Introduction
Retroperitoneal sarcomas (RPS) provide a unique set of challenges for oncologic management. Due to the deep location, presentation is often delayed until the tumors reach a very large size or abut/invade vital abdominal viscera or neurovascular structures. More than 60% percent of RPS in large institutional series from Memorial Sloan Kettering Cancer Center (MSKCC) and the University of Michigan measured larger than 10 cm, while 45% of patients in a French series had tumors larger than 20 cm [1, 2]. The median tumor size in a Surveillance, Epidemiology, and End Results (SEER) database analysis was 17 cm [3].
Available data demonstrate a strong relationship between R0 resection and both survival and local recurrence. However, the size of the tumor and its proximity to adjacent organs create technical challenges with regards to achieving a complete resection, and en bloc resection of involved viscera is typically required in about three-fourths of patients [1, 2, 4]. It has been noted that, in general, complicated surgeries are best performed in high-volume centers [5, 6•]. This is also important for rare conditions like soft-tissue sarcomas (STSs) including RPS [7, 8••] where higher rates of R0 resection are observed in high-volume centers. However, even at high-volume institutions employing aggressive surgical strategies, about 12–58% patients will have R1 resection and up to 20–25% patients will have R2 resection or unresectable disease [8••–15]. More recently, there have been several reports in the surgical literature describing improved outcomes with a more aggressive, extended surgical approach to include all adjacent organs, regardless of whether they appear to be involved with tumor or not [8••, 13••]. Although overall survival and local recurrence outcomes appear promising with this extended surgical approach, morbidity is increased and available data is limited by comparison to historical controls and necessary selection bias. This area of research is continuing to evolve.
Even with optimal surgery, rates of local recurrence remain high (25–50%) [16]. Local-recurrence accounts for nearly 75% of RPS-related deaths [17]. This establishes the rationale for additional therapy with local efficacy, either pre- or post-operatively. The high incidence of R1 resections suggests a potential role for preoperative therapies to achieve sterilization of microscopic disease and increase R0 resection rates, while also reducing recurrence rates in resectable patients.
Radiotherapy (RT) has a long history in RPS management. Many older series failed to demonstrate an impact on local control or survival with the addition of radiotherapy [18–23]. This may be due to delivery of sub-therapeutic doses as allowed by the available radiotherapy techniques. However, improvements in radiotherapy technology now permit delivery of higher doses while limiting toxicity. Multiple, more contemporary, series have demonstrated a dose-response relationship suggesting improved outcomes with higher doses [1, 2, 24–27].
This review provides an update of literature on the role of radiotherapy in the management of RPS, and highlights future directions required to improve outcomes in this disease.
TREATMENT
Indications for RT in RPS
Postoperative radiotherapy (PORT) is typically recommended for close or positive margins and high-grade disease. Additional factors that are considered include tumor size, location, difficulty in surgical excision/intra-operative impression, tolerance to surgery, performance status and institutional preference/experience. PORT is usually not offered in the setting of postoperative complications resulting in poor performance status or delayed recovery, significant post-surgical residual disease and metastatic disease [2].
Preoperative radiotherapy (PreRT) is typically employed when tumors are deemed unresectable or marginally resectable. The primary goals of PreRT are to improve resectability and local control. However, PreRT has several other advantages that include decreased intra-operative seeding, superior radiotherapy targeting by natural displacement of adjacent normal tissue and lower equi-efficacious doses due to better oxygenated tissue [16]. Experience from extremity STS has clearly outlined a role for PreRT that allows delivery of lower total dose while maintaining equivalent local control and survival outcomes at the expense of a higher risk of wound complications [28]. This has significant implications in the management of RPS wherein the deliverable RT dose is highly limited by the presence of adjacent, radiosensitive normal structures.
In light of the advantages of PreRT over PORT, this approach has become increasingly represented in modern series regardless of RPS resectability [see Table 1 and 2].
Intra-operative RT (IORT) has been conventionally employed by itself or in combination with PORT with the goal of radiotherapy dose-escalation. Two primary forms are available: intraoperative electrons (IOERT) and brachytherapy (BRT). Physical displacement of sensitive normal tissue, use of individualized shielding and the limited penetration of electrons permits the use of a highly biologically effective, single large fraction of radiation with IOERT. BRT, in contrast, involves placement of interstitial catheters through the surgical bed with intra-operative assistance followed by remote afterloading for treatment using high-dose-rate (HDR) techniques or placement of permanent radiation sources for treatment using low-dose-rate (LDR) techniques. The sharp dose-fall off from BRT forms the basis of using this approach. In either case, IORT is somewhat limited by availability of techniques and experience. There is also concern that intraoperative techniques may raise risks of dose-limiting neuropathy [24]. In the PreRT era, IORT may be particularly attractive when intra-operative evaluation suggests R0 resection is implausible. In such cases, IORT may allow selective radiotherapy boost to the surgical bed without a prolonged delay from completion of PreRT.
Despite improvements in surgical techniques, about 20% of patients remain unresectable at presentation typically due to involvement of neurovascular structures of the retroperitoneum or extensive involvement of the viscera [1, 2]. Definitive RT alone, especially with highly conformal radiotherapy techniques or use of particle beam therapy may be employed in these scenarios, primarily to slow the rate of progression and, potentially, provide symptomatic relief.
Table 1.
Independent prognostic factors for local recurrence (LR) and overall survival (OS) in the RT era; Literature summary between the year 2000 to 2009
| Institution [reference], year, patient numbers | RT sequence (percent of analyzed patients) | Negative Prognostic factor# for Local recurrence at 5- years | Negative Prognostic factor# for OS at 5-years |
|---|---|---|---|
| MSKCC [1], 2000, 32 | IORT + PORT (78%) IORT (22%) |
|
|
| FNCLCC [32], 2001, 145 | PORT (56%) |
|
|
| Institut Bergonie [2], 2002, 45 | PORT alone (62%) IORT alone (7%) IORT + PORT (31%) |
|
|
| Wayne State* [10], 2002, 60 | PORT (73%) PORT + BRT (27%) |
|
|
| U. Heidelberg [12], 2006, 67 | PORT + IORT (67%) IORT (33%) |
|
|
| MGH [9], 2001, 37 | PreRT alone (46%) PreRT + IORT (54%) |
|
|
| PMH [54], 2002, 55 | PreRT (38%) PreRT + BRT (38%) |
|
|
| PMH + MDACC [48], 2006, 54 | PreRT (37%) PreRT + IORT (41%) |
|
|
| Tom Baker [55], 2007, 27 | PreRT (93%) |
|
|
| Mayo Rochester [33], 2002, 87 | PreRT + IORT (60%) PORT + IORT (14%) Both + IORT (14%) IORT alone (12%) |
|
|
| U. Florida [11], 2005, 40 | PORT (37.5%) PreRT (62.5%)^ |
|
|
| U. Michigan* [26], 2007, 88 | PORT (60%) PreRT (20%) Both (5%) Definitive RT (15%) |
|
|
| MDACC [27], 2007, 83 | PORT (40%) PreRT (60%) IORT + EBRT (22% of whole) |
|
|
| Istituto Nazionale Tumori [13], Italy 2009, 288 | RT (31%) |
|
|
| French Multi-institutional [8], 2009, 382 | PORT (29%) PreRT (3%) IORT + EBRT (5% of whole) |
|
|
OS: Overall survival, MSKCC: Memorial Sloan Kettering Cancer Center, FNCLCC: French Federation of Cancer Centers Sarcoma Group database, MGH: Massachusetts General Hospital, PMH: Princess Margaret Hospital, MDACC: MD Anderson Cancer Center, PORT: Postoperative RT, PreRT: Preoperative RT, EBRT: External Beam RT, BRT: Brachytherapy, IORT: Intra-operative RT, MVA: Multivariate analysis,
On multivariate analysis
Deep trunk tumors (deep tumors of the chest wall, flank, spinal, paraspinal regions and tumors of the pelvic wall) were also included in this series (37% at Wayne state and 20% at University of Michigan).
