Abstract
Anterior cruciate ligament (ACL) deficiency increases the risk of early osteoarthritis (OA). Studies of ACL deficient knee kinematics would be important to reveal the disease process and therefore to find mechanisms which would potentially slow OA progression. The purpose of this study was to determine if in vivo kinematics of the anterior cruciate ligament deficient (ACLD) knee during a wide-based squat activity differ from kinematics of the contralateral intact knee. Thirty-three patients with a unilateral ACLD knee consented to participate in this institutional review board approved study with the contralateral intact knee serving as the control. In vivo knee kinematics during the wide-based squat were analyzed using a 2D/3D registration technique utilizing CT-based bone models and lateral fluoroscopy. Comparisons were performed using values between 0 and 100° flexion both in flexion and extension phases of the squat activity. Both the ACLD and intact knees demonstrated increasing tibial internal rotation with knee flexion, and no difference was observed in tibial rotation between the groups. The tibia in the ACLD knee was more anterior than that of the contralateral knees at 0 and 5° flexion in both phases (p < 0.05). Tibiofemoral medial contact points of the ACLD knees were more posterior than that of the contralateral knees at 5, 10 and 15° of knee flexion in the extension phase of the squat activity (p < 0.05). Tibiofemoral lateral contact points of the ACLD knees were more posterior than that of the contralateral knees at 0° flexion in the both phases (p < 0.05). The kinematics of the ACLD and contralateral intact knees were similar during the wide-based squat except at the low flexion angles. Therefore, we conclude the wide-based squat may be recommended for the ACLD knee by avoiding terminal extension.
Key points
In vivo knee kinematics during the wide-based squat was analyzed using a 2D/3D registration technique utilizing CT-based bone models and lateral fluoroscopy.
Significant differences of in vivo knee kinematics between the ACLD and contralateral knees were detected at low flexion angles.
The wide-based squat is considered a safe exercise for the ACLD knee.
Key words: 2D/3D registration technique, anterior cruciate ligament deficient knee, in vivo knee kinematics, wide-based squat activity.
Introduction
Anterior cruciate ligament (ACL) deficiency increases the risk of early knee osteoarthritis (OA). Radiographic OA changes developed in 50 to 70% of ACL deficient (ACLD) knees within ten to twenty years after ACL injury (Gillquist and Messner, 1999, Roos et al., 1995). All of the ACLD knees demonstrated some evidence of OA changes such as joint space narrowing within 12 years following their ACL injury (Segawa et al., 2001). Since rotational and anterior instability after ACL injury are considered to contribute to early development of knee OA, studies of ACLD knee kinematics would be important to reveal the disease process and gather information which would support mechanisms to potentially slow OA progression.
In vivo knee kinematics during weight-bearing activities in the ACLD knee have been reported in recent years. Studies agreed that the tibia translates anteriorly to a greater degree in the ACLD knee than in the intact knee (Van De Velde et al., 2009, Defrate, 2006, Yamaguchi et al., 2009). Medial and lateral contact points of the tibiofemoral joint in the ACLD knee were located more posteriorly than in the intact knee (Li, 2006, Logan et al., 2004). However, sample sizes in these studies were insufficient (under ten knees) to draw firm conclusions and a further study with sufficient sample size is needed. The aim of this study was to determine if there were any differences in the kinematics of the ACLD knee and the contralateral intact knee during a weight-bearing activity.
Knee kinematics are activity dependent, and several types of weight-bearing activities have been examined in previous studies (Van De Velde et al., 2009, Defrate, 2006, Yamaguchi et al., 2009). The front lunge is commonly chosen in kinematic studies using lateral fluoroscopy to avoid overlapping of the contralateral knee (Van De Velde et al., 2009, Defrate, 2006). However, a narrow base of support in this activity may not be suitable for an early rehabilitation phase of the ACLD knee. In contrast, a wide-based squat activity, with the feet oriented at 90° to each other and positioned 1.5 times shoulder-width apart, is considered to be a more stable rehabilitation exercise with wider base-of-support than that of the lunge activity for the ACLD knee and this does allow for lateral fluoroscopy. In addition, it is easy, stable and reproducible due to bearing the weight evenly on both feet and a lower center of mass with a greater base area. In a previous study, the wide-based squat activity involves increased anterior translation at - 10~80° flexion angles and internal rotation of the tibia at low knee flexion angles (Yamaguchi et al., 2009). On the other hand, some studies reported that the tensile force in the ACL during squat was minimal or absent even at lower flexion angles, needless to say the tensile force was also minimal or absent at deep knee flexion angles (Escamilla, 2012, Escamilla et al., 2009, Beynnon et al., 2002, Toutoungi, 2000). Though these results may not allow direct comparisons, it is still unclear at which knee flexion angles tibial anterior translation during squat activity places the knee at risk.
