Abstract
Our bodies are continuously exposed to various types of infectious pathogens. Vaccinations are the most cost effective way to protect our bodies against a variety of infectious microbes. The efficacy of most vaccines relies on protective antibody production and generation of memory B cells. These two key components develop mostly from B cells that participate in germinal center reactions. Recent efforts have highlighted the critical role of follicular helper CD4 T (Tfh) cells in the generation of germinal centers. Given that Bcl6 is a major transcription factor for Tfh differentiation, here we review recent developments in the understanding of signaling molecules that regulate Bcl6 expression in CD4 T cells, as a potential target for development of more efficacious vaccines.
Introduction
In 1918, the world witnessed the “Spanish flu”, which led to more deaths than World War I [1]. For the last three decades, we have been confronted by another deadly virus, the human immunodeficiency virus (HIV), which causes acquired immunodeficiency syndrome (AIDS) and leads to almost 2 million deaths every year [2,3]. Vaccinations provide one of the most cost effective medical preventions against infectious diseases. The smallpox vaccine, the first successful vaccine, developed by Edward Jenner [4], resulted in the eradication of smallpox in 1980 through a worldwide vaccination program of the World Health Organization (WHO) [5]. Tremendous progress has been made in the field of immunology since the time of Edward Jenner. The efficacy of almost all licensed vaccines relies heavily on the capacity of our bodies to generate protective antibodies and memory B cells against infectious microbes [6]. Protective antibody production and memory B cells mostly develop from germinal center reactions, during which two critical immunological processes occur in B cells – affinity maturation and immunoglobulin isotype switching [7]. Random mutations in the immunoglobulin V region gene of B cells are generated and then selected for by limiting T cell help and expanding higher affinity B cell receptors (BCRs) recognizing microbes, while antibody effector functions are also gained through isotype switching [8,9]. These immunological events occur mostly in a helper CD4 T cell dependent manner.
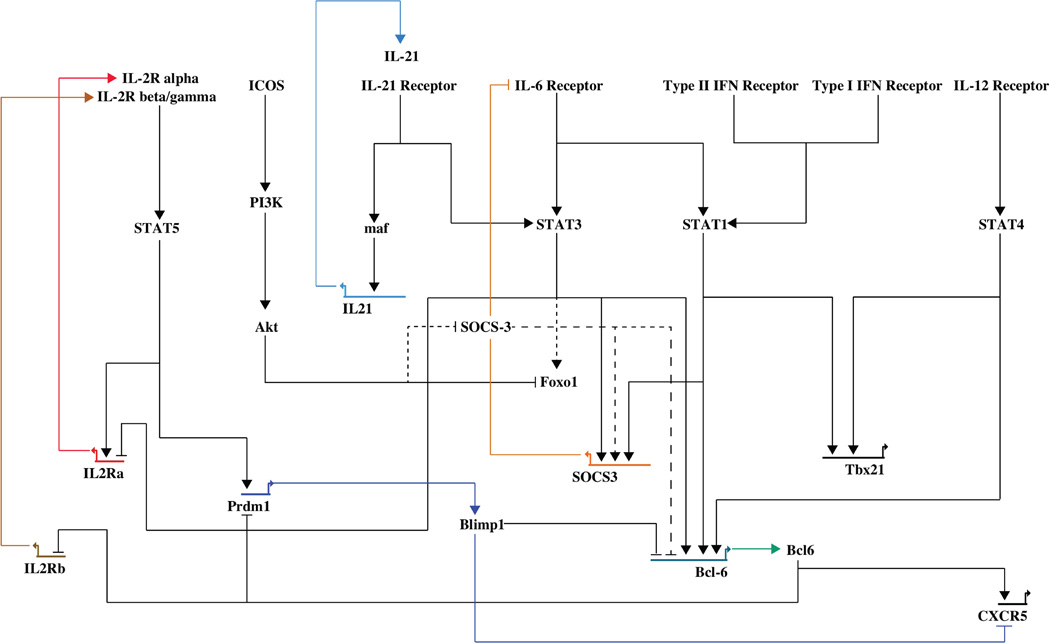
The effector CD4 T cells with specialized B cell help functions were defined relatively recently, and are called follicular helper T (Tfh) cells [10]. The transcriptional repressor Bcl6 was identified as a “master regulator” transcription factor for Tfh differentiation [11–13]. Bcl6 is regulated by a complicated signaling circuitry in Tfh differentiation, and here we discuss recent findings regarding the multiple layers of both positive and negative signaling pathways that regulate Bcl6 expression (Figure 1).
Figure 1. Signaling pathways in regulation of Bcl6 expression of CD4 T cells.
In antigen primed CD4 T cells, Bcl6 expression is regulated by various stimuli (i.e., ICOS, IL-6, IL-12, etc.). Signaling circuits are depicted with positive (arrows) and negative (bars) functions shown for signaling molecules that regulate Bcl6 expression or related processes. The solid lines indicate defined in the literature, whereas the dotted lines describe signaling pathways that are potentially involved in regulation of Bcl6 expression in CD4 T cells. Direct lines don’t imply direct binding.