Table 2.
Independent prognostic factors for local control and survival in the RT era; Literature summary between the year 2010 to current
| Institution, year, patient numbers (reference) | Radiotherapy sequence (percent of analyzed patients) | Radiotherapy dose: Median (Range) | Prognostic factor# for Local recurrence at 5-years | Prognostic factor# for overall survival at 5-years |
|---|---|---|---|---|
| UCLA [34], 2010, 55 | PreRT (56%) PORT (44%) |
50 Gy |
|
|
| U. Ulsan [50], 2011, 40 | PORT (100%) | 55.9 Gy (36 to 65 Gy) |
|
|
| City of Hope [56], 2011, 33 | IORT +PORT (61%) PORT alone (39%) |
91% received 57–65 Gy |
|
|
| Centre Alexis Vautrin [37], 2012, 50 | PORT (56%) | 45 Gy (40–56 Gy) |
|
|
| Peter MacCallum [15], 2012, 24 | PreRT (100%) | 45–50.4 Gy |
|
|
| U. Florida [36], 2012, 58 | PreRT (72%) PORT (28%) |
PreRT: 50.4 Gy (36–65 Gy), BID PORT 49.6 Gy (20–70 Gy), OD |
|
|
| Institut Gustave Roussy [38], 2013, 110 | PORT (44%) | 50.4 Gy |
|
|
| Dana-Farber [14], 2013, 33 | PreRT (70%) PreRT + I-125 BRT (30%) |
EBRT: 50 Gy (43.2 – 50.4 Gy) I-125: 77.5 Gy (70–90 Gy) LDR |
|
|
MVA: Multivariate analysis, UCLA: University of California, Los Angeles, U.: University, PreRT: Preoperative radiotherapy, PORT: Postoperative radiotherapy, IORT: Intra-operative radiotherapy, BRT: Brachytherapy, LDR: Low-dose rate, MFH: Malignant fibrous histiocytoma
Other factors significant on MVA not reported
Usage of RT
Multiple SEER analyses and multi-institutional assessments demonstrate that 23–26% of RPS patients diagnosed in the 1990’s and 2000’s, received radiotherapy of whom 80% received PORT and about 5% received PreRT [3, 29–31]. As discussed above, a trend toward offering radiotherapy preoperatively is observed in more modern series. Moreover, radiotherapy use appears to be significantly more common in high volume centers. For example, radiotherapy was used in 62% of RPS patients treated at MSKCC and in 72–77% in the French experience [1, 2, 32].
Treatment Outcomes: Role of RT in RPS
Historically, reported five-year overall survival (OS) for patients with RPS has varied over a broad range of 10–65% [16]. In general, outcomes are worse than those observed in extremity STS. To date, no RCT data unequivocally defines the role of radiation therapy in the management of RPS hence treatment decision-making must rely on knowledge of prognostic factors derived from institutional and population-level data. Table 1 summarizes published studies from the years 2000–2009, describing prognostic factors for local control and survival, in the three dimensional-conformal RT (3D-CRT) era. Usual PreRT, intra-operative RT (IORT) and PORT doses used were 45–50.4, 12–15 and 50–66 Gy, respectively. In these studies, the 5-year local control ranged from 40–80% and 5-year OS ranged from 34–90%. Surgical margin was noted to be a critical prognostic factor for local control and survival in most series. Other poor prognostic factors that have been inconsistently identified include high-grade histology, large tumor size, male gender, older age and avoidance of RT.
Limited data exist to inform decisions regarding the timing of RT for RPS. At the University of Florida, local control and OS were not statistically different between preoperative and postoperative groups [11]. However, the time to local recurrence was different (PORT: 1 year vs PreRT: 2.5 years). In a recent update, there continues to be no local control distinction between the two sequences (described later in this review). At the University of Michigan, recurrences were higher in the PORT group compared to PreRT group (p = 0.046), though this was not a significant factor on multivariate analysis [26]. Similarly, in series from MD Anderson Cancer Center (MDACC) and the Mayo Clinic, no differences in local control or survival were noted between PreRT versus PORT [27, 33].
The study populations in these series are extremely heterogenous with respect to presentation, histologies and radiotherapy sequencing. In addition, due to the retrospective nature, inherent institutional and patient selection biases are unavoidable. Hence, a definite recommendation regarding the role and timing of RT is not possible. In the absence of randomized studies specifically addressing RPS, the role of radiotherapy is largely defined based on treatment principles for extremity STS.
Literature update (2010- current)
In the last three years, several institutions have published their experiences in RPS management. Table 2 describes RT use and factors impacting local control and OS in these studies. The reported 5-year DFS and OS ranged from 19–52% and 32–76%, respectively. There continues to be variations in RT use and sequencing based on institutional preferences. Pertinent features of a few of the larger series are described below:
At the University of California, Los Angeles all patients received neoadjuvant chemotherapy, though timing of RT was variable [34]. The 25% of patients in whom the surgical specimen demonstrated > 95% necrosis had significantly better disease-specific survival (DSS) than the remaining 75% of patients (83% vs 34%, p = 0.002) and also had significantly better outcomes than that predicted by the MSKCC sarcoma nomogram (39%, p = 0.018) [35]. PreRT, however, was not associated with improved DSS.
The University of Florida recently updated their experience with multimodality therapy in RPS [36]. In 58 patients, the 5-year overall survival, cause-specific survival, and local control rates were 49%, 58%, and 62%, respectively. The small size of the postoperatively treated cohort (N = 16) limited the ability to detect significant efficacy or toxicity differences between PreRT and PORT groups.
Fifty patients with primary RPS treated with curative surgery at Centre Alexis Vautrin were analyzed; 5-year OS and disease-free survival (DFS) were 46% and 19%, respectively [37]. PORT was associated with longer time to recurrence (27 vs 13 months, p = 0.05) and had a favorable impact on OS in univariate analysis, however, the significance was lost in multivariate analysis.
At Dana-Farber Cancer Institute, 33 patients with RPS treated with PreRT and radical/en bloc resection were analyzed [14]. 3-year OS, freedom-from progression (FFP) and local recurrence rates (LRR) were 64%, 45% and 37% respectively. The risk of LRR was lower in the group of patients that received I-125 mesh BRT, though this did not reach statistical significance (42% vs 35%, p = NS).
