Abstract
Background
The role of bisphosphonates (BP) in early breast cancer (BC) has been considered controversial. We performed a meta-analysis of all randomized controlled trials (RCTs) that appraised the effects of BP on survival in early BC.
Methods
RCTs were identified by searching the Cochrane Library, MEDLINE databases and conference proceedings. Hazard ratios (HRs) of overall survival (OS), disease-free survival (DFS) and relative risks of adverse events were estimated and pooled.
Results
Thirteen trials met the inclusion criteria, evaluating a total of 15,762 patients. Meta-analysis of ten trials which reported OS revealed no statistically significant benefit in OS for BP (HR 0.89, 95% CI = 0.79 to 1.01). Meta-analysis of nine trials which reported the DFS revealed no benefit in DFS (HR 0.95 (0.81–1.12)). Meta-analysis upon menopausal status showed a statistically significant better DFS in the BP-treated patients (HR 0.81(0.69–0.95)). In meta-regression, chemotherapy was negatively associated with HR of survival.
Conclusions
Our meta-analysis indicates a positive effect for adjuvant BP on survival only in postmenopausal patients. Meta-regression demonstrated a negative association between chemotherapy use BP effect on survival. Further large scale RCTs are warranted to unravel the specific subgroups that would benefit from the addition of BP in the adjuvant setting.
Introduction
Breast cancer is the most common malignancy among women, accounting for nearly 1 in 3 cancers diagnosed and the second leading cause of cancer death among women in the United States [1]. Survival rates are highly correlated with the extent of the disease at diagnosis whereas early stage disease confers superior survival rates. The majority of patients with advanced breast cancer eventually develop bone metastases. Breast cancer has a particular propensity for the bone; likewise, it has been previously demonstrated that the relationship between cancer cells and the bone microenvironment is mediated by a reciprocal interaction between cancer cells and normal bone cells [2]–[4]. Due to their beneficial effects on bone turnover, bisphosphonates have been evaluated for the prevention and treatment of bone metastases in women with breast cancer [5]–[8]. Two large population-based cohort studies demonstrated a significant reduction in the incidence of breast cancer in women who were treated with BP for non-cancer indications for more than a year [9]–[10]. Both pre-clinical and clinical evidence indicate that BP exhibit anti-metastatic and anti-tumor properties, including the inhibition of angiogenesis, and a unique effect in the bony niche – inhibition of cancer cell invasion and adhesion and the induction of apoptosis [11]–[12]. The role of BP in addition to standard adjuvant treatment has been evaluated in several studies, yielding inconsistent evidence regarding the antitumor effect of BP [13]–[29].
In view of the conflicting data from randomized clinical trials, we performed a systematic review of the literature and a meta-analysis of all randomized trials to evaluate the impact of BP administration on survival in patients with early breast cancer in the adjuvant setting.
Methods
Data Sources
We searched the Cochrane Central Register of Controlled Trials, published in The Cochrane Library, PubMed (1966 to 4.2012); EMBASE (1974 to 7.2012); LILACS (1982 to 7.2012); the database of clinical trials in non-metastatic breast cancer; conference proceedings of the American Society of Clinical Oncology (1995–7.2012), San Antonio Breast Cancer Symposium (1995–2012), proceedings of the European Society of Medical Oncology (ESMO); and databases of ongoing and unpublished trials: http://www.clinicaltrials.gov and ,http://www.clinicaltrials.nci.nih.gov. The terms (early OR adjuvant) AND (breast OR mammary) AND (tumor OR malign* OR carcinoma* OR cancer) AND (biphosphonates OR bisphosphonates OR clodronate OR pamidronate OR zoledronic acid OR ibandronate) were cross-searched. We scanned references of all included trials and reviews identified for additional studies.
Study Selection
We included randomized controlled trials that compared BP adjuvant therapy in addition to the standard therapy (cytotoxic or hormonal) with placebo in patients with early breast cancer in the adjuvant setting. We included trials regardless of publication status, date of publication, and language. Two authors (IBA and LV) independently inspected each reference title identified by the search and applied the inclusion criteria.
Data Extraction and Quality Assessment
Trials that fulfilled the inclusion criteria were assessed for methodological quality by two authors (IBA and LV). Information about randomization and allocation concealment, blinding, sample size, exclusions after randomization, and the length of follow-up were recorded [30]. The same two authors independently extracted the data from publications of included trials. The data extraction was discussed, decisions were documented, and, if necessary, the authors of the trials were contacted for clarification. Authors of included trials were contacted for all data relevant to the primary and secondary outcomes (survival and safety data) of this study and quality variables. In case of several publications for the same trial, the most updated one was extracted. Safety outcomes were pooled from the most updated publication of every trial.
Outcome Measures
The primary outcome was overall survival, which was defined as time from randomization to death. Secondary outcomes were disease-free survival (defined as the time from randomization to earliest occurrence of relapse or death from any cause), and toxicity (defined as grade 3 or 4 hematological and nonhematological adverse events).
Data Synthesis and Statistical Analysis
Hazard ratios (HRs) and variances for time-to-event outcomes were estimated as described by Parmar et al. [31] and pooled according to inverse of variance method (Review Manager [RevMan], version 5.1(Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2011). A HR less than 1.0 was in favor of bisphosphonate therapy. Relative risks (RRs) and 95% confidence intervals (CIs) for dichotomous data were estimated using the Mantel – Haenszel method [31]. We used a fixed effect model. In case of performing a sensitivity analysis for the primary outcomes we repeated the above analysis using a random effects model (the DerSimonian and Laird method) [32]. Since the Mantel-Haenszel odds ratio method using a 0.5 zero-cell correction were shown to be biased when events are rare, we therefore performed for rare events as osteonecrosis of the jaw (ONJ) a sensitivity analysis by using Peto method [33]. We assessed heterogeneity of trial results by the chi test of heterogeneity and the I 2 statistic of inconsistency. Statistically significant heterogeneity was defined as P less than .1 or an I 2 statistic greater than 50% [32]. In case of a significant heterogeneity (I 2>50%) we used a random-effects model. Potential sources of heterogeneity were explored through stratifying by type of induction therapy (use of chemotherapy and endocrine therapy, endocrine therapy only, menopausal status), bisphosphonate's schedule and type of BP, allocation concealment, blinding, and size of studies. All statistical tests were two-sided.
