Abstract
Purpose
The joint effects of genetic, ocular, and environmental variables were evaluated and predictive models for prevalence and incidence of AMD were assessed.
Methods
Participants in the multicenter Age-Related Eye Disease Study (AREDS) were included in a prospective evaluation of 1446 individuals, of which 279 progressed to advanced AMD (geographic atrophy or neovascular disease) and 1167 did not progress during 6.3 years of follow-up. For prevalent AMD, 509 advanced cases were compared with 222 controls. Covariates for the incidence analysis included age, sex, education, smoking, body mass index (BMI), baseline AMD grade, and the AREDS vitamin–mineral treatment assignment. DNA specimens were evaluated for six variants in five genes related to AMD. Unconditional logistic regression analyses were performed for prevalent and incident advanced AMD. An algorithm was developed and receiver operating characteristic curves and C statistics were calculated to assess the predictive ability of risk scores to discriminate progressors from nonprogressors.
Results
All genetic polymorphisms were independently related to prevalence of advanced AMD, controlling for genetic factors, smoking, BMI, and AREDS treatment. Multivariate odds ratios (ORs) were 3.5 (95% confidence interval [CI], 1.7–7.1) for CFH Y402H; 3.7 (95% CI, 1.6 – 8.4) for CFH rs1410996; 25.4 (95% CI, 8.6 –75.1) for LOC387715 A69S (ARMS2); 0.3 (95% CI, 0.1– 0.7) for C2 E318D; 0.3 (95% CI, 0.1– 0.5) for CFB; and 3.6 (95% CI, 1.4 –9.4) for C3 R102G, comparing the homozygous risk/protective genotypes to the referent genotypes. For incident AMD, all these variants except CFB were significantly related to progression to advanced AMD, after controlling for baseline AMD grade and other factors, with ORs from 1.8 to 4.0 for presence of two risk alleles and 0.4 for the protective allele. An interaction was seen between CFH402H and treatment, after controlling for all genotypes. Smoking was independently related to AMD, with a multiplicative joint effect with genotype on AMD risk. The C statistic for the full model with all variables was 0.831 for progression to advanced AMD.
Conclusions
Factors reflective of nature and nurture are independently related to prevalence and incidence of advanced AMD, with excellent predictive power.
Knowledge about genetic and epidemiologic associations with the leading cause of blindness among the elderly, age-related macular degeneration, has grown exponentially in recent years. Several genetic variants with strong and consistent associations with AMD have recently been identified.1–15 We also know that in addition to age, ethnicity, and family history, there are modifiable factors: smoking,16 –19 nutritional antioxidants and omega-3 fatty acid intake,20 –27 and overall and abdominal adiposity.28 –30 However, it remains unknown whether all these genetic and environmental factors act independently or jointly and to what extent they as a group can predict the occurrence of AMD or progression to advanced AMD from early and intermediate stages. Such information may be useful for screening those at high risk due to a positive family history or having signs of early or intermediate disease, among whom some progress to advanced stages of AMD with visual loss. Early detection could reduce the growing societal burden due to AMD by targeting and emphasizing modifiable habits earlier in life and recommending more frequent surveillance for those highly susceptible to the disease. Treatment trials will also benefit from such information by enrolling participants with more homogeneous risks of developing the outcome in question. To develop and evaluate such a model, we assessed the relative contribution of genetic and environmental factors toward prevalence and incidence of advanced AMD in a well-characterized multicenter study population. We also developed an algorithm and receiver operating curve models to discriminate between individuals who developed advanced AMD and those who did not.
Methods
Phenotypic Data
The Age-Related Eye Disease Study (AREDS) included a randomized clinical trial to assess the effect of antioxidant and mineral supplements on risk of AMD and cataract and a longitudinal study of AMD that ended in December 2005. The study procedures have been reported.20 Based on ocular examination and AREDS reading center photographic grading of fundus photographs, Caucasian participants in the present study were defined as AREDS category 1 in both eyes (essentially free of age-related macular abnormalities), category 2 in the worst eye (mild changes including multiple small drusen, nonextensive intermediate drusen, and/or pigment abnormalities), category 3 in the worst eye (at least one large drusen of at least 125-μm diameter, extensive intermediate drusen, and/or noncentral geographic atrophy), category 4 in one eye (advanced AMD, either neovascular or central geographic atrophy, or visual loss due to AMD regardless of phenotype), or category 4 in both eyes. Non-Caucasians were excluded because the distribution of advanced AMD in that population differs considerably from that among Caucasians.30 Since group 4 in the original AREDS classification included both advanced forms of AMD and visual loss regardless of phenotype,20 we reclassified this group into grades 4 and 5: grade 4 including both noncentral and central geographic atrophy and grade 5 including neovascular disease, independent of visual acuity level using the Clinical Age-Related Maculopathy Grading System (CARMS),31 to determine whether results differed between these two advanced dry and wet phenotypes. Comparisons were also made between unilateral or bilateral advanced AMD according to the AREDS system.20 Cases were defined as having advanced AMD and progression was defined as incident advanced AMD based on the AMD grade at the end of the clinical trial in 2001, with a mean follow-up time of 6.3 years. Progressors were those individuals with early or intermediate AMD at baseline who progressed to advanced AMD during follow-up, and individuals with advanced AMD in one eye at baseline who progressed to advanced AMD in both eyes.
Demographic and risk factor data, including education, smoking history, and body mass index, were obtained at the baseline visit from questionnaires and height and weight measurements. Antioxidant status was defined as taking antioxidants (antioxidants alone or antioxidants and zinc) or no antioxidants (placebo or zinc alone) in the clinical trial. AREDS treatment groups included placebo, antioxidants alone, zinc, and antioxidants plus zinc. The research protocol was approved by institutional review boards, and all participants signed informed consent statements. Research adhered to the tenets of the Declaration of Helsinki.
Genotyping
DNA samples, which were obtained beginning in 1998, were obtained from the AREDS Genetic Repository. The following six common single-nucleotide polymorphisms (SNPs) associated with AMD were evaluated: (1) complement factor H (CFH) Y402H (rs1061170) in exon 9 of the CFH gene on chromosome 1, region q31, with the change 1277T>C resulting in a substitution of a histidine for a tyrosine at codon 402 of the CFH protein; (2) CFH rs1410996, an independently associated SNP within intron 14 of CFH; (3) LOC387715 A69S, ARMS2, (rs10490924 in the LOC387715/HTRA1 region of chromosome 10), a nonsynonymous coding SNP variant in exon 1 of LOC387715, resulting in a substitution of the amino acid serine for alanine at codon 69; (4) complement factor 2 or C2 E318D (rs9332739), the nonsynonymous coding SNP variant in exon 7 of C2 resulting in the amino acid glutamic acid changing to aspartic acid at codon 318; (5) complement factor B or CFB R32Q (rs641153), the nonsynonymous coding SNP variant in exon 2 of CFB resulting in the amino acid glutamine changing to arginine at codon 32; and (6) complement factor 3 or C3 R102G (rs2230199), the nonsynonymous coding SNP variant in exon 3 of C3 resulting in the amino acid change of glycine to arginine at codon 102. For the genetic variant on chromosome 10, LOC387715 A69S, whether the gene HTRA1 adjacent to it is in fact the AMD-susceptibility gene on 10q269 –11 remains debatable; however, the relevant SNPs in these two genes have been reported to be nearly perfectly correlated. Thus, although the other SNP is a promising candidate variant, rs10490924 used in this study can be considered a surrogate for the causal variant that resides in this region. For the C2/CFB genes, there are two independent associations to the C2/CFB locus, but because of linkage disequilibrium, we do not know which of the two genes or both are functionally affected. Genotyping was performed using primer mass extension and MALDI-TOF MS analysis (MassEXTEND methodology of Sequenom, San Diego, CA) at the Broad Institute Center for Genotyping and Analysis (Cambridge, MA).
