Abstract
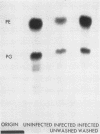
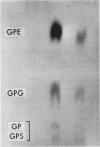
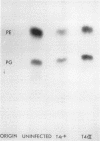
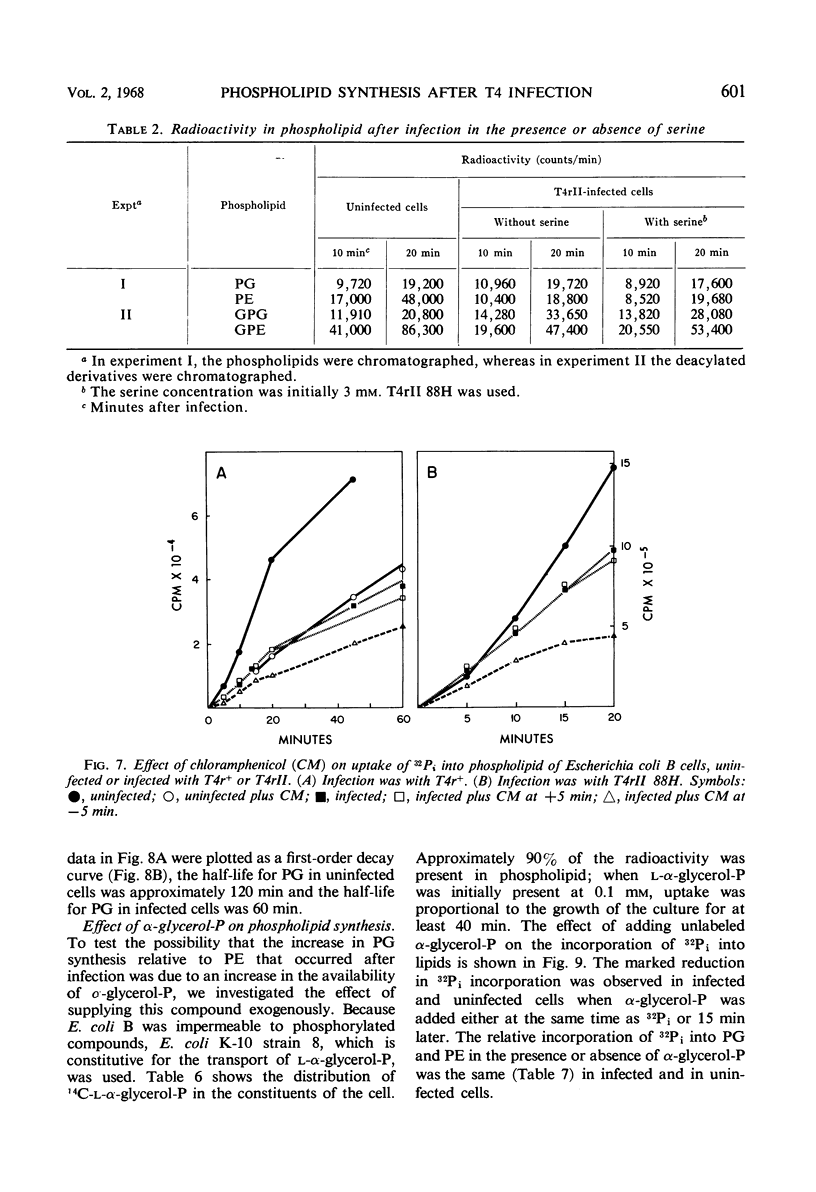
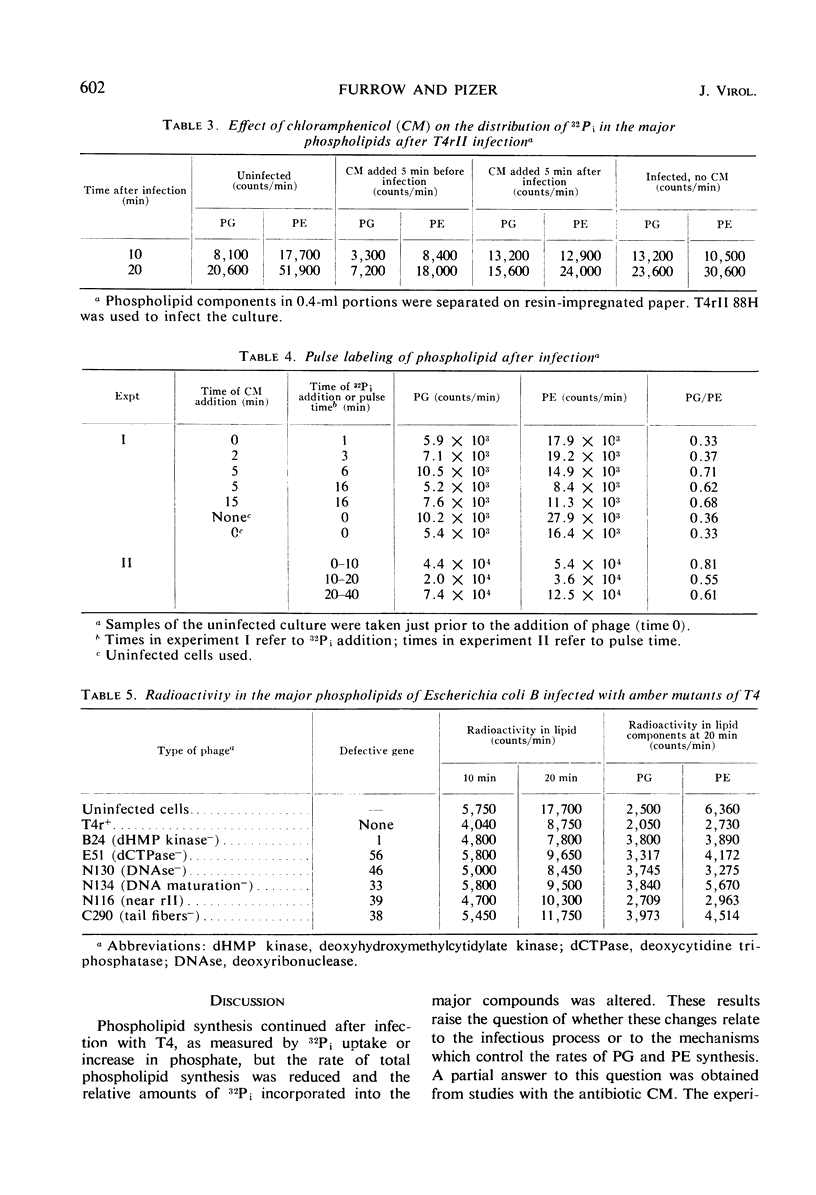
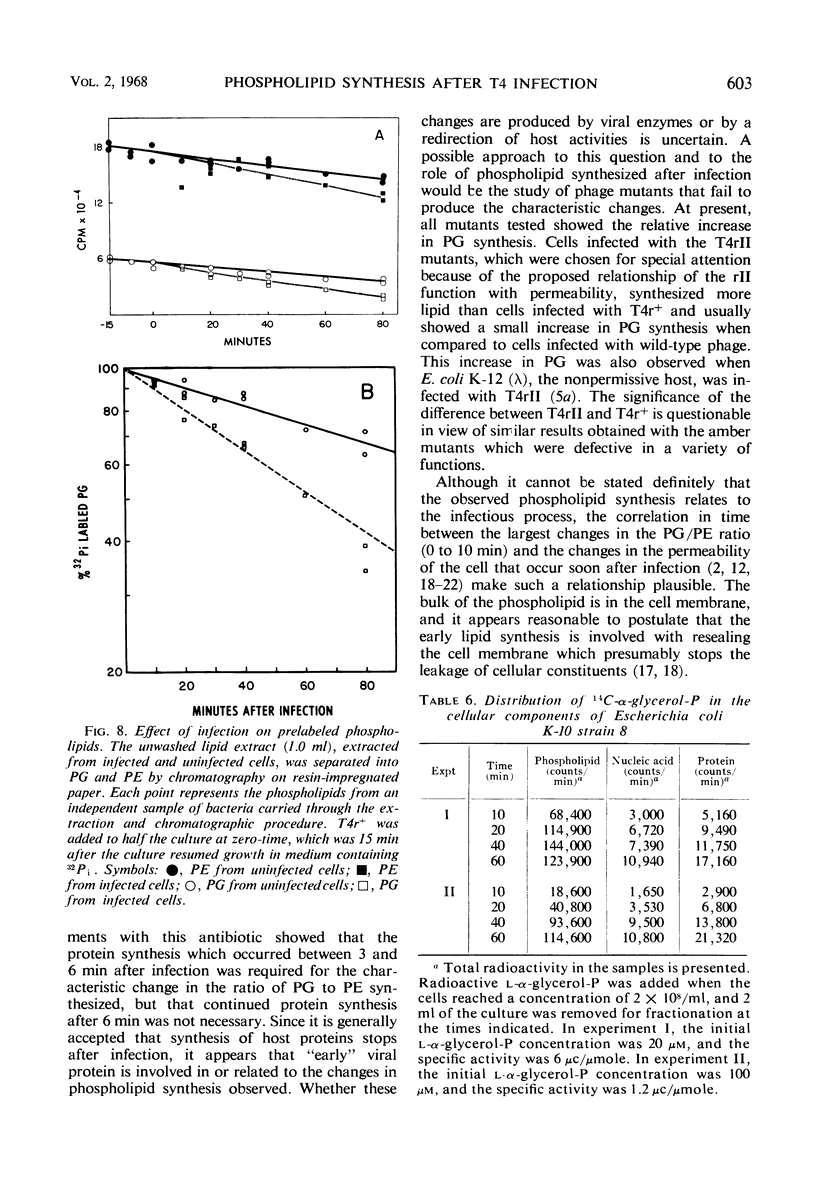
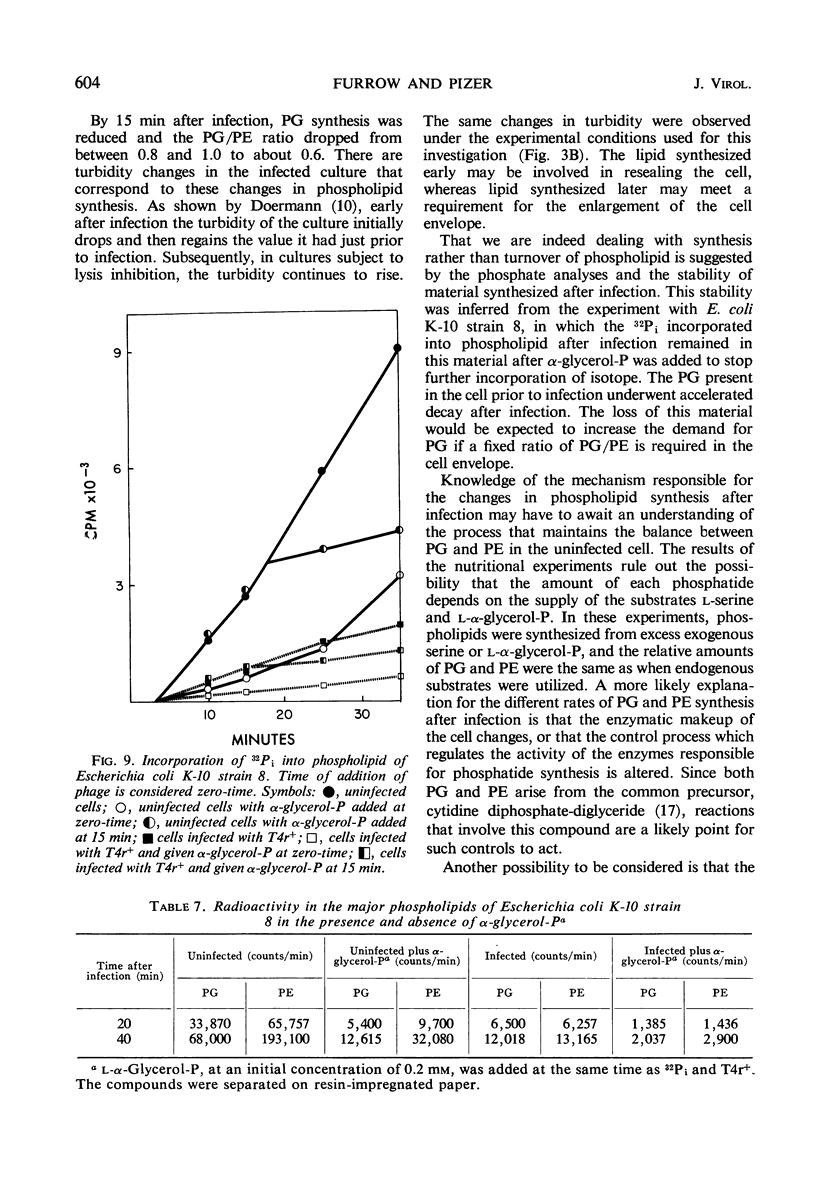
After infection of Escherichia coli with T4 phage, phospholipid synthesis continued but at a reduced rate. The same phospholipid components were synthesized as in uninfected cells; however, the relative rates of 32Pi incorporation into phosphatidylglycerol (PG) and phosphatidylethanolamine (PE) were altered. This alteration was most pronounced during the first 10 min after infection. Under these conditions, the isotope incorporated into PG equaled or exceeded that found in PG from uninfected cells. Chloramphenicol (CM) added before, but not 5 min after, infection inhibited the relative increase in PG synthesis, and CM added at different times after infection indicated that a protein synthesized between 3 and 6 min was required for this change to occur. Supplies of exogenous l-serine or l-α-glycerol-P failed to affect the relative rates of 32Pi incorporation into PG and PE by infected or uninfected cells. Phospholipid synthesis was somewhat higher after infection with T4rII mutants than after infection with wild-type phage. After infection with these mutants or several amber mutants, the relative synthesis of PG and PE was characteristic of T4r+-infected cells. The phospholipid synthesized after infection did not rapidly turn over, but infection accelerated the loss of PG synthesized prior to infection.
