Abstract
Background and Objectives
Metabolic syndrome (MetS) is an important risk factor for cardiovascular disease. However, the clinical outcome of acute myocardial infarction (AMI) with MetS has not been well examined. The purpose of this study was to evaluate the clinical outcomes of AMI patients with MetS.
Subjects and Methods
We evaluated a total of 6352 AMI patients who had successful percutaneous coronary interventions and could be identified for MetS between 2005 and 2008 at 51 hospitals participating in the Korea Acute Myocardial Infarction Registry. They were divided into 2 groups according to the presence of MetS: the MetS group (n=2493, 39.2%) versus the Non-MetS group (n=3859, 60.8%). In addition, 4049 AMI patients with high levels of low density lipoprotein-cholesterol (LDL-C) (≥100 mg/dL) among them, were divided into the MetS group (n=1561, 38.6%) versus the Non-MetS group (n=2488, 61.4%).
Results
In the overall population, there was no significant difference in 12-month the major adverse cardiac events (MACE) rate between the 2 groups. However, the MetS group showed a significantly higher 12-month MACE rate in the high LDL-C population. Multivariate analysis showed that MetS was an independent prognostic factor for 12-month MACE {hazard ratio (HR) 1.607, 95% confidence interval (CI) 1.027 to 2.513, adjusted p=0.038} and for 12-month target vessel revascularization (HR 1.564, 95% CI 1.092 to 2.240, adjusted p=0.015) in the high LDL-C population.
Conclusion
MetS patients with AMI in the overall population showed no significant difference in 12-month clinical outcomes. However, in patients with higher LDL-C ≥100 mg/dL, they showed significantly worse clinical outcome than Non-MetS patients. Therefore, it is important to ascertain the presence of MetS in AMI patients, and more aggressive therapy should be strongly considered for AMI patient with MetS.
Keywords: Metabolic syndrome, Myocardial infarction, Low density lipoprotein-cholesterol
Introduction
These days, the prognosis of acute myocardial infarction (AMI) remains unsatisfactory despite the development of treatments, necessitating the discovery of causes and their appropriate correction. Searching for patients who are susceptible to poor outcomes after AMI and providing them with more optimal treatment could be a solution to improving the prognosis of AMI patients.
Metabolic syndrome (MetS) is a major cause of AMI, with a rapidly increasing tendency in prevalence. It has multiple cardiovascular risk factors consisting of abdominal obesity, high blood pressure, impaired blood glucose, high triglyceride, and a low level of high density lipoprotein-cholesterol (HDL-C).1),2) There have been many studies of the relationship between MetS and cardiovascular disease.3),4) However, there remains controversy over the impact of MetS on prognosis after AMI. Furthermore, there have been relatively few studies of the impact of MetS on patients with a high level of low density lipoprotein-cholesterol (LDL-C) (more than 100 mg/dL), who have been known to be associated with an increased risk of AMI among Asian populations.5)
Therefore, our study investigated the impact of MetS on the long-term prognosis among AMI patients who underwent successful percutaneous coronary intervention (PCI), especially in patients with high levels of LDL-C (more than 100 mg/dL), using the database of the Korea Acute Myocardial Infarction Registry (KAMIR).
Subjects and Methods
Patient population
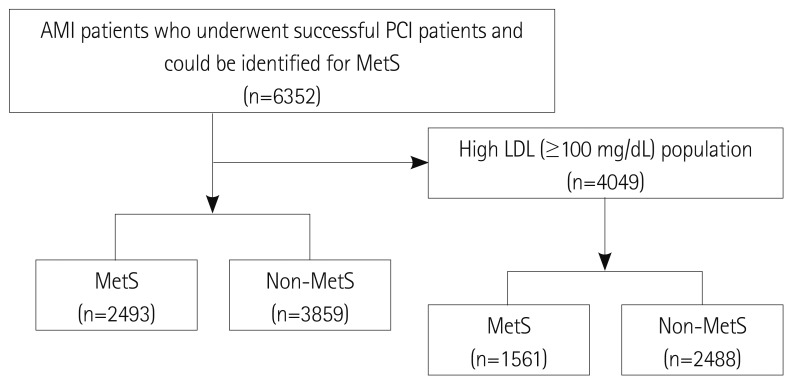
We evaluated a total of 6352 AMI patients who had successful PCI and could be identified for the presence of MetS between November 2005 and January 2008 at 51 hospitals participating in the KAMIR. They were divided into 2 groups according to presence of MetS: the MetS group (n=2493, 39.2%) versus the Non-MetS group (n=3859, 60.8%). Among them, 4049 AMI patients who had high levels of LDL-C (more over 100 mg/dL) were selected and divided into the MetS group (n=1561, 38.6%) versus the Non-MetS group (n=2488, 61.4%). The patient flow chart is shown in Fig. 1.
Fig. 1.
Patient flow chart. A total of 6352 AMI patients who had successful PCI and could be identified for MetS between November 2005 and January 2008 at 51 hospitals participating in the Korea Acute Myocardial Infarction Registry were divided into 2 groups according to the presence of MetS: the MetS group versus the Non-MetS group. Among them, 4049 AMI patients who had high LDL-C levels (more than 100 mg/dL) were divided into the MetS group versus the Non-MetS group. AMI: acute myocardial infarction, PCI: percutaneous coronary intervention, MetS: metabolic syndrome, LDL-C: low density lipoprotein-cholesterol.
The KAMIR is the largest multicenter data collection registry in Korea, designed to evaluate the outcomes of AMI patients.6) The KAMIR included 51 community and teaching hospitals and contained data for AMI patients from November 2005 to January 2008.
