Abstract
Physiological studies were conducted to determine the optimum cultural conditions for maximal carboxymethyl cellulase (CMCase) formation by Aspergillus terreus DSM 826. Shaking condition at 150 rpm is favorable for the production of CMCase from rice straw and sugar cane bagasse. The highest enzyme yield was obtained at the third day of incubation at 30 °C for both cases; however CMCase formation occurred at a broad range of pH values, with maximal formation of A. terreus DSM 826 CMCase at pH 4.5 and 5.0 when rice straw and sugar cane bagasse were used as sole carbon source, respectively. Carboxymethyl cellulose (CMC) was found to be a good inducer for CMCase formation in both agricultural wastes with CMC concentrations of 0.5 and 1.0 % (w/v) in case of rice straw and sugar cane bagasse, respectively. High level of enzyme formation was obtained with the addition of ammonium chloride as nitrogen source in both cases and at a concentration of 0.4 % (v/v Tween-80) as an addition to medium containing rice straw. However this addition did not influence the production of CMCase in case of using sugar cane bagasse as carbon source.
Keywords: Aspergillus terreus, carboxymethyl cellulase (CMCase), rice straw, sugar cane bagasse
INTRODUCTION
The bioconversion of cellulosic materials has been receiving a great attention in recent years and the development of large-scale bioconversion process would alleviate shortages of food and animal feeds, solve modern waste disposal problems and diminish the dependence on fossil fuels by providing an energy source in the form of glucose. Lignocellulolytic enzymes also have significant potential applications in agriculture and in various industries including chemicals, fuel, food, brewery, wine, animal feed, textile, laundry, pulp and paper (9). On the other hand, lignocelluloses are abundant source of renewable biopolymers in the world, which are continually replenished by photosynthetic reduction of carbon dioxide with the help of sunlight as energy source (1, 14). For this purpose research on the enzymatic degradation of cellulose and hemicellulose has been developed in the last years (17).
Enormous amounts of agricultural, industrial and municipal cellulosic wastes have been accumulating or used inefficiently due to the high cost of their utilization processes (7, 21, 24). In Egypt, more than 30 million tons of agricultural wastes are annually produced (42). Most of these materials are generally burnt in the field, resulting in low energy utilization efficiency (10 %) and environmental pollution (4, 6). On the other hand, food shortage, energy crisis, and environmental pollution are now major problems all over the world, which makes the utilization of cellulose quite urgent (4).
A number of fungi and bacteria capable of utilizing cellulose as a carbon source have been identified (5, 21). Cellulases produced by fungi such as the Aspergillus, Rhizopus and Trichoderma species have been extensively studied by several researchers (33, 41). Aspergillus sp. is known to produce a variety of cellulolytic enzymes including carboxymethyl cellulase (CMCase) or endo ß-1,4-glucanase (37), although several other fungi such as Penicillium funiculosum (30), Pestalotiopsis versicolor (40) have also been studied.
Cellulases are composed of a complex mixture of enzyme proteins with different specificities to hydrolyze glycosidic bonds. Cellulases can be divided into three major enzyme activity classes (15, 38, 39) including endoglucanases or endo-1, 4-ß-glucanase (EC 3.2.1.4), cellobio-hydrolase (EC 3.2.1.91) and ß-glucosidase (EC 3.2.1.21). Endo-glucanases, or carboxymethyl cellulases (CMCase), are proposed to initiate attack randomly at multiple internal sites in the amorphous regions of the cellulose fiber opening-up sites for subsequent attack by the cellobio-hydrolases (46). Cellobio-hydrolase, or an exoglucanase, is the major component of the fungal cellulase system accounting for 40–70 % of the total cellulase proteins and can hydrolyze highly crystalline cellulose (11). ß-glucosidase hydrolyze glucose dimmers and in some cases cellooligo-saccharides to glucose.
Previous studies reported that the agricultural wastes of lignocellulosics are used as a carbohydrate source to produce commercially important products such as ethanol, glucose and single cell protein (45). We previously described a comparison between cellulase activities of sixteen fungi utilizing corn stover as substrate (10). In the present work, physiological studies were made to determine the optimum cultural conditions for maximum production of CMCase by a filamentous fungus using rice straw or sugar cane bagasse as a sole carbon source.
METHODS
Microorganism
Aspergillus terreus DSM 826 was obtained from Deutsche Sammlung Von Mikroorganismen (Gottingen, Germany) and maintained on slants of modified solid Czapek-Dox’s medium (3) at 4°C.