We explored potential ground of heterogeneity through meta-regression, assessing the effect of percentage of patients receiving chemotherapy in addition to endocrine therapy in each study on the effect estimates for the primary outcome. Meta-regression was performed on the HR (Comprehensive Meta Analysis, version 2.2; BioStat, Englewood, NJ). The regression slope with its SE and significance of the model are reported.
Results
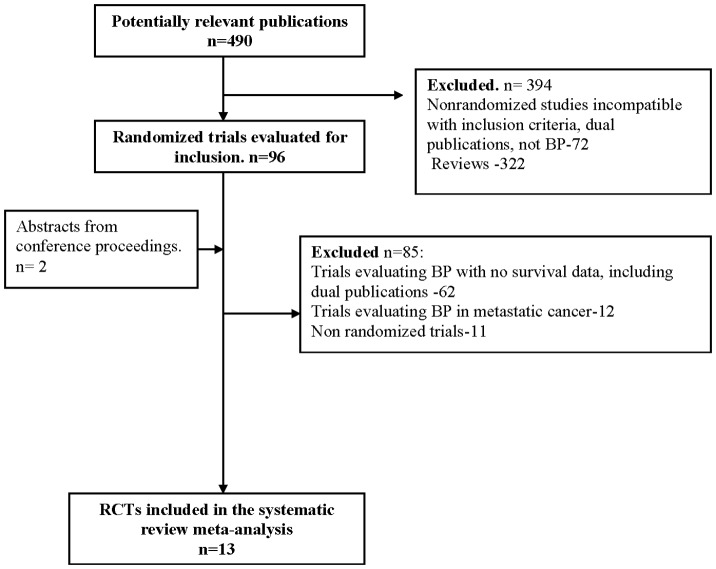
The literature search identified 490 trials up until 7.2012, of which 96 were considered potentially relevant. Additional trials were identified by searching conference proceedings and electronic resources of ongoing trials. Figure 1 illustrates the process of study selection. Thirteen trials were designed to evaluate the effect of bisphosphonates on survival and fulfilled the inclusion criteria for published studies ([13]–[29]; including safety reports) including two recently published abstracts of large scale randomized controlled trials [28]–[29].
Figure 1. Randomized controlled trials search and selection.
Studies' characteristics
A total of 15,762 patients were randomly assigned in the thirteen trials included in this meta-analysis. Nevertheless, in 3 studies disease free survival has been the primary endpoint with no report of overall survival data [18], [20]–[21], [26]–[27].
Trial Design
In six trials [18]–[20], [22], [24], [26], patients were randomly assigned to zoledronic acid in addition to standard- of-care therapy. Four trials used clodronate, [14]–[15], [17], [28], one trial used pamidronate [16] and one risedronate [13] and one ibandronate [29]; the schedule varied as reflected in Table 1.
Table 1. Included studies' characteristics.
| First Author, Year, Publication status | BP, Schedule | Duration of Treatment | Number of Patients BP/Placebo (or delayed) | Follow Up (median, moths) | Menopausal Status (% - post) | Adjuvant Chemotherapy (%, mean) | Adjuvant Endocrine Treatment |
| Delmas, 1997 | PO Risedronate | 2 y | 27/26 | 36 m | 100 | 100 | Tamoxifen |
| Published PR | 30 mg/d for 2 wk then 10 w off | ||||||
| Saarto, 2004 | PO Clodronate | 3 y | 139/143 | 120 m | 47.7 | 54 | Tamoxifen |
| Published PR | 1600 mg/d | ||||||
| Powels, 2006 | PO Clodronate | 2 y | 530/539 | 66 m | 50 | 74 | NS |
| Published PR | 1600 mg/d | ||||||
| Diel, | PO Clodronate | 2 y | 157/145 | 103 m | 62.5 | 42.1 | Tamoxifen |
| 2008 | 1600 mg/d | ||||||
| Published PR | |||||||
| Kristensen, 2008 | PO Pamidronate | 4 y | 460/493 | 120 m | 33 | 100 | NS |
| Published PR | 150bid | ||||||
| Brufsky, | Upfront IV zolendronic acid vs. Delayed zolendronic acid | 5 y | 300/300 | 61 m | 100 | 47 | Letrozole |
| Z-FAST, 2011 | 4 mg q6m | ||||||
| Published PR | |||||||
| Coleman EZO-FAST, 2009 | Upfront IV zolendronic acid vs. Delayed zolendronic acid | 5 y | 252/270 | 36 m | 100 | 53 | Letrozole |
| Conference proceeding | 4 mg q6m | ||||||
| Leal, | IV zolendronic acid | 1 y | 36/32 | 96 m | 100 | 90 | Tamoxifen |
| 2010??? | 4 mg q3m | (AI – 15%) | |||||
| Published PR | |||||||
| Eidtmann, 2010, | Upfront IV zolendronic acid vs. Delayed zolendronic acid | 5 y | 532/533 | 60 m | 100 | 54 | Letrozole |
| ZO-FAST | 4 mg q6m | ||||||
| Published PR | |||||||
| + update from SABCS 2011 | |||||||
| Gnant, ABCSG12, 2011 | IV zolendronic acid | 3 y | 899/904 | 84 m | 0 | 5.5 | Tamoxifen |
| Published PR | 4 mg q6m | Anastrazole | |||||
| + update from SABCS 2011 | GnRH* agonist | ||||||
| Coleman, AZURE, 2011 | IV zolendronic acid | 5 y | 1680/1680 | 59 m | 55 | 96 | NS |
| Published PR | 4 mg q6m | ||||||
| NSABP B34, 2012 | PO Clondronate | 3 y | 1662/1661 | 101 m | 64 | 68 | NS |
| 1600 mg/d | |||||||
| GAIN, | PO Ibandronate | 2 y | 1996/998 | 39 m | 52 | 100 | NS |
| 2011, Conference proceeding | 50 mg/d |
PO – Per os (oral); IV - intravenous; PR – peer review; NS- not specified; GnRH – Gonadotropin Releasing Hormone AI – Aromatase inhibitor.