The diagnosis of AMI was based on a clinical symptom consistent with AMI and at least one of the following: specific changes on the electrocardiography, serial increases of serum cardiac markers of myocardial necrosis, and/or an angiographic finding of coronary artery disease (CAD). For the diagnosis of MetS, we used modified National Cholesterol Education Program-Adult Treatment Panel III criteria.7) The diagnostic criteria for MetS were elevated blood pressure (systolic ≥130 mm Hg, or diastolic ≥85 mm Hg) or drug treatment for hypertension; impaired blood glucose or drug treatment for elevated glucose or a history of diabetes; reduced HDL-C (<40 mg/dL in men, <50 mg/dL in women) or drug treatment for dyslipidemia; elevated triglyceride (≥150 mg/dL) or drug treatment for dyslipidemia; and abdominal obesity (waist circumference >90 cm in men and >80 cm in women). Our study included a history of drug treatment for hypertension instead of blood pressure and a history of drug treatment for diabetes instead of blood glucose for the diagnosis of MetS. Coronary artery angiography and stent insertion were performed using standard methods. The decision for detailed treatment was left up to the physician's discretion. A clinical follow-up was performed during a period of 12 months after enrollment.
The primary end points were cardiac death and a composite of major adverse clinical events (MACE) during the 12-month follow-up period. MACE included cardiac death, myocardial infarction, and target vessel revascularization (TVR). Cardiac death was defined as death from pump failure, arrhythmia, ventricular septal rupture, or free wall rupture.
Statistical analysis
The Statistical Package for the Social Sciences (SPSS) for Windows version 18.0 (SPSS Inc., Chicago, IL, USA) was used for all analyses. Continuous variables were expressed as mean±standard deviation or median (interquartile range), and compared with an independent t-test or Mann-Whitney U test. Categorical variables were expressed as percentage and compared with a chi-square test. Missing values were coded as "missing" in SPSS. Cox regression analysis was performed to analyze the independent impact of MetS on 12-month clinical outcomes in AMI patients who had undergone successful PCI. All variables with p<0.2 in univariate analysis were entered into a multivariate analysis. The results are presented as adjusted hazard ratios (HRs) with 95% confidence intervals (CIs) and p. Co-variables, which were used to adjust for the difference in baseline characteristics in 12-month MACE, were age, gender, atypical symptoms, chest pain, low blood pressure, heart rate, cerebrovascular disease, smoking, multi-vessel disease, drug eluting stent (DES), left ventricular ejection fraction, creatinine clearance rate (CrCl), N-terminal pro-B type natriuretic peptide (NT-proBNP), high-sensitivity C-reactive protein (hsCRP), troponin T, heparin, glycoprotein IIb/IIIa (GP2b3a) inhibitors, angiotensin-converting enzyme inhibitors or angiotensin receptor blockers, beta blockers, calcium channel blockers (CCBs), and statin.
All variables were considered to be statistically significant when the p was less than 0.05.
Ethics statement
Our study was conducted according to the Declaration of Helsinki. The study protocol was approved by the institutional review board of all centers, and the approval number was 05-49 of Chonnam National University Hospital. Written informed consent was obtained from all patients.
Results
Baseline characteristics
A total of 6352 patients were included in the overall population; 2493 (39.2%) patients with MetS and 3859 (60.8%) patients without MetS. Among them, a total of 4049 patients were included in the high LDL-C population (≥100 mg/dL); 1561 (38.6%) patients with MetS and 2488 (61.4%) patients without MetS.
The baseline characteristics are summarized in Table 1. In the overall population, compared to the Non-MetS group, the MetS group was older, more likely to be female, and had more atypical symptoms, a higher Killip class (class>II) and a higher tendency of non-ST-elevation myocardial infarction (NSTEMI). Both symptom to door time and door to balloon time were longer in the MetS group. The MetS group had more comorbidity factors, such as diabetes, hypertension, dyslipidemia, and a past history of CAD. In addition, hs-CRP and NT-proBNP were higher, but creatine kinase-MB and troponin I, and CrCl were lower in the MetS group. Triglyceride and blood sugar were higher and HDL-C was lower in the MetS group. There were no significant differences in the level of LDL-C between the 2 groups. Statin was similarly prescribed in both groups, but less GP2b3A inhibitor and more CCBs were prescribed in the MetS group.
Table 1.
