Abstract
Cerebral cortical progenitor cells can be classified into several different types, and each progenitor type integrates cell-intrinsic and cell-extrinsic cues to regulate neurogenesis. On one hand, cell-intrinsic mechanisms that depend upon appropriate apical-basal polarity are established by adherens junctions and apical complex proteins and are particularly important in progenitors with apical processes contacting the lateral ventricle. The apical protein complexes themselves are con-centrated at the ventricular surface, and apical complex proteins regulate mitotic spindle orientation and cell fate. On the other hand, remarkably little is known about how cell-extrinsic cues signal to progenitors and couple with cell-intrinsic mechanisms to instruct neurogenesis. Recent research shows that the cerebrospinal fluid, which contacts apical progenitors at the ventricular surface and bathes the apical complex of these cells, provides growth- and survival-promoting cues for neural progenitor cells in developing and adult brain. This review addresses how the apical-basal polarity of progenitor cells regulates cell fate and allows progenitors to sample diffusible signals distributed by the cere-brospinal fluid. We also review several classes of signaling factors that the cerebrospinal fluid distributes to the developing brain to instruct neurogenesis.
Keywords: adherens junctions, apical complex, cerebral cortex, neural progenitor cell, polarity, cerebral spinal fluid
INTRODUCTION
The complexity of the human brain is due in part to the flexibility of fundamental processes that govern neural progenitor proliferation (Rakic 2009). The challenge that evolution faced, and has solved so elegantly, is to provide developmental mechanisms that vary the size of the cortex phylogenetically while ensuring that changes in cortical size still provide a functionally integrated structure. The solution involves separating proliferating cells from postmitotic neurons. The proliferating cells control cortical size, whereas independent mechanisms control the functional architecture of the cortex. The flexibility in the control of cortical size and architecture seems to be provided in part by the presence of multiple types of neural progenitor cells (Figure 1; Angevine et al. 1970, Fietz & Huttner 2011, Kriegstein & Alvarez-Buylla 2009), whose patterns of cell division and cell specification are also controlled in interacting, but partially separable, ways. Although our understanding of the mechanisms underlying mammalian cerebral cortical development has certainly benefited from studies in lower vertebrates and invertebrates, humans have also proven to be a robust genetic system for the identification of key genes underlying cortical neurogenesis, many of which subserve the mitotic spindle. The mechanisms underlying the action of these microcephaly genes have been the subject of several recent reviews (Manzini & Walsh 2011, Thornton & Woods 2009). Here, we focus on how cell-extrinsic cues may help regulate neurogenesis by interacting with or complementing the mechanisms that regulate the mitotic spindle.
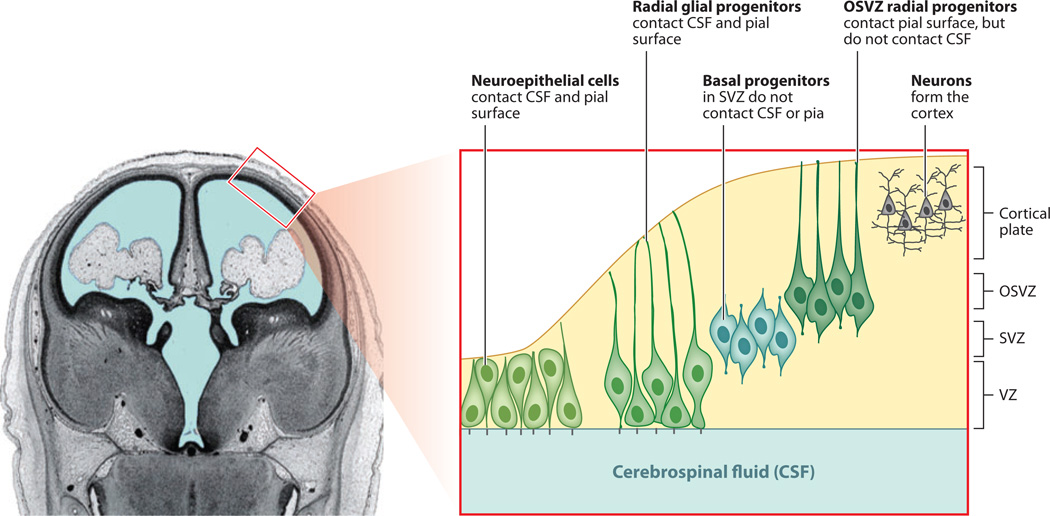
Figure 1.