Statistical Analyses
Individuals with advanced AMD were compared to the control group of persons with no AMD, and progressors were compared to nonprogressors with regard to genotype and risk factor data. Multivariate unconditional logistic regression analysis was performed to evaluate the relationships between prevalence or progression of AMD and all the genotypes plus various risk factors, controlling for age (70 years or older versus younger than 70), sex, and education (high school or less versus more than high school), cigarette smoking (never, past, or current), and body mass index (BMI), which was calculated as the weight in kilograms divided by the square of the height in meters (<25, 25–29.9, and 30+). Baseline AMD grade was included in the progression models. The AREDS assignment in the randomized clinical trial was also added to the multivariate model (taking a supplement containing antioxidants or taking study supplements containing no antioxidants for the case– control comparison, or the four treatment groups for the progression analyses). Tests for multiplicative interactions between each of the genotypes versus smoking and BMI were calculated by using cross product terms according to genotype and the individual risk factors. Similar analyses were performed to assess gene–gene interactions for each combination of genes. ORs and 95% CIs were calculated for each risk factor and within the three genotype groups. Tests for trend for the number of risk alleles for each genetic variant (0, 1, or 2) were calculated.32 Sensitivities and specificities for a variety of risk score cutoffs were evaluated to assess the optimal use of the model for individual risk prediction (i.e., sensitivities and specificities of at least 80%). The method for calculation of the AMD progression risk score based on all ocular, demographic, behavioral, genetic and treatment factors is explained in Table 1, and is illustrated for a randomly selected progressor and nonprogressor.
Table 1.
Calculation of AMD Progression Risk Score where βi and Xi are given as follows
| i | Variable Name (Xi) | Regression Coefficient βi | Code | Nonprogression Subject A (XAi) | βiXAi | Progression Subject B (XBi) | βiXBi |
|---|---|---|---|---|---|---|---|
| Intercept (α) | −5.78 | 1 | −5.78 | 1 | −5.78 | ||
| 1 | Age 70+ years | 0.4116 | 1 | 0.41 | 0 | 0 | |
| 2 | Sex | 0.0688 | 1, M/0, F | 0 | 0 | 0 | 0 |
| 3 | Education | −0.128 | 1, Some college/0, high school or less | 1 | −0.13 | 1 | −0.13 |
| Baseline AMD Grade | |||||||
| 4 | 3 | 2.3944 | 1, Yes/0, No | 0 | 0 | 1 | 2.39 |
| 5 | 4 | 2.9521 | 1, Yes/0, No | 0 | 0 | 0 | 0 |
| 6 | Current Smoking | 1.1261 | 1, Yes/0, No | 0 | 0 | 0 | 0 |
| 7 | Past Smoking | 0.1211 | 1, Yes/0, No | 0 | 0 | 1 | 0.12 |
| 8 | BMI 25–29 | 0.517 | 1, Yes/0, No | 0 | 0 | 0 | 0 |
| 9 | BMI 30+ | 0.4754 | 1, Yes/0, No | 1 | 0.48 | 0 | 0 |
| 10 | Antioxidant | −0.1299 | 1, Yes/0, No | 0 | 0 | 0 | 0 |
| 11 | Zinc | −0.3897 | 1, Yes/0, No | 1 | −0.39 | 0 | 0 |
| 12 | Antioxidant/zinc | −0.4973 | 1, Yes/0, No | 0 | 0 | 0 | 0 |
| 13 | CFH:rs1061170 (Y402H) CT | 0.2644 | 1, Yes/0, No | 0 | 0 | 0 | 0 |
| 14 | CFH:rs1061170 (Y402H) CC | 0.6778 | 1, Yes/0, No | 0 | 0 | 1 | 0.68 |
| 15 | LOC387715:rs10490924 (A69S) GT | 0.8396 | 1, Yes/0, No | 0 | 0 | 0 | 0 |
| 16 | LOC387715:rs10490924 (A69S) TT | 1.3837 | 1, Yes/0, No | 0 | 0 | 0 | 0 |
| 17 | CFH:rs1410996 CT | 0.5251 | 1, Yes/0, No | 1 | 0.53 | 0 | 0 |
| 18 | CFH:rd1410996 CC | 0.8606 | 1, Yes/0, No | 0 | 0 | 1 | 0.86 |
| 19 | C2:rs9332739 (E318D) CG or CC | −1.051 | 1, Yes/0, No | 0 | 0 | 0 | 0 |
| 20 | CFB:rs641153 (R32Q) CT or TT | −0.2147 | 1, Yes/0, No | 0 | 0 | 0 | 0 |
| 21 | C3:rs2230199 (R102G) CG | 0.3679 | 1, Yes/0, No | 1 | 0.37 | 0 | 0 |
| 22 | C3:rs2230199 (R102G) GG | 0.597 | 1, Yes/0, No | 0 | 0 | 0 | 0 |
| Risk score* | −4.52 | −1.85 |
Includes the intercept of −5.7776. The probability of progression = exp(risk score)/[1 + exp(risk score)] For example, for the above two subjects of whom subject A did not progress and subject B progressed, the probability of progression = exp(−4.52)/[1 + exp(−4.52)] = .01 for subject A and exp(−1.85)/[1 + exp(−1.85)] = .14 for subject B. α is the intercept and βi is the regression coefficient for the ith risk factor
The area under the receiver operating characteristic (ROC) curve was obtained. In addition, an age-adjusted concordance (C) statistic based on the ROC curve was calculated for different combinations of these factors, to assess the probability that the risk score based on the group of risk factors in that model from a random progressor was higher than the corresponding risk score from a random nonprogressor within the same 10-year age group.33 We obtained standard errors of estimated C statistics and compared C statistics from alternative risk prediction models, by using correlated ROC curve methods.34
Results
The mean ages (±SD) of cases, controls and progressors, non-progressors were 69.1 (±5.2), 66.8 (±4.2) years and 69.2 (±5.0), 67.7 (±5.0) years, respectively. There were no statistically significant associations between any of the genetic variants and the demographic, behavioral, or treatment variables. Relationships between pairs of genes among the controls were evaluated. CFH Y402H rs1061170 and CFH rs1410996 were significantly related (P < 0.001) as a result of linkage disequilibrium between these sites, and CFB R32Q rs641153 was weakly related to C3 R102G rs2230199 (P = 0.03). No other associations between pairs of genetic variants were statistically significant.
Table 2 displays the unadjusted association between demographic, environmental, and genetic variables and incident advanced AMD as well as the sample sizes within the groups. All factors except gender were related to progression. Baseline macular status was strongly related to progression. Both modifiable factors (smoking and BMI) and genetic variants were also associated with worsening of macular disease over time. The antioxidant/mineral treatment group had a lower rate of progression.
Table 2.