Full text
PDF

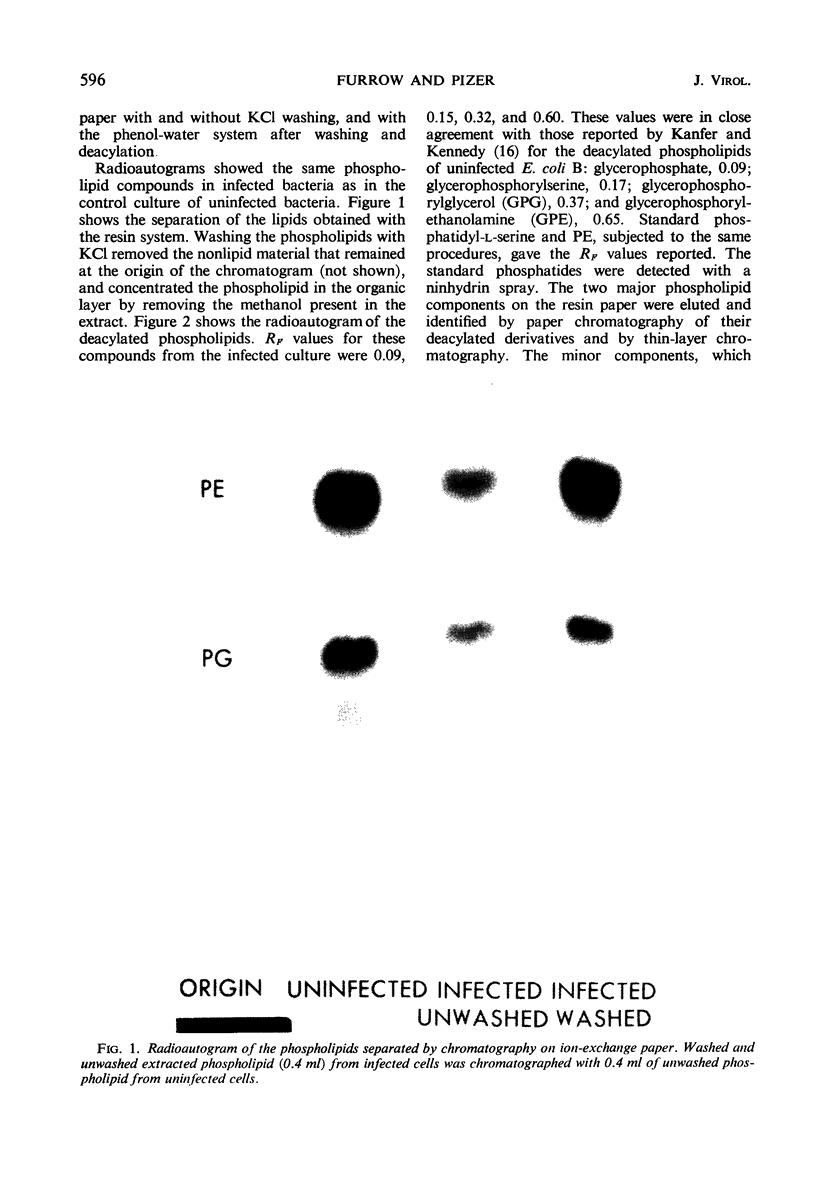
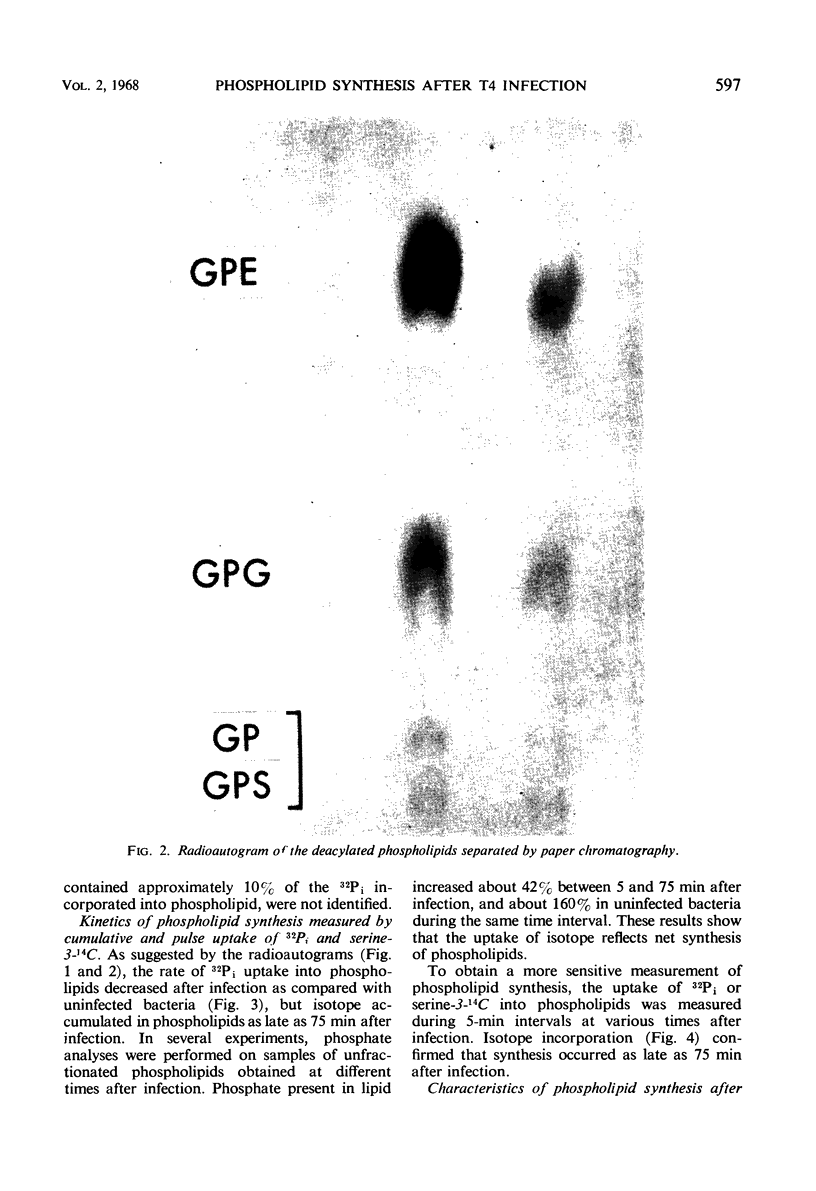
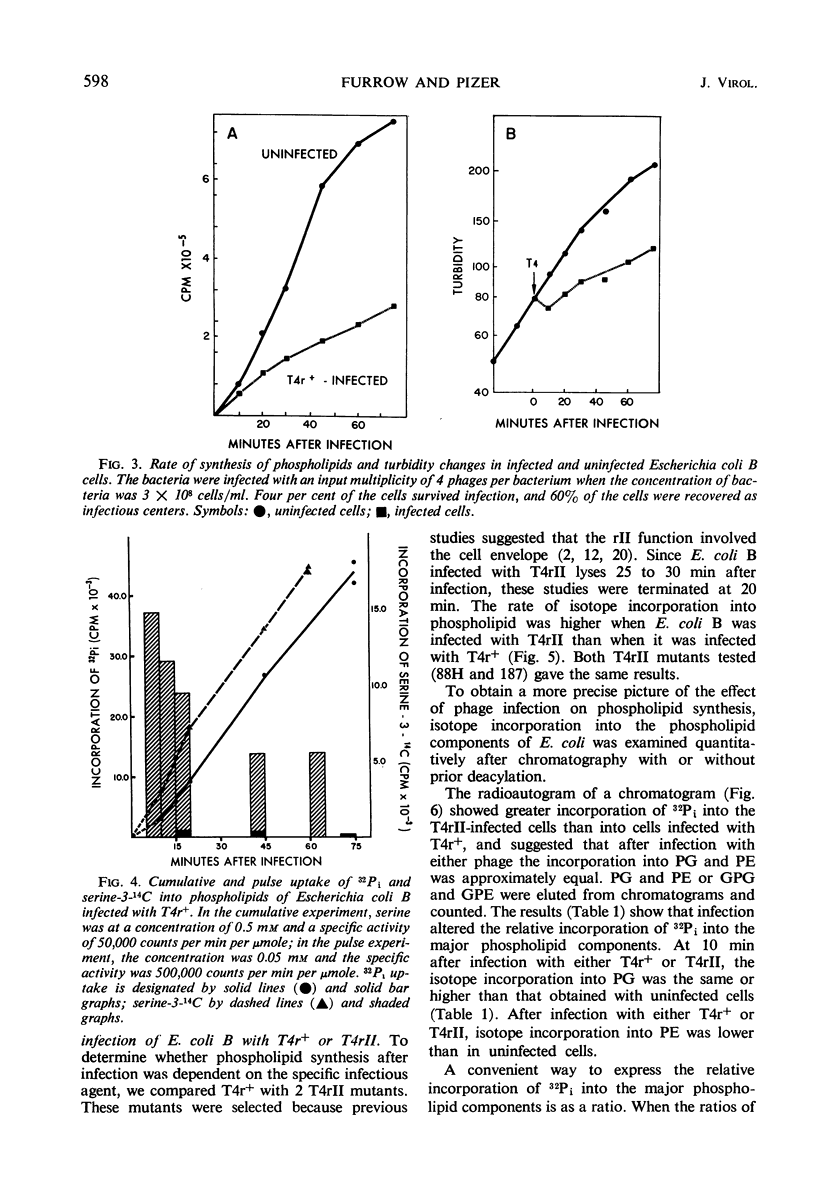
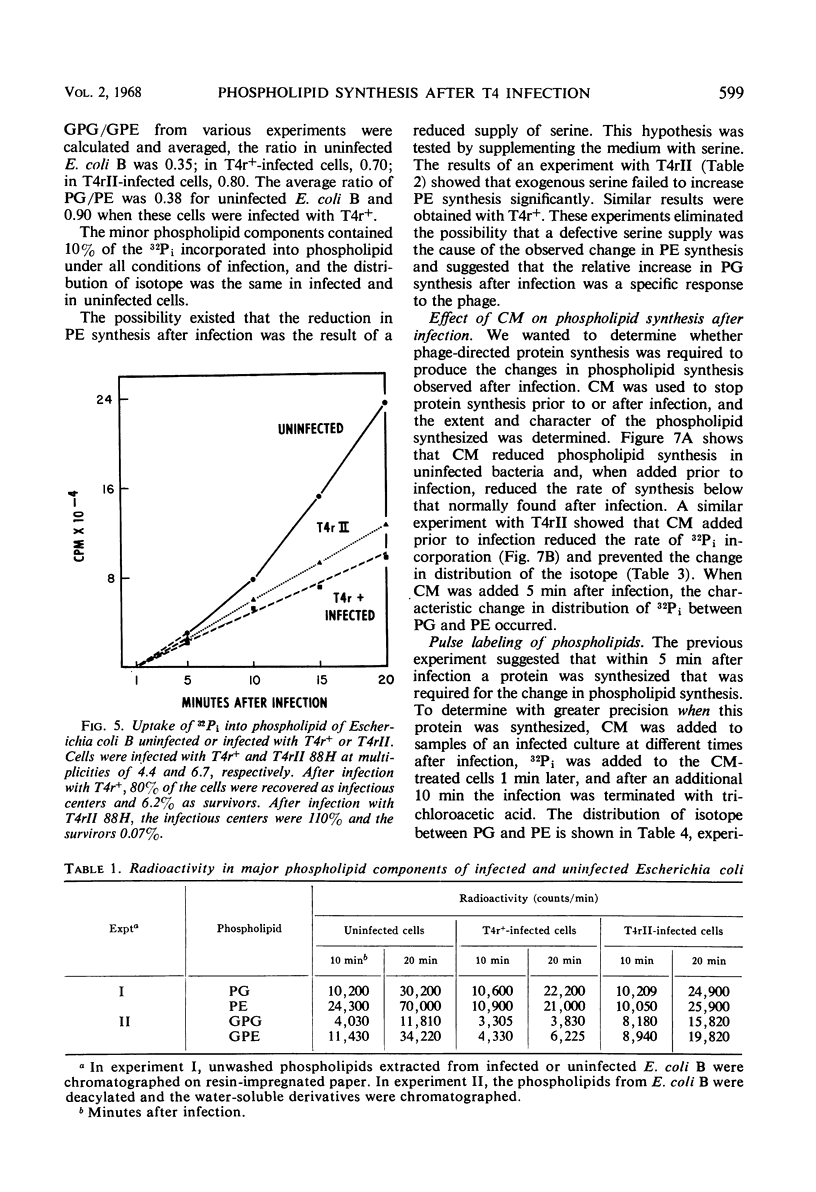
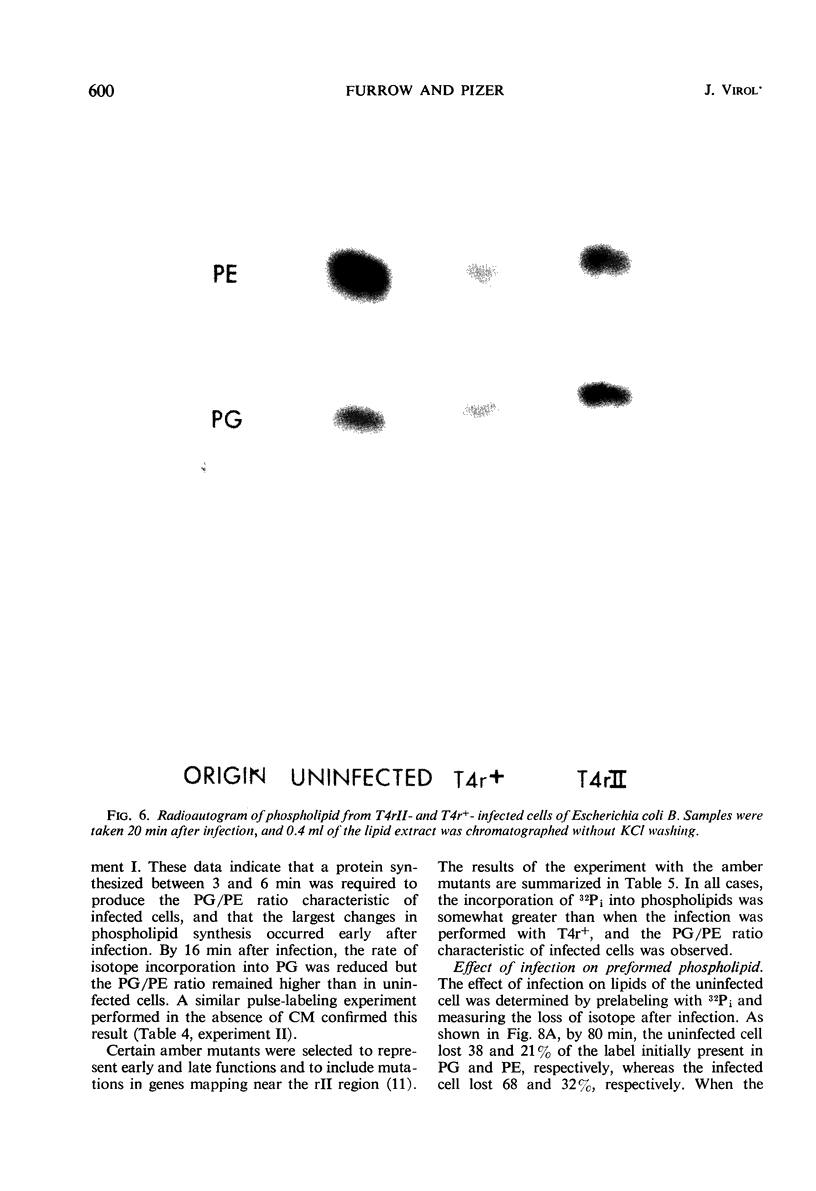

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BALLOU C. E., VILKAS E., LEDERER E. Structural studies on the myo-inositol phospholipids of Mycobacterium tuberculosis (var. bovis, strain BCG). J Biol Chem. 1963 Jan;238:69–76. [PubMed] [Google Scholar]
- BARTLETT G. R. Phosphorus assay in column chromatography. J Biol Chem. 1959 Mar;234(3):466–468. [PubMed] [Google Scholar]
- BUBLITZ C., KENNEDY E. P. Synthesis of phosphatides in isolated mitochondria. III. The enzymatic phosphorylation of glycerol. J Biol Chem. 1954 Dec;211(2):951–961. [PubMed] [Google Scholar]
