Abstract
Fogo selvagem (FS), the endemic form of pemphigus foliaceus, is a cutaneous autoimmune disease characterized by subcorneal blistering of the epidermis and the production of autoantibodies against the desmosomal antigen desmoglein-1 (Dsg1). Previously, we showed that mice injected with autoantibodies from FS patients develop a skin disease that reproduces the clinical, histological, and immunological features of FS, indicating that autoantibodies play an essential role in the development of this disease. The purpose of this study was to characterize the autoimmune T-cell response associated with FS. We provide here the first evidence, to our knowledge, that the great majority of FS patients have circulating T lymphocytes that specifically proliferate in response to the extracellular domain of Dsg1. Long-term T cells developed from these patients also responded to Dsg1, and this antigen-specific response was shown to be restricted to HLA-DR molecules. These Dsg1-reactive FS T cells exhibited a CD4-positive memory T-cell phenotype and produced a T helper 2–like cytokine profile. These findings represent the initial steps in defining the role of T cells in FS autoimmunity.
Introduction
Fogo selvagem (FS) is the endemic form of pemphigus foliaceus that has been reported in certain regions of Brazil since the beginning of this century (1). An endemic form of pemphigus foliaceus has also been described in certain parts of Colombia (2, 3) and Tunisia (4, 5). The clinical, histological, and immunological features of FS are indistinguishable from those of the nonendemic form of pemphigus foliaceus, which occurs sporadically in other parts of the world (6, 7). The distinctive features of FS, however, are its unique epidemiology, the high frequency of cases among young adults and children, and the common occurrence of familial cases (8). FS is strongly associated with certain HLA-DRB1 alleles, such as DRB1*0102, *0404, *1402, and *1406. Individuals carrying these alleles have a relative risk as high as 14. Therefore, it has been hypothesized that these HLA-DRB1 alleles confer susceptibility to the development of FS (9, 10).
Although the epidemiological data strongly suggest an environmental etiology for FS, the agent(s) precipitating the disease remain unknown. Recently, we characterized a human settlement with a high prevalence and incidence of FS (11). This new FS focus area is located on the Limao Verde Reservation in the State of Mato Grosso do Sul, Brazil, and is populated primarily by native Amerindians belonging to the Terena tribe. The incidence of FS on this reservation is 3%. A recent study by our group (12) revealed a correlation between the geographic distribution of the sites where patients live and the distributions of certain species of simuliids (black flies). Work is in progress to define the possible etiologic role of blood-feeding insects in FS in this unique human settlement.
FS is an autoimmune disease that specifically targets the epidermis, sparing mucosal and other epithelial tissues. The typical skin lesions are superficial vesicles that rupture and leave large areas of denuded skin (13). These intraepidermal vesicles are formed as a result of a process of epidermal cell detachment known as acantholysis (14). The majority of these patients possess anti–desmoglein-1 (Dsg1) IgG autoantibodies that are predominantly of the IgG4 subclass (15). Passive transfer of FS anti-Dsg1 IgG into neonatal mice reproduces the key features of the human disease in the epidermis of these experimental animals (16, 17). Dsg1 is a desmosomal glycoprotein that belongs to the cadherin family of cell adhesion molecules (18, 19). The epitope(s) recognized by pathogenic FS autoantibodies are Ca2+ dependent and are localized to the ectodomain of Dsg1 (20–23). The precise nature of these epitopes and the molecular mechanisms of acantholysis remain unknown.
The diagnostic and pathologic relevance of anti-Dsg1 autoantibodies in FS has been well established (24). However, very little is known about the role that T cells play in the onset and progression of the disease. In general, antibody production by B cells requires collaboration of T helper cells in the T cell–dependent antibody responses (25–28). Because FS is an autoimmune disease mediated by autoantibodies, it is postulated that T lymphocytes also participate in the pathogenesis of this disease in the stage leading to the production of pathogenic autoantibodies. T cells have been implicated in other autoimmune diseases, such as multiple sclerosis (29), myasthenia gravis (30), Graves’ disease (31), herpes gestationis (32), and pemphigus vulgaris (33). Disease-specific T-cell lines and clones have been isolated from these patients. These T cells recognize self antigens and may play an important role in the initiation and the progression of the respective autoimmune diseases.
The purpose of this study was to identify and characterize the immune response of T lymphocytes from FS patients living in the Limao Verde Reservation (12). We have demonstrated for the first time that T cells from the majority of these FS patients proliferate when stimulated with recombinant Dsg1 (rDsg1). T-cell lines and clones derived from 5 patients were shown to express a CD4-positive memory T-cell phenotype and a T helper 2–like (Th2-like) cytokine profile. Moreover, the response of these T cells to Dsg1 was HLA-DR restricted. This information will be useful in dissecting the cellular events leading to production of pathogenic anti-Dsg1 autoantibodies in FS patients.
Methods
FS patients and controls.