Univariate Association between Baseline Demographic, Environmental, and Genetic Variables and Incident Advanced AMD
| Progressors, n (%) | Nonprogressors, n (%) | Incidence Rates (%) | OR (95% CI) | P* | |
|---|---|---|---|---|---|
| Total patients | 279 | 1167 | |||
| Age (y) | |||||
| <70 | 137 (49) | 743 (64) | 16 | 1.0 | |
| 70+ | 142 (51) | 424 (36) | 25 | 1.8 (1.4–2.4) | <0.001† |
| Sex | |||||
| Female | 163 (58) | 694 (59) | 19 | 1.0 | 0.74 |
| Male | 116 (42) | 473 (41) | 20 | 1.0 (0.8–1.4) | |
| Education | |||||
| ≤High school | 119 (43) | 383 (33) | 24 | 1.0 | 0.002 |
| >High school | 160 (57) | 784 (67) | 17 | 0.7 (0.5–0.9) | |
| Baseline AMD Grades | |||||
| 2 | 8 (3) | 446 (38) | 2 | 1.0 | |
| 3 | 161 (58) | 566 (48) | 22 | 15.9 (7.7–32.6) | <0.001† |
| 4 | 110 (39) | 155 (13) | 42 | 39.6 (18.9–83.0) | |
| Smoking | |||||
| Never | 110 (39) | 557 (48) | 16 | 1.0 | |
| Past | 137 (49) | 564 (48) | 20 | 1.2 (0.9–1.6) | 0.14 |
| Current | 32 (11) | 46 (4) | 41 | 3.5 (2.1–5.8) | <0.001 |
| BMI | |||||
| <25 | 69 (25) | 416 (36) | 14 | 1.0 | |
| 25–29 | 130 (47) | 484 (41) | 21 | 1.6 (1.2–2.2) | 0.003 |
| 30+ | 80 (29) | 267 (23) | 23 | 1.8 (1.3–2.6) | 0.001 |
| Treatment Group | |||||
| Placebo | 74 (27) | 264 (23) | 22 | 1.0 | |
| Antioxidants | 77 (28) | 295 (25) | 21 | 0.9 (0.7–1.3) | 0.70 |
| Zinc | 67 (24) | 294 (25) | 19 | 0.8 (0.6–1.2) | 0.27 |
| Antioxidants and zinc | 61 (22) | 314 (27) | 16 | 0.7 (0.5–1.0) | 0.056 |
| CFH: rs1061170 (Y402H) | |||||
| TT | 39 (14) | 366 (31) | 10 | 1.0 | |
| CT | 116 (42) | 521 (45) | 18 | 2.1 (1.4–3.1) | |
| CC | 124 (44) | 280 (24) | 31 | 4.1 (2.8–6.1) | <0.001† |
| LOC387715:rs10490924 (A69S)(ARMS2) | |||||
| GG | 67 (24) | 612 (52) | 10 | 1.0 | |
| GT | 138 (49) | 446 (38) | 24 | 2.8 (2.1–3.9) | <0.001† |
| TT | 74 (27) | 109 (9) | 40 | 6.2 (4.2–9.1) | |
| CFH:rs1410996 | |||||
| TT | 8 (3) | 158 (14) | 5 | 1.0 | |
| CT | 74 (27) | 472 (40) | 14 | 3.1 (1.5–6.6) | <0.001† |
| CC | 197 (71) | 537 (46) | 27 | 7.2 (3.5–15.0) | |
| C2:rs9332739 (E318D) | |||||
| GG | 271 (97) | 1075 (92) | 20 | 1.0 | |
| CG/CC | 8 (3) | 92 (8) | 6 | 0.3 (0.2–0.7) | 0.005 |
| CFB:rs641153 (R32Q) | |||||
| CC | 256 (92) | 1023 (88) | 20 | 1.0 | |
| CT/TT | 23 (8) | 143 (12) | 14 | 0.6 (0.4–1.0) | 0.06 |
| C3:rs2230199 (R102G) | |||||
| CC | 124 (44) | 652 (56) | 16 | 1.0 | |
| CG | 130 (47) | 456 (39) | 22 | 1.5 (1.1–2.0) | |
| GG | 25 (9) | 59 (5) | 30 | 2.2 (1.3–3.7) | <0.001† |
OR, odds ratio; CI, confidence interval. AMD grade 2 was category 2 in one or both eyes (worst eye), grade 3 was AMD category 3 in one or both eyes (worst eye), grade 4 was AMD category 4 in one eye (the worst eye).
Referent category is that listed first under each variable.
Test for linear trend.
Tables 3 and 4 show the multivariate adjusted models for prevalent and incident advanced AMD, respectively, after adjustment for demographic and behavioral factors and all six genetic variants.
Table 3.
Association between Prevalent Advanced AMD and Demographic, Environmental, and Genetic Risk Factors
| Variable | All Advanced AMD
|
Unilateral Advanced AMD*
|
Bilateral Advanced AMD*
|
Geographic Atrophy†
|
Neovascular AMD†
|
|||||
|---|---|---|---|---|---|---|---|---|---|---|
| OR (95% CI) | P | OR (95% CI) | P | OR (95% CI) | P | OR (95% CI) | P | OR (95% CI) | P | |
| Cases/Controls (n) | 509/222 | 202/222 | 307/222 | 136/222 | 373/222 | |||||
| Age (y) | ||||||||||
| <70 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| ≥70 | 2.8 (1.8–4.2) | <0.0001 | 2.3 (1.4–3.8) | 0.001 | 3.7 (2.2–6.2) | <0.0001 | 2.6 (1.5–4.6) | 0.001 | 3.1 (1.9–4.9) | <0.0001 |
| Sex | ||||||||||
| Female | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| Male | 0.9 (0.6–1.4) | 0.62 | 1.0 (0.6–1.5) | 0.85 | 0.9 (0.5–1.4) | 0.55 | 1.0 (0.6–1.8) | 0.89 | 0.9 (0.5–1.3) | 0.5 |
| Education | ||||||||||
| ≤High school | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| >High school | 0.6 (0.4–0.9) | 0.01 | 0.5 (0.3–0.9) | 0.01 | 0.6 (0.4–1.0) | 0.07 | 0.7 (0.4–1.2) | 0.18 | 0.6 (0.3–0.9) | 0.01 |
| Smoking | ||||||||||
| Never | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| Past | 1.9 (1.2–2.9) | 0.004 | 2.2 (1.3–3.6) | 0.002 | 1.6 (0.9–2.6) | 0.09 | 1.8 (1.0–3.1) | 0.06 | 1.9 (1.2–3.1) | 0.01 |
| Current | 3.9 (1.7–8.9) | 0.001 | 3.7 (1.5–9.6) | 0.01 | 4.0 (1.5–10.7) | 0.01 | 2.7 (0.8–8.9) | 0.11 | 4.4 (1.9–10.4) | 0.001 |
| BMI | ||||||||||
| <25 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| 25–29 | 1.1 (0.7–1.8) | 0.72 | 1.2 (0.7–2.1) | 0.53 | 1.0 (0.6–1.8) | 0.99 | 1.0 (0.5–1.9) | 0.97 | 1.1 (0.7–1.9) | 0.65 |
| 30+ | 1.5 (0.9–2.6) | 0.11 | 1.7 (0.9–3.2) | 0.09 | 1.5 (0.8–2.9) | 0.25 | 1.3 (0.6–2.8) | 0.44 | 1.8 (1.0–3.2) | 0.06 |
| Antioxidant | ||||||||||
| No | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| Yes | 1.3 (0.8–1.9) | 0.25 | 1.3 (0.8–2.1) | 0.29 | 1.2 (0.7–2.0) | 0.42 | 1.1 (0.6–1.9) | 0.77 | 1.4 (0.9–2.2) | 0.14 |
| CFH:rs1061170(Y402H) | ||||||||||
| TT | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| CT | 1.8 (1.1–3.1) | 0.02 | 2.2 (1.2–4.2) | 0.01 | 1.8 (0.9–3.4) | 0.08 | 1.7 (0.8–3.4) | 0.16 | 2.3 (1.3–4.2) | 0.01 |
| CC | 3.5 (1.7–7.1) | 0.001 | 4.2 (1.8–9.5) | 0.0001 | 3.4 (1.5–7.7) | 0.003 | 3.3 (1.4–8.2) | 0.01 | 4.1 (1.9–8.7) | 0.0003 |
| P trend | 0.0003 | 0.001 | 0.002 | 0.01 | 0.0001 | |||||
| LOC387715:rs10490924(A69S)(ARMS2) | ||||||||||
| GG | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | |||||
| GT | 3.1 (2.0–4.7) | <0.0001 | 2.1 (1.3–3.5) | 0.002 | 5.1 (3.0–8.5) | <0.0001 | 2.7 (1.6–4.7) | 0.001 | 3.7 (2.3–5.8) | <0.0001 |
| TT | 25.4 (8.6–75.1) | <0.0001 | 13.4 (3.9–45.6) | <0.0001 | 64.9 (19.6–214.6) | <0.0001 | 21.7 (5.7–82.7) | <0.0001 | 31.2 (10.2–95.2) | <0.0001 |
| P trend | <0.0001 | <0.0001 | <0.0001 | <0.0001 | <0.0001 | |||||
| CFH:rs1410996 | ||||||||||
| TT | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| CT | 1.6 (0.8–3.5) | 0.2 | 1.4 (0.6–3.6) | 0.46 | 1.6 (0.6–4.6) | 0.34 | 2.0 (0.6–6.7) | 0.28 | 1.3 (0.6–3.1) | 0.53 |
| CC | 3.7 (1.6–8.4) | 0.002 | 2.4 (0.9–6.6) | 0.08 | 5.5 (1.9–15.7) | 0.002 | 4.2 (1.2–14.9) | 0.03 | 3.3 (1.3–8.2) | 0.01 |
| P trend | 0.0003 | 0.04 | <0.0001 | 0.01 | 0.001 | |||||
| C2:rs9332739(E318D) | ||||||||||
| GG | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| CG/CC | 0.3 (0.1–0.7) | 0.003 | 0.5 (0.2–1.2) | 0.11 | 0.1 (0.04–0.5) | 0.002 | 0.20 (0.05–0.8) | 0.02 | 0.4 (0.2–0.8) | 0.02 |
| CFB:rs641153(R32Q) | ||||||||||
| CC | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| CT or TT | 0.3 (0.1–0.5) | <0.0001 | 0.3 (0.1–0.5) | 0.0001 | 0.2 (0.1–0.5) | 0.0001 | 0.2 (0.1–0.5) | 0.0004 | 0.3 (0.1–0.5) | <0.0001 |
| C3:rs2230199(R102G) | ||||||||||
| CC | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| CG | 1.6 (1.1–2.5) | 0.02 | 1.5 (0.9–2.5) | 0.08 | 1.9 (1.2–3.2) | 0.01 | 1.9 (1.1–3.3) | 0.03 | 1.7 (1.1–2.7) | 0.03 |
| GG | 3.6 (1.4–9.4) | 0.01 | 5.0 (1.7–14.5) | 0.003 | 3.7 (1.1–12.5) | 0.03 | 4.9 (1.4–18.0) | 0.02 | 4.3 (1.5–12.2) | 0.01 |
| P trend | 0.001 | 0.003 | 0.003 | 0.003 | 0.001 | |||||
OR, odds ratio; CI, confidence interval. OR’s adjusted for age (<70, ≥70), sex, education (≤high school, >high school), smoking (never, past, current), BMI (25, 25–29, 30+), antioxidant treatment (yes, no), and all genetic variants and associated genotypes as listed in table.