Role of Radiotherapy in the Era of Aggressive Surgical Resection
As discussed, the benefit of RT with conventional surgical approaches remains ill-evidenced. Controversial, in and of itself, is a role for highly aggressive upfront surgical resection that has been suggested to improve outcomes in recent retrospective analyses. We envision an emerging area of controversy regarding the role of RT in RPS management when such highly aggressive surgical approaches are employed. Early experiences describing the impact of RT in the setting of highly aggressive surgical approaches are described below.
In their experience (N = 288) comparing aggressive surgical excision with simple complete resection, the Istituto Nazionale Tumori reported that aggressive resection resulted in a significant reduction in adjusted 5-year local recurrence rate (48% to 29%, HR 0.64, p = 0.02) despite only a modest increase in the rates of R0/R1 resections (87.5% to 90.8%) [13]. In the entire population, the use of RT was an independent factor with a favorable impact on OS (p = 0.008) and recurrence free survival (RFS) (p = 0.057) (table 1). However, between the two groups, the absolute magnitude of OS benefit with RT was smaller with more aggressive surgery, while the benefit in RFS was similar.
The French Association of Surgery reported their multi-institutional experience (N = 382 patients) and showed that in the setting of complete compartmental resection (systematic resection of uninvolved contiguous organs to obtain wide margins), RT did not have an impact on RFS or OS (table 1) [8].
At Institut Gustave Roussy, 48 patients who received adjuvant radiotherapy after having undergone front-line aggressive surgery were compared with 62 patients who underwent surgery alone (table 2) [38• •]. Omentoplasty or placement of expanders was performed to push bowel away from the surgical bed. PORT was offered typically for unfavorable prognostic factors like difficult resections, positive/close margins on frozen section or high-grade tumors. Despite these features, five-year RFS (60% vs 47%, p = 0.02) and the rate of abdominal relapse (22% vs 36%, p = 0.18) appeared better in the RT group. The overall survival was 76% at 5-years for the entire population. The authors noted that improved OS could be attributed to more aggressive surgical resection with 98% having a R0/R1 resection.
As surgical approaches to RPS evolve, the interplay between RT and surgical extent for both oncologic outcomes and quality of life will mandate continuous reassessment.
Recent Reports from Population Databases
As a result of the inherent limitations associated with interpreting outcome data from single institutions, several groups have examined population-level databases.
Four reports have been published using the Surveillance, Epidemiology, and End Results (SEER) database to assess the impact of RT on RPS outcomes. In summary, mixed findings were noted on the survival benefit observed with RT [3, 30, 39, 40•]. There was some suggestion of a benefit for subgroups in individual studies (malignant fibrous histiocytoma [MFH] histology [30•], stage I disease [39••]). As seen in single institution studies, older age, male gender, non-liposarcoma histology, higher grade and incomplete surgery were observed to be associated with poorer outcomes [3]. It is important to note that detailed margin status is unavailable within the SEER database and therefore not included in any of these analyses; thus, it is possible that any benefit of radiation may be masked by this important, unexamined variable.
One registry-based sample used a proprietary cancer registry to examine outcomes in 261 RPS patients with negative or microscopically positive margins (clinical R0 resections) [31••]. 5-year cause-specific and local-failure free survival (LFFS) were 73% and 66% respectively. On multivariate analysis, use of RT in combination with surgery significantly improved LFFS (hazard ratio= 0.42; 95% CI, 0.21–0.86, P<0.05). This improvement was noted mainly for patients undergoing simple resection (88% versus 69% p<0.05); LFFS was similar, with or without RT, for patients undergoing wide resection (67% versus 64% p>0.05). By examining only patients with clinical R0 resections, this study overcomes some of the limitations associated with SEER database analyses and provides supporting evidence for the use of RT in the management of RPS. However, these data also raise the possibility that RT benefits are reduced with more aggressive surgical resection.
Modern Radiotherapy Techniques
The constant evolution of radiotherapy technologies permits new opportunities in RPS management. As noted in the sections above, the major obstacle to the delivery of adjuvant radiotherapy is the dose limitations of surrounding normal tissues. Newer techniques like Intensity Modulated Radiotherapy (IMRT) and particle beam therapy such as proton beam radiotherapy (PBRT) and carbon ion radiotherapy (CIRT) allow more conformal dose distributions to irregularly shaped target volumes and oncologist-directed tailoring of radiation doses. IMRT employs modulated photon beams to achieve better conformality, however, it is limited by the gradual dose fall off of photons (Figure 1). Nonetheless, IMRT is a major advance beyond conventional three-dimensional conformal radiotherapy (3D-CRT) wherein beam shaping was the only tool available to conform radiation doses. In contrast, particle beam therapies exploit physical and/or radiobiological properties to achieve an improved therapeutic index. PBRT achieves highly conformal dose-distributions as a result of the radically reduced exit dose afforded by the Bragg Peak. In RPS, where target volumes abut multiple critical adjacent viscera, PBRT may have significant dosimetric advantages and allow better normal tissue sparing. At the same time multiple air-tissue interfaces and the positional variability of mobile bowel loops adds to the dosimetric uncertainty in actual dose delivered. Techniques for PBRT are still developing and will hopefully reduce these potential limitations. Carbon ions display similar radical reductions in exit dose due to the Bragg Peak. In addition, due to significantly higher linear energy transfer compared to photons and protons, they have higher radiobiological effectiveness, which may be particularly useful for large, hypoxic tumors.
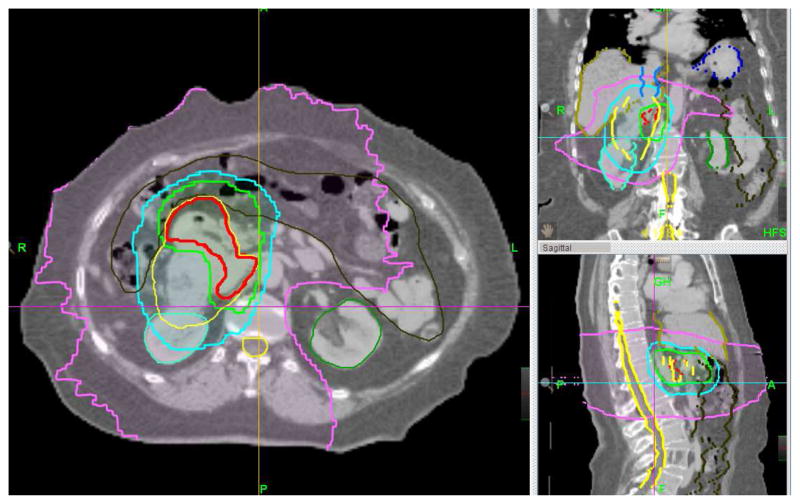
FIGURE 1.
