Abstract
A Multiplex Polymerase Chain Reaction (PCR) assay to be used as an alternative to the conventional culture method in detecting Shigella and enteroinvasive Escherichia coli (EIEC) virulence genes ipaH and ial in lettuce was developed. Efficacy and rapidity of the molecular method were determined as compared to the conventional culture. Lettuce samples were inoculated with different Shigella flexneri concentrations (from 10 CFU/ml to 107 CFU/ml). DNA was extracted directly from lettuce after inoculation (direct-PCR) and after an enrichment step (enrichment PCR). Multiplex PCR detection limit was 104CFU/ml, diagnostic sensitivity and specificity were 100% accurate. An internal amplification control (IAC) of 100 bp was used in order to avoid false negative results. This method produced results in 1 to 2 days while the conventional culture method required 5 to 6 days. Also, the culture method detection limit was 106 CFU/ml, diagnostic sensitivity was 53% and diagnostic specificity was 100%. In this study a Multiplex PCR method for detection of virulence genes in Shigella and EIEC was shown to be effective in terms of diagnostic sensitivity, detection limit and amount of time as compared to Shigella conventional culture.
Keywords: PCR, Shigella, rapid method, lettuce, food
INTRODUCTION
According to the World Health Organization, foodborne diseases represent a major public health problem which has increased in the last decade. The ingestion of pathogenic and virulent microorganisms generally results in foodborne illnesses affecting populations in both developed and developing countries (28).
Shigella spp. is a virulent bacterium that belongs to the Enterobacteriaceae family. The infectious dose of Shigella is as low as 10 bacterial cells (1,2,9). The transmission occurs person to person through the faecal-oral pathway, and also by contaminated food and water (4). The symptoms of Shigella infection (shigellosis) range from mild watery diarrhea to severe bacillary dysentery with fever, abdominal pain, blood and mucus in stool samples (21).
Only three out of the four recognized Shigella species (S. flexneri, S. boydii and S.sonnei) are known to be etiological agents of foodborne gastroenteritis (3, 13). S. dysenteriae was described by Shiga in 1898, and is the primary cause of bacillary dysentery (9).
Foodborne diseases, including shigellosis, are substantially underestimated, particularly in developing countries (9). Although 99% of the cases of shigellosis occur in the developing world, industrialized countries such as Norway, Denmark, Australia, Portugal and the United States, among others, have reported outbreaks of foodborne shigellosis in the last decade (9, 13, 28). In the United States, Shigella is reported as the third highest cause of foodborne disease since 1997, after Salmonella and Campylobacter (26).
In Costa Rica, Shigella has been isolated from faecal, water and food samples (7,8,27). Other studies confirm that Shigella spp. is an endemic pathogen in Central American countries (R.Achí et al.,manuscript in preparation), causing severe diarrheal disease that requires hospitalization in young children (2).
Traditional microbiological assays for detection of Shigella in food include enrichment, culture, and biotype- and serotype identification steps; all of which are time consuming, requiring several days to obtain results. On the other hand, Shigella is considered a fastidious pathogen for bacteriological isolation, which in the context of indigenous micro flora and other substances makes detection less feasible (9,16,19). However, the advent of molecular biology assays, such as the Polymerase Chain Reaction (PCR), has made bacterial detection possible without the need for bacteria isolation. PCR has become a powerful diagnostic tool for detection of microorganisms in food and clinical samples (5, 16, 19). lt is considered a fast, highly sensitive and specific assay that quickly amplifies specific sequences of the target DNA from bacterial pathogens such as Shigella spp. The target DNA is a specific sequence from a gene related to virulence mechanisms. However, despite the advantages compared to conventional culture, PCR-based methods must be reliable according to international criteria such as selectivity (inclusivity and exclusivity), good detection level and the mandatory use of internal amplification controls (IAC) (6, 11, 7).
For the present study, a PCR assay for detection of the invasion antigen loci (ial) region and invasion plasmid antigen H (ipa H) genes of Shigella and enteroinvasive Escherichia coli (EIEC) in lettuce samples was developed. These target sequences are related to the cell invasion process in both pathogens (6,22–25). The ial region is encoded in the invasion plasmid while the ipaH genes are present in both the bacterial chromosome and the invasion plasmid (9,15,25). This study compared a Multiplex PCR technique to conventional culture by using lettuce samples spiked with serial dilutions of the control strain of Shigella flexneri and the diagnostic accuracy was calculated.
