Abstract
Inflammation is a major factor in the progression of damage after stroke and in the clinic, current therapies treat the clot, not the resulting damage. We have developed a novel method of protein delivery that exploits the migration ability of leukocytes after ischemic stroke (transient middle cerebral artery occlusion; tMCAO). In our studies, ex vivo-derived dendritic cells (exDCs) migrate to the inflamed rat brain soon after tMCAO onset and the number of cells that remain in the brain after injection is significantly correlated with the amount of local inflammation at the injury site. In addition, exDCs transduced to overexpress soluble tumor necrosis factor (TNF) receptor1 (sTNFR1) produce functional cargo that is secreted and that blocks TNF-α bioavailability in vitro. When delivered at 6 hours post-tMCAO reperfusion, sTNFR1-exDC-treated rats show significantly smaller infarct size and decreased inflammation compared with animals treated with exDCs transduced with GFP lentivirus. These studies indicate that cell-mediated delivery of proteins may be a promising new approach to reduce brain damage after acute neurologic insult.
Keywords: acute stroke, cell tracking, chemokines, immunology, inflammation
Introduction
Stroke affects over 700,000 Americans annually and the current leading intervention, tissue plasminogen activator, has significant temporal limitations.1 When administered beyond 4.5 hours, tissue plasminogen activator is ineffective or even detrimental.2 Novel treatments using genes or proteins that target stroke damage progression may prove to be more effective. A significant hurdle in the use of gene or protein therapy is directing the spatial delivery of the therapeutic cargo as systemic delivery distributes the therapy to non-target organs, causing the material to be wasted and increasing the chance of side effects. Alternatively, one can directly inject protein or viral vector into the brain parenchyma, an obviously extremely invasive protocol. As an alternative, we have designed a novel delivery system using leukocytes that allow for protein delivery in a minimally invasive manner.3
Leukocyte-mediated delivery of proteins shows temporal and spatial selectivity, presenting a promising, new method of targeted protein delivery to reduce stroke damage.3 Because leukocytes respond to proinflammatory factors and the migration of endogenous immune cells to the injured brain is concomitant with the increase in proinflammatory factors after stroke, leukocytes are an ideal vehicle for delivery of therapeutic proteins. After ischemia, messenger RNA expression of potent inflammatory cytokines and chemokines TNF-α, IL-1β, and MCP-1 increases in the brain within 1 to 3 hours and peaks between 24 and 36 hours.4, 5, 6, 7, 8, 9, 10 Increases in protein levels of these factors occur by 6 hours after stroke (ref. 4 and our data) in neurons, glia, peripheral immune organs, and circulating immune cells.11 Shortly after stroke onset, increased cytokine and chemokine production is also associated with increased and rapid leukocyte migration to the injured hemisphere. In a mouse model of temporary middle cerebral artery occlusion, neutrophils and macrophages arrived at the ischemic hemisphere within 12 hours after reperfusion, while dendritic cells (DCs) arrived within 24 hours.12 However, in a rat permanent MCAO model, DCs were observed in the brain within one hour.13
As DCs migrate rapidly after stroke, they can be suitable as protein carriers. Ex vivo-derived DCs (exDCs) overexpressing the anti-apoptotic gene BH4 fused to Tat (transactivator of transcription) produced and secreted their protein cargo, migrated to the injury site at 3 hours poststroke, and reduced damage after stroke.3 In this study, we expanded these findings by clarifying the temporal limits of administration of exDCs, identifying the possible mechanisms of exDC migration, and demonstrating that exDCs overexpressing soluble TNF receptor1 (sTNFR1) decrease stroke damage.
Materials and Methods
Subjects
Procedures were approved by the Stanford University Administrative Panel on Laboratory Care and the Association for Assessment of Laboratory Animal Care and are in compliance with the NIH (National Institutes of Health, Bethesda, MD, USA) Guide for the Care and Use of Laboratory Animals. Adult male Sprague-Dawley rats (Charles River, Hollister, CA, USA) were housed with a 12-hour light–dark schedule. After 5 to 9 days of acclimation, animals (320 to 360 g) underwent temporary middle cerebral artery occlusion (tMCAO) at a physiologic temperature and respiration rate.14 Briefly, using 2% isoflurane anesthetic, the left common carotid artery, external carotid artery (ECA), and internal carotid artery were exposed through a midline incision. The ECA was ligated, coagulated, and cut just proximal to the lingual and maxillary artery branches. Next, a silicon-tipped 4-0 monofilament (Doccol, Sharon, MA, USA) was inserted into the ECA and advanced ∼18 mm into the internal carotid artery until resistance was felt to occlude the MCA. After 1 hour, the monofilament was withdrawn, the ECA branch was sutured, blood flow restored, and the wound closed; subjects were given the analgesic buprenorphine (0.05 mg/kg subcutaneously; Nationwide Medical/Surgical, Agoura Hills, CA, USA).
Intra-Arterial Delivery of Cultured Dendritic Cells
For in vivo migration studies (Figure 1), a catheter was inserted into the ECA 3, 6, 12, or 24 hours postreperfusion onset and 2 × 106 transgenic GFP+ cells were infused (0.3 mL over 1 minute). For inflammation correlation studies (Figures 2 and 3), the catheter was inserted 6 hours postreperfusion and 2.5 × 106 GFP+ cells were injected over 1 minute. For protection and inflammation studies (Figures 4 and 5), vehicle (50% RPMI 1640/50% complete media without cytokines) or 2 × 106 lentivirus (LV)-transduced cells were injected 6 hours poststroke over 1 minute. After infusion, the catheter was removed, the ECA sutured, and the wound closed.
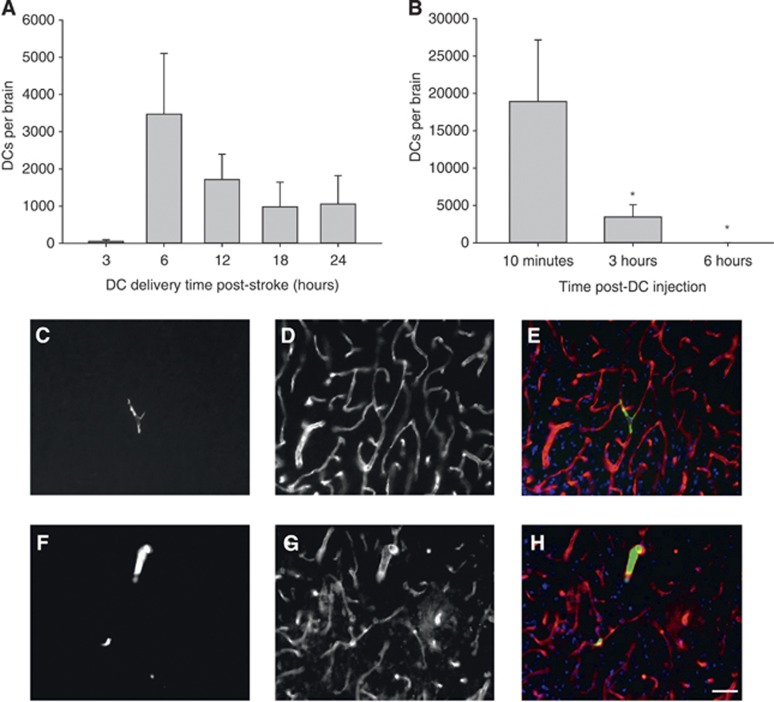
Figure 1.
(A) Animals subjected to transient middle cerebral artery occlusion (tMCAO) were injected with green fluorescent protein (GFP+) ex vivo-derived dendritic cells(exDCs) at 3 (N=4), 6 (N=7), 12 (N=5), 18 (N=6) and 24 hours (N=5) hours poststroke, then killed 3 hours postinjection. The total number of exDCs in serial sections spanning the stroke region is indicated and error bars indicate s.e.m. (B) Animals subjected to tMCAO were injected with exDCs at 6 hours poststroke and killed at 10 minutes (N=7), 3 hours (N=7), or 6 hours (N=6) postinjection. One-way analysis of variance and S–N–K post hoc revealed that the number of exDCs at 10 minutes is significantly higher than 3 hours (P=0.043) and 6 hours postinjection (P=0.05). Error bars indicate s.e.m. (C–H) Animals injected with GFP+ exDCs (green) at 6 hours poststroke were killed at 10 minutes (N=2) or 3 hours (N=2) poststroke. Forty-micrometer-thick serial slices were stained for the endothelial marker RECA-1 (red) and counterstained with the nuclear marker 4',6-diamidino-2-phenylindole I (DAPI) (blue). (C) Representative section of 10 minutes poststroke GFP+ exDC (D) RECA-1 stain (E) overlay with DAPI (N=2 animals, 69 total cells counted). (F) Representative section of 3 hours poststroke GFP+ exDC (G) RECA-1 stain (H) overlay with DAPI (N=2 animals, 50 cells counted). Scale bar 20 μm.