A case demonstrating highly conformal radiotherapy dose-distributions achieved with intensity modulated radiotherapy (IMRT) as displayed in axial, sagittal and coronal images. A 79-year old woman presented with deep abdominal discomfort and lower limb edema leading to a diagnosis of intermediate grade leiomyosarcoma of the inferior vena cava. After multidisciplinary assessment, preoperative radiotherapy using TomoTherapy-based IMRT was offered. A differential dose of 50 Gy to the surgeon-defined, at-risk margin (red contour) and 45 Gy to the entire GTV plus PTV margin (yellow contour) was delivered sparing contralateral kidney (green contour), bowel/peritoneal cavity (black contour), liver and spinal cord. Ipsilateral kidney (light blue) was sacrificed as part of the planned surgical resection. 50 Gy, 40 Gy and 15 Gy isodose lines are shown in green, blue and pink, respectively. The patient remains disease- and toxicity-free two and half years after treatment.
Intensity Modulated Radiotherapy (IMRT) experience
In a small City of Hope experience, 14 patients (42.5%) were treated with IMRT-based PORT while 19 patients (57.5%) received conventional RT [41]. The tumor surgical bed clinical target volume (CTV-2) with PTV margins was planned to a total dose of 60–65 Gy depending on degree of resection, while a surrounding larger volume of resection bed plus 5 cm margins modified around natural barriers (CTV-1) was planned to 45 Gy. IORT was used to plan the boost in 20 (61%) patients. None of the patients planned with IMRT developed grade 3/4 acute GI toxicity, compared with 16% patients treated with 3D conformal techniques (p = NS). IMRT reduced patient weight loss during treatment (−0.1% with IMRT vs −3.5% for conventional RT, p=0.009). Amongst patients with a minimum 15 months follow-up, absolute incidence of late grade 3/4 toxicities was also lower in IMRT treated patients (10% vs 27%, p = NS).
The University of Alabama experience with IMRT and selective integrated boosting is described below [42].
At University Hospitals Gasthuisberg, an interesting approach was employed to use IMRT to deliver meaningful radiation dose (50 Gy) only to the surface of contact between the tumor mass and the posterior abdominal wall [43]. This is the highest risk area for relapse, often due to challenges achieving negative surgical margins. No intentional attempt was made to include the entire gross target volume (GTV) in the treatment volume. The abdominal wall posterior to the great vessels and the ipsilateral paravertebral space were included in the CTV, which was expanded by 3 cm to generate a PTV. Of 18 patients treated on the pilot study, 11% experienced grade 3, acute anorexia during RT while one patient (5.5%) developed peri-operative gastric hemorrhage. Local recurrence was noted in two patients (11%). Overall, the authors supported this approach to treat RPS preoperatively. In addition to the clinical study, a dosimetric study compared 3DCRT and IMRT plans and demonstrated significantly reduced kidney doses and improved PTV doses when IMRT was employed.
Paumier et al reported a dosimetric comparison of 3DCRT, 6-field coplanar IMRT (6b-IMRT) and 9-field coplanar IMRT (9b-IMRT) for adjuvant RT in 10 RPS patients [44•]. Interestingly, PTV underdosing was more common in IMRT plans though the overall conformity index was twice as good when compared with 3DCRT. Intestinal cavity, stomach and contralateral kidney volumes receiving moderate-to-high doses were significantly lower with IMRT plans though the low dose spread with IMRT resulted in significantly higher volumes receiving low doses. No significant differences were noted in mean liver doses.
Particle Beam Radiotherapy: Proton and Carbon ion experience
MGH recently reported their experience in 28 patients treated with IMRT or PBRT [45]. Twenty (71%) patients were planned for PreRT to a median dose of 50 Gy. Six patients (21%) received PORT to a median dose of 50.2 Gy. Following PreRT, patients underwent aggressive surgical resection with nearly 80% requiring removal of contiguous organs. Positive surgical margins were identified in 54% of resected patients. 43% of patients received IORT (median dose, 11 Gy). Radiotherapy related complications were noted in 14% of patients including ureteral stricture, bleeding, entero-cutaneous fistula and infected seroma, LR developed in 18% patients with a median time to LR of 12.3 months. On univariate analysis, pelvic site of disease and recurrent disease at presentation were associated with increased risk of LR. 3-year LRFS for primary and recurrent tumors was 90% and 30%, respectively. Interestingly, margin status was not a significant prognostic factor for local recurrence. 3-year OS was 87%.
Swanson et al performed a preoperative, dosimetric study comparing 3D conformal proton RT (3DCPT), 3D conformal photon RT (3DCRT) and IMRT, in 8 patients with retroperitoneal or intra-abdominal sarcomas [46•]. Target volume coverage was comparable. The conformity index was significantly better for IMRT and 3DCPT compared to 3DCRT and IMRT was borderline superior to 3DCPT (p = 0.052). 3DCPT plans delivered the lowest bowel, contralateral kidney and liver doses.
There is limited experience with heavy ions in the management of RPS. The National Institute of Radiological Sciences in Chiba, reported 24 patients with unresectable RPS who were treated with carbon ion RT (CIRT) to a dose of 52.8 to 73.6 GyE in 16 fractions over four weeks [47]. An encouraging 5-year survival of 50% and local control of 69% was noted despite unresectable disease. No significant GI toxicities were seen.
Radiotherapy Dose-response
For extremity STS, doses of 60–66 Gy are recommended to treat the postoperative surgical bed. Preoperatively, however, a dose of 50 Gy appears adequate [28]. There has been some suggestion of a dose-response relationship in RPS. A National Cancer Institute, prospective, randomized trial in 35 patients, compared IORT (20 Gy) and low-dose PORT (35–40 Gy) with conventional PORT (50–55 Gy) [24]. Reduced local failures and lower GI toxicities were noted in the IORT arm; however, peripheral neuropathy was more common in the IORT arm. Even in the MGH retrospective experience, there was a trend towards improved local control with addition of IOERT to PreRT (83% vs 61% at 5-years), although this did not reach statistical significance [9]. The IOERT dose in this series was dependent on surgical margins (R0: 10 Gy, R1: 12.5–15 Gy, R2: 15–20 Gy). In a combined MDACC and Princess Margaret Hospital (PMH) experience, the use of IORT (BRT: 25 Gy, IOERT: 15 Gy) in addition to PreRT (median 45 Gy) did not significantly impact DFS or OS [48]. In small institutional series, a dose threshold of 50 Gy (MDACC, Institut Bergonié) and 55 Gy (Fox Chase, MSKCC and U. Michigan) has been noted [1, 2, 25–27]. The shape of the dose-response curve is even more pertinent in the era of modern, highly conformal radiation that safely allows delivery of higher doses while limiting normal tissue toxicity.
Prospective dose-escalation studies
Tzeng and colleagues at the University of Alabama reported outcomes of 16 patients prospectively treated with preoperative, selective dose-escalation to the margin at presumed risk [42]. An IMRT-based, simultaneous integrated boost technique was used to prescribe 57.5 Gy to the margin at risk while delivering 45 Gy to lower risk volumes in 25 fractions. The treatment was well tolerated with only one patient having acute grade 3, GI toxicity. Nearly 90% patients had complete macroscopic resection, with two patients unable to undergo resection due to detection of peritoneal implants at laparotomy. Only two patients amongst those who had complete macroscopic resection developed recurrent disease, with a 2-year actuarial local control rate of 80%. A companion treatment planning study to assess dosimetric feasibility of further dose-escalation to the margin at risk suggested the high-risk planning target volume (PTV) could be dose-escalated to 70 Gy without compromising a small bowel dose limit of V54 Gy < 20 cc.