We hypothesized that abnormal knee kinematics during wide-based squat are present at lower flexion angles in the ACLD knee as compared with the contralateral intact knee. A 2D/3D registration technique with lateral fluoroscopy was utilized to analyze dynamic knee kinematics in this cross-sectional study with sufficient sample size to test this hypothesis.
Methods
Materials
This study is a laboratory-based, cross-sectional study. The study protocol was approved by a local institutional review board. Subjects were recruited from patients visiting our hospital. Selection criteria included (1) males aged over 20 years old, (2) unilateral, isolated ACLD confirmed by MRI, (3) signs of knee instability by manual physical examinations, (4) symptoms of knee instability during daily activities, (5) no evidence of past knee injuries except for the ACL injury, (6) over 100 degrees range of motion (ROM) arc in bilateral knees during squat, (7) no previous injury or OA changes in the contralateral knee, and (8) written informed consent. Females were not enrolled in this study to avoid radiation exposures for potentially pregnant women. Thirty-three patients aged 29.2 ± 7.9 years (range 20-51 years) with unilateral ACLD and contralateral intact knees were enrolled in this study. The mean term between the ACL injury and the testing was 47.4 weeks (range 3.3 - 450 weeks, median ten weeks). Full passive knee range of motion arc and resolution of knee swelling were achieved for all subjects before testing began. All of the ACLD knees underwent ACL reconstruction following data acquisition due to persistent severe knee instability. All had a positive Lachman test, 31 knees had positive pivot-shift tests, and none had radiographic OA changes. All subjects were examined with anterior-drawer test using a KT- 2000 arthrometer (MEDmetric Corp, San Diego, California) under anesthesia during the ACL reconstruction surgery. Anterior laxity ranged from 13.1 ± 2.3 mm in the ACLD knees to 7.3 ± 2.1 mm in the contralateral non-involved knees. There was a significant difference (p < 0.001, Student’s t-test).
Overview of analysis
Knee kinematics were analyzed using the 2D/3D registration technique utilizing CT scan and lateral fluoroscopy proposed by Banks et al. (Banks and Hodge, 1996, Hoff et al., 1998). Three-dimensional positions of femoral and tibial bone models in virtual space were obtained and knee kinematics were determined in Cardan angles (Andriacchi and Dyrby, 2005). Fregly et al. reported the accuracy of this technique was 0.42 mm for in-plane translation, 5.6 mm for out-of-plane translation, 1.3° for rotations with the 3D bone model and single-plane fluoroscopy (Fregly et al., 2005). Komistek et al. reported values of 0.45 mm for in-plane translation and 0.66° for rotations?(Komistek et al., 2003). The best-case accuracy was reported to be 0.53 mm for in-plane translation, 1.6 mm for out-of-plane translation and 0.54° for rotations (Moro-Oka et al., 2007). These data indicate that the accuracy of this technique is sufficient to investigate knee kinematics for in-plane translation and rotations.
Activity and fluoroscopic imaging
The wide-based squat (Figure 1) was chosen for knee kinematic analyses. First, the subjects stood on the pre-set foot prints, so that everyone maintained the same stance width. It allowed their feet to be oriented at 90° to each other and positioned 1.5 times shoulder-width apart. Then, the subjects performed the squat with their hips at 45° external rotation, during which the knee over the toe position was maintained. The maximal flexion angle during squat was instructed to be "as deep as possible". They kept their back straight and nearly upright (within 20° of forward tilt) throughout the activity. Subjects were not allowed forward knee movement beyond the toes during knee bending so that the knee would not protrude from the fluoroscopic screen. Instructions given to the subjects were to maintain equal weight-bearing on both legs as possible, and to perform a slow squat motion. One squat cycle was defined as movement from maximal extension to maximal flexion and then returning to maximal extension. Subjects practiced the activity until the motion became smooth and they felt comfortable before testing began.
Figure 1.

Wide-based squat. The subjects performed the squat with their hips at 45° external rotation, allowing their feet to be oriented at 90° to each other and positioned 1.5 times shoulder-width apart. The flat panel detector was on the lateral side of the knee and X-ray emitted from left to right on the photo. This activity can avoid overlapping of the contralateral knee.
The fluoroscopy unit (Shimadzu, Cvision Safire, Kyoto, Japan) used in this study was equipped with a square, 17-inch flat-panel screen. The imaging frame rate was at 5 Hz and the image size was 1024 × 1024 pixels. Fluoroscopic surveillance was performed for only one squat cycle for the sake of minimal radiation exposure. The above one cycle means movement from maximal extension to maximal flexion and then returning to maximal extension. The kinematic data during squat involved flexion and extension phases which were analyzed separately.
Our validation study revealed that our techniques for embedding the femoral and tibial coordinate systems are highly repeatable. Intra-researcher root mean square (RMS) of the translations/rotations was less than 0.5 mm/0.75 degrees for the femur and less than 0.4 mm/0.6 degrees for the tibia. Inter-researcher RMS of the translations/rotations was less than 0.5 mm/1.1 degrees for the femur and less than 1.1 mm/1.3 degrees for the tibia (Ikuta, 2012).