Medium for carboxymethyl cellulase production
Aspergillus terreus DSM 826 used in this study was grown on modified Czapek-Dox’s liquid medium containing rice straw or sugar cane bagasse pith 2% as a carbon source for fungal growth. This medium contains (g/l): rice straw or sugar cane bagasse, 20.0; NaNO3, 2.0; KH2PO4, 1.0; MgSO4.7H2O, 0.5; KCl, 0.5. The medium was adjusted to an initial pH value of 5. The medium were sterilized by autoclaving at 1.5 atmospheres and 121°C for 20 min. Rice straw was brought from Ash-Sharqiyah Governorate fields, while sugar cane bagasse was brought from Al-Giza Governorate.
Cultivation of microorganism
Conidia were scrapped from mycelia, which were grown on slants for 7days at 30°C and suspended by hand shaking in sterile cold distilled water. Five mL aliquots of this suspension (1.3 x 104 CFU/ml) were used to inoculate, under aseptic conditions, 250mL Erlenmeyer flasks each containing 50mL of sterile medium. The flasks were incubated at 30°C for three days either statically or under shaking condition (150rpm) using shaker (New Brunswick scientific Co. Inc. Edison N. J. USA). At the end of the incubation period, the cultures were filtrated using Whatman No.1 filter paper. The culture filtrate was used directly for enzyme activity determination.
Pretreatment of lignocellulosic wastes
Rice straw and sugar cane bagasse were ground and soaked in NaOH (1%) solution (w/v) for 1 h at room temperature [100g waste /1L NaOH (1%) solution] in 3L conical flask. Wastes were then filtered and washed using running tap water until neutralization (pH 7), then washed with distilled water, dried in an oven at 60°C to be used as a carbon source in the basal salt medium.
Analytical methods
Protein content was estimated by Lowry et al. (25). Reducing sugars released were determined by the dinitrosalicylic acid method (DNS) (29).
Enzyme assay
Carboxymethyl cellulase (CMCase, Endo-1,4-P-D-glucanase, E.C.3.2.1.4) activity was determined according to the method described by Adsul et al. (2). The total reaction mixture of 1mL contained 0.5mL of 1% (w/v) CMC solution in citrate buffer (50mM, pH 4.8) and 0.5mL of the enzyme. The reaction mixture was incubated at 50°C for 30min. One unit of enzyme activity is defined as the amount of protein that catalyzed the formation of one µmol of glucose under the experimental conditions. All presented data are average values of triplicates. Statistical analysis was carried out according to the method described by Kenney and Keeping (20).
RESULTS
Effect of static and shaking conditions
Results shown in Table 1 indicated that CMCase was formed mainly under shaking condition in case of using rice straw or sugar cane bagasse as a carbon source. Table 1 shows that, shaking conditions are favorable for the production of CMCase from rice straw and sugar cane bagasse and the specific activities of CMCase were increased under shaking condition by 152 and 203%, respectively as compared with the corresponding static condition. In all the successive experiments, the culture was grown under shaking condition and the fermented medium (broth) was employed as the source of extracellular CMCase.
Table 1.
Effect of static and shaking conditions on the formation of CMCase by A. terreus DSM 826
| Aeration condition | Rice straw | Sugar cane bagasse | ||
|---|---|---|---|---|
| CMCase activity (U/ml) | Specific activity (U/mg protein) | CMCase activity (U/ml) | Specific activity (U/mg protein) | |
| Static | 1.67 ± 0.334 | 0.56 ± 0.012 | 1.00 ± 0.056 | 0.84 ± 0.044 |
| Shaking | 2.36 ± 0.000 | 0.85 ± 0.003 | 2.28 ± 0.042 | 1.71 ± 0.015 |
Effect of incubation period
A. terreus DSM 826 was grown on a medium containing rice straw or sugar cane bagasse as carbon source at pH 5 for 8 days. Samples of the fermented medium were collected daily for the determination of CMCase activity. Results cited in Figure 1 illustrated that the highest specific activity was obtained at the third day of growth in both cases then the enzyme level began to decrease. In all the following experiments, the cultures were harvested at the third day of incubation at which CMCase formation reaches its maximum value.
Figure 1.
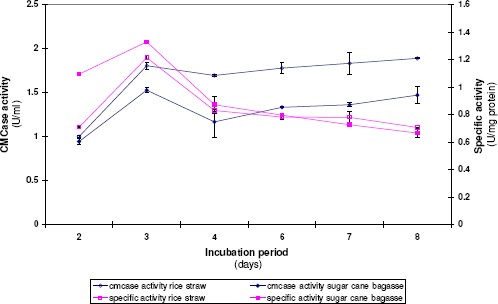
Effect of incubation period on CMCase formation by A. terreus DSM 826
Effect of inoculum size
Inocula with size ratios ranging from 5% to 25% of the total cultural volume were used. Results in Figure 2 indicated that as the percentage of the introduced inoculum size (1.3 x 104 CFU/ml) was raised from 5 to 20%, CMCase progressively increased with rice straw as a carbon source. With regard to sugar cane bagasse, the optimal percent of inoculum size was found to be 10 %. Further increase in inoculum size reduced the level of the formed enzyme.
