Significance
EV71 infection with severe neurological complications has become a serious public health concern. However, suitable small animal models to study human EV71 pathogenesis are not available. We have generated a Tg mouse model by expressing the human EV71 receptor, Scavenger receptor B2, and found it to be susceptible to EV71 infection. This Tg mouse model exhibits neurological disease and pathology very similar to that observed in humans. The results confirm that the Scavenger receptor B2 receptor is important for EV71 infection in vivo. Further development of this new small animal model should greatly contribute toward investigation of EV71 pathogenesis and development of vaccines and antiviral drugs.
Keywords: picornavirus, neurotropism, viral receptor
Abstract
Enterovirus 71 (EV71) typically causes mild hand-foot-and-mouth disease in children, but it can also cause severe neurological disease. Recently, epidemic outbreaks of EV71 with significant mortality have been reported in the Asia-Pacific region, and EV71 infection has become a serious public health concern worldwide. However, there is little information available concerning EV71 neuropathogenesis, and no vaccines or anti-EV71 drugs have been developed. Previous studies of this disease have used monkeys and neonatal mice that are susceptible to some EV71 strains as models. The monkey model is problematic for ethical and economical reasons, and mice that are more than a few weeks old lose their susceptibility to EV71. Thus, the development of an appropriate small animal model would greatly contribute to the study of this disease. Mice lack EV71 susceptibility due to the absence of a receptor for this virus. Previously, we identified the human scavenger receptor class B, member 2 (hSCARB2) as a cellular receptor for EV71. In the current study, we generated a transgenic (Tg) mouse expressing hSCARB2 with an expression profile similar to that in humans. Tg mice infected with EV71 exhibited ataxia, paralysis, and death. The most severely affected cells were neurons in the spinal cord, brainstem, cerebellum, hypothalamus, thalamus, and cerebrum. The pathological features in these Tg mice were generally similar to those of EV71 encephalomyelitis in humans and experimentally infected monkeys. These results suggest that this Tg mouse could represent a useful animal model for the study of EV71 infection.
Enterovirus 71 (EV71) is a human enterovirus species A of the genus Enterovirus within the Picornaviridae family, and it is known to be one of the causative agents of hand-foot-and-mouth disease (HFMD) (1, 2). HFMD is generally considered to be a mild exanthematous disease. However, in some infants and young children, after a few days of prodromal illness, HFMD caused predominantly by EV71 can be complicated by neurological manifestations, including ataxia, tremor, myoclonus, polio-like paralysis, encephalomyelitis, cardiopulmonary failure, and death (3, 4). In humans with fatal EV71 encephalomyelitis, inflammation and viral antigens in neurons were observed mainly in the spinal cord, brainstem, hypothalamus, cerebellar dentate nucleus, and cerebrum (5, 6). Since the 1970s, HFMD outbreaks with significant mortality have been reported throughout the world, including in Bulgaria in 1975 [44 deaths (7)], Hungary in 1978 [47 deaths (8)], Malaysia in 1997 [29 deaths (9)], Taiwan in 1998 [78 deaths (3)], China in 2008–2011 [1,894 deaths (10), www.wpro.who.int/china/mediacentre/factsheets/hfmd/en/)], Vietnam in 2011 [166 deaths (www.wpro.who.int/emerging_diseases/HFMD/en/index.html)], and Cambodia in 2012 (www.wpro.who.int/mediacentre/releases/2012/20120713/en/index.html). Thus, EV71 infection has the potential to become the most serious public health issue caused by neurotropic picornaviruses after the eradication of poliovirus (PV). Unfortunately, there is still very little information concerning EV71 neuropathogenesis, and vaccines or anti-EV71 drugs have yet to be developed.
Appropriate animal models are needed to better understand EV71 neuropathogenesis and to facilitate the development of effective vaccines and drugs. EV71-infected cynomolgus monkeys developed neurological complications similar to those observed in human cases, including ataxia, tremor, and flaccid paralysis (11–15), and pathological lesions were observed in the spinal cord, brainstem, cerebellar dentate nucleus, and other parts of the brain (14, 15). However, the use of monkeys to model EV71 infection is difficult for both ethical and economic reasons. Other investigators have developed neonatal mouse models of EV71 infection using mouse-adapted virus strains or mice deficient in IFN receptors (16–20). Unfortunately, EV71 infection in neonatal mice is significantly different from that in humans, as the major viral replication sites in the mouse include the muscle and adipose tissues, which are not featured in human infection. Moreover, mice that are more than a few weeks old are generally not susceptible to EV71. Thus, it is necessary to establish a new experimental animal model that can overcome these limitations.
Viral receptors determine the host range specificity for certain enteroviruses (21). The transgenic (Tg) expression of the human PV receptor (PVR), CD155, and the major group human rhinovirus receptor, intracellular adhesion molecule-1 (ICAM-1) in mice conferred susceptibility to PV and major group human rhinoviruses, respectively (22–24). These Tg mice are therefore good small animal models to study the pathogenesis of these viruses. Recently, sialylated glycans (25), annexin II (26), human P-selectin glycoprotein ligand-1 (PSGL-1) (27), and human scavenger receptor class B, member 2 (hSCARB2) (28) were reported as candidate receptors for EV71. SCARB2, also known as lysosomal integral membrane protein-2, localizes mainly to lysosomes (29) and acts as a receptor for lysosomal targeting of β-glucocerebrosidase (30). Mouse cells transformed with hSCARB2 are rendered susceptible to all EV71 strains (31, 32), facilitating virus binding, internalization and uncoating (33), whereas mouse Scarb2 does not function as a receptor for EV71. Importantly, SCARB2 is widely expressed in many human tissues and cell types, including neurons in the CNS (34–36). Moreover, there is no evidence that other candidates can support viral infection as efficiently as SCARB2. Transgenic expression of human PSGL-1 in mice was not sufficient to permit EV71 infection (37). Together, these observations suggest that the expression of hSCARB2 in mice may confer susceptibility to EV71 infection. Here, we describe the generation and characterization of a Tg mouse expressing hSCARB2 and demonstrate its utility as a small animal model for the study of EV71 pathogenesis.
Results
Generation of hSCARB2-Tg Mice.
To generate Tg mice expressing hSCARB2, two bacterial artificial chromosome (BAC) clones (RP11-54D17 and RP11-628A4) containing the complete human scavenger receptor class B, member 2 (hSCARB2) gene and endogenous promoter were used (Fig. S1A). Genomic DNA was isolated from the resulting transgenic mice and subjected to PCR genotyping using the four primer sets indicated in Fig. S1B and Table S1. Four founder mice [C57BL/6 Tg(hSCARB2)10, 16, 22, and 24] and two founder mice [C57BL/6 Tg(hSCARB2)49 and 75] were obtained from the RP11-54D17–injected and RP11-628A4–injected groups, respectively. Using each primer set, the expected PCR product was amplified from Tg mice and positive control human rhabdomyosarcoma (RD) cells, but not from non-Tg mice (Fig. S1B). These results indicated that the Tg mice carried the entire hSCARB2 gene in their genome. The analysis of the hemizygous C57BL/6 Tg(hSCARB2)10 mouse, which we refer to as hSCARB2-Tg10, is presented in this article. Similar results were also obtained using hemizygous hSCARB2-Tg16, 22, and 49 mice.
