Abstract
Despite the availability of the National Comprehensive Cancer Network’s (NCCN) evidence-based practice guidelines for the assessment and management of Cancer-Related Fatigue (CRF), assessment of CRF still is not routinely performed at many institutions and oncology practice settings. Numerous patient-, provider-, and system-related barriers exist that hinder the translation of these guidelines into practice by oncology nurses and other health care providers. Oncology nurses can play vital roles in decreasing these barriers to promote the translation of these guidelines into practice to insure that CRF is routinely assessed, managed, and documented. This article briefly reviews the NCCN CRF guidelines for CRF assessment and discusses many of the common barriers that exist that hinder the translation of the CRF guidelines into practice settings. Current assessment/screening and measurement scales validated in patients with cancer are highlighted, and case studies reflect the vital roles that oncology nurses can play in the assessment, documentation, and ongoing monitoring of a patient with CRF.
Introduction
Despite the availability of the NCCN evidence-based practice guidelines for the assessment and management of CRF (Mock et al, 2007; Mock, Atkinson et al, 2007), assessment of CRF still is not performed routinely at many institutions and oncology practice settings (Knowles, Borthwick, McNamara, Miller, & Leggot, 2000). Numerous patient-, provider-, and system-related barriers exist that hinder the translation of these guidelines into practice settings by oncology nurses and other health care providers. Oncology nurses can play vital roles in decreasing these barriers to promote the translation of the guidelines into practice to insure that CRF is routinely assessed, managed, and documented. This article briefly reviews the NCCN CRF guidelines for CRF assessment and discusses many of the common barriers that hinder the translation of the guidelines into practice settings. Assessment/screening and measurement scales validated in patients with cancer are highlighted, and case studies reflect the vital roles that oncology nurses can play in the assessment, documentation, and ongoing monitoring of a patient with CRF.
Barriers to the Translation of CRF Guidelines Into Practice
Many barriers that hinder the translation of the NCCN guidelines into practice for the assessment and management of CRF are comparable to patient-, provider-, and system-related barriers experienced with assessing and managing cancer-related pain, as there are inherent similarities between these symptoms (NIH, 2002). Each of these barriers is discussed in the following sections.
Patient-Related Barriers. Patients with cancer often do not initiate discussion with their health care providers about their fatigue for several reasons. They may not want to bother busy health care providers, and they may not want to be perceived as complainers. They may only volunteer that they have fatigue when it becomes overwhelming or severely limits their functioning and activities of daily living (Borneman et al., 2007). Patients may fear that their medical treatment might be negatively affected if they report their fatigue to their provider. They may also fear that the fatigue they are experiencing may mean that their disease is not responding to treatment or that their disease is getting worse. They also may believe that their fatigue is an inevitable part of dealing with cancer and its therapies, and they just have to live with it because there is nothing that can be done to treat it (Borneman et al, 2007; Cella, Peterman, Passik, Jacobsen, & Breitbart, 1998; NIH, 2002).
Provider-Related Barriers. Health care providers may not be aware of just how severe or prevalent CRF is in their patients unless the patients bring up the subject themselves for discussion. Since CRF is not routinely assessed in many clinical settings (Knowles et al., 2000; Nail, 2002), it goes underreported, underdiagnosed, and undertreated (Mock et al., 2007). Health care providers may believe that CRF is not any different than usual tiredness (Payne, Piper, Rabinowitz, & Zimmerman, 2006; Wu & McSweeney, 2007) and that symptoms other than CRF, such as pain or nausea, may be more important to treat (Vogelzang, Breitbart, & Cella, 1997). They also may not be comfortable initiating a discussion about CRF themselves particularly if they are unaware that effective treatments exist to manage CRF or they do not know what may be causing fatigue or its underlying mechanisms (Borneman et al., 2007; NIH, 2002). Another barrier to the translation of the guidelines into practice settings may be the perception by providers that the guidelines are too complex and not feasible to implement in busy practice settings.
System-Related Barriers. In general, symptom assessment and the translation of evidence-based guidelines into practice are relatively recent phenomena in clinical practice. Pain is the only symptom currently assessed, documented, and managed in clinical settings, and this required a mandate from The Joint Commission. Documentation of CRF in the medical record is not a common practice and is not required by The Joint Commission as is pain. Thus, CRF assessment and management is currently not viewed as a practice priority despite its prevalence (Prue, Rankin, Allen, Gracey, & Cramp, 2006); providers are not routinely reminded to document its presence, severity, or management (Borneman et al., 2007); and as patients transition between clinical settings (ie, outpatient clinics, office practice, or inpatient settings), it is not consistently assessed or documented. Additional barriers include the time it may take to obtain a physician’s order for CRF supportive care referrals (ie, physical therapy, nutrition, psychological support) and the type of health care coverage and reimbursements available that affect prescription practices and referral patterns (Borneman et al, 2007).
As part of a 5-year prospective and ongoing clinical trial sponsored by the National Cancer Institute (NCI) designed to translate the NCCN CRF and pain guidelines into practice (R01-CA-115323) (ie, the Barriers study), many of the above-mentioned barriers were confirmed during phase 1 or the usual care phase of the study that was implemented prior to the study’s intervention phases. The most frequent patient-related barrier documented in the study was the patient’s belief the physician would ask about fatigue if it was important, followed the patient’s desire to play the “good patient” role (Borneman et al., 2007). The most common provider- and system-related barriers documented by medical record audits performed by the research team were the lack of guideline adherence indicators, CRF assessment documentation, and CRF supportive care referrals.
NCCN Guidelines for CRF Assessment
Current NCCN guidelines for CRF assessment state that CRF is a subjective experience that needs to be assessed systematically using the patient’s own self-report or perception of fatigue and other sources of information (Mock et al, 2007). Thus, patients need to be routinely and systematically asked by oncology nurses and health care providers to describe their fatigue in their own words using a standard format that is also documented in the medical record. Health care providers should not wait for patients to volunteer that they are fatigued, as waiting for patients to initiate the subject is a known barrier (Homsi et al, 2006), and may contribute to patients’ experiencing even more severe fatigue by the time they mention it.
Screening assessments by health care providers should include asking patients if they have fatigue (ie, its presence or absence) and, if present, asking patients to rate fatigue’s intensity by using a simple numeric rating scale (NRS) such as a 0 to 10 intensity scale (ie, 0 = no fatigue; 10 = worst fatigue you can imagine). Mild fatigue is indicated by a score of 1 to 3, moderate fatigue as 4 to 6, and severe fatigue as 7 to 10. If patients are unable to rate their fatigue using the 0–10 NRS, as noted occasionally with cancer pain assessment in some patients, the provider should ask patients to rate their fatigue by using the words “none, mild, moderate, or severe.” The provider should document which scale patients prefer, and use this same scale thereafter to screen/reassess patients’ fatigue. For children aged less than 6 to 7 years, the guidelines recommend using the words “tired” or “not tired.”
While there are a number of validated scales for patients with cancer that can be used to screen and measure CRF (see tables and discussion that follow), the NCCN guidelines recommend that adult patients be asked to rate their fatigue on a 0 to 10 scale over the past 7 days (Mock, Atkinson et al, 2007). While this method relies on patient recall and requires patients to average their fatigue intensity over the past 7 days, this recommendation was made by the NCCN expert panel members because patients may tend to underestimate their fatigue level during their actual visits. More research needs to be done to validate this recommendation.
If the initial screening assessment indicates that CRF is absent or at a mild level, the patient and family should receive education about fatigue and common strategies for its management. Inherent in this education is teaching the patient and family about common patient-related barriers, including the importance of reporting fatigue and not waiting for providers to bring up the subject. Health care providers need to teach patients and families to treat fatigue as if it were the “6th vital sign,” and to stress the importance of reporting fatigue so health care providers can monitor it! All patients should be screened for fatigue at their initial visit and be rescreened at appropriate intervals thereafter, including during and following active cancer treatment, in long-term follow-up, at end of life, and whenever clinically indicated.
