Abstract
Transgenic expression of just four defined transcription factors (c-Myc, Klf4, Oct4, and Sox2) is sufficient to reprogram somatic cells to a pluripotent state1-4. The resulting induced pluripotent stem (iPS) cells resemble ES cells in their properties and potential to differentiate into a spectrum of adult cells types. Current reprogramming strategies involve retroviral1, lentiviral5, adenoviral6 and plasmid7 transfection to deliver reprogramming factor transgenes. Although the latter two methods are transient and minimize the potential for insertion mutagenesis, they are currently limited by diminished reprogramming efficiencies. piggyBac (PB) transposition is host-factor independent, and has recently been demonstrated to be functional in various human and mouse cell lines8-11. The PB transposon/transposase system requires only the inverted terminal repeats flanking a transgene and transient expression of the transposase enzyme to catalyze insertion or excision events12. Here we demonstrate successful and efficient reprogramming of murine and human embryonic fibroblasts using doxycycline inducible transcription factors delivered by PB transposition13. Stable iPS cells thus generated express hallmark pluripotency markers and succeed in a series of rigorous differentiation assays. By taking advantage of the natural propensity of the PB system for seamless excision12, we show that the individual P insertions can be removed from established iPS cell lines, providing an invaluable tool for discovery. In addtion, we have demonstrated the traceless removal of reprogramming factors joined with 2A sequences14 delivered by a single transposon from murine iPS lines. We anticipate that the unique properties of this virus-independent simplification of iPS cell production will accelerate the movement of this field further towards full exploration of the reprogramming process and future cell-based therapies.
Keywords: iPS, induced pluripotent stem cell, embryonic stem cell, fibroblast, mice/mouse, human, differentiation, reprogramming, transposon, transposition, piggyBac, non-viral
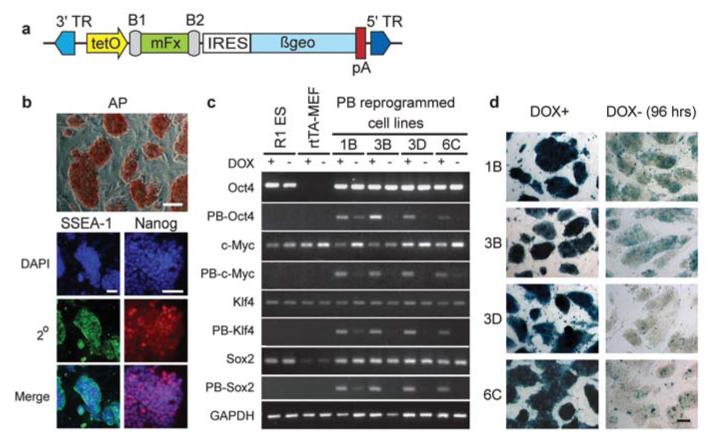
To explore the utility of PB as a vector for somatic cell reprogramming, it was necessary to achieve temporal expression control and transgene silencing, as PB transposons are not purposefully subjected to the same natural silencing process that diminishes retroviral (and less extensively lentiviral) expression15. The four mouse factors (mFx: c-Myc, Klf4, Oct4, and Sox2)1 were transferred into the PB-TET transposon plasmid (Fig. 1a) under the transcriptional control of the tetO2 tetracycline/doxycycline (dox) inducible promoter16. All were linked to βgeo through and IRES sequence to allow monitoring the tightness of dox regulation and later demonstration of the reprogrammed cells’ capacity for exogenous factor independent maintenance. The reverse tetracycline transactivator (rtTA) protein was provided by parental mouse embryonic fibroblasts (rtTA-MEFs) established from 15.5dpc ROSA26 rtTA-IRES-GFP knock-in17 transgenic embryos. rtTA-MEFs were transfected with circular PB-TET-mFx plasmid DNA ‘cocktails’ in conjunction with a PB transposase expression plasmid11. From the time of transfection onwards, fibroblasts were maintained in dox-containing ES cell culture conditions without passage. Mouse fibroblasts underwent ES cell-like colony formation, which resulted in the derivation of self-renewing cell lines displaying key characteristics of reprogramming (Supplementary Fig. S1).
Figure 1.
Cell lines generated by PB-mediated factor transposition are reprogrammed. (a) The PB-TET transposon vector used to deliver inducible (tet-O), reporter-linked (IRES-ßgeo-pA) mouse factors (mFx). 3′/5′TR, PB terminal repeats; B1/B2, post-Gateway cloning sites. (b) Stable dox-independent cell lines activate alkaline phosphatase (AP), SSEA1 and Nanog. Representative images are from a single cell line (1B). (c) Dox regulation of PB-delivered factors as monitored by transgene-specific RT-PCR analysis (vector-based reverse primer). Reprogrammed cell lines and controls were grown in the presence (+) or absence (−) of dox for 2 passages (~96 hrs). The induction (Oct4, Sox2) or maintenance (c-Myc, Klf4) of endogenous gene expression was determined by RT-PCR using 3′UTR-directed reverse primers. (d) Residual transgene expression in reprogrammed cell lines visualized as lacZ activity from the transcription-linked βgeo reporter gene (Fig. 1a). The level and mosaicism of lacZ after dox withdrawal correlates roughly with basal level transcription detected by RT-PCR. Scale bars are 200 μm.
