Abstract
Dendritic cells (DCs) are permissive to murine norovirus (MNV) infection in vitro and in vivo. However, their roles during infection in vivo are not well defined. To determine the role of DCs during infection, conventional DCs were depleted from CD11c-DTR mice and infected with a persistent MNV strain. Viral titres in the intestine and secondary lymphoid organs were determined at early time points during infection, and anti-MNV antibody responses were analysed later during infection. Depletion of conventional DCs resulted in increased viral loads in intestinal tissues, impaired generation of antibody responses, and a failure of MNV to efficiently infect lymphoid tissues. These data suggest that DCs play multiple roles in MNV pathogenesis, in both innate immunity and the efficient generation of adaptive immune responses against MNV, as well as by promoting the dissemination of MNV to secondary lymphoid tissues. This is the first study to probe the roles of DCs in controlling and/or facilitating a norovirus infection in vivo and provides the basis for further studies aimed at defining mechanisms by which DCs control MNV replication and promote viral dissemination.
Introduction
Murine norovirus (MNV) is a small non-enveloped, plus-sense RNA virus in the genus Norovirus within the Caliciviridae family (Karst et al., 2003). This gastrointestinal virus is the single most widespread pathogen found in research mouse colonies worldwide (Pritchett-Corning et al., 2009). In vitro, MNV productively infects mononuclear antigen-presenting cells including macrophages, dendritic cells (DCs) (Wobus et al., 2004) and microglia (Cox et al., 2009). In vivo, MNV alters DC populations (Chen et al., 2011). Although wild-type mice show little to no outward signs of MNV infection (Mumphrey et al., 2007), confounding effects of several MNV strains on biomedical research have been described (Cadwell et al., 2010; Lencioni et al., 2008; Paik et al., 2011). Several of these confounding effects have been linked to disrupted DC and macrophage function in MNV-infected animals (Lencioni et al., 2008; Paik et al., 2011).
The earliest description of MNV established a critical role for interferon (IFN) signalling in providing protection from a potentially lethal MNV-1 infection (Karst et al., 2003). More recent studies have found that IFN-α/β responses limit the replication of the tissue-culture adapted strain MNV-1 in lysozyme M- (e.g. macrophages) and CD11c- (i.e. DCs) expressing cells in vivo (Thackray et al., 2012). Antibodies are required for clearance of MNV-1 (Chachu et al., 2008b), whereas CD4+ and CD8+ T-lymphocytes limit peak levels of MNV-1 replication and facilitate the efficient clearance of MNV, respectively, but are not absolutely required for clearance (Chachu et al., 2008a). However, the role(s) of DCs during MNV infection remain unknown.
DCs, the most potent class of antigen-presenting cells, are widely distributed throughout the body and are found in lymphoid tissues, such as the spleen and lymph nodes, as well as in peripheral tissues including the intestinal tract. Within intestinal tissues, DCs exist within Peyer’s patches and isolated lymphoid follicles and are scattered throughout the lamina propria (Rescigno, 2010). Intestinal DCs have critical roles in promoting immunity to intestinal pathogens but must avoid generating pathological immune responses against food antigens and commensal microbes (Denning et al., 2007). In addition, certain subsets of DCs extrude dendrites between intestinal epithelial cells to the intestinal lumen and may provide an initial point-of-contact for incoming pathogens (Niess et al., 2005). Despite the importance of DCs within the intestine and their ability to support MNV replication, the roles of DCs in MNV infection in vivo are unknown. As cells with critical functions in innate antiviral immunity in general (Kassim et al., 2006) and MNV in particular (Thackray et al., 2012), and the ability to prime adaptive immune responses in vivo (Jung et al., 2002), DCs likely play important roles in controlling MNV replication and inducing anti-MNV immune responses. Conversely, while both macrophages and DCs are found in subepithelial tissues, DCs are the only cell type permissive for MNV known to make direct contact with the intestinal lumen (Niess et al., 2005) and may have critical roles in the establishment and dissemination of MNV infection.
The goal of this study was to determine the role(s) of conventional DCs (cDCs) during MNV infection by studying MNV pathogenesis in transgenic CD11c-DTR mice. These mice express a high-affinity simian diphtheria toxin (DT) receptor under the control of the murine CD11c promoter (Jung et al., 2002). CD11c is an integrin that is predominantly expressed by DCs, and when exogenous DT is administered to CD11c-DTR mice, DCs are systemically ablated (Jung et al., 2002). Importantly, cDCs, which are most classically known for their antigen-presenting functions, are efficiently depleted in these mice, whereas plasmacytoid DCs, which express low levels of CD11c and are known for their ability to rapidly secrete large quantities of type I IFN, remain intact (Sapoznikov et al., 2007). In this study, cDCs were depleted from mice, and MNV infection was analysed in the absence of cDCs. The results demonstrated that following depletion, MNV viral titres in intestinal tissues were increased early during infection, and mice had defects in the subsequent generation of anti-MNV antibody responses. In addition, cDCs were absolutely required for the efficient dissemination of MNV to secondary lymphoid tissues within the first 48 h following infection. Together, these data suggest that while cDCs play important roles in immunity to norovirus infection, MNV exploits the natural biology of cDCs to rapidly disseminate within the host.