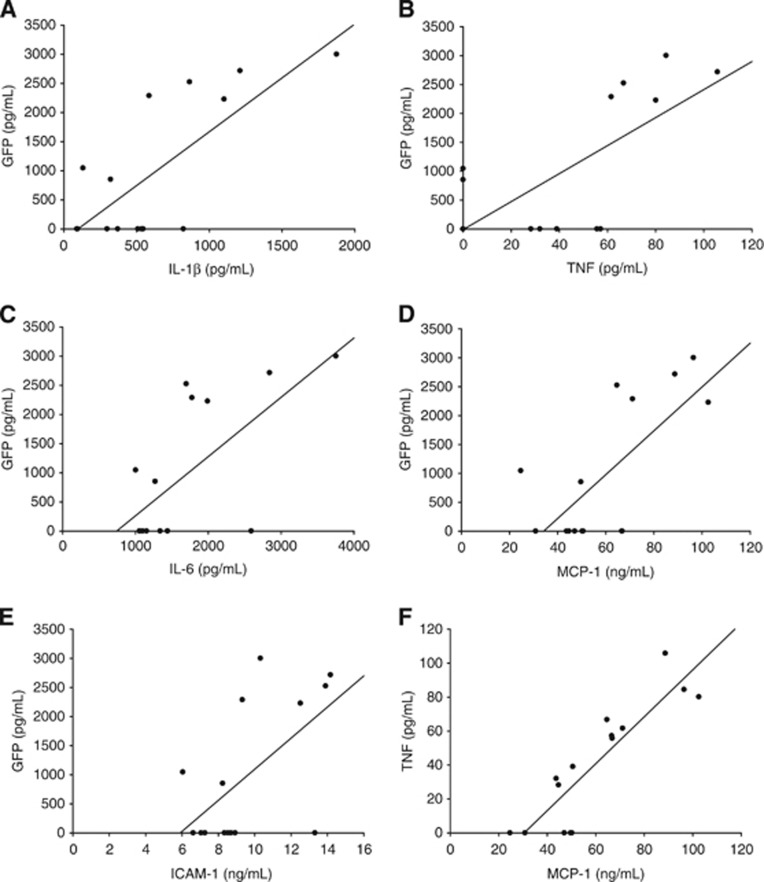
Figure 2.
(A–F) Animals subjected to transient middle cerebral artery occlusion (tMCAO) were injected with GFP+ ex vivo-derived dendritic cells (exDCs) at 6 hours poststroke, and then killed 10 minutes postinjection. Brain homogenate was normalized to overall protein levels and run on a green fluorescent protein GFP ELISA to determine relative GFP+ exDC migration. GFP (DCs) was highly correlated with brain levels of (A) IL-1β (R2=0.558, P=0.0014), (B) TNF-α (R2=0.511, P=0.0027), (C) IL-6 (R2=0.446, P=.0065), (D) MCP-1 (R2=0.510, P=0.0028), (E) ICAM-1 (R2=0.355, P=0.0191). (F) All inflammatory factors were significantly correlated with each other, TNF and MCP-1 showed the strongest correlation (R2=0.776, P=0.000014).
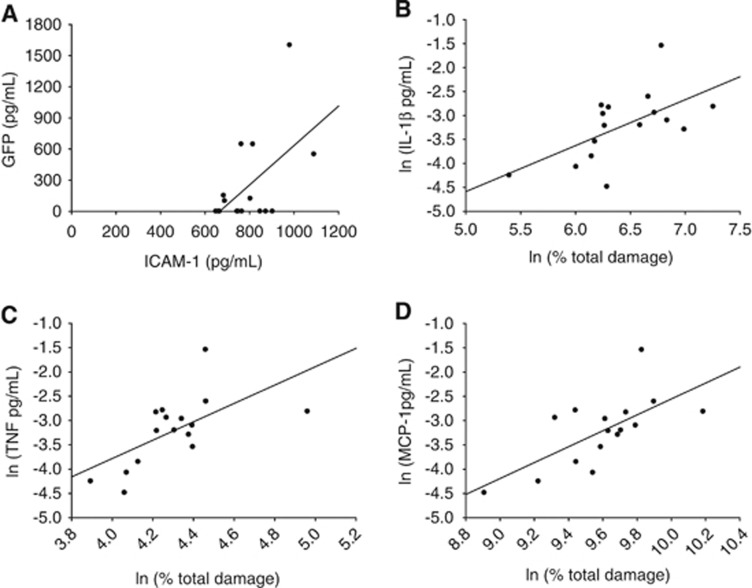
Figure 3.
Animals subjected to transient middle cerebral artery occlusion were injected with green fluoroscent protein (GFP+) ex vivo-derived dendritic cells at 6 hours post-stroke and killed 10 minutes postinjection. Two millimeter slices were taken and slices one, three, and five were analyzed for protein, both sides of slices two and four were analyzed for stroke damage using 2,3,5-triphenyltetrazolium chloride. Brain homogenate was run on a GFP ELISA and 11-plex polystyrene bead inflammatory protein assay. (A) GFP ELISA and ICAM-1 multiplex values were significantly correlated (R2=0.291, P=0.0314). (B) Total damage was significantly correlated with IL-1β (R2=0. 353, P=0.0152), (C) TNF (R2=0.386, P=0.0102), and (D) MCP-1 (R2=0.454, P=0.00421) after log-linear analysis.
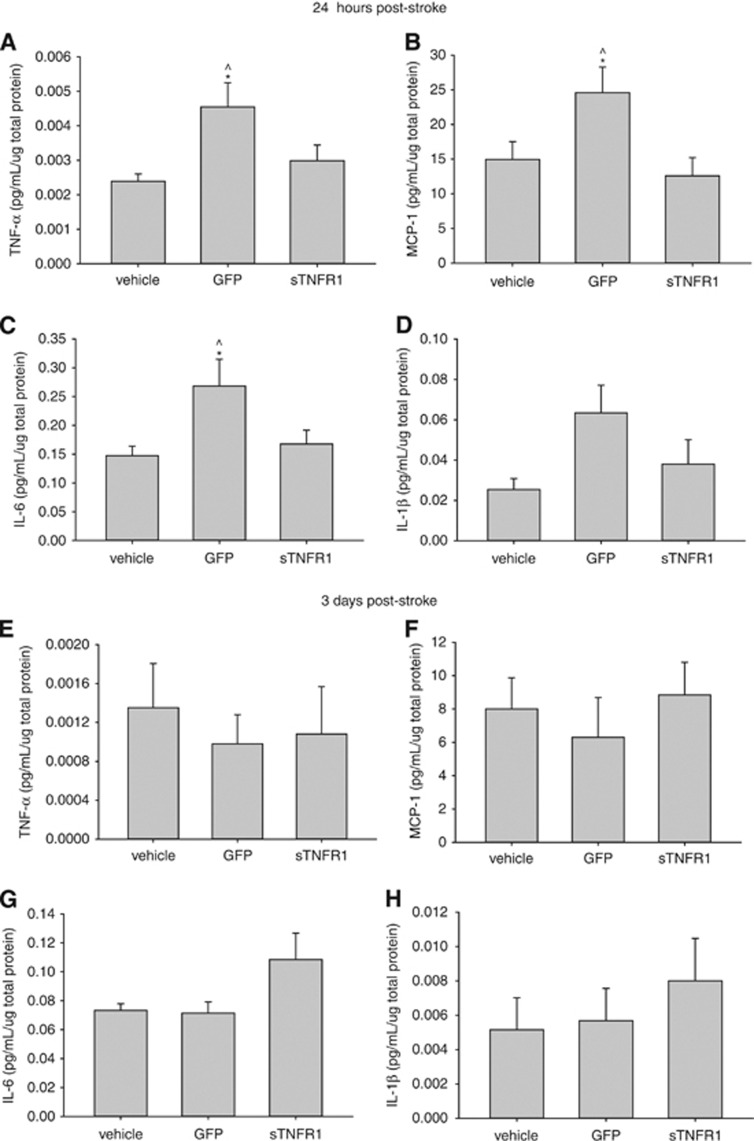
Figure 4.
(A–D) Animals killed 24 hours post-stroke. One-way analysis of variance and S-N-K post hoc revealed a significant increase in inflammation in green fluoroscent protein (GFP)-ex vivo-derived dendritic cells (exDC)-treated subjects compared with vehicle and sTNFR1-exDC-treated animals. (A) Tumor necrosis factor (TNF) (P=0.016), (B) MCP-1 (P=0.022), (C) IL-6 (P=0.028), (D) IL-1β (P=0.064). (E–H) Subjects killed 3 days poststroke showed no significant differences in these inflammatory factors. *P<0.05 compared with vehicle ^P<0.05 compared with sTNFR1. At 24 hours vehicle (N=9), GFP (N=9), sTNFR1 (N=9) and 3 days vehicle (N=9), GFP (N=10), sTNFR1 (N=10). Error bars indicate s.e.m.
