Abstract
Cerebral blood flow is controlled by two crucial processes, cerebral autoregulation (CA) and neurovascular coupling (NVC) or functional hyperemia. Whereas CA ensures constant blood flow over a wide range of systemic pressures, NVC ensures rapid spatial and temporal increases in cerebral blood flow in response to neuronal activation. The focus of this review is to discuss the cellular mechanisms by which astrocytes contribute to the regulation of vascular tone in terms of their participation in NVC and, to a lesser extent, CA. We discuss evidence for the various signaling modalities by which astrocytic activation leads to vasodilation and vasoconstriction of parenchymal arterioles. Moreover, we provide a rationale for the contribution of astrocytes to pressure-induced increases in vascular tone via the vasoconstrictor 20-HETE (a downstream metabolite of arachidonic acid). Along these lines, we highlight the importance of the transient receptor potential channel of the vanilloid family (TRPV4) as a key molecular determinant in the regulation of vascular tone in cerebral arterioles. Finally, we discuss current advances in the technical tools available to study NVC mechanisms in the brain as it relates to the participation of astrocytes.
Keywords: astrocytes, cerebral autoregulation, neurovascular coupling, parenchymal arteriole, vascular tone
this article is part of a collection on Unique Features of Cerebral Circulation. Other articles appearing in this collection, as well as a full archive of all collections, can be found online at http://ajpheart.physiology.org/.
Introduction
Given its limited energy reserves, the brain requires constant perfusion for proper function. This is accomplished through three fundamental processes, cerebral autoregulation (CA), neurovascular coupling (NVC) or functional hyperemia (FH) (88), and endothelium-mediated signaling (8). Whereas autoregulation ensures constant blood flow over a wide range of systemic pressures, FH ensures rapid spatial and temporal increases in cerebral blood flow (CBF) in response to neuronal activation. In addition, endothelial cells (ECs) release both vasodilators [nitric oxide (NO), endothelium-derived hyperpolarizing factor, prostacyclin, and prostaglandin E2 (PGE2)] and vasoconstrictors (endothelin-1, thromboxane A2, and prostaglandin F2α), which are capable of modulating cerebrovascular tone (8). In the next sections, we will expand on the mechanisms by which astrocytes participate in NVC and, to a lesser extent, CA. The signaling mechanisms arising from neurons, in particular, interneurons, have been reviewed previously (9, 42, 76). These pathways are an important component of the hyperemic response and must not be underestimated, especially given that they constitute the principal mechanism controlling CBF in some brain areas such as the cerebellum (169). Moreover, the intimate anatomical association of cortical neurons and cerebral blood vessels via neuronal-astrocyte-vascular appositions also lends support to the idea that vasoactive peptides released from neurons act directly on the vasculature (2, 22, 26, 50, 153, 159). While neuronal signaling is undoubtedly involved in NVC mechanisms, experimental evidence also demonstrates the involvement of astrocytes in NVC. In this review, we focus on the mechanisms by which astrocytes contribute to the regulation of vascular tone during FH or NVC (21, 148).
The Neurovascular Unit
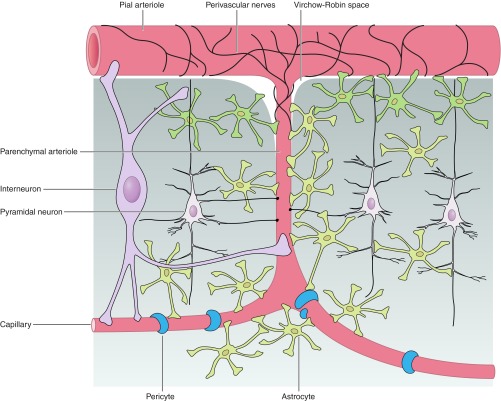
Blood supply to the brain is conducted by the internal carotid and vertebral arteries (28, 48). The vertebral arteries give rise to the basilar artery, which along with the internal carotid and communicating arteries form the circle of Willis (28). The circle of Willis further divides, giving rise to the anterior, middle, and posterior cerebral arteries that then continue to branch into smaller arteries and arterioles (28). For a detailed description of the neurovascular control of the cerebral arteries that do not penetrate the brain parenchyma, we refer the readers to excellent review articles (15, 73, 77). As arterioles outside the brain branch into pial arterioles that surround the surface of the brain, they then penetrate the brain parenchyma as penetrating or parenchymal arterioles (48) (Fig. 1). Parenchymal arterioles branch into an extensive capillary network with a heterogeneous density distribution that is dependent on the network proximity to neuronal populations (48). Higher capillary density is observed in the gray matter relative to the white matter regions (23). Accordingly, the neurovascular control of the cerebral circulation varies depending on the location and caliber of the vessels. The density of peripheral nerve terminals decreases as they penetrate the brain parenchyma and pass the Virchow-Robin space (25, 29, 31, 77); consequently, parenchymal microvessels are primarily regulated by local interneurons and neuronal terminals from a central origin (intrinsic innervation) such as the basal forebrain, raphe nucleus, and locus coeruleus (73, 77, 85).
Fig. 1.
Hypothetical illustration of the major cellular components comprising the neurovascular unit in the cerebral cortex. As pial arterioles penetrate the brain parenchyma, they gradually lose innervations from extracellular nerves originating from peripheral ganglia. The vascular tone of parenchymal arterioles is chiefly regulated by signals derived from neurons and astrocytes that encase much of the abluminal surface of the arterioles.
In recent years, much attention has been placed on the participation of glial cells, primarily astrocytes, in the regulation of cerebrovascular tone (9, 21, 72, 88, 91). The growing interest to further our understanding of the mechanisms underlying NVC was prompted by the large amount of evidence of the highly sophisticated arrangement of various cells types at the neuronal-glial-vessel interface or the neurovascular unit (NVU). The NVU (Fig. 1) is comprised of vascular cells [ECs, pericytes, and vascular smooth muscle cells (VSMCs)], neuron terminals or varicosities, astrocytes and their specialized end-foot processes, and microglia. The functional role of these various cell types varies throughout development and in disease conditions. For example, astrocytes establish a tight association with ECs during vessel formation/maturation (1, 132, 133) and with neurons during the establishment of new synapses and circuit organization (122, 145). The interaction between these various cell types aids in blood brain barrier development/maintenance (1) and CBF control. This fine-tuned anatomical organization is disrupted in disease conditions such as Alzheimer's disease (69, 86, 172), hypertension (69, 87), and stroke (7, 38, 39, 69) to name a few. Cell-to-cell communication among astrocytes (67) and/or between astrocytes-ECs (104) is supported and propagated by structural components such as gap junctions (60, 141) and anchoring proteins [e.g., integrins (38)]. It has been reported that astrocytic end feet cover about 99% of the abluminal surface of the vessel wall (92, 141). It is not clear, however, whether this extensive end-foot coverage is homogeneous among all vessel subtypes (arterioles, venules, capillaries) and/or throughout different brain regions, and if not, what functional implications varying degrees of end-foot coverage of the vasculature may have on NVC mechanisms. Clearly, the structural and functional organization of the cells comprising the NVU is critical for optimal CBF distribution in the brain.
On a larger scale than that of the NVU, additional functional organization is provided by glial networks (66). Specialized gap junctions connect neighboring astrocytes forming a syncytium (149) that is capable of efficiently modulating the activity of large neuronal populations (66) and, of particular relevance to this review, vascular networks as well. For example, the gap junction protein connexin 43 functionally links astrocytic end feet surrounding parenchymal arterioles to the glia limitans, the thick layer of astrocytic processes surrounding pial vessels and separating it from the underlying neuropil (60, 167). This functional coupling allows local information from these cells to be transmitted upstream to pial arterioles and thus vessels that supply the brain circulation (167), thereby ensuring an efficient increase in CBF during FH. Paisansathan et al. (126) have shown that adenosine and K+ mediate this upstream pial arteriole dilation following neuronal activation.
NVC and Astrocytes
An increase in neuronal activity triggers the activation of a number of pathways originating from both neurons and astrocytes that elicit rapid spatiotemporal delivery of glucose and oxygen to working neurons via increased CBF. Studies in the cortex and hippocampus suggest that neuronal activity-induced astrocytic activation occurs primarily through glutamatergic signaling pathways (58, 173). In a seminal study, Carmignoto's group (173) showed that activation of metabotropic glutamate receptors (mGluRs) induced an increase in astrocytic Ca2+, resulting in arteriolar vasodilation. The study provided mechanistic evidence that a cyclooxygenase product downstream from arachidonic acid (AA) metabolism (9), likely PGE2-mediated astrocyte-induced arteriole dilation (173). Additional studies followed that further supported the role of astrocytes in the control of vascular tone via signals released from AA metabolism. Specifically, in vivo data obtained using two-photon laser-scanning microscopy provided evidence for a signal derived from the cyclooxygenase-1 pathway in astrocyte-induced arteriole vasodilation (148). In addition to prostaglandin production (21, 35, 36, 95, 148, 173, 174), increased intracellular Ca2+ in astrocytes (3, 33) also leads to the formation and release of other vasoactive signals including NO (21, 27, 37, 94, 105, 119, 134, 164), epoxyeicosatrienoic acids (EETs) (4, 14, 21, 82, 95, 108, 119, 128), glutamate, adenosine, and ATP (6, 21, 95, 101, 137, 141, 154), all which are capable of altering the vascular tone of parenchymal arterioles (55, 61, 173). Table 1 lists some of the reported astrocyte-derived vasoactive signals acting on cerebral vessels.
Table 1.