Results
DC depletion increases MNV titres within intestinal tissues
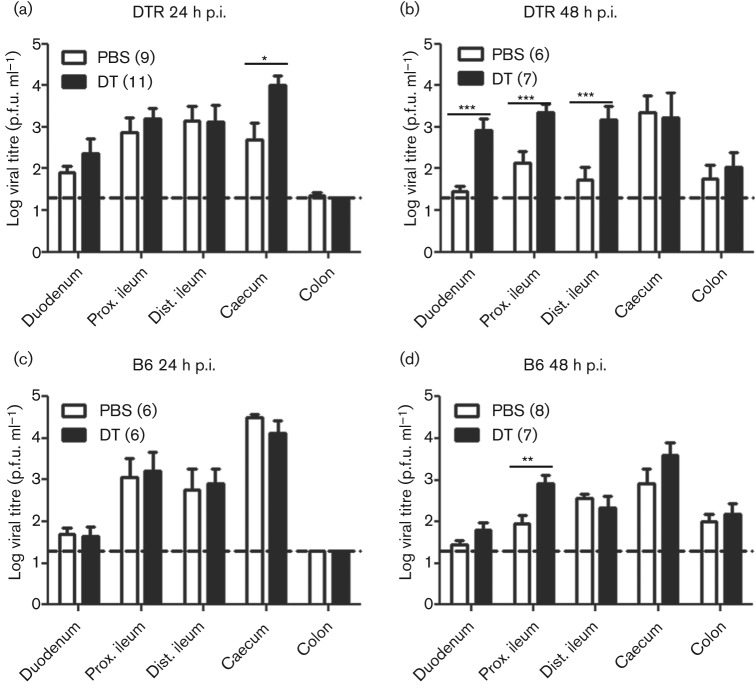
To determine the role of cDCs during MNV infection, CD11c-DTR mice were depleted of cDCs and orally infected with MNV strain CR3. CR3 was isolated from the faeces of mice and is shed from mice for at least 35 days (Thackray et al., 2007). Efficient cDC depletion in these mice initially occurs 18 h after DT administration (Probst et al., 2005). To confirm the efficient depletion of DCs from lymphoid and intestinal tissues throughout the experimental time course, CD11c-DTR mice were injected with DT or PBS and the presence of DCs in spleens, mesenteric lymph nodes (MLNs), and intestinal tissues was determined by flow cytometry after 66 h [i.e. the time point equivalent to 48 h post-infection (p.i.)]. The 66 h time point was selected because this is the time by which DCs are reported to begin repopulating tissues by differentiating from the bone marrow (Probst et al., 2005), and because this was the latest time point employed in studies of acute MNV infection (see below). Therefore, the depletion observed at the 66 h time point represented the minimal depletion that occurs during the acute MNV experiments. By administering a single intraperitoneal injection of 100 ng DT per mouse, CD11c+ DCs were efficiently depleted from spleens (~70 % depletion), MLNs (~55 % depletion) and small and large intestinal tissues (~75 % depletion) (Fig. 1).
Fig. 1.
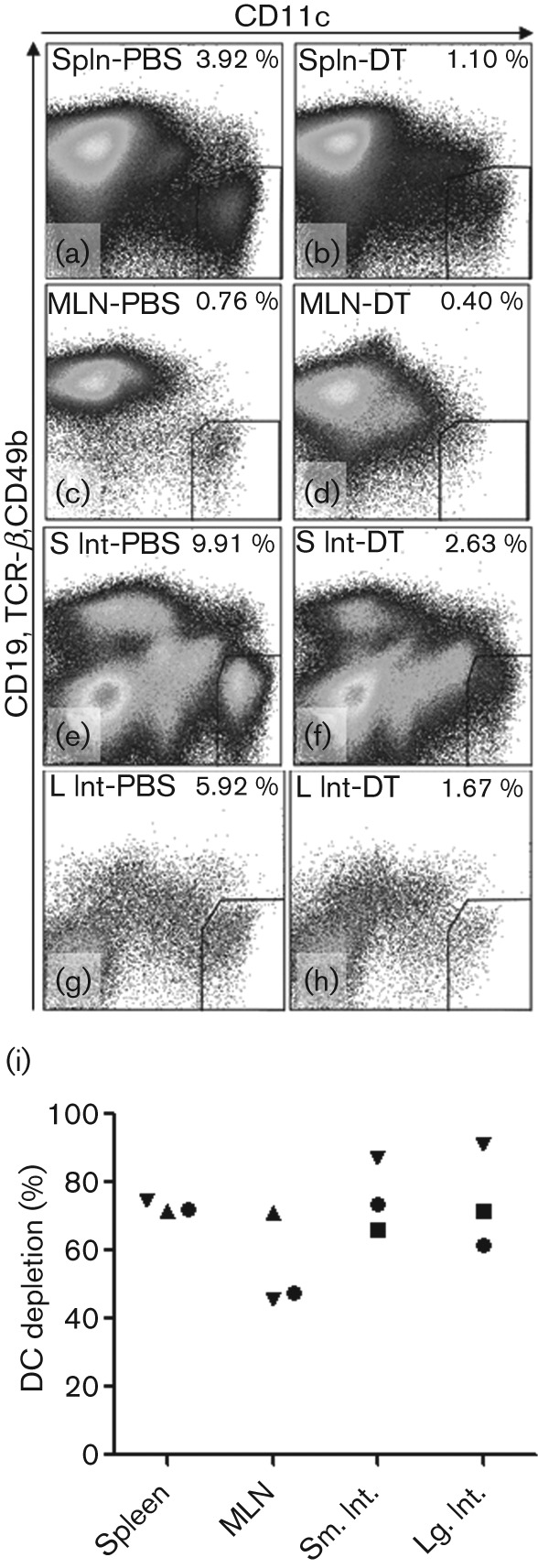
DT efficiently depleted DCs from lymphoid and gastrointestinal tissues of CD11c-DTR mice. (a–h) Mice were treated with 100 ng DT or 50 µl PBS by i.p. injection. Spleen (Spln), mesenteric lymph nodes (MLN), small (S) and large (L) intestinal (Int) tissues were removed 66 h later and single-cell suspensions stained for CD19, CD49b, and TCR-β (to exclude B-, NK and T-cells) and CD11c to identify DCs. Representative flow plots from PBS-treated (a, c, e, g) and DT-treated (b, d, f, h) mice are shown. (i) The efficiency of depletion in DT-treated mice relative to PBS-treated controls is shown, with each symbol representing the average depletion of the indicated tissue from an independent experiment.
