Abstract
Purpose
To identify the frequency of bacterial isolates in early-onset neonatal sepsis (EONS) and their antimicrobial resistance pattern.
Methods
A retrospective study of EONS was conducted at the Beni Suef University Hospital from September 2008 to September 2012. A case of EONS was defined as an infant who had clinical signs of infection or who was born to a mother with risk factors for infection, and in whom blood culture obtained within 72 hours of life grew a bacterial pathogen.
Results
Of 673 neonates screened, there were 138 positive blood cultures (20.5%) (confirmed EONS). Of the recovered isolates, 86.2% were gram-negative pathogens. Klebsiella pneumoniae (42.8%), Enterobacter cloacae (22.5%), and Escherichia coli (13.8%) were the commonest isolated organisms. The most common gram-positive microorganism was Staphylococcus aureus accounting for only 12 isolates (8.7%). All Klebsiella isolates and 93% of Enterobacter isolates were resistant to ampicillin. Gram-negative pathogens had the maximum overall sensitivity to imipenem, cefepime, and ciprofloxacin; whereas, gram-positive isolates were most susceptible to vancomycin, imipenem, and piperacillin.
Conclusion
K. pneumoniae was the predominant causative bacteria of EONS followed by E. cloacae and E. coli. There was a high resistance to ampicillin. Imipenem had the maximum overall activity against the causative bacteria. Continuous surveillance is needed to monitor the changing epidemiology of pathogens and antibiotic sensitivity.
Keywords: Drug Resistance, Newborn, Sepsis
Introduction
Newborn infants are uniquely susceptible to infectious diseases, especially bacterial infections (BI). Serious BI in newborns is the main cause of mortality and morbidity, including sepsis, bacteremia, bacterial meningitis, urinary tract infections, pneumonia, and bacterial gastroenteritis. In developing countries, the incidence of neonatal sepsis is about 3.5-4.3 cases per 1,000 live births1).
Two types of neonatal sepsis have been observed: early-onset disease, when features of sepsis appear during the first 72 hours of birth; and late-onset disease, where the disease manifests beyond 72 hours2).
Unlike early-onset neonatal sepsis (EONS), which are acquired mainly from maternal genital tract microorganisms, most late-onset sepsis are caused by hospital-acquired organisms3). The bacterial pathogens responsible for neonatal sepsis vary with geographical areas, and tend to change over time4).
The extensive use of antibiotics has resulted in the emergence of resistant bacterial strains such as gentamicin-resistant Klebsiella species, third-generation cephalosporin-resistant gram-negative organisms, and methicillin-resistant Staphylococcus aureus (MRSA)5).
Data on the causative organisms and their susceptibility pattern to the antibiotics conventionally used in neonatal intensive care units (NICUs) are scarce in the developing countries. To establish a rational treatment strategy, the ongoing changes of bacterial resistance require repeated local revisions of susceptibility patterns to antibiotic agents. Current study aimed to find out the main pathogens and their antibiotics resistance pattern in neonates with EONS in Beni Suef University Hospital.
Materials and methods
The Present study was carried out in the NICU of Beni Suef University Hospital from September 2008 to August 2012. It is a referral hospital which serve about 2.5 million people in Beni Suef Governorate which is located about 120 km South of Cairo. This neonatal unit is categorized as level 3. We retrospectively evaluated the case records of neonates who had EONS.
A case of EONS was defined as an infant who had clinical signs of infection or those who were born to mothers with risk factors for infection, in whom blood culture obtained within 72 hours of life, grew a bacterial pathogen. Maternal risk factors included prolonged rupture of membranes, fever, urinary tract infection, chorioamnionitis, and meconium stained amniotic fluid. Clinical signs of infection included poor feeding, lethargy, respiratory problems (such as tachypnea, apnea, grunting, cyanosis, and retraction), temperature instability (such as hyperthermia and hypothermia), gastrointestinal problems (as vomiting, abdominal distension, diarrhea, and abnormal gastric residual), and central nervous system symptoms (such as convulsion, hypotonia, and irritability).
Prolonged rupture of membranes is defined as rupture of membranes for ≥18 hours before labor6). All neonates admitted to the NICU with suspected EONS (neonates with clinical signs of infection or those with maternal risk factors for infection) were enrolled in the study. Neonates aged more than 72 hours and those receiving antibiotics before withdrawal of blood culture were excluded from the study.