MATERIALS AND METHODS
Bacteria strains
Bacterial strains selected to test the diagnostic specificity of PCR included: Aeromonas hydrophila, Citrobacter freundi, Citrobacter youngae, Edwardisella tarda, Enterobacter cloacae, Escherichia coli ATCC 64111, Escherichia coli ATCC 25922, Escherichia coli ATCC 13706, Escherichia coli O155, Enterococcus faecalis ATCC 10741, Klebsiella onithynolitica, Plesiomonas shigelloides, Pseudomonas aeruginosae ATCC 14502, Pseudomonas fluorescens, Providencia spp., Proteus spp, Salmonella serovar Enteritidis ATCC 13076, Salmonella serovar Typhimurium ATCC14028, Salmonella serovar Indiana, Salmonella serovar Typhi, Staphylococcus aureus ATCC 6538, Shigella flexneri, Shigella sonnei, Shigella boydii, Shigella dysenteriae and Yersinia enterocolytica.
DNA Extraction
DNA from the four species of Shigella and non Shigella strains was obtained by the phenol-chloroform extraction method: 1.5 ml of an overnight broth culture (Tripticase Soy Broth, and GN broth) at 35ºC were centrifuged at 2700 g for 5 min (microcentrifuge 5417C Eppendorf Hamburg, Germany). The pellet was resuspended in 567 ml of TE Buffer, 30 μl of 10% SDS and 3 μl of Proteinase K 20 mg/ml (Promega). The mixture was incubated at 37ºC for 1 h. After incubation 100 μl of 5M NaCl was added and mixed thoroughly. After incubation, 80 μl of CTAB/NaCl was added, mixed and incubated for 10 min at 65ºC. Finally, DNA was collected by centrifugation after phenol-chloroform precipitation, washed with 70% ethanol and dried with air.
Primers and Internal amplification control (IAC)
The sequences of ipaH 600 bp product primer pair (fw 5’GCCGGTCAGCCACCCT CTGAGACTAC-3’ and rv 5’-GTTCCTTGACCGCCTTTCCGTACCGT C-3’) (15), and ial 320 bp product primer pair (fw 5’-CTGGATGGTATGGTG AGG-3’ and rv 5’-GG AGGCCAACAATTATTTCC-3’) (1), were prepared by Innovagen (Lund, Sweden). An internal amplification control (IAC) of 100 bp using ipaH primers sequence was designed and purchased from Invitrogen (USA): 5’ – GTTCCTTGACCGCCTTTCCGTACCGTCTCT GCACGCAATACCTCCGGACAGAAGTATGAGATGCTGGAGAATGAGTACTCTCAGAGGGTGGCTGACCGGC-3`. The amplification of IAC was performed with ten-fold dilutions of IAC (from 1 μg/ml to 0.01 ng/μl) with two defined amounts of target (Shigella) DNA at 50 ng/μl and 100 ng/μl.
PCR protocol
Comparable PCR protocols were standardized using extracted DNA from both Shigella (S.dysenteriae, S.flexneri, S.boydii and S.sonnei) culture and lettuce samples spiked with bacterial dilutions. A 25 μl reaction volume was used, containing 1X Buffer, 2.0 mM MgCl, 0.5 U Taq polymerase (Promega), 5% DMSO (Sigma Aldrich) 0.2 mM each dNTPs (Applied Biosystems), 0.004 mM ipaH primers, 0.016 mM ial primers and 10 μl of DNA template. The reaction mixture was placed in a thermocycler (Verity, Applied Biosystems) for 30 cycles, 10 cycles in PCR touch down (each cycle consisting of 1 min at 94ºC, 30 s at 65ºC decreasing to 55ºC, and 1 min at 72ºC) and last 20 cycles in normal PCR (1 min at 94ºC, 30 s at 55ºC and 1 min at 72ºC). An additional step at 5 min at 72ºC for primer extension was added at the end of the reaction.