To determine the role of cDCs in controlling MNV within intestinal tissues, DT- or PBS-treated CD11c-DTR mice were orally infected with CR3 18 h after DT treatment, and tissues of the intestinal tract were collected at 24 and 48 h p.i. (or 42 and 66 h after DT administration). By 24 h p.i., the amount of infectious virus within the caeca of mice that had been depleted of cDCs was significantly increased (~20-fold) (Fig. 2a). By 48 h p.i., titres in the caeca were similar in DC- and mock-depleted mice, but DT-treated mice showed >15- to 30-fold increases in the amount of infectious virus throughout the small intestine (Fig. 2b). No significant changes were observed in viral titres in the colon at either time point in both treatment groups (Fig. 2a, b). Furthermore, there was no effect of DT administration in wild-type mice at 24 h (Fig. 2c) or at 48 h with the exception of the proximal ileum (Fig. 2d). MNV titres in the proximal ilea of DT-treated wild-type mice were slightly increased (<9-fold) relative to PBS-treated animals (Fig. 2d), but titres in the proximal ilea of CD11c-DTR mice were increased to a greater degree (>15-fold) in DT-treated mice (Fig. 2b). This suggested that the majority of the toxin’s effects observed in CD11c-DTR mice were mediated by DC depletion. Taken together, these data showed that DC depletion increases MNV replication within intestinal tissues and suggests that DCs are critical for limiting MNV replication in the intestine.
Fig. 2.
DCs control MNV replication within the intestinal tract. CD11c-DTR mice (a, b) or littermates (c, d) were treated with PBS (white) or DT (black) 18 h prior to infection with 7.5×106 p.f.u. CR3. Intestinal tissues were harvested 24 (a, c) or 48 (b, d) h p.i. Viral titres were determined by plaque assay. The number of mice in each group is indicated in parentheses. Data are shown as mean±sem for three independent experiments. The dashed line indicates the limit of detection. *P<0.05, **P<0.01, ***P<0.001.
DCs are required for dissemination of MNV to secondary lymphoid tissues
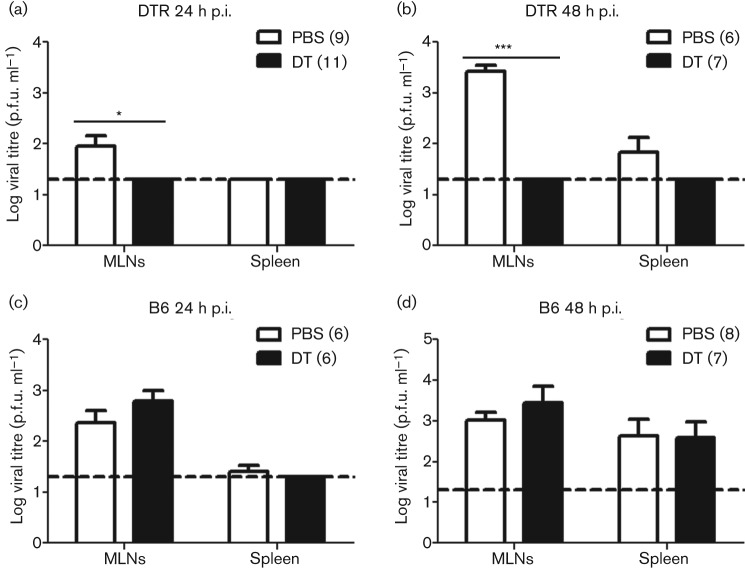
Since cDC depletion led to increased MNV replication in the intestine, we next sought to determine the extent to which cDCs control MNV replication in secondary lymphoid tissues, specifically the spleen and the MLNs, which drain the intestinal tract. Infectious virus was measured in the spleen and MLNs from the same animals described above. Interestingly, no infectious virus was ever recovered from either the spleen or MLNs of CD11c-DTR mice depleted of DCs (Fig. 3a, b). This was in contrast to up to 3.5 logs infectious virus in the MLNs of PBS-depleted CD11c-DTR mice at 48 h p.i. (Fig. 3b). While similar trends were observed for the spleen, MNV titres in the spleens of depleted and non-depleted CD11c-DTR mice did not reach statistical significance at 24 or 48 h p.i. (Fig. 3b). DT administration did not impair dissemination of MNV to lymphoid tissues in wild-type littermates (Fig. 3c, d). These data suggested that dissemination of MNV to secondary lymphoid organs required, and was mediated by, cDCs.
Fig. 3.
DCs are required for efficient dissemination of MNV to secondary lymphoid tissues. CD11c-DTR mice (a, b) or littermates (c, d) were treated with PBS (open bars) or DT (filled bars) 18 h prior to infection with 7.5×106 p.f.u. CR3. Spleens and MLN were harvested at 24 (a, c) or 48 (b, d) h p.i. and viral titres determined by plaque assay. The number of mice in each group is indicated in parentheses. Data are shown as means±sem for three independent experiments. The dashed line indicates the limit of detection. *P<0.05, ***P<0.001.
DC-depleted mice have increased MNV shedding in faeces
The cell type and location of MNV shedding are unknown. To determine whether cDCs were an important source of transmissible virus, cDC-depleted and non-depleted CD11c-DTR mice were infected with MNV as described above, and viral shedding in faeces was determined by measuring viral genomes in faeces by quantitative reverse transcription PCR (qRT-PCR) at 1, 2, 4, 7, 21 and 28 days p.i. (Fig. 4). Viral shedding in cDC-depleted mice was significantly increased at 2 days p.i., but became similar to shedding in non-depleted animals thereafter. These data demonstrate that transient DC depletion does not prevent shedding of MNV in faeces. Instead, increased numbers of viral genome were shed in the faeces of depleted mice, which is likely a result of increased viral replication within the intestinal tract.
Fig. 4.