Baseline clinical characteristics
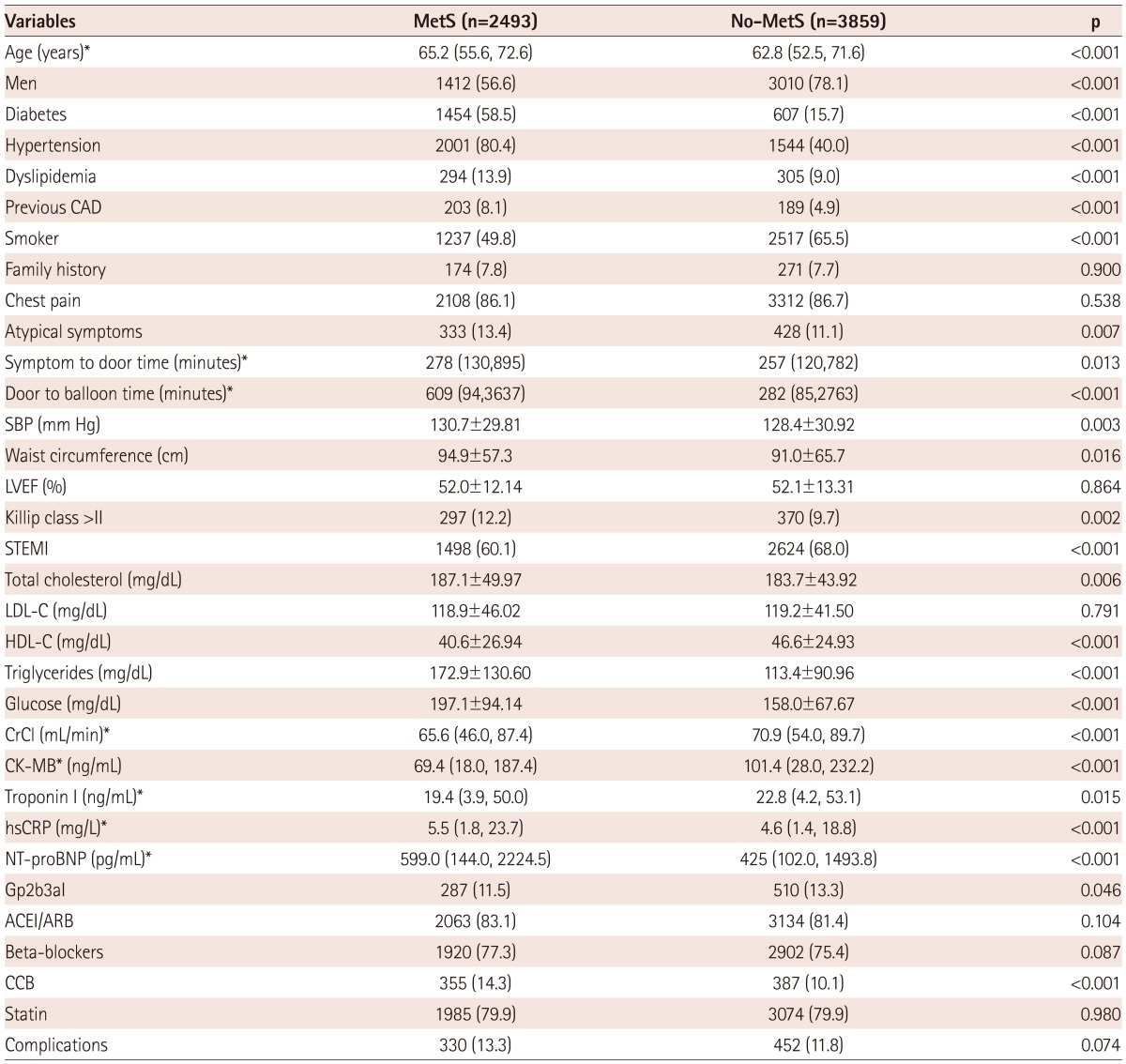
Data are presented as the n (%) of patients or mean±SD. *Values are expressed as the median (interquartile range). MetS: metabolic syndrome, CAD: coronary artery disease, SBP: systolic blood pressure, LVEF: left ventricular ejection fraction, STEMI: ST elevation myocardial infarction, LDL-C: low density lipoprotein-cholesterol, HDL-C: high density lipoprotein-cholesterol, CrCl: creatinine clearance, CK-MB: creatine kinase-MB, hsCRP: high-sensitivity C-reactive protein, NT-proBNP: N-terminal pro-B type natriuretic peptide, Gp2b3aI: glycoprotein IIb/IIIa inhibitor, ACEI: angiotensin-converting enzyme inhibitors, ARB: angiotensin receptor blockers, CCB: calcium channel blocker
Coronary angiographic findings
Coronary angiographic findings are summarized in Table 2. Multi-vessel involvement and right coronary artery involvement were more common in the MetS group. Pre-PCI Thrombolysis in Myocardial Infarction (TIMI) flow grade 0 was less common in the MetS group. There were no differences in the American College of Cardiology/American Heart Association lesion type, post-PCI TIMI flow grade, and stent sizes between the MetS and Non-MetS groups. The stent number was higher, and stent diameter was smaller in the MetS group.
Table 2.
Coronary angiographic findings
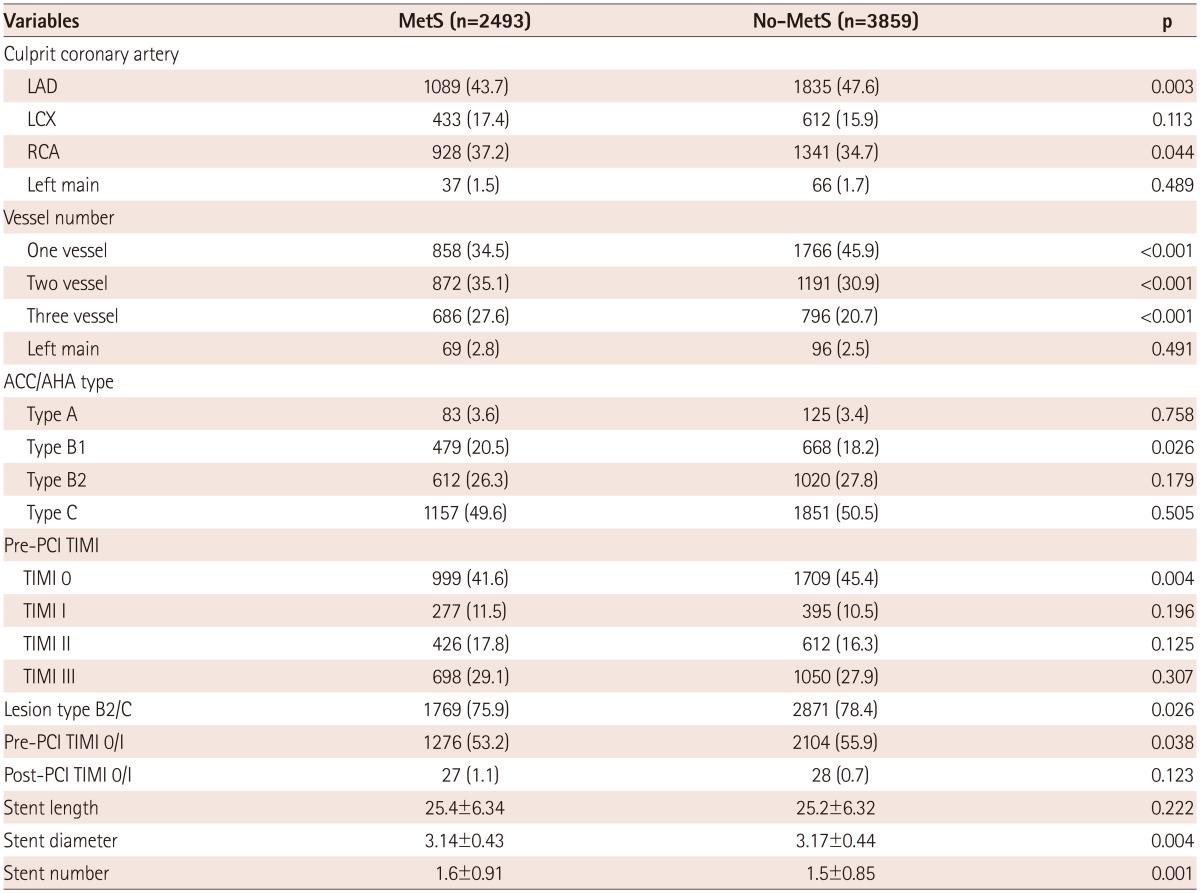
Data are presented as the n (%) of patients or mean±SD. MetS: metabolic syndrome, LAD: left anterior descending artery, LCX: left circumflex artery, RCA: right coronary artery, ACC/AHA: American College of Cardiology/American Heart Association, PCI: percutaneous coronary intervention, TIMI: Thrombolysis in Myocardial Infarction
Clinical outcomes at 12 months in patients with AMI
A total of 6352 patients were included in the overall population, and a total of 4049 patients among them were included in the high LDL-C population.
