Abstract
Background
BRAF mutations occur in approximately 8% of all human cancers and approach 50% in melanoma and papillary carcinoma of thyroid. These mutations provide potentially valuable diagnostic, prognostic and treatment response prediction markers. A sensitive, specific, low-cost assay to detect these mutations is needed.
Results
To detect BRAF V600E mutation in formalin-fixed, paraffin-embedded (FFPE) tissue, we developed a method using Amplification Refractory Mutation System (ARMS)-PCR. This method was designed to amplify three products in a single reaction tube: a 200 bp common product serving as an amplification control, a 144 bp BRAF V600E specific product, and a 97 bp wild-type (wt) specific product. The sensitivity of this method was determined to be as low as 0.5% for the BRAF V600E allele in a wild-type background. This method was successfully validated in 72 thyroid tumors. It detected V600E mutation in 22 out of 33 (67%) of the conventional papillary thyroid carcinoma (PTC), 8 out of 12 (75%) of the tall-cell variant of PTC, whereas none of the 10 follicular variant of PTC showed BRAF V600E mutation. In addition, none of the 14 follicular adenomas and 3 follicular carcinomas had BRAF V600E mutation. As a comparison method, direct dideoxy sequencing found only 27 out of 30 (90%) mutations detected by ARMS-PCR method, suggesting that this ARMS-PCR method has higher sensitivity.
Conclusions
Our ARMS-PCR method provides a new tool for rapid detection of BRAF V600E mutation. Our results indicate that ARMS-PCR is more sensitive than automated dideoxy sequencing in detecting low BRAF V600E allele burdens in FFPE tumor specimen. The strategy of this ARMS-PCR design may be adapted for early detection of point mutations of a variety of biomarker genes.
Background
BRAF gene encodes a serine/threonine protein kinase that functions downstream of RAS in the RAS-RAF-MEK-ERK signaling pathway, also known as mitogen activated protein kinase (MAPK) pathway, which is important in regulating cellular responses to extracellular signals including epidermal growth factor (EGF) [1]. Upon activation by receptor binding, RAS proteins recruit RAF to the cellular membrane where RAF is phosphorylated [2]. Activated RAF proteins further phosphorylate MEK1 and MEK2, which then phosphorylate ERK1 and ERK2. Phosphorylated ERK proteins regulate cellular functions through activation of transcription factors including p53, SMAD4, ELK1, c-Myc and c-Fos [2,3]. Activating mutations in RAS and BRAF permit constitutive MAPK pathway activation independent of growth factor stimulation, thereby causing deregulation in cellular growth and survival. BRAF mutations occur in approximately 8% of all human cancers, with high mutation frequency in malignant melanoma (50-70%), classic papillary carcinoma of the thyroid (40-70%), colorectal cancer (CRC, 5-15%), ovarian cancer and hairy cell leukemia (5-100%) [4,5]. A T1799A transversion resulting in valine-to-glutamate substitution at codon 600 (V600E) accounts for over 80% of all BRAF mutations [6,7], and almost all mutations in thyroid tumors [8].
Molecular testing for BRAF mutation can provide valuable diagnostic information, treatment selection, and may help predict prognosis. Like KRAS mutations, CRC patients with activating BRAF mutations do not respond to anti-EGFR monoclonal antibody (MoAb) treatment [9,10]. Because BRAF mutations occur in 5-15% of CRC and are usually mutually exclusive with KRAS mutations [11], a substantial number of these CRC patients may benefit from BRAF mutation testing before initiating MoAb treatment [9]. In thyroid, BRAF V600E mutation is restricted to malignant tumors, and is associated with papillary thyroid cancer (PTC), and PTC-derived poorly-differentiated and anaplastic carcinoma [7,8]. Therefore, BRAF mutation testing can be helpful as an adjunct method to improve diagnostic accuracy in difficult cases, such as thyroid FNA samples with indeterminate and atypical cytology [12,13].