Vasoactive astrocyte-derived signals altering pial and/or parenchymal arteriole vascular tone
| Signals | Vascular Response | Possible Mechanism | References |
|---|---|---|---|
| EETs | VD | BK channel activation on VSMC | 4, 14, 21, 82, 95, 108, 119, 128 |
| 20-HETE | VC | BK channel inhibition on VSMC | 119, 121 |
| Nitric oxide | VD/VC | likely via its interaction with prostanoids | 21, 27, 37, 94, 105, 119, 134, 164 |
| Lactate | VD | PGE2 levels | 71 |
| PGE2 | VD/VC | VD via prostanoid (EP4) receptor | 21, 35, 36, 95, 148, 173, 174 |
| VC via EP1 | |||
| K+ | VD/VC (Conc. dependent) | VD via VSMC Kir channel activation | 62, 68 |
| VC via VSMC depolarization | |||
| Ca2+ | VD/VC (Conc. dependent) | VD via VSMC BK channel activation | 61, 68 |
| VC via VSMC depolarization | |||
| ATP→AMP→adenosine | VD | via A2R in VSMC | 6, 21, 95, 101, 137, 141, 154 |
EETs, epoxyeicosatrienoic acids; VD, vasodilation; VC, vasoconstriction; Conc, concentration; BK, large conductance Ca2+-activated and voltage-dependent K+ channels; VSMC, vascular smooth muscle cells; PGE2, prostaglandin E2; EP, E-prostanoid; Kir, inwardly rectifying K+ channel; A2R, adenosine type 2 receptor.
Research investigating the effects of astrocyte-derived signals on brain arterioles was promptly complicated by the fact that different laboratories used a variety of technical approaches and parameters, which led to variability in the types of vascular responses observed. One such parameter is the age of the animal used for study. In young or neonatal animals (which are often used for NVC studies), astrocytes may have a different resting dynamic state and arterioles may not be fully differentiated leading to variability in the resting level of tone. To this end, our group showed that the polarity of vascular responses following astrocyte stimulation could be altered by the resting level of arteriole tone (14). Astrocytic stimulation induced constrictions in arterioles with little or no basal tone and dilations in arterioles with 30% or greater basal tone (14). In addition, a recent study by Sun et al. (147) demonstrated that astrocytic expression of mGluR5, a commonly targeted receptor in NVC studies, is developmentally regulated. Thus studies addressing NVC mechanisms using mGluR agonists must take into consideration shifts in the expression/pattern of these receptors in older animals. Another factor that may account for variability in vascular responses is that some of the approaches used to stimulate astrocytes or neurons may have been too strong, thus eliciting nonphysiological responses as may be the case with Ca2+ uncaging or electrical field stimulation (68, 121). Finally, experimental conditions such as temperature and oxygen gradients may have a strong impact on the activity of astrocytes as well as the levels of vascular tone, particularly as it relates to in vitro studies. For example, Gordon et al. (71) suggested that the polarity of the vascular response to glia-derived vasoactive signals is coupled to oxygen concentration levels, which modulate the metabolic state of the tissue, specifically extracellular lactate and adenosine concentrations. The authors proposed that when Po2 levels are low, the increased lactate (following neuronal stimulation) inhibits the activity of PGE2 transporters, raising extracellular PGE2 levels and thus favoring vasodilation (24). They also concluded that vasoconstriction occurs under hyperoxic conditions, where lactate levels are low and extracellular PGE2 availability is decreased (71). These observations were challenged by Lindauer et al. (106) when they demonstrated that CBF responses to electrical forepaw stimulation or cortical spreading depression were independent of O2 levels in anesthetized rats under conditions of hyperbaric oxygenation. Along these lines, Newman and colleagues (120) showed that O2 levels altered the polarity of vascular responses in vitro but had little effect in vivo in the retina. Similarly, Metea and Newman (119) attributed the polarity of astrocyte-induced vascular responses to NO availability and its interactions with AA metabolites, such that vasoconstriction is likely favored under conditions of increased NO due to NO-mediated inhibition of cytochrome P-450 (152) and the subsequent decrease in EET formation (119). In agreement with these data, Rancillac et al. (134) demonstrated that glutamate-induced neuronal NO release elicited vasoconstriction of cerebellar microvessels via a prostanoid and endothelin-dependent mechanism.
The contribution of NO signaling to NVC is likely brain region specific and dependent on the intensity of neuronal stimulation. NO has been reported as the primary NVC signal in the cerebellum (168, 169), yet it is a suggested modulatory signal in the cortex (107). Parenchymal arterioles in hippocampal brain slices constricted in response to NO synthase inhibition, suggesting that in this brain region NO has a tonic vasodilatory impact on vascular tone (56). Chisari et al. (27) provided evidence for the involvement of astrocyte-derived NO in NVC using a coculture system in which NO originating from LPS-activated glia induced basilar artery dilation. Furthermore, de Labra et al. (37) suggested that vascular or glial-derived NO was the primary mediator of low frequency-induced vasodilation, whereas neuronal NO was the primary mediator of vasodilation following intense stimulation.
An additional gas messenger molecule, carbon monoxide (CO), has also been shown to participate in glutamate-induced cerebral arteriole dilation (63). CO, derived from vascular cells (63, 102) and astrocytes (103, 127), relaxes VSMCs through its effect on Ca2+ sparks and the subsequent activation of large conductance Ca2+-activated and voltage-dependent K+ channels (BK) (90, 166). As with other glial and neuronal-derived signals (e.g., NO, K+, Ca2+), the polarity of the vascular response to CO may be modulated by its effect on NO production (89).
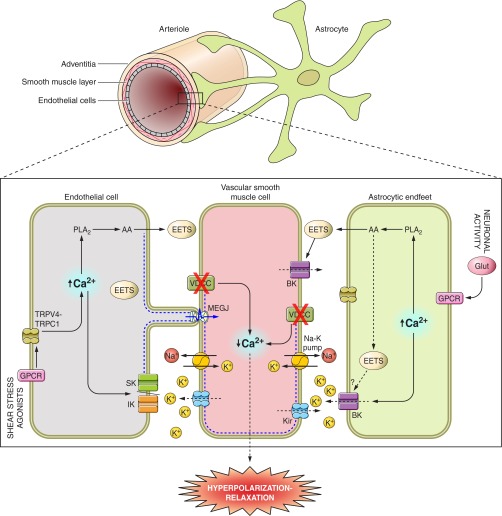
Potassium has long been shown to regulate vascular tone (Fig. 2). Activation of endothelial intermediate and small conductance K+ channels (IK and SK, respectively) can trigger a hyperpolarizing current from the endothelium to VSMC, via myoendothelial gap junctions (143), resulting in arteriole dilation. Secondary to gap junction activation, K+ efflux from IK and SK channels generate a K+ cloud (78) that hyperpolarizes neighboring VSMCs via activation of both inwardly rectifying (Kir) channels and the Na+/K+ pump (117), further contributing to endothelial-VSMC interactions. The contribution of Kir channels to VSMC hyperpolarization may be secondary to the activation of other K+ conductances as Smith et al. (142) showed blunted vasodilation to pinacidil (ATP-sensitive K+ channel activator) in the presence of the Kir channel blocker Ba2+; the authors suggested that Kir channels participate as electrical amplifiers of responses initiated by EC K+ channels. Similarly, we showed that following mGluR-mediated astrocyte activation, there is a significant increase in BK channel activity in astrocytic end-feet processes, leading to an increase in extracellular K+ that also hyperpolarizes VSMCs (62). In astrocytes, BK channels are selectively expressed in astrocytic end feet (131), thus providing a potentially high density of K+ currents in these structures and eliciting rapid changes in extracellular K+ concentrations at the gliovascular interface upon their activation. Experimental evidence suggests that the polarity of vascular responses to K+ (vasodilation vs. vasoconstriction) is dependent on the degree of K+ efflux from astrocytes. Girouard et al. (68) demonstrated that increasing the concentration of uncaged Ca2+ in astrocytes shifted dilations to constrictions and that these responses were mediated by BK channels. The putative mechanisms leading to responses of opposing polarity are the activation of VSMC Kir channels to modest K+ elevations (<20 mM) and closure of VSMC voltage-gated calcium channels resulting in membrane hyperpolarization for dilations (62) and depolarization in response to K+ elevations > 20 mM for constrictions (45). The contribution of K+ signaling to NVC is further supported by the observation that EETs increase outward K+ currents in astrocytes (84). This would suggest that upon the release of AA metabolites, K+ signaling is either enhanced or prolonged, thus contributing to hyperemia in the brain. While Higashimori et al. (84) demonstrated that outward K+ currents in astrocytes are mediated exclusively by BK and SK channels, Longden et al. (109) showed that IK channels also contribute to K+ efflux from astrocytes following electrical field stimulation. Along with the observation that extracellular K+ affects the polarity of the vascular response to astrocyte stimulation (68), our group demonstrated the importance of resting vascular tone in the K+-induced vascular response (14). We showed a positive correlation between the magnitude of K+-induced vasodilation and the level of arteriolar tone before dilation (14). K+ signaling studies suggest that, similarly to ECs, astrocytes modulate vascular tone through the efflux of K+ from various K+ channel subtypes (BK, SK and IK) and that this mechanism can induce both dilation and constriction of parenchymal arterioles in a K+-concentration manner.
Fig. 2.
K+ signaling mechanisms at the neurovascular unit. Schematic representation of the signaling mechanism by which endothelial cell (EC) and astrocyte signaling induce membrane potential (Vm) hyperpolarization of vascular smooth muscle cells (VSMCs) and arteriole vasodilation. Shear stress or agonist-induced activation of G protein-coupled receptors (GPCR) increases Ca2+ in ECs via transient receptor potential (TRP) channel-dependent mechanisms resulting in the activation of intermediate conductance (IK) and small conductance (SK) potassium channels. The hyperpolarization mediated by these channels spreads to the VSMCs through myoendothelial gap junctions (MEGJ). Moreover, the resulting K+ efflux from ECs activates VSMC inwardly rectifying K+ channel (Kir) channels and the Na+/K+ pump inducing Vm hyperpolarization and closure of voltage-dependent calcium channels (VDCC), decreased intracellular Ca2+, and VSMC relaxation. On the other hand, glutamate released during neuronal activation binds to GPCRs on astrocytes, triggering an increase in Ca2+ that, in turn, activates large conductance Ca2+ activated potassium channels (BK) on astrocytic end feet (EF). Astrocytic BK channel activation causes potassium efflux into the perivascular space and, again, VSMC Kir channel activation followed by relaxation. TRP of the vanilloid family (TRPV4) channel activation has recently been shown to elicit increases in astrocytic EF Ca2+ as well. The intracellular Ca2+ increase in ECs and astrocytes also results in PLA2 activation, arachidonic acid (AA) metabolism, and the production/release of various vasoactive substances including epoxyeicosatrienoic acids (EETs), which in turn can modulate BK channels in astrocytes and vascular cells, further contributing to the regulation of vascular tone. TRPC1, transient receptor potential canonical-1.