Bone-model preparations
All knees underwent CT (Siemens, SOMATOM Definition, Germany) scanning with a 0.5 mm slice pitch spanning approximately 150 mm above and below the knee joint line. Geometric bone models of the femur and tibia were created from the CT images. Exterior cortical bone edges were segmented using 3D-Doctor software (Able Software Corp., Lexington, MA) and were converted into polygonal surface models using Geomagic Studio software (Geomagic, Research Triangle Park, NC, USA). Local coordinate systems were embedded in each bone using the custom VHKneeFitter program (University of Colorado Health Sciences Center, Aurora, Co) that allows all the procedures detailed below to be performed in a virtual space with high reproducibility.
The cylindrical axis (CA) (Eckhoff et al., 2001) was utilized as the reference line for the femoral coordinate system. The femoral origin was defined as the midpoint of the medial and lateral ends of the CA, which were points on the CA crossing the bony surfaces medially and laterally. A plane through the origin perpendicular to the CA was defined as the sagittal plane on which the vertical axis (VA) and anteroposterior axis (APA) would be located. The distal one-third of the femoral shaft was projected onto the sagittal plane and the central line of the projected femoral shaft was drawn. A line through the origin parallel to this central line on the sagittal plane was defined as the VA. The APA was obtained as the cross product of the CA and VA (Figure 2).
Figure 2.
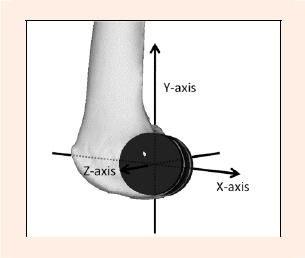
Femoral coordinate system. A cylinder is fitted into the femoral medial and lateral condyle. Z-axis: Cylinder Axis (CA): the co-axis of the cylinders. Y-axis: Vertical Axis (VA): A line through the origin parallel to the central line on the femoral shaft projected onto the sagittal plane. X-axis: Anteroposterior Axis (APA): the cross product of the CA and VA.
The tibial coordinate system was defined around a virtual rectangle fitted onto the contour of the tibial plateau. The rectangle was fitted at the level of the top of the fibular head parallel to the tibial plateau plane in order to avoid fitting onto highly variable morphology at the posterior contour of the tibial plateau. The four lines of the rectangle were fitted onto (1) the co-tangent of the posterior contours of the medial and lateral tibial condyles, (2) the medial tangent of the medial tibial condyle, (3) the lateral tangent of the lateral tibial condyle, and (4) the anterior tangent of the medial tibial condyle. Then, the rectangle was translated superiorly so that it fitted the bottoms of both tibial plateaus. The center of the rectangle was defined as the tibial origin, through which the medial/lateral and anteroposterior axes were defined as two axes of the tibial coordinate system. The vertical axis of the tibia was, by definition, the cross product of these two axes proximally (Figure 3).
Figure 3.
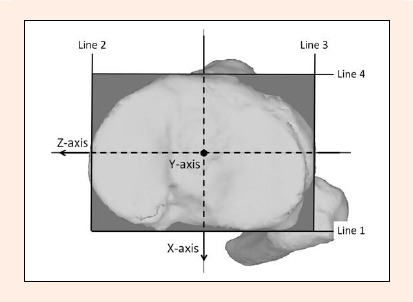
Tibial coordinate system. A virtual rectangle is fitted onto the contour of the tibial plateau (bounded by the line 1~4). Line 1: the co-tangent of the posterior contours of the medial and lateral tibial condyles. Line 2: the medial tangent of the medial tibial condyle. Line 3: the lateral tangent of the lateral tibial condyle. Line 4: the anterior tangent of the medial tibial condyle. Z-axis: medial/lateral axis. X-axis: anteroposterior axis. Y-axis: vertical axis.
Model registration and data processing
In vivo three dimensional femoral and tibial positions and orientations were determined using a 2D/3D registration technique (Banks and Hodge, 1996, Fregly et al., 2005, Moro-Oka et al., 2007). The bone model was projected onto the distortion-corrected fluoroscopic image, and its three dimensional pose was iteratively adjusted to match its silhouette with the silhouette of the bones on the fluoroscopic images with the custom JointTrack program (sourceforge.net/projects/jointtrack). Manual matching was first performed, and then an automated matching procedure was performed using the nonlinear least-squares (modified Levenberg-Marquardt) technique.
Six degrees-of-freedom (DOF) joint kinematics were computed using the 3D-JointManager commercial software (GLAB Inc., Higashi-Hiroshima, Japan) once the registration procedures were completed for the activity sequence. The joint coordinate system utilized in this software was based on the projection angles of the fixed tibial coordinate system (Andriacchi et al., 2005). Kinematics were analyzed in 5° increments of knee flexion angles after B-spline curve approximation was performed. Comparisons were performed using values between 0 and 100° of knee flexion.