Figure 2.
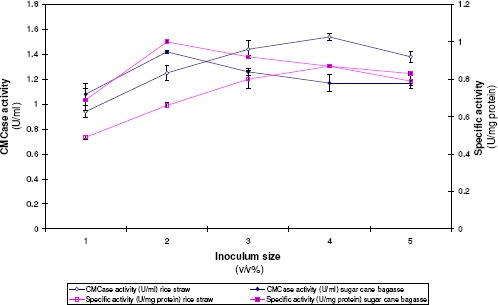
Effect of inoculum size on CMCase formation by A. terreus DSM 826
Effect of incubation temperature
A. terreus DSM 826 was grown at various degrees of temperatures ranging from 25 to 60°C. Results cited in Figure 3 demonstrated that A. terreus DSM 826 was able to grow and produce CMCase within a broad range of incubation temperatures from 25 to 50 °C. The optimal temperature for CMCase formation was found to be 30 °C with both rice straw and sugar cane bagasse. Results also indicated that, by increasing the incubation temperature above 30 °C, a gradual decrease in CMCase formation occurred until 50 °C. No growth was obtained at 60 °C and subsequently no enzyme formation could be detected at that degree of temperature.
Figure 3.
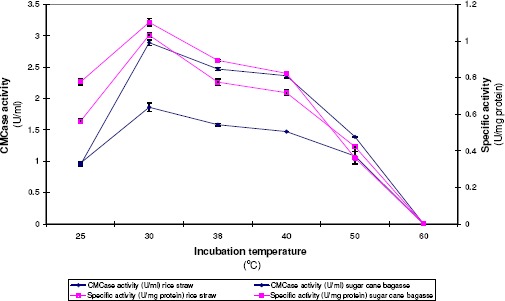
Effect of incubation temperature on CMCase production by A. terreus DSM 826
Influence of pH values
Seven pH values ranging from 3.0 to 8.0 were selected for this study. Results obtained are shown in Table 2. It was clear that maximal formation of A. terreus DSM 826 CMCase took place at pH 4.5 when rice straw was used as a sole carbon source wherein CMCase formation occurred at a broad range of pH values. In case of using sugar cane bagasse, optimal formation of CMCase occurred at pH 5, whereas considerably low levels of enzyme were obtained at pH values of 7 and 8.
Table 2.
Influence of different pH values on the formation of A. terreus DSM 826 CMCase
| Initial pH | Rice straw | Sugar cane bagasse | ||||
|---|---|---|---|---|---|---|
| Final pH | CMCase activity (U/ml) | Specific activity (U/mg protein) | Final pH | CMCase activity (U/ml) | Specific activity (U/mg protein) | |
| 3.0 | 5.0 | 1.67 ± 0.028 | 0.70 ± 0.104 | 6.0 | 1.14 ± 0.014 | 0.62 ± 0.005 |
| 4.0 | 6.5 | 1.70 ± 0.069 | 0.71 ± 0.034 | 6.5 | 1.19 ± 0.000 | 0.68 ± 0.037 |
| 4.5 | 6.5 | 1.70 ± 0.014 | 0.77 ± 0.075 | 6.5 | 1.22 ± 0.014 | 0.74 ± 0.013 |
| 5.0 | 6.0 | 1.67 ± 0.056 | 0.71 ± 0.023 | 6.5 | 1.25 ± 0.097 | 0.85 ± 0.064 |
| 6.0 | 7.0 | 1.56 ± 0.056 | 0.68 ± 0.061 | 7.0 | 1.00 ± 0.014 | 0.65 ± 0.009 |
| 7.0 | 7.0 | 1.14 ± 0.014 | 0.64 ± 0.004 | 7.0 | 0.77 ± 0.000 | 0.30 ± 0.000 |
| 8.0 | 7.0 | 1.06 ± 0.069 | 0.51 ± 0.002 | 7.0 | 0.22 ± 0.139 | 0.13 ± 0.036 |
Effect of agitation speed
This experiment was carried out to investigate the effect of agitation speed on enzyme formation. A. terreus DSM 826 cultures were grown on rice straw or sugar cane bagasse under shaking conditions at different speed levels ranging from 50 to 200rpm. Results in Figure 4 demonstrated that increasing agitation speed up to 150 rpm enhanced CMCase formation for both wastes. Further increase in shaking speed reflected on reducing the level of the formed enzyme. Consequently, shaking condition at an agitation speed of 150rpm was optimal for CMCase formation and used in the following experiments.
Figure 4.
