Abstract
It is now generally accepted that vitamin D deficiency is a worldwide health problem that affects not only musculoskeletal health but also a wide range of acute and chronic diseases. However, there remains cynicism about the lack of randomized controlled trials to support the association studies regarding the nonskeletal health benefits of vitamin D. This review was obtained by searching English-language studies in PubMed, MEDLINE, and the Cochrane Central Register of Controlled Trials and focuses on recent challenges regarding the definition of vitamin D deficiency and how to achieve optimal serum 25-hydroxyvitamin D concentrations from dietary sources, supplements, and sun exposure. The effect of vitamin D on fetal programming epigenetics and gene regulation could potentially explain why vitamin D has been reported to have such wide-ranging health benefits throughout life. There is potentially a great upside to increasing the vitamin D status of children and adults worldwide for improving musculoskeletal health and reducing the risk of chronic illnesses, including some cancers, autoimmune diseases, infectious diseases, type 2 diabetes mellitus, neurocognitive disorders, and mortality.
Vitamin D deficiency has been recognized as a pandemic with a myriad of health consequences.1,2 Low vitamin D status has been associated with an increased risk of type 1 diabetes mellitus, cardiovascular disease, certain cancers, cognitive decline, depression, pregnancy complications, autoimmunity, allergy, and even frailty.1–4 Low prenatal and neonatal vitamin D status may also increase susceptibility to schizophrenia, type 1 diabetes, and multiple sclerosis (MS) in later life via specific target organ effects, including the immune system, or through epigenetic modification.5
Despite the many important health benefits of vitamin D, there is controversy regarding the definition of vitamin D deficiency and what the vitamin D requirement should be.2,6,7 In addition, critical windows of exposure to adequate vitamin D levels during fetal maturation remain to be defined5,6 owing, in part, to the lack of well-designed controlled clinical trials with long-term follow-up.5–7
This review, obtained, in part, from searching English-language studies in PubMed, MEDLINE, and the Cochrane Central Register of Controlled Trials, focuses on recent challenges about how to achieve an optimal serum level of 25-hydroxyvitamin D (25[OH]D) from dietary sources, supplements, and sun exposure and evidence-based benefits for skeletal and nonskeletal health. Also, we explore fetal programming and epigenomic mechanisms that could potentially explain why vitamin D has been reported to have such wide-ranging health benefits throughout life.
VITAMIN D METABOLISM AND BIOLOGICAL FUNCTIONS
Vitamin D (D represents D2, D3, or both) is a secosterol produced endogenously in the skin from sun exposure or obtained from foods that naturally contain vitamin D, including cod liver oil and fatty fish (eg, salmon, mackerel, and tuna); UV-irradiated mushrooms; foods fortified with vitamin D; and supplements.2,7
During exposure to sunlight, 7-dehydrocholesterol (7-DHC) in the skin is converted to previtamin D3. The 7-DHC is present in all the layers of human skin.7–9 Approximately 65% of 7-DHC is found in the epidermis, and greater than 95% of the previtamin D3 that is produced is in the viable epidermis and, therefore, cannot be removed from the skin when it is washed.9 Once previtamin D3 is synthesized in the skin, it can undergo either a photoconversion to lumisterol, tachysterol, and 7-DHC or a heat-induced membrane-enhanced isomerization to vitamin D3 (Figure 1).7,8 The cutaneous production of previtamin D3 is regulated. Solar photoproducts (tachysterol and lumisterol) inactive on calcium metabolism are produced at times of prolonged exposure to solar UVB radiation, thus preventing sun-induced vitamin D intoxication.7,8 Vitamin D3 is also sensitive to solar irradiation and is, thereby, inactivated to suprasterol 1 and 2 and to 5,6-trans-vitamin D3.7 Cutaneous vitamin D3 production is influenced by skin pigmentation, sunscreen use, time of day, season, latitude, altitude, and air pollution.1,2,7,8 An increase in the zenith angle of the sun during winter and early morning and late afternoon results in a longer path for the solar UVB photons to travel through the ozone layer, which efficiently absorbs them. This is the explanation for why above and below approximately 33° latitude little if any vitamin D3 is made in the skin during winter.10,11 This is also the explanation for why—whether being at the equator and in the far northern and southern regions of the world in summer, where the sun shines almost 24 hours a day—vitamin D3 synthesis occurs only between approximately 10 AM and 3 PM.1,11 Similarly, in urban areas, such as Los Angeles, California, and Mexico City, Mexico, where nitrogen dioxide and ozone levels are high, few vitamin D3-producing UVB photons reach the people living in these cities.7,11 Similarly, because glass absorbs all UVB radiation, no vitamin D3 is produced in the skin when the skin is exposed to sunlight that passes through glass.
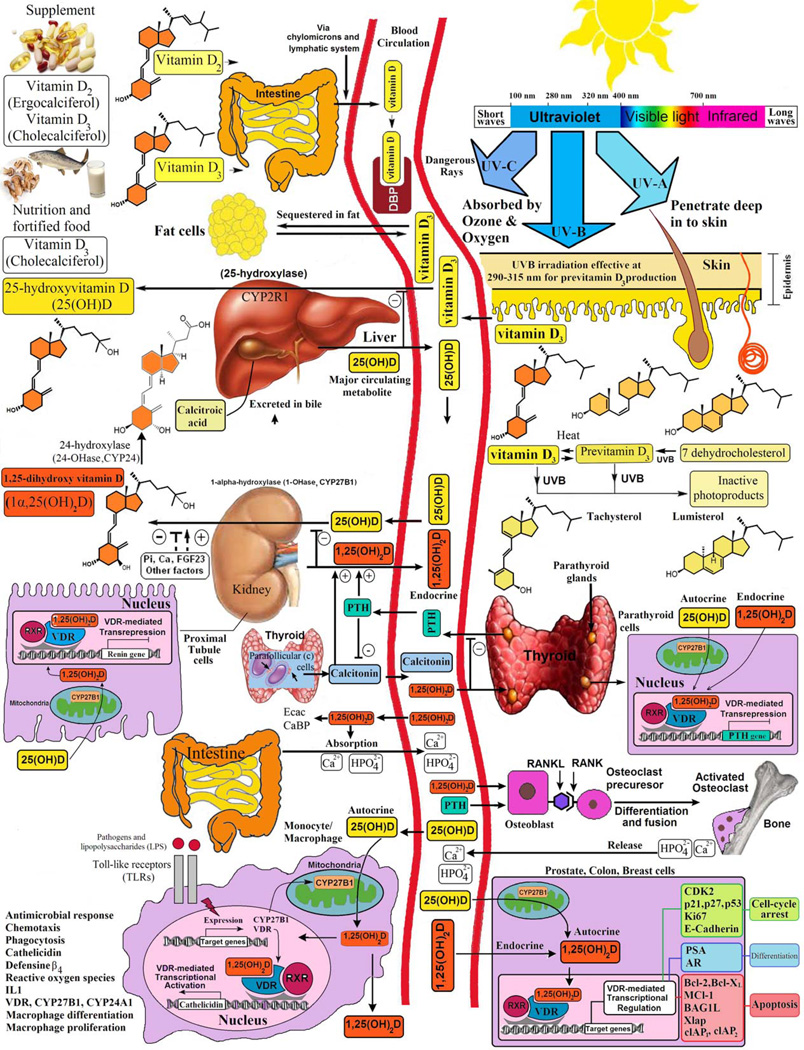
FIGURE 1.
Schematic representation of the synthesis and metabolism of vitamin D for skeletal and nonskeletal function. 1-OHase = 25-hydroxyvitamin D-1a–hydroxylase; 24-OHase = 25-hydroxyvitamin D-24-hydroxylase; 25(OH)D = 25-hydroxyvitamin D; 1,25(OH)2D = 1,25-dihydroxyvitamin D; CaBP = calciumbinding protein; DBP = vitamin D-binding protein; ECaC = epithelial calcium channel; FGF-23 = fibroblast growth factors; PTH = parathyroid hormone; RANK = receptor activator of the NF-kB; RANKL = receptor activator of the NF-kB ligand; RXR = retinoic acid receptor; TLR2/1 = Toll-like receptor 2/1; VDR = vitamin D receptor; vitamin D = vitamin D2 or vitamin D3. Copyright Holick 2013, reproduced with permission.
Once formed, vitamin D3 is ejected out of the keratinocyte plasma membrane and is drawn into the dermal capillary bed by the vitamin D binding protein (DBP).7,8 Vitamin D that is ingested is incorporated into chylomicrons, which are released into the lymphatic system, and enters the venous blood,2,7 where it binds to DBP and lipoproteins transported to the liver.1,2,7 Vitamin D2 and vitamin D3 are 25-hydroxylated by the liver vitamin D-25-hydroxylase (CYP2R1) to produce the major circulating vitamin D metabolite, 25(OH)D, which is used to determine a patient’s vitamin D status.1,2,7 This metabolite undergoes further hydroxylation by the 25(OH)D-1α-hydroxylase (CYP27B1) in the kidneys to form the secosteroid hormone 1α,25-dihydroxyvitamin D (1,25[OH]2D) (Figure 1).2,7,12 The 25(OH)D bound to DBP is filtered in the kidneys and is reabsorbed in the proximal renal tubules by megalin cubilin receptors.6,12 The renal 1α-hydroxylation is closely regulated, being enhanced by parathyroid hormone (PTH), hypocalcemia, and hypophosphatemia and inhibited by hyperphosphatemia, fibroblast growth factor-23, and 1,25(OH)2D itself.7,13,14
The 1,25(OH)2D performs many of its biologic functions by regulating gene transcription through a nuclear high-affinity vitamin D receptor (VDR).15,16 This active metabolite of vitamin D binds to the nuclear VDR, which binds retinoic acid X receptor to form a heterodimeric complex that binds to specific nucleotide sequences in the DNA known as vitamin D response elements. Once bound, a variety of transcription factors attach to this complex, resulting in either up- or down-regulation of the gene’s activity.2,7,17 There are estimated to be 200 to 2000 genes that have vitamin D response elements or that are influenced indirectly, possibly by epigenetics, to control a multitude of genes across the genome.2,16 A recent microarray study on the influence of vitamin D status and vitamin D3 supplementation on genome-wide expression in white blood cells before and after vitamin D3 supplementation found that an improved serum 25(OH)D concentration was associated with at least a 1.5-fold alteration in the expression of 291 genes.17 This study suggested that any improvement in vitamin D status will significantly affect the expression of genes that have a variety of biologic functions of more than 80 pathways linked to cancer, autoimmune disorders, and cardiovascular disease, which have been associated with vitamin D deficiency.17
One of the major physiologic functions of vitamin D is to maintain serum calcium and phosphorus levels in a healthy physiologic range to maintain a variety of metabolic functions, transcription regulation, and bone metabolism (Figure 1).2,7 The 1,25(OH)2D interacts with its VDR in the small intestine to increase the efficiency of intestinal calcium absorption from approximately 10% to 15% up to 30% to 40% and intestinal phosphorus absorption from approximately 60% to 80%.7 It also interacts with VDR in osteoblasts to stimulate a receptor activator of nuclear factor κB ligand, which, in turn, interacts with receptor activator of nuclear factor κB on immature preosteoclasts, stimulating them to become mature bone-resorbing osteoclasts (Figure 1).7,18 The mature osteoclast removes calcium and phosphorus from the bone to maintain blood calcium and phosphorus levels. In the kidneys, 1,25(OH)2D stimulates calcium reabsorption from the glomerular filtrate.2,7
The VDR is present in most tissues and cells in the body.1,2,7,19–25 Many of these organs and cells, including the brain, vascular smooth muscle, prostate, breast, and macrophages, not only have a VDR but also have the capacity to produce 1,25(OH)2D.1,2,7,19–25 This production probably depends on the availability of circulating 25(OH)D, indicating the biological importance of sufficient blood levels of this vitamin D metabolite.2,15,26
The estimated 2000 genes that are directly or indirectly regulated by 1,25(OH)2D2,7,17–24,26 have a wide range of proven biological actions, including inhibiting cellular proliferation and inducing terminal differentiation, inhibiting angiogenesis, stimulating insulin production, inducing apoptosis, inhibiting renin production, and stimulating macrophage cathelicidin production.1,2,7,16,17,26 In addition, 1,25(OH)2D stimulates its own destruction in the kidneys and in cells that have a VDR and responds to 1,25(OH)2D by enhancing expression of the 25(OH)D–24-hydroxylase (CYP24A1) to metabolize 25(OH)D and 1,25(OH)2D into water-soluble inactive forms that are exerted in the bile (Figure 1).1,7,19,27
VITAMIN D METABOLISM DURING PREGNANCY
Vitamin D metabolism is enhanced during pregnancy and lactation. The placenta is formed at 4 weeks of gestation.2,25 From this time to term, 25(OH)D is transferred across the placenta, and the fetal cord blood concentration of 25(OH)D is correlated with the mother’s concentration.27 However, the active metabolite 1,25(OH)2D does not readily cross the placenta.25,27 The fetal kidneys and the placenta provide the fetal circulation with 1,25(OH)2D by expressing CYP27B1 (Figure 2).28
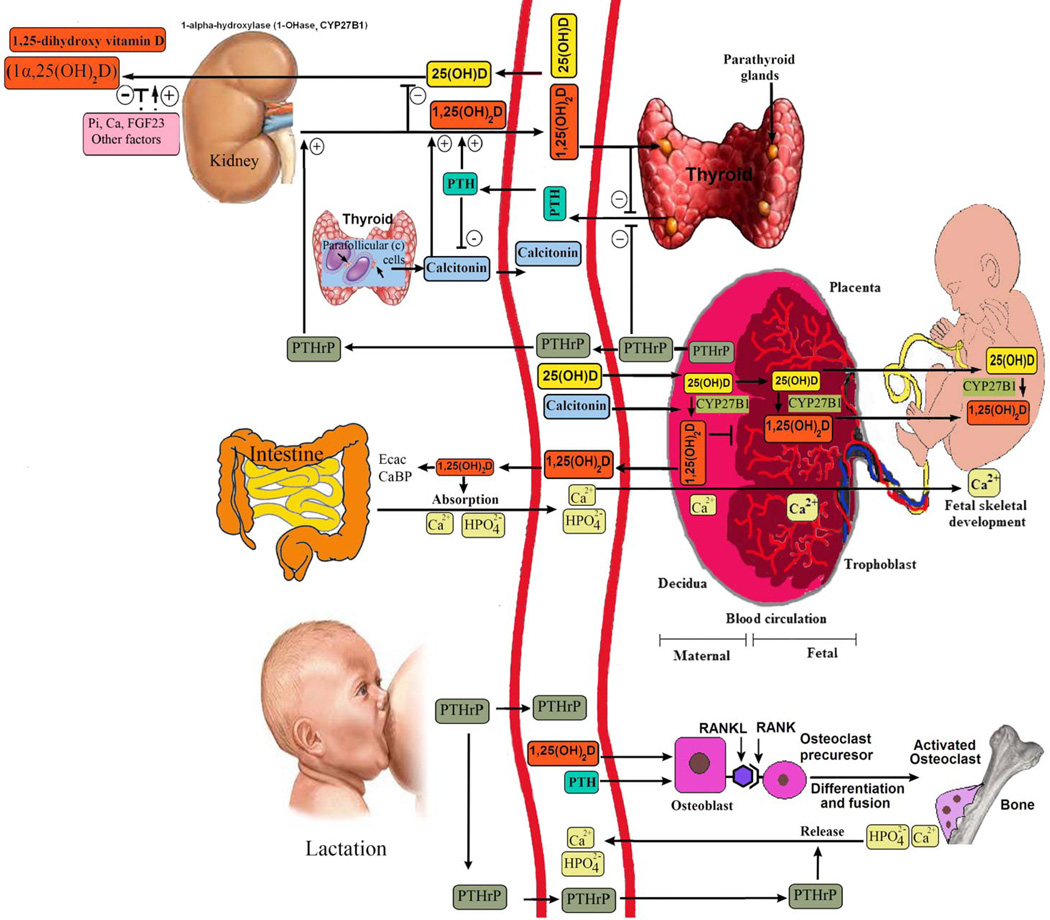
FIGURE 2.
Vitamin D metabolism during pregnancy and lactation. Maternal 25(OH)D is thought to freely cross the human placenta. The placenta expresses vitamin D receptors (VDR) and also produces 1-OHase to convert 25(OH)D to 1,25(OH)2D. 1,25(OH)2D does not readily cross the placenta, and fetal 1,25(OH)2D levels are normally lower than maternal serum levels. The low fetal concentrations of 1,25(OH)2D reflect the low fetal PTH and high phosphorus concentrations, which suppress renal 1-OHase. Although PTHrP is elevated in the fetal circulation, it appears to be less able to stimulate the renal 1-OHase than PTH. Total (free and bound) 1,25(OH)2D concentrations double or triple in the maternal circulation starting in the first trimester, but studies have only shown increased free concentrations during the third trimester. This increase is due to maternal synthesis by the renal 1-OHase. Vitamin D passes readily into breast milk, 25(OH)D passes very poorly, and 1,25(OH)2D does not appear to pass at all.2 1,25(OH)2D levels fall rapidly after pregnancy and are normal during lactation.7 Near-exclusive breastfeeding for 6 months leads, on average, to maternal calcium loss 4 times higher than in pregnancy. Phosphorus can rise above the normal range, probably because of accelerated resorption from the skeleton. PTHrP levels are higher than PTH concentrations in nonpregnant women and show some pulsatility in response to suckling. PTHrP (produced by the lactating breast) in combination with low estradiol concentrations appears to drive the main physiologic adaptation to meet the calcium demands of lactation. Suckling and prolactin both inhibit ovarian function and stimulate PTHrP. Together, PTHrP and low estradiol concentrations stimulate skeletal resorption. Renal calcium reabsorption rates increase, presumably due to PTHrP, which mimics the actions of PTH on the renal tubules. Copyright Holick 2013, reproduced with permission.
The maternal (decidual) and fetal placental (trophoblastic) components of the placenta have CYP27B1 activity; cultured human syncytiotrophoblasts and decidual cells synthesize 1,25(OH)2D3.27 The spatiotemporal organization of placental CYP27B1 and the VDR across gestation has also been characterized, confirming that the enzyme and receptor are localized to the maternal and fetal parts of the placenta.29 Serum levels of DBP increase 46% to 103% during pregnancy, suggesting that DBP may play a role in directing vitamin D metabolism and function during pregnancy.2,27,30 The DBP has a much higher binding affinity for 25(OH)D than for 1,25(OH)2D, and in kidney epithelial cells, DBP plays a pivotal role in conserving 25(OH)D by facilitating the recovery of 25(OH)D from the glomerular filtrate.31,32
Transplacental transfer of calcium to the fetus is also facilitated by expression of all the key mediators of vitamin D metabolism in the placenta. Hormones involved in fetal growth and that influence CYP27B1 activity include insulinlike growth factor 1 and human placental lactogen, PTH-related protein (PTHrP), estradiol, and prolactin.2,31,33,34 The PTHrP acts as a calciotropic hormone during fetal life and in lactation.35–37 The exact role of circulating PTHrP in pregnancy is unknown, but its rise may stimulate renal CYP27B1 and contribute to the increase in 1,25(OH)2D concentration and, indirectly, the suppression of PTH levels.27,35,37 The PTHrP arises from several sources, including the breast, myometrium, decidua, amnion, and fetal parathyroids.36 Several roles of PTHrP are postulated from animal studies, including fetal chondrocyte maturation, fetal calcium transfer, and stimulation of CYP27B1 activity.33,34,36 Furthermore, the carboxy terminal of PTHrP (osteostatin) may suppress osteoclastic activity and may have a possible bone protection role in the mother during pregnancy.32,35–37
Calcitonin, an important component of calcium homeostasis during pregnancy,38,39 is known to promote transcription of CYP27B140 and may, therefore, be a key determinant of placental vitamin D metabolism.41 Thus, PTHrP and calcitonin, as well as other factors, cause 1,25(OH)2D levels to increase, being 2-fold higher in serum of women in the third trimester of pregnancy than in nonpregnant or postpartum women.2,27 Normally, 1,25(OH)2D regulates its own metabolism via a feedback loop such that elevated concentrations induce the expression of CYP24A1, with concomitant down-regulation of CYP27B1.7,25,42 This process results in a reduction of 25(OH)D and 1,25(OH)2D levels.9,29,30 However, during pregnancy, this process becomes uncoupled, resulting in elevated maternal concentrations of circulating 1,25(OH)2D.27,43 The placental methylation of the CYP24A1 promoter reduces the capacity for CYP24A1 induction and down-regulates basal promoter activity and abolishes vitamin D–mediated feedback activation. This epigenetic decoupling of vitamin D feedback catabolism also plays an important role in enhancing 1,25(OH)2D bioavailability at the fetomaternal interface.44
VITAMIN D, PLACENTA DEVELOPMENT, FETAL PROGRAMMING, AND EPIGENETIC MODIFICATION
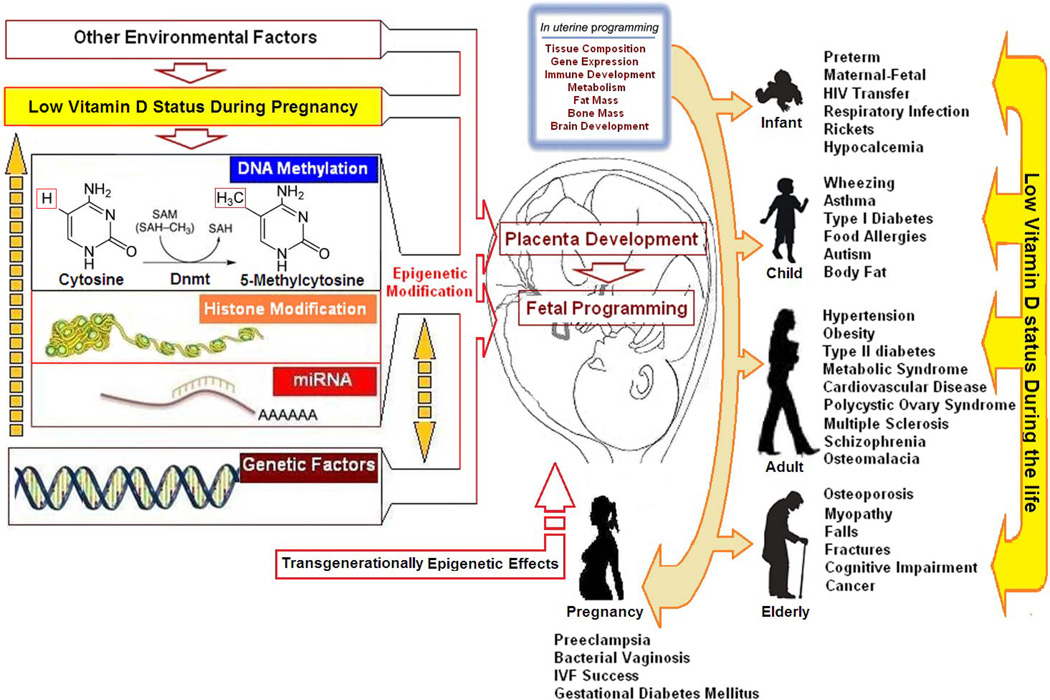
Epidemiologic evidence has suggested a link between fetal life events and susceptibility to disease in adult life.45–47 This paradigm, referred to as “fetal programming” or “developmental origins of health and disease,” may have a profound effect on public health strategies for the prevention of major illnesses.2,48 The role of vitamin D in implantation tolerance and placental development has been studied. The 1,25(OH)2D3 regulates key target genes associated with implantation, such as Homeobox A10 (HOXA10), whereas the potent immunosuppressive effects of 1,25(OH)2D3 suggest a role in placental development.49 Increasing expression of CYP27B1 and VDR in first-trimester human trophoblasts and deciduas50 may be related to the immunosuppressive effects of 1,25(OH)2D3 and may help improve implantation tolerance. Placental development plays a critical role in pregnancy health, and its link to maternal vitamin D deficiency may explain related adverse outcomes.5,45 In neonatal rats exposed prenatally to low maternal serum 25(OH)D levels, there was a general slowing of cardiac development, with significantly lower heart weights, decreased citrate synthase and 3-hydroxyacyl CoA dehydrogenase activity, and a 15% lower myofibrillar protein content.46 A 2-month-old human infant with dilated cardiomyopathy and severe vitamin D deficiency had dramatic improvement of her ejection fraction (17%–66%) after vitamin D supplementation.47 In addition, maternal vitamin D deficiency in rats stimulated nephrogenesis in offspring, with a 20% increase in nephron number but a decrease in renal corpuscle size observed between replete and deficient rats, despite there being no difference in body weight or kidney weight and volume.5,51 These findings support the role of vitamin D influencing fetal programming and placental development.