Expression Profile of hSCARB2 in Human and Tg Mouse Tissues.
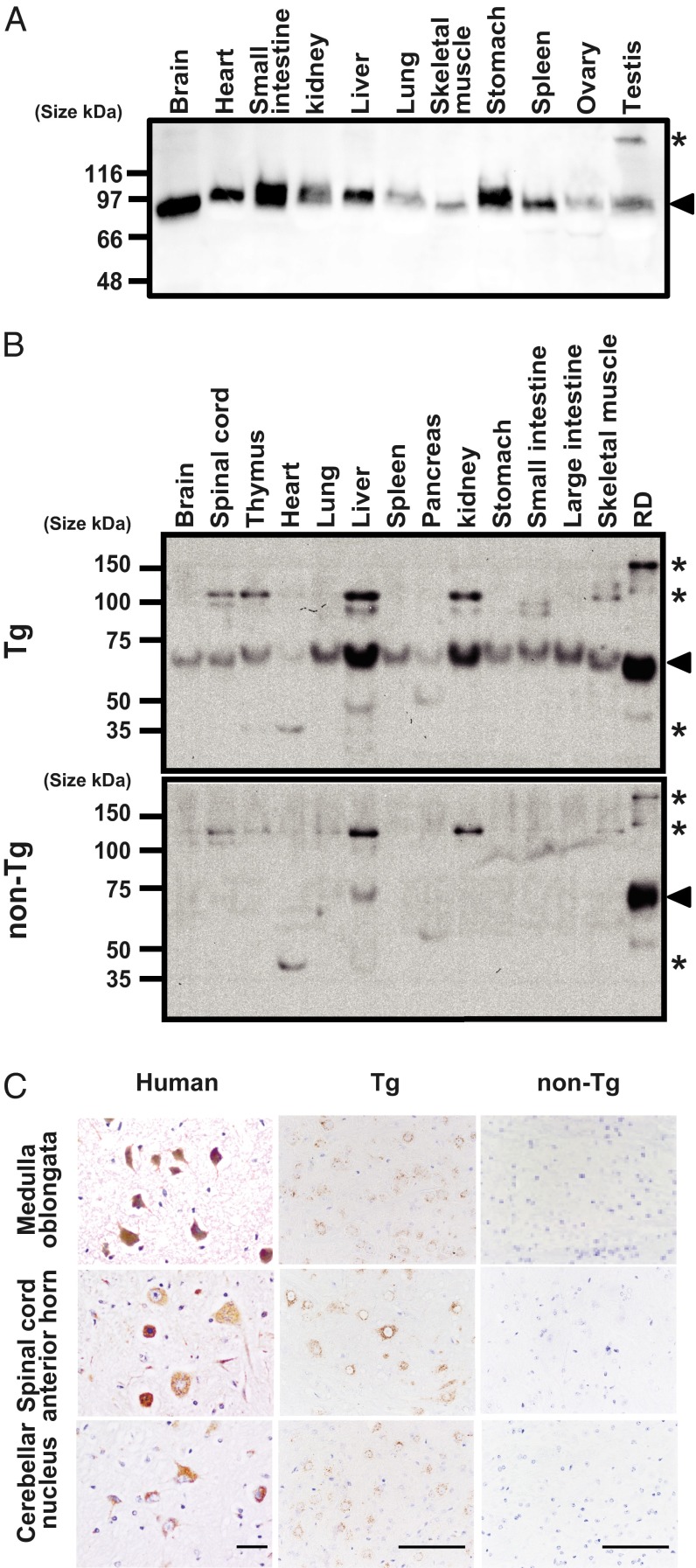
We performed Western blot analysis of human organ samples using a goat anti-hSCARB2 polyclonal antibody (R&D Systems). This antibody detected hSCARB2 in Western blots of human RD cells and cross-reacted weakly with mouse Scarb2 in mouse L929 cells (31). hSCARB2 signals with apparent molecular weights ranging from 66 to 97 kDa were detected in the Western blots of all tested human tissues (Fig. 1A), suggesting that hSCARB2 was expressed in all tissues. Western blot analysis showed marked expression of hSCARB2 in all tested organs of the Tg mouse (Fig. 1B). Together, these data suggest that the human transgene was expressed in all organs of the Tg mouse.
Fig. 1.
Expression profiles of hSCARB2 in human and hSCARB2-Tg10 mouse tissues. (A) Expression of hSCARB2 in various human tissues detected using an anti-hSCARB2 antibody. (B) Expression of hSCARB2 in various tissues in Tg and non-Tg mice. Tissues were homogenized and subjected to SDS/PAGE, and hSCARB2 was detected using an anti-hSCARB2 antibody. Arrowheads indicate the positions of hSCARB2 and mScarb2. Asterisks indicate nonspecific bands. (C) Immunohistochemical analysis of hSCARB2 expression in human, Tg, and non-Tg mouse CNS tissues stained with an anti-human SCARB2 antibody (brown) and counterstained with hematoxylin. (Scale bars, 50 and 100 μm in human and mouse tissue images, respectively.)
We also examined hSCARB2 expression by immunohistochemistry (IHC). The antibody used for Western blotting did not discriminate between endogenous mouse Scarb2 and hSCARB2 in paraffin sections. We therefore used a different anti-hSCARB2 antibody (Sigma) that does not cross-react with mouse Scarb2 in paraffin sections (Fig. 1C). These two antibodies produced indistinguishable staining profiles in human tissues (Fig. S2A). When this antibody was used for IHC, SCARB2 expression was observed in all human and Tg mouse tissues examined but not in non-Tg mouse tissues. Although the staining intensity of hSCARB2 varied among cell types, importantly, hSCARB2 was clearly detected in most CNS neurons in humans and Tg mice but not in non-Tg mice (Fig. 1C; Fig. S2B). The SCARB2 staining for other CNS cell types, such as glial cells and endothelial cells, was much lower in intensity than in neurons. High levels of hSCARB2 expression were also observed in lung pneumocytes, hepatocytes, renal tubular epithelium, splenic germinal centers, and intestinal epithelium (Fig. S2B). Similar expression patterns were observed in hSCARB2-Tg16 and 22 mice (Fig. S3). These observations suggest that the expression profile of hSCARB2, including its cell type specificity, is similar in Tg mice and humans.
Susceptibility of hSCARB2-Tg Mice to EV71 Infection.
