Abstract
Aims
The aim of this study was to evaluate the combined contribution of 8 polymorphisms to the risk of Alzheimer's disease (AD).
Methods
Through a comprehensive literature search for genetic variants involved in the AD association study, we harvested a total of 6 genes (8 polymorphisms) for the current meta-analyses. These genes consisted of A2M (5bp I/D and V1000I), ABCA2 (rs908832), CHAT (1882G >A, 2384G >A), COMT (Val158Met), HTR6 (267C >T) and LPL (Ser447Ter).
Results
A total of 33 studies among 9,453 cases and 10,833 controls were retrieved for the meta-analyses of 8 genetic variants. It was showed that A2M V1000I (odd ratio (OR) = 1.26, 95% confidence interval (CI) = 1.07–1.49, P = 0.007), rs908832 allele of ABCA2 (OR = 1.55, 95% CI = 1.12–2.16, P = 0.009), 2384G >A of CHAT (OR = 1.22, 95% CI = 1.00–1.49, P = 0.05) and Ser447Ter of LPL in the Northern-American population (OR = 0.56, 95% CI = 0.35–0.91, P = 0.02) were significantly associated with the risk of AD. No association was found between the rest of the 5 polymorphisms and the risk of AD.
Conclusion
Our results showed that A2M V1000I polymorphism in German, Korean, Chinese, Spanish, Italian and Polish populations, rs90883 of ABCA2 gene in French, American, Swiss, Greek and Japanese populations, 2384G >A of CHAT gene in British and Korean populations and LPL Ser447Ter in the Northern-American population were associated with the risk of AD.
Introduction
Alzheimer's disease (AD) is the most common form of dementia among people over 65 years of age [1]. AD is predicted to affect 1 in 85 people globally by 2050 [1]. As an incurable degenerative disease, AD gets worse gradually and eventually leads to death. The features of AD development consist of loss of cognitive functions such as thinking, remembering, and reasoning, and ultimately leading to death. The averaged life expectancy after AD diagnosis is seven years [2]. Although hundreds of clinical trials have been conducted to find ways to treat the disease, none has claimed its effect of stopping or reversing the progressive symptoms. Because AD patients rely on others for assistance, it has imposed great economic costs on society [3], [4].
The cause for most AD cases is still largely unknown, although attempts have been made to explain AD by using the hypotheses based on acetylcholine [5], amyloid [6], [7], tau [7] and etc. In the present study, we performed meta-analyses for the variants on 6 protein encoding genes, including choline O-acetyltransferase (CHAT), catechol-O-methyltransferase (COMT), alpha-2-macroglobulin (A2M), 5-hydroxytryptamine receptor 6 (HTR6), ATP-binding cassette, sub-family A, member 2 (ABCA2), lipoprotein lipase (LPL). CHAT is an important enzyme catalyzing the biosynthesis of the neurotransmitter acetylcholine [8]. Altered protein levels of CHAT in neurons are shown to affect the symptoms of AD containing mild cognitive impairment [9], [10]. As one of the serotonin receptors, HTR6 plays a pivotal role in cognitive and memory processes [11] that are gradually damaged along with the AD progression. Involved in dopamine system, COMT is an important enzyme catalyzing the transfer of a methyl group from S-adenosylmethionine to catecholamines in the synapse [12]. COMT gene variant is associated with the volumes of ventral tegmental area where the gray matter correlates with cognitive and behavioral deficits in AD patients [13]. A2M encodes a protease inhibitor and cytokine transporter [14], [15]. A2M is important for the clearance and degradation of beta-amyloid [16], [17] which may lead to the pathogenesis of AD through the induction of tau phosphorylation [18]–[20]. ABCA2 was cholesterol-responsive gene encoded a member of the superfamily of ATP-binding cassette (ABC) transporters [21]. Over-expression of ABCA2 causes increased protein levels of amyloid beta precursor protein (APP) and beta-amyloid, both of which are important determinants of AD [22]. As a key enzyme to transfer fatty acids from triacylglyceride-rich lipoproteins, LPL is especially important in the process of cholesterol transport in neurons [23]. AD might be related to LPL protein that is one of component of amyloid plaques [24].
Associations between 8 single-nucleotide polymorphisms (SNPs) of the above 6 genes and AD have been reported in different ethnic groups [11], [25]–[57]. The results of these case-control studies for the above 8 genetic variants with AD differ across different groups. In the present study, we aimed to evaluate the combined contribution of the SNPs in these genes to AD susceptibility in different populations using a meta-analysis approach.
Materials and Methods
Literature search and study selection
Literatures were searched through the online databases from 1999 to 2012 using the following key words: “alzheimer's disease, association, SNP or polymorphism or variant or variation or mutation”. The involved databases include PubMed, Chinese National Knowledge infrastructure (CNKI), Embase, SpringerLink, and ScienceDirect. Reference lists in the harvested literatures were explored for additional case-control studies. The criteria for the selection of literatures in the meta-analyses were as followed: (1) the study was case-control association study; (2) allele or genotype information is available; (3) the involved genetic variants have not been studied in previous meta-analysis. The retrieved information consisted of the first author, the year of publication, the number of participants with the different allele (patients and healthy controls), and the odds ratios (ORs) values with 95% confidence intervals (CIs). In total, 33 publications addressing 8 polymorphisms were included in the current meta-analyses.
Statistical Analysis
The meta-analyses were done using the Review Manager 5.0 software [58]. Total ORs with 95% CIs were estimated to evaluate the strength of the association between polymorphisms and AD risk. Heterogeneity was tested by the Cochran's Q statistic and I2 test [59]. A I2<50% denoted a non-signficant heterogeneity among the involved studies in the meta-analysis and fixed-effect model was used in the meta-analyses. The funnel plot was used to evaluate the publication bias in the meta-analysis. A two-sided P value <0.05 in the Z-test was treated as significant.