Blood cultures were collected before starting antibiotics from all neonates with risk factors of EONS and whenever relevant clinical signs were present. Blood samples were obtained under strict aseptic conditions .The skin site was cleansed with 70% alcohol and povidone iodine (1%) followed by 70% alcohol again. One- to two-mililiter blood samples were withdrawn and injected in aerobic broth bottles then incubated at 37℃ for 7 days and observed daily for any turbidity due to bacterial growth. Subculture was done on sheep blood agar and MacConkey Agar routinely after 48 hours and 7 days. Subculture was also done in between if visible turbidity appeared.
Bacterial isolates were identified by conventional biochemical and serological methods. Antimicrobial susceptibility test was performed using the standard disc diffusion (Kirbey-Bauer) method.
Coagulase negative staphylococci (CONS) was considered pathogenic if the baby was clinically septic, and had at least one abnormal hematology test (abnormally high or low peripheral neutrophil count or abnormal immature to total white cell ratio, based on the reference ranges of Manroe et al.7) or thrombocytopenia <150×109/L)8).
Antibiotic treatment is initiated immediately among neonates with suspected infection. Empirical antibiotic treatment usually starts with ampicillin and amikacin, but later on the treatment is guided by the results of blood cultures.
Demographic data, clinical manifestations, causative organisms and their antibiotics susceptibility were identified and recorded. Data were analyzed using the SPSS ver. 11.5 (SPSS Inc., Chicago, IL, USA). Ethical clearance was obtained from the ethical committee of the hospital (Institutional Review Board, IRB). Approved IBR number is 1308N.
Results