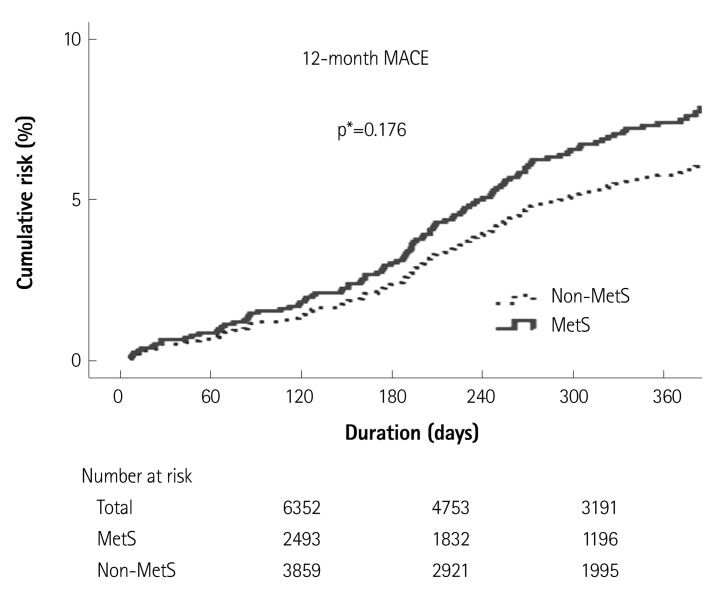
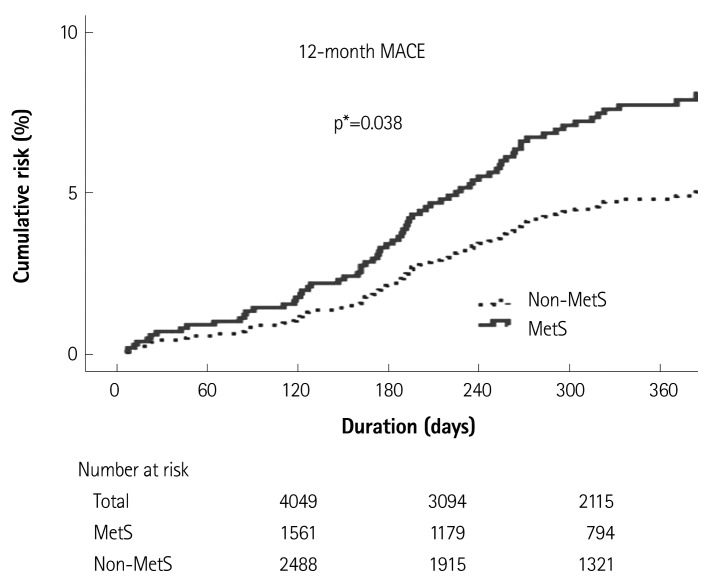
Clinical outcomes in the overall population and in the high LDL-C population are listed in Table 3 and 4. In the overall population, the in-hospital mortality rate was higher in the MetS group, but there was no significant difference. In addition, the 12-month MACE rates in the MetS group versus the Non-MetS groups of the overall population were 8.8% vs. 6.2%, respectively. The numbers of patients included in the final Cox regression analysis for the 12-month MACE were 1666 in the overall population, and there was no significant difference in the 12-month MACE rates (adjusted p=0.176) between the MetS group and Non-MetS groups (Table 3, Fig. 2). However, in the high LDL-C population, the 12-month MACE rates of the MetS group versus the Non-MetS group were 8.2% vs. 5.5%, and the 12-month TVR rates were 3.9% vs. 2.9%, respectively. The numbers of patients included in the final Cox regression analysis for the 12-month MACE rates were 1152 in the high LDL population, and there was a significant difference in 12-month MACE rates (adjusted p=0.038) as well as 12-month TVR rates (adjusted p=0.015) (Table 4, Fig. 3).
Table 3.