The EV71 Isehara/Japan/99 (Isehara) strain causes paralysis and death in WT neonatal mice but not in those older than 3 wk of age. To assess the SCARB2-dependent susceptibility of the hSCARB2-Tg10 mice to EV71 infection, we first inoculated 3-wk-old Tg and non-Tg mice intracerebrally (i.c.) with EV71 at several different doses and observed for development of clinical signs. A number of the Tg mice inoculated with the Isehara strain at doses of 102–106 tissue culture infectious dose (TCID50) showed ataxia, paralysis, or death at days 4–14 after infection (Table 1; Fig. S4A). In contrast, no neurological manifestations were observed in non-Tg mice (Table 1). Tg mice older than 6 wk were also susceptible to EV71 infection, and the development of disease was similar to that of 3-wk-old mice (Fig. S4B). hSCARB2-Tg10 mice were also inoculated via i.v., i.p., and intragastric (i.g.) routes. Inoculation with EV71 doses greater than 102 and 103 TCID50 via the i.v. and i.p. routes, respectively, led to the development of neurological symptoms, although dose dependency was not clear (Table 1). When Tg mice were inoculated i.g. with EV71 at a dose of 106 or 107 TCID50 per animal, neurological signs were observed in 1 of 20 mice (Table 1). The other Tg mouse strains, hSCARB2-Tg16, 22, and 49 mice, were also susceptible to EV71 infection (Fig. S4 C and D). These results showed that adult Tg mice acquired susceptibility to EV71 through the expression of hSCARB2 and that EV71 is able to disseminate from the peripheral tissues to the CNS.
Table 1.
Incidence of neurological signs in mice after EV71 inoculation
| Inoculation route | Mouse | Viral inoculation dose (log10 TCID50/animal) |
|||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | ||
| i.c. | Non-Tg | — | — | — | — | — | 0/18* | — | — |
| Tg | 0/18 | 2/18 | 13/18 | 11/18 | 17/18 | 18/18 | — | — | |
| i.v. | Non-Tg | — | — | — | — | — | 0/18 | — | — |
| Tg | 0/18 | 7/18 | 4/18 | 4/18 | 4/18 | 16/18 | — | — | |
| i.p. | Non-Tg | — | — | — | — | — | 0/18 | — | — |
| Tg | — | 0/18 | 2/18 | 2/18 | 5/18 | 7/18 | — | — | |
| i.g. | Non-Tg | — | — | — | — | — | — | — | 0/10 |
| Tg | — | — | — | — | — | 1/20 | 1/20 | 0/10 | |
Three-week-old mice were inoculated i.c., i.v, i.p., or i.g. with the EV71 Isehara strain at the indicated dose. After viral inoculation, the mice were observed daily for 2 wk. —, not performed.
Number of mice showing ataxia, paralysis, and death/total number of inoculated mice.
Next, we inoculated 3-wk-old hSCARB2-Tg10 mice i.c. with several different strains of EV71 [BrCr/USA/70 (BrCr), SK-EV006/Malaysia/97 (SK-EV006), and C7/Japan/97 (C7) strains] and a Coxsackie virus A (CVA) 16 G-10 strain. These virus strains have been shown to use hSCARB2 as receptor (32), and it is known that CVA7, 14, and 16 also cause neurological diseases in humans, albeit infrequently (32, 38–40). Infection with the BrCr, SK-EV006, and C7 strains caused ataxia, paralysis, or death at days 4–10 after infection in some mice at more than 106, 103, and 104 TCID50, respectively, but some mice remained resistant to disease even after inoculation with the highest dose (106 TCID50; Table 2). Similarly, some of the Tg mice inoculated with CVA16 G-10 strain at more than 104 TCID50 also showed ataxia, paralysis, or death at days 7–10 after infection. The severity of the disease caused by these virus strains was similar to that caused by the Isehara strain, but much larger virus doses were required. These results suggest that Tg mice are susceptible to EV71 and related viruses that use SCARB2 as receptor and that the relative sensitivity to each virus strain was different.
Table 2.
Incidence of neurological signs in Tg mice after i.c. inoculation with EV71 and CVA16
| Virus | Strain | Viral inoculation dose (log10 TCID50/animal) |
||||
| 2 | 3 | 4 | 5 | 6 | ||
| EV71 | Isehara* | 2/18† | 13/18 | 11/18 | 17/18 | 18/18 |
| BrCr | 0/10 | 0/10 | 0/10 | 0/10 | 4/10 | |
| SK-EV006 | 0/10 | 2/10 | 3/10 | 3/10 | 3/10 | |
| C7 | 0/10 | 0/10 | 2/10 | 7/10 | 5/10 | |
| CVA16 | G-10 | 0/10 | 0/10 | 1/10 | 1/10 | 2/10 |
Three-week-old mice were inoculated i.c. with one of four EV71 strains or the CVA16 G-10 strain at the indicated dose. After viral inoculation, the mice were observed daily for 2 wk.
The results shown for mice infected with the Isehara strain are the same results as in Table 1.
Number of mice showing ataxia, paralysis, or death/total number of inoculated mice.
Viral Replication Sites of EV71 in Tg Mice.
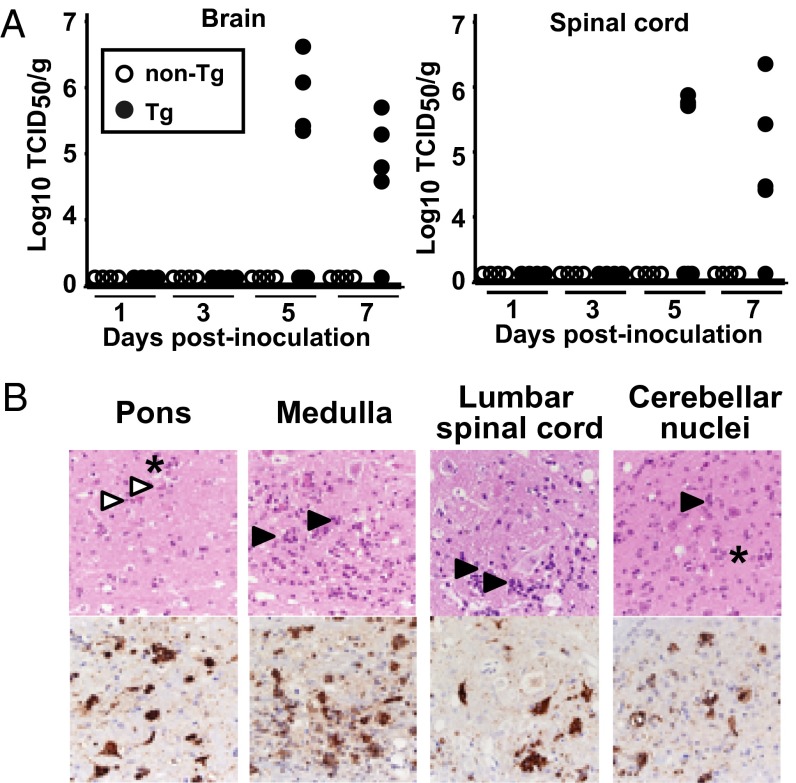
To identify the viral replication sites in 3-wk-old Tg mice, we measured the viral titers in major visceral organs at several time points after i.v. inoculation with EV71. The brains and spinal cords of symptomatic mice had high viral titers at days 5 and 7 after inoculation. The viral titers from asymptomatic Tg and non-Tg mice were below detectable levels (Fig. 2A). The viral titers in non-CNS organs were also below detection levels in all animals examined. These results suggest that the CNS is the major replication site for EV71 in hSCARB2-Tg10 mice, whereas replication in other organs remained limited.