During the 4-year study period, there were a total of 673 neonates with suspected EONS. Of these 673 neonates screened for EONS, there were 138 positive blood cultures (20.5%) (confirmed EONS).
Among the patients with confirmed EONS, 23 (16.7%) were diagnosed as pneumonia by chest X-ray, 6 patients (4.3%) were diagnosed as meningitis due to a positive CSF culture and 8 (5.8%) had a positive urine culture (samples were collected using urinary catheter).
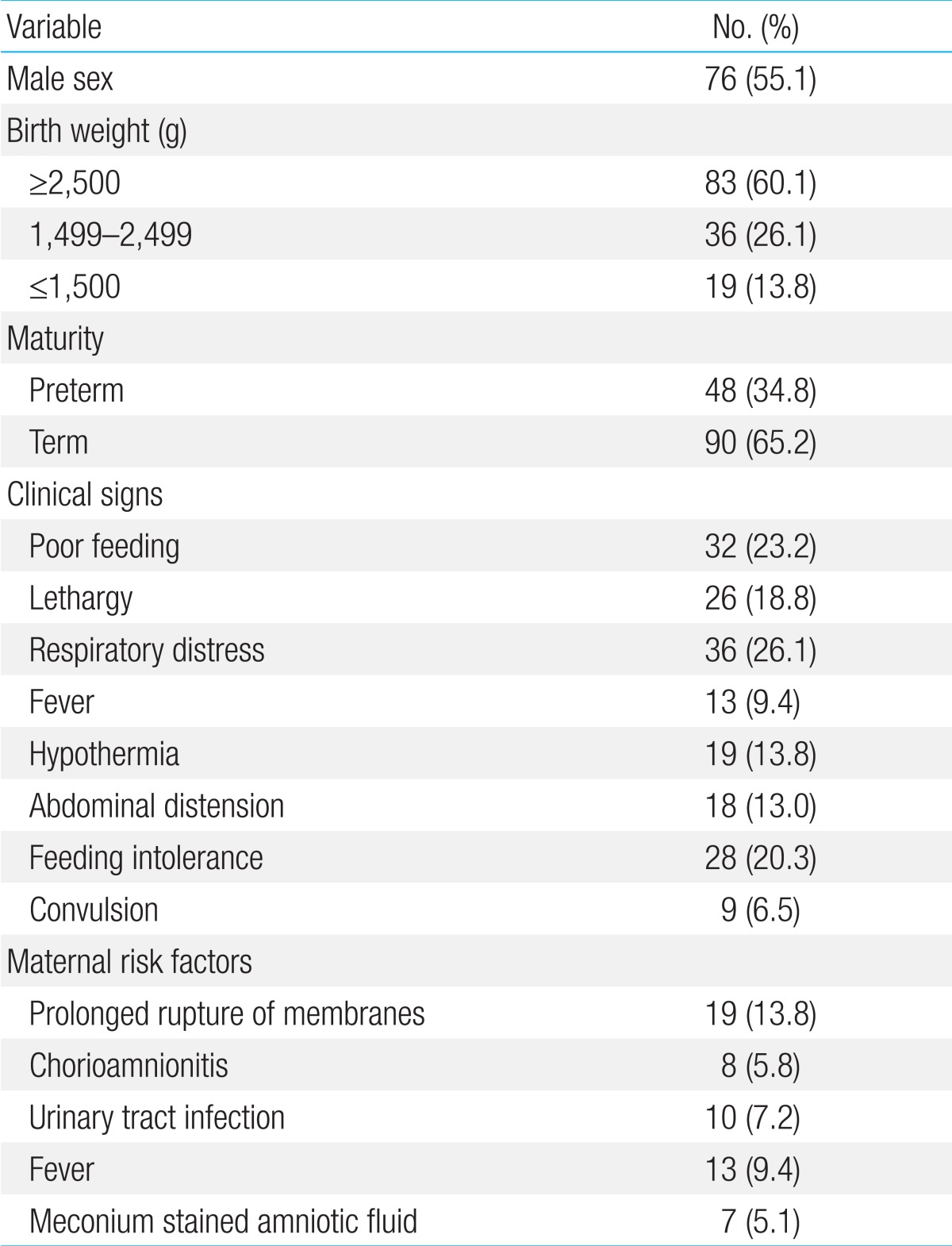
The demographic and clinical characteristics of patients are illustrated in Table 1. Their age ranged between zero and three days, 86 (62.3%) were inborn and 52 (37.7%) were outborn. Seventy-two babies (52.2%) were delivered by CS whereas 66 (47.8%) were delivered vaginally. Overall mortality rate was 18.8% (26 out of 138 neonates died). Clinical signs significantly associated with worst prognosis (death) were lethargy, respiratory distress and convulsions.
Table 1.
Demographic and clinical data of infected neonates (n=138)
The microorganisms identified in blood cultures of 138 infants are shown in Table 2. A total of 119 of 138 recovered isolates (86.2%) were Gram-negative organisms and only 19 isolates (13.8%) were Gram-positive. Klebsiella pneumoniae was the most common pathogen, accounting for 59 of 138 isolates (42.8%) whereas the most common Gram-positive microorganism was S. aureus accounting for 12 isolates (8.7%).
Table 2.
Distribution of pathogens isolated from neonates with confirmed EONS (n=138)
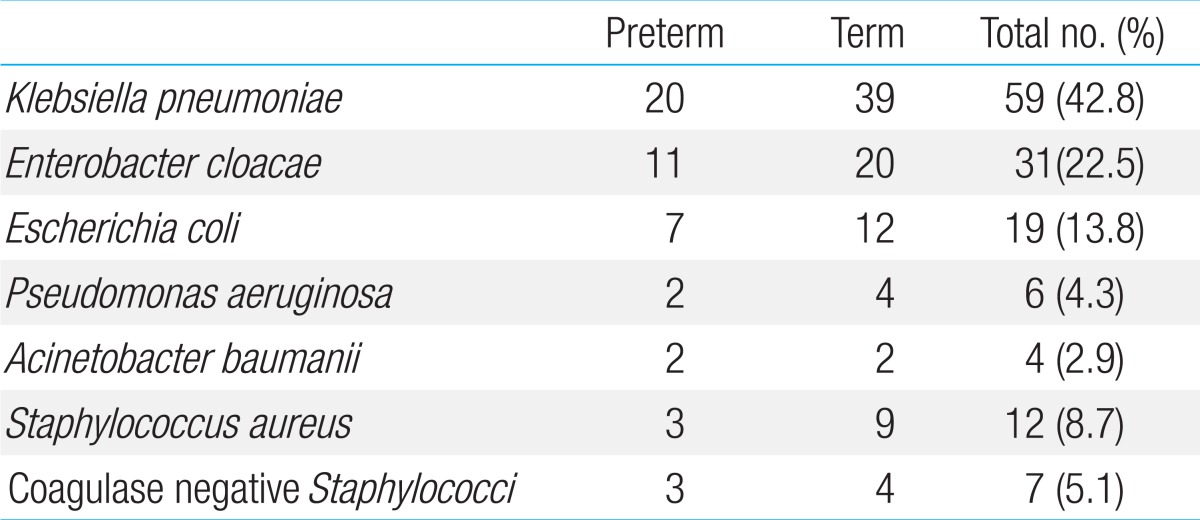
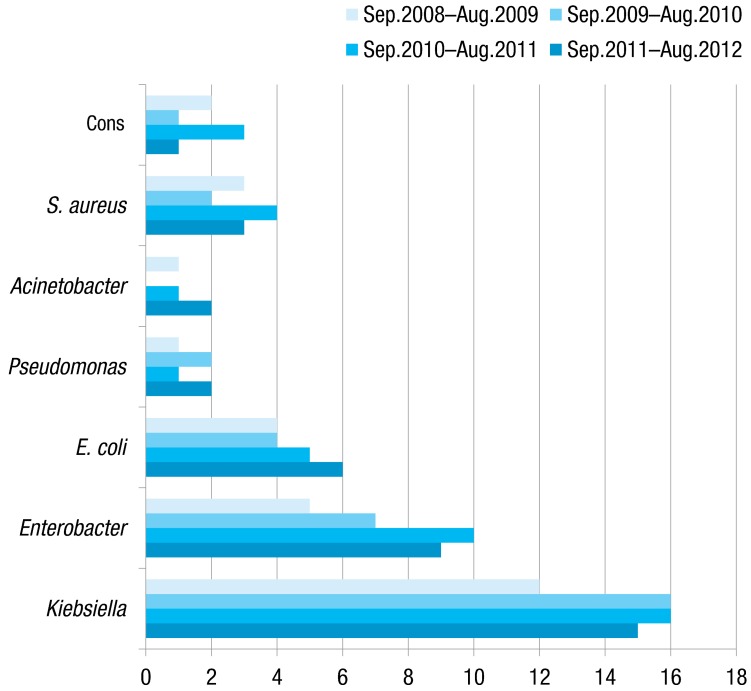