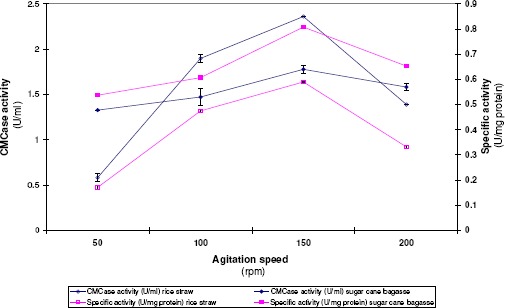
Effect of agitation speed on CMCase formation by A. terreus DSM 826
Effect of additive carbon sources
Different carbon sources were added at a concentration of 1% with rice straw or sugar cane bagasse (2%) as the main carbon source. As shown in Table 3 CMC was found to be the only inducer for CMCase formation in both agricultural wastes. Alternatively, D-glucose, D-mannose, maltose, sucrose and cellobiose significantly repressed CMCase formation in case of using rice straw and sugar cane bagasse as carbon sources.
Table 3.
Effect of the addition of some carbon sources on CMCase formation by A. terreus DSM 826.
| Additive carbon source (1%) | Rice straw | Sugar cane bagasse | ||
|---|---|---|---|---|
| CMCase activity (U/ml) | Specific activity (U/mg protein) | CMCase activity (U/ml) | Specific activity (U/mg protein) | |
| Control | 1.68 ± 0.014 | 0.87 ± 0.042 | 1.24 ± 0.014 | 0.92 ± 0.011 |
| D-Glucose | 0.78 ± 0.084 | 0.39 ± 0.042 | 0.74 ± 0.070 | 0.52 ± 0.027 |
| D-Mannose | 0.96 ± 0.014 | 0.39 ± 0.006 | 0.89 ± 0.028 | 0.58 ± 0.018 |
| Maltose | 0.90 ± 0.097 | 0.45 ± 0.049 | 1.01 ± 0.014 | 0.68 ± 0.010 |
| Sucrose | 0.70 ± 0.056 | 0.25 ± 0.021 | 0.60 ± 0.014 | 0.47 ± 0.011 |
| Cellobiose | 0.69 ± 0.000 | 0.37 ± 0.000 | 0.49 ±0.014 | 0.33 ± 0.010 |
| CMC | 1.76 ± 0.070 | 0.98 ± 0.040 | 2.03 ± 0.056 | 1.36 ± 0.038 |
CMCase formation as a function of CMC concentrations
The influences of different concentrations of CMC on CMCase activity were shown in Figure 5. CMC (0.25–2 %) was amended to the culture medium of rice straw or sugar cane bagasse as an inducing and dispersing agent. Results obtained indicate that the enzyme activity increased by about 17% and 82% in case of amending the culture medium containing rice straw and sugar cane bagasse by 0.5% and 1% CMC respectively. No further effect on enzyme level was observed by increasing CMC concentration up to 2%.
Figure 5.
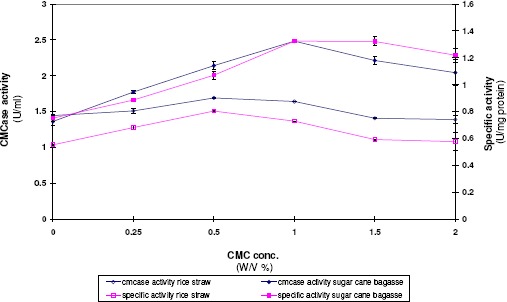
CMCase formation as a function of CMC concentration in the culture medium of A. terreus DSM 826
Effect of the addition of various nitrogen sources
Various nitrogen compounds were added to the culture medium in amounts equivalent (on nitrogen basis) to the amount of nitrogen in sodium nitrate in Czapek-Dox’s medium. Table 4 demonstrated that A. terreus DSM 826 can grow well on the all tested additive nitrogen sources. It was obvious also from the results that the highest level of enzyme formation expressed in terms of specific activity was obtained with ammonium chloride with both rice straw and sugar cane bagasse. Other nitrogen sources namely ammonium sulphate, potassium nitrate and ammonium oxalate gave also considerable amounts of CMCase with both wastes as compared with sodium nitrate (control).
Table 4.