Fig. 2.
Replication of EV71 in the CNS of hSCARB2-Tg10 mice. (A) Viral titers in the brain and spinal cord. After i.v. inoculation of the EV71 Isehara strain at a dose of 1 × 106 TCID50 in 3-wk-old animals (non-Tg; n = 4 in each time point, Tg; n = 4 at day 1 and day 3, n = 6 at day 5, n = 5 at day 7), the mice were euthanized at the indicated times, the tissues were collected, and viral titers were measured using the microtiter method. The open and closed circles show the viral titers in individual non-Tg and Tg mice, respectively. The viral titer detection limit is less than 103 TCID50/g (B) Histopathological changes and viral antigen expression in the CNS of 3-wk-old Tg mice inoculated i.v. with the EV71 Isehara strain (Table 1; Table S2). The pons, medulla, lumbar spinal cord, and cerebellar nuclei are shown (Upper, H&E staining; Lower, immunohistochemistry). Open arrowheads, closed arrowheads, and asterisks indicate degenerated neurons, neuronophagia, and gliosis, respectively.
Consistent with the virus isolation results (Fig. 2A), we observed pathological changes in the CNS of mice with clinical signs of infection (Fig. 2B; Fig. S5A; Table S2). For example, we observed cellular damage (such as degeneration, necrosis, and neuronophagia) and inflammatory changes (such as gliosis and perivascular cuffing) in the spinal cord, brainstem, hypothalamus, thalamus, cerebellum, cerebrum, and dorsal root ganglia. Viral antigens were also detected in the affected neurons in these CNS regions (Fig. 2B; Fig. S5A; Table S2), whereas other nonneuronal cell types were negative for viral antigens. We did not detect antigen-positive cells or pathological changes, such as inflammation and pulmonary edema, in regions outside the CNS in any of the mice (Table S2). None of the mice inoculated with EV71 demonstrated cutaneous lesions, regardless of inoculation route. Pathological changes and EV71 antigens in the neurons were also detected in hSCARB2-Tg16, 22, and 49 mice (Fig. S5B).
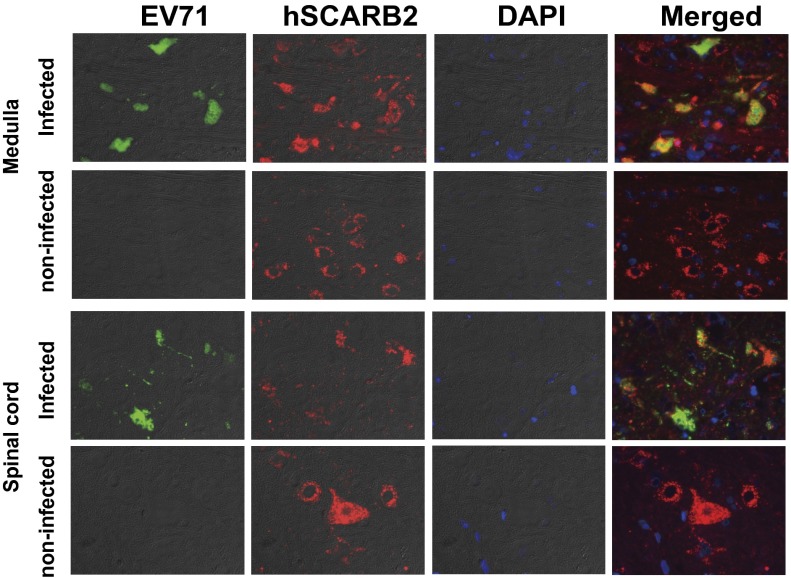
To confirm that EV71 infection in the neurons of Tg mice was dependent on hSCARB2 expression, we performed double immunofluorescence staining for hSCARB2 and viral antigen. In the medulla oblongata and spinal cord, all viral antigen-positive neurons expressed hSCARB2 (Fig. 3). These data suggest that the expression of hSCARB2 in neurons conferred susceptibility to EV71 infection and led to the neurological symptoms observed in the Tg mice.
Fig. 3.
EV71 infection in hSCARB2-expressing neurons in Tg mice. The medulla and spinal cord sections from a Tg mouse showing neurological signs (Table S2) were double-stained with an anti-EV71 antibody (green) and anti-hSCARB2 antibody (red). Nuclei were stained with DAPI (blue).
Viral replication sites in neonatal Tg mice were also examined by s.c. inoculation of 1-d-old non-Tg and Tg mice with the Isehara strain. As expected, infection with the Isehara strain caused paralysis in both non-Tg and Tg mice (Table S3). Viral antigens were detected in the CNS and skeletal muscle of both non-Tg and Tg mice (Table S3). Interestingly, EV71 antigens were also observed in the lungs, in some oral mucosal epithelium cells in the lip and a few skin epidermal cells on terminal parts of limbs, but only in Tg mice (Fig. S5C; Table S3). Viral antigens were not detected in the skin of the trunk. These results suggested that the replication of EV71 in these sites occurred in a SCARB2-dependent manner and that EV71 displays tropism to these cells in neonatal mice.
Discussion
Here, we demonstrated that the exogenous expression of hSCARB2 in mice was sufficient to confer susceptibility to EV71 infection and subsequent development of neuropathogenesis, which suggests that hSCARB2 also functions as a cellular receptor for EV71 infection in vivo. EV71 can cause different neurological manifestations in humans such as encephalomyelitis, polio-like flaccid paralysis, and meningitis. Although a certain manifestation can be prevalent during a given outbreak, various manifestations can be combined in a patient or different patients in the same outbreak may manifest differently (4, 7, 41–44). Despite these variations, inflammation and EV71 antigens have been invariably detected in the brainstem, spinal cord, hypothalamus, and cerebellar dentate nuclei in fatal human encephalomyelitis (5, 6), suggesting that EV71 has tropism to neurons found therein. In the cynomolgus monkey model, neurological manifestations were observed following i.v. and intraspinal EV71 inoculation (105.5–106 TCID50) (14, 15). Histological and viral examinations confirmed viral replication in the spinal cord, brainstem, cerebellar dentate nuclei, and cerebrum. Quantitative scoring of these lesions showed that the major foci of replication included the brainstem and dentate nuclei. In comparison, hSCARB2-Tg10 mice infected with EV71 via the i.c., i.v., i.p. or i.g. routes demonstrated paralysis and ataxia, and severe lesions and/or EV71 antigens were observed in the brainstem, spinal cord, hypothalamus, thalamus, cerebellar dentate nuclei, and Purkinje layer in most animals (Fig. 2B; Fig. S5A; Table S2). These results indicate that the spinal cord, brainstem, and cerebellar dentate nuclei in this Tg mouse model were most severely impacted by EV71 infection regardless of the inoculation route. Our findings highlighted the similarities in EV71 neurotropism observed in humans, monkeys, and Tg mice. Nonetheless, there were some differences between the Tg mice and humans. Compared with humans, Tg mice were less susceptible to oral infection. Adult Tg mice did not display the typical cutaneous lesions found in human HFMD. Pulmonary edema, which has been reported to be one of the most severe clinical outcomes of human EV71 infection, was not observed in the Tg mouse model. The reasons for these differences remain to be clarified.