Relevant to K+ signaling and NVC mechanisms is the K+ buffering/siphoning mechanism in which astrocytes remove K+ from the synapse during neuronal activity and redistribute it to areas with lower K+ concentrations such as the gliovascular space to prevent excessive neuronal excitability induced by continued neuronal depolarization (96). However, evidence from the retina suggests that this passive mechanism does not hold true as direct depolarization of Müller cells does not induce Kir-mediated efflux of K+ at the gliovascular space or elicit dilation (118). Nevertheless, alternative mediators, such as other classes of K+ channels, may underlie a K+ buffering/siphoning mechanism that contributes to NVC. Furthermore, in a recent study, Nedergaard's group (158) showed that neuronal stimulation activates the Na+/K+ pump. Activation of the Na+/K+ pump will result in hyperpolarization of the astrocytic membrane, which would then facilitate the influx of K+ through Kir channels on perisynaptic astrocytic processes. Moreover, upstream events (e.g., activation of Na+-Ca2+ exchanger and/or G protein-coupled receptors) triggering increases in astrocytic Ca2+ would activate BK (62) or other potassium channels at the end feet, thereby contributing to vasodilation and the removal of K+ from the synapse (K+ buffering). Thus we suggest that the K+ siphoning hypothesis and its contribution to NVC be revisited, keeping in mind variations in brain regions as well as the contribution of other classes of K+ channels at the synapse and the gliovascular space.
CA and Astrocytes
CA constitutes a critical physiological mechanism of cerebral arterioles that ensures constant perfusion throughout a broad range of systemic pressures (50–150 mmHg) (17, 28). It is well established that CA involves at least three major components: myogenic, neurogenic, and metabolic. In the myogenic component of CA, the increased transmural pressure in cerebral arterioles increases vascular resistance through a number of mechanisms, including activation of L-type calcium channels, membrane potential (Vm) depolarization, and increased production of vasoconstrictors (e.g., 20-HETE) downstream from inositol 1,4,5-triphosphate and diacylglycerol activation (81, 151). 20-HETE induces VSMC constriction via the inhibition of BK channels followed by Vm depolarization and opening of calcium channels (80). In the cerebral circulation, the AA metabolite 20-HETE can be produced by VSMCs (65) and the endothelium (124). Work by Faraci et al. (52) demonstrated that autoregulatory responses persist following endothelial injury, further supporting the myogenic component of CA. While much has been discovered concerning the myogenic constriction of cerebral vessels, less is understood on the contribution of the metabolic and neurogenic pathways. Even fewer studies have addressed the potential mechanisms by which cells within the brain parenchyma (e.g., astrocytes) contribute to the regulation of arteriole tone during CA.
Within the intricate structural arrangement of the NVU, VSMCs are situated between two very different cell types, ECs on the luminal side and astrocytes on the abluminal side. The literature suggests that many of the endothelial vasoactive pathways that allow for EC-VSMC interactions (e.g., AA metabolites, NO, EETs, adenosine, K+) (53, 54) are present in astrocytes as well, thus potentially contributing to astrocyte-VSMC interactions and the regulation of vascular tone (Fig. 1). Although speculative at this moment, one reason for such a structural arrangement may stem from the need to maintain brain blood volume within physiological levels. Because the brain is encased in a closed cranium with no space to expand, astrocytes may participate in the control of CBF as an additional system to prevent overexpansion of brain volume. They may also participate in the signaling necessary (besides NVC/FH) (148) to maintain a level of vascular tone that allows for optimal perfusion to the needed areas. Thus astrocytes may be constant monitors of both neuronal activity and the degree of basal CBF. While the contributions of astrocytes to NVC and FH-induced increases in CBF are well documented, little is known as to whether astrocytes contribute to resting CBF. The ability of astrocytes to both dilate and constrict arterioles favors this idea since their intimate contact with active neurons places them in an ideal position to regulate local vascular tone. Thus astrocytes may be key contributors to CA mechanisms as well (82). However, critical questions remain unanswered, particularly whether astrocytes, as well as neurons, have the ability to sense autoregulatory-mediated adjustments in perfusion/energy supply and thus participate in bidirectional communication at the NVU.
Studies performed in cannulated cerebral arterioles showed increased flow/pressure-induced vasoconstriction in parenchymal arterioles in the presence or absence of a functional endothelium (19, 20, 70). While a significant portion of this pressure/flow-induced response is mediated by the intrinsic properties of the VSMCs, it is possible that signals released from activated astrocytes also contribute to the constriction. As mentioned earlier, the abluminal surface of parenchymal cerebral vessels is almost completely covered by astrocytic end-feet processes (92, 141); these specialized structures are unique in that they express a distinct subset of channels including BK, aquaporin-4, and the recently characterized transient receptor potential channel of the vanilloid subfamily, TRPV4. Using brain slices, Mulligan and MacVicar (121) showed that astrocytic stimulation via uncaging of Ca2+ (in the absence of neuronal stimulation) induced parenchymal arteriole vasoconstriction. This observation provided evidence that in addition to contributing to NVC during increases in neuronal activity, astrocytes may also be capable of constricting arterioles, a response that may be useful to the brain under conditions of increased perfusion pressures. Importantly, the anchoring of some of the channels mentioned is also dependent on the presence of mechanosensory proteins such as integrins (40). The polarized expression of specific proteins and channels in astrocytic end feet is ideal for continuous monitoring and subsequent adjustment of vascular tone. While evidence for this mechanism is lacking, there is indeed evidence that TRPV4 channels, which upon activation cause an increase in intracellular Ca2+ (43), are expressed in astrocytic end feet. Thus it is reasonable to suggest that in addition to a myogenic component, signals from perivascular cells may also contribute to increases in vascular tone in response to increases in luminal pressure and/or flow (79, 81, 151).
TRPV4 Channels as Regulators of Vascular Tone
Structurally, the TRPV4 protein is comprised of six transmembrane segments with a cation pore located between segments 5 and 6 (51). Four subunits are required for the formation of a functional TRPV4 channel (51). In addition to forming homotetramers, recent studies suggest that TRPV4 can also heteromerize with transient receptor potential canonical-1 and transient receptor potential polycystin-2 (43, 112). Functionally, these cation channels are moderately selective for Ca2+ with a permeability ratio of 5.8–6.9 PCa/PNa (146, 155, 160). TRPV4 channels are activated by both chemical [endocannabinoids (162), AA (162) and 4-α-phorbol esters (5, 160, 163)] and physical stimuli [cell swelling (146), heat (74, 163) and mechanical displacement (125)]. EETs, which are produced by both ECs and astrocytes, are potent endogenous activators of TRPV4 channels (46, 47, 130, 156, 162); cytochrome P-450 epoxygenase metabolism of AA to EETs mediates TRPV4 activation by endocannabinoids and cell swelling (157, 162).
TRPV4 channels are important mediators of vascular tone. VSMC and EC TRPV4 expression has been demonstrated in multiple tissues including aorta and extraaveolar vessels as well as carotid, cerebral, mesenteric, and pulmonary arteries (5, 64, 97, 113, 114, 156, 160, 161, 165, 170). In VSMCs, EET-induced activation of TRPV4 channels initiates VSMC hyperpolarization and vascular relaxation via the subsequent activation of BK channels (46, 47). In ECs, increases in intracellular Ca2+ induced by TRPV4 activation initiate both NO and endothelial-derived hyperpolarizing factor-mediated vasodilation (98, 144).
In the brain, TRPV4 expression has been demonstrated in both neuronal and nonneuronal cell types, including astrocytes and microglia in addition to cerebral artery VSMCs and ECs (12, 13, 30, 46, 51, 100, 113, 139). TRPV4 channels expressed in both cerebral artery VSMCs (46) and ECs (113) have been shown to contribute to cerebral arteriole tone. In astrocytes, TPRV4 channel expression is localized primarily to astrocytic end feet (12, 13) that envelop the cerebral vasculature, an ideal location for monitoring and/or regulating vascular tone. The importance of astrocytic TRPV4 channels in modulating vascular tone has been demonstrated in a recent study by Dunn et al. (44) in which they show that astrocytic TRPV4 channel activation enhances vasodilation during NVC. Whereas they show that EETs are capable of inducing TRPV4-mediated increases in astrocytic end-foot Ca2+, EETs do not seem to mediate this TRPV4-induced amplification of NVC; instead, the response is induced by the subsequent activation of inositol 1,4,5-trisphosphate receptors (44).
Other indirect evidence also suggests that TRPV4 channels may play a more comprehensive role in monitoring and/or regulating vascular tone. Higashimori et al. (84) demonstrated that the synthetic EET analog 11-nonyloxy-undec-8 (Z)-enoic acid increased both Ca2+ oscillation frequency and BK channel currents in astrocytes. Given that EETs are endogenous TRPV4 agonists (46, 47, 130, 156, 162) and astrocytic K+ signaling is an important mediator of vascular tone (62), these data support the hypothesis that EET-induced activation of TRPV4 channels and subsequent increases in intracellular Ca2+ may contribute to astrocyte K+ signaling and the regulation of cerebral vascular tone. Upon their activation, astrocytic TRPV4 channels also engage inositol 1,4,5-trisphosphate receptors to increase Ca2+ as recently demonstrated (44), providing an additional mechanism by which TRPV4 channel-induced Ca2+ signaling in astrocytes may contribute to the regulation of vascular tone. Furthermore, TRPV4 channel activation is associated with the production of NO (41, 171), which itself can elicit sustained increases in astrocytic Ca2+ (11). These data suggest that TRPV4 channels may also survey and/or modulate vascular tone via an NO-dependent signaling mechanism. Given their strategic expression in astrocytic end feet combined with their capacity to respond to a multitude of signals, TRPV4 channels are ideal candidates to mediate a bidirectional communication modality within the NVU.