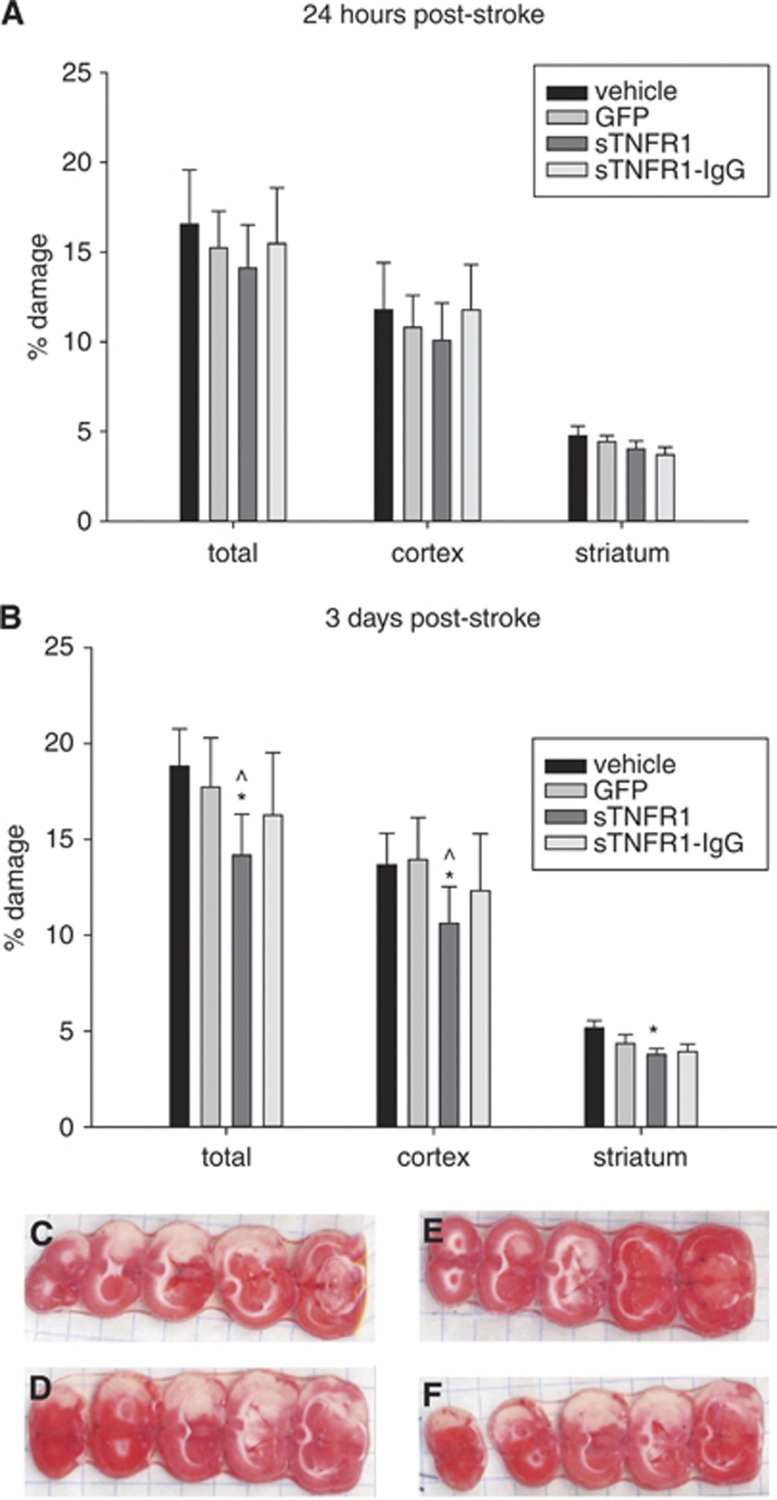
Figure 5.
(A) No significant differences in damage at 24 hours poststroke. (B) At 3 days poststroke, there was a significant reduction in percent total (P=0.008), percent cortex (P=0.039), and percent striatum (P=0.006) versus vehicle damage, and versus green fluorescent protein (GFP)-ex vivo-derived dendritic cell treated animals in percent total (P=0.038) and percent cortex (P=0.044) damage *P<0.05 in 2-way analysis of variance (ANOVA) (group, slice) for each brain area compared with vehicle; ^P<0.05 in 2-way ANOVA (group, slice) for each brain area compared with GFP control. 24 hours vehicle (N=14), GFP (N=16), sTNFR1 (N=14), sTNFR1-IgG (N=11) and 3 days vehicle (N=16), GFP (N=13), sTNFR1 (N=13), sTNFR1-IgG (N=9). Error bars indicate s.e.m.. Representative photos of 2,3,5-triphenyltetrazolium chloride staining for day 3 infarct size assessment for (C) vehicle, (D) GFP-exDCs, (E) sTNFR1-exDCs, (F) sTNFR1-IgG-exDCs.
Ex vivo-Derived Dendritic Cell Cultures
A DC-enriched population was generated from bone marrow as described,15 with slight modifications. GFP transgenic rats were a gift from G. Steinberg and cells from GFP +/− rats were used for GFP+ groups. Of the exDCs isolated from GFP +/− transgenic rats, 100% were GFP+. Bone marrow from Sprague-Dawley or GFP +/− transgenic rat pups (postnatal day 15 to 17) was isolated and passed through a 70 μm cell strainer. After RBC lysis and washing with RPMI (Gibco, Grand Island, NY, USA), cells were resuspended in freezing media (Gibco), and stored in liquid nitrogen. Upon rapid thawing, cells were washed with RPMI (Gibco) and seeded in 6-well plates at a concentration of 2 × 106 cells/mL in complete media: RPMI 1640 (Gibco), 10% fetal bovine serum (fetal bovine serum, Gibco), 2 mmol/L L-glutamine (Gibco), 1% nonessential amino acids (Gibco), 1 mmol/L sodium pyruvate (Gibco), 1% penicillin–streptomycin and the cytokines, interleukin-4, granulocyte–macrophage colony stimulating factor, Flt-3 ligand (all at 5 ng/mL/cytokine, RD Systems, Minneapolis, MN, USA). On the third day in vitro (DIV 3), 1 mL/well of complete media was added. On DIV 4, media containing non-adherent cells was removed and replaced with 0.75 mL complete media containing 10 μg/mL protamine sulfate (Sigma, St Louis, MO, USA) and one of the LV vectors (multiplicity of infection (MOI)=10 to 30). Eighteen hours post LV transduction, virus-containing media was replaced with a combination of 75% complete media and 25% spun-down conditioned media from DIV 4. On DIV 6 to 7, cultures were harvested in their media, spun at 1,300 r.p.m. at room temperature, resuspended in RPMI.
Perfusion and Tissue Processing
To fix brain tissue for DC migration assessment and RECA-1 staining, animals were transcardially perfused with ice cold 0.9% saline followed by ice cold 4% paraformaldehyde (PFA) in phosphate buffered saline (PBS) (pH 7.2 to 7.4). Isolated brains were cryoprotected in 20% sucrose/4% paraformaldehyde for 24 hours at 4°C, and then snap frozen and stored at −80°C. Thirty micrometer coronal sections were cut on a cryostat, dry-mounted on Superfrost Plus slides (Fisher Scientific, Waltham, MA, USA; VWR), and re-stored at −80°C until staining or coverslipping. For RECA staining, 40 μm sections were cut and stored in freezing media (30% ethylene glycol, 25% glycerol in saline, pH 7.2 to 7.4) at −20°C until staining. Sets of six coronal sections were collected corresponding to the infarct region (anterior/posterior to bregma: +2.5 to −2.0 mm).
Tissue for Protein and Damage
For inflammation and migration correlations (Figure 2), animals were sacrificed 10 minutes postinjection with an overdose of isoflurane and an injection of Euthasol, and the brain was cut into 2 mm slices in a brain matrix. Five slices corresponding to the infarct region were collected and the first slice began at +2.5 mm to bregma. The ipsilateral and contralateral hemispheres were separated and snap frozen. For inflammation, damage, and migration correlations (Figure 3), animals were sacrificed 10 minutes postinjection (as above); slices one, three, and five were separated into ipsilateral and contralateral hemispheres and snap frozen. Slices two and five were placed into a 1% (w/v) solution of 2,3,5-triphenyltetrazolium chloride (Sigma), in 0.9% saline for 10 minutes at 37°C. After incubation in triphenyltetrazolium chloride, slices were immediately placed between glass slides (VWR) and both sides of the slice were imaged on a flat-bed scanner. A blinded observer manually assessed damage using ImageJ (NIH) by outlining the infarct area in the cortex, sub-cortical region (striatum), and total slice area to determine percent damage per slice. For inflammation assessment (Figure 4), animals were killed (as above) 24 or 76 hours postreperfusion; slices were separated into ipsilateral and contralateral hemispheres and snap frozen. Some animals died before the survival time point (sTNFR1 N=1 out of 10 for 24 hours, vehicle N=1 out of 10 for 24 hours, GFP N=1 out of 11 for 3 days, sTNFR1 N=2 out of 12 for 3 days, vehicle N=1 out of 10 for 3 days). For protection assessment (Figure 5), animals were killed 24 or 76 hours postreperfusion; slices were incubated in triphenyltetrazolium chloride (as above). Animals that showed no damage (presumably due to incorrect filament placement) were excluded (for 24-hour time point N=3 GFP out of 19, sTNFR1 N=2 out of 16). Some animals died before the 3-day survival time point (sTNFR1 N=1 out of 14, sTNFR1-IgG N=2 out of 11, vehicle N=2 out of 18).
