Abstract
Mild cognitive impairment (MCI), defined as episodic memory impairment beyond what is expected in normal aging, is often associated with hippocampal atrophy (HA) and may represent incipient Alzheimer’s disease. However, recent studies suggest that MCI is very heterogeneous and multiple etiologies likely exist. One possibility is small vessel cerebrovascular disease (CVD). Specifically, we hypothesized that white matter hyperintensities(WMH),an MRI marker for CVD, would lead to impairments in executive control processes critical for working memory that may, in turn, result in episodic memory impairment. To test this hypothesis, we examined a group of subjects clinically diagnosed with MCI and used MRI to further subcategorize individuals as either MCI with severe white matter hyperintensities (MCI-WMH) or MCI with severe hippocampal atrophy (MCI-HA). MCI-WMH, MCI-HA, and matched control subjects each performed a battery of working memory and episodic memory tasks. Results showed that MCI-HA and MCI-WMH were equally impaired on the episodic memory task relative to controls, but MCI-WMH were additionally impaired on tests tapping verbal and spatial working memory abilities and attentional control processes. These results suggest that CVD and hippocampal dysfunction are associated with distinct neuropsychological profiles. Although both syndromes are associated with episodic memory deficits, CVD is additionally associated with working memory and executive control deficits. © 2005 Elsevier Ltd. All rights reserved.
Keywords: Aging, Dementia, White matter hyperintensities, Working memory, Cerebrovascular disease, Hippocampus
1. Introduction
Even in the absence of dementia, many elderly persons develop a degree of cognitive loss beyond what is expected in normal aging. There are many characterizations of this intermediate stage of cognition, one of which is mild cognitive impairment (MCI) (Petersen et al., 1999). Individuals diagnosed with MCI typically have severe episodic memory deficits with otherwise relatively preserved cognitive and functional abilities. MCI was originally defined to identify individuals who are in the preclinical stage of Alzheimer’s disease (AD). Given that the hippocampus plays a central role in episodic memory and is the initial target of AD pathology, many studies have focused on hippocampal dysfunction as an etiology of MCI. Indeed, there is evidence that individuals with MCI show distinct hippocampal activation patterns (Machulda et al., 2003; Small, Perera, DeLaPaz, Mayeux, & Stern, 1999) and that individuals with MCI and hippocampal atrophy (HA) have greatly increased risk of developing AD (Jack et al., 1999). Such results suggest a strong link between hippocampal dysfunction and MCI.
However, it is has also become clear that some individuals diagnosed with MCI do not have hippocampal atrophy and are not in the preclinical stages of AD, suggesting that other pathological processes are at work. Epidemiological studies suggest that MCI is heterogeneous and likely arises from multiple etiologies (DeCarli, 2003a). One possible etiology is small vessel cerebrovascular disease (CVD). Small vessel CVD is commonly seen in elderly individuals and has been associated with increased risk for MCI (DeCarli et al., 2001; Lopez et al., 2003). Rather than causing large cortical strokes, small vessel CVD is associated with small subcortical infarcts and white matter abnormalities.
These white matter changes appear on MRI as white matter hyperintensities (WMH) and are used as a marker for small vessel CVD severity in this study. WMH appear as areas of high signal intensity in deep or periventricular white matter on proton density and T2-weighted MRIs. The underlying pathology is non-specific and includes multiple types of injury to white matter such as reduction in myelination of axons, narrowing of small vessels, and gliosis (see Bronge, 2002, for review). WMH have been associated with hypertension, diabetes mellitus, and history of stroke, three risk factors for CVD (Breteler et al., 1994b; DeCarli et al., 1995).
We propose that WMH related to small vessel CVD may play a role in the episodic memory impairment characteristic of MCI. Given the evidence that WMH may be associated with frontal lobe dysfunction (Breteler et al., 1994a; DeCarli et al., 1995; Gunning-Dixon & Raz, 2003), we predicted that WMH may compromise executive control processes that are critical for working memory, which in turn may lead to episodic memory deficits and a diagnosis of MCI. This theory presupposes that if information cannot be actively maintained and manipulated at an immediate or short-term level, episodic encoding and retrieval impairments may arise. Thus, whereas hippocampal dysfunction may be associated with isolated episodic memory impairments, small vessel CVD may lead to a distinct pattern of deficits that includes both episodic memory impairment and deficits in executive control processes.
To test our hypothesis, we examined a group of individuals who were clinically diagnosed with MCI and used MRI to identify two subgroups of subjects: (1) those with severe WMH without hippocampal atrophy (MCI-WMH), and (2) those with severe hippocampal atrophy without extensive WMH (MCI-HA). Cognitive performance for each of these groups was compared to a group of age-matched control subjects. Importantly, these specific subgroups of MCI subjects were selected for the purpose of trying to understand the different mechanisms by which WMH and HA may lead to episodic memory impairment in MCI. Although there is increasing evidence suggesting that cerebrovascular disease and degenerative processes associated with AD often co-occur, the nature of the interaction is unclear and complex to study due to the difficulty of disentangling the two in standard clinical samples. Thus, we examined a highly selected sample in order to begin to understand the separate roles that each type of brain lesion may play in producing memory impairment.
The study is divided into two parts. First, we compared performance of MCI patients and controls on the neuropsychological tests that were used to diagnose MCI. This allowed us to determine whether standard neuropsychological tests used widely in clinical practice could distinguish between two MCI groups with different underlying brain pathologies. Second, we compared the performance of these subjects on a battery of behavioral tasks used widely in the cognitive neuroscience literature. The purpose of using these tasks was to attempt to understand the different cognitive mechanisms that underlie memory loss in MCI. The battery included an episodic memory task, two working memory tasks, and a version of the continuous performance test (CPT). We predicted that both groups of MCI participants would show deficits on the episodic memory task, but that the MCI-WMH group would show additional impairments on the working memory tasks and on the CPT.
2. Methods
2.1. Participant selection
This study was approved by the UC Davis IRB. All participants were recruited from the UC Davis Alzheimer’s Disease Center (ADC) and were over the age of 65 years, in stable health. Exclusion criteria were limited to clinical depression, history of cortical strokes, and red–green color blindness. Five of the 11 MCI-HA participants and 6 of the 11 MCI-WMH were on stable doses of cholinesterase inhibitors. All participants received a clinical diagnosis through the ADC of either normal cognition or mild cognitive impairment based on neurological exams and neuropsychological evaluations. The diagnoses of either normal or MCI were adjudicated at a multidisciplinary case conference, based upon all available clinical information. Subjects with MCI all met criteria for an amnestic form of MCI (Petersen et al., 1999) as all had memory complaints (usually verified by an informant), performed poorly on neuropsychological tests of verbal memory (Memory Assessment Scales (MAS) List Learning, Logical Memory I and II, see below), had normal general cognitive function, and intact activities of daily living. All of these MCI subjects were recruited from a pool of individuals who presented to the ADC for cognitive evaluation of their memory complaints. Control subjects were recruited either from the community through advertising or word of mouth, or from spouses of patients seen at the ADC. MRIs obtained within 6 months (mean = 121 days) of clinical diagnosis were then used to categorize participants in the MCI group as either MCI-WMH or MCI-HA. After examination of MRIs, out of a total of 30 individuals clinically diagnosed with MCI, 3 were excluded based on having neither severe HA or severe WMH and 5 were excluded based on the presence of both severe HA and severe WMH. As an additional measure to confirm the presence of cerebrovascular disease in the MCI-WMH group, we also examined the prevalence of hypertension and type II diabetes (see Table 1).