At the German Cancer Research Center (DKFZ), Heidelberg, a phase I/II study is being conducted evaluating preoperative, dose-escalated IMRT (50–56 Gy to GTV plus 1.5 cm expansion to generate an anatomically modified CTV) to be followed by IORT boost (10–12 Gy to tumor bed) in patients with resectable or borderline resectable RPS [49•]. A wider PTV was prescribed to 45–50 Gy. Final results of this study are awaited.
Radiotherapy Volumes and Dose-constraints
Optimal radiotherapy volumes for RPS treatment are not well-defined. Generally a policy to include the surgical bed with appropriate margins to cover microscopic disease while avoiding critical structures has been followed for PORT. In the 3D-planning era, PORT is typically targeted to a 3–5 cm expansion on the tumor bed with appropriate anatomic modifications [9, 26, 37, 50]. When planning PreRT, the GTV is expanded using similar margins to generate a PTV [9, 26].
With increasing use of highly conformal radiotherapy techniques, careful radiation planning, attention to normal tissue tolerances and reproducible positioning of both target and normal tissues are critical. In the University of Alabama study, the high risk target volume and the contralateral kidney (V23Gy < 33%) were given the highest priority followed by spinal cord (Dmax < 45 Gy) then low risk target volume, liver (whole liver < 33 Gy) and small bowel (Dmax < 45 Gy) although the latter had an absolute dose limit of V54Gy < 20 cc [42]. In the MGH study, normal tissue dose constraints were cord Dmax < 48 Gy, contralateral kidney V20 < 33%, liver V25Gy < 50% and V30 Gy < 40%, small bowel Dmax preferably < 50.4 Gy, though small volumes were allowed to reach 57.6 Gy if the dose was falling across the bowel, stomach Dmax < 55 Gy and colon Dmax < 60 Gy [45]. The planning dose constraints for IMRT in the Paumier et al dosimetric study for a planned dose of 50.4 Gy/28 fractions were V95% > 95% of PTV, cord Dmax < 45 Gy, contralateral kidney mean dose < 6 Gy and V10Gy < 5%, bilateral kidney V20Gy < 50%, median dose to intestinal cavity < 25 Gy with V50Gy < 5%, stomach Dmax < 54 Gy with V50Gy < 5% and liver mean < 26 Gy with V30Gy< 50% [44]. The dosimetric goals in the Swanson et al study were to achieve a CTV coverage such that V100% > 95%, V97% > 99% and V110% < 20% [46]. Normal tissue constraints were to limit spinal cord V50Gy < 0.1cc, each kidney V20 Gy < 50% and V35 Gy < 33% (if not part of CTV), mean liver dose < 30 Gy, bowel space minus CTV V40Gy < 170 cc, V45Gy < 100 cc, and V50Gy < 33 cc.
Importance of Multidisciplinary Input for RT planning
Inputs from the operating surgeon and diagnostic radiologists are critical for PReRT planning. With modern techniques that allow tailoring of radiation dose distributions, several surgical factors can guide modification of radiation plans. Diagnostic studies should be employed to assess involvement of adjacent normal viscera. Based on this information, the team should develop an operative plan prior to initiation of radiation. For example, if nephrectomy is anticipated, exquisite attention must be given to ensuring adequate contralateral kidney function is present and preserved. If escalated radiation doses are contemplated, high-risk areas for potentially positive margins should be defined in concert with diagnostic radiologists and the operating surgeon. If complex anastomoses are anticipated, radiation doses may be tailored to limit dose to the regions of interest and reduce risk of anastomotic leak.
For patients taken directly for resection, the radiation oncologist should be included in upfront treatment planning. Surgeons may improve PORT planning by placement of surgical clips in areas of suspected positive margins. Moreover, surgeons may perform omentoplasty or placement of tissue expanders to displace bowel and improve normal tissue dosimetry with PORT.
Future Trends
For RPS, prospective studies that unequivocally define the optimal extent of surgical resection, establish the need for radiotherapy and clarify the ideal sequencing of radiotherapy are lacking. The American College of Surgeons Oncology Group (ACOSOG) initiated a phase 3 randomized trial that compared preoperative RT and surgery with surgery alone. Unfortunately, the trial had to be closed early due to poor accrual. This question is currently being studied in a randomized controlled trial through the European Organization for the Research and Treatment of Cancer (EORTC 62092-22092), which, if completed, will significantly advance our approach to RPS. Clearly, one of the key factors in addressing the unanswered questions in RPS management and advancing our approach to this disease will be the support and initiation of additional, tractable clinical trials.
Similarly, roles for chemotherapy in RPS management are unclear. It is generally believed that chemotherapy efficacy is more dependent on histology and biological behavior than anatomic location. Thus, results from studies in non-retroperitoneal soft-tissue sarcoma may be applicable. However, an impact of anatomic location on soft-tissue sarcoma chemosensitivity cannot be completely disregarded [51–53]. Moreover, with the very large tumors characteristic of RPS, issues of bioavailability may require consideration. Finally, the use of systemic therapy as a radiosensitizer must also be cautiously approached, as normal tissue constraints in RPS are more pressing than in most other soft-tissue sarcoma sites.
Opinion statement.
Retroperitoneal sarcomas form a group of rare malignancies that require expertise in every aspect of management. Patients benefit from referral to cancer centers that can provide comprehensive, multi-disciplinary, oncologic management. The role of radiation in retroperitoneal sarcoma management is, appropriately, the subject of considerable controversy due to the absence of high-level evidence proving its efficacy. Nonetheless, the preponderance of available data suggest radiation therapy likely improves local control and may, in some settings, favorably impact resectability and survival. These outcome observations coupled with the lower doses (45–54 Gy) and normal tissue displacement characteristic of preoperative radiation therapy leads us to favor preoperative radiotherapy followed by oncologic resection for most retroperitoneal sarcomas. This strategy appears to provide the highest chance of safe and successful delivery of multi-modal therapy, which can otherwise be hindered by postoperative complications as a result of technically challenging surgery and normal tissue radiation dose tolerances. Dose-escalation and selective integrative boosts to “at risk” margins are attractive strategies that merit, and arguably require, further clinical evaluation. We believe postoperative radiotherapy should be reserved for very high-risk cases and should be treated to a dose of ≥ 60 Gy respecting normal tissue dose tolerances. An additional approach we consider in the postoperative setting is close surveillance with consideration of preoperative radiotherapy at recurrence before repeat surgical resection. Highly conformal radiotherapy techniques like IMRT with IGRT should be employed to minimize dose to normal tissues and thereby allow delivery of efficacious radiation doses. If feasible, referral to a treatment facility with proton beam therapy should be discussed with the patient, especially if normal tissue dose constraints cannot be met using IMRT/IGRT. Participation in prospective studies should be highly encouraged.