Epigenetic modification refers to heritable changes in gene expression that are not mediated by alterations in DNA sequence.52 This hypothesis, first articulated by Barker et al,53 postulated that in utero epigenetic fetal programming (as a result of environmental events during pregnancy) induced specific genes and genomic pathways that controlled fetal development and subsequent disease risk. The role of vitamin D in epigenetic modification and fetal programming could potentially explain why vitamin D has been reported to have such wide-ranging health benefits. Recent studies have suggested that epigenetic decoupling of vitamin D feedback catabolism plays an important role in maximizing 1,25(OH)D bioavailability at the fetomaternal interface.25,44 Modified expression of the genes encoding placental calcium transporters, by epigenetic regulation by 1,25(OH)2D, might represent the means whereby maternal vitamin D status could influence bone mineral accrual in the neonate.54,55 Vitamin D deficiency during pregnancy may, therefore, not only impair maternal skeletal preservation and fetal skeletal formation but also influence fetal “imprinting” that may affect chronic disease susceptibility soon after birth as well as later in life (Figure 3).15,56 Transgenerational hormonal imprinting caused by vitamin D treatment of newborn rats has been previously reported.57 A recent study reported that VDR binds to the ε germline gene promoter and exhibits transrepressive activity.58 Inhibition of IgE production by 1,25(OH)2D was mediated by its transrepressive activity through the VDR-corepressor complex, affecting chromatin compacting around the Iε region.58 Also, the associations of early-life sun exposure and germline variation in VDR and CYP24A1 with non-Hodgkin lymphoma risk was reported in a clinic-based case-control study.59
FIGURE 3.
Vitamin D, placenta development, fetal programming, and epigenetic modification. Copyright Holick 2013, reproduced with permission.
DEFINITION OF VITAMIN D DEFICIENCY
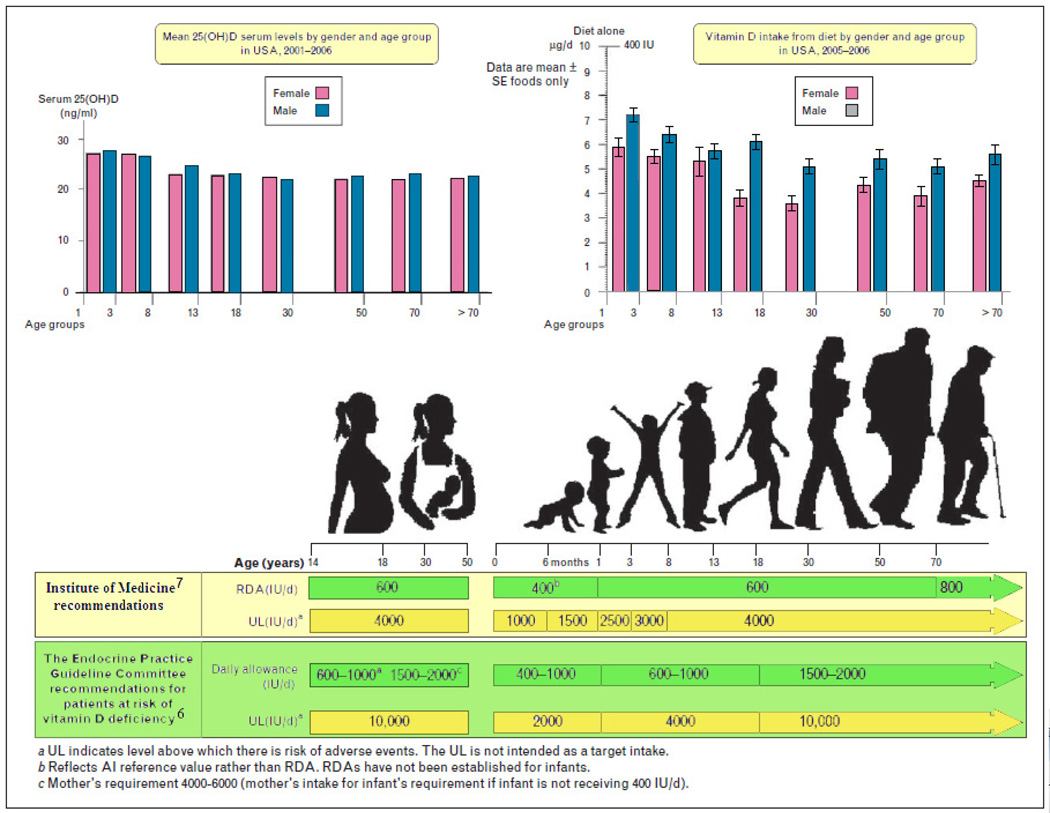
The blood level of 25(OH)D is the best method to determine vitamin D status. Although 1,25(OH)2D is the biologically active form, it provides no information about vitamin D status because it is often normal or even elevated in children and adults who are vitamin D deficient.7,15 Recently, the Institute of Medicine (IOM) and the Endocrine Society released separate guidelines for vitamin D requirements.60,61 The recommended dietary allowances (RDAs) of the IOM and the Endocrine Society guidelines for vitamin D intake are summarized in Figure 4.
FIGURE 4.
Vitamin D intakes recommended by the IOM and the Endocrine Practice Guidelines Committee. IOM, Institute of Medicine. Copyright Holick 2013, reproduced with permission.
The revised guidelines by the IOM stress that the daily requirements for vitamin D are generally met by most of the population and are appropriate to reach the “sufficient” level of 20 ng/mL (to convert to nmol/L, multiply by 2.496).62 The IOM guidelines used a population model to prevent vitamin D deficiency in 97.5% of the general population. Also, note that the IOM report focuses only on bone health (calcium absorption, bone mineral density, and osteomalacia/rickets) and found no evidence that a serum 25(OH)D concentration greater than 20 ng/mL had beneficial effects at a population level. However, considering the available evidence on skeletal and extraskeletal effects of vitamin D, the few negative studies, and the lack of toxicity potential of vitamin D supplementation at recommended doses, the US Endocrine Society, which used a medical model, recommended that serum 25(OH)D levels of 30 ng/mL should be attained to avoid other risks connected with an inadequate vitamin D status.6,63 Therefore, the Endocrine Society recommended that vitamin D deficiency be defined as a 25(OH)D level of 20 ng/mL or less, vitamin D insufficiency as 21 to 29 ng/mL, and vitamin D sufficiency as 30 ng/mL or greater for children and adults.61 It suggested that maintenance of a 25(OH)D level of 40 to 60 ng/mL is ideal (this takes into account assay variability) and that up to 100 ng/mL is safe.61
MUSCULOSKELETAL CONSEQUENCES OF VITAMIN D DEFICIENCY
According to current evidence from biochemical testing, observational studies, and randomized controlled trials (RCTs), serum 25(OH)D levels of at least 20 ng/mL are required for normalization of PTH levels, to minimize the risk of osteomalacia, and for optimal bone and muscle function, with many experts regarding 30 ng/mL as the threshold for optimal bone health.7,16,64–66 The skeletal consequences of 25(OH)D insufficiency include secondary hyperparathyroidism, increased bone turnover and bone loss, and increased risk of low-trauma fractures.7,15,61,64
The most common etiology of rickets, historically and presently, is vitamin D deficiency. Low maternal 25(OH)D levels were found to correlate with increased fetal distal femoral splaying, determined by ultrasonography measurements of femoral length and metaphyseal width.63,65 Children begin to manifest classical clinical signs of rickets between 6 months and 1.5 years that include rachitic rosary, widened epiphyseal plates at the end of long bones, and bowing deformities of the legs.66 A common early symptom in newborns is excessive sweating due to neuromuscular irritability,66 and a 25(OH)D level less than 20 ng/mL is common in children presenting with vague limb or back pain.
From a skeletal perspective for adults, evidence from RCTs suggests that vitamin D may be considered a threshold nutrient, with little bone benefit observed at levels of 25(OH)D above which PTH is normalized.62,67 A literature review of 70 studies generally found a threshold for a decline in serum PTH levels with increasing serum 25(OH)D levels, but there was no consistency in the threshold level of serum 25(OH)D, which varied from 20 to 50 ng/mL.68 A study of 4100 older adults (>60 years old) from the Third National Health and Nutrition Examination Survey (NHANES III) found that higher 25(OH)D levels were associated with better lower-extremity function.61 Much of the improvement occurred at 25(OH)D levels ranging from 9 to 16 ng/mL, but was continued to be seen up to 40 ng/mL.69 A systematic review revealed that supplemental vitamin D at daily doses of 800 to 1000 IU consistently had beneficial effects on muscle strength and balance.70 Several RCTs have reported positive effects of vitamin D supplementation on muscle function and fall prevention.71–73 Adequate calcium intake is imperative to gain optimal benefit from improving the vitamin D status in those with insufficient 25(OH)D levels.67 In contrast, in a study of 173 young Asian-Indian females revealed that after supplementation with vitamin D3 (60,000 IU/wk for 8 weeks followed by 60,000 IU for 2 weeks) and calcium (500 mg twice per day for 6 months), and despite significant improvement in serum 25(OH)D levels, there was no significant change in their skeletal muscle strength.74 Thus, age, baseline and final 25(OH)D concentrations, and whether and how much calcium supplementation was included in the clinical trial could affect outcome measures related to muscle performance and vitamin D status.
Proximal muscle weakness is a prominent clinical feature of vitamin D deficiency.7,60 The relative contributions of vitamin D and calcium to reducing fracture risk remain unclear75 because improving calcium intake is also associated with suppression of PTH levels independent of vitamin D status.67,76,77 A meta-analysis of data from RCTs found a dose-response relationship between a higher vitamin D dose and higher achieved serum 25(OH)D levels, with prevention of falls and fractures.73 The greatest benefit was observed at 700 to 1000 IU/d or a mean serum 25(OH)D concentration of 30 to 44 ng/mL.71,73 Similar results were reported in a more recent meta-analysis of pooled participant-level data from 11 double-blind RCTs of oral vitamin D supplementation, with or without calcium, compared with placebo or calcium alone in persons 65 years or older.78 Reduction in the risk of fracture occurred only at the highest vitamin D intake level (median, 800 IU/d; range, 792–2000 IU), with a 30% reduction in the risk of hip fracture and a 14% reduction in the risk of any nonvertebral fracture.77 This reduction was independent of the assigned treatment dose of vitamin D, age group, sex, type of dwelling, and study.78 Several previous meta-analyses have suggested that the dose of vitamin D is irrelevant when vitamin D is combined with calcium.79–82 In contrast, a pooled subgroup analysis of the 8 double-blind RCTs that used vitamin D combined with calcium indicated that with combined supplementation, the risk of fracture was reduced only at the highest actual intake level of vitamin D. These findings support that a 25(OH)D level of more than 24 ng/mL may be most beneficial for reducing the risk of fractures.78
With a similar tone and theme, a report from the US Preventive Services Task Force concluded that current evidence is insufficient to assess the balance of benefits and harms of combined vitamin D and calcium supplementation for the primary prevention of fractures in premenopausal women or in men. Furthermore, they concluded that there was insufficient evidence to assess the balance of benefits and harms of daily supplementation with greater than 400 IUs of vitamin D3 and greater than 1000 mg of calcium for primary prevention of fractures in noninstitutionalized postmenopausal women. They recommended against daily supplementation with 400 IU or less of vitamin D3 and 1000 mg or less of calcium for the primary prevention of fractures in noninstitutionalized postmenopausal women. They also stated that it was unclear whether higher doses of vitamin D and calcium are effective in preventing fractures in postmenopausal women, younger women, or men.83 The Task Force, however, concluded that vitamin D supplementation is effective in preventing falls in community-dwelling adults 65 years or older, which, in turn, reduces the risk of fracture. This could help explain the observation by the Women’s Health Initiative (WHI) that, in the subgroup of long-adherent women who took their calcium and vitamin D, there was a reduced risk of hip but not total fractures. Therefore, what is still unknown is whether adequate intake of calcium, especially from dietary sources, and maintenance of serum 25(OH)D levels of at least 20 ng/mL as recommended by the IOM62 or at least 30 ng/mL as recommended by the Endocrine Society60 throughout life will reduce the risk of fracture. Most evidence suggests that adequate calcium and vitamin D intake along with exercise during childhood will maximize bone mineral content that can be sustained in young and middle-aged adults as long as they also have a healthy lifestyle, adequate calcium intake, and a healthy vitamin D status.60–62,84–86 Accruing maximum bone mineral content during childhood, and maintaining peak bone mineral density in young and middle-aged adults, will likely reduce the risk of fracture later in life, when there is a disruption in bone remodeling due to menopause and aging.
Recent recommendations of Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO)87 for the optimal management of elderly and postmenopausal women regarding vitamin D supplementation have also indicated that patients with serum 25(OH)D levels less than 20 ng/mL have increased bone turnover, bone loss, and, possibly, mineralization defects compared with patients with serum 25(OH)D levels of 20 ng/mL or greater. Similar relationships have been reported for frailty, nonvertebral and hip fracture, and all-cause mortality, with poorer outcomes at less than 20 ng/mL.87 Thus, ESCEO recommended that 20 ng/mL be the minimal serum 25(OH)D concentration at the population level and in patients with osteoporosis to ensure optimal bone health. Also, in fragile elderly individuals who are at elevated risk for falls and fractures, ESCEO recommended a minimal serum 25(OH)D level of 30 ng/mL for the greatest effect on fracture.87 This coincides with the recommendation from the Endocrine Society6 and with the observation of Primel et al,79 who reported that of 675 presumed healthy adults (aged 20–90+ years) who died in an accident, 36% had evidence of osteomalacia. However, Primel et al79 observed no osteomalacia in those who had a 25(OH)D level greater than 30 ng/mL.
EVIDENCE-BASED SKELETAL AND NONSKELETAL HEALTH BENEFITS OF VITAMIN D
Observational studies have found a decreased risk of many disorders, including certain types of cancer, mental disorders, infection disease, cardiovascular disease, type 2 diabetes mellitus, and autoimmune disorders, associated with serum 25(OH)D levels greater than 28 to 32 ng/mL.7,60,67 It has, therefore, been argued that 25(OH)D levels should be in the range of 28 to 40 ng/mL to maximize these nonskeletal benefits.1,2,7,19,60,69
The results of some clinical trials provide evidence confirming the results of observational and association studies, whereas others do not. The Table summarizes the meta-analyses on vitamin D supplementation, comparing the beneficial and nonbeneficial effects of vitamin D supplementation in randomized trials for musculoskeletal and nonskeletal outcomes. The Table provides the foundation for clinical decision making for recommending vitamin D supplementation and identifies gaps in our knowledge that require additional RCTs to provide insights as to whether vitamin D supplementation has nonskeletal health benefits.
VITAMIN D AND NONSKELETAL HEALTH ASSOCIATIONS AND MECHANISMS
Cancers
Association studies have related higher serum levels of 25(OH)D to reduced incidence of many types of cancers. It has been hypothesized that the local conversion of 25(OH)D to 1,25(OH)2D in healthy cells in the colon, breast, and prostate can help prevent malignancy by inducing cellular maturation, inducing apoptosis, and inhibiting angiogenesis while enhancing the expression of genes including P21 and P27 to control cellular proliferation (Figure 1).1,2,7,19,30,80–82 Another vitamin D–regulated gene is CYP3A4, whose protein product detoxifies the bile acid lithocholic acid.114 Lithocholic acid is believed to damage the DNA of intestinal cells, and it may promote colon carcinogenesis. Stimulating the production of a detoxifying enzyme by 1,25(OH)2D could explain a protective role for improving vitamin D status against colon cancer.115 Because vitamin D regulates a gamut of physiologic processes, including immune modulation, resistance to oxidative stress, and modulation of other hormones, it is not surprising that low vitamin D status has been associated with increased risk of several cancers and cancer mortality.84–86,94–97 As the importance of noncoding RNAs has emerged, the ability of 1,25(OH)2D to regulate microRNAs (miRNAs) had been found in several cancer cell lines, patient tissues, and sera. In vitamin D3 intervention trials, significant differences in miRNAs were observed between treatment groups or between baseline and follow-up.116 In patient sera from population studies, specific miRNA differences were associated with serum levels of 25(OH)D. The findings thus far indicate that increasing vitamin D3 intake in patients and 1,25(OH)2D3 in vitro not only regulates specific miRNA(s) but also up-regulates global miRNA levels.116
Epidemiologic studies have suggested that adequate levels of 25(OH)D are critical for the prevention of various solid tumors, including prostate, breast, ovarian, and colon cancers.97,114,115,117–120 A meta-analysis for the US Preventive Services Task Force regarding vitamin D supplementation concluded that each 4-ng/mL increase in blood 25(OH)D levels was associated with a 6% reduced risk of colorectal cancer but not with statistically significant dose-response relationships for prostate and breast cancer.97 In a large prospective study of lethal prostate cancer (1260 cases vs 1331 controls), men with the highest quartile of plasma 25(OH)D levels had less than half the risk of lethal prostate cancer compared with men who were in the lowest quartile of plasma 25(OH)D levels.115 A meta-analysis including 1822 colon and 868 rectal cancers reported an inverse association between circulating 25(OH)D levels and colorectal cancer, with a stronger association for rectal cancer.97,121 Participants in the WHI who had a baseline 25(OH)D level less than 12 ng/mL and who took 400 IU of vitamin D3 and 1000 mg of calcium daily had a 253% increased risk of colorectal cancer compared with women who took the same amount of vitamin D3 and calcium for 7 years and had baseline serum 25(OH)D levels greater than 24 ng/mL.122
Although cross-sectional data have many limitations, the findings are hypothesis generating and can be used to develop protocols for RCTs.123,124 The findings from prospective case-control cohort studies in which blood collection occurred many years before diagnosis add another dimension to the evidence.118 The results of these studies generally support vitamin D supplementation in those with “low” vitamin D status. However, some have argued for caution before increasing 25(OH)D levels and associated dosing regimens beyond quantities clearly supported by RCTs and meta-analyses.7,95,103 There are now several observational studies reporting a U- or J-shaped association between some cancers and serum 25(OH)D and latitude or UVB radiation levels, in which those in the highest percentiles have an inverse risk compared with those in the lowest.118,125–127 Many RCTs that were evaluated for nonskeletal benefits of vitamin D had problems with a high incidence of nonadherence, misinterpretation of the original data, and use of doses of vitamin D below the 2010 IOM recommendations.62,123,124,128 A good example is the WHI.129 More than 50% of participants in the WHI admitted not taking their calcium and vitamin D daily, and blood concentrations of 25(OH)D were often not measured at baseline or at study end.124,129 Furthermore, the authors acknowledged that the 400 IU of vitamin D was inadequate to raise the blood level of 25(OH)D above 30 ng/mL, which most studies have suggested is required to reduce cancer risk and other nonskeletal acute and chronic diseases.7,26,61,127,128 A reanalysis of the WHI concluded that in 15,646 women (43%) who were not taking personal calcium or vitamin D supplements at randomization, the calcium and vitamin D intervention significantly decreased the risk of total, breast, and invasive breast cancers by 14% to 20% and the risk of colorectal cancer by 17%.123 In another RCT, a 60% reduction in all cancers was observed in postmenopausal women who ingested 1100 IU of vitamin D3 and 1500 mg of elemental calcium daily for 4 years.130 There is conflicting evidence about vitamin D’s relationship with risk of pancreatic cancer. A study of more than 120,000 men and women from the Health Professionals Follow-up Study and the Nurses’ Health Study found that participants with higher dietary intake of vitamin D had a progressively lower risk of pancreatic cancer compared with those who had the lowest intake.131 In a study of men and women enrolled in the Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial,132 no association between serum 25(OH)D levels and pancreatic cancer risk was observed. A pooled analysis of 5 nested case-control studies reported an inverse association between plasma levels of 25(OH)D and the subsequent risk of pancreatic cancer.133 Compared with individuals with 25(OH)D levels less than 20 ng/mL, those with 25(OH)D levels greater than or equal to 20 ng/mL experienced an approximately 30% lower risk of pancreatic cancer.
Grant120,134,135 reported that more than 13 cancers were reduced by adequate exposure to solar UVB radiation. He calculated that in a span of 24 years (1970–1994), 566,400 Americans died of cancer because of inadequate exposure to solar UVB radiation.120,134–136 He also estimated that 50,000 to 63,000 Americans and 19,000 to 25,000 British citizens in the United Kingdom die prematurely of cancer each year due to vitamin D deficiency.135,136 A large collaborative effort analyzed data from 10 prospective cohort studies to examine whether serum 25(OH)D levels were associated with 7 rare cancers.137 The National Cancer Institute Cohort Consortium Vitamin D Pooling Project of Rarer Cancers included information on serum 25(OH)D levels and the incidence of rare cancers in a subset of more than 12,000 men and women. The researchers matched participants on date and season of blood collection and used other statistical techniques to adjust for seasonal variation in serum 25(OH)D levels. When the data from the different studies were pooled, there was no overall association between vitamin D status and risk of non-Hodgkin lymphoma or cancers of the endometrium, esophagus, stomach, kidney, or ovary.137 In contrast, a recent review of ecological studies associating solar UVB exposure–vitamin D and cancers found strong inverse correlations with solar UVB irradiance for 15 types of neoplasms: bladder, breast, cervical, colon, endometrial, esophageal, gastric, lung, ovarian, pancreatic, rectal, renal, and vulvar cancers and additionally Hodgkin and non-Hodgkin lymphoma.138 Weaker evidence was observed for 9 other types of cancer: brain, gallbladder, laryngeal, oral/pharyngeal, prostate, and thyroid cancers; leukemia; melanoma; and myeloma.138 Although there was compelling evidence for the association between vitamin D intake and cancer risk, a meta-analysis by Buttigliero et al98 found no effect of vitamin D supplementation on survival in patients with cancer.
Mortality
Vitamin D deficiency is associated with an increased risk of total mortality.139 Most, but not all, studies documented increased mortality rates in patients with low 25(OH)D concentrations. IA study of 247,574 individuals from the primary care sector, a reverse J-shaped relation was reported between serum level of 25(OH)D and all-cause mortality, with the lowest mortality rate at 20 to 24 ng/mL.126 This finding underscores the importance of not only including the very low (4 ng/mL) but also the higher (56 ng/mL) levels of 25(OH)D in the analysis.126 It also raises several questions. How do patients who had a 25(OH)D level greater than 50 ng/mL attain such as high level, which is usually observed only in Africans living outdoors?128,140 These people are likely taking megadoses of vitamin D and possibly other supplements or are being treated for vitamin D deficiency. In some instances, studies are misrepresented or misinterpreted. For example, the IOM reported in their overview that there was evidence of increased mortality for those with a 25(OH)D level greater than 30 ng/mL. However, one of the studies used to support this IOM conclusion actually stated that mortality rates were reduced until the blood level of 25(OH)D reached 50 ng/mL and that mortality rates possibly increased only in women who had 25(OH)D levels greater than 50 ng/mL.141 When these J curves are plotted, we are not informed what percentage of study participants had a 25(OH)D level greater than 50 ng/mL. By some estimates, less than 10% of patients have a 25(OH)D level greater than 50 ng/mL, and in a recent meta-analysis,126 only 1.5% of participants had a 25(OH)D level greater than 50 ng/mL. This raises questions about the validity of the so-called J–U curve analyses.