Age-Related Eye Disease Study grades 4 (unilateral) and 5 (bilateral) advanced AMD.
Clinical Age-Related Maculopathy Grading System grades 4 (noncentral and central geographic atrophy) and grade 5 (neovascular disease).
Table 4.
Multivariate Association between Demographic, Environmental, and Genetic Risk Factors and Incident Advanced AMD
| Variable | Overall
|
Unilateral Advanced
|
Bilateral Advanced
|
Geographic Atrophy
|
Neovascular AMD
|
|||||
|---|---|---|---|---|---|---|---|---|---|---|
| OR (95% CI)* | P | OR (95% CI) | P | OR (95% CI) | P | OR (95% CI) | P | OR (95% CI) | P | |
| Progressors/nonprogressors (n) | 279/1167 | 123/1012 | 156/1167 | 64/584 | 119/1053 | |||||
| Age (y) | ||||||||||
| <70 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| ≥70 | 1.5 (1.1–2.0) | 0.008 | 1.4 (0.9–2.2) | 0.091 | 1.6 (1.1–2.5) | 0.021 | 1.1 (0.6–1.9) | 0.80 | 1.8 (1.2–2.7) | 0.008 |
| Sex | ||||||||||
| Female | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| Male | 1.1 (0.8–1.5) | 0.68 | 0.9 (0.6–1.5) | 0.81 | 1.3 (0.8–2.0) | 0.29 | 1.3 (0.7–2.4) | 0.35 | 1.1 (0.7–1.7) | 0.63 |
| Education | ||||||||||
| ≤High school | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| >High school | 0.9 (0.6–1.2) | 0.42 | 1.1 (0.7–1.7) | 0.66 | 0.7 (0.5–1.0) | 0.082 | 0.7 (0.4–1.2) | 0.21 | 0.9 (0.6–1.5) | 0.82 |
| Baseline Grade | ||||||||||
| 2 | 1.0 | 1.0 | 1.0 | 1.0 | ||||||
| 3 | 11.0 (5.3–22.8) | <0.001 | 9.2 (4.2–20.2) | <0.001 | 24.4 (3.3–179.7) | 0.002 | † | 8.4 (3.6–19.8) | <0.001 | |
| 4 | 19.1 (8.9–41.2) | <0.001 | 163.9 (22.3–>999.9 | <0.001 | 9.1 (2.4–34.9) | 0.001 | ||||
| Smoking | ||||||||||
| Never | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| Past | 1.1 (0.8–1.6) | 0.47 | 1.2 (0.8–1.8) | 0.48 | 1.1 (0.7–1.7) | 0.75 | 1.0 (0.5–1.7) | 0.90 | 1.3 (0.8–2.0) | 0.30 |
| Current | 3.1 (1.7–5.6) | <0.001 | 2.7 (1.1–6.7) | 0.034 | 3.0 (1.4–6.3) | 0.005 | 1.5 (0.4–6.1) | 0.54 | 3.4 (1.4–8.7) | 0.009 |
| BMI | ||||||||||
| <25 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| 25–29 | 1.7 (1.2–2.4) | 0.006 | 1.7 (1.0–2.7) | 0.042 | 1.7 (1.0–2.9) | 0.038 | 1.8 (0.9–3.6) | 0.09 | 1.6 (1.0–2.7) | 0.059 |
| 30+ | 1.6 (1.1–2.4) | 0.024 | 1.6 (0.9–2.8) | 0.11 | 1.5 (0.9–2.7) | 0.16 | 2.5 (1.2–5.1) | 0.016 | 1.5 (0.8–2.6) | 0.19 |
| Treatment Group | ||||||||||
| Placebo | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| Antioxidants | 0.9 (0.6–1.3) | 0.54 | 1.0 (0.6–1.9) | 0.90 | 0.8 (0.4–1.4) | 0.40 | 0.6 (0.3–1.4) | 0.23 | 1.1 (0.6–2.0) | 0.68 |
| Zinc | 0.7 (0.4–1.0) | 0.075 | 1.3 (0.7–2.3) | 0.40 | 0.4 (0.2–0.7) | 0.001 | 1.2 (0.6–2.5) | 0.63 | 0.8 (0.4–1.5) | 0.49 |
| Antioxidants and zinc | 0.6 (0.4–0.9) | 0.023 | 0.9 (0.5–1.6) | 0.65 | 0.4 (0.2–0.8) | 0.004 | 0.8 (0.4–1.7) | 0.58 | 0.7 (0.4–1.3) | 0.28 |
| CFH:rs1061170(Y402H) | ||||||||||
| TT | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| CT | 1.3 (0.8–2.1) | 0.29 | 1.6 (0.8–3.4) | 0.22 | 1.1 (0.6–2.1) | 0.77 | 1.0 (0.4–2.5) | 0.98 | 1.5 (0.7–3.1) | 0.28 |
| CC | 2.0 (1.1–3.5) | 0.019 | 2.7 (1.2–6.3) | 0.019 | 1.5 (0.7–3.1) | 0.33 | 1.9 (0.7–5.5) | 0.21 | 2.1 (0.9–4.6) | 0.08 |
| P trend | 0.014 | 0.011 | 0.27 | 0.110 | 0.08 | |||||
| LOC387715:rs10490924(A69S)(ARMS2) | ||||||||||
| GG | 1.00 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| GT | 2.3 (1.6–3.3) | <0.001 | 2.8 (1.7–4.4) | <0.001 | 2.0 (1.2–3.3) | 0.007 | 1.9 (1.1–3.5) | 0.03 | 2.7 (1.6–4.4) | <0.001 |
| TT | 4.0 (2.6–6.1) | <0.001 | 3.1 (1.7–5.8) | <0.001 | 4.6 (2.6–8.2) | <0.001 | 1.8 (0.8–4.2) | 0.16 | 5.2 (2.9–9.4) | <0.001 |
| P trend | <0.001 | <0.001 | <0.001 | 0.06 | <0.001 | |||||
| CFH:rs1410996 | ||||||||||
| TT | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| CT | 1.7 (0.7–4.0) | 0.23 | 1.1 (0.4–3.5) | 0.84 | 2.7 (0.7–10.8) | 0.15 | 2.0 (0.4–10.4) | 0.39 | 1.9 (0.5–7.2) | 0.36 |
| CC | 2.4 (1.0–5.8) | 0.061 | 1.9 (0.6–6.2) | 0.29 | 3.3 (0.8–13.6) | 0.098 | 3.5 (0.6–19.4) | 0.15 | 3.0 (0.8–12.0) | 0.12 |
| P trend | 0.029 | 0.083 | 0.16 | 0.09 | 0.037 | |||||
| C2:rs9332739(E318D) | ||||||||||
| GG | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| CG/CC | 0.4 (0.2–0.8) | 0.010 | 0.5 (0.2–1.4) | 0.19 | 0.2 (0.1–0.8) | 0.021 | 0.2 (0.03–1.7) | 0.15 | 0.4 (0.1–1.3) | 0.12 |
| CFB:rs641153(R32Q) | ||||||||||
| CC | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| CT or TT | 0.8 (0.5–1.4) | 0.42 | 0.8 (0.4–1.6) | 0.47 | 1.0 (0.5–1.9) | 0.92 | 0.8 (0.3–2.1) | 0.60 | 0.9 (0.5–1.9) | 0.87 |
| C3:rs2230199(R102G) | ||||||||||
| CC | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |||||
| CG | 1.4 (1.1–2.0) | 0.022 | 1.3 (0.8–2.0) | 0.26 | 1.6 (1.0–2.4) | 0.044 | 1.2 (0.7–2.1) | 0.59 | 1.7 (1.1–2.7) | 0.015 |
| GG | 1.8 (1.0–3.2) | 0.044 | 2.1 (1.0–4.5) | 0.052 | 1.6 (0.7–3.6) | 0.24 | 1.9 (0.7–5.3) | 0.22 | 2.5 (1.2–5.4) | 0.015 |
| P trend | 0.006 | 0.050 | 0.049 | 0.31 | 0.002 | |||||