Pathways that induce Bcl6 expression
IL-6 and cooperative roles of STAT1 and STAT3
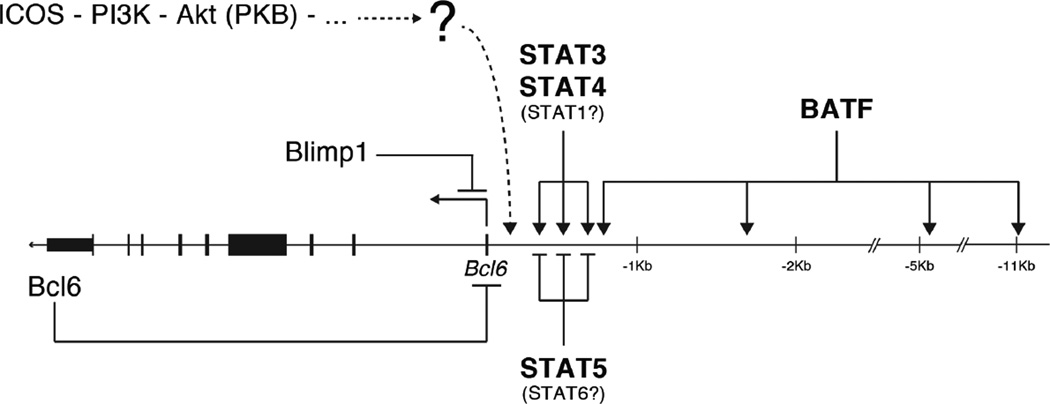
IL-6 is a proinflammatory cytokine, produced by a variety of cell types, both non-hematopoietic and hematopoietic, including dendritic cells [14]. In vitro stimulation of CD4 T cells in the presence of IL-6 was reported to induce Tfh features to some degree [12]. We recently demonstrated that IL-6 is a key signal in vivo for CD4 T cells to initiate Bcl6 induction and the Tfh differentiation program [15]. Bcl6+CXCR5+ Tfh cells were not found in the absence of IL-6 during the DC priming stage of Tfh differentiation [15]. STAT3 is JAK phosphorylated and activated in response to IL-6 stimulation [16], and as such STAT3 was reported as an important molecule for Tfh differentiation [17]. However, we found that STAT3 deficiency failed to recapitulate the severe IL-6 dependent early Tfh differentiation defect seen in an acute viral infection model [18], implying that another signaling molecule acts downstream of the IL-6 receptor during Tfh differentiation. STAT1 is phosphorylated and activated in response to IL-6 stimulation selectively in CD4 T cells [19]. Thus, we tested whether STAT1 participates in IL-6 mediated Tfh differentiation pathway and found that STAT1 cooperates with STAT3 to convey IL-6 mediated Bcl6 induction signals downstream of IL-6R [18]. These recent findings may provide mechanistic insights into human genetic diseases. In human patients with dominant negative mutations in STAT3, Th17 differentiation is completely abrogated [20], whereas CXCR5+ Tfh cell frequencies in peripheral blood CD45RO+ CD4 T cells were reduced by half in comparison to healthy controls [21]. The remaining Tfh cell differentiation may result from compensatory roles of STAT1 [18]. In human with genetic defects in STAT1, the abundance of memory Tfh cells seems to correlate with STAT1 function [21]; more or fewer CXCR5+ Tfh cells appeared to be present in patients with dominant negative or gain of function mutation in STAT1, respectively. STAT1-STAT3 mediated Bcl6 induction appears to result from a direct binding of STAT molecules to the Bcl6 gene, as STAT3 binds to the promoter regulatory element region of Bcl6 in CD4 T cells (Figure 2) [22]. Further studies are required to reveal whether STAT1 directly binds Bcl6 (Figure 2). Collectively, STAT1 and STAT3 deliver IL-6 mediated Bcl6 induction signals during the Tfh priming stage (Figure 1).
Figure 2. Direct interaction of transcription factors with Bcl6 promoter regulatory region.
Shown is the Bcl6 locus including −11 Kb promoter regulatory region. Transcription factors that bind to the Bcl6 promoter regulatory region are depicted. Arrows and bars indicate positive and negative roles of the transcription factors for Bcl6 expression, respectively. An unidentified signaling pathway is represented by a dotted line. Molecules in parentheses indicate transcription factors associated with Bcl6 expression regulation, but not confirmed to bind to Bcl6 gene.
Intriguingly, Tfh cells do differentiate in IL-6 deficient mice later in the immune responses to acute viral infections and protein immunization [18,23–25]. For instance, Bcl6+CXCR5+ Tfh cells begin to appear starting at day 4 after acute LCMV infection (Choi and Crotty, unpublished data). Hence, germinal center formation is minimally affected by a lack of IL-6 signals in multiple experimental models. This indicates that induction of Tfh differentiation can be compensated by other signaling molecules. Multiple layers of redundancy are consistent with the fact that T cell help to B cells is evolutionarily conserved and highly important for protective immunity [26]. Identification of the compensatory signals may facilitate the development of robust in vitro Tfh cell culture conditions, since currently Tfh differentiation cannot be induced with high reproducibility in vitro [26]. In the following sections, we discuss additional molecules that contribute to Bcl6 induction signaling circuitry in CD4 T cells (Figure 1).
IL-21 – STAT3
IL-6 signaling induces IL-21 production by CD4 T cells [27]. IL-21 binding to the IL-21 receptor common γ chain activates STAT3 through Jak-mediated phosphorylation [28]. As such, IL-21 was reported to induce Bcl6 expression in in vitro stimulated CD4 T cells [12]. One study showed that CD4 T cell development into Tfh cells was severely impaired in immunized IL-21 deficient mice [17]. In contrast, several studies found that Tfh cells are present at normal frequencies in the absence of IL-21 signaling, both in the Tfh priming stage of immune response to an acute LCMV infection [18] and at the peak of the immune responses to viral infections and protein immunizations [23–25,29]. IL-21 directly acts on B cells to enhance germinal center B cell proliferation [24]. As germinal center B cells are major APCs for maintaining Tfh cells [11,30,31], defective Tfh frequencies in IL-21 deficient mice may occur over time as an indirect effect of reduced numbers of cognate B cells as APCs, due to lack of IL-21 signaling to germinal center B cells. Nevertheless, CD4 T cells express IL-21R and IL-21 seems to be a compensatory Tfh differentiation signal in IL-6 deficient mice, as a lack of IL-21 production--in the absence of IL-6--results in a reduction in Tfh differentiation [23,24]. IL-21 is produced also by IL-12 mediated signals through STAT3 activation in human CD4 T cells [21,32]. As such, IL-6 (IL-12) → STAT3 → IL-21, and then IL-21 → STAT3 → Bcl6 signaling axes partially contribute to Bcl6 induction in CD4 T cells (Figure 1).
