Abstract
Purpose:
We evaluated whether existing ultra-low vision tests are suitable for measuring outcomes using sensory substitution. The BrainPort is a vision assist device coupling a live video feed with an electrotactile tongue display, allowing a user to gain information about their surroundings.
Methods:
We enrolled 30 adult subjects (age range 22–74) divided into two groups. Our blind group included 24 subjects (n = 16 males and n = 8 females, average age 50) with light perception or worse vision. Our control group consisted of six subjects (n = 3 males, n = 3 females, average age 43) with healthy ocular status. All subjects performed 11 computer-based psychophysical tests from three programs: Basic Assessment of Light Motion, Basic Assessment of Grating Acuity, and the Freiburg Vision Test as well as a modified Tangent Screen. Assessments were performed at baseline and again using the BrainPort after 15 hours of training.
Results:
Most tests could be used with the BrainPort. Mean success scores increased for all of our tests except contrast sensitivity. Increases were statistically significant for tests of light perception (8.27 ± 3.95 SE), time resolution (61.4% ± 3.14 SE), light localization (44.57% ± 3.58 SE), grating orientation (70.27% ± 4.64 SE), and white Tumbling E on a black background (2.49 logMAR ± 0.39 SE). Motion tests were limited by BrainPort resolution.
Conclusions:
Tactile-based sensory substitution devices are amenable to psychophysical assessments of vision, even though traditional visual pathways are circumvented.
Translational Relevance:
This study is one of many that will need to be undertaken to achieve a common outcomes infrastructure for the field of artificial vision.
Keywords: sensory substitution, BrainPort, outcomes
Introduction
Blindness is a severe disability affecting over 36 million people worldwide, including 694,000 in the United States.1 The retinal implant is currently the frontrunner of artificial vision technology, though its use is limited to those with functioning optic nerves.2–4 Sensory substitution devices aim to circumvent primary visual pathways and try to provide visual information through nonvisual, afferent circuits.5–8 Sensory substitution devices exploit input from the remaining intact auditory or tactile senses, and may be an option for patients who no longer have eyes or functional ocular components. With rehabilitation and appropriate training, the brain can learn to translate this input into visual qualia.9,10 In addition, our work, as well as that of others, shows that the visual cortex is activated by sensory substitution.10–14 While there is some evidence that sensory substitution might be a viable method to improve appreciation of the environment among the blind, there are no studies that attempt to systematically evaluate outcomes measures for this modality of “vision” restoration.
Presently, the resolution of artificial vision and sensory substitution devices provide what is termed “ultra-low vision,” allowing for crude perceptions of larger, high contrast objects.9,15,16 Because current artificial vision technology cannot recapitulate the complexity of normal vision, traditional clinical methods of assessing visual function are not appropriate. Establishing objective methods to determine improvement in function is vital to gauge performance improvement, to compare subsequent device iterations, and for regulatory approval. Moreover, if such outcomes can be standardized, comparisons between different types of artificial vision devices would be facilitated17 (www.nei.nih.gov/news/meetings/fDA_2011.asp). Several studies have used various techniques such as light detection, square pointing, object recognition, mobility tasks, and resolution as ways to show improvement over baseline for patients using artificial vision (Friberg TNA, et al. IOVS. 2011;52:ARVO E-Abstract 109; Nau A, et al. IOVS. 2011;52:ARVO E-Abstract 461).4,18–21 At the present time, there is no agreement among researchers working in these nascent fields regarding the most appropriate outcomes tools. Some aspect of functional ability should be considered, and is recognized in the field of low vision.22 In addition, good arguments can be made that outcomes assessments need to go a step further to incorporate patient reported outcomes and quality of life measures.23 To date, there has not been a comprehensive attempt to systematically measure the sensory experiences that are enabled by visual sensory substitution devices. Interesting questions arise as to whether perceptions mediated through nonstandard pathways produce sensations that can be measured with tests designed to quantify vision, per se. In spite of the proven activation of visual cortex with sensory substitution, bootstrapping visual information onto an alternative afferent stream could elicit atypical or aberrant outputs. There is limited proof beyond anecdotal reports that any improvement can be conferred by use of these devices, partially because there are no accepted outcomes measures with which to demonstrate efficacy. Additional gaps in knowledge include whether the existing outcomes measures used for some retinal implant chips (also producing states of ultra-low vision) might be useful for sensory substitution, as well as also how to set up and administer the tests. This paper describes our efforts to systematically investigate whether existing, validated tests of ultra-low vision would need to be modified when used in the context of sensory substitution.
The BrainPort is a vision assist device that uses an electrotactile sensory substitution strategy.8,15,24 The unit consists of a custom design, grayscale image sensor with a wide angle lens (Aptima Imaging Corporation, San Jose, CA) mounted in the center of a pair of sunglasses (Oakley, Inc., Foothill Ranch, CA) that sends live video to a handheld processor with user controls. The processor, in turn, down samples the video and displays the altered images as an electrotactile sensation on the surface of the tongue. The stimulus is presented via a square array of 400 electrodes embedded in a tethered, removable resin lollipop called the intra oral device (IOD). The BrainPort has now received CE Mark clearance and has advanced to the point of pending Food and Drug Administration approval. Several investigators have shown that it cannot only confer some functional improvement, but also that it can activate visual cortex (Friberg TNA, et al. IOVS. 2011;52:ARVO E-Abstract 109; Nau A, et al. IOVS. 2011;52:ARVO E-Abstract 461).9,5,13,15 The fact that the device is completely noninvasive permits enrollment of sufficient numbers of study subjects to generate statistically robust results. It, therefore, provided an ideal tool for initial testing of outcomes assessments. Further work will have to be done to determine whether these same tests are generalizable to other sensory substitution devices such as The vOICe (MetaModal, LLC, Pasadena, CA) or Aux Deco (EyePlusPlus, Inc., Toyko, Japan).