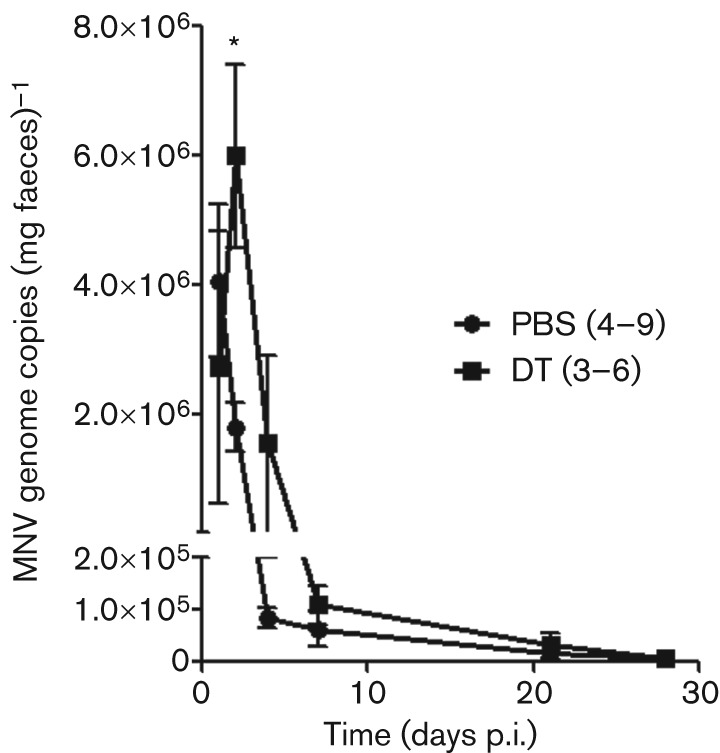
DC depletion transiently increases MNV shedding. CD11c-DTR mice were treated with PBS (•) or DT (▪) 18 h prior to infection with 7.5×106 p.f.u. CR3. Faeces were collected from mice at 1, 2, 4, 7, 21 and 28 days p.i. and MNV RNA was quantified by qRT-PCR. The number of mice in each group is indicated in parentheses. Data are shown as means±sem for three independent experiments. *P<0.05.
DCs are required for the efficient generation of antibodies against MNV
In addition to their functions in innate immunity, DCs are important stimulators of adaptive immune responses (Jung et al., 2002). To determine whether cDCs contribute to adaptive immune responses against MNV, DC-depleted and non-depleted CD11c-DTR mice were infected with MNV, and anti-MNV IgG in the serum was measured prior to infection and 4, 7, 14, 21 and 28 days p.i. by ELISA. In the absence of cDCs, mice had significantly less anti-MNV IgG for the first 2 weeks following infection (Fig. 5a). In addition, when sera from CD11c-DTR mice depleted of cDCs were tested for their ability to neutralize MNV infection in tissue culture, sera had significantly decreased ability to neutralize MNV at 4 and 7 days p.i. (Fig. 5b). These data suggested that the generation of neutralizing antibodies was delayed and indicated that cDCs are critical for the efficient generation of antibody responses against MNV.
Fig. 5.
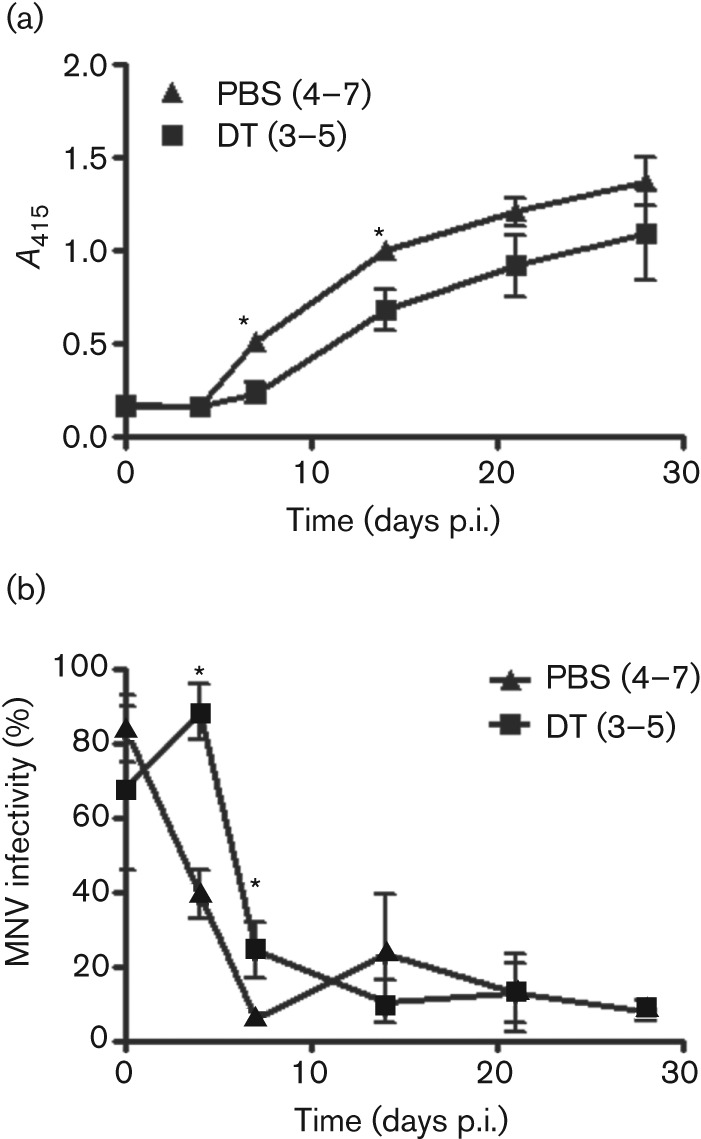
DCs facilitate the efficient generation of anti-MNV antibodies. Mice were treated with PBS or DT 18 h prior to infection with 7.5×106 p.f.u. CR3. Blood was collected from mice prior to infection and 4, 7, 14, 21, and 28 days p.i. (a) Anti-MNV IgG was measured by ELISA using 1 : 100 dilutions of sera. (b) To measure the ability of serum to neutralize MNV, 104 p.f.u. CR3 was incubated with a 1 : 100 dilution of serum in a volume of 100 µl. Infectious virus was detected by plaque assay and is presented as a percentage of input virus. The number of mice in each group is indicated in parentheses. Data are shown as mean±sem for three independent experiments. *P<0.05.