Tools to Study NVC in the Brain
The role of astrocytes in the control of vascular tone has been addressed with a number of in vivo and in vitro techniques. Both include their own set of advantages and disadvantages. It is reasonable to state that in vivo simultaneous vascular and astrocyte recordings are ideal as the approach provides an intact system including pressurized and perfused vessels with the complete anatomical organization of astrocytes and neurons in their surroundings. Moreover, in vivo techniques have been complemented with advances in the use of genetically engineered animals, adenoviruses, and Ca2+ sensors (e.g., GCamP), as well as additional stimulation modalities such as uncaging and optogenetics (59, 115, 138). Although these approaches provide powerful data, confounding limitations lie in the fact that imaging is confined to the first few hundred microns below the surface of the brain, acquisition rates are generally slower, and pharmacological approaches are limited given the need to use higher drug concentrations. In addition, the use of anesthesia must be taken into consideration given that it can have profound effects on constituents of the NVU (10, 150). Nonetheless, in vivo two-photon laser scanning microscopy is among the most sophisticated tools to study astrocyte-vascular interactions.
Among the in vitro approaches, the most commonly used technique is the brain slice model. Slices are an ideal model as all of the constituents of the NVU are intact allowing for the study of neuronal-to-astrocyte-to-vessel communication. Moreover, the fact that virtually any portion of the brain can be sliced allows for the study of NVC in different brain regions which may shed light on unexplored mechanisms that may differ from those in commonly studied areas, namely the cortex and hippocampus. Earlier studies that lacked some of the important steps we know today must be taken into account when studying arterioles that are not pressurized and perfused. To this end, it is clear now that to attain a physiological vascular response, arterioles must have tone. The latter can be achieved by either perfusing the slice with a vasoconstrictor such as the thromboxane A2 receptor agonist U-46619 (14, 61, 62, 110, 111) or NO synthase inhibitors (57, 173), as previously reported, or by cannulating and pressurizing the arterioles as previously performed in excised cerebral vessels (123). The latter approach consists of introducing a cannula into the open end of the arteriole at the pial surface and perfusing the arteriole (93, 110) to attain physiological levels of luminal pressure and shear stress (19, 151). Given that this approach allows cerebral arterioles to develop myogenic tone, arteriole cannulation in brain slices is an ideal approach for studying the cellular mechanisms underlying NVC in the brain.
In addition to vascular tone, the role and effects of tissue O2 tension on NVC have been debated and clearly deserve further consideration. Extensive studies were performed on brain slices to optimize the conditions by which the tissue would remain viable for several hours (18, 32, 75). Perfusing brain slices with a bicarbonate buffered artificial cerebrospinal fluid (aCSF) gassed with 95% O2-5% CO2 clearly aids in prolonging tissue viability (83, 135). However, while these solutions have led to successful electrophysiological and imaging recordings from both neurons and astrocytes, a closer look at the constituents of the aCSF is needed. For example, glucose concentrations used by most researchers (10–25 mM) exceeds that of the actual CSF (∼2.5 mM) (140). Moreover, the use of 95% O2 may result in variable levels of neuronal excitability as well as superoxide production and cell death (34, 83). Additionally, tissue Po2 levels may also have profound effects on vascular responses since it may alter the levels of vasoactive agonists at the gliovascular interface (71). Similarly, high O2 levels may have direct effects on NO availability (99), also interfering with the vasodilator/vasoconstrictor ratios acting on the arterioles. Thus future considerations must be made as to the need to switch brain slice studies (at least for NVC mechanisms) to lower glucose and O2 concentrations that approximate physiological levels without compromising the viability of the tissue.
In summary, it is clear that NVC is mediated by a multiplicity of signals and conditions including astrocyte release of vasodilatory signals. However, to better understand the physiology of how astrocytes regulate vascular tone, we must first take into consideration the conditions under which these mechanisms are being studied and the questions elicited by previous studies. Consequently, when studying NVC, one must consider that the predominant vascular response is highly dependent on the resting levels of vascular tone, O2 gradients, anesthesia, and strength/duration/type of stimulus. Future studies addressing whether astrocytes participate in regulating basal vascular tone will shed light on the role these cells have in CA (82, 136).
In light of the large amount of knowledge gained on the signaling modalities by which astrocytes and neurons control vascular tone, new questions have arisen. For example, what are the functional differences between astrocyte signaling to arterioles, venules, and capillaries? To this end, Peppiatt et al. (129) demonstrated norepinephrine-induced constrictions of pericytes and glutamate-induced dilations providing evidence for the control of blood flow at the level of the capillaries. Thus it is possible to speculate that given the intimate association between astrocytes and pericytes (16, 116), astrocyte-induced pericyte dilation/constriction may also contribute to the regulation in flow at the level of the capillaries. A mechanism of this nature may provide very tight regulation of CBF without the need to involve upstream vessels. The potential for regulatory diversity within the brain microcirculation itself highlights the necessity of continuing research, using a range of advanced methodologies to reveal the intricate cellular signaling modalities involved in NVC and CBF autoregulation.
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the author (s).
AUTHOR CONTRIBUTIONS
J.A.F. prepared figures; J.A.F. and J.A.I. drafted manuscript; J.A.F. and J.A.I. edited and revised manuscript; J.A.F. and J.A.I. approved final version of manuscript.
ACKNOWLEDGMENTS
This work was funded by a National Heart, Lung, and Blood Institute (NHLBI) Grant R01-HL089067-02 (to J. A. Filosa), as well as American Heart Association Predoctoral Fellowship 11PRE7400037, and NHLBI Multidisciplinary Predoctoral Training Grant T32-HL-076146 (to J. A. Iddings).
REFERENCES
- 1.Abbott NJ, Ronnback L, Hansson E. Astrocyte-endothelial interactions at the blood-brain barrier. Nat Rev Neurosci 7: 41–53, 2006 [DOI] [PubMed] [Google Scholar]
- 2.Abounader R, Hamel E. Associations between neuropeptide Y nerve terminals and intraparenchymal microvessels in rat and human cerebral cortex. J Comp Neurol 388: 444–453, 1997 [PubMed] [Google Scholar]
- 3.Aguado F, Espinosa-Parrilla JF, Carmona MA, Soriano E. Neuronal activity regulates correlated network properties of spontaneous calcium transients in astrocytes in situ. J Neurosci 22: 9430–9444, 2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Alkayed NJ, Narayanan J, Gebremedhin D, Medhora M, Roman RJ, Harder DR. Molecular characterization of an arachidonic acid epoxygenase in rat brain astrocytes. Stroke 27: 971–979, 1996 [DOI] [PubMed] [Google Scholar]
- 5.Alvarez DF, King JA, Weber D, Addison E, Liedtke W, Townsley MI. Transient receptor potential vanilloid 4-mediated disruption of the alveolar septal barrier: a novel mechanism of acute lung injury. Circ Res 99: 988–995, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Anderson CM, Bergher JP, Swanson RA. ATP-induced ATP release from astrocytes. J Neurochem 88: 246–256, 2004 [DOI] [PubMed] [Google Scholar]
- 7.Anderson MF, Blomstrand F, Blomstrand C, Eriksson PS, Nilsson M. Astrocytes and stroke: networking for survival? Neurochem Res 28: 293–305, 2003 [DOI] [PubMed] [Google Scholar]
- 8.Andresen J, Shafi NI, Bryan RM., Jr Endothelial influences on cerebrovascular tone. J Appl Physiol 100: 318–327, 2006 [DOI] [PubMed] [Google Scholar]
- 9.Attwell D, Buchan AM, Charpak S, Lauritzen M, Macvicar BA, Newman EA. Glial and neuronal control of brain blood flow. Nature 468: 232–243, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ayata C, Dunn AK, Gursoy OY, Huang Z, Boas DA, Moskowitz MA. Laser speckle flowmetry for the study of cerebrovascular physiology in normal and ischemic mouse cortex. J Cereb Blood Flow Metab 24: 744–755, 2004 [DOI] [PubMed] [Google Scholar]
- 11.Bal-Price A, Moneer Z, Brown GC. Nitric oxide induces rapid, calcium-dependent release of vesicular glutamate and ATP from cultured rat astrocytes. Glia 40: 312–323, 2002 [DOI] [PubMed] [Google Scholar]