Clinical outcomes in the overall population
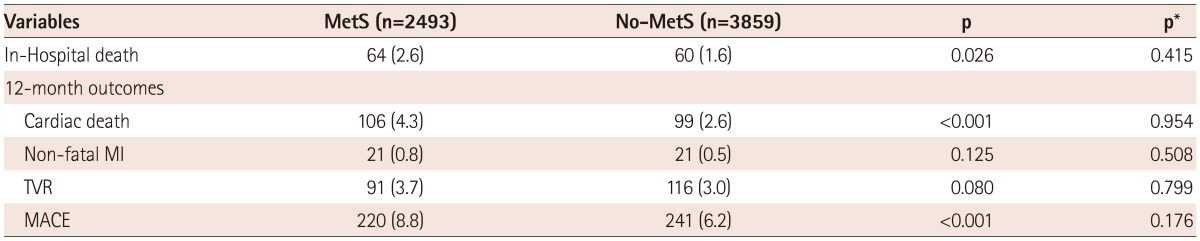
Values are n (%). All comparisons were made using the chi-square test. P was calculated by log rank analysis, and p* was calculated by multivariate Cox regression analysis. Major adverse cardiac events included cardiac deaths, recurrent myocardial infarction, and target vessel revascularization. p*: adjusted p, MetS: metabolic syndrome, MI: myocardial infarction, TVR: target vessel revascularization, MACE: major adverse cardiac event(s)
Table 4.
Clinical outcomes in the high LDL-C population
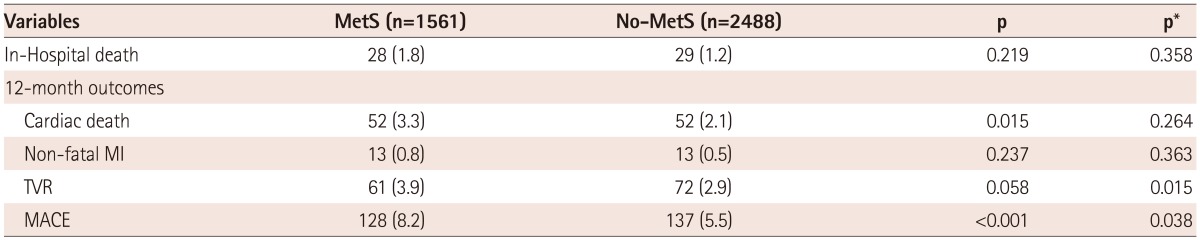
Values are n (%). All comparisons were made using the chi-square test. P was calculated by log rank analysis, and p* was calculated by multivariate Cox regression analysis. Major adverse cardiac events included cardiac deaths, recurrent myocardial infarction, and target vessel revascularization. p*: adjusted p, LDL-C: low density lipoprotein-cholesterol, MetS: metabolic syndrome, MI: myocardial infarction, TVR: target vessel revascularization, MACE: major adverse cardiac event(s)
Fig. 2.
Twelve-month MACE in the overall population. In the overall population, 12-month MACE rates were higher in the MetS group than in the Non-MetS group, but there was no significant difference between the 2 groups. MACE: major adverse cardiac events, MetS: metabolic syndrome, p*: adjusted p.
Fig. 3.
Twelve-month clinical outcomes in the high LDL-C population. In the high LDL-C population, 12-month MACE rates were higher in the MetS group than in the Non-MetS group, and there was a significant difference between the 2 groups. LDL-C: low density lipoprotein-cholesterol, MACE: major adverse cardiac events, MetS: metabolic syndrome, p*: adjusted p.
Predictors of major adverse cardiovascular events
Multivariate analysis using the Cox proportional hazard model in the overall population showed that MetS was not an independent predictor of the 12-month MACE rate (HR 1.284, 95% CI 0.894 to 1.843, adjusted p=0.176) in AMI. However, multivariate analysis in the high LDL-C population, which is shown in Table 5, showed that MetS was an independent predictor of 12-month MACE rate (HR 1.607, 95% CI 1.027 to 2.513, adjusted p=0.038) and of the 12-month TVR (HR 1.564, 95% CI 1.092 to 2.240, adjusted p=0.015) in the high LDL-C population of AMI.
Table 5.
Multivariate analysis of 12-month MACE in the high LDL-C population
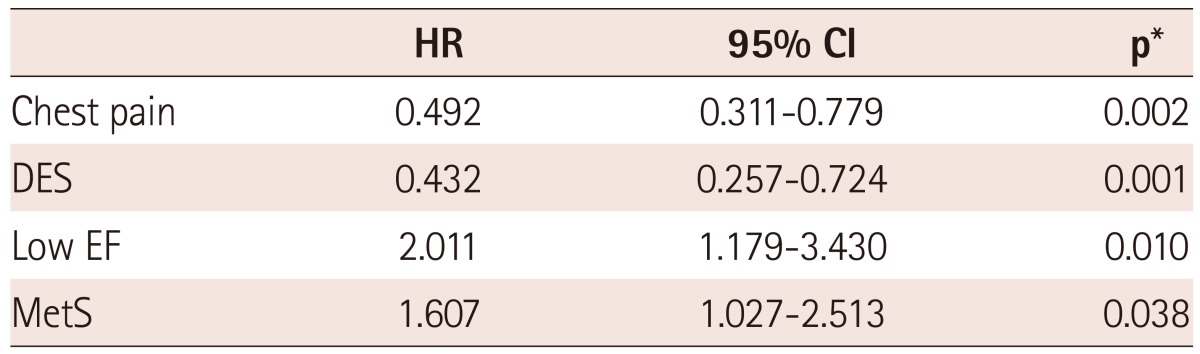
p* value was calculated using the multivariate Cox regression analysis. MACE: major adverse cardiac events, LDL-C: low density lipoprotein-cholesterol, HR: hazard ratio, CI: confidence interval, p*: adjusted p, DES: drug-eluting stent, EF: ejection fraction, MetS: metabolic syndrome
Discussion
The main finding of this study was that MetS was an independent predictor for 12-month MACE and TVR in the high LDL-C population, although it was not an independent predictor in the overall population. There were no significant differences in the incidences of cardiac death and Non-fatal myocardial infarction between the MetS group and the Non-MetS groups in the high LDL-C population.