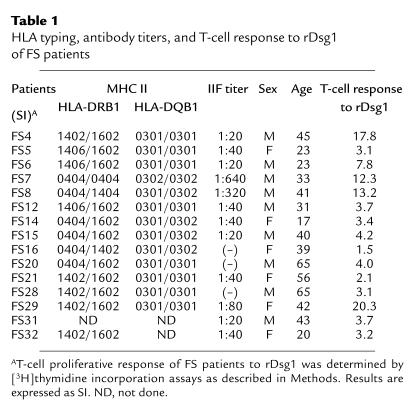
Peripheral blood was obtained from 15 well-characterized FS patients (9 males and 6 females). These patients were followed at the Terena Indian Limao Verde Reservation and have been described in a previous publication (12). The heparinized blood samples were transported in less than 24 hours to our laboratories at the Medical College of Wisconsin following guidelines posted by the Centers for Disease Control, Atlanta, Georgia. The FS patients included in this study showed positive histological and immunofluorescent findings of FS, and blood from all patients demonstrated Dsg1 recognition by immunoprecipitation (23). Further information about these patients, such as age, sex, and indirect IF titers, is shown in Table 1. The indirect IF titers of the patients tested ranged from less than 1:20 to 1:640; this may represent various disease stages of FS (Table 1). Patients with other cutaneous autoimmune diseases, such as bullous pemphigoid (n = 4), systemic lupus erythematosus (n = 1), and psoriasis (n = 2) were included along with normal volunteers from the USA (n = 7) and normal individuals (n = 8) from the Limao Verde Reservation as controls.
Table 1.
HLA typing, antibody titers, and T-cell response to rDsg1 of FS patients
Preparation of recombinant Dsg1, Dsg3, and control BP180 fusion proteins.
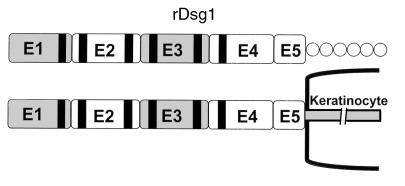
To evaluate the response of T cells and autoantibodies of FS patients to Dsg1, the entire extracellular domain of human Dsg1 was expressed in a baculovirus expression system as described (34) (Figure 1). Briefly, the appropriate Dsg1 cDNA segments were isolated by PCR amplification from a human keratinocyte cDNA library. The construct contains the 5′ end of the Dsg1 coding region, including the flanking Kozak sequence. To facilitate purification of rDsg1 by nickel column chromatography, a sequence of 6 histidine codons was engineered immediately downstream of the Dsg1 sequence. The insert was subcloned into the pVL1393 vector (Invitrogen Corp., Carlsbad, California, USA), and recombinant baculoviruses were generated by homologous recombination using standard protocol provided by the manufacturer. Subsequently, the rDsg1 was purified by nickel column chromatography according to the procedure provided by the manufacturer (QIAGEN Inc., Chatsworth, California, USA). A similar procedure was used to construct and produce rDsg3 (23).
Figure 1.
The structure of desmoglein-1. Dsg1 contains 5 major cadherin-like domains on its extracellular portion. The black strips are the Ca2+ binding sites. The recombinant Dsg1 used in this study comprises the entire ectodomain of the molecule and is represented by the horizontal bar. There is a 6-histidine tail immediately downstream of the rDsg1, represented by circles.
The BP180 fusion protein that covers the NC16A region was used in this study (32). To generate this fusion protein, the human BP180 cDNA segment encoding the NC16A region was PCR amplified and then subcloned into the bacterial expression vector pGEX2T (Pharmacia Biotech Inc., Piscataway, New Jersey, USA) as described (35). These recombinant proteins containing glutathione S-transferase (GST) were expressed in Escherichia coli strain DH5α and purified by glutathione-agarose affinity chromatography (36). The purified fusion proteins were dialyzed against PBS, concentrated by ultrafiltration, and filter sterilized. The protein concentration was determined by Bradford protein assays (Bio-Rad Laboratories Inc., Hercules, California, USA).
MHC II expression of FS patients.
HLA-DR and HLA-DQ typing of FS patients was conducted in the Immunogenetic Laboratory at the Fundação Pro-Sangue, São Paulo, Brazil, and the Department of Internal Medicine, Southwestern Medical Center, University of Texas, using PCR sequence–specific primers as described (10, 37). The HLA typing result of these FS patients is shown in Table 1.
Purification of PBMC and T-cell proliferation assays.
PBMC were isolated by Ficoll-Hypaque (Pharmacia Biotech Inc.) density gradient separation (32). T cells were then purified by E rosetting using sheep red blood cells (Colorado Serum Co., Denver, Colorado, USA) treated with 2-aminoethylisothiouronium bromide (Sigma Chemical Co., St. Louis, Missouri, USA) (38). The purity of the isolated T cells was greater than 95%, as determined by FACS® analysis (not shown). The purified T cells were washed with medium 3 times, and then resuspended in RPMI-1640 medium supplemented with 10% human AB serum (NABI, Miami, Florida, USA) for the T-cell proliferation assays.
T-cell responses to rDsg1, rDsg3, and GST fusion proteins were determined by proliferation assays as described (32). Briefly, 105 T cells/mL were cultured with 105 cells/mL of irradiated autologous PBMC as antigen-presenting cells in 96-well U-bottom plates for 7 days. Phytohemagglutinin (0.25 μg/mL; Sigma Chemical Co.) or IL-2 (10 U/mL; Collaborative Research Inc., Bedford, Massachusetts, USA) served as positive control in all T-cell proliferation experiments. Cells in individual wells were pulsed with 1 μCi of [3H]thymidine (ICN Pharmaceuticals Inc., Costa Mesa, California, USA) during the last 18 hours of incubation, and were then harvested using an automated cell harvester (Inotech Biosystems International Inc., Lansing, Michigan, USA). Proliferation of T cells was determined by measuring [3H]thymidine uptake on a beta counter (Wallac Inc., Gaithersburg, Maryland, USA). Data were presented as stimulation index (SI): cpm of cells treated with antigen/cpm of cells treated with no antigen or GST at the same concentration. An SI greater than or equal to 3 was considered a positive response.
