Abstract
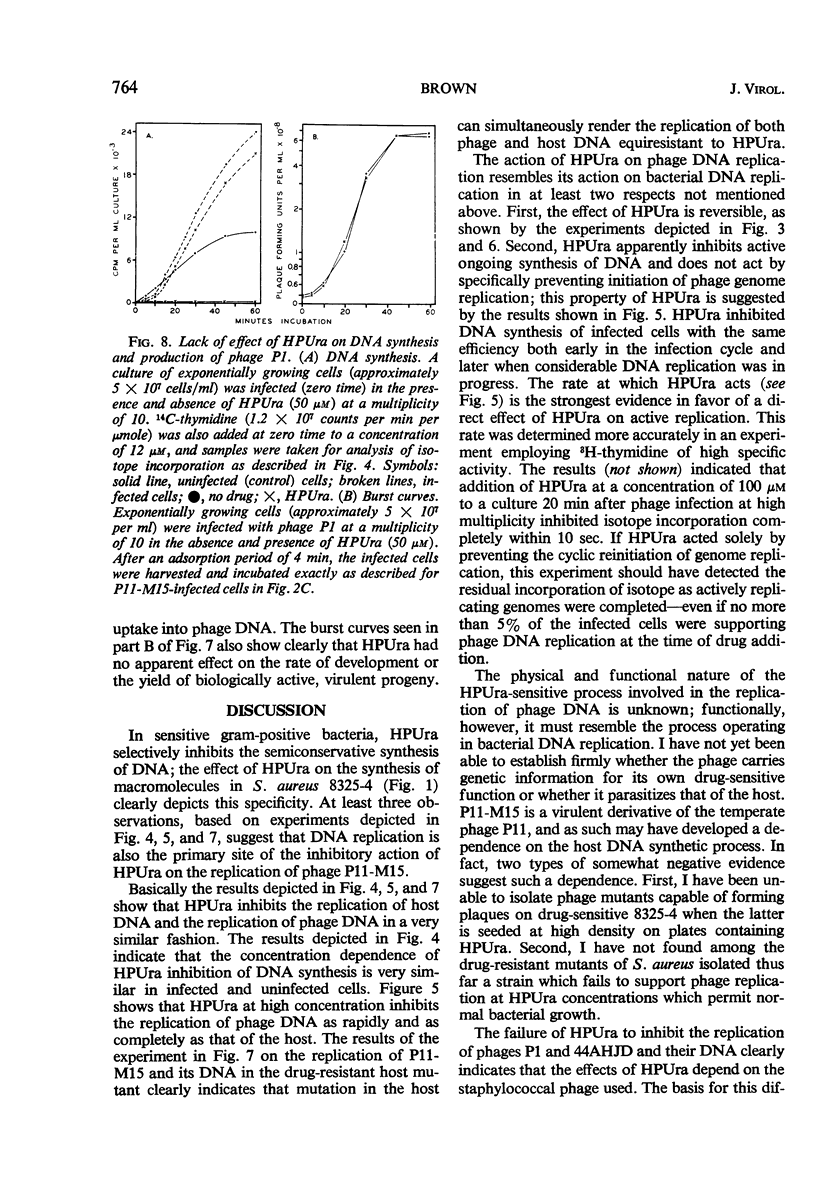
6(p-Hydroxyphenylazo)-uracil (HPUra), a selective inhibitor of the semiconservative replication of deoxyribonucleic acid (DNA) of gram-positive bacteria, was found to inhibit the replication of DNA of bacteriophage P11-M15, a virulent derivative of the temperate Staphylococcus aureus bacteriophage P11. At appropriate concentration, HPUra inhibited DNA synthesis by P11-M15-infected S. aureus immediately and completely, regardless of the stage of the lytic cycle at which infected cells were exposed to drug. The effect of HPUra was reversible since the capacity of inhibited, infected cells to replicate phage DNA and produce mature phage could be restored by removal of HPUra from incubation media. Concentrations of HPUra which completely inhibited the replication of P11-M15 in its drug-sensitive host did not inhibit the replication of this phage or its DNA in several drug-resistant host mutants. HPUra also did not inhibit the replication of two other serologically distinct, virulent staphylococcal bacteriophages, P1 and 44AHJD, in drug-sensitive hosts.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Brown N. C. 6-(p-hydroxyphenylazo)-uracil: a selective inhibitor of host DNA replication in phage-infected Bacillus subtilis. Proc Natl Acad Sci U S A. 1970 Nov;67(3):1454–1461. doi: 10.1073/pnas.67.3.1454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown N. C., Handschumacher R. E. Inhibition of the synthesis of deoxyribonucleic acid in bacteria by 6-(p-hydroxyphenylazo)-2,4-dihydroxypyrimidine. I. Metabolic studies in Streptococcus fecalis. J Biol Chem. 1966 Jul 10;241(13):3083–3089. [PubMed] [Google Scholar]
- Brown N. C. Inhibition of bacterial DNA replication by 6-(p-hydroxyphenylazo)-uracil: differential effect on repair and semi-conservative synthesis in Bacillus subtilis. J Mol Biol. 1971 Jul 14;59(1):1–16. doi: 10.1016/0022-2836(71)90409-8. [DOI] [PubMed] [Google Scholar]
- MAALOE O., HANAWALT P. C. Thymine deficiency and the normal DNA replication cycle. I. J Mol Biol. 1961 Apr;3:144–155. doi: 10.1016/s0022-2836(61)80041-7. [DOI] [PubMed] [Google Scholar]
- Marcus M., Newlon M. C. Control of DNA synthesis in Bacillus subtilis by phage phi e. Virology. 1971 Apr;44(1):83–93. doi: 10.1016/0042-6822(71)90155-3. [DOI] [PubMed] [Google Scholar]
- NOVICK R. P. ANALYSIS BY TRANSDUCTION OF MUTATIONS AFFECTING PENICILLINASE FORMATION IN STAPHYLOCOCCUS AUREUS. J Gen Microbiol. 1963 Oct;33:121–136. doi: 10.1099/00221287-33-1-121. [DOI] [PubMed] [Google Scholar]
- Novick R. Properties of a cryptic high-frequency transducing phage in Staphylococcus aureus. Virology. 1967 Sep;33(1):155–166. doi: 10.1016/0042-6822(67)90105-5. [DOI] [PubMed] [Google Scholar]
- ROODYN D. B., MANDEL H. G. A simple membrane fractionation method for determining the distribution of radioactivity in chemical fractions of Bacillus cereus. Biochim Biophys Acta. 1960 Jun 17;41:80–88. doi: 10.1016/0006-3002(60)90371-1. [DOI] [PubMed] [Google Scholar]
- ROSENBLUM E. D., TYRONE S. SEROLOGY, DENSITY, AND MORPHOLOGY OF STAPHYLOCOCCAL PHAGES. J Bacteriol. 1964 Dec;88:1737–1742. doi: 10.1128/jb.88.6.1737-1742.1964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spizizen J. TRANSFORMATION OF BIOCHEMICALLY DEFICIENT STRAINS OF BACILLUS SUBTILIS BY DEOXYRIBONUCLEATE. Proc Natl Acad Sci U S A. 1958 Oct 15;44(10):1072–1078. doi: 10.1073/pnas.44.10.1072. [DOI] [PMC free article] [PubMed] [Google Scholar]