There have been many studies that have reported an impact of MetS on cardiovascular morbidity and mortality in AMI patients, and there have been some disagreements formed. Some studies showed that MetS was associated with poor outcomes after AMI, including severe heart failure, by Zeller et al.8) and in-hospital death in patients with ST elevation myocardial infarction (STEMI), by Lee et al.9) Other studies, however, have found that MetS was not associated with poor outcomes after PCI. Kim et al.10) reported that MetS was not an independent predictor of increased in-stent restenosis and MACE in patients with angina and significant coronary artery stenosis, excluding STEMI. Hoffmann et al.11) also reported that MetS patients without diabetes did not have an increased risk for MACE during a 12-month follow-up period after stent placement. Rana et al.12) also showed that MetS was not associated with TVR after PCI in patients with stable angina, non-ST-elevation acute coronary syndromes, or silent ischemia with PCI. However, meta-analysis showed that MetS was associated with a significant increased risk of post-stent all-cause mortality and MACE in CAD patients with DES.13)
Our results in the overall population were consistent with previous studies by Kim et al.10) and Rana et al.12) However, our results in the high LDL-C population showed similar trends in results to the previous meta-analysis, although there are some differences. MetS in the high LDL-C population was associated with an increased risk of MACE and TVR in our study, while an increased risk of all-cause mortality and MACE in the meta-analysis. These discrepancies between meta-analysis findings and our study may be due to the dissimilarities of patient populations, which are differences in the species of stents, and follow-up periods. In addition, our study included diabetic patients, while other studies did not. Coming together with these earlier studies, our study might suggest that MetS could be an important risk factor in some populations of AMI patients.
There are possible mechanisms to explain how MetS could have an adverse influence on the clinical outcomes in AMI patients. First, there is a large difference in the dyslipidemia pattern between the MetS group and the Non-MetS group. The MetS group had atherogenic dyslipidemia, which consists of high triglyceride and lower HDL-C, as well as qualitative dyslipidemia, which consists of small dense LDL-C (sd-LDL-C) and lipoprotein (Lp) (a).14),15) Sd-LDL-C and Lp (a), rather than LDL-C, have been known to be major risk factors for atherogenic dyslipidemia16) as well as for the severity of CAD,17),18) which causes oxidative stress, endothelial dysfunction, and the inflammation of vessels.19)
Second, inflammation caused by obesity has been known to play an important role in the pathogenesis of MetS-associated re-stenosis by releasing inflammatory adipocytokines, such as leptin, tumor necrosis factor-α, plasminogen activator inhibitor-1, and interleukins 1 and 6.20),21) In the present study, hsCRP, a biomarker for inflammation that adds prognostic information to MetS, was higher in the MetS group than in the Non-MetS group. Moreover, the rate of abdominal obesity was higher in MetS patients in the high LDL-C population than in those of the overall population (83.8% vs. 81.6%), respectively.
Finally, statin is more effective in AMI patients with MetS than in those without MetS, because it not only has an effect on the atherogenic dyslipidemia associated with MetS in hypercholesterolemic patients,22) which reduces sd-LDL-C as well as LDL-C,23),24) but also a characteristic pleiotropic effect, such as an anti-inflammatory, antioxidant effect, and the modulation of endothelial dysfunction,25-27) resulting in enhanced myocardial perfusion.28),29) In the present study, however, statin was underprescribed and similarly prescribed to both the MetS and Non-MetS groups in AMI patients with high LDL-C, without any consideration of possible benefits according to statin treatment in MetS patients. Therefore, these factors, consisting of atherogenic dyslipidemia, inflammation, and the suboptimal use of statin, may play a crucial role in worsening the outcome of AMI patients with MetS in the high LDL-C population over those without MetS.
Some aspects of our results should be of concern. First of all, the present study showed that door to balloon time was longer and GP-2b3A inhibitor was less prescribed in the MetS group. This might mean hesitation toward aggressive treatment in AMI patients with MetS, reminding us that there are certain factors involved in less aggressive treatment. Our study showed that MetS patients were more likely to be female and had poor features such as older age, a higher Killip class, or lower CrCl than those without MetS. Atypical presentation as well as NSTEMI was more common among them. These factors may be related to the reluctance for, or delay of, aggressive treatment and, subsequently, suboptimal management in AMI patients with MetS, resulting in worsened clinical outcomes. Therefore, we suggest that careful consideration for the presence of MetS in AMI patients, especially in patients with poor characteristics or in patients with atypical presentation or NSTEMI, should be made, and more rapid and aggressive treatment should be given to those patients to improve their clinical outcomes.
Second, the present study reveals the state of statin treatment for AMI patients in real-world clinical practice. Statin has been indicated in AMI patients, regardless of LDL-C level, by recent guidelines.30) However, our study showed that statin treatment rates were suboptimal and there was no significant difference in the statin prescription rate between the MetS group and the Non-MetS group. The inference from this finding is that most physicians might pay close attention to only LDL-C level, without any consideration of MetS, an examination of which may benefit from statin treatment, leading to similar prescription rates of statin in real-world clinical practice. Therefore, we suggest that statin treatment rates should be enhanced in AMI patients, especially among patients with MetS.
Finally, symptom to door time was longer in AMI patients with MetS than among those without MetS in the present study, which might negatively influence the clinical outcomes. What might be the reason for this? MetS patients had a higher prevalence of diabetes and were older. They were also more likely to be female. These factors could, in turn, have led to more atypical symptoms with a higher rate in MetS patients than in Non-MetS patients (13.4% vs. 11.1%, p=0.007, respectively). These factors might be related to delays in hospital visits, because it is not easy for most patients to recognize that atypical symptoms are associated with CAD. Therefore, we suggest that meticulous education in atypical symptoms, as a type of possible presentation of AMI, should be given to patients who are older or hypertensive or diabetic in outpatient clinics before the onset of AMI, in addition to conventional treatment.