Neural progenitor cells in the developing mammalian cerebral cortex. Cerebral cortical progenitor cells divide in three principal locations in the developing mammalian brain: the apical ventricular zone (VZ), the inner subventricular zone (SVZ), and the outer subventricular zone (OSVZ). Progenitors in the apical progenitor pool can be divided into two main types of progenitor cells: neuroepithelial cells and radial glial cells. The progenitors in the SVZ constitute the basal progenitor cell pool as well as the most recently discovered class of progenitors, the radial-type progenitors that lie in the OSVZ, which have radial morphology with a long basal process but lack the apical process contacting the ventricle. They are more apparent in mammals with larger brains but are seen in mice as well (Fietz et al. 2010, Hansen et al. 2010, Reillo et al. 2011, Wang et al. 2011). Coronal image of human fetal brain reproduced and adapted from O’Rahilly & Müller (1994), copyright © 1994, Wiley-Liss, Inc.
PROLIFERATIVE ELEMENTS IN THE DEVELOPING CEREBRAL CORTEX
The cell bodies of apical progenitors lie adjacent to the ventricular surface, which is also defined as the apical surface. These apical progenitors include the initial population of neuroepithelial progenitors that compose the early neuroepithelium as well as later-appearing radial glial cells. Apical progenitors share many features with other epithelial cells, such as apical-basal polarity and side-to-side contacts with neighboring cells via adherens junctions. Neuroepithelial and radial glial cell processes generally extend from the apical ventricular surface to the basal lamina at the pial surface, although a population of short apical neural precursors with basal processes of more variable length have also been described in the ventricular zone (VZ) (Gal et al. 2006, Stancik et al. 2010). As apical progenitors proliferate, the single-cell-thick neuroepithelium rapidly adopts a pseudostratified structure. Apical progenitors undergo interkinetic nuclear migration, which involves the apical-basal migration of their nuclei within the pseudo-stratified VZ during the cell cycle; the cells undergo mitosis only at the apical ventricular surface (Kriegstein & Alvarez-Buylla 2009, Taverna & Huttner 2010). Neuroepithelial progenitors tend to divide symmetrically to generate pairs of daughter cells with progenitor cell fate early in development, although some early neurons or intermediate progenitors may be generated as well (Haubensak et al. 2004; Noctor et al. 2004, 2007). During peak neurogenesis, radial glial progenitors favor asymmetric cell division, which leads to pairs of daughter cells with distinct progenitor or early neuronal cell fate (Kriegstein & Alvarez-Buylla 2009). Because symmetric cell divisions producing two proliferative daughter cells provide exponential expansion of cerebral cortical cell numbers (1, 2, 4, 8, 16, 32 …), the symmetry of division of apical progenitors is a crucial control on ultimate cerebral cortical size.
Basal progenitors [also called intermediate progenitors, subventricular zone (SVZ) progenitors, or nonsurface progenitors] are defined by their lack of prominent apical or basal processes and their location relatively basal to the apical surface. Basal progenitors are derived from apical progenitors, but they localize their nuclei in the SVZ and divide almost exclusively in a symmetric fashion to generate pairs of postmitotic daughter neurons (Kriegstein et al. 2006, Martinez-Cerdeno et al. 2006, Noctor et al. 2008, Pontious et al. 2008). As cortical progenitor cells transition from apical and basal progenitors to postmitotic neurons, they sequentially express molecular markers defining their identity including Pax6 (apical progenitors), Tbr2 (basal progenitors), and Tbr1 (neurons) (Englund et al. 2005, Kawaguchi et al. 2008). However, more complex transcrip-tional profiles have been proposed to underlie these transitions as well (Kawaguchi et al. 2008).
Recently, a third general type of progenitor has been identified that has a modified radial morphology but localizes to the SVZ. These outer SVZ (OSVZ) progenitors appear to be present in all mammals to varying extents but are enriched in mammals with larger cerebral cortices, in which an expanded OSVZ appears during mid-gestation, coinciding with the onset of neurogenesis (Fietz et al. 2010, Hansen et al. 2010, Reillo et al. 2011, Shitamukai et al. 2011, Smart et al. 2002, Wang et al. 2011). The OSVZ progenitors show radial morphology and express classic markers of radial glial progenitor cells including Pax6, phospho-vimentin, GFAP (glial fibrillary acidic protein), and BLBP (brain lipid-binding protein), which distinguishes them from typical basal progenitors. OSVZ progenitors appear to have random planes of cleavage and, like apical progenitors, can undergo proliferative and self-renewing cell divisions or can divide asymmetrically to generate an apical daughter as well as an intermediate progenitor (Fietz et al. 2010, Hansen et al. 2010, Reillo et al. 2011). Although OSVZ progenitors do not extend apical processes to the VZ, they retain their basal processes throughout mitosis (Fietz et al. 2010, Hansen et al. 2010).