Effect of the addition various nitrogen sources to the medium on the CMCase formation by A. terreus DSM 826
| Nitrogen source | Rice straw | Sugar cane bagasse | ||
|---|---|---|---|---|
| CMCase activity (U/ml) | Specific activity (U/mg protein) | CMCase activity (U/ml) | Specific activity (U/mg protein) | |
| NaNO3 (control) | 1.67 ± 0.190 | 0.81 ± 0.058 | 1.33 ± 0.070 | 0.94 ± 0.017 |
| NH4Cl | 2.25 ± 0.190 | 1.40 ± 0.051 | 1.03 ± 0.000 | 1.10 ± 0.000 |
| (NH4)2SO4 | 1.81 ± 0.475 | 1.08 ± 0.142 | 0.83 ± 0.010 | 0.78 ± 0.002 |
| KNO3 | 1.86 ± 0.000 | 0.99 ± 0.001 | 1.31 ± 0.000 | 0.92 ± 0.002 |
| (NH4)2C2O4.H2O | 2.64 ± 0.127 | 0.84 ± 0.028 | 1.50 ± 0.060 | 0.83 ± 0.018 |
Effect of the addition of Tween-80
A. terreus DSM 826 was grown on modified Czapek-Dox’s liquid medium supplemented with different concentrations of Tween-80 (0.1–0.5 %, v/v) to define the optimal concentration needed to induce maximum CMCase yield. The flasks were incubated at shaking condition (150 rpm) for 3 days at 30 °C. The data obtained were recorded in Table 5, which showed that the enzyme production expressed as relative activity was increased by increasing Tween-80 concentrations to a definite limit till reaching its maximum value at a concentration of 0.4% (v/v Tween-80) for rice straw. However in case of sugar cane bagasse the addition of Tween-80 did not influence CMCase production. The results obtained also indicate that the CMCase relative activity in case of rice straw increased by about 78% than control. In addition, it was found that increasing Tween-80 concentrations above these values resulted in a gradual decrease in CMCase formation for both agricultural wastes.
Table 5.
Effect of different concentrations of Tween-80 on CMCase formation by A. terreus DSM 826.
| Tween-80 (v/v %) | Relative activity (%) | |
|---|---|---|
| Rice straw | Sugar cane bagasse | |
| Control | 100 ± 0.056 | 100 ± 0.052 |
| 0.1 | 128 ± 0.040 | 103 ± 0.042 |
| 0.2 | 133 ± 0.052 | 94 ± 0.052 |
| 0.4 | 178 ± 0.000 | 92 ± 0.030 |
| 0.5 | 167 ± 0.034 | 80 ± 0.042 |
DISCUSSION
Several studies on physiological aspects of CMCase production were carried out using submerged culture among them are Chang et al. (4), Ikram-ul-Hag et al. (18), Muthuvelayudham and Viruthagiri (32) and Kansoh et al. (19). In the present work, the maximum production of CMCase was recorded after 3 days of incubation in both cases of rice straw and sugar cane bagasse, after that a gradual decrease in enzyme level was observed. This result can be interpreted by the depletion of macro and micronutrients in the fermentation medium with the lapse in time, which stressed the fungal physiology resulting in the inactivation of secretary machine of the enzyme. This result was similar to that reported by Chang et al. (4) with Aspergillus glaucus and Fadel (12) with A. niger. On the other hand, Silva et al. (44) reported the maximum enzyme level was obtained at the 4th day of incubation.
The results obtained in the present work showed that 20 % and 10 % inoculum size (1.3 x 104 CFU/ml) were the optimum for CMCase production from rice straw and sugar cane bagasse, respectively. Sharma et al. (43) reported that inoculum size controls and shortens the initial lag phase, as smaller inoculum size increased the lag phase. When the inoculum’s ratio was lower, superfluous nutrients resulted in the excessive growth of mycelia and delayed enzyme production. However, insufficient nutrients and increasing temperatures in the later period resulting from the rapid growth of mycelia inhibited enzyme production through a higher inoculum’s ratio. In agreement with our results, Fadel (12) reported that maximum CMCase production was obtained with inoculum size of 10 % from A. niger F.119. On the other hand, Chang et al. (4) reported that maximal production of CMCase occurred with the inoculum ratio of 6% of Aspergillus glaucus XC9.
Optimum cultivation temperature depends on the growth kinetics of the microorganism employed rather than on the enzyme produced (22, 26). In this study optimum CMCase production was recorded at 30ºC. Above 30ºC, a gradual decrease in enzyme formation was occurred. This can be interpreted by the alteration of cell membrane composition and stimulation of protein catabolism. Many authors also reported that 30 ºC was the best for CMCase production by Penicillium citrinum (36), consortium of A. niger and T. viride (18), A. glaucus XC9 (4), T. reesei NRRL 3653 (19). On the other hand, Thermoascus auranticus showed their maximum CMCase production at 50ºC (44).
In the present investigation, the maximal formation of A. terreus DSM 826 CMCase took place at pH 4.5 and 5.0 when rice straw and sugar cane bagasse were used as a sole carbon source in the medium, respectively. Most of fungal cultures prefer a slightly acidic pH in the medium for growth and enzyme biosynthesis (16) in agreement with the results obtained. In the literature optimum CMCase production was obtained with Trichoderma reesei strain QM-9414 (pH 3.5) (23), Rhodotorula glutinis and A. terreus (pH 5.0) (8, 35) and Aspergillus glaucus (pH 5.5) (4). Increasing agitation speed of the shake culture medium up to 150rpm enhanced CMCase formation in both cases. This can be explained by the fact that agitation increased the dissolved oxygen in the medium and uniform distribution of the medium contents such as foodstuffs and catabolites. The accumulation of these compounds and products in the medium contributes to large extent in the process of catabolite repression (13). This observation was in accordance with the results of Wood et al. (47). Mountfort and Asher (31) reported that CMCase formation was improved by shaking the culture media of the anaerobe Neocallimastix frontalis PN-1.