Immunofluorescent Staining
Free-floating sections were stained with the anti-endothelial antibody RECA-1 to label blood vessels at 10 minutes or 3 hours postcell infusion. Sections were washed 3 × with 0.3% Triton X-100/PBS, blocked in 5% goat serum/0.3% Triton X-100/PBS for 1 hour at room temperature, and incubated in RECA-1 (1:200, AbCam, Cambridge, MA, USA) overnight at 4°C. After 3 × washes, sections were incubated with the secondary antibody, goat anti-mouse IgG:Alexa 568 (1/200, Invitrogen, Grand Island, NY, USA) for 1 hour at room temperature. Sections were washed 2 × with wash buffer, nuclei were counterstained with 4',6-diamidino-2-phenylindole in PBS (1/1,000, Sigma), washed 2 × , and coverslipped in PVA mounting medium (Sigma). For GFP+ DC quantification, slides were rinsed in PBS and coverslipped. Sections were imaged with an Olympus IX70 microscope (Center Valley, PA, USA) and Hamamatsu camera (Bridgewater, NJ, USA; model C4742-95) powered by a Xenon fluorescent lamp. Using Metamorph Imaging Software (Molecular Devices, Sunnyvale, CA, USA), GFP fluorescence and its relative colocalization with RECA-1 staining was imaged.
Lentivirus Vector Construction and Purification
Transgene-containing LV particles were generated using a self-inactivating, three-plasmid system (a gift of G. Fathman). The first plasmid contained the transgene, internal ribosomal entry site, and enhanced green fluorescent protein (GFP). The other two plasmids included the packaging proteins plasmid (p-delta) and the envelope protein plasmid (encoding vesicular stomatitis virus glycoprotein). Transfer vectors were generated with one of two transgenes inserted in the multiple cloning sites (MCS, upstream of internal ribosomal entry site) of pHR-IG, while the GFP transfer vector contained only the eGFP sequence without multiple cloning sites or internal ribosomal entry site (pHR-G). The sTNFR1 transgene sequence (Genebank: NM_013091.1, 205 to 824 bp) was amplified from a rat spleen cDNA library (OriGene) with PCR: 94°C for 2 minutes; 94°C for 15 seconds, 51°C for 1 minute, and 68°C for 1 minute for 35 cycles; 68°C for 5 minutes; stored at 4°C. Forward primer 5′-(TGGGGGAGTGAGAGAGGCCA)-3′ preceded by a SalI cut site and the reverse primer 5′-(GGCACAGCTTCATACATTCCTGAT-3′) was followed immediately by a stop codon and SacII cut site. STNFR1-pHRIG ligation was confirmed by DNA sequencing (Sequetech). The sTNFR1-hIgG sequence contained SalI/BamHI cut sites and the inverse complement was produced by overlapping oligo synthesis by Genewiz (South Plainfield, NJ, USA) to produce the rat sTNFR1 sequence immediately followed by the sequence for human immunoglobulin G (IgG; Genebank: BC018747; 777 to 1,472 bp). Lentiviral particles containing pHR-sTNFR1-IG, pHRsTNFR1-IgG-IG, or pHR-G were produced by standard calcium-phosphate precipitation (chemicals from Sigma) with human embryonic kidney 293T cells as the packaging cell line as previously described,16 modified for production in 175 cm2 flasks. Viral particles were harvested from the culture supernatant 72 hours posttransfection, passed through a 0.45μm filter and concentrated approximately 100 × g by ultracentrifugation. Viral pellets were resuspended in RPMI 1640 (GIBCO) and stored at −80°C until use. Vectors made with pHR-G, pHR-sTNFR1-IG, and pHR-sTNFR1-IgG-IG were designated LV-GFP, LV-sTNFR1, LV-sTNFR1-IgG, respectively.
ELISA or Multiplex Protein Analysis
Brain tissue was homogenized on ice in cell lysis buffer using either a glass homogenizer (Wheaton—for Figure 2) or an electric homogenizer (Biospec (Bartlesville, OK, USA) Tissue-Tearor – for Figures 3 and 4). Cell lysis buffer contained 1 × lysis buffer (Cell Signaling, Danvers, MA, USA), 1:1,000 phosphatase inhibitor cocktail I (Sigma), 1:1,000 phosphatase inhibitor cocktail II (Sigma), and 1 Roche (Pleasanton, CA, USA) Complete Mini Pill per 10 mL lysis buffer. For Figures 2 and 4, the brain was sliced on a brain matrix in 2-mm slices; five slices were homogenized, corresponding to those used for damage analysis, in 1 mL lysis buffer. For Figure 3, 0.5 mL Procarta (Santa Clara, CA, USA) cell lysis buffer was used for three slices (slice one, three, and five). Lysate was pelleted at 13,000 r.p.m. for 10 minutes at 4°C, and the supernatant was collected and spun again. The final supernatant was transferred to a new tube and frozen at −80°C. Total protein was assessed using Bradford reagent (Bio-Rad, Hercules, CA, USA) and ELISA results were normalized across total protein levels. The following homogenate incubation times, dilutions of homogenate, and ELISA kits were used according to manufacturer's directions unless otherwise noted: IL-6 (overnight 4°C; 1:5; DuoSet, R&D Systems), Il-1β (2 hours room temperature; 1:2, Quantikine, R&D Systems), TNF-α (overnight 4°C; 1; Ready, Set, Go! eBioscience, San Diego, CA, USA), ICAM-1 (2 hours room temperature; 1:20; AbCam), MCP-1 (overnight 4°C; 1:50; BD OptEIA Set, BD Biosciences, San Jose, CA, USA), and GFP (overnight 4°C; 1:2.5; Cell Biolabs, San Diego, CA, USA). An 11-plex polystyrene bead kit was custom ordered from Procarta (ICAM-1, sVCAM-1, IL-6, MCP-1, MIP-1a, TNF-α, IL-1β, RANTES, GROα, IFN-γ, and IP-10; Affymetrix, Santa Clara, CA, USA). Tissue homogenate was incubated for 2 hours at room temperature, then overnight at 4°C, and the secondary antibody was incubated at room temperature for 1 hour.
Detection of sTNFR1 by ELISA
Dendritic cell production of cargo was measured by mouse sTNFR1 ELISA (cross-reactive with rat sTNFR1; R&D Systems). Bone marrow was plated in 12-well plates in the same density and conditions used for in vivo studies. On DIV 4, cells were transduced with LV-GFP, LV-sTNFR1, or LV-sTNFR1-IgG (MOI=10–30), and media was changed on DIV 5. Conditioned media was collected at 24, 48, and 72 hours after the transduction period, any floating cells and debris were pelleted at 1,300 r.p.m. for 5 minutes at 4°C, and supernatant was transferred to a new tube and frozen at −20°C until use. After the first media collection, the number of DCs per well were counted to normalize sTNFR1 production per 100,000 cells. Media was collected from three separate transductions and run on the same ELISA plate in duplicate.
TNF-α Bioactivity Assay
The bioactivity of TNF-α in the presence of sTNFR1 and sTNFR1-IgG compared with GFP was assessed with WEHI-164 clone 13 cells (Sigma). HEK293T cells at a density of approximately 1.5 × 105 cells/cm2 in a 75 cm2 flask were transduced with LV-GFP, LV-sTNFR1, or LV-sTNFR1-IgG (MOI=1 to 2) in the presence of 10 μg/mL protamine sulfate. After 18 hours, the media was changed with 7 mL of new media and the conditioned media was harvested after an additional 48 hours, spun at 2,000 r.p.m. for 5 minutes to remove debris, and transferred to a new tube. Media was stored at −80°C until use. Media was assayed on the mouse sTNFR1 ELISA and the concentrations of sTNFR1 and sTNFR1-IgG were normalized based on the ELISA results. Green fluorescent protein media was assayed undiluted. WEHI cells (3 × 104 per well) were incubated with 1 μg/mL Actinomycin-D (Sigma) and with 0, 0.01, 0.1, or 1.0 pg/mL rat recombinant TNF-α (BioLegend, San Diego, CA, USA) in a 96-well plate. In addition, one of four two-fold dilutions of GFP, sTNFR1, or sTNFR1-IgG conditioned media were added to each well and all combinations were run in triplicate. Cells incubated for 20 hours at 37°C and were assessed for cell viability using MTS according to manufacturer's directions (CellTiter 96 Aqueous Non-Radioactive Cell Proliferation Assay; Promega, Sunnyvale, CA, USA). Results are displayed as percent viability compared with control WEHI-164 cells (0 pg/mL TNF, 0 μg/mL Actinomycin-D, control (unconditioned) media).
Flow Cytometry
On DIV 7, cultures were harvested in their own media and washed in Fluorescence-activated cell sorting buffer (1 mmol/L EDTA, 1% fetal bovine serum in PBS). The following steps were performed at 4°C. Cells were incubated with primary antibody for 30 minutes on ice (mouse anti-rat Ox42-biotin 1:100 (AbD Serotec, Bio-Rad)), mouse anti-rat CD11c (clone 8A2 1:20 (AbCam)), mouse anti-rat MHC class II (RT1B 1:50 (AbD Serotec)), mouse anti-rat CD80-biotin (1:20 (eBioscience)), mouse anti-rat CCR2-APC (1:10 (R&D Systems)), mouse anti-rat CCR5-APC (1:10 (R&D Systems)), mouse anti-rat CD54 (1:20 (ICAM-1) (BDPharmingen, Sunnyvale, CA, USA)) and mouse IgG1 negative control-Alexa Fluor 647 (1:10 (AbD Serotec)). For unconjugated antibodies, cells were washed and incubated with fluorophore-conjugated secondary antibody for 30 minutes (streptavidin-PECy7 (1:500 (Invitrogen)) or goat anti-mouse IgG-RPE ([STAR76] 1:10 (AbD Serotec))). Cells were washed again, resuspended in Fluorescence-activated cell sorting buffer, and run immediately on an LSR II flow cytometer (BDBiosciences, Sunnyvale, CA, USA). Results were graphed with FlowJo Software (Tree Star, Ashland, OR, USA). Flow cytometry was run twice, with separate batches of cells, and a representative run is shown as both runs produced similar results.