Statistical analyses
Statistical analyses for kinematic data were performed using the Student’s t-test, repeated measures analysis of variance (ANOVA) and post-hoc pair-wise comparison (Tukey Kramer test) to compare between the two groups. The level of significance was set at p < 0.05. Statcel-The Useful Addin Forms on Excel-2nd ed. (The Publisher OMS Ltd., Saitama, Japan) and G*Power ver. 3 (University of Kiel, Kiel, Germany) were used to complete a power analysis. The effect size was set at 0.33 estimated from our preliminary study using ten ACLD knees. The sample size necessary for achieving alpha = 0.05 and beta = 0.20 was 26.
Results
Tibial rotation
Flexion phase: The average magnitude of the tibial internal/external rotation during 0 to 100° knee flexion angles for the ACLD and contralateral knees were 15.9 ± 5.7° and 19.3 ± 7.2°, respectively (Table 1) demonstrating no significant difference. The tibial internal rotation increased with knee flexion for both groups, but there was no significant difference at any flexion angles between the groups (Figure 4).
Table 1.
Characteristics of the ACLD and contralateral intact knees.
| Group | phase | ACLD | Contralateral |
|---|---|---|---|
| Tibial Rotation | Flexion phase | 15.9 ± 5.7 | 19.3 ± 7.2 |
| Extension phase | 16.0 ± 5.7 | 20.0 ± 6.8 | |
| Tibial Translation | Flexion phase | 5.4 ± 2.4 | 5.4 ± 2.7 |
| Extension phase | 4.7 ± 2.5 | 4.9 ± 2.7 | |
| Medial condyle Contact point | Flexion phase | 9.3 ± 3.4 | 10.7 ± 3.8 |
| Extension phase | 8.3 ± 3.2 | 10.1 ± 3.7 | |
| Lateral condyle Contact point | Flexion phase | 10.8 ± 3.6 * | 13.3 ± 4.4 |
| Extension phase | 10.6 ± 3.4 * | 13.6 ± 4.0 |
Tibial Rotation: average magnitude of tibial internal/external rotation (°). Tibial Translation: average magnitude of tibial anterior/posterior translation (mm). Medial condyle Contact point: average magnitude of anterior/posterior translation of tibiofemoral contact points on medial tibial condyle (mm). Lateral condyle Contact point: average magnitude of anterior/posterior translation of tibiofemoral contact points on lateral tibial condyle (mm).There were significant differences in Lateral condyle Flexion and Extension phase.
Asterisk denotes P value < 0.05 by repeated measures ANOVA. Tukey’s test was used for post-hoc pair wise comparisons (p < 0.05).
Figure 4.
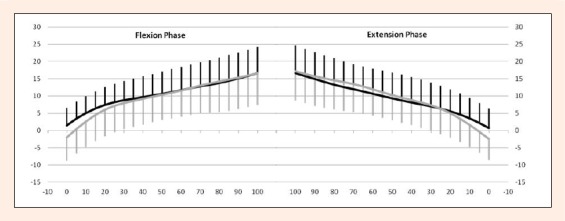
Comparisons of tibial rotation between the ACLD and contralateral intact knees. Y-axis: tibial internal rotation (°). X-axis: knee flexion (°). Left graph: Flexion phase. Right graph: Extension phase. Thick line: ACLD knee. Thin line: Contralateral knee. There were no significant differences by repeated measures ANOVA. Error bars represent ± 1 standard deviation.
Extension phase: The average magnitude of the tibial internal/external rotation for the ACLD and contralateral knees was 16.0 ± 5.7° and 20.0 ± 6.8° respectively (Table 1), also demonstrating no significant difference. Tibial external rotation increased with knee extension for both groups, but there was no significant difference between the groups at any flexion angles (Figure 4).
Tibial translation
Flexion phase: The average magnitude of the tibial anterior/posterior translation for the ACLD and contralateral knees during 0 to 100° knee flexion motion were 5.4 ± 2.4 mm and 5.4 ± 2.7 mm, respectively (Table 1) revealing no significant difference. The tibia of the ACLD knee presented with significantly greater anterior translation (p < 0.05) than that of the contralateral knee at 0 and 5° of knee flexion (Figure 5).
Figure 5.
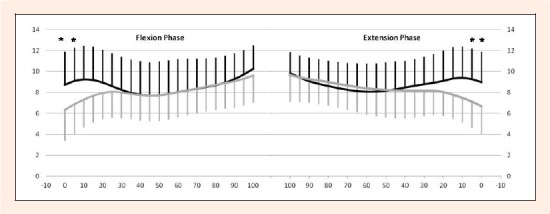
Comparisons of tibial anterior/posterior translation between the ACLD and contralateral intact knees. Y-axis: tibial anterior translation (mm). X-axis: knee flexion (°). Left graph: Flexion phase. Right graph: Extension phase. Thick line: ACLD knee. Thin line: Contralateral knee. There were significant differences at 0 and 5° of knee flexion in both phases. Asterisk denotes p value < 0.05 by repeated measures ANOVA. Tukey’s test was used for post-hoc pair wise comparisons. Error bars represent ± 1 standard deviation.