For patients who have moderate to severe fatigue (4 to 10 on the severity scale), it is necessary to supplement education with further assessment and a workup that includes a more in-depth fatigue assessment (including onset, pattern, duration, change over time, aggravating or alleviating factors, and interference in functioning) and a more focused history and physical exam that considers the patients’ current disease and treatment status, medications, and a 2review of systems (Mock, Atkinson et al, 2007). This more in-depth fatigue assessment might also include the use of a more multidimensional CRF measurement scale validated in patients with cancer (see multidimensional measurement scales discussed in the following section). In addition, the guidelines recommend that assessment of 7 treatable contributing factors be performed (ie, pain, emotional distress [anxiety, depression], sleep disturbance, anemia, nutrition, activity level, and comorbidities). These 7 treatable contributing factors are informally referred to as “the Gang of 7.”
CRF Assessment/Screening Scales for Practice and Research
There are several excellent reviews that describe the various CRF assessment/screening scales for use in practice. These reviews also describe CRF measurement scales used in research studies that might also be used as part of a more in-depth workup should the patient have moderate to severe CRF on screening (ie, 4 or more on a 0 to 10 scale) (Ahlberg, Ekman, Gaston-Johansson, & Mock, 2003; Dittner, Wessely, & Brown, 2004; Hjollund, Andersen, & Bech, 2007; Jacobsen, 2004; Jean-Pierre et al, 2007; Mota & Pimenta, 2006; Piper, 2004; Prue et al, 2006; Wu & McSweeney, 2001).
Table 1 describes some CRF screening scales that can be used in practice settings. These include single-item numeric rating scales (NRS), Likert-type rating scales, and visual analog rating scales (VAS) (Waltz, Strickland, & Lenz, 2005). For the most part, these scales are somewhat similar in design in that they each measure a single dimension of CRF (ie, intensity), and vary in length from a 10-cm or 100-mm horizontal or vertical line (VAS) to a horizontal 0 to 5 or 0 to 10 Likert-type or NRS with word anchors on each end of the scale (ie, 0 = not at all to 10 = a great deal). Likert-type scales may sometimes include specific wording that accompanies each numeric value (ie, 1–5) that represents varying degrees of intensities. One 0–10 NRS item taken from a multidimensional CRF scale has been used as a measure of intensity in a series of studies when more frequent or daily CRF measurements are necessary (Berger, Farr, Kuhn, Fischer, & Agrawal, 2007).
Table 1.
Single-Item/Single Dimension Measures for CRF
| Scale/Instrument | Description | Practice Recommendations | References |
|---|---|---|---|
| NRS Examples: NCCN Intensity Scale(s) |
Item(s): 1 Dimension(s): Severity Scaling: How would you rate your fatigue on a scale of 0 to 10 over the past 7 days? Or: 0 = no fatigue to 10 = worst fatigue you can imagine Or: “none, mild, moderate, severe.” Or: “tired, not tired.” |
All NCCN intensity scales are recommended for practice and research screening. | (Mock, Atkinson et al., 2007; Piper, 2004; Stone, Richards, & Hardy, 1998) |
| The Fatigue Intensity Scale (FIS) | Item(s): 1 Dimension(s): Intensity Scaling: 0 = no fatigue to 10 = overwhelming fatigue; similar to NCCN’s screening scale. This face, content, and strong concurrent validity estimates with the Piper Fatigue Scale-Revised (PFS-R) and strong criterion validity estimates. (Borneman et al., 2007). |
Can be recommended for practice and research screening | (Borneman et al., 2007; Piper et al., 1999) |
| Rhoten Fatigue Scale | Item(s): 1 Dimension(s): Severity Scaling: 0 = not tired, full of energy to 10 = totally exhausted. |
Can be used for practice screening | (Blesch et al., 1991; Pickard-Holley, 1991; Rhoten, 1982; Winstead-Fry, 1998; Wu & McSweeney, 2001) |
| VAS Example: Visual Analog Scale for Fatigue (VAS-F) |
Item(s): 1 Dimension(s): Severity Scaling: 10-cm, 0–100-mm horizontal line 0 = “I do not feel tired.” 100 = “I feel totally exhausted.” |
Can be recommended for practice and research screening; has been primarily tested in patients with cancer in Switzerland and Germany. As with any VAS, may have measurement characteristics that need to be considered when using it in practice or research settings (Waltz et al., 2005). | (A. Glaus, 1993; Sutherland, Lockwood, & Cunningham, 1989) |
| Likert-Type Scale(s) Example: NCI Common Terminology Criteria for Adverse Events V.3.0 (CTCAE) |
Item(s): 1 Dimension(s): Severity Scaling: 1–5 Likert scale (1 = mild fatigue over baseline; 2 = moderate or causing difficulty performing some activities of daily living (ADL); 3 = severe fatigue interfering with ADL; 4 = disabling; 5 = death) |
Not recommended for practice until further studies establish its use as a reliable and valid “patient-reported outcome” measure | (Basch et al., 2006; Huschka & Burger, 2006) |
Additional CRF Measurement Scales for Practice and Research
Additional CRF scales have been validated in patients with cancer in research studies but have not been evaluated in practice settings. These scales, as previously suggested, might be used as part of a more in-depth assessment of fatigue once a single-item screening intensity scale indicates moderate to severe fatigue exists. Table 2 describes the multi-item, single-dimension intensity measures available to measure CRF. Each of these scales has been validated in patients with cancer but has not been tested in practice settings. Additional studies are needed to evaluate whether these multi-item, single-dimension scales provide additional data useful for treatment workup and planning over and above the single-item intensity rating scales described in Table 1.
Table 2.
Multi-Item, Single Dimension Measures for CRF
| Scale/Instrument | Description | Advantages/Disadvantages | References |
|---|---|---|---|
| Brief Fatigue Inventory (BFI) | Item(s): 9 Dimension(s): Intensity/Severity Scaling: 0–10 NRS (0 = no fatigue, 10 = as bad as you can imagine); includes fatigue now, usual and worst fatigue during the past 24 hours, and interference in general activity, mood, walking ability, normal work, relations with others, and enjoyment of life; averaging 9 items gives a global fatigue severity score; cut scores for mild (1–3), moderate (4–6), and severe (7–10) established, although strongest evidence is for the severe cut score, similar to what others have reported (Piper et al., 1999) |
Has not been tested in practice settings but is widely used in national and international clinical trials | (Jean-Pierre et al., 2007; Mendoza et al., 1999) |
| Cancer-Related Fatigue Distress Scale | Item(s): 20 Dimension(s): Distress Scaling: Each item rated on a 0–10 Likert scale, during the past 7 days/week |
Validated in cancer survivors; written at a third grade reading level | (Holley, 2000a; Holley, 2000b; Piper, 2004) |
| Pearson-Byars Fatigue Feeling Tone Checklist | Item(s): 10 Dimension(s): Intensity/Severity Scaling: Adjective Rating Scale. Subjects rate whether they feel the “same as”, “worse than,” or “better than.” Adjectives are rated on a 1 = “very peppy” to 10 = “ready to drop” scale; numbers are summed to give a total fatigue score (8–20). |
Colloquial wording; patients may have difficulty knowing how to respond to certain items; developed originally for use in pilot studies; 3-point rating scale may not provide enough variability to assess different levels of fatigue. | (Graydon, Bubela, Irvine, & Vincent, 1995; Irvine, Vincent, Graydon, Bubela, & Thompson, 1994; Pearson & Byars, 1956; Piper, 2004) |
| Wu Cancer Fatigue Scale | Item(s): 9 Dimension(s): Intensity/severity Scaling: 1–5, 1 = not at all, 5 = very much. |
Easy to use and score in practice and research settings | (Wu & McSweeney, 2004; Wu, Wyrwich, & McSweeney, 2006) |
Table 3 describes the multidimensional CRF measures that have been validated in patients with cancer but have not yet been tested in practice settings. These scales might be used as part of a more in-depth assessment of fatigue once a single-item intensity screening scale indicates that moderate to severe fatigue exists. Additional studies are needed to evaluate whether these multidimensional CRF scales provide additional data useful for treatment workup and planning over and above the single-item or multi-item single-dimension rating scales described in Tables 1 and 2.
Table 3.