In several laboratories, neonatal mice have been used as models for studying EV71 pathogenesis and for the development of vaccines and antiviral drugs (17, 18, 45–47). However, EV71 infection in the neonatal mouse model is significantly different from that in humans as the major viral replication sites in the mouse system include muscle and adipose tissues. EV71 infection in WT neonatal mice may be mediated by unknown molecule(s), and increased viral pathogenicity in neonatal mice may have resulted from adaptation of the virus to these molecules acting as receptors. It is possible to avoid this bias by using Tg mice that are more than 3 wk old. Our stocks of EV71 BrCr and C7 strains were not pathogenic to neonatal mice but were pathogenic to hSCARB2-Tg mice, suggesting that the viruses did not need to be adapted to rodents for infection to occur (Table 2). Furthermore, susceptibility was still observed in older mice. These results indicate that the hSCARB2-Tg mouse model can be used to overcome the problems associated with traditional neonatal mouse models.
The results in Tg mice suggested the existence of molecular mechanisms that determine EV71 tropism. Recently, another hSCARB2 Tg mouse model was reported (48) in which hSCARB2 was expressed ubiquitously using the human EF-1α promoter. This Tg mouse was susceptible to EV71 only up to 2 wk of age, and the main viral replication sites were in the muscle and the CNS. Therefore, the pathological features of this Tg mouse model were similar to those of the WT neonatal mouse model. In contrast, hSCARB2 expression in our hSCARB2-Tg10 mice older than 3 wk of age was similar to that in humans (Fig. 1C; Fig. S2B), and the majority of viral replication took place in CNS neurons (Fig. 2B; Fig. S5A). These results suggest that differences in virus susceptibility and tissue tropism may be strongly influenced by the expression pattern of hSCARB2. However, high levels of SCARB2 expression were also observed in many nonneural tissues, such as the lung, liver, kidney, and intestine of hSCARB2-Tg10 mice, in which EV71 replication was not observed. Together, these results suggest that accurate hSCARB2 expression is necessary for the acquisition of susceptibility but other, as yet unknown, factors could also contribute to efficient viral replication in non-CNS tissues.
The recent large outbreaks of EV71-related HFMD associated with severe neurological consequences represent a major public health concern, especially in the Asia-Pacific region. It can be speculated that circulating EV71 may further increase in neurovirulence. We tested four strains of EV71 and a CVA16 and found that the Isehara strain was associated with the most severe clinical signs among the strains tested. The BrCr, SK-EV006, and C7 strains and the CVA16 G-10 strain were associated with less severe signs, although their virulence levels could not be determined accurately because of the low susceptibility of the mice and poor dose-dependent response (Table 2). The Isehara strain, which was isolated from a HFMD patient, showed an apparent higher neurovirulence in the Tg mouse compared with the other EV71 strains (BrCr, SK-EV006, and C7) isolated from patients with neurological diseases. The CVA16 G-10 strain showed similar virulence level to BrCr (Table 2). One possible explanation for these paradoxical results could be the genetic changes that may have occurred in these viruses following serial passages in cell cultures after the initial isolation. In addition, severity of the human disease may be influenced by multiple factors including amount of virus ingested and the immunological condition and genetic background of the infected person. The neurovirulence level of each virus strain should be determined experimentally using the same background. The neurovirulence levels of these viruses in the monkey model have not been determined yet. It is important to investigate whether there is a correlation between the neurovirulence levels determined using the monkey and the Tg mouse models and to investigate if the virulence levels determined in these models reflect the virulence levels in humans.
In the process of establishing the PVR-Tg mouse model, an appropriate Tg mouse strain (PVR-Tg21) that showed good sensitivity and clear dose-dependent response was chosen (49, 50). In addition, many other investigations were performed, including correlation of neurovirulence levels of viruses in monkey and Tg mouse models (50, 51) and selection of suitable inoculation routes (52). Consequently, PVR-Tg mice became widely used to determine the neurovirulence levels of attenuated and neurovirulent strains of PV (50, 51, 53–55). Analogous to another PVR-Tg mouse strain that expressed PVR at low levels and therefore did not show good sensitivity or dose-dependent response (49), it should be possible to improve the sensitivity of our hSCARB2-Tg mice by developing a new strain with higher hSCARB2 expression levels. With similar efforts conducted for PVR-Tg mice research, our hSCARB2-Tg mice could be used to experimentally evaluate the neurovirulence of field isolates of EV71 with greater statistical significance in the future.
Materials and Methods
Ethics Statements.
Experiments using recombinant DNA and pathogens were approved by the Committee for Experiments using Recombinant DNA and Pathogens at the Tokyo Metropolitan Institute of Medical Science. Experiments using mice were approved by the Animal Use and Care Committee and performed in accordance with the Guidelines for the Care and Use of Animals (Tokyo Metropolitan Institute of Medical Science, 2011). All human histological samples used in this study were obtained ethically, and the protocols were approved by the relevant ethics committee of University of Malaya.
Cells and Viruses.
Human RD and African green monkey Vero cells were cultured in DMEM supplemented with 5% (vol/vol) FCS, l-glutamine, and penicillin-streptomycin solution [5% (vol/vol) FCS-DMEM]. The prototype strain of EV71, BrCr (genogroup A), was isolated from a patient with aseptic meningitis in 1970 (1). Two isolates, SK-EV006 (genogroup B) (14) and C7 (genogroup B) (56), were obtained from fatal encephalitis cases in 1997. Isehara (genogroup C) was isolated from a case of HFMD (57). The CVA16 G-10 strain was isolated in South Africa (58). We propagated these strains in RD cells.
Virus Purification.
RD cells were infected with EV71 and CVA16 G-10 at a multiplicity of infection (MOI) of 0.01. The cells and media were frozen at 48 h after infection. After thawing, the cell debris were removed by centrifugation at 10,000 × g for 20 min at room temperature. Subsequently, 100 mM NaCl, 2 mM EDTA, and 1% sarkosyl was added to the supernatant, and the supernatant was centrifuged at 14,000 × g for 2.5 h at room temperature. The pellet was resuspended in PBS and then centrifuged through 30% (wt/vol) sucrose at 14,000 × g overnight at room temperature. The viral pellet was suspended in 5% (vol/vol) FCS-DMEM.
Virus Titration.
The viral titers of stock viruses and infected mouse tissues were measured with a microtitration assay using Vero cells as previously described (28).