Extension phase: The average magnitude of the tibial anterior/posterior translation for the ACLD and contralateral knees were 4.7 ± 2.5 and 4.9 ± 2.7 mm respectively (Table 1), demonstrating no significant difference. The tibia of the ACLD knee presented with significantly greater anterior translation (p < 0.05) than that of the contralateral knee at 0 and 5° flexion (Figure 5).
Medial contact point
Flexion phase: The average magnitude of the anterior/posterior translation of the medial contact points for the ACLD and contralateral knees were 9.3 ± 3.4 mm and 10.7 ± 3.8 mm, respectively (Table 1) demonstrating no significant difference. There were no significant differences at any flexion angles between the groups for the tibiofemoral contact point on the medial tibial condyle (Figure 6).
Figure 6.
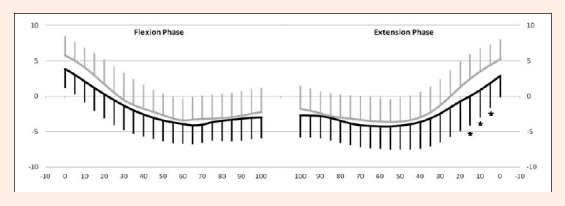
Comparisons of tibiofemoral contact points on the medial tibial condyle between the ACLD and contralateral intact knees. Y-axis: anterior translation of contact points (mm) , X-axis: knee flexion (°). Left graph: Flexion phase. Right graph: Extension phase. Thick line: ACLD knee. Thin line: Contralateral knee. There were significant differences at 5, 10, 15° of knee flexion in the extension phase. Asterisk denotes P value < 0.05 by repeated measures ANOVA. Tukey’s test was used for post-hoc pair wise comparisons. Error bars represent ± 1 standard deviation.
Extension phase: The average magnitude of the anterior/posterior translation of the medial contact points for the ACLD and contralateral knees were 8.3 ± 3.2 mm and 10.1 ± 3.7 mm, respectively (Table 1) demonstrating no significant difference. The tibiofemoral contact point on the medial tibial condyle translated posteriorly with knee flexion for both groups. The contact points in the ACLD knee were located more posteriorly (p < 0.05) at 5, 10 and 15° flexion than that of the contralateral knees (Figure 6).
Lateral contact point
Flexion phase: The average magnitude of the anterior/posterior translation of the lateral contact points for the ACLD and contralateral knees were 10.8 ± 3.6 mm and 13.3 ± 4.4 mm, respectively (Table 1) demonstrating a significant difference (p < 0.05). The tibiofemoral contact points on the lateral tibial condyle translated posteriorly with knee flexion for both groups. The contact points in the ACLD knee were located more posteriorly at 0° flexion than that of the contralateral knee (p < 0.05) (Figure 7, 8).
Figure 7.
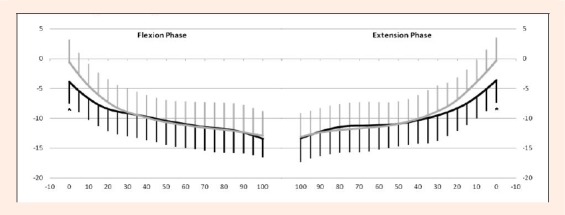
Comparisons of tibiofemoral contact points on the lateral tibial condyle between the ACLD and contralateral intact knees. Y-axis: anterior translation of contact points (mm), X-axis: knee flexion (°). Left graph: Flexion phase. Right graph: Extension phase. Thick line: ACLD knee. Thin line: Contralateral knee. There were significant differences at 0° of knee flexion in both phases. Asterisk denotes P value < 0.05 by repeated measures ANOVA. Tukey’s test was used for post-hoc pair wise comparisons. Error bars represent ± 1 standard deviation.
Figure 8.
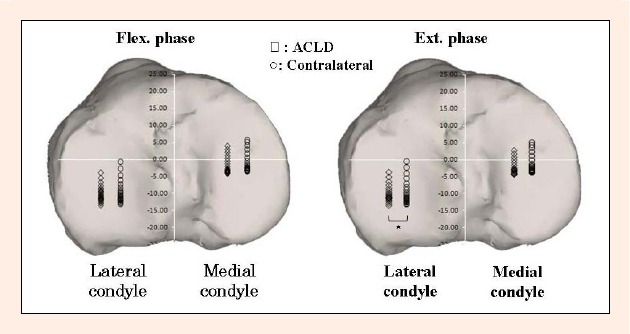
The medial and lateral tibiofemoral contact locations on the tibial plateau between 0 to 100° of knee flexion are shown. The contact locations are slightly posterior in the ACLD knee on the both condyles in the both phases. There was a significant difference on the average magnitudes of the anterior/posterior translation of the lateral contact points between the ACLD and contralateral knees in the extension phase (p < 0.05, denoted by an asterisk).