A meta-analysis of prospective cohort studies including 5562 deaths of 62,548 participants suggested a nonlinear decrease in mortality risk as circulating 25(OH)D concentration increases, with optimal outcomes occurring at concentrations of approximately 30 to 35 ng/mL.142 In a similar meta-analysis, vitamin D intake and blood 25(OH)D levels were inversely associated with risk of colorectal cancer, and a 10-ng/mL increase in blood 25(OH)D levels conferred a risk rate (RR) of 0.74.143 A meta-analysis of prospective studies of 6853 patients with chronic kidney disease found that the mortality risk decreased by 14% per 10-ng/mL increase in 25(OH)D levels.144 The major cause of mortality was cardiovascular disease.
In a recent meta-analysis with 70,528 randomized participants (86.8% females) with a median age of 70 years, vitamin D supplementation with or without calcium reduced mortality by 7%. However, vitamin D supplementation alone did not affect mortality, but risk of death was reduced if vitamin D was given with calcium.145 The Ludwigshafen Risk and Cardiovascular Health study is a cohort study of patients referred for coronary angiography between 1997 and 2000, from which 1801 with the metabolic syndrome were investigated. Mortality was tracked for a median of 7.7 years.146 Multivariable survival analysis was used to estimate the association between serum 25(OH)D levels and mortality. After full adjustment, including the metabolic syndrome components, patients with optimal 25(OH)D levels had a substantial reduction in all-cause (hazard ratio [HR], 0.25; 95% CI, 0.13–0.46) and cardiovascular disease (HR, 0.33; 95% CI, 0.16–0.66) mortality rates compared with those with severe vitamin D deficiency. For specific cardiovascular disease mortality, there was a strong reduction in sudden death (HR, 0.15; 95% CI, 0.04–0.63) and congestive heart failure (HR, 0.24; 95% CI, 0.06–1.04) but in for myocardial infarction. The reduction in the mortality rate was dose dependent for each of these causes.146 Consistent with the beneficial effect of vitamin D on risk of mortality, a meta-analysis of 50 randomized trials by Bjelakovic et al99 found a positive effect of vitamin D3 on mortality.
Cardiovascular Disorders and Type 2 Diabetes Mellitus
Observational studies in humans found that 25(OH)D and 1,25(OH)2D levels are inversely related to coronary artery calcifications147,148 and are lower in patients with myocardial infarction.149 An in vitro study suggested that low 25(OH)D levels influence the activity/expression of macrophages and lymphocytes in atherosclerotic plaques, thus promoting chronic inflammation in the artery wall.150 Additionally, 1,25(OH)2D3 inhibited foam cell formation and promoted angiogenesis in endothelial colony-forming cells in vitro, possibly due to an increase in vascular endothelial growth factor expression and pro–matrix metalloproteinase-2 activity.4,151 A short course of treatment with vitamin D (4000 IU for 5 days) effectively attenuated the increase in circulating levels of inflammatory cytokines after an acute coronary event.150 These findings provide support for the anti-inflammatory effects of vitamin D on the vascular system and suggest mechanisms that mediate some of its cardioprotective properties.4,150 In addition, low 25(OH)D concentrations result in elevations in PTH levels, which have been linked to insulin resistance and significant increases in the serum levels of many acute phase proteins.149
Wang et al152 studied 1739 Framingham Offspring Study participants (mean age, 59 years; 55% women; all of white race) without previous cardiovascular disease. During mean follow-up of 5.4 years, 120 individuals developed a first cardiovascular event. Individuals with 25(OH)D levels less than 15 ng/mL had a multivariable-adjusted HR of 1.62 for incident cardiovascular events compared with those with 25(OH)D levels of 15 ng/mL or greater. This effect was evident in participants with hypertension (HR, 2.13; 95% CI, 1.30–3.48) but not in those without hypertension.152 The observational studies indicated that a serum 25(OH)D level less than 30 ng/mL was strongly associated with hypertension and metabolic syndrome.153 This effect is thought to be partly mediated through regulation of the renin-angiotensin-aldosterone axis.154 The Intermountain Heart Collaborative Study Group prospectively analyzed a large electronic medical records database that contained 41,504 patient records. Serum 25(OH)D levels less than 30 ng/mL were associated with highly significant increases in the prevalence of diabetes, hypertension, hyperlipidemia, and peripheral vascular disease. Serum 25(OH)D levels were also highly associated with coronary artery disease, myocardial infarction, heart failure, and stroke and with incident death, heart failure, coronary artery disease/myocardial infarction, stroke, and their composite.153 Black normotensive children who received 2000 IU/d of vitamin D3 were compared with those who received 400 IU/d for 16 weeks in an RCT. Teenagers who received 400 IU/d of vitamin D3 increased their mean ± SD plasma levels of 25(OH)D from 13.6±4.2 to 23.9±7.2 ng/mL and showed no reduction in arterial stiffness. In contrast, teenagers who received 2000 IU/d of vitamin D3 increased their mean ± SD plasma levels of 25(OH)D from 13.2±3.4 to 34.2±12.1 ng/mL and significantly lowered their arterial wall stiffness.155 This is supported by the observation that serum 25(OH)D levels less than 30 ng/mL were strongly associated with hypertension, elevated blood glucose, and metabolic syndrome in adolescents.156
Children who were vitamin D deficient or insufficient had a 2.5-fold higher risk of an elevated blood glucose level, a 2.4-fold increased risk of elevated blood pressure, and a 4-fold increased risk of metabolic syndrome, a prelude to type 2 diabetes.156
A meta-analysis of 11 prospective studies involving 3612 cases and 55,713 noncase participants provided the largest and most comprehensive assessment thus far of the association between circulating 25(OH)D levels and type 2 diabetes. It suggested a strong inverse association between serum 25(OH)D concentration and incidence of type 2 diabetes. The combined RR of 0.59 suggested that the risk of future diabetes may be reduced by 41% (95% CI, 33%–48%) by having a serum 25(OH)D level greater than 32 ng/mL compared with a serum 25(OH)D level less than 19.5 ng/mL at baseline.157 The MIDSPAN family study was a prospective study of 1040 men and 1298 women from the West of Scotland recruited in 1996 and followed up for a median of 14.4 years.158 Plasma levels of 25OHD less than 15 ng/mL were not associated with a risk of cardiovascular disorders in this cohort with very low 25(OH)D levels. The median plasma 25(OH)D level was 18.6 ng/mL, and the median vitamin D intake was 3.2 µg/d (128 IU/d). However, there was some evidence that a 25(OH)D level less than 15 ng/mL was associated with all-cause mortality.158 There was an association between 25(OH)D levels and incidence of type 2 diabetes, but there was no evidence in this study of a beneficial effect of vitamin D supplementation on type 2 diabetes outcomes.106 A meta-analysis of 15 trials by George et al106 did not find sufficient evidence to recommend vitamin D supplementation for improving glycemia or insulin resistance in patients with diabetes, normal fasting glucose levels, or impaired glucose tolerance. Similarly, Wang et al109 and Pittas et al110 concluded in their meta-analyses that evidence from limited data suggested that vitamin D supplements at moderate to high doses may reduce the risk of cardiovascular disease,109 but most studies that used lower doses found no clinically meaningful effect.110
Autoimmune Diseases
Vitamin D has been defined as a natural immune modulator. Epidemiologic, genetic, and basic science studies indicate a potential role of vitamin D in the pathogenesis of certain systemic and organ-specific autoimmune diseases, such as type 1 diabetes mellitus, MS, rheumatoid arthritis (RA), and Crohn disease (CD).159 Vitamin D’s effects on the innate immune system are predominantly through the toll-like receptors and on the adaptive immune system through T-cell differentiation, particularly the type 17 T helper cell (TH17) response. Because TH17 cells are critical in the pathogenesis of RA, this has led to an interest in the effects of vitamin D deficiency in RA.160 Vitamin D inhibits immune reactions in general, but it enhances the transcription of endogenous antibiotics, such as cathelicidin and defensins.26,161 Vitamin D suppresses autoimmune disease pathology by regulating the differentiation and activity of CD4+ T cells, resulting in a more balanced TH1/TH2 response that favors less development of self-reactive T cells and autoimmunity.162 The TH1-dependent autoimmune diseases, including MS, type 1 diabetes, CD, and RA, are also inhibited by 1,25(OH)2D3 owing to inhibition of antigen presentation, reduced polarization of TH0 cells to TH1 cells, and reduced production of cytokines from the latter cells.161 The 1,25(OH)2D3 down-regulated the proinflammatory cytokine (interleukin 1-β, interleukin 6, and tumor necrosis factor) production in human activated macrophages by significantly decreasing the aromatase activity, especially in the presence of an estrogenic milieu, such as in RA synovial tissue.163 A prospective cohort study of 29,368 women aged 55 to 69 years without a history of RA found an inverse association between vitamin D intake and RA after 11 years of follow-up.164 There was a 34% reduction in the development of RA with greater vitamin D intake. Women using a multivitamin with 400 IU of vitamin D reduced their risk of RA by 40%.164 Use of a high-dose vitamin D3 analogue resulted in improvement of symptoms of RA in 89% of patients, with 45% of patients entertaining a complete remission.165 Recent evidence has suggested a significant inverse relationship between serum 25(OH)D levels and visual analog scale scores in patients with RA.166 Very low serum 25(OH)D levels (≤6 ng/mL) were characterized by patients being positive for rheumatoid factor, a high percentage of patients with very high disease activity, and a high percentage of patients requiring treatment with at least 3 disease-modifying antirheumatic drugs.167
There is a large body of evidence linking a lack of vitamin D early in life to the development of type 1 diabetes.168 Vitamin D supplementation during infancy was reported to confer partial protection against β-cell autoimmunity.169 There is consistent evidence from observational studies for potential long-term programming effects of vitamin D supplementation on immunologic diseases, such as type 1 diabetes, MS, asthma, and allergic diseases.5 There was a 63% decreased risk of islet cell antibodies in offspring with a single standard deviation (156 IU) increase in recalled maternal dietary vitamin D intake during pregnancy.5 Similarly, higher maternal cod liver oil (a source of vitamin D) intake during pregnancy was associated with a decreased risk of type 1 diabetes in offspring, and fetal exposure to vitamin D deficiency was linked to a higher metabolic and cardiovascular disease risk in adult life.170
A Finnish study (10,366 children) found that children who regularly took the recommended dose of vitamin D (2000 IU/d) had a rate ratio of 0.22 (95% CI, 0.05–0.89) compared with those who regularly received less than the recommended amount.171 The 1,25(OH)2D3 has been reported in animal models and in cultured cells to improve insulin production, modulate T- and β-cell activity, enhance phagocytic killing activity, improve vascular smooth muscle resistance, and reduce the risk of autoimmune diseases.7,97,145 In contrast, in healthy youth (aged 8–18 years), plasma 25(OH)D concentrations had no independent relationship with parameters of glucose homeostasis and in vivo insulin sensitivity and β-cell function relative to insulin sensitivity.172 It remains to be determined whether in youth with dysglycemia the relationships are different and whether vitamin D optimization enhances insulin sensitivity and β-cell function.172
Evidence continues to accumulate supporting a protective role for vitamin D in MS risk and progression. Notable recent findings are that high 25(OH)D levels at the time of a first demyelinating event predicts a lower MS risk and a decreased risk of MS in offspring whose mothers had high 25(OH)D levels.173 An American study of more than 187,000 women followed up for 10 to 20 years reported promising results with women taking at least 400 IU of supplemental vitamin D daily. The risk of MS was decreased by 41%.174 An epigenetic study in lymphoblastoid cell lines reported relevant insights into how vitamin D may influence the immune system and the risk of MS through VDR interactions with the chromatin state inside MS-associated genomic regions.175 Higher 25(OH)D levels were associated with decreased exacerbation risk in relapsing-remitting MS.175 However, the literature is limited by small study sizes, heterogeneity of dosing, form of vitamin D tested, and clinical outcome measures.171 Whether vitamin D3 immunomodulatory effects can be translated into clinical benefits in patients with MS is still a matter of debate.176 High doses of vitamin D3 (up to 280,000 IU/wk for 6 weeks) have been used safely in patients with MS.177,178 Levels of 25(OH)D rose to a mean of 154 ng/mL without causing hypercalcemia. The progression and activity of MS were not affected in this study, but the number of gadolinium-enhancing lesions per patient assessed by nuclear magnetic brain scan was significantly reduced.177 A trial using high-dose vitamin D2 to achieve 25(OH)D levels of 52 to 78 ng/mL did not reduce magnetic resonance imaging lesions in relapsing-remitting MS.178 In a trial using escalating doses up to 40,000 IU/d of vitamin D3 for 28 weeks followed by 10,000 IU/d for 12 weeks, there were no significant adverse events, and there seemed to be significantly less progression of disability in the treatment group.179
A chromatin immunoprecipitation sequencing–defined genome-wide map of VDR binding reported that there were 2776 “binding sites” on the human genome with at least 229 genes associated with type 1 diabetes and CD.180 A clinical trial in patients with CD in remission using 1200 IU of vitamin D3 daily increased mean ± SD serum 25(OH)D levels from 27.6±12.4 to 38.4±10.8 ng/mL after 3 months.181 The relapse rate was numerically lower in patients treated with vitamin D3 (6 of 46 or 13%) than in patients treated with placebo (14 of 48 or 29%), although this did not quite reach significance (P=.06). Monocyte-derived dendritic cells (DCs) from 20 patients with CD were cultured with either 25(OH)D3 or 1,25(OH)2D3 and were matured with lipopolysaccharide (LPS).182 After stimulation with 25(OH)D3, DCs from patients with CD displayed a reduced response to LPS with a diminished capability to activate T cells compared with DCs stimulated with LPS alone. Compared with LPS alone, both metabolites of vitamin D3 reduced the ability of DCs to activate lymphocytes. These data indicate that intrinsic activation of 25(OH)D3 to 1,25(OH)2D3 occurs in DCs from patients with CD and provides evidence that higher serum 25(OH)D3 levels can potentially modulate DC function in CD.182 Although several studies reported the immunomodulatory effects of vitamin D on biological functions and developing processes of autoimmune diseases, there is no strong evidence for recommending vitamin D supplementation to prevent or manage the autoimmune diseases on the basis of the results of some short-term clinical trials.177,178,181
Respiratory Tract Diseases and Wheezing Disorders
At the turn of the past century, children with rickets were at higher risk for upper respiratory tract infections and for dying of them.26,183 Macrophages have a VDR, and when they ingest an infectious agent, such as tuberculosis bacillus, the toll-like receptors are activated, resulting in signal transduction to increase the expression of VDR and CYP27B1.7,26,145 In turn, 25(OH)D is converted to 1,25(OH)2D, which signals the nucleus to increase the expression of cathelicidin, a defensin protein that kills infective agents, such as tuberculosis bacillus.7,26
Cord blood 25(OH)D levels were associated with tolerogenic immune regulation and fewer respiratory tract infections in newborns.184 Also, high 25(OH)D levels during maternity were associated with a decrease in childhood wheezing by nearly 50% compared with low maternal 25(OH)D levels. Newborns with 25(OH)D levels less than 10 ng/mL were twice as likely to develop respiratory tract infections compared with those with levels of 30 ng/mL or greater, and every 4-ng/mL increase in the cord blood 25(OH)D level lowered the cumulative risk of wheezing by age 5 years.184 Serum concentrations of 25(OH)D in 198 healthy adults revealed that a concentration of 38 ng/mL or higher reduced the risk of acute viral respiratory tract infections and number of days ill by 2-fold.185 Japanese children who received 1200 IU/d of vitamin D from December through March compared with those who received placebo reduced their risk of influenza A by 42%.181 It was also observed that children who took vitamin D daily had a relative risk reduction of 93% for having an asthma attack compared with children who did not take a vitamin D supplement.186 Vitamin D has also been implicated in the reversal of corticosteroid resistance and in airway remodeling, which are the hallmarks of chronic obstructive pulmonary disease and severe asthma. Dietary vitamin D may regulate epigenetic events, in particular on genes that are responsible for chronic obstructive pulmonary disease susceptibility.187
The potential role of vitamin D in reducing the risk of allergies also may be related to epigenetic regulation.188,189 Misdirected epigenetic programming offered an explanation for why vitamin D deficiency in pregnancy may be associated with increased allergy rates in the offspring. The cord blood level of 25(OH)D found a U-shaped association, with a 2.4-fold odds ratio (OR) of low and a 4-fold OR of high levels of 25(OH)D to develop allergen-specific IgE.188,190 Eczema was significantly more likely in those with 25(OH)D levels less than 20 ng/mL compared with those with 25(OH)D levels of 30 ng/mL or greater (OR, 2.66; 95% CI, 1.24–5.72; P=.01).189 On a molecular level, maternal vitamin D intake during pregnancy increased the mRNA levels of immunoglobulin-like transcript (ILT) 3 and ILT4 in umbilical cord blood.191 Because ILT3 and ILT4 are critical for the generation of T suppressor cells and the induction of immunologic tolerance, this finding may point toward an early induction of tolerogenic immune responses by maternal vitamin D intake in the developing child. In addition, vitamin D stimulates natural killer cells that are known to play an immunoregulatory role in the prevention of autoimmune diseases.2 Thus, although vitamin D can favorably influence several pathways associated with respiratory tract diseases, there are few clinical trials to support the beneficial effect of vitamin D supplementation for these patients. Meta-analyses on respiratory outcomes111 and recovery from tuberculosis112 did not report a beneficial effect of supplementation for patients with cystic fibrosis or tuberculosis, respectively.
Neurologic Disorders
The brain has a VDR and has the ability to produce 1,25(OH)2D3. In vivo mouse studies found that in utero hypovitaminosis D impairs brain development and leads to persistent changes in the adult brain.192 The 1,25(OH)2D3 is rapidly incorporated into embryonic hippocampal cells, moves into the nucleus, and then returns to the cytoplasm.193 These events delay cell proliferation and induce cell differentiation characterized by the expression of differentiation markers, modification of soma lengthening, and increase in neurite length and branching.193 At birth, rats with prenatal vitamin D deficiency had heavier and longer brains, enlarged lateral ventricles, and decreased cortical thickness.5,173–175 Evidence from human studies is scanty. One recent study found that higher maternal serum 25(OH)D levels in late pregnancy (<12 vs >30 ng/mL) were associated with larger head circumference of offspring at 9 years old but not with measures of cognition or psychological health.5,194 In addition, there may be a critical window during late gestation in which vitamin D insufficiency precipitates an altered adult behavioral phenotype.195 In rats, offspring of vitamin D–deficient mothers had significant impairment of latent inhibition (ability to ignore irrelevant stimuli), a feature often associated with schizophrenia, whereas those transiently depleted had subtle and discrete alterations in learning and memory.196 In a Finnish birth cohort study, 9114 individuals were drawn from the northern Finland 1966 birth cohort.197 In males, the use of at least 2000 IU of vitamin D during the first year of life was associated with a reduced risk of schizophrenia (RR, 0.23, 95% CI, 0.06–0.95) compared with those taking lower doses.197
There is minimal evidence for an association of low maternal vitamin D status with risk of autism.5 Children of dark-skinned mothers, particularly immigrants to low–ambient UV radiation locations, such as Minnesota, may be at increased risk, but this finding has been inconsistent.198
The 1,25(OH)2D3 seems to have a neuroprotective role, inducing remyelination by endogenous progenitor cells and stimulation of amyloid-β clearance by macrophages of patients with Alzheimer disease.199 A vitamin D3–enriched diet correlated with a decrease in the number of amyloid plaques and inflammation in the brains of AβPP mice.199 These observations suggest that a vitamin D3–enriched diet may reduce the risk of Alzheimer disease as well as depression and neurocognitive disorders. An Australian study of 743 white pregnant women found that maternal vitamin D insufficiency during pregnancy is significantly associated with offspring language impairment.200 Vitamin D deficiency was also associated with prominent changes in behavior and brain neurochemistry in the adult mouse.201 In the follow-up of a British birth cohort (n=7401), current and subsequent risk of depression in middle adulthood was associated with low serum 25(OH)D levels. This study provides support for a lower risk of depression with serum 25(OH)D levels between 20 and 34 ng/mL.202 A meta-analysis of cohort studies reported that there was a significantly increased HR of depression for the lowest vs highest vitamin D categories (HR, 2.21; 95% CI, 1.40–3.49).203 In a community setting, depressed adults had significantly lower 25(OH)D levels than those without depression.204 A variety of studies found an association between a low level of 25(OH)D and a high depression score.205,206 Patients who received 400 to 800 IU of vitamin D with calcium for 6 to 12 months did not have an improvement in their mental health scores. However, patients who received 400 to 800 IU of vitamin D for 5 days with calcium or a single 100,000 IU dose of vitamin D had an improvement in the assessments of depression.207,208 Although there is a strong association between risk of neurologic disorders and serum 25(OH)D concentrations, there are only a few short-term clinical trials of vitamin D in patients with MS that have not reported benefit and no clinical trials evaluating other neurologic disorders.177,178
Adverse Pregnancy Outcomes
A recent meta-analysis of data from 24 studies found that women with circulating 25(OH)D levels less than 20 ng/mL in pregnancy experienced an increased risk of preeclampsia (OR, 2.09; 95% CI, 1.50–2.90), gestational diabetes mellitus (OR, 1.38; 95% CI, 1.12–1.70), preterm birth (OR, 1.58; 95% CI, 1.08–2.31), and small for gestational age (OR, 1.52; 95% CI, 1.08–2.15).209 However, many of these outcomes are rare and require a large sample size to study, representing a challenge for cohorts with a limited number of preserved samples. Experimental studies have provided evidence of disrupted vitamin D metabolic homeostasis in the preeclamptic placenta and have suggested that increased oxidative stress could be a causative factor of altered vitamin D metabolism in preeclamptic placentas.50 In normal placenta, DBP, CYP24A1, and VDR expressions were localized mainly in trophoblasts, whereas CYP2R1 and CYP27B1 expressions were localized mainly in villous core fetal vessel endothelium.50 Protein expression of CYP2R1 and VDR were reduced, but CYP27B1 and CYP24A1 expressions were elevated in preeclamptic compared with normotensive placentas.50 A similar pattern was observed in an in vitro model that found that hypoxia induced down-regulation of DBP, CYP2R1, and VDR and up-regulation of CYP27B1 and CYP24A1.50 These data indicate that fetal (trophoblastic) autocrine synthesis of 1,25(OH)2D3 may play a pivotal role in controlling placental inflammation and preeclampsia.
One of the main pathogenic features of preeclampsia is maternal endothelial dysfunction that results from impaired angiogenesis and reduced endothelial repair capacity. The 1,25(OH)2D3 improves the angiogenic properties of endothelial progenitor cells. These findings could explain the positive influence of vitamin D3 in reducing preeclampsia risk.151
There was an inverse association with having a cesarean delivery and serum 25(OH)D levels. In a case-control study, after adjustment for race, age, educational level, insurance status, and alcohol use, women with 25(OH)D levels less than 15 ng/mL were almost 4 times as likely to have a cesarean delivery than were women with 25(OH)D levels of at least 15 ng/mL.210
A meta-analysis of 3 trials involving 463 women suggested that women who received vitamin D supplements during pregnancy less frequently had a baby with a birth weight less than 2500 g than did those who received no treatment or placebo; the statistical significance was borderline.89 In terms of other conditions, there were no significant differences in adverse effects, including nephrotic syndrome, stillbirths, and neonatal deaths, between women who received vitamin D supplements and women who received no treatment or placebo.89 A meta-analysis indicated a significant inverse relation between serum 25(OH)D level and the incidence of gestational diabetes mellitus. Overall, vitamin D deficiency (25[OH]D <20 ng/mL) in pregnancy was significantly related to the incidence of gestational diabetes mellitus, with an OR of 1.61.211 However, it remains unclear whether this association is causal owing to the observational design of the studies. Recently, meta-analyses by Thorne-Lyman and Fawzi88 and De-Regil et al89 reported a similar beneficial effect of vitamin D supplementation on birth weight but no significant effect on other maternal and neonatal outcomes.