- 12.Benfenati V, Amiry-Moghaddam M, Caprini M, Mylonakou MN, Rapisarda C, Ottersen OP, Ferroni S. Expression and functional characterization of transient receptor potential vanilloid-related channel 4 (TRPV4) in rat cortical astrocytes. Neuroscience 148: 876–892, 2007 [DOI] [PubMed] [Google Scholar]
- 13.Benfenati V, Caprini M, Dovizio M, Mylonakou MN, Ferroni S, Ottersen OP, Amiry-Moghaddam M. An aquaporin-4/transient receptor potential vanilloid 4 (AQP4/TRPV4) complex is essential for cell-volume control in astrocytes. Proc Natl Acad Sci USA 108: 2563–2568, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Blanco VM, Stern JE, Filosa JA. Tone-dependent vascular responses to astrocyte-derived signals. Am J Physiol Heart Circ Physiol 294: H2855–H2863, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bleys RL, Cowen T. Innervation of cerebral blood vessels: morphology, plasticity, age-related, and Alzheimer's disease-related neurodegeneration. Microsc Res Tech 53: 106–118, 2001 [DOI] [PubMed] [Google Scholar]
- 16.Bonkowski D, Katyshev V, Balabanov RD, Borisov A, Dore-Duffy P. The CNS microvascular pericyte: pericyte-astrocyte crosstalk in the regulation of tissue survival. Fluids Barriers CNS 8: 8, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bor-Seng-Shu E, Kita WS, Figueiredo EG, Paiva WS, Fonoff ET, Teixeira MJ, Panerai RB. Cerebral hemodynamics: concepts of clinical importance. Arq Neuropsiquiatr 70: 352–356, 2012 [DOI] [PubMed] [Google Scholar]
- 18.Brahma B, Forman RE, Stewart EE, Nicholson C, Rice ME. Ascorbate inhibits edema in brain slices. J Neurochem 74: 1263–1270, 2000 [DOI] [PubMed] [Google Scholar]
- 19.Bryan RM, Jr, Marrelli SP, Steenberg ML, Schildmeyer LA, Johnson TD. Effects of luminal shear stress on cerebral arteries and arterioles. Am J Physiol Heart Circ Physiol 280: H2011–H2022, 2001 [DOI] [PubMed] [Google Scholar]
- 20.Bryan RM, Jr, Steenberg ML, Marrelli SP. Role of endothelium in shear stress-induced constrictions in rat middle cerebral artery. Stroke 32: 1394–1400, 2001 [DOI] [PubMed] [Google Scholar]
- 21.Carmignoto G, Gomez-Gonzalo M. The contribution of astrocyte signalling to neurovascular coupling. Brain Res Rev 63: 138–148, 2010 [DOI] [PubMed] [Google Scholar]
- 22.Cauli B, Hamel E. Revisiting the role of neurons in neurovascular coupling. Front Neuroenergetics 2: 9, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cavaglia M, Dombrowski SM, Drazba J, Vasanji A, Bokesch PM, Janigro D. Regional variation in brain capillary density and vascular response to ischemia. Brain Res 910: 81–93, 2001 [DOI] [PubMed] [Google Scholar]
- 24.Chan BS, Endo S, Kanai N, Schuster VL. Identification of lactate as a driving force for prostanoid transport by prostaglandin transporter PGT. Am J Physiol Renal Physiol 282: F1097–F1102, 2002 [DOI] [PubMed] [Google Scholar]
- 25.Chedotal A, Hamel E. Serotonin-synthesizing nerve fibers in rat and cat cerebral arteries and arterioles: immunohistochemistry of tryptophan-5-hydroxylase. Neurosci Lett 116: 269–274, 1990 [DOI] [PubMed] [Google Scholar]
- 26.Chedotal A, Umbriaco D, Descarries L, Hartman BK, Hamel E. Light and electron microscopic immunocytochemical analysis of the neurovascular relationships of choline acetyltransferase and vasoactive intestinal polypeptide nerve terminals in the rat cerebral cortex. J Comp Neurol 343: 57–71, 1994 [DOI] [PubMed] [Google Scholar]
- 27.Chisari M, Salomone S, Laureanti F, Copani A, Sortino MA. Modulation of cerebral vascular tone by activated glia: involvement of nitric oxide. J Neurochem 91: 1171–1179, 2004 [DOI] [PubMed] [Google Scholar]
- 28.Cipolla MJ. The Cerebral Circulation. San Rafael, CA: Morgan & Claypool Life Sciences, 2009 [PubMed] [Google Scholar]
- 29.Cipolla MJ, Li R, Vitullo L. Perivascular innervation of penetrating brain parenchymal arterioles. J Cardiovasc Pharmacol 44: 1–8, 2004 [DOI] [PubMed] [Google Scholar]
- 30.Cohen DM. The transient receptor potential vanilloid-responsive 1 and 4 cation channels: role in neuronal osmosensing and renal physiology. Curr Opin Nephrol Hypertens 16: 451–458, 2007 [DOI] [PubMed] [Google Scholar]
- 31.Cohen Z, Bovento G, Lacombe P, Seylaz J, MacKenzie ET, Hamel E. Cerebrovascular nerve fibers immunoreactive for tryptophan-5-hydroxylase in the rat: distribution, putative origin and comparison with sympathetic noradrenergic nerves. Brain Res 598: 203–214, 1992 [DOI] [PubMed] [Google Scholar]
- 32.Collingridge GL. The brain slice preparation: a tribute to the pioneer Henry McIlwain. J Neurosci Methods 59: 5–9, 1995 [DOI] [PubMed] [Google Scholar]
- 33.Cornell-Bell AH, Finkbeiner SM, Cooper MS, Smith SJ. Glutamate induces calcium waves in cultured astrocytes: long-range glial signaling. Science 247: 470–473, 1990 [DOI] [PubMed] [Google Scholar]
- 34.D'Agostino DP, Putnam RW, Dean JB. Superoxide (*O2-) production in CA1 neurons of rat hippocampal slices exposed to graded levels of oxygen. J Neurophysiol 98: 1030–1041, 2007 [DOI] [PubMed] [Google Scholar]
- 35.Dabertrand F, Hannah RM, Pearson JM, Hill-Eubanks DC, Brayden JE, Nelson MT. Prostaglandin E2, a postulated astrocyte-derived neurovascular coupling agent, constricts rather than dilates parenchymal arterioles. J Cereb Blood Flow Metab 33: 479–482, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Davis RJ, Murdoch CE, Ali M, Purbrick S, Ravid R, Baxter GS, Tilford N, Sheldrick RL, Clark KL, Coleman RA. EP4 prostanoid receptor-mediated vasodilatation of human middle cerebral arteries. Br J Pharmacol 141: 580–585, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.de Labra C, Rivadulla C, Espinosa N, Dasilva M, Cao R, Cudeiro J. Different sources of nitric oxide mediate neurovascular coupling in the lateral geniculate nucleus of the cat. Front Syst Neurosci 3: 9, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.del Zoppo GJ. The neurovascular unit in the setting of stroke. J Intern Med 267: 156–171, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.del Zoppo GJ, Hallenbeck JM. Advances in the vascular pathophysiology of ischemic stroke. Thromb Res 98: 73–81, 2000 [DOI] [PubMed] [Google Scholar]
- 40.del Zoppo GJ, Milner R. Integrin-matrix interactions in the cerebral microvasculature. Arterioscler Thromb Vasc Biol 26: 1966–1975, 2006 [DOI] [PubMed] [Google Scholar]
- 41.Ding XL, Wang YH, Ning LP, Zhang Y, Ge HY, Jiang H, Wang R, Yue SW. Involvement of TRPV4-NO-cGMP-PKG pathways in the development of thermal hyperalgesia following chronic compression of the dorsal root ganglion in rats. Behav Brain Res 208: 194–201, 2010 [DOI] [PubMed] [Google Scholar]
- 42.Drake CT, Iadecola C. The role of neuronal signaling in controlling cerebral blood flow. Brain Lang 102: 141–152, 2007 [DOI] [PubMed] [Google Scholar]
- 43.Du J, Wong WY, Sun L, Huang Y, Yao X. Protein kinase G inhibits flow-induced Ca2+ entry into collecting duct cells. J Am Soc Nephrol 23: 1172–1180, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Dunn KM, Hill-Eubanks DC, Liedtke WB, Nelson MT. TRPV4 channels stimulate Ca2+-induced Ca2+ release in astrocytic endfeet and amplify neurovascular coupling responses. Proc Natl Acad Sci USA 110: 6157–6162, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Dunn KM, Nelson MT. Potassium channels and neurovascular coupling. Circ J 74: 608–616, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Earley S, Heppner TJ, Nelson MT, Brayden JE. TRPV4 forms a novel Ca2+ signaling complex with ryanodine receptors and BKCa channels. Circ Res 97: 1270–1279, 2005 [DOI] [PubMed] [Google Scholar]
- 47.Earley S, Pauyo T, Drapp R, Tavares MJ, Liedtke W, Brayden JE. TRPV4-dependent dilation of peripheral resistance arteries influences arterial pressure. Am J Physiol Heart Circ Physiol 297: H1096–H1102, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Edvinsson L, MacKenzie ET. General and comparative anatomy of the cerebral circulation. In: Cerebral Blood Flow and Metabolism (2nd ed), edited by Edvinsson LM, Krause DN. Philadelphia: Lippincott Williams & Wilkins, 2002 [Google Scholar]
- 50.Estrada C, DeFelipe J. Nitric oxide-producing neurons in the neocortex: morphological and functional relationship with intraparenchymal microvasculature. Cereb Cortex 8: 193–203, 1998 [DOI] [PubMed] [Google Scholar]
- 51.Everaerts W, Nilius B, Owsianik G. The vanilloid transient receptor potential channel TRPV4: from structure to disease. Prog Biophys Mol Biol 103: 2–17, 2010 [DOI] [PubMed] [Google Scholar]
- 52.Faraci FM, Baumbach GL, Heistad DD. Myogenic mechanisms in the cerebral circulation. J Hypertens Suppl 7: S61–S65, 1989 [PubMed] [Google Scholar]
- 53.Feletou M, Tang EH, Vanhoutte PM. Nitric oxide the gatekeeper of endothelial vasomotor control. Front Biosci 13: 4198–4217, 2008 [DOI] [PubMed] [Google Scholar]
- 54.Feletou M, Vanhoutte PM. EDHF: an update.Clin Sci (Lond) 117: 139–155, 2009 [DOI] [PubMed] [Google Scholar]