Discussion
MNV represents a versatile model for the analysis of mechanisms of pathogenesis and immunity for a norovirus in its natural host (Wobus et al., 2006). While the roles of IFN, B-cells and T-cells have been described previously (Chachu et al., 2008a; Chachu et al., 2008b; Mumphrey et al., 2007; Thackray et al., 2012), this is the first study to describe the dual role of cDCs in both controlling and promoting MNV infection. Furthermore, this study elucidated distinct roles for cDCs in innate and adaptive immunity and the dissemination of MNV.
A main finding of this study was that MNV titres in the intestinal tract were increased significantly for the first 48 h following MNV infection in mice depleted of cDCs (Fig. 2). While cDCs are best known for their ability to present antigen and prime T-cells (Jung et al., 2002), they also possess important roles in innate immunity (Kassim et al., 2006; Loof et al., 2007). For example, cDCs are important producers of type I IFN at early times following adenovirus infection (Fejer et al., 2008). Since IFN are important for protection from MNV lethality (Karst et al., 2003), and type I IFN responses within macrophages and DCs limit MNV infection in vivo (Thackray et al., 2012), it is likely that a lack of cDC-derived IFN in the intestinal tissues resulted in the observed increase in MNV replication. Due to the short time frame, it is unlikely that the observed increases in viral titres were the result of impaired T-cell-mediated immunity from a lack of antigen presentation.
An alternative explanation for the increased viral titres within the intestinal tract is that MNV could have increased replication within cells that are recruited to tissues in which DCs have been depleted. However, we think that this alternative is less likely. First, it has recently been shown that CD11c-DTR mice undergo a strong neutrophilia within the first 72 h following DC depletion (Tittel et al., 2012); however, there is no evidence that MNV is able to replicate within neutrophils. Secondly, CD11b+ CD11c− myeloid cell populations (which could include macrophages, monocytes and neutrophils) were quantified in PBS- and DT-treated mice in the current study, and although the proportion of these cells was increased by roughly threefold in the spleen and MLNs, intestinal tissues had only a slight increase (1.2–1.4-fold) in the proportion of CD11b+ CD11c− cells (data not shown). In contrast, viral titres were increased 15–30-fold in intestinal tissues. If the increased viral titres were due to a greater frequency of permissive cells, then one would expect a significantly more robust recruitment of cells into intestinal tissues.
In contrast to the increased viral titres seen in the intestinal tract, no infectious MNV was detected in MLNs in the absence of cDCs for the first 48 h following MNV infection (Fig. 3). The most plausible explanation for this finding is that cDCs were infected by MNV in either the lamina propria or in the Peyer’s patches and subsequently migrated to the draining MLNs of non-depleted mice while carrying infectious virus. CD103+ DCs constitutively migrate from the intestine to MLNs and are thought to play a role in maintaining tolerance to commensal bacteria and/or food-derived antigens (Coombes et al., 2007). These cells have previously been implicated in facilitating the dissemination of a non-invasive strain of Salmonella from the intestinal tract to the MLNs (Bogunovic et al., 2009). In addition, migratory DCs have been demonstrated or implicated to promote dissemination of viral pathogens as well, including influenza virus (Moltedo et al., 2011), human immunodeficiency virus (Geijtenbeek et al., 2000) and the enteric pathogen mouse mammary tumour virus (Courreges et al., 2007). Taken together, these studies support the hypothesis whereby migratory DCs provide a mechanism by which MNV disseminates from the initial sites of replication to the draining MLNs. However, future studies are needed to test whether CD103+ DCs or other subsets of migratory DCs provide the mechanism for dissemination of MNV in vivo.
Our data also provide evidence that MNV replication in DCs is not required for MNV shedding. Similar or higher amounts of MNV genomes were detected in the faeces of DC-depleted mice compared with non-depleted mice (Fig. 4), suggesting transient DC depletion was insufficient to limit MNV transmission. In addition, replication of MNV in the MLNs was not required for MNV shedding early during the infection since MNV was unable to replicate in the MLNs during the first 48 h following infection (Fig. 3) at times when MNV shedding occurred (Fig. 4). Interestingly, the significant increase in MNV shedding observed on day 2 was temporally associated with an increased amount of infectious MNV within the intestines (compare Figs 2 and 4). This transient increase in shedding was likely due to the transience of cDC depletion in the CD11c-DTR model (Probst et al., 2005). Thus, as cDCs repopulated the tissues over time, ‘normal’ control of MNV replication was probably restored and MNV shedding reduced to amounts found in the faeces of non-depleted mice. Future studies are needed to determine the mechanism of MNV shedding.
Beyond the immediate effects of transient cDC depletion on innate immunity to MNV and dissemination to secondary lymphoid tissues, DCs were also required for the efficient generation of an antibody response against MNV (Fig. 5). There are several possible mechanisms that may underlie the observed impairment of an antibody response against MNV. Since DCs are important for priming CD4+ T-cell responses (Lemos et al., 2003), it is likely that T-cell responses against MNV are impaired in DC-depleted mice. However, CD4+ T-cells are not required for clearance of the acute, laboratory-adapted strain MNV-1 (Chachu et al., 2008a), while B-cells are required (Chachu et al., 2008b), suggesting that B-cell responses to MNV can occur in the absence of CD4+ T-cell help. If this is the case, then it is possible that the impaired antibody response observed in depleted animals was not due to impaired antigen presentation. However, it is important to note that this study used the field isolate, CR3, which is not cleared from wild-type mice (Thackray et al., 2007). Thus, the role of T-cells in B-cell activation may differ between MNV strains. In our study, we found that serum concentrations of anti-MNV IgG were reduced in DT-treated mice, while the ability of their sera to neutralize MNV occurred to a similar extent, albeit with delayed kinetics, relative to PBS-treated controls (Fig. 5b). This supports a role for DC in the generation of a T-cell-dependent antibody response. It is also possible that B-cell responses were impaired because of a lack of MNV antigen within the MLNs in DC-depleted mice. If this is the case, then the ‘failure’ of MNV to disseminate may not have been beneficial for the host, but instead might have resulted in weaker control of MNV by the adaptive immune system. Alternatively, DCs may produce cytokines that are necessary for the optimal activation of B-cell responses (Zhu et al., 2007). DC-dependent antibody responses to influenza (Gonzalez et al., 2010) and Friend murine leukemia virus (Browne & Littman, 2009) have been described to require diverse DC functions including pathogen detection and antigen transport. However, the exact role of DCs in promoting B-cell responses to MNV remains unknown.
