Abstract
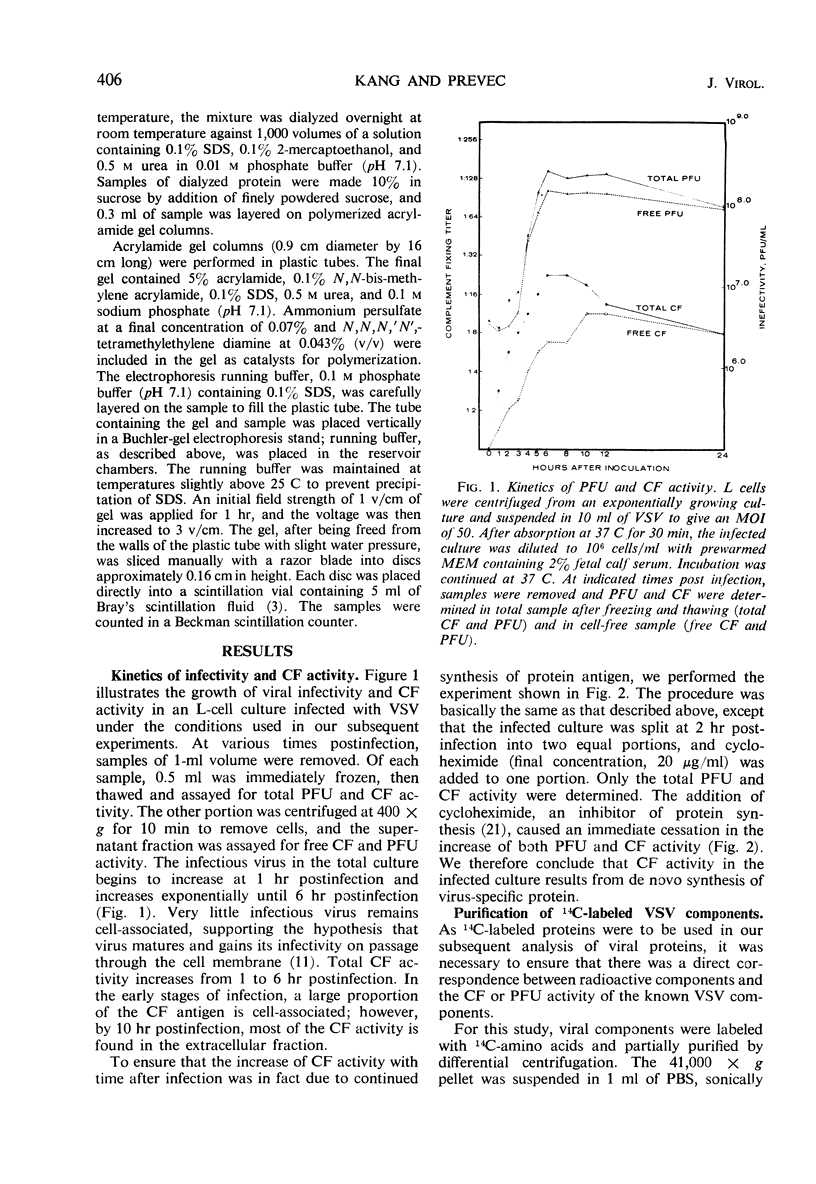
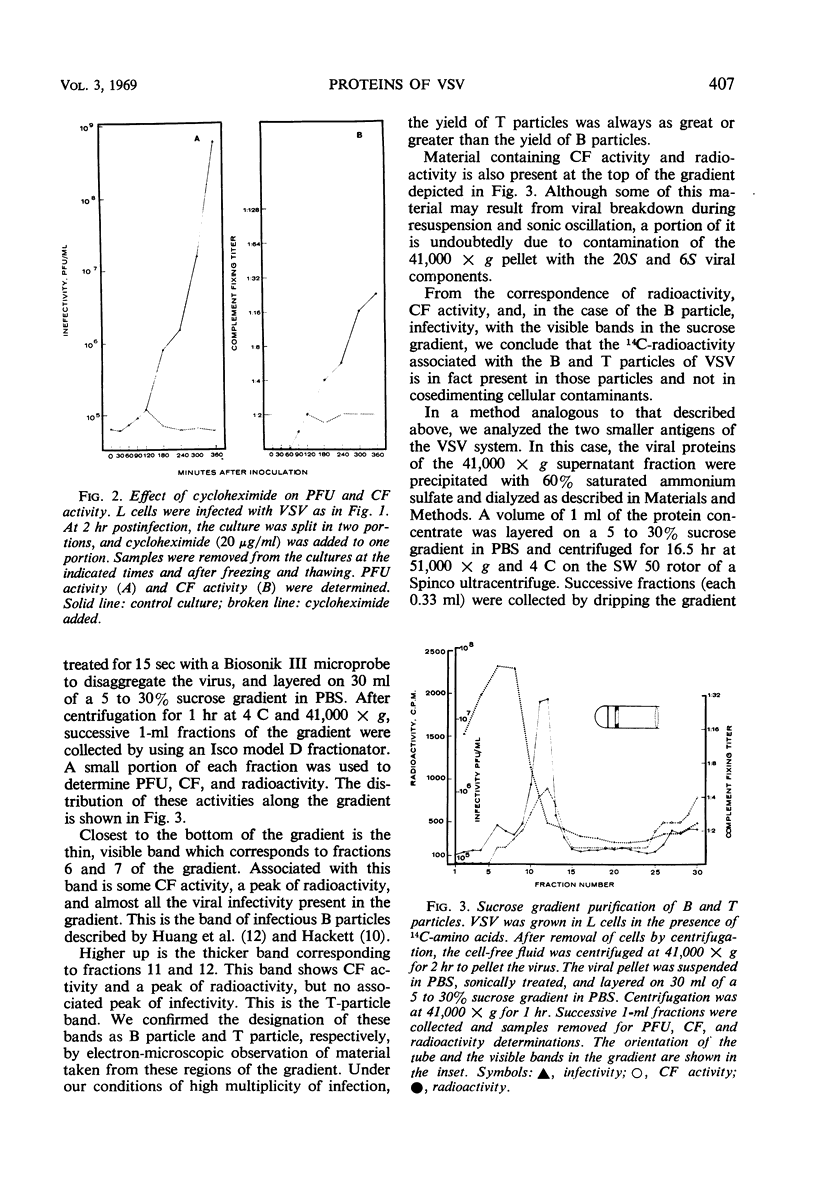
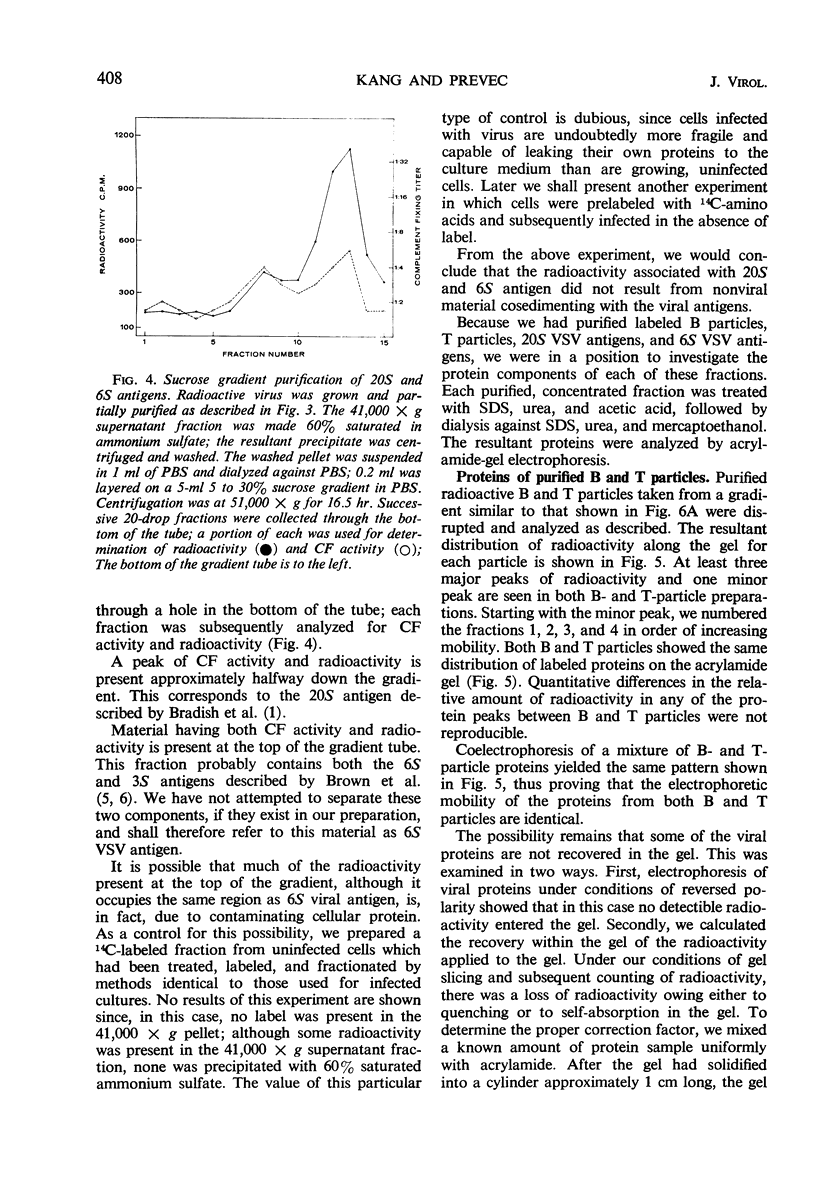
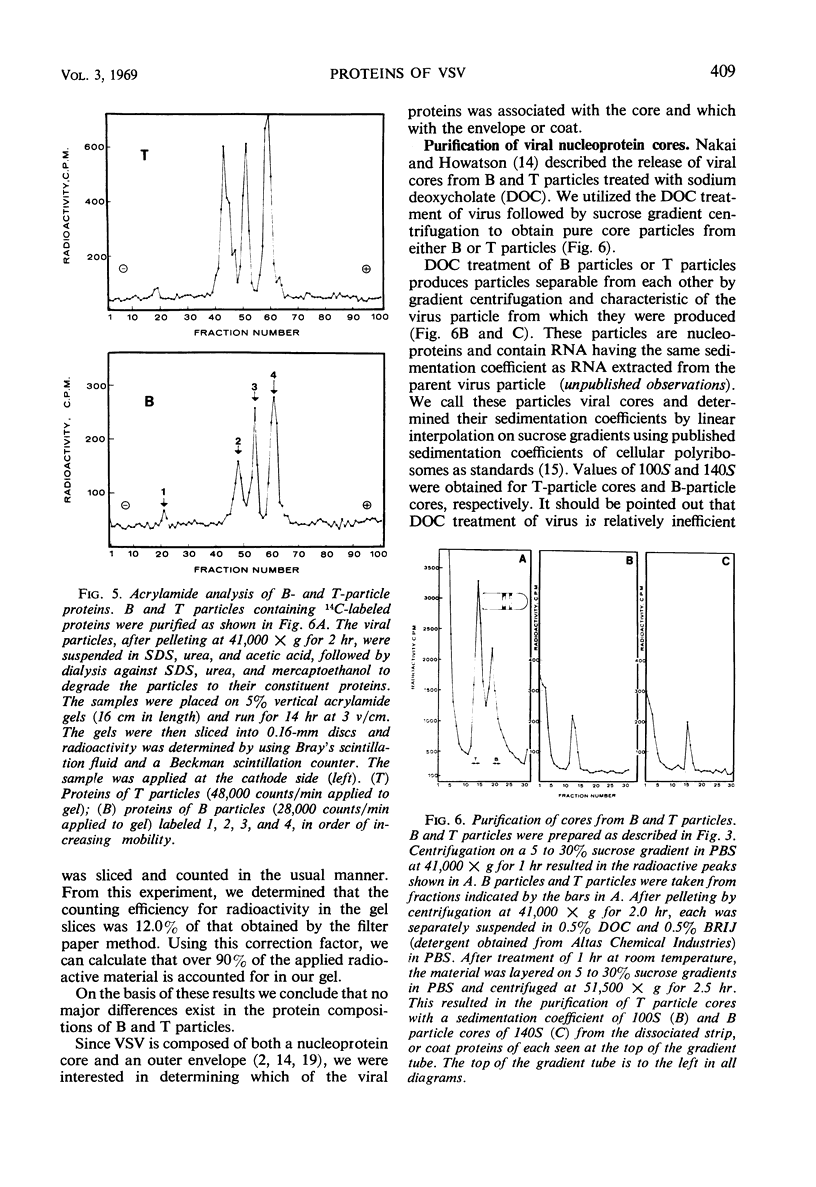
Infection of L cells with vesicular stomatitis virus results in the release, into the cell-free fluid, of four antigenic components separable by rate zonal centrifugation on sucrose gradients. The largest antigens are the infectious (B) particle and a shorter noninfectious, autointerfering (T) particle. The two small antigens are characterized by sedimentation coefficients of approximately 20S and 6S. Treatment of purified B or T particles with sodium deoxycholate results in the release from the particle of a nucleoprotein core which can be purified on sucrose gradient and which has a sedimentation coefficient characteristic of the virus from which it arose. Utilizing purified antigens labeled with 14C-amino acids during growth, we examined the protein constituents of each antigen by acrylamide-gel electrophoresis. The proteins of B and T particles are identical, each containing one minor (virus protein 1) and three major (virus proteins 2, 3, and 4) proteins, numbered in order of increasing