There are several methods for BRAF V600E mutation testing, including dideoxy sequencing, colorimetric Mutector assay, allele-specific real-time PCR, pyrosequencing, high resolution melting (HRM) analysis and COLD-PCR [14-17]. These methods vary in their sensitivity, assay complexity and costs. Automated dideoxy sequencing is considered the “gold standard” method for mutation testing. However, it is limited by relatively complex procedure, overall high costs and low analytical sensitivity of detecting approximately 15-20% mutant allele in a wild-type background [18]. ARMS-PCR is a highly sensitive, specific and low-cost mutation detection method with reported analytical sensitivity ranging from 0.1% to 2% for JAK2 V617F mutation detection [19,20]. Here we report the development of a highly sensitive and specific ARMS-PCR assay to detect BRAF V600E mutation in formalin-fixed, paraffin-embedded (FFPE) tissue with sensitivity as low as 0.5%. We successfully used this assay to characterize the frequency of BRAF V600E in thyroid tumors.
Methods
Sample source
Cell lines HT-29 (heterozygous for BRAF V600E) and K-562 (wt BRAF without V600E mutation) were cultured in RPMI1640 supplemented with 10% fetal calf serum. Anonymized FFPE thyroid tumor tissue blocks were obtained from the Department of Pathology for clinical protocol development.
DNA extraction
3–5 sections of FFPE tissues (5 micron/section) were deparaffinized by xylene and ethanol treatment, and digested with proteinase K at 56°C overnight. Genomic DNA was extracted using ZR genomic DNA I kit (ZYMO Research Corp., Orange, CA) as recommended. DNA extraction from culture cells was otherwise the same as above except no deparaffinization was performed. DNA was then qualified/quantified with ND-1000 spectrophotometer (NanoDrop Products, Wilmington, DE).
ARMS-PCR and sequencing
The ARMS-PCR primer sequences were forward (Fo): 5′-CTCTTCATAATGCTTGCTCTGATAG-3′; reverse (Ro): 5′-GCCTCAATTCTTACCATCCAC-3′; forward wild-type identifying (Fiwt): 5′-GTGATTTTGGTCTAGCTACAGT-3′ and reverse mutation identifying (Rimut):5′-CCCACTCCATCGAGATTTCT-3′. PCR was performed in a 25 μl final volume containing 1 × Buffer, 2 mM MgCl2, 1 unit of Hotstar Taq DNA polymerase (Qiagen Science, Valencia, CA), 200 μM each dNTP, 400 nM primer Fo, 200 nM primer Ro and Fiwt, 800 nM primer Rimut and 30 ng genomic DNA template. PCR amplification was carried out by denaturation at 95°C for 5 min, followed by 40 cycles of 94°C for 20 sec, 68°C for 20 sec and 72°C for 20 sec with a final extension at 72°C for 5 min. PCR products were analyzed by 2% agarose gel electrophoresis. Automated dideoxy sequencing was performed by Macrogen USA (Rockville, MD) after PCR amplification using the Fo-Ro primer pair and the ampliocons purification using QIAquick PCR purification kit (Qiagen).
Assay sensitivity study
HT-29 cell DNA with heterozygous BRAF V600E mutation was serial diluted with DNA from a wt BRAF cell line K-562, to generate a sensitivity panel consisting of 0, 0.1%, 0.25%, 0.5%, 1%, 2.5%, 5%, 10%, 20% and 50% of BRAF V600E allele in a wild-type background. The PCR was performed under the optimized condition as above.
Results
Method establishment
The BRAF V600E ARMS-PCR assay contains 4 primers in a single PCR reaction tube with the two outside primers designed to amplify a common fragment of 200 bp flanking the mutation site which can serve as an internal amplification control (Figure 1A). The two internal primers are either specific for the mutant sequence or the wild-type sequence. The mutant and wild-type sequences are distinguishable by the difference of fragment size, as the amplified wild-type fragment is 97 bp, while the mutant fragment is 144 bp. The concentrations of PCR primers and magnesium, the annealing temperature and other cycling parameters were determined by multiple exploring studies, and the condition given here is the final optimized one.
Figure 1.