IL-12 – STAT4
IL-12 is a key molecule for instruction of Th1 differentiation through STAT4 mediated expression of T-bet, which potentiates Th1 differentiation by inducing expression of IFN-γ and other Th1 genes [33]. Interestingly, IL-12 is capable of inducing human CD4 T cells to acquire some Tfh phenotypes, including IL-21 production and CXCR5 expression, suggesting IL-12 can participate in Tfh differentiation [32]. Recently, O’Shea and colleagues showed that IL-12 stimulation of murine CD4 T cells leads to PD-1hiCXCR5+ Tfh differentiation [34]. Tfh differentiation appeared to result from STAT4 mediated induction of Bcl6, but not through the STAT4 → T-bet signaling axis, as T-bet is not required for Tfh differentiation [34]. STAT4 acted as a positive regulator of Bcl6 expression. It remains to be revealed whether STAT4 mediated Bcl6 induction depends on STAT4 binding to the Bcl6 promoter (Figure 2). Both Tfh cell numbers and germinal center B cell counts were normal at the peak of immune response in STAT4 deficient mice [34], which implies STAT4 deficiency could be compensated by other signaling pathways. Interestingly, IL-12 mediated IL-21 production in CD4 T cells is compromised in the absence of STAT3 in both human [21] and mice [34]. As such, IL-12 mediated STAT3 activation may compensate for STAT4 deficiency, and/or IL-6 mediated STAT1 and STAT3 activation may provide all of the necessary signals for Tfh differentiation in the absence of STAT4 (Figure 1).
IFN-γ – STAT1
A recent study by Vinuesa and colleagues demonstrated that excessive IFN-γ expression, due to a lack of post-transcriptional negative regulation of IFN-γ mRNA in Sanroque mice (R3ch1san/san), contributes to abnormal Tfh differentiation [35]. The atypical Tfh differentiation is associated with aberrant formation of germinal center B cells and autoantibody accumulation in Sanroque mice [36]. Removal of IFN-γ signaling in Sanroque mice reduced spleen size and ameliorated aberrant Tfh formation and autoantibody production [35]. Reduced Bcl6 induction in CD4 T cells in the absence of IFN-γ signaling indicates a positive role of IFN-γ in Bcl6 induction or maintenance [35]. STAT1 is strongly activated by signaling through the IFN-γ receptor [37]. As such, IFN-γ receptor → STAT1 crosstalk may be involved in Bcl6 induction downstream of IFN-γ signals in Sanroque mice, given the capacity of STAT1 for Bcl6 induction downstream of IL-6R [18] (Figure 1).
ICOS – PI3K
Following immunization and infection, Tfh differentiation and germinal center formation is highly dependent on ICOS expression by CD4 T cells in both humans and mice [10]. We now know that ICOS mediated signals are required not only for CD4 T cells to initiate Bcl6 induction during the Tfh priming stage but are also necessary for primed Tfh cells to maintain Bcl6 expression through interacting with ICOS-L expressing germinal center B cells [17,30,38,39]. The PI3K pathway appears to deliver Bcl6 induction signals downstream of ICOS, as Tfh cells are not present in mice that harbor a point mutation of cytoplasmic tail of ICOS (Y181F), to which PI3K cannot be recruited [38] (Figure 1). Furthermore, Tfh cells are lost in p110δ−/− mice [40]. The PI3K signaling pathway is also activated downstream of OX40, CD28, and other receptors expressed on T cells [41,42]. As such, further research is needed to clarify the selective action by which ICOS mediates PI3K dependent Tfh differentiation (Figure 1).
Other molecules
In addition to the molecules discussed above, several other molecules have been reported to participate in Tfh differentiation. For instance, Batf is involved in Tfh differentiation. Batf can bind to the Bcl6 gene and induce Bcl6 expression [43]. Batf also binds to the Maf gene and induces its expression. Maf is involved in Tfh differentiation, as Tfh differentiation was impaired in Maf deficient mice [44]. However, constitutive expression of Maf in human tonsillar CD4 T cells was not able to induce Bcl6 [45]. Maf is involved in ICOS mediated IL-21 production by Tfh cells [44], and may regulate multiple other aspects of Tfh cell biology either in cooperation with Bcl6 or independently of Bcl6 [45]. IRF4 is also involved in Tfh differentiation [46]. The mechanism for how IRF4 supports Tfh differentiation is not clear. IRF4 is known to enhance IL-21 mediated STAT3 binding to the Prdm1 gene [47], and Blimp1, the product of the Prdm1 gene, potently inhibits Tfh differentiation [11]. Therefore, the regulator of Tfh differentiation by IRF4 must be complex. This is not unexpected, as IRF4 binds many genes upon T cell activation, and has a plethora of effects on CD4 T cell differentiation.
In summary, multiple signaling pathways contribute to Bcl6 induction in CD4 T cells (Figure 1). The complexity of signaling pathways involved in inducing Bcl6 expression in CD4 T cells most likely results from an evolutionarily evolved mechanism for the immune system to ensure the generation of Tfh cells and protective antibody responses no matter what type of pathogen is the source of the infection. Future studies are necessary to completely define the major regulators of Tfh differentiation. Currently, there are no in vitro Tfh differentiation systems that are highly reproducible, in contrast to culture systems available for the generation of other effector CD4 T cells. This may be due to a lack of information of other signaling molecule(s) that induce Bcl6 or other Tfh features. Alternatively, it may result from missing information regarding inhibitory pathways in Tfh differentiation. Understanding both positive and negative regulators of Bcl6 is important for understanding how to harness Tfh cells in rational vaccine design [48]. In the following section, we will discuss factors involved in negative regulation of Bcl6 induction in CD4 T cells.