Results
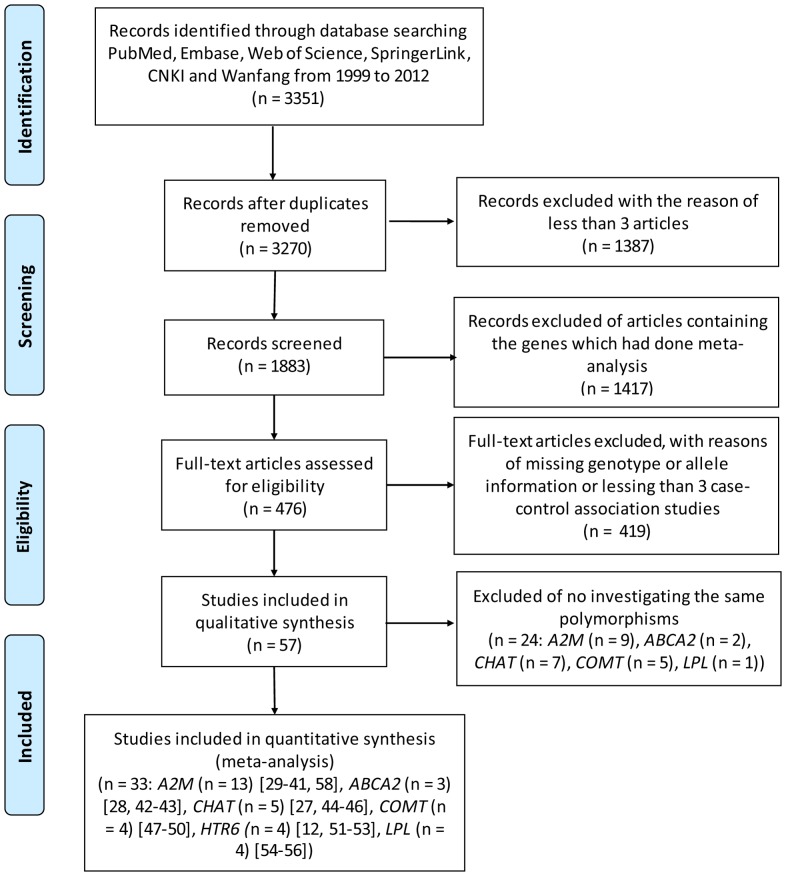
As shown in Figure 1, our search for the case-control studies of AD retrieved 3,351 articles from PubMed, Embase, Web of Science, CNKI and Wanfang from 2000 to 2012. After removing the duplicated publications, we harvested 3270 articles. Among them, 1417 studies were discarded for their involvement in the previous meta-analyses. For the rest 476 studies, we filtered out a total of 428 articles because they failed to accumulate at least three independent genotypic datasets for the same genetic variants. At last, there were 33 case-control studies with 8 polymorphisms for the current meta-analyses (Figure 1).
Figure 1. Flow diagram of the 8 meta-analyses.
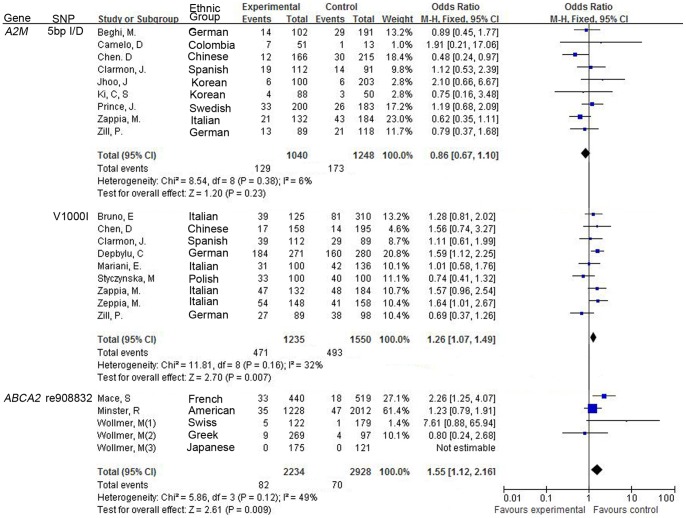
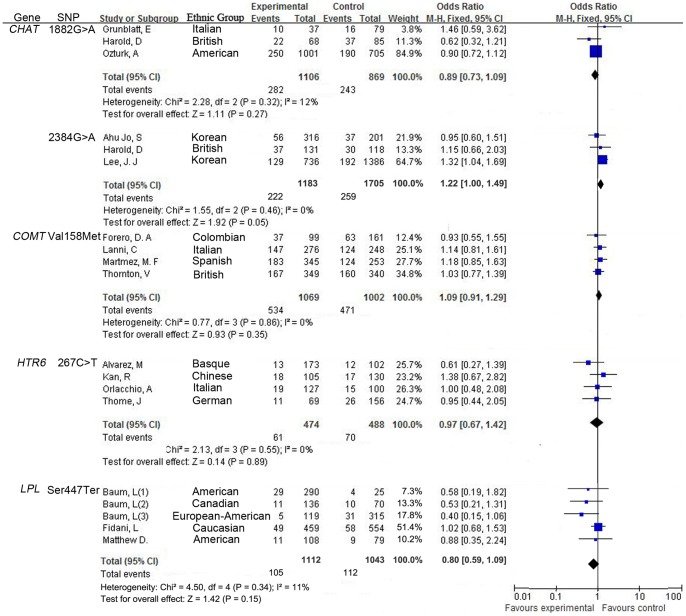
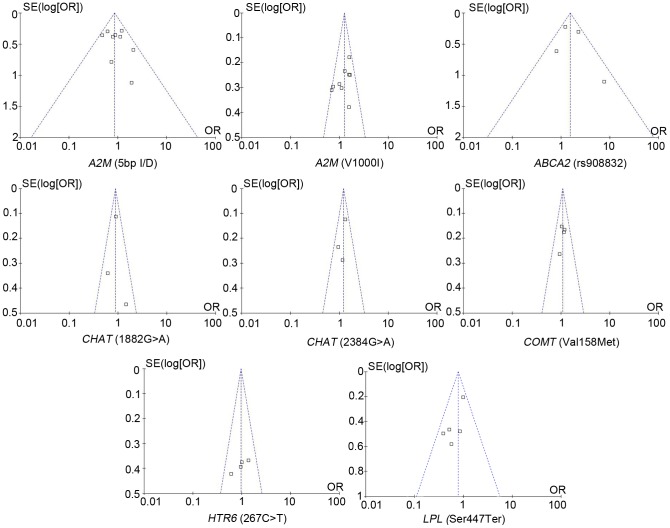
There was no evidence of statistical heterogeneity for all the SNPs in our meta-analysis. Minimal heterogeneity was observed for A2M 5bp-del (I2 = 4%, Figure 2), CHAT 1882G >A (I2 = 12%, Figure 3), CHAT 2384G >A (I2 = 0%, Figure 3), COMT Val158Met (I2 = 0%, Figure 3), HTR6 267C >T (I2 = 0%, Figure 3) and LPL Ser447Ter polymorphism (I2 = 11%, Figure 3). There was moderate heterogeneity for A2M V1000I (I2 = 32%, Figure 2) and ABCA2 rs908832 polymorphism (I2 = 49%, Figure 2). As shown in the funnel plot, no obvious publication bias was observed for the 8 meta-analyses (Figure 4). The details were presented in the Tables 1 and 2.
