Abstract
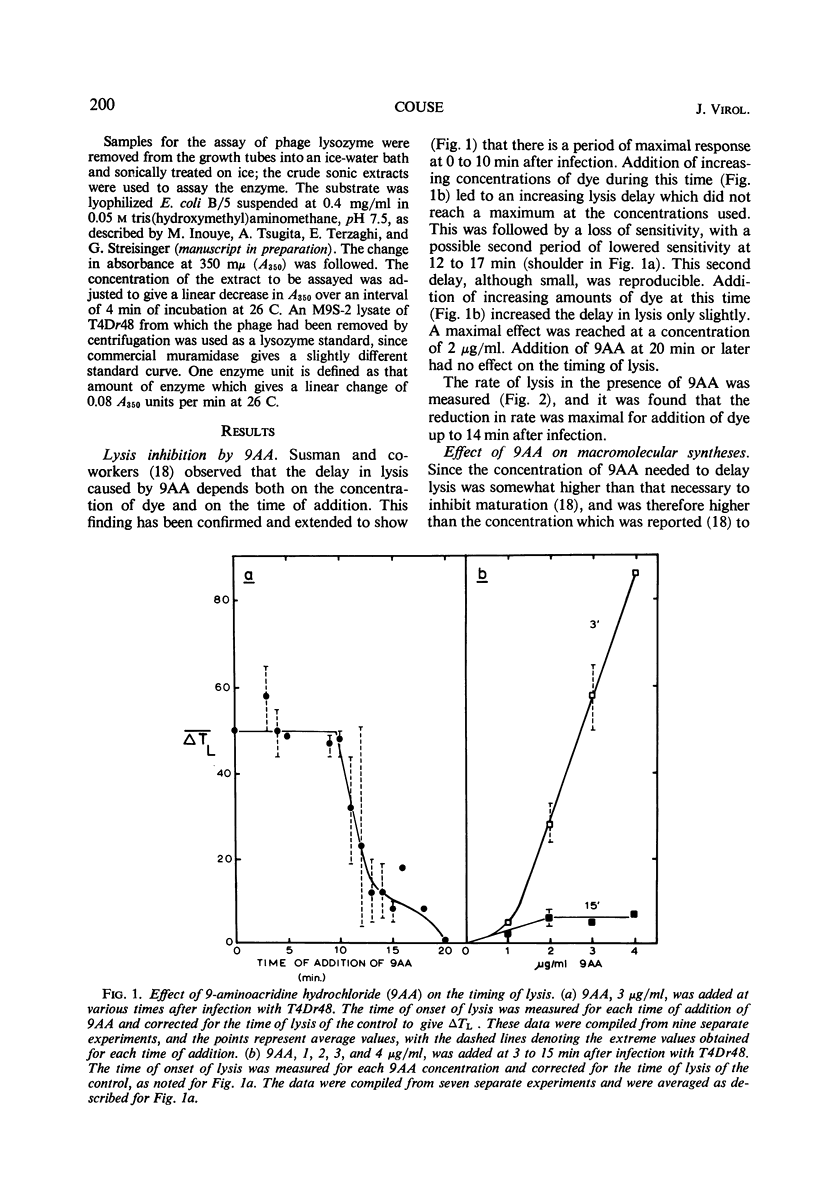
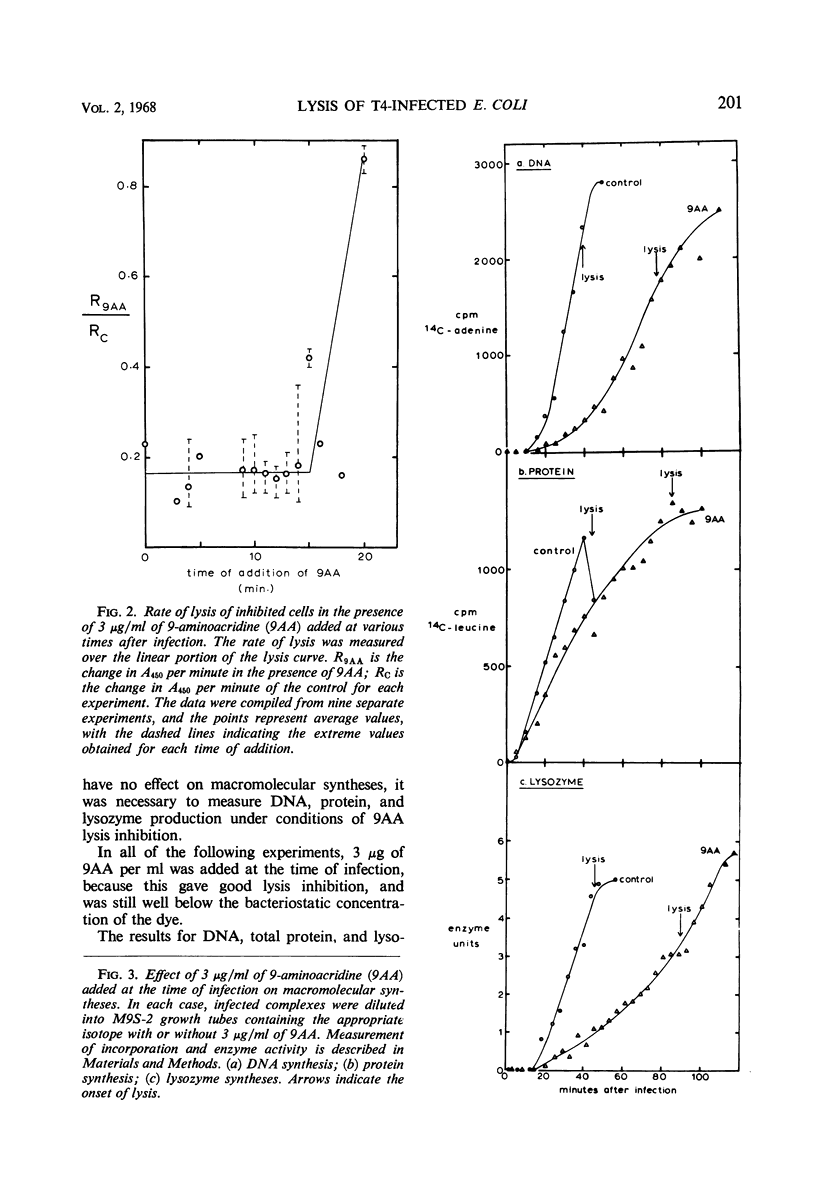
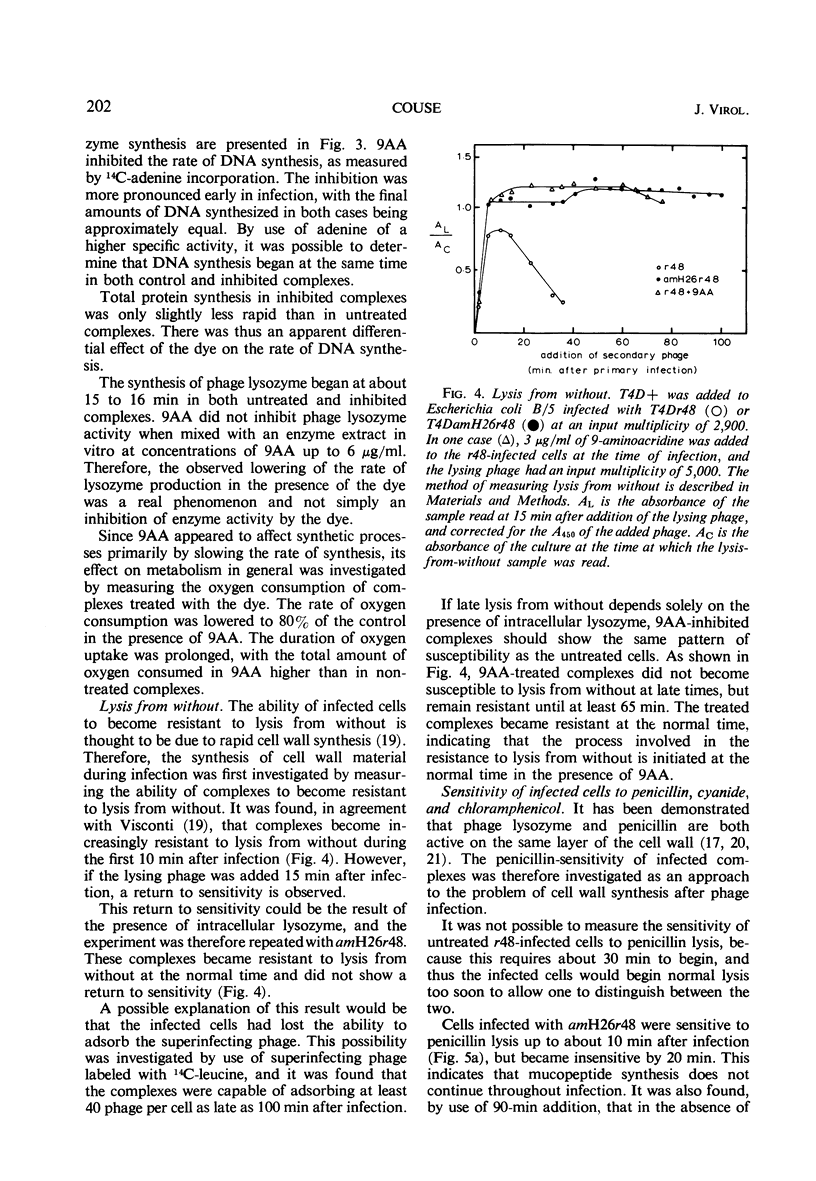
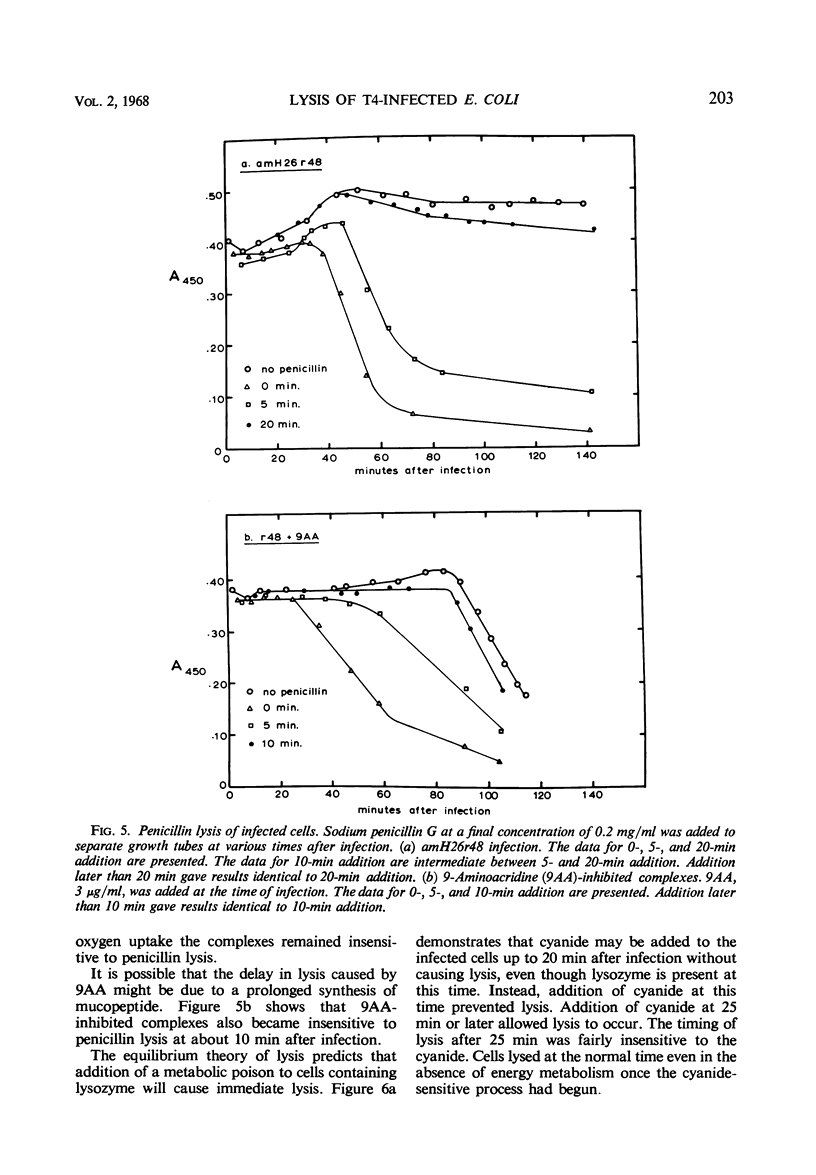
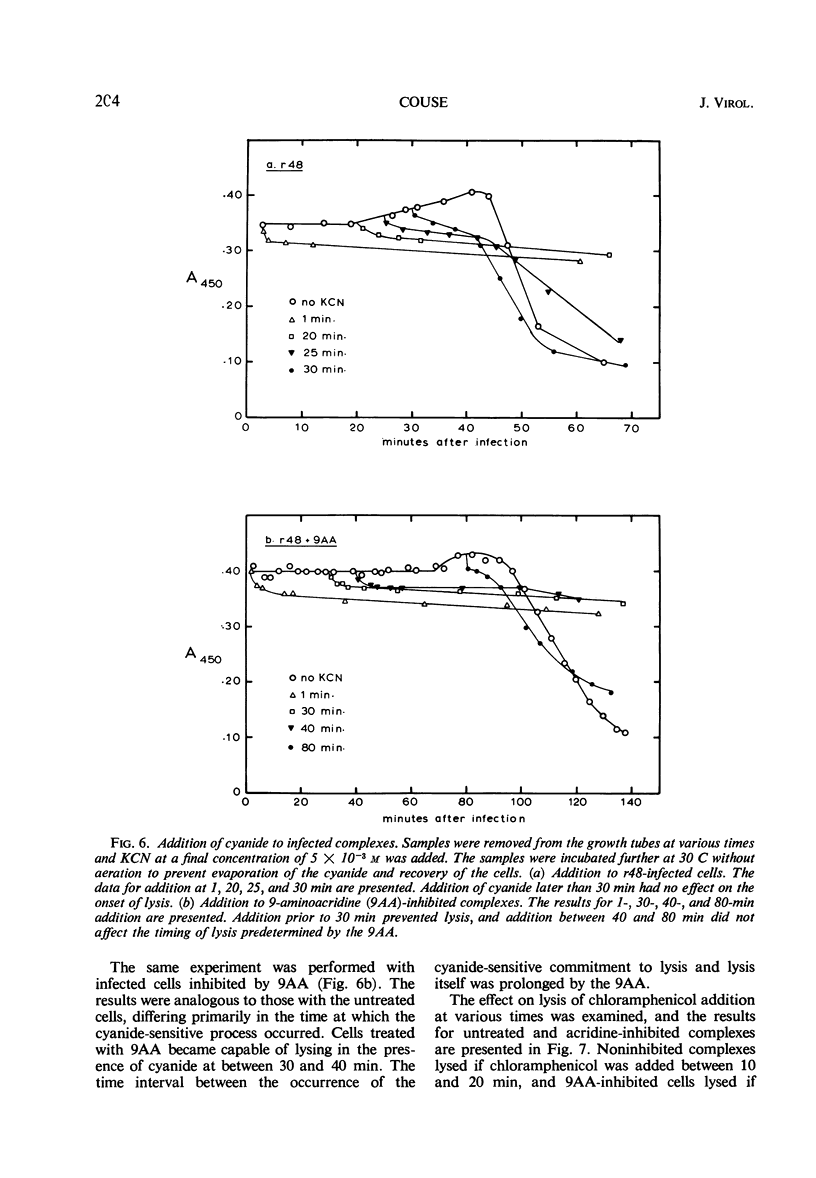
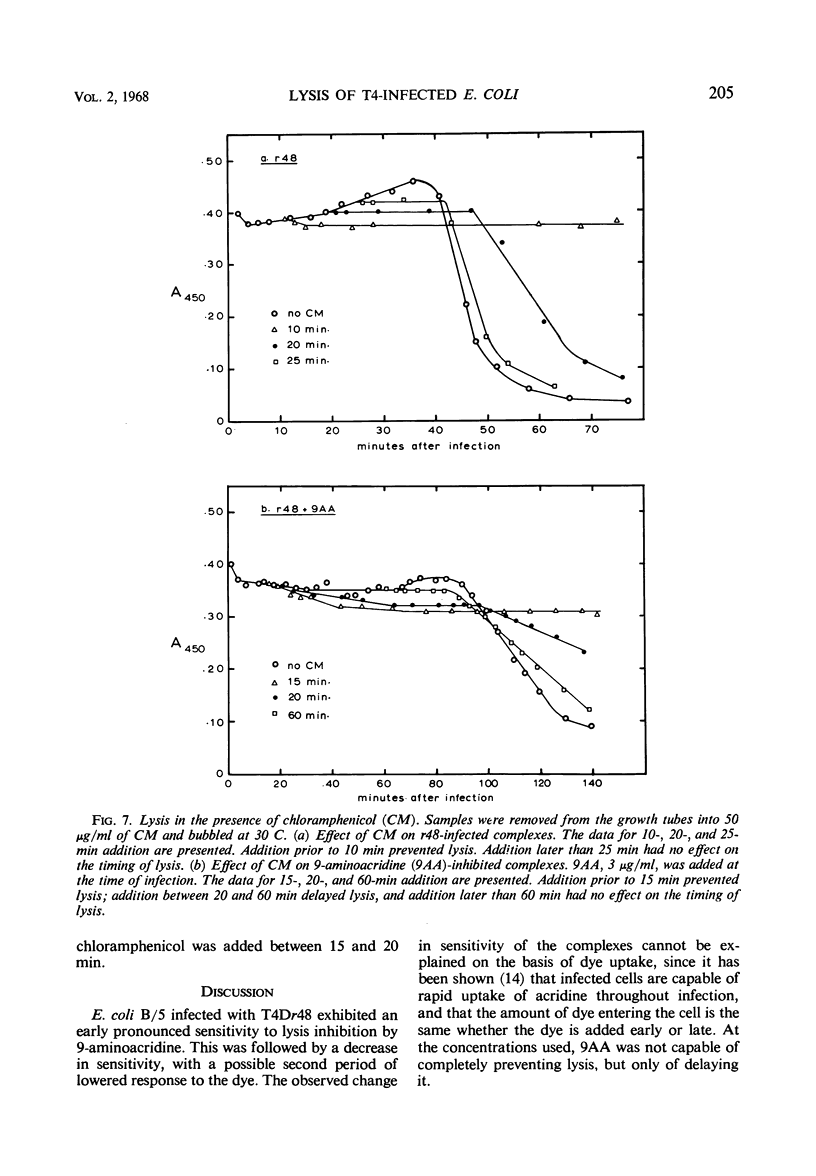
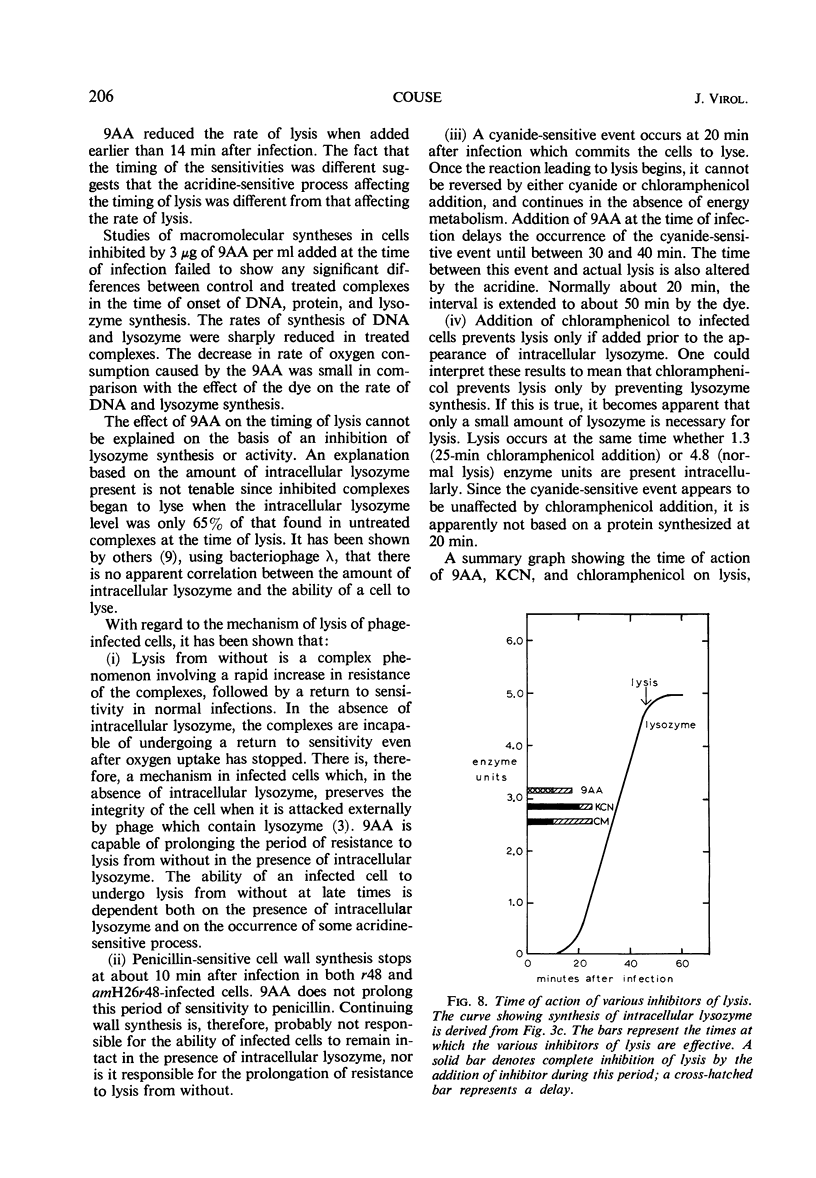
The lysis of Escherichia coli B/5 infected with T4Dr48 could be delayed by addition of 9-aminoacridine (9AA). Infected cells showed an early period of maximal response followed by a decline in sensitivity. The ultimate rate of lysis was also affected by the dye. Deoxyribonucleic acid (DNA), protein, and lysozyme synthesis began at the normal time in complexes inhibited by 9AA addition. The rates of synthesis of these macromolecules were lower in the presence of the dye, with DNA