Negative signaling pathways that inhibit Bcl6 expression
Blimp1
Within the first few cycles of cell division, a distinct population of differentiated antigen-specific Tfh cells are observable following infection of mice with acute LCMV or Listeria [30,49], demonstrating that much of Tfh differentiation can be determined during the earliest DC priming stage. We recently demonstrated that the virus specific CD4 T cells become fate committed very early, with IL-2Rαhi cells becoming Blimp1+T-bethiIFN-γhi Th1 cells and IL-2Rαlo cells becoming Bcl6hiCXCR5hi Tfh cells [50]. IL-2 receptor signaling activates STAT5 [51] and induces Blimp1 [52]. Blimp1 is a potent negative regulator of Tfh differentiation through transcriptional repression of Bcl6 [53] (Figure 1 and Figure 2). As such, an IL-2 → STAT5 → Blimp1 signaling axis prevents Tfh differentiation. This was shown recently by both in vivo and in vitro studies. A two-fold reduction in IL-2Rα expression (IL-2Rα+/−) resulted in a significantly higher frequency of Bcl6+CXCR5+ Tfh cells development after acute viral infection [53]. Downstream of strong IL-2 signals, STAT5 mediated Blimp1 induction appears to inhibit Tfh differentiation, as a constitutively active STAT5 molecule did not prevent Tfh differentiation in Prdm1 (the gene that encodes Blimp1) deficient CD4 T cells [53]. IL-2 mediated inhibition of Tfh differentiation was also reported during in vitro CD4 T cell differentiation. High concentrations of IL-2 in Th1 cell culture media inhibits Bcl6 expression [54], which is discussed more in detail below. Blimp1 was also found to not only inhibit Bcl6, but also directly repress multiple genes associated with Tfh cell differentiation, including CXCR5 [54].
Foxo1
Forkhead box O (FOXO) transcription factors regulate a variety of important cellular processes, such as cell cycle progression, DNA repair, and glucose metabolism [55,56]. Unexpectedly, CD4 T cell specific deletion of FOXO1 leads to spontaneous formation of germinal centers and anti-DNA antibody production in mice, which was strongly associated with aberrant accumulation of Tfh cells [57]. This study indicated a potential role of FOXO1 as a negative regulator of Bcl6 expression. Intriguingly, another study suggested that FOXO1 could participate in regulation of Bcl6 expression as a positive regulator. In low IL-2 concentrations, FOXO1 was found to bind to Bcl6 promoter in CD4 T cells, which was associated with higher Bcl6 expression compare to high IL-2 conditions, indicating positive roles of FOXO1 for Bcl6 induction [54]. As such, further studies are necessary to address more clearly the function of FOXO1 for Tfh differentiation. Moreover, given that PI3K activation leads to Akt mediated phosphorylation of FOXO1, inactivating FOXO1 [58,59], it will be interesting to study whether and/or how FOXO1 mediated modulation of Bcl6 expression may intersect with signaling pathway downstream of ICOS (Figure 1).
SOCS3
SOCS proteins negatively regulate cytokine mediated activation signals, and as such are involved in many aspects of immune system regulation [60]. SOCS-3 is known to function as a potent negative feedback loop of IL-6 signal transducer (gp130) mediated STAT activation [61,62]. IL-6 mediated STAT1-STAT3 activation conveys Bcl6 induction signals for early Tfh differentiation [18]. Hence, IL-6 mediated SOCS-3 activation has a potential to inhibit Tfh differentiation. Recently, we observed SOCS-3 indeed negatively regulates Tfh differentiation (Choi and Crotty, unpublished data). Retroviral overexpression of SOCS-3 shRNAmir in antigen-specific CD4 T cells resulted in enhanced Tfh differentiation during the DC priming stage after an acute LCMV infection (Choi and Crotty, unpublished data). SOCS-3 presumably inhibits Tfh differentiation by curtailing IL-6 → STAT1-STAT3 → Bcl6 signaling axes (Figure 1). However, how SOCS-3, or other negative regulators of cytokine signaling, regulates Tfh differentiation remains to be understood.
T-bet
IL-12 induces Bcl6 expression in in vitro stimulated CD4 T cells through STAT4 activation [34]. The importance of IL-12 for T-bet induction and Th1 differentiation has been known for decades [63]. Thus, it is intriguing that a single cytokine induces master regulators for two different effector CD4 T cells. T-bet appears to inhibit Bcl6 expression, as augmented Bcl6 expression was observed after IL-12 stimulation in the absence of T-bet [34]. A recent study by Weinmann and colleagues provided mechanistic insight for how T-bet and Bcl6 interact in determining Tfh or Th1 cell differentiation. In Th1 culture conditions, both Bcl6 and T-bet are expressed and form a complex [54]. Bcl6 function was modulated in response to changing IL-2 concentrations. In the presence of high IL-2 concentrations, Bcl6 failed to repress its target genes because T-bet bound and masked the DNA binding domain of Bcl6. Conversely, when the Bcl6 to T-bet ratio was inverted under low IL-2 conditions, the CD4 T cells acquired a partial Tfh gene expression profile, including Bcl6 mediated Blimp1 repression. Therefore, the Bcl6:T-bet ratio is key. More IL-2 signals enhance Th1 differentiation, whereas strong IL-6, IL-21, or ICOS mediated signals result in Tfh differentiation (Figure 1).