Figure 2. Forest plots for the relationship between SNPs (5bp I/D, V1001I, rs908832), and AD in the meta-analyses.
Figure 3. Forest plots for the relationship between SNPs (1882G >A, 2384G >A, Val158Met, 267C >T, rs2233678, rs2233679, Ser447Ter) and AD in the meta-analyses.
Figure 4. Funnel plots for the relationship between 8 SNPs and AD in the meta-analyses.
Table 1. The characteristics of the enrolled SNPs (5bp I/D, V1001I, and rs908832).
| Gene | SNP | Year | Author | Ethnic group | No. case/control | Genotype (case/control) | Allele (case/control) | |||
| A2M | 5bpI/D | I/I | I/D | D/D | I | D | ||||
| 2000 | Beghi, M | German | 102/191 | 75/139 | 27/47 | 0/5 | 177/325 | 27/57 | ||
| 2004 | Camelo, D | Colombia | 51/13 | 39/11 | 11/2 | 1/0 | 89/24 | 13/2 | ||
| 2004 | Chen. D | Chinese | 166/215 | 150/175 | 8/20 | 8/20 | 308/370 | 24/60 | ||
| 2003 | Clarmon, J | Spanish | 112/91 | 77/65 | 33/25 | 2/1 | 187/155 | 37/27 | ||
| 2001 | Jhoo, J | Korean | 100/203 | 89/192 | 10/11 | 1/0 | 188/395 | 12/11 | ||
| 2001 | Ki, C, S | Korean | 88/50 | 81/45 | 6/5 | 1/0 | 168/95 | 8/5 | ||
| 2001 | Prince, J | Swedish | 200/183 | 139/137 | 56/40 | 5/6 | 334/314 | 66/52 | ||
| 2002 | Zappia, M | Italian | 132/184 | 95/105 | 32/72 | 5/7 | 222/282 | 42/86 | ||
| 2000 | Zill, P | German | 89/118 | 65/78 | 22/39 | 2/1 | 152/195 | 26/41 | ||
| V1000I | AA | AG | GG | A | G | |||||
| 2010 | Bruno, E | Italian | 125/310 | 57/174 | 58/111 | 10/25 | 172/459 | 78/161 | ||
| 2004 | Chen, D | Chinese | 158/195 | 127/168 | 29/26 | 2/1 | 283/362 | 33/28 | ||
| 2003 | Clarmon, J | Spanish | 112/89 | 45/42 | 56/36 | 11/11 | 146/83 | 78/58 | ||
| 2006 | Depbylu, C | German | 271/280 | 24/45 | 127/151 | 120/84 | 175/241 | 367/319 | ||
| 2006 | Mariani, E | Italian | 100/136 | 49/70 | 41/49 | 10/17 | 139/189 | 61/83 | ||
| 2003 | Styczynska, M | Polish | 100/100 | 49/40 | 37/41 | 14/19 | 135/121 | 65/79 | ||
| 2002 | Zappia, M | Italian | 132/184 | 61/98 | 48/77 | 23/9 | 119/163 | 94/95 | ||
| 2004 | Zappia, M | Italian | 148/158 | 65/85 | 58/65 | 25/8 | 188/235 | 108/81 | ||
| 2000 | Zill, P | German | 89/98 | 44/32 | 37/56 | 8/10 | 125/120 | 53/76 | ||
| ABCA2 | rs908832 | CC | CT | TT | C | T | ||||
| 2005 | Mace, S | French | 440/519 | 376/484 | 63/35 | 1/0 | 815/1003 | 65/35 | ||
| 2009 | Minster, R | American | 1228/2012 | 1160/1920 | 67/90 | 1/2 | 2387/3930 | 69/94 | ||
| 2006 | Wollmer, M | Swiss | 122/179 | 113/177 | 9/2 | 0/0 | 235/356 | 9/2 | ||
| Greek | 269/97 | 253/89 | 15/8 | 1/0 | 521/186 | 17/8 | ||||
| Japanese | 175/121 | 175/121 | 0/0 | 0/0 | 350/242 | 0/0 | ||||
a:N.A. denotes not available.
Table 2. The characteristics of the enrolled SNPs (1882G >A, 2384G >A, Val158Met, 267C >T, and Ser447Ter)a.
| Gene | SNP | Year | Author | Ethnic group | No. case/control | Genotype (case/control) | Allele (case/control) | |||
| CHAT | 1882G >A | GG | GA | AA | G | A | ||||
| 2011 | Grunblatt, E | Italian | 37/79 | 24/51 | 7/26 | 6/2 | 55/128 | 19/32 | ||
| 2003 | Harold, D | British | 68/85 | 34/49 | 25/33 | 9/3 | 93/131 | 43/73 | ||
| 2006 | Ozturk, A | American | 1001/705 | 563/369 | 376/292 | 62/44 | 1502/1030 | 500/380 | ||
| 2384G >A | GG | GA | AA | G | A | |||||
| 2006 | Ahu Jo, S | Korean | 316/201 | 211/192 | 99/7 | 6/2 | 521/454 | 111/74 | ||
| 2003 | Harold, D | British | 131/118 | 69/65 | 51/47 | 11/6 | 189/177 | 73/59 | ||
| 2012 | Lee, J. J | Korean | 736/1386 | 505/1023 | 205/342 | 26/21 | 1215/2388 | 257/384 | ||
| COMT | Val158Met | AA | AG | GG | A | G | ||||
| 2006 | Forero, D. A | Colombian | 99/161 | 41/53 | 43/90 | 15/18 | 125/196 | 73/126 | ||
| 2012 | Lanni, C | Italian | 276/248 | 61/57 | 141/131 | 74/60 | 259/248 | 293/248 | ||
| 2009 | Martmez, M. F | Spanish | 345/253 | 74/67 | 176/125 | 95/61 | 324/259 | 366/247 | ||
| 2011 | Thornton, V | British | 349/340 | 99/105 | 167/151 | 83/84 | 365/361 | 333/319 | ||
| HTR6 | 267C >T | CC | CT | TT | C | T | ||||
| 2003 | Alvarez, M | Basque | 173/102 | 149/81 | 23/18 | 1/3 | 321/180 | 25/24 | ||
| 2004 | Kan, R | Chinese | 105/130 | 70/101 | 35/25 | 0/4 | 175/227 | 35/33 | ||
| 2002 | Orlacchio, A | Italian | 127/100 | 92/76 | 32/19 | 3/5 | 216/171 | 38/29 | ||
| 2001 | Thome, J | German | 69/156 | 50/107 | 17/47 | 2/2 | 117/261 | 21/51 | ||
| LPL | Ser447Ter | Ser/Ser | Ser/Ter | Ter/Ter | Ser | Ter | ||||
| 2000 | Baum, L | American | 290/25 | N.A | N.A | N.A | 522/43 | 58/7 | ||
| Canadian | 136/70 | N.A | N.A | N.A | 250/121 | 22/19 | ||||
| 1999 | Baum, L | European-American | 119/315 | N.A | N.A | N.A | 229/568 | 9/62 | ||
| 2002 | Fidani, L | Caucasian | 459/554 | 368/444 | 85/104 | 6/6 | 821/992 | 97/116 | ||
| 2002 | Matthew D | American | 108/79 | N.A | N.A | N.A | 194/140 | 22/18 | ||
a: N.A. denotes not available.