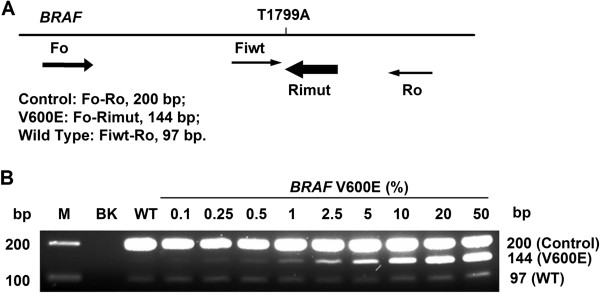
BRAF V600E ARMS-PCR.A, Four PCR primers are included in one PCR tube. Fo-Ro primer pair generates a common fragment of 200 bp flanking the mutation site. Fo-Rimut primer pair generates the 144 bp fragment specific to BRAF V600E. Fiwt-Ro primer pair generates a 97 bp fragment specific to wild-type BRAF. B, Sensitivity study. Agarose gel electrophoresis showed band pattern of a mixing study using BRAF V600E positive HT-29 cell DNA spiked into K-562 cell DNA at wide range of ratio namely 0, 0.1%, 0.25%, 0.5%, 1%, 2.5%, 5%, 10%, 20% and 50%. The assay can detect 0.5% BRAF V600E allele in a wild-type background. M, 100 bp DNA marker; BK, blank (no DNA) control.
Assay sensitivity
To assess the analytical sensitivity of the assay, BRAF V600E mutation containing DNA was spiked into wild-type BRAF DNA, the PCR was performed and showed that the assay we used could detect as low as, if not better than, 0.5% BRAF V600E allele in the background of wild-type DNA (Figure 1B). Therefore, the analytical sensitivity of the assay is 0.5%.
Assay validation and comparison with direct dideoxy sequencing using thyroid tumor specimens
We then analyzed BRAF V600E in thyroid tumors using the ARMS-PCR method. A total of 72 thyroid tumors were tested, including 55 PTCs and 17 follicular tumors. Among 55 PTCs, 33 were conventional type, 12 were tall-cell variant, and 10 were follicular variant (Table 1). ARMS-PCR results showed that 22 out of 33 (67%) of the conventional PTC, 8 out of 12 (75%) of the tall-cell variant of PTC, and none of the 10 follicular variant of PTC harbored BRAF V600E mutation. Interestingly, none of the 14 follicular adenomas and 3 follicular carcinomas showed BRAF V600E mutation.
Table 1.
BRAF V600E mutation rate in thyroid tumors
| Tumor | Sample number | Positive | Positive rate (%) |
|---|---|---|---|
|
Papillary Thyroid Carcinoma |
55 |
30 |
55 |
| Conventional Type |
33 |
22 |
67 |
| Tall-cell Variant |
12 |
8 |
75 |
| Follicular Variant |
10 |
0 |
0 |
|
Follicular Tumor |
17 |
0 |
0 |
| Follicular Adenoma |
14 |
0 |
0 |
| Follicular Carcinoma | 3 | 0 | 0 |
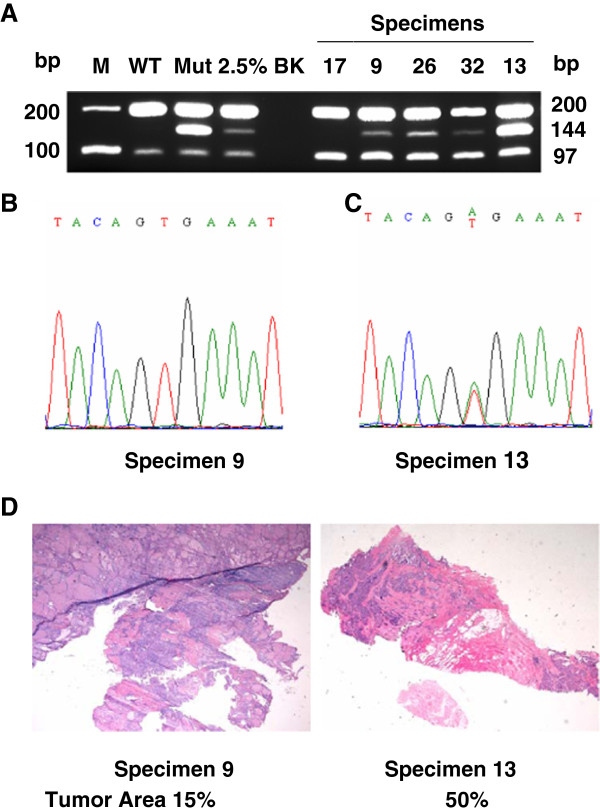
As a comparison, automated dideoxy sequencing was used to analyze 42 tumors including all 30 BRAF V600E positive tumors and 12 of the BRAF V600E negative tumors from ARMS-PCR assayed samples. Among the 30 BRAF V600E positive tumors detected with ARMS-PCR assay, direct sequencing identified the mutation in 27 tumors, but failed to identify 3 tumors that had BRAF V600E mutation, possibly due to the lower sensitivity of detection of automated dideoxy sequencing. Figure 2A depicts the representative ARMS-PCR results, showing samples 9, 26 and 32 with faint, but clearly visible BRAF V600E bands (144 bp). This is comparable to the mutant band intensity of the 2.5% V600E allele burden positive control included in the study. Figure 2B depicts the representative sequencing result for sample 9, showing the mutant signal was below the detection limit of the assay. While ARMS-PCR for sample 13 showed a strong V600E mutant band, and direct sequencing showed A/T double peaks with approximately equal height (Figure 2C). Histologic study of specimen 9 showed that the tumor constituted only a small fraction of the tissue area and sample 13 had a high tumor percentage (Figure 2D). These two cases illustrate the importance of high assay sensitivity of our ARMS-PCR assay to detect low BRAF V600E allele burdens in tumor samples. The assay results for the 12 BRAF V600E negative tumors were concordant between the ARMS-PCR and direct sequencing. All 14 cases of follicular adenomas had no mutated band detected by ARMS-PCR, showing the specificity of our assay was 100% in this validation study.