Multidimensional CRF Measures
| Scale/Instrument | Description | Advantages/Disadvantages | References |
|---|---|---|---|
| Cancer Fatigue Scale | Item(s): 15 Dimension(s): Physical, cognitive, affective Scaling: 5 point Likert (1 = not at all) to 5 (very much); scores range 0–28 (physical), 0–16 (affective), 0–16 (cognitive); maximum score 60 |
Simple and easily completed in 2 minutes, even by patients with advanced cancer; primarily tested in patients with cancer in Japan; needs cross-cultural validation (Wu & McSweeney, 2001). | (Okuyama, Akechi, Kugaya, Okamura, Shima et al., 2000; Piper, 2004) |
| Chalder Fatigue Questionnaire | Item(s): 11 Dimension(s): Physical and mental fatigue Scaling: 0–3 Likert, 0 = less than usual, 1 = same as usual, 3 = much more than usual; scores are summed to give a total fatigue score. |
Easy to administer and score | (Chalder et al., 1993; Knobel et al., 2003; Piper, 2004) |
| Fatigue Assessment Questionnaire | Item(s): 20 Dimension(s): Physical, affective, cognitive Scaling: 4-point rating scale that measures intensity and distress during the past week and previous month; 0 = not at all, 1 = hardly, 2 = moderately, 3 = strongly; plus 3 additional VASs that measure amount of fatigue and distress |
Developed to measure fatigue in patients with cancer; has thus far been tested primarily in Switzerland and in Germany in patients with cancer. Needs further testing in other cultures/countries | (Beutel, Hinz, Albani, & Brahler, 2006; A. Glaus, Crowe, & Bohme, 1996; A. Glaus & Muller, 2001; Piper, 2004) |
| Fatigue Symptom Checklist | Item(s): 30 Dimension(s): Decreased motivation/mental fatigue, general fatigue, feelings of incongruity Scaling: Dichotomous (ie, yes/no), 1–5 Likert and 0–10 NRS. |
Originally developed in healthy industrial workers in Japan; has been tested in patients with cancer | (Haylock & Hart, 1979; Piper, 2004; Saito, Kogi, & Kashiwagi, 1970; Yoshitake, 1971; Yoshitake, 1978) |
| Lee Fatigue Scale (Formerly referred to as the Visual Analog Scale for Fatigue [VAS-F]) | Item(s): 13 Dimension(s): Energy, fatigue Scaling: Fatigue scale is rated on a 0–10 NRS; has “cut” scores established |
Easy to use and score | (Breitbart, Rosenfeld, Kaim, & Funesti-Esch, 2001; Lee, Hicks, & Nino-Murcia, 1991; Meek et al., 2000; Okuyama, Akechi, Kugaya, Okamura, Imoto et al., 2000; Okuyama, Akechi, Kugaya, Okamura, Shima et al., 2000; Winstead-Fry, 1998) |
| Multidimensional Assessment of Fatigue (MAF)/Global Fatigue Index (GFI) | Item(s): 16 (MAF); 15 (GFI) Dimension(s): Supposedly measures 4 dimensions of fatigue (intensity, distress, interference in ADLs, and timing); Scaling: 0–10; frequency item is 0–4. |
Developed from the original version of the Piper Fatigue Scale; dimensions/factor structure may not be stable across studies (ie, validity issue) | (Meek et al., 2000; Piper, 2004) |
| Multidimensional Fatigue Inventory (MFI-20) | Item(s): 20 Dimension(s): general, physical, mental, reduced activity, reduced motivation Scaling: 5-point Likert; no summated grand fatigue total score |
Tested in patients with cancer receiving radiation therapy; needs to be tested in other populations of patients with cancer (Ahlberg et al., 2003). | (Jean-Pierre et al., 2007; Piper, 2004; Schneider, 1998a; Schneider, 1998b; Smets, Garssen, Bonke, & De Haes, 1995; Smets, Garssen, Cull, & de Haes, 1996; Smets et al., 1998; Stone et al., 1998) |
| Fatigue Symptom Inventory (FSI) | Item(s): 14 Dimension(s): Severity, frequency, diurnal variation, perceived interference with quality of life Scaling: 5-point scale in the past week (severity, 4 items: most, least, average, and current in the past week; frequency, 2 items; diurnal variation, 1 item; interference, 7 items) |
Validated in patients with cancer. | (Hann et al., 1998; Hann, Winter, & Jacobsen, 1999; Hann, Denniston, & Baker, 2000; Jacobsen, 2004; Stein, Martin, Hann, & Jacobsen, 1998) |
| Revised Piper Fatigue Scale (PFS-R) | Item(s): 22 (plus 5 additional open-ended items related to the temporal dimension of fatigue, perceived cause, effect, relief, and additional symptoms not included in the scoring) Dimension(s): Behavioral/severity, affective meaning, sensory, cognitive/mood Scaling: 0–10 NRS; Total and subscale mean scores are derived by summing the individual items and dividing by the number of items in the subscale/total scale to maintain the 0–10 scaling; “cut” scores are comparable to what others have reported (Mendoza et al., 1999; Yoshitake, 1969) with stronger evidence for the severe category (ie, 0 = none, 1–3 = mild, 4–6 = moderate, 7–10 = severe); this takes 2–5 minutes to complete (Berger et al., 2002). Behavioral severity subscale has been used as a fatigue interference scale (de Jong, Candel, Schouten, Abu-Saad, & Courtens, 2006). |
One of the most well-developed and widely used CRF multidimensional scales (Wu & McSweeney, 2001); developed initially to measure fatigue in patients with cancer, now also used in other clinical, nonclinical, and healthy groups nationally and internationally | (Berger et al., 2007; Mock et al., 2005; Ostlund, Gustavsson, & Furst, 2007; Piper et al., 1998) |
| Revised Schwartz Cancer Fatigue Scale (SCFS-6) | Item(s): 6 Dimension(s): Physical, perceptual, Scaling: 5-point NRS, 1 = not at all, 5 = extremely past 2 to 3 days |
Easy to use and score | (Schwartz, 1998; Schwartz & Meek, 1999; Wu et al., 2006) |
Table 4 describes some of the single-item and multi-item measures of CRF that are embedded in other scales that for the most part measure various symptoms, mood states, and/or quality of life. These scales are not meant to be an exhaustive review of these types of measures. They are included here as exemplars only in this era of symptom management and symptom cluster research (Barsevick, 2007), where patients may frequently present with more than one symptom that may need to be assessed, treated, and documented in the medical or electronic medical record. More studies are needed to determine their suitability for use in practice settings.
Table 4.