ORs adjusted for age (<70, ≥70), sex, education (≤high school, >high school), smoking (never, past, current), baseline AMD grade, BMI (<25, 25–29, 30+), and treatment groups (placebo, antioxidants, zinc, and antioxidants plus zinc), and all six genetic variants and associated genotypes as listed in the table.
Analysis restricted to initial grade of 3.
As seen in Table 3, there were positive associations between both CFH variants and the combined advanced AMD case group compared with the controls (Y402H: OR = 3.5, 95% CI = 1.7–7.1, Ptrend = 0.0003; rs1410996: OR = 3.7, Ptrend = 0.0003). In addition, there were positive associations between AMD and the LOC388715 A69S variant (OR = 25.4, Ptrend < 0.0001) and C3 (OR = 3.6, Ptrend = 0.001) and between both the C2 variant (OR = 0.3, P = 0.003) and the CFB variant (OR = 0.3, P < 0.0001). There were positive independent associations with older age (OR = 2.8, P < 0.0001), current smoking (OR = 3.9, P = 0.001), and past smoking (OR = 1.9, P = 0.004). A protective effect of higher education (OR = 0.6, P = 0.01) was shown. A borderline positive association with BMI was present (OR = 1.5, P = 0.11), and no significant association with sex or antioxidant treatment was seen. In general, similar associations between genes and AMD were seen for all subtypes of AMD, including unilateral and bilateral advanced AMD and dry and wet types of advanced AMD, although associations varied slightly for specific types of advanced AMD.
Table 4 displays the multivariate adjusted ORs for incident advanced AMD and shows that, after adjustment for genotypes, older age, smoking, and higher BMI were related to a higher rate of progression. Baseline grade was a strong predictor of incident advanced AMD, and antioxidant–mineral treatment was protective. The two CFH variants each independently increased risk of progression about two- to threefold, with similar increased risk for C3, comparing the homozygous risk and nonrisk genotypes. Variants in the two complement genes C2 and CFB reduced risk, although the association with CFB was not significant for progression to incident advanced AMD. LOC387715 (ARMS2) increased risk and was more strongly associated with progression to neovascular disease (OR = 5.2, 95% CI = 2.9 –9.4, Ptrend < 0.001) than to geographic atrophy (OR = 1.8, 95% CI = 0.8 – 4.2).
An assessment of interactions between genotypes and treatment groups on risk of progression to incident advanced AMD for all genetic variants is shown in Table 5. Only the CFHY402H variant showed a possible interaction with antioxidant–mineral treatment, with P = 0.057 for the analysis including three genotype groups defined categorically and P = 0.016 for analysis of number of alleles as a continuous variable. This result suggests that the antioxidant–mineral supplement is more effective for the homozygous low-risk or heterozygous CFHY402H genotype and less effective for the homozygous risk genotype, with adjustment for the other five genotypes and other nongenetic factors.
Table 5.
Assessment of Genotype-Treatment Interaction Effects on Risk of Incident Advanced AMD
| Treatment Groups
|
P-interaction (6 df)‡ | P-interaction (3 df)§ | ||||
|---|---|---|---|---|---|---|
| Placebo OR (95% CI)* | Antioxidant OR (95% CI) | Zinc OR (95% CI) | Antioxidant-Zinc OR (95% CI) | |||
| CFH rs:1061170(Y402H) | ||||||
| TT | 1.0 | 0.8 (0.3–2.1) | 0.2 (0.05–0.5) | 0.2 (0.07–0.6) | 0.057 | 0.016 |
| CT | 0.7 (0.3–1.5) | 0.7 (0.3–1.5) | 0.6 (0.3–1.3) | 0.4 (0.2–1.0) | ||
| CC | 0.9 (0.4–2.2) | 0.8 (0.3–1.9) | 0.9 (0.4–2.0) | 0.9 (0.4–2.1) | ||
| LOC387715 rs:10490924(A69S) (ARMS2) | ||||||
| GG | 1.0 | 5.8 (2.5–13.5) | 4.1 (1.7–9.8) | 2.0 (0.8–4.8) | 0.37 | 0.18 |
| GT | 3.5 (1.7–7.1) | 2.5 (1.2–4.9) | 1.9 (1.0–3.8) | 1.7 (0.8–3.3) | ||
| TT | 5.0 (2.1–11.8) | 1.0 (0.5–2.2) | 0.8 (0.4–1.8) | 1.1 (0.5–2.4) | ||
| CFH:rs1410996 | ||||||
| TT | 1.0 | 0.4 (0.06–2.6) | † | 0.4 (0.06–2.3) | 0.88 | 0.28 |
| CT | 0.9 (0.3–3.3) | 0.9 (0.2–3.2) | 0.5 (0.1–1.8) | 0.5 (0.1–1.9) | ||
| CC | 1.1 (0.3–4.0) | 1.0 (0.3–3.7) | 1.0 (0.3–3.4) | 0.7 (0.2–2.7) | ||
| C2:rs9332739(E318D) | ||||||
| GG | 1.0 | 0.9 (0.6–1.3) | 0.7 (0.4–1.0) | 0.6 (0.4–0.9) | 0.68 | |
| CG/CC | 0.2 (0.02–1.3) | 0.2 (0.05–1.2) | 0.2 (0.05–1.2) | 0.4 (0.1–1.5) | ||
| CFB:rs641153(R32Q) | ||||||
| CC | 1.0 | 0.7 (0.2–1.9) | 0.6 (0.2–1.9) | 0.6 (0.3–1.5) | ||
| CT/TT | 0.3 (0.09–1.2) | 0.8 (0.5–1.3) | 0.6 (0.4–1.0) | 0.5 (0.3–0.9) | 0.45 | |
| C3:rs2230199(R102G) | ||||||
| CC | 1.0 | 1.0 (0.5–1.8) | 0.7 (0.4–1.3) | 0.7 (0.4–1.4) | 0.56 | 0.54 |
| CG | 1.7 (0.9–3.3) | 1.2 (0.6–2.2) | 1.1 (0.6–2.0) | 1.0 (0.5–1.8) | ||
| GG | 1.5 (0.5–4.8) | 2.7 (1.0–7.9) | 2.0 (0.6–6.5) | 0.4 (0.07–1.9) | ||
ORs adjusted for age (<70, ≥70), sex, education (≤high school, >high school), smoking (never, past, current), baseline AMD grade, BMI (<25, 25–29, 30+), and treatment groups (placebo, antioxidants, zinc, and antioxidants plus zinc), and all genotypes.