Meta-analysis of A2M V1000I polymorphism involved 9 studies among 1235 cases and 1550 controls. As shown in Figure 2, V1000I was risk factors to AD onsets (OR = 1.26, 95% CI = 1.07–1.49, P = 0.007, Figure 2). A strong association between rs908832 of ABCA2 gene and AD was observed in the meta-analysis of 5 studies among 2234 cases and 2928 controls (OR = 1.55, 95% CI = 1.12–2.16, P = 0.009, Figure 2). Moderate association was found between the CHAT 2384G >A polymorphism and AD in the meta-analysis of 3 studies among 1183 cases and 1705 controls (OR = 1.22, 95% CI = 1.00–1.49, P = 0.05, Figure 3). As shown in Figure 3, no association of LPL Ser447Ter polymorphism with AD was found in the meta-analysis of 5 studies among 1112 cases and 1043 controls (OR = 0.8, 95% CI = 0.59–1.09, P = 0.16). However, the subgroup analysis by ethnicity found that LPL Ser447Ter polymorphism in the Northern-American population was associated with the risk of AD (OR = 0.56, 95% CI = 0.35–0.91, P = 0.02, Figure S1).
In order to test the robustness of the results in the meta-analyses, we perform a series of subgroup meta-analyses by excluding each study in turn, and the results showed that there was a significant association between the 2 SNPs (A2M V1001I, ABCA2 rs908832) with AD (P<0.05), except for the exclusion of Depbylu's study (A2M V1001I) and the Mace's study (ABCA2 rs908832) (Table S1). For LPL Ser447Ter, the subgroup meta-analysis by excluding Fidani's study found a significant association between LPL Ser447Ter and the risk of AD (Z = 2.33, P = 0.02). Moreover, subgroup meta-analyses by ethnicity were also performed to prevent the bias among different ethnic populations (Figure S1). Our subgroup meta-analyses indicated that A2M V1000I was a risk factor of AD in Italian population among 171 cases and 212 controls (OR = 1.37, 95% CI = 1.07–1.75, P = 0.01), and LPL Ser447Ter polymorphism was likely to be a protective factor of AD in Northern-American population (OR = 0.56, 95% CI = 0.35–0.91, P = 0.02, Figure S2).
Discussion
In the present study, we carried out a systematic overview of case-control association studies for the susceptibility of AD. We screened all the available studies to harvest the eligible SNPs that were involved in at least three independent datasets. In the end, 8 SNPs of 6 AD candidate genes were included in the current meta-analyses. Our results showed significant evidence for 2 AD susceptibility SNPs (A2M V1000I polymorphism (OR = 1.26, 95% CI = 1.07–1.49, P = 0.007), ABCA2 rs908832 polymorphism (OR = 1.55, 95% CI = 1.12–2.16, P = 0.009). We also observed a moderate association of AD for CHAT 2384G >A polymorphism (OR = 1.22, 95% CI = 1.00–1.49, P = 0.05), and an association of AD for LPL Ser447Ter polymorphism in the Northern-American population (OR = 0.56, 95% CI = 0.35–0.91, P = 0.02). No significant associations were found between the rest 4 SNPs and AD.
Large ethnic differences were observed for some of SNPs such as V1000I (20.8% in Germans versus 4.6% in Chinese), and Val158Met (12.4% in Colombians versus 34.8% in British). Under a moderate risk of AD (OR = 1.2), power analysis showed that there might be a lack of power for the meta-analyses of the 4 SNPs, including 5bp I/D of A2M (59.6%), Ser447Ter (56.6%) of LPL, rs908832 of ABCA2 (31.6%) and 267C >T of HTR6 (29.9%). These might partly explain our failure to observe significant results for the meta-analyses of most polymorphisms.
A2M is the one of the key ligands for low density lipoprotein receptor-related protein (LRP) which modulates the critical step for the clearance of A-beta, the major component of beta-amyloid [60]. A2M may regulate AD progression through its ability to mediate the degradation of A-beta [61]. V1000I polymorphism is located near the C-terminal region of A2M which inhibits the β-sheet formation and fibril-formation activities of beta-amyloid [15]. V1000I polymorphism has been shown to increase beta-amyloid directly [34]. Moreover, our meta-analysis has confirmed that A2M V1000I polymorphism is associated with a 26% increase in the risk of AD (P = 0.007), although validation of this finding is warranted among other ethnic populations.
As a member in the Sub-family A, ABCA2 may regulate cholesterol homeostasis and LDLR metabolism in neuronal cells [62]–[64]. ABCA2 expression has been shown to increase endogenous expression of amyloid precursor protein (APP) and the production of Aβ fragment that is a key player in AD progression [65]. SNP rs908832 is C-T polymorphism in exon 14 of the ABCA2 gene. Our result showed a significant contribution of ABCA2 rs908832 polymorphism to the susceptibility of AD (OR = 1.55, 95% CI = 1.12–2.16, P = 0.009). Future research is needed to clarify the mechanistic details of this polymorphism.
As the enzyme responsible for the biosynthesis of acetylcholine [66], CHAT protein is a marker of evaluating the function of basal forebrain cholinergic cells [67], the dementia severity in Alzheimer's disease [68], [69] and the density of senile plaques [70]. By modulating levels of acetylcholine, CHAT influences a wide range of cholinergic-dependent neurophysiological functions including cognitive ability [44], [71]. In early stage of AD, reduction in CHAT activity is a more sensitive indicator than the loss of cholinergic neurons in AD brains [72]. In the current study, two SNPs of CHAT gene were analyzed, but only 1882G >A was shown a moderate association with AD (OR = 1.22, 95% CI = 1.00–1.49, P = 0.05).