Epstein-Barr virus–transformed B-cell lines and antibodies.
Epstein-Barr virus cell lines from FS patients were developed as described (39). These cell lines were cultured in medium supplemented with 10% FBS (HyClone Laboratories, Salt Lake City, Utah, USA) and were used as antigen-presenting cells in the maintenance of T-cell clones and T-cell proliferation assays.
Anti-CD3, -CD4, -CD8, CD19, –HLA-DR (clone B-F1), and -CD45RA Ig, and fluorescein-labeled goat F(ab′)2 anti-mouse Ig were purchased from BioSource International (Camarillo, California, USA). Anti–HLA-DQ, anti–HLA-DP, and anti-TCRα/β antibodies were obtained from Becton Dickinson Immunocytometry Systems (San Jose, California, USA). Anti-CD45RO was purchased from Immunotech (Westbrook, Maine, USA). Negative control mouse antibodies IgG2a and IgG1 were obtained from DAKO A/S (Glostrup, Denmark).
Development and characterization of rDsg1-specific T-cell lines and clones.
The Dsg1-specific T-cell lines and clones were developed from 5 FS patients using rDsg1, following the in vitro repeat stimulation protocol described previously (33). The cell surface expression of CD3, CD4, CD8, CD19, CD45RA, CD45RO, and TCRα/β on Dsg1-specific T cells was examined by flow cytometric analysis using a FACScan flow cytometer (Becton Dickinson Immunocytometry Systems) and specific monoclonal antibodies. Mouse IgG was used as a negative control. Fluorescein-conjugated F(ab′)2 goat anti-mouse Ig was used as the secondary antibody.
The antigen specificity of derived T-cell lines and clones was examined by culturing T cells with irradiated autologous PBMC or Epstein-Barr virus–transformed B cells (5 × 104 cells/mL of each) pulsed with 20 μg/mL of antigens for 5 days at 37°C in 96-well plates (Fisher Scientific Co., Pittsburgh, Pennsylvania, USA) at a final volume of 200 μL. T cells in each well were pulsed with 1 μCi of [3H]thymidine during the last 18 hours of incubation. An SI greater than or equal to 3 was considered a positive response.
Cytokine profile analysis of Dsg1-specific T cells.
For cytokine profile analysis, 106 T cells per well were cultured in a 24-well plate in the presence of 10 ng/mL of phorbol myristate acetate (Sigma Chemical Co.) and 100 ng/mL of anti-CD3 antibodies (40). Stimulated cells were collected and washed before total RNA was extracted using TRIzol reagent (GIBCO BRL, Grand Island, New York, USA). Subsequently, first-strand cDNAs was synthesized using the reverse transcription kit SuperScript (GIBCO BRL), following the manufacturer’s protocol. The messages of cytokines expressed by rDsg1-specific T lymphocytes were amplified by PCR using specific sets of primers (41) and Taq polymerase (GIBCO BRL). The PCR products were separated by a 1.5% agarose gel and were revealed with ethidium bromide staining under UV light. The sequences of the cytokine PCR primer set were: β-actin: 5′-TCCTGTGGCATC-CACGAAACT-3′ and 5′-GAAGCATTTGCGG-TGGACGAT-3′; IL-2: 5′-AA-CTCCTGTCTTGCATTGCA-3′ and 5′-GTGTTGAGATGATGCT-TTGAC-3′; IL-4: 5′-CAACTTT-GCTGGACTTCAACA-3′ and 5′-TCCAACGTACTCTGGTT-GG-3′; IL-5: 5′-AGGATGCTTCTG-CATTTGAG-3′ and CTATTA-TCCATCTCGGTGTTC-3′; IL-6: 5′-AACTCCTTCTCCATAAGC-G-3′ and 5′-TGGACTGC-AGGAACTCCTT-3′; TGF-β: 5′-AACATGATCGTGCGCTCCTGCA-AGTGCAGC-3′ and 5′-AAGGAATAGTGCAGACAG-GCAGGA-3′; and IFN-γ: 5′-ATGAAATAT-ACAATTATATC-3′ and 5′-TTACTGGGATGCTCT-TCGACCTCGAAACAGCAT-3′. The expected sizes of the PCR products were: β-actin: 314 bp; IL-2: 441 bp; IL-4: 345 bp; IL-5: 394 bp; IL-6: 610 bp; TGF-β: 200 bp; and IFN-γ: 501 bp. The presence of IL-2, IL-4, IL-5, IL-6, and IFN-γ in the supernatant of stimulated T cells was verified by commercial human cytokine ELISA kits (Cytimmune Sciences Inc., College Park, Maryland, USA).
Results
FS patients express limited MHC II alleles.
As reported in Table 1, almost all FS patients who were included in this study expressed at least 1 of the FS-associated HLA-DRB1 alleles: DRB1*0404, *1402, or *1602. Interestingly, all of these patients also expressed either the HLA-DQB1*0301 or the *0302 allele.
Immune responses of T lymphocytes from FS patients.