A battery of computer-based tests developed as quantitative examination tools for very low vision assessment was chosen to evaluate function and acuity, namely the Basic Light and Motion Test (BaLM),25 the Basic Grating Acuity Test (BaGA),18 and the Freiburg Acuity and Contrast Test (FrACT).24,26 Our rationale for using these specific tests as opposed to alternative tests of very low vision states27,28 was that they had also been used recently by Zrenner et al. to quantify vision with a subretinal artificial vision chip.18 Additionally, we describe the evaluation of the extent of visual field using a modified tangent screen.
Materials and Methods
We enrolled 30 adult subjects in this observational cohort pilot study (age range 22–74) divided into two groups. Our blind group included 24 subjects (n = 16 males and n = 8 females) who were bilaterally blind with light perception or worse vision from a variety of noncortical etiologies (Table 1). Subjects in the blind group were further subdivided based on the duration of blindness. We defined blindness as the year that light perception or worse vision occurred bilaterally. Exclusion criteria included cortical blindness, current smoking, oral lesions or piercings, tongue abnormalities, and any contraindication to neuroimaging. Testing of psychophysical outcomes described in this study were part of a larger research program that also included visual evoked potential testing, obstacle course navigation, depression screens, quality of life questionnaires, and neuroimaging studies. Because there is no predicate data using our tests with sensory substitution, this study based the power calculations on data gleaned from the literature regarding neuroimaging studies of sensory substitution. Means and SDs were used to calculate the effect size that was needed for the appropriate statistical test for our positron emission tomography (PET) and magnetic resonance imagining (MRI) scans. All power calculations were performed using PASW 18.0 software (IBM, Armonk, NY). For our hypotheses, we would have 85% power to detect significant correlations of large effect size (r = 0.5) with 24 blind patients.
Table 1.
Categories of Subject Duration of Blindness, Number of Subjects in Each Group, and Etiologies of Blindness Per Category

Within the blind group, we created subgroups according to duration of blindness, which is detailed in Table 1. The average age for the blind group was 50 years. Our control group consisted of the remaining six subjects (n = 3 males, n = 3 females) with healthy vision and healthy ocular health status. Their average age was 43 years. All subjects were recruited from the University of Pittsburgh Medical Center (UPMC) Eye Center Sensory Substitution Lab's research registry. The registry is comprised of individuals who contacted the laboratory in order to participate in artificial vision research studies. Our protocol adhered to the tenets of the Declaration of Helsinki, was approved by University of Pittsburgh internal review board, and all subjects gave written informed consent before participation in the study.
The subjects performed 11 computer-based psychophysical tests from three programs: BaLM, BaGA, and FrACT according to the standard, published methods.24–26,29 These assessments, as well as a modified tangent screen were performed at baseline without the BrainPort (sighted controls were blindfolded), and again with the BrainPort after approximately 15 hours of structured training over the course of five consecutive days. The training regimen was not focused on recognition of the testing stimuli. Rather, it is a standard training protocol we have developed in cooperation with occupational therapists for attaining core skill acquisition with the BrainPort device that would allow for real world applications. The specific details of the training protocol are beyond the scope of this paper, but are briefly summarized. The training was broken up into 3 hour sessions occurring over the course of 5 days. Patients were introduced to the features of the BrainPort vision device including turning the device off and on, field of view/zoom features, intensity control, inversion controls, and so on. We demonstrated maintenance issues such as how to change batteries, how to care for the device, and troubleshooting strategies. Limitations of the technology such as lack of color, lack of three-dimensional depth perception, and that the stimulus is primarily a tactile percept were discussed. The next session involved creating a high contrast, virtually blank environment by seating the subject at a table covered in a piece of black felt, with a background wall similarly covered. The initial training task was to have the subject hold a white foam bar in their hand against the black background and experience and describe how the sensation on the tongue display changes as they change the orientation and/or distance of the bar. Building complexity, we progressed to identifying white felt shapes. Subjects were then introduced to perceiving, recognizing, and categorizing more complex shapes and symbols that are seen in activities of daily living such as letters and numbers of various sizes. Once they could identify individual letters, then multiple letters can be used to form words. Subjects were introduced to complex three-dimensional objects tasks, which explored the effects of perspective, shadows, and contrast. They were taught to confirm the location of these objects by reaching toward and grasping the objects, sorting, and stacking. In this stage, we were retraining the concepts of egocentric location and position and its relation to head movements. Subjects were taught two-dimensional depth perception strategies including object size, relative location of objects (i.e., higher or lower), and comparisons to known reference points. We introduced commonly encountered symbols such as exit signs, stop signs, men's and women's restroom signs, and so on, to teach them how to categorize similarly shaped symbols into groups to aid in identification. We subsequently introduced the skill of recognizing landmark information. Spatial relationships between oneself and an object of regard while ambulating were presented (i.e., is the object near or far) (Fig. 1).