Overall, we have found evidence that DCs have differing roles at distinct anatomical sites during MNV infection. DC depletion led to impaired control of MNV replication within the intestinal tract and weaker and slower antibody responses, but DCs were required for the rapid dissemination of MNV to secondary lymphoid tissues. These studies highlight a critical innate role for cDCs in controlling norovirus replication within intestinal tissues, while reinforcing the idea that enteric viruses can exploit cDCs to facilitate their dissemination within the host.
Methods
Cell culture, mice, and virus.
RAW 264.7 cells were purchased from the ATCC and maintained as described previously (Wobus et al., 2004). Wild-type C57BL/6 mice were purchased from the Jackson Laboratory (000664). CD11c-DTR mice (Jung et al., 2002) were purchased from the Jackson Laboratory (004509) and bred at the University of Michigan animal facilities. All mice in this study were cared for and used in accordance with federal and university guidelines. All mice were tested for anti-MNV antibodies by ELISA as described previously (Wobus et al., 2004) and were seronegative. MNV strain CR3 (GV/CR3/2005/USA) was used at passage 6 (Thackray et al., 2007). For in vivo infections, CR3 lysate was concentrated by ultracentrifugation and resuspended in PBS as previously described (Chachu et al., 2008b). A mock lysate was similarly concentrated and used as control.
DC depletion and MNV infection.
DCs were depleted from CD11c-DTR mice by intraperitoneal injection of 100 ng DT in a volume of 50 µl PBS per mouse (John et al., 2009). Control mice received an injection of PBS instead. Eighteen hours after DT or PBS treatment, mice were orally infected with 7.5×106 p.f.u. CR3 in a volume of 25 µl PBS or an equal volume of concentrated mock lysate.
Leukocyte isolation.
To determine depletion efficiency, spleens, MLNs and small and large intestines were removed from DT- or PBS-treated CD11c-DTR mice 66 h after DT administration. Spleens and MLNs were finely minced, digested using 2 mg collagenase D ml−1 (Roche) for 30 min at 37 °C, and disrupted by pushing through 40 µm cell strainers to obtain single-cell suspensions. Red cells were lysed by treating splenocytes with ACK lysing buffer (BioWhittaker) for 5 min at room temperature. To obtain intestinal leukocytes, intestinal tissues were washed extensively and minced. Lamina propria mononuclear cells (LPMCs) were isolated from large intestines as described previously (Franchi et al., 2012). LPMCs were isolated from small intestines in a similar manner. Small intestinal tissues were treated with 1 mM dithiothreitol for 15 min to remove mucus. Intestinal tissues were then washed three times with 5 mM EDTA at 37 °C for a total of 90 min to remove epithelial cells. Intestinal tissues were then digested using 200 U collagenase, type 3 ml−1 and 0.01 mg DNase I ml−1 (both from Worthington) for 90–180 min. Digested intestinal cell suspensions were filtered through 70 µm cell strainers, centrifuged on a 40 : 75 % Percoll gradient (GE Life Sciences), and LPMCs were collected from the interface, similar to cells from the large intestine.
Flow cytometry.
Splenocytes, MLN cells and intestinal leukocytes were treated with 2.4G2 hybridoma supernatant to block FC receptor-mediated antibody binding (Unkeless, 1979). Cells were then stained for markers of B-cells (CD19, clone 6D5; BioLegend), NK cells (CD49b, clone DX5; BioLegend) and T-cells (TCR-β, clone H57-597; BioLegend) to exclude these cells from further analysis and CD11c (clone N418; eBioscience) to identify DCs. Additional myeloid cells (monocytes, macrophages, or neutrophils) were identified from the CD11c− population by staining for CD11b (clone M1/70, BD Pharmingen). Flow cytometry was performed using a FACSCanto (Becton Dickinson) and data were analysed using FlowJo software (Treestar). DCs were identified as CD11c+, CD19−, CD49b−, TCR-β− cells.
Quantification of MNV.
At 24 or 48 h p.i., tissues were removed, including duodenum, proximal and distal ileum, caecum, colon, spleen and MLN. The entire MLN, one-half of the caecum and spleen, and 1 cm of various portions of the intestinal tract were homogenized in 1 ml medium. To measure infectious virus, plaque assays were performed on RAW 264.7 cells as previously described (Gonzalez-Hernandez et al., 2012; Mumphrey et al., 2007; Wobus et al., 2004). Titres are presented as p.f.u. ml−1. To measure genome titres in faeces, samples were submitted to the Research Animal Diagnostic Services (Charles River Laboratories, Wilmington, MA) for qRT-PCR and analysed as described previously (Thackray et al., 2007).
Serology.
Serum was collected from DT- or PBS-treated mice prior to and at various times following CR3 infection by retro-orbital bleeding. CR3 and MNV-1 are part of a single serotype (Thackray et al., 2007). Thus, ELISA was performed on serially diluted sera using plates coated with CsCl-purified MNV-1 as described (Wobus et al., 2004). For microneutralization experiments, a 1 : 100 dilution of serum was incubated with 104 p.f.u. CR3 in a volume of 100 µl for 30 min at room temperature. Infectious virus was then measured by plaque assay as previously described (Gonzalez-Hernandez et al., 2012). The percentage infectivity was calculated by dividing the observed titre of serum-treated virus by the titre of virus incubated with PBS.