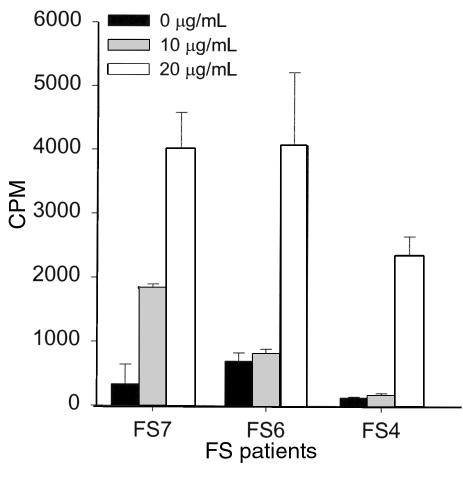
To determine whether T lymphocytes from FS patients responded to Dsg1, T cells were purified from PBMC of patients by E rosetting, and were subsequently used in a proliferation assay. As shown in Figure 2, total T cells from FS patients proliferated in response to rDsg1 in a dose-dependent manner. We found that T cells from 13 of 15 FS patients from Limao Verde showed a proliferation response to rDsg1 at an antigen concentration of 20 μg/mL, with an SI ranging from 3.1 to 17.8 (Table 1). The proliferative responses of FS T cells to rDsg1, however, do not tightly correlate to the indirect IF titers in the sera of patients. The immune response of FS T cells to Dsg1 was specific: the cells did not proliferate when incubated with BP180 fusion proteins or rDsg3 (not shown). T cells from a control group of individuals, which included cells from patients with bullous pemphigoid (n = 4), systemic lupus erythematosus (n = 1), and psoriasis (n = 2), as well as healthy volunteers (n = 7) and normal individuals from Limao Verde Reservation (n = 8), did not proliferate when incubated with rDsg1, indicating that the proliferative response to Dsg1 is specific to FS patients in this study.
Figure 2.
T lymphocytes of FS patients respond to rDsg1. The proliferative responses of FS T cells to rDsg1 were examined using proliferation assays as described. The responses of 3 representative patients are shown here. Data are expressed as average cpm ± SD.
Antigen specificity of rDsg1-specific T cells from FS patients.
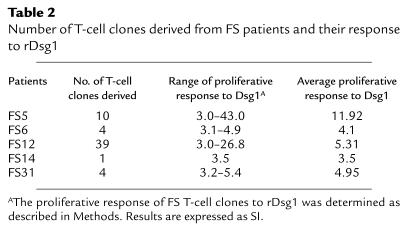
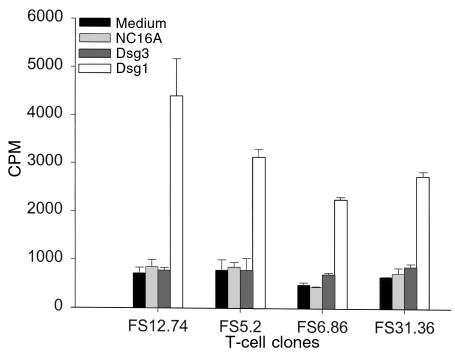
To further analyze the properties of rDsg1-specific T cells, long-term T cells were developed from 5 FS patients using an in vitro stimulation method as described previously (33). T-cell clones were subsequently derived from cell lines by limiting dilution assays. The clones that responded to rDsg1 with an SI greater than or equal to 3 were expanded by repeated stimulation in vitro. In total, we selected 58 T-cell clones for further property characterization. The number of clones derived from each patient and their average responses to rDsg1 are presented in Table 2. The antigen specificity of these T cells to rDsg1 was confirmed by T-cell proliferation assays. As shown in Figure 3, representative FS T-cell clones strongly proliferate in response to rDsg1, but not in response to the BP180 fusion protein or rDsg3, indicating that these FS T-cell clones respond specifically to rDsg1.
Table 2.
Number of T-cell clones derived from FS patients and their response to rDsg1
Figure 3.
FS T-cell clones specifically respond to rDsg1. The antigen specificity of T cells developed from FS patients was examined using the proliferation assay as described. Results from analyses of 4 representative T-cell clones are presented here. The data are expressed as average cpm ± SD.
Immune response of FS T-cell clones to rDsg1 is restricted to HLA-DR molecules.
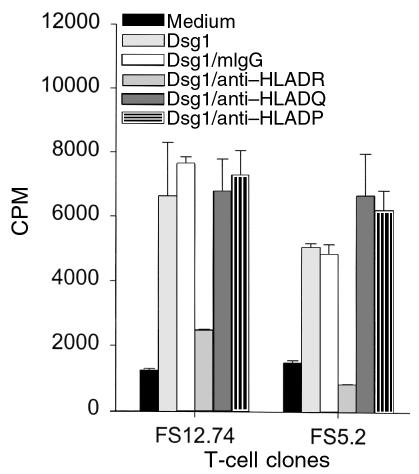
To investigate MHC II restriction of the response of FS T cells to Dsg1, anti–HLA-DR, anti–HLA-DQ, and anti–HLA-DP antibodies were used in the proliferation assays as described previously (32). Briefly, T cells were cultured with rDsg1-pulsed antigen-presenting cells in the presence of anti–MHC II antibodies or mouse isotype control antibodies for 5 days. Cell proliferation was subsequently determined by the [3H]thymidine incorporation assay. As demonstrated in Figure 4, only the anti–HLA-DR, and not the anti–HLA-DQ or anti–HLA-DP antibodies, inhibited the proliferation responses of FS T cells to rDsg1. This result clearly demonstrates that the response of FS T cells to Dsg1 requires HLA-DR molecules on antigen-presenting cells.
Figure 4.