No cases among the zinc-TT genotype group.
Interaction of treatment group by genotype group.
Interaction of treatment group by number of risk/protective alleles.
Interactions between each genotype versus smoking (ever/ never) and BMI (25+/<25) for prevalent AMD in the case–control comparison were evaluated, but no significant interactions were found between any of the genotypes and smoking or BMI (data not shown). However, there was a weak nonsignificant trend for a smaller effect of BMI among those with genotype CFH Y402H TT and an adverse effect of BMI for those with a risk allele (the CC and CT genotypes). Furthermore, within a given genotype, smoking and higher BMI increased risk of advanced AMD. For example, for the homozygous GG risk genotype for C3, the OR for advanced AMD was 3.3 (1.0 –10.9) for never smokers and increased threefold to 9.8 (2.0 – 47.5) for individuals who had ever smoked, compared with the CC genotype among nonsmokers, indicating that there are main effects of both smoking and C3 genotype, and the joint effects are consistent with a multiplicative model.
In Table 6, we present C statistics for models predicting progression to incident advanced AMD with different combinations of genetic, ocular, demographic, behavioral, and treatment variables.
Table 6.
C Statistics for Incident Advanced AMD Based on Models with Different Combinations of Genetic, Environmental, and Treatment Variables
| Model | Genetic Variables | Demographic, Environmental, Treatment Variables | C Statistic (±SE)* |
|---|---|---|---|
| 1 | Age, gender, education, baseline grade | 0.732 ± 0.017 | |
| 2 | Age, gender, education, baseline grade, smoking, BMI | 0.757 ± 0.016 | |
| 3 | CFH Y402H, LOC387715 A69S(ARMS2), CFH 1410996, C2E318D, CFB R32Q, C3 R102G | Age, gender, education, baseline grade, smoking, BMI | 0.821 ± 0.014 |
| 4 | CFH Y402H, LOC387715 A69S(ARMS2), CFH 1410996, C2E318D, CFB R32Q, C3 R102G | Age, gender, education, baseline grade, smoking, BMI, treatment assignment | 0.822 ± 0.014 |
| 5 | LOC387715 A69S(ARMS2), CFH 1410996, C2E318D, CFB R32Q, C3 R102G, interaction between CFHY402H and treatment assignment | Age, gender, education, baseline grade, smoking, BMI | 0.831 ± 0.013 |
Model 2 vs. 1, P = 0.028; 3 vs. 2, P < 0.001; 4 vs. 3, P = 0.52; 5 vs. 4, P = 0.018; 5 vs. 3, P = 0.019.
There was a significant improvement in the C statistic when the behavioral factors smoking and BMI were added as additional risk variables in model 2 with a C statistic of 0.757 ± 0.016 (model 1 vs. 2, P = 0.028). In model 3, we added all six variants in this report, together with baseline AMD grade, age, sex, education, smoking, and BMI and found a C statistic of 0.821 ± 0.014 which was a significant improvement over the model without genotypes (model 2 vs. 3, P < 0.001). When treatment was added to model 3, the C statistic was unchanged with C statistic of 0.822 ± 0.014. When the treatment–CFH Y402H interaction term was added to the full model, the C statistic significantly improved to 0.831 (±0.013), but the change in the C statistic was small. Of note, these C statistics for prediction of AMD progression are similar to the Framingham risk score prediction model results for coronary heart disease (CHD).35
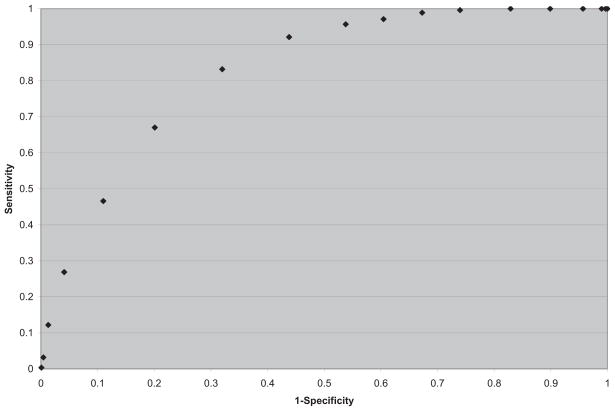
We then considered the possibility of using model 4 as shown in Table 6, for purposes of individual risk prediction. We calculated the sensitivity and specificity of model 4 in Table 6 using different cutoffs to denote potential screen-positive criteria. The ROC curve is presented in Figure 1.
Figure 1.
Sensitivities and specificities for a variety of risk score cutoffs and ROC curve for prediction of incident advanced age-related macular degeneration.
Our goal was to identify a cutoff, if possible, where both the sensitivity and specificity would be at least 70%. This cutoff was approximately identified for risk score ≥ −1.5 as screen positive, < −1.5 as screen negative, which yielded a sensitivity of 83% (232/279) and specificity of 68% (794/1167). Some examples of using this risk prediction algorithm on a random progressor and nonprogressor are given in Table 1.
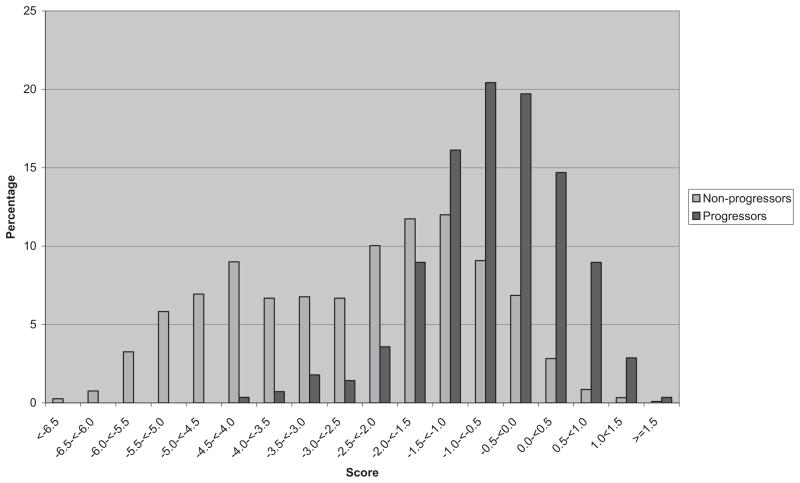
Plotted histograms of scores for progressors and nonprogressors are shown in Figure 2, indicating a good separation between these two groups. Risk score distributions were substantially different with progressor scores tending to be higher than nonprogressors, although there was some overlap.
Figure 2.
Incident advanced age-related macular degeneration. Risk scores for progressors and nonprogressors are based on all genetic variants as well as demographic, ocular, environmental, and treatment variables.
Discussion
This article, for the first time to our knowledge, shows the independent association of six genetic variants with both prevalence and incidence of advanced AMD, with adjustment for all the genetic variants in addition to ocular, demographic, behavioral, and treatment factors. Discrimination between progressors and nonprogressors was excellent for the overall risk score. The predictive power of this composite of risk factors for progression to advanced AMD, with a C statistic score of 0.83, is comparable to the Framingham risk functions for CHD in which the C statistics were 0.79 for white men and 0.83 for white women in the Framingham study cohort and somewhat lower in several replication samples.35 Clearly, genetic factors play a major role in this disease, as demonstrated by the large and consistent estimates of the effects of the genetic variants on various groups of advanced AMD, with adjustment for other known risk factors. However, modifiable factors also have an impact. Cigarette smoking increased risk for all genotypes. For example, risk of advanced AMD increased from over 3-fold for nonsmokers to almost 10-fold for smokers among individuals with the same homozygous C3 risk genotype compared with nonsmokers with the nonrisk genotype. Higher BMI also contributed to the risk profile for all genotypes.