Mice.
To generate Tg mice expressing hSCARB2, two BACs carrying hSCARB2 genes, RP11-54D17 and RP11-628A4, were purchased from the Children’s Hospital Oakland Research Institute BACPAC Resources Center. The BAC clones were purified using a Large-Construct Kit (Qiagen) according to the manufacturer’s instructions and suspended in TE buffer (10 mM Tris⋅HCl and 0.1 mM EDTA, pH 7.5). The purified BAC clones were microinjected into the pronuclei of fertilized mouse eggs (ARK Resource) and then transplanted into pseudopregnant CD1 (ICR) mice (Charles River). The resulting mice were screened by PCR analysis using PCR primer sets (Table S1) for the hSCARB2 gene and genomic DNA extracted from tail biopsies as a template.
Western Blot.
To investigate the expression of SCARB2 in human tissues, we used a ready-for-use Western blot system with human tissue lysates (INSTA-Blot; Imgenex). To prepare the mouse organ protein samples, tissues (100 mg) was homogenized in 1 mL PBS containing a protease inhibitor mixture (Complete Mini; Roche) with a Polytron homogenizer. The protein concentrations in the samples were determined using a DC protein assay kit (Bio-Rad). The homogenates (5 µg of protein equivalents) were subjected to SDS/PAGE on 12% (wt/vol) gels (Bio-Rad), followed by transfer to the PVDF membrane (Millipore). After blocking, the membranes were incubated for 1 h with a goat anti-human SCARB2 antibody (R&D Systems), followed by incubation with an anti-goat HRP-conjugated antibody (Jackson ImmunoResearch) and SuperSignal West Pico Chemiluminescent Substrate (Thermo Scientific).
Infection of EV71 in Mice.
Mice (3 wk old or adults) were inoculated with the viruses at the indicated doses and routes and were observed for clinical signs (Tables 1 and 2; Table S2). To evaluate the pathogenesis of EV71 infection in neonatal mice, 1-d-old non-Tg or Tg10 mice were inoculated s.c. with EV71 (Fig. S5C; Table S3). To determine the viral titers in mouse tissues, 3-wk-old mice were inoculated i.v. with the Isehara strain at a dose of 1 × 106 TCID50. The infected mice were killed at the appropriate time after inoculation. Blood was collected from the heart, and the tissues were removed, frozen at −80 °C, thawed, and then homogenized in MEM containing 5% (vol/vol) FCS. After centrifugation for 10 min at 3,000 × g, the viral titer of the supernatant was determined in TCID50 per gram tissues. Mice were fixed with formalin-buffered saline by perfusion. The removed organs were subjected to IHC.
IHC.
Antigen retrieval of formalin-fixed human tissue sections was performed using a rice cooker (30 min, 96–98 °C, citrate buffer). Antigen retrieval of formalin-fixed mouse tissue sections was performed by autoclaving at 121 °C for 10 min in a retrieval solution of pH 6.0 (Nichirei) for the detection of viral antigens and a retrieval solution of pH 9.0 (Nichirei) for the detection of SCARB2 expression. SCARB2 and EV71 antigens were detected in a standard immunoperoxidase procedure using a rabbit anti-human SCARB2 antibody (SIGMA) and anti-EV71 antibody (14), respectively.
Double Immunofluorescence Staining.
Paraffin-embedded sections were stained to evaluate the expression of SCARB2 in virus antigen-positive cells. Following the retrieval reaction by autoclaving in a retrieval solution at pH 9.0, a goat anti-SCARB2 antibody (R&D Systems) and rabbit anti-EV71 antibody were incubated overnight at 4 °C. EV71 and SCARB2 were detected following incubation with Alexa Fluor 488 goat anti-rabbit IgG or Alexa Fluor 568 goat anti-mouse IgG (Molecular Probes) for 60 min at 37 °C, respectively. The sections were mounted in SlowFade Gold antifade reagent containing DAPI (Molecular Probes), and images were captured using a fluorescence microscope (IX71; Olympus) equipped with a Hamamatsu high-resolution digital B/W CCD camera (ORCA2; Hamamatsu Photonics).
Supplementary Material
Acknowledgments
We thank H. Shimizu for helpful discussions and critical review of the manuscript. We also thank A. Ohkubo, Y. Ichinokawa, A. Harashima, and M. Fujino for excellent technical assistance. This work was supported by Japan Society for the Promotion of Science KAKENHI Grants 23390116 and 23790518, Ministry of Education, Culture, Sports, Science and Technology, Japan KAKENHI Grant 24115006, and a Grant-in-Aid for Research on Emerging and Re-emerging Infectious Diseases from the Ministry of Health, Labor and Welfare of Japan. This study was also partly supported by High Impact Research Grant UM.C/625/1/HIR/MOHE/MED/06 from the University of Malaya.
Footnotes
The authors declare no conflict of interest.
This article is a PNAS Direct Submission. K.C. is a guest editor invited by the Editorial Board.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1217563110/-/DCSupplemental.
References
- 1.Schmidt NJ, Lennette EH, Ho HH. An apparently new enterovirus isolated from patients with disease of the central nervous system. J Infect Dis. 1974;129(3):304–309. doi: 10.1093/infdis/129.3.304. [DOI] [PubMed] [Google Scholar]
- 2. Pallansch M, Roos R (2007) Enteroviruses: Polioviruses, Coxsackieviruses, Echoviruses, and newer Enteroviruses. Fields Virology, eds Knipe DM et al. (Lippincott Williams & Wilkins, Philadelphia), 5th Ed, Vol 1, pp 839–893.