Figure 1.
Early ambulation/spatial relations task training.
Advanced activities included tossing a bean bag into a bucket, discerning how far large objects are from one self as well as from each other, and walking along a path. Mobile scenarios were presented while incorporating head scanning and tracing. The concept of shore lining was discussed and demonstrated by navigating a hallway with turns or locating structures such as doorways and windows.
Testing Conditions
Our team spent the first year of the study conducting pilot tests on sighted, blindfolded subjects in order to optimize the testing conditions.
Set Up of BrainPort and Computer-Based Tests
The electrode array of the BrainPort IOD is arranged as 20 rows of 20 stainless steel electrodes of 0.762-mm diameter, spaced 0.32-mm apart on a tongue display unit that measures 29.5 mm × 33.8 mm × 7 mm overall. The BrainPort allows users to zoom the camera from 73° of field to less than 5° (diameters). To avoid variables of differing zoom across subjects, and the potential loss of target stimulus through reduced field or inaccurate aiming of the camera, we fixed the zoom at 44° for the entire computer testing conditions, which made the full computer display of the BaLM, BaGA, and FrACT tests correspond directly to the full tongue display. The BrainPort camera was mounted to a tripod to standardize the testing stimuli, and avoid the confounders related to unregistered scanning motion, which can cause confusion for untrained users of the BrainPort.
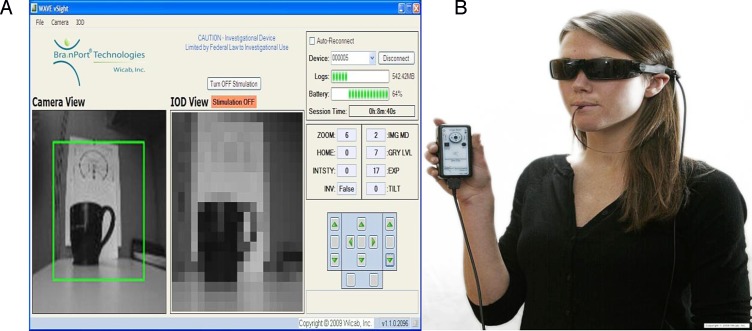
For all computer tests, the subjects were seated 0.5 m away from a 17 inch liquid crystal display (LCD) computer screen (HP Compaq La1951g, Hewlett Packard Co., Palo Alto, CA) in a darkened room, and responded to the onscreen stimulus via the nine key number pad on the keyboard. Error due to pixel resolution was not present because all measured acuity scores were well above the resolution limitations of the computer screen. The test administrators were equipped with a companion laptop wirelessly connected to the BrainPort, running a program (Vsight, Wicab, Inc. Middleton, WI) that allowed them to simultaneously view the video captured by the camera and the corresponding pixilated electrotactile stimulus presented to the subject's tongue (Fig. 2).
Figure 2.
(A) Screen shot of laptop, which is wirelessly connected to the BrainPort vision device. This Vsight Program allows researchers to visualize what is being relayed to the tongue in real time. (B) Shows the BrainPort as worn by a blind user during training. For outcomes testing described here, the camera/glasses were mounted to a tripod.
Light perception, time resolution, light localization, and motion detection were tested using the BaLM computer program. BaLM reported the percent correct and mean reaction time after 24 randomly ordered trials within each test. Light perception and time resolution use a two-alternative forced-choice method (2-AFC) where the subject has to enter a response within 1 minute or their response is counted as incorrect.
Light perception testing began with an auditory cue, then either a white full screen illumination lasting 200 ms was displayed or the screen remained black. This was repeated 12 times for each condition in random order. The subject responded whether or not they perceived an illumination after the cue using the number pad as described above. The time resolution task was similar, except the subject responded to a stimulus of either one 200 ms illumination or two 200 ms illuminations, separated by 200 ms on a black screen. Twelve trials of single illumination and 12 trials of double illumination were repeated in random order. The subject responded as having perceived either one or two illuminations, again using the keypad.
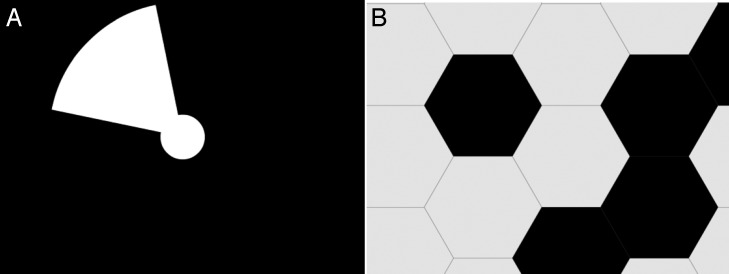
Location and motion tests used an 8-AFC with the same 1 minute timeout period as the light perception and time resolution tests. The location task measured the subject's ability to perceive the direction of projected light. The screen always contained a 5° central white fixation disk on a black background. Figure 3A shows the 15° white wedge stimulus that was projected from the central disk in one of eight directions (top, bottom, left, right, upper-left, lower-left, upper-right, and lower-right). The subject responded geographically via the number pad. Figure 3B demonstrates the motion stimulus, which consisted of a moving pattern of random 6.6° width black and white hexagons. The subject responded according to which of the eight directions the pattern was moving toward, again using the keypad with geographic reference.