Extension phase: The average magnitude of the anterior/posterior translation of the lateral contact points for the ACLD and contralateral knees were 10.6 ± 3.4 mm and 13.6 ± 4.0 mm, respectively (Table 1), indicating a significant difference (p < 0.05). The tibiofemoral contact points on the lateral tibial condyle translated posteriorly with knee flexion for both groups. The contact points in the ACLD knee were located more posteriorly at 0° flexion than that of the contralateral knee (p < 0.05) (Figure 7, 8).
Discussion
We hypothesized that there were different knee kinematics at low flexion angles between the ACLD and contralateral intact knees during a wide-based squat activity. The results of this study were consistent with our hypothesis. Significant differences were detected at 0 and 5° flexion in tibial anterior/posterior translation, at 5, 10 and 15° of knee flexion in the contact points on the medial tibial plateau in the extension phase, and at 0° flexion in the contact points on the lateral tibial plateau. The ACLD knees demonstrated a smaller magnitude of anterior/posterior translation of the lateral contact points in the extension phase than that of the contralateral knees (Figure 8).
The kinematic differences at low flexion angles observed in this study were consistent with previous reports. Van de Velde et al. (Van De Velde et al., 2009) demonstrated increased anterior translation and internal rotation of the ACLD tibia at low flexion angles when compared with the contralateral intact knees during quasi-static front lunge activity. Li et al. (Li, 2006) reported tibiofemoral contact points on the medial tibial condyle in the ACLD knee were significantly more posterior than that of the contralateral intact knee at 0 and 15° of knee flexion. These two studies used highly accurate dual-orthogonal fluoroscopy and MRI-based bone-cartilage models, though the knee motions were not truly dynamic. In vivo strain gauge studies demonstrated that the ACL tensile forces were minimal, but greater at low knee flexion angles than at higher knee flexion angles (Toutoungi, 2000, Escamilla, 2012). Although the ACL force in the intact knee does not directly correlate with the knee kinematics in the ACLD knee, an increased ACL force at low flexion angles in the intact knees suggests an increased anterior shear force, which may cause greater tibial anterior translation in the ACLD knees. Therefore, our results are supported by the previous kinematic as well as strain gauge studies. However, Yamaguchi et al. analyzed dynamic wide-based squat identical to our activity in a single-plane fluoroscopic study with eight ACLD knees (Yamaguchi et al., 2009). He reported greater anterior translation at -10 through 80° of knee flexion and greater internal rotation at terminal extension in the ACLD knee. The reason for this discrepancy in findings is unknown.
Strength in internal validity should be pointed out. The 2D/3D registration technique using lateral fluoroscopy or radiography is a well-established measurement method of in vivo dynamic weight-bearing knee kinematics with standard errors within 2.2 mm for translations and 1.8 degrees for rotation (Banks and Hodge). The wide-based squat, with greater area of support, should reduce loading pattern variability and relative pelvic positions and therefore should improve knee kinematic consistency, providing greater sensitivity to detect small kinematic changes. Moreover, knee kinematics were computed based on the bony coordinate systems that had been assigned using a software program specially coded for assigning femoral and tibial coordinate systems with high reproducibility. Errors caused by potential morphological variations should have been significantly diminished by using the CA for the femur and the posterior co-tangent of both tibial condyles at the top of the fibular head for the tibia as reference lines for knee rotation. The data presented in this study should be reliable with good internal validity.
Several issues should be pointed out regarding external validity. Knee kinematics observed in this study should be activity specific, and should not be generalized to the other activities such as gait. Andriacchi and Dyrby (Andriacchi and Dyrby, 2005) noted the kinematic changes associated with loss of the ACL occurred primarily during the terminal portion of the swing phase of the gait cycle where the tibia had reduced external rotation and anterior translation as the knee extended prior to heel strike in the ACL deficient knee. The wide-based squat requiring continuous loading of the knee with continuous hamstring activity (Isear et al., 1997) demonstrated little abnormal kinematics, and therefore this activity is considered a safe exercise for the ACLD knee (Escamilla, 2012).
Sufficient statistical power, accurate analyses, and reproducible activity are strengths of this study. However, there are several limitations in this study. First, the 2D/3D registration method using single-plane fluoroscopy provides limited measurement accuracy for out-of-plane motions (Moro-Oka et al., 2007). We did not report medial/lateral translations in this paper, and important kinematic differences might have been missed. Second, the fluoroscopic surveillance was performed for only one squat cycle, so that the radiation dose was minimized. Although subjects were advised to practice the specific squat motion before the testing, potential variations of kinematics might have been discarded. Third, we did not compare between the wide-based squat and other weight-bearing activities such as the front lunge. Since external control is available in literature, we decided not to perform internal comparisons to avoid an extra radiation dose.