Encoding lipoprotein lipase, LPL functions as an Aβ-binding protein promoting cellular uptake and subsequent degradation of Aβ [73]. Lipoprotein lipase genes such as APOE-ε and LPL, are known to be involved in AD pathogenesis [74] [75]. LPL has a neuroprotective effect on AD by participating in the pathophysiological effects of oxidative stress [76]. Our meta-analysis indicates that LPL Ser447Ter polymorphism is a protective factor of AD in the Northern-American population (OR = 0.56, 95% CI = 0.35–0.91, P = 0.02) and thus supports this above speculation.
There were several limitations in our meta-analyses. Firstly, for some SNPs such as Ser447Ter of LPL gene, the involved samples were only limited in a few populations. The results of our meta-analyses may not stand for all ethnic populations. Future investigations in other populations are needed to clarify the contribution of the SNPs of interest to AD susceptibility. Secondly, we didn't probe the interaction of the two positive SNPs (A2M V1000I and ABCA2 rs908832) and two less significant SNPs (CHAT 2384G >A and LPL Ser447Ter) with APOE-ε4 genotype which is the strongest risk factor of AD. Thus, we can't exclude the possibility that our findings are dependent on APOE-ε4 genotype. Thirdly, according to the disease onset age, there are two subtypes of AD, early-onset of AD (EOAD) and late-onset of AD (LOAD). A potential stratification by age may exist in the current meta-analyses, although no significant heterogeneity was found for all the 8 meta-analyses (Table S2 and S3). Among all the studies, we are able to get only three study mentioning the age of onset A2M 5bp I/D polymorphism. There are a total 111 EOAD and 235 LOAD cases and 129 controls younger than 65 and 338 controls with age equal to or over 65. The subgroup analysis has shown that A2M 5bp I/D polymorphism is not associated with AD in both young (Figure S2, OR = 0.78, 95% CI = 0.28–2.16, P = 0.64) and old (OR = 1.41, 95% CI = 0.62–3.18, P = 0.41) subgroups. As shown in the funnel plot of Figure S2, no obvious publication bias is shown for the above two meta-analyses. Fourthly, the incidence of female is higher than male in clinical and over half of the subjects participating in all the studies are female [1]. As an important factor of AD, gender should be considered as a stratifying variable for the further study exploring the diversity among the results of different studies. Due to a paucity of gender-related information, we are unable to perform the subgroup meta-analyses by gender. Fifthly, as shown in the Table S4, we have performed a thorough scanning for the criteria used to determine AD diagnosis. Among all the studies, only one study by Kan did the subgroup analysis of clinic and pathology. Due to a lack of informative subgroup analysis in the involved studies, we discontinue the subgroup analysis by the diagnosis criteria. Sixthly, as shown in the Table S5, there are inconsistencies in the presentation of the score of Mini Mental State Examination (MMSE) for controls. This may lead to the discrepancy in the association studies worldwide. Lastly but not least, we didn't include the genomewide association studies into our meta-analyses. There are a total of 33 GWA studies on AD in the GWAS catalog (http://www.genome.gov/page.cfm?pageid=26525384#searchForm) and 14 studies in the NCBI dbGap dataset (http://www.ncbi.nlm.nih.gov/gap/?term=alzheimer). All the loci in our meta-analyses are not presented among the strongest loci in those GWA studies.
In conclusion, we identified significant associations between 2 SNPs (A2M V1000I and ABCA2 rs908832) and AD. Meta-analysis among 1235 cases and 1550 controls has confirmed that A2M V1000I is a risk factor of AD in German, Korean, Chinese, Spanish, Italian and Polish populations. Further, meta-analysis among 2234 cases and 2928 controls has confirmed that rs908832 of ABCA2 gene is a risk factor of AD in French, American, Swiss, Greek and Japanese populations. In addition, meta-analysis among 222 cases and 259 controls indicates a moderate association of CHAT 2384G >A with AD in British and Korean populations. Another meta-analysis among 1112 cases and 1043 controls indicates that LPL Ser447Ter polymorphism is likely to be associated with a reduced risk of AD in the Northern-American population (OR = 0.56, 95% CI = 0.35–0.91, P = 0.02).
Supporting Information
Subgroup analysis by ethnicity between SNPs (5bp I/D, V1001I, rs908832, Ser447Ter).
(TIF)
Subgroup analysis by mean age of AD patient.
(TIF)
The meta-analysis results of excluding each study in turn.
(DOC)
The stratifying variables of the enrolled SNPs (5bp I/D, V1001I, and rs908832).
(DOC)
The stratifying variables of the enrolled SNPs (1882G >A, 2384G >A, Val158Met, 267C>T, and Ser447Ter).
(DOC)
Subgroup analysis by AD diagnosis criteria.
(DOC)
Subgroup analysis by Mini Mental State Examination (MMSE) in control population.
(DOC)
PRISMA Checklist.