Figure 3.
(A) BaLM directional stimulus. (B) BaLM motion stimulus, which was not well detected by the BrainPort due to lack of spatial resolution.
Grating acuity was measured using the BaGA computer program. BaGA reported the percentage of correct responses to the orientation of a square wave grating in one of four orientations (horizontal, vertical, upper left to lower right diagonal, and lower left to upper right diagonal). Twenty four random stimuli were presented, totaling six presentations for each orientation. BaGA was used as a 4-AFC with a 1 minute timeout. We set our grating stimuli to a suprathreshold level of 0.1 cycle per degree (cpd) for this study, where each bar in the grating was 46-mm thick and corresponded to 2.9 mm on the tongue. Subjects used the keypad to record their responses.
Visual acuity and contrast sensitivity were measured with the FrACT computer program. FrACT measured decimal acuity using both Landolt C and Tumbling E optotypes. The Tumbling E optotype was added to the FrACT program partway through our study, and as a result it was used with fewer total subjects (control n = 4, blind n = 22). We presented the Landolt C and Tumbling E tests both as a single white optotype on a black background and as a black optotype on a white background for a total of four independent tests. For each test, FrACT correspondingly enlarged or shrank the optotype depending on the subject's responses over 24 trials using Best Parameter Estimation by Sequential Testing (best-PEST) method to determine threshold acuity.24
Contrast sensitivity was measured with a 300 arcmin Landolt C as both a light optotype on a dark background and a dark optotype on a light background. For each variation, the contrast was increased or decreased depending on the subject's responses over 24 trials to determine contrast sensitivity threshold. All FrACT tests were used as 8-AFC with a 1 minute timeout. Again, as in all tests, subjects used the keypad to record their responses.
Visual Field Testing
Pilot results revealed that the Humphrey Field Analyzer (Carl Zeiss Meditech, Inc., Dublin, CA), Octopus perimeter (Haag Streit USA, Inc., Mason, OH), Humphrey Frequency Doubling Perimetry (Carl Zeiss Meditech, Inc.), and Goldmann perimeter (Marco, Jacksonville, FL) did not display a stimulus large enough for reliable perception with the BrainPort. The standard size targets had to be enlarged to be consistently perceived by BrainPort users. In addition, the overall size of the screen was enlarged so as to encompass the full field of the BrainPort camera in the default (i.e., nonzoomed) setting. Ultimately, a 2 × 2 m black tangent screen with a kinetic 50 mm white stimulus at a distance of 0.5 m was used to evaluate visual field. The test administrators were equipped with a companion laptop (Dell Mini, Dell, Inc., Round Rock, TX) wirelessly connected to the BrainPort, running the VSight (program that allowed them to simultaneously view the video captured by the camera and the corresponding pixilated electrotactile stimulus presented to the subject's tongue. For extent of visual field testing, we fixed the zoom at 73°, the widest angle of the BrainPort's camera.
Results
Our results demonstrate that psychophysical assessments of vision can be administered with tactile-based sensory substitution vision assist devices. The success threshold for identifying the stimulus correctly in the light perception, time resolution, location perception, motion perception, and grating acuity tests was established as halfway between chance rate (1/number of response alternatives) and 100% correct. Thus, a subject achieved success on a particular test when their percent of correct responses was at least halfway between chance rate and a perfect score. This success threshold corresponds to rates for 2AFC, 4AFC, and 8AFC at 75.0%, 62.5%, and 56.25%, respectively. This criterion for 24 trials results in the probability of exceeding the threshold by chance at 1.1%, 0.011%, and 0.000013% for 2-AFC, 4-AFC, and 8-AFC,25 respectively.
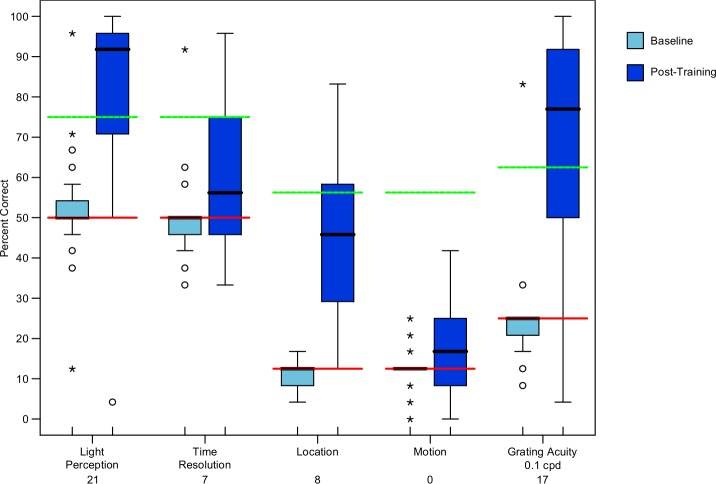
The Shapiro-Wilk test showed that our data were not normally distributed. We employed nonparametric statistical tests to conduct our analysis. Figure 4 is a box plot that summarizes our results for BaLM (light perception, time resolution, location, and motion tasks) and BaGA grating acuity tasks. The data indicate that subjects performed at chance level at baseline, which would be expected for a blind cohort. It should be noted that two of the subjects had light perception in one eye only and two had light perception bilaterally, and so for the light perception test, the performance in aggregate is slightly better than chance. The four subjects with light perception scored an average of 81.2% correct. These same subjects had no differences when compared with their totally blind counterparts for the other tests in the BaLM or BaGA programs. Motion perception, which contained aspects of both spatial and temporal resolution, and therefore was considered the most difficult test, was not perceived above success threshold by any of our subjects.