We have previously presented the combined risk of AMD as a function of five of these genetic variants alone ranging from a less than 1% to a more than 50% lifetime risk.7 This report expands and refines those observations in important and meaningful ways by adding a new genetic variant; incorporating ocular, demographic, behavioral. and treatment factors; calculating C statistics for advanced AMD based on models with different combinations of genetic and other variables; and evaluating the ability of the resultant risk scores to discriminate between individuals who progress and those who do not.
It is well known that the presence of intermediate AMD is related to progression to advanced AMD,20 and results in our report underscore the importance of baseline pathologic features in the macula, including drusen and pigment irregularities. However, we have shown that two genetic variants also predict progression to advanced disease independent of the fundus appearance.12 This current report expands on those results by adding four additional known genetic variants and adds new analyses. We show that a combined model with six genetic factors significantly contributes to the prediction of AMD progression, above and beyond knowledge of the ocular phenotype, as shown by the higher C statistic score when the genetic variables are added (P < 0.001). In other words, the prediction of advanced AMD progressively improves beyond knowledge of only the demographic and ocular factors when the behavioral factors smoking and BMI are added to the model, and improves even more when genetic factors are known.
The relationship between treatment effect and two of these genotypes was evaluated previously.36 In the current manuscript, we have expanded our set of genetic risk factors to include four additional SNPs that are associated with AMD. We also considered interaction effects between genetic and environmental factors. We did find a significant interaction between the number of risk alleles for the CFH Y402H variant and treatment, whereby patients with the CC (i.e., high risk) genotype are less likely to benefit from the antioxidant–mineral supplementation than are subjects with the TT and CT genotypes. However, inclusion of this interaction term increased the C statistic by less than 1% and had a small effect (approximately 1%) on the sensitivity and specificity, when a cutoff for test positive of ≥ −1.5 was used. Therefore, for the sake of simplicity, we elected not to include this interaction term in our risk prediction algorithm. It remains to be seen whether this interaction effect is reproducible in other study populations evaluated for progression of AMD.
Unique features of this study include the evaluation of the predictive power of several genetic and other variables based on a large, well-characterized population of Caucasian patients from various geographic regions around the United States. Further strengths include the standardized collection of risk factor information, direct measurements of height and weight, and classification of maculopathy by standardized ophthalmic examinations and grading of fundus photographs. Misclassification was unlikely, because grades were assigned without knowledge of risk factors or genotype. We controlled for known AMD risk factors, including age, education, BMI, smoking, baseline AMD grade, and treatment assignment in assessing the relationship between genetic variants and advanced AMD. Both the environmental and genetic risk factors were independently associated with AMD, when considered simultaneously. There may be some other unmeasured and therefore uncontrolled factors that confound these relationships, but, to explain these results, they would have to be highly related to genotype, smoking, BMI, and treatment assignment and a strong risk factor for AMD. Although this is a selected population, the subjects probably represent the typical patient with advanced AMD, and the overall population is similar to other clinic populations in this age range in terms of smoking and prevalence of obesity as well as the distribution of the genotypes. This large and well-characterized population provided a unique opportunity to evaluate gene– environment associations and interactions. Furthermore, the biologic effects of the genetic variants do not appear to differ in major ways among various Caucasian populations with AMD.
These analyses and results indicate the potential for individual prediction of risk for AMD. For example, in calculating the risk score, one could estimate “points” from the regression coefficients (Table 1) for current smoking (1.1), BMI 30+ (0.48), antioxidants and zinc (−0.50) and the various genetic variants (ranging from −1.0 to +1.4), to obtain an overall risk for an individual to progress to advanced AMD. This process could be refined as new genetic and environmental predictors are established. Targeting high-risk individuals could lead to heightened awareness and more frequent surveillance and clinical examinations, as well as identification of high-risk individuals for inclusion in clinical trials of new therapies. Other advantages of knowing such a risk score include the possibility of more targeted education and counseling about known modifiable factors. Screening would identify high-risk individuals who would be encouraged to follow a healthy lifestyle by not smoking, eating vegetables and fish, maintaining a normal weight and getting exercise, and taking antioxidant–mineral supplements. All of these factors are known to influence the inflammatory and immune pathways that are involved in the pathogenesis of AMD.37,38
Acknowledgments
Supported by Grant R01-EY11309 from the National Institutes of Health (NIH) National Eye Institute; the Foundation Fighting Blindness, Owing Mills, MD; the Age-Related Macular Degeneration Research Fund, Ophthalmic Epidemiology and Genetics Service, Tufts Medical Center, Tufts University School of Medicine; the Massachusetts Lions Eye Research Fund Inc., Northboro, MA; Grant U54 RR020278 from the National Center for Research Resources to the Broad Institute Center for Genotyping and Analysis; and unrestricted grants from Research to Prevent Blindness Inc., New York, NY.
The authors thank the EMMES Corporation for their work on the AREDS Genetic Repository, and the AREDS research group; Jay S. Duker, MD, Chairman, Department of Ophthalmology, Tufts Medical Center, Tufts University School of Medicine, for his support; Daniel Mirel, PhD, for assistance with genotyping; and Marion McPhee, BEd (Harvard University Channing Laboratory, Boston, MA), for programming assistance.
Footnotes
Disclosure: J.M. Seddon, P; R. Reynolds, None; J. Maller, None; J.A. Fagerness, None; M.J. Daly, None; B. Rosner, None
References
- 1.Edwards AO, Ritter R, Abel KJ, Manning A, Panhuysen C, Farrer LA. Complement factor H polymorphism and age-related macular degeneration. Science. 2005;308:421–424. doi: 10.1126/science.1110189. [DOI] [PubMed] [Google Scholar]
- 2.Klein RJ, Zeiss C, Chew E, et al. Complement factor H polymorphism in age-related macular degeneration. Science. 2005;308:385–389. doi: 10.1126/science.1109557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Haines JL, Hauser MA, Schmidt S, et al. Complement factor H variant increases the risk of age-related macular degeneration. Science. 2005;308:419–421. doi: 10.1126/science.1110359. [DOI] [PubMed] [Google Scholar]
- 4.Hageman GS, Anderson DH, Johnson LV, et al. A common haplo-type in the complement regulatory gene factor H (HF1/CFH) predisposes individuals to age-related macular degeneration. Proc Natl Acad Sci USA. 2005;102:7227–7232. doi: 10.1073/pnas.0501536102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gold B, Merriam JE, Zernant J, et al. Variation in factor B (BF) and complement component 2 (C2) genes is associated with age-related macular degeneration. Nat Genet. 2006;38:458–462. doi: 10.1038/ng1750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Jakobsdottir J, Conley Y, Weeks DE, et al. Susceptibility genes for age-related maculopathy on chromosome 10q26. Am J Hum Gen. 2005;77(3):389–407. doi: 10.1086/444437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Maller J, George S, Purcell S, et al. Common variation in three genes, including a noncoding variant in CFH, strongly influences risk of age-related macular degeneration. Nat Genet. 2006;38:1055–1059. doi: 10.1038/ng1873. [DOI] [PubMed] [Google Scholar]
