Abstract
Tumor infiltrating lymphocytes (TILs) convey clinically relevant information for primary breast cancers (PBCs). However, limited data characterizing the immunobiology of metastatic breast cancers (MBCs) are available. Here, we examine TILs in surgically resected MBCs relative to their matched PBCs. Tissue microarrays of PBCs and MBCs were labeled for CD3 (total T -cells), CD4 (helper T-cells), CD8 (cytotoxic T-cells), FoxP3 (regulatory T-cells), and CD20 (B-cells) to characterize TILs. Expression of estrogen receptor (ER), progesterone receptor (PR) and HER-2 classified the tumors as luminal (ER+/PR+/HER-2−), triple negative (ER−/PR−/HER-2−), or HER-2+ (ER−/PR−/HER-2+). These analyses reveal five novel findings. First, MBCs overall contained fewer TILs (mean 35.1 CD3+ TILs/HPF) than their matched PBCs (mean 23.6 CD3+ TILS/HPF), with fewer CD20+ cells than CD3+ cells in PBC and MBC (p=0.0247). Second, the number of CD3+, CD8+, CD4+, and FoxP3 TILs were decreased in triple negative MBCs relative to matched PBCs, whereas only CD8+ TILs were decreased in luminal MBCs relative to matched PBCs. Third, triple negative MBCs contain fewer TILS (mean 16 CD3+ TILs/HPF) than luminal MBCs (mean 21.7 CD3+ TILs/HPF). Fourth, brain metastases contained fewer TILs relative to MBC from other sites. Finally, in this series a CD8+/FoxP3+ T-cell ratio ≥3 in PBCs was associated with improved overall survival from diagnosis, whereas a CD8+/FoxP3+ T-cell ratio <3 in MBCs at first relapse was associated with improved overall survival. These findings suggest that evaluating the immunologic microenvironment of both PBCs and MBCs may yield important clinical information to guide breast cancer prognosis and therapy.
Keywords: breast cancer, tumor infiltrating lymphocytes (TILs), regulatory T cells (Treg), FoxP3
1. INTRODUCTION
Breast cancers have historically been considered immunologically silent. More recently, accumulating data have highlighted an important role for tumor infiltrating lymphocytes (TILs) in relaying clinically relevant prognostic and predictive information for primary breast cancers (PBCs). High-risk breast tumors (characterized by lack of estrogen receptor (ER) and progesterone receptor (PR) expression, high grade, and/or lymph node (LN) involvement) typically have higher CD3+, CD8+, and FoxP3+ T cell infiltrates at diagnosis than low-risk breast tumors [1]. In PBCs, higher numbers of both CD8+ T lymphocytes and CD20+ B lymphocytes are an independent predictor of improved patient survival [2, 3]. Conversely, higher numbers of FoxP3+ regulatory T cells (Tregs) are associated with shorter relapse-free survival [4]. Notably, the presence of TILs can predict complete pathologic response (cPR) to neoadjuvant chemotherapy [5, 6]. More specifically, T cell infiltrates enriched for CD8+ T lymphocytes (cytotoxic T cells) relative to FoxP3+ Tregs are associated with cPR to neoadjuvant chemotherapy for breast cancer [1] and with improved disease-free survival (DFS) and overall survival (OS) [7]. However, there are few data characterizing the immune infiltrates of metastatic breast tumors, and no prior studies have characterized TIL populations present in metastatic breast tumors relative to their matched PBCs. Here, we profile the phenotype of TILs in a group of surgically resected metastatic breast cancers (MBCs) representing first relapse relative to their matched PBCs. First, we compared the level of lymphocyte infiltration between primary breast cancers and their paired metastases. Second, we analyzed differences between lymphocyte infiltrates present within triple negative breast cancers (TNBCs) and luminal breast cancers. Third, we evaluated metastases for possible differences in lymphocyte infiltrates across organ sites. Fourth, we evaluated the prognostic value of immune infiltrates in predicting outcome.
2. MATERIALS AND METHODS
2.1 Tissue microarray construction
This study was reviewed and approved by the Institutional Review Board of the Johns Hopkins Medical Institutions. Tissue microarrays (TMAs) were constructed from archived paraffin tissue blocks of PBCs and surgically-resected matched MBCs from 16 patients as previously described [8]. One of these samples was lost in processing (SPC6), leaving 15 evaluable cases. Each TMA consisted of 99 spots, each measuring 1.4 mm in diameter, and 5–10 spots per tumor were taken to minimize sampling error. In total, the 3 TMAs contained 135 spots of PBC and 135 spots of MBC from the original 16 cases. With the loss of one case in processing, 125 spots of PBC and 125 spots of MBC were evaluable for these studies.