ASSESSING VITAMIN D STATUS
Although the generally accepted measure of vitamin D status is circulating 25(OH)D concentration, there is little consensus on which assay method should be used. Commonly used assays include competitive protein-binding assay, radioimmunoassay, enzyme immunoassay, chemiluminescence immunoassay, high-performance liquid chromatography, and liquid chromatography–tandem mass spectrometry (LC-MS/MS), each with its own advantages and disadvantages.212,213 Binkley et al214 reported that 25(OH)D results differed widely depending on the laboratory and the method used, with the mean result (from 10 healthy adults) varying from 17.1 to 35.6 ng/mL. A study conducted by the Vitamin D External Quality Assessment Scheme found a 31% overestimation by one immunoassay method.215 Its specificity needs to exclude significant interferences from the C-3 epimer of 25(OH)D, which is more prevalent in infants younger than 1 year.216
DiaSorin radioimmunoassays (DiaSorin Corp) used in the NHANES III had a mean bias of greater than 12% comparing the vitamin D status of the US population of 1988–1994 with that of 2000–2004. This difference was probably caused by changes in reagents and calibration lots performed by the manufacturer.217,218 This makes diagnostic and therapeutic decisions on the basis of absolute cutoff values for vitamin D deficiency extremely problematic219,220 and hinders the comparability of results from prospective and epidemiologic studies. Since November 2010, efforts have been made to recalibrate 25(OH)D measurements from all NHANES samples with LC-MS/MS. This effort is within the context of an international approach to standardization of 25(OH)D measurements in national surveys, the Vitamin D Standardization Program, and publication of the results is planned for the middle of 2013.221 To ensure that laboratories are providing accurate testing results, it is important that their vitamin D testing method measures total 25(OH)D levels (25[OH]D2 and 25[OH]D3) and has acceptable precision. The immunoassay remains the predominant mode of measurement for 25(OH)D. Most, if not all, of these assays have problems with equimolar recovery of the 25(OH)D2 and 25(OH)D3 levels.222 The level of 25(OH)D2 is underestimated by 20% to 80% and can vary for different patients who received vitamin D2 in the same assay. Standardization of all assays has been improved but not resolved with the currently available reference materials, as evidenced by the Vitamin D External Quality Assessment Scheme.222 The choice of method for each laboratory remains a balance mainly among turnaround time, convenience, cost, and the specificity and accuracy of the information obtained.208 Recognizing the importance of a 25(OH)D2 and 25(OH)D3 reference material, the National Institute of Standards and Technology released a 4-level Standard Reference Material set, SRM972.221 Treatment with vitamin D2, therefore, may not be accurately monitored using many of the commercial assays because these antibody assays often underestimate 25(OH)D2 levels and, thus, total 25(OH)D levels, which is what they record in the report. This issue is most important in patients who experience no improvement with replacement therapy with vitamin D2 (the most commonly used Food and Drug Administration–approved pharmaceutical form of vitamin D); the absence of a rise in the total 25(OH)D level may indicate nonadherence or malabsorption.223 For these patients, use of the gold standard LC-MS/MS would quantitatively report 25(OH)D2, 25(OH)D3, and total 25(OH)D levels.
VITAMIN D STATUS DURING PREGNANCY, BIRTH, AND CHILDHOOD
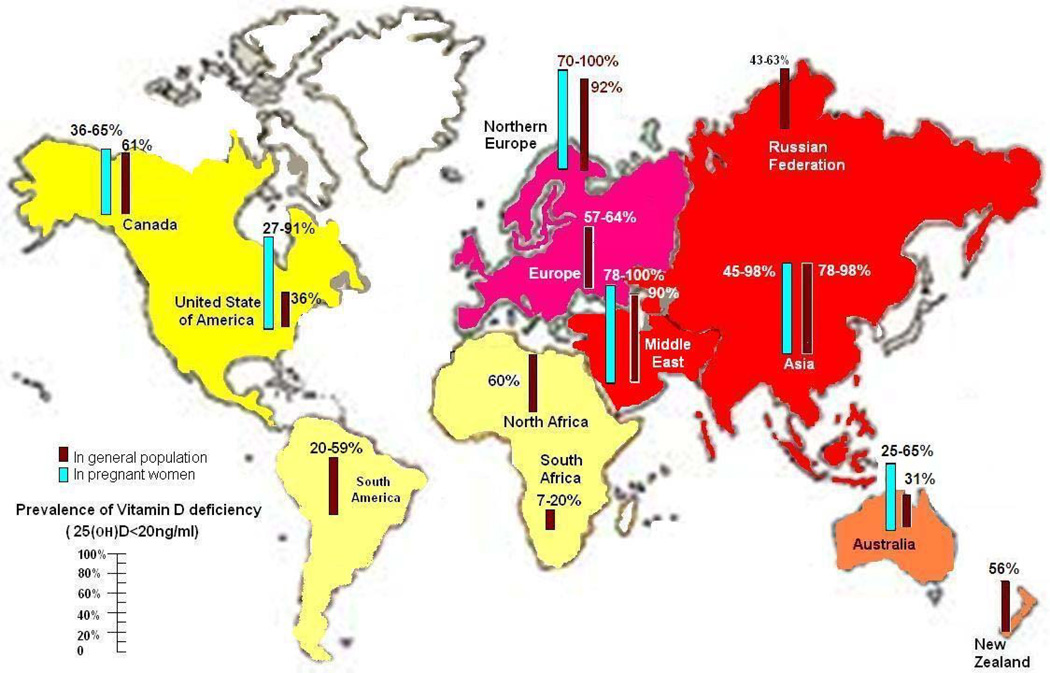
Maternal vitamin D deficiency predisposes to low vitamin D stores in the newborn and increases infantile rickets224 because the mother is the only source of vitamin D during pregnancy. The prevalence of vitamin D deficiency and insufficiency during pregnancy is of special concern and ranges from 8% to 100%, depending on the country of residence and the definitions of vitamin D deficiency and insufficiency (Figure 5).2 In the United States, vitamin D deficiency and insufficiency is estimated to be 27% to 91% in pregnant women.2 As shown in Figure 5, this rate is estimated to be 39% to 65% in Canada, 45% to 100% in Asia, 19% to 96% in Europe, and 25% to 87% in Australia and New Zealand.
FIGURE 5.
Reported incidence of vitamin D deficiency defined as a 25-hydroxyvitamin D level below 20 ng/ml around the globe in pregnant women and general population. To convert 25(OH)D values to nmol/L, multiply by 2.496. Copyright Holick 2013, reproduced with permission.
The prevalence of vitamin D deficiency and insufficiency in children in China was high, especially in children aged 6 to 16 years.211 In the United States, it is estimated that 50% of children aged 1 to 5 years and 70% of children aged 6 to 11 years are vitamin D deficient or insufficient.156 Recent studies reported that adolescents and young adults are also at risk for vitamin D deficiency.200–205 Also, a high prevalence of vitamin D deficiency was reported in a cross-sectional study conducted at a tertiary care center in western India.225
Evidence suggests that children and adults in the United States are becoming more vitamin D deficient and insufficient because of an increase in the incidence of obesity, a decrease in milk consumption, and an increase in sun protection.211,215 This recent evidence emphasizes the high prevalence of vitamin D deficiency throughout the world, not only in at-risk groups (Figure 5).226,227
PREVALENCE OF VITAMIN D DEFICIENCY IN ADOLESCENTS AND ADULTS
It has been estimated that 20% to 80% of US, Canadian, and European men and women are vitamin D deficient.228,229 The prevalence of serum 25(OH)D levels less than 20 ng/mL was almost one-third of the US population (32%). More than 70% of non-Hispanic black individuals and more than 40% of Hispanic/Mexican individuals were at risk for a 25(OH)D level less than 20 ng/mL.228 In a national Canadian cohort, serum 25(OH)D levels less than 30 ng/mL were evident in 57.5% of men and in 60.7% of women, and they rose to 73.5% in spring (men) and 77.5% in winter (women).229 In the Healthy Lifestyle in Europe by Nutrition in Adolescence study, 25(OH)D levels less than 30 ng/mL were reported to be approximately 80% in adolescents from the 9 European countries.230 Levels of 25(OH)D were higher in northern Europe than in southern Europe and were higher in western Europe than in eastern Europe.230 The higher levels in northern Europe were also observed in some multicenter studies in which a single laboratory facility was used.
The higher serum 25(OH)D levels in Norway and Sweden are probably due to a high intake of fatty fish and cod liver oil. The lower serum 25(OH)D levels in Spain, Italy, and Greece may be due to more skin pigmentation, sunshine-avoiding behavior, and air pollution with ozone and nitrogen dioxide, which reduce sun-induced vitamin D production.226,231
In the Middle East and Asia, vitamin D deficiency in children and adults is highly prevalent.214,216 Children and adults of color are especially at high risk owing to the inefficient cutaneous production of vitamin D3.1,2,7 In a study on the vitamin D status of Australian adults, vitamin D deficiency (25[OH]D <20 ng/mL) was 31% (22% in men and 39% in women); 73% had 25(OH)D levels less than 30 ng/mL.226 Women who practice purdah (ie, the use of clothing and other approaches to screen themselves from men and strangers) and children and adults who avoid all sun exposure or wear sunscreen protection are equally at high risk.7,232
CAUSES OF VITAMIN D DEFICIENCY AND RISK FACTORS
Traditional risk groups for vitamin D deficiency include pregnant women, children, older persons, the institutionalized, and nonwestern immigrants.7,228 The major source of vitamin D for children and adults is exposure to natural sunlight.7,61 The Maasai and Hadzabe tribes in Tanzania (East Africa) with traditional lifestyles, living in the presumed cradle of humankind, who are exposed daily to tropical sunlight had a mean circulating 25(OH)D levels of 46 ng/mL.140
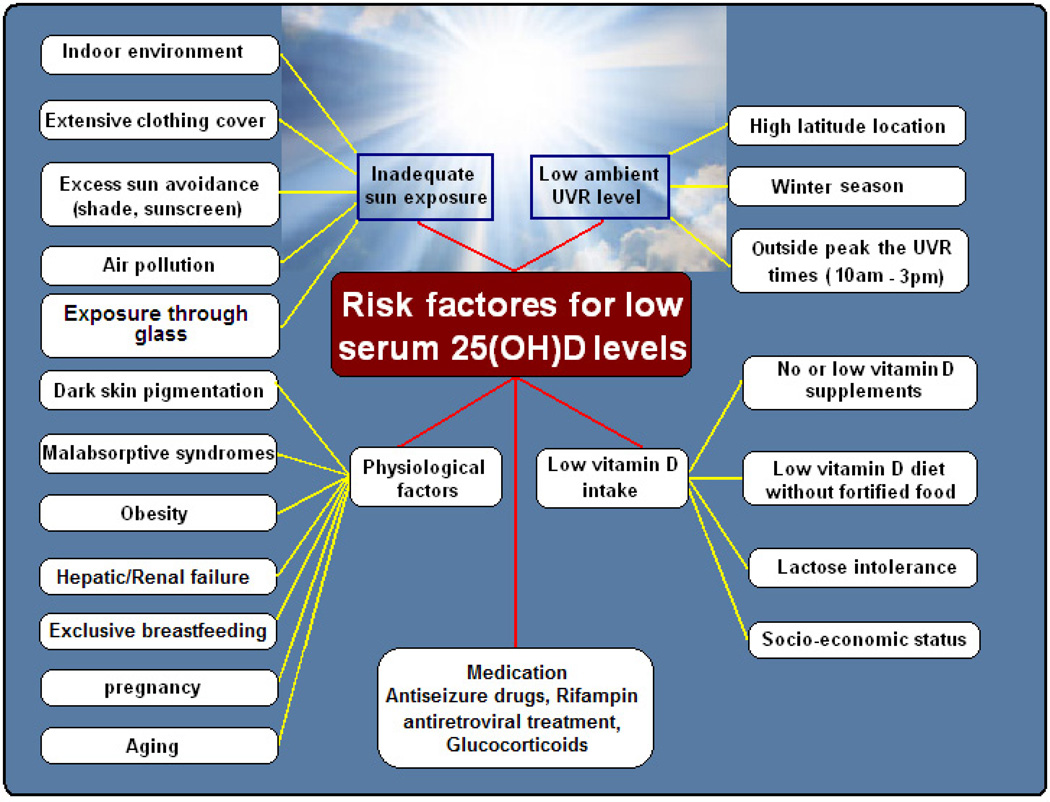
A variety of factors influence the cutaneous production of vitamin D. A sunscreen with a sun protection factor of 30 applied properly reduces the ability of the skin to produce vitamin D by as much as 95% to 99%. People of color who have natural sunscreen protection from their increased melanin pigment are less efficient by more than 90% in producing vitamin D in their skin compared with white individuals.233 In addition, air pollution with increased ozone and nitrogen dioxide levels (both known to compromise several health outcomes)absorbs UVB radiation and is an often-neglected risk factor for hypovitaminosis D.61,231,234 Important risk factors for vitamin D deficiency are shown in Figure 6.
FIGURE 6.
Risk factors of low vitamin D status. Copyright Holick 2013, reproduced with permission.
The prevalence of vitamin D deficiency and insufficiency was affected by seasonal variation and latitude. The prevalence increased in late winter/spring and decreased in summer.235 A study of the effect of education on vitamin D status found that low-educated women had lower 25(OH)D levels compared with high-educated women, and women in the lowest 25(OH)D quartile had a higher risk of small-for-gestational-age offspring.236
The elderly population is particularly at risk for clinical complications related to low 25(OH)D levels. With increasing age, solar exposure is usually limited because of changes in lifestyle factors, such as clothing and less outdoor activity. Diet may also become less varied, with a lower natural vitamin D content. Most important, however, the cutaneous production of vitamin D after exposure to solar UVB radiation decreases with age because of atrophic skin changes, with a reduced amount of its precursor 7-DHC.237,238 A comparison of the amount of previtamin D3 produced in skin from 8- and 18-year-old individuals with the amount produced in skin from 77- and 82-year-old individuals revealed that aging can decrease by greater than 2-fold the capacity of the skin to produce previtamin D3.238
Although the heritability of vitamin D status seems considerable, the specific genetic determinants of 25(OH)D levels are only beginning to be identified. A recent examination of 141 single nucleotide polymorphisms (SNPs) in a discovery cohort of 1514 white participants from the community-based Cardiovascular Health Study found that lower serum 25(OH)D levels were associated with HRs for the risk of the composite outcome of 1.40 for those who had 1 minor allele at rs7968585 (in VDR) and 1.82 for those with 2 minor alleles.215,216 This candidate gene study indicates that known associations of low serum 25(OH)D levels with clinical outcomes may vary according to genetic differences in the VDR. In black patients, there were significant associations in 3 SNPs in vitamin D pathway genes (rs2282679, rs2298849, and rs10877012), all of which replicate earlier findings in populations of European ancestry.239 Included among these was rs2282679, a highly significant result from 2 recent genome-wide association studies (GWASs),240,241 one of which reported a 49% increased risk of vitamin D deficiency (25[OH]D <20 ng/mL) associated with the rs2282679 minor allele in white individuals.218 In another study of genetic predictors of 25(OH)D in black individuals involved 513 participants from 42 families in Los Angeles, California, and evaluated 30 SNPs in GC, VDR, and CYP27B1.242 Recent epigenomic findings confirmed 3 genes (DHCR7, CYP2R1, and CYP24A1) of the 4 genes in the GWAS findings, which reinforces the crucial roles played by those 3 genes in vitamin D metabolism.243 DHCR7 encodes the enzyme 7-DHC reductase, which converts 7-DHC to cholesterol, thereby removing the substrate from the synthetic pathway of vitamin D3.239 DHCR7 is a novel gene for association with 25(OH)D levels, as identified in 2 recent GWASs.228,229 CYP24A1, which encodes 25(OH)D–24-hydroxylase, has been identified as a candidate gene for vitamin D insufficiency in one GWAS but not in the other.217,218 This mitochondrial protein initiates the degradation of 1,25(OH)2D3 and plays a role in calcium homeostasis and vitamin D metabolism. These epigenomic findings suggest that individuals with vitamin D deficiency are more likely to have reduced synthesis and increased catabolism of 25(OH)D and 1,25(OH)2D.243
The genetic contributions to circulating 25(OH)D represents a complex trait for which family studies have estimated heritability ranging from 43% to 80%.231 Genomic and epigenomic data integration provided greater understanding of the physiology and etiology of the complex traits. Further elucidation of the genetic architecture of this complex trait beyond environmental determinants of 25(OH)D has the potential to identify those at risk for vitamin D insufficiency.244 It may also provide a useful proxy for lifetime vitamin D exposure that may be applied in instrumental variable analyses investigating the association between vitamin D and common complex diseases. However, a recent GWAS of prospectively collected 25(OH)D data in 5 studies with 5575 individuals reported that known GWAS-associated SNPs explain only a fraction of the observed variance in circulating 25(OH)D levels (ie, approximately 5.2%).244 On rare occasions, some patients who deny taking a vitamin D supplement have unexplained high normal 25(OH)D levels in the range of 40 to 80 ng/mL. It is believed that this is due to a genetic mutation of the cyp24p1 that reduces the catabolism of 25(OH)D and 1,25(OH)2D and can be a cause of infantile hypercalcemia.231,245 Therefore, these recent genomic and epigenetic data provide additional evidence of genetic-environmental interactions and their effects on circulating 25(OH)D levels.
TREATMENT AND PREVENTION OF VITAMIN D DEFICIENCY AND INSUFFICIENCY WITH SUN EXPOSURE AND UVB IRRADIATION
Humans obtain a considerable amount of their vitamin D requirement from sun exposure.7,140 Although excessive exposure to sunlight increases the risk of nonmelanoma skin cancer, which is easy to detect and easy to treat, there is no evidence that sensible sun exposure, as our hunter-gatherer forefathers likely experienced, increases risk.124 More importantly, the most deadly form of melanoma skin cancer that occurs on the least sun-exposed areas is less likely to occur in adults who have outdoor occupations.1,124,141,246,247 Therefore, it is not unreasonable to consider sensible sun exposure as a good source of vitamin D.7,124 An adult in a bathing suit exposed to 1 minimal erythemal dose (slight pinkness to the skin 24 hours after exposure) is equivalent to taking approximately 20,000 IU (500 µg) of vitamin D2 orally.7,15 Thus, exposure of arms and legs to 0.5 minimal erythemal dose is equivalent to ingesting approximately 3000 IU of vitamin D3.7,60 Adults who frequented a tanning salon had robust levels of 25(OH)D, on average 46 ng/mL, and had higher bone mineral density in their hips compared with healthy adults who did not go to a tanning salon in Boston, Massachusetts, at the end of winter.248 It was estimated that if all the people in the United States were to double their solar UVB irradiance to raise their serum 25(OH)D levels to 45 ng/mL, the net result could be as many as 400,000 reduced deaths compared with only 11,000 increased deaths from melanoma and other skin cancer.249 Time of day during sun exposure, season, latitude, and degree of skin pigmentation dictate how much vitamin D3 is produced during sun exposure. Exposure of the arms and legs (abdomen and back when possible) to sunlight 2 to 3 times a week for approximately 25% to 50% of the time it would take to develop a mild sunburn (minimal erythemal dose) will cause the skin to produce enough vitamin D. For a white person, if 30 minutes of June noontime sun would cause a mild sunburn, then 10 to 15 minutes of exposure followed by good sun protection should be sufficient to produce adequate vitamin D.7 There is no need to ever expose the face because although it is the most sun exposed of all the body areas, it provides little vitamin D3. A free app, dminder, provides the user with information about sensible sun exposure and vitamin D production. For patients with fat malabsorption syndromes that render oral consumption of supplemental vitamin D ineffective, exposure to a lamp that emits UVB radiation can be effective in raising blood levels of 25(OH)D.250
Food Sources
Very few foods naturally contain vitamin D; examples of foods with ample vitamin D stores include wild-caught salmon and UV-exposed mushrooms.7 Foods fortified with vitamin D usually contain 100 IU per serving. An analysis of the vitamin D intake of children and adults in the United States revealed that they were unable to achieve the RDA for vitamin D from any dietary sources.251
Vitamin D intake can be increased by eating foods fortified with vitamin D. A recent systematic review found that food fortification with vitamin D (especially in milk) is effective in significantly increasing 25(OH)D levels in the population.7,252 Other foods include some cereals, juices, other dairy products, and some margarines. A mean individual intake of approximately 11 µg/d (440 IU/d) from fortified foods (range, 120–1000 IU/d) increased 25(OH)D concentrations by 7.7 ng/mL, corresponding to a 0.48 ng/mL increase in 25(OH)D for each 40 IU (1 µg) ingested.252
Vitamin D Supplementation
The RDA for vitamin D and tolerable upper limit levels vary in different age groups and in certain circumstances.7,60,61 Although it is recommended that RDAs of 600 to 800 IU daily should meet the requirements to optimize bone health62 in most of the population, higher vitamin D intakes (1000–2000 IU) are needed to reach and maintain 25(OH)D levels greater than 30 ng/mL.7,60 It is recognized that for every 100 IU of vitamin D ingested, the blood level of 25(OH)D increases by approximately 0.6 to 1 ng/mL.253 When the serum 25(OH)D level is less than 15 ng/mL, 100 IU of vitamin D will increase the 25(OH)D level by as much as 2 to 3 ng/mL.7,71 An effective strategy to treat vitamin D deficiency and insufficiency in children and adults is to give them 50,000 IU of vitamin D2 once a week for 6 and 8 weeks, respectively.60,254 To prevent recurrence of vitamin D deficiency in children, administration of 600 to 1000 IU/d is effective.60 For adults, to prevent recurrence of vitamin D deficiency, administration of 50,000 IU of vitamin D2 every 2 weeks is effective.7,60,128 This strategy was shown to be effective in maintaining blood levels of 25(OH)D at approximately 40 to 60 ng/mL for up to 6 years without any evidence of toxic effects.255
Vitamin D can be administered daily, weekly, monthly, or every 4 months to sustain an adequate serum 25(OH)D concentration.7,232–234 A bolus of high doses of vitamin D (up to 300,000 IU) can be initially used in persons with extreme vitamin D deficiency. Repeated boluses of high-dose vitamin D at 6- to 12-month intervals have been used in a nursing home setting, but a steady state serum 25(OH)D concentration is likely to be maintained by more frequent, lower doses of vitamin D. One study has suggested that a 500,000-IU bolus dose of vitamin D3 increases the risk of fracture within 3 months,256 but other studies have reported reduced risk of fracture.257,258
Vitamin D Supplementation During Pregnancy and Lactation
The 25(OH)D passes from the placenta into the bloodstream of the fetus. Because the half-life for 25(OH)D is approximately 2 to 3 weeks, the infant can remain vitamin D sufficient for several weeks after birth, as long as the mother is vitamin D sufficient.2
In a study of 40 mostly black pregnant women who were documented to be ingesting approximately 600 IU of vitamin D a day, at the time that they gave birth, 76% were vitamin D deficient as defined by the IOM cutoff value of a 25(OH)D level less than 20 ng/mL.259 Eighty-one percent of their newborns were vitamin D deficient.259 Maternal supplementation with 2000 and 4000 IU/d of vitamin D during pregnancy improved the maternal/neonatal vitamin D status.256 None of the pregnant women developed significant changes in their serum calcium or 24-hour urine calcium levels. Evidence of risk reduction in infection, preterm labor, and preterm birth was suggestive, requiring additional studies powered for these end points.260
Human breast milk and unfortified cow’s milk have little vitamin D.7,260,261 Only after lactating women were given 4000 to 6000 IU/d of vitamin D was enough vitamin D transferred in breast milk to satisfy the infant’s requirement.259–261
Vitamin D Supplementation in Special Conditions
Because body fat can sequester vitamin D, it is now recognized that children and adults who are obese require 2 to 5 times more vitamin D to treat and prevent vitamin D deficiency.7,60 Patients taking antiseizure medications, AIDS medications, and glucocorticoids often require more vitamin D to satisfy their requirements.7,60 However, patients with granulomatous disorders, such as sarcoidosis and tuberculosis, are at risk for hypercalciuria and hypercalcemia when blood levels of 25(OH)D are greater than 30 ng/mL owing to the increased serum levels of 1,25(OH)2D produced in the macrophages in the granulomas.7 Therefore, their vitamin D intake needs to be carefully monitored and controlled.7,60 Hence, daily requirements of vitamin D to reach and maintain the desired serum 25(OH)D level can be estimated from the baseline 25(OH)D concentration. Supplemental vitamin D is preferentially administered orally or intramuscularly (not available in the United States), and the vitamin D–producing Sperti lamp can be used, where available, in patients with malabsorption syndromes.248,262
Type of Vitamin D Supplementation
Either vitamin D2 or vitamin D3 can be used for vitamin D supplementation, although there is controversy regarding vitamin D3 vs vitamin D2 for achieving and maintaining higher serum 25(OH)D levels. Although a recent meta-analysis indicated that vitamin D3 is more efficacious at raising serum 25(OH)D concentrations than is vitamin D2,2,102 several prospective studies have found them to be equally effective in raising and maintaining serum 25(OH)D levels in children and adults.263,264 Holick and colleagues264 found that an 11-week course of treatment with 1000 IU/d of vitamin D2, 1000 IU/d of vitamin D3, or a combination of 500 IU of vitamin D2 and 500 IU of vitamin D3 daily caused an equivalent increase in serum total 25(OH)D levels. Furthermore, the group that received vitamin D2 did not experience a significant change in serum 25(OH)D3 levels. Gordon et al265 and Thacher et al266 also found that in infants and toddlers treated for 6 weeks, 2000 IU of vitamin D2 and 2000 IU of vitamin D3 daily or a single dose of 50,000 IU of vitamin D2 or vitamin D3 were equally effective in increasing the serum total 25(OH)D level. The bioavailability of vitamin D3 is well established, and the bioavailability of vitamin D2 from mushrooms in humans has been found to be comparable with that of a vitamin D2 supplement.267 Finally, adults treated with vitamin D2 not only raised their total blood levels of 25(OH)D but also maintained total blood levels of 1,25(OH)2D to the same degree as adults who received the same dose of vitamin D3.263
SAFETY AND INTOXICATION
Vitamin D intoxication is characterized by hypercalcemia, hypercalciuria, and hyperphosphatemia, which, in turn, are responsible for soft-tissue and vascular calcifications and nephrolithiasis in the long-term. Serum 25(OH)D levels are usually markedly elevated (>150 ng/mL) in individuals with vitamin D intoxication.7,60,90 Daily doses of vitamin D3 up to 10,000 IU were safe in healthy males, and there was no evidence of hypercalcemia or hypercalciuria for 5 months.253,268 This amount is far above the tolerable upper level indicated in the IOM guidelines (4000 IU). Higher doses of vitamin D (up to 40,000 IU/d) are still safe provided that a serum 25(OH)D concentration of 200 ng/mL is not exceeded. A recent report of an infant inadvertently receiving 12,000 IU of vitamin D3 daily for 20 days and achieving a serum 25(OH)D level of 425 ng/mL had no signs of vitamin D intoxication. Once the vitamin D use was stopped, the serum 25(OH)D level was less than 100 ng/mL within 2 months.269
CONCLUSION
Vitamin D deficiency is a common underdiagnosed condition that has received increasing attention in the world. The US Endocrine Society guidelines and the IOM recommend screening only in populations at risk, as no evidence currently exists to support screening at a population level. Candidates for vitamin D screening include those who are at specific risk for vitamin D deficiency and patients who are experiencing or are at risk for specific medical conditions associated with hypovitaminosis D.7,60,90
Recent evidence from hundreds of studies has suggested that vitamin D is important for reducing the risk of a variety of chronic illnesses. The identification of a VDR in most tissues and cells and the observation that a multitude of genes may be directly or indirectly regulated by 1,25(OH)2D have provided a rationale for the nonskeletal health benefits of vitamin D. A study in healthy adults who received either 400 or 2000 IU/d of vitamin D3 for 3 months in winter reported that 291 genes were either up- or down-regulated. That these genes affected as many as 160 different metabolic pathways (from immune modulation to enhanced antioxidant activity) emphasizes the importance of improving the world’s vitamin D status.17 The observation that 1,25(OH)2D may also influence epigenetics provides additional support for the concept that there is no downside to increasing the vitamin D status of children and adults. Vitamin D deficiency during pregnancy may adversely influence placental development and fetal programming. Vitamin D deficiency in both parents may influence adverse pregnancy outcomes and susceptibility to developing disease in adult life and even into the next generation.