Footnotes
Conflicts of Interest
Pranshu Mohindra reports no conflicts of interest.
Heather B. Neuman reports no conflicts of interest.
Kevin R. Kozak reports no conflicts of interest.
Contributor Information
Pranshu Mohindra, Email: pmohindra@uwhealth.org, Department of Human Oncology, University of Wisconsin Hospital and Clinics, 600 Highland Avenue, K4/B100, Madison, Wisconsin, 53792, Tel: 608-263-8500, Fax: 608-263-9167.
Heather B. Neuman, Email: neuman@surgery.wisc.edu, Department of Surgery, University of Wisconsin School of Medicine and Public Health, 600 Highland, Avenue, K4/B100, Madison, Wisconsin, 53792, Tel: 608-265-0500, Fax: 608-263-9167.
Kevin R. Kozak, Email: kevin_kozak@yahoo.com, Department of Human Oncology, University of Wisconsin School of Medicine and Public Health, 600 Highland Avenue, K4/B100, Madison, Wisconsin, 53792, Tel: 608-263-8500, Fax: 608-263-9167.
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as:
• Of importance
•• Of major importance
- 1.Lewis JJ, Leung D, Woodruff JM, Brennan MF. Retroperitoneal soft-tissue sarcoma: analysis of 500 patients treated and followed at a single institution. Ann Surg. 1998;228(3):355–65. doi: 10.1097/00000658-199809000-00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gilbeau L, Kantor G, Stoeckle E, et al. Surgical resection and radiotherapy for primary retroperitoneal soft tissue sarcoma. Radiother Oncol. 2002;65(3):137–43. doi: 10.1016/s0167-8140(02)00283-9. [DOI] [PubMed] [Google Scholar]
- 3.Nathan H, Raut CP, Thornton K, et al. Predictors of survival after resection of retroperitoneal sarcoma: a population-based analysis and critical appraisal of the AJCC staging system. Ann Surg. 2009;250(6):970–6. doi: 10.1097/SLA.0b013e3181b25183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mullinax JE, Zager JS, Gonzalez RJ. Current diagnosis and management of retroperitoneal sarcoma. Cancer Control. 2011;18(3):177–87. doi: 10.1177/107327481101800305. [DOI] [PubMed] [Google Scholar]
- 5.Chowdhury MM, Dagash H, Pierro A. A systematic review of the impact of volume of surgery and specialization on patient outcome. Br J Surg. 2007;94(2):145–61. doi: 10.1002/bjs.5714. [DOI] [PubMed] [Google Scholar]
- 6•.Learn PA, Bach PB. A decade of mortality reductions in major oncologic surgery: the impact of centralization and quality improvement. Med Care. 2010;48(12):1041–9. doi: 10.1097/MLR.0b013e3181f37d5f. Mortality reduction seen in the analysis presented in this study stresses the need to refer patients needing complicated surgery to tertiary care centers. Significant mortality reductions is noted. [DOI] [PubMed] [Google Scholar]
- 7.Gutierrez JC, Perez EA, Franceschi D, et al. Outcomes for soft-tissue sarcoma in 8249 cases from a large state cancer registry. J Surg Res. 2007;141(1):105–14. doi: 10.1016/j.jss.2007.02.026. [DOI] [PubMed] [Google Scholar]
- 8••.Bonvalot S, Rivoire M, Castaing M, et al. Primary retroperitoneal sarcomas: a multivariate analysis of surgical factors associated with local control. J Clin Oncol. 2009;27(1):31–7. doi: 10.1200/JCO.2008.18.0802. This multi-institutional, French experience describes importance of aggressive surgical resection but unlike the Italian experience (Gronchi et al.) raises questions on the role of radiotherapy in this setting. [DOI] [PubMed] [Google Scholar]
- 9.Gieschen HL, Spiro IJ, Suit HD, et al. Long-term results of intraoperative electron beam radiotherapy for primary and recurrent retroperitoneal soft tissue sarcoma. Int J Radiat Oncol Biol Phys. 2001;50(1):127–31. doi: 10.1016/s0360-3016(00)01589-3. [DOI] [PubMed] [Google Scholar]
- 10.Youssef E, Fontanesi J, Mott M, et al. Long-term outcome of combined modality therapy in retroperitoneal and deep-trunk soft-tissue sarcoma: analysis of prognostic factors. Int J Radiat Oncol Biol Phys. 2002;54(2):514–9. doi: 10.1016/s0360-3016(02)02942-5. [DOI] [PubMed] [Google Scholar]
- 11.Zlotecki RA, Katz TS, Morris CG, et al. Adjuvant radiation therapy for resectable retroperitoneal soft tissue sarcoma: the University of Florida experience. Am J Clin Oncol. 2005;28(3):310–6. doi: 10.1097/01.coc.0000158441.96455.31. [DOI] [PubMed] [Google Scholar]
- 12.Krempien R, Roeder F, Oertel S, et al. Intraoperative electron-beam therapy for primary and recurrent retroperitoneal soft-tissue sarcoma. Int J Radiat Oncol Biol Phys. 2006;65(3):773–9. doi: 10.1016/j.ijrobp.2006.01.028. [DOI] [PubMed] [Google Scholar]
- 13••.Gronchi A, Lo Vullo S, Fiore M, et al. Aggressive surgical policies in a retrospectively reviewed single-institution case series of retroperitoneal soft tissue sarcoma patients. J Clin Oncol. 2009;27(1):24–30. doi: 10.1200/JCO.2008.17.8871. This is a modern, single-institutional experience describing impact of aggressive surgical resection in improving local control rates for RPS. Survival benefit was seen with addition of RT in the population, though benefit was smaller with more aggressive resection. [DOI] [PubMed] [Google Scholar]
- 14•.McBride SM, Raut CP, Lapidus M, et al. Locoregional Recurrence After Preoperative Radiation Therapy for Retroperitoneal Sarcoma: Adverse Impact of Multifocal Disease and Potential Implications of Dose Escalation. Ann Surg Oncol. 2013 doi: 10.1245/s10434-013-2868-y. Epub ahead of print. In this recent series, poor prognostic implication of multifocality is noted on multivariate analysis for RFS. [DOI] [PubMed] [Google Scholar]
- 15.Alford S, Choong P, Chander S, et al. Outcomes of preoperative radiotherapy and resection of retroperitoneal sarcoma. ANZ J Surg. 2012 doi: 10.1111/j.1445-2197.2012.06211.x. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 16.Van De Voorde L, Delrue L, van Eijkeren M, De Meerleer G. Radiotherapy and surgery-an indispensable duo in the treatment of retroperitoneal sarcoma. Cancer. 2011;117(19):4355–64. doi: 10.1002/cncr.26071. [DOI] [PubMed] [Google Scholar]