The PCR products were detected by 2% agarose gel electrophoresis (140V, at 45 min) in 1X TBE Buffer at pH 8.3 and stained with 0.5 μg/ml ethidium bromide. The products were visualized as 600 bp (ipaH), 380 bp (ial) and 100bp (IAC) bands using an UVITEC transiluminator (Cambridge, UK).
DNA sequences of the ipaH and ial products were determined (310 ABI Genetic Analyzer, Applied Biosystems) to confirm amplification of the correct gene.
Inoculation of lettuce samples
Portions of lettuce (Lactuca sativa Var. Capitata) were aseptically added to sterile bags and spiked with serial dilutions of S. flexneri. Only spiked lettuce samples with Shigella were used.
One ml of each serial dilution of S. flexneri in sterile peptone buffer (107 CFU/ml to 10 CFU/ml) was inoculated into three portions of lettuce, two of 10 g and one of 25 g. The inoculation experiment was performed to complete 10 repetitions at the detection levels for Enriched-PCR and culture (5).Bacterial dilutions were prepared as follows: A S.flexneri strain was inoculated onto Tripticase soy agar (TSA) and incubated overnight at 35ºC. After incubation, single colonies from TSA were inoculated into 10 ml of sterile peptone buffer to a concentration of 108 CFU/ml, as determined by the Pour Plate technique(10).
Conventional Culture Samples: Lettuces (25 g) were homogenized for 2 min and 225 ml of GN broth were added. After 1 hour pH was measured and samples were incubated at 35ºC for 24 hrs. After incubation they were streaked onto T7, XLD and DC agars (14).
Direct-PCR Samples: Lettuces (10 g) were rinsed with 4 ml of peptone buffer. After 1 min homogenization, 3 ml buffer were recovered with a sterile Pasteur pipette into a polyethylene sterile tube for further centrifugation and DNA extraction for detection of Shigella as previously described.
Enriched-PCR Samples: Lettuces (10 g) were rinsed with 4 ml of peptone buffer. After 1 min homogenization, 3 ml of buffer were recovered with a sterile Pasteur pipette into a stomacher sterile bag (Labplas, Canada) with 45 ml of GN broth. These samples were incubated at 35ºC for 24 h. After incubation, 1.5 ml portion of enrichment broth was used to DNA extraction for detection of Shigella as previously described.
Lettuce inoculation experiment (culture, direct-PCR and enriched-PCR) was performed by triplicate in each dilution (10 to 107 CFU/ml) and with 10 repetitions for dilutions 103, 104 and 105 CFU/ml (Enriched-PCR) and 105, 106 and 107CFU/ml (culture). DNA extracted was used to detect ipaH (600 bp) and ial (320 bp) products by PCR.
PCR and culture accuracy
Diagnostic accuracy of both methods was established according to definitions and formulas as follows:
Diagnostic specificity is defined as a measure of the degree to which the method is affected by non-target microorganisms present in a food matrix, which may result in false positive responses (17, 18).
Diagnostic sensitivity is defined as a measure of the degree to detect the target pathogen in the food matrix, which may result in false negative responses (17, 18).
RESULTS
Internal amplification control (IAC)
The IAC was co-amplified with the target DNA, resulting in a 100 bp amplicon (Figure 1). The lowest IAC concentration which showed a PCR band, visible by gel electrophoresis and ethidium bromide stain, was 0.1 ng/μl per 25 μl reaction. IAC was used in all PCR analysis to detect false negative results because of failure of the amplification reaction. This internal control is highly recommended when PCR-based methods are used to detect bacterial pathogens (5,11,17,18).
Figure 1.
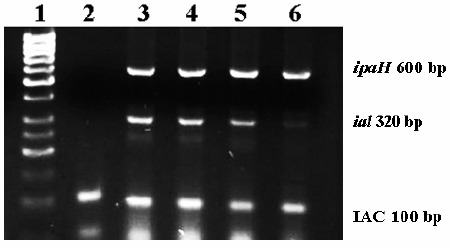
Multiplex PCR standardization results: 2% agarose gel electrophoresis stained with ethidium bromide. Lane 1: molecular size marker; lane 2: negative control, lane 3 and 4: Shigella flexneri strain 50(ipaH+/ial+); lanes 5 and 6: Shigella flexneri strain 01 (ipaH+/ial+).