The signaling circuits that regulate Bcl6 expression: Could they be targeted in new vaccine strategies?
Successful vaccines depend on many parameters, including the antigens and epitopes incorporated in the vaccine, the route of administration, and the type of adjuvant used. Adjuvants contribute to adaptive immunity by shaping the magnitude and quality of innate immune responses [64]. Many different adjuvants have been tested using animal models for their ability to bias CD4 T cell differentiation in one direction or another. Mycobacterial products in complete Freund’s adjuvant strongly elicit Th1 response. Incomplete Freund’s adjuvant induces a less biased response. Monophosphoryl lipid A, a detoxified version of LPS, augments Th1 responses [65]. Alum induces a strong Th2 response in mice; however, alum elicits a largely unbiased CD4 T cell response in humans. An approach for enhancing Tfh differentiation by candidate vaccines would be to modulate positive and negative signaling pathways to induce Bcl6 expression in CD4 T cells. However, there is no simple way to accomplish this as of yet. Regulation of Bcl6 in CD4 T cells is controlled by multiple layers of signaling pathways, some of which are shared with non-Tfh cells or engage potent negative feedback loops. Given the depth of mechanistic insights revealed about Bcl6 regulation in relatively short period of time since the importance of Bcl6 in Tfh differentiation was first identified, it is likely that studies in the near future will clarify the complex signaling circuitries regulating Bcl6 induction in CD4 T cells. Then, hopefully, an understanding of how Bcl6 expression is regulated in CD4 T cells could be applied to facilitate rational vaccine design for more powerful B cell immunity against infectious diseases.
Highlights.
-
-
Bcl6 expression is regulated complex signaling circuitries in CD4 T cells.
-
-
The signaling pathways that regulate Bcl6 expression in positive and negative ways are discussed in detail.
-
-
Future studies are suggested to reveal more clearly how Bcl6 is regulated in CD4 T cells, which will contribute to better vaccine development.
Acknowledgements
We would like to thank Simon Belanger, Joyce Hu, and John Nance for helpful comments and suggestion on the manuscript.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References and recommended reading
Papers of particular interest, published within the period of review, have been highlighted as:
* of special interest
** of outstanding interest
- 1.Taubenberger JK, Reid AH, Janczewski TA, Fanning TG. Integrating historical, clinical and molecular genetic data in order to explain the origin and virulence of the 1918 Spanish influenza virus. Philosophical transactions of the Royal Society of London. Series B, Biological sciences. 2001;356:1829–1839. doi: 10.1098/rstb.2001.1020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Burton DR, Ahmed R, Barouch DH, Butera ST, Crotty S, Godzik A, Kaufmann DE, McElrath MJ, Nussenzweig MC, Pulendran B, et al. A Blueprint for HIV Vaccine Discovery. Cell Host & Microbe. 2012;12:396–407. doi: 10.1016/j.chom.2012.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Stamatatos L. HIV vaccine design: the neutralizing antibody conundrum. Current Opinion in Immunology. 2012;24:316–323. doi: 10.1016/j.coi.2012.04.006. [DOI] [PubMed] [Google Scholar]
- 4.Jenner E. An inquiry into the causes and effects of the variolae vaccinae. London: 1798. [Google Scholar]
- 5.Behbehani AM. The smallpox story: life and death of an old disease. Microbiological reviews. 1983;47:455–509. doi: 10.1128/mr.47.4.455-509.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Amanna IJ, Slifka MK, Crotty S. Immunity and immunological memory following smallpox vaccination. Immunological reviews. 2006;211:320–337. doi: 10.1111/j.0105-2896.2006.00392.x. [DOI] [PubMed] [Google Scholar]
- 7.Victora GD, Nussenzweig MC. Germinal centers. Annual Review of Immunology. 2012;30:429–457. doi: 10.1146/annurev-immunol-020711-075032. [DOI] [PubMed] [Google Scholar]
- 8.Stavnezer J, Guikema JEJ, Schrader CE. Mechanism and regulation of class switch recombination. Annual Review of Immunology. 2008;26:261–292. doi: 10.1146/annurev.immunol.26.021607.090248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Muramatsu M, Kinoshita K, Fagarasan S, Yamada S, Shinkai Y, Honjo T. Class switch recombination and hypermutation require activation-induced cytidine deaminase (AID), a potential RNA editing enzyme. Cell. 2000;102:553–563. doi: 10.1016/s0092-8674(00)00078-7. [DOI] [PubMed] [Google Scholar]
- 10.Crotty S. Follicular helper CD4 T cells (TFH) Annual Review of Immunology. 2011;29:621–663. doi: 10.1146/annurev-immunol-031210-101400. [DOI] [PubMed] [Google Scholar]
- 11.Johnston RJ, Poholek AC, Ditoro D, Yusuf I, Eto D, Barnett B, Dent AL, Craft J, Crotty S. Bcl6 and Blimp-1 Are Reciprocal and Antagonistic Regulators of T Follicular Helper Cell Differentiation. Science. 2009;325:1006–1010. doi: 10.1126/science.1175870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Nurieva RI, Chung Y, Martinez GJ, Yang XO, Tanaka S, Matskevitch TD, Wang Y-H, Dong C. Bcl6 mediates the development of T follicular helper cells. Science. 2009;325:1001–1005. doi: 10.1126/science.1176676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Yu D, Rao S, Tsai LM, Lee SK, He Y, Sutcliffe EL, Srivastava M, Linterman M, Zheng L, Simpson N, et al. The transcriptional repressor Bcl-6 directs T follicular helper cell lineage commitment. Immunity. 2009;31:457–468. doi: 10.1016/j.immuni.2009.07.002. The first demonstration that Bcl6, the transcription repressor, is a master regulator of Tfh differentiation of CD4 T cells.