Our analysis faces several limitations. First, although these results come from a large cohort study, this was a multi-center trial and retrospective study, so there is the possibility of selection bias. Second, there are many missing data features, including fasting blood sugar, sd-LDL-C, Lp (a), and 12-month follow-up LDL level in our registry. Third, our data for blood pressure and blood glucose were checked at admission. The checked level of blood glucose (mean; 173.4±81.38 mg/dL) and blood pressure (mean; systolic 129.3±30.51 mg/dL) were higher than the usual condition because of the unstable and emergent condition. Thus, it seemed to be inappropriate to use those data points for the diagnosis of MetS. Our study might have overcome this limitation by involving patients with a known history of hypertension and diabetes instead of blood pressure and blood glucose. The present study might be a reference for MetS patients with AMI in the Korean population, because, to date, the data for MetS patients with AMI is relatively insufficient. More specialized long-term follow up studies are required in this area.
In conclusion, AMI patients with MetS had significantly worse clinical outcomes when compared with Non-MetS patients, in patients with higher LDL-C of more than 100 mg/dL, although those in the overall population showed no significant difference in 12-month clinical outcomes. Therefore, it is important to ascertain the presence of MetS in AMI patients, and provide more aggressive therapy, including the early initiation of PCI, as well as the greater use of GP2b3A inhibitors. Greater statin treatment should also be strongly considered.
Acknowledgments
This study was conducted with the support of the Korean Circulation Society in commemoration of its 50th Anniversary.
Footnotes
The authors have no financial conflicts of interest.
References
- 1.Skilton MR, Moulin P, Sérusclat A, Nony P, Bonnet F. A comparison of the NCEP-ATPIII, IDF and AHA/NHLBI metabolic syndrome definitions with relation to early carotid atherosclerosis in subjects with hypercholesterolemia or at risk of CVD: evidence for sex-specific differences. Atherosclerosis. 2007;190:416–422. doi: 10.1016/j.atherosclerosis.2006.02.019. [DOI] [PubMed] [Google Scholar]
- 2.Lorenzo C, Okoloise M, Williams K, Stern MP, Haffner SM San Antonio Heart Study. The metabolic syndrome as predictor of type 2 diabetes: the San Antonio heart study. Diabetes Care. 2003;26:3153–3159. doi: 10.2337/diacare.26.11.3153. [DOI] [PubMed] [Google Scholar]
- 3.Girman CJ, Rhodes T, Mercuri M, et al. The metabolic syndrome and risk of major coronary events in the Scandinavian Simvastatin Survival Study (4S) and the Air Force/Texas Coronary Atherosclerosis Prevention Study (AFCAPS/TexCAPS) Am J Cardiol. 2004;93:136–141. doi: 10.1016/j.amjcard.2003.09.028. [DOI] [PubMed] [Google Scholar]
- 4.Selcuk H, Temizhan A, Selcuk MT, et al. Impact of metabolic syndrome on future cardiovascular events in patients with first acute myocardial infarction. Coron Artery Dis. 2009;20:370–375. doi: 10.1097/MCA.0b013e32832ed31e. [DOI] [PubMed] [Google Scholar]
- 5.Karthikeyan G, Teo KK, Islam S, et al. Lipid profile, plasma apolipoproteins, and risk of a first myocardial infarction among Asians: an analysis from the INTERHEART Study. J Am Coll Cardiol. 2009;53:244–253. doi: 10.1016/j.jacc.2008.09.041. [DOI] [PubMed] [Google Scholar]
- 6.Sim DS, Jeong MH, Kang JC. Current management of acute myocardial infarction: experience from the Korea Acute Myocardial Infarction Registry. J Cardiol. 2010;56:1–7. doi: 10.1016/j.jjcc.2010.04.002. [DOI] [PubMed] [Google Scholar]
- 7.Grundy SM, Cleeman JI, Daniels SR, et al. Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation. 2005;112:2735–2752. doi: 10.1161/CIRCULATIONAHA.105.169404. [DOI] [PubMed] [Google Scholar]
- 8.Zeller M, Steg PG, Ravisy J, et al. Prevalence and impact of metabolic syndrome on hospital outcomes in acute myocardial infarction. Arch Intern Med. 2005;165:1192–1198. doi: 10.1001/archinte.165.10.1192. [DOI] [PubMed] [Google Scholar]
- 9.Lee MG, Jeong MH, Ahn Y, et al. Impact of the metabolic syndrome on the clinical outcome of patients with acute ST-elevation myocardial infarction. J Korean Med Sci. 2010;25:1456–1461. doi: 10.3346/jkms.2010.25.10.1456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kim JS, Lee HC, Choi BK, et al. Impact of metabolic syndrome on instent restenosis and clinical outcomes after percutaneous coronary stent implantation. Diabetes Res Clin Pract. 2010;88:e38–e41. doi: 10.1016/j.diabres.2010.03.024. [DOI] [PubMed] [Google Scholar]
- 11.Hoffmann R, Stellbrink E, Schröder J, et al. Impact of the metabolic syndrome on angiographic and clinical events after coronary intervention using bare-metal or sirolimus-eluting stents. Am J Cardiol. 2007;100:1347–1352. doi: 10.1016/j.amjcard.2007.06.021. [DOI] [PubMed] [Google Scholar]