mobility. Virus protein 3 originates from the nucleoprotein core, whereas proteins 2 and 4 come from the coat. The origin of virus protein 1 is not known. The 20S antigen contains a single protein equivalent to virus protein 3, whereas the 6S antigen shows a single protein which is similar to, but probably distinct from, virus protein 2.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BELLETT A. J., COOPER P. D. Some properties of the transmissible interfering component of vesicular stomatitis virus preparations. J Gen Microbiol. 1959 Dec;21:498–509. doi: 10.1099/00221287-21-3-498. [DOI] [PubMed] [Google Scholar]
- BRADISH C. J., BROOKSBY J. B., DILLON J. F., Jr Biophysical studies of the virus system of vesicular stomatitis. J Gen Microbiol. 1956 Apr;14(2):290–314. doi: 10.1099/00221287-14-2-290. [DOI] [PubMed] [Google Scholar]
- Bradish C. J., Kirkham J. B. The morphology of vesicular stomatitis virus (Indiana C) derived from chick embroyos or cultures of BHK21/13 cells. J Gen Microbiol. 1966 Sep;44(3):359–371. doi: 10.1099/00221287-44-3-359. [DOI] [PubMed] [Google Scholar]
- Brown F., Cartwright B., Almeida J. D. The antigens of vesicular stomatitis virus. I. Separation and immunogenicity of three complement-fixing components. J Immunol. 1966 Mar;96(3):537–545. [PubMed] [Google Scholar]
- Brown F., Cartwright B., Smale C. J. The antigens of vesicular stomatitis virus. 3. Structure and immunogenicity of antigens derived from the virion by treatment with Tween and ether. J Immunol. 1967 Jul;99(1):171–177. [PubMed] [Google Scholar]
- Brown F., Cartwright B. The antigens of vesicular stomatitis virus. II. The presence of two low molecular weight immunogens in virus suspensions. J Immunol. 1966 Nov;97(5):612–620. [PubMed] [Google Scholar]
- Brown F., Martin S. J., Cartwright B., Crick J. The ribonucleic acids of the infective and interfering components of vesicular stomatitis virus. J Gen Virol. 1967 Oct;1(4):479–486. doi: 10.1099/0022-1317-1-4-479. [DOI] [PubMed] [Google Scholar]
- Cartwright B., Pearce C. A. Evidence for a host cell component in vesicular stomatitis virus. J Gen Virol. 1968 Mar;2(2):207–214. doi: 10.1099/0022-1317-2-2-207. [DOI] [PubMed] [Google Scholar]
- HACKETT A. J. A POSSIBLE MORPHOLOGIC BASIS FOR THE AUTOINTERFERENCE PHENOMENON IN VESICULAR STOMATITIS VIRUS. Virology. 1964 Sep;24:51–59. doi: 10.1016/0042-6822(64)90147-3. [DOI] [PubMed] [Google Scholar]