Figure 2.
A, Representative BRAF V600E ARMS-PCR results. Specimen 9, 26 and 32 had a low abundance of BRAF V600E mutation with band intensity similar to that of the 2.5% sensitivity control. Specimen 13 was strongly positive for BRAF V600E mutation. B, Direct dideoxy sequencing of specimen 9 failed to identify BRAF V600E mutation as the mutant allele burden was below the limit of detection. C, Direct dideoxy sequencing of specimens 13 showed that the mutant allele (A) was more abundant than the wild-type allele (T). D, H&E stain of specimens 9 and 13 showed that tumor areas accounted for ~15% and 50% of the total specimen areas, respectively.
Overall, our results indicate that ARMS-PCR is more sensitive than automated dideoxy sequencing in detecting low BRAF V600E allele burdens in FFPE tumor specimen.
Discussion
For the development and implementation of personalized medicine, a sensitive, specific, low-cost and easy-to-implement assay to identify various genetic alterations such as BRAF V600E mutation is needed. By selecting primer pair and optimizing PCR cycling conditions, we achieved this goal by using ARMS based PCR. One of the advantages of ARMS-PCR is that the assay is designed to amplify a relative larger common fragment of DNA that flanks the mutation site in all samples regardless of their mutation status. This common fragment conveniently serves as an internal control for template DNA quality as well as potential PCR inhibition. The mutant or wild-type specific PCR amplifications take place in the same reaction tube, thereby allowing the mutant or wild-type specific PCR primers to compete for binding to very limited templates. To increase the sensitivity for detecting this mutation, we tried to change the ratio of four primers and found that the combination of 400 nM primer Fo, 200 nM primer Ro and Fiwt, 800 nM primer Rimut resulted the strongest mutant band. The primer binding to template varies based on their annealing conditions, but relative high Fo and the highest Rimut primer concentrations might favor the production of mutant product in our design. In addition, relative low concentration for Fiwt and Ro primers might reduce full length and wild type product formation by the competition effect of each primer. By such a design, the three products were produced at reasonably comparable levels. Otherwise, the wild type or whole length bands might overshadow the weak mutant band if the mutant BRAF V600E abundance is low, which is frequently the case for clinical specimens. With this study, we improved assay sensitivity to as low as 0.5% mutant allele level in the background of wild-type DNA, which is quite sensitive among ARMS-PCR methods [21]. This sensitivity is very difficult to reach with direct dideoxy sequencing which has the sensitivity of 15-20%, and with pyrosequencing which has greatly increased sensitivity to about 2% [22,23]. Considering DNA samples obtained from FFPE tissue is often fragmented due to damage by formalin fixation, we designed our PCR product size to be small (no more than 200 bp) to maximize the chance of successful amplification. This was proven to be helpful since all 72 FFPE samples had successful amplification.