Acknowledgements
This work was supported by NIH grants AI080611 to C. E. W. and DK091191 to G. N. M. D. E. was supported by the University of Michigan Experimental Immunology Training Grant (2 T32 AI007413-18). We would like to thank the University of Michigan Unit for Laboratory Animal Medicine and the technical services team for assistance with animal husbandry and blood collection, and Thomas Liu, Tracey Schultz and Marta J. Gonzalez-Hernandez for assistance with experiments. We are also grateful to Dr Mengxi Jiang (University of Michigan) for critical reading of the manuscript.
References
- Bogunovic M., Ginhoux F., Helft J., Shang L., Hashimoto D., Greter M., Liu K., Jakubzick C., Ingersoll M. A. & other authors (2009). Origin of the lamina propria dendritic cell network. Immunity 31, 513–525 10.1016/j.immuni.2009.08.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Browne E. P., Littman D. R. (2009). Myd88 is required for an antibody response to retroviral infection. PLoS Pathog 5, e1000298 10.1371/journal.ppat.1000298 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cadwell K., Patel K. K., Maloney N. S., Liu T. C., Ng A. C., Storer C. E., Head R. D., Xavier R., Stappenbeck T. S., Virgin H. W. (2010). Virus-plus-susceptibility gene interaction determines Crohn’s disease gene Atg16L1 phenotypes in intestine. Cell 141, 1135–1145 10.1016/j.cell.2010.05.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chachu K. A., LoBue A. D., Strong D. W., Baric R. S., Virgin H. W. (2008a). Immune mechanisms responsible for vaccination against and clearance of mucosal and lymphatic norovirus infection. PLoS Pathog 4, e1000236 10.1371/journal.ppat.1000236 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chachu K. A., Strong D. W., LoBue A. D., Wobus C. E., Baric R. S., Virgin H. W., IV (2008b). Antibody is critical for the clearance of murine norovirus infection. J Virol 82, 6610–6617 10.1128/JVI.00141-08 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen X., Leach D., Hunter D. A., Sanfelippo D., Buell E. J., Zemple S. J., Grayson M. H. (2011). Characterization of intestinal dendritic cells in murine norovirus infection. Open Immunol J 4, 22–30 10.2174/1874226201104010022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coombes J. L., Siddiqui K. R., Arancibia-Cárcamo C. V., Hall J., Sun C. M., Belkaid Y., Powrie F. (2007). A functionally specialized population of mucosal CD103+ DCs induces Foxp3+ regulatory T cells via a TGF-β and retinoic acid-dependent mechanism. J Exp Med 204, 1757–1764 10.1084/jem.20070590 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Courreges M. C., Burzyn D., Nepomnaschy I., Piazzon I., Ross S. R. (2007). Critical role of dendritic cells in mouse mammary tumor virus in vivo infection. J Virol 81, 3769–3777 10.1128/JVI.02728-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cox C., Cao S., Lu Y. (2009). Enhanced detection and study of murine norovirus-1 using a more efficient microglial cell line. Virol J 6, 196 10.1186/1743-422X-6-196 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Denning T. L., Wang Y. C., Patel S. R., Williams I. R., Pulendran B. (2007). Lamina propria macrophages and dendritic cells differentially induce regulatory and interleukin 17-producing T cell responses. Nat Immunol 8, 1086–1094 10.1038/ni1511 [DOI] [PubMed] [Google Scholar]
- Fejer G., Drechsel L., Liese J., Schleicher U., Ruzsics Z., Imelli N., Greber U. F., Keck S., Hildenbrand B. & other authors (2008). Key role of splenic myeloid DCs in the IFN-αβ response to adenoviruses in vivo. PLoS Pathog 4, e1000208 10.1371/journal.ppat.1000208 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franchi L., Kamada N., Nakamura Y., Burberry A., Kuffa P., Suzuki S., Shaw M. H., Kim Y. G., Núñez G. (2012). NLRC4-driven production of IL-1β discriminates between pathogenic and commensal bacteria and promotes host intestinal defense. Nat Immunol 13, 449–456 10.1038/ni.2263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geijtenbeek T. B., Kwon D. S., Torensma R., van Vliet S. J., van Duijnhoven G. C., Middel J., Cornelissen I. L., Nottet H. S., KewalRamani V. N. & other authors (2000). DC-SIGN, a dendritic cell-specific HIV-1-binding protein that enhances trans-infection of T cells. Cell 100, 587–597 10.1016/S0092-8674(00)80694-7 [DOI] [PubMed] [Google Scholar]
- Gonzalez S. F., Lukacs-Kornek V., Kuligowski M. P., Pitcher L. A., Degn S. E., Kim Y. A., Cloninger M. J., Martinez-Pomares L., Gordon S. & other authors (2010). Capture of influenza by medullary dendritic cells via SIGN-R1 is essential for humoral immunity in draining lymph nodes. Nat Immunol 11, 427–434 10.1038/ni.1856 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonzalez-Hernandez M. B., Bragazzi Cunha J., Wobus C. E. (2012). Plaque assay for murine norovirus. J Vis Exp 66, e4297. 10.3791/4297 [DOI] [PMC free article] [PubMed] [Google Scholar]
- John B., Harris T. H., Tait E. D., Wilson E. H., Gregg B., Ng L. G., Mrass P., Roos D. S., Dzierszinski F. & other authors (2009). Dynamic Imaging of CD8+ T cells and dendritic cells during infection with Toxoplasma gondii. PLoS Pathog 5, e1000505 10.1371/journal.ppat.1000505 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jung S., Unutmaz D., Wong P., Sano G., De los Santos K., Sparwasser T., Wu S., Vuthoori S., Ko K. & other authors (2002). In vivo depletion of CD11c+ dendritic cells abrogates priming of CD8+ T cells by exogenous cell-associated antigens. Immunity 17, 211–220 10.1016/S1074-7613(02)00365-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karst S. M., Wobus C. E., Lay M., Davidson J., Virgin H. W., IV (2003). STAT1-dependent innate immunity to a Norwalk-like virus. Science 299, 1575–1578 10.1126/science.1077905 [DOI] [PubMed] [Google Scholar]