- HOWATSON A. F., WHITMORE G. F. The development and structure of vesicular stomatitis virus. Virology. 1962 Apr;16:466–478. doi: 10.1016/0042-6822(62)90228-3. [DOI] [PubMed] [Google Scholar]
- Huang A. S., Greenawalt J. W., Wagner R. R. Defective T particles of vesicular stomatitis virus. I. Preparation, morphology, and some biologic properties. Virology. 1966 Oct;30(2):161–172. doi: 10.1016/0042-6822(66)90092-4. [DOI] [PubMed] [Google Scholar]
- Huang A. S., Wagner R. R. Defective T particles of vesicular stomatitis virus. II. Biologic role in homologous interference. Virology. 1966 Oct;30(2):173–181. doi: 10.1016/0042-6822(66)90093-6. [DOI] [PubMed] [Google Scholar]
- Nakai T., Howatson A. F. The fine structure of vesicular stomatitis virus. Virology. 1968 Jun;35(2):268–281. doi: 10.1016/0042-6822(68)90267-5. [DOI] [PubMed] [Google Scholar]
- Noll H. Characterization of macromolecules by constant velocity sedimentation. Nature. 1967 Jul 22;215(5099):360–363. doi: 10.1038/215360a0. [DOI] [PubMed] [Google Scholar]
- PREVEC L., WHITMORE G. F. PURIFICATION OF VESICULAR STOMATITIS VIRUS AND THE ANALYSIS OF P32-LABELED VIRAL COMPONENTS. Virology. 1963 Jul;20:464–471. doi: 10.1016/0042-6822(63)90095-3. [DOI] [PubMed] [Google Scholar]