This review focuses mainly on the control of cell proliferation in the apical progenitors, because the ultimate size of the cerebral cortex, both developmentally and evolutionarily, is so dependent upon even small changes in apical progenitor proliferation. Recent research on the control of proliferation of these cells has focused on the role of cyto-plasmic proteins [Notch, Numb, β-catenin, and Par3/Par6/aPKC (atypical protein kinase C)] and short-acting signaling pathways (e.g., Wnt, Delta, Jagged). In addition, we survey recent work that suggests that the apical complex also integrates cell-extrinsic cues carried in the cerebrospinal fluid (CSF), some derived from extremely distant sources, that may provide global, age-related controls over neural proliferation.
ESTABLISHING PROGENITOR CELL POLARITY
Apical cortical progenitor cells are highly polarized; their apical membranes form the ventricular surface of the developing brain, facing the lumen of the lateral ventricle that is filled with CSF, whereas their basal processes reach all the way to the outer, pial surface of the developing brain (Chenn et al. 1998, Mission et al. 1991, Takahashi et al. 1990). Apical-basal polarity is established and maintained in part by adherens junctions, which are ring-like, cadherin-containing contacts located between cells that regulate calcium-dependent cell-cell adhesion (Aaku-Saraste et al. 1997, Gotz & Huttner 2005, Harris & Tepass 2010, Stoykova et al. 1997). Cadherins connect to the catenins, including β-catenin and α-catenin, and together they regulate actin filament dynamics. In this manner, adherens junctions asymmetrically distribute proteins in progenitor cells while also anchoring the apical endfeet of adjacent radial glial cells to the ventricular surface (Figure 2).
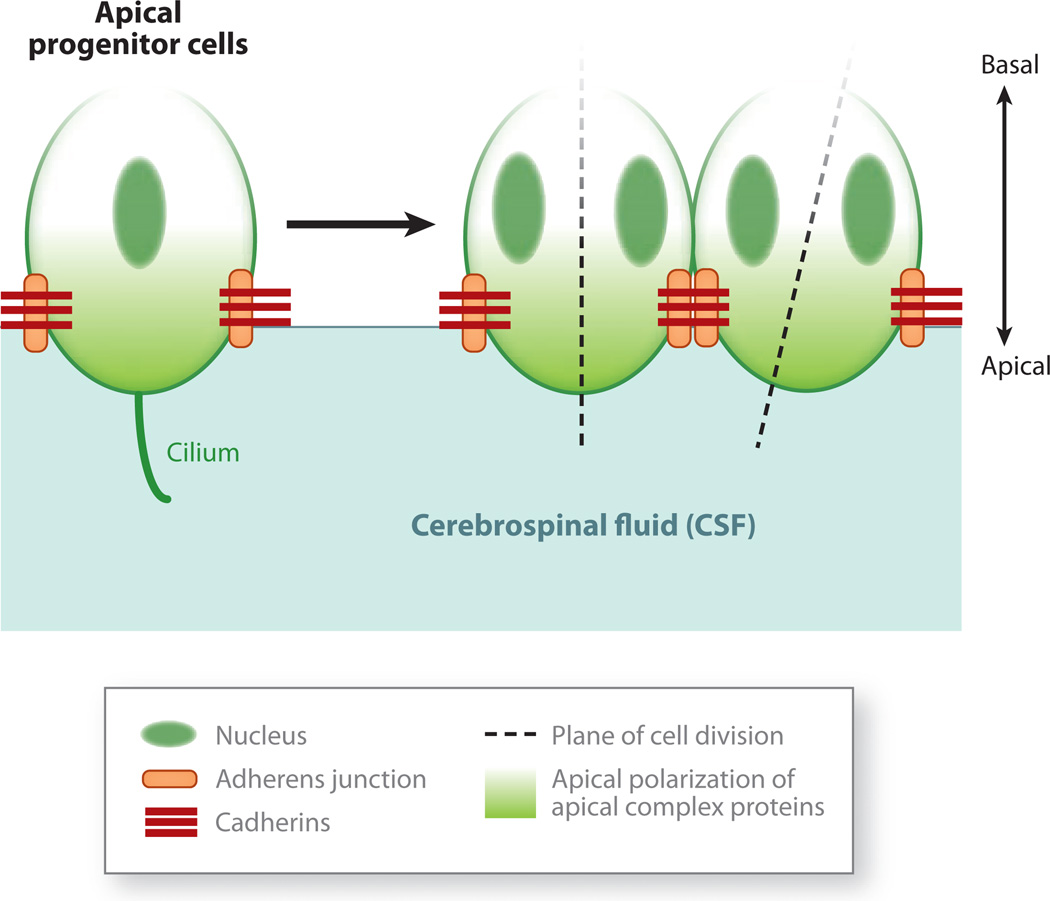
Figure 2.