2.2 Immunohistochemistry and expression scoring
The TMAs were labeled by immunohistochemistry for CD3 (mouse monoclonal, clone PS1, catalogue no. ORG-8982, prediluted, Leica Microsystems; Bannockburn, IL), CD4 (rabbit monoclonal, clone Sp35, catalogue no. 790-4423, prediluted, Ventana Medical Systems Inc, Tucson, AZ), CD8 (mouse monoclonal, clone C8/C8144B, catalogue no. 760-4250, dilution: 5 μg, Cell Marque; Rockin, CA), and CD20 (monoclonal, clone MS/L26, catalogue no. 760-2531, dilution: 5ML/5UG, Ventana Medical Systems Inc, Tucson, AZ) using standard methods. Briefly, unstained 5-μm sections were cut from paraffin TMA blocks; slides were de-paraffinized by routine techniques, steamed for 30 min at 90°C in 1X sodium citrate buffer, cooled for 5 min, then incubated with the primary antibody.
The TMAs were stained by immunohistochemistry (IHC) for FoxP3 using a mouse monoclonal anti-FoxP3 antibody (eBioscience catalog# 14-4777-80; clone 236A/E7) at a dilution of 1:250 using routine techniques. Briefly, unstained 5-μm sections were cut from paraffin TMA blocks; slides were de-paraffinized, rehydrated and heat-induced epitope retrieval pre-treatment was conducted by immersing the slides in a commercial retrieval buffer (DAKO catalog# S170084-2), and steaming for 40 minutes. Endogenous peroxidase activity was blocked by hydrogen peroxide treatment and the primary antibody was applied and incubated overnight at 4 degrees Celsius. Secondary detection was accomplished using an HRP/DAB system (EnVision Plus Detection System; DAKO catalog #K4007) followed by light nuclear counterstaining with Mayers hematoxylin.
The lymphocytes were characterized as tumor infiltrating if they were located within one high power field (hpf; 40x) of the epithelial tumor cells. FoxP3 positivity was defined as any nuclear labeling within lymphocytes. The total number of positive lymphocytes per immunostain was counted in one hpf per TMA core and averaged to result in total number lymphocytes/hpf. The IHC was scored separately and blindly of the clinicopathologic patient characteristics. Fisher’s exact tests were performed to assess statistical significance of the categorical data.
In addition, the TMAs were also labeled for estrogen receptor (ER) (monoclonal, clone 611, catalogue no. ORG-8871, prediluted, LEICA Microsystems; Bannockburn, IL), progesterone receptor (PR) (monoclonal, clone 16, catalogue no. ORG-8721 prediluted, LEICA Microsystems; Bannockburn, IL), and HER-2 (Herceptest kit, Dakocytomation, CA) using standard methods as previously described [8]. ER and PR expression were scored both by labeling intensity (none, weak, moderate or strong) and the percentage of nuclear labeling (0%–100%). Any labeling was considered a positive result. HER-2 expression was scored using established criteria from 0 to 3+ using labeling intensity and proportion of complete membranous staining. The level of hormone receptor and HER-2 expression was scored individually on each TMA spot, and an average of all spots per site was calculated.
The hormone receptor and HER-2 expression was used to classify cases into the following categories: luminal (ER+/PR+/HER2−, ER+/PR−/HER-2−, or ER−/PR+/HER-2+), triple negative (TNBC) (ER−/PR−/HER-2−), HER-2+ (ER−/PR−/HER-2+), and luminal loss (loss of ER or PR from PBC to MBC) using a previously described and validated IHC surrogate for molecular breast cancer subtype [9, 10].
2.3 Statistical methods
Survival curves and median time of survival were calculated using the Kaplan-Meier method. The Log-Rank test was used to compare two survival distributions. All P values are reported as 2-sided and all analyses were conducted using SAS software 9.2 (SAS Institute, Cary, NC).
3. RESULTS
3.1. Cohort of Primary Breast Cancers and Paired Metastases
The clinicopathologic characteristics of the cohort of patients with primary breast cancers and paired metastases resected at first relapse are shown in Table 1. To summarize, the primary breast cancers included 8 luminal (ER+/PR+/HER-2−, ER+/PR−/HER-2−, or ER−/PR+/HER-2−), 2 HER-2+ (ER−/PR−/HER-2+), and 6 triple negative (ER−/PR−/HER-2−) immunohistochemical subtypes. One HER-2+ case was lost in processing, leaving a total of 15 cases with matched PBC and MBC evaluable for analysis. The results of the ER, PR, and HER-2 staining of the PBCs performed on the TMAs were completely concordant with those performed on whole tumor sections at the time of diagnosis and documented in the surgical pathology reports. There were no cases of luminal loss (loss of ER or PR expression with transition from PBC to MBC).
Table 1.