- 55.Fellin T, Carmignoto G. Neurone-to-astrocyte signalling in the brain represents a distinct multifunctional unit. J Physiol 559: 3–15, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Fergus A, Jin Y, Thai QA, Kassell NF, Lee KS. Tonic protein kinase C-mediated vasoconstriction is unmasked when nitric oxide synthase is inhibited in cerebral microvessels. Neuroscience 74: 927–934, 1996 [DOI] [PubMed] [Google Scholar]
- 57.Fergus A, Jin Y, Thai QA, Kassell NF, Lee KS. Vasodilatory actions of calcitonin gene-related peptide and nitric oxide in parenchymal microvessels of the rat hippocampus. Brain Res 694: 78–84, 1995 [DOI] [PubMed] [Google Scholar]
- 58.Fergus A, Lee KS. Regulation of cerebral microvessels by glutamatergic mechanisms. Brain Res 754: 35–45, 1997 [DOI] [PubMed] [Google Scholar]
- 59.Figueiredo M, Lane S, Tang F, Liu BH, Hewinson J, Marina N, Kasymov V, Souslova EA, Chudakov DM, Gourine AV, Teschemacher AG, Kasparov S. Optogenetic experimentation on astrocytes. Exp Physiol 96: 40–50, 2011 [DOI] [PubMed] [Google Scholar]
- 60.Figueroa XF, Duling BR. Gap junctions in the control of vascular function. Antioxid Redox Signal 11: 251–266, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Filosa JA, Bonev AD, Nelson MT. Calcium dynamics in cortical astrocytes and arterioles during neurovascular coupling. Circ Res 95: e73–e81, 2004 [DOI] [PubMed] [Google Scholar]
- 62.Filosa JA, Bonev AD, Straub SV, Meredith AL, Wilkerson MK, Aldrich RW, Nelson MT. Local potassium signaling couples neuronal activity to vasodilation in the brain. Nat Neurosci 9: 1397–1403, 2006 [DOI] [PubMed] [Google Scholar]
- 63.Fiumana E, Parfenova H, Jaggar JH, Leffler CW. Carbon monoxide mediates vasodilator effects of glutamate in isolated pressurized cerebral arterioles of newborn pigs. Am J Physiol Heart Circ Physiol 284: H1073–H1079, 2003 [DOI] [PubMed] [Google Scholar]
- 64.Gao F, Wang DH. Hypotension induced by activation of the transient receptor potential vanilloid 4 channels: role of Ca2+-activated K+ channels and sensory nerves. J Hypertens 28: 102–110, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Gebremedhin D, Lange AR, Lowry TF, Taheri MR, Birks EK, Hudetz AG, Narayanan J, Falck JR, Okamoto H, Roman RJ, Nithipatikom K, Campbell WB, Harder DR. Production of 20-HETE and its role in autoregulation of cerebral blood flow. Circ Res 87: 60–65, 2000 [DOI] [PubMed] [Google Scholar]
- 66.Giaume C, Koulakoff A, Roux L, Holcman D, Rouach N. Astroglial networks: a step further in neuroglial and gliovascular interactions. Nat Rev Neurosci 11: 87–99, 2010 [DOI] [PubMed] [Google Scholar]
- 67.Giaume C, Liu X. From a glial syncytium to a more restricted and specific glial networking. J Physiol (Paris) 106: 34–39, 2012 [DOI] [PubMed] [Google Scholar]
- 68.Girouard H, Bonev AD, Hannah RM, Meredith A, Aldrich RW, Nelson MT. Astrocytic endfoot Ca2+ and BK channels determine both arteriolar dilation and constriction. Proc Natl Acad Sci USA 107: 3811–3816, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Girouard H, Iadecola C. Neurovascular coupling in the normal brain and in hypertension, stroke, and Alzheimer disease. J Appl Physiol 100: 328–335, 2006 [DOI] [PubMed] [Google Scholar]
- 70.Golding EM, Robertson CS, Bryan RM., Jr Comparison of the myogenic response in rat cerebral arteries of different calibers. Brain Res 785: 293–298, 1998 [DOI] [PubMed] [Google Scholar]
- 71.Gordon GR, Choi HB, Rungta RL, Ellis-Davies GC, MacVicar BA. Brain metabolism dictates the polarity of astrocyte control over arterioles. Nature 456: 745–749, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Gordon GR, Mulligan SJ, MacVicar BA. Astrocyte control of the cerebrovasculature. Glia 55: 1214–1221, 2007 [DOI] [PubMed] [Google Scholar]
- 73.Gulbenkian S, Uddman R, Edvinsson L. Neuronal messengers in the human cerebral circulation. Peptides 22: 995–1007, 2001 [DOI] [PubMed] [Google Scholar]
- 74.Guler AD, Lee H, Iida T, Shimizu I, Tominaga M, Caterina M. Heat-evoked activation of the ion channel, TRPV4. J Neurosci 22: 6408–6414, 2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Hajos N, Mody I. Establishing a physiological environment for visualized in vitro brain slice recordings by increasing oxygen supply and modifying aCSF content. J Neurosci Methods 183: 107–113, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Hamel E. Cholinergic modulation of the cortical microvascular bed. Prog Brain Res 145: 171–178, 2004 [DOI] [PubMed] [Google Scholar]
- 77.Hamel E. Perivascular nerves and the regulation of cerebrovascular tone. J Appl Physiol 100: 1059–1064, 2006 [DOI] [PubMed] [Google Scholar]
- 78.Hannah RM, Dunn KM, Bonev AD, Nelson MT. Endothelial SK (Ca) and IK (Ca) channels regulate brain parenchymal arteriolar diameter and cortical cerebral blood flow. J Cereb Blood Flow Metab 31: 1175–1186, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Harder DR, Campbell WB, Roman RJ. Role of cytochrome P-450 enzymes and metabolites of arachidonic acid in the control of vascular tone. J Vasc Res 32: 79–92, 1995 [DOI] [PubMed] [Google Scholar]
- 80.Harder DR, Lange AR, Gebremedhin D, Birks EK, Roman RJ. Cytochrome P450 metabolites of arachidonic acid as intracellular signaling molecules in vascular tissue. J Vasc Res 34: 237–243, 1997 [DOI] [PubMed] [Google Scholar]
- 81.Harder DR, Narayanan J, Gebremedhin D. Pressure-induced myogenic tone and role of 20-HETE in mediating autoregulation of cerebral blood flow. Am J Physiol Heart Circ Physiol 300: H1557–H1565, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Harder DR, Zhang C, Gebremedhin D. Astrocytes function in matching blood flow to metabolic activity. News Physiol Sci 17: 27–31, 2002 [DOI] [PubMed] [Google Scholar]
- 83.Hájos N, Mody I. Establishing a physiological environment for visualized in vitro brain slice recordings by increasing oxygen supply and modifying aCSF content. J Neurosci Methods 183: 107–113, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Higashimori H, Blanco VM, Tuniki VR, Falck JR, Filosa JA. Role of epoxyeicosatrienoic acids as autocrine metabolites in glutamate-mediated K+ signaling in perivascular astrocytes. Am J Physiol Cell Physiol 299: C1068–C1078, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Iadecola C. Neurogenic control of the cerebral microcirculation: is dopamine minding the store? Nat Neurosci 1: 263–265, 1998 [DOI] [PubMed] [Google Scholar]
- 86.Iadecola C. Neurovascular regulation in the normal brain and in Alzheimer's disease. Nat Rev Neurosci 5: 347–360, 2004 [DOI] [PubMed] [Google Scholar]
- 87.Iadecola C, Davisson RL. Hypertension and cerebrovascular dysfunction. Cell Metab 7: 476–484, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Iadecola C, Nedergaard M. Glial regulation of the cerebral microvasculature. Nat Neurosci 10: 1369–1376, 2007 [DOI] [PubMed] [Google Scholar]
- 89.Ishikawa M, Kajimura M, Adachi T, Maruyama K, Makino N, Goda N, Yamaguchi T, Sekizuka E, Suematsu M. Carbon monoxide from heme oxygenase-2 Is a tonic regulator against NO-dependent vasodilatation in the adult rat cerebral microcirculation. Circ Res 97: e104–e114, 2005 [DOI] [PubMed] [Google Scholar]
- 90.Jaggar JH, Leffler CW, Cheranov SY, Tcheranova D, ES , Cheng X. Carbon monoxide dilates cerebral arterioles by enhancing the coupling of Ca2+ sparks to Ca2+-activated K+ channels. Circ Res 91: 610–617, 2002 [DOI] [PubMed] [Google Scholar]
- 91.Jakovcevic D, Harder DR. Role of astrocytes in matching blood flow to neuronal activity. Curr Top Dev Biol 79: 75–97, 2007 [DOI] [PubMed] [Google Scholar]
- 92.Kacem K, Lacombe P, Seylaz J, Bonvento G. Structural organization of the perivascular astrocyte endfeet and their relationship with the endothelial glucose transporter: a confocal microscopy study. Glia 23: 1–10, 1998 [PubMed] [Google Scholar]
- 93.Kim KJ, Filosa JA. Advanced in vitro approach to study neurovascular coupling mechanisms in the brain microcirculation. J Physiol 590: 1757–1770, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Kitaura H, Uozumi N, Tohmi M, Yamazaki M, Sakimura K, Kudoh M, Shimizu T, Shibuki K. Roles of nitric oxide as a vasodilator in neurovascular coupling of mouse somatosensory cortex. Neurosci Res 59: 160–171, 2007 [DOI] [PubMed] [Google Scholar]
- 95.Koehler RC, Roman RJ, Harder DR. Astrocytes and the regulation of cerebral blood flow. Trends Neurosci 32: 160–169, 2009 [DOI] [PubMed] [Google Scholar]
- 96.Kofuji P, Newman EA. Potassium buffering in the central nervous system. Neuroscience 129: 1045–1056, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Kohler R, Heyken WT, Heinau P, Schubert R, Si H, Kacik M, Busch C, Grgic I, Maier T, Hoyer J. Evidence for a functional role of endothelial transient receptor potential V4 in shear stress-induced vasodilatation. Arterioscler Thromb Vasc Biol 26: 1495–1502, 2006 [DOI] [PubMed] [Google Scholar]
- 98.Kohler R, Hoyer J. Role of TRPV4 in the mechanotransduction of shear stress in endothelial cells. In: TRP Ion Channel Function in Sensory Transduction and Cellular Signaling Cascades, edited by Liedtke WB, Heller S. Boca Raton FL: Taylor & Francis, 2007 [PubMed] [Google Scholar]