and lysozyme synthesis being more strongly affected than total protein synthesis. Penicillin-sensitive cell wall synthesis stopped at about 10 min after infection. Inhibition of oxidative metabolism by early potassium cyanide addition prevented lysis in the presence of intracellular lysozyme. The cyanide-sensitive event occurred at about 20 min in normal infections, and between 30 and 40 min in 9AA-inhibited infections. 9AA could alter both the time at which the cyanide-sensitive event occurred and the time of lysis. Addition of chloramphenicol did not prevent lysis once intracellular lysozyme was present. Lysis from without of infected cells consisted of three phases: an initial sensitivity, followed by a short period of resistance, and then a return to sensitivity in normal infections. The demonstration of the late return to sensitivity depended on the presence of intracellular lysozyme, and could be delayed by 9AA addition.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- APPLEYARD R. K., MCGREGOR J. F., BAIRD K. M. Mutation to extended host range and the occurrence of phenotypic mixing in the temperate coliphage lambda. Virology. 1956 Aug;2(4):565–574. doi: 10.1016/0042-6822(56)90012-5. [DOI] [PubMed] [Google Scholar]
- BARRINGTON L. F., KOZLOFF L. M. Action of bacteriophage on isolated host cell walls. J Biol Chem. 1956 Dec;223(2):615–627. [PubMed] [Google Scholar]
- CHAMPE S. P. BACTERIOPHAGE REPRODUCTION. Annu Rev Microbiol. 1963;17:87–114. doi: 10.1146/annurev.mi.17.100163.000511. [DOI] [PubMed] [Google Scholar]