Conclusion
In conclusion, knee kinematics between the ACLD and contralateral intact knees are similar during a wide-based squat activity except at the low knee flexion angles. This further indicates that this activity is recommended for ACLD knees during pre-operative rehabilitation and potentially during the early post-operative phase as well. This should be validated in future studies. This research project is part of a series which has currently been extended to open chain knee extension and kneeling and the results will be reported shortly. We also have been gathering patients with long-term-ACLD knees and OA changes, and hope that our series of studies can help reveal the process of OA changes in ACLD knees.
Acknowledgments
This work was supported, in part, by Arthrex inc. and Minato Medical Science Co. Ltd. We thank Scott Banks, PhD (University of Florida) for significant academic and technical supports including providing the JointTrack program. We also appreciate the considerable technical support given by the radiology technicians in our hospital.
Biographies
Takeshi Miyaji
Employment
Department of Orthopaedic Surgery, Graduate School of Biomedical Science, Nagasaki University, Nagasaki, Japan
Degree
MD
Research interests
Knee biomechanics
E-mail: takemiya@k9.dion.ne.jp
Kazuyoshi Gamada
Employment
Medical Engineering and Technology, Graduate School of Medical Technology and Health Welfare Sciences, Hiroshima International University, Hiroshima, Japan
Degree
PhD, PT
Research interests
Knee biomechanics
E-mail: kazgamada@ortho-pt.com
Kenichi Kidera
Employment
Department of Orthopaedic Surgery, Graduate School of Biomedical Science, Nagasaki University, Nagasaki, Japan
Degree
MD
Research interests
Knee biomechanics
E-mail: sayufu@mac.com
Futoshi Ikuta
Employment
Medical Engineering and Technology, Graduate School of Medical Technology and Health Welfare Sciences, Hiroshima International University, Hiroshima, Japan
Degree
MS, PT
Research interests
Knee osteoarthritis
E-mail: futoshi.ikuta@gmail.com
Kei Yoneta
Employment
Department of Rehabilitation, Kobayashi Hospital, Kitami, Japan
Degree
MS, PT
Research interests
Knee osteoarthritis
E-mail: einstein_syndrome@yahoo.co.jp
Hiroyuki Shindo
Employment
Department of Orthopaedic Surgery, Graduate School of Biomedical Science, Nagasaki University, Nagasaki, Japan
Degree
MD, PhD
Research interests
Biomechanics, bone and cartilage metabolism, arthroplasty
E-mail: shindo@nagasaki-u.ac.jp
Makoto Osaki
Employment
Department of Orthopaedic Surgery, Graduate School of Biomedical Science, Nagasaki University, Nagasaki, Japan
Degree
MD, PhD
Research interests
Biomechanics, bone and cartilage metabolism, arthroplasty
E-mail: mosaki@net.nagasaki-u.ac.jp
Akihiko Yonekura
Employment
Department of Orthopaedic Surgery, Graduate School of Biomedical Science, Nagasaki University, Nagasaki, Japan
Degree
MD, PhD
Research interests
Knee biomechanics
E-mail: yonekura@nagasaki-u.ac.jp
References
- Andriacchi T.P., Johnson T.S., Hurwitz D.E., Matarajan R.N.(2005)Musculoskeletal Dynamics, Locomotion, and Clinical Applications. In: Basic Orthopaedic Biomechanics & Mechano-Biology. Eds: Mow V.C., Huiskes R.Third Edition Philadelphia: Lippincott Williams & Wilkins; [Google Scholar]
- Andriacchi T.P., Dyrby C.O.(2005) Interactions between kinematics and loading during walking for the normal and ACL deficient knee. Journal of Biomechanical Engineering 38(2), 293-298 [DOI] [PubMed] [Google Scholar]
- Banks S.A., Hodge W.A.(1996) Accurate measurement of three-dimensional knee replacement kinematics using single-plane fluoroscopy. IEEE Transactions on Biomedical Engineering 43(6), 638-649 [DOI] [PubMed] [Google Scholar]
- Beynnon B.D., Johnson R.J., Fleming B.C.(2002) The science of anterior cruciate ligament rehabilitation. Clinical Orthopaedics and Related Research Sep(402), 9-20 [DOI] [PubMed] [Google Scholar]
- Defrate L.E.(2006) The 6 degrees of freedom kinematics of the knee after anterior cruciate ligament deficiency: An in vivo imaging analysis. American Journal of Sports Medicine 34(8), 1240-1246 [DOI] [PubMed] [Google Scholar]