Single-Item/Multi-Item CRF Measures Embedded Other Scales
| Scale/Instrument | Description | Advantages/Disadvantages | References |
|---|---|---|---|
| Profile of Mood States-Fatigue-Inertia (POMS-F) and Vigor (POMS-V) Subscales | Item(s): POMS-F: 5–7 POMS-V: 8-item Dimension(s): Intensity Scaling: 5-point Likert, during the past week |
Both scales include colloquialisms (ie, bushed, worn out, full of pep); may have floor and ceiling effects (ie, may not be sensitive to the wide range of scores that patients may want to indicate (Buchanan et al., 2007) | (McNair, Lorr, & Droppleman, 1992; Meek et al., 2000; Mendoza et al., 1999) |
| Rotterdam Symptom Checklist | Item(s): 1 Dimension(s): Distress Scaling: 4-point rating scale (ie, have you been bothered by tiredness, lack of energy, difficulty sleeping; not at all, a little, quite a bit, very much) |
Simple to administer and score | (de Haes, van Knippenborg, & Neijt, 1990; Jean-Pierre et al., 2007) |
| Symptom Distress Scale | Item(s): 1 Dimension(s): Distress Scaling: 1–5 Likert. |
Simple to administer and score | (Boehmke, 2004; Jean-Pierre et al., 2007; McCorkle & Quint-Benoliel, 1983) |
| European Organization for Research and Treatment of Cancer 30-Item Quality of Life Scale (EORTC-QLQ-C30) Fatigue Subscale | Item(s): 3 Dimension(s): Intensity Scaling: 1–4 Likert (during the past week, did you need to rest, have you felt weak, were you tired; 1 = not at all, 2 = a little, 3 = quite a bit, 4 = very much) |
Used in many clinical trials particularly in Europe; scores need to be converted to 0 to 100 scaling | (Aaronson et al., 1993; Jean-Pierre et al., 2007; Stone et al., 1998) |
| Functional Assessment of Cancer Therapy Fatigue Subscale (FACT-F) | Item(s): 13 Dimension(s): Intensity/severity Scaling: 0–4 Likert |
Used in many clinical trials nationally and internationally; higher scores denote less fatigue | (Cella & Webster, 1997; Stone et al., 1998; Yellen, Cella, Webster, Blendowski, & Kaplan, 1997) |
| Medical Outcome Study Short Form-36 (MOS-36) Vitality Subscale | Item(s): 4 Dimension(s): Vitality Scaling: Over the past 4 weeks, Did you feel full of pep? Did you have a lot of energy? Did you feel worn out? Did you feel tired? Vitality scores above 50 = well-being; scores below 50 = fatigue. |
Includes colloquialisms (ie, full of pep, worn out); scores need to be converted to 0–100 scaling. | (Jean-Pierre et al., 2007; McHorney, Ware, & Raczek, 1993; Piper, 2004) |
| Fatigue Severity Scale | Item(s): 9 Dimension(s): Fatigue impact Scaling: 7-point Likert (1 = strongly disagree, to 7 = strongly agree) |
Simple to administer and score | (Krupp, LaRocca, Muir-Nash, & Steinberg, 1989; Stone, Richards, A’Hern, & Hardy, 2001) |
| MD Anderson Symptom Inventory (MDASI) | Item(s): 1 Dimension(s): Intensity, interference Scaling: 0 = not present, to 10 = as bad as you can imagine your fatigue (tiredness) at its worst during the past 24 hours |
Simple to administer and score | (Cleeland et al., 2000; Cleeland, 2000; Jean-Pierre et al., 2007) |
| Edmonton Symptom Assessment System (ESAS) | Item(s): 1 Dimension(s): Intensity Scaling: 0–10, 0 = no fatigue; 10 = worst fatigue |
Simple to administer; developed to measure fatigue in palliative care, end-of-life patients | (Bruera et al., 2007; Chow, Davis, Holden, Tsao, & Danjoux, 2005; Reddy, Bruera, Pace, Zhang, & Reyes-Gibby, 2007) |
| Linear Analogue Self-Assessment Scale (LASA) | Item(s): 1 Dimension(s): Severity Scaling: “Your level of fatigue on the average” 0–10 |
Simple to administer; like any VAS, may have certain measurement characteristics that need to be considered if used in practice/research settings (Waltz et al., 2005) | (Locke et al., 2007) |
In the research literature, much discussion has ensued as to whether CRF is a symptom or a syndrome (a complex of signs and symptoms that indicate an abnormal condition or a disease state (Cella et al., 1998). Currently there is a lack of clearly defined criteria for identifying a CRF “case” diagnostically for research, disability, and reimbursement purposes. Based on 2 national telephone surveys, (Curt et al., 2000; Vogelzang et al., 1997) a group of fatigue scientists put forth criteria for the syndrome of CRF to be included in the International Classification of Diseases (ICD-10). These criteria, displayed in Table 5, are undergoing further research and debate to determine their validity and applicability clinically.
Role of the Oncology Nurse in Translating the Guidelines
Oncology nurses can play vital roles in translating the NCCN fatigue assessment guidelines into practice. One role can include identifying and breaking down the patient-related barriers that may exist. For example, in addition to the NCCN recommendations for patient education inherent in the guidelines, the oncology nurse, by being aware of the patient-related barriers that exist, can “coach” the patients and family members about the importance of bringing up fatigue for discussion with their health care providers without waiting for the provider to bring up the subject. Teaching patients about the importance of viewing CRF as the “6th vital sign” can emphasize this symptom’s importance and significance.
Another role that the oncology nurse can play is recognizing the many provider- and system-related barriers that exist and working with others in a systematic and collaborative fashion within the system to decrease these barriers and begin to incorporate a simple intensity scale for CRF assessment/screening, documentation, and ongoing monitoring. Quick and Fonteyn describe the process that they undertook in an attempt to improve CRF assessment in their clinical setting (2005). In deciding which scale(s) to use in a specific practice setting, they consider whether the setting is inpatient or outpatient as that may dictate the frequency of the screening and the type of scale used. Clearly, patient burden regardless of setting must be a consideration as the measure should not be burdensome and cause fatigue in and of itself. Lastly, patient characteristics such as age, educational level, and sensory or motor limitations as well as scale characteristics (Waltz et al, 2005) might affect which scale is most appropriate to use for screening.
When a patient has moderate to severe fatigue and further workup is necessary, the oncology nurse can work collaboratively with other multidisciplinary health care team members, such as advance practice nurses, physicians, cancer care committees, and others, to insure the use of NCCN recommendations for workup and referrals. When assessing this type of patient, it is important to do a more in-depth fatigue assessment that includes eliciting from the patient/family member more information about fatigue’s onset, pattern, duration, change over time, and perceived aggravating and alleviating factors and the perceived effect of fatigue on functioning. The oncology nurse must remember to assess the “Gang of 7 (ie, pain, emotional distress [anxiety, depression], sleep disturbance, anemia, nutrition, activity level, and comorbidities) that may affect workup, treatment, and supportive care referrals. The following case studies are provided as exemplars to enhance the skills of oncology nurses in translating the CRF guidelines into practice for their patients.
Case Studies
Fatigue Case Study #1
Carol was seen in your practice setting in October 2006. A review of her history reveals that: She is a 35-year-old African American woman with stage IV bilateral adenocarcinoma of the breast metastatic to the lung that she discovered through self-detection in December 2005. Her tumor is ER/PR negative, HER-2/neu positive. In February 2006, she underwent a modified radical mastectomy of the right breast and a skin-sparing mastectomy of the left breast. Postsurgical pathology revealed only one node was involved. Her tumor was staged as T3N1. She was referred to medical oncology for adjuvant therapy. A CT scan done prior to the start of her chemotherapy initiation found a pulmonary nodule. In March 2006, Carol began adjuvant treatment with doxorubicin hydrochloride/cyclophosphamide (Adriamycin/Cytoxan) X 4 cycles. In June 2006, her regimen was changed to docetaxel/trastuzumab (Taxotere/Herceptin) X 12 cycles. After 1 cycle, Carol developed a neutropenic fever and was hospitalized for 14 days. In August 2006, she again was hospitalized for C. difficile infection with diarrhea and bloody stool. In October 2006, she was referred to the Barriers study and agreed to participate. At that time, she had received 6 cycles of docetaxel/trastuzumab.
Question #1: From a fatigue standpoint, what do you want to assess first?
Assess for the presence of comorbidities
Assess for the presence of anemia
Determine if she has any pain, insomnia, nutritional problems, or emotional distress
Determine her current activity pattern
Ask Carol about her level of fatigue severity
The preferred response is e. Ask Carol about her level of fatigue severity. Your first priority should be to screen for the presence of fatigue and determine its severity on a 0–10 NRS. Carol rates her fatigue as a “7” on a 0 to 10 scale.
Question #2: How do you now proceed in your fatigue assessment? Since Carol has severe fatigue, conduct a more in-depth fatigue assessment (onset, pattern, duration, change over time, associated and alleviating factors, and interference with functioning) and assess for any patient-related fatigue barriers she may have. She tells you that she wasn’t aware that fatigue could be managed. She thought that she just had to learn to live with it as it is an expected side effect of her treatment. You then perform a focused history and physical examination, consider her disease and treatment status (ie, stage IV, on active treatment), perform a review of systems, and assess the “Gang of 7” (ie, pain, emotional distress [anxiety, depression], sleep disturbance, anemia, nutrition, activity level, and comorbidities). Your workup, history, physical, and review of systems is negative except for her lack of physical activity and taking 3 to 4 naps per day that last 2 to 3 hours. You counsel her to begin to use walking as a form of exercise and counsel her to keep her naps short in order to maintain nighttime sleep patterns. After talking with Carol and her physician, supportive care referrals for occupational therapy (OT) and physical therapy (PT) are initiated as is an order for an as-needed sleep medication. In October 2006, Carol underwent a wedge resection to remove her pulmonary nodule, and later in November, she was started on trastuzumab every 3 weeks for a year. Her fatigue levels gradually decreased from 7 to 6 to 4 over the course of 3 months, as she continued her walking program.
Fatigue Case Study #2
Lydia is an 83 year old Filipino woman originally diagnosed with stage I left breast cancer in 2004 that was discovered through routine mammography. In October 2004, she underwent a left breast segmentectomy. The tumor was ER/PR positive, HER 2/neu negative, so her postsurgical staging was T1aN0. She was started on letrozole (Femora) for the breast cancer. In October 2006, she was referred to your practice and to the Barriers study and agreed to participate. At that time, she had been on letrozole for 2 years.