- 14.Diehl S, Rincon M. The two faces of IL-6 on Th1/Th2 differentiation. Molecular immunology. 2002;39:531–536. doi: 10.1016/s0161-5890(02)00210-9. [DOI] [PubMed] [Google Scholar]
- 15.Choi YS, Eto D, Yang JA, Lao C, Crotty S. Cutting Edge: STAT1 Is Required for IL-6-Mediated Bcl6 Induction for Early Follicular Helper Cell Differentiation. Journal of Immunology. 2013;190:3049–3053. doi: 10.4049/jimmunol.1203032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Heinrich PC, Behrmann I, Müller-Newen G, Schaper F, Graeve L. Interleukin-6-type cytokine signalling through the gp130/Jak/STAT pathway. Biochem J. 1998;334:297–314. doi: 10.1042/bj3340297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nurieva RI, Chung Y, Hwang D, Yang XO, Kang HS, Ma L, Wang Y-h, Watowich SS, Jetten AM, Tian Q, et al. Generation of T follicular helper cells is mediated by interleukin-21 but independent of T helper 1, 2, or 17 cell lineages. Immunity. 2008;29:138–149. doi: 10.1016/j.immuni.2008.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Choi YS, Eto D, Yang JA, Lao C, Crotty S. STAT1 is required for IL-6 mediated Bcl6 induction for early Tfh differentiation. J Immunol. 2013 doi: 10.4049/jimmunol.1203032. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hotson AN, Hardy JW, Hale MB, Contag CH, Nolan GP. The T cell STAT signaling network is reprogrammed within hours of bacteremia via secondary signals. Journal of Immunology. 2009;182:7558–7568. doi: 10.4049/jimmunol.0803666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Milner JD, Brenchley JM, Laurence A, Freeman AF, Hill BJ, Elias KM, Kanno Y, Spalding C, Elloumi HZ, Paulson ML, et al. Impaired T(H)17 cell differentiation in subjects with autosomal dominant hyper-IgE syndrome. Nature. 2008;452:773–776. doi: 10.1038/nature06764. This study demonstrated that STAT3 is required for Th17 cell differentiation of human CD4 T cells.
- 21.Ma CS, Avery DT, Chan A, Batten M, Bustamante J, Boisson-Dupuis S, Arkwright PD, Kreins AY, Averbuch D, Engelhard D, et al. Functional STAT3 deficiency compromises the generation of human T follicular helper cells. Blood. 2012;119:3997–4008. doi: 10.1182/blood-2011-11-392985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Liao W, Lin J-X, Wang L, Li P, Leonard WJ. Modulation of cytokine receptors by IL-2 broadly regulates differentiation into helper T cell lineages. Nature Immunololgy. 2011;12:551–559. doi: 10.1038/ni.2030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Eto D, Lao C, Ditoro D, Barnett B, Escobar TC, Kageyama R, Yusuf I, Crotty S. IL-21 and IL-6 are critical for different aspects of B cell immunity and redundantly induce optimal follicular helper CD4 T cell (Tfh) differentiation. PloS one. 2011;6:e17739. doi: 10.1371/journal.pone.0017739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Karnowski A, Chevrier S, Belz GT, Mount A, Emslie D, D'Costa K, Tarlinton DM, Kallies A, Corcoran LM. B and T cells collaborate in antiviral responses via IL-6, IL-21, and transcriptional activator and coactivator, Oct2 and OBF-1. Journal of Experimental Medicine. 2012;209:2049–2064. doi: 10.1084/jem.20111504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Poholek AC, Hansen K, Hernandez SG, Eto D, Chandele A, Weinstein JS, Dong X, Odegard JM, Kaech SM, Dent AL, et al. In vivo regulation of Bcl6 and T follicular helper cell development. Journal of Immunology. 2010;185:313–326. doi: 10.4049/jimmunol.0904023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Crotty S. The 1-1-1 fallacy. Immunol Rev. 2012;247:133–142. doi: 10.1111/j.1600-065X.2012.01117.x. [DOI] [PubMed] [Google Scholar]
- 27.Suto A, Kashiwakuma D, Kagami S-i, Hirose K, Watanabe N, Yokote K, Saito Y, Nakayama T, Grusby MJ, Iwamoto I, et al. Development and characterization of IL-21-producing CD4+ T cells. Journal of Experimental Medicine. 2008;205:1369–1379. doi: 10.1084/jem.20072057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Asao H, Okuyama C, Kumaki S, Ishii N, Tsuchiya S, Foster D, Sugamura K. Cutting edge: the common gamma-chain is an indispensable subunit of the IL-21 receptor complex. Journal of Immunology. 2001;167:1–5. doi: 10.4049/jimmunol.167.1.1. [DOI] [PubMed] [Google Scholar]
- 29.Linterman MA, Beaton L, Yu D, Ramiscal RR, Srivastava M, Hogan JJ, Verma NK, Smyth MJ, Rigby RJ, Vinuesa CG. IL-21 acts directly on B cells to regulate Bcl-6 expression and germinal center responses. Journal of Experimental Medicine. 2010;207:353–363. doi: 10.1084/jem.20091738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Choi YS, Kageyama R, Eto D, Escobar TC, Johnston RJ, Monticelli L, Lao C, Crotty S. ICOS receptor instructs T follicular helper cell versus effector cell differentiation via induction of the transcriptional repressor Bcl6. Immunity. 2011;34:932–946. doi: 10.1016/j.immuni.2011.03.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Goenka R, Barnett LG, Silver JS, O'Neill PJ, Hunter CA, Cancro MP, Laufer TM. Cutting edge: dendritic cell-restricted antigen presentation initiates the follicular helper T cell program but cannot complete ultimate effector differentiation. Journal of Immunology. 2011;187:1091–1095. doi: 10.4049/jimmunol.1100853. These studies demonstrated that dendritic cell priming of CD4 T cells is required and sufficient for initiation of Tfh cell differentiation.