Figure 4.
A box plot that demonstrates the percent correct among our subjects for the BaLM and BaGA tests. Red lines show chance level, green lines show success threshold, and black lines show median scores. The number below the test on the x axis represents the number of subjects that achieved our success threshold for the given test. Light blue boxes indicate baseline and dark blue boxes indicate post training.
The FrACT acuity tests used in this study had a maximum logMAR score of 2.59, 2.60, or 2.70. This variance in maximum scores is due to the use of different 17 inch computer screens in the clinic, which were independently calibrated with the FrACT program. Because of this variation, all logMAR acuity scores at or above 2.59 were considered beyond the measurable threshold.
At baseline all subjects scored at or above the 2.59 logMAR threshold on all FrACT acuity tests and 100% on the light on dark and dark on light contrast tests. A significant increase in acuity scores was found for blind subjects after training when testing with a white Tumbling E on a black background (+0.14 logMAR [P = 0.001]). No significant increase in acuity or contrast sensitivity was found for the blind or control groups after training with the other FrACT tests. Our results show that subjects repeatedly were unable to correctly discriminate the rotating Landolt C stimulus both at baseline and after training. In addition, our data revealed that successful interpretation of the Tumbling E stimuli is heavily dependent on contrast, as performance with the dark E optotype was poorer than with the white E stimuli (Table 2). Based on these data, we recommend only using the white E on the black background at the 100% contrast setting. Subjects can use the inverse function on the BrainPort device if they wish to try the opposite contrast.
Table 2.
Median Percent Correct for Light Perception, Time Resolution, Location, Motion, and Grating Acuity
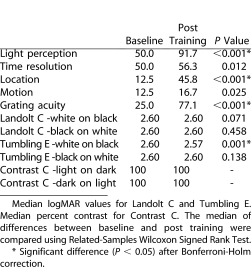
Table 2 summarizes the median of differences for all subjects on the computer-based tests between baseline condition and BrainPort condition. Significant improvements after Bonferroni-Holm correction are indicated by an asterisk. For a number of tests, we found statistically significant improvements after training. Nearly all subjects were able to detect light, over 75% were able to detect the direction of a sine wave grating and almost half could detect the location of a light stimulus. More complicated tests of time and motion did not show such robust improvements, but the tests were nonetheless measuring some change in performance. As mentioned, only the white E optotype on a dark background was useful for the FrACT test.
We examined whether duration of blindness had any effect on performance scores for the BaLM, BaGA, and FrACT tests. Table 3 shows that among our blind subjects, there is no apparent difference in performance between those with less than 10 years of blindness or more than 10 years of blindness.
Table 3.
Comparison of Outcomes According to Duration of Blindness
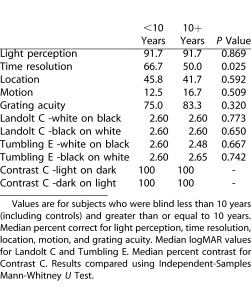
Finally, visual field testing results showed that no subjects perceived the tangent screen stimulus at baseline. The mean extent of visual field after training was 56.1° (SD 2.5), with 58.1° (SD 2.7) on the horizontal axis, and 54° (SD 2.5) on the vertical axis. There was no significant difference in scores among the blind when separated into duration of blindness subgroups using Independent-Samples Mann-Whitney U Test (P = 0.805). The mean tangent screen visual field (56°) was found to be less than the technical maximum of 73° field of view for the current BrainPort camera. We conclude that extent of visual field testing for ultra-low vision is best done using a large tangent screen with larger stimuli.
Discussion
This is the first study to systematically evaluate how to objectively evaluate a sensory substitution device using vision based psychophysical assessments. We found that the BrainPort provided a unique opportunity for testing different kinds of outcomes measures in large numbers of subjects with many different kinds and durations of blindness. In our cohort, the BaLM, BaGA, FrACT tests and enlarged tangent screen were tests that can, with some exceptions, be used for outcomes testing with the BrainPort. Tests of motion and contrast as well as use of the Landolt C optotypes could not be measured well. The resolution capabilities of the existing BrainPort were the main barriers to using these particular stimuli. For example, the BaLM motion stimuli resembled a Gaussian noise image when viewed with the IOD. Similarly, many of the subjects reported that the Landolt C stimulus was an “O,” and only high contrast settings need to be used for the BaGA test. Subsequent iterations of the IOD with higher electrode densities and/or training to the particular stimuli would likely increase performance. In the short term, our laboratory is actively developing alternative methods for testing motion.