- 3.Ho M, et al. Taiwan Enterovirus Epidemic Working Group An epidemic of enterovirus 71 infection in Taiwan. N Engl J Med. 1999;341(13):929–935. doi: 10.1056/NEJM199909233411301. [DOI] [PubMed] [Google Scholar]
- 4.Huang CC, et al. Neurologic complications in children with enterovirus 71 infection. N Engl J Med. 1999;341(13):936–942. doi: 10.1056/NEJM199909233411302. [DOI] [PubMed] [Google Scholar]
- 5.Wong KT, et al. Enterovirus 71 encephalomyelitis and Japanese encephalitis can be distinguished by topographic distribution of inflammation and specific intraneuronal detection of viral antigen and RNA. Neuropathol Appl Neurobiol. 2012;38(5):443–453. doi: 10.1111/j.1365-2990.2011.01247.x. [DOI] [PubMed] [Google Scholar]
- 6.Wong KT, et al. The distribution of inflammation and virus in human enterovirus 71 encephalomyelitis suggests possible viral spread by neural pathways. J Neuropathol Exp Neurol. 2008;67(2):162–169. doi: 10.1097/nen.0b013e318163a990. [DOI] [PubMed] [Google Scholar]
- 7.Shindarov LM, et al. Epidemiological, clinical, and pathomorphological characteristics of epidemic poliomyelitis-like disease caused by enterovirus 71. J Hyg Epidemiol Microbiol Immunol. 1979;23(3):284–295. [PubMed] [Google Scholar]
- 8.Nagy G, Takátsy S, Kukán E, Mihály I, Dömök I. Virological diagnosis of enterovirus type 71 infections: Experiences gained during an epidemic of acute CNS diseases in Hungary in 1978. Arch Virol. 1982;71(3):217–227. doi: 10.1007/BF01314873. [DOI] [PubMed] [Google Scholar]
- 9.AbuBakar S, et al. Identification of enterovirus 71 isolates from an outbreak of hand, foot and mouth disease (HFMD) with fatal cases of encephalomyelitis in Malaysia. Virus Res. 1999;61(1):1–9. doi: 10.1016/s0168-1702(99)00019-2. [DOI] [PubMed] [Google Scholar]
- 10.Wang Y, et al. Hand, foot, and mouth disease in China: Patterns of spread and transmissibility. Epidemiology. 2011;22(6):781–792. doi: 10.1097/EDE.0b013e318231d67a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hashimoto I, Hagiwara A, Kodama H. Neurovirulence in cynomolgus monkeys of enterovirus 71 isolated from a patient with hand, foot and mouth disease. Arch Virol. 1978;56(3):257–261. doi: 10.1007/BF01317855. [DOI] [PubMed] [Google Scholar]
- 12.Hashimoto I, Hagiwara A. Studies on the pathogenesis of and propagation of enterovirus 71 in Poliomyelitis-like disease in monkeys. Acta Neuropathol. 1982;58(2):125–132. doi: 10.1007/BF00691653. [DOI] [PubMed] [Google Scholar]
- 13.Hashimoto I, Hagiwara A. Pathogenicity of a poliomyelitis-like disease in monkeys infected orally with enterovirus 71: A model for human infection. Neuropathol Appl Neurobiol. 1982;8(2):149–156. doi: 10.1111/j.1365-2990.1982.tb00269.x. [DOI] [PubMed] [Google Scholar]
- 14.Nagata N, et al. Pyramidal and extrapyramidal involvement in experimental infection of cynomolgus monkeys with enterovirus 71. J Med Virol. 2002;67(2):207–216. doi: 10.1002/jmv.2209. [DOI] [PubMed] [Google Scholar]
- 15.Nagata N, et al. Differential localization of neurons susceptible to enterovirus 71 and poliovirus type 1 in the central nervous system of cynomolgus monkeys after intravenous inoculation. J Gen Virol. 2004;85(Pt 10):2981–2989. doi: 10.1099/vir.0.79883-0. [DOI] [PubMed] [Google Scholar]
- 16.Chumakov M, et al. Enterovirus 71 isolated from cases of epidemic poliomyelitis-like disease in Bulgaria. Arch Virol. 1979;60(3-4):329–340. doi: 10.1007/BF01317504. [DOI] [PubMed] [Google Scholar]
- 17.Chen YC, et al. A murine oral enterovirus 71 infection model with central nervous system involvement. J Gen Virol. 2004;85(Pt 1):69–77. doi: 10.1099/vir.0.19423-0. [DOI] [PubMed] [Google Scholar]
- 18.Wang YF, et al. A mouse-adapted enterovirus 71 strain causes neurological disease in mice after oral infection. J Virol. 2004;78(15):7916–7924. doi: 10.1128/JVI.78.15.7916-7924.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wang W, et al. A mouse muscle-adapted enterovirus 71 strain with increased virulence in mice. Microbes Infect. 2011;13(10):862–870. doi: 10.1016/j.micinf.2011.04.004. [DOI] [PubMed] [Google Scholar]
- 20.Khong WX, et al. A non-mouse-adapted enterovirus 71 (EV71) strain exhibits neurotropism, causing neurological manifestations in a novel mouse model of EV71 infection. J Virol. 2012;86(4):2121–2131. doi: 10.1128/JVI.06103-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bergelson JM. Receptors. In: Ehrenfeld E, Domingo E, Roos RP, editors. The Picornaviruses. Washington, DC: ASM Press; 2010. pp. 73–86. [Google Scholar]
- 22.Ren RB, Costantini F, Gorgacz EJ, Lee JJ, Racaniello VR. Transgenic mice expressing a human poliovirus receptor: A new model for poliomyelitis. Cell. 1990;63(2):353–362. doi: 10.1016/0092-8674(90)90168-e. [DOI] [PubMed] [Google Scholar]
- 23.Koike S, et al. Transgenic mice susceptible to poliovirus. Proc Natl Acad Sci USA. 1991;88(3):951–955. doi: 10.1073/pnas.88.3.951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bartlett NW, et al. Mouse models of rhinovirus-induced disease and exacerbation of allergic airway inflammation. Nat Med. 2008;14(2):199–204. doi: 10.1038/nm1713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yang B, Chuang H, Yang KD. Sialylated glycans as receptor and inhibitor of enterovirus 71 infection to DLD-1 intestinal cells. Virol J. 2009;6:141. doi: 10.1186/1743-422X-6-141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Yang SL, Chou YT, Wu CN, Ho MS. Annexin II binds to capsid protein VP1 of enterovirus 71 and enhances viral infectivity. J Virol. 2011;85(22):11809–11820. doi: 10.1128/JVI.00297-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Nishimura Y, et al. Human P-selectin glycoprotein ligand-1 is a functional receptor for enterovirus 71. Nat Med. 2009;15(7):794–797. doi: 10.1038/nm.1961. [DOI] [PubMed] [Google Scholar]
- 28.Yamayoshi S, et al. Scavenger receptor B2 is a cellular receptor for enterovirus 71. Nat Med. 2009;15(7):798–801. doi: 10.1038/nm.1992. [DOI] [PubMed] [Google Scholar]
- 29.Kuronita T, et al. A role for the lysosomal membrane protein LGP85 in the biogenesis and maintenance of endosomal and lysosomal morphology. J Cell Sci. 2002;115(Pt 21):4117–4131. doi: 10.1242/jcs.00075. [DOI] [PubMed] [Google Scholar]
- 30.Reczek D, et al. LIMP-2 is a receptor for lysosomal mannose-6-phosphate-independent targeting of beta-glucocerebrosidase. Cell. 2007;131(4):770–783. doi: 10.1016/j.cell.2007.10.018. [DOI] [PubMed] [Google Scholar]