The reported prevalence of BRAF V600E mutation for conventional PTC varied from 36% to 67% in the US and European studies [7,8,24-27], and was 83% in a Korean study [28]. The difference in prevalence could be due to different study population or different methodologies used. The prevalence of BRAF V600E mutation in the tall-cell variant PTC is in general higher than that of the conventional type, with a mean prevalence of 77% in several studies [8]. On the contrary, the follicular variant of PTC is rarely involved by BRAF V600E mutation, with a mean prevalence of 12% [8]. Using ARMS-PCR, we tested 72 thyroid tumors and determined that the frequencies of BRAF V600E mutation in the conventional, tall-cell and follicular variants of PTC were 66%, 75% and 0%, respectively. Our frequency of BRAF V600E mutation in conventional PTC was at the high end of the reported results, possibly reflecting our improved assay sensitivity compared with automated DNA sequencing, which was the method used in many of the reported studies. Our study showed that DNA sequencing method missed 3 out of 30 PTC samples with BRAF V600E detected by ARMS-PCR, resulting in a false negative rate of 10%. In our validation study, the assay specificity is 100% (14/14) based on negative mutation in follicular adenoma. This is consistent with previous reports since benign follicular adenoma has not been found to harbor the BRAF mutation [8].
BRAF V600E mutation testing has demonstrated utility in helping select CRC patients who are considering monoclonal antibody therapy as wild-type BRAF is required for response to anti-EGFR antibodies [9] and improve diagnostic accuracy in thyroid FNA samples [12,13]. In addition, BRAF V600E mutation is associated with sporadic microsatellite instable CRC, but not hereditary non-polyposis colorectal cancer (HNPCC) syndrome [29,30]. Therefore, the presence of BRAF V600E mutation is an exclusion criteria for HNPCC genetic testing [29,30]. BRAF V600E mutation testing can also help facilitate clinical studies of BRAF-targeted therapies [31]. The improved understanding of the role BRAF mutations in cancer diagnosis, prognosis and treatment has increased the need for BRAF mutation testing [4,32].
New methods are constantly being developed for BRAF V600E mutation detection. Shifted termination assay (STA) developed by TrimGen Corporation declared the sensitivity of 1-5% [33]. Lang et al. developed an allele-specific, also known as ARMS, real-time PCR using Taqman probe to increase the sensitivity to 1% and using internal control to exclude false negative results [34]. Morandi et al. developed an allele specific locked nucleic acid (LNA) quantitative PCR assay using LNA-modified allele specific primers and LNA-modified beacon probes to achieve sensitivity of 0.1% [35]. Dual-priming oligonucleotide (DPO)-based multiplex PCR was commercially available from Seegene (Seoul, Korea) and has a sensitivity of 2% [36]. Qiagen developed a BRAF mutation detection PCR kit with a sensitivity of 1.27%. Our method using ARMS-PCR therefore appears to have high sensitivity (0.5%) for BRAF V600E mutation detection.
Conclusions
We have developed a sensitive, specific and low-cost ARMS-PCR assay to detect BRAF V600E mutation. Comparative study showed that this assay is superior to automated dideoxy sequencing in terms of assay sensitivity, turn-around time and costs. The ARMS-PCR assay can be easily implemented by many molecular laboratories for BRAF V600E mutation testing. Our method provides one of the most sensitive methods for BRAF V600E gene mutation detection. The principal of our study design can be potentially adapted to detect other low abundance point mutations such as tumors with rich background stroma and post-treatment tumor samples.
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
TH: design, data analysis, paper writing submission. JZ: design experiments and implement. WWZ: design study, sample collection, data analysis and paper writing. All authors read and approved the final manuscript.
Contributor Information
Tiangui Huang, Email: huangt@wcmc.com.
Jian Zhuge, Email: zhugej@wcmc.com.
Wenyong W Zhang, Email: wenyongzhang@gmail.com.
Acknowledgments
We thank Dr. Minghao Zhong for critical reading of the manuscript.