There is potentially a great upside (in terms of improving overall health and well-being) to increasing serum 25(OH)D levels above 30 ng/mL. An effective strategy to prevent vitamin D deficiency and insufficiency is to obtain some sensible sun exposure, ingest foods that contain vitamin D, and take a vitamin D supplement.
Supplementary Material
TABLE.
Summary of Meta-analyses of Vitamin D Supplementation
| Reference, year | Included studies | Sample size (No.) |
Participants | Dose/duration | Outcomes | Effects |
|---|---|---|---|---|---|---|
| Thorne-Lyman and Fawzi,88 2012 |
5 randomized trials 2 observational studies |
28,943 | Pregnant women |
Vitamin D2 and vitamin D3 Various doses and patterns of intake during pregnancy |
Perinatal and infant health |
Positive effect on low birth weight No effect on small-for- gestational age (2 trials) No effect on preterm delivery |
| De-Regil et al,89 2012 |
6 trials | 1023 | Women during pregnancy |
Vitamin D (1200 IU/d) alone or combined with 375 mg of elemental calcium |
Safely improve maternal and neonatal outcomes |
No effect on preeclampsia Positive effect on concentrations of 25(OH)D in serum Positive effect on birth weight No effect on adverse effects |
| Bischoff-Ferrari et al,78 2012 |
11 double-blind RCTs |
31,022 | Persons aged ≥65 y |
Oral vitamin D supplementation with or without calcium |
Fracture reduction |
No effect on risk of hip fracture until 800 IU/d Positive effect on hip and any nonvertebral fracture by highest intake level according to quartiles |
| Lai et al,90 2010 | 7 eligible RCTs and 17 identified case-control studies |
801 | Persons aged 74.8–85 y |
Vitamin D2 and vitamin D3 (400–1100 IU) |
Hip fracture risk | No effect on hip fracture risk |
| Bergman et al,802010 |
8 controlled trials | 12,658 | Postmenopaus al women |
Vitamin D3 supplementation (800 IU/d) with or without calcium |
Increasing BMD Preventing fractures |
Positive effect on nonvertebral and hip fractures |
| Bischoff-Ferrari et al,81 2009 |
12 double- blindRCTs |
83,165 | Older individuals (≥65 y) |
>400 IU/d | Preventing nonvertebral and hip fractures |
Positive effect on nonvertebral fracture prevention with vitamin D is dose dependent (only high dose) |
| Avenell et al,82 2009 |
45 trials | 83,741 | Older people | Vitamin D or related compounds |
Preventing fractures |
Positive effect by vitamin D with calcium on hip fracture No effect by vitamin D alone on hip fracture |
| Abrahamsen et al,75 2010 |
7 major randomized trials |
68,517 | Persons aged 47–107 y |
Vitamin D2 and vitamin D3 (10 µg/d to 300,000 IU/12 mo) |
Antifracture efficacy |
Positive effect of vitamin D with calcium on fracture No effect of vitamin D alone |
| Izaks,91 2007 | 11 trials | NA | General population |
Vitamin D2 and vitamin D3; follow-up >1 y |
Fracture risk | High-dose vitamin D may be effective in institutionalized persons but probably is not effective in the general population |
| Jackson et al,92 2007 |
9 studies | 2410 | Postmenopaus al women |
Vitamin D3 (excluding the potential effect of calcium supplementation) |
Risk of fall and fracture |
Positive effect on risk of fall in patients treated with vitamin D3 |
| Boonen et al,932007 |
10 RCTs | 54,592 | Postmenopaus al women and/or older men (≥50 y) |
Oral vitamin D with or without calcium vs placebo/no treatment |
Prevention of hip fractures |
Positive effect of oral vitamin D on reducing the risk of hip fractures only with calcium supplementation |
| Avenell et al,94 2005 |
57 trials | 82,986 | Older people | Vitamin D or an analogue, alone or with calcium, vs placebo |
Fracture | Positive effect of vitamin D with calcium supplements on hip and other nonvertebral fractures |
| Bischoff-Ferrari et al,84 2005 |
Five RCTs for hip fracture 7 RCTs for nonvertebral fracture risk |
19,114 | Older people | Oral vitamin D supplementation (cholecalciferol, ergocalciferol) with or without calcium supplementation vs calcium supplementation |
Preventing hip and nonvertebral fractures |
Positive effect (700–800 IU/d) on hip and any nonvertebral fractures in ambulatory or institutionalized elderly persons No effect (400 IU/d) on fracture prevention |
| Winzenberg et al,85 2011 |
6 studies | 884 | Healthy children and adolescents (aged 1 mo to <20 y) |
Vitamin D supplementation vs placebo for ≥3 mo |
Improving BMD (effects vary with factors such as vitamin D dose and vitamin D status) |
No effect on total body BMC or on hip or forearm BMD Positive small effect on lumbar spine BMD Positive effect with low serum vitamin D on total body BMC and lumbar spine bone |
| Huncharek et al,86 2008 |
21RCTs | NA | Children | Dietary calcium/dairy supplementation |
BMC | Positive effect of dietary calcium/dairy products, with and without vitamin D, on total body and lumbar spine BMC in children (with low baseline intakes) |
| Bischoff-Ferrari et al,95 2009 |
8 RCTs | 2426 | Older individuals |
Vitamin D2 and vitamin D3 (200–1000 IU) with or without calcium |
Preventing falls | Positive effect of supplemental vitamin D (700–1000 IU/d) on the risk of falling No effect at a dose <700 IU |
| Kalyani et al,96 2010 |
10 articles | 2932 | Older adults (aged ≥60 y) |
200–1000 IU/d of vitamin D for 1–36 mo |
Fall prevention | Positive effect on fall prevention |
| Chung et al,97 2011 | 19 RCTs (3 for cancer and 16 for fracture outcomes) 28 observational studies (for cancer outcomes) |
NA | Adults | Vitamin D with or without calcium (limited data from RCTs assessed high- dose vitamin D [1000 IU/d]) |
Benefits and harms of vitamin D with or without calcium supplementatio n on clinical outcomes of cancer and fractures |
Positive effect of high- dose vitamin D on reduced risk of total cancer Positive effect on fracture Negative effect on renal and urinary tract stones |
| Buttigliero et al,98 2011 |
25 studies (3 randomized trials involving patients with advanced prostate cancer explored the |
1273 | Cancer patients | 1 trial: doxercalciferol 2 trials: calcitriol Duration: 11.7–18.32 mo |
Influence of hypovitaminosi s D on prognosis of cancer Improvement |
No effect on survival |
| prognostic role of vitamin D supplementation ) |
outcome of vitamin D supplementatio n |
|||||
| Bjelakovic et al,99 2011 |
50 randomized trials |
94,148 | Adults Most trials included elderly women (>70 y) |
Supplemental vitamin D (vitamin D3 [cholecalciferol] or vitamin D2 [ergocalciferol]) or an active form of vitamin D (1α- hydroxyvitamin D [alfacalcidol] or 1,25- dihydroxyvitamin D [calcitriol]) at any dose, duration, and route of administration vs placebo or no intervention |
Beneficial and harmful effects of vitamin D for prevention of mortality |
Positive effect of vitamin D3 on mortality Negative effect of vitamin D3 combined with calcium on nephrolithiasis Negative effect on hypercalcemia |
| Irlam et al,100 2010 | 16 additional trials (only 1 trial was single supplements of vitamin D) |
22,120 participa nts in trials |
Adults and children with HIV infection |
NA | Reducing mortality and morbidity |
No effect |
| Autier et al,101 2012 |
76 trials | 6207 | White persons aged >50 y |
Doses of 5–250 µg/d (median, 20 µg/d) |
Circulating 259OH)D level |
Positive effect of vitamin D3 intake without calcium on serum 25(OH)D concentrations No effect of concomitant use of calcium supplementation and high 25(OH)D concentration at baseline |
| Bjorkman et al,102 2009 |
52 clinical trials | 6290 | Chronically immobile patients |
Vitamin D supplementation |
Responses of parathyroid hormone |
Positive effect in chronically immobile patients on 25(OH)D levels but a slight effect on PTH decrease |
| Tripkovic et al,103 2012 |
17 studies | 1016 | Persons aged 18–97 y |
Varying dosages and treatment periods |
Compared the effects of vitamin D2 and vitamin D3 on serum 25(OH)D concentrations |
Positive effect of vitamin D3 compared with vitamin D2 in the raising of serum 25(OH)D concentrations |
| Kandula et al,104 2011 |
22 studies | 264 | Patients with non–dialysis- dependent CKD, dialysis- dependent CKD, and renal transplant |
Ergocalciferol or cholecalciferol |
Benefits and harms of vitamin D supplementatio n |
Positive effect on 25(OH)D and PTH levels |
| Song et al,105 2013 | 21 prospective studies |
81,216 | Healthy individuals and patients with type 2 diabetes |
Circulating 25(OH)D | Association between blood levels of 25(OH)D and risk of incident type 2 diabetes |
Inverse and significant association between circulating 25(OH)D levels and risk of type 2 diabetes |
| George et al,106 2012 |
15 trials | NA | Nondiabetes to diabetes |
Vitamin D or analogues |
Glycemia, insulin resistance, progression to diabetes, and complications of diabetes |
No effect on fasting glucose, hemoglobin A1c, or insulin resistance Small positive effect on fasting glucose and insulin resistance in patients with diabetes or impaired glucose tolerance No effect on glycated hemoglobin in diabetic patients |
| Bath-Hextall et al,107 2012 |
11 studies | 596 | Atopic eczema/dermat itis |
Vitamin D vs vitamin E |
Treating established atopic eczema/dermat itis |
Negative effect at high doses |
| Muir et al,70 2011 | 13 trials | NA | Older adults (≥60 y) |
Vitamin D (800–1000 IU/d) |
Muscle strength, gait, and balance |
Positive effect on balance and muscle strength |
| Annweiler et al,108 2009 |
16 trials | 24- 33,067 |
Persons aged ≥80 y |
NA | Muscle, balance, and gait performance |
No significant effect on balance and gait Positive/no effect on muscle strength No effect on sit-to- stand test |
| Wang et al,109 2010 |
18 trials | NA | Adults | NA | Reduce the risk of cardiovascular events |
No effect on cardiovascular disease risk |
| Pittas et al,110 2010 | 18 trials | 37,162 | Generally healthy adults |
Vitamin D (400–8571 IU/d) with or without calcium |
Cardiometaboli c outcomes |
No effect on glycemia or incident diabetes, blood pressure, and cardiovascular outcomes |
| Ferguson and Chang,111 2009 |
3 double-blind randomized crossover trial |
41 | Adults and children with cystic fibrosis |
800 and1600 IU of vitamin D alone with or without 1 g of calcium |
Respiratory outcomes |
No adequate evidence of benefit or harm |
| Abba et al,112 2008 | 12 trials | 3393 | Patients with tuberculosis |
Several vitamins and minerals and diets |
Promote the recovery of tuberculosis |
No effect on number of deaths or number of participants with positive sputum test results |
| Autier and Gandini,113 2007 |
18 independent RCTs |
57,311 | At risk for dying of any cause |
Vitamin D supplements varied from 300 to 2000 IU/d |
Any health condition |
Not enough evidence for effective decision |
BMC = bone mineral content; BMD = bone mineral density; CKD = chronic kidney disease; HIV = human immunodeficiency virus; NA = not available; PTH = parathyroid hormone; RCT = randomized controlled trial; 25(OH)D = .
ARTICLE HIGHLIGHTS.
Comprehensive evidence-based review of the musculoskeletal and noncalcemic health benefits of vitamin D.
A review and perspective regarding the definitions of vitamin D deficiency, insufficiency, and sufficiency.
Recommendations for the prevention and treatment of vitamin D deficiency in pregnant and lactating women, children, and adults.
An update on the role of vitamin D in broad gene expression in human immune cells and exploration of how vitamin D may influence epigenetics.
Review of biochemical mechanisms that may help explain the broad range of health benefits associated with vitamin D.
Acknowledgments
Grant Support: This work was supported, in part, by grant UL-1-RR-25711 from the National Institutes of Health and by the Mushroom Council.
Abbreviations and Acronyms
- CD
Crohn disease
- DBP
vitamin D binding protein
- DC
dendritic cell
- ESCEO
genome-wide association study
- GWAS
Crohn disease
- HR
hazard ratio
- ILT
immunoglobulin-like transcript
- IOM
Institute of Medicine
- LC-MS/MS
liquid chromatography–tandem mass spectrometry
- LPS
lipopolysaccharide
- miRNA
microRNA
- MS
multiple sclerosis
- NHANES
National Health and Nutrition Examination Survey
- OR
odds ratio
- PTH
parathyroid hormone
- PTHrP
Crohn disease
- parathyroid hormone–related protein
Crohn disease
- RA
rheumatoid arthritis
- RDA
recommended dietary allowance
- RR
risk rate
- SNP
single nucleotide polymorphism
- TH
T helper cell
- VDR
vitamin D receptor
- WHI
Women’s Health Initiative
- 1,25(OH)2D
1α,25-dihydroxyvitamin D
- 7-DHC
7-dehydrocholesterol
- 25(OH)D
25-hydroxyvitamin D
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Potential Competing Interests: The authors report no competing interests.