- 8.Li M, Atmaca-Sonmez P, Othman M, et al. CFH haplotypes without the Y402H coding variant show strong association with susceptibility to age-related macular degeneration. Nat Genet. 2006;38:1049–1054. doi: 10.1038/ng1871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dewan A, Liu M, Hartman S, et al. HTRA1 promoter polymorphism in wet age-related macular degeneration. Science. 2006;314:989–992. doi: 10.1126/science.1133807. [DOI] [PubMed] [Google Scholar]
- 10.Yang Z, Camp NJ, Sun H, et al. A variant of the HTRA1 gene increases susceptibility to age-related macular degeneration. Science. 2006;314:992–993. doi: 10.1126/science.1133811. [DOI] [PubMed] [Google Scholar]
- 11.Kanda A, Chen W, Othman M, et al. A variant of mitochondrial protein LOC387715/ARMS2, not HTRA1, is strongly associated with age-related macular degeneration. Proc Natl Acad Sci USA. 2007;104:16227–16232. doi: 10.1073/pnas.0703933104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Seddon JM, Francis PJ, George S, Schultz DW, Rosner B, Klein ML. Association of CFH Y402H and LOC387715 A69S with progression of age-related macular degeneration. JAMA. 2007;297:1793–1800. doi: 10.1001/jama.297.16.1793. [DOI] [PubMed] [Google Scholar]
- 13.Maller JB, Fagerness JA, Reynolds RC, Neale BM, Daly MJ, Seddon JM. Variation in complement factor 3 is associated with risk of age-related macular degeneration. Nat Gen. 2007;39:1200–1201. doi: 10.1038/ng2131. [DOI] [PubMed] [Google Scholar]
- 14.Yates JRW, Sepp T, Matharu BK, et al. Complement C3 variant and the risk of age-related macular degeneration. NEJM. 2007;357:553–561. doi: 10.1056/NEJMoa072618. [DOI] [PubMed] [Google Scholar]
- 15.Haddad S, Chen CA, Santangelo SL, Seddon JM. The genetics of age-related macular degeneration: a review of progress to date. Surv Ophthalmol. 2006;50:306–363. doi: 10.1016/j.survophthal.2006.05.001. [DOI] [PubMed] [Google Scholar]
- 16.Seddon JM, Willett WC, Speizer FE, Hankinson SE. A prospective study of cigarette smoking and age-related macular degeneration in women. JAMA. 1996;276:1141–1146. [PubMed] [Google Scholar]
- 17.Tomany SC, Wang JJ, Van Leeuwen R, et al. Risk factors for incident age-related macular degeneration: pooled findings from 3 continents. Ophthalmology. 2004;111:1280–1287. doi: 10.1016/j.ophtha.2003.11.010. [DOI] [PubMed] [Google Scholar]
- 18.Seddon JM, George S, Rosner B, Klein ML. CFH gene variant, Y402H, and smoking, body mass index, environmental associations with advanced age-related macular degeneration. Hum Hered. 2006;61:157–165. doi: 10.1159/000094141. [DOI] [PubMed] [Google Scholar]
- 19.Francis PJ, George S, Schultz DW, et al. The LOC387715 gene, smoking, body mass index, environmental associations with advanced age-related macular degeneration. Hum Hered. 2007;63:212–218. doi: 10.1159/000100046. [DOI] [PubMed] [Google Scholar]
- 20.Age-Related Eye Disease Study Research Group. A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and beta carotene, and zinc for age-related macular degeneration and vision loss. AREDS Report No. 8. Arch Ophthalmol. 2001;119:1417–1436. doi: 10.1001/archopht.119.10.1417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Seddon JM, Ajani UA, Sperduto RD, et al. Dietary carotenoids, vitamins A, C, and E and advanced age-related macular degeneration: a multicenter study. JAMA. 1994;272:1413–1420. [PubMed] [Google Scholar]
- 22.Age-Related Eye Disease Study Research Group. The relationship of dietary carotenoid and vitamin A, E, and C intake with age-related macular degeneration in a case-control study. AREDS Report No. 22. Arch Ophthalmol. 2007;125:1225–1232. doi: 10.1001/archopht.125.9.1225. [DOI] [PubMed] [Google Scholar]
- 23.Mares Perlman JA, Fisher AI, Klein R, et al. Lutein and zeaxanthin in the diet and serum and their relation to age-related maculopathy in the third national health and nutrition examination survey. Am J Epidemiol. 2001;153:424–432. doi: 10.1093/aje/153.5.424. [DOI] [PubMed] [Google Scholar]
- 24.Seddon JM, Cote J, Rosner B. Progression of age-related macular degeneration: association with dietary fat, transunsaturated fat, nuts, and fish intake. Arch Ophthalmol. 2003;121:1728–1737. doi: 10.1001/archopht.121.12.1728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Seddon JM, George S, Rosner B. Cigarette smoking, fish consumption, omega-3 fatty acid intake, and associations with age-related macular degeneration: the US Twin Study of Age-Related Macular Degeneration. Arch Ophthalmol. 2006;124:995–1001. doi: 10.1001/archopht.124.7.995. [DOI] [PubMed] [Google Scholar]
- 26.Schaumberg DA, Hankinson SE, Guo Q, Rimm E, Hunter DJ. A prospective study of 2 major age-related macular degeneration susceptibility alleles and interactions with modifiable factors. Arch Ophthalmol. 2007;125:55–62. doi: 10.1001/archopht.125.1.55. [DOI] [PubMed] [Google Scholar]
- 27.Age-Related Eye Disease Study Group. The relationship of dietary lipid intake and age-related macular degeneration in a case-control study. AREDS Report No. 20. Arch Ophthalmol. 2007;125:671–679. doi: 10.1001/archopht.125.5.671. [DOI] [PubMed] [Google Scholar]
- 28.Seddon JM, Cote J, Davis N, Rosner B. Progression of age-related macular degeneration: Association with body mass index, waist circumference and waist-hip ratio. Arch Ophthalmol. 2003;121:785–792. doi: 10.1001/archopht.121.6.785. [DOI] [PubMed] [Google Scholar]
- 29.Age-Related Eye Disease Study Group. Risk factors for the incidence of advanced age-related macular degeneration in the AREDS. AREDS Report No. 19. Ophthalmol. 2005;112:533–539. doi: 10.1016/j.ophtha.2004.10.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Seddon J, Sobrin L. Epidemiology of age-related macular degeneration AMD. In: Albert DM, Miller J, editors. The Principles and Practice of Ophthalmology. 3. Philadelphia: WB Saunders; 2007. [Google Scholar]
- 31.Seddon JM, Sharma S, Adelman RA. Evaluation of the clinical age-related maculopathy staging system. Ophthalmology. 2006;113:260–266. doi: 10.1016/j.ophtha.2005.11.001. [DOI] [PubMed] [Google Scholar]
- 32.Rosner B. Fundamentals of Biostatistics. 6. Boston: Duxbury Press; 2005. [Google Scholar]
- 33.Hanley JA, McNeil BJ. The meaning and use of the area under the receiver operating characteristic (ROC) curve. Radiology. 1982;143:29–36. doi: 10.1148/radiology.143.1.7063747. [DOI] [PubMed] [Google Scholar]
- 34.Rosner B, Glynn RJ. Power and sample size estimation for the Wilcoxon rank sum test with application to comparisons of C statistics from alternative prediction models. Biometrics. 2009;65:188–197. doi: 10.1111/j.1541-0420.2008.01062.x. [DOI] [PubMed] [Google Scholar]
- 35.D’Agostino RB, Grundy S, Sullivan LM, Wilson P for the CHD Risk Prediction Group. Validation of the Framingham coronary heart disease prediction scores: results of a multiple ethnic groups investigation. JAMA. 2001;286:180–187. doi: 10.1001/jama.286.2.180. [DOI] [PubMed] [Google Scholar]
- 36.Klein ML, Francis PJ, Rosner B, et al. CFH and LOC387715/ARMS2 genotypes and treatment with antioxidants and zinc for age-related macular degeneration. Ophthalmology. 2008;115:1019–1025. doi: 10.1016/j.ophtha.2008.01.036. [DOI] [PubMed] [Google Scholar]
- 37.Seddon JM, Gensler G, Klein ML, Milton RC. C-reactive protein and homocysteine are associated with dietary and behavioral risk factors for age-related macular degeneration. Nutrition. 2006;22:441–443. doi: 10.1016/j.nut.2005.12.004. [DOI] [PubMed] [Google Scholar]
- 38.Nussenblatt RB, Ferris F. Age-related macular degeneration and the immune response: implications for therapy. Am J Ophthalmol. 2007;144:618–626. doi: 10.1016/j.ajo.2007.06.025. [DOI] [PMC free article] [PubMed] [Google Scholar]