Clinicopathologic Characteristics of Matched Primary Breast Cancer and Paired Metastasis Cohort
| Case | Age at Diagnosis (years) | Pathologic Stage at Diagnosis | Primary Tumor Type and Grade | Phenotype | Interval to Metastasis (years) | Months until death | Metastasis Location |
|---|---|---|---|---|---|---|---|
| SPC1 | 33 | T3N1M1 | IDC, Grade 3 | HER-2 | At presentation | 22 | Lung |
| SPC2 | 50 | T2N1 | IDC, Grade 3 | Luminal | 6 | ** | Lung |
| SPC3 | 34 | T2N2M1 | IDC, Grade 3 | TNBC | At presentation | 9 | Brain |
| SPC4 | 38 | T1cN0 | ILC, Grade 2 | Luminal | 3 | 65 | Ovary |
| SPC5 | 45 | T1aN1 | IDC, Grade 3 | Luminal | 7 | *** | Lung |
| SPC6 | 36 | yT1cN0 | IDC, Grade 2 | HER-2 | 4 | 58 | Brain |
| SPC7 | 39 | T2N0 | IDC, Grade 3 | TNBC | 4 | 67 | Brain |
| SPC8 | 53 | T3N1 | IDC, Grade 3 | TNBC | 1 | 24 | Brain |
| SPC9 | 38 | T1cN0 | IDC, Grade 3 | TNBC | 5 | 62 | Brain |
| SPC10 | 53 | T2N2 | IDC, Grade 3 | Luminal | 5 | 38 | Brain |
| SPC11 | 58 | T2N0 | ILC, Grade 2 | Luminal | 7 | 90 | GI (Bowel) |
| SPC12 | 56 | TXNXM1 | ILC, Grade 2 | Luminal | At presentation | 21 | GI (Bowel) |
| SPC13 | 44 | T2N0 | IDC, Grade 3 | TNBC | 1 | *** | Lung |
| SPC14 | 33 | yT2N1 | IDC, Grade 2 | Luminal | 2 | *** | GI (Pancreas) |
| SPC15 | 57 | T1N0 | IDC, Grade 3 | TNBC | 6 | *** | Lung |
| SPC16 | 36 | yT1cN0 | IDC, Grade 3 | Luminal | 2 | ** | Brain |
Abbreviations: IDC=invasive ductal carcinoma; ILC=invasive lobular carcinoma; TNBC=triple negative breast cancer.
Case SPC6 was lost in processing and was therefore not evaluable for analysis.
No recent follow up data available
Patients remain alive/living with relapsed disease
Three patients had invasive lobular carcinoma (ILC; SPC 4, 11, and 12), and the remaining 12 patients had invasive ductal carcinoma (IDC). The sites of resected metastases represented solitary metastases and included the brain (n=6), the lung (n=5), the gastrointestinal tract (two bowel and one pancreas, n=3), and the ovary (n=1). The 15 patients ranged in age from 33 to 58 years (average 44 years). Three patients presented with Stage 4 disease at the time of diagnosis (SPC 1, 3, and 12). For the remaining 12 patients, the average time to the development of metastatic disease was 4.1 years (range 1–7 years). The primary breast tumors evaluated on the tissue microarray for cases SPC6, SPC14 and SPC16 were status-post neoadjuvant therapy. All 12 patients who presented with early stage breast cancer received adjuvant chemotherapy prior to disease relapse; chemotherapy drugs included doxorubicin, cyclophosphamide, taxanes, and carboplatinum. Of the 8 patients with luminal breast cancer, 7 were treated with either tamoxifen or an aromatase inhibitor. No patients were treated with ovarian ablation/suppression. The remaining patient with luminal disease declined endocrine therapy.
3.2 Metastatic Breast Cancers Have Lower Numbers of Tumor Infiltrating Lymphocytes than Matched Primary Breast Cancers
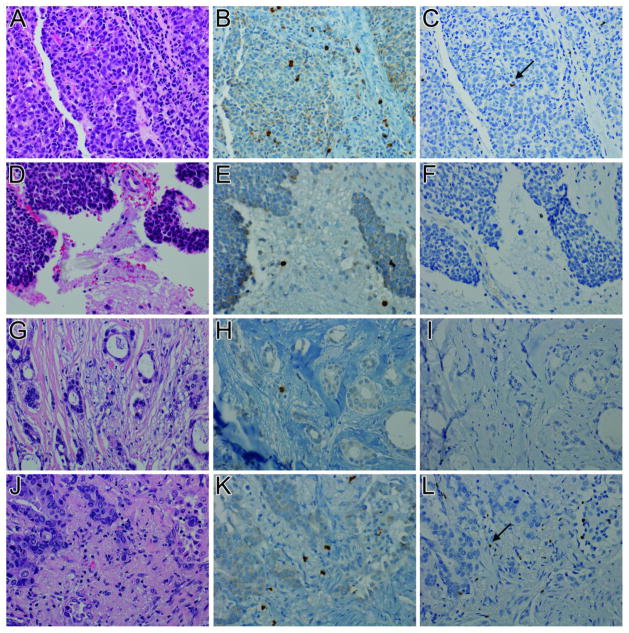
Tumor infiltrating lymphocyte markers were assessed by immunohistochemistry in the TMAs constructed from PBC and MBC for each subject. Examples of immunostaining for CD8 and FoxP3 in primary and metastatic triple negative and luminal breast cancers are shown with one representative case each (Figure 1). Taken overall, metastatic tumors had fewer numbers of infiltrating CD3+, CD8+ CD4+ and FoxP3+ T lymphocytes than their matched primary tumors (Figure 2). Four patients had greater numbers of TILs in their metastases than in their primary breast tumors (SPC1, 2, 5, and 12); these cases consisted of 3 luminal phenotypes and 1 HER-2 phenotype, with metastases to lung (3 cases) and brain (1 case). There were fewer tumor infiltrating CD20+ B cells within metastases than within primary tumors (Figure 2), and there were overall many fewer tumor infiltrating CD20+ B cells than tumor infiltrating CD3+ T cells in both primary and metastatic tumors (p value = 0.0249) (Figure 2). Finally, of the 3 patients who presented with MBC at the time of diagnosis (SPC1, 3, and 12), two (SPC1 and 3) had high levels of FoxP3+ cells per high power field (hpf), defined as ≥15 cells/hpf within their primary breast cancer [4]; no other patients met this criteria for high Treg levels. Overall, these data suggest that the immunologic milieu in patients with MBC is more suppressed than in patients with early stage PBC.