Table 1.
Demographics, brain structure variables, and prevalence of vascular risk factors
| Controls | MCI-HA | MCI-WMH | |
|---|---|---|---|
| n | 20 | 11 | 11 |
| Age | 78.65 (6.34) | 74.64 (5.70) | 77.64 (3.56) |
| Education | 15.6 (2.80) | 15.8 (3.46) | 13.5 (1.51) |
| Gender (M/F) | 3/14 | 4/8 | 5/6 |
| Normalized left Hippocampal volume |
.151 (.03) | .101 (.03)a | .145 (.02) |
| Normalized right Hippocampal volume |
.155 (.02) | .107 (.03)a | .147 (.01) |
| White matter Hyperintensity Load |
1.66 (2.39) | .371 (.327) | 3.89 (1.34)a |
| Hypertension (%) | 47 | 45 | 82 |
| Type II diabetes (%) |
12 | 0 | 27 |
Normalized hippocampal volumes= (hippocampal volume/total cranial volume)× 100. White matter hyperintensity load is expressed as percent of abnormal white matter. Data are expressed as mean (S.D.).
Differs from all other groups, p < .01.
2.2. Imaging
All MR images were collected on a GE 1.5T Signa Horizon LX NV/i System. Three sequences were obtained: a sagittal fast spin echo T2 weighted pulse sequence (TR 3000 ms, TE 94 ms, fov 24 cm, slice thickness 5 mm, slice gap 1 mm, matrix 256 × 224), an axial oblique spin echo T2 weighted sequence (TR 2420 ms, TE 20ms and 90ms, 44 slices, fov 24 cm × 24 cm, slice thickness 3 mm, slice gap 0 mm, matrix 256 ×192), and a T1-weighted, coronal 3D spoiled gradient recalled echo, inversion recovery prepped (3DSPGR, IR-prepped) sequence (TE 1.9 ms, flip angle 20°, fov 24 cm, matrix 256 × 256, 124 contiguous slices, slice thickness 1.6 mm).
Hippocampal volumes were quantified using a region of interest approach as described previously (Wu et al., 2002). In brief, the sampled hippocampus included the CA1–CA4 fields, dentate gyrus, and the subicular complex. Hippocampal boundaries were manually traced on contiguous 1.6 mm T1-weighted coronal slices by a single rater. Intrarater reliability was determined using intraclass correlation coefficients (ICC). ICCs for right and left hippocampi were .98 and .96, respectively. Hippocampal volumes were normalized to total intracranial volume to control for differences in head size.
White matter hyperintensities were rated on proton density weighted images using a semi-quantitative scale (Wu et al., 2002). Ratings were made on contiguous 5mm axial proton density images by a single rater. Intrarater reliability was high (ICC 0.97).
Percentile cutoffs, from a separate population based study of 122 individuals, were used to categorize participants as either having severe hippocampal atrophy or severe white matter hyperintensities (Wu et al., 2002). A 25th percentile cutoff, which corresponded to a normalized value of .126 (hippocampal volume/intracranial volume ×100), was used to determine severity of HA. Individuals with hippocampal volumes below this value were classified as having severe HA. A 75th percentile cutoff was used to classify individuals with severe WMH. For this cutoff, WMH volume was assessed as percent of total white matter; individuals with a value greater than 19.375 were classified as having severe WMH. For this study, individuals with neither severe HA or severe WMH were excluded as were individuals with both severe HA and severe WMH.
2.3. Neuropsychological testing
Participants received clinical neuropsychological testing at the UC Davis ADC. The testing was administered as part of a clinical work up used to determine the clinical diagnosis of MCI or cognitively normal. The time interval between neuropsychological testing and behavioral testing in this study ranged from 0 to 896 days (median =86) for the control subjects and 27 to 365 days (median = 107) for subjects diagnosed with MCI. To ensure that no cognitive decline had occurred during that time interval, the next available clinical assessment for subjects with a greater than 6-month gap was evaluated to confirm that there was no change in clinical status. Three control subjects did not receive neuropsychological testing. Data from the remaining 17 control subjects are reported.
The neuropsychological battery included Mini Mental State Exam (MMSE), Wechsler Memory Scale-Revised (WMS-R) Logical Memory I and II, Memory Assessment Scales List Learning, Boston Naming, Block Design, Digit Span, Animal Fluency, and the Geriatric Depression Scale (Yesavage, 1988).
2.4. Behavioral testing
Participants were tested in a single session using a laptop computer, either in the participant’s home or at the ADC, a familiar setting to all participants. Tasks were always administered in the same order: episodic memory study and immediate test, 2-item recognition, 4-item recognition, 6-item recognition, spatial item recognition, 1-back, 2-back, CPT, and delayed object–color association test. Left/right responses for the episodic memory task and working memory tasks were counterbalanced across subject groups.
In general, MCI patients were able to complete behavioral testing without much difficulty or frustration. No subjects were excluded based on poor performance or inability to complete testing. One MCI-HA subject received an incomplete battery of working memory tests due to time constraints (missing data include spatial item recognition, 1-back, 2-back, and CPT). In addition, technical difficulties lead to missing data for one control and one MCI-HA on the delayed episodic memory test and one MCI-WMH on the 1-back task.
2.4.1. Episodic memory task
The episodic memory task was an object–color association task that has been shown in functional imaging studies to be sensitive to hippocampal and prefrontal function (Yonelinas, Hopfinger, Buonocore, Kroll, & Baynes, 2001). Participants were presented with 36 study items, half in red and half in green and were instructed to remember the color of each object. Study items were clip-art images of familiar objects. To facilitate the association between the object and color, participants were instructed to generate an explanation for why the object was presented in that particular color. Immediately after the study session, a memory test was given in which all of the previously studied items were shown in black and the participant was asked to indicate by a left/right button press which color it had been presented in previously. Items were presented in a randomized order. The immediate test was self-paced, and participants received immediate feedback on each item. During the delayed test session, 60 min later, all of the previously studied items were again shown in black at a rate of 3500 ms per picture and participants made left/right button press responses to indicate the studied color.
2.4.2. Working memory tasks
Participants were tested on two verbal working memory tasks and a spatial working memory task that have been widely used in functional imaging studies of working memory processes (Nystrom et al., 2000; Rypma, Prabhakaran, Desmond, Glover, & Gabrieli, 1999; Smith & Jonides, 1999) and have been shown to be sensitive to prefrontal function.
2.4.2.1. Verbal item recognition
Three versions of the item recognition task were used, a low memory load condition (2 letters), a medium load condition (4 letters), and a high memory load condition (6 letters). Letters were chosen randomly from a set of 17 consonants. Each trial consisted of a 2500 ms study presentation (letter set), a 2000 ms delay, and a 2500 ms probe presentation (single letter), followed by a 2000 ms inter-trial interval. Participants were instructed to make a left/right button press to indicate whether the probe letter had appeared in the study set. Each testing session consisted of 32 trials.
2.4.2.2. Verbal n-back
Two versions of the n-back task were used, 1-back and 2-back. Single letters appeared sequentially with a 3000 ms presentation followed by 3000 ms inter-trial interval. During the 1-back task, participants were instructed to make a left/right response to indicate whether the current letter was the same or different as the letter that they saw one back. During the 2-back task, participants made a left/right response to indicate whether the current letter matched the letter that they say two letters back. Each task consisted of 56 trials. Approximately 30% of trials were targets.