Quality of Trials
Allocation concealment was reported as adequate in five trials [15], [22], [24], [28]–[29] and was not reported in the other eight trials. Few of the trials have been open-labeled. The quality assessment of the included trials has been performed according to the Cochrane risk of bias tool, as described in detail in Table S1. We appraised the rate of patients lost to follow up and in the majority of the studies the rate was <10%. In three trials the rate was higher (Range – 17–42%; [19]–[20], [28]).
Overall Survival
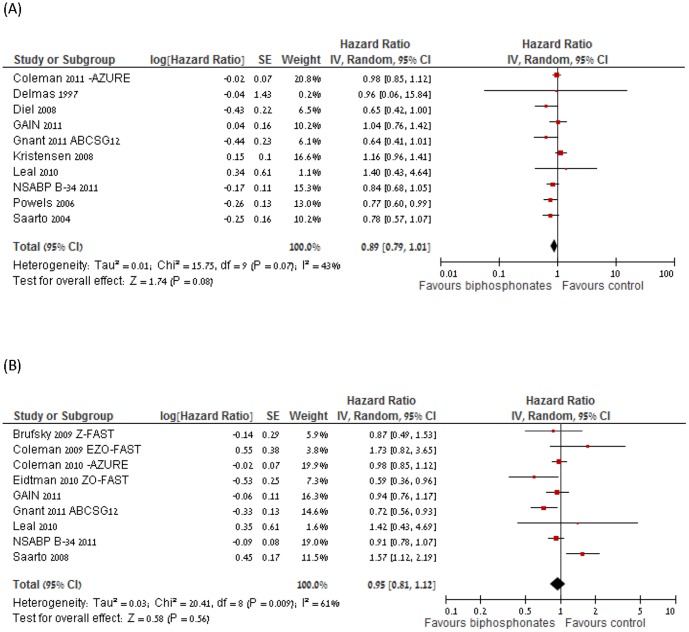
Ten trials (13,571 patients) were eligible for the meta-analysis of overall survival [13]–[17], [19], [22], [24], [28]–[29]. The numbers of randomly assigned and analyzed patients in each included trial are described in Table 1. Patients who were treated with BP therapy had no statistically significant different overall survival than patients in the control group (HR of death = 0.89, 95% CI = 0.79 to 1.01; I2 of heterogeneity −43%, random effect model) (Figure 2a). Sensitivity analysis of two trials that had short duration of follow up [13], [19] demonstrated that HR remained robust lacking these trials (HR of death = 0.89, 95% CI = 0.77 to 1.01, compared with 0.89, 0.79–1.01), presented in Figure S1). The funnel plot of the primary outcome did not support a publication bias.
Figure 2. Forest plot of hazard ratios (HRs) comparing (A) overall survival or (B) disease-free survial (DFS) for patients who received BP in addition to standard therapy vs. those who received standard therapy only.
Hazard ratios for each trail are represented by the squares, the size of the square represents the weight of the trial in the meta-analysis, and the horizontal line crossing the square represents the 95% confidence interval (CI). The diamonds represent the estimated overall effect based on the meta-analysis random effect of all trials.
Disease Free Survival
Nine trials (12,167 patients) were eligible for the meta-analysis of DFS [14], [18]–[20], [22], [24], [26], [28]–[29]. The numbers of randomly assigned and analyzed patients in each included trial are described in Table 1. Patients who were treated with BPtherapy had no statistically significant better DFS than patients in the control group (HR of death = 0.95, 95% CI = 0.81 to 1.12; I2 of heterogeneity −61%, random effect model) (Figure 2b). The funnel plot of the primary outcome did not support a publication bias.
Subgroup analysis according to menopausal status
Six trials reported the DFS outcome stratified by menopausal status [18]–[20], [24], [26], [29]. Postmenopausal patients who were treated with BP therapy had statistically significant better DFS than patients in the control group (HR of death = 0.81, 95% CI = 0.69 to 0.95; I2 of heterogeneity −34%, fixed effect model) (Figure 3). Meta-analysis for overall survival stratified by postmenopausal status was feasible for two trials only, and indicated a favorable outcome for the intervention arm (HR of death = 0.74, 95% CI = 0.57 to 0.98).
Figure 3. Forest plot of hazard ratios comparing disease-free survival for most-menopausal patients who received BP in addition to standard therapy vs. those who received standard therapy only.
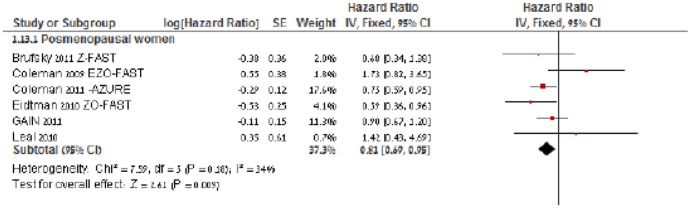
Hazard ratios for each trail are represented by the squares, the size of the square represents the weight of the trial in the meta-analysis, and the horizontal line crossing the square represents the 95% confidence interval (CI). The diamonds represent the estimated overall effect based on the meta-analysis random effect of all trials.