PCR standardization
The products from DNA amplification by Multiplex PCR are shown in Figure 1. In the amplification reaction, bands of 100 bp (IAC), 320 bp (ial) and 600 bp (ipaH) were observed with S.flexneri DNA. The ipaH and ial products were not observed with the other bacterial strains (data not shown).
Detection level
Table 1 shows the results of the number of positive repetitions/total of repetitions for each bacterial dilution (10 to 107 CFU/ml), from enrichment broth (Enriched-PCR), direct rinse (Direct-PCR) and conventional culture. The detection level of S. flexneri in lettuce by PCR was better than culture (104to 107CFU/sample by PCR and 106 to 107CFU/sample by culture). It was detected an inhibitory effect in the PCR-Direct samples since IAC was negative for all dilutions with 3 repetitions (Table 1, Figure 3), probably because of the matrix used. For this reason, only 3 repetitions were tested in the PCR-Direct experiment and these results were excluded to calculate PCR diagnostic accuracy.
Table 1.
Detection level of the PCR assay (Direct and Enriched) as compared to the conventional culture, using serial dilutions of Shigella flexneri spiked to lettuce samples.
| Inoculum Shigella (CFU/sample) | Multiplex PCR positive results | Culture positive results | |
|---|---|---|---|
| Enriched-PCR samples | Direct-PCR samples | ||
| 107 | 3/3 | 0/3 | 10/10 |
| 106 | 3/3 | 0/3 | 5/10 |
| 105 | 10/10 | 0/3 | 1/10 |
| 104 | 10/10 | 0/3 | 0/3 |
| 103 | 0/10 | 0/3 | 0/3 |
| 102 | 0/3 | 0/3 | 0/3 |
| 10 | 0/3 | 0/3 | 0/3 |
| Noninoculated | 0/10 | 0/3 | 0/10 |
Figure 3.
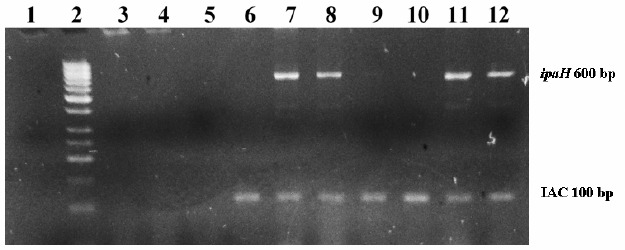
Multiplex PCR results from direct-PCR samples at different concentrations of Shigella flexneri. 2% agarose gel electrophoresis stained with ethidium bromide. Lane 1: empty; lane 2: molecular size marker; lane 3: 107 Shigella direct-PCR sample, lane 4: 106Shigella direct –PCR sample; lane 5: 105Shigella direct-PCR sample; lanes 6 and 9: negative control; lanes 7 and 11, 105Shigella enriched sample; lane 8 and 12: 106Shigella enriched sample; lane 10: Escherichia coli.
PCR diagnostic accuracy
According to Table 1, diagnostic sensitivity and specificity of Multiplex PCR and conventional culture were calculated using the number of repetitions from all dilutions that showed at least one positive result: for Multiplex PCR 104CFU/ml to 107CFU/ml and for conventional 105 CFU/ml to 107CFU/ml. Multiplex PCR showed diagnostic specificity and sensitivity of 100% to detect Shigella flexneri. Conventional culture showed a diagnostic specificity of 100%, but sensitivity of 53% from a detection limit of S. flexneri 105 CFU/ml.
DISCUSSION
In this study, a Multiplex PCR assay for detection of virulence genes of Shigella spp. and enteroinvasive Escherichia coli (EIEC) was standardized and compared to the reference culture method to detect Shigella spp. PCR detection of virulence genes in Shigella, such as ipaH, are considered a good and specific diagnostic tool of Shigella from clinical and food samples (9,15,23"–25). Although ipaH and ial genes are also carried by EIEC, Shigella has been considered the most incident pathogen related to foodborne outbreaks in Costa Rica during 2005 and 2006 (12).