2.4.3. Spatial item recognition
This task was a spatial variant of the verbal item recognition task. The study set consisted of four asterisks placed randomly in the field of view around a central fixation point. After a delay, a single probe asterisk appeared on the screen and the participant was instructed to make a button press indicating whether the probe had appeared in the study set. Timing parameters were the same as the verbal item recognition tasks.
2.4.4. Continuous performance test
The CPT has been widely used to test prefrontal cortex function in many areas of clinical research, including schizophrenia and attention deficit hyperactivity disorder (Beck, Bransome, Mirsky, Rosvold, & Sarason, 1956; Riccio, Reynolds, Lowe, & Moore, 2002). This version of the CPT, the AX-CPT, was designed to test sustained attention and impulsivity. Randomly chosen letters were presented sequentially on the computer screen at a rate of 750 ms for 500 msduration. Subjects were instructed to make a button press when they saw the target letter X, but only when it was preceded by the cue letter A. One hundred letter pairs were presented. The frequency of A–X trials was 70%, with the remaining 30% of trials divided among three distracter conditions: B–X, A–Y, and B–Y (B denotes any letter other than A, and Y denotes any letter other than X).
2.5. Statistical analyses
ANCOVA and Fisher’s protected least significant difference (PLSD) post hoc analyses were used to compare performance measures for all tasks across groups. Age and education were included as covariates.
3. Results
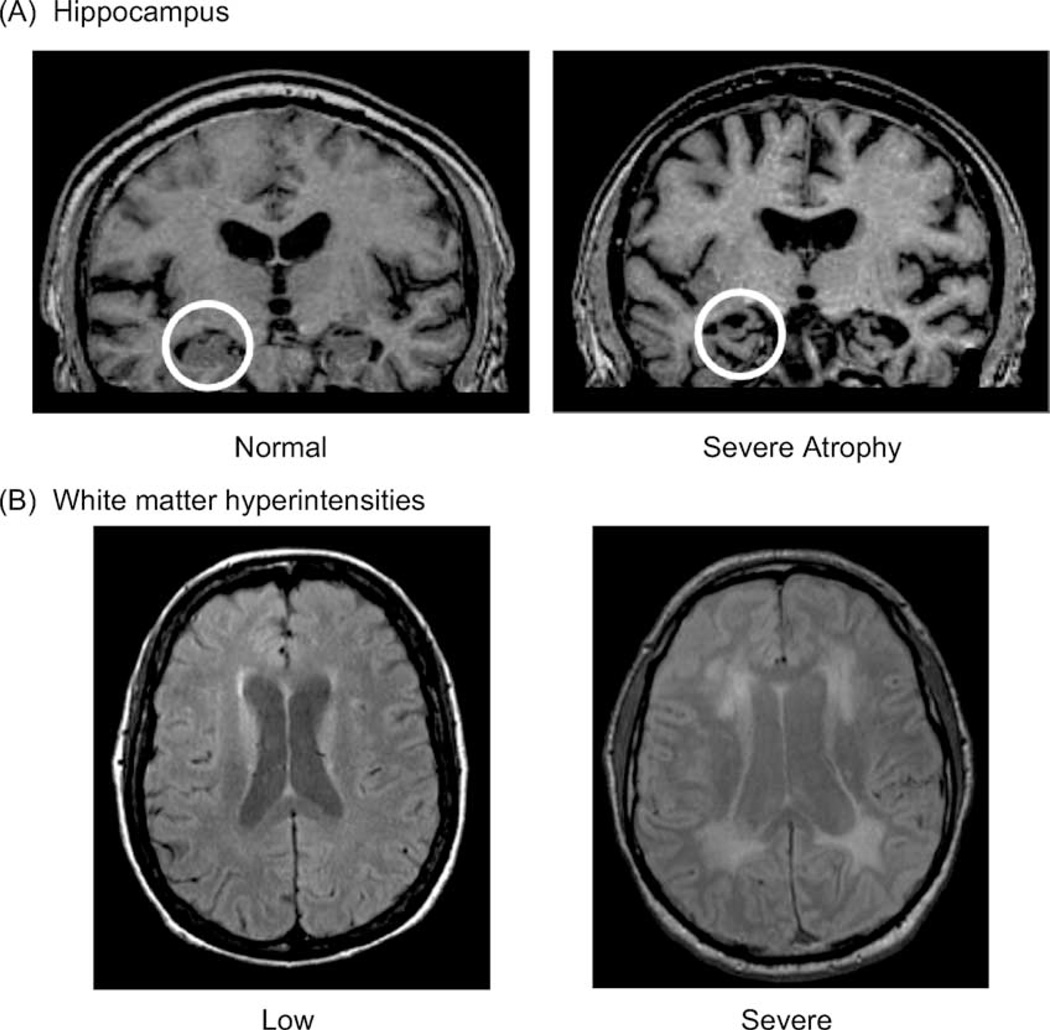
Demographic information, MRI measures of hippocampal volumes and WMH load, and incidence of vascular risk factors for each group are shown in Table 1. Fig. 1 depicts examples of the MRI measures used to subcategorize MCI subjects. Because the gender distribution was not balanced, especially for the control group, we looked for potential gender effects on performance of all of the tasks and did not observe any. Therefore, gender was not controlled for statistically. Although age (F = 2.353, p = .11) and education (F = 2.476, p = .10) did not differ across groups, they were included as covariates in all further analyses because of trend level differences.
Fig. 1.
MRI variables used to classify MCI subjects with either hippocampal atrophy or cerebrovascular disease. (A) The white circles outline a normal hippocampus and a severely atrophic hippocampus. (B) Minimal and severe white matter hyperintensities.
3.1. Neuropsychological assessment
As part of the clinical assessment that lead to diagnosis of cognitive status,10 MCI-WMH,11MCI-HA, and 17 age and education matched elderly controls underwent standardized neuropsychological testing with a battery that included tests of episodic memory, executive function, visuo-spatial ability, attention, semantic memory, global cognition, and depression. Table 2 shows performance of MCI-HA, MCI-WMH, and controls on the neuropsychological tests as well as main effect F- and p-values. The two MCI groups were equally impaired on all episodic memory tests relative to controls: WMS-R Logical Memory I and II, and MAS List Learning, Immediate Recall and Delayed Recall. The two MCI groups did not differ from each other or controls on Digit Span or Boston Naming. MCI-WMH performed worse than the MCI-HA and control groups on Block Design. In addition, the MCI-WMH group performed worse than controls, but not different from the MCI-HA group on Animal Fluency. There were no group differences on the Geriatric Depression Scale.
Table 2.