Interaction with potential confounders
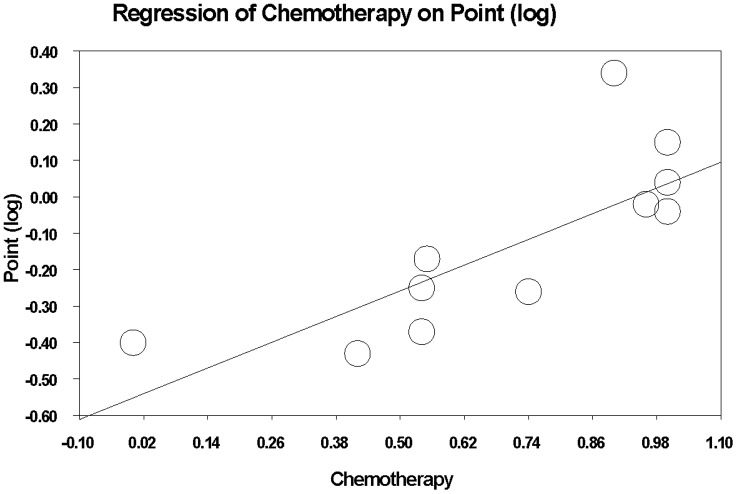
In meta-regression (Figure 4), chemotherapy was negatively associated with HR of overall survival (coefficient, −0.23; standard error, 0.144).
Figure 4. Meta regression of hazard ratios of overall survival in individual studies assessing the effect of percentage of patients receiving chemotherapy in addition to endocrine therapy.
Adverse Events
The rate of grade 3 or 4 adverse events was reported in thirteen trials [14]–[16], [18], [20], [22], [25]–[26], [28], [56]–[58], including three studies [56]–[58] that evaluated BP for reducing skeletal events without survival assessment, as depicted in Table 2. Bisphosphonate therapy resulted in a lower rate of bone fractures in the intervention arm (RR = 0.59, 95% CI = 0.42 to 0.83). The rate of osteonecrosis of the jaw (ONJ) was higher in the BP arm than in the control arm (RR = 7.53 95% CI = 2.91 to 19.50). To note, that RR was similar by using either the Mantel-Haenszel or Peto method. Pyrexia was also significantly higher in the BP arm (RR = 3.36, 95% CI = 2.61 to 4.32). The incidence of all constitutional grade 3 to 4 adverse events was not statistically significantly affected by BP therapy compared to control as depicted in Table 2.
Table 2. Relative risks (RRs) of grade 3–4 adverse events for patients who received BP in addition to standard therapy vs those who received standard therapy only.
| ONJ | 7.53 [2.91, 19.50] |
| Fractures | 0.59 [0.42, 0.83] |
| Arthralgia | 0.98 [0.76, 1.26] |
| Vomiting | 1.14 [0.82, 1.58] |
| Pyrexia | 3.36 [2.61, 4.32] |
| Hot flushes | 0.95 [0.86, 1.05] |
| Fatigue | 0.83 [0.61, 1.11] |
| Bone pain | 1.35 [0.98, 1.87] |
Discussion
The results of our meta-analysis demonstrate a positive effect for BP on survival outcomes when administered in the adjuvant setting, only in post-menopausal patients with breast cancer. Meta-regression appraised the effect of confounders such as chemotherapy on the interaction between BP use and overall survival, and showed a negative association between chemotherapy use and the effect of BP on survival.
The primary endpoint of many BP trials in the setting of early breast cancer was a reduction in skeletal related events, while survival had not been an integral endpoint of the study. We attempted to avoid publication bias by searching for and including in our meta-analysis conference proceedings, databases of ongoing trials, and unpublished data, and indeed in the recent San-Antonio Breast Cancer Symposium (2011) two large scale trials [28]–[29], have been primarily presented (one of which has been fully published later on) and were included in our meta-analysis.
Bisphosphonates have been proven to display high affinity for bone and to inhibit bone resorption by osteoclasts [34]–[35]. Newer generation, nitrogen-containing bisphosphonates, such as alendronate, risedronate, pamidronate, ibandronate, and zoledronic acid are considered an integral part of treatment to reduce the risk for skeletal related events in patients with bone metastases [36]–[39]. Many in-vitro studies have portrayed the effect of a variety of BP (both nitrogen containing and non-nitrogen bisphosphonates) to hinder tumor cell adhesion, invasion, proliferation and interplay with the bone microenvironment components such as matrix-metalloproteinases (MMPs) [40]–[41]. Moreover in their well-recognized target, the osteoclast, BP interfere with signaling pathways (e.g. Rac and Ras) required for osteoclast function and survival [42]. Breast cancer cells have a unique predilection to metastasize to bone, whereas the microenvironment is highly receptive to metastatic tumor cells [4].
Two large cohort studies evaluated breast cancer incidence in healthy postmenopausal women treated with bisphosphonates to prevent bone loss and showed a 29–32% statistically significant reduction in the incidence of invasive breast cancer, lending credence for a potential chemo- preventive effect for BP [9]–[10]. The benefit was observed both in ER-positive and ER-negative tumors, implying that the mechanisms may be hormonally-independent and might involve pathways such as angiogenesis inhibition, modulation of the immune system or a direct effect on the bone microenvironment.
The biological rationale for the benefit of BP in post-menopausal patients lies in the natural estrogen deficiency at postmenopause which leads to increased osteoclast activity, resulting in decreased bone mineral density (BMD), yet is still considered hypothetical. Bisphosphonates have been shown to be effective in numerous studies in preventing cancer treatment-associated bone loss in postmenopausal women with early breast cancer [42]–[44] mainly due to their effect on the highly activated osteoclasts. Of note, the increase in bone loss is 5-fold higher following treatment with aromatase inhibitors (AIs) than physiological bone loss observed in postmenopausal women with established osteoporosis in the absence of AI therapy, and further induce bone turnover than documented during tamoxifen use [45]–[49]. A recent study by Cheung indicated that the effect of aromatase inhibitors (exemestane) on bone density may be underestimated and that exemestane increases cortical bone permeability thus predisposing the patient to loss of bone strength and non-vertebral fractures [50]–[51].