Theoretically, the detection limit of one bacterial cell per PCR reaction is equivalent of 103–104 cells per ml of preenriched sample, and our results showed a detection limit for the PCR method of 104 CFU/sample, comparable to this criterion(18). We also obtained superior results using an enrichment step prior to PCR analysis rather than the direct PCR analysis of samples. The use of enrichment provides advantages such as multiplication of the bacterial target levels to obtain detectable concentrations, dilution of inhibitory substances present in food and dilution of dead target cells, which provides some assurance that the detected DNA belongs from viable target cells (16).
According to international standards, the use of an internal amplification control (IAC) is mandatory in any PCR-based method to detect foodborne pathogens (5, 11,17). The IAC band should always be present in both negative and positive samples since IAC specific primers are added to the sample tube and to be coamplified at same time with the target sequence (11). In this study, addition of the IAC allowed the differentiation of a true negative result from an inhibitory effect in Direct PCR samples, since no IAC band was obtained (Figure 3), conversely to Enriched samples, which showed the IAC band even in negative samples (Figure 2). These results confirm the importance of these controls as indicators of failure of the amplification reaction; therefore this aspect should be considered as part of a good diagnostic performance of any PCR detection method (5, 11, 17).
Figure 2.
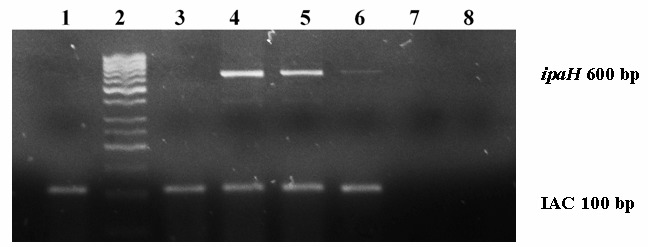
Multiplex PCR results from Enriched-PCR samples at different concentrations of Shigella flexneri. 2% agarose gel electrophoresis stained with ethidium bromide. Lane 1: negative control, lane 2: molecular size marker; lane 3: Escherichia coli; lane 4: Shigella 106 enriched-PCR sample; lane 5: Shigella 105 enriched-PCR sample; lane 6: Shigella 104 enriched-PCR sample; lanes 7 and 8: empty.
Some protocols have described a simplex PCR method specific to the ial region to detect Shigella from food. In this study the ial product was detected only from pure culture but not from enriched samples. The ial region is encoded into the Shigella invasion plasmid, and it has been described that avirulent variants (pINV-) often arise owing to the loss or deletion of large fragments of the pINV spontaneously during growth at in-vitro conditions (9). Schuch et al. have described the stability of invasive phenotype at 30ºC but not at 37ºC, presumably attributed to absence of high level activity and/or repression of virulence genes expression into pINV(22). The instability of the Shigella invasion plasmid may contribute also to false-negative result because this extrachromosomal element is prone to loss or deletions (9,20,25). Invasive phenotype Shigella colonies could also be directly detected from culture by addition of Congo red dye to the agar medium (9). Virulent strains absorb the dye from agar and thus appear pigmented or Congo red positive (Crb+), which allows identification by culture of potentially invasive colonies (9,22,25). In this study the growth temperature at 35ºC could possibly affect the detection from enrichment samples by PCR of the ial target. The use of the Congo red dye will be included in future studies for identification of Shigella invasive phenotype in vitro when these virulence genes are the targets to be detected.
Both Shigella Multiplex PCR and conventional culture showed good analytical and diagnostic accuracy, which means that non target microorganisms tested were negative both by PCR and culture while all Shigella strains were positive by both methods. The molecular method showed a higher diagnostic sensitivity and a faster result as compared to the conventional culture. Even though conventional culture is considered the gold standard for Shigella detection, the PCR method is an useful tool which complements detection of foodborne pathogens such as Shigella.
ACKNOWLEDGEMENTS
We thank the participants of INISA-CDRC University of Miami Scientific Publications Workshop, especially to Harlan Wolfe and Mary Comerford for their valuable help reading this manuscript.