- 99.Kojima H, Hirata M, Kudo Y, Kikuchi K, Nagano T. Visualization of oxygen-concentration-dependent production of nitric oxide in rat hippocampal slices during aglycemia. J Neurochem 76: 1404–1410, 2001 [DOI] [PubMed] [Google Scholar]
- 100.Konno M, Shirakawa H, Iida S, Sakimoto S, Matsutani I, Miyake T, Kageyama K, Nakagawa T, Shibasaki K, Kaneko S. Stimulation of transient receptor potential vanilloid 4 channel suppresses abnormal activation of microglia induced by lipopolysaccharide. Glia 60: 761–770, 2012 [DOI] [PubMed] [Google Scholar]
- 101.Kusano Y, Echeverry G, Miekisiak G, Kulik TB, Aronhime SN, Chen JF, Winn HR. Role of adenosine A2 receptors in regulation of cerebral blood flow during induced hypotension. J Cereb Blood Flow Metab 30: 808–815, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Leffler CW, Balabanova L, Fedinec AL, Waters CM, Parfenova H. Mechanism of glutamate stimulation of CO production in cerebral microvessels. Am J Physiol Heart Circ Physiol 285: H74–H80, 2003 [DOI] [PubMed] [Google Scholar]
- 103.Leffler CW, Parfenova H, Jaggar JH, Wang R. Carbon monoxide and hydrogen sulfide: gaseous messengers in cerebrovascular circulation. J Appl Physiol 100: 1065–1076, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Leybaert L, Cabooter L, Braet K. Calcium signal communication between glial and vascular brain cells. Acta Neurol Belg 104: 51–56, 2004 [PubMed] [Google Scholar]
- 105.Li N, Sul JY, Haydon PG. A calcium-induced calcium influx factor, nitric oxide, modulates the refilling of calcium stores in astrocytes. J Neurosci 23: 10302–10310, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Lindauer U, Leithner C, Kaasch H, Rohrer B, Foddis M, Fuchtemeier M, Offenhauser N, Steinbrink J, Royl G, Kohl-Bareis M, Dirnagl U. Neurovascular coupling in rat brain operates independent of hemoglobin deoxygenation. J Cereb Blood Flow Metab 30: 757–768, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Lindauer U, Megow D, Matsuda H, Dirnagl U. Nitric oxide: a modulator, but not a mediator, of neurovascular coupling in rat somatosensory cortex. Am J Physiol Heart Circ Physiol 277: H799–H811, 1999 [DOI] [PubMed] [Google Scholar]
- 108.Liu X, Li C, Gebremedhin D, Hwang SH, Hammock BD, Falck JR, Roman RJ, Harder DR, Koehler RC. Epoxyeicosatrienoic acid-dependent cerebral vasodilation evoked by metabotropic glutamate receptor activation in vivo. Am J Physiol Heart Circ Physiol 301: H373–H381, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Longden TA, Dunn KM, Draheim HJ, Nelson MT, Weston AH, Edwards G. Intermediate-conductance calcium-activated potassium channels participate in neurovascular coupling. Br J Pharmacol 164: 922–933, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Lovick TA, Brown LA, Key BJ. Neuronal activity-related coupling in cortical arterioles: involvement of astrocyte-derived factors. Exp Physiol 90: 131–140, 2005 [DOI] [PubMed] [Google Scholar]
- 111.Lovick TA, Brown LA, Key BJ. Neurovascular relationships in hippocampal slices: physiological and anatomical studies of mechanisms underlying flow-metabolism coupling in intraparenchymal microvessels. Neuroscience 92: 47–60, 1999 [DOI] [PubMed] [Google Scholar]
- 112.Ma X, Qiu S, Luo J, Ma Y, Ngai CY, Shen B, Wong CO, Huang Y, Yao X. Functional role of vanilloid transient receptor potential 4-canonical transient receptor potential 1 complex in flow-induced Ca2+ influx. Arterioscler Thromb Vasc Biol 30: 851–858, 2010 [DOI] [PubMed] [Google Scholar]
- 113.Marrelli SP, O'Neil RG, Brown RC, Bryan RM., Jr PLA2 and TRPV4 channels regulate endothelial calcium in cerebral arteries. Am J Physiol Heart Circ Physiol 292: H1390–H1397, 2007 [DOI] [PubMed] [Google Scholar]
- 114.Martin E, Dahan D, Cardouat G, Gillibert-Duplantier J, Marthan R, Savineau JP, Ducret T. Involvement of TRPV1 and TRPV4 channels in migration of rat pulmonary arterial smooth muscle cells. Pflügers Arch 464: 261–272, 2012 [DOI] [PubMed] [Google Scholar]
- 115.Maschio MD, Beltramo R, De Stasi AM, Fellin T. Two-photon calcium imaging in the intact brain. Adv Exp Med Biol 740: 83–102, 2012 [DOI] [PubMed] [Google Scholar]
- 116.Mathiisen TM, Lehre KP, Danbolt NC, Ottersen OP. The perivascular astroglial sheath provides a complete covering of the brain microvessels: an electron microscopic 3D reconstruction. Glia 58: 1094–1103, 2010 [DOI] [PubMed] [Google Scholar]
- 117.McCarron JG, Halpern W. Potassium dilates rat cerebral arteries by two independent mechanisms. Am J Physiol Heart Circ Physiol 259: H902–H908, 1990 [DOI] [PubMed] [Google Scholar]
- 118.Metea MR, Kofuji P, Newman EA. Neurovascular coupling is not mediated by potassium siphoning from glial cells. J Neurosci 27: 2468–2471, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Metea MR, Newman EA. Glial cells dilate and constrict blood vessels: a mechanism of neurovascular coupling. J Neurosci 26: 2862–2870, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Mishra A, Hamid A, Newman EA. Oxygen modulation of neurovascular coupling in the retina. Proc Natl Acad Sci USA 108: 17827–17831, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Mulligan SJ, MacVicar BA. Calcium transients in astrocyte endfeet cause cerebrovascular constrictions. Nature 431: 195–199, 2004 [DOI] [PubMed] [Google Scholar]
- 122.Nedergaard M, Ransom B, Goldman SA. New roles for astrocytes: redefining the functional architecture of the brain. Trends Neurosci 26: 523–530, 2003 [DOI] [PubMed] [Google Scholar]
- 123.Ngai AC, Winn HR. Modulation of cerebral arteriolar diameter by intraluminal flow and pressure. Circ Res 77: 832–840, 1995 [DOI] [PubMed] [Google Scholar]
- 124.Nithipatikom K, Grall AJ, Holmes BB, Harder DR, Falck JR, Campbell WB. Liquid chromatographic-electrospray ionization-mass spectrometric analysis of cytochrome P450 metabolites of arachidonic acid. Anal Biochem 298: 327–336, 2001 [DOI] [PubMed] [Google Scholar]
- 125.O'Neil RG, Heller S. The mechanosensitive nature of TRPV channels. Pflügers Arch 451: 193–203, 2005 [DOI] [PubMed] [Google Scholar]
- 126.Paisansathan C, Xu H, Vetri F, Hernandez M, Pelligrino DA. Interactions between adenosine and K+ channel-related pathways in the coupling of somatosensory activation and pial arteriolar dilation. Am J Physiol Heart Circ Physiol 299: H2009–H2017, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Parfenova H, Tcheranova D, Basuroy S, Fedinec AL, Liu J, Leffler CW. Functional role of astrocyte glutamate receptors and carbon monoxide in cerebral vasodilation response to glutamate. Am J Physiol Heart Circ Physiol 302: H2257–H2266, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Peng X, Carhuapoma JR, Bhardwaj A, Alkayed NJ, Falck JR, Harder DR, Traystman RJ, Koehler RC. Suppression of cortical functional hyperemia to vibrissal stimulation in the rat by epoxygenase inhibitors. Am J Physiol Heart Circ Physiol 283: H2029–H2037, 2002 [DOI] [PubMed] [Google Scholar]
- 129.Peppiatt CM, Howarth C, Mobbs P, Attwell D. Bidirectional control of CNS capillary diameter by pericytes. Nature 443: 700–704, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Plant TD, Strotmann RT. RPV4. In: Handb Exp Pharmacol (2007/01/16 ed.), 2007, p. 189–205 [DOI] [PubMed] [Google Scholar]
- 131.Price DL, Ludwig JW, Mi H, Schwarz TL, Ellisman MH. Distribution of rSlo Ca2+-activated K+ channels in rat astrocyte perivascular endfeet. Brain Res 956: 183–193, 2002 [DOI] [PubMed] [Google Scholar]
- 132.Quaegebeur A, Lange C, Carmeliet P. The neurovascular link in health and disease: molecular mechanisms and therapeutic implications. Neuron 71: 406–424, 2011 [DOI] [PubMed] [Google Scholar]
- 133.Ramsauer M, Krause D, Dermietzel R. Angiogenesis of the blood-brain barrier in vitro and the function of cerebral pericytes. FASEB J 16: 1274–1276, 2002 [DOI] [PubMed] [Google Scholar]
- 134.Rancillac A, Rossier J, Guille M, Tong XK, Geoffroy H, Amatore C, Arbault S, Hamel E, Cauli B. Glutamatergic Control of Microvascular Tone by Distinct GABA Neurons in the Cerebellum. J Neurosci 26: 6997–7006, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Reid KH, Edmonds HL, Jr, Schurr A, Tseng MT, West CA. Pitfalls in the use of brain slices. Prog Neurobiol 31: 1–18, 1988 [DOI] [PubMed] [Google Scholar]
- 136.Rossier J. Wiring and plumbing in the brain. Front Hum Neurosci 3: 2, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Shi Y, Liu X, Gebremedhin D, Falck JR, Harder DR, Koehler RC. Interaction of mechanisms involving epoxyeicosatrienoic acids, adenosine receptors, and metabotropic glutamate receptors in neurovascular coupling in rat whisker barrel cortex. J Cereb Blood Flow Metab 28: 111–125, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Shigetomi E, Kracun S, Khakh BS. Monitoring astrocyte calcium microdomains with improved membrane targeted GCaMP reporters. Neuron Glia Biol 6: 183–191, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Shirakawa H, Nakagawa T, Kaneko S. [Pathophysiological roles of transient receptor potential channels in glial cells]. Yakugaku Zasshi 130: 281–287, 2010 [DOI] [PubMed] [Google Scholar]