Moreover, in vitro studies have indicated there may be a synergistic effect between zoledronic acid and letrozole on breast cancer cell lines [52]. This has been the leading rationale for the design of the Z-FAST, ZO-FAST, E-ZO-FAST that evaluated zoledronic acid in combination with AI [18], [20], [26]. Premenopausal patient who become pharmacologically-induced postmenopausal by Gonadotropin Relasing Hormone agonists (GnRHa) may benefit from the addition of bisphosphonates, since bone turnover is further enhanced in premenopausal women who undergo chemical ovarian suppression' as indicated in the ABCSG-12 trial [22]. Meta-regression that assessed the effect of chemotherapy on the association between BP use and survival indicated that the use of chemotherapy may lessen the effect of BP on survival. This result may elucidate the lack of survival benefit in few of the included studies in which chemotherapy was administered in a major portion of the study population [19], [24], [28]–[29]. On the contrary, in the ABSCG-12 which demonstrated an impressive survival benefit for the addition of zoledronic acid, less than 5% of the patients received chemotherapy.
Meta-analysis of the adverse events as reported in the included studies revealed a significant increase in pyrexia as well as osteonecrosis of the jaw (ONJ). Nevertheless, the incidence of ONJ is extremely low (0.24%, according to a recent meta-analysis [53]) and is therefore still considered a non-frequent adverse effect of BP. Although oral bisphosphonates are often associated with gastrointestinal adverse effects while intravenous bisphosphonates have been associated with infusion rate-dependent effect on renal function, the incidence of these adverse events was not statistically significant in all of the studies. Adequate hydration, serum creatinine monitoring and dose reductions in patients with renal impairment prior to BP administration are mandatory in the oncology setting and may support the lack of renal toxicity in the studies.
Limitations
Several limitations of this analysis must be acknowledged. The study population varies between trials with regard to the risk of recurrence: whereas in few of the large-scale trials most patients had positive lymph nodes at diagnosis [24], [28]–[29], indicating an a-priori worse prognosis than for patients with N0 disease as in the ABCSG-12 trial. Chemotherapy administration differs among studies; in the AZURE trial almost all patients received chemotherapy, in few trials all patients received chemotherapy [16], [19], [29] and in several studies more than half of the patients received chemotherapy [15], [17]–[18], [20], [26], [28]. On the contrary, in the ABSCG-12 less than 5% received chemotherapy, also reflecting the good prognosis of the included population. This heterogeneity prompted us to perform meta-regression which indeed demonstrated that chemotherapy administration correlates with a lack of effect to BP. It should be acknowledged that the result of the meta-regression may reflect more the higher risk group in which chemotherapy was used and less so the effect of chemotherapy itself; along with the known limitations of a meta-regression [31]. Although meta-analysis of the subgroup of post-menopausal patients was feasible due to a separate report on the outcome of this population, in some of the trials the study population was not stratified by menopausal status. Moreover, the trials differ by the specific BP that had been employed. Upon in vitro studies, zoledronic acid may exhibit more potent anti-cancer effect than other BP.
The schedule and duration of the intervention varies between studies and could account for the apparent discrepancy in survival. Pre-clinical studies have demonstrated the activation of T cells by bisphosphonate as their key anti-tumor effect which is paradoxically reduced with each subsequent administration of BP. Thus, frequent administration as in few of the trials may result in a reduced anti-tumor effect [54]–[55].
Implication for research
As indicated by our results, the group of patients that tends to benefit the most from the addition of BP in the adjuvant setting is the post-menopausal population, especially those who are not treated with chemotherapy. In the era of tailored therapy, the use of molecular techniques enables to individualize prognostic and predictive genetic traits by the 21-gene recurrence score (RS) assay (Oncotype Dx) or a microarray of 70-gene expression profile (Mammaprint) identifies subsets of patients with estrogen receptor positive tumors, who are not treated with chemotherapy; it would therefore be of value to elucidate the role of BP in a randomized trial in this population. This design may define clearly the potential benefit of bisphosphonates while the use of chemotherapy would not hamper their potential anti-tumor effect.
Implications for practice
Our meta-analysis provides a reflection of the new mounting and highly significant data published in the last 3 years which shed new light on the results of a former published meta-analysis on this matter both regarding efficacy and safety of BP in early breast cancer [53]. In view of the lack of evidence for the effectiveness of BP on survival in patients treated with chemotherapy or in premenopausal patients, this treatment should be considered in addition to endocrine treatment only in a subset of postmenopausal patients who are not scheduled for chemotherapy or as part of a clinical trial. Furthermore, the protective effect of BP on bone loss associated with AIs in postmenopausal women with early breast cancer as demonstrated in former studies further rationalize adopting these agents in this population. The addition of BP to the armamentarium of adjuvant treatments for early breast cancer should be further delineated in large scale RCTs which may unravel the specific subgroup that may benefit from this intervention.
Supporting Information
Checklist items according to PRISMA criteria.
(DOC)
Sensitivity analysis of survival according to median follow up duration. Hazard ratios for each trial are represented by the squares, the size of the square represents the weight of the trial in the meta-analysis, and the horizontal line crossing the square represents the 95% confidence interval (CI). The diamonds represent the estimated overall effect based on the meta-analysis random effect of all trials.
(TIFF)
Quality assessment of included studies according to Cochrane criteria. Quality assessment: Low risk-adequate (central randomization; numbered or coded bottles or containers; drugs prepared by the pharmacy; serially numbered sealed opaque envelopes; other convincing) Unclear – not reported.