Apical-basal polarity in cortical progenitor cells. Apical progenitor cells have a distinct apical-basal polarity. Their apical surface contacts the cerebrospinal fluid (CSF) that fills the ventricles, whereas their basal processes extend to and contact the meninges, basal lamina, and vasculature. The adhesion of adjacent progenitor cells to each other is maintained by adherens junctions, which are cadherin-containing contacts between cells. The adherens junctions also define the border of the apical membrane domain that contacts the CSF. The adherens junctions and apical membrane are home to the apical complex proteins, which play an active role in polarizing cellular proteins. The unequal inheritance of the apical membrane and associated proteins appears to regulate whether dividing cells generate pairs of daughter cells with the same, symmetric cell fate (e.g., two progenitor cells) or cells with distinct, asymmetric cell fates (i.e., one progenitor and one neuron). The progenitors of the outer subventricular zone do not appear to show the same expression of apical complex proteins (Fietz et al. 2010).
The distinctive polarity of cortical progenitor cells invites comparison with similar polarized protein distributions in other cell division processes. In particular, genetic control of asymmetric cell divisions has been extensively studied in the Caenorhabditis elegans zygote and Drosophila larval neuroblasts. In the asymmetric cell divisions of the Drosophila neu-roblast, an apical protein complex consisting of the Par3/Par6/aPKC proteins is distributed in a highly polarized fashion. Some of these asymmetrically expressed proteins, including Par3/Bazooka as well as Bazooka-interacting proteins Pins and Inscuteable, then serve to orient the mitotic spindle; this orientation specifies whether daughter cells inherit similar complements of cytoplasmic determinants (in which case the daughters tend to adopt similar cell fates, a symmetric cell division), or inherit dissimilar complements of proteins and adopt distinct (asymmetric) fates (Siller & Doe 2009). Principal among the cytoplasmic regulators of cell fate is the Numb protein, which negatively regulates the neurogenic gene, Notch. Inheritance of Numb tends to cause cells to adopt a more differentiated fate, whereas absence of Numb causes cells to remain neuroblasts (Rhyu et al. 1994, Wirtz-Peitz et al. 2008). Thus, the mechanism of asymmetric cell division in these systems requires cytoplasmic regulators of cell fate and controlled orientation of the mitotic spindle in relationship to these cytoplasmic proteins (Siller & Doe 2009, Tajbakhsh et al. 2009). Although mammalian cerebral cortical progenitor cells express many orthologous proteins with similar effects on proliferation and cortical size, in strikingly asymmetric patterns, a major unanswered question involves the extent to which orientation of the mitotic spindle specifies inheritance of these cytoplasmic determinants.
ADHERENS JUNCTIONS IN NEURAL PROGENITOR CELLS
Proteins associated with adherens junctions in the developing cerebral cortex have been well documented to regulate progenitor cell pool and brain size (Bilder et al. 2000, Chae et al. 2004, Chenn & McConnell 1995, Chenn & Walsh 2002, Junghans et al. 2005, Kim et al. 2010, Lien et al. 2006, Machon et al. 2003). One challenge with these types of studies is that the integrity of neuroepithelial architecture is so intimately linked to progenitor proliferation. On the one hand, for example, disruption of adherens junctions via genetic deletion of α-E-catenin in cortical progenitor cells leads to an activation of Sonic hedgehog (Shh) signaling associated with accelerated cell cycle progression and decreased apoptosis, which together lead to hyperplasia (Lien et al. 2006). On the other hand, focal reduction of α-E-catenin expression in a wild-type background promotes premature exit from the cell cycle and a decrease in β-catenin signaling (Stocker & Chenn 2009). β-catenin also has dual roles in progenitors as an integral component of adherens junctions as well as a transcriptional coactivator that interacts with the TCF/LEF family of transcription factors to transduce Wnt signals (Clevers 2006). Overexpression of a truncated, and hence activated, β-catenin expands the progenitor cell pool by limiting cell cycle exit (Chenn & Walsh 2002, Machon et al. 2007, Wrobel et al. 2007). This increased β-catenin activity considerably expands brain size in the lateral direction, leading to the generation of additional folds in the cortex (Chenn & Walsh 2002). Cortical thickness is markedly reduced in these brains, which suggests that β-catenin overexpression leads, in effect, to an exchange of neurons for progenitors. Interestingly, β-catenin expression is developmentally regulated such that β-catenin signaling decreases in the progenitor pool during development (Mutch et al. 2009). Consistent with the inside-out model of cortical development in which the earliest neurons form the deepest cortical layers, higher β-catenin activity favors the generation of deep-layer neurons, whereas lower β-catenin activity favors the development of upper layer neurons (Mutch et al. 2009, Wrobel et al. 2007). Reintroduction of β-catenin signaling in neural progenitors during mid-neurogenesis at a time when more upper layer neurons are typically generated can partially extend deep-layer neuron production (Mutch et al. 2009). Although radial glial progenitors appear to use β-catenin as a mechanism for retaining apical progenitor cell fate (Hirabayashi et al. 2004, Mutch et al. 2010, Woodhead et al. 2006, Zhang et al. 2010), its activity is also tightly regulated in intermediate progenitors (Kuwahara et al. 2010, Munji et al. 2011).