We performed pilot testing for one year on blindfolded, sighted subjects to determine how to control our testing paradigms before enrolling our blind subjects. We found that several preset conditions were needed to achieve reproducible results. Some of the questions we needed to answer included determining what size stimuli could be perceived by the BrainPort and the appropriate testing distance for each proposed test. In addition, because in untrained subjects, motion of the head for scanning causes considerable confusion as the movement is not registered, we had to determine whether to mount the camera on a tripod or to allow the subject to manipulate the camera unaided. We also struggled with whether to control the field of view of the camera, which, if not preset to the perimeter of a computer (or tangent) screen, can confuse a subject with stimulus irrelevant information. Our ultimate decision to mount the camera for these specific tests was predicated on the need to properly center and zoom in the camera to make identical intersubject stimuli, and precedent in the literature.15 Other tests of object and word recognition as well as mobility that are being conducted in our laboratory do not use a mounted camera testing condition. The relative merits of each condition can be debated, but it is probably best to have a combination of both. Other issues were whether ambient illumination mattered. We found that changing the illumination of the room was not relevant since the BrainPort is equipped with luminance averaging software. This may not be the case for other devices.
Another critical consideration when attempting to measure acuity with a camera fed sensory substitution device is the potential to zoom in and out, which can skew acuity scores. For example, magnifying an image by zooming in will provide the user with greater detail but will also result in a loss of field of view. For instance, our blind subjects had a mean acuity score of 2.44 logMAR (20/5500 Snellen Equivalent) with a white tumbling E on a black background at a camera zoom limiting their field to 44°. This setup corresponded to an optotype gap size on the tongue of 2.6 mm. If the camera were zoomed in to 12°, the same measured acuity of 2.44 logMAR would correspond to a gap size on the tongue of 9.8 mm, over three times as large of a stimulus, but with less than a third of the visual field, providing higher spatial resolution. Two previous studies have tested the acuity of the tongue using more rudimentary electrotactile tongue devices with grids of 100 and 144 electrodes.9,15 These studies used Tumbling E optotypes. The study by Chebat, using a camera displaying 12° of field, found the average measured acuity of their 15 early blind participants to be 2.34 logMAR (20/4375 Snellen equivalent) after an average of 10 minutes of task-oriented training.9 The optotype gap on the tongue at that acuity would be 5.7 mm, which is over twice as large as the smallest gap of 2.6 mm resolved on average by our blind subjects. It should be noted that while the acuity score for our subjects was worse (2.44 logMAR) despite the less than half-sized letter on the tongue, our subjects had an over 3.5 times larger field of view. It is not surprising that our subjects were able to resolve a letter nearly half the size as that of Chebat's subjects because the resolution of the tongue display on the most current BrainPort has nearly twice as many electrodes in both the horizontal and vertical axes. The study by Sampaio measured visual acuity in one control and one congenitally blind to be 20/240 (1.07 LogMAR) after 9 hours of stimulus directed training.15 Unfortunately, it was not possible to usefully compare their results with ours, as they did not report the field of view of the camera used or the sizes of the optotypes on the tongue. The incongruity of the measurement scores among three studies highlights the need to account for both acuity and visual field extent when assessing functional perception in artificial vision devices. Indeed, allowing user to manipulate the zoom function and engage in modified visual search techniques in an unmounted camera may be more representative of real world conditions and show results that are different from those described in this study.
Recently, Striem-Amit et al. studied the visual acuity potential of an auditory based sensory substitution device called the VoICE.30 Their results, based on eight subjects trained for this task for an average of 73 hours, showed Tumbling E's could be resolved at the 20/200 level in some cases. Based on our own experience of training subjects, many are able to read individual letters and simple words after only 15 hours of training (data not shown). It is likely that with continued practice, our FrACT acuity scores would improve. It should be noted that our study subjects did not specifically train to the testing stimuli, and were exposed to the FrACT test at baseline and then again only on the last day of the study. It must be stressed that careful attention to the experimental conditions must be exercised when designing and comparing study results.
Temporal resolution testing with the BaLM test was hampered by the inherent temporal sampling capabilities of the BrainPort (200 ms), which places constraints on time resolution. It was noted by the test administrators while watching the companion laptop that many of the double flashes were displayed by the BrainPort as a longer, single flash. A subsequent pilot study performed by our lab with six blindfolded sighted controls suggests that an interstimulus interval of approximately 300 ms is necessary for reliable discrimination using the BrainPort (data not shown). Future outcomes measures for temporal resolution must have provisions to modify the sampling frequency so as to account for the inherent sampling limits of individual devices.
When performing a standard visual field evaluation in a sighted individual, the patient is instructed to fix their gaze at center of the fixation target. We attempted to mimic this procedure by aligning the center of the camera on a tripod with the center of the screen, which was confirmed by the sighted research subject using our linked workstation device. Yet, our subjects using the BrainPort could additionally move their tongues around on the IOD, effectively searching for the stimulus. The stimulus may move farther toward the center during this search process while the subject does not have their tongue in the correct location. While tongue movement may result in overly conservative fields, it may also suggest a more functional field in real life environments. A maximum field of 56° is still notably greater than the minimum field of around 30° required for normal mobility in sighted subjects that were limited to pixilated vision from a 25 × 25 array.31 This is an important finding in that it has direct implications for rehabilitation training, obstacle avoidance, and visual search techniques. For several subjects, however, the measured field of perception approached 70° and resembled the square shape of the IOD. The inward movement of the stimulus during a kinetic tangent screen evaluation may have led to an underestimation of field. Further testing with inward and outward directional kinetic stimuli will be undertaken to explore this hypothesis.
