Abstract
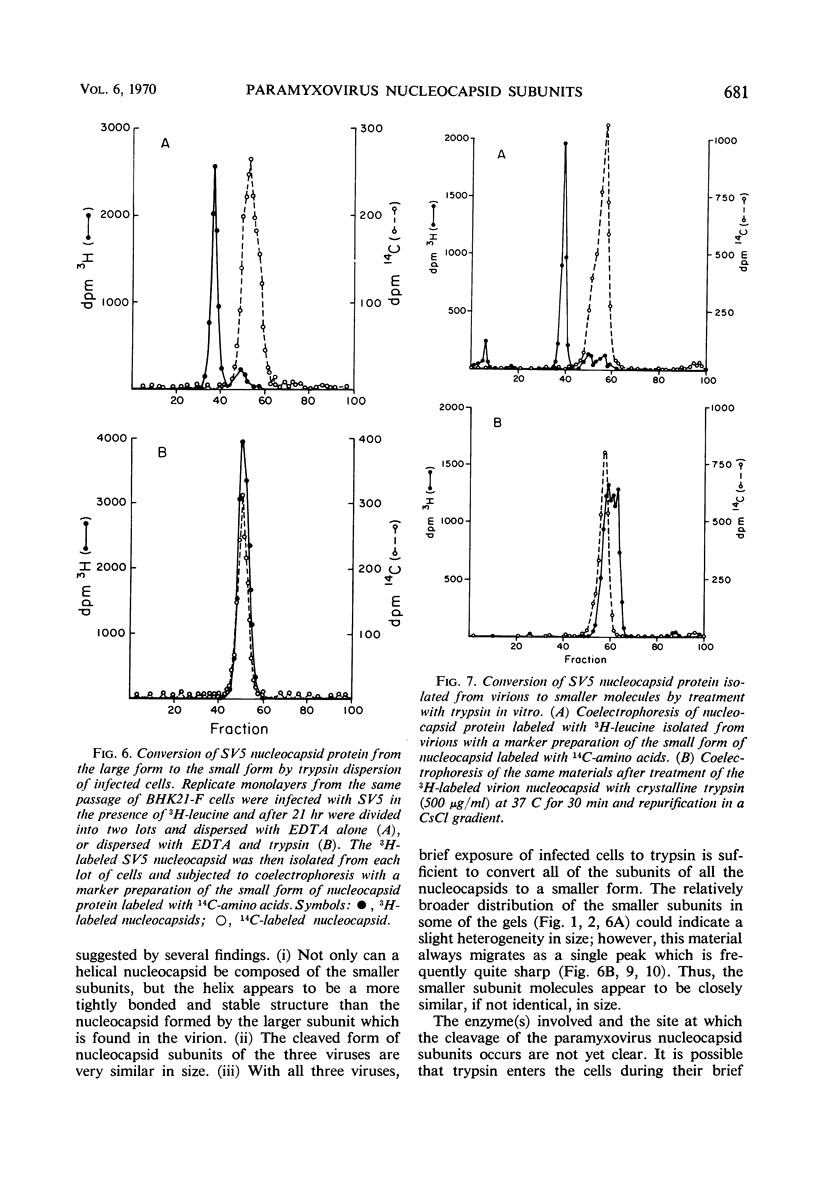
Helical nucleocapsids of each of the paramyxoviruses simian virus 5 (SV5), Newcastle disease virus (NDV), and Sendai virus have been isolated in two different forms. One form contains larger protein subunits and is obtained from mature virions or infected cells dispersed by ethylenediaminetetraacetic acid. The other form possesses smaller subunits and is obtained from infected cells dispersed by trypsin. The estimated molecular weights of the larger subunits in the three viruses are similar: SV5, 61,000; Sendai virus, 60,000; NDV, 56,000. The smaller nucleocapsid subunits are also very similar: SV5, 43,000; Sendai virus, 46,000; NDV, 47,000. The helical nucleocapsid composed of the smaller subunit appears to be less flexible and more stable than that formed by the larger subunit. There is suggestive evidence that conversion of the larger subunit to the smaller by proteolytic cleavage may occur intracellularly. The possibility that such a mechanism could be involved in the accumulation of nucleocapsid in cells persistently infected with paramyxoviruses is discussed.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BABLANIAN R., EGGERS H. J., TAMM I. STUDIES ON THE MECHANISM OF POLIOVIRUS-INDUCED CELL DAMAGE. I. THE RELATION BETWEEN POLIOVIRUS,-INDUCED METABOLIC AND MORPHOLOGICAL ALTERATIONS IN CULTURED CELLS. Virology. 1965 May;26:100–113. doi: 10.1016/0042-6822(65)90030-9. [DOI] [PubMed] [Google Scholar]
- Barry R. D., Bukrinskaya A. G. The nucleic acid of Sendai virus and ribonucleic acid synthesis in cells infected by Sendai virus. J Gen Virol. 1968 Jan;2(1):71–79. doi: 10.1099/0022-1317-2-1-71. [DOI] [PubMed] [Google Scholar]
- Bikel I., Duesberg P. H. Proteins of Newcastle disease virus and of the viral nucleocapsid. J Virol. 1969 Oct;4(4):388–393. doi: 10.1128/jvi.4.4.388-393.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blair C. D., Robinson W. S. Replication of Sendai virus. II. Steps in virus assembly. J Virol. 1970 May;5(5):639–650. doi: 10.1128/jvi.5.5.639-650.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CHOPPIN P. W. MULTIPLICATION OF A MYXOVIRUS (SV5) WITH MINIMAL CYTOPATHIC EFFECTS AND WITHOUT INTERFERENCE. Virology. 1964 Jun;23:224–233. doi: 10.1016/0042-6822(64)90286-7. [DOI] [PubMed] [Google Scholar]
- Caliguiri L. A., Klenk H. D., Choppin P. W. The proteins of the parainfluenza virus SV5. 1. Separation of virion polypeptides by polyacrylamide gel electrophoresis. Virology. 1969 Nov;39(3):460–466. doi: 10.1016/0042-6822(69)90094-4. [DOI] [PubMed] [Google Scholar]
- Choppin P. W. Replication of influenza virus in a continuous cell line: high yield of infective virus from cells inoculated at high multiplicity. Virology. 1969 Sep;39(1):130–134. doi: 10.1016/0042-6822(69)90354-7. [DOI] [PubMed] [Google Scholar]
- Compans R. W., Choppin P. W. Isolation and properties of the helical nucleocapsid of the parainfluenza virus SV5. Proc Natl Acad Sci U S A. 1967 Apr;57(4):949–956. doi: 10.1073/pnas.57.4.949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Compans R. W., Choppin P. W. The length of the helical nucleocapsid of Newcastle disease virus. Virology. 1967 Oct;33(2):344–346. doi: 10.1016/0042-6822(67)90153-5. [DOI] [PubMed] [Google Scholar]