Performance on neuropsychological battery
| Controls (n = 17) | MCI-HA (n=11) | MCI-WMH (n=10) | F | p | |
|---|---|---|---|---|---|
| MMSE | 29.6 (.502) | 27.5 (1.70)a | 27.4 (2.38)a | 9.803 | .0004 |
| WMS-R | |||||
| Logical Memory I | 26.8 (6.53) | 13.8 (6.21)a | 13.6 (7.54)a | 15.9 | <.0001 |
| Logical Memory II | 24.6 (6.41) | 5.4 (6.27)a | 7.1 (8.49)a | 30.1 | <.0001 |
| MAS | |||||
| List Learning | 61.6 (6.97) | 41.9 (11.2)a | 44.9 (10.68)a | 15.5 | <.0001 |
| Immediate Recall | 10.9 (1.03) | 3.5 (3.48)a | 5.2 (4.09)a | 22.4 | <.0001 |
| Delayed Recall | 10.9 (.854) | 4.3 (3.34)a | 5.8 (5.22)a | 16.1 | <.0001 |
| Boston Naming | 55.5 (4.20) | 52.9 (5.32) | 49.5 (9.48) | 2.1 | .143 |
| Block Design | 26.6 (6.94) | 25.8 (10.07) | 15.4 (7.86)b | 3.3 | .05 |
| Animal Fluency | 16.6 (3.95) | 14.0 (2.97) | 12.6 (4.17)a | 3.7 | .03 |
| Digit Span | 14.4 (2.99) | 14.1 (3.34) | 12.2 (2.64) | 1.5 | .225 |
| Geriatric Depression Scale | 1.9 (2.02) | 2.6 (1.80) | 1.5 (1.27) | 0.9 | .43 |
Data are expressed as mean (S.D.).
Differs from controls, p< .005.
Differs from controls and MCI-HA, p < .005.
3.2. Behavioral assessment
We next investigated whether tasks used in cognitive neuroscience studies to assess episodic memory, working memory, and attentional control might be able to differentiate between MCI-WMH and MCI-HA subjects. Participants were tested on an episodic memory task, two verbal working memory tasks, one spatial working memory task, and a continuous performance test.
3.2.1. Episodic memory
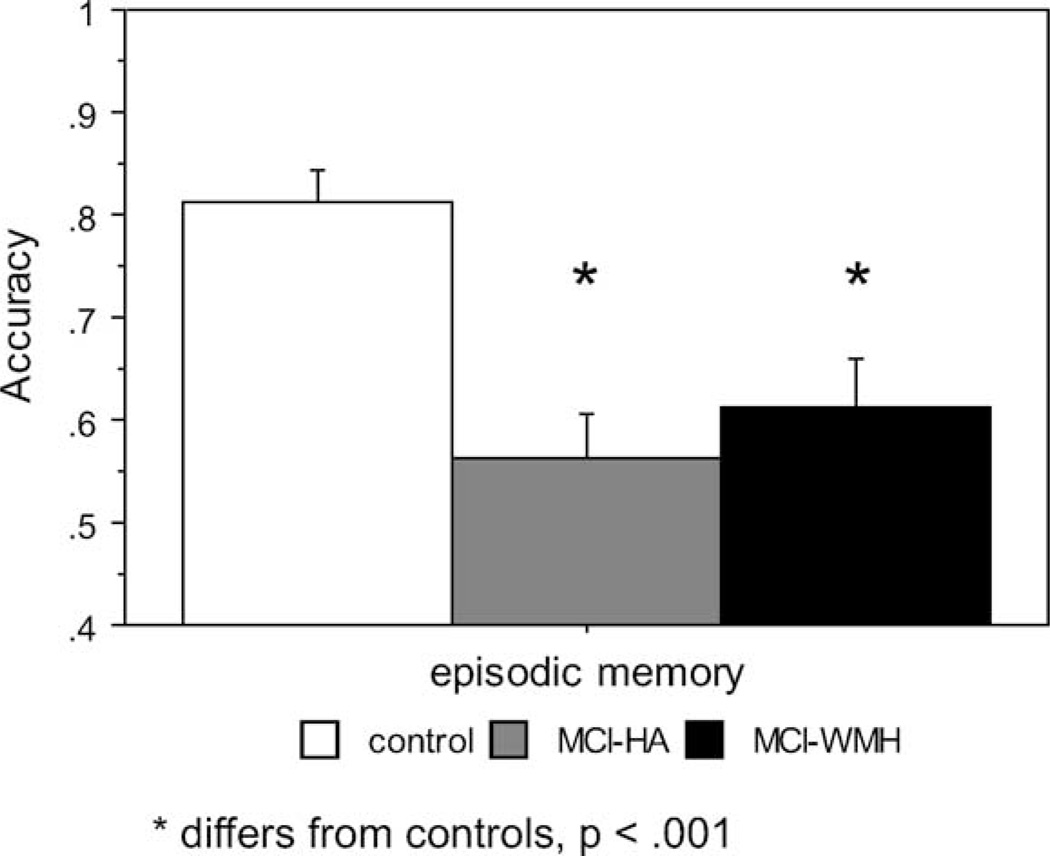
We observed a significant between-group difference in accuracy at both the immediate (F = 14.010, p < .0001) and delayed test (F = 11.159, p < .0001). As shown in Fig. 2, both MCI groups performed worse than the controls but were not different from each other. No significant between-group differences were observed in mean per picture study time or in reaction time for test responses.
Fig. 2.
Performance on episodic memory task. Both MCI groups are impaired relative to controls. Error bars represent standard error.
3.2.2. Working memory
3.2.2.1. Verbal item recognition
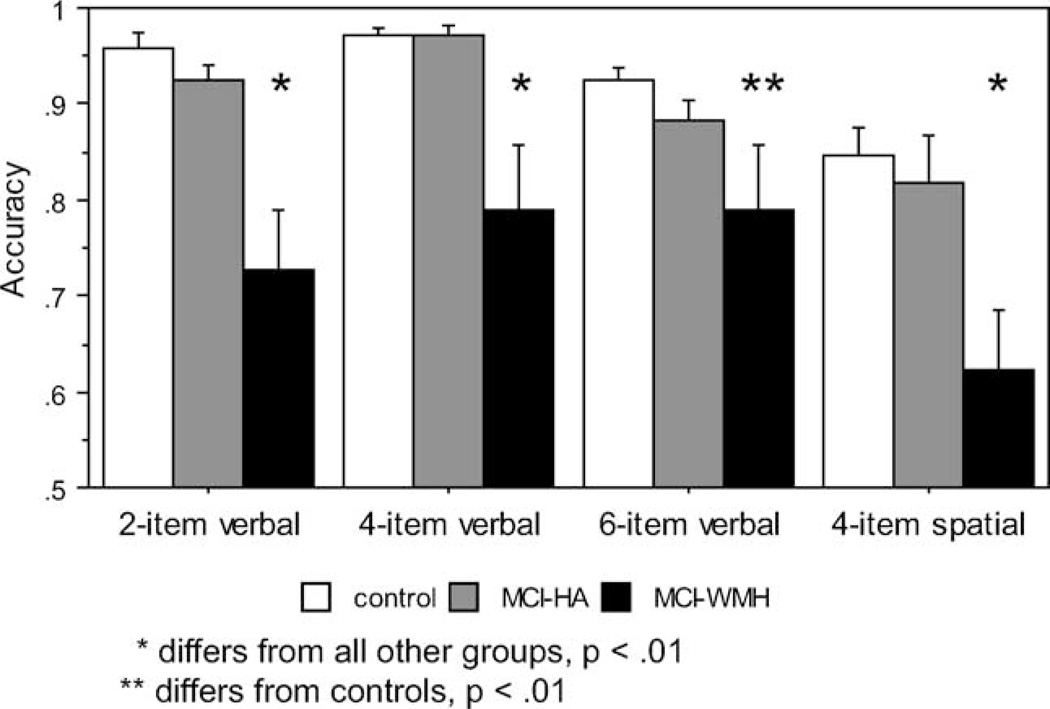
As shown in Fig. 3, there was a between-group difference in accuracy on each of the three load conditions: 2-item (F = 11.325, p = .0001), 4-item (F = 7.847, p = .0014), and 6-item (F = 3.768, p = .0324). For the 2- and 4-item loads, the MCI-WMH group performed worse than the controls and the MCI-HA groups (p’s< .01). The MCI-HA group did not differ from the controls. For the 6-item load, the MCI-WMH group performed worse than controls (p < .01) and the MCI-HA group was intermediate to controls and MCI-WMH, but did not differ from either group. The MCI-WMH group made more omission errors on the 2- and 4- load conditions than either the MCI-HA or control group (2-item: F = 5.835, p= .0068, post hoc p’s < .01; 4-item: F = 3.542, p = .0005, post hoc p’s < .02). However, even when trials for which no response was made were excluded, all group differences reported above remained significant (2-item: F= 5.207, p= .01; 4-item: F = 9.443, p = .04). Reaction times for correct responses on the 2- and 4-item tasks differed across group (F = 4.301, p = .02;F = 3.475,p = .04). The MCI-WMH group was slower to respond than both the MCI-HA and control groups (p’s <.05). There were no differences in reaction time or omission errors on the 6-item task.