Colonies produced from rtTA-MEFs with PB-TET-mFx cocktails were scored on the basis of morphology, with early foci formation noted on d6-8 and colonies forming around d8-10. These were mostly ES cell-like (Supplementary Fig. S1a), while a handful displayed diverse morphologies (data not shown). Adjusting the dox concentration above or below the standard of 1.5μg/mL resulted in subtle declines (150ng/mL) or drastic decreases (15ng/mL or 15μg/mL) in colony numbers (Supplementary Fig. S1b), supporting the notion that factor expression level affects reprogramming rates. In the absence of dox, no reprogramming was noted, nor was any cells found positive for AP staining (data not shown).
Forty-eight PB-TET-mFx induced colonies were picked from fibroblast induction fields and passaged on inactivated fibroblast feeder layers. Surviving clones were maintained in dox during establishment, until found to be dox independent in replicate wells. Dox independence occurred as early as d12 for 3 lines (4D, 4E, 6C), and was achieved by most clones by d24 (7/39=18% on d15, 21/39=54% on d19, 31/39=80% on d21, 33/39=86% on d23). During establishment, cultures which had not yet achieved autonomous maintenance of reprogramming rapidly flattened and returned to a fibroblast-like state upon dox withdrawal (Supplementary Fig. S2a, b) as observed in a prior report5. Dox-independence was sustained for at least an additional 18 passages, with no apparent disturbance of growth characteristics compared to replica cultures maintained in 1.5μg/mL dox. Doubling time and morphology of PB-TET clones were indistinguishable from that of R1mouse ES cells18 (data not shown).
It has been shown that stable reprogramming by exogenous factor expression results in the sequential activation of ES cell markers3,5,19. On d16 (p2-3) the seven dox-independent lines passed the alkaline phosphatase (AP) staining criterion (Fig. 1b). These clones tested positive on d20-22 for the cell-surface marker SSEA1 and nuclear-localized Nanog protein (Fig. 1b). Semi-quantitative reverse transcription polymerase chain reaction (RT-PCR) revealed the expression of ES cell pluripotency markers20-22, such as Dax1, ERas, Fbxo, FoxD3, Nanog, Rex1 and Zfp296 (Supplementary Fig. S2c). Using 3′UTR-specific reverse primers (Supplementary Table S1), the endogenous counterparts of the four mFx transgenes were also shown to be active (Fig. 1c).
PB-TET-reprogrammed clones were screened for lacZ expression to determine general transgene activity in the presence and absence of dox (Fig. 1d). RT-PCR, which allows further distinction of individual transgene activity, revealed variable trace expression levels of some transgenes in the uninduced (dox -) state (Fig. 1c). Oct4 transgene expression could be detected in 1B and to a lesser extent in 3B, and c-Myc was maintained minimally in both 1B and 6C. For clones 1B and 3D Sox2 expression could be detected, while Klf4 appeared silent in all lines except for 1B. Note that RT-PCR detection of mFx transcripts reflects average expression levels for each cell population, and does not expose mosaic expression patterns (as revealed in Fig. 1d).
Genomic Southern analysis was used to determine transgene copy number in selected dox-independent mouse PB-TET lines (Supplementary Fig. S3a). The average estimated PB transposon copy number was 9, although co-migrating bands which may obscure the exact total could not be ruled out. Line 3D contains only five transposon insertions. As all four PB-TET-mFx transgenes are represented in 3D (Fig. 1c), three of the four factors are sufficient in single copy for reprogramming.