The obtained results showed that CMC was found to be the only inducer for CMCase formation. Addition of D-glucose, D-mannose, maltose, sucrose and cellobiose significantly repressed CMCase formation. Similarly, CMC also stimulated the highest yield of cellulase in the case of a consortium of A. niger and T. viride (18), Penicillium citrinum (36), and A. niger (34). In this connection Chellapandi and Himanshu reported that the extracellular endoglucanase synthesis is cyclic dependent and therefore subject to catabolite repression by soluble sugar accumulation (5). CMCase formation was increased with increasing CMC concentration up to 0.5 and 1.0% in case of rice straw and sugar cane bagasse, then a gradual decrease take place. This is probably due to the high viscosity of the medium, which decreases the oxygen supply to the cells. High viscosity leads to retard cell division, resulted in low production metabolism and cellulase extraction (13) in agreement with our results.
The obtained data revealed that the best inorganic additive nitrogen compounds stimulated CMCase production was found to be ammonium chloride; whileas ammonium sulphate was recorded in case of Pleurotus dryinus, Aspergillus niger, and Aspergillus glaucus (4, 9, 12). In case of Bacillus subtilis the rate of CMCase production increased with the addition of sodium nitrate to the medium followed by ammonium sulphate (22).
In this study, CMCase expressed as relative activity was increased by increasing Tween 80 concentrations till reaching its maximum value at 0.4% (v/v) for rice straw, however in case of sugar cane bagasse, the increase of Tween 80 led to a gradual decrease of cellulase production (Table5). The stimulatory effect of surfactants may be a consequence of its action on cell membranes causing increased permeability and/or by promoting the release of cell-bound enzymes and increase cellulase stability and prevent denaturizing of enzyme by desorbing it from cellulose substrate (48). In accordance with our results in case of using rice straw, Tween 80 also induced CMCase production by Aspergillus glaucus, Trichoderma viride (4, 27). On the other hand, Micales (28) reported that the presence of Tween-80 did not influence the production or secretion of CMCase of Postia placenta.
REFERENCES
- 1.Adsul M.G., Bastawde K.B., Varma A.J., Gokhale D.V. Strain improvement of Penicillium janthinellum NCIM 1171 for increased cellulase production. Bioresour. Technol. 2007;98:1467–1473. doi: 10.1016/j.biortech.2006.02.036. [DOI] [PubMed] [Google Scholar]
- 2.Adsul M.G., Ghule J.E., Singh R., Shaikh H., Bastawde K.B., Gokhale D.V., Varma A.J. Polysaccharides from bagasse: applications in cellulase and xylanase production. Carbohydr. Polymers. 2004;57:67–72. [Google Scholar]
- 3.Anon A.O. 9. Difco Laboratories, Detroit, Michigan, USA: 1972. Difco Manual of dehydrated culture media and reagents for microbiological and clinical laboratories procedures; pp. 212–231. [Google Scholar]
- 4.Chang X., Minnan L., Xiaobing W., Huijuan X., Zhongan C., Fengzhang Z., Liangshu X. Screening and characterization of the high cellulase producing strain Aspergillus glaucus XC9. Front. Biol. China. 2006;1:35–40. [Google Scholar]
- 5.Chellapandi P., Himanshu M.J. Production of endoglucanase by the native strains of Streptomyces isolates in submerged fermentation. Braz. J. Microbiol. 2008;39:122–127. doi: 10.1590/S1517-838220080001000026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cheng H.Z., Li Z.H. Microorganisms grown on cellulosic materials and utilization of biomass Sci. Technol. Mark. Chem. 2001;5:17–20. [Google Scholar]
- 7.Cunha-Santino M.B., Bianchini Jr. I. Cellulase activities during decomposition of a submerged aquatic macrophyte (Utricularia breviscapa): A microcosm assay. Braz. J. Microbiol. 2007;38:230–236. [Google Scholar]
- 8.D’Souza J., Volfava O. The effect of pH on the production of cellulases in Aspergillus terreus. Eur. J. Appl. Microbiol. 1982;16:123–125. [Google Scholar]