References
- Ciardiello F, Tortora G. EGFR antagonists in cancer treatment. N Engl J Med. 2008;358:1160–1174. doi: 10.1056/NEJMra0707704. [DOI] [PubMed] [Google Scholar]
- Zhang BH, Guan KL. Activation of B-Raf kinase requires phosphorylation of the conserved residues Thr598 and Ser601. EMBO J. 2000;19:5429–5439. doi: 10.1093/emboj/19.20.5429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turjanski AG, Vaque JP, Gutkind JS. MAP kinases and the control of nuclear events. Oncogene. 2007;26:3240–3253. doi: 10.1038/sj.onc.1210415. [DOI] [PubMed] [Google Scholar]
- Pratilas CA, Solit DB. Therapeutic strategies for targeting BRAF in human cancer. Rev Recent Clin Trials. 2007;2:121–134. doi: 10.2174/157488707780599393. [DOI] [PubMed] [Google Scholar]
- Tiacci E, Trifonov V, Schiavoni G, Holmes A, Kern W, Martelli MP, Pucciarini A, Bigerna B, Pacini R, Wells VA. et al. BRAF mutations in hairy-cell leukemia. N Engl J Med. 2011;364:2305–2315. doi: 10.1056/NEJMoa1014209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davies H, Bignell GR, Cox C, Stephens P, Edkins S, Clegg S, Teague J, Woffendin H, Garnett MJ, Bottomley W. et al. Mutations of the BRAF gene in human cancer. Nature. 2002;417:949–954. doi: 10.1038/nature00766. [DOI] [PubMed] [Google Scholar]
- Nikiforova MN, Kimura ET, Gandhi M, Biddinger PW, Knauf JA, Basolo F, Zhu Z, Giannini R, Salvatore G, Fusco A. et al. BRAF mutations in thyroid tumors are restricted to papillary carcinomas and anaplastic or poorly differentiated carcinomas arising from papillary carcinomas. J Clin Endocrinol Metab. 2003;88:5399–5404. doi: 10.1210/jc.2003-030838. [DOI] [PubMed] [Google Scholar]
- Xing M. BRAF mutation in thyroid cancer. Endocr Relat Cancer. 2005;12:245–262. doi: 10.1677/erc.1.0978. [DOI] [PubMed] [Google Scholar]
- Di Nicolantonio F, Martini M, Molinari F, Sartore-Bianchi A, Arena S, Saletti P, De Dosso S, Mazzucchelli L, Frattini M, Siena S, Bardelli A. Wild-type BRAF is required for response to panitumumab or cetuximab in metastatic colorectal cancer. J Clin Oncol. 2008;26:5705–5712. doi: 10.1200/JCO.2008.18.0786. [DOI] [PubMed] [Google Scholar]
- Siena S, Sartore-Bianchi A, Di Nicolantonio F, Balfour J, Bardelli A. Biomarkers predicting clinical outcome of epidermal growth factor receptor-targeted therapy in metastatic colorectal cancer. J Natl Cancer Inst. 2009;101:1308–1324. doi: 10.1093/jnci/djp280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Normanno N, Tejpar S, Morgillo F, De Luca A, Van Cutsem E, Ciardiello F. Implications for KRAS status and EGFR-targeted therapies in metastatic CRC. Nature reviews. 2009;6:519–527. doi: 10.1038/nrclinonc.2009.111. [DOI] [PubMed] [Google Scholar]
- Salvatore G, Giannini R, Faviana P, Caleo A, Migliaccio I, Fagin JA, Nikiforov YE, Troncone G, Palombini L, Basolo F, Santoro M. Analysis of BRAF point mutation and RET/PTC rearrangement refines the fine-needle aspiration diagnosis of papillary thyroid carcinoma. J Clin Endocrinol Metab. 2004;89:5175–5180. doi: 10.1210/jc.2003-032221. [DOI] [PubMed] [Google Scholar]
- Cohen Y, Rosenbaum E, Clark DP, Zeiger MA, Umbricht CB, Tufano RP, Sidransky D, Westra WH. Mutational analysis of BRAF in fine needle aspiration biopsies of the thyroid: a potential application for the preoperative assessment of thyroid nodules. Clin Cancer Res. 2004;10:2761–2765. doi: 10.1158/1078-0432.CCR-03-0273. [DOI] [PubMed] [Google Scholar]