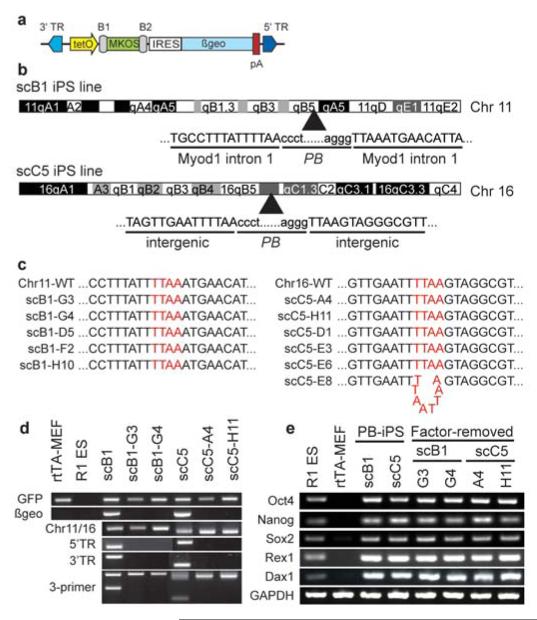
Through additional transient transposase expression, we exploited the “cut” step of transposition to remove the transgenes following complete reprogramming. For clone 1B, a broad range was observed in the mobilization rate for each transposon (Supplementary Fig. S3b, S4); transposon #8 was excised only once (1/38=2.5%) while transposon #9 was excised 11 times (11/38=29%). Such high variability suggests that the excision frequency is integration site-dependent. To simplify complete transgene removal we inserted the MKOS sequence (c-Myc, Klf4, Oct4 and Sox2 ORFs linked with 2A peptide sequences14) into PB-TET (Fig. 2a). Similar to four-factor reprogramming, induction of rtTA-MEFs with PB-TET-MKOS in the presence of dox yielded colonies after 10-14 days. Out of forty-eight iPS cell-like colonies, characterized by pluripotency marker expression and diagnostic Southern analysis (not shown), two single-copy cell lines, designated as scB1 and scC5, were established. Both lines maintained tight regulation of the MKOS reprogramming factors through indirect measurement of the lacZ reporter (Supplementary Fig. S5). Splinkerette PCR11 determined that PB inserted into the first intron of the Myo1d gene (Chr11) in scB1, and into an intragenic region of Chr16 in scC5 (Fig. 2b). The insertion events were confirmed by locus-specific genomic PCR (Fig. 2d). For both scB1 and scC5, transient PB transposase expression led to the removal of the linked reprogramming factors in >2% of the exposed cells as estimated by the ratio of lacZ negative sublines in the presence of dox (Supplementary Fig. S5). Sequence analysis across the original insertion sites revealed that in 10/11 subclones the original transposon integration site was reverted to wild type (Fig. 2c). Complete loss of the PB-TET-MKOS transposon was further confirmed by locus- and transgene-specific genomic PCR revealing subclones which were negative for both genome-transposon junction and internal transposon-specific PCRs (Fig. 2d). Even after the exogenous factors were removed, the endogenous pluripotency genes remained actively expressed (Fig. 2e).
Figure 2.
Seamless factor removal from iPS cells using transposase-stimulated PB excision. (a) Schematic of the MKOS-containing PB-TET transposon. (b) Genomic integration site of the individual transposons in scB1 and scC5 lines. Capital letters represent flanking genomic sequences while lower case letters are transposon TR sequences. (c) Sequence analyses revealed that no mutation was left behind following transposon-mediated removal in the majority of sublines (10 of 11). One single C5 subclone harbored a TTAA duplication at the excision site. (d) Molecular demonstration of transposon removal in representative subclones. From top to bottom: GFP PCR ensures all cell lines are derivatives of rtTA MEFs (R1 ES negative control); βgeo PCR detects the presence of transposons regardless of genomic location; Chr11- and Chr16-specific PCR across the TTAA tetranucleotide insertion site. scB1 and scC5 are hemizygous for the transposon and amplify the wild type allele; 5′ and 3′ junction PCR; 3-primer PCR for the wild type allele and the transposon-genome junction. (e) RT-PCR analysis of the single transposon induced iPS cell lines and their factor-removed derivatives reveals maintenance of hallmark pluripotency gene expression. R1 ES cells and parental rtTA-MEFs serve as positive and negative controls, respectively.
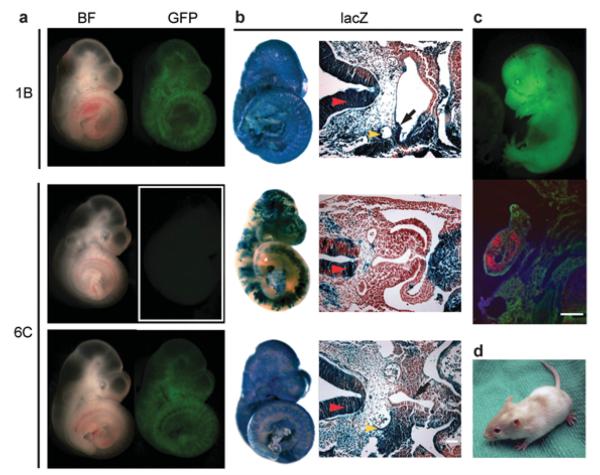
Pluripotency of PB-reprogrammed cell lines (1B, 3D, 6C, scB1 and scC5) was demonstrated by their contribution to chimaera development (Fig. 3a and Supplementary Table S2) shown). Embryos were dissected between 10.5dpc and 15.5dpc, and scored for GFP fluorescence indicating contribution of reprogrammed ROSA26-rtTA-IRES-GFP parental MEFs. To reveal chimaeric contribution at the cellular level in tissues representative of the three germ layers we treated recipient dams with dox 20h prior to dissection, and then lacZ stained and sectioned embryos (Fig. 3b, Supplementary Table S2). Interestingly, for cell line 1B, many embryos demonstrated high-level chimaeric contribution despite low-level residual factor expression (Fig. 1c, d), suggesting a threshold that permits differentiation of these cells in the embryonic environment. Pluripotency was stringently confirmed by the production of completely iPS cell-derived 13.5dpc embryos (Fig. 3c) via tetraploid embryo complementation assay18, including germ cell formation detected by Vasa expression23 in the genital ridge (Fig. 3c). Germ cell formation from iPS cells was also detected in genital ridges of standard diploid chimaeric embryos (Supplementary Fig. S6a). Teratomas containing derivatives from all three embryonic germ layers are additional proof that these cells are pluripotent and able to differentiate to complex tissues (Supplementary Fig. S6b). The chimaeric contribution of iPS cells to postnatal animals (Fig. 3d) is a clear testament to PB transposon-assisted complete reprogramming of fibroblasts to iPS cells (PB-iPS) with the capability of building functional adult tissues.