Question #1: From a fatigue standpoint, what do you want to assess first?
Assess for the presence of comorbidities
Assess for the presence of anemia
Determine if she has any pain, insomnia, nutritional problems, or emotional distress
Determine her current activity pattern
Ask Lydia about her level of fatigue severity
The preferred response is e. Ask Lydia about her level of fatigue severity. Lydia rates her fatigue at a “5” level on a 0 to 10 scale. Since Lydia has moderate fatigue, conduct an in-depth fatigue assessment (onset, pattern, duration, change over time, associated and alleviating factors, and interference with functioning), perform a focused history, physical examination, review systems and medications, and consider her disease and treatment status (ie, stage IV, on letrozole). You assess the “Gang of 7” (ie, pain, emotional distress [anxiety, depression], sleep disturbance, anemia, nutrition, activity level, and comorbidities). Based on your workup, you discover that she has several comorbidities (gastroesophageal reflux disease, osteoarthritis, hypertension, congestive heart failure, and hypothyroidism). For these she is on pantoprazole (Protonix), amlodipine (Norvasc), candesartan (Atacand), and levothyroxine (Synthroid). She states that she is pretty much confined to a wheelchair because of her arthritis and is taking celecoxib (Celebrex) for related pain. She has no evidence of anemia, insomnia, or nutritionally related problems. In assessing patient-related barriers, Lydia tells you that she has limited physical activity secondary to her arthritis (wheelchair bound). Her primary caregiver is her daughter who is a nurse. The daughter tells you that she encourages her mother to get out of the wheelchair, as her mother doesn’t need it all the time, but it is challenging to get her mother to do this. Her mother is very attached to her wheelchair and would rather not aggravate her osteoarthritis by moving around. A consultation and referral was initiated to Lydia’s outside physician regarding her arthritis treatment, inactivity, and pain management. A recommendation to consider a supportive care referral for OT and PT was included to determine appropriate wheelchair exercises that could be performed. Follow-up at 1 and 3 months thereafter revealed that her fatigue was remaining stable at a moderate level (ie, 4 to 5 on a 0 to 10 scale).
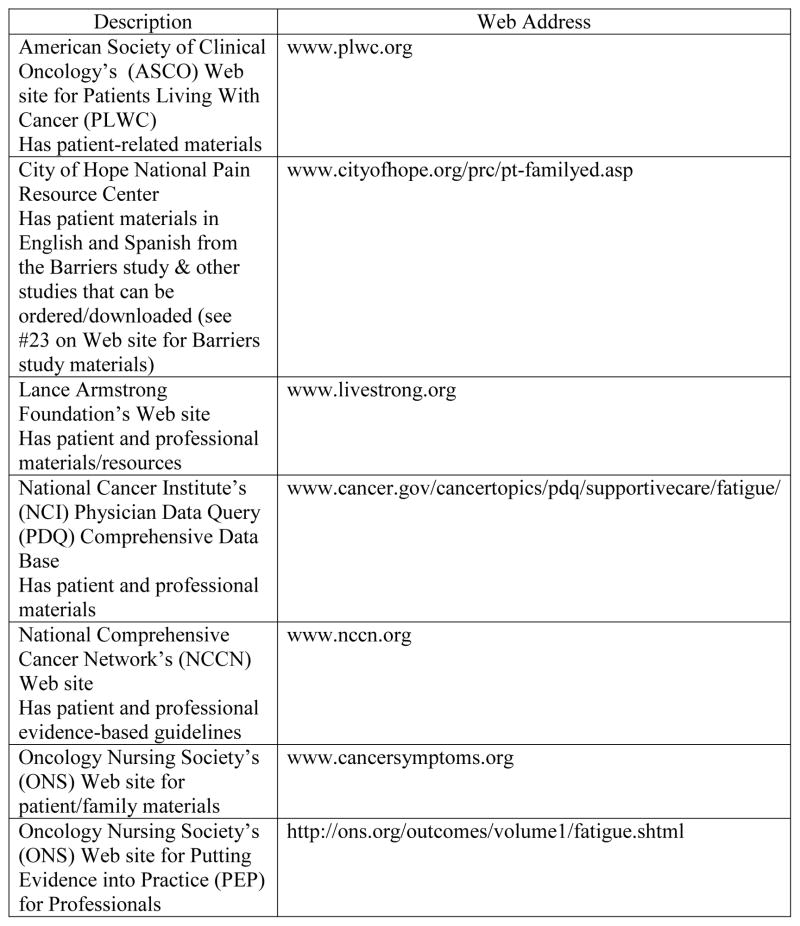
In summary, there are a number of CRF Web sites that oncology nurses can access for educational materials about CRF for themselves and their patients (see Table 6).
By using these and other resources, oncology nurses can play significant roles in the translation of the CRF NCCN’s evidence-based practice guidelines into their practice settings. The important thing to remember is “just do it!”
Figure 1.
Proposed International Classification of Diseases-10 (ICD-10) Criteria for Cancer-Related Fatigue
Figure 2.
Cancer-Related Fatigue Web Sites for Patients and Oncology Nurses
At a Glance.
NCCN CRF guidelines for the assessment of CRF are reviewed.
Barriers to the translation of the NCCN CRF guidelines into practice are discussed as are the roles oncology nurses play in decreasing these barriers.
CRF assessment/screening and measurement scales are highlighted, and case studies underscore the roles oncology nurses play in the assessment and management of CRF.
Footnotes
Disclosure Statements
Dr. Piper has been a scientific consultant for Cephalon, Inc. She will receive an honorarium for writing this article as part of the Cancer-Related Fatigue supplement. The other coauthors have nothing to financially disclose. No non–FDA-approved or investigational uses of any products/devices are mentioned in this article.
Authorship Contributions
Dr. Piper is the article’s corresponding author. She presented similar content at a Cephalon-sponsored symposium: Cancer-Related Fatigue: The 6th Vital Sign at the Oncology Nursing Society’s 32nd Annual Congress in Las Vegas, Nevada, April 24, 2007.
Ms. Borneman, Ms. Sun, and Drs. Koczywas, Piper, and Uman are co-investigators with Dr. Ferrell (principal investigator) on an NCI-funded study designed to decrease barriers to the management of pain and fatigue in patients with cancer (NCI R01-CA115323). Each has contributed to the writing of this manuscript, the collection and analysis of barrier data, and the case studies cited in this article.
Ms. James assisted in the literature review, table(s) development, obtaining permissions and reference citations, and reviewing and editing this manuscript.
Contributor Information
Barbara F. Piper, Email: bpiper@shc.org, bpiper@nursing.arizona.edu, Professor and Chair of Nursing Research, Scottsdale Healthcare/University of Arizonaa, 10460 N. 92nd Street, Suite 206, Scottsdale, AZ 85258, Phone: (480) 323-1243; Fax: (480) 323-1560.
Tami Borneman, Senior Research Specialist, Department of Nursing Research & Education, Division of Population Sciences, Beckman Research Institute, City of Hope National Medical Center, Duarte, California.
Virginia Chih-Yi Sun, Senior Research Specialist, Department of Nursing Research & Education, Division of Population Sciences, Beckman Research Institute, City of Hope National Medical Center, Duarte, California.
Marianna Koczywas, Department of Medical Oncology & Therapeutics Research, City of Hope National Medical Center, Duarte, California.
Gwen Uman, Senior & Founding Partner: Vital Research, LLC, Los Angeles, California.
Betty Ferrell, Research Scientist, Department of Nursing Research & Education, Division of Population Sciences, Beckman Research Institute, City of Hope National Medical Center, Duarte, California.
Raysenia L. James, Program Coordinator for Research, Scottsdale Healthcare/University of Arizona, 10460 N. 92nd Street, Suite 206, Scottsdale, AZ 85258.