- 9.Elisashvili V., Penninckx M., Kachlishvili E., Asatiani M., Kvesitadze G. Use of Pleurotus dryinus for lingocellulolytic enzymes production in submerged fermentation of mandarin peels and tree leaves. Enzyme Microb. Technol. 2006;38:998–1004. [Google Scholar]
- 10.Elshafei A.M., Vega J.L., Klasson K.T., Clausen E.C., Gaddy J.L. Cellulase and hemicellulase formation by fungi using corn stover as the substrate. Biol. Wastes. 1990;32:209–218. [Google Scholar]
- 11.Esterbauer H., Steiner W., Labudova I. Production of Trichoderma cellulase in laboratory and pilot scale. Bioresour. Technol. 1991;36:51–65. [Google Scholar]
- 12.Fadel M. Production Physiology of Cellulases and β-glucosidase Enzymes of Aspergillus niger Grown under Solid State Fermentation Conditions. J. Biol. Sci. 2000;1(5):401–411. [Google Scholar]
- 13.Fritsche W. 2. Germany: Spektrum Akademischer Verlag; 1999. Mikrobiologie. [Google Scholar]
- 14.Fan L.T., Gharpuray M.M., Lee Y.H. Spinger-Verlag, Berlin, Germany: 1987. Cellulose hydrolysis; pp. 1–68. [Google Scholar]
- 15.Goyal A., Ghosh B., Eveleigh D. Characterization of fungal cellulases. Bioresour. Technol. 1991;36:37–50. [Google Scholar]
- 16.Haltrich D., Nidetzky B., Kulbe K.D., Steiner W., Zupancic S. Production of fungal xylanases. Bioresour. Technol. 1996;58:137–161. [Google Scholar]
- 17.Heck J.X., Hertz P.F., Ayub M.A.Z. Cellulase and xylanase production by isolated Amazon Bacillus strains using soybean industrial residue based solid-state cultivation. Braz. J. Microbiol. 2002;33:213–218. [Google Scholar]
- 18.Ikram-ul-Hag, Javed M.M., Khan T.S. An innovative approach for hyperproduction of cellulolytic and hemicellulolytic enzymes by consortium of Aspergillus niger MSK-7 and Trichoderma viride MSK-10. Afr. J. Biotechnol. 2006;5(8):609–614. [Google Scholar]
- 19.Kansoh A.L., Essam S.A., Zeinat A.N. Biodegradation and utilization of bagasse with Trichoderma reesei. Polym. Degrad. Stabil. 1999;62:273–278. [Google Scholar]
- 20.Kenney J.F., Keeping E.S. 3. Princeton, NJ, Van Nostrand: 1962. The standard deviation and calculation of the standard deviation” In Mathematics of statistics; pp. 77–80. [Google Scholar]
- 21.Kim K.C., Yoo S.S., Oh Y.A., Kim S.J. Isolation and characteristics of Trichoderma harzianum FJ1 producing cellulases and xylanase. J. Microbiol. Biotechnol. 2003;13:1–8. [Google Scholar]
- 22.Krishna C. Production of bacterial cellulases by solid state bioprocessing of banana wastes. Bioresour. Technol. 1999;69:231–239. [Google Scholar]
- 23.Krishna S.H., Rao K.C., Babu J.S., Reddy D.S. Studies on the production and application of cellulase from Trichoderma reesei QM-9414. Biop. Eng. 2000;22:467–470. [Google Scholar]
- 24.Lee Y.J., Kim B.K., Lee B.H., Jo K.I., Lee N.K., Chung C.H., Lee Y.C., Lee J.W. Purification and characterization of cellulase produced by Bacillus amyloliquefaciens DL-3 utilizing rice hull. Bioresour. Technol. 2008;99(2):378–386. doi: 10.1016/j.biortech.2006.12.013. [DOI] [PubMed] [Google Scholar]
- 25.Lowry O.H., Rosebrough N.J., Farr A.L., Randall R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951;193:265–275. [PubMed] [Google Scholar]
- 26.Lonsane B.K., Ghild N.P., Budeatman S., Kamakrishna S.V. Engineering aspects of solid state fermentation. Enzyme Microb. Technol. 1985;7:258–265. [Google Scholar]
- 27.Liu J., Yuan X., Zeng G., Shi J., Chen S. Effect of biosurfactant on cellulase and xylanase production by Trichoderma viride in solid substrate fermentation. Process. Biochem. 2006;41:2347–2351. [Google Scholar]
- 28.Micales J.A. Berlin: 1991. Increased recovery of ß-D-glucosidase from Postia placenta in presence of tween surfactants. Sonderdruck aus: material und organismen 26. Bd. 1991 Heft 1, Verlag Duncker and Humblot, 1000; p. 41. [Google Scholar]
- 29.Miller G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 1959;31:426–428. [Google Scholar]
- 30.Mishra C., Rao M., Seeta R., Srinivasan M.C., Deshpande V. Hydrolysis of lignocelluloses by Penicillium funiculosum cellulase. Biotechnol. Bioeng. 1985;26:370–373. doi: 10.1002/bit.260260412. [DOI] [PubMed] [Google Scholar]