- Jin L, Sebo TJ, Nakamura N, Qian X, Oliveira A, Majerus JA, Johnson MR, Lloyd RV. BRAF mutation analysis in fine needle aspiration (FNA) cytology of the thyroid. Diagn Mol Pathol. 2006;15:136–143. doi: 10.1097/01.pdm.0000213461.53021.84. [DOI] [PubMed] [Google Scholar]
- Pichler M, Balic M, Stadelmeyer E, Ausch C, Wild M, Guelly C, Bauernhofer T, Samonigg H, Hoefler G, Dandachi N. Evaluation of high-resolution melting analysis as a diagnostic tool to detect the BRAF V600E mutation in colorectal tumors. J Mol Diagn. 2009;11:140–147. doi: 10.2353/jmoldx.2009.080100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jarry A, Masson D, Cassagnau E, Parois S, Laboisse C, Denis MG. Real-time allele-specific amplification for sensitive detection of the BRAF mutation V600E. Mol Cell Probes. 2004;18:349–352. doi: 10.1016/j.mcp.2004.05.004. [DOI] [PubMed] [Google Scholar]
- Pinzani P, Santucci C, Mancini I, Simi L, Salvianti F, Pratesi N, Massi D, De Giorgi V, Pazzagli M, Orlando C. BRAFV600E detection in melanoma is highly improved by COLD-PCR. Clin Chim Acta. 2011;412:901–905. doi: 10.1016/j.cca.2011.01.014. [DOI] [PubMed] [Google Scholar]
- Monzon FA, Ogino S, Hammond ME, Halling KC, Bloom KJ, Nikiforova MN. The role of KRAS mutation testing in the management of patients with metastatic colorectal cancer. Arch Pathol Lab Med. 2009;133:1600–1606. doi: 10.5858/133.10.1600. [DOI] [PubMed] [Google Scholar]
- Chen Q, Lu P, Jones AV, Cross NC, Silver RT, Wang YL. Amplification refractory mutation system, a highly sensitive and simple polymerase chain reaction assay, for the detection of JAK2 V617F mutation in chronic myeloproliferative disorders. J Mol Diagn. 2007;9:272–276. doi: 10.2353/jmoldx.2007.060133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melanie JP, Mary Frances M. The V617F JAK2 mutation and the myeloproliferative disorders. Hematol Oncol. 2005;23:91–93. doi: 10.1002/hon.761. [DOI] [PubMed] [Google Scholar]
- Ellison G, Donald E, McWalter G, Knight L, Fletcher L, Sherwood J, Cantarini M, Orr M, Speake G. A comparison of ARMS and DNA sequencing for mutation analysis in clinical biopsy samples. J Exp Clin Cancer Res. 2010;29:132. doi: 10.1186/1756-9966-29-132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan YH, Liu Y, Eu KW, Ang PW, Li WQ, Salto-Tellez M, Iacopetta B, Soong R. Detection of BRAF V600E mutation by pyrosequencing. Pathology. 2008;40:295–298. doi: 10.1080/00313020801911512. [DOI] [PubMed] [Google Scholar]
- Lee ST, Kim SW, Ki CS, Jang JH, Shin JH, Oh YL, Kim JW, Chung JH. Clinical implication of highly sensitive detection of the BRAF V600E mutation in fine-needle aspirations of thyroid nodules: a comparative analysis of three molecular assays in 4585 consecutive cases in a BRAF V600E mutation-prevalent area. J Clin Endocrinol Metab. 2012;97:2299–2306. doi: 10.1210/jc.2011-3135. [DOI] [PubMed] [Google Scholar]
- Kimura ET, Nikiforova MN, Zhu Z, Knauf JA, Nikiforov YE, Fagin JA. High prevalence of BRAF mutations in thyroid cancer: genetic evidence for constitutive activation of the RET/PTC-RAS-BRAF signaling pathway in papillary thyroid carcinoma. Cancer Res. 2003;63:1454–1457. [PubMed] [Google Scholar]