References
- Aaronson NK, Ahmedzai S, Bergman B, Bullinger M, Cull A, Duez NJ, et al. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. Journal of the National Cancer Institute. 1993;85:365–376. doi: 10.1093/jnci/85.5.365. [DOI] [PubMed] [Google Scholar]
- Ahlberg K, Ekman T, Gaston-Johansson F, Mock V. Assessment and management of cancer-related fatigue in adults. Lancet. 2003;362:640–650. doi: 10.1016/S0140-6736(03)14186-4. [DOI] [PubMed] [Google Scholar]
- Barsevick AM. The elusive concept of the symptom cluster. Oncology Nursing Forum. 2007;34:971–980. doi: 10.1188/07.ONF.971-980. [DOI] [PubMed] [Google Scholar]
- Basch E, Iasonos A, McDonough T, Barz A, Culkin A, Kris MG, et al. Patient versus clinician symptom reporting using the National Cancer Institute Common Terminology Criteria for Adverse Events: results of a questionnaire-based study. The Lancet Oncology. 2006;7:903–909. doi: 10.1016/S1470-2045(06)70910-X. [DOI] [PubMed] [Google Scholar]
- Berger AM, Farr LA, Kuhn BR, Fischer P, Agrawal S. Values of sleep/wake, activity/rest, circadian rhythms, and fatigue prior to adjuvant breast cancer therapy. Journal of Pain and Symptom Management. 2007;33:398–409. doi: 10.1016/j.jpainsymman.2006.09.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berger AM, VonEssen S, Kuhn BR, Piper BF, Farr L, Agrawal S, et al. Feasibility of a sleep intervention during adjuvant breast cancer chemotherapy. Oncology Nursing Forum. 2002;29:1431–1441. doi: 10.1188/02.ONF.1431-1441. [DOI] [PubMed] [Google Scholar]
- Beutel ME, Hinz A, Albani C, Brahler E. Fatigue Assessment Questionnaire: Standardization of a cancer-specific instrument based on the general population. Oncology. 2006;70:351–357. doi: 10.1159/000098108. [DOI] [PubMed] [Google Scholar]
- Blesch KS, Paice JA, Wickham R, Harte N, Schnoor DK, Purl S, et al. Correlates of fatigue in people with breast or lung cancer. Oncology Nursing Forum. 1991;18:81–87. [PubMed] [Google Scholar]
- Boehmke MM. Measurement of symptom distress in women with early-stage breast cancer. Cancer Nursing. 2004;27:144–152. doi: 10.1097/00002820-200403000-00008. [DOI] [PubMed] [Google Scholar]
- Borneman T, Piper BF, Sun VC, Koczywas M, Uman G, Ferrell B. Implementing the fatigue guidelines at one NCCN member institution: Process and outcomes. Journal of the National Comprehensive Cancer Network : JNCCN. 2007;5:1092–1101. doi: 10.6004/jnccn.2007.0090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breitbart W, Rosenfeld B, Kaim M, Funesti-Esch J. A randomized, double-blind, placebo-controlled trial of psychostimulants for the treatment of fatigue in ambulatory patients with human immunodeficiency virus disease. Archives of Internal Medicine. 2001;161:411–420. doi: 10.1001/archinte.161.3.411. [DOI] [PubMed] [Google Scholar]
- Bruera E, El Osta B, Valero V, Driver LC, Pei BL, Shen L, et al. Donepezil for cancer fatigue: A double-blind, randomized, placebo-controlled trial. Journal of Clinical Oncology. 2007;25:3475–3481. doi: 10.1200/JCO.2007.10.9231. [DOI] [PubMed] [Google Scholar]
- Buchanan DR, O’Mara AM, Kelaghan JW, Sgambati M, McCaskill-Stevens W, Minasian L. Challenges and recommendations for advancing the state-of-the-science of quality of life assessment in symptom management trials. Cancer. 2007;110:1621–1628. doi: 10.1002/cncr.22893. [DOI] [PubMed] [Google Scholar]
- Cella D, Peterman A, Passik S, Jacobsen P, Breitbart W. Progress toward guidelines for the management of fatigue. Oncology (Williston Park) 1998;12:369–377. [PubMed] [Google Scholar]
- Cella D, Webster K. Linking outcomes management to quality-of-life measurement. Oncology (Williston Park) 1997;11:232–235. [PubMed] [Google Scholar]
- Chalder T, Berelowitz G, Pawlikowska T, Watts L, Wessely S, Wright D, et al. Development of a fatigue scale. Journal of Psychosomatic Research. 1993;37:147–153. doi: 10.1016/0022-3999(93)90081-p. [DOI] [PubMed] [Google Scholar]
- Chow E, Davis L, Holden L, Tsao M, Danjoux C. Prospective assessment of patient-rated symptoms following whole brain radiotherapy for brain metastases. Journal of Pain and Symptom Management. 2005;30:18–23. doi: 10.1016/j.jpainsymman.2005.02.009. [DOI] [PubMed] [Google Scholar]
- Cleeland CS. Cancer-related symptoms. Seminars in Radiation Oncology. 2000;10:175–190. doi: 10.1053/srao.2000.6590. [DOI] [PubMed] [Google Scholar]
- Cleeland CS, Mendoza TR, Wang XS, Chou C, Harle MT, Morrissey M, et al. Assessing symptom distress in cancer patients: The M.D. Anderson Symptom Inventory. Cancer. 2000;89:1634–1646. doi: 10.1002/1097-0142(20001001)89:7<1634::aid-cncr29>3.0.co;2-v. [DOI] [PubMed] [Google Scholar]
- Curt GA, Breitbart W, Cella D, Groopman JE, Horning SJ, Itri LM, et al. Impact of cancer-related fatigue on the lives of patients: new findings from the Fatigue Coalition. Oncologist. 2000;5:353–360. doi: 10.1634/theoncologist.5-5-353. [DOI] [PubMed] [Google Scholar]
- de Haes JC, van Knippenborg FC, Neijt JP. Measuring psychological and physical distress in cancer patients: Structure and application of the Rotterdam Symptom Checklist. British Journal of Cancer. 1990;62:1034–1038. doi: 10.1038/bjc.1990.434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Jong N, Candel MJ, Schouten HC, Abu-Saad HH, Courtens AM. Course of the fatigue dimension “activity level” and the interference of fatigue with daily living activities for patients with breast cancer receiving adjuvant chemotherapy. Cancer Nursing. 2006;29:E1–E13. doi: 10.1097/00002820-200609000-00013. [DOI] [PubMed] [Google Scholar]
- Dittner AJ, Wessely SC, Brown RG. The assessment of fatigue: a practical guide for clinicians and researchers. Journal of Psychosomatic Research. 2004;56:157–170. doi: 10.1016/S0022-3999(03)00371-4. [DOI] [PubMed] [Google Scholar]
- Glaus A, Crowe R, Bohme C. Development of a fatigue assessment questionnaire (FAQ) for cancer patients. Annals of Oncology. 1996;7(Suppl 5):134. [Google Scholar]
- Glaus A. Assessment of fatigue in cancer and non-cancer patients and in healthy individuals. Supportive Care in Cancer. 1993;1:305–315. doi: 10.1007/BF00364968. [DOI] [PubMed] [Google Scholar]