APICAL COMPLEX PROTEINS, NUMB/NUMBL, AND NOTCH SIGNALING IN PROGENITOR CELLS
Adherens junctions also serve as docking sites for the proteins of the apical complex, also known as the Par protein complex in invertebrates (Manabe et al. 2002). Highly conserved across species, the Par proteins have a well-established role in determining polarity and cell division (Jan & Jan 2001, Kemphues et al. 1988, Knoblich 2008, Siller & Doe 2009, Wodarz 2005). The mammalian apical complexes assemble as Par3/Par6/aPKC, Crb/Pals1/Patj, and Mals/Pals1 (Margolis & Borg 2005). Perhaps the best understood of these, the Par3/Par6/aPKC complex, is assembled by the PDZ-containing scaffold Par3, which then recruits Par6, a CRIB (Cdc42/Rac interactive binding) and PDZ domain–containing protein that inhibits aPKC activity. When bound to Cdc42 or Rac1, Par6 no longer inhibits aPKC, allowing aPKC to phosphorylate downstream target proteins, which leads to their selective exclusion from the apical domain of the progenitor (Siller & Doe 2009).
Several apical complex proteins, including Par3, Par6, and Pals1 [Protein associated with Lin7, a member of the MAGUK (membrane-associated guanylate kinase) family of scaffolding proteins (Kamberov et al. 2000)], have been shown to regulate proliferation and cell fate of apical progenitors when overexpressed or interfered with in mammals, suggesting that the pattern of apical complex inheritance helps define symmetric or asymmetric cell divisions, although this is not completely settled (Bultje et al. 2009, Costa et al. 2008, Kim et al. 2010, Manabe et al. 2002, Srinivasan et al. 2008). For example, the OSVZ radial progenitors show asymmetrical cell divisions but seem not to express the apical complex proteins at all, which suggests that the apical complex may not be universally required for asymmetrical cell divisions or all radial progenitors. Nonetheless, in apical progenitors, many apical complex proteins localize strikingly along the apical ventricular surface, and a daughter cell that inherits more apical surface from the mother cell (Kosodo et al. 2004, Noctor et al. 2008), including Par3/aPKC (Marthiens & ffrench-Constant 2009) or Pals1 (Kim et al. 2010), appears more likely to remain a progenitor cell (Figure 2). Similarly in chicks, aPKCζ/λ is associated with the apical surface of neural precursors in the developing chick neural tube (Ghosh et al. 2008). Thus, apical complex proteins regulate progenitor proliferation and cell fate in the developing apical progenitors of the mammalian brain.
In invertebrates, the apical complex regulates the distribution of a protein called Numb, which regulates neuronal cell fate, and genetic studies in vertebrates have uncovered many roles for the Numb orthologs Numb and Numb-like (Numb/Numbl) in neurogenesis (Bultje et al. 2009; Li et al. 2003; Nishimura & Kaibuchi 2007; Petersen et al. 2002, 2004; Rasin et al. 2007; Shen et al. 2002; Tajbakhsh et al. 2009). In mammalian neural progenitors, Numb/Numbl localize to the basolateral regions of progenitor endfeet in the VZ (Rasin et al. 2007). Numb/Numbl are required for continued cell division during development, as their deletion in the developing brain leads to premature exit from the cell cycle and disrupts cortical development, which leads to a mature cortex with severe neocortical thinning and diminished hippocampal size (Li et al. 2003, Petersen et al. 2004). At the cellular level, Numb/Numbl interact with several adherens junction components, including Cdh1 (E-cadherin), Cdh2 (N-cadherin), and the catenins (α-E-catenin, β-catenin). In Numb/Numbl-deficient cells, the cadherins are mistargeted to the apical membrane, which leads to a disruption of adherens junctions (Rasin et al. 2007). Numb is an endocytic adaptor protein known to regulate Notch endocytosis (Berdnik et al. 2002), to associate directly with recycling endosomes, and to be regulated by the Golgi complex adapter protein ACBD3 (Zhou et al. 2007). Thus, Numb may play an important role in trafficking adherens junction components.