Secondary to our primary purpose of studying outcomes assessments for states of ultra-low vision in the context of sensory substitution, our results showed that mean success rates increased over baseline for all of our tests except contrast sensitivity when subjects used the BrainPort. This increase was statistically significant for the tests of light perception, localization, grating orientation, and white Tumbling E on a black background. This small pilot study suggests that that sensory substitution may eventually be a viable, noninvasive alternative for blind persons to enhance appreciation of certain aspects of their environment. Our analysis failed to detect performance differences according to duration of blindness, suggesting that with training, sensory substitution may work with both the newly blind and those who have been blind for long periods of time. Our laboratory is currently conducting functional MRI studies of cross modal plasticity between different age groups to test this hypothesis. It should be noted that we did not include individuals with cortical blindness in this study, but further experiments with this population are in progress.
In conclusion, the Tangent screen and the BaGA, BaLM, and FrACT programs proved to be useful methods for assessing the basic visual capabilities of a tactile-based sensory substitution device. No modifications of the tests were required, but careful calibration of testing parameters is a critical aspect of being able to compare results between trials as well as between individuals. Other tests will need to be developed to test motion discrimination, object and word recognition, mobility, and other functional improvements. Further testing with other sensory substitution devices should be undertaken to determine the generalizability of these tests. As devices become more advanced, additional tests will likely need to be developed. A collaboration among researchers to standardize a mutually agreed upon set of visual and functional outcomes assessments tools is needed to establish a clinical infrastructure for the field of artificial vision.
Acknowledgments
This work was supported by a grant from the State of Pennsylvania (DCED).
Disclosure: A. Nau, Board Member, Association of Optometrists at Academic Medical Centers (S); M. Bach, None; C. Fisher, None
References
- 1.Resnikoff S, Pascolini D, Etya'ale D, et al. Global data on visual impairment in the year 2002. Bull World Health Organ. 2004;82:844–851. [PMC free article] [PubMed] [Google Scholar]
- 2.Ahuja AKDJ, Caspi A, McMahon MJ, et al. Blind subjects implanted with the Argus II retinal prosthesis are able to improve performance in a spatial motor task. Br J Ophthalmol. 2011;95:539–543. doi: 10.1136/bjo.2010.179622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Humayun MSDJ, Ahuja AK, Caspi A, et al. Preliminary 6 month results from the Argus II epiretinal prosthesis feasibility study. Conf Proc IEEE Eng Med Biol Soc. 2009;2009:4566–4568. doi: 10.1109/IEMBS.2009.5332695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Humayun MSDJ, da Cruz L, Dagnelie G, et al. Interim results from the international trial of Second Sight's visual prosthesis. Ophthalmology. 2012;119:779–788. doi: 10.1016/j.ophtha.2011.09.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Danilov YTM. Brainport: an alternative input to the brain. J Integr Neurosci. 2005;4:537–550. doi: 10.1142/s0219635205000914. [DOI] [PubMed] [Google Scholar]
- 6.Kärcher SMFS, Hartmann D, Nagel SK, König P. Sensory augmentation for the blind. Front Hum Neurosci. 2012;6:37. doi: 10.3389/fnhum.2012.00037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Collignon O, Voss P, Lassonde M, Lepore F. Cross-modal plasticity for the spatial processing of sounds in visually deprived subjects. Exp Brain Res. 2009;192:343–358. doi: 10.1007/s00221-008-1553-z. [DOI] [PubMed] [Google Scholar]
- 8.Bach-y-Rita P. Tactile sensory substitution studies. Ann N Y Acad Sci. 2004;1013:83–91. doi: 10.1196/annals.1305.006. [DOI] [PubMed] [Google Scholar]
- 9.Chebat DR, Rainville C, Kupers R, Ptito M. Tactile-‘visual’ acuity of the tongue in early blind individuals. Neuroreport. 2007;18:1901–1904. doi: 10.1097/WNR.0b013e3282f2a63. [DOI] [PubMed] [Google Scholar]
- 10.Poirier C, De Volder AG, Scheiber C. What neuroimaging tells us about sensory substitution. Neurosci Biobehav Rev. 2007;31:1064–1070. doi: 10.1016/j.neubiorev.2007.05.010. [DOI] [PubMed] [Google Scholar]