- DOERMANN A. H. The intracellular growth of bacteriophages. I. Liberation of intracellular bacteriophage T4 by premature lysis with another phage or with cyanide. J Gen Physiol. 1952 Mar;35(4):645–656. doi: 10.1085/jgp.35.4.645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doermann A H, Hill M B. Genetic Structure of Bacteriophage T4 as Described by Recombination Studies of Factors Influencing Plaque Morphology. Genetics. 1953 Jan;38(1):79–90. doi: 10.1093/genetics/38.1.79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doermann A. H. Lysis and Lysis Inhibition with Escherichia coli Bacteriophage. J Bacteriol. 1948 Feb;55(2):257–276. doi: 10.1128/jb.55.2.257-276.1948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GROMAN N. B., SUZUKI G. Relation of endolysin to lysis by lambda bacteriophages. J Bacteriol. 1962 Sep;84:596–597. doi: 10.1128/jb.84.3.596-597.1962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HEAGY F. C. The effect of 2,4-dinitrophenol and phage T2 on Escherichia coli B. J Bacteriol. 1950 Mar;59(3):367–373. doi: 10.1128/jb.59.3.367-373.1950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- JACOB F., FUERST C. R. The mechanism of lysis by phage studied with defective lysogenic bacteria. J Gen Microbiol. 1958 Apr;18(2):518–526. doi: 10.1099/00221287-18-2-518. [DOI] [PubMed] [Google Scholar]
- Mukai F., Streisinger G., Miller B. The mechanism of lysis in phage T4-infected cells. Virology. 1967 Nov;33(3):398–404. doi: 10.1016/0042-6822(67)90115-8. [DOI] [PubMed] [Google Scholar]