- Xu X, Quiros RM, Gattuso P, Ain KB, Prinz RA. High prevalence of BRAF gene mutation in papillary thyroid carcinomas and thyroid tumor cell lines. Cancer Res. 2003;63:4561–4567. [PubMed] [Google Scholar]
- Cohen Y, Xing M, Mambo E, Guo Z, Wu G, Trink B, Beller U, Westra WH, Ladenson PW, Sidransky D. BRAF mutation in papillary thyroid carcinoma. J Natl Cancer Inst. 2003;95:625–627. doi: 10.1093/jnci/95.8.625. [DOI] [PubMed] [Google Scholar]
- Trovisco V, de Castro Vieira I, Soares P, Maximo V, Silva P, Magalhaes J, Abrosimov A, Guiu XM, Sobrinho-Simoes M. BRAF mutations are associated with some histological types of papillary thyroid carcinoma. J Pathol. 2004;202:247–251. doi: 10.1002/path.1511. [DOI] [PubMed] [Google Scholar]
- Kim KH, Kang DW, Kim SH, Seong IO, Kang DY. Mutations of the BRAF gene in papillary thyroid carcinoma in a Korean population. Yonsei Med J. 2004;45:818–821. doi: 10.3349/ymj.2004.45.5.818. [DOI] [PubMed] [Google Scholar]
- Domingo E, Laiho P, Ollikainen M, Pinto M, Wang L, French AJ, Westra J, Frebourg T, Espin E, Armengol M. et al. BRAF screening as a low-cost effective strategy for simplifying HNPCC genetic testing. J Med Genet. 2004;41:664–668. doi: 10.1136/jmg.2004.020651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Domingo E, Niessen RC, Oliveira C, Alhopuro P, Moutinho C, Espin E, Armengol M, Sijmons RH, Kleibeuker JH, Seruca R. et al. BRAF-V600E is not involved in the colorectal tumorigenesis of HNPCC in patients with functional MLH1 and MSH2 genes. Oncogene. 2005;24:3995–3998. doi: 10.1038/sj.onc.1208569. [DOI] [PubMed] [Google Scholar]
- Tsai J, Lee JT, Wang W, Zhang J, Cho H, Mamo S, Bremer R, Gillette S, Kong J, Haass NK. et al. Discovery of a selective inhibitor of oncogenic B-Raf kinase with potent antimelanoma activity. Proc Natl Acad Sci U S A. 2008;105:3041–3046. doi: 10.1073/pnas.0711741105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nikiforova MN, Nikiforov YE. Molecular genetics of thyroid cancer: implications for diagnosis, treatment and prognosis. Expert Rev Mol Diagn. 2008;8:83–95. doi: 10.1586/14737159.8.1.83. [DOI] [PubMed] [Google Scholar]
- Benoit NE, Goldenberg D, Deng SX, Rosenbaum E, Cohen Y, Califano JA, Shackelford WH, Wang XB, Sidransky D. Colorimetric approach to high-throughput mutation analysis. Biotechniques. 2005;38:635–639. doi: 10.2144/05384PF01. [DOI] [PubMed] [Google Scholar]
- Lang AH, Drexel H, Geller-Rhomberg S, Stark N, Winder T, Geiger K, Muendlein A. Optimized allele-specific real-time PCR assays for the detection of common mutations in KRAS and BRAF. J Mol Diagn. 2011;13:23–28. doi: 10.1016/j.jmoldx.2010.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morandi L, de Biase D, Visani M, Cesari V, De Maglio G, Pizzolitto S, Pession A, Tallini G. Allele Specific Locked Nucleic Acid Quantitative PCR (ASLNAqPCR): an accurate and cost-effective assay to diagnose and quantify KRAS and BRAF mutation. PLoS One. 2012;7:e36084. doi: 10.1371/journal.pone.0036084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee ST, Kim SW, Ki CS, Jang JH, Shin JH, Oh YL, Kim JW, Chung JH. Clinical implication of highly sensitive detection of the BRAF V600E mutation in fine-needle aspirations of thyroid nodules: a comparative analysis of three molecular assays in 4585 consecutive cases in a BRAF V600E mutation-prevalent area. J Clin Endocrinol Metab. 2012;97:2299–2306. doi: 10.1210/jc.2011-3135. [DOI] [PubMed] [Google Scholar]