REFERENCES
- 1.Holick MF. Vitamin D: extraskeletal health. Rheum Dis Clin North Am. 2012;38(1):141–160. doi: 10.1016/j.rdc.2012.03.013. [DOI] [PubMed] [Google Scholar]
- 2.Hossein-nezhad A, Holick MF. Optimize dietary intake of vitamin D: an epigenetic perspective. Curr Opin Clin Nutr Metab Care. 2012;15(6):567–579. doi: 10.1097/MCO.0b013e3283594978. [DOI] [PubMed] [Google Scholar]
- 3.Smit E, Crespo CJ, Michael Y, et al. The effect of vitamin D and frailty on mortality among non-institutionalized US older adults. Eur J Clin Nutr. 2012;66(9):1024–1028. doi: 10.1038/ejcn.2012.67. [DOI] [PubMed] [Google Scholar]
- 4.Holick MF. Nutrition: D-iabetes and D-eath D-efying vitamin D. Nat Rev Endocrinol. 2012;8(7):388–390. doi: 10.1038/nrendo.2012.84. [DOI] [PubMed] [Google Scholar]
- 5.Lucas RM, Ponsonby AL, Pasco JA, Morley R. Future health implications of prenatal and early-life vitamin D status. Nutr Rev. 2008;66(12):710–720. doi: 10.1111/j.1753-4887.2008.00126.x. [DOI] [PubMed] [Google Scholar]
- 6.Brannon PM, Picciano MF. Vitamin D in pregnancy and lactation in humans. Annu Rev Nutr. 2011;31:89–115. doi: 10.1146/annurev.nutr.012809.104807. [DOI] [PubMed] [Google Scholar]
- 7.Holick MF. Vitamin D deficiency. N Engl J Med. 2007;357(3):266–281. doi: 10.1056/NEJMra070553. [DOI] [PubMed] [Google Scholar]
- 8.Holick MF. Sunlight and vitamin D for bone health and prevention of autoimmune diseases, cancers, and cardiovascular disease. Am J Clin Nutr. 2004;80(6) suppl:1678S–1688S. doi: 10.1093/ajcn/80.6.1678S. [DOI] [PubMed] [Google Scholar]
- 9.Holick MF, MacLaughlin JA, Clark MB, et al. Photosynthesis of previtamin D3 in human skin and the physiologic consequences. Science. 1980;210(4466):203–205. doi: 10.1126/science.6251551. [DOI] [PubMed] [Google Scholar]
- 10.Webb AR, Kline L, Holick MF. Influence of season and latitude on the cutaneous synthesis of vitamin D3: exposure to winter sunlight in Boston and Edmonton will not promote vitamin D3 synthesis in human skin. J Clin Endocrinol Metab. 1988;67(2):373–378. doi: 10.1210/jcem-67-2-373. [DOI] [PubMed] [Google Scholar]
- 11.Holick MF, Chen TC, Lu Z, Sauter E. Vitamin D and skin physiology: a D-lightful story. J Bone Miner Res. 2007;22(suppl 2):V28–V33. doi: 10.1359/jbmr.07s211. [DOI] [PubMed] [Google Scholar]
- 12.Negri AL. Proximal tubule endocytic apparatus as the specific renal uptake mechanism for vitamin D-binding protein/25-(OH)D3 complex. Nephrology. 2006;11(6):510–515. doi: 10.1111/j.1440-1797.2006.00704.x. [DOI] [PubMed] [Google Scholar]
- 13.Portale A, Perwad F. Calcium and phosphorus. Pediatr Nephrol. 2009:231–265. [Google Scholar]
- 14.Nair R, Maseeh A. Vitamin D: the “sunshine” vitamin. J Pharmacol Pharmacother. 2012;3(2):118–126. doi: 10.4103/0976-500X.95506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Holick MF. The D-lightful vitamin D for child health. JPEN J Parenter Enteral Nutr. 2012;36(1) suppl:9S–19S. doi: 10.1177/0148607111430189. [DOI] [PubMed] [Google Scholar]
- 16.Nagpal S, Na S, Rathnachalam R. Noncalcemic actions of vitamin D receptor ligands. Endocr Rev. 2005;26(5):662–687. doi: 10.1210/er.2004-0002. [DOI] [PubMed] [Google Scholar]
- 17.Hossein-nezhad A, Spira A, Holick MF. Influence of vitamin D status and vitamin D3 supplementation on genome wide expression of white blood cells: a randomized double-blind clinical trial. PLoS One. 2013;8(3):e58725. doi: 10.1371/journal.pone.0058725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kitazawa S, Kajimoto K, Kondo T, Kitazawa R. Vitamin D3 supports osteoclastogenesis via functional vitamin D response element of human RANKL gene promoter. J Cell Biochem. 2003;89(4):771–777. doi: 10.1002/jcb.10567. [DOI] [PubMed] [Google Scholar]
- 19.Cui X, Pelekanos M, Liu PY, Burne TH, McGrath JJ, Eyles DW. The vitamin D receptor in dopamine neurons; its presence in human substantia nigra and its ontogenesis in rat midbrain. Neuroscience. 2013;236:77–87. doi: 10.1016/j.neuroscience.2013.01.035. [DOI] [PubMed] [Google Scholar]
- 20.Gonzalez-Parra E, Rojas-Rivera J, Tunon J, Praga M, Ortiz A, Egido J. Vitamin D receptor activation and cardiovascular disease. Nephrol Dial Transplant. 2012;27(suppl 4):v17–iv21. doi: 10.1093/ndt/gfs534. [DOI] [PubMed] [Google Scholar]
- 21.Nowak R, Szota J, Mazurek U. Vitamin D receptor gene (VDR) transcripts in bone, cartilage, muscles and blood and microarray analysis of vitamin D responsive genes expression in paravertebral muscles of juvenile and adolescent idiopathic scoliosis patients. BMC Musculoskelet Disord. 2012;13(259):1471–2474. doi: 10.1186/1471-2474-13-259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Doig CL, Singh PK, Dhiman VK, et al. Recruitment of NCOR1 to VDR target genes is enhanced in prostate cancer cells and associates with altered DNA methylation patterns. Carcinogenesis. 2013;34(2):248–256. doi: 10.1093/carcin/bgs331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Alimirah F, Peng X, Yuan L, et al. Crosstalk between the peroxisome proliferator-activated receptor γ (PPAR γ) and the vitamin D receptor (VDR) in human breast cancer cells: PPARγ binds to VDR and inhibits 1 α,25-dihydroxyvitamin D3 mediated transactivation. Exp Cell Res. 2012;318(19):2490–2497. doi: 10.1016/j.yexcr.2012.07.020. [DOI] [PubMed] [Google Scholar]
- 24.Nagy L, Szanto A, Szatmari I, Szeles L. Nuclear hormone receptors enable macrophages and dendritic cells to sense their lipid environment and shape their immune response. Physiol Rev. 2012;92(2):739–789. doi: 10.1152/physrev.00004.2011. [DOI] [PubMed] [Google Scholar]
- 25.Kaludjerovic J, Vieth R. Relationship between vitamin D during perinatal development and health. J Midwifery Womens Health. 2010;55(6):550–560. doi: 10.1016/j.jmwh.2010.02.016. [DOI] [PubMed] [Google Scholar]
- 26.Adams JS, Hewison M. Update in vitamin D. J Clin Endocrinol Metab. 2010;95(2):471–478. doi: 10.1210/jc.2009-1773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Shin JS, Choi MY, Longtine MS, Nelson DM. Vitamin D effects on pregnancy and the placenta. Placenta. 2010;31(12):1027–1034. doi: 10.1016/j.placenta.2010.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Adams JS, Hewison M. Extrarenal expression of the 25-hydroxyvitamin D-1-hydroxylase. Arch Biochem Biophys. 2012;523(1):95–102. doi: 10.1016/j.abb.2012.02.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Grayson R, Hewison M. Vitamin D and human pregnancy. Fetal Matern Med Rev. 2011;22(1):67–90. [Google Scholar]
- 30.Powe CE, Seely EW, Rana S, et al. First trimester vitamin D, vitamin D binding protein, and subsequent preeclampsia. Hypertension. 2010;56(4):758–763. doi: 10.1161/HYPERTENSIONAHA.110.158238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zella LA, Shevde NK, Hollis BW, Cooke NE, Pike JW. Vitamin D-binding protein influences total circulating levels of 1,25-dihydroxyvitamin D3 but does not directly modulate the bioactive levels of the hormone in vivo. Endocrinology. 2008;149(7):3656–3667. doi: 10.1210/en.2008-0042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Liu NQ, Hewison M. Vitamin D, the placenta and pregnancy. Arch Biochem Biophys. 2012;523(1):37–47. doi: 10.1016/j.abb.2011.11.018. [DOI] [PubMed] [Google Scholar]
- 33.Ardawi MS, Nasrat HA, BA’Aqueel HS. Calcium-regulating hormones and parathyroid hormone-related peptide in normal human pregnancy and postpartum: a longitudinal study. Eur J Endocrinol. 1997;137(4):402–409. doi: 10.1530/eje.0.1370402. [DOI] [PubMed] [Google Scholar]
- 34.Weisser J, Riemer S, Schmidl M, et al. Four distinct chondrocyte populations in the fetal bovine growth plate: highest expression levels of PTH/PTHrP receptor, Indian hedgehog, and MMP-13 in hypertrophic chondrocytes and their suppression by PTH (1–34) and PTHrP (1–40) Exp Cell Res. 2002;279(1):1–13. doi: 10.1006/excr.2002.5580. [DOI] [PubMed] [Google Scholar]
- 35.Kovacs CS, Kronenberg HM. Maternal-fetal calcium and bone metabolism during pregnancy, puerperium, and lactation. Endocr Rev. 1997;18(6):832–872. doi: 10.1210/edrv.18.6.0319. [DOI] [PubMed] [Google Scholar]
- 36.Mahadevan S, Kumaravel V, Bharath R. Calcium and bone disorders in pregnancy. Indian J Endocrinol Metab. 2012;16(3):358–363. doi: 10.4103/2230-8210.95665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kovacs CS. Vitamin D in pregnancy and lactation: maternal, fetal, and neonatal outcomes from human and animal studies. Am J Clin Nutr. 2008;88(2):520S–528S. doi: 10.1093/ajcn/88.2.520S. [DOI] [PubMed] [Google Scholar]
- 38.Kovacs CS. Calcium and bone metabolism disorders during pregnancy and lactation. Endocrinol Metab Clin North Am. 2011;40(4):795–826. doi: 10.1016/j.ecl.2011.08.002. [DOI] [PubMed] [Google Scholar]
- 39.Moller UK, Streym S, Mosekilde L, et al. Changes in calcitropic hormones, bone markers and insulin-like growth factor I (IGF-I) during pregnancy and postpartum: a controlled cohort study. Osteoporos Int. 2013;24(4):1307–1320. doi: 10.1007/s00198-012-2062-2. [DOI] [PubMed] [Google Scholar]
- 40.Zhong Y, Armbrecht HJ, Christakos S. Calcitonin, a regulator of the 25-hydroxyvitamin D3 1α -hydroxylase gene. J Biol Chem. 2009;284(17):11059–11069. doi: 10.1074/jbc.M806561200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kovacs CS. Bone development in the fetus and neonate: role of the calciotropic hormones. Curr Osteoporos Rep. 2011;9(4):274–283. doi: 10.1007/s11914-011-0073-0. [DOI] [PubMed] [Google Scholar]
- 42.Jones G, Kano K, Yamada S, Furusawa T, Takayama H, Suda T. Identification of 24,25,26,27-tetranor-23-hydroxyvitamin D3 as a product of the renal metabolism of 24,25-dihydroxyvitamin D3 . Biochemistry. 1984;23(16):3749–3754. doi: 10.1021/bi00311a028. [DOI] [PubMed] [Google Scholar]
- 43.Papapetrou PD. The interrelationship of serum 1,25-dihydroxyvitamin D, 25-hydroxyvitamin D and 24,25-dihydroxyvitamin D in pregnancy at term: a meta-analysis. Hormones. 2010;9(2):136–144. doi: 10.14310/horm.2002.1263. [DOI] [PubMed] [Google Scholar]
- 44.Novakovic B, Sibson M, Ng HK, et al. Placenta-specific methylation of the vitamin D 24-hydroxylase gene: implications for feedback autoregulation of active vitamin D levels at the fetomaternal interface. J Biol Chem. 2009;284(22):14838–14848. doi: 10.1074/jbc.M809542200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Dror DK. Vitamin D status during pregnancy: maternal, fetal, and postnatal outcomes. Curr Opin Obstet Gynecol. 2011;23(6):422–426. doi: 10.1097/GCO.0b013e32834cb791. [DOI] [PubMed] [Google Scholar]
- 46.Morris GS, Zhou Q, Hegsted M, Keenan MJ. Maternal consumption of a low vitamin D diet retards metabolic and contractile development in the neonatal rat heart. J Mol Cell Cardiol. 1995;27(6):1245–1250. doi: 10.1016/s0022-2828(05)82386-7. [DOI] [PubMed] [Google Scholar]
- 47.Kim BG, Chang SK, Kim SM, Hwang JS, Jung JW. Dilated cardiomyopathy in a 2 month-old infant: a severe form of hypocalcemia with vitamin d deficient rickets. Korean Circ J. 2010;40(4):201–203. doi: 10.4070/kcj.2010.40.4.201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Jansson T, Powell TL. Role of the placenta in fetal programming: underlying mechanisms and potential interventional approaches. Clin Sci. 2007;113(1):1–13. doi: 10.1042/CS20060339. [DOI] [PubMed] [Google Scholar]
- 49.Evans KN, Bulmer JN, Kilby MD, Hewison M. Vitamin D and placental-decidual function. J Soc Gynecol Investig. 2004;11(5):263–271. doi: 10.1016/j.jsgi.2004.02.002. [DOI] [PubMed] [Google Scholar]
- 50.Ma R, Gu Y, Zhao S, Sun J, Groome LJ, Wang Y. Expressions of vitamin D metabolic components VDBP, CYP2R1, CYP27B1, CYP24A1, and VDR in placentas from normal and preeclamptic pregnancies. Am J Physiol Endocrinol Metab. 2012;303(7):E928–E935. doi: 10.1152/ajpendo.00279.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Maka N, Makrakis J, Parkington HC, Tare M, Morley R, Black MJ. Vitamin D deficiency during pregnancy and lactation stimulates nephrogenesis in rat offspring. Pediatr Nephrol. 2008;23(1):55–61. doi: 10.1007/s00467-007-0641-9. [DOI] [PubMed] [Google Scholar]
- 52.Stefanska B, Karlic H, Varga F, Fabianowska-Majewska K, Haslberger A. Epigenetic mechanisms in anti-cancer actions of bioactive food components: the implications in cancer prevention. Br J Pharmacol. 2012;167(2):279–297. doi: 10.1111/j.1476-5381.2012.02002.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Barker DJ, Eriksson JG, Forsen T, Osmond C. Fetal origins of adult disease: strength of effects and biological basis. Int J Epidemiol. 2002;31(6):1235–1239. doi: 10.1093/ije/31.6.1235. [DOI] [PubMed] [Google Scholar]
- 54.Karlic H, Varga F. Impact of vitamin D metabolism on clinical epigenetics. Clin Epigenetics. 2011;2(1):55–61. doi: 10.1007/s13148-011-0021-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Martin R, Harvey NC, Crozier SR, et al. Placental calcium transporter (PMCA3) gene expression predicts intrauterine bone mineral accrual. Bone. 2007;40(5):1203–1208. doi: 10.1016/j.bone.2006.12.060. [DOI] [PubMed] [Google Scholar]
- 56.Lapillonne A. Vitamin D deficiency during pregnancy may impair maternal and fetal outcomes. Med Hypotheses. 2010;74(1):71–75. doi: 10.1016/j.mehy.2009.07.054. [DOI] [PubMed] [Google Scholar]
- 57.Tekes K, Gyenge M, Hantos M, Csaba G. Transgenerational hormonal imprinting caused by vitamin A and vitamin D treatment of newborn rats: alterations in the biogenic amine contents of the adult brain. Brain Dev. 2009;31(9):666–670. doi: 10.1016/j.braindev.2008.10.007. [DOI] [PubMed] [Google Scholar]
- 58.Milovanovic M, Heine G, Hallatschek W, Opitz B, Radbruch A, Worm M. Vitamin D receptor binds to the epsilon germline gene promoter and exhibits transrepressive activity. J Allergy Clin Immunol. 2010;126(5):1016–1023. doi: 10.1016/j.jaci.2010.08.020. [DOI] [PubMed] [Google Scholar]
- 59.Kelly JL, Drake MT, Fredericksen ZS, et al. Early life sun exposure, vitamin D-related gene variants, and risk of non-Hodgkin lymphoma. Cancer Causes Control. 2012;23(7):1017–1029. doi: 10.1007/s10552-012-9967-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Holick MF, Binkley NC, Bischoff-Ferrari HA, Gordon CM, Hanley DA, Heaney RP, Murad MH, Weaver CM. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(7):1911–1930. doi: 10.1210/jc.2011-0385. [DOI] [PubMed] [Google Scholar]
- 61.Bischoff-Ferrari HA, Giovannucci E, Willett WC, Dietrich T, Dawson-Hughes B. Estimation of optimal serum concentrations of 25-hydroxyvitamin D for multiple health outcomes. Am J Clin Nutr. 2006;84(1):18–28. doi: 10.1093/ajcn/84.1.18. [DOI] [PubMed] [Google Scholar]
- 62.Ross AC, Manson JE, Abrams SA, et al. The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: what clinicians need to know. J Clin Endocrinol Metab. 2011;96(1):53–58. doi: 10.1210/jc.2010-2704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Hewison M, Adams JS. Vitamin D insufficiency and skeletal development in utero. J Bone Miner Res. 2010;25(1):11–13. doi: 10.1002/jbmr.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Sanders KM, Nicholson GC, Ebeling PR. Is high dose vitamin D harmful? Calcif Tissue Int. 2013;92(2):191–206. doi: 10.1007/s00223-012-9679-1. [DOI] [PubMed] [Google Scholar]
- 65.Mahon P, Harvey N, Crozier S, et al. Low maternal vitamin D status and fetal bone development: cohort study. J Bone Miner Res. 2010;25(1):14–19. doi: 10.1359/jbmr.090701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Holick MF, Lim R, Dighe AS. Case records of the Massachusetts General Hospital: case 3–2009: a 9-month-old boy with seizures. N Engl J Med. 2009;360(4):398–407. doi: 10.1056/NEJMcpc0807821. [DOI] [PubMed] [Google Scholar]
- 67.Nowson CA, McGrath JJ, Ebeling PR, et al. Vitamin D and health in adults in Australia and New Zealand: a position statement. Med J Aust. 2012;196(11):686–687. doi: 10.5694/mja11.10301. [DOI] [PubMed] [Google Scholar]
- 68.Sai AJ, Walters RW, Fang X, Gallagher JC. Relationship between vitamin D, parathyroid hormone, and bone health. J Clin Endocrinol Metab. 2011;96(3):E436–E446. doi: 10.1210/jc.2010-1886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Bischoff-Ferrari HA, Dietrich T, Orav EJ, et al. Higher 25-hydroxyvitamin D concentrations are associated with better lower-extremity function in both active and inactive persons aged ≥60 y. Am J Clin Nutr. 2004;80(3):752–758. doi: 10.1093/ajcn/80.3.752. [DOI] [PubMed] [Google Scholar]
- 70.Muir SW, Montero-Odasso M. Effect of vitamin D supplementation on muscle strength, gait and balance in older adults: a systematic review and meta-analysis. J Am Geriatr Soc. 2011;59(12):2291–2300. doi: 10.1111/j.1532-5415.2011.03733.x. [DOI] [PubMed] [Google Scholar]
- 71.Pramyothin P, Holick MF. Vitamin D supplementation: guidelines and evidence for subclinical deficiency. Curr Opin Gastroenterol. 2012;28(2):139–150. doi: 10.1097/MOG.0b013e32835004dc. [DOI] [PubMed] [Google Scholar]
- 72.Pfeifer M, Begerow B, Minne HW, Suppan K, Fahrleitner-Pammer A, Dobnig H. Effects of a long-term vitamin D and calcium supplementation on falls and parameters of muscle function in community-dwelling older individuals. Osteoporos Int. 2009;20(2):315–322. doi: 10.1007/s00198-008-0662-7. [DOI] [PubMed] [Google Scholar]
- 73.Bischoff-Ferrari HA, Shao A, Dawson-Hughes B, Hathcock J, Giovannucci E, Willett WC. Benefit-risk assessment of vitamin D supplementation. Osteoporos Int. 2010;21(7):1121–1132. doi: 10.1007/s00198-009-1119-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Goswami R, Vatsa M, Sreenivas V, et al. Skeletal muscle strength in young Asian Indian females after vitamin D and calcium supplementation: a double-blind randomized controlled clinical trial. J Clin Endocrinol Metab. 2012;97(12):4709–4716. doi: 10.1210/jc.2012-2340. [DOI] [PubMed] [Google Scholar]
- 75.Abrahamsen B, Masud T, Avenell A, et al. Patient level pooled analysis of 68 500 patients from seven major vitamin D fracture trials in US and Europe. BMJ. 2010;12(340):b5463. doi: 10.1136/bmj.b5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Elders PJ, Lips P, Netelenbos JC, et al. Long-term effect of calcium supplementation on bone loss in perimenopausal women. J Bone Miner Res. 1994;9(7):963–970. doi: 10.1002/jbmr.5650090702. [DOI] [PubMed] [Google Scholar]
- 77.Reid IR, Mason B, Horne A, et al. Randomized controlled trial of calcium in healthy older women. Am J Med. 2006;119(9):777–785. doi: 10.1016/j.amjmed.2006.02.038. [DOI] [PubMed] [Google Scholar]
- 78.Bischoff-Ferrari HA, Willett WC, Orav EJ, et al. A pooled analysis of vitamin D dose requirements for fracture prevention. N Engl J Med. 2012;367(1):40–49. doi: 10.1056/NEJMoa1109617. [DOI] [PubMed] [Google Scholar]
- 79.Priemel M, von Domarus C, Klatte TO, et al. Bone mineralization defects and vitamin D deficiency: histomorphometric analysis of iliac crest bone biopsies and circulating 25-hydroxyvitamin D in 675 patients. J Bone Miner Res. 2010;25(2):305–312. doi: 10.1359/jbmr.090728. [DOI] [PubMed] [Google Scholar]
- 80.Bergman GJ, Fan T, McFetridge JT, Sen SS. Efficacy of vitamin D3 supplementation in preventing fractures in elderly women: a meta-analysis. Curr Med Res Opin. 2010;26(5):1193–1201. doi: 10.1185/03007991003659814. [DOI] [PubMed] [Google Scholar]
- 81.Bischoff-Ferrari HA, Willett WC, Wong JB, et al. Prevention of nonvertebral fractures with oral vitamin D and dose dependency: a meta-analysis of randomized controlled trials. Arch Intern Med. 2009;169(6):551–561. doi: 10.1001/archinternmed.2008.600. [DOI] [PubMed] [Google Scholar]
- 82.Avenell A, Gillespie WJ, Gillespie LD, O’Connell D. Vitamin D and vitamin D analogues for preventing fractures associated with involutional and post-menopausal osteoporosis. Cochrane Database Syst Rev. 2009;(2):CD000227. doi: 10.1002/14651858.CD000227.pub3. [DOI] [PubMed] [Google Scholar]
- 83.Moyer VA. U.S. Preventive Services Task Force. Vitamin D and calcium supplementation to prevent fractures in adults: u.s. Preventive services task force recommendation statement. Ann Intern Med. 2013;158(9):691–696. doi: 10.7326/0003-4819-158-9-201305070-00603. [DOI] [PubMed] [Google Scholar]
- 84.Bischoff-Ferrari HA, Willett WC, Wong JB, Giovannucci E, Dietrich T, Dawson-Hughes B. Fracture prevention with vitamin D supplementation: a meta-analysis of randomized controlled trials. JAMA. 2005;293(18):2257–2264. doi: 10.1001/jama.293.18.2257. [DOI] [PubMed] [Google Scholar]
- 85.Winzenberg T, Powell S, Shaw KA, Jones G. Effects of vitamin D supplementation on bone density in healthy children: systematic review and meta-analysis. BMJ. 2011;342:c7254. doi: 10.1136/bmj.c7254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Huncharek M, Muscat J, Kupelnick B. Impact of dairy products and dietary calcium on bone-mineral content in children: results of a meta-analysis. Bone. 2008;43(2):312–321. doi: 10.1016/j.bone.2008.02.022. [DOI] [PubMed] [Google Scholar]
- 87.Rizzoli R, Boonen S, Brandi ML, et al. Vitamin D supplementation in elderly or postmenopausal women: a 2013 update of the 2008 recommendations from the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO) Curr Med Res Opin. 2013;29(4):305–313. doi: 10.1185/03007995.2013.766162. [DOI] [PubMed] [Google Scholar]
- 88.Thorne-Lyman A, Fawzi WW. Vitamin D during pregnancy and maternal, neonatal and infant health outcomes: a systematic review and meta-analysis. Paediatr Perinat Epidemiol. 2012;1:75–90. doi: 10.1111/j.1365-3016.2012.01283.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.De-Regil LM, Palacios C, Ansary A, Kulier R, Pena-Rosas JP. Vitamin D supplementation for women during pregnancy. Cochrane Database Syst Rev. 2012;(2):CD008873. doi: 10.1002/14651858.CD008873.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Lai JK, Lucas RM, Clements MS, Roddam AW, Banks E. Hip fracture risk in relation to vitamin D supplementation and serum 25-hydroxyvitamin D levels: a systematic review and meta-analysis of randomised controlled trials and observational studies. BMC Public Health. 2010;10(331):1471–2458. doi: 10.1186/1471-2458-10-331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Izaks GJ. Fracture prevention with vitamin D supplementation: considering the inconsistent results. BMC Musculoskelet Disord. 2007;8:26. doi: 10.1186/1471-2474-8-26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Jackson C, Gaugris S, Sen SS, Hosking D. The effect of cholecalciferol (vitamin D3) on the risk of fall and fracture: a meta-analysis. QJM. 2007;100(4):185–192. doi: 10.1093/qjmed/hcm005. [DOI] [PubMed] [Google Scholar]