Statistics
Results were assessed and graphed using SigmaPlot 12.0 (Systat, Chicago, IL, USA). For time-course studies (Figures 1A and 1B), the number of GFP+ DCs was counted across six sections and multiplied by 30 (30, 30 μm slices between each section). A one-way analysis of variance (ANOVA) compared the time points. For inflammation and migration analysis (Figure 2), a Pearson correlation compared all inflammatory proteins and GFP (exDC) levels. For inflammation, damage, and migration analysis (Figure 3), data were transformed into natural log plots for damage and inflammatory proteins and a Pearson correlation assessed significance across all groups. A standard linear regression was used to compare GFP and all inflammatory proteins because of 0 values for GFP for some subjects. For ELISA measurement of sTNFR1 production and release into the media (Figure 6A), a two-way ANOVA and Student–Newman–Keuls (S–N–K) post hoc were used. For WEHI-164 cell death analysis (Figure 6B), a two-way ANOVA compared conditioned media treatment and pg/mL TNF. For inflammation assessment (Figure 4), a one-way ANOVA compared protein values across groups at 24 hours (vehicle (N=9), GFP (N=9), sTNFR1 (N=9)) and 3 days (vehicle (N=9), GFP (N=10), sTNFR1 (N=10)). For damage assessment (Figure 5), a two-way ANOVA and S-N-K post hoc compared each of five slices across all groups at 24 hours (vehicle (N=14), GFP (N=16), sTNFR1 (N=14), sTNFR1-IgG (N=11)) and 3 days (vehicle (N=16), GFP (N=13), sTNFR1 (N=13), sTNFR1-IgG (N=9)).
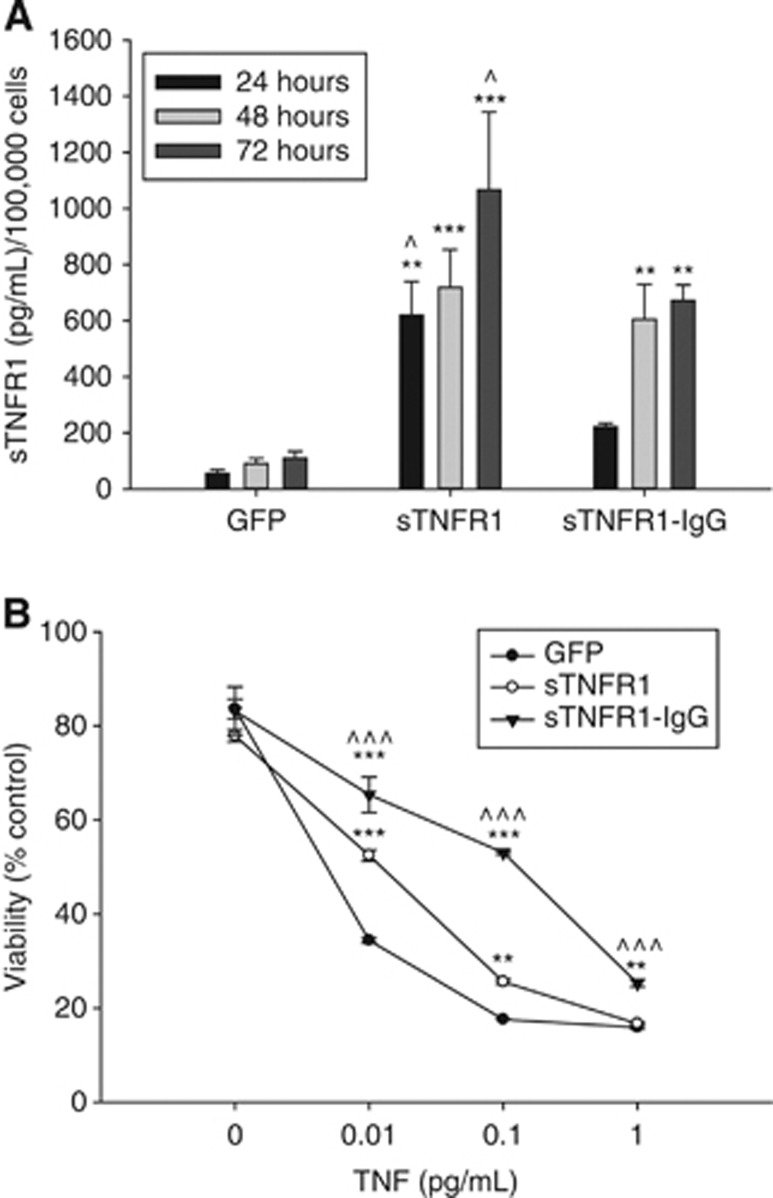
Figure 6.
(A) ExDCs transduced with LV-sTNFR1 show overexpression of sTNFR1 protein compared with ex vivo-derived dendritic cells (exDCs) transduced with LV-green fluoroscent protein (GFP). Ex vivo-derived dendritic cells (exDCs) transduced with LV-sTNFR1-IgG show decreased production compared with exDCs transduced with LV-sTNFR1. Two way analysis of varience (ANOVA) (day, conditioned media) and S-N-K post hoc test. *P<0.05, **P<0.01, and ***P<0.001 compared with GFP-conditioned media; ^P<0.05 compared with sTNFR1-IgG conditioned media; error bars indicate s.e.m. (B) sTNFR1 and sTNFR1-IgG conditioned media significantly block the effects of tumor necrosis factor (TNF) and Actinomycin-D at moderate doses of TNF in WEHI-164 cells. sTNFR1-IgG shows improved blockage of TNF bioavailability compared with sTNFR1- and GFP-conditioned media at all doses of TNF. Two-way ANOVA (TNF, conditioned media) and S-N-K post-hoc test. *P<0.05, **P<0.01, and ***P<0.001 comparedwith GFP-conditioned media; ^P<0.05 ^^^P<0.001compared to sTNFR1-conditioned media; error bars indicate s.e.m.
Results
Ex vivo-Derived Dendritic Cells Migrate to the Inflamed Brain and Leave Within 6 Hours
We have shown previously that ex vivo-derived DCs (exDCs) migrate to the ischemic area of the brain when injected 3 hours post-tMCAO reperfusion.3 To define the broader therapeutic window for this approach, we characterized the time-course of exDC recruitment to the injury site after stroke. ExDCs from the bone marrow of postnatal day 15 to 17 GFP+/− transgenic rats were isolated and cultured. Two million GFP+ exDCs were injected via the carotid artery 3, 6, 12, 18, or 24 postreperfusion and animals were killed 3 hours postinjection. Twenty-four hours was chosen as the maximal time point for this study as most damage occurs by then.17 We quantified the number of GFP+ cells in six representative 30 μm slices in the infarcted hemisphere with a 900 μm interval between each slice. This number was then multiplied by 30 to extrapolate the total GFP+ exDC number across the stroke hemisphere. Of the time points tested, animals injected at 6 hours post-tMCAO reperfusion showed the highest numbers of GFP+ exDCs in the ipsilateral hemisphere; however, this increase in exDCs did not differ from the other time points investigated, which also exhibited substantial exDC migration (Figure 1A). No exDCs were observed in the contralateral hemisphere (data not shown).
We next examined how long and where exDCs remain in the brain after injection. Animals were subjected to tMCAO and then injected with 2 million GFP+ exDCs at 6 hours poststroke. At 10 minutes, 3 hours, or 6 hours postinjection, animals were killed and the number of GFP+ cells was quantified. ExDCs were observed in the brain within 10 minutes of injection, were dramatically reduced by 3 hours, and were absent by 6 hours (Figure 1B). We then determined whether exDCs crossed the blood–brain barrier (BBB) into the brain parenchyma. Brain sections were stained with the endothelial marker RECA-1 and all of GFP+ exDCs colocalized with RECA-1 staining, appearing to fill the vessel, indicating the cells did not leave the brain vasculature to cross into the brain parenchyma, however entry into the glia limitans was not ruled out (Figures 1C–1H); this is in agreement with our prior findings.3
Ex vivo-Derived Dendritic Cells Migration Correlates with Inflammation Levels in the Brain
While exDCs do not enter the brain parenchyma, the production of inflammatory factors such as TNF-α and IL-1β by cells in the infarcted region directly affect vascular environment. For example, TNF-α increases expression of ICAM-1 on the endothelial cell surface18 and in vitro human endothelial migration models have shown that ICAM-1 directly mediates exDC firm adhesion and migration across endothelial cells.19 We next determined whether levels of inflammation in the post-tMCAO brain were associated with high or low exDC retention in ischemic areas. After tMCAO, animals were injected with 2.5 million GFP+ exDCs at 6 hours poststroke and killed after 10 minutes. The entire injured ipsilateral hemisphere was homogenized and used to quantify and compare the number of exDCs to the amount of proinflammatory factors. The proinflammatory cytokines IL-1β, TNF-α, and IL-6 and chemokine MCP-1 correlated with the number of exDCs (GFP; Figures 2A–2D). In addition, levels of the vascular adhesion molecule ICAM-1 (Figure 2E) correlated with the number of exDCs. The levels of each inflammatory factor correlated with each other as well, most strikingly with TNF and MCP-1 (Figure 2F). Thus, the acute inflammatory response initiated by ischemic stroke is associated with exDC recruitment to the site of injury.