The response of FS T-cell clones to rDsg1 is restricted to the expression of HLA-DR molecules. To examine the MHC II restriction of the FS T-cell response to Dsg1, anti–HLA-DR, anti–HLA-DQ, and anti–HLA-DP, monoclonal antibodies (and mouse isotype control antibodies) were incorporated into the proliferation assays as described. The results using 2 T-cell clones, FS12.74 and FS5.2, in 1 of the representative experiments are shown here. The data are expressed as average cpm ± SD.
Cell surface phenotype and cytokine profile of Dsg1-specific T cells.
Flow cytometric analysis was used to determine the cell-surface phenotype of rDsg1-specific T-cell clones developed from the FS patients. We found that Dsg1-specific T-cell clones expressed CD3, CD4, CD45RO, and TCRα/β, but were negative for CD8, CD45RA, and the B-cell marker CD19 (not shown), indicating that they are CD4-positive memory T cells.
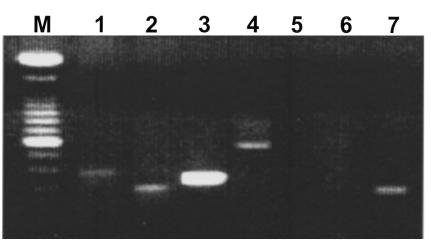
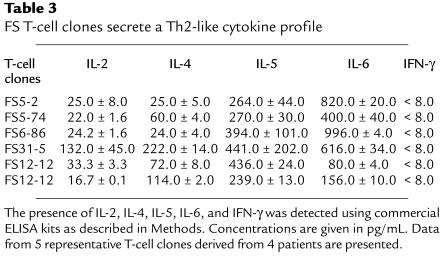
The cytokine profiles of the T cells from FS patients were determined as follows. T cells were activated by anti-CD3 antibodies in the presence of phorbol myristate acetate for 30 hours. The expression of IL-2, IL-4, IL-5, IL-6, TGF-β, and IFN-γ mRNA messages in these T cells was then examined by RT-PCR as described previously (41). As shown in Figure 5, a representative rDsg1-specific T-cell clone derived from an FS patient produced IL-2, IL-4, IL-5, and IL-6, but not IFN-γ or TGF-β. These results were confirmed by human cytokine–specific ELISA kits (Table 3). Although the IL-2 mRNA and IL-2 can be detected in these activated T cells, the message for this cytokine is weaker when compared with that of IL-4. Because the IFN-γ message is not detectable in these cell clones, it is suggested that FS-specific T cells express a Th2-type cytokine profile.
Figure 5.
FS T cells express a Th2-like cytokine profile. To examine the cytokine mRNA messages produced by FS T cells, total RNA was isolated from the cells and subjected to RT-PCR analysis. After production of first-strand cDNAs using reverse transcriptase, the mRNAs of cytokines were amplified using specific primer sets as described (40). The RT-PCR products were separated on a 1.5% agarose gel and viewed using ethidium bromide staining. Lane M, molecular weight marker; lane 1, IL-2; lane 2, IL-4; lane 3, IL-5; lane 4, IL-6; lane 5, IFN-γ; lane 6, TGF-β; lane 7, β-actin.
Table 3.
FS T-cell clones secrete a Th2-like cytokine profile
Discussion
The data presented in this paper clearly demonstrate that T cells from FS patients specifically proliferate when incubated with the extracellular domain of Dsg1. This study thus provides the first evidence that T lymphocytes from FS patients recognize the same autoantigen targeted by autoantibodies. T cells from 13 of 15 FS patients responded to rDsg1. Interestingly, the 2 patients whose T cells did not proliferate in response to Dsg1 were undergoing steroid therapy and were in partial remission at the time their lymphocytes were isolated. The proliferation of FS T cells to rDsg1 was antigen specific: they did not respond when incubated with other epidermal antigens such as BP180. T cells from other control groups, such as patients with BP, lupus, and psoriasis, and normal individuals from both the USA and Limao Verde, Brazil, remained unresponsive to rDsg1.
To further characterize the properties of Dsg1-responsive T cells from FS patients, cell clones were developed from 5 patients. These T-cell clones specifically responded to Dsg1, but not to other epidermal antigens such as Dsg3 and BP180. From the phenotype analysis, it was revealed that these T cells express CD3, CD4, CD45RO, and TCRα/β, but not CD8, CD19, or CD45RA, suggesting that they are CD4-positive memory T cells, a cell population shown to regulate the production of antibodies (33). This result further emphasizes the possibility that the Dsg1-reactive T cells participate in the autoimmune response of FS.
It is also known that the isotype of antibodies produced by a given B cell is dependent on the type of helper T lymphocytes that it encounters during the T-B cell interaction (42). For example, T cells that secrete Th1 cytokines are capable of stimulating B cells to produce IgG1, whereas Th2-type cytokines induce B cells to secrete IgG4 (43, 44). Because IgG4 is the predominant isotype of the anti-Dsg1 autoantibodies of FS, it is thought that T cells of the Th2 helper cell lineage may be relevant in this autoimmune disease. Our study supports this hypothesis, because T-cell clones derived from the study’s FS patients produce IL-4, IL-5, and IL-6, but not IFN-γ, suggesting that they secrete a Th2-like cytokine profile. The cytokines expressed by autoimmune FS T cells may promote the production of anti-Dsg1 autoantibodies of the IgG4 subclass in these patients. However, all of the T-cell clones analyzed in this study also express IL-2 mRNA and IL-2. Whether IL-2 plays a role in modulating the anti-Dsg1 production in these FS patients is not clear.