- 140.Silver IA, Erecinska M. Extracellular glucose concentration in mammalian brain: continuous monitoring of changes during increased neuronal activity and upon limitation in oxygen supply in normo-, hypo-, and hyperglycemic animals. J Neurosci 14: 5068–5076, 1994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Simard M, Arcuino G, Takano T, Liu QS, Nedergaard M. Signaling at the gliovascular interface. J Neurosci 23: 9254–9262, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Smith PD, Brett SE, Luykenaar KD, Sandow SL, Marrelli SP, Vigmond EJ, Welsh DG. KIR channels function as electrical amplifiers in rat vascular smooth muscle. J Physiol 586: 1147–1160, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Sokoya EM, Burns AR, Setiawan CT, Coleman HA, Parkington HC, Tare M. Evidence for the involvement of myoendothelial gap junctions in EDHF-mediated relaxation in the rat middle cerebral artery. Am J Physiol Heart Circ Physiol 291: H385–H393, 2006 [DOI] [PubMed] [Google Scholar]
- 144.Sonkusare SK, Bonev AD, Ledoux J, Liedtke W, Kotlikoff MI, Heppner TJ, Hill-Eubanks DC, Nelson MT. Elementary Ca2+ signals through endothelial TRPV4 channels regulate vascular function. Science 336: 597–601, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Stevens B. Neuron-astrocyte signaling in the development and plasticity of neural circuits. Neurosignals 16: 278–288, 2008 [DOI] [PubMed] [Google Scholar]
- 146.Strotmann R, Harteneck C, Nunnenmacher K, Schultz G, Plant TD. OTRPC4, a nonselective cation channel that confers sensitivity to extracellular osmolarity. Nat Cell Biol 2: 695–702, 2000 [DOI] [PubMed] [Google Scholar]
- 147.Sun W, McConnell E, Pare JF, Xu Q, Chen M, Peng W, Lovatt D, Han X, Smith Y, Nedergaard M. Glutamate-dependent neuroglial calcium signaling differs between young and adult brain. Science 339: 197–200, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Takano T, Tian GF, Peng W, Lou N, Libionka W, Han X, Nedergaard M. Astrocyte-mediated control of cerebral blood flow. Nat Neurosci 9: 260–267, 2006 [DOI] [PubMed] [Google Scholar]
- 149.Theis M, Sohl G, Eiberger J, Willecke K. Emerging complexities in identity and function of glial connexins. Trends Neurosci 28: 188–195, 2005 [DOI] [PubMed] [Google Scholar]
- 150.Thrane AS, Rangroo Thrane V, Zeppenfeld D, Lou N, Xu Q, Nagelhus EA, Nedergaard M. General anesthesia selectively disrupts astrocyte calcium signaling in the awake mouse cortex. Proc Natl Acad Sci USA 109: 18974–18979, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Toth P, Rozsa B, Springo Z, Doczi T, Koller A. Isolated human and rat cerebral arteries constrict to increases in flow: role of 20-HETE and TP receptors. J Cereb Blood Flow Metab 31: 2096–2105, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Udosen IT, Jiang H, Hercule HC, Oyekan AO. Nitric oxide-epoxygenase interactions and arachidonate-induced dilation of rat renal microvessels. Am J Physiol Heart Circ Physiol 285: H2054–H2063, 2003 [DOI] [PubMed] [Google Scholar]
- 153.Vaucher E, Tong XK, Cholet N, Lantin S, Hamel E. GABA neurons provide a rich input to microvessels but not nitric oxide neurons in the rat cerebral cortex: a means for direct regulation of local cerebral blood flow. J Comp Neurol 421: 161–171, 2000 [PubMed] [Google Scholar]
- 154.Vetri F, Xu H, Mao L, Paisansathan C, Pelligrino DA. ATP hydrolysis pathways and their contributions to pial arteriolar dilation in rats. Am J Physiol Heart Circ Physiol 301: H1369–H1377, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Voets T, Prenen J, Vriens J, Watanabe H, Janssens A, Wissenbach U, Bodding M, Droogmans G, Nilius B. Molecular determinants of permeation through the cation channel TRPV4. J Biol Chem 277: 33704–33710, 2002 [DOI] [PubMed] [Google Scholar]
- 156.Vriens J, Owsianik G, Fisslthaler B, Suzuki M, Janssens A, Voets T, Morisseau C, Hammock BD, Fleming I, Busse R, Nilius B. Modulation of the Ca2 permeable cation channel TRPV4 by cytochrome P450 epoxygenases in vascular endothelium. Circ Res 97: 908–915, 2005 [DOI] [PubMed] [Google Scholar]
- 157.Vriens J, Watanabe H, Janssens A, Droogmans G, Voets T, Nilius B. Cell swelling, heat, and chemical agonists use distinct pathways for the activation of the cation channel TRPV4. Proc Natl Acad Sci USA 101: 396–401, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Wang F, Smith NA, Xu Q, Fujita T, Baba A, Matsuda T, Takano T, Bekar L, Nedergaard M. Astrocytes modulate neural network activity by Ca2+-dependent uptake of extracellular K+. Sci Signal 5: ra26, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Wang H, Hitron IM, Iadecola C, Pickel VM. Synaptic and vascular associations of neurons containing cyclooxygenase-2 and nitric oxide synthase in rat somatosensory cortex. Cereb Cortex 15: 1250–1260, 2005 [DOI] [PubMed] [Google Scholar]
- 160.Watanabe H, Davis JB, Smart D, Jerman JC, Smith GD, Hayes P, Vriens J, Cairns W, Wissenbach U, Prenen J, Flockerzi V, Droogmans G, Benham CD, Nilius B. Activation of TRPV4 channels (hVRL-2/mTRP12) by phorbol derivatives. J Biol Chem 277: 13569–13577, 2002 [DOI] [PubMed] [Google Scholar]
- 161.Watanabe H, Murakami M, Ohba T, Takahashi Y, Ito H. TRP channel and cardiovascular disease. Pharmacol Ther 118: 337–351, 2008 [DOI] [PubMed] [Google Scholar]
- 162.Watanabe H, Vriens J, Prenen J, Droogmans G, Voets T, Nilius B. Anandamide and arachidonic acid use epoxyeicosatrienoic acids to activate TRPV4 channels. Nature 424: 434–438, 2003 [DOI] [PubMed] [Google Scholar]
- 163.Watanabe H, Vriens J, Suh SH, Benham CD, Droogmans G, Nilius B. Heat-evoked activation of TRPV4 channels in a HEK293 cell expression system and in native mouse aorta endothelial cells. J Biol Chem 277: 47044–47051, 2002 [DOI] [PubMed] [Google Scholar]
- 164.Wiencken AE, Casagrande VA. Endothelial nitric oxide synthetase (eNOS) in astrocytes: another source of nitric oxide in neocortex. Glia 26: 280–290, 1999 [PubMed] [Google Scholar]
- 165.Willette RN, Bao W, Nerurkar S, Yue TL, Doe CP, Stankus G, Turner GH, Ju H, Thomas H, Fishman CE, Sulpizio A, Behm DJ, Hoffman S, Lin Z, Lozinskaya I, Casillas LN, Lin M, Trout RE, Votta BJ, Thorneloe K, Lashinger ES, Figueroa DJ, Marquis R, Xu X. Systemic activation of the transient receptor potential vanilloid subtype 4 channel causes endothelial failure and circulatory collapse: Part 2. J Pharmacol Exp Ther 326: 443–452, 2008 [DOI] [PubMed] [Google Scholar]
- 166.Xi Q, Tcheranova D, Basuroy S, Parfenova H, Jaggar JH, Leffler CW. Glutamate-induced calcium signals stimulate CO production in piglet astrocytes. Am J Physiol Heart Circ Physiol 301: H428–H433, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Xu HL, Mao L, Ye S, Paisansathan C, Vetri F, Pelligrino DA. Astrocytes are a key conduit for upstream signaling of vasodilation during cerebral cortical neuronal activation in vivo. Am J Physiol Heart Circ Physiol 294: H622–H632, 2008 [DOI] [PubMed] [Google Scholar]
- 168.Yang G, Chen G, Ebner TJ, Iadecola C. Nitric oxide is the predominant mediator of cerebellar hyperemia during somatosensory activation in rats. Am J Physiol Regul Integr Comp Physiol 277: R1760–R1770, 1999 [DOI] [PubMed] [Google Scholar]
- 169.Yang G, Huard JM, Beitz AJ, Ross ME, Iadecola C. Stellate neurons mediate functional hyperemia in the cerebellar molecular layer. J Neurosci 20: 6968–6973, 2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Yang XR, Lin AH, Hughes JM, Flavahan NA, Cao YN, Liedtke W, Sham JS. Upregulation of osmo-mechanosensitive TRPV4 channel facilitates chronic hypoxia-induced myogenic tone and pulmonary hypertension. Am J Physiol Lung Cell Mol Physiol 302: L555–L568, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Zhang DX, Mendoza SA, Bubolz AH, Mizuno A, Ge ZD, Li R, Warltier DC, Suzuki M, Gutterman DD. Transient receptor potential vanilloid type 4-deficient mice exhibit impaired endothelium-dependent relaxation induced by acetylcholine in vitro and in vivo. Hypertension 53: 532–538, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Zlokovic BV. Neurovascular pathways to neurodegeneration in Alzheimer's disease and other disorders. Nat Rev Neurosci 12: 723–738, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Zonta M, Angulo MC, Gobbo S, Rosengarten B, Hossmann KA, Pozzan T, Carmignoto G. Neuron-to-astrocyte signaling is central to the dynamic control of brain microcirculation. Nat Neurosci 6: 43–50, 2003 [DOI] [PubMed] [Google Scholar]
- 174.Zonta M, Sebelin A, Gobbo S, Fellin T, Pozzan T, Carmignoto G. Glutamate-mediated cytosolic calcium oscillations regulate a pulsatile prostaglandin release from cultured rat astrocytes. J Physiol 553: 407–414, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]