(DOC)
Funding Statement
The research was supported by the grants from the National Natural Science Foundation of China (31100919, 81070873, and 81271209), Natural Science Foundation of Zhejiang Province (LR13H020003), K. C. Wong Magna Fund in Ningbo University, Ningbo social development research projects (2012C50032), Science and Technology Innovation team of Ningbo (2011B82014), the neurobiological mechanisms of drug-reward: role of the habenula National Natural Science Foundation of China (81171257), and Research Fund in Ningbo University (XKL11D2117). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Vina J, Lloret A (2010) Why women have more Alzheimer's disease than men: gender and mitochondrial toxicity of amyloid-beta peptide. J Alzheimers Dis 20 Suppl 2S527–533. [DOI] [PubMed] [Google Scholar]
- 2. Molsa PK, Marttila RJ, Rinne UK (1986) Survival and cause of death in Alzheimer's disease and multi-infarct dementia. Acta Neurol Scand 74: 103–107. [DOI] [PubMed] [Google Scholar]
- 3. Bonin-Guillaume S, Zekry D, Giacobini E, Gold G, Michel JP (2005) [The economical impact of dementia]. Presse Med 34: 35–41. [DOI] [PubMed] [Google Scholar]
- 4.Meek PD, McKeithan K, Schumock GT (1998) Economic considerations in Alzheimer's disease. Pharmacotherapy 18: 68–73; discussion 79–82. [PubMed]
- 5. Francis PT, Palmer AM, Snape M, Wilcock GK (1999) The cholinergic hypothesis of Alzheimer's disease: a review of progress. J Neurol Neurosurg Psychiatry 66: 137–147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Hardy J, Allsop D (1991) Amyloid deposition as the central event in the aetiology of Alzheimer's disease. Trends Pharmacol Sci 12: 383–388. [DOI] [PubMed] [Google Scholar]
- 7. Mudher A, Lovestone S (2002) Alzheimer's disease-do tauists and baptists finally shake hands? Trends Neurosci 25: 22–26. [DOI] [PubMed] [Google Scholar]
- 8. Mubumbila V, Sutter A, Ptok U, Heun R, Quirin-Stricker C (2002) Identification of a single nucleotide polymorphism in the choline acetyltransferase gene associated with Alzheimer's disease. Neurosci Lett 333: 9–12. [DOI] [PubMed] [Google Scholar]
- 9. Kooi EJ, Prins M, Bajic N, Belien JA, Gerritsen WH, et al. (2011) Cholinergic imbalance in the multiple sclerosis hippocampus. Acta Neuropathol 122: 313–322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Mengel-From J, Christensen K, Thinggaard M, McGue M, Christiansen L (2011) Genetic variants in the choline acetyltransferase (ChAT) gene are modestly associated with normal cognitive function in the elderly. Genes Brain Behav 10: 876–882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Orlacchio A, Kawarai T, Paciotti E, Stefani A, Sorbi S, et al. (2002) Association study of the 5-hydroxytryptamine(6) receptor gene in Alzheimer's disease. Neurosci Lett 325: 13–16. [DOI] [PubMed] [Google Scholar]
- 12. Guldberg HC, Marsden CA (1975) Catechol-O-methyl transferase: pharmacological aspects and physiological role. Pharmacol Rev 27: 135–206. [PubMed] [Google Scholar]
- 13. Gennatas ED, Cholfin JA, Zhou J, Crawford RK, Sasaki DA, et al. (2012) COMT Val158Met genotype influences neurodegeneration within dopamine-innervated brain structures. Neurology 78: 1663–1669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Blacker D, Wilcox MA, Laird NM, Rodes L, Horvath SM, et al. (1998) Alpha-2 macroglobulin is genetically associated with Alzheimer disease. Nat Genet 19: 357–360. [DOI] [PubMed] [Google Scholar]
- 15. Hughes SR, Khorkova O, Goyal S, Knaeblein J, Heroux J, et al. (1998) Alpha2-macroglobulin associates with beta-amyloid peptide and prevents fibril formation. Proc Natl Acad Sci U S A 95: 3275–3280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Narita M, Holtzman DM, Schwartz AL, Bu G (1997) Alpha2-macroglobulin complexes with and mediates the endocytosis of beta-amyloid peptide via cell surface low-density lipoprotein receptor-related protein. J Neurochem 69: 1904–1911. [DOI] [PubMed] [Google Scholar]
- 17. Herring A, Yasin H, Ambree O, Sachser N, Paulus W, et al. (2008) Environmental enrichment counteracts Alzheimer's neurovascular dysfunction in TgCRND8 mice. Brain Pathol 18: 32–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Jin M, Shepardson N, Yang T, Chen G, Walsh D, et al. (2011) Soluble amyloid beta-protein dimers isolated from Alzheimer cortex directly induce Tau hyperphosphorylation and neuritic degeneration. Proc Natl Acad Sci U S A 108: 5819–5824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Haass C, Kaether C, Thinakaran G, Sisodia S (2012) Trafficking and Proteolytic Processing of APP. Cold Spring Harb Perspect Med 2: a006270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Lambert MP, Sabo S, Zhang C, Enam SA, Klein WL (1995) Constitutive Alzheimer's-type tau epitopes in a neuritogenic rat CNS cell line. Neurobiol Aging 16: 583–589. [DOI] [PubMed] [Google Scholar]
- 21. Kaminski WE, Piehler A, Pullmann K, Porsch-Ozcurumez M, Duong C, et al. (2001) Complete coding sequence, promoter region, and genomic structure of the human ABCA2 gene and evidence for sterol-dependent regulation in macrophages. Biochem Biophys Res Commun 281: 249–258. [DOI] [PubMed] [Google Scholar]
- 22. Chen ZJ, Vulevic B, Ile KE, Soulika A, Davis W Jr, et al. (2004) Association of ABCA2 expression with determinants of Alzheimer's disease. FASEB J 18: 1129–1131. [DOI] [PubMed] [Google Scholar]
- 23. Nunez M, Peinado-Onsurbe J, Vilaro S, Llobera M (1995) Lipoprotein lipase activity in developing rat brain areas. Biol Neonate 68: 119–127. [DOI] [PubMed] [Google Scholar]
- 24. Ben-Zeev O, Doolittle MH, Singh N, Chang CH, Schotz MC (1990) Synthesis and regulation of lipoprotein lipase in the hippocampus. J Lipid Res 31: 1307–1313. [PubMed] [Google Scholar]
- 25. Lee JJ, Jo SA, Park JH, Lee SB, Jo I, et al. (2012) Choline acetyltransferase 2384G >a polymorphism and the risk of Alzheimer disease. Alzheimer Dis Assoc Disord 26: 81–87. [DOI] [PubMed] [Google Scholar]
- 26. Harold D, Peirce T, Moskvina V, Myers A, Jones S, et al. (2003) Sequence variation in the CHAT locus shows no association with late-onset Alzheimer's disease. Hum Genet 113: 258–267. [DOI] [PubMed] [Google Scholar]