- 31.Yamayoshi S, Koike S. Identification of a human SCARB2 region that is important for enterovirus 71 binding and infection. J Virol. 2011;85(10):4937–4946. doi: 10.1128/JVI.02358-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Yamayoshi S, et al. Human SCARB2-dependent infection by coxsackievirus A7, A14, and A16 and enterovirus 71. J Virol. 2012;86(10):5686–5696. doi: 10.1128/JVI.00020-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yamayoshi S, Ohka S, Fujii K, Koike S. Functional comparison of SCARB2 and PSGL1 as receptors for enterovirus 71. J Virol. 2013;87(6):3335–3347. doi: 10.1128/JVI.02070-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tabuchi N, Akasaki K, Sasaki T, Kanda N, Tsuji H. Identification and characterization of a major lysosomal membrane glycoprotein, LGP85/LIMP II in mouse liver. J Biochem. 1997;122(4):756–763. doi: 10.1093/oxfordjournals.jbchem.a021820. [DOI] [PubMed] [Google Scholar]
- 35.Gamp AC, et al. LIMP-2/LGP85 deficiency causes ureteric pelvic junction obstruction, deafness and peripheral neuropathy in mice. Hum Mol Genet. 2003;12(6):631–646. [PubMed] [Google Scholar]
- 36.Ritsch A, et al. Molecular characterization of rabbit scavenger receptor class B types I and II: Portal to central vein gradient of expression in the liver. J Lipid Res. 2004;45(2):214–222. doi: 10.1194/jlr.M300353-JLR200. [DOI] [PubMed] [Google Scholar]
- 37.Liu J, et al. Transgenic expression of human P-selectin glycoprotein ligand-1 is not sufficient for enterovirus 71 infection in mice. Arch Virol. 2012;157(3):539–543. doi: 10.1007/s00705-011-1198-2. [DOI] [PubMed] [Google Scholar]
- 38.Ranzenhofer ER, Dizon FC, Lipton MM, Steigman AJ. Clinical paralytic poliomyelitis due to Coxsackie virus group A, type 7. N Engl J Med. 1958;259(4):182. doi: 10.1056/NEJM195807242590408. [DOI] [PubMed] [Google Scholar]
- 39.Helin I, Widell A, Borulf S, Walder M, Ulmsten U. Outbreak of coxsackievirus A-14 meningitis among newborns in a maternity hospital ward. Acta Paediatr Scand. 1987;76(2):234–238. doi: 10.1111/j.1651-2227.1987.tb10453.x. [DOI] [PubMed] [Google Scholar]
- 40.Xu W, et al. Distribution of enteroviruses in hospitalized children with hand, foot and mouth disease and relationship between pathogens and nervous system complications. Virol J. 2012;9(1):8. doi: 10.1186/1743-422X-9-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Samuda GM, Chang WK, Yeung CY, Tang PS. Monoplegia caused by Enterovirus 71: An outbreak in Hong Kong. Pediatr Infect Dis J. 1987;6(2):206–208. doi: 10.1097/00006454-198702000-00013. [DOI] [PubMed] [Google Scholar]
- 42.Melnick JL. Enterovirus type 71 infections: A varied clinical pattern sometimes mimicking paralytic poliomyelitis. Rev Infect Dis. 1984;6(Suppl 2):S387–S390. doi: 10.1093/clinids/6.supplement_2.s387. [DOI] [PubMed] [Google Scholar]
- 43.Lum LC, et al. Fatal enterovirus 71 encephalomyelitis. J Pediatr. 1998;133(6):795–798. doi: 10.1016/s0022-3476(98)70155-6. [DOI] [PubMed] [Google Scholar]
- 44.Wang SM, et al. Clinical spectrum of enterovirus 71 infection in children in southern Taiwan, with an emphasis on neurological complications. Clin Infect Dis. 1999;29(1):184–190. doi: 10.1086/520149. [DOI] [PubMed] [Google Scholar]
- 45.Dong C, et al. Immunoprotection elicited by an enterovirus type 71 experimental inactivated vaccine in mice and rhesus monkeys. Vaccine. 2011;29(37):6269–6275. doi: 10.1016/j.vaccine.2011.06.044. [DOI] [PubMed] [Google Scholar]
- 46.Chang JY, et al. Selection and characterization of vaccine strain for Enterovirus 71 vaccine development. Vaccine. 2012;30(4):703–711. doi: 10.1016/j.vaccine.2011.11.087. [DOI] [PubMed] [Google Scholar]
- 47.Ong KC, Devi S, Cardosa MJ, Wong KT. Formaldehyde-inactivated whole-virus vaccine protects a murine model of enterovirus 71 encephalomyelitis against disease. J Virol. 2010;84(1):661–665. doi: 10.1128/JVI.00999-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lin YW, et al. Human SCARB2 transgenic mice as an infectious animal model for enterovirus 71. PLoS ONE. 2013;8(2):e57591. doi: 10.1371/journal.pone.0057591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Koike S, et al. Characterization of three different transgenic mouse lines that carry human poliovirus receptor gene—influence of the transgene expression on pathogenesis. Arch Virol. 1994;139(3-4):351–363. doi: 10.1007/BF01310797. [DOI] [PubMed] [Google Scholar]
- 50.Abe S, et al. Studies on neurovirulence in poliovirus-sensitive transgenic mice and cynomolgus monkeys for the different temperature-sensitive viruses derived from the Sabin type 3 virus. Virology. 1995;210(1):160–166. doi: 10.1006/viro.1995.1327. [DOI] [PubMed] [Google Scholar]
- 51.Horie H, et al. Transgenic mice carrying the human poliovirus receptor: New animal models for study of poliovirus neurovirulence. J Virol. 1994;68(2):681–688. doi: 10.1128/jvi.68.2.681-688.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Abe S, et al. Neurovirulence test for oral live poliovaccines using poliovirus-sensitive transgenic mice. Virology. 1995;206(2):1075–1083. doi: 10.1006/viro.1995.1030. [DOI] [PubMed] [Google Scholar]
- 53.Dragunsky E, et al. Transgenic mice as an alternative to monkeys for neurovirulence testing of live oral poliovirus vaccine: Validation by a WHO collaborative study. Bull World Health Organ. 2003;81(4):251–260. [PMC free article] [PubMed] [Google Scholar]
- 54.Kew O, et al. Outbreak of poliomyelitis in Hispaniola associated with circulating type 1 vaccine-derived poliovirus. Science. 2002;296(5566):356–359. doi: 10.1126/science.1068284. [DOI] [PubMed] [Google Scholar]
- 55.Jegouic S, et al. Recombination between polioviruses and co-circulating Coxsackie A viruses: Role in the emergence of pathogenic vaccine-derived polioviruses. PLoS Pathog. 2009;5(5):e1000412. doi: 10.1371/journal.ppat.1000412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Shimizu H, et al. Enterovirus 71 from fatal and nonfatal cases of hand, foot and mouth disease epidemics in Malaysia, Japan and Taiwan in 1997-1998. Jpn J Infect Dis. 1999;52(1):12–15. [PubMed] [Google Scholar]
- 57.Shimizu H, et al. Molecular epidemiology of enterovirus 71 infection in the Western Pacific Region. Pediatr Int. 2004;46(2):231–235. doi: 10.1046/j.1442-200x.2004.01868.x. [DOI] [PubMed] [Google Scholar]
- 58.Sickles GM, Mutterer M, Feorino P, Plager H. Recently classified types of Coxsackie virus, group A: Behavior in tissue culture. Proc Soc Exp Biol Med. 1955;90(2):529–531. doi: 10.3181/00379727-90-22088. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.