- 32.Ma CS, Suryani S, Avery DT, Chan A, Nanan R, Santner-Nanan B, Deenick EK, Tangye SG. Early commitment of naïve human CD4+ T cells to the T follicular helper (TFH) cell lineage is induced by IL-12. Immunology and cell biology. 2009;87:590–600. doi: 10.1038/icb.2009.64. [DOI] [PubMed] [Google Scholar]
- 33.Watford WT, Hissong BD, Bream JH, Kanno Y, Muul L, O'Shea JJ. Signaling by IL-12 and IL-23 and the immunoregulatory roles of STAT4. Immunological reviews. 2004;202:139–156. doi: 10.1111/j.0105-2896.2004.00211.x. [DOI] [PubMed] [Google Scholar]
- 34.Nakayamada S, Kanno Y, Takahashi H, Jankovic D, Lu KT, Johnson TA, Sun H-w, Vahedi G, Hakim O, Handon R, et al. Early Th1 Cell Differentiation Is Marked by a Tfh Cell-like Transition. Immunity. 2011;35:919–931. doi: 10.1016/j.immuni.2011.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Lee SK, Silva DG, Martin JL, Pratama A, Hu X, Chang P-P, Walters G, Vinuesa CG. Interferon-γ Excess Leads to Pathogenic Accumulation of Follicular Helper T Cells and Germinal Centers. Immunity. 2012;37:880–892. doi: 10.1016/j.immuni.2012.10.010. These studies discuss that Tfh differentiation program shares signaling pathways of Th1 cell differentiation.
- 36.Vinuesa CG, Cook MC, Angelucci C, Athanasopoulos V, Rui L, Hill KM, Yu D, Domaschenz H, Whittle B, Lambe T, et al. A RING-type ubiquitin ligase family member required to repress follicular helper T cells and autoimmunity. Nature. 2005;435:452–458. doi: 10.1038/nature03555. [DOI] [PubMed] [Google Scholar]
- 37.O'Shea JJ, Plenge R. JAK and STAT signaling molecules in immunoregulation and immune-mediated disease. Immunity. 2012;36:542–550. doi: 10.1016/j.immuni.2012.03.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Gigoux M, Shang J, Pak Y, Xu M, Choe J, Mak TW, Suh W-K. Inducible costimulator promotes helper T-cell differentiation through phosphoinositide 3-kinase. Proceedings of the National Academy of Sciences of the United States of America. 2009;106:20371–20376. doi: 10.1073/pnas.0911573106. This study demonstrates that PI3K activation downstream of ICOS receptor on CD4 T cells is critical for Tfh differentiation.
- 39.Hu H, Wu X, Jin W, Chang M, Cheng X, Sun S-C. Noncanonical NF-kappaB regulates inducible costimulator (ICOS) ligand expression and T follicular helper cell development. Proceedings of the National Academy of Sciences of the United States of America. 2011;108:12827–12832. doi: 10.1073/pnas.1105774108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Rolf J, Bell SE, Kovesdi D, Janas ML, Soond DR, Webb LMC, Santinelli S, Saunders T, Hebeis B, Killeen N, et al. Phosphoinositide 3-kinase activity in T cells regulates the magnitude of the germinal center reaction. Journal of Immunology. 2010;185:4042–4052. doi: 10.4049/jimmunol.1001730. [DOI] [PubMed] [Google Scholar]
- 41.So T, Choi H, Croft M. OX40 complexes with phosphoinositide 3-kinase and protein kinase B (PKB) to augment TCR-dependent PKB signaling. Journal of Immunology. 2011;186:3547–3555. doi: 10.4049/jimmunol.1003156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Rudd CE, Schneider H. Unifying concepts in CD28, ICOS and CTLA4 co-receptor signalling. Nature Reviews Immunology. 2003;3:544–556. doi: 10.1038/nri1131. [DOI] [PubMed] [Google Scholar]
- 43.Ise W, Kohyama M, Schraml BU, Zhang T, Schwer B, Basu U, Alt FW, Tang J, Oltz EM, Murphy TL, et al. The transcription factor BATF controls the global regulators of class-switch recombination in both B cells and T cells. Nature Immunology. 2011;12:536–543. doi: 10.1038/ni.2037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Bauquet AT, Jin H, Paterson AM, Mitsdoerffer M, Ho I-C, Sharpe AH, Kuchroo VK. The costimulatory molecule ICOS regulates the expression of c-Maf and IL-21 in the development of follicular T helper cells and TH-17 cells. Nature Immunology. 2009;10:167–175. doi: 10.1038/ni.1690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Kroenke MA, Eto D, Locci M, Cho M, Davidson T, Haddad EK, Crotty S. Bcl6 and Maf cooperate to instruct human follicular helper CD4 T cell differentiation. Journal of Immunology. 2012;188:3734–3744. doi: 10.4049/jimmunol.1103246. This study demonstrates that Bcl6 instructs Tfh differentitaion program in human CD4 T cells.