(PDF)
Funding Statement
The authors have no external support or funding to report.
References
- 1. Ferlay J, Shin HR, Bray F, Forman D, Mathers C, et al. (2010) Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 127: 2893–2917. [DOI] [PubMed] [Google Scholar]
- 2. Coleman RE (2006) Clinical features of metastatic bone disease and risk of skeletal morbidity. Clin Cancer Res 12: 6243s–6249s. [DOI] [PubMed] [Google Scholar]
- 3. Boissier S, Ferreras M, Peyruchaud O, Magnetto S, Ebetino FH, et al. (2000) Bisphosphonates inhibit breast and prostate carcinoma cell invasion, an early event in the formation of bone metastases. Cancer Res 60: 2949–2954. [PubMed] [Google Scholar]
- 4. Van der Pluijm G, Vloedgraven H, van Beek E, van der Wee-Pals L, Löwik C, et al. (1996) Bisphosphonates inhibit the adhesion of breast cancer cells to bone matrices in vitro. J Clin Invest 98: 698–705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Gallo M, De Luca A, Lamura L, Normanno N (2011) Zoledronic acid blocks the interaction between mesenchymal stem cells and breast cancer cells: implications for adjuvant therapy of breast cancer. Ann Oncol 1: 1–7. [DOI] [PubMed] [Google Scholar]
- 6. Holen I, Coleman RE (2011) Bisphosphonates as treatment of bone metastases. Curr Pharm Des 16: 1262–1271. [DOI] [PubMed] [Google Scholar]
- 7. Wong R, Wiffen PJ (2002) Bisphosphonates for the relief of pain secondary to bone metastases. Cochrane Database Syst Rev 2: CD002068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Pavlakis N, Schmidt R, Stockler M (2005) Bisphosphonates For Breast Cancer. Cochrane Database Syst Rev 3: CD003474. [DOI] [PubMed] [Google Scholar]
- 9. Rennert G, Pinchev M, Rennert HS (2010) Use of bisphosphonates and risk of postmenopausal breast cancer. J Clin Oncol 28: 3577–3581. [DOI] [PubMed] [Google Scholar]
- 10. Chlebowski RT, Chen Z, Cauley JA, Anderson G, Rodabough RJ, et al. (2010) Oral bisphosphonate use and breast cancer incidence in postmenopausal women. J Clin Oncol 28: 3582–3590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Daubine F, Le Gall C, Gasser J, Green J, Clézardin P (2007) Antitumor effects of clinical dosing regimens of bisphosphonates in experimental breast cancer bone metastasis. J Natl Cancer Inst 99: 322–330. [DOI] [PubMed] [Google Scholar]
- 12. Ottewell PD, Woodward JK, Lefley DV (2009) Anticancer mechanisms of doxorubicin and zoledronic acid in breast cancer tumor growth in bone. Mol Cancer Ther 8: 2821–2832. [DOI] [PubMed] [Google Scholar]
- 13. Delmas PD, Balena R, Confravreux E, Hardouin C, Hardy P, et al. (1997) Bisphosphonate risedronate prevents bone loss in women with artificial menopause due to chemotherapy of breast cancer: a double-blind, placebo-controlled study. J Clin Oncol 15: 955–962. [DOI] [PubMed] [Google Scholar]
- 14. Saarto T, Vehmanen L, Virkkunen P, Blomqvist C (2004) Ten-year follow-up of a randomized controlled trial of adjuvant clodronate treatment in node-positive breast cancer patients. Acta Oncol 43: 650–656. [DOI] [PubMed] [Google Scholar]
- 15. Powles T, Paterson S, Kanis JA, McCloskey E, Ashley S, et al. (2006) Reduction in bone relapse and improved survival with oral clodronate for adjuvant treatment of operable breast cancer. Breast Cancer Res 82: R13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Kristensen B, Ejlertsen B, Mouridsen HT, Jensen MB, Andersen J, et al. (2008) Bisphosphonate treatment in primary breast cancer: results from a randomised comparison of oral pamidronate versus no pamidronate in patients with primary breast cancer. Acta Oncol 47: 740–746. [DOI] [PubMed] [Google Scholar]
- 17. Diel IJ, Jaschke A, Solomayer EF, Gollan C, Bastert G, et al. (2008) Adjuvant oral clodronate improves the overall survival of primary breast cancer patients with micrometastases to the bone marrow: a long-term follow-up. Ann Oncol 19: 2007–2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Coleman R, Bundred N, de Boer R, Llombarto A, Campbell I, et al.. (2009) on behalf of the Z-FAST, ZO-FAST, and E-ZO-FAST Study Groups. Impact of Zoledronic Acid in Postmenopausal women with early breast cancer receiving Letrozole: Z-FAST, ZO-FAST and EZO-FAST. Presented at the San Antonio Breast Cancer Symposium; San Antonio, TX
- 19. Leal T, Tevaarwerk A, Love R, Stewart J, Binkley N, et al. (2010) Randomized trial of adjuvant zoledronic acid in postmenopausal women with high-risk breast cancer. Clin Breast Cancer 10: 471–476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Eidtmann H, de Boer R, Bundred N, Llombart-Cussac A, Davidson N, et al. (2010) Efficacy of zoledronic acid in postmenopausal women with early breast cancer receiving adjuvant letrozole: 36-month results of the ZO-FAST Study. Ann Oncol 21: 2188–2194. [DOI] [PubMed] [Google Scholar]
- 21.de Boer R, Bundred N, Eidtmann H, Neven P, von Minckwitz G, et al.. Long-term survival outcomes among postmenopausal women with hormone receptor-positive early breast cancer receiving adjuvant letrozole and zoledronic acid: 5-year follow-up of ZO-FAST [abstract S1–3]. Presented at the San Antonio Breast Cancer Symposium; San Antonio, TX; 2011.