Figure 1. The pattern of tumor infiltrating T-cell lymphocytes in matched primary and metastatic breast carcinomas differs in triple negative and luminal breast carcinoma subtypes.
Triple negative primary breast carcinomas (A, H&E) have more CD8+ (B) and Foxp3+ (C, arrow) T cell tumor infiltrating lymphocytes (TILs) than their matched metastases (D, H&E; E, CD8; F, FoxP3). No FoxP3+ cells are seen in this field of metastatic triple negative breast carcinoma (F). Triple negative primary breast carcinomas also have more T cell TILS than luminal primary breast carcinomas (G, H&E; H, CD8; I, FoxP3). No FoxP3+ cells are seen in this field of primary luminal breast carcinoma (I). In contrast, luminal primary breast carcinomas have fewer T cell TILS than their matched metastatic breast carcinomas (J, H&E; K, CD8; L, FoxP3 with arrow). (x20)
Figure 2. Enumeration of tumor infiltrating lymphocytes in primary (p) and metastatic (m) breast tumors.
Data are represented at number of lymphocyte counts per high power field (hpf, x40), and given as the median and range. CD3=T cells; CD8=cytotoxic T cells; CD4=helper T cells; FoxP3=regulatory T cells; CD20=B cells.
3.3 Triple Negative Breast Cancers Have Greater Numbers of Tumor Infiltrating Lymphocytes than Luminal Breast Cancers
We also examined the relative lymphocyte infiltration of the distinct subtypes of breast cancer. Here, we focused only on TNBCs and luminal breast cancers, as only one HER-2+ tumor/metastasis pair was available for analysis. Overall, primary TNBCs had greater numbers of infiltrating CD3+, CD4+, CD8+, and FoxP3+ TILs than primary luminal cancers (Figures 1 and 3). In contrast, the metastatic TNBCs had fewer TILs than the metastatic luminal cancers. In addition, there were fewer tumor infiltrating CD20+ lymphocytes in metastatic triple negative breast cancers as compared to both primary triple negative breast cancers and metastatic luminal breast cancers (data not shown). These data suggest that primary triple negative breast cancers may be more permissive for an immune response than their paired metastases.
Figure 3. Enumeration of tumor infiltrating lymphocytes by breast tumor subtype and lymphocyte phenotype.
Data are represented at number of cell counts per high power field (hpf, x40), and given as the median and range. CD3=T cells; CD8=cytotoxic T cells; CD4=helper T cells; FoxP3=regulatory T cells; CD20=B cells; pTNBC=primary triple negative breast cancer; mTNBC=metastatic TNBC; pLBC=primary luminal breast cancer; mLBC=metastatic luminal breast cancer.
3.4 Brain Metastases May Uniquely Have Fewer Infiltrating Lymphocytes Compared to the Primary Tumor
We compared changes in infiltrating cells between the PBC and its associated metastases across the distinct metastatic sites (Table 2). Although numbers are small, there was a trend toward fewer lymphocytes of all types in breast cancer metastases to the brain relative to the matched PBC. In contrast, there was no apparent change in the extent of lymphocyte infiltration between the PBC and its associated metastasis when the metastasis was located in the lung, the ovary, or the gastrointestinal tract. This suggests the possibility of a unique immunologic environment within the brain. These findings are consistent with the widely held view of the central nervous system as an immunologically privileged site.
Table 2.