- 93.Boonen S, Lips P, Bouillon R, Bischoff-Ferrari HA, Vanderschueren D, Haentjens P. Need for additional calcium to reduce the risk of hip fracture with vitamin d supplementation: evidence from a comparative metaanalysis of randomized controlled trials. J Clin Endocrinol Metab. 2007;92(4):1415–1423. doi: 10.1210/jc.2006-1404. [DOI] [PubMed] [Google Scholar]
- 94.Avenell A, Gillespie WJ, Gillespie LD, O’Connell DL. Vitamin D and vitamin D analogues for preventing fractures associated with involutional and post-menopausal osteoporosis. Cochrane Database Syst Rev. 2005;(3):CD000227. doi: 10.1002/14651858.CD000227.pub2. [DOI] [PubMed] [Google Scholar]
- 95.Bischoff-Ferrari HA, Dawson-Hughes B, Staehelin HB, et al. Fall prevention with supplemental and active forms of vitamin D: a meta-analysis of randomised controlled trials. BMJ. 2009;339:b3692. doi: 10.1136/bmj.b3692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Kalyani RR, Stein B, Valiyil R, Manno R, Maynard JW, Crews DC. Vitamin D treatment for the prevention of falls in older adults: systematic review and meta-analysis. J Am Geriatr Soc. 2010;58(7):1299–1310. doi: 10.1111/j.1532-5415.2010.02949.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Chung M, Lee J, Terasawa T, Lau J, Trikalinos TA. Vitamin D with or without calcium supplementation for prevention of cancer and fractures: an updated meta-analysis for the U.S. Preventive Services Task Force. Ann Intern Med. 2011;155(12):827–838. doi: 10.7326/0003-4819-155-12-201112200-00005. [DOI] [PubMed] [Google Scholar]
- 98.Buttigliero C, Monagheddu C, Petroni P, et al. Prognostic role of vitamin D status and efficacy of vitamin D supplementation in cancer patients: a systematic review. Oncologist. 2011;16(9):1215–1227. doi: 10.1634/theoncologist.2011-0098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Bjelakovic G, Gluud LL, Nikolova D, et al. Vitamin D supplementation for prevention of mortality in adults. Cochrane Database Syst Rev. 2011;(7):CD007470. doi: 10.1002/14651858.CD007470.pub2. [DOI] [PubMed] [Google Scholar]
- 100.Irlam JH, Visser MM, Rollins NN, Siegfried N. Micronutrient supplementation in children and adults with HIV infection. Cochrane Database Syst Rev. 2010;(12): CD003650. doi: 10.1002/14651858.CD003650.pub3. [DOI] [PubMed] [Google Scholar]
- 101.Autier P, Gandini S, Mullie P. A systematic review: influence of vitamin D supplementation on serum 25-hydroxyvitamin D concentration. J Clin Endocrinol Metab. 2012;97(8):2606–2613. doi: 10.1210/jc.2012-1238. [DOI] [PubMed] [Google Scholar]
- 102.Bjorkman M, Sorva A, Tilvis R. Responses of parathyroid hormone to vitamin D supplementation: a systematic review of clinical trials. Arch Gerontol Geriatr. 2009;48(2):160–166. doi: 10.1016/j.archger.2007.12.005. [DOI] [PubMed] [Google Scholar]
- 103.Tripkovic L, Lambert H, Hart K, et al. Comparison of vitamin D2 and vitamin D3 supplementation in raising serum 25-hydroxyvitamin D status: a systematic review and meta-analysis. Am J Clin Nutr. 2012;95(6):1357–1364. doi: 10.3945/ajcn.111.031070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Kandula P, Dobre M, Schold JD, Schreiber MJ, Jr., Mehrotra R, Navaneethan SD. Vitamin D supplementation in chronic kidney disease: a systematic review and meta-analysis of observational studies and randomized controlled trials. Clin J Am Soc Nephrol. 2011;6(1):50–62. doi: 10.2215/CJN.03940510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Song Y, Wang L, Pittas AG, et al. Blood 25-hydroxy vitamin D levels and incident type 2 diabetes: a meta-analysis of prospective studies. Diabetes Care. 2013;36(5):1422–1428. doi: 10.2337/dc12-0962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.George PS, Pearson ER, Witham MD. Effect of vitamin D supplementation on glycaemic control and insulin resistance: a systematic review and meta-analysis. Diabet Med. 2012;29(8):e142–e150. doi: 10.1111/j.1464-5491.2012.03672.x. [DOI] [PubMed] [Google Scholar]
- 107.Bath-Hextall FJ, Jenkinson C, Humphreys R, Williams HC. Dietary supplements for established atopic eczema. Cochrane Database Syst Rev. 2012;(2):CD005205. doi: 10.1002/14651858.CD005205.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Annweiler C, Schott AM, Berrut G, Fantino B, Beauchet O. Vitamin D-related changes in physical performance: a systematic review. J Nutr Health Aging. 2009;13(10):893–898. doi: 10.1007/s12603-009-0248-x. [DOI] [PubMed] [Google Scholar]
- 109.Wang L, Manson JE, Song Y, Sesso HD. Systematic review: vitamin D and calcium supplementation in prevention of cardiovascular events. Ann Intern Med. 2010;152(5):315–323. doi: 10.7326/0003-4819-152-5-201003020-00010. [DOI] [PubMed] [Google Scholar]
- 110.Pittas AG, Chung M, Trikalinos T, et al. Systematic review: vitamin D and cardiometabolic outcomes. Ann Intern Med. 2010;152(5):307–314. doi: 10.1059/0003-4819-152-5-201003020-00009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Ferguson JH, Chang AB. Vitamin D supplementation for cystic fibrosis. Cochrane Database Syst Rev. 2009;(4):CD007398. doi: 10.1002/14651858.CD007298.pub2. [DOI] [PubMed] [Google Scholar]
- 112.Abba K, Sudarsanam TD, Grobler L, Volmink J. Nutritional supplements for people being treated for active tuberculosis. Cochrane Database Syst Rev. 2008;(4):CD006086. doi: 10.1002/14651858.CD006086.pub2. [DOI] [PubMed] [Google Scholar]
- 113.Autier P, Gandini S. Vitamin D supplementation and total mortality: a meta-analysis of randomized controlled trials. Arch Intern Med. 2007;167(16):1730–1737. doi: 10.1001/archinte.167.16.1730. [DOI] [PubMed] [Google Scholar]
- 114.Harris DM, Go VL. Vitamin D and colon carcinogenesis. J Nutr. 2004;134(12) suppl:3463S–3471S. doi: 10.1093/jn/134.12.3463S. [DOI] [PubMed] [Google Scholar]
- 115.Shui IM, Mucci LA, Kraft P, et al. Vitamin D-related genetic variation, plasma vitamin D, and risk of lethal prostate cancer: a prospective nested case-control study. J Natl Cancer Inst. 2012;104(9):690–699. doi: 10.1093/jnci/djs189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Giangreco AA, Nonn L. The sum of many small changes: microRNAs are specifically and potentially globally altered by vitamin D(3) metabolites. J Steroid Biochem Mol Biol. 2013;16(13):00005–00008. doi: 10.1016/j.jsbmb.2013.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Holick MF. Vitamin D and sunlight: strategies for cancer prevention and other health benefits. Clin J Am Soc Nephrol. 2008;3(5):1548–1554. doi: 10.2215/CJN.01350308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Freedman DM, Looker AC, Abnet CC, Linet MS, Graubard BI. Serum 25-hydroxyvitamin D and cancer mortality in the NHANES III study (1988–2006) Cancer Res. 2010;70(21):8587–8597. doi: 10.1158/0008-5472.CAN-10-1420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Garland CF, Gorham ED, Mohr SB, et al. Vitamin D and prevention of breast cancer: pooled analysis. J Steroid Biochem Mol Biol. 2007;103(3–5):708–711. doi: 10.1016/j.jsbmb.2006.12.007. [DOI] [PubMed] [Google Scholar]
- 120.Grant WB. An estimate of premature cancer mortality in the U.S. due to inadequate doses of solar ultraviolet-B radiation. Cancer. 2002;94(6):1867–1875. doi: 10.1002/cncr.10427. [DOI] [PubMed] [Google Scholar]
- 121.Lee JE, Li H, Chan AT, et al. Circulating levels of vitamin D and colon and rectal cancer: the Physicians’ Health Study and a meta-analysis of prospective studies. Cancer Prev Res. 2011;4(5):735–743. doi: 10.1158/1940-6207.CAPR-10-0289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Holick MF. Calcium plus vitamin D and the risk of colorectal cancer. N Engl J Med. 2006;354(21):2287–2288. doi: 10.1056/NEJMc060753. [DOI] [PubMed] [Google Scholar]
- 123.Bolland MJ, Grey A, Gamble GD, Reid IR. Calcium and vitamin D supplements and health outcomes: a reanalysis of the Women’s Health Initiative (WHI) limited-access data set. Am J Clin Nutr. 2011;94(4):1144–1149. doi: 10.3945/ajcn.111.015032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Holick MF. Evidence-based D-bate on health benefits of vitamin D revisited. Dermatoendocrinol. 2012;4(2):183–190. doi: 10.4161/derm.20015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Grant WB. Geographic variation of prostate cancer mortality rates in the United States: implications for prostate cancer risk related to vitamin D. Int J Cancer. 2004;111(3):470–471. doi: 10.1002/ijc.20220. [DOI] [PubMed] [Google Scholar]
- 126.Durup D, Jorgensen HL, Christensen J, Schwarz P, Heegaard AM, Lind B. A reverse J-shaped association of all-cause mortality with serum 25-hydroxyvitamin D in general practice: the CopD study. J Clin Endocrinol Metab. 2012;97(8):2644–2652. doi: 10.1210/jc.2012-1176. [DOI] [PubMed] [Google Scholar]
- 127.Vieth R. Enzyme kinetics hypothesis to explain the U-shaped risk curve for prostate cancer vs. 25-hydroxyvitamin D in nordic countries. Int J Cancer. 2004;111(3):468. doi: 10.1002/ijc.20218. [DOI] [PubMed] [Google Scholar]
- 128.Holick MF, Binkley NC, Bischoff-Ferrari HA, et al. Guidelines for preventing and treating vitamin D deficiency and insufficiency revisited. J Clin Endocrinol Metab. 2012;97(4):1153–1158. doi: 10.1210/jc.2011-2601. [DOI] [PubMed] [Google Scholar]
- 129.Wactawski-Wende J, Kotchen JM, Anderson GL, et al. Calcium plus vitamin D supplementation and the risk of colorectal cancer. N Engl J Med. 2006;354(7):684–696. doi: 10.1056/NEJMoa055222. [DOI] [PubMed] [Google Scholar]
- 130.Lappe JM, Travers-Gustafson D, Davies KM, Recker RR, Heaney RP. Vitamin D and calcium supplementation reduces cancer risk: results of a randomized trial. Am J Clin Nutr. 2007;85(6):1586–1591. doi: 10.1093/ajcn/85.6.1586. [DOI] [PubMed] [Google Scholar]
- 131.Skinner HG, Michaud DS, Giovannucci E, Willett WC, Colditz GA, Fuchs CS. Vitamin D intake and the risk for pancreatic cancer in two cohort studies. Cancer Epidemiol Biomarkers Prev. 2006;15(9):1688–1695. doi: 10.1158/1055-9965.EPI-06-0206. [DOI] [PubMed] [Google Scholar]
- 132.Stolzenberg-Solomon RZ, Hayes RB, Horst RL, Anderson KE, Hollis BW, Silverman DT. Serum vitamin D and risk of pancreatic cancer in the prostate, lung, colorectal, and ovarian screening trial. Cancer Res. 2009;69(4):1439–1447. doi: 10.1158/0008-5472.CAN-08-2694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Wolpin BM, Ng K, Bao Y, et al. Plasma 25-hydroxyvitamin D and risk of pancreatic cancer. Cancer Epidemiol Biomarkers Prev. 2012;21(1):82–91. doi: 10.1158/1055-9965.EPI-11-0836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Holick MF. Vitamin d, sunlight and cancer connection. Anticancer Agents Med Chem. 2013;13(1):70–82. [PubMed] [Google Scholar]
- 135.Grant WB. An estimate of the global reduction in mortality rates through doubling vitamin D levels. Eur J Clin Nutr. 2011;65(9):1016–1026. doi: 10.1038/ejcn.2011.68. [DOI] [PubMed] [Google Scholar]
- 136.Grant WB. Lower vitamin-D production from solar ultraviolet-B irradiance may explain some differences in cancer survival rates. J Natl Med Assoc. 2006;98(3):357–364. [PMC free article] [PubMed] [Google Scholar]
- 137.Helzlsouer KJ. Overview of the Cohort Consortium Vitamin D Pooling Project of Rarer Cancers. Am J Epidemiol. 2010;172(1):4–9. doi: 10.1093/aje/kwq119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Grant WB. Ecological studies of the UVB-vitamin D-cancer hypothesis. Anticancer Res. 2012;32(1):223–236. [PubMed] [Google Scholar]
- 139.Pilz S, Tomaschitz A, Marz W, et al. Vitamin D, cardiovascular disease and mortality. Clin Endocrinol. 2011;75(5):575–584. doi: 10.1111/j.1365-2265.2011.04147.x. [DOI] [PubMed] [Google Scholar]
- 140.Luxwolda MF, Kuipers RS, Kema IP, Dijck-Brouwer DA, Muskiet FA. Traditionally living populations in East Africa have a mean serum 25-hydroxyvitamin D concentration of 115 nmol/l. Br J Nutr. 2012;108(9):1557–1561. doi: 10.1017/S0007114511007161. [DOI] [PubMed] [Google Scholar]
- 141.Melamed ML, Michos ED, Post W, Astor B. 25-Hydroxyvitamin D levels and the risk of mortality in the general population. Arch Intern Med. 2008;168(15):1629–1637. doi: 10.1001/archinte.168.15.1629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Zittermann A, Iodice S, Pilz S, Grant WB, Bagnardi V, Gandini S. Vitamin D deficiency and mortality risk in the general population: a meta-analysis of prospective cohort studies. Am J Clin Nutr. 2012;95(1):91–100. doi: 10.3945/ajcn.111.014779. [DOI] [PubMed] [Google Scholar]
- 143.Ma Y, Zhang P, Wang F, Yang J, Liu Z, Qin H. Association between vitamin D and risk of colorectal cancer: a systematic review of prospective studies. J Clin Oncol. 2011;29(28):3775–3782. doi: 10.1200/JCO.2011.35.7566. [DOI] [PubMed] [Google Scholar]
- 144.Pilz S, Iodice S, Zittermann A, Grant WB, Gandini S. Vitamin D status and mortality risk in CKD: a meta-analysis of prospective studies. Am J Kidney Dis. 2011;58(3):374–382. doi: 10.1053/j.ajkd.2011.03.020. [DOI] [PubMed] [Google Scholar]
- 145.Rejnmark L, Avenell A, Masud T, et al. Vitamin D with calcium reduces mortality: patient level pooled analysis of 70,528 patients from eight major vitamin D trials. J Clin Endocrinol Metab. 2012;97(8):2670–2681. doi: 10.1210/jc.2011-3328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Thomas GN, o Hartaigh B, Bosch JA, et al. Vitamin D levels predict all-cause and cardiovascular disease mortality in subjects with the metabolic syndrome: the Ludwigshafen Risk and Cardiovascular Health (LURIC) Study. Diabetes Care. 2012;35(5):1158–1164. doi: 10.2337/dc11-1714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Doherty TM, Tang W, Dascalos S, et al. Ethnic origin and serum levels of 1α,25-dihydroxyvitamin D3 are independent predictors of coronary calcium mass measured by electron-beam computed tomography. Circulation. 1997;96(5):1477–1481. doi: 10.1161/01.cir.96.5.1477. [DOI] [PubMed] [Google Scholar]
- 148.Watson KE, Abrolat ML, Malone LL, et al. Active serum vitamin D levels are inversely correlated with coronary calcification. Circulation. 1997;96(6):1755–1760. doi: 10.1161/01.cir.96.6.1755. [DOI] [PubMed] [Google Scholar]
- 149.Cigolini M, Iagulli MP, Miconi V, Galiotto M, Lombardi S, Targher G. Serum 25-hydroxyvitamin D3 concentrations and prevalence of cardiovascular disease among type 2 diabetic patients. Diabetes Care. 2006;29(3):722–724. doi: 10.2337/diacare.29.03.06.dc05-2148. [DOI] [PubMed] [Google Scholar]
- 150.Arnson Y, Itzhaky D, Mosseri M, et al. Vitamin D inflammatory cytokines and coronary events: a comprehensive review. Clin Rev Allergy Immunol. doi: 10.1007/s12016-013-8356-0. [published online January 12, 2013] [DOI] [PubMed] [Google Scholar]
- 151.Grundmann M, Haidar M, Placzko S, et al. Vitamin D improves the angiogenic properties of endothelial progenitor cells. Am J Physiol Cell Physiol. 2012;303(9):29. doi: 10.1152/ajpcell.00030.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Wang TJ, Pencina MJ, Booth SL, et al. Vitamin D deficiency and risk of cardiovascular disease. Circulation. 2008;117(4):503–511. doi: 10.1161/CIRCULATIONAHA.107.706127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Anderson JL, May HT, Horne BD, et al. Relation of vitamin D deficiency to cardiovascular risk factors, disease status, and incident events in a general healthcare population. Am J Cardiol. 2010;106(7):963–968. doi: 10.1016/j.amjcard.2010.05.027. [DOI] [PubMed] [Google Scholar]
- 154.Tamez H, Kalim S, Thadhani RI. Does vitamin D modulate blood pressure? Curr Opin Nephrol Hypertens. 2013;22(2):204–209. doi: 10.1097/MNH.0b013e32835d919b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Dong Y, Stallmann-Jorgensen IS, Pollock NK, et al. A 16-week randomized clinical trial of 2000 international units daily vitamin D3 supplementation in black youth: 25-hydroxyvitamin D, adiposity, and arterial stiffness. J Clin Endocrinol Metab. 2010;95(10):4584–4591. doi: 10.1210/jc.2010-0606. [DOI] [PubMed] [Google Scholar]
- 156.Kumar J, Muntner P, Kaskel FJ, Hailpern SM, Melamed ML. Prevalence and associations of 25-hydroxyvitamin D deficiency in US children: NHANES 2001–2004. Pediatrics. 2009;124(3):e362–e370. doi: 10.1542/peds.2009-0051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Forouhi NG, Ye Z, Rickard AP, et al. Circulating 25-hydroxyvitamin D concentration and the risk of type 2 diabetes: results from the European Prospective Investigation into Cancer (EPIC)-Norfolk cohort and updated meta-analysis of prospective studies. Diabetologia. 2012;55(8):2173–2182. doi: 10.1007/s00125-012-2544-y. [DOI] [PubMed] [Google Scholar]
- 158.Welsh P, Doolin O, McConnachie A, et al. Circulating 25OHD, dietary vitamin D, PTH, and calcium associations with incident cardiovascular disease and mortality: the MIDSPAN Family Study. J Clin Endocrinol Metab. 2012;97(12):4578–4587. doi: 10.1210/jc.2012-2272. [DOI] [PubMed] [Google Scholar]
- 159.Agmon-Levin N, Theodor E, Segal RM, Shoenfeld Y. Vitamin D in systemic and organ-specific autoimmune diseases. Clin Rev Allergy Immunol. doi: 10.1007/s12016-012-8342-y. [published online December 14, 2012] [DOI] [PubMed] [Google Scholar]
- 160.Higgins MJ, Mackie SL, Thalayasingam N, Bingham SJ, Hamilton J, Kelly CA. The effect of vitamin D levels on the assessment of disease activity in rheumatoid arthritis. Clin Rheumatol. doi: 10.1007/s10067-013-2174-x. [published online January 23, 2013] [DOI] [PubMed] [Google Scholar]
- 161.Szekely JI, Pataki A. Effects of vitamin D on immune disorders with special regard to asthma, COPD and autoimmune diseases: a short review. Expert Rev Respir Med. 2012;6(6):683–704. doi: 10.1586/ers.12.57. [DOI] [PubMed] [Google Scholar]
- 162.Cantorna MT, Mahon BD. Mounting evidence for vitamin D as an environmental factor affecting autoimmune disease prevalence. Exp Biol Med. 2004;229(11):1136–1142. doi: 10.1177/153537020422901108. [DOI] [PubMed] [Google Scholar]
- 163.Villaggio B, Soldano S, Cutolo M. 1,25-dihydroxyvitamin D3 downregulates aromatase expression and inflammatory cytokines in human macrophages. Clin Exp Rheumatol. 2012;30(6):934–938. [PubMed] [Google Scholar]
- 164.Merlino LA, Curtis J, Mikuls TR, Cerhan JR, Criswell LA, Saag KG. Vitamin D intake is inversely associated with rheumatoid arthritis: results from the Iowa Women’s Health Study. Arthritis Rheum. 2004;50(1):72–77. doi: 10.1002/art.11434. [DOI] [PubMed] [Google Scholar]
- 165.Andjelkovic Z, Vojinovic J, Pejnovic N, et al. Disease modifying and immunomodulatory effects of high dose 1 a (OH) D3 in rheumatoid arthritis patients. Clin Exp Rheumatol. 1999;17(4):453–456. [PubMed] [Google Scholar]
- 166.Sen D, Ranganathan P. Vitamin D in rheumatoid arthritis: panacea or placebo? Discov Med. 2012;14(i78):311–319. [PubMed] [Google Scholar]
- 167.Haga HJ, Schmedes A, Naderi Y, Moreno AM, Peen E. Severe deficiency of 25-hydroxyvitamin D3 (25-OH-D3) is associated with high disease activity of rheumatoid arthritis. Clin Rheumatol. 2013;15:15. doi: 10.1007/s10067-012-2154-6. [DOI] [PubMed] [Google Scholar]
- 168.Schwalfenberg GK. Solar radiation and vitamin D: mitigating environmental factors in autoimmune disease. J Environ Public Health. 2012;2012:619381. doi: 10.1155/2012/619381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Knip M, Akerblom HK. Early nutrition and later diabetes risk. Adv Exp Med Biol. 2005;569:142–150. doi: 10.1007/1-4020-3535-7_21. [DOI] [PubMed] [Google Scholar]
- 170.Stene LC, Ulriksen J, Magnus P, Joner G. Use of cod liver oil during pregnancy associated with lower risk of type I diabetes in the offspring. Diabetologia. 2000;43(9):1093–1098. doi: 10.1007/s001250051499. [DOI] [PubMed] [Google Scholar]
- 171.Hypponen E, Laara E, Reunanen A, Jarvelin MR, Virtanen SM. Intake of vitamin D and risk of type 1 diabetes: a birth-cohort study. Lancet. 2001;358(9292):1500–1503. doi: 10.1016/S0140-6736(01)06580-1. [DOI] [PubMed] [Google Scholar]
- 172.Rajakumar K, de las Heras J, Lee S, Holick MF, Arslanian SA. 25-Hydroxyvitamin D concentrations and in vivo insulin sensitivity and β-cell function relative to insulin sensitivity in black and white youth. Diabetes Care. 2012;35(3):627–633. doi: 10.2337/dc11-1825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Simon KC, Munger KL, Ascherio A. Vitamin D and multiple sclerosis: epidemiology, immunology, and genetics. Curr Opin Neurol. 2012;25(3):246–251. doi: 10.1097/WCO.0b013e3283533a7e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Munger KL, Zhang SM, O’Reilly E, et al. Vitamin D intake and incidence of multiple sclerosis. Neurology. 2004;62(1):60–65. doi: 10.1212/01.wnl.0000101723.79681.38. [DOI] [PubMed] [Google Scholar]
- 175.Runia TF, Hop WC, de Rijke YB, Buljevac D, Hintzen RQ. Lower serum vitamin D levels are associated with a higher relapse risk in multiple sclerosis. Neurology. 2012;79(3):261–266. doi: 10.1212/WNL.0b013e31825fdec7. [DOI] [PubMed] [Google Scholar]
- 176.Weinstock-Guttman B, Mehta BK, Ramanathan M, et al. Vitamin D and multiple sclerosis. Neurologist. 2012;18(4):179–183. doi: 10.1097/NRL.0b013e31825bbf35. [DOI] [PubMed] [Google Scholar]
- 177.Kimball SM, Ursell MR, O’Connor P, Vieth R. Safety of vitamin D3 in adults with multiple sclerosis. Am J Clin Nutr. 2007;86(3):645–651. doi: 10.1093/ajcn/86.3.645. [DOI] [PubMed] [Google Scholar]
- 178.Stein MS, Liu Y, Gray OM, et al. A randomized trial of high-dose vitamin D2 in relapsing-remitting multiple sclerosis. Neurology. 2011;77(17):1611–1618. doi: 10.1212/WNL.0b013e3182343274. [DOI] [PubMed] [Google Scholar]
- 179.Burton JM, Kimball S, Vieth R, et al. A phase I/II dose-escalation trial of vitamin D3 and calcium in multiple sclerosis. Neurology. 2010;74(23):1852–1859. doi: 10.1212/WNL.0b013e3181e1cec2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Ramagopalan SV, Heger A, Berlanga AJ, et al. A ChIP-seq defined genome-wide map of vitamin D receptor binding: associations with disease and evolution. Genome Res. 2010;20(10):1352–1360. doi: 10.1101/gr.107920.110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Jorgensen SP, Agnholt J, Glerup H, et al. Clinical trial: vitamin D3 treatment in Crohn’s disease: a randomized double-blind placebo-controlled study. Aliment Pharmacol Ther. 2010;32(3):377–383. doi: 10.1111/j.1365-2036.2010.04355.x. [DOI] [PubMed] [Google Scholar]