- 12.Rana JS, Monraats PS, Zwinderman AH, et al. Metabolic syndrome and risk of restenosis in patients undergoing percutaneous coronary intervention. Diabetes Care. 2005;28:873–877. doi: 10.2337/diacare.28.4.873. [DOI] [PubMed] [Google Scholar]
- 13.Xu D, Guo Y, Wang H, et al. The angiographic and clinical outcomes after coronary stenting in patients with metabolic syndrome. Atherosclerosis. 2012;221:416–421. doi: 10.1016/j.atherosclerosis.2011.12.016. [DOI] [PubMed] [Google Scholar]
- 14.Ascaso J, Gonzalez Santos P, Hernandez Mijares A, et al. Management of dyslipidemia in the metabolic syndrome: recommendations of the Spanish HDL-Forum. Am J Cardiovasc Drugs. 2007;7:39–58. doi: 10.2165/00129784-200707010-00004. [DOI] [PubMed] [Google Scholar]
- 15.Fukushima Y, Hirayama S, Ueno T, et al. Small dense LDL cholesterol is a robust therapeutic marker of statin treatment in patients with acute coronary syndrome and metabolic syndrome. Clin Chim Acta. 2011;412:1423–1427. doi: 10.1016/j.cca.2011.04.021. [DOI] [PubMed] [Google Scholar]
- 16.Koba S, Yokota Y, Hirano T, et al. Small LDL-cholesterol is superior to LDL-cholesterol for determining severe coronary atherosclerosis. J Atheroscler Thromb. 2008;15:250–260. doi: 10.5551/jat.e572. [DOI] [PubMed] [Google Scholar]
- 17.Moon JY, Kwon HM, Kwon SW, et al. Lipoprotein(a) and LDL particle size are related to the severity of coronary artery disease. Cardiology. 2007;108:282–289. doi: 10.1159/000099097. [DOI] [PubMed] [Google Scholar]
- 18.Yazdandoust S, Parizadeh SM, Moohebati M, et al. Serum small dense low-density lipoprotein concentrations are elevated in patients with significant coronary artery stenosis and are related to features of the metabolic syndrome. Lipids. 2012;47:963–972. doi: 10.1007/s11745-012-3706-x. [DOI] [PubMed] [Google Scholar]
- 19.Goswami B, Rajappa M, Singh B, Ray PC, Kumar S, Mallika V. Inflammation and dyslipidaemia: a possible interplay between established risk factors in North Indian males with coronary artery disease. Cardiovasc J Afr. 2010;21:103–108. [PMC free article] [PubMed] [Google Scholar]
- 20.Greenberg AS, Obin MS. Obesity and the role of adipose tissue in inflammation and metabolism. Am J Clin Nutr. 2006;83:461S–465S. doi: 10.1093/ajcn/83.2.461S. [DOI] [PubMed] [Google Scholar]
- 21.Goyal SN, Bharti S, Krishnamurthy B, Agrawal Y, Ojha SK, Arya DS. Impact of metabolic syndrome on re-stenosis development: role of drug-eluting stents. Diab Vasc Dis Res. 2012;9:177–188. doi: 10.1177/1479164111430336. [DOI] [PubMed] [Google Scholar]
- 22.Deedwania PC, Hunninghake DB, Bays HE, et al. Effects of rosuvastatin, atorvastatin, simvastatin, and pravastatin on atherogenic dyslipidemia in patients with characteristics of the metabolic syndrome. Am J Cardiol. 2005;95:360–366. doi: 10.1016/j.amjcard.2004.09.034. [DOI] [PubMed] [Google Scholar]
- 23.Baldassarre S, Scruel O, Deckelbaum RJ, Dupont IE, Ducobu J, Carpentier YA. Beneficial effects of atorvastatin on sd LDL and LDL phenotype B in statin-naive patients and patients previously treated with simvastatin or pravastatin. Int J Cardiol. 2005;104:338–345. doi: 10.1016/j.ijcard.2005.01.006. [DOI] [PubMed] [Google Scholar]
- 24.Bahadir MA, Oguz A, Uzunlulu M, Bahadir O. Effects of different statin treatments on small dense low-density lipoprotein in patients with metabolic syndrome. J Atheroscler Thromb. 2009;16:684–690. doi: 10.5551/jat.1123. [DOI] [PubMed] [Google Scholar]
- 25.Devaraj S, Chan E, Jialal I. Direct demonstration of an antiinflammatory effect of simvastatin in subjects with the metabolic syndrome. J Clin Endocrinol Metab. 2006;91:4489–4496. doi: 10.1210/jc.2006-0299. [DOI] [PubMed] [Google Scholar]
- 26.Ray KK, Cannon CP. Intensive statin therapy in acute coronary syndromes: clinical benefits and vascular biology. Curr Opin Lipidol. 2004;15:637–643. doi: 10.1097/00041433-200412000-00003. [DOI] [PubMed] [Google Scholar]
- 27.Filardi PP, Cecere M, Savarese G, et al. [Inflammation and lipids in the coronary pathology. Risk factors, causes or therapeutic target?] G Ital Cardiol (Rome) 2010;11(12 Suppl 3):10S–15S. [PubMed] [Google Scholar]
- 28.Ulus T, Parspour A, Cavusoglu Y, Entok E, Uslu I, Demirustu C. Statins improve myocardial perfusion in metabolic syndrome patients who have perfusion defects on myocardial perfusion imaging and angiographically normal coronary arteries. Eur Rev Med Pharmacol Sci. 2012;16:328–334. [PubMed] [Google Scholar]
- 29.Murrow JR, Sher S, Ali S, et al. The differential effect of statins on oxidative stress and endothelial function: atorvastatin versus pravastatin. J Clin Lipidol. 2012;6:42–49. doi: 10.1016/j.jacl.2011.08.006. [DOI] [PubMed] [Google Scholar]
- 30.Task Force on the management of ST-segment elevation acute myocardial infarction of the European Society of Cardiology (ESC) Steg PG, James SK, et al. ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur Heart J. 2012;33:2569–2619. doi: 10.1093/eurheartj/ehs215. [DOI] [PubMed] [Google Scholar]