- SILVER S. ACRIFLAVINE RESISTANCE: A BACTERIOPHAGE MUTATION AFFECTING THE UPTAKE OF DYE BY THE INFECTED BACTERIAL CELLS. Proc Natl Acad Sci U S A. 1965 Jan;53:24–30. doi: 10.1073/pnas.53.1.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- STEINBERG C. M., EDGAR R. S. A critical test of a current theory of genetic recombination in bacteriophage. Genetics. 1962 Feb;47:187–208. doi: 10.1093/genetics/47.2.187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- STREISINGER G., MUKAI F., DREYER W. J., MILLER B., HORIUCHI S. Mutations affecting the lysozyme of phage T4. Cold Spring Harb Symp Quant Biol. 1961;26:25–30. doi: 10.1101/sqb.1961.026.01.007. [DOI] [PubMed] [Google Scholar]
- SUSMAN M., PIECHOWSKI M. M. STUDIES ON PHAGE DEVELOPMENT. I. AN ACRIDINE-SENSITIVE CLOCK. Virology. 1965 Jun;26:163–174. doi: 10.1016/0042-6822(65)90043-7. [DOI] [PubMed] [Google Scholar]
- VISCONTI N. Resistance to lysis from without in bacteria infected with T2 bacteriophage. J Bacteriol. 1953 Sep;66(3):247–253. doi: 10.1128/jb.66.3.247-253.1953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WEIDEL W., PRIMOSIGH J. Biochemical parallels between lysis by virulent phage and lysis by penicillin. J Gen Microbiol. 1958 Apr;18(2):513–517. doi: 10.1099/00221287-18-2-513. [DOI] [PubMed] [Google Scholar]
- Wise E. M., Jr, Park J. T. Penicillin: its basic site of action as an inhibitor of a peptide cross-linking reaction in cell wall mucopeptide synthesis. Proc Natl Acad Sci U S A. 1965 Jul;54(1):75–81. doi: 10.1073/pnas.54.1.75. [DOI] [PMC free article] [PubMed] [Google Scholar]