Patterns of Lymphocyte Infiltration Across Metastatic Sites*
| Metastatic Site | CD3 | CD8 | CD4 | FoxP3 | CD20 | N | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Decr | Incr | Decr | Incr | Decr | Incr | Decr | Incr | Decr | Incr | ||
| Brain | 5 | 1 | 6 | 0 | 5 | 1 | 4 | 2 | 6 | 0 | 6 |
| GYN | 1 | 0 | 1 | 0 | 1# | 0 | 1# | 0 | 0 | 1# | 1 |
| GI | 2 | 1 | 2 | 1# | 2 | 1 | 3# | 0 | 1 | 2## | 3 |
| Lung | 2 | 3 | 2 | 3 | 2 | 3 | 3 | 2 | 2# | 3# | 5 |
| TOTAL | 10 | 5 | 11 | 4 | 10 | 5 | 11 | 4 | 9 | 6 | 15 |
Abbreviations: Decr=decreased; Incr=increased; GYN=gynecologic metastasis; GI=gastrointestinal metastasis; N = number.
Comparisons are numbers of cases compared by number of cells per high power field.
One case each shows almost no change;
Two cases show almost no change.
3.5 Distinct Ratios of CD8+/FoxP3+ T Cells in Primary as Compared to Metastatic Breast Tumors May Be Associated with Improved Overall Survival
Finally, we examined the association of the magnitude of the CD8+/FoxP3+ T cell ratio with clinical outcome. For the group overall, the median overall survival (OS) was 66.6 months, and the median progression free survival (PFS) was 45 months. The median ratio of CD8+/FoxP3+ T cells in both the PBCs and MBCs was 3, so this value was chosen as the cut-off to determine the association of this ratio with overall survival. A CD8+/FoxP3+ T cell ratio ≥3 in the PBC was associated with improved overall survival from the time of diagnosis relative to a CD8+/FoxP3+ T cell ratio<3 (Figure 4A). Interestingly, the reverse pattern was seen when examining the CD8+/FoxP3+ T cell ratio in MBCs. Here, a CD8+/FoxP3+ T cell ratio <3 was associated with improved overall survival as measured from the time of first relapse (e.g., the development of the metastasis) (Figure 4B). The difference in survival remained relevant even when adjusted for patient age at diagnosis and the distinct biology of the triple negative and luminal breast cancer subtypes. Of the 3 patients who presented with metastatic disease (SPC 1,3 and 12), all 3 had a CD8+/FoxP3+ T cell ratio <3 in the PBC, which is the ratio in the PBC associated with decreased survival; interestingly, two of these cases (SPC 1 and SPC12) had a CD8+/FoxP3+ cell ratio >3 in the synchronous MBC, which while reversed from the PBC is the pattern in the MBC also associated with decreased survival. All three of these patients had a survival time from diagnosis of less than 22 months. These findings together are consistent with the concept that higher numbers of effector CD8+ T cells and lower numbers of FoxP3+ Tregs represent a productive antitumor immune response that may promote longer tumor control in the PBC. However, the findings also suggest that the immunologic milieu may be altered in the metastatic site, where a lower ratio of CD8+ T cells to FoxP3+ T cells may be beneficial to patient survival.
Figure 4. Distinct ratios of CD8+/FoxP3+ T cells in primary and metastatic breast carcinomas may predict overall survival.
Median overall and progression free survival for the group of fifteen patients overall was 66.6 months and 45 months respectively (data not shown). A. Primary breast carcinomas with a CD8+/FoxP3+ T cell ratio >3 may have improved overall survival as measured from the time of diagnosis compared to those with a ratio of <3. B. Metastatic breast carcinomas with a CD8+/FoxP3+ T cell ratio of <3 may have improved overall survival as measured by survival from first relapse compared to those with a ratio <3.
4. DISCUSSION
Accumulating data have revealed an important role for the immune system in breast cancer growth and progression and in response to therapy. Most of these studies have focused on PBCs. In this study, we analyzed the presence of TILs within a cohort of PBCs matched with their surgically resected metastases at first relapse. To the best of our knowledge, this is the first report evaluating how the pattern of lymphocyte infiltration within breast tumors might change with progression from early stage to relapsed disease.
Previous studies characterizing TILs in PBCs have shown that high numbers of CD8+ T lymphocytes and low numbers of FoxP3+ T cells are associated with better prognosis [1,2,7]. Here, we observed that metastatic breast tumors overall have lower numbers of TILs (CD3+, CD8+, CD4+, and FoxP3+) than their parent primary tumors. In addition, we found that 2 of 3 patients who presented with metastatic disease at the outset had high levels of FoxP3+ cells within their PBC. Primary breast cancers contained very few infiltrating B cells (CD20+), and their associated metastases contained virtually none. These findings together are consistent with the concept that cancers evoke increasing immune suppression with progressive growth and metastasis.