Fig. 3.
Performance on verbal item recognition and spatial item recognition. The MCI-WMH group is impaired on all of the item recognition working memory tasks. Error bars represent standard error.
3.2.2.2. Verbal n-back
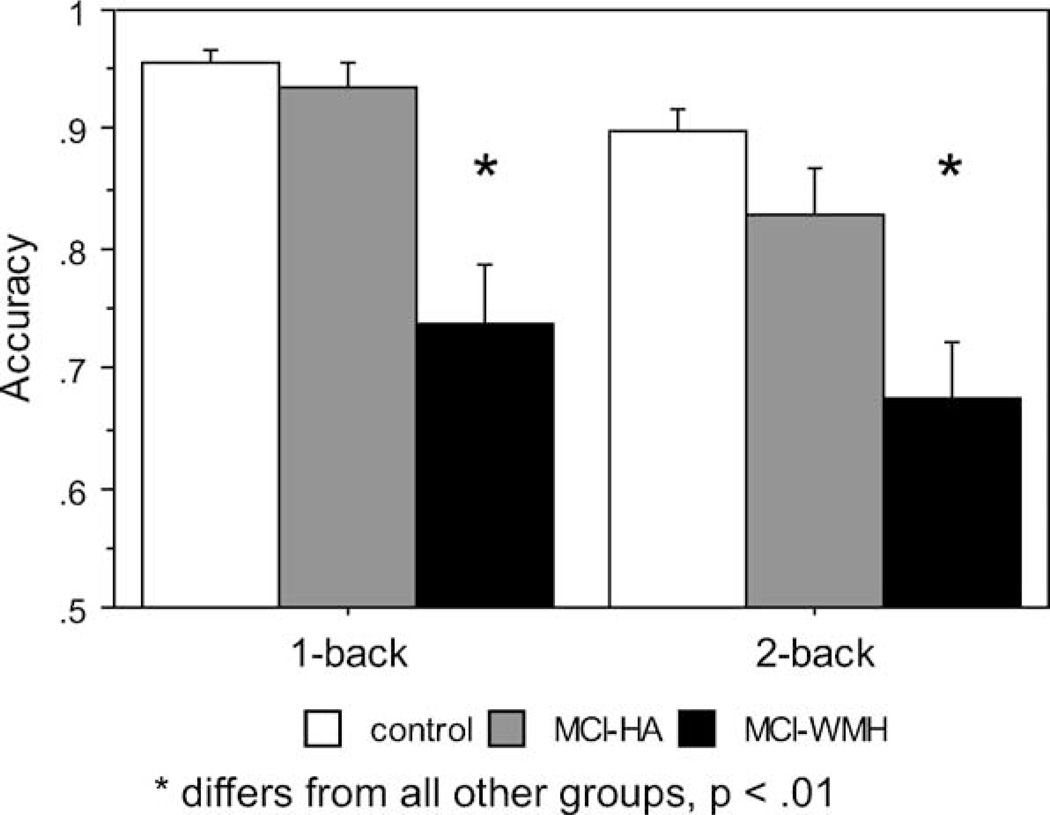
As shown in Fig. 4, we also observed a between-group difference in accuracy on both 1-back and 2-back versions of this task (1-back: F = 5.257, p = .0108; 2-back: F= 9.340, p= .0006). The MCI-WMH group performed worse than both the control and MCI-HA groups on both tasks (p’s < .0001). The MCI-HA group did not differ from the controls on either version of the task. Again, the MCI-WMH group made more omission errors, but group differences remain even after excluding omission trials. When error types were categorized as either type I (missed targets) or type II (false positives), MCI-WMH showed increases in both types of errors on both the 1-back (p < .01) and 2-back tasks (p< .01).
Fig. 4.
Performance on 1- and 2-back. The MCI-WMH group is impaired relative to the controls and MCI-HA group on both tasks. Error bars represent standard error.
3.2.2.3. Spatial item recognition
Also depicted in Fig. 3, we observed a between-group difference in accuracy (F = 6.694, p=.0039) with the MCI-WMH performing worse than both the MCI-HA and control groups (p’s< .02) (Fig. 3). Again, the MCI-WMH group made more omission errors (F= 6.307, p = .0052). When omission trials were excluded, the group differences remained (F = 4.654, p = .01, post hoc p’s < .05).
3.2.2.4. Continuous performance test
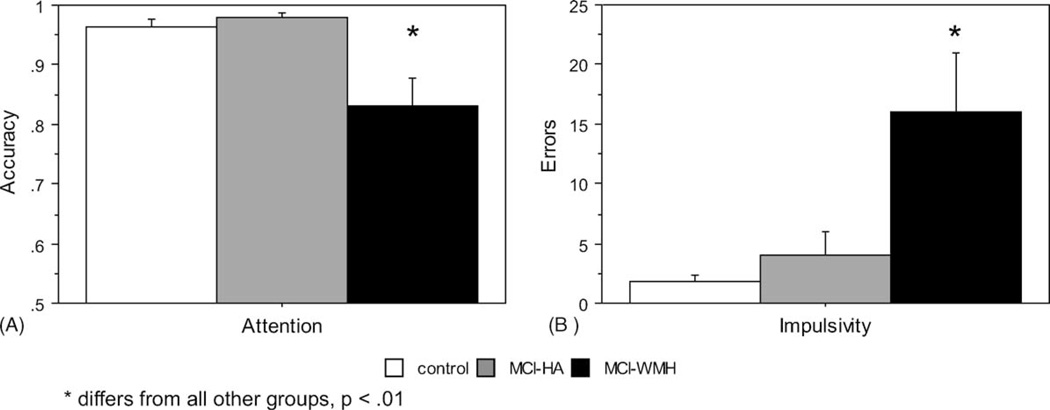
As shown in Fig. 5, the MCI-WMH group performed worse than controls and MCI-HA on the measure of sustained attention (number target hits/total number of targets) (F = 6.396, p = .0047, post hoc p’s<.001). In addition, the MCI-WMH group made more commission errors (response to any item that was not a target) than the controls and MCI-HA groups (F = 6.016, p = .0063, post hoc p’s< .007) (Fig. 5). The MCI-HA group did not differ from controls on either measure. Of the different types of commission errors, MCI-WMH made more BY errors (F = 3.452, p = .044) and A errors (responding to the cue) (F = 6.479, p = .0045) than controls and MCI-HA. MCI-WMH also made more BX errors than controls (F= 3.691, p = .0425) with MCI-HA intermediate and not different from either group. Error rates for AY trials did not differ across group.