- Eckhoff D.G., Dwyer T.F., Bach J.M., Spitzer V.M., Reinig K.D.(2001) Three-dimensional morphology of the distal part of the femur viewed in virtual reality. The Journal of Bone and Joint Surgery. American volume 83-A(Suppl 2), 43-50 [DOI] [PubMed] [Google Scholar]
- Escamilla R.F.(2012) Anterior cruciate ligament strain and tensile forces for weight-bearing and non-weight-bearing exercises: a guide to exercise selection. Journal of Orthopaedic and Sports Physical Therapy 442(3), 208-220 [DOI] [PubMed] [Google Scholar]
- Escamilla R.F., Zheng N., Imamura R., Macleod T.D., Edwards W.B., Hreljac A., Fleisig G.S., Wilk K.E., Moorman C.T., Andrews J.R.(2009) Cruciate ligament force during the wall squat and the one-leg squat. Medicine & Science in Sports & Exercise 41(2), 408-417 [DOI] [PubMed] [Google Scholar]
- Fregly B.J., Rahman H.A., Banks S.A.(2005) Theoretical accuracy of model-based shape matching for measuring natural knee kinematics with single-plane fluoroscopy. Journal of Biomechanical Engineering 127(4), 692-699 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gillquist J., Messner K.(1999) Anterior cruciate ligament reconstruction and the long-term incidence of gonarthrosis. Sports Medicine 27(3), 143-156 [DOI] [PubMed] [Google Scholar]
- Hoff W.A., Komistek R.D., Dennis D.A., Gabriel S.M., Walker S.A.(1998) Three-dimensional determination of femoral-tibial contact positions under in vivo conditions using fluoroscopy. Clinical Biomechanics (Bristol, Avon) 13(7), 455-472 [DOI] [PubMed] [Google Scholar]
- Ikuta F.Y., Yoneta K., Gamada K.(2012)Validity of local coordinate systems embedded to the distal femur and proximal tibia. In: Transaction of the 2012 ORS Annual meeting, February 4-7, San Francisco CA: Poster number 2014 [Google Scholar]
- Isear J.A., Jr., Erickson J.C., Worrell T.W.(1997) EMG analysis of lower extremity muscle recruitment patterns during an unloaded squat. Medicine and Science in Sports and Exercise 29(4), 532-539 [DOI] [PubMed] [Google Scholar]
- Komistek R.D., Dennis D.A., Mahfouz M.(2003) In vivo fluoroscopic analysis of the normal human knee. Clinical Orthopaedics and Related Research May(410), 69-81 [DOI] [PubMed] [Google Scholar]
- Li G.(2006) Anterior cruciate ligament deficiency alters the in vivo motion of the tibiofemoral cartilage contact points in both the anteroposterior and mediolateral directions. The Journal of Bone and Joint Surgery 88(8), 1826-1834 [DOI] [PubMed] [Google Scholar]
- Logan M., Dunstan E., Robinson J., Williams A., Gedroyc W., Freeman M.(2004) Tibiofemoral kinematics of the anterior cruciate ligament (ACL)-deficient weightbearing, living knee employing vertical access open "interventional" multiple resonance imaging. The American Journal of Sports Medicine 32(3), 720-726 [DOI] [PubMed] [Google Scholar]
- Moro-Oka T.A., Hamai S., Miura H., Shimoto T., Higaki H., Fregly B.J., Iwamoto Y., Banks S.A.(2007)Can magnetic resonance imaging-derived bone models be used for accurate motion measurement with single-plane three-dimensional shape registration? Journal of Orthopaedic Research 25(7), 867-872 [DOI] [PubMed] [Google Scholar]
- Roos H., Adalberth T., Dahlberg L., Lohmander L.S.(1995) Osteoarthritis of the knee after injury to the anterior cruciate ligament or meniscus: the influence of time and age. Osteoarthritis and Cartilage 3(4), 261-267 [DOI] [PubMed] [Google Scholar]
- Segawa H., Omori G., Koga Y.(2001) Long-term results of non-operative treatment of anterior cruciate ligament injury. The Knee 8(1), 5-11 [DOI] [PubMed] [Google Scholar]
- Toutoungi D.E.(2000) Cruciate ligament forces in the human knee during rehabilitation exercises. Clinical Biomechanics (Bristol, Avon) 115(3), 176-187 [DOI] [PubMed] [Google Scholar]
- Van De Velde S.K., Gill T.J., Li G.(2009) Evaluation of kinematics of anterior cruciate ligament-deficient knees with use of advanced imaging techniques, three-dimensional modeling techniques, and robotics. The Journal of Bone and Joint Surgery. American volume 91(Suppl 1), 108-114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamaguchi S., Gamada K., Sasho T., Kato H., Sonoda M., Banks S.(2009) In vivo kinematics of anterior cruciate ligament deficient knees during pivot and squat activities. Clinical Biomechanics 24, 71-76 [DOI] [PubMed] [Google Scholar]