- 11.Merabet LB, Battelli L, Obretenova S, Maguire S, Meijer P, Pascual-Leone A. Functional recruitment of visual cortex for sound encoded object identification in the blind. Neuroreport. 2009;20:132–138. doi: 10.1097/WNR.0b013e32832104dc. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Amedi A, Stern WM, Camprodon JA, et al. Shape conveyed by visual-to-auditory sensory substitution activates the lateral occipital complex. Nat Neurosci. 2007;10:687–689. doi: 10.1038/nn1912. [DOI] [PubMed] [Google Scholar]
- 13.Ptito M, Moesgaard SM, Gjedde A, Kupers R. Cross-modal plasticity revealed by electrotactile stimulation of the tongue in the congenitally blind. Brain. 2005;128:606–614. doi: 10.1093/brain/awh380. [DOI] [PubMed] [Google Scholar]
- 14.Ptito M, Matteau I. Zhi Wang A, Paulson OB, Siebner HR, Kupers R. Crossmodal recruitment of the ventral visual stream in congenital blindness. Neural Plast. 2012;2012:304045. doi: 10.1155/2012/304045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sampaio E, Maris S, Bach-y-Rita P. Brain plasticity: “visual” acuity of blind persons via the tongue. Brain Res. 2001;908:204–207. doi: 10.1016/s0006-8993(01)02667-1. [DOI] [PubMed] [Google Scholar]
- 16.Chader GJ, Weiland J, Humayun MS. Artificial vision: needs, functioning, and testing of a retinal electronic prosthesis. Prog Brain Res. 2009;175:317–332. doi: 10.1016/S0079-6123(09)17522-2. [DOI] [PubMed] [Google Scholar]
- 17.Schneck MEDG. Prosthetic Vision Assessment: Physiology, Bioengineering, Rehabilitation. New York, New York: Springer;; 2011. [Google Scholar]
- 18.Wilke RBM, Wilhelm B, Durst W, Trauzettel-Klosinski S, Zrenner E. Testing Visual Functions in Patients with Visual Prostheses. New York, New York: Springer;; 2008. [Google Scholar]
- 19.Zrenner E, Bartz-Schmidt KU, Benav H, et al. Subretinal electronic chips allow blind patients to read letters and combine them to words. Proc Biol Sci. 2011;278:1489–1497. doi: 10.1098/rspb.2010.1747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yanai D, Weiland JD, Mahadevappa M, Greenberg RJ, Fine I, Humayun MS. Visual performance using a retinal prosthesis in three subjects with retinitis pigmentosa. Am J Ophthalmol. 2007;143:820–827. doi: 10.1016/j.ajo.2007.01.027. [DOI] [PubMed] [Google Scholar]
- 21.Humayun MS, Weiland JD, Fujii GY, et al. Visual perception in a blind subject with a chronic microelectronic retinal prosthesis. Vision Res. 2003;43:2573–2581. doi: 10.1016/s0042-6989(03)00457-7. [DOI] [PubMed] [Google Scholar]
- 22.Dougherty BE, Martin SR, Kelly CB, Jones LA, Raasch TW, Bullimore MA. Development of a battery of functional tests for low vision. Optom Vis Sci. 2009;86:955–963. doi: 10.1097/OPX.0b013e3181b180a6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lepri BP. Is acuity enough? Other considerations in clinical investigations of visual prostheses. J Neural Eng. 2009;6:035003. doi: 10.1088/1741-2560/6/3/035003. [DOI] [PubMed] [Google Scholar]
- 24.Bach M. The Freiburg Visual Acuity Test- automatic measurement of visual acuity. Optom Vis Sci. 1996;73:49–53. doi: 10.1097/00006324-199601000-00008. [DOI] [PubMed] [Google Scholar]
- 25.Bach MWM, Wilhelm B, Zrenner E, Wilke R. Basic quantitative assessment of visual performance in patients with very low vision. Invest Ophthalmol Vis Sci. 2010;51:1255–1260. doi: 10.1167/iovs.09-3512. [DOI] [PubMed] [Google Scholar]
- 26.Bach M. The Freiburg Visual Acuity Test-Variability unchanged by post-hoc reanalysis. Graefes Arch Clin Exp Ophthalmol. 2007;245:956–971. doi: 10.1007/s00417-006-0474-4. [DOI] [PubMed] [Google Scholar]
- 27.Bailey IL, Jackson AJ, Minto H, Greer RB, Chu MA. The Berkeley Rudimentary Vision Test. Optom Vis Sci. 2012;89:1257–1264. doi: 10.1097/OPX.0b013e318264e85a. [DOI] [PubMed] [Google Scholar]
- 28.Bittner AKJP, Dagnelie G. Grating acuity and contrast tests for clinical trials of severe vision loss. Optom Vis Sci. 2011;88:1153–1163. doi: 10.1097/OPX.0b013e3182271638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Schulze-Bonsel K, Feltgen N, Burau H, Hansen L, Bach M. Visual acuities “hand motion” and “counting fingers” can be quantified with the freiburg visual acuity test. Invest Ophthalmol Vis Sci. 2006;47:1236–1240. doi: 10.1167/iovs.05-0981. [DOI] [PubMed] [Google Scholar]
- 30.Ella Striem-Amit MG, Amir Amedi. ‘Visual' acuity of the congenitally blind using visual-to-auditory sensory substitution. PLOS One. 2012;7:e33136. doi: 10.1371/journal.pone.0033136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Cha K, Horch KW, Normann RA. Mobility performance with a pixelized vision system. Vision Res. 1992;32:1367–1372. doi: 10.1016/0042-6989(92)90229-c. [DOI] [PubMed] [Google Scholar]