Figure 3.
Cell lines reprogrammed by PB-mediated factor transposition are pluripotent. (a) Contribution of PB-TET cell lines to embryonic development. Chimaeras dissected at 10.5dpc with the strongest contribution of PB-iPS-derived cells were easily detected as GFP positive (BF - bright field). (b) Whole mount lacZ staining of chimaeric embryos and their sections show contribution of PB-iPS lacZ positive cells to derivatives of all three embryonic germ layers. Red arrowhead – neural tube (ectoderm); yellow arrowhead – dorsal aorta (mesoderm); black arrow foregut epithelium (endoderm). (c) Completely iPS cell-derived (1B) 13.5 dpc embryo generated by tetraploid embryo complementation. Immunohistochemistry on sections through its genital ridge shows the iPS cell contribution to germ cells (Vasa+, red). (d) Adult chimaera obtained by aggregating 1B PB-iPS cells with diploid eight cell stage ICR (albino) embryos. Scale bars are 100 μm.
We successfully employed a PB-CAG-rtTA transposon together with PB-TET-mFx for dox-regulated reprogramming of normal human embryonic fibroblasts. Colonies induced via transposition were picked between d14 and d28. Four out of five AP positive clones became dox independent one week after isolation. Factor expression in the uninduced state was not detectable by lacZ staining (Supplementary Fig. S7a) and only trace amounts of transcription were detected in colonies by semi-quantitative RT-PCR (Supplementary Fig. S7b). Expression of the endogenous forms of the inducer genes, however, reached levels characteristic of human ES cells (Supplementary Fig. S7b) along with hallmark pluripotency marker genes (Supplementary Fig. S7c) including nuclear-localized NANOG (Supplementary Fig. S8). Spontaneous differentiation following bFGF withdrawal from the medium gave rise to cystic embryoid bodies and to various differentiated cell types, including AFP (endoderm), SMA and vWF (mesoderm) or ßIII TUBULIN and GFAP (ectoderm) positive cells (Supplementary Fig. S9). In accordance to an earlier report24, our observations indicate that mouse factors effectively reprogram human cells, underlining the functional conservation of these transcription factors between the two species.
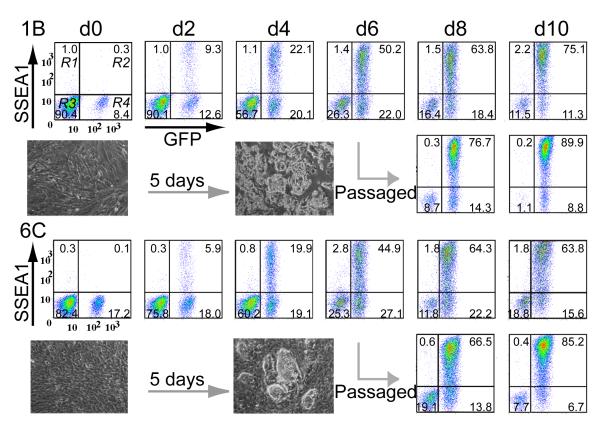
Diploid chimaeras from three mouse iPS cell lines (1B, 6C and 3D) were used to derive secondary MEFs, tracking the iPS cell-derived component (2°F/1B, 2°F/6C and 2°F/3D) by GFP expression (Fig. 3a and Fig. 4). Dox treatment had a dramatic effect on 2°F/1B and 2°F/6C, which initiated early signs of reprogramming within two days, including cellular aggregation (Supplementary Movie). Proliferation also increased dramatically, (Fig. 4 and Supplementary Fig. S10a) as initial contribution (10-18% on d0) reached 90% by d10. These physical changes were reflected at the molecular level by the activation of AP (not shown) and SSEA1. The latter showed very rapid activation; detected as early as d2, expressed by nearly 50% of 2°F/1B and 2°F/6C cells on d4 and greater than 80% on d8 (Fig. 4, Supplementary Fig. S10b). Nanog activation occurred four days earlier (by d9) in 2°F/1B than in 2°F/6C (Supplementary Fig. S11), highlighting clonal variation and flexibility in the reprogramming process. Overall, PB-iPS cell derived fibroblasts displayed a much more rapid initial response to reprogramming factor induction than that previously reported for the dox inducible lentivirus system25, which may reflect a high level of instability for lentivirus-inserted transgene expression. Single cell sorting of 2°F/6C cells into 96-well plates revealed that 56 of 192 single cells (29%) were capable of forming colonies. Thirty-nine out of forty-four (89%) established single-cell clones evaluated for Nanog expression were found positive on d13 of induction. In contrast, line 2°F/3D was inefficient at 2° iPS cell production, as few dox-induced colonies formed and expression of AP and SSEA1 was delayed (not shown). The PB-TET-Oct4 transgene was noticeably less active in 2°F/3D as compared to 2°F /1B and 2°F /6C (Supplementary Fig. S10c), signifying the need for a sufficient level of Oct4 expression even in secondary iPS cell induction.