- 46.Bollig N, Brüstle A, Kellner K, Ackermann W, Abass E, Raifer H, Camara B, Brendel C, Giel G, Bothur E, et al. Transcription factor IRF4 determines germinal center formation through follicular T-helper cell differentiation. Proceedings of the National Academy of Sciences of the United States of America. 2012;109:8664–8669. doi: 10.1073/pnas.1205834109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kwon H, Thierry-Mieg D, Thierry-Mieg J, Kim H-P, Oh J, Tunyaplin C, Carotta S, Donovan CE, Goldman ML, Tailor P, et al. Analysis of interleukin-21-induced Prdm1 gene regulation reveals functional cooperation of STAT3 and IRF4 transcription factors. Immunity. 2009;31:941–952. doi: 10.1016/j.immuni.2009.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Streeck H, D'Souza MP, Littman DR, Crotty S. Harnessing CD4(+) T cell responses in HIV vaccine development. Nature Medicine. 2013;19:143–149. doi: 10.1038/nm.3054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Pepper M, Pagán AJ, Igyártó BZ, Taylor JJ, Jenkins MK. Opposing signals from the bcl6 transcription factor and the interleukin-2 receptor generate T helper 1 central and effector memory cells. Immunity. 2011;35:583–595. doi: 10.1016/j.immuni.2011.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Choi YS, Yang JA, Yusuf I, Johnston RJ, Greenbaum J, Peters B, Crotty S. Bcl6 expressing follicular helper CD4 T cells are fate committed early and have the capacity to form memory. J Immunol. 2013 doi: 10.4049/jimmunol.1202963. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Gilmour KC, Pine R, Reich NC. Interleukin 2 activates STAT5 transcription factor (mammary gland factor) and specific gene expression in T lymphocytes. Proceedings of the National Academy of Sciences of the United States of America. 1995;92:10772–10776. doi: 10.1073/pnas.92.23.10772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gong D, Malek TR. Cytokine-dependent Blimp-1 expression in activated T cells inhibits IL-2 production. Journal of immunology. 2007;178:242–252. doi: 10.4049/jimmunol.178.1.242. [DOI] [PubMed] [Google Scholar]
- 53.Johnston RJ, Choi YS, Diamond JA, Yang JA, Crotty S. STAT5 is a potent negative regulator of TFH cell differentiation. Journal of Experimental Medicine. 2012;209:243–250. doi: 10.1084/jem.20111174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Oestreich KJ, Mohn SE, Weinmann AS. Molecular mechanisms that control the expression and activity of Bcl-6 in T(H)1 cells to regulate flexibility with a T(FH)-like gene profile. Nature Immunology. 2012;13:405–411. doi: 10.1038/ni.2242. This study demonstrates how CD4 T cells determine differentiation pathways towards Th1 or Tfh effector CD4 T cells under Th1 biased cell culture condition.
- 55.Stahl M, Dijkers PF, Kops GJPL, Lens SMA, Coffer PJ, Burgering BMT, Medema RH. The forkhead transcription factor FoxO regulates transcription of p27Kip1 and Bim in response to IL-2. Journal of Immunology. 2002;168:5024–5031. doi: 10.4049/jimmunol.168.10.5024. [DOI] [PubMed] [Google Scholar]
- 56.Kousteni S. FoxO1, the transcriptional chief of staff of energy metabolism. Bone. 2012;50:437–443. doi: 10.1016/j.bone.2011.06.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kerdiles YM, Stone EL, Beisner DL, Mcgargill MA, Ch'en IL, Stockmann C, Katayama CD, Hedrick SM. Foxo Transcription Factors Control Regulatory T Cell Development and Function. Immunity. 2010;33:890–904. doi: 10.1016/j.immuni.2010.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Brunet A, Bonni A, Zigmond MJ, Lin MZ, Juo P, Hu LS, Anderson MJ, Arden KC, Blenis J, Greenberg ME. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell. 1999;96:857–868. doi: 10.1016/s0092-8674(00)80595-4. [DOI] [PubMed] [Google Scholar]
- 59.Kps GJ, de Ruiter ND, De Vries-Smits AM, Powell DR, Bos JL, Burgering BM. Direct control of the Forkhead transcription factor AFX by protein kinase B. Nature. 1999;398:630–634. doi: 10.1038/19328. [DOI] [PubMed] [Google Scholar]
- 60.Alexander WS. Suppressors of cytokine signalling (SOCS) in the immune system. Nature Reviews Immunology. 2002;2:410–416. doi: 10.1038/nri818. [DOI] [PubMed] [Google Scholar]
- 61.Kim H, Hawley TS, Hawley RG, Baumann H. Protein tyrosine phosphatase 2 (SHP-2) moderates signaling by gp130 but is not required for the induction of acute-phase plasma protein genes in hepatic cells. Molecular and cellular biology. 1998;18:1525–1533. doi: 10.1128/mcb.18.3.1525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Nicholson SE, De Souza D, Fabri LJ, Corbin J, Willson TA, Zhang JG, Silva A, Asimakis M, Farley A, Nash AD, et al. Suppressor of cytokine signaling-3 preferentially binds to the SHP-2-binding site on the shared cytokine receptor subunit gp130. Proceedings of the National Academy of Sciences of the United States of America. 2000;97:6493–6498. doi: 10.1073/pnas.100135197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Jacobson NG, Szabo SJ, Weber-Nordt RM, Zhong Z, Schreiber RD, Darnell JE, Murphy KM. Interleukin 12 signaling in T helper type 1 (Th1) cells involves tyrosine phosphorylation of signal transducer and activator of transcription (Stat)3 and Stat4. Journal of Experimental Medicine. 1995;181:1755–1762. doi: 10.1084/jem.181.5.1755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Wilson-Welder JH, Torres MP, Kipper MJ, Mallapragada SK, Wannemuehler MJ, Narasimhan B. Vaccine adjuvants: current challenges and future approaches. Journal of pharmaceutical sciences. 2009;98:1278–1316. doi: 10.1002/jps.21523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Mata-Haro V, Cekic C, Martin M, Chilton PM, Casella CR, Mitchell TC. The vaccine adjuvant monophosphoryl lipid A as a TRIF-biased agonist of TLR4. Science. 2007;316:1628–1632. doi: 10.1126/science.1138963. [DOI] [PubMed] [Google Scholar]