- 31.Mountfort D.O., Asher R.A. Production and regulation of cellulase by two strains of the rumen anaerobic fungus Neocallimastix frontalis. Appl. Environ. Microbiol. 1985;49:1314–1322. doi: 10.1128/aem.49.5.1314-1322.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Muthuvelayudham R., Viruthagiri T. Fermentative production and kinetics of cellulase protein on Trichoderma reesei using sugarcane bagasse and rice straw. Afr. J. Biotechnol. 2006;5(20):1873–1881. [Google Scholar]
- 33.Murashima K., Nishimura T., Nakamura Y., Koga J., Moriya T., Sumida N., Yaguchi T., Kono T. Purification and characterization of new endo-1, 4- β -D- glucanases from Rhizopus oryzae. Enzyme Microb. Technol. 2002;30:319–326. [Google Scholar]
- 34.Narasimha G., Sridevi A., Buddolla V., Subhosh C.M., Rajasekhar R.B. Nutrient effects on production of cellulolytic enzymes by Aspergillus niger. Afr. J. Biotechnol. 2006;5(5):472–476. [Google Scholar]
- 35.Oikawa T., Tsukagawa Y., Soda K. Endo-ß-glucanase secreted by a psychrotrophic yeast: Purification and characterization. Biotechnol. Biochem. 1998;62:1751–1756. doi: 10.1271/bbb.62.1751. [DOI] [PubMed] [Google Scholar]
- 36.Olutiola P.O. Cellulase complex in culture filtrates of Penicillium citrinum. Can. J. Microbiol. 1976;22:1153–1159. doi: 10.1139/m76-167. [DOI] [PubMed] [Google Scholar]
- 37.Onsori H., Zamani M.R., Motallebi M., Zarghami N. Identification of over producer strain of endo-P-1,4-glucanase in Aspergillus Species: Characterization of crude carboxymethyl cellulase. Afr. J. Biotechnol. 2005;4(1):26–30. [Google Scholar]
- 38.Rabinovich M.L., Melnik M.S., Bolobova A.V. Microbial cellulases: A review. Appl. Biochem. Microbiol. 2002a;38(4):305–321. [Google Scholar]
- 39.Rabinovich M.L., Melnik M.S., Bolobova A.V. The structure and mechanism of action of cellulolytic enzymes. Biochem.(Moscow) 2002;67(8):850–871. doi: 10.1023/a:1019958419032. [DOI] [PubMed] [Google Scholar]
- 40.Rao M.N., Mithal B.M., Thakur R.N., Sastry K.S. Production of cellulase from Pestalotiopsis versicolor. Biotechnol. Bioeng. 1983;25:2395–2408. doi: 10.1002/bit.260251009. [DOI] [PubMed] [Google Scholar]
- 41.Saito K., Kawamura Y., Oda Y. Role of the pectinolytic enzyme in the lactic acid fermentation of potato pulp by Rhizopus oryzae. J. Ind. Microbiol. Biotechnol. 2003;30:440–444. doi: 10.1007/s10295-003-0071-z. [DOI] [PubMed] [Google Scholar]
- 42.Sedrak M.T. Cairo University, Egypt: 2000. Biochemical studies on ethanol production from lignocellulosic wastes by using some microorganisms. M.Sc. (Agric.Sc.) thesis, Faculty of Agriculture. [Google Scholar]
- 43.Sharma D.K., Tiwari M., Behere B.K. Solid state fermentation of new substrates for production of cellulase and other biopolymer hydrolyzing enzymes. Appl. Biochem. Biotechnol. 1996;15:495–500. [Google Scholar]
- 44.Silva R.da, Lago E.S., Merheb C.W., Macchione M.M., Park K.Y., Gomes E. Production of xylanase and CMCase on solid-state fermentation in different residues by Thermoascus aurantiacus Miehe. Braz. J. Microbiol. 2005;36:235–241. [Google Scholar]
- 45.Solomon B.O., Amigun B., Betiku E., Ojumu T., Layokun S.K. Optimization of cellulase production by Aspergillus flavus Linn isolate NSPR 101 grown on bagasse. JNSChE. 1999;16:61–68. [Google Scholar]
- 46.Wood T.M. Haigler . Biosynthesis and Biodegradation of cellulose. Macel Dekker Inc., New York.: 1991. Fungal cellulases; pp. 491–534. [Google Scholar]
- 47.Wood W.E., Neubauer D.G., Stutzenberger F.J. Cyclic AMP levels during induction and repression of cellulase biosynthesis in Thermomonospora curvata. J. Bacteriol. 1984;160:1047–1054. doi: 10.1128/jb.160.3.1047-1054.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zeng G.M., Shi J.G., Yuan X.Z., Liu J., Zhang Z.B., Huang G.H., Li J.B., Xi B.D., Liu H.L. Effects of Tween 80 and rhamnolipid on the extracellular enzymes of Penicillium simplicissimum isolated from compost. Enzyme Microb. Technol. 2006;39:1451–1456. [Google Scholar]