Fig. 1 describes the distribution of causative organisms over time. Tables 3 and 4 show antimicrobial resistance pattern of organisms isolated form blood culture. All Klebsiella isolates were resistant to ampicillin and amoxicillin while the resistance of Klebsiella isolates to Imipenem and cefepime was low (12% and 17%, respectively). Enterobacter revealed high susceptibility to imipenem and cefepime (level of resistance was 16%). All Pseudomonas isolates were resistant to ampicillin, amoxicillin, gentamicin, tobramycin, cefotaxime, ceftazidime, and ceftriaxone. All Gram-positive isolates were susceptible to vancomycin.
Fig. 1.
The distribution of causative organisms over time. CONS, coagulase negative Staphylococci; S. aureus, Staphylococcus aureus; E. coli, Escherichia coli.
Table 3.
Antibiotic resistance pattern of Gram-negative isolates (n=119)

Values are presented as number (%).
Table 4.
Antibiotic resistance pattern of Gram-positive isolates (n=19)

Values are presented as number (%).
CONS, coagulase negative Staphylococci.
Discussion
EONS has significant morbidity and mortality. Improvement in outcome and successful treatment depends on early initiation of appropriate antibiotic therapy. The pattern of causative organisms has been constantly changing and the frequent emergence of resistant bacteria compounds the problem further9).
Egypt is a developing country where the problem of infection is more evident due to lack of health services and hygiene practices. Also, infection control practices are also underdeveloped and are known to contribute to the risk of antibiotic resistance.
Beni Suef is the capital of a governorate in Upper Egypt with high unemployment and poor living condition which make the problem of infection more complicated. NICU of Beni Suef University Hospital is the only level 3 unit in that governorate.
To our knowledge, this study is the first published one from Egypt to report the bacterial etiology of EONS, and to identify their antibiotic resistance pattern.