- Buller C. S., Astrachan L. Replication of T4rII bacteriophage in Escherichia coli K-12 (lambda). J Virol. 1968 Apr;2(4):298–307. doi: 10.1128/jvi.2.4.298-307.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- COHEN S. S. The enzymes of the host cell as a requirement for virus synthesis. Ann N Y Acad Sci. 1952 Jul 10;54(6):902–908. doi: 10.1111/j.1749-6632.1952.tb39965.x. [DOI] [PubMed] [Google Scholar]
- COHEN S. S. Virus-induced acquisition of metabolic function. Fed Proc. 1961 Jul;20:641–649. [PubMed] [Google Scholar]
- DAWSON R. M. The measurement of 32P labelling of individual kephalins and lecithin in a small sample of tissue. Biochim Biophys Acta. 1954 Jul;14(3):374–379. doi: 10.1016/0006-3002(54)90195-x. [DOI] [PubMed] [Google Scholar]
- Doermann A. H. Lysis and Lysis Inhibition with Escherichia coli Bacteriophage. J Bacteriol. 1948 Feb;55(2):257–276. doi: 10.1128/jb.55.2.257-276.1948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GAREN A. Physiological effects of rII mutations in bacteriophage T4. Virology. 1961 Jun;14:151–163. doi: 10.1016/0042-6822(61)90190-8. [DOI] [PubMed] [Google Scholar]
- HAYASHI S., KOCH J. P., LIN E. C. ACTIVE TRANSPORT OF L-ALPHA-GLYCEROPHOSPHATE IN ESCHERICHIA COLI. J Biol Chem. 1964 Sep;239:3098–3105. [PubMed] [Google Scholar]
- HERSHEY A. D. An upper limit to the protein content of the germinal substance of bacteriophage T2. Virology. 1955 May;1(1):108–127. doi: 10.1016/0042-6822(55)90009-x. [DOI] [PubMed] [Google Scholar]
