardi L, et al. Diuretics in obstructive sleep apnea with diastolic heart failure. Chest. 2007;132:440–446. doi: 10.1378/chest.07-0311. [DOI] [PubMed] [Google Scholar]
- 54.Logan AG, Tkacova R, Perlikowski SM, Leung RS, Tisler A, Floras JS, et al. Refractory hypertension and sleep apnoea: effect of CPAP on blood pressure and baroreflex. Eur Respir J. 2003;21:241–247. doi: 10.1183/09031936.03.00035402. [DOI] [PubMed] [Google Scholar]
- 55.Martínez-García MA, Gómez-Aldaraví R, Soler-Cataluña JJ, Martínez TG, Bernácer-Alpera B, Román-Sánchez P. Positive effect of CPAP treatment on the control of difficult-to-treat hypertension. Eur Respir J. 2007;29:951–957. doi: 10.1183/09031936.00048606. [DOI] [PubMed] [Google Scholar]