Ex Vivo-Derived Dendritic Cells Migration and Damage Correlate with Inflammation
While 6 hours poststroke is a time when both damage and ischemia-induced inflammation are rising quickly, it is not clear if these two factors are closely correlated at such an early time point poststroke.4, 17 At 24 hours poststroke, peripheral inflammatory levels, particularly TNF-α, correlate with stroke severity;20 however, studies have not identified whether several inflammatory factors in the ischemic region of the CNS closely correlate with the damage within animals during this early, rapidly changing phase. To determine whether the extent of inflammation and/or exDC migration correlate with the amount of damage after stroke, we used a combination of 2,3,5-triphenyltetrazolium chloride (TTC) staining for damage assessment and ELISAs for protein measurement. Animals were injected with 2.5 million GFP+ exDCs at 6 hours poststroke reperfusion and killed within 10 minutes of the injection. We divided the brain into 2-mm slices on a brain matrix and used slices one, three, and five for protein analysis and slices two and four for damage assessment. We quantified the proinflammatory cytokines TNF-α, IL-6, IL-1β, CXCL1 (GRO-α; CINC-1), IFN-γ, CXCL10 (IP-10); chemokines MCP-1, CCL3 (MIP-1 α), and CCL5 (RANTES); and vascular adhesion molecules ICAM-1 and soluble VCAM-1. We then compared the proinflammatory profile to numbers of exDCs (GFP) and the volume of ischemia in the cortex, striatum, and whole hemisphere for each animal. Of the factors measured, levels of ICAM-1 and exDCs correlated (Figure 3A). In addition, total damage and TNF-α, IL-1β, and MCP-1 correlated after log-linear analysis (Figures 3B–3D). Levels of damage in areas measured (total, cortical, and striatal) and the number of exDCs were not correlated (R2=−0.023, P=0.575; data not shown). The sensitivity may not be as high in this 11-plex assay, however, since slices two and four were not run for protein analysis, and caution should be taken when comparing previous ELISA results with this study. Thus, the extent of inflammation and exDC migration correlate, and at this early time point of 6 hours poststroke, levels of proinflammatory factors IL-1β, TNF-α, and MCP-1 are also correlated with the extent of damage.
sTNFR1 is Produced by Ex vivo-Derived Dendritic Cells and Blocks Tumor Necrosis Factor Bioavailability
As higher inflammation brings greater numbers of exDCs and higher inflammation is associated with more severe infarct,4 we next hypothesized that exDC-mediated delivery of an anti-inflammatory therapy might reduce damage after stroke. Reducing the bioavailability of TNF-α has shown promise in animal studies of acute brain injury. For example, intracerebroventricular delivery of anti-TNF antibody at the onset of stroke decreases damage and expression of ICAM-1 in the stroke hemisphere.21 First, we designed a lentiviral construct that allowed constitutive overexpression of rat soluble TNF receptor I (LV-sTNFR1) or rat soluble TNF receptor I fused to the Fc portion of human immunoglobulin G (IgG; LV-sTNFR1-IgG). The sTNFR1-IgG fusion protein forms a dimer and has been shown to have improved binding ability to TNF-α compared with sTNFR1 alone.22 To confirm that exDCs release sTNFR1 and sTNFR1-IgG into the media after lentiviral transduction and to determine the relative production of sTNFR1- or sTNFR1-IgG-transduced cells, conditioned media from GFP-, sTNFR1-, or sTNFR1-IgG-transduced exDCs was run on a sTNFR1 ELISA. As expected, the amount of sTNFR1 or sTNFR1-IgG increases in the media over time, but interestingly, sTNFR1-IgG-exDCs produce lower amounts of cargo compared with sTNFR1-exDCs (Figure 6A). A two-way ANOVA (conditioned media type, time post-transduction) and Student-Newman-Keuls (S-N-K) post hoc test determined that sTNFR1-exDCs produce more sTNFR1 than sTNFR1-IgG-exDCs at 24 hours and 72 hours post-transduction.
Next, we determined the functionality of the sTNFR1 and sTNFR1-IgG cargo in vitro. TNF-sensitive WEHI-164 cells were incubated with Actinomycin-D, varying levels of rat recombinant TNF, and conditioned media from GFP, sTNFR1, or sTNFR1-IgG-transfected cells. At 0.01, 0.1, and 1.0 pg/mL TNF, sTNFR1-conditioned media protected the WEHI-164 cells from TNF-induced death, while sTNFR1-IgG conditioned media showed improved binding ability to TNF compared with sTNFR1 (Figure 6B). Two-way ANOVA (conditioned media type, TNF concentration) and S-N-K post hoc test indicated significant protection by sTNFR1 media compared with GFP media at 0.01 and 0.1 pg/mL TNF and sTNFR1-IgG media at 0.01, 0.1, and 1.0 pg/mL TNF. Overall, both sTNFR1 and sTNFR1-IgG were effective at moderate TNF concentrations, while sTNFR1-IgG showed improved cell viability compared with sTNFR1 at all TNF concentrations.
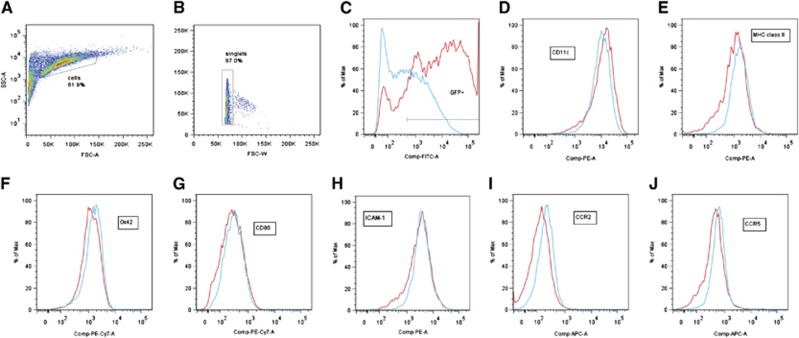
To determine whether exDCs have an altered phenotype after LV-sTNFR1 transduction compared with LV–GFP transduction, sTNFR1-exDCs and GFP-exDCs were stained for DC-related markers, and we observed no differences across the GFP+ populations (successfully transduced) for Ox42 (CD11b/c), CD80, MHC class II, and ICAM-1 (Figure 7). We observed a slight increase in CD11c+ cells in the sTNFR1-DC population (Figure 7D) and a slight increase in CCR2+ (Figure 7I) and CCR5+ (Figure 7J) cells in the GFP–DC population. To determine whether these surface protein expression differences presented a functional difference in migration, we injected sTNFR1- or GFP-exDCs at 6 hours postreperfusion and killed the animals within 10 minutes of injection to measure GFP+ cell counts. We observed no differences in migration between the two groups (P=0.196, GFP N=12, sTNFR1 N=14; data not shown).
Figure 7.
(A–J) Representative flow cytometry for lentiviral (LV)-green fluorescent protein GFP- (red) and LV-sTNFR1-transduced (blue) exDCs. Gating strategy for (A) live dendritic cells (DCs), (B) singlets, and (C) GFP+ transduced population. (D) The percent CD11c+ cells shows slight increase for GFP-ex vivo-derived dendritic cells (exDCs). No difference in (E) MHC class II, (F) Ox42, (G) CD80, or (H) ICAM-1 expression. The percent (I) CCR2+ and (J) CCR5+ cells shows a slight increase for sTNFR1-exDCs.
sTNFR1-Ex vivo-Derived Dendritic Cells-Treated Animals Show Decreased Inflammation
After confirmation that sTNFR1-exDCs are phenotypically similar to GFP-exDCs after lentiviral transduction and sTNFR1 reduces TNF bioavailability, we determined whether delivery of sTNFR1-exDCs reduces overall levels of inflammation in the brain 24 hours and 3 days poststroke. Animals were subjected to tMCAO, injected with vehicle or GFP- or sTNFR1-exDCs at 6 hours poststroke, and killed at either 24 hours or 3 days poststroke. A one-way ANOVA at 24 hours poststroke revealed that animals treated with GFP-exDCs demonstrated increased levels of TNF-α, MCP-1, and IL-6 compared with sTNFR1-exDC and vehicle-treated animals (Figures 4A–4C). Levels of IL-1β were not different across the groups (Figure 4D). At 3 days poststroke, there were no differences across animals for TNF-α, MCP-1, IL-6, and IL-1β (Figures 4E–4H). Thus, sTNFR1-exDC-treated animals had reduced inflammation in the brain within hours after injection.
sTNFR1-Ex vivo-Derived Dendritic Cells Reduce Stroke Damage
As sTNFR1-exDC-treated animals showed less inflammation compared with GFP-exDC-treated animals, we next determined whether delivery of sTNFR1- or sTNFR1-IgG-exDCs would reduce damage in the stroke brain. Animals were subjected to tMCAO and vehicle or GFP-, sTNFR1-, or sTNFR1-IgG-exDCs were injected at 6 hours postreperfusion. Animals were killed at 24 hours or 3 days postreperfusion and assessed for damage in the cortex, striatum, or total injured hemisphere. A two way-ANOVA (slice number, cell type/vehicle) confirmed that delivery of sTNFR1- or sTNFR1-IgG-exDCs did not reduce damage 24 hours poststroke (Figure 5A). However, sTNFR1-exDC-treated animals killed at 3 days poststroke had 25% less total and cortical percent damage compared with vehicle- and GFP-exDC-treated animals (Figures 5B–5F). In addition, sTNFR1-exDC-treated animals had less damage in the striatum compared with vehicle-treated animals. STNFR1-IgG-exDC-treated animals showed a trend toward protection at 3 days poststroke. Overall, sTNFR1-exDC-treated animals had less long-term damage compared with control animals.