- 17.Strauss DC, Hayes AJ, Thomas JM. Retroperitoneal tumours: review of management. Ann R Coll Surg Engl. 2011;93(4):275–80. doi: 10.1308/003588411X571944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bevilacqua RG, Rogatko A, Hajdu SI, Brennan MF. Prognostic factors in primary retroperitoneal soft-tissue sarcomas. Arch Surg. 1991;126(3):328–34. doi: 10.1001/archsurg.1991.01410270072012. [DOI] [PubMed] [Google Scholar]
- 19.Singer S, Corson JM, Demetri GD, et al. Prognostic factors predictive of survival for truncal and retroperitoneal soft-tissue sarcoma. Ann Surg. 1995;221(2):185–95. doi: 10.1097/00000658-199502000-00009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kilkenny JW, 3rd, Bland KI, Copeland EM., 3rd Retroperitoneal sarcoma: the University of Florida experience. J Am Coll Surg. 1996;182(4):329–39. [PubMed] [Google Scholar]
- 21.Shiloni E, Szold A, White DE, Freund HR. High-grade retroperitoneal sarcomas: role of an aggressive palliative approach. J Surg Oncol. 1993;53(3):197–203. doi: 10.1002/jso.2930530315. [DOI] [PubMed] [Google Scholar]
- 22.McGrath PC, Neifeld JP, Lawrence W, Jr, et al. Improved survival following complete excision of retroperitoneal sarcomas. Ann Surg. 1984;200(2):200–4. doi: 10.1097/00000658-198408000-00014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Glenn J, Sindelar WF, Kinsella T, et al. Results of multimodality therapy of resectable soft-tissue sarcomas of the retroperitoneum. Surgery. 1985;97(3):316–25. [PubMed] [Google Scholar]
- 24.Sindelar WF, Kinsella TJ, Chen PW, et al. Intraoperative radiotherapy in retroperitoneal sarcomas. Final results of a prospective, randomized, clinical trial. Arch Surg. 1993;128(4):402–10. doi: 10.1001/archsurg.1993.01420160040005. [DOI] [PubMed] [Google Scholar]
- 25.Fein DA, Corn BW, Lanciano RM, et al. Management of retroperitoneal sarcomas: does dose escalation impact on locoregional control? Int J Radiat Oncol Biol Phys. 1995;31(1):129–34. doi: 10.1016/0360-3016(94)E0302-Z. [DOI] [PubMed] [Google Scholar]
- 26.Feng M, Murphy J, Griffith KA, et al. Long-term outcomes after radiotherapy for retroperitoneal and deep truncal sarcoma. Int J Radiat Oncol Biol Phys. 2007;69(1):103–10. doi: 10.1016/j.ijrobp.2007.02.041. [DOI] [PubMed] [Google Scholar]
- 27.Ballo MT, Zagars GK, Pollock RE, et al. Retroperitoneal soft tissue sarcoma: an analysis of radiation and surgical treatment. Int J Radiat Oncol Biol Phys. 2007;67(1):158–63. doi: 10.1016/j.ijrobp.2006.08.025. [DOI] [PubMed] [Google Scholar]
- 28.O’Sullivan B, Davis AM, Turcotte R, et al. Preoperative versus postoperative radiotherapy in soft-tissue sarcoma of the limbs: a randomised trial. Lancet. 2002;359(9325):2235–41. doi: 10.1016/S0140-6736(02)09292-9. [DOI] [PubMed] [Google Scholar]
- 29.Porter GA, Baxter NN, Pisters PW. Retroperitoneal sarcoma: a population-based analysis of epidemiology, surgery, and radiotherapy. Cancer. 2006;106(7):1610–6. doi: 10.1002/cncr.21761. [DOI] [PubMed] [Google Scholar]
- 30•.Tseng WH, Martinez SR, Do L, et al. Lack of survival benefit following adjuvant radiation in patients with retroperitoneal sarcoma: a SEER analysis. J Surg Res. 2011;168(2):e173–80. doi: 10.1016/j.jss.2011.02.004. In this SEER analysis, survival advantage was suggested with use of RT in intermediate grade and MFH histology patients. [DOI] [PubMed] [Google Scholar]
- 31••.Sampath S, Hitchcock YJ, Shrieve DC, et al. Radiotherapy and extent of surgical resection in retroperitoneal soft-tissue sarcoma: multi-institutional analysis of 261 patients. J Surg Oncol. 2010;101(5):345–50. doi: 10.1002/jso.21474. In this large multi-institutional experience, use of RT improved local control rates when performing a simple surgical resection, while no benefit was seen if a wide resection wsa performed. [DOI] [PubMed] [Google Scholar]
- 32.Stoeckle E, Coindre JM, Bonvalot S, et al. Prognostic factors in retroperitoneal sarcoma: a multivariate analysis of a series of 165 patients of the French Cancer Center Federation Sarcoma Group. Cancer. 2001;92(2):359–68. doi: 10.1002/1097-0142(20010715)92:2<359::aid-cncr1331>3.0.co;2-y. [DOI] [PubMed] [Google Scholar]
- 33.Petersen IA, Haddock MG, Donohue JH, et al. Use of intraoperative electron beam radiotherapy in the management of retroperitoneal soft tissue sarcomas. Int J Radiat Oncol Biol Phys. 2002;52(2):469–75. doi: 10.1016/s0360-3016(01)02595-0. [DOI] [PubMed] [Google Scholar]
- 34.Donahue TR, Kattan MW, Nelson SD, et al. Evaluation of neoadjuvant therapy and histopathologic response in primary, high-grade retroperitoneal sarcomas using the sarcoma nomogram. Cancer. 2010;116(16):3883–91. doi: 10.1002/cncr.25271. [DOI] [PubMed] [Google Scholar]
- 35.Kattan MW, Leung DH, Brennan MF. Postoperative nomogram for 12-year sarcoma-specific death. J Clin Oncol. 2002;20(3):791–6. doi: 10.1200/JCO.2002.20.3.791. [DOI] [PubMed] [Google Scholar]
- 36.Paryani NN, Zlotecki RA, Swanson EL, et al. Multimodality local therapy for retroperitoneal sarcoma. Int J Radiat Oncol Biol Phys. 2012;82(3):1128–34. doi: 10.1016/j.ijrobp.2011.04.009. [DOI] [PubMed] [Google Scholar]
- 37.Fuks D, Verhaeghe JL, Marchal F, et al. Surgery and postoperative radiation therapy in primary retroperitoneal sarcomas: experience of the cancer centre Alexis-Vautrin. Cancer Radiother. 2012;16(3):194–200. doi: 10.1016/j.canrad.2011.11.006. [DOI] [PubMed] [Google Scholar]
- 38••.Le Pechoux C, Musat E, Baey C, et al. Should adjuvant radiotherapy be administered in addition to front-line aggressive surgery (FAS) in patients with primary retroperitoneal sarcoma? Ann Oncol. 2013;24(3):832–7. doi: 10.1093/annonc/mds516. This is a modern series that describes use of PORT in setting of aggressive surgical resection. Despite selection bias with unfavorble histological features in the RT arm, relapse-free survival and abdominal relapse was significantly lower in the RT arm. In this series, tissue expanders or omentoplasty was used to displace bowel from surgical bed. [DOI] [PubMed] [Google Scholar]
- 39••.Zhou Z, McDade TP, Simons JP, et al. Surgery and radiotherapy for retroperitoneal and abdominal sarcoma: both necessary and sufficient. Arch Surg. 2010;145(5):426–31. doi: 10.1001/archsurg.2010.70. In this SEER analysis, after stage adjustment, survival advantage was noted with use of RT in stage I but not for stage II/III. [DOI] [PubMed] [Google Scholar]