- 182.Bartels LE, Jorgensen SP, Bendix M, et al. 25-Hydroxyvitamin D3 modulates dendritic cell phenotype and function in Crohn’s disease. Inflammopharmacology. 2013;21(2):177–186. doi: 10.1007/s10787-012-0168-y. [DOI] [PubMed] [Google Scholar]
- 183.Holick MF. Resurrection of vitamin D deficiency and rickets. J Clin Invest. 2006;116(8):2062–2072. doi: 10.1172/JCI29449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Camargo CA, Jr., Ingham T, Wickens K, et al. Cord-blood 25-hydroxyvitamin D levels and risk of respiratory infection, wheezing, and asthma. Pediatrics. 2011;127(1):e180–e187. doi: 10.1542/peds.2010-0442. [DOI] [PubMed] [Google Scholar]
- 185.Sabetta JR, DePetrillo P, Cipriani RJ, Smardin J, Burns LA, Landry ML. Serum 25-hydroxyvitamin D and the incidence of acute viral respiratory tract infections in healthy adults. PLoS One. 2010;5(6):0011088. doi: 10.1371/journal.pone.0011088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Urashima M, Segawa T, Okazaki M, Kurihara M, Wada Y, Ida H. Randomized trial of vitamin D supplementation to prevent seasonal influenza A in schoolchildren. Am J Clin Nutr. 2010;91(5):1255–1260. doi: 10.3945/ajcn.2009.29094. [DOI] [PubMed] [Google Scholar]
- 187.Sundar IK, Rahman I. Vitamin D and susceptibility of chronic lung diseases: role of epigenetics. Front Pharmacol. 2011;2:50. doi: 10.3389/fphar.2011.00050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Wjst M. Is vitamin D supplementation responsible for the allergy pandemic? Curr Opin Allergy Clin Immunol. 2012;12(3):257–262. doi: 10.1097/ACI.0b013e3283535833. [DOI] [PubMed] [Google Scholar]
- 189.Tolppanen AM, Williams D, Henderson J, Lawlor DA. Serum 25-hydroxy-vitamin D and ionised calcium in relation to lung function and allergen skin tests. Eur J Clin Nutr. 2011;65(4):493–500. doi: 10.1038/ejcn.2011.6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Jones AP, Palmer D, Zhang G, Prescott SL. Cord blood 25-hydroxyvitamin D3 and allergic disease during infancy. Pediatrics. 2012;130(5):e1128–e1135. doi: 10.1542/peds.2012-1172. [DOI] [PubMed] [Google Scholar]
- 191.Rochat MK, Ege MJ, Plabst D, et al. Maternal vitamin D intake during pregnancy increases gene expression of ILT3 and ILT4 in cord blood. Clin Exp Allergy. 2010;40(5):786–794. doi: 10.1111/j.1365-2222.2009.03428.x. [DOI] [PubMed] [Google Scholar]
- 192.Eyles DW, Burne TH, McGrath JJ. Vitamin D, effects on brain development, adult brain function and the links between low levels of vitamin D and neuropsychiatric disease. Front Neuroendocrinol. 2013;34(1):47–64. doi: 10.1016/j.yfrne.2012.07.001. [DOI] [PubMed] [Google Scholar]
- 193.Bartoccini E, Marini F, Damaskopoulou E, et al. Nuclear lipid microdomains regulate nuclear vitamin D3 uptake and influence embryonic hippocampal cell differentiation. Mol Biol Cell. 2011;22(17):3022–3031. doi: 10.1091/mbc.E11-03-0196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Gale CR, Robinson SM, Harvey NC, et al. Maternal vitamin D status during pregnancy and child outcomes. Eur J Clin Nutr. 2008;62(1):68–77. doi: 10.1038/sj.ejcn.1602680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.O’Loan J, Eyles DW, Kesby J, Ko P, McGrath JJ, Burne TH. Vitamin D deficiency during various stages of pregnancy in the rat; its impact on development and behaviour in adult offspring. Psychoneuroendocrinology. 2007;32(3):227–234. doi: 10.1016/j.psyneuen.2006.12.006. [DOI] [PubMed] [Google Scholar]
- 196.Becker A, Eyles DW, McGrath JJ, Grecksch G. Transient prenatal vitamin D deficiency is associated with subtle alterations in learning and memory functions in adult rats. Behav Brain Res. 2005;161(2):306–312. doi: 10.1016/j.bbr.2005.02.015. [DOI] [PubMed] [Google Scholar]
- 197.McGrath J, Saari K, Hakko H, et al. Vitamin D supplementation during the first year of life and risk of schizophrenia: a Finnish birth cohort study. Schizophr Res. 2004;67(2–3):237–245. doi: 10.1016/j.schres.2003.08.005. [DOI] [PubMed] [Google Scholar]
- 198.Newschaffer CJ, Croen LA, Daniels J, et al. The epidemiology of autism spectrum disorders. Annu Rev Public Health. 2007;28:235–258. doi: 10.1146/annurev.publhealth.28.021406.144007. [DOI] [PubMed] [Google Scholar]
- 199.Yu J, Gattoni-Celli M, Zhu H, et al. Vitamin D3-enriched diet correlates with a decrease of amyloid plaques in the brain of AβPP transgenic mice. J Alzheimers Dis. 2011;25(2):295–307. doi: 10.3233/JAD-2011-101986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Whitehouse AJ, Holt BJ, Serralha M, Holt PG, Kusel MM, Hart PH. Maternal serum vitamin D levels during pregnancy and offspring neurocognitive development. Pediatrics. 2012;129(3):485–493. doi: 10.1542/peds.2011-2644. [DOI] [PubMed] [Google Scholar]
- 201.Groves NJ, Kesby JP, Eyles DW, McGrath JJ, Mackay-Sim A, Burne TH. Adult vitamin D deficiency leads to behavioural and brain neurochemical alterations in C57BL/6J and BALB/c mice. Behav Brain Res. 2013;241:120–131. doi: 10.1016/j.bbr.2012.12.001. [DOI] [PubMed] [Google Scholar]
- 202.Maddock J, Berry DJ, Geoffroy MC, Power C, Hypponen E. Vitamin D and common mental disorders in mid-life: cross-sectional and prospective findings. Clin Nutr. 2013;21(13):00030–00037. doi: 10.1016/j.clnu.2013.01.006. [DOI] [PubMed] [Google Scholar]
- 203.Anglin RE, Samaan Z, Walter SD, McDonald SD. Vitamin D deficiency and depression in adults: systematic review and meta-analysis. Br J Psychiatry. 2013;202:100–107. doi: 10.1192/bjp.bp.111.106666. [DOI] [PubMed] [Google Scholar]
- 204.Hoogendijk WJ, Lips P, Dik MG, Deeg DJ, Beekman AT, Penninx BW. Depression is associated with decreased 25-hydroxyvitamin D and increased parathyroid hormone levels in older adults. Arch Gen Psychiatry. 2008;65(5):508–512. doi: 10.1001/archpsyc.65.5.508. [DOI] [PubMed] [Google Scholar]
- 205.Armstrong DJ, Meenagh GK, Bickle I, Lee AS, Curran ES, Finch MB. Vitamin D deficiency is associated with anxiety and depression in fibromyalgia. Clin Rheumatol. 2007;26(4):551–554. doi: 10.1007/s10067-006-0348-5. [DOI] [PubMed] [Google Scholar]
- 206.Jorde R, Waterloo K, Saleh F, Haug E, Svartberg J. Neuropsychological function in relation to serum parathyroid hormone and serum 25-hydroxyvitamin D levels: the Tromso study. J Neurol. 2006;253(4):464–470. doi: 10.1007/s00415-005-0027-5. [DOI] [PubMed] [Google Scholar]
- 207.Lansdowne AT, Provost SC. Vitamin D3 enhances mood in healthy subjects during winter. Psychopharmacology. 1998;135(4):319–323. doi: 10.1007/s002130050517. [DOI] [PubMed] [Google Scholar]
- 208.Gloth FMIII, Alam W, Hollis B. Vitamin D vs broad spectrum phototherapy in the treatment of seasonal affective disorder. J Nutr Health Aging. 1999;3(1):5–7. [PubMed] [Google Scholar]
- 209.Wei SQ, Qi HP, Luo ZC, Fraser WD. Maternal vitamin D status and adverse pregnancy outcomes: a systematic review and meta-analysis. J Matern Fetal Neonatal Med. 2013;26(9):889–899. doi: 10.3109/14767058.2013.765849. [DOI] [PubMed] [Google Scholar]
- 210.Merewood A, Mehta SD, Chen TC, Bauchner H, Holick MF. Association between vitamin D deficiency and primary cesarean section. J Clin Endocrinol Metab. 2009;94(3):940–945. doi: 10.1210/jc.2008-1217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Poel YH, Hummel P, Lips P, Stam F, van der Ploeg T, Simsek S. Vitamin D and gestational diabetes: a systematic review and meta-analysis. Eur J Intern Med. 2012;23(5):465–469. doi: 10.1016/j.ejim.2012.01.007. [DOI] [PubMed] [Google Scholar]
- 212.Hollis BW, Horst RL. The assessment of circulating 25(OH)D and 1,25(OH)2D: where we are and where we are going. J Steroid Biochem Mol Biol. 2007;103(3–5):473–476. doi: 10.1016/j.jsbmb.2006.11.00. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Schottker B, Jansen EH, Haug U, Schomburg L, Kohrle J, Brenner H. Standardization of misleading immunoassay based 25-hydroxyvitamin D levels with liquid chromatography tandem-mass spectrometry in a large cohort study. PLoS One. 2012;7(11):e48774. doi: 10.1371/journal.pone.0048774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Binkley N, Krueger D, Cowgill CS, et al. Assay variation confounds the diagnosis of hypovitaminosis D: a call for standardization. J Clin Endocrinol Metab. 2004;89(7):3152–3157. doi: 10.1210/jc.2003-031979. [DOI] [PubMed] [Google Scholar]
- 215.Carter GD, Carter R, Jones J, Berry J. How accurate are assays for 25-hydroxyvitamin D? data from the international vitamin D external quality assessment scheme. Clin Chem. 2004;50(11):2195–2197. doi: 10.1373/clinchem.2004.040683. [DOI] [PubMed] [Google Scholar]
- 216.Singh RJ, Taylor RL, Reddy GS, Grebe SK. C-3 epimers can account for a significant proportion of total circulating 25-hydroxyvitamin D in infants, complicating accurate measurement and interpretation of vitamin D status. J Clin Endocrinol Metab. 2006;91(8):3055–3061. doi: 10.1210/jc.2006-0710. [DOI] [PubMed] [Google Scholar]
- 217.Looker AC, Johnson CL, Lacher DA, Pfeiffer CM, Schleicher RL, Sempos CT. Vitamin D status: United States, 2001–2006. NCHS Data Brief. 2011;59:1–8. [PubMed] [Google Scholar]
- 218.Centers for Disease Control and Prevention (CDC) Revised analytical note for NHANES 2000–2006 and NHANES III (1988–1994) 25-hydroxyvitamin D analysis. http://www.cdc.gov/nchs/data/nhanes/nhanes3/VitaminD_analyticnote.pdf. Revised November 2010. [Google Scholar]
- 219.Carter GD. 25-Hydroxyvitamin D assays: the quest for accuracy. 2009;55(7):1300–1302. doi: 10.1373/clinchem.2009.125906. [DOI] [PubMed] [Google Scholar]
- 220.Sempos CT, Vesper HW, Phinney KW, Thienpont LM, Coates PM. Vitamin D status as an international issue: national surveys and the problem of standardization. Scand J Clin Lab Invest Suppl. 2012;243:32–40. doi: 10.3109/00365513.2012.681935. [DOI] [PubMed] [Google Scholar]
- 221.Yetley EA, Pfeiffer CM, Schleicher RL, et al. NHANES monitoring of serum 25-hydroxyvitamin D: a roundtable summary. J Nutr. 2010;140(11):2030S–2045S. doi: 10.3945/jn.110.121483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Fraser WD, Milan AM. Vitamin D assays: past and present debates, difficulties, and developments. Calcif Tissue Int. 2013;92(2):118–127. doi: 10.1007/s00223-012-9693-3. [DOI] [PubMed] [Google Scholar]
- 223.Glendenning P, Taranto M, Noble JM, et al. Current assays overestimate 25-hydroxyvitamin D3 and underestimate 25-hydroxyvitamin D2 compared with HPLC: need for assay-specific decision limits and metabolite-specific assays. Ann Clin Biochem. 2006;43(pt 1):23–30. doi: 10.1258/000456306775141650. [DOI] [PubMed] [Google Scholar]
- 224.Thandrayen K, Pettifor JM. Maternal vitamin D status: implications for the development of infantile nutritional rickets. Rheum Dis Clin North Am. 2012;38(1):61–79. doi: 10.1016/j.rdc.2012.03.007. [DOI] [PubMed] [Google Scholar]
- 225.Shivane VK, Sarathi V, Bandgar T, Menon P, Shah NS. High prevalence of hypovitaminosis D in young healthy adults from the western part of India. Postgrad Med J. 2011;87(1030):514–518. doi: 10.1136/pgmj.2010.113092. [DOI] [PubMed] [Google Scholar]
- 226.van Schoor NM, Lips P. Worldwide vitamin D status. Best Pract Res Clin Endocrinol Metab. 2011;25(4):671–680. doi: 10.1016/j.beem.2011.06.007. [DOI] [PubMed] [Google Scholar]
- 227.Fields J, Trivedi NJ, Horton E, Mechanick JI. Vitamin D in the Persian Gulf: integrative physiology and socioeconomic factors. Curr Osteoporos Rep. 2011;9(4):243–250. doi: 10.1007/s11914-011-0071-2. [DOI] [PubMed] [Google Scholar]
- 228.Ganji V, Zhang X, Tangpricha V. Serum 25-hydroxyvitamin D concentrations and prevalence estimates of hypovitaminosis D in the U.S. population based on assay-adjusted data. J Nutr. 2012;142(3):498–507. doi: 10.3945/jn.111.151977. [DOI] [PubMed] [Google Scholar]
- 229.Greene-Finestone LS, Berger C, de Groh M, et al. 25-Hydroxyvitamin D in Canadian adults: biological, environmental, and behavioral correlates. Osteoporos Int. 2011;22(5):1389–1399. doi: 10.1007/s00198-010-1362-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Gonzalez-Gross M, Valtuena J, Breidenassel C, et al. Vitamin D status among adolescents in Europe: the Healthy Lifestyle in Europe by Nutrition in Adolescence study. Br J Nutr. 2012;107(5):755–764. doi: 10.1017/S0007114511003527. [DOI] [PubMed] [Google Scholar]
- 231.Schlingmann KP, Kaufmann M, Weber S, et al. Mutations in CYP24A1 and idiopathic infantile hypercalcemia. N Engl J Med. 2011;365(5):410–421. doi: 10.1056/NEJMoa1103864. [DOI] [PubMed] [Google Scholar]
- 232.Daly RM, Gagnon C, Lu ZX, et al. Prevalence of vitamin D deficiency and its determinants in Australian adults aged 25 years and older: a national, population-based study. Clin Endocrinol. 2012;77(1):26–35. doi: 10.1111/j.1365-2265.2011.04320.x. [DOI] [PubMed] [Google Scholar]
- 233.Clemens TL, Adams JS, Henderson SL, Holick MF. Increased skin pigment reduces the capacity of skin to synthesise vitamin D3 . Lancet. 1982;1(8263):74–76. doi: 10.1016/s0140-6736(82)90214-8. [DOI] [PubMed] [Google Scholar]
- 234.Manicourt DH, Devogelaer JP. Urban tropospheric ozone increases the prevalence of vitamin D deficiency among Belgian postmenopausal women with outdoor activities during summer. J Clin Endocrinol Metab. 2008;93(10):3893–3899. doi: 10.1210/jc.2007-2663. [DOI] [PubMed] [Google Scholar]
- 235.Holick MF. Vitamin D: evolutionary, physiological and health perspectives. Curr Drug Targets. 2011;12(1):4–18. doi: 10.2174/138945011793591635. [DOI] [PubMed] [Google Scholar]
- 236.van den Berg G, van Eijsden M, Vrijkotte TG, Gemke RJ. Suboptimal maternal vitamin D status and low education level as determinants of small-for-gestational-age birth weight. Eur J Nutr. 2013;52(1):273–279. doi: 10.1007/s00394-012-0327-3. [DOI] [PubMed] [Google Scholar]
- 237.Godar DE, Pope SJ, Grant WB, Holick MF. Solar UV doses of young Americans and vitamin D3 production. Environ Health Perspect. 2012;120(1):139–143. doi: 10.1289/ehp.1003195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.MacLaughlin J, Holick MF. Aging decreases the capacity of human skin to produce vitamin D3 . J Clin Invest. 1985;76(4):1536–1538. doi: 10.1172/JCI112134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Signorello LB, Shi J, Cai Q, et al. Common variation in vitamin D pathway genes predicts circulating 25-hydroxyvitamin D levels among African Americans. PLoS One. 2011;6(12):e28623. doi: 10.1371/journal.pone.0028623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Ahn J, Yu K, Stolzenberg-Solomon R, et al. Genome-wide association study of circulating vitamin D levels. Hum Mol Genet. 2010;19(13):2739–2745. doi: 10.1093/hmg/ddq155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Wang TJ, Zhang F, Richards JB, et al. Common genetic determinants of vitamin D insufficiency: a genome-wide association study. Lancet. 2010;376(9736):180–188. doi: 10.1016/S0140-6736(10)60588-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Engelman CD, Fingerlin TE, Langefeld CD, et al. Genetic and environmental determinants of 25-hydroxyvitamin D and 1,25-dihydroxyvitamin D levels in Hispanic and African Americans. J Clin Endocrinol Metab. 2008;93(9):3381–3388. doi: 10.1210/jc.2007-2702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Zhu H, Wang X, Shi H, et al. A genome-wide methylation study of severe vitamin D deficiency in African American adolescents. J Pediatr. 2013;162(5):1004–1009. doi: 10.1016/j.jpeds.2012.10.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Hiraki LT, Major JM, Chen C, et al. Exploring the genetic architecture of circulating 25-hydroxyvitamin D. Genet Epidemiol. 2013;37(1):92–98. doi: 10.1002/gepi.21694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Holick M. Vitamin D and health: evolution, biologic functions, and recommended dietary intakes for vitamin D. Clin Rev Bone Miner Metab. 2009;7:2–19. [Google Scholar]
- 246.Kennedy C, Bajdik CD, Willemze R, De Gruijl FR, Bouwes Bavinck JN. The influence of painful sunburns and lifetime sun exposure on the risk of actinic keratoses, seborrheic warts, melanocytic nevi, atypical nevi, and skin cancer. J Invest Dermatol. 2003;120(6):1087–1093. doi: 10.1046/j.1523-1747.2003.12246.x. [DOI] [PubMed] [Google Scholar]
- 247.Garland FC, White MR, Garland CF, Shaw E, Gorham ED. Occupational sunlight exposure and melanoma in the U.S. Navy. Arch Environ Health. 1990;45(5):261–267. doi: 10.1080/00039896.1990.10118743. [DOI] [PubMed] [Google Scholar]
- 248.Tangpricha V, Turner A, Spina C, Decastro S, Chen TC, Holick MF. Tanning is associated with optimal vitamin D status (serum 25-hydroxyvitamin D concentration) and higher bone mineral density. Am J Clin Nutr. 2004;80(6):1645–1649. doi: 10.1093/ajcn/80.6.1645. [DOI] [PubMed] [Google Scholar]
- 249.Grant WB. In defense of the sun: an estimate of changes in mortality rates in the United States if mean serum 25-hydroxyvitamin D levels were raised to 45 ng/mL by solar ultraviolet-B irradiance. Dermatoendocrinol. 2009;1(4):207–214. doi: 10.4161/derm.1.4.9841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Dabai NS, Pramyothin P, Holick MF. The effect of ultraviolet radiation from a novel portable fluorescent lamp on serum 25-hydroxyvitamin D3 levels in healthy adults with Fitzpatrick skin types II and III. Photodermatol Photoimmunol Photomed. 2012;28(6):307–311. doi: 10.1111/phpp.12000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Moore C, Murphy MM, Keast DR, Holick MF. Vitamin D intake in the United States. J Am Diet Assoc. 2004;104(6):980–983. doi: 10.1016/j.jada.2004.03.028. [DOI] [PubMed] [Google Scholar]
- 252.Black LJ, Seamans KM, Cashman KD, Kiely M. An updated systematic review and meta-analysis of the efficacy of vitamin D food fortification. J Nutr. 2012;142(6):1102–1108. doi: 10.3945/jn.112.158014. [DOI] [PubMed] [Google Scholar]
- 253.Heaney RP, Davies KM, Chen TC, Holick MF, Barger-Lux MJ. Human serum 25-hydroxycholecalciferol response to extended oral dosing with cholecalciferol. Am J Clin Nutr. 2003;77(1):204–210. doi: 10.1093/ajcn/77.1.204. [DOI] [PubMed] [Google Scholar]
- 254.Demetriou ET, Travison TG, Holick MF. Treatment with 50,000 IU vitamin D2 every other week and effect on serum 25-hydroxyvitamin D2, 25-hydroxyvitamin D3, and total 25-hydroxyvitamin D in a clinical setting. Endocr Pract. 2012;18(3):399–402. doi: 10.4158/EP11268.OR. [DOI] [PubMed] [Google Scholar]
- 255.Pietras SM, Obayan BK, Cai MH, Holick MF. Vitamin D2 treatment for vitamin D deficiency and insufficiency for up to 6 years. Arch Intern Med. 2009;169(19):1806–1808. doi: 10.1001/archinternmed.2009.361. [DOI] [PubMed] [Google Scholar]
- 256.Sanders KM, Stuart AL, Williamson EJ, et al. Annual high-dose oral vitamin D and falls and fractures in older women: a randomized controlled trial. JAMA. 2010;303(18):1815–1822. doi: 10.1001/jama.2010.594. [DOI] [PubMed] [Google Scholar]
- 257.Heikinheimo RJ, Inkovaara JA, Harju EJ, et al. Annual injection of vitamin D and fractures of aged bones. Calcif Tissue Int. 1992;51(2):105–110. doi: 10.1007/BF00298497. [DOI] [PubMed] [Google Scholar]
- 258.Trivedi DP, Doll R, Khaw KT. Effect of four monthly oral vitamin D3 (cholecalciferol) supplementation on fractures and mortality in men and women living in the community: randomised double blind controlled trial. BMJ. 2003;326(7387):469. doi: 10.1136/bmj.326.7387.469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Lee JM, Smith JR, Philipp BL, Chen TC, Mathieu J, Holick MF. Vitamin D deficiency in a healthy group of mothers and newborn infants. Clin Pediatr. 2007;46(1):42–44. doi: 10.1177/0009922806289311. [DOI] [PubMed] [Google Scholar]
- 260.Hollis BW, Wagner CL. Vitamin D and pregnancy: skeletal effects, nonskeletal effects, and birth outcomes. Calcif Tissue Int. 2013;92(2):128–139. doi: 10.1007/s00223-012-9607-4. [DOI] [PubMed] [Google Scholar]
- 261.Hollis BW, Wagner CL. Vitamin D requirements during lactation: high-dose maternal supplementation as therapy to prevent hypovitaminosis D for both the mother and the nursing infant. Am J Clin Nutr. 2004;80(6) suppl:1752S–1758S. doi: 10.1093/ajcn/80.6.1752S. [DOI] [PubMed] [Google Scholar]
- 262.Chandra P, Wolfenden LL, Ziegler TR, et al. Treatment of vitamin D deficiency with UV light in patients with malabsorption syndromes: a case series. Photodermatol Photoimmunol Photomed. 2007;23(5):179–185. doi: 10.1111/j.1600-0781.2007.00302.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Biancuzzo RM, Clarke N, Reitz RE, Travison TG, Holick MF. Serum concentrations of 1,25-dihydroxyvitamin D2 and 1,25-dihydroxyvitamin D3 in response to vitamin D2 and vitamin D3 supplementation. J Clin Endocrinol Metab. 2013;98(3):973–979. doi: 10.1210/jc.2012-2114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Holick MF, Biancuzzo RM, Chen TC, et al. Vitamin D2 is as effective as vitamin D3 in maintaining circulating concentrations of 25-hydroxyvitamin D. J Clin Endocrinol Metab. 2008;93(3):677–681. doi: 10.1210/jc.2007-2308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 265.Gordon CM, Williams AL, Feldman HA, et al. Treatment of hypovitaminosis D in infants and toddlers. J Clin Endocrinol Metab. 2008;93(7):2716–2721. doi: 10.1210/jc.2007-2790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Thacher TD, Fischer PR, Obadofin MO, Levine MA, Singh RJ, Pettifor JM. Comparison of metabolism of vitamins D2 and D3 in children with nutritional rickets. J Bone Miner Res. 2010;25(9):1988–1995. doi: 10.1002/jbmr.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Keegan R, Lu Z, Bogusz J, Holick M. Photobiology of vitamin D in mushrooms and its bioavailability in humans. Dermato-Endocrinology. 2013;5:1. doi: 10.4161/derm.23321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 268.Hathcock JN, Shao A, Vieth R, Heaney R. Risk assessment for vitamin D. Am J Clin Nutr. 2007;85(1):6–18. doi: 10.1093/ajcn/85.1.6. [DOI] [PubMed] [Google Scholar]
- 269.Rajakumar K, Reis EC, Holick MF. Dosing error with over-the-counter vitamin D supplement: a risk for vitamin D toxicity in infants. Clin Pediatr. 2013;52(1):82–85. doi: 10.1177/0009922812439245. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.