We also evaluated for potential differences in immune infiltrates by molecular breast cancer subtype by applying a validated method of classifying breast cancers into molecular subtypes using a panel of immunohistochemical surrogate markers [9]. We found that primary TNBCs contained greater numbers of TILs than primary luminal cancers, and that primary TNBCs contained greater numbers of CD3+, CD8+, CD4+, and FoxP3+ TILs than their paired metastases, whereas only CD8+ T cells decreased between matched primary luminal and metastatic breast cancers. With only one HER-2+ case, we are unable to make preliminary observations about this tumor subtype. These findings are consistent with previous reports of higher numbers of CD8+ T lymphocytes and FoxP3+ Tregs within more aggressive tumors [1,6,11,12].
Published data examining differences in lymphocyte infiltrates specific to the distinct breast cancer subtypes are conflicting. One study suggests that high levels of infiltrating CD8+ T cells are a favorable prognostic indicator in basal-like breast cancer [13]. Another suggests that high Treg levels are preferentially associated with the basal phenotype, although high Treg infiltration correlated with shorter survival for HER-2+, luminal, and basal-type breast cancers [14]. A third study suggests that luminal breast cancers are more likely to contain large numbers of TILs than non-luminal breast cancers [15,16]. Complementing these findings, medullary breast cancer is a histologic subtype of invasive breast cancer characterized by large anaplastic tumor cells and an associated dense inflammatory infiltrate. It typically has a favorable prognosis relative to conventional ductal and lobular histologic subtypes [17]. Medullary breast cancers have high numbers of CD8+ T lymphocytes relative to FoxP3+ T cells when compared to conventional ductal and lobular breast cancers [18], and also have fewer Tregs [19]. More work is necessary to characterize the subtype-specific immunobiology of the distinct molecular and histologic subtypes of breast cancer.
We also compared changes in infiltrating lymphocytes between the primary breast cancer and its associated metastases across distinct metastatic sites. Although numbers are small, we noted a trend toward fewer lymphocytes infiltrating brain metastases as compared to the primary tumor relative to matched primary and metastatic breast tumors from other sites. This is consistent with recently published data that minimal B- and T-lymphocytes are found in brain metastases from solid tumors, including breast cancer [20].
Finally, we found that higher ratios of CD8+/FoxP3+ T cells in the PBC may be associated with improved overall survival. This is consistent with a previous report suggesting that tumors containing lymphocyte infiltrates enriched for CD8+ T cells relative to FoxP3+ Tregs are associated with improved disease-free and overall survival [7]. Immune infiltrates appeared to predict outcomes more effectively than classical predictive factors, and a score derived from a combination of the CD8+/FoxP3+ T cell ratio and traditional pathologic staging identified a subgroup of patients with long term overall survival of 100% [7]. These findings are relevant to multiple tumor types, including melanoma and colorectal carcinoma, and have led to an effort to standardize the histologic analysis of tumor-associated immune infiltrates and incorporate an immune assessment of an “immune score” into the standard histologic evaluation and staging of tumors [21].
The CD8+/FoxP3+ T cell ratio has been examined previously in MBC. Interestingly, our findings are the first to suggest that lower ratios of CD8+/FoxP3+ T cells in the MBC may be associated with improved survival from the time of development of the metastasis. The association between higher numbers of both CD8+ T cells and FoxP3+ Tregs and improved survival has previously been described in primary colorectal carcinoma [22, 23], and more recently in ER− breast cancer [24]. The association of higher numbers of Tregs with improved survival was unexpected, given their widely recognized role in suppressing tumor immunity. However, a recent report suggested that the influence of CD8+ T cells and Tregs on clinical outcome is interdependent, where FoxP3+ Tregs exert a favorable influence on survival only in colon cancers with low numbers of CD8+ TILs [25]. These findings, together with our observation that the influence of CD8+ T cells and FoxP3+ Tregs may be different in primary and metastatic breast tumors, highlight the importance of immunologic context in influencing clinical outcome [26].
The limitations of this study include the potential for sampling error inherent in the TMA methodology, the subjective nature of scoring cellular infiltrates, and the relatively small sample size (15 evaluable patients) including the lack of a sizeable HER-2 cohort to complement the luminal and TNBC subgroups. To minimize these limitations, five to ten large cores (1.4-mm) were taken per tumor to minimize sampling error and account for regional differences in immune infiltrates. Furthermore, independent review of all tumor grades and immunohistochemical staining was performed, and ER, PR, and HER-2 staining was repeated and re-scored on each tumor on the TMAs rather than reported from the initial surgical pathology diagnosis. Three of the primary tumors on the TMA were status-post neoadjuvant therapy, but unfortunately the primary core tissue was unavailable for review. Although we cannot exclude the possibility that the neoadjuvant therapy altered the immune milieu of these primary tumors, the tumors themselves were large with no evidence of treatment effect (e.g., fully viable carcinoma cells with no stromal fibrosis, histiocytic replacement or other evidence of tumor regression).