This study was supported by Consejo Nacional para Investigaciones Científicas y Tecnológicas-Costa Rica (FORINVES-CONICIT), Vicerrectoría de Investigación, Universidad de Costa Rica, NETROPICA –SIDA/SAREC and Internacional Foundation for Science (IFS), Swedeen.
REFERENCES
- 1.Achí R., Lindberg A. Rapid and sensitive detection of Shigella sonnei in feces by the use of an O-specific monoclonal antibody in immunomagnetic separation-polymerase chain reaction combined assay. Clin Microbial Infect. 1996;2:55–58. [PubMed] [Google Scholar]
- 2.Achí R., Siles X., Mata L., Lindberg A.A. Shigellae assays based on immunomagnetic separation in children from urban marginal communities of Costa Rica. J Infect. 1996;32:211–218. doi: 10.1016/s0163-4453(96)80021-1. [DOI] [PubMed] [Google Scholar]
- 3.Anonymous . Foodborne gastroenteritis caused by Salmonella and Shigella. In: Jay J.M., Loessner M.J., Golden D.A., editors. Modern Food Microbiology. New York: Springer Science + Business Media Inc; 2005. pp. 619–634. [Google Scholar]
- 4.Anonymous Ray B., Bhunia A. Fundamental Food Microbiology. Boca Raton: CRC Press Taylor & Francis Group; 2008. Foodborne infections; pp. 298–301. [Google Scholar]
- 5.Anonymous . Protocol for the validation of alternative methods. (ISO 16140:2003 E) International Organization for Standardization. Geneva, Switzerland: 2003. Microbiology of food and animal feeding stuffs. [Google Scholar]
- 6.Aranda K.R.S., Fagunde-Neto U., Scaletsky I.C.A. Evaluation of Multiplex PCRs for diagnosis of Infection with diarrheagenic Escherichia coli and Shigella spp. J Clin Microbiol. 2004;12:5849–5853. doi: 10.1128/JCM.42.12.5849-5853.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Barrantes K., Achí R., Bolaños S., Cerdas M., Cortés X. Calidad microbiológica y aislamiento de Shigella flexneri en vegetales frescos del Área Metropolitana de Costa Rica, 2001–2002. Boletín Avances de Investigación en Seguridad Alimentaria y Nutricional (SAN). NCAP, ME/11. 2006:42–48. [Google Scholar]
- 8.Barrantes K., Veko P., Achí R. Rev Costarric Cienc Méd. Vol. 25. San José, Costa Rica: 2004. Brote de diarrea asociado a Shigella sonnei debido a contaminación hídrica; pp. 15–22. 2001. [Google Scholar]
- 9.Germani Y., Sansonetti P.J. Dworkin M, Falkow S, Rosenberg E, Schleifer KH, Stackebrandt E. The Prokaryotes. A handbook of the biology of bacteria. New York: Springer Science + Business Media Inc; 2006. The genus Shigella; pp. 99–116. [Google Scholar]
- 10.Hall P.A., Ledenbach L., Flowers R.S. Culture methods for enumeration of microorganisms. In: Downes FP, Ito K., editors. Compendium of methods for the microbiological examination of foods. Washington: American Public Health Association; 2001. pp. 53–58. [Google Scholar]
- 11.Hoorfar J., Malorny B., Abdulmawjood A., Cook N., Wagner M., Fach P. Practical considerations in design of internal amplification controls for diagnostic PCR assays. J Clin Microbiol. 2004;5:1863–1868. doi: 10.1128/JCM.42.5.1863-1868.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kopper G., Calderón G., Schneider S., Domínguez W., Gutiérrez G. Organización de las Naciones Unidas para la Agricultura y la Alimentación (FAO) 2009. Enfermedades transmitidas por alimentos y su impacto socioeconómico Estudio de caso en Costa Rica, El Salvador, Guatemala, Honduras y Nicaragua. Informe técnico sobre ingeniería agrícola y alimentaria; pp. 13–39. [Google Scholar]
- 13.Kotloff K.L., Winickoff J.P., Ivanoff B., Clemens J.D., Swerdlow D.L, Sansonetti P.J., et al. Global burden of Shigella infections: Implications for the development of vaccines and application of control strategies. Bull World Health Organ. 1999;77:651–666. [PMC free article] [PubMed] [Google Scholar]