A male predominance was found in our study which agrees with previous reports5,9). Forty-eight neonates (34.8%) with EONS were premature, which is comparable to other study in Iran by Shahian et al.4), who reported that prematurity was found in 21% of septic neonates. In our study, the most prominent clinical finding of EONS was respiratory distress, which is consistent with other reports4,10).
During the study period, the rate of confirmed EONS (positive blood cultures) in neonates screened for EONS was 20.5%. In a similar study in Iran, the rate of proven sepsis was 28.9%4), but in India it was17.8%9).
The most common organism identified in our study was K. pneumoniae which is similar to other studies from India11) and Pakistan12). However, group B Streptococcys which are common pathogens, causing early neonatal sepsis in the western countries were not seen in this study.
E. cloacae were the 2nd most common pathogen in our study representing 22.5% of all isolates. This is higher than the incidence reported from other developing countries as Iran (7.7%)4) and Kuwait (2.6%)13).
A Canadian study by Sgro et al.14) reported that the association of group B Streptococcys with EONS declined over a period of 6 years and there was a trend to an increase in the rate of infection with other organisms. They also found that E. coli was the predominant organism in preterm infants. In our study E. coli accounted for 13.8% of EONS. We also found that gram-positive organisms (including S. aureus and CONS) were responsible for only small proportion (13.8%) of the cases which is consistent with other studies in developing countries as in Pakistan12) and India9). This is in contrast to the studies from developed countries where gram-positive isolates are the predominant agents15,16).
Predominance of Gram-negative organisms in our study may be due to the indiscriminate and inappropriate use of antibiotics, lack of hygienic practices at the place of delivery, poor cord care, and unhygienic newborn care practices.
Antibiotic resistance is now a global problem. Reports of multiresistant bacteria causing neonatal infection in developing countries are increasing, particularly in intensive care17).
In the present study, Klebsiella isolates were best susceptible to imipenem, cefepime, ceftriaxone, and ciprofloxacin whereas all Klebsiella isolates were resistant to ampicillin and amoxicillin. More than 65% of Klebsiella isolates were resistant to aminoglycosides. Thaver et al.18) reported that almost all Klebsiella species were resistant to ampicillin. Shahian et al.4) found that about 33% of Klebsiella isolates were sensitive to ampicillin. High susceptibility to imipenem (98%) and ciprofloxacin (96%) was found by Macharashvili et al.19). Because quinolones have been implicated as interfering with cartilage growth in animals, these drugs should be used with caution and only in the absence of other options5). Also, since β-lactam antibiotics are prone to rapid evolvement of bacterial resistance, imipenem usually is reserved for treatment of the most severe and resistant infections.
A single hospital study performed in Chicago found an increase in the proportion of early-onset E. coli infections that were caused by ampicillin-resistant strains20). According to our data all cases of neonatal early-onset E. coli sepsis were resistant to ampicillin but they were best susceptible to imipenem. High susceptibility to imipenem (91%) was also found by Macharashvili et al.19).
Intestinal colonization of the newborn with multiresistant pathogen could pose a risk of Enterobacter sepsis even in the absence of obvious invasive procedures. This may have been the case in newborns that developed Enterobacter sepsis within 72 hours of birth, suggesting that the organism may have been acquired from the environment either during or soon after birth21). In our study, 35% and 48% of infections caused by Enterobacter were resistant to amikacin and gentamicin, respectively, in comparison to 19% and 33% in another study done in Pakistan21).
According to the results of the present study, it seems that piperacillin is the best effective agent against Pseudomonas . In contrast, Bhat et al.9) found that only 24% of Pseudomonas isolates were susceptible to piperacillin.
In the recent years, Acinetobacter species are gaining importance as potential pathogens in neonatal septicemia because of their multidrug resistance. In the present study, Acinetobacter baumanii contributed to 2.9% of total septicemia cases and imipenem resistance is observed in only 25% of them. A recent study from India has reported almost 41% imipenem resistance in Acinetobacter isolates22).
Our study revealed high resistance of S. aureus and CONS to cephalosporins, whereas not a single case of resistant was found against vancomycin. Also, it seems that imipenem had good activity against gram-positive isolates. In developing countries, it may be necessary to cover for Staphylococci from birth, whereas Staphylococcal infections are rare in developed countries before the third day of age23). Empiric vancomycin is not recommended unless MRSA is common, and it is important to restrict vancomycin use in all countries because of the risk of selecting for vancomycin resistant organisms24).