- 27. Mace S, Cousin E, Ricard S, Genin E, Spanakis E, et al. (2005) ABCA2 is a strong genetic risk factor for early-onset Alzheimer's disease. Neurobiol Dis 18: 119–125. [DOI] [PubMed] [Google Scholar]
- 28. Depboylu C, Lohmuller F, Du Y, Riemenschneider M, Kurz A, et al. (2006) Alpha2-macroglobulin, lipoprotein receptor-related protein and lipoprotein receptor-associated protein and the genetic risk for developing Alzheimer's disease. Neurosci Lett 400: 187–190. [DOI] [PubMed] [Google Scholar]
- 29. Chen D, Zhang JW, Zhang ZX, Wu YN, Qu QM (2004) Association of alpha 2-macroglobulin polymorphisms and Alzheimer disease in Mainland Han Chinese. J Neurol Sci 217: 13–15. [DOI] [PubMed] [Google Scholar]
- 30. Jhoo JH, Kim KW, Lee DY, Lee KU, Lee JH, et al. (2001) Association of alpha-2-macroglobulin deletion polymorphism with sporadic Alzheimer's disease in Koreans. J Neurol Sci 184: 21–25. [DOI] [PubMed] [Google Scholar]
- 31. Ki CS, Na DL, Kim HJ, Kim JW (2001) Alpha-1 antichymotrypsin and alpha-2 macroglobulin gene polymorphisms are not associated with Korean late-onset Alzheimer's disease. Neurosci Lett 302: 69–72. [DOI] [PubMed] [Google Scholar]
- 32. Camelo D, Arboleda G, Yunis JJ, Pardo R, Arango G, et al. (2004) Angiotensin-converting enzyme and alpha-2-macroglobulin gene polymorphisms are not associated with Alzheimer's disease in Colombian patients. J Neurol Sci 218: 47–51. [DOI] [PubMed] [Google Scholar]
- 33. Clarimon J, Bertranpetit J, Calafell F, Boada M, Tarraga L, et al. (2003) Joint analysis of candidate genes related to Alzheimer's disease in a Spanish population. Psychiatr Genet 13: 85–90. [DOI] [PubMed] [Google Scholar]
- 34. Zappia M, Cittadella R, Manna I, Nicoletti G, Andreoli V, et al. (2002) Genetic association of alpha2-macroglobulin polymorphisms with AD in southern Italy. Neurology 59: 756–758. [DOI] [PubMed] [Google Scholar]
- 35. Prince JA, Feuk L, Sawyer SL, Gottfries J, Ricksten A, et al. (2001) Lack of replication of association findings in complex disease: an analysis of 15 polymorphisms in prior candidate genes for sporadic Alzheimer's disease. Eur J Hum Genet 9: 437–444. [DOI] [PubMed] [Google Scholar]
- 36. Bagli M, Papassotiropoulos A, Jessen F, Schmitz S, Rao ML, et al. (2000) Identical distribution of the alpha 2-macroglobulin pentanucleotide deletion in subjects with Alzheimer disease and controls in a German population. Am J Med Genet 96: 775–777. [DOI] [PubMed] [Google Scholar]
- 37. Styczynska M, Religa D, Pfeffer A, Luczywek E, Wasiak B, et al. (2003) Simultaneous analysis of five genetic risk factors in Polish patients with Alzheimer's disease. Neurosci Lett 344: 99–102. [DOI] [PubMed] [Google Scholar]
- 38. Zappia M, Manna I, Serra P, Cittadella R, Andreoli V, et al. (2004) Increased risk for Alzheimer disease with the interaction of MPO and A2M polymorphisms. Arch Neurol 61: 341–344. [DOI] [PubMed] [Google Scholar]
- 39. Mariani E, Seripa D, Ingegni T, Nocentini G, Mangialasche F, et al. (2006) Interaction of CTSD and A2M polymorphisms in the risk for Alzheimer's disease. J Neurol Sci 247: 187–191. [DOI] [PubMed] [Google Scholar]
- 40. Bruno E, Quattrocchi G, Nicoletti A, Le Pira F, Maci T, et al. (2010) Lack of interaction between LRP1 and A2M polymorphisms for the risk of Alzheimer disease. Neurosci Lett 482: 112–116. [DOI] [PubMed] [Google Scholar]
- 41. Minster RL, DeKosky ST, Kamboh MI (2009) No association of DAPK1 and ABCA2 SNPs on chromosome 9 with Alzheimer's disease. Neurobiol Aging 30: 1890–1891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Wollmer MA, Kapaki E, Hersberger M, Muntwyler J, Brunner F, et al. (2006) Ethnicity-dependent genetic association of ABCA2 with sporadic Alzheimer's disease. Am J Med Genet B Neuropsychiatr Genet 141B: 534–536. [DOI] [PubMed] [Google Scholar]
- 43. Ozturk A, DeKosky ST, Kamboh MI (2006) Genetic variation in the choline acetyltransferase (CHAT) gene may be associated with the risk of Alzheimer's disease. Neurobiol Aging 27: 1440–1444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Grunblatt E, Reif A, Jungwirth S, Galimberti D, Weber H, et al. (2011) Genetic variation in the choline O-acetyltransferase gene in depression and Alzheimer's disease: the VITA and Milano studies. J Psychiatr Res 45: 1250–1256. [DOI] [PubMed] [Google Scholar]
- 45. Ahn Jo S, Ahn K, Kim JH, Kang BH, Kim E, et al. (2006) ApoE-epsilon 4-dependent association of the choline acetyltransferase gene polymorphisms (2384G >A and 1882G >A) with Alzheimer's disease. Clin Chim Acta 368: 179–182. [DOI] [PubMed] [Google Scholar]
- 46. Forero DA, Benitez B, Arboleda G, Yunis JJ, Pardo R, et al. (2006) Analysis of functional polymorphisms in three synaptic plasticity-related genes (BDNF, COMT AND UCHL1) in Alzheimer's disease in Colombia. Neurosci Res 55: 334–341. [DOI] [PubMed] [Google Scholar]
- 47. Martinez MF, Martin XE, Alcelay LG, Flores JC, Valiente JM, et al. (2009) The COMT Val158 Met polymorphism as an associated risk factor for Alzheimer disease and mild cognitive impairment in APOE 4 carriers. BMC Neurosci 10: 125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lanni C, Garbin G, Lisa A, Biundo F, Ranzenigo A, et al.. (2012) Influence of COMT Val158Met Polymorphism on Alzheimer's Disease and Mild Cognitive Impairment in Italian Patients. J Alzheimers Dis. [DOI] [PubMed]
- 49. Thornton V, Warden D, Talbot C, Mastana SS, Bandelow S, et al. (2011) Modification of estrogen's association with Alzheimer's disease risk by genetic polymorphisms. Brain Res 1379: 213–223. [DOI] [PubMed] [Google Scholar]
- 50. Thome J, Retz W, Baader M, Pesold B, Hu M, et al. (2001) Association analysis of HTR6 and HTR2A polymorphisms in sporadic Alzheimer's disease. J Neural Transm 108: 1175–1180. [DOI] [PubMed] [Google Scholar]
- 51. Alvarez-Alvarez M, Galdos L, Fernandez-Martinez M, Gomez-Busto F, Garcia-Centeno V, et al. (2003) 5-Hydroxytryptamine 6 receptor (5-HT(6)) receptor and apolipoprotein E (ApoE) polymorphisms in patients with Alzheimer's disease in the Basque Country. Neurosci Lett 339: 85–87. [DOI] [PubMed] [Google Scholar]
- 52. Kan R, Wang B, Zhang C, Yang Z, Ji S, et al. (2004) Association of the HTR6 polymorphism C267T with late-onset Alzheimer's disease in Chinese. Neurosci Lett 372: 27–29. [DOI] [PubMed] [Google Scholar]