Figure 4.
Properties of secondary fibroblast (2°F) reprogramming. FACS analysis establishing the dynamics of SSEA1 activation in 2°F/1B and 2°F/6C cell lines. The inset shows colony formation as early as d5. On d6 of reprogramming gene induction, the cultures were also passed by standard trypsinization and analyzed in parallel to eliminate the negative effect of cell overgrowth.
Successful transposon-based reprogramming of fibroblasts to iPS cells represents significant advance in current methods of transgene delivery. First, PB transposition permits technical simplification and improved accessibility of reprogramming methodology by making use of effortless plasmid DNA preparation and commercial transfection products for delivery. This eliminates the need for specialized biohazard containment facilities or the production of high-titer, limited-lifetime viral stocks26. Second, the range of somatic cell types that could be used for reprogramming are not limited by a decreased susceptibility to viral infection26. Third, PB-mediated delivery will allow the option of xeno-free production of human iPS cells contrary to current viral production protocols that use xenobiotic conditions. Finally, accurate transgene removal through transposase expression has been demonstrated in various cell lines8,11,12,27-29. We have harnessed this potential and show here that the reprogramming factors can be removed without a trace from iPS cells once exogenous expression becomes dispensable. These four key characteristics of PB transgenesis mark important advances towards achieving clinically acceptable methods of deriving reprogrammed cells. Complementary, secondary fibroblasts or alternate secondary cell types with a combinatorial removal of proven reprogramming transgene insertions may provide an enormously powerful tool for high throughput screening to further explore the mechanisms which play roles in the reprogramming process.
METHODS
Plasmid vector construction
PB-TET was constructed by exchanging the 5′/3′TR-flanked PGK-neo cassette of PB-PGK-neo-bpA11 with a blunt NotI/HindIII fragment containing tetO2 (provided by S. Mohammadi) via digestion with NheI and HindIII (both polished), resulting in PB-tetO2. The NheI and SalI (polished) IRES-βgeo-bpA element from pIFS (provided by J. Dixon) was ligated into PB-tetO2 prepared by NheI and SmaI digestion to yield PB-tetO2-IRES-βgeo. Finally, a Gateway RfA cassette (Invitrogen) maintained in the polished NotI site of pBluescriptKS+ (Stratagene), was inserted by digestion with SacII and SpeI into PB-tetO2-IRES-βgeo digested with SacII and NheI. For PB-CAG, the PGK-neo cassette was removed by digestion with NheI (polished) and HindIII and replaced with a CBA-RfA-rβgpA cassette released by OliI and HindIII digestion from a constitutive gene expression construct (I.P.M., K.W. and A.N., unpublished data). The four reprogramming factors were Gateway cloned from retroviral backbones (Addgene) into the PB-TET transposon vector using pDONR221 (Invitrogen) as an intermediate. The Tet transactivator was amplified from pTet-On Advanced (Clontech) using attB1/2 primers (Supplementary Table S2) and similarly Gateway cloned into PB-CAG. The MKOS element from pCAG2LMKOSimO14 was cloned into pENTR2B using EcoRI prior to Gateway shuttling. The Tet transactivator was amplified from pTet-On Advanced (Clontech) using attB1/2 primers (Supplementary Table S1).
Fibroblast isolation
15.5dpc ROSA26-rtTA-IRES-GFP embryos (from Gt(ROSA)26Sortm1.1(rtTA,EGFP)Nagy) were decapitated, eviscerated, dissociated with 0.25% trypsin, 0.1% EDTA, and plated in DMEM, 10% FBS, penicillin-streptomycin, glutamax. HEFs were isolated from 12 week-old abortuses and maintenaned in DMEM, 15% human serum, 10ng/mL bFGF, penicillin-streptomycin, glutamax, β-mercaptoethanol, NEAA.
PB transfection and cell culture
MEFs were seeded in DMEM, 15% FBS, penicillin-streptomycin, glutamax, β-mercaptoethanol, sodium-pyruvate, non-essential amino acids, LIF on gelatinized (0.1%) 6-well dishes at a density of 1.25×105cells/10cm2. After 24hrs culture, FugeneHD (Roche) was used to transfect cells with 10ng, 100ng, or 400ng of each mFx transposon (25ng, 50ng, or 100ng for PB-TET-MKOS) plus 100ng of pCyL43 PB transposase plasmid11 (normalized to 2μg total DNA with empty pBluescriptKS+) at a Fugene:DNA ratio of 8uL:2μg. After 24 hours, the media was supplemented with dox (d0), and changed entirely 48hrs post-transfection. Cells were fed daily with dox containing media (1.5μg/mL, unless otherwise indicated). Colonies were picked in 96-well format over d10-14 and cultivated on mitomycin-c arrested MEFs. For PB-TET induced clones, dox treatment was maintained until d16-24. iPS cells for DNA or RNA preparation were grown on gelatin. Established iPS cells were passaged 1:6 every 48 hours. Transfection of HEFs was performed similarly, except fibroblasts were initially seeded in DMEM supplemented with 15% human serum, 10ng/mL bFGF, penicillin-streptomycin, glutamax, non-essential amino acids at a density of 6.25×104cells/10cm2, and grown in HEScGRO (Millipore) 48 hours after transfection. Doxycycline (1.5μg/ml) was added 24h post transfection and withdrawn a week after picking. Colonies were initially passaged mechanically 1:2, and later with TripLE Select (Invitrogen) 1:4 every 7 days. Human iPS cells were maintained on inactivated MEFs in KO-DMEM, 20% serum replacement, 10ng/mL bFGF, penicillin-streptomycin, glutamax, non-essential amino acids.