Fig. 5.
Performance on CPT. (A) Sustained attention measure; (B) impulsivity measure. The MCI-WMH group is impaired relative to controls and MCI-HA on both measures. Error bars represent standard error.
3.3. Summary of results
Standard clinical neuropsychological testing revealed that both MCI groups have significant episodic memory deficits. The MCI groups did not differ from controls or from one another on most of the tests, demonstrating that the MCI groups are indeed very mildly impaired, with deficits generally limited to episodic memory. These findings suggest that these two MCI groups, with differential structural brain abnormalities, were comparable in overall level of cognitive impairment and were broadly similar in their cognitive symptomatology.
As additional confirmation of episodic memory impairment in these MCI subjects, behavioral testing indicated that both MCI groups were equally impaired on the episodic memory tasks relative to controls. The MCI-WMH group was additionally impaired on both verbal and spatial working memory tasks while the MCI-HA group was generally unimpaired on the working memory tasks. The MCI-WMH group was also impaired relative to MCI-HA and controls on measures of sustained attention and response control as measured by the AX-CPT task.
4. Discussion
In the present study, we tested the hypothesis that among individuals diagnosed with MCI, small vessel CVD and hippocampal dysfunction give rise to different profiles of cognitive deficits. We found that although these two groups were similar on standard neuropsychological tests administered at the time of diagnosis of MCI, more detailed testing revealed reliable differences between the two subgroups of MCI subjects. Whereas MCI-HA patients exhibited relatively specific episodic memory impairment, MCI-WMH patients exhibited deficits on episodic memory, working memory, and attentional control tasks. These findings suggest that MCI-WMH subjects, in contrast to MCI-HA subjects, suffered from impaired executive control processes that affect a wide variety of cognitive domains. We discuss these findings and their implications below.
4.1. MCI, WMH, and clinical neuropsychological testing
In this study,we examined the performance of the two MCI groups on standard measures from a battery of neuropsychological tests used widely in clinical practice to screen for cognitive impairment. While the MCI-WMH group showed mild deficits in Animal Fluency and Block Design, these were not judged to be clinically significant and were felt compatible with the clinical diagnosis of MCI, which is made according to clinical criteria rather than a specific set of neuropsychological cutoffs. Thus, despite having very different underlying brain pathologies, MCI-WMH and MCI-HA groups had similar clinical neuropsychological profiles according to the neuropsychological battery used in this study to diagnose MCI.
However, it is important to note that this battery did not include specific tests of executive function, such as the Trail Making Test—parts A and B, Wisconsin Card Sort Test, or the Stroop Test. It is possible, and even likely, that a neuropsychological battery with more extensive tests of executive function would have differentiated the two MCI groups. Although such tests of executive function are not always included in clinical assessment batteries, the recent focus on clinical differentiation of subtypes of MCI (Lopez et al., 2003) will hopefully support a more thorough assessment of such cognitive functions. We recommend that including neuropsychological tests of executive function may prove useful in providing a more complete clinical diagnosis.
Indeed, previous studies addressing the relationship between WMH and cognition have used neuropsychological tests that tap executive function. Several studies report a relationship between WMH and impairment on Trails A and B, Verbal Fluency, and Wisconsin Card Sort Test (Gunning-Dixon & Raz, 2003). However, some inconsistencies exist, with other studies reporting no relationship between WMH and performance on Verbal Fluency and Stroop tasks (Smith, Snowdon, Wang, & Markesbery, 2000; Soderlund, Nyberg, Adolfsson, Nilsson, & Launer, 2003). It is possible that Trails A and B and WCST are more sensitive to the types of executive deficits related to WMH than Verbal Fluency and Stroop, which may tap into various cognitive processes that may be difficult to dissociate and therefore lack sensitivity to detect specific deficits (Boone, Ponton, Gorsuch, Gonzalez, & Miller, 1998). Regardless, it is likely that some standardized neuropsychological tests of executive function would distinguish between the two groups of MCI subjects described in this study. Further research into exactly which clinical tests are bestat differentiating subtypes of MCI from one another is clearly needed, as are investigations into the more routine assessment of working memory and attention tasks as we used.
4.2. MCI, WMH, and prefrontal cortex
Although episodic memory has historically been linked to the hippocampus and surrounding cortices, substantial evidence from neuropsychological and neuroimaging studies suggests that the prefrontal cortex plays a critical role in implementing executive control processes that contribute to normal episodic memory functioning (Ranganath & Knight, 2003). In this study, MCI-WMH subjects were impaired not only on episodic memory tasks, but also on a battery of working memory tasks in both verbal and spatial domains as well as an attentional control task. Our interpretation is that episodic memory failure in MCI-WMH subjects is secondary to a more general impairment in executive control processes.
The underlying mechanism of prefrontal cortex impairment in MCI-WMH is currently unknown. Results from one previous study suggest that abnormalities in frontal lobe metabolism may underlie cognitive deficits in subjects with cognitive impairment and cerebrovascular disease (Reed, Eberling, Mungas, Weiner, & Jagust, 2000). Another recent study found that WMH related to CVD are predominant in the frontal lobes, and that regardless of regional distribution throughout the brain, WMH most severely affects frontal lobe metabolism (Tullberg et al., 2004).
We hypothesize that WMH may reflect disruption of the white matter tracts that connect dorsolateral prefrontal cortex (DLPFC) with its targets. Disruption of these neural circuits, in turn may lead to deficits in executive control processes that impact a wide range of cognitive domains, including episodic memory. Multiple neural circuits exist that if disrupted, may lead to the behavioral deficits observed the MCI-WMH group. For example, lesions affecting connections between the dorsolateral prefrontal cortex and its subcortical targets (Alexander, DeLong, & Strick, 1986) or lesions affecting the long cortico-cortical connections between prefrontal and posterior parietal cortex (Cavada & Goldman-Rakic, 1989; Selemon & Goldman-Rakic, 1988) would be expected to result in impaired working memory and executive control processes (Burruss et al., 2000; Cummings, 1993; Tekin & Cummings, 2002). Disconnection of the prefrontal, retrosplenial, hippocampal circuit may also give rise to deficits observed in MCI (Morris, Pandya, & Petrides, 1999; Petrides & Pandya, 1999).
4.3. Caveats
In this study, we used WMH as a marker for small vessel CVD, and there is substantial evidence supporting this link. WMH are associated with various cerebrovascular risk factors such as hypertension, atherosclerosis, smoking, diabetes, and presence of subcortical lacunar infarcts (see Bronge, 2002, for review). Moreover, recent evidence suggests that WMH may be a stronger correlate of cognitive function than lacunar infarcts, the other commonly used marker for small vessel CVD (Fein et al., 2000; Kwan et al., 1999). We recognize, however, that WMH are non-specific MRI findings for which the underlying etiology is varied, particularly amongst younger individuals (Osborn, 1994). Therefore, our findings likely do not apply to all subjects with extensive WMH. Pathological studies indicate that WMH from vascular disease are common to older individuals (DeCarli, 2003b) and are likely to be associated with MCI (Lopez et al., 2003). Our selection of older subjects with known vascular risk factors, therefore, increases the likelihood that the WMH seen here do accurately reflect small vessel CVD.