In summary, we analyzed the presence of infiltrating lymphocytes, including T cells, B cells, and Tregs, within 15 matched primary and metastatic breast tumors, where 8 tumors were luminal, 6 were triple negative, and 1 was HER-2+. We found that metastatic breast tumors overall contained fewer TILs than their matched primary breast cancers. Two patients who presented with Stage 4 disease had high levels of Tregs present in their primary tumors. In addition, we found that metastatic TNBCs contained lower numbers of all types of TILs relative to their matched primary breast tumors, whereas only CD8+ TILs were decreased in metastatic luminal breast cancers relative to their matched primary breast tumors. Compared to metastases to other organs, breast cancer metastases to the brain were characterized by a paucity of infiltrating lymphocytes. Finally, higher numbers of CD8+ T cells relative to FoxP3+ Tregs within primary tumors appeared to predict improved clinical outcome, whereas the opposite ratio was favorable when present in the metastatic tumors. Our findings, together with the published literature, suggest that further evaluation of the immunologic milieu of both primary breast cancers and their associated metastases may inform both prognosis and the likelihood of response to therapy. Larger studies to evaluate these questions are warranted.
Acknowledgments
This work was supported by the Department of Defense (Clinical Translational Research Award W81XWH-07-1-0485), the American Cancer Society (RSG CCE 112685), the Specialized Programs in Research Excellence (SPORE) in Breast Cancer (P50CA88843), Genentech Incorporated, the Gateway Foundation, the Avon Foundation, and the V Foundation. The authors would also like to acknowledge Jessica Hicks for technical assistance with staining of the tissue microarrays.
Footnotes
CONFLICT OF INTEREST
Dr. Emens receives research funding from Genentech, Incorporated, and has received honoraria for participating on regional advisory panels for Genentech, Incorporated, Roche Incorporated, and Bristol Myers Squibb. Under a licensing agreement between Aduro, Incorporated and the Johns Hopkins University, the University is entitled to milestone payments and royalty on sales of the GM-CSF-secreting breast cancer vaccine. The terms of these arrangements are being managed by Johns Hopkins University in accordance with its conflict of interest policies. All other authors report no conflict of interest to disclose.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Ladoire S, Arnould L, Apetoh L, et al. Pathologic complete response to neoadjuvant chemotherapy of breast carcinoma is associated with the disappearance of tumor-infiltrating FoxP3+ regulatory T cells. Clin Cancer Res. 2008;14:2413–2420. doi: 10.1158/1078-0432.CCR-07-4491. [DOI] [PubMed] [Google Scholar]
- 2.Mahmoud SM, Paish EC, Powe DG, et al. Tumor-infiltrating CD8+ T lymphocytes predict clinical outcome in breast cancer. J Clin Oncol. 2011;29:1949–1955. doi: 10.1200/JCO.2010.30.5037. [DOI] [PubMed] [Google Scholar]
- 3.Mahmoud SM, Lee AH, Paish EC, et al. The prognostic significance of B lymphocytes in invasive carcinoma of the breast. Breast Cancer Res Treat. 2012;132:545–553. doi: 10.1007/s10549-011-1620-1. [DOI] [PubMed] [Google Scholar]
- 4.Bates GJ, Fox SB, Han C, et al. Quantification of regulatory T cells enables the identification of high-risk breast cancer patients and those at risk of late relapse. J Clin Oncol. 2006;24:5373–5380. doi: 10.1200/JCO.2006.05.9584. [DOI] [PubMed] [Google Scholar]
- 5.Denkert C, Loibi S, Noske A, et al. Tumor-associated lymphocytes as an independent predictor of response to neoadjuvant chemotherapy in breast cancer. J Clin Oncol. 2010;28:105–113. doi: 10.1200/JCO.2009.23.7370. [DOI] [PubMed] [Google Scholar]
- 6.West NR, Milne K, Truong PT, et al. Tumor-infiltrating lymphocytes predict response to anthracycline-based chemotherapy in estrogen receptor-negative breast cancer. Breast Cancer Res. 2011;13:R126. doi: 10.1186/bcr3072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ladoire S, Mignot G, Dabakuyo S, et al. In situ immune response after neoadjuvant chemotherapy predicts survival. J Pathol. 2011;224:389–400. doi: 10.1002/path.2866. [DOI] [PubMed] [Google Scholar]
- 8.Cimino-Mathews A, Hicks JL, Illei PB, et al. Androgen receptor expression is usually maintained in initial surgically resected breast cancer metastases but is often lost in end-stage metastases found at autopsy. Hum Pathol. 2012;43:1003–1011. doi: 10.1016/j.humpath.2011.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Carey LA, Perou CM, Livasy CA, et al. Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. JAMA. 2006;295:2492–2502. doi: 10.1001/jama.295.21.2492. [DOI] [PubMed] [Google Scholar]