- 22. Gnant M, Mlineritsch B, Stoeger H, Luschin-Ebengreuth G, Heck D, et al. (2011) Adjuvant endocrine therapy plus zoledronic acid in premenopausal women with early-stage breast cancer: 62-month follow-up from the ABCSG-12 randomised trial. Lancet Oncol 12: 631–641. [DOI] [PubMed] [Google Scholar]
- 23.Gnant M, Mlineritsch B, Luschin-Ebengreuth G, Stoeger H, Dubsky P, et al.. Long-term follow-up in ABCSG-12: Significantly improved overall survival with adjuvant zoledronic acid in premenopausal patients with endocrine-receptor-positive early breast cancer [abstract S1–2]. Presented at the San Antonio Breast Cancer Symposium; San Antonio, TX; 2011.
- 24. Coleman RE, Marshall H, Cameron D, Dodwell D, Burkinshaw R, et al. (2011) AZURE Investigators. Breast-cancer adjuvant therapy with zoledronic acid. N Engl J Med 365: 1396–1405. [DOI] [PubMed] [Google Scholar]
- 25. Coleman R, Woodward E, Brown J, Cameron D, Bell R, et al. (2011) Safety of zoledronic acid and incidence of osteonecrosis of the jaw (ONJ) during adjuvant therapy in a randomised phase III trial (AZURE: BIG 01-04) for women with stage II/III breast cancer. Breast Cancer Res Treat 127: 429–438. [DOI] [PubMed] [Google Scholar]
- 26. Brufsky AM, Bosserman LD, Caradonna RR, Haley BB, Jones CM, et al. (2009) Zoledronic acid effectively prevents aromatase inhibitor-associated bone loss in postmenopausal women with early breast cancer receiving adjuvant letrozole: Z-FAST study 36-month follow-up results. Clin Breast Cancer 9: 77–85. [DOI] [PubMed] [Google Scholar]
- 27. Brufsky AM, Harker WG, Beck JT, Carroll R, Tan-Chiu E, et al. (2011) Final 5-year results of Z-FAST trial: Adjuvant Zoledronic Acid Maintains Bone Mass in Postmenopausal Breast Cancer Patients Receiving Letrozole. Cancer doi: 10.1002/cncr.26313 [DOI] [PubMed] [Google Scholar]
- 28. Paterson AH, Anderson SJ, Lembersky BC, Fehrenbacher L, Falkson CI, et al. (2012) Oral clodronate for adjuvant treatment of operable breast cancer (National Surgical Adjuvant Breast and Bowel Project protocol B-34): a multicentre, placebo-controlled, randomised trial. Lancet Oncol 13: 734–742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Möbus V, Thomssen C, Harbeck N, Diel IJ, Elling D, et al.. GAIN (German Adjuvant Intergroup Node Positive) study: A phase-III multicenter trial to compare dose dense, dose intense ETC (iddETC) vs. EC-TX and Ibandronate vs. observation in patients with node-positive primary breast cancer - 1st interim efficacy analysis [abstract S2–4]. Presented at the San Antonio Breast Cancer Symposium; San Antonio, TX; 2011.
- 30.Higgins JPT, Green S (2008) Cochrane Handbook for Systematic Reviews of Interventions Version 5.0. The Cochrane Collaboration. Available: www.cochrane-handbook.org. Accessed August 10, 2011
- 31. Mantel N, Haenszel W (1959) Statistical aspects of the analysis of data from retrospective studies of disease. J Natl Cancer Inst 22: 719–748. [PubMed] [Google Scholar]
- 32. DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Control Clin Trials 7: 177–188. [DOI] [PubMed] [Google Scholar]
- 33. Sweeting MJ, Sutton AJ, Lambert PC (2004) What to add to nothing? Use and avoidance of continuity corrections in meta-analysis of sparse data. Statistics in Medicine 23: 1351–1375. [DOI] [PubMed] [Google Scholar]
- 34. Fisher JE, Rogers MJ, Halasy JM, Luckman SP, Hughes DE, et al. (1999) Alendronate mechanism of action: geranylgeraniol, an intermediate in the mevalonate pathway, prevents inhibition of osteoclast formation, bone resorption, and kinase activation in vitro. Proc Natl Acad Sci U S A 96: 133–138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Rodan GA (1998) Mechanisms of action of bisphosphonates. Annu Rev Pharmacol Toxicol 38: 375–388. [DOI] [PubMed] [Google Scholar]
- 36. Holen I, Coleman RE (2010) Bisphosphonates as treatment of bone metastases. Curr Pharm Design 16: 1262–1271. [DOI] [PubMed] [Google Scholar]
- 37. Kanis JA, Powles T, Paterson AH, McCloskey EV, Ashley S, et al. (1996) Clodronate decreases the requency of skeletal metastases in women with breast cancer. Bone 19: 663–667. [DOI] [PubMed] [Google Scholar]
- 38. Kavanagh KL, Guo K, Dunford JE, Wu X, Knapp S, et al. (2006) The molecular mechanism of nitrogen-containing bisphosphonates as antiosteoporosis drugs. Proc Natl Acad Sci USA 103: 7829–7834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Bianchi G, Sambrook P (2008) Oral nitrogen-containing bisphosphonates: a systematic review of randomized clinical trials and vertebral fractures. Curr Med Res Opin 24: 2669–2677. [DOI] [PubMed] [Google Scholar]
- 40. Neville-Webbe HL, Gnant M, Coleman RE (2010) Potential anticancer properties of bisphosphonates. Semin Oncol 37 Suppl 1: S53–65. [DOI] [PubMed] [Google Scholar]
- 41. Winter MC, Holen I, Coleman RE (2008) Exploring the anti-tumour activity of bisphosphonates in early breast cancer. Cancer Treat Rev 34: 453–475. [DOI] [PubMed] [Google Scholar]