Southern blotting
Ten micrograms of genomic DNA from R1 ES cells, PB-iPS lines or rtTA-MEFs was digested with BamHI overnight, resolved by gel electrophoresis, and transferred to Hybond N+ (GE Healthcare). A neo probe PCR fragment prepared with DIG High Prime DNA Labeling and Detection Kit II (Roche) was used to detect transposon insertions (~25ng probe/mL hybridization solution).
Splinkerette, Genomic, and RT-PCR
Splinkerette PCR to determine PB genomic integration sites was performed as described11. TA-cloned PCR products were sequenced bidirectionally with M13 forward and reverse primers. PB insertion loci were determined using BLAST. Genomic PCR on factor-removed PB-iPS lines was performed using primers described in Supplementary Table S1. Approximately 100ng of genomic template DNA was amplified using Qiagen Taq (Qiagen) with the inclusion of Q-Solution. Highly repetitive sequences on chromosome 16 required nested PCR. Three-primer PCR amplification used PB-3F in conjunction with the chromosome-specific primer set. Standard PCR conditions were: 95°C for 30 sec, 55°C for 30sec, 72°C for 45sec; x35 cycles. RNA was collected using RNeasy Mini Kit (Qiagen), quantified and treated with gDNA WipeOut and cDNA prepared with QuantiTect Reverse Transcription Kit (Qiagen). For each RT-PCR reaction, we used 50-100 ng of cDNA and primers listed in Supplementary Table S1. Standard PCR conditions were: 94°C for 30 sec, 58-62°C for 30sec, 72°C for 15-30sec; x30-35 cycles.
PB transgene removal
Stable PB-TET clones were transiently transfected with pCyL43 transposase (2ug DNA:8uL FugeneHD). After Puromycin (1ug/mL) treatment for 3 days to eliminate non-transfected cells, the cells were plated at clonal density (~500-1000cells/60cm2) on inactivated MEFs. Clones were picked after 5 days further culture.
lacZ/AP staining
cells and embryos were fixed with 0.25% glutaraldehyde, rinsed in wash buffer (2mM MgCl2, 0.01% Sodium deoxycholate, and 0.02% Nonidet-P40 in PBS) and stained overnight (~16hrs) in lacZ staining solution: 20mM MgCl2, 5mM K3Fe(CN)6, 5mM K4Fe(CN)6 and 1mg/mL X-gal in PBS. Embryos were embedded in paraffin, sectioned and counterstained with neutral red. Staining for alkaline phosphatase activity was performed on cells without fixation using the Vector Red Kit (Vector Labs).
Immunostaining and flow cytometry
Cells were grown on inactivated MEFs on gelatin-coated Lab-Tek borosilicate glass slides (Nunc) or 4-well dishes containing gelatinized or Matrigel-treated glass coverslip inserts. Samples were washed with PBS, fixed in 4% PFA/PBS for 10min at 25°C, permeabilized with 0.3% Triton X-100 in PBS for 10 min at 25°C, blocked in 5% goat serum for 1hr, and primary Ab was added overnight at 4°C (mNanog, AB5731, Chemicon, 1:1000; hNanog, 0002P-F, ReproCell, 1:200; SSEA1, SSEA4, Tra1-60, Tra1-81, 1:5, provided by P. Andrews; Muscle Actin, M0635, DakoCytomation, 1:100; βIII-Tubulin, TU-20, Millipore, 1:100; HNF-3β, sc-9187, Santa Cruz, 1:100; GFAP, Z0334, DakoCytomation, 1:200; AFP, MAB1369, R&D Systems, 1:200; vWF, A 0082, DakoCytomation, 1:200). Samples were washed in PBS and secondary Ab (cy3 IgG, 1:1000; Alexa488 IgG or IgM, 1:400; Alexa594 IgG, 1:200) was added for 1hr at 25°C. After washing, samples were mounted in Vectashield with DAPI (Vector Labs). Genital ridges were pre-fixed with 4% paraformaldehyde in PBS for 1 hour at 25°C, embedded and cryosectioned at 30μm thickness, washed, blocked, and incubated overnight at 4°C with rabbit anti-primordial germ cell marker (DDX4/MVH, ab13840, abcam, 1:400). Sections were washed in PBS and secondary Ab (cy3 IgG, 1:500) was added for 2hrs at 25°C. Immunofluorescence was visualized and acquired using a Zeiss LSM 510 confocal microscope. Dox-induced fibroblast pools from PB-iPS chimaeric mice were diluted to ~15% GFP positive 2°F/1B and 2°F/6C representation with wildtype ICR MEFs prior to seeding. Cells were washed once in PBS containing 5% FBS, incubated with anti-SSEA1 antibody (1:200) for 30min on ice, washed twice in PBS:5% FBS, incubated with Alexa647 conjugated secondary Ab for 30min on ice, washed twice and resuspended in PBS:5% FBS for analysis on a FACS-Calibur. Single GFP positive cell plating from PB-iPS chimeric MEF pools was done with a FACS-Aria.