- Kassim S. H., Rajasagi N. K., Zhao X., Chervenak R., Jennings S. R. (2006). In vivo ablation of CD11c-positive dendritic cells increases susceptibility to herpes simplex virus type 1 infection and diminishes NK and T-cell responses. J Virol 80, 3985–3993 10.1128/JVI.80.8.3985-3993.2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lemos M. P., Fan L., Lo D., Laufer T. M. (2003). CD8alpha+ and CD11b+ dendritic cell-restricted MHC class II controls Th1 CD4+ T cell immunity. J Immunol 171, 5077–5084 [DOI] [PubMed] [Google Scholar]
- Lencioni K. C., Seamons A., Treuting P. M., Maggio-Price L., Brabb T. (2008). Murine norovirus: an intercurrent variable in a mouse model of bacteria-induced inflammatory bowel disease. Comp Med 58, 522–533 [PMC free article] [PubMed] [Google Scholar]
- Loof T. G., Rohde M., Chhatwal G. S., Jung S., Medina E. (2007). The contribution of dendritic cells to host defenses against Streptococcus pyogenes. J Infect Dis 196, 1794–1803 10.1086/523647 [DOI] [PubMed] [Google Scholar]
- Moltedo B., Li W., Yount J. S., Moran T. M. (2011). Unique type I interferon responses determine the functional fate of migratory lung dendritic cells during influenza virus infection. PLoS Pathog 7, e1002345 10.1371/journal.ppat.1002345 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mumphrey S. M., Changotra H., Moore T. N., Heimann-Nichols E. R., Wobus C. E., Reilly M. J., Moghadamfalahi M., Shukla D., Karst S. M. (2007). Murine norovirus 1 infection is associated with histopathological changes in immunocompetent hosts, but clinical disease is prevented by STAT1-dependent interferon responses. J Virol 81, 3251–3263 10.1128/JVI.02096-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niess J. H., Brand S., Gu X., Landsman L., Jung S., McCormick B. A., Vyas J. M., Boes M., Ploegh H. L. & other authors (2005). CX3CR1-mediated dendritic cell access to the intestinal lumen and bacterial clearance. Science 307, 254–258 10.1126/science.1102901 [DOI] [PubMed] [Google Scholar]
- Paik J., Fierce Y., Mai P. O., Phelps S. R., McDonald T., Treuting P., Drivdahl R., Brabb T., LeBoeuf R. & other authors (2011). Murine norovirus increases atherosclerotic lesion size and macrophages in Ldlr−/− mice. Comp Med 61, 330–338 [PMC free article] [PubMed] [Google Scholar]
- Pritchett-Corning K. R., Cosentino J., Clifford C. B. (2009). Contemporary prevalence of infectious agents in laboratory mice and rats. Lab Anim 43, 165–173 10.1258/la.2008.008009 [DOI] [PubMed] [Google Scholar]
- Probst H. C., Tschannen K., Odermatt B., Schwendener R., Zinkernagel R. M., Van Den Broek M. (2005). Histological analysis of CD11c-DTR/GFP mice after in vivo depletion of dendritic cells. Clin Exp Immunol 141, 398–404 10.1111/j.1365-2249.2005.02868.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rescigno M. (2010). Functional specialization of antigen presenting cells in the gastrointestinal tract. Curr Opin Immunol 22, 131–136 10.1016/j.coi.2009.12.007 [DOI] [PubMed] [Google Scholar]
- Sapoznikov A., Fischer J. A., Zaft T., Krauthgamer R., Dzionek A., Jung S. (2007). Organ-dependent in vivo priming of naive CD4+, but not CD8+, T cells by plasmacytoid dendritic cells. J Exp Med 204, 1923–1933 10.1084/jem.20062373 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thackray L. B., Wobus C. E., Chachu K. A., Liu B., Alegre E. R., Henderson K. S., Kelley S. T., Virgin H. W., IV (2007). Murine noroviruses comprising a single genogroup exhibit biological diversity despite limited sequence divergence. J Virol 81, 10460–10473 10.1128/JVI.00783-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thackray L. B., Duan E., Lazear H. M., Kambal A., Schreiber R. D., Diamond M. S., Virgin H. W. (2012). Critical role for interferon regulatory factor 3 (IRF-3) and IRF-7 in type I interferon-mediated control of murine norovirus replication. J Virol 86, 13515–13523 10.1128/JVI.01824-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tittel A. P., Heuser C., Ohliger C., Llanto C., Yona S., Hämmerling G. J., Engel D. R., Garbi N., Kurts C. (2012). Functionally relevant neutrophilia in CD11c diphtheria toxin receptor transgenic mice. Nat Methods 9, 385–390 10.1038/nmeth.1905 [DOI] [PubMed] [Google Scholar]
- Unkeless J. C. (1979). Characterization of a monoclonal antibody directed against mouse macrophage and lymphocyte Fc receptors. J Exp Med 150, 580–596 10.1084/jem.150.3.580 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wobus C. E., Karst S. M., Thackray L. B., Chang K. O., Sosnovtsev S. V., Belliot G., Krug A., Mackenzie J. M., Green K. Y., Virgin H. W. (2004). Replication of Norovirus in cell culture reveals a tropism for dendritic cells and macrophages. PLoS Biol 2, e432 10.1371/journal.pbio.0020432 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wobus C. E., Thackray L. B., Virgin H. W., IV (2006). Murine norovirus: a model system to study norovirus biology and pathogenesis. J Virol 80, 5104–5112 10.1128/JVI.02346-05 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu J., Huang X., Yang Y. (2007). Type I IFN signaling on both B and CD4 T cells is required for protective antibody response to adenovirus. J Immunol 178, 3505–3510 [DOI] [PubMed] [Google Scholar]