- 42. Hadji P, Bundred N (2007) Reducing the risk of cancer treatmentassociated bone loss in patients with breast cancer. Semin Oncol 34 Suppl 4: S4–10. [DOI] [PubMed] [Google Scholar]
- 43. Greenspan SL, Brufsky A, Lembersky BC, Bhattacharya R, Vujevich KT, et al. (2008) Risedronate prevents bone loss in breast cancer survivors: a 2-year, randomized, double-blind, placebo-controlled clinical trial. J Clin Oncol 26: 2644–2652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Lester JE, Dodwell D, Purohit OP, Gutcher SA, Ellis SP, et al. (2008) Prevention of anastrazole-induced bone loss with monthly oral ibandronate during adjuvant aromatase inhibitor therapy for breast cancer. Clin Cancer Res 14: 6336–6342. [DOI] [PubMed] [Google Scholar]
- 45. Coleman RE, Banks LM, Girgis SI, Kilburn LS, Vrdoljak E, et al. (2007) Skeletal effects of exemestane on bone-mineral density, bone biomarkers, and fracture incidence in postmenopausal women with early breast cancer participating in the Intergroup Exemestane Study (IES): a randomised controlled study. Lancet Oncol 8: 119–127. [DOI] [PubMed] [Google Scholar]
- 46. Perez EA, Josse RG, Pritchard KI, Ingle JN, Martino S, et al. (2006) Effect of letrozole versus placebo on bone mineral density in women with primary breast cancer completing 5 or more years of adjuvant tamoxifen: a companion study to NCIC CTG MA.17. J Clin Oncol 24: 3629–3635. [DOI] [PubMed] [Google Scholar]
- 47. Hadji P, Ziller M, Kieback DG, Dornoff W, Tessen HW, et al. (2009) Effects of exemestane and tamoxifen on bone health within the Tamoxifen Exemestane Adjuvant Multicentre (TEAM) trial: results of a German, 12-month, prospective, randomised substudy. Ann Oncol 20: 1203–1209. [DOI] [PubMed] [Google Scholar]
- 48. Hadji P, Body JJ, Aapro MS, Brufsky A, Coleman RE, et al. (2008) Practical guidance for the management of aromatase inhibitor-associated bone loss. Ann Oncol 19: 1407–1416. [DOI] [PubMed] [Google Scholar]
- 49. Safra T, Bernstein-Molho R, Greenberg J, Pelles-Avraham S, Stephansky I, et al. (2011) The Protective Effect of Zoledronic Acid on Bone Loss in Postmenopausal Women with Early Breast Cancer Treated with Sequential Tamoxifen and Letrozole: A Prospective, Randomized, Phase II Trial. Oncology 81: 298–305. [DOI] [PubMed] [Google Scholar]
- 50. Cheung AM, Tile L, Cardew S, Pruthi S, Robbins J, et al. (2012) Bone density and structure in healthy postmenopausal women treated with exemestane for the primary prevention of breast cancer: a nested substudy of the MAP.3 randomised controlled trial. Lancet Oncol Feb 6. [DOI] [PubMed] [Google Scholar]
- 51. Cauley JA (2012) Bone loss associated with prevention of breast cancer. Lancet Oncol Feb 6. [DOI] [PubMed] [Google Scholar]
- 52. Neville-Webbe HL, Coleman RE (2010) Bisphosphonates and RANK ligand inhibitors for the treatment and prevention of metastatic bone disease. Eur J Cancer 46: 1211–1222. [DOI] [PubMed] [Google Scholar]
- 53. Mauri D, Valachis A, Polyzos IP, Polyzos NP, Kamposioras K, et al. (2009) Osteonecrosis of the jaw and use of bisphosphonates in adjuvant breast cancer treatment: a meta-analysis. Breast Cancer Res Treat 116: 433–439. [DOI] [PubMed] [Google Scholar]
- 54. Kunzmann V, Bauer E, Wilhelm M (1999) Gamma/delta T-cell stimulation by pamidronate. N Engl J Med 340: 737–738. [DOI] [PubMed] [Google Scholar]
- 55. van der Vliet HJ, Verheul HM (2012) Breast-cancer adjuvant therapy with zoledronic acid. N Engl J Med 366: 188–189. [DOI] [PubMed] [Google Scholar]
- 56. Lester JE, Dodwell D, Purohit OP, Gutcher SA, Ellis SP, et al. (2008) Prevention of anastrozole-induced bone loss with monthly oral ibandronate during adjuvant aromatase inhibitor therapy for breast cancer. Clin Cancer Res 14: 6336–6342. [DOI] [PubMed] [Google Scholar]
- 57. Greenspan SL, Brufsky A, Lembersky BC, Bhattacharya R, Vujevich KT, et al. (2008) Risedronate prevents bone loss in breast cancer survivors: a 2-year, randomized, double-blind, placebo-controlled clinical trial. J Clin Oncol 26: 2644–2652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Van Poznak C, Hannon RA, Mackey JR, Campone M, Apffelstaedt JP, et al. (2010) Prevention of aromatase inhibitor-induced bone loss using risedronate: the SABRE trial. J Clin Oncol 28: 967–975. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Checklist items according to PRISMA criteria.
(DOC)
Sensitivity analysis of survival according to median follow up duration. Hazard ratios for each trial are represented by the squares, the size of the square represents the weight of the trial in the meta-analysis, and the horizontal line crossing the square represents the 95% confidence interval (CI). The diamonds represent the estimated overall effect based on the meta-analysis random effect of all trials.
(TIFF)
Quality assessment of included studies according to Cochrane criteria. Quality assessment: Low risk-adequate (central randomization; numbered or coded bottles or containers; drugs prepared by the pharmacy; serially numbered sealed opaque envelopes; other convincing) Unclear – not reported.
(PDF)