Numb is phosphorylated in a Par3/Par6/ aPKC-dependent manner (Klezovitch et al. 2004, Nishimura & Kaibuchi 2007, Smith et al. 2007), and these apical complex proteins are essential for self-renewal of neural progenitors in the developing mammalian cortex. Knockdown and overexpression studies of Par3 and Par6 promote premature differentiation and excessive cell division, respectively (Costa et al. 2008). Trafficking of Par3 and Par6 is regulated by Cdc42, a Rho GTPase family member, so that conditional removal of Cdc42 leads to loss of adherens junctions and misdirects mitotic progenitors basally, away from the apical surface (Cappello et al. 2006). Although these newly relocated progenitors continue to cycle, they do so with an increase in basal progenitor cell fate.
Recent work suggests that Par3 is segregated asymmetrically in some mammalian cortical progenitor cells and serves as a cell-autonomous regulator of Notch signaling (Bultje et al. 2009, Manabe et al. 2002). Par3 is dynamically localized in progenitors; it associates with the lateral membrane domain of ventricular endfeet during interphase and then disperses as the cell cycle progresses. Asymmetric Par3 expression during the cell cycle leads to asymmetric Par3 segregation in daughter cells, which promotes differential Notch signaling via Numb/Numbl that drives distinct cell fate in daughter cells. Ultimately, the daughter cell with greater Notch signaling remains a radial glial progenitor, whereas the cell with lesser Notch activity adopts neuronal or intermediate progenitor cell fate (Bultje et al. 2009). The degree to which Notch signaling may also influence the inheritance of the basal process will be interesting to examine. Quantification of the cell fate of dividing progenitors recently revealed that inheritance of both apical and basal processes by a progenitor cell may be essential for maintaining its apical progenitor identity (Konno et al. 2008), but the signaling proteins present in the basal process that might regulate this effect are not known.
The asymmetric distribution of Notch by Par3 adds an interesting mechanistic twist to the Notch literature, as Notch is already well known for maintaining the neural stem cell pool during mammalian neurogenesis (Chenn & McConnell 1995, Gaiano et al. 2000, Imayoshi et al. 2010, Mizutani & Saito 2005, Mizutani et al. 2007, Tajbakhsh et al. 2009). Notch activity maintains progenitor proliferation, and its removal promotes the generation of upper layer neurons. Thus, although progenitors retain competence to generate neurons, they lose the ability to generate lower-layer-fated cells, which essentially are skipped (Mizutani & Saito 2005). Importantly, Notch selectively promotes neural stem cell fate through activation of the canonical Notch effector C-promoter binding factor 1 (CBF1), which is attenuated in intermediate progenitor cells. Although decreased CBF1 activity converts neural stem cells to intermediate progenitors, the converse experiment in which CBF1 is artificially activated in intermediate progenitors fails to revert cell fate, which indicates that Notch signaling plays a key role in specifying the lineage commitment of prospective neurons (Mizutani et al. 2007). Oscillations in Notch activity via Hes1, which modulate expression of proneural genes including Ngn2 and Dll1, Notch ligands, and cell cycle regulators, have also been suggested to promote neural maintenance (Hirataetal.2002,Shimojo et al. 2008). Notch was also recently shown to play a dual role in the maintenance of apical mitoses and apical-basal polarity via interactions with the Crumbs/Moe apical complex in ze-brafish neuroepithelial cells (Ohata et al. 2011).
Despite these recent advances, the biochemical interactions between adherens junctions and interacting proteins as well as the regulation of downstream effector pathways are complex and remain poorly understood. Surprisingly, deletion of aPKCλ in mice at E15, midway through neurogenesis, does not clearly affect cell fate decisions in a manner comparable with its role in invertebrates (Imai et al. 2006). In addition, mouse Lgl1 (Lethal giant larva 1) mutants exhibited hyperprolifer-ation of progenitors in the brain in conjunction with Numb mislocalization (Klezovitch et al. 2004). Therefore, the role of apical complex proteins in progenitors likely extends well beyond the regulation of proliferation and cell fate to other biological functions as well.