- SEVER J. L. Application of a microtechnique to viral serological investigations. J Immunol. 1962 Mar;88:320–329. [PubMed] [Google Scholar]
- Shapiro A. L., Viñuela E., Maizel J. V., Jr Molecular weight estimation of polypeptide chains by electrophoresis in SDS-polyacrylamide gels. Biochem Biophys Res Commun. 1967 Sep 7;28(5):815–820. doi: 10.1016/0006-291x(67)90391-9. [DOI] [PubMed] [Google Scholar]
- Simpson R. W., Hauser R. E. Structural components of vesicular stomatitis virus. Virology. 1966 Aug;29(4):654–667. doi: 10.1016/0042-6822(66)90289-3. [DOI] [PubMed] [Google Scholar]
- Summers D. F., Maizel J. V., Jr, Darnell J. E., Jr Evidence for virus-specific noncapsid proteins in poliovirus-infected HeLa cells. Proc Natl Acad Sci U S A. 1965 Aug;54(2):505–513. doi: 10.1073/pnas.54.2.505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watanabe Y., Kudo H., Graham A. F. Selective inhibition of reovirus ribonucleic acid synthesis by cycloheximide. J Virol. 1967 Feb;1(1):36–44. doi: 10.1128/jvi.1.1.36-44.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]