According to the results of our study, gram-negative pathogens had best overall sensitivity to imipenem, cefepime and ciprofloxacin whereas gram-positive isolates were best susceptible to vancomycin,imipenem and piperacillin. It seems that imipenem is the appropriate antibiotic for empiric treatment of EONS, but its extensive use will select for beta lactamase producing organisms as well as for organisms resistant to imipenem25).
In conclusion, K. pneumoniae and E. cloacae were the predominant isolates in early-onset sepsis and showed high resistance to ampicillin. Imipenem had the best overall activity against the causative bacteria yet it is difficult to use it as an empiric treatment. The emergence of resistant strains to commonly used antibiotics is a serious problem and necessitates the implementation of a rational empirical treatment strategy based on local susceptibility data. In the face of the ongoing changes of microbial prevalence and resistance, local treatment protocol should be reviewed and assessed periodically.
Acknowledgments
The author would like to thank all his colleagues in the NICU and microbiology laboratory, Beni Suef University Hospital, for their valuable cooperation.
Footnotes
No potential conflict of interest relevant to this article was reported.
References
- 1.Yang YN, Tseng HI, Yang SN, Lu CC, Chen HL, Chen CJ. A strategy for reduction of antibiotic use in new patients admitted to a neonatal intensive care unit. Pediatr Neonatol. 2012;53:245–251. doi: 10.1016/j.pedneo.2012.06.009. [DOI] [PubMed] [Google Scholar]
- 2.Basu S, Dewangan S, Shukla RC, Anupurva S, Kumar A. Cerebral blood flow velocity in early-onset neonatal sepsis and its clinical significance. Eur J Pediatr. 2012;171:901–909. doi: 10.1007/s00431-011-1643-y. [DOI] [PubMed] [Google Scholar]
- 3.Hammoud MS, Al-Taiar A, Thalib L, Al-Sweih N, Pathan S, Isaacs D. Incidence, aetiology and resistance of late-onset neonatal sepsis: a five-year prospective study. J Paediatr Child Health. 2012;48:604–609. doi: 10.1111/j.1440-1754.2012.02432.x. [DOI] [PubMed] [Google Scholar]
- 4.Shahian M, Pishva N, Kalani M. Bacterial etiology and antibiotic sensitivity patterns of early-late onset neonatal sepsis among newborns in Shiraz, Iran 2004-2007. Iran J Med Sci. 2010;35:293–298. [Google Scholar]
- 5.Bromiker R, Arad I, Peleg O, Preminger A, Engelhard D. Neonatal bacteremia: patterns of antibiotic resistance. Infect Control Hosp Epidemiol. 2001;22:767–770. doi: 10.1086/501860. [DOI] [PubMed] [Google Scholar]
- 6.Blumenfeld YJ, Lee HC, Gould JB, Langen ES, Jafari A, El-Sayed YY. The effect of preterm premature rupture of membranes on neonatal mortality rates. Obstet Gynecol. 2010;116:1381–1386. doi: 10.1097/AOG.0b013e3181fe3d28. [DOI] [PubMed] [Google Scholar]
- 7.Manroe BL, Weinberg AG, Rosenfeld CR, Browne R. The neonatal blood count in health and disease. I. Reference values for neutrophilic cells. J Pediatr. 1979;95:89–98. doi: 10.1016/s0022-3476(79)80096-7. [DOI] [PubMed] [Google Scholar]
- 8.Isaacs D Australasian Study Group For Neonatal Infections. A ten year, multicentre study of coagulase negative staphylococcal infections in Australasian neonatal units. Arch Dis Child Fetal Neonatal Ed. 2003;88:F89–F93. doi: 10.1136/fn.88.2.F89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bhat Y R, Lewis LE, K E V. Bacterial isolates of early-onset neonatal sepsis and their antibiotic susceptibility pattern between 1998 and 2004: an audit from a center in India. Ital J Pediatr. 2011;37:32. doi: 10.1186/1824-7288-37-32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ojukwu JU, Abonyi LE, Ugwu J, Orji IK. Neonatal septicemia in high risk babies in South-Eastern Nigeria. J Perinat Med. 2006;34:166–172. doi: 10.1515/JPM.2006.030. [DOI] [PubMed] [Google Scholar]