We have previously demonstrated that certain HLA-DRB1 alleles, such as DRB1*0404, *1402, *1406, and *1602 are prevalent in FS patients (10). As shown in Table 1, all of the FS patients recruited from the Limao Verde Reservation express at least 1 of the disease-associated alleles. Although these patients also express limited HLA-DQ alleles, such as DQB1*0301 and *0302, these 2 alleles are not significantly associated with FS (9, 10). In this study, we demonstrate that the proliferative response of FS T-cell clones was blocked by anti–HLA-DR antibodies, but not by anti–HLA-DQ or anti–HLA-DP antibodies (Figure 4), further indicating that the HLA-DR molecules play an important role in the Dsg1-specific immune reaction of FS T cells.
Previous investigations on rheumatoid arthritis–associated DRB1 alleles have shown that several groups of amino acid residues on the DRβ chain may regulate the binding of antigenic peptides (45). Among these key clusters, DRβ67–71 at the P4 position seems to play an essential role in the selection of the sequence of the binding peptides. Other positions, such as the anchor residues P1 and P6, also appear to be relevant in determining the affinity for sequences of peptides presented by DR molecules (46). Based on this information, it has been possible to predict Dsg3 antigenic peptides that are recognized by T cells from pemphigus vulgaris patients (47), and certain viral or bacterial peptides that stimulate myelin basic protein–specific T cells (48). In FS, the DRβ hypervariable domain from amino acids 67 to 74 of the alleles DRB1*0404, *1402, *1406, and *1602 share the same sequence (LLEQRRAA). Hence, it is possible that in FS these MHC II molecules may be involved in presenting the same Dsg1 peptides to autoimmune T cells. This possibility is currently under investigation.
In conclusion, our findings provide strong evidence that FS patients develop autoimmune T cells that specifically respond to the ectodomain of Dsg1, and this antigen-specific response requires the expression of HLA-DR. These Dsg1-reactive FS T cells exhibited a CD4-positive memory T-cell phenotype and produced a Th2-like cytokine profile. These T cells may stimulate autoimmune B cells in the production of pathogenic anti-Dsg1 autoantibodies. Further characterization of detailed Dsg1 epitopes that stimulate these T cells may help to elucidate the pathogenic mechanisms underlying the development of this disease.
Acknowledgments
We acknowledge with gratitude the support of the Cooperative Group on Fogo Selvagem Research. This work was supported in part by US Public Health Service Grants RO1-HL47145, UO1-AI34621 (P. Stastny), R01-AR40410 (G.J. Giudice), R37-AR30281, and RO1-AR32599 (L.A. Diaz), a Merit Award from the Veterans Administration Central Office (L.A. Diaz), a Dermatology Foundation Career Development Award, and a Dermatology Foundation Research grant (M.S. Lin).
References
- 1.Diaz LA, et al. Endemic pemphigus foliaceus (fogo selvagem). II. Current and historic epidemiologic studies. J Invest Dermatol. 1989;92:4–12. doi: 10.1111/1523-1747.ep13070394. [DOI] [PubMed] [Google Scholar]
- 2.Martins-Castro R, Proenco N, deSalles-Gomez LF. On the association of some dermatoses with South American pemphigus foliaceus. Int J Dermatol. 1974;13:271–275. doi: 10.1111/j.1365-4362.1974.tb05080.x. [DOI] [PubMed] [Google Scholar]
- 3.Robledo MA, Prada S, Jaramillo D, Leon W. South American pemphigus foliaceus: study of an epidemic in El Barge and Nechi, Colombia 1982 to 1986. Br J Dermatol. 1988;118:737–744. doi: 10.1111/j.1365-2133.1988.tb02590.x. [DOI] [PubMed] [Google Scholar]
- 4.Bastuji-Garin S, et al. Comparative epidemiology of pemphigus in Tunisia and France: unusual incidence of pemphigus foliaceus in young Tunisian women. J Invest Dermatol. 1995;104:302–305. doi: 10.1111/1523-1747.ep12612836. [DOI] [PubMed] [Google Scholar]
- 5.Morini JP, et al. Pemphigus foliaceus in young women. An endemic focus in the sousse area of Tunisia. Arch Dermatol. 1995;129:69–73. doi: 10.1001/archderm.129.1.69. [DOI] [PubMed] [Google Scholar]
- 6.Diaz LA, et al. Endemic pemphigus foliaceus (fogo selvagem). I. Clinical features and immunopathology. J Am Acad Dermatol. 1989;20:657–669. doi: 10.1016/s0190-9622(89)70079-7. [DOI] [PubMed] [Google Scholar]