Discussion
Current therapies in patients with ischemic stroke are designed to treat the clot, but new therapies are needed to treat the resulting damage. Systemic delivery of therapeutic proteins or viral vectors is inefficient and can lead to off-target effects, while direct injection into the brain parenchyma is extremely invasive. Our novel delivery system exploits the migratory ability of exDCs toward proinflammatory factors emanating from the region of injury. In this study, we show that exDC injury homing correlates with the relative expression of proinflammatory factors in the injured hemisphere. In addition, we demonstrate that delivery of the anti-inflammatory protein therapy sTNFR1 via exDCs reduces damage and inflammation after stroke.
Ex vivo-Derived Dendritic Cells Respond to Inflammatory Factors in vivo
During the first 6 hours of reperfusion after temporary middle cerebral artery occlusion (tMCAO), the levels of proinflammatory factors are rising and damage is increasing quickly. Ischemic stroke triggers the activation of transcription factors such as nuclear factor κB (NF-κB), which causes an increase in the expression of many proinflammatory genes such as TNF-α and IL-1β.4 Protein levels of cytokines and chemokines, most of which are normally produced at very low or undetectable levels in the brain, show increasing concentrations after a stroke because of the production by endothelial cells, migrating immune cells, and glia.4 At this early time point, the number of endogenous leukocytes is very low, and it is not clear whether these leukocytes enter the parenchyma.12, 13 Our data show that when injected at 6 hours poststroke, exDCs home to the ischemic hemisphere, but do not enter the brain parenchyma. In addition, our current and previous data indicate exDCs are an immature phenotype because of the low CD80 expression, even after LV transduction.3 Upon lipopolysaccharide stimulation, exDCs become mature with increased CD80 expression and decreased retention in the injury site.3 Previous studies in our lab have also shown that after clearing from the brain vasculature, exDCs delivered at 6 hours poststroke migrate to the lung, liver, spleen, and gut over time.3
Previously published in vitro studies have shown that exDCs respond to chemoattractants such as MCP-1 (ref. 23), and therefore, would likely be responsive to the inflamed brain in a similar manner as endogenous leukocytes. To determine which factors mediate exDC migration, we measured the correlation of proinflammatory factors and exDC migration in the brain 6 hours poststroke. The number of exDCs correlated with levels of all the factors we measured in multiple ELISAs, including IL-6, TNF-α, IL-1β, MCP-1, and ICAM-1. We also determined that infarct size and proinflammatory factors TNF-α, IL-1β, and MCP-1 are also correlated in a log-linear manner. Previous studies have observed a logarithmic correlation between stroke severity, as measured by the Scandanavian Stroke Scale and Barthel Index, and inflammation as measured in the serum and cerebrospinal fluid of human patients.20 Therefore, our study shows that even during this early time point after the onset of stroke, inflammation and damage and inflammation and exDC migration are already correlated within the subject.
Ex vivo-Derived Dendritic Cells Reduce Inflammation and Damage Poststroke
Because the exDCs appeared responsive to the level of inflammation in the poststroke brain, we hypothesized that they would be well poised to deliver variable levels of anti-inflammatory proteins that would be tailored to the specific level of inflammation in a given animal. Tumor necrosis factor was chosen as the target for our anti-inflammatory therapy since it has multiple effects on the brain after stroke and has a significant role in proinflammatory processes. Tumor necrosis factor receptor 1 (TNFR1), one of the two receptors for TNF, is expressed on most cell types in the body. Signaling via this receptor can lead to activation of the transcription factor NF-κB and expression of a variety of anti- and proinflammatory, anti- and pro-apoptotic, and cytoprotective and cytotoxic proteins.24 In addition, TNF can cause pro-coagulant effects, stimulate production of reactive oxygen species, increase BBB permeability, and activate immune cells in the area, all of which can lead to increased leukocyte recruitment at the injury site.24 We developed a lentiviral vector that successfully transduced exDCs to constitutively produce the soluble TNFR1 protein and the resulting sTNFR1 cargo reduced the bioactivity of TNF in vitro.
Animals treated with sTNFR1-exDCs showed a significant reduction in proinflammatory factors TNF-α, IL-6, and MCP-1 at 24 hours poststroke compared with GFP-exDC-treated animals. This reduction in inflammation also corresponded with a trend toward damage reduction at the same time point. In addition, we did not observe any significant differences in TNF-α, IL-6, IL-1β, and MCP-1 measured at 3 days poststroke between vehicle-, GFP-exDC-, or sTNFR1-exDC-treated animals. Importantly, sTNFR1-exDC-treated animals showed a significant, 25% reduction in long-term damage compared with vehicle- and GFP-exDC-treated animals. It is possible we did not observe a significant reduction in inflammation at 3 days poststroke because different proinflammatory factors are important at later time points compared with 24 hours poststroke. Levels of TNF, IL-6, IL-1β, and MCP-1 are associated with the early progression of brain damage and decrease after 24 hours. It is conceivable that levels of other proinflammatory factors or other pathways of TNF-mediated damage the brain, such as BBB permeability or reactive oxygen species production, would be reduced in the sTNFR1-exDC-treated animals.
Several previous studies have shown successful reduction of damage and inflammation with the delivery of anti-TNF therapies at or before the onset of stroke.21, 25 Sumbria and colleagues;26 showed protection as far out as 90 minutes poststroke; however, our study is the first showing successful reduction of damage when the anti-TNF intervention is delivered at much later time points after stroke onset. In addition, a 25% reduction in lesion volume is similar in magnitude of damage reduction observed in previous studies employing anti-TNF therapies.21, 25, 26 The relationship between infarct and inflammation reduction and functional improvement is less clear, however, and depends on the timing of the intervention, the severity of the damage, and location of the insult.27 On the one hand, Barone and colleagues 25 showed no effect on behavior with damage-reducing anti-TNF treatment,25 while Sumbria and colleagues showed a significant reduction in neurologic deficit behavior with their anti-TNF treatment.26 It should be noted, however, that even a modest reduction in lesion volume has been shown to promote functional improvement. For example, treatment with tissue plasminogen activator and minocycline after stroke resulted in a non-significant, 20% reduction in damage, but animals demonstrated a significant improvement in behavioral tests compared with controls.28 Future studies should address the behavior of animals treated with anti-inflammatory exDCs to determine the extent of functional sparing.
Interestingly, despite the superior binding ability of sTNFR1-IgG cargo to TNF-α in vitro, exDCs transduced with sTNFR1-IgG LV did not show significant protection in vivo in the stroke brain. This lack of protection is likely due to the decreased production by exDCs of the sTNFR1-IgG cargo. The efficacy of sTNFR1-IgG in reducing the bioavailability of TNF in the WEHI-164 assay did not directly translate to increased efficacy in the more complex in vivo environment. Also, the role of human sTNFR1-IgG (etanercept) in protection after acute injury has been controversial with some,29 but not all,26 showing protection.
In conclusion, our data demonstrate that the use of exDCs as a delivery vehicle for protein therapy shows promise as a new tool in the treatment of stroke. We show that exDCs are capable of delivering a variety of anti-inflammatory cargo and past studies have also demonstrated the efficacy of delivering damage reducing anti-apoptotic factors at the earlier time point of 3 hours poststroke.3 In the future, cells could be isolated and prepared before procedures such as cardiac surgery, where the chance of stroke is as high as 16%, and injected into patients if a stroke occurs.30 Finally, this technology may be applicable to many types of acute, proinflammatory neurological insults, the subject of future studies.
Acknowledgments
We thank Nathan Manley for his guidance and technical assistance, the Stanford FACS Facility for their technical assistance, and Sara Michie, Susan McConnell, and Tony Wyss-Coray for their guidance.
The authors declare no conflict of interest.
Footnotes
This work was generously supported by John A and Cynthia Fry Gunn. Salary support for MGW was provided by the Gabilan Stanford Graduate Fellowship. Salary support for JBK was provided by the Stanford Vice Provost for Undergraduate Education.