- 14.Lampel K. Shigella. In: Downes F.P., Ito K., editors. Compendium of methods for the microbiological examination of foods. Washington: American Public Health Association; 2001. pp. 381–385. [Google Scholar]
- 15.Lampel K., Orlandi P. Polymerase chain reaction detection of invasive Shigella and Salmonella enterica in food. Methods in Molecular Biology. 2002;179:235–237. doi: 10.1385/1-59259-238-4:235. [DOI] [PubMed] [Google Scholar]
- 16.Lee M.D., Fairchild M.S. Sample preparation for PCR. In: Maurer J., editor. PCR Methods in Foods. New York: Springer Science + Business Media Inc; 2006. pp. 41–49. [Google Scholar]
- 17.Malorny B., Hoorfar J., Bunge C., Helmuth R. Multicenter validation of the analytical accuracy of Salmonella PCR: towards an International Standard. App Envirom Microbiol. 2003;1:290–296. doi: 10.1128/AEM.69.1.290-296.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Malorny B., Tassios P.T., Radstrom P., Cook N., Wagner M., Hoorfar J. Standardization of diagnostic PCR for the detection of foodborne pathogens. Int J Food Microbiol. 2003:39–48. doi: 10.1016/s0168-1605(02)00322-7. [DOI] [PubMed] [Google Scholar]
- 19.Naraveneni R., Jamil K. Rapid detection of foodborne pathogens by using molecular techniques. J Med Microbiol. 2005;54:51–54. doi: 10.1099/jmm.0.45687-0. [DOI] [PubMed] [Google Scholar]
- 20.Sayeed S., Brendler T., Davis M., Reaves L., Austin S. Surprising dependence on postsegregational killing of host cells for maintenance of the large virulence plasmid of Shigella flexneri. J Bacteriol. 2005;8:2768–2773. doi: 10.1128/JB.187.8.2768-2773.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Schroeder G., Hilbi H. Molecular pathogenesis of Shigella spp: Controlling host cell signalling, invasion, and death by Type III Secretion. Clin Microbiol Rev. 2008;21:134–156. doi: 10.1128/CMR.00032-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schuch R., Maurelli A. Virulence plasmid instability in Shigella flexneri 2a is induced by virulence gene expression. Infec Immun. 1997;9(65):3686–3692. doi: 10.1128/iai.65.9.3686-3692.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Silva T., Nogueira P.A., Magalhaes G.F., Grava A.F., Pereira da Silva L.H., Orlandi P.P., et al. Mem Inst Oswaldo Cruz. Vol. 7. Brazil: 2008. Characterization of Shigella spp. by antimicrobial resistance and PCR detection of ipa genes in an infantile population from Porto Velho (Western Amazon region) pp. 731–733. [DOI] [PubMed] [Google Scholar]
- 24.Thiem V.D., Sethabutr O., von Seidlein L., Tung T.V., Canh D.G., Chien B.T., et al. Detection of Shigella by a PCR assay targeting the ipaH gene suggests increased prevalence of shigellosis in Nha Trang, Vietnam. J Clin Microbiol. 2004;5:2031–2035. doi: 10.1128/JCM.42.5.2031-2035.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Thong K.L., Hoe S.L.L., Puthucheary S.D., Yasin R.M. Detection of virulence genes in Malaysian Shigella species by multiplex PCR assay. BMC. 2005;5:8. doi: 10.1186/1471-2334-5-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.U.S. Centres of Diseases Control and Prevention . [Accesed 03 June, 2009]. Foodnet Reports. Available at: http://www.cdc.gov./foodnet/reports.htm. [Google Scholar]
- 27.Valiente C., Mora D. Estudio bacteriológico del agua asociado a brotes de diarrea en Costa Rica, 1999–2005. Rev Colegio Microb Quim Clin Costa Rica. 2005;(11suppl):11–14. [Google Scholar]
- 28.World Health Organization . Food Safety Programme. Regional Office for the Western Pacific; 2003. Food Safety: issues-public health significance of foodborne illnesses. [Google Scholar]