In vitro differentiation assays
Human PB-iPS lines were dissociated and used to generate embryoid bodies (EBs) by aggregation in AggreWell 400 plates (StemCell Technologies) in 15% FBS DMEM with all additives except LIF and bFGF, in the absence of dox. After 14 days growth, EBs were plated on Matrigel coated cover slips or 4 chamber slides. Immunohistochemistry was performed after an additional 10 days culture.
Teratoma formation
Cell lines were suspended in DMEM containing 10% FBS, and 100uL (1×106cells) injected subcutaneously into both dorsal flanks of nude mice (CByJ.Cg-Foxn1nu/J) anesthetized with isoflurane. Six weeks after injection, teratomas were dissected, fixed overnight in 10% buffered formalin phosphate and embedded in paraffin. Sections were stained with hematoxylin and eosin.
Generation of chimaeras
Dox-independent PB-TET iPS clones were collected as cell clumps of ~8-15 cells from gelatinized dishes by gentle trypsinization. For diploid chimeras, 2.5dpc Hsd:ICR(CD-1) or C57BL/6 embryos were aggregated with PB-iPS cell clumps, and cultured overnight at 37°C in 5% CO2 in KSOM medium (Millipore). For tetraploid embryo complementation, two-cell stage Hsd:ICR(CD-1) embryos (1.5dpc) were electrofused with a CF-150B Pulse Generator (BLS) and cultured overnight, and aggregated as described above. All embryos were transferred into pseudopregnant recipient ICR females 24hrs later30. Dissected embryos were examined for GFP using Leica MZ16 FA stereomicroscope, or left to term to verify coat color contribution. For lacZ detection, pregnant dams were treated with dox (1.5μg/mL dox; 5% sucrose in water) 20hrs prior to dissection.
Acknowledgements
We thank Jason Moffat for time lapse image acquisition, Pier-Andre Pentilla for cell sorting, Marthe-Sandrine Eiymo for assisting with initial PB test vector construction, Janice Ure and Malgosia Kownacka for technical assistance, Mark Kibschull for establishing human embryonic fibroblasts, Aaron Cheung for discussions, and Kristina Vintersten for critical reading of the manuscript. This work was supported by the Wellcome Trust to P.L., and grants awarded to A.N. from the Canadian Stem Cell Network and JDRF.
Footnotes
Full Methods and any associated references are available in the online version of the paper at www.nature.com/nature.
Supplementary Information is linked to the online version of the paper at www.nature.com/nature.
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions.
The authors declare no conflict of interest
References
- 1.Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126(4):663–676. doi: 10.1016/j.cell.2006.07.024. [DOI] [PubMed] [Google Scholar]
- 2.Maherali N, et al. Directly reprogrammed fibroblasts show global epigenetic remodeling and widespread tissue contribution. Cell Stem Cell. 2007;1(1):55–70. doi: 10.1016/j.stem.2007.05.014. [DOI] [PubMed] [Google Scholar]
- 3.Okita K, Ichisaka T, Yamanaka S. Generation of germline-competent induced pluripotent stem cells. Nature. 2007;448(7151):313–317. doi: 10.1038/nature05934. [DOI] [PubMed] [Google Scholar]
- 4.Meissner A, Wernig M, Jaenisch R. Direct reprogramming of genetically unmodified fibroblasts into pluripotent stem cells. Nat Biotechnol. 2007;25(10):1177–1181. doi: 10.1038/nbt1335. [DOI] [PubMed] [Google Scholar]
- 5.Brambrink T, et al. Sequential expression of pluripotency markers during direct reprogramming of mouse somatic cells. Cell Stem Cell. 2008;2(2):151–159. doi: 10.1016/j.stem.2008.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Stadtfeld M, Nagaya M, Utikal J, Weir G, Hochedlinger K. Induced pluripotent stem cells generated without viral integration. Science. 2008;322(5903):945–949. doi: 10.1126/science.1162494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Okita K, Nakagawa M, Hyenjong H, Ichisaka T, Yamanaka S. Generation of mouse induced pluripotent stem cells without viral vectors. Science. 2008;322(5903):949–953. doi: 10.1126/science.1164270. [DOI] [PubMed] [Google Scholar]
- 8.Ding S, et al. Efficient transposition of the piggyBac (PB) transposon in mammalian cells and mice. Cell. 2005;122(3):473–483. doi: 10.1016/j.cell.2005.07.013. [DOI] [PubMed] [Google Scholar]