- 7.Stanley JR, Klaus-Kovtun V, Sampaio SAP. Antigenic specificity of fogo selvagem autoantibodies is similar to North American pemphigus foliaceus and distinct from pemphigus vulgaris. J Invest Dermatol. 1986;87:197–201. doi: 10.1111/1523-1747.ep12695334. [DOI] [PubMed] [Google Scholar]
- 8.Sequiquera HL, et al. Serologic abnormalities in patients with endemic pemphigus foliaceus (fogo selvagem), their relatives, and normal donors from endemic and non-endemic areas of Brazil. J Invest Dermatol. 1988;91:189–191. doi: 10.1111/1523-1747.ep12464490. [DOI] [PubMed] [Google Scholar]
- 9.Cerna M, et al. Genetic markers for susceptibility to endemic Brazilian pemphigus foliaceus (fogo selvagem) in Xavante Indians. Tissue Antigens. 1993;42:138–140. doi: 10.1111/j.1399-0039.1993.tb02180.x. [DOI] [PubMed] [Google Scholar]
- 10.Moraes ME, et al. An epitope in the third hypervariable region of the DRB1 gene is involved in the susceptibility to endemic pemphigus foliaceus (fogo selvagem) in three different Brazilian populations. Tissue Antigens. 1997;49:35–40. doi: 10.1111/j.1399-0039.1997.tb02707.x. [DOI] [PubMed] [Google Scholar]
- 11.Hans-Filho G, et al. An active focus of high prevalence of fogo selvagem on an Amerindian reservation in Brazil. J Invest Dermatol. 1996;107:68–75. doi: 10.1111/1523-1747.ep12298213. [DOI] [PubMed] [Google Scholar]
- 12.Eaton DP, et al. Comparison of black fly species (Diptera: Simuliidae) on an Amerindian reservation with a high prevalence of fogo selvagem to neighboring disease-free sites in the State of Mato Grosso do Sul, Brazil. The Cooperative Group on Fogo Selvagem Research. 1998. J Med Entomol. 1998;35:120–131. doi: 10.1093/jmedent/35.2.120. [DOI] [PubMed] [Google Scholar]
- 13.Lin MS, Mascaro JM, Jr, Liu Z, España A, Diaz LA. The desmosome and hemidesmosome in cutaneous autoimmunity. Clin Exp Immunol. 1997;107:9–15. [PubMed] [Google Scholar]
- 14.Lever WF. Pemphigus. Medicine. 1953;32:1–123. doi: 10.1097/00005792-195302000-00001. [DOI] [PubMed] [Google Scholar]
- 15.Rock B, et al. The pathogenic effect of IgG4 autoantibodies in endemic pemphigus foliaceus (fogo selvagem) N Engl J Med. 1989;320:1463–1469. doi: 10.1056/NEJM198906013202206. [DOI] [PubMed] [Google Scholar]
- 16.Roscoe JT, et al. Brazilian pemphigus foliaceus autoantibodies are pathogenic to BALB/c mice by passive transfer. J Invest Dermatol. 1985;85:538–541. doi: 10.1111/1523-1747.ep12277362. [DOI] [PubMed] [Google Scholar]
- 17.Rock B, Labib RS, Diaz LA. Monovalent Fab′ immunoglobulin fragments from endemic pemphigus foliaceus autoantibodies reproduce the human disease in neonatal Balb/c mice. J Clin Invest. 1990;85:296–299. doi: 10.1172/JCI114426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wheeler GN, et al. Desmosomal glycoprotein DGI, a component of intercellular desmosome junctions, is related to the cadherin family of cell adhesion molecules. Proc Natl Acad Sci USA. 1991;88:4796–4800. doi: 10.1073/pnas.88.11.4796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Buxton RS, et al. Nomenclature of the desmosomal cadherin. J Cell Biol. 1993;121:481–483. doi: 10.1083/jcb.121.3.481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Olague-Alcala M, Diaz LA. The epitopes on bovine pemphigus foliaceus antigen are calcium-dependent and located on the peptide backbone of this glycoprotein. Chronica Dermatologica. 1993;2:189–209. [Google Scholar]
- 21.Emery DJ, et al. Pemphigus foliaceus and pemphigus vulgaris autoantibodies react with the extracellular domain of desmoglein-1. J Invest Dermatol. 1995;104:323–328. doi: 10.1111/1523-1747.ep12665364. [DOI] [PubMed] [Google Scholar]
- 22.Amagai M, Tsunoda K, Zillikens D, Nagai T, Nishikawa T. The clinical phenotype of pemphigus is defined by the anti-desmoglein autoantibody profile. J Am Acad Dermatol. 1999;40:167–170. doi: 10.1016/s0190-9622(99)70183-0. [DOI] [PubMed] [Google Scholar]
- 23.Ding X, Diaz LA, Fairley JA, Giudice GJ, Liu Z. The anti-desmoglein 1 autoantibodies in pemphigus vulgaris sera are pathogenic. J Invest Dermatol. 1999;112:739–743. doi: 10.1046/j.1523-1747.1999.00585.x. [DOI] [PubMed] [Google Scholar]
- 24.Lin, M.S., Liu, Z., Drolet, B.A., and Diaz, L.A. 1998. Cutaneous autoimmune diseases. In The autoimmune diseases. 3rd edition. N.R. Rose and I.R. Mackay, editors. Academic Press. San Diego, CA. 545–570.