- 11.Ananthakrishnan S, Gunasekaran D. Etiology and risk factors for early onset neonatal sepsis. Indian J Med Microbiol. 2009;27:279. doi: 10.4103/0255-0857.53221. [DOI] [PubMed] [Google Scholar]
- 12.Mahmood A, Karamat KA, Butt T. Neonatal sepsis: high antibiotic resistance of the bacterial pathogens in a neonatal intensive care unit in Karachi. J Pak Med Assoc. 2002;52:348–350. [PubMed] [Google Scholar]
- 13.Al-Taiar A, Hammoud MS, Thalib L, Isaacs D. Pattern and etiology of culture-proven early-onset neonatal sepsis: a five-year prospective study. Int J Infect Dis. 2011;15:e631–e634. doi: 10.1016/j.ijid.2011.05.004. [DOI] [PubMed] [Google Scholar]
- 14.Sgro M, Shah PS, Campbell D, Tenuta A, Shivananda S, Lee SK, et al. Early-onset neonatal sepsis: rate and organism pattern between 2003 and 2008. J Perinatol. 2011;31:794–798. doi: 10.1038/jp.2011.40. [DOI] [PubMed] [Google Scholar]
- 15.Kuhn P, Dheu C, Bolender C, Chognot D, Keller L, Demil H, et al. Incidence and distribution of pathogens in early-onset neonatal sepsis in the era of antenatal antibiotics. Paediatr Perinat Epidemiol. 2010;24:479–487. doi: 10.1111/j.1365-3016.2010.01132.x. [DOI] [PubMed] [Google Scholar]
- 16.Cordero L, Sananes M, Ayers LW. Bloodstream infections in a neonatal intensive-care unit: 12 years' experience with an antibiotic control program. Infect Control Hosp Epidemiol. 1999;20:242–246. doi: 10.1086/501619. [DOI] [PubMed] [Google Scholar]
- 17.Vergnano S, Sharland M, Kazembe P, Mwansambo C, Heath PT. Neonatal sepsis: an international perspective. Arch Dis Child Fetal Neonatal Ed. 2005;90:F220–F224. doi: 10.1136/adc.2002.022863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Thaver D, Ali SA, Zaidi AK. Antimicrobial resistance among neonatal pathogens in developing countries. Pediatr Infect Dis J. 2009;28(1 Suppl):S19–S21. doi: 10.1097/INF.0b013e3181958780. [DOI] [PubMed] [Google Scholar]
- 19.Macharashvili N, Kourbatova E, Butsashvili M, Tsertsvadze T, McNutt LA, Leonard MK. Etiology of neonatal blood stream infections in Tbilisi, Republic of Georgia. Int J Infect Dis. 2009;13:499–505. doi: 10.1016/j.ijid.2008.08.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Joseph TA, Pyati SP, Jacobs N. Neonatal early-onset Escherichia coli disease: the effect of intrapartum ampicillin. Arch Pediatr Adolesc Med. 1998;152:35–40. doi: 10.1001/archpedi.152.1.35. [DOI] [PubMed] [Google Scholar]
- 21.Bhutta ZA. Enterobacter sepsis in the newborn: a growing problem in Karachi. J Hosp Infect. 1996;34:211–216. doi: 10.1016/s0195-6701(96)90068-7. [DOI] [PubMed] [Google Scholar]
- 22.De AS, Rathi MR, Mathur MM. Mortality audit of neonatal sepsis secondary to acinetobacter. J Glob Infect Dis. 2013;5:3–7. doi: 10.4103/0974-777X.107165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Isaacs D. Rationing antibiotic use in neonatal units. Arch Dis Child Fetal Neonatal Ed. 2000;82:F1–F2. doi: 10.1136/fn.82.1.F1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Isaacs D. Unnatural selection: reducing antibiotic resistance in neonatal units. Arch Dis Child Fetal Neonatal Ed. 2006;91:F72–F74. doi: 10.1136/adc.2005.074963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Isaacs D. Neonatal sepsis: the antibiotic crisis. Indian Pediatr. 2005;42:9–13. [PubMed] [Google Scholar]