POTENTIAL ROLES OF APICAL COMPLEX PROTEINS IN CELL SURVIVAL
Recent evidence indicates that the apical complex is essential for survival of neural progenitors and newly differentiated neurons. Conditional deletion of the apical complex protein Pals1 in progenitor cells not only causes the expected premature withdrawal from the cell cycle that is coupled with excessive generation of early born postmitotic neurons, but is also followed by the rapid death of these newly generated neurons (Kim et al. 2010). Together, these two effects lead to the abrogation of essentially the entire cerebral cortex. Loss of Pals1 blocks essential cell survival signals, including the mammalian target of rapamycin (mTOR) pathway, such that concomitant mTORC1 activation via conditional Tsc2 deletion partially restores the Pals1 deficiency (Kim et al. 2010).
The Pals1 cell death phenotype reveals a previously unidentified role for the apical complex in promoting survival during the transition from progenitor to neuron. Intriguingly, this is akin to conditional deletion of Notch1 and Notch3, which also result in a profound death of neural progenitors and newly differentiating neurons (Mason et al. 2006). Although the exact mechanism underlying the Notch phenotypes remains unclear, Notch-dependent survival in the developing brain is thought to occur by a Hes-independent mechanism (Mason et al. 2006). The shared phenotype between Pals1 and Notch conditional mutants suggests that Pals1 and Notch may either signal in the same pathway or converge on a shared set of targets to regulate survival of newly differentiated neurons.
POTENTIAL ROLES OF APICAL COMPLEX PROTEINS IN GROWTH FACTOR SIGNALING
The prominent cell death phenotype of the Pals1 mutants, and the finding that upregu-lation of mTOR signaling partially restores the phenotype, suggests that growth factor signaling pathways are disrupted in apical complex mutants (Kim et al. 2010). Growth factor signaling, especially via the type 1 insulinlike growth factor (IGF) receptor (IGF1R), mediates powerful, age-dependent effects on the development and maintenance of many organ systems, including the brain, through the regulation of progenitor cell division (Baker et al. 1993, Hodge et al. 2004, Liu et al. 2009, Popken et al. 2004, Randhawa & Cohen 2005), but the mechanisms coordinating the availability of IGF ligands to cortical progenitors have remained unclear. Interestingly, growth factor receptors including IGF1R (Lehtinen et al. 2011) and epidermal growth factor receptor (EGFR) (Sun et al. 2005), as well as phosphotyrosine (Chenn et al. 1998) and phospho-ERK1/2 (extracellular-signal-regulated kinase 1/2) (Toyoda et al. 2010), have enriched expression along the apical ventricular surface of progenitors. Could the apical-basal polarity of progenitors ensure the apical localization of receptors for sampling cell-extrinsic growth factors emanating from the CSF?
A direct interaction between Par3 and Pten (phosphatase and tensin homolog) (Feng et al. 2008, Pinaletal.2006,von Stein et al. 2005, Wu et al. 2007) also suggests that the apical complex interacts with growth factor signaling pathways. Disrupting the apical complex via Pals1 deletion in progenitors abolishes the apical enrichment of IGF1R and attenuates growth factor signaling assessed by pS6 activity (Lehtinen et al. 2011). Consistent with a genetic interaction between the apical complex and growth factor signaling, artificial activation of growth factor signaling by conditional deletion of Pten in Pals1 heterozygous progenitor cells largely restores brain size, owing in part to an expansion of the IGF1R signaling domain, which partly restores the proportions of proliferating apical progenitor cells (Lehtinen et al. 2011). IGF1 is thought to promote S-phase commitment of apical progenitors via PI3K (phosphatidyl-inositol 3-kinase) signaling (Mairet-Coello et al. 2009). Thus, it will be important to determine the nature of the biochemical interaction between the apical complex and Pten. The disruption of IGF1R localization in Pals1 conditional mutants is similar to that of C. elegans LIN-2A, mutation of which disrupts the LET-23 (EGF) receptor and blocks its appropriate cellular localization and signaling (Hoskins et al. 1996, Simske et al. 1996).