- 9.Wu SC, et al. piggyBac is a flexible and highly active transposon as compared to sleeping beauty, Tol2, and Mos1 in mammalian cells. Proc Natl Acad Sci U S A. 2006;103(41):15008–15013. doi: 10.1073/pnas.0606979103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cadinanos J, Bradley A. Generation of an inducible and optimized piggyBac transposon system. Nucleic Acids Res. 2007;35(12):e87. doi: 10.1093/nar/gkm446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wang W, et al. Chromosomal transposition of PiggyBac in mouse embryonic stem cells. Proc Natl Acad Sci U S A. 2008;105(27):9290–9295. doi: 10.1073/pnas.0801017105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fraser MJ, Ciszczon T, Elick T, Bauser C. Precise excision of TTAA-specific lepidopteran transposons piggyBac (IFP2) and tagalong (TFP3) from the baculovirus genome in cell lines from two species of Lepidoptera. Insect Mol Biol. 1996;5(2):141–151. doi: 10.1111/j.1365-2583.1996.tb00048.x. [DOI] [PubMed] [Google Scholar]
- 13.Cary LC, et al. Transposon mutagenesis of baculoviruses: analysis of Trichoplusia ni transposon IFP2 insertions within the FP-locus of nuclear polyhedrosis viruses. Virology. 1989;172(1):156–169. doi: 10.1016/0042-6822(89)90117-7. [DOI] [PubMed] [Google Scholar]
- 14.Kaji K, et al. Virus-free induction of pluripotency enabling subsequent excision of the reprogramming factors. Nature. 2009 doi: 10.1038/nature07864. (accompanying) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ellis J. Silencing and variegation of gammaretrovirus and lentivirus vectors. Hum Gene Ther. 2005;16(11):1241–1246. doi: 10.1089/hum.2005.16.1241. [DOI] [PubMed] [Google Scholar]
- 16.Agha-Mohammadi S, et al. Second-generation tetracycline-regulatable promoter: repositioned tet operator elements optimize transactivator synergy while shorter minimal promoter offers tight basal leakiness. J Gene Med. 2004;6(7):817–828. doi: 10.1002/jgm.566. [DOI] [PubMed] [Google Scholar]
- 17.Belteki G, et al. Conditional and inducible transgene expression in mice through the combinatorial use of Cre-mediated recombination and tetracycline induction. Nucleic Acids Res. 2005;33(5):e51. doi: 10.1093/nar/gni051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Nagy A, Rossant J, Nagy R, Abramow-Newerly W, Roder JC. Derivation of completely cell culture-derived mice from early-passage embryonic stem cells. Proc Natl Acad Sci U S A. 1993;90(18):8424–8428. doi: 10.1073/pnas.90.18.8424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Stadtfeld M, Maherali N, Breault DT, Hochedlinger K. Defining molecular cornerstones during fibroblast to iPS cell reprogramming in mouse. Cell Stem Cell. 2008;2(3):230–240. doi: 10.1016/j.stem.2008.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kaji K, et al. The NuRD component Mbd3 is required for pluripotency of embryonic stem cells. Nat Cell Biol. 2006;8(3):285–292. doi: 10.1038/ncb1372. [DOI] [PubMed] [Google Scholar]
- 21.Kim JB, et al. Pluripotent stem cells induced from adult neural stem cells by reprogramming with two factors. Nature. 2008;454(7204):646–650. doi: 10.1038/nature07061. [DOI] [PubMed] [Google Scholar]
- 22.Mikkelsen TS, et al. Dissecting direct reprogramming through integrative genomic analysis. Nature. 2008;454(7200):49–55. doi: 10.1038/nature07056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Fujiwara Y, et al. Isolation of a DEAD-family protein gene that encodes a murine homolog of Drosophila vasa and its specific expression in germ cell lineage. Proc Natl Acad Sci U S A. 1994;91(25):12258–12262. doi: 10.1073/pnas.91.25.12258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Huangfu D, et al. Induction of pluripotent stem cells from primary human fibroblasts with only Oct4 and Sox2. Nat Biotechnol. 2008;26(11):1269–1275. doi: 10.1038/nbt.1502. [DOI] [PubMed] [Google Scholar]
- 25.Wernig M, et al. A drug-inducible transgenic system for direct reprogramming of multiple somatic cell types. Nat Biotechnol. 2008;26(8):916–924. doi: 10.1038/nbt1483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Takahashi K, Okita K, Nakagawa M, Yamanaka S. Induction of pluripotent stem cells from fibroblast cultures. Nat Protoc. 2007;2(12):3081–3089. doi: 10.1038/nprot.2007.418. [DOI] [PubMed] [Google Scholar]
- 27.Dafa’alla TH, et al. Transposon-free insertions for insect genetic engineering. Nat Biotechnol. 2006;24(7):820–821. doi: 10.1038/nbt1221. [DOI] [PubMed] [Google Scholar]
- 28.Wilson MH, Coates CJ, George AL., Jr. PiggyBac transposon-mediated gene transfer in human cells. Mol Ther. 2007;15(1):139–145. doi: 10.1038/sj.mt.6300028. [DOI] [PubMed] [Google Scholar]
- 29.Mitra R, Fain-Thornton J, Craig NL. piggyBac can bypass DNA synthesis during cut and paste transposition. Embo J. 2008;27(7):1097–1109. doi: 10.1038/emboj.2008.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Nagy A. Manipulating the mouse embryo: A Laboratory Manual. 3rd ed Cold Spring Harbor Laboratory Press; 2003. [Google Scholar]