- Glaus A, Müller S. Measuring fatigue of cancer patients in the German-speaking region: development of the Fatigue Assessment Questionnaire. [Messung der Mudigkeit bei Krebskranken im Deutschen Sprachraum: Die Entwicklung des Fatigue Assessment Questionnaires]. Pflege. 2001;14:161–170. doi: 10.1024/1012-5302.14.3.161. [DOI] [PubMed] [Google Scholar]
- Graydon JE, Bubela N, Irvine D, Vincent L. Fatigue-reducing strategies used by patients receiving treatment for cancer. Cancer Nursing. 1995;18:23–28. [PubMed] [Google Scholar]
- Hann DM, Denniston MM, Baker F. Measurement of fatigue in cancer patients: Further validation of the Fatigue Symptom Inventory. Quality of Life Research. 2000;9:847–854. doi: 10.1023/a:1008900413113. [DOI] [PubMed] [Google Scholar]
- Hann DM, Jacobsen PB, Azzarello LM, Martin SC, Curran SL, Fields KK, et al. Measurement of fatigue in cancer patients: development and validation of the Fatigue Symptom Inventory. Quality of Life Research. 1998;7:301–310. doi: 10.1023/a:1024929829627. [DOI] [PubMed] [Google Scholar]
- Hann D, Winter K, Jacobsen P. Measurement of depressive symptoms in cancer patients: evaluation of the Center for Epidemiological Studies Depression Scale (CES-D) Journal of Psychosomatic Research. 1999;46:437–443. doi: 10.1016/s0022-3999(99)00004-5. [DOI] [PubMed] [Google Scholar]
- Haylock PJ, Hart LK. Fatigue in patients receiving localized radiation. Cancer Nursing. 1979;2:461–467. [PubMed] [Google Scholar]
- Hjollund NH, Andersen JH, Bech P. Assessment of fatigue in chronic disease: a bibliographic study of fatigue measurement scales. Health & Quality of Life Outcomes. 2007;5:12. doi: 10.1186/1477-7525-5-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holley S. Cancer-related fatigue: Suffering a different fatigue. Cancer Practice. 2000a;8:87–95. doi: 10.1046/j.1523-5394.2000.82007.x. [DOI] [PubMed] [Google Scholar]
- Holley SK. Evaluating patient distress from cancer-related fatigue: an instrument development study. Oncology Nursing Forum. 2000b;27:1425–1431. [PubMed] [Google Scholar]
- Homsi J, Walsh D, Rivera N, Rybicki LA, Nelson KA, Legrand SB, et al. Symptom evaluation in palliative medicine: patient report vs systematic assessment. Supportive Care in Cancer. 2006;14:444–453. doi: 10.1007/s00520-005-0009-2. [DOI] [PubMed] [Google Scholar]
- Huschka M, Burger K. Does QOL provide the same information as toxicity data? Current Problems in Cancer. 2006;30:244–252. doi: 10.1016/j.currproblcancer.2006.08.003. [DOI] [PubMed] [Google Scholar]
- Irvine D, Vincent L, Graydon JE, Bubela N, Thompson L. The prevalence and correlates of fatigue in patients receiving treatment with chemotherapy and radiotherapy. A comparison with the fatigue experienced by healthy individuals. Cancer Nursing. 1994;17:367–378. [PubMed] [Google Scholar]
- Jacobsen PB. Assessment of fatigue in cancer patients. Journal of the National Cancer Institute Monographs. 2004;32:93–97. doi: 10.1093/jncimonographs/lgh010. [DOI] [PubMed] [Google Scholar]
- Jean-Pierre P, Figueroa-Moseley CD, Kohli S, Fiscella K, Palesh OG, Morrow GR. Assessment of cancer-related fatigue: implications for clinical diagnosis and treatment. The Oncologist. 2007;12(Suppl 1):11–21. doi: 10.1634/theoncologist.12-S1-11. [DOI] [PubMed] [Google Scholar]
- Knobel H, Loge JH, Brenne E, Fayers P, Hjermstad MJ, Kaasa S. The validity of EORTC QLQ-C30 fatigue scale in advanced cancer patients and cancer survivors. Palliative Medicine. 2003;17:664–672. doi: 10.1191/0269216303pm841oa. [DOI] [PubMed] [Google Scholar]
- Knowles G, Borthwick D, McNamara S, Miller M, Leggot L. Survey of nurses’ assessment of cancer-related fatigue. European Journal of Cancer Care. 2000;9:105–113. doi: 10.1046/j.1365-2354.2000.00197.x. [DOI] [PubMed] [Google Scholar]
- Krupp LB, LaRocca NG, Muir-Nash J, Steinberg AD. The Fatigue Severity Scale. Application to patients with multiple sclerosis and systemic lupus erythematosus. Archives of Neurology. 1989;46:1121–1123. doi: 10.1001/archneur.1989.00520460115022. [DOI] [PubMed] [Google Scholar]
- Lee KA, Hicks G, Nino-Murcia G. Validity and reliability of a scale to assess fatigue. Psychiatry Research. 1991;36:291–298. doi: 10.1016/0165-1781(91)90027-m. [DOI] [PubMed] [Google Scholar]
- Locke DE, Decker PA, Sloan JA, Brown PD, Malec JF, Clark MM, et al. Validation of single-item linear analog scale assessment of quality of life in neuro-oncology patients. Journal of Pain and Symptom Management. 2007;34:628–638. doi: 10.1016/j.jpainsymman.2007.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCorkle R, Quint-Benoliel J. Symptom distress, current concerns and mood disturbance after diagnosis of life-threatening disease. Social Science & Medicine. 1983;17:431–438. doi: 10.1016/0277-9536(83)90348-9. [DOI] [PubMed] [Google Scholar]
- McHorney CA, Ware JE, Jr, Raczek AE. The MOS 36-item Short-Form Health Survey (SF-36): II. Psychometric and clinical tests of validity in measuring physical and mental health constructs. Medical Care. 1993;31:247–263. doi: 10.1097/00005650-199303000-00006. [DOI] [PubMed] [Google Scholar]
- McNair D, Lorr M, Droppleman L. The Manual for the Profile of Mood States. San Diego, CA: Education and Industrial Testing Service; 1992. [Google Scholar]
- Meek PM, Nail LM, Barsevick A, Schwartz AL, Stephen S, Whitmer K, et al. Psychometric testing of fatigue instruments for use with cancer patients. Nursing Research. 2000;49:181–190. doi: 10.1097/00006199-200007000-00001. [DOI] [PubMed] [Google Scholar]
- Mendoza TR, Wang XS, Cleeland CS, Morrissey M, Johnson BA, Wendt JK, et al. The rapid assessment of fatigue severity in cancer patients: use of the Brief Fatigue Inventory. Cancer. 1999;85:1186–1196. doi: 10.1002/(sici)1097-0142(19990301)85:5<1186::aid-cncr24>3.0.co;2-n. [DOI] [PubMed] [Google Scholar]
- Mock V, Abernathy AP, Atkinson A, Barsevick AM, Berger AM, Cella D, et al. NCCN clinical practice guidelines in oncology: Cancer-related fatigue. 2007 Retrieved February 16, 2008, from http://www.nccn.org/professional/physician_gls/default.asp.