- 10.Nielsen TO, Hsu FD, Jensen K, et al. Immunohistochemical and clinical characterization of the basal-like subtype of invasive breast carcinoma. Clin Cancer Res. 2004;10:5367–5374. doi: 10.1158/1078-0432.CCR-04-0220. [DOI] [PubMed] [Google Scholar]
- 11.Bohling SD, Allison KH. Immunosuppressive regulatory T cells are associated with aggressive breast cancer subtypes: a potential therapeutic target. Mol Pathol. 2008;21:1527–1532. doi: 10.1038/modpathol.2008.160. [DOI] [PubMed] [Google Scholar]
- 12.Kim ST, Jeong H, Woo OH, et al. Tumor-infiltrating lymphocytes, tumor characteristics, and recurrence in patients with early breast cancer. Am J Clin Oncol. 2012 doi: 10.1097/COC.0b013e3182467d90. epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 13.Liu S, Lachapelle J, Leung S, et al. CD8+ T lymphocyte infiltration is an independent favorable prognostic indicator in basal-like breast cancer. Breast Cancer Res. 2012;14:R48. doi: 10.1186/bcr3148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yan M, Jene N, Byrne D, et al. Recruitment of regulatory T cells is correlated with hypoxia-induced CXCR4 expression, and is associated with poor prognosis in basal-like breast cancers. Breast Cancer Res. 2011;13:R47. doi: 10.1186/bcr2869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dong DD, Yie SM, Li K, et al. Importance of HLA-G expression and tumor infiltrating lymphocytes in molecular subtypes of breast cancer. Hum Immunol. 2012;73:998–1004. doi: 10.1016/j.humimm.2012.07.321. [DOI] [PubMed] [Google Scholar]
- 16.Liu F, Lang R, Zhao J, et al. CD8+ cytotoxic T cell and FoxP3+ regulatory T cell infiltration in relation to breast cancer survival and molecular subtypes. Breast Cancer Res Treat. 2011;130:645–655. doi: 10.1007/s10549-011-1647-3. [DOI] [PubMed] [Google Scholar]
- 17.Kuroda H, Tamaru J, Sakamoto G, Ohnisi K, Itoyama S. Immunophenotype of lymphocytic infiltration in medullary carcinoma of the breast. Virchows Arch. 2005;446:10–14. doi: 10.1007/s00428-004-1143-9. [DOI] [PubMed] [Google Scholar]
- 18.Anz D, Eiber S, Scholz C, et al. In breast cancer, a high ratio of tumor-infiltrating intraepithelial CD8+ to FoxP3+ cells is characteristic or the medullary subtype. Histopathology. 2011;59:965–974. doi: 10.1111/j.1365-2559.2011.04040.x. [DOI] [PubMed] [Google Scholar]
- 19.Vong JS, Yu AM, Ng DC, et al. Reduced numbers of regulatory T cells in breast carcinoma with medullary features. Histopathol. 2011;59:345–349. doi: 10.1111/j.1365-2559.2011.03903.x. [DOI] [PubMed] [Google Scholar]
- 20.Berghoff AS, Lassmann H, Preusser M, Hoftberger R. Characterization of the inflammatory response to solid cancer metastases to the brain. Clin Exp Metastasis. 2013;30:69–81. doi: 10.1007/s10585-012-9510-4. [DOI] [PubMed] [Google Scholar]
- 21.Galon J, Pages F, Marincola FM, et al. The immune score as a new possible approach for the classification of cancer. J Transl Med. 2012;10:1–3. doi: 10.1186/1479-5876-10-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Salama P, Phillips M, Grieu F, et al. Tumor-infiltrating FOXP3+ T regulatory cells show strong prognostic significance in colorectal cancer. J Clin Oncol. 2009;27:186–92. doi: 10.1200/JCO.2008.18.7229. [DOI] [PubMed] [Google Scholar]
- 23.Ladoire S, Martin F, Ghiringhelli F. Prognostic role of FOXP3+ regulatory T cells infiltrating human carcinomas: the paradox of colorectal cancer. Cancer Immunol Immunother. 2011;60:909–18. doi: 10.1007/s00262-011-1046-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.West NR, Kost SE, Martin SD, et al. Tumour-infiltrating FOXP3(+) lymphocytes are associated with cytotoxic immune responses and good clinical outcome in oestrogen receptor-negative breast cancer. Br J Cancer. 2013;108:155–62. doi: 10.1038/bjc.2012.524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yoon HH, Orrock JM, Foster NR, et al. Prognostic impact of FoxP3+ regulatory T cells in relation to CD8+ T lymphocyte density in human colon carcinomas. PLoS One. 2012;7:e42274. doi: 10.1371/journal.pone.0042274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.deLeeuw RJ, Kost SE, Kakal JA, Nelson BH. The prognostic value of FoxP3+ tumor-infiltrating lymphocytes in cancer: a critical review of the literature. Clin Cancer Res. 2012;18:3022–29. doi: 10.1158/1078-0432.CCR-11-3216. [DOI] [PubMed] [Google Scholar]