- 53. Fidani L, Compton D, Hardy J, Petersen RC, Tangalos E, et al. (2002) No association between the lipoprotein lipase S447X polymorphism and Alzheimer's disease. Neurosci Lett 322: 192–194. [DOI] [PubMed] [Google Scholar]
- 54. Baum L, Wiebusch H, Pang CP (2000) Roles for lipoprotein lipase in Alzheimer's disease: an association study. Microsc Res Tech 50: 291–296. [DOI] [PubMed] [Google Scholar]
- 55. Martin-Rehrmann MD, Cho HS, Rebeck GW (2002) Lack of association of two lipoprotein lipase polymorphisms with Alzheimer's disease. Neurosci Lett 328: 109–112. [DOI] [PubMed] [Google Scholar]
- 56. Baum L, Chen L, Masliah E, Chan YS, Ng HK, et al. (1999) Lipoprotein lipase mutations and Alzheimer's disease. Am J Med Genet 88: 136–139. [DOI] [PubMed] [Google Scholar]
- 57. Zill P, Burger K, Behrens S, Hampel H, Padberg F, et al. (2000) Polymorphisms in the alpha-2 macroglobulin gene in psychogeriatric patients. Neurosci Lett 294: 69–72. [DOI] [PubMed] [Google Scholar]
- 58. Estrela C, Silva JA, de Alencar AH, Leles CR, Decurcio DA (2008) Efficacy of sodium hypochlorite and chlorhexidine against Enterococcus faecalis–a systematic review. J Appl Oral Sci 16: 364–368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Coory MD (2010) Comment on: Heterogeneity in meta-analysis should be expected and appropriately quantified. Int J Epidemiol 39: 932; author reply 933. [DOI] [PubMed]
- 60. Kang DE, Pietrzik CU, Baum L, Chevallier N, Merriam DE, et al. (2000) Modulation of amyloid beta-protein clearance and Alzheimer's disease susceptibility by the LDL receptor-related protein pathway. J Clin Invest 106: 1159–1166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Mettenburg JM, Webb DJ, Gonias SL (2002) Distinct binding sites in the structure of alpha 2-macroglobulin mediate the interaction with beta-amyloid peptide and growth factors. J Biol Chem 277: 13338–13345. [DOI] [PubMed] [Google Scholar]
- 62. Smith JD, Le Goff W, Settle M, Brubaker G, Waelde C, et al. (2004) ABCA1 mediates concurrent cholesterol and phospholipid efflux to apolipoprotein A-I. J Lipid Res 45: 635–644. [DOI] [PubMed] [Google Scholar]
- 63. Davis W Jr (2011) The ATP-binding cassette transporter-2 (ABCA2) regulates cholesterol homeostasis and low-density lipoprotein receptor metabolism in N2a neuroblastoma cells. Biochim Biophys Acta 1811: 1152–1164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Ricciarelli R, Canepa E, Marengo B, Marinari UM, Poli G, et al. (2012) Cholesterol and Alzheimer's disease: a still poorly understood correlation. IUBMB Life 64: 931–935. [DOI] [PubMed] [Google Scholar]
- 65. Davis W Jr (2010) The ATP-binding cassette transporter-2 (ABCA2) increases endogenous amyloid precursor protein expression and Abeta fragment generation. Curr Alzheimer Res 7: 566–577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Oda Y (1999) Choline acetyltransferase: the structure, distribution and pathologic changes in the central nervous system. Pathol Int 49: 921–937. [DOI] [PubMed] [Google Scholar]
- 67. Gil-Bea FJ, Garcia-Alloza M, Dominguez J, Marcos B, Ramirez MJ (2005) Evaluation of cholinergic markers in Alzheimer's disease and in a model of cholinergic deficit. Neurosci Lett 375: 37–41. [DOI] [PubMed] [Google Scholar]
- 68. Bierer LM, Haroutunian V, Gabriel S, Knott PJ, Carlin LS, et al. (1995) Neurochemical correlates of dementia severity in Alzheimer's disease: relative importance of the cholinergic deficits. J Neurochem 64: 749–760. [DOI] [PubMed] [Google Scholar]
- 69. DeKosky ST, Ikonomovic MD, Styren SD, Beckett L, Wisniewski S, et al. (2002) Upregulation of choline acetyltransferase activity in hippocampus and frontal cortex of elderly subjects with mild cognitive impairment. Ann Neurol 51: 145–155. [DOI] [PubMed] [Google Scholar]
- 70. Wilcock GK, Esiri MM, Bowen DM, Smith CC (1982) Alzheimer's disease. Correlation of cortical choline acetyltransferase activity with the severity of dementia and histological abnormalities. J Neurol Sci 57: 407–417. [DOI] [PubMed] [Google Scholar]
- 71. Kanju PM, Parameshwaran K, Sims-Robinson C, Uthayathas S, Josephson EM, et al. (2012) Selective cholinergic depletion in medial septum leads to impaired long term potentiation and glutamatergic synaptic currents in the hippocampus. PLoS One 7: e31073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Nunes-Tavares N, Santos LE, Stutz B, Brito-Moreira J, Klein WL, et al. (2012) Inhibition of choline acetyltransferase as a mechanism for cholinergic dysfunction induced by amyloid-beta peptide oligomers. J Biol Chem 287: 19377–19385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Nishitsuji K, Hosono T, Uchimura K, Michikawa M (2011) Lipoprotein lipase is a novel amyloid beta (Abeta)-binding protein that promotes glycosaminoglycan-dependent cellular uptake of Abeta in astrocytes. J Biol Chem 286: 6393–6401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Xie C, Wang ZC, Liu XF, Yang MS (2010) The common biological basis for common complex diseases: evidence from lipoprotein lipase gene. Eur J Hum Genet 18: 3–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Papassotiropoulos A, Wollmer MA, Tsolaki M, Brunner F, Molyva D, et al. (2005) A cluster of cholesterol-related genes confers susceptibility for Alzheimer's disease. J Clin Psychiatry 66: 940–947. [PubMed] [Google Scholar]
- 76. Paradis E, Clement S, Julien P, Ven Murthy MR (2003) Lipoprotein lipase affects the survival and differentiation of neural cells exposed to very low density lipoprotein. J Biol Chem 278: 9698–9705. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Subgroup analysis by ethnicity between SNPs (5bp I/D, V1001I, rs908832, Ser447Ter).
(TIF)
Subgroup analysis by mean age of AD patient.
(TIF)
The meta-analysis results of excluding each study in turn.
(DOC)
The stratifying variables of the enrolled SNPs (5bp I/D, V1001I, and rs908832).
(DOC)
The stratifying variables of the enrolled SNPs (1882G >A, 2384G >A, Val158Met, 267C>T, and Ser447Ter).
(DOC)
Subgroup analysis by AD diagnosis criteria.
(DOC)
Subgroup analysis by Mini Mental State Examination (MMSE) in control population.
(DOC)
PRISMA Checklist.
(DOC)