- 25.Coffman RL, et al. The role of helper T cell products in mouse B cell differentiation and isotype regulation. Immunol Rev. 1988;102:5–28. doi: 10.1111/j.1600-065x.1988.tb00739.x. [DOI] [PubMed] [Google Scholar]
- 26.Barlett WC, et al. Cognate interactions between helper T cells and B cells. II. Dissection of cognate help by using class II-restricted, antigen-specific, IL-2-dependent helper T cell clone. J Immunol. 1989;143:1745–1754. [PubMed] [Google Scholar]
- 27.Hamano T, et al. Direct interaction between an antigen-specific B cell clones and an MHC class II-reactive T helper cell clone. J Leukoc Biol. 1992;52:89–96. doi: 10.1002/jlb.52.1.89. [DOI] [PubMed] [Google Scholar]
- 28.Stevens TL, et al. Regulation of antibody isotype secretion by subsets of antigen-specific helper T cells. Nature. 1988;334:255–258. doi: 10.1038/334255a0. [DOI] [PubMed] [Google Scholar]
- 29.Allegretta M, Nicklas JA, Siram S, Albertini RJ. T cells responsive to myelin basic protein in patients with multiple sclerosis. Science. 1990;247:718–721. doi: 10.1126/science.1689076. [DOI] [PubMed] [Google Scholar]
- 30.Melms A, et al. Autoimmune T lymphocytes in myasthenia gravis. Determination of target epitopes using T cell lines and recombinant products of the mouse nicotinic acetylcholine receptor gene. J Clin Invest. 1989;83:785–790. doi: 10.1172/JCI113958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mullins RJ, et al. Identification of thyroid stimulating hormone receptor-specific T cells in Graves’ disease thyroid using autoantigen-transfected Epstein-Barr virus-transformed B cells lines. J Clin Invest. 1995;96:30–37. doi: 10.1172/JCI118034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lin MS, Gharia MA, Swartz SJ, Diaz LA, Giudice GJ. Identification and characterization of epitopes recognized by T lymphocytes and autoantibodies from patients with herpes gestationis. J Immunol. 1999;162:4991–4997. [PubMed] [Google Scholar]
- 33.Lin MS, et al. Development and characterization of desmoglein-3 specific T cells from patients with pemphigus vulgaris. J Clin Invest. 1997;99:31–40. doi: 10.1172/JCI119130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Amagai M, Hashimoto T, Green KJ, Shimizu N, Nishikawa T. Antigen-specific immunoadsorption of pathogenic autoantibodies in pemphigus foliaceus. J Invest Dermatol. 1995;104:895–901. doi: 10.1111/1523-1747.ep12606168. [DOI] [PubMed] [Google Scholar]
- 35.Liu Z, et al. Molecular mapping of a pathogenically relevant BP180 epitope associated with experimentally induced murine bullous pemphigoid. J Immunol. 1995;155:5449–5454. [PubMed] [Google Scholar]
- 36.Liu Z, Diaz LA, Haas L, Giudice GJ. cDNA cloning of a novel human ubiquitin carrier protein. An antigenic domain specifically recognized by endemic pemphigus foliaceus autoantibodies is encoded in a secondary reading frame of this human epidermal transcript. J Biol Chem. 1992;267:15829–15835. [PubMed] [Google Scholar]
- 37.Lazaro, A., et al. Evolution of HLA class I compared to HLA class II polymorphism in Terena, a South American Indian tribe. Hum. Immunol. In press. [DOI] [PubMed]
- 38.Indiveri F, Huddlestone J, Pellegrino MA, Ferrone S. Isolation of human T lymphocytes: comparison between wool filtration and rosetting with neuraminidase (VCN) and 2-aminoethylisothiouronium bromide (AET)-treated sheep red blood cells. J Immunol Methods. 1980;34:107–115. doi: 10.1016/0022-1759(80)90164-7. [DOI] [PubMed] [Google Scholar]
- 39.Ohlin M, Danielsson L, Carlsson R, Borrebaeck AK. The effect of leucyl-leucine methyl ester on proliferation and Ig secretion of EBV-transformed human B lymphocytes. Immunology. 1989;66:485–490. [PMC free article] [PubMed] [Google Scholar]
- 40.Del Prete G, et al. Preferential expression of CD30 by human CD4+ T cells producing Th2-type cytokines. FASEB J. 1995;9:81–86. [PubMed] [Google Scholar]
- 41.Brenner CA, et al. Message amplification phenotyping (MAPPing): a technique to simultaneously measure multiple mRNAs from small numbers of cells. Biotechniques. 1989;7:1096–1101. [PubMed] [Google Scholar]
- 42.Lin MS, Chen YW. B cell differentiation. II. Isotype potential of a single B cells. Cell Immunol. 1993;150:343–352. doi: 10.1006/cimm.1993.1202. [DOI] [PubMed] [Google Scholar]
- 43.Armitage RJ, MacDuff BM, Spriggs MK, Fanslow WC. Human B cell proliferation and Ig secretion induced by recombinant CD40 ligand are modulated by soluble cytokines. J Immunol. 1993;150:3671–3680. [PubMed] [Google Scholar]
- 44.Romagnani S. Human Th1 and Th2 subsets: regulation of differentiation and role in proliferation and immunopathology. Int Arch Allergy Appl Immunol. 1992;98:279–285. doi: 10.1159/000236199. [DOI] [PubMed] [Google Scholar]
- 45.Hammer J, et al. Peptide binding to HLA-DR4 molecules: correlation with rheumatoid arthritis association. J Exp Med. 1995;181:1847–1855. doi: 10.1084/jem.181.5.1847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Hammer J, et al. Promiscuous and allele-specific anchors in HLA-DR binding peptides. Cell. 1993;74:197–203. doi: 10.1016/0092-8674(93)90306-b. [DOI] [PubMed] [Google Scholar]
- 47.Wucherpfennig KW, Strominger JL. Molecular mimicry in T cell-mediated autoimmunity: viral peptides activate human T cell clones specific for myelin basic protein. Cell. 1995;80:695–705. doi: 10.1016/0092-8674(95)90348-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wucherpfennig KW, Strominger JL. Selective binding of self peptides to disease-associated major histocompatibility complex (MHC) molecules: a mechanism for MHC-linked susceptibility to human autoimmune diseases. J Exp Med. 1995;181:1597–1601. doi: 10.1084/jem.181.5.1597. [DOI] [PMC free article] [PubMed] [Google Scholar]