- Compans R. W., Choppin P. W. The nucleic acid of the parainfluenza virus SV5. Virology. 1968 Jun;35(2):289–296. doi: 10.1016/0042-6822(68)90269-9. [DOI] [PubMed] [Google Scholar]
- Compans R. W., Holmes K. V., Dales S., Choppin P. W. An electron microscopic study of moderate and virulent virus-cell interactions of the parainfluenza virus SV5. Virology. 1966 Nov;30(3):411–426. doi: 10.1016/0042-6822(66)90119-x. [DOI] [PubMed] [Google Scholar]
- DULBECCO R., VOGT M. Plaque formation and isolation of pure lines with poliomyelitis viruses. J Exp Med. 1954 Feb;99(2):167–182. doi: 10.1084/jem.99.2.167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duc-Nguyen H., Rosenblum E. N. Immuno-electron microscopy of the morphogenesis of mumps virus. J Virol. 1967 Apr;1(2):415–429. doi: 10.1128/jvi.1.2.415-429.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duesberg P. H., Robinson W. S. Isolation of the nucleic acid of Newcastle disease virus (NDV). Proc Natl Acad Sci U S A. 1965 Sep;54(3):794–800. doi: 10.1073/pnas.54.3.794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans M. J., Kingsbury D. W. Separation of Newcastle disease virus proteins by polyacrylamide gel electrophoresis. Virology. 1969 Apr;37(4):597–604. doi: 10.1016/0042-6822(69)90277-3. [DOI] [PubMed] [Google Scholar]
- Finch J. T., Gibbs A. J. Observations on the structure of the nucleocapsids of some paramyxoviruses. J Gen Virol. 1970 Jan;6(1):141–150. doi: 10.1099/0022-1317-6-1-141. [DOI] [PubMed] [Google Scholar]
- Haslam E. A., Cheyne I. M., White D. O. The structural proteins of Newcastle disease virus. Virology. 1969 Sep;39(1):118–129. doi: 10.1016/0042-6822(69)90353-5. [DOI] [PubMed] [Google Scholar]
- Holmes K. V., Choppin P. W. On the role of the response of the cell membrane in determining virus virulence. Contrasting effects of the parainfluenza virus SV5 in two cell types. J Exp Med. 1966 Sep 1;124(3):501–520. doi: 10.1084/jem.124.3.501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hosaka Y. Isolation and structure of the nucleocapsid of HVJ. Virology. 1968 Jul;35(3):445–457. doi: 10.1016/0042-6822(68)90223-7. [DOI] [PubMed] [Google Scholar]
- Kingsbury D. W., Darlington R. W. Isolation and properties of Newcastle disease virus nucleocapsid. J Virol. 1968 Mar;2(3):248–255. doi: 10.1128/jvi.2.3.248-255.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakai T., Shand F. L., Howatson A. F. Development of measles virus in vitro. Virology. 1969 May;38(1):50–67. doi: 10.1016/0042-6822(69)90127-5. [DOI] [PubMed] [Google Scholar]
- Payne F. E., Baublis J. V., Itabashi H. H. Isolation of measles virus from cell cultures of brain from a patient with subacute sclerosing panencephalitis. N Engl J Med. 1969 Sep 11;281(11):585–589. doi: 10.1056/NEJM196909112811103. [DOI] [PubMed] [Google Scholar]
- Shapiro A. L., Viñuela E., Maizel J. V., Jr Molecular weight estimation of polypeptide chains by electrophoresis in SDS-polyacrylamide gels. Biochem Biophys Res Commun. 1967 Sep 7;28(5):815–820. doi: 10.1016/0006-291x(67)90391-9. [DOI] [PubMed] [Google Scholar]
- Tellez-Negal I., Harter D. H. Subacute sclerosing leukoencephalitis: ultrastructure of intranuclear and intracytoplasmic inclusions. Science. 1966 Nov 18;154(3751):899–901. doi: 10.1126/science.154.3751.899. [DOI] [PubMed] [Google Scholar]