- KANFER J., KENNEDY E. P. METABOLISM AND FUNCTION OF BACTERIAL LIPIDS. I. METABOLISM OF PHOSPHOLIPIDS IN ESCHERICHIA COLI B. J Biol Chem. 1963 Sep;238:2919–2922. [PubMed] [Google Scholar]
- KANFER J., KENNEDY E. P. METABOLISM AND FUNCTION OF BACTERIAL LIPIDS. II. BIOSYNTHESIS OF PHOSPHOLIPIDS IN ESCHERICHIA COLI. J Biol Chem. 1964 Jun;239:1720–1726. [PubMed] [Google Scholar]
- Kaback H. R., Stadtman E. R. Proline uptake by an isolated cytoplasmic membrane preparation of Escherichia coli. Proc Natl Acad Sci U S A. 1966 Apr;55(4):920–927. doi: 10.1073/pnas.55.4.920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- PUCK T. T., LEE H. H. Mechanism of cell wall penetration by viruses. I. An increase in host cell permeability induced by bacteriophage infection. J Exp Med. 1954 May 1;99(5):481–494. doi: 10.1084/jem.99.5.481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- PUCK T. T., LEE H. H. Mechanism of cell wall penetration by viruses. II. Demonstration of cyclic permeability change accompanying virus infection of Escherichia coli B cells. J Exp Med. 1955 Feb 1;101(2):151–175. doi: 10.1084/jem.101.2.151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sekiguchi M. Studies on the physiological defect in rII mutants of bacteriophage T4. J Mol Biol. 1966 Apr;16(2):503–522. doi: 10.1016/s0022-2836(66)80188-2. [DOI] [PubMed] [Google Scholar]
- Silver S. Acridine sensitivity of bacteriophage T2: a virus gene affecting cell permeability. J Mol Biol. 1967 Oct 14;29(1):191–202. doi: 10.1016/0022-2836(67)90190-8. [DOI] [PubMed] [Google Scholar]