Another caveat is that in this initial examination of the effects of HA and WMH in MCI, we limited our sample to individuals who had either severe HA or severe WMH and excluded those with neither or both conditions. This amounted to the exclusion of 27% of potential subjects diagnosed with MCI. It is important to emphasize that this sample was not selected to be clinically representative. Rather, the purpose of selecting these specific subgroups was to examine the separate contribution of either hippocampal dysfunction or small vessel CVD to memory impairment in MCI. Inclusion of subjects with both underlying pathologies would have confounded the results, especially given recent evidence suggesting that CVD may interact with Alzheimer’s disease pathology to hasten the onset of dementia (Snowdon et al., 1997; Wu et al., 2002). While investigating this interaction merits further study, it is first important to understand the consequences of each pathology separately.
4.4. Implications for MCI and normal aging
Understanding the pathophysiology of mild cognitive impairment in old age is important for many reasons, most important among them prognosis and treatment. The present results suggest that small vessel CVD can produce the syndrome of MCI by disrupting frontal/executive systems. Longitudinal follow up of these patients is necessary to determine whether or not they are at elevated risk for dementia.
Moreover, the results from this study may be more broadly applicable to normal aging given that CVD is highly prevalent in the aging population. Healthy elderly individuals generally experience some decline in memory processing capabilities and many functional imaging studies have implicated frontal lobe function in contributing to the decline in memory performance (see Grady & Craik, 2000, for review). Importantly, WMH are commonly observed in cognitively normal elderly individuals (Breteler et al., 1994b; de Leeuw et al., 2001), and many studies suggest that compromised frontal lobe function is a consequence of normal aging (Grady & Craik, 2000). Accordingly, it is possible that white matter abnormalities secondary to CVD may be a mechanism for “normal” age-related changes in memory functioning. Further studies, using behavioral methods such as those used here, along with functional and structural neuroimaging techniques, will be necessary to more conclusively link white matter abnormalities to cognitive deficits in the healthy elderly.
Acknowledgements
The authors wish to thank Jean Coleman and Esther Lara for their assistance in recruiting participants for this study. This project was supported by NIH grants P30 AG10129 and R01 AG021028 and in part by funding from the NIMH predoctoral National Research Service Award (MH-065082).
References
- Alexander GE, DeLong MR, Strick PL. Parallel organization of functionally segregated circuits linking basal ganglia and cortex. Annual Review of Neuroscience. 1986;9:357–381. doi: 10.1146/annurev.ne.09.030186.002041. [DOI] [PubMed] [Google Scholar]
- Beck LH, Bransome ED, Jr., Mirsky AF, Rosvold HE, Sarason I. A continuous performance test of brain damage. Journal of Consulting Psychology. 1956;20(5):343–350. doi: 10.1037/h0043220. [DOI] [PubMed] [Google Scholar]
- Boone KB, Ponton MO, Gorsuch RL, Gonzalez JJ, Miller BL. Factor analysis of four measures of prefrontal lobe functioning. Archives of Clinical Neuropsychology. 1998;13(7):585–595. [PubMed] [Google Scholar]
- Breteler MM, van Amerongen NM, van Swieten JC, Claus JJ, Grobbee DE, van Gijn J, et al. Cognitive correlates of ventricular enlargement and cerebral white matter lesions on magnetic resonance imaging. The Rotterdam Study. Stroke. 1994;25(6):1109–1115. doi: 10.1161/01.str.25.6.1109. [DOI] [PubMed] [Google Scholar]
- Breteler MM, van Swieten JC, Bots ML, Grobbee DE, Claus JJ, van den Hout JH, et al. Cerebral white matter lesions, vascular risk factors, and cognitive function in a population-based study: The Rotterdam Study. Neurology. 1994;44(7):1246–1252. doi: 10.1212/wnl.44.7.1246. [DOI] [PubMed] [Google Scholar]
- Bronge L. Magnetic resonance imaging in dementia. A study of brain white matter changes. Acta Radiologica Supplementum. 2002;428:1–32. doi: 10.1034/j.1600-0455.43.s.428.1.x. [DOI] [PubMed] [Google Scholar]
- Burruss JW, Hurley RA, Taber KH, Rauch RA, Norton RE, Hayman LA. Functional neuroanatomy of the frontal lobe circuits. Radiology. 2000;214(1):227–230. doi: 10.1148/radiology.214.1.r00ja43227. [DOI] [PubMed] [Google Scholar]
- Cavada C, Goldman-Rakic PS. Posterior parietal cortex in rhesus monkey. II. Evidence for segregated corticocortical networks linking sensory and limbic areas with the frontal lobe. The Journal of Comparative Neurology. 1989;287(4):422–445. doi: 10.1002/cne.902870403. [DOI] [PubMed] [Google Scholar]
- Cummings JL. Frontal-subcortical circuits and human behavior. Archives of Neurology. 1993;50(8):873–880. doi: 10.1001/archneur.1993.00540080076020. [DOI] [PubMed] [Google Scholar]
- de Leeuw FE, de Groot JC, Achten E, Oudkerk M, Ramos LM, Heijboer R, et al. Prevalence of cerebral white matter lesions in elderly people: A population based magnetic resonance imaging study. The Rotterdam Scan Study. Journal of Neurology, Neurosurgery, and Psychiatry. 2001;70(1):9–14. doi: 10.1136/jnnp.70.1.9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeCarli C. Mild cognitive impairment: Prevalence, prognosis, aetiology, and treatment. The Lancet Neurology. 2003a;2(1):15–21. doi: 10.1016/s1474-4422(03)00262-x. [DOI] [PubMed] [Google Scholar]
- DeCarli C. The role of cerebrovascular disease in dementia. Neurologist. 2003b;9(3):123–136. doi: 10.1097/00127893-200305000-00001. [DOI] [PubMed] [Google Scholar]
- DeCarli C, Miller BL, Swan GE, Reed T, Wolf PA, Carmelli D. Cerebrovascular and brain morphologic correlates of mild cognitive impairment in the National Heart, Lung, and Blood Institute Twin Study. Archives of Neurology. 2001;58(4):643–647. doi: 10.1001/archneur.58.4.643. [DOI] [PubMed] [Google Scholar]
- DeCarli C, Murphy DG, Tranh M, Grady CL, Haxby JV, Gillette JA, et al. The effect of white matter hyperintensity volume on brain structure, cognitive performance, and cerebral metabolism of glucose in 51 healthy adults. Neurology. 1995;45(11):2077–2084. doi: 10.1212/wnl.45.11.2077. [DOI] [PubMed] [Google Scholar]
- Fein G, Di Sclafani V, Tanabe J, Cardenas V, Weiner MW, Jagust WJ, et al. Hippocampal and cortical atrophy predict dementia in subcortical ischemic vascular disease. Neurology. 2000;55(11):1626–1635. doi: 10.1212/wnl.55.11.1626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grady CL, Craik FI. Changes in memory processing with age. Current Opinion in Neurobiology. 2000;10(2):224–231. doi: 10.1016/s0959-4388(00)00073-8. [DOI] [PubMed] [Google Scholar]