- Mock V, Atkinson A, Barsevick AM, Berger AM, Cimprich B, Eisenberger MA, et al. Cancer-related fatigue. Clinical practice guidelines in oncology. Journal of the National Comprehensive Cancer Network : JNCCN. 2007;5:1054–1078. doi: 10.6004/jnccn.2007.0088. [DOI] [PubMed] [Google Scholar]
- Mock V, Frangakis C, Davidson NE, Ropka ME, Pickett M, Poniatowski B, et al. Exercise manages fatigue during breast cancer treatment: a randomized controlled trial. Psycho-Oncology. 2005;14:464–477. doi: 10.1002/pon.863. [DOI] [PubMed] [Google Scholar]
- Mota DD, Pimenta CA. Self-report instruments for fatigue assessment: A systematic review. Research and Theory for Nursing Practice. 2006;20:49–78. doi: 10.1891/rtnp.20.1.49. [DOI] [PubMed] [Google Scholar]
- Nail LM. Fatigue in patients with cancer. Oncology Nursing Forum. 2002;29:537. doi: 10.1188/onf.537-546. [DOI] [PubMed] [Google Scholar]
- NIH. NIH State-of-the-Science Statement on symptom management in cancer: Pain, depression, and fatigue. NIH Consensus and State-of-the-Science Statements. 2002;19:1–29. [PubMed] [Google Scholar]
- Okuyama T, Akechi T, Kugaya A, Okamura H, Imoto S, Nakano T, et al. Factors correlated with fatigue in disease-free breast cancer patients: application of the Cancer Fatigue Scale. Supportive Care in Cancer. 2000;8:215–222. doi: 10.1007/s005200050288. [DOI] [PubMed] [Google Scholar]
- Okuyama T, Akechi T, Kugaya A, Okamura H, Shima Y, Maruguchi M, et al. Development and validation of the Cancer Fatigue Scale: a brief, three-dimensional, self-rating scale for assessment of fatigue in cancer patients. Journal of Pain and Symptom Management. 2000;19:5–14. doi: 10.1016/s0885-3924(99)00138-4. [DOI] [PubMed] [Google Scholar]
- Ostlund U, Gustavsson P, Fürst CJ. Translation and cultural adaptation of the Piper Fatigue Scale for use in Sweden. European Journal of Oncology Nursing. 2007;11:133–140. doi: 10.1016/j.ejon.2006.04.034. [DOI] [PubMed] [Google Scholar]
- Payne J, Piper B, Rabinowitz I, Zimmerman B. Biomarkers, fatigue, sleep, and depressive symptoms in women with breast cancer: a pilot study. Oncology Nursing Forum. 2006;33:775–783. doi: 10.1188/06.ONF.775-783. [DOI] [PubMed] [Google Scholar]
- Pearson RG, Byars GE. The development and validation of a checklist for measuring subjective fatigue. Randolph Air Force Base, Texas: United States Air Force School of Aviation Medicine; 1956. pp. 56–115. [Google Scholar]
- Pickard-Holley S. Fatigue in cancer patients. A descriptive study. Cancer Nursing. 1991;14:13–19. [PubMed] [Google Scholar]
- Piper BF. Measuring fatigue. In: Frank-Stromberg M, Olsen SJ, editors. Instruments for clinical health-care research. 3. Boston, MA: Jones & Bartlett; 2004. pp. 538–553. [Google Scholar]
- Piper BF, Dibble SL, Dodd MJ, Weiss MC, Slaughter RE, Paul SM. The revised Piper Fatigue Scale: psychometric evaluation in women with breast cancer. Oncology Nursing Forum. 1998;25:677–684. [PubMed] [Google Scholar]
- Piper BF, Dodd MJ, Ream E, Richardson A, Berger AM, Kessinger AM, et al. Better Health Through Nursing Research: State of the Science Congress Proceedings. American Nurses’ Association; Washington, D.C: 1999. Improving the clinical measurement of cancer treatment-related fatigue; p. 99. [Google Scholar]
- Prue G, Rankin J, Allen J, Gracey J, Cramp F. Cancer-related fatigue: a critical appraisal. European Journal of Cancer (Oxford, England : 1990) 2006;42:846–863. doi: 10.1016/j.ejca.2005.11.026. [DOI] [PubMed] [Google Scholar]
- Quick M, Fonteyn M. Development and implementation of a clinical survey for cancer-related fatigue assessment. Clinical Journal of Oncology Nursing. 2005;9:435–439. doi: 10.1188/05.CJON.435-439. [DOI] [PubMed] [Google Scholar]
- Reddy S, Bruera E, Pace E, Zhang K, Reyes-Gibby CC. Clinically important improvement in the intensity of fatigue in patients with advanced cancer. Journal of Palliative Medicine. 2007;10:1068–1075. doi: 10.1089/jpm.2007.0007. [DOI] [PubMed] [Google Scholar]
- Rhoten D. Concept clarification in nursing. 1. Rockville, MD: Aspen; 1982. Fatigue in the postsurgical patient; pp. 277–300. [Google Scholar]
- Saito Y, Kogi K, Kashiwagi S. Factors underlying subjective feelings of fatigue. Journal of the Science of Labor. 1970;46:205–224. [Google Scholar]
- Schneider RA. Concurrent validity of the Beck Depression Inventory and the Multidimensional Fatigue Inventory-20 in assessing fatigue among cancer patients. Psychology Reports. 1998a;82:883–886. doi: 10.2466/pr0.1998.82.3.883. [DOI] [PubMed] [Google Scholar]
- Schneider RA. Reliability and validity of the Multidimensional Fatigue Inventory (MFI-20) and the Rhoten Fatigue Scale among rural cancer outpatients. Cancer Nursing. 1998b;21:370–373. doi: 10.1097/00002820-199810000-00009. [DOI] [PubMed] [Google Scholar]
- Schwartz A. The Schwartz Cancer Fatigue Scale: Testing reliability and validity. Oncology Nursing Forum. 1998;25:711–717. [PubMed] [Google Scholar]
- Schwartz A, Meek P. Additional construct validity of the Schwartz Cancer Fatigue Scale. Journal of Nursing Measurement. 1999;7:35–45. [PubMed] [Google Scholar]
- Smets EM, Garssen B, Bonke B, De Haes JC. The Multidimensional Fatigue Inventory (MFI) psychometric qualities of an instrument to assess fatigue. Journal of Psychosomatic Research. 1995;39:315–325. doi: 10.1016/0022-3999(94)00125-o. [DOI] [PubMed] [Google Scholar]
- Smets EM, Garssen B, Cull A, de Haes JC. Application of the Multidimensional Fatigue Inventory (MFI-20) in cancer patients receiving radiotherapy. British Journal of Cancer. 1996;73:241–245. doi: 10.1038/bjc.1996.42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smets EM, Visser MR, Willems-Groot AF, Garssen B, Schuster-Uitterhoeve AL, de Haes JC. Fatigue and radiotherapy: (B) experience in patients 9 months following treatment. British Journal of Cancer. 1998;78:907–912. doi: 10.1038/bjc.1998.600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stein KD, Martin SC, Hann DM, Jacobsen PB. A multidimensional measure of fatigue for use with cancer patients. Cancer Practice. 1998;6:143–152. doi: 10.1046/j.1523-5394.1998.006003143.x. [DOI] [PubMed] [Google Scholar]
- Stone P, Richards M, A’Hern R, Hardy J. Fatigue in patients with cancers of the breast or prostate undergoing radical radiotherapy. Journal of Pain and Symptom Management. 2001;22:1007–1015. doi: 10.1016/s0885-3924(01)00361-x. [DOI] [PubMed] [Google Scholar]
- Stone P, Richards M, Hardy J. Fatigue in patients with cancer. European Journal of Cancer. 1998;34:1670–1676. doi: 10.1016/s0959-8049(98)00167-1. [DOI] [PubMed] [Google Scholar]
- Sutherland HJ, Lockwood GA, Cunningham AJ. A simple, rapid method for assessing psychological distress in cancer patients: Evidence of validity for linear analog scales. Journal of Psychosocial Oncology. 1989;7:31–43. [Google Scholar]
- Vogelzang NJ, Breitbart W, Cella D, Curt GA, Groopman JE, Horning SJ, et al. Patient, caregiver, and oncologist perceptions of cancer-related fatigue: results of a tripart assessment survey. The Fatigue Coalition. Seminars in Hematology. 1997;34(3 Suppl 2):4–12. [PubMed] [Google Scholar]
- Waltz CF, Strickland OL, Lenz ER. Measurement in nursing and health research. 3. New York: Springer Publishing Company; 2005. [Google Scholar]
- Winstead-Fry P. Psychometric assessment of four fatigue scales with a sample of rural cancer patients. Journal of Nursing Measurement. 1998;6:111–122. [PubMed] [Google Scholar]
- Wu HS, McSweeney M. Measurement of fatigue in people with cancer. Oncology Nursing Forum. 2001;28:1371–1384. quiz 1385–1386. [PubMed] [Google Scholar]
- Wu HS, McSweeney M. Assessing fatigue in persons with cancer: an instrument development and testing study. Cancer. 2004;101:1685–1695. doi: 10.1002/cncr.20540. [DOI] [PubMed] [Google Scholar]
- Wu HS, McSweeney M. Cancer-related fatigue: “It’s so much more than just being tired. European Journal of Oncology Nursing. 2007;11:117–125. doi: 10.1016/j.ejon.2006.04.037. [DOI] [PubMed] [Google Scholar]
- Wu HS, Wyrwich KW, McSweeney M. Assessing fatigue in persons with cancer: Further validation of the Wu Cancer Fatigue Scale. Journal of Pain and Symptom Management. 2006;32:255–265. doi: 10.1016/j.jpainsymman.2006.06.001. [DOI] [PubMed] [Google Scholar]
- Yellen SB, Cella DF, Webster K, Blendowski C, Kaplan E. Measuring fatigue and other anemia-related symptoms with the Functional Assessment of Cancer Therapy (FACT) measurement system. Journal of Pain and Symptom Management. 1997;13:63–74. doi: 10.1016/s0885-3924(96)00274-6. [DOI] [PubMed] [Google Scholar]
- Yoshitake H. Rating the feelings of fatigue. Journal of the Science of Labor. 1969;45:422–432. [Google Scholar]
- Yoshitake H. Relations between the symptoms and the feeling of fatigue. Ergonomics. 1971;14:175–186. doi: 10.1080/00140137108931236. [DOI] [PubMed] [Google Scholar]
- Yoshitake H. Three characteristic patterns of subjective fatigue symptoms. Ergonomics. 1978;21:231–233. doi: 10.1080/00140137808931718. [DOI] [PubMed] [Google Scholar]