- 40•.Choi AH, Barnholtz-Sloan JS, Kim JA. Effect of radiation therapy on survival in surgically resected retroperitoneal sarcoma: a propensity score-adjusted SEER analysis. Ann Oncol. 2012;23(9):2449–57. doi: 10.1093/annonc/mdr616. To correct for heterogeneity in patient populatioin noted in all SEER data analyses, the authors performed a propensity-score adjusted, matched-pair analysis to demonstrate that RT did not contribute to survival. Impact on local control was not analysable in this database. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Pezner RD, Liu A, Han C, et al. Dosimetric comparison of helical tomotherapy treatment and step-and-shoot intensity-modulated radiotherapy of retroperitoneal sarcoma. Radiother Oncol. 2006;81(1):81–7. doi: 10.1016/j.radonc.2006.08.025. [DOI] [PubMed] [Google Scholar]
- 42.Tzeng CW, Fiveash JB, Popple RA, et al. Preoperative radiation therapy with selective dose escalation to the margin at risk for retroperitoneal sarcoma. Cancer. 2006;107(2):371–9. doi: 10.1002/cncr.22005. [DOI] [PubMed] [Google Scholar]
- 43.Bossi A, De Wever I, Van Limbergen E, Vanstraelen B. Intensity modulated radiation-therapy for preoperative posterior abdominal wall irradiation of retroperitoneal liposarcomas. Int J Radiat Oncol Biol Phys. 2007;67(1):164–70. doi: 10.1016/j.ijrobp.2006.08.023. [DOI] [PubMed] [Google Scholar]
- 44•.Paumier A, Le Pechoux C, Beaudre A, et al. IMRT or conformal radiotherapy for adjuvant treatment of retroperitoneal sarcoma? Radiother Oncol. 2011;99(1):73–8. doi: 10.1016/j.radonc.2011.02.002. In this dosimetric study, superiority of IMRT over 3D-conformal RT to reduce radiation dose to normal tissues is demonstrated. Dose-constraints presented in the study may be referred to for IMRT planning. [DOI] [PubMed] [Google Scholar]
- 45•.Yoon SS, Chen YL, Kirsch DG, et al. Proton-beam, intensity-modulated, and/or intraoperative electron radiation therapy combined with aggressive anterior surgical resection for retroperitoneal sarcomas. Ann Surg Oncol. 2010;17(6):1515–29. doi: 10.1245/s10434-010-0935-1. A modern series describing clinical experiene with use of IMRT and Proton beam RT in management of RPS. Dose-constraints presented in the study may be referred to for IMRT and proton beam RT planning. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46•.Swanson EL, Indelicato DJ, Louis D, et al. Comparison of three-dimensional (3D) conformal proton radiotherapy (RT), 3D conformal photon RT, and intensity-modulated RT for retroperitoneal and intra-abdominal sarcomas. Int J Radiat Oncol Biol Phys. 2012;83(5):1549–57. doi: 10.1016/j.ijrobp.2011.10.014. This study demonstrates dosimetric superiority of 3D-conformal proton beam RT over IMRT and 3D-conformal photon RT. Dose-constraints presented in the study may be referred to for IMRT planning. [DOI] [PubMed] [Google Scholar]
- 47.Serizawa I, Kagei K, Kamada T, et al. Carbon ion radiotherapy for unresectable retroperitoneal sarcomas. Int J Radiat Oncol Biol Phys. 2009;75(4):1105–10. doi: 10.1016/j.ijrobp.2008.12.019. [DOI] [PubMed] [Google Scholar]
- 48.Pawlik TM, Pisters PW, Mikula L, et al. Long-term results of two prospective trials of preoperative external beam radiotherapy for localized intermediate- or high-grade retroperitoneal soft tissue sarcoma. Ann Surg Oncol. 2006;13(4):508–17. doi: 10.1245/ASO.2006.05.035. [DOI] [PubMed] [Google Scholar]
- 49•.Roeder F, Schulz-Ertner D, Nikoghosyan AV, et al. A clinical phase I/II trial to investigate preoperative dose-escalated intensity-modulated radiation therapy (IMRT) and intraoperative radiation therapy (IORT) in patients with retroperitoneal soft tissue sarcoma. BMC Cancer. 2012;12:287. doi: 10.1186/1471-2407-12-287. This is a prospective phase I/II study aiming to use dose-escalated IMRT along with IORT to improve treatment outcomes. Results of this study are awaited. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Lee HJ, Song SY, Kwon TW, et al. Treatment outcome of postoperative radiotherapy for retroperitoneal sarcoma. Radiat Oncol J. 2011;29(4):260–8. doi: 10.3857/roj.2011.29.4.260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Hensley ML, Maki R, Venkatraman E, et al. Gemcitabine and docetaxel in patients with unresectable leiomyosarcoma: results of a phase II trial. J Clin Oncol. 2002;20(12):2824–31. doi: 10.1200/JCO.2002.11.050. [DOI] [PubMed] [Google Scholar]
- 52.Maki RG, Wathen JK, Patel SR, et al. Randomized phase II study of gemcitabine and docetaxel compared with gemcitabine alone in patients with metastatic soft tissue sarcomas: results of sarcoma alliance for research through collaboration study 002 [corrected] J Clin Oncol. 2007;25(19):2755–63. doi: 10.1200/JCO.2006.10.4117. [DOI] [PubMed] [Google Scholar]
- 53.Pautier P, Floquet A, Penel N, et al. Randomized multicenter and stratified phase II study of gemcitabine alone versus gemcitabine and docetaxel in patients with metastatic or relapsed leiomyosarcomas: a Federation Nationale des Centres de Lutte Contre le Cancer (FNCLCC) French Sarcoma Group Study (TAXOGEM study) Oncologist. 2012;17(9):1213–20. doi: 10.1634/theoncologist.2011-0467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Jones JJ, Catton CN, O’Sullivan B, et al. Initial results of a trial of preoperative external-beam radiation therapy and postoperative brachytherapy for retroperitoneal sarcoma. Ann Surg Oncol. 2002;9(4):346–54. doi: 10.1007/BF02573869. [DOI] [PubMed] [Google Scholar]
- 55.White JS, Biberdorf D, DiFrancesco LM, et al. Use of tissue expanders and pre-operative external beam radiotherapy in the treatment of retroperitoneal sarcoma. Ann Surg Oncol. 2007;14(2):583–90. doi: 10.1245/s10434-006-9139-0. [DOI] [PubMed] [Google Scholar]
- 56.Pezner RD, Liu A, Chen YJ, et al. Full-dose adjuvant postoperative radiation therapy for retroperitoneal sarcomas. Am J Clin Oncol. 2011;34(5):511–6. doi: 10.1097/COC.0b013e3181f4796d. [DOI] [PubMed] [Google Scholar]