References
- Killer M, Ladurner G, Kunz AB, Kraus J. Current endovascular treatment of acute stroke and future aspects. Drug Discov Today. 2010;15:640–647. doi: 10.1016/j.drudis.2010.04.007. [DOI] [PubMed] [Google Scholar]
- Lansberg MG, Bluhmki E, Thijs VN. Efficacy and safety of tissue plasminogen activator 3 to 4.5 hours after acute ischemic stroke. Stroke. 2009;40:2438–2441. doi: 10.1161/STROKEAHA.109.552547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manley NM, Caso JR, Works MG, Cutler AB, Zemlyak I, Sun G, et al. Derivation of injury-responsive dendritic cells for acute brain targeting and therapeutic protein delivery in the stroke-injured rat. PLoS One. 2013;8:e61789. doi: 10.1371/journal.pone.0061789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Q, Tang XN, Yenari MA. The inflammatory response in stroke. J Neuroimmunol. 2007;184:53–57. doi: 10.1016/j.jneuroim.2006.11.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu T, Clark RK, McDonnel PC, Young PR, White RF, Barone FC, et al. Tumor necrosis factor-alpha expression in ischemic neurons. Stroke. 1994;25:1481–1488. doi: 10.1161/01.str.25.7.1481. [DOI] [PubMed] [Google Scholar]
- Buttini M, Sauter A, Boddeke NW. Induction of interleukin-1 beta mRNA after focal cerebral ischemia in the rat. Mol Brain Res. 1994;23:126–134. doi: 10.1016/0169-328x(94)90218-6. [DOI] [PubMed] [Google Scholar]
- Davies CA, Loddick SA, Toulmond S, Stroemer PP, Hunt J, Rothwell NJ. The progression and topographic distribution of interleukin-1beta expression after permament middle cerebral artery occlusion in the rat. J Cereb Blood Flow Metab. 1999;19:87–98. doi: 10.1097/00004647-199901000-00010. [DOI] [PubMed] [Google Scholar]
- Caso JR, Moro MA, Lorenzo P, Lizasoain I, Leza JC. Involvement of IL-1 beta in acute stress induces worsening of cerebral ischemia in rats. Eur J Neuropsychopharmacol. 2007;17:600–607. doi: 10.1016/j.euroneuro.2007.02.009. [DOI] [PubMed] [Google Scholar]
- Yamagami S, Tamura M, Hayashi M, Endo N, Tanabe H, Katsuura Y, et al. Differential production of MCP-1 and cytokine-induced neutrophil chemoattractant in the ischemic brain after transient focal ischemia in rats. J Leukoc Biol. 1999;66:744–749. doi: 10.1002/jlb.65.6.744. [DOI] [PubMed] [Google Scholar]
- Nishi T, Maier CM, Hayashi T, Saito A, Chan PH. Superoxide dismutase 1 overexpression reduces MCP-1 and MIP-1α expression after transient focal cerebral ischemia. J Cereb Blood Flow Metab. 2005;25:1312–1324. doi: 10.1038/sj.jcbfm.9600124. [DOI] [PubMed] [Google Scholar]
- Offner H, Vandenbark AA, Hurn PD. Experimental stroke induces massive, rapid activation of the peripheral immune system. J Cereb Blood Flow Metab. 2006;26:654–665. doi: 10.1038/sj.jcbfm.9600217. [DOI] [PubMed] [Google Scholar]
- Gelderblom M, Leypoldt F, Steinbach K, Behrens D, Choe C, Siler DA, et al. Temporal and spatial dynamics of cerebral immune cell accumulation in stroke. Stroke. 2009;40:1849–1857. doi: 10.1161/STROKEAHA.108.534503. [DOI] [PubMed] [Google Scholar]
- Kostulas N, Li H, Xiao B, Huang Y, Kostulas V, Link H. Dendritic cells are present in the ischemic brain after permanent middle cerebral artery occlusion in the rat. Stroke. 2002;33:1129–1134. doi: 10.1161/hs0402.105379. [DOI] [PubMed] [Google Scholar]
- Belayev L, Alonsa OF, Busto R, Zhao W, Ginsberg MD. Middle cerebral artery occlusion in the rat by intraluminal suture neurological and pathological evaluation of an improved model. Stroke. 1996;27:1616–1623. doi: 10.1161/01.str.27.9.1616. [DOI] [PubMed] [Google Scholar]
- Grauer O, Wohlleben G, Seubert S, Weishaupt A, Kampgen E, Gold R. Analysis of maturation states of rat bone marrow-derived dendritic cells using an improved culture technique. Histochem Cell Biol. 2002;117:351–362. doi: 10.1007/s00418-002-0384-4. [DOI] [PubMed] [Google Scholar]
- Naldini L, Blomer U, Gallay P, Ory D, Mulligan R, Gage FH, et al. In vivo gene delivery and stable transduction of nondividing cells by a lentiviral vector. Science. 1996;272:263–267. doi: 10.1126/science.272.5259.263. [DOI] [PubMed] [Google Scholar]
- Xu X, Zhang S, Yan W, Li X, Zhang H, Zheng X. Development of cerebral infarction, apoptotic cell death and expression of X-chromosome-linked inhibitor of apoptosis protein following focal cerebral ischemia in rats. Life Sci. 2006;78:704–712. doi: 10.1016/j.lfs.2005.05.080. [DOI] [PubMed] [Google Scholar]
- Fabry Z, Waldschmidt MM, Hendrickson D, Keiner J, Love-Homan L, Takei F, et al. Adhesion molecules on murine brain microvascular endothelial cells: expression and regulation of ICAM-1 and Lgp55. J Neuroimmunol. 1992;36:1–11. doi: 10.1016/0165-5728(92)90026-h. [DOI] [PubMed] [Google Scholar]
- Arjmandi A, Liu K, Dorovini-Zis K. Dendritic cell adhesion to cerebral endothelium: role of endothelial cells adhesion molecules and their ligands. J Neuropathol Exp Neurol. 2009;68:300–313. doi: 10.1097/NEN.0b013e31819a8dd1. [DOI] [PubMed] [Google Scholar]
- Zaremba J, Skrobanski P, Losy J. Tumour necrosis factor-alpha is increased in the cerebrospinal fluid and serum of ischaemic stroke patients and correlates with the volume of evolving brain infaract. Biomed Pharmacother. 2001;55:258–263. doi: 10.1016/s0753-3322(01)00058-0. [DOI] [PubMed] [Google Scholar]
- Yang GY, Gong C, Qin Z, Ye W, Mao Y, Bertz AL. Inhibition of TNF-alpha attenuates infarct volume and ICAM-1 expression in ischemic mouse brain. NeuroReport. 1998;9:2131–2134. doi: 10.1097/00001756-199806220-00041. [DOI] [PubMed] [Google Scholar]
- Kapadia S, Torre-Amione G, Yokoyama T, Mann DL. Soluble TNF binding proteins modulate the negative inotropic properties of TNF-alpha in vitro. Am J Physiol. 1995;268:H517–H525. doi: 10.1152/ajpheart.1995.268.2.H517. [DOI] [PubMed] [Google Scholar]
- Vecchi A, Massimiliano L, Ramponi S, Luini W, Bernasconi S, Bonecchi R, et al. Differential responsiveness to constitutive versus inducible chemokines of immature and mature mouse dendritic cells. J Leukoc Biol. 1999;66:489–494. doi: 10.1002/jlb.66.3.489. [DOI] [PubMed] [Google Scholar]
- Hallenbeck J. The many faces of tumor necrosis factor in stroke. Nat Med. 2002;8:1363–1368. doi: 10.1038/nm1202-1363. [DOI] [PubMed] [Google Scholar]
- Barone FC, Arvin B, White RF, Miller A, Webb CL, Willette RN, et al. Tumor necrosis factor-alpha. A mediator of focal ischemic brain injury. Stroke. 1997;28:1233–1244. doi: 10.1161/01.str.28.6.1233. [DOI] [PubMed] [Google Scholar]
- Sumbria RK, Boado RJ, Pardridge WM. Brain protection from stroke with intravenous TNF-alpha decoy receptor-Trojan horse fusion protein. J Cereb Blood Flow Metab. 2012;32:1933–1938. doi: 10.1038/jcbfm.2012.97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dumas TC, Sapolsky RM. Gene therapy against neurological insults: sparing neurons versus sparing function. Trends Neurosci. 2001;24:695–700. doi: 10.1016/s0166-2236(00)01956-1. [DOI] [PubMed] [Google Scholar]
- Machado LS, Sazonova IY, Kozak A, Wiley DC, El-Remessy AB, Ergul A, et al. Minocycline and tissue-type plasminogen activator for stroke asssssment of interaction potential. Stroke. 2009;40:3028–3033. doi: 10.1161/STROKEAHA.109.556852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chio C, Lin J, Chang M, Wang C, Kuo J, Yang C, et al. Therapeutic evaluation of etanercept in a model of traumatic brain injury. J Neurochem. 2010;115:921–929. doi: 10.1111/j.1471-4159.2010.06969.x. [DOI] [PubMed] [Google Scholar]
- Wolman RL, Nussmeier NA, Aggarwal A, Kanchuger MS, Roach GW, Newman MF, et al. Cerebral injury after cardiac surgery: identification of a group at extraordinary risk. Stroke. 1999;30:514–522. doi: 10.1161/01.str.30.3.514. [DOI] [PubMed] [Google Scholar]