- Gunning-Dixon FM, Raz N. Neuroanatomical correlates of selected executive functions in middle-aged and older adults: A prospective MRI study. Neuropsychologia. 1929–1941;41(14) doi: 10.1016/s0028-3932(03)00129-5. [DOI] [PubMed] [Google Scholar]
- Jack CR, Petersen RC, Xu YC, O’Brien PC, Smith GE, Ivnik RJ, et al. Prediction of AD with MRI-based hippocampal volume in mild cognitive impairment. Neurology. 1999;52(7):1397–1403. doi: 10.1212/wnl.52.7.1397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwan LT, Reed BR, Eberling JL, Schuff N, Tanabe J, Norman D, et al. Effects of subcortical cerebral infarction on cortical glucose metabolism and cognitive function. Archives of Neurology. 1999;56(7):809–814. doi: 10.1001/archneur.56.7.809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lopez OL, Jagust WJ, Dulberg C, Becker JT, DeKosky ST, Fitzpatrick A, et al. Risk factors for mild cognitive impairment in the Cardiovascular Health Study Cognition Study: Part 2. Archives of Neurology. 2003;60(10):1394–1399. doi: 10.1001/archneur.60.10.1394. [DOI] [PubMed] [Google Scholar]
- Machulda MM, Ward HA, Borowski B, Gunter JL, Cha RH, O’Brien PC, et al. Comparison of memory fMRI response among normal, MCI, and Alzheimer’s patients. Neurology. 2003;61(4):500–506. doi: 10.1212/01.wnl.0000079052.01016.78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morris R, Pandya DN, Petrides M. Fiber system linking the mid-dorsolateral frontal cortex with the retrosplenial/presubicular region in the rhesus monkey. The Journal of Comparative Neurology. 1999;407(2):183–192. doi: 10.1002/(sici)1096-9861(19990503)407:2<183::aid-cne3>3.0.co;2-n. [DOI] [PubMed] [Google Scholar]
- Nystrom LE, Braver TS, Sabb FW, Delgado MR, Noll DC, Cohen JD. Working memory for letters, shapes, and locations: fMRI evidence against stimulus-based regional organization in human prefrontal cortex. NeuroImage. 2000;11(5 Pt 1):424–446. doi: 10.1006/nimg.2000.0572. [DOI] [PubMed] [Google Scholar]
- Osborn AC, editor. Diagnostic Neuroradiology. London: Mosby; 1994. [Google Scholar]
- Petersen RC, Smith GE, Waring SC, Ivnik RJ, Tangalos EG, Kokmen E. Archives of Neurology. 3. Vol. 56. published erratum appears in Archives of Neurology; 1999. Mild cognitive impairment: Clinical characterization and outcome; pp. 303–308. (1999), 56(June (6)), 760. [DOI] [PubMed] [Google Scholar]
- Petrides M, Pandya DN. Dorsolateral prefrontal cortex: Comparative cytoarchitectonic analysis in the human and the macaque brain and corticocortical connection patterns. The European Journal of Neuroscience. 1999;11(3):1011–1036. doi: 10.1046/j.1460-9568.1999.00518.x. [DOI] [PubMed] [Google Scholar]
- Ranganath C, Knight R. Prefrontal cortex and episodic memory: Integrating findings from neuropscyhology and functional brain imaging. In: Amanda Parker TB, Wilding Edward, editors. Memory encoding and retrieval: A cognitive neuroscience perspective. Psychology Press; 2003. [Google Scholar]
- Reed BR, Eberling JL, Mungas D, Weiner MW, Jagust WJ. Memory failure has different mechanisms in subcortical stroke and Alzheimer’s disease. Annals of Neurology. 2000;48(3):275–284. [PMC free article] [PubMed] [Google Scholar]
- Riccio CA, Reynolds CR, Lowe P, Moore JJ. The continuous performance test: A window on the neural substrates for attention? Archives of Clinical Neuropsychology. 2002;17(3):235–272. [PubMed] [Google Scholar]
- Rypma B, Prabhakaran V, Desmond JE, Glover GH, Gabrieli JD. Load-dependent roles of frontal brain regions in the maintenance of working memory. NeuroImage. 1999;9(2):216–226. doi: 10.1006/nimg.1998.0404. [DOI] [PubMed] [Google Scholar]
- Selemon LD, Goldman-Rakic PS. Common cortical and subcortical targets of the dorsolateral prefrontal and posterior parietal cortices in the rhesus monkey: Evidence for a distributed neural network subserving spatially guided behavior. The Journal of Neuroscience. 1988;8(11):4049–4068. doi: 10.1523/JNEUROSCI.08-11-04049.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Small SA, Perera GM, DeLaPaz R, Mayeux R, Stern Y. Differential regional dysfunction of the hippocampal formation among elderly with memory decline and Alzheimer’s disease. Annals of Neurology. 1999;45(4):466–472. doi: 10.1002/1531-8249(199904)45:4<466::aid-ana8>3.0.co;2-q. [DOI] [PubMed] [Google Scholar]
- Smith CD, Snowdon DA, Wang H, Markesbery WR. White matter volumes and periventricular white matter hyperintensities in aging and dementia. Neurology. 2000;54(4):838–842. doi: 10.1212/wnl.54.4.838. [DOI] [PubMed] [Google Scholar]
- Smith EE, Jonides J. Storage and executive processes in the frontal lobes. Science. 1999;283(5408):1657–1661. doi: 10.1126/science.283.5408.1657. [DOI] [PubMed] [Google Scholar]
- Snowdon DA, Greiner LH, Mortimer JA, Riley KP, Greiner PA, Markesbery WR. Brain infarction and the clinical expression of Alzheimer disease. The Nun Study. The Journal of the American Medical Association. 1997;277(10):813–817. [PubMed] [Google Scholar]
- Soderlund H, Nyberg L, Adolfsson R, Nilsson LG, Launer LJ. High prevalence of white matter hyperintensities in normal aging: Relation to blood pressure and cognition. Cortex. 2003;39(4–5):1093–1105. doi: 10.1016/s0010-9452(08)70879-7. [DOI] [PubMed] [Google Scholar]
- Tekin S, Cummings JL. Frontal-subcortical neuronal circuits and clinical neuropsychiatry: An update. Journal of Psychosomatic Research. 2002;53(2):647–654. doi: 10.1016/s0022-3999(02)00428-2. [DOI] [PubMed] [Google Scholar]
- Tullberg M, Fletcher E, DeCarli C, Mungas D, Reed BR, Harvey DJ, et al. White matter lesions impair frontal lobe function regardless of their location. Neurology. 2004;63(2):246. doi: 10.1212/01.wnl.0000130530.55104.b5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu CC, Mungas D, Petkov CI, Eberling JL, Zrelak PA, Buonocore MH, et al. Brain structure and cognition in a community sample of elderly Latinos. Neurology. 2002;59(3):383–391. doi: 10.1212/wnl.59.3.383. [DOI] [PubMed] [Google Scholar]
- Yesavage JA. Geriatric Depression Scale. Psychopharmacology Bulletin. 1988;24(4):709–711. [PubMed] [Google Scholar]
- Yonelinas AP, Hopfinger JB, Buonocore MH, Kroll NE, Baynes K. Hippocampal, parahippocampal and occipital-temporal contributions to associative and item recognition memory: An fMRI study. Neuroreport. 2001;12(2):359–363. doi: 10.1097/00001756-200102120-00035. [DOI] [PubMed] [Google Scholar]