EXTRINSIC REGULATION OF NEUROGENESIS AT THE APICAL MEMBRANE
How might the apical ventricular surface receive extrinsic growth factors? In addition to bathing their apical domains in CSF, progenitor cells extend primary cilia directly into it (Cohen et al. 1988, Dubreuil et al. 2007, Han & Alvarez-Buylla 2010, Hinds & Ruffett 1971). Cilia transduce extracellular signals to the cell body, as has been well established for Shh (Han & Alvarez-Buylla 2010, Han et al. 2008, Rohatgi et al. 2007). Although the details of cilia-dependent signaling are not fully understood, primary cilia are critical for normal brain patterning and development (Breunig et al. 2008, Chizhikov et al. 2007, Gorivodsky et al. 2009, Spassky et al. 2008, Stottmann et al. 2009, Willaredt et al. 2008, Louvi & Grove 2011). While vascular sources of secreted proliferative signals are well characterized (Palmer et al. 2000; Shen et al. 2004, 2008; Tavazoie et al. 2008), the apical surfaces of early cortical precursors and their primary cilia directly contact the CSF but not the vasculature (Cohen et al. 1988, Lehtinen et al. 2011). The location of cilia and their immersion in the CSF suggest that the CSF may distribute and synchronize signals regulating neurogenesis, potentially over long distances because the CSF represents a large fluid space without known barriers to diffusion. These possibilities have recently spurred interest in the growth-promoting features of the CSF and the dynamic regulation of the CSF proteome during the period of neurogenesis.
Several studies have investigated potential developmental roles for the CSF beyond provision of a fluid cushion for the central nervous system (CNS) and maintenance of extracellular ionic balance (Dziegielewska et al. 1981, Parada et al. 2005a, Zappaterra et al. 2007). In the developing chick brain, CSF facilitates the retention of midbrain markers including Otx2 and Fgf8 (Parada et al. 2005b), and CSF-fibroblast growth factor 2 (FGF2) promotes precursor proliferation (Martin et al. 2006). In the mouse cerebellum, CSF-distributed Shh has been suggested to stimulate proliferation of cerebellar granule neuron precursors (Huang et al. 2010). Most recently, the embryonic CSF, and in particular CSF-IGF2, was found to provide essential growth- and survival-promoting factors for the developing rodent cortex (Lehtinen et al. 2011). These and other studies support the model that the CSF contains a large and dynamic library of proteins that may instruct many aspects of neuronal development.
THE CEREBROSPINAL FLUID DURING TIMES OF NEUROGENESIS
The study of CSF dates back to the Greeks, who provided the first account of fluid in the brain (reviewed in Tascioglu & Tasciolgu 2005). Galen of Pergamon proposed that the cerebral fluid provided energy for the entire body, but it was the work of Leonardo da Vinci and Andreas Vesalius during the Renaissance that produced the first accurate models of the mammalian cerebroventricular system. Surprisingly, although the Greeks had originally described a fluid within the ventricles, few accounts of CSF had been actually been made since that time. CSF was finally documented in the 1700s, but not until the early twentieth century did Harvey Cushing observe secretion of CSF by the choroid plexus (Cushing 1914).
The primitive cerebroventricular system emerges at neural tube closure, occurring at embryonic day (E)8.5–9 during mouse development, at which point the amniotic fluid trapped in the neural tube serves as the developing brain’s initial CSF (Lowery & Sive 2009). The surrounding neuroepithelium initially secretes additional factors into the CSF and helps to maintain ventricular pressure. The choroid plexus tissues, which actively generate CSF in the mature brain, develop over the course of the following several days to form modified ependyma that project into the brain’s ventricles (Zheng & Chodobski 2005). The choroid plexus develops sequentially in each ventricle in the brain such that the hindbrain/fourth ventricle choroid plexus develops first (mouse E11–12) and is quickly followed by the lateral ventricle choroid plexi (E11–12); the mesen-cephalic/third ventricle choroid plexus is the last to develop, by E14.5 (Zheng & Chodobski 2005). In the human embryo the choroid plexus begins to develop in the fourth and lateral ventricles at Carnegie stage (CS) 18–19, approximately 44 days postovulation (O’Rahilly & Muller 1994). The first appearance of cerebral cortical neurons in the human embryo occurs at CS21, shortly following the appearance of the choroid plexus and the production of CSF (O’Rahilly & Muller 1994), and a similar temporal sequence is observed in several species including mice and rats (Figure 3). Thus, the secretory choroid plexus of the lateral ventricles ultimately derives from the roof plate of the forebrain, which is the known source of many key secreted patterning factors, including many bone morphogenic proteins (Bmps), Wnt proteins, and FGF8 and other FGF family members (Currle et al. 2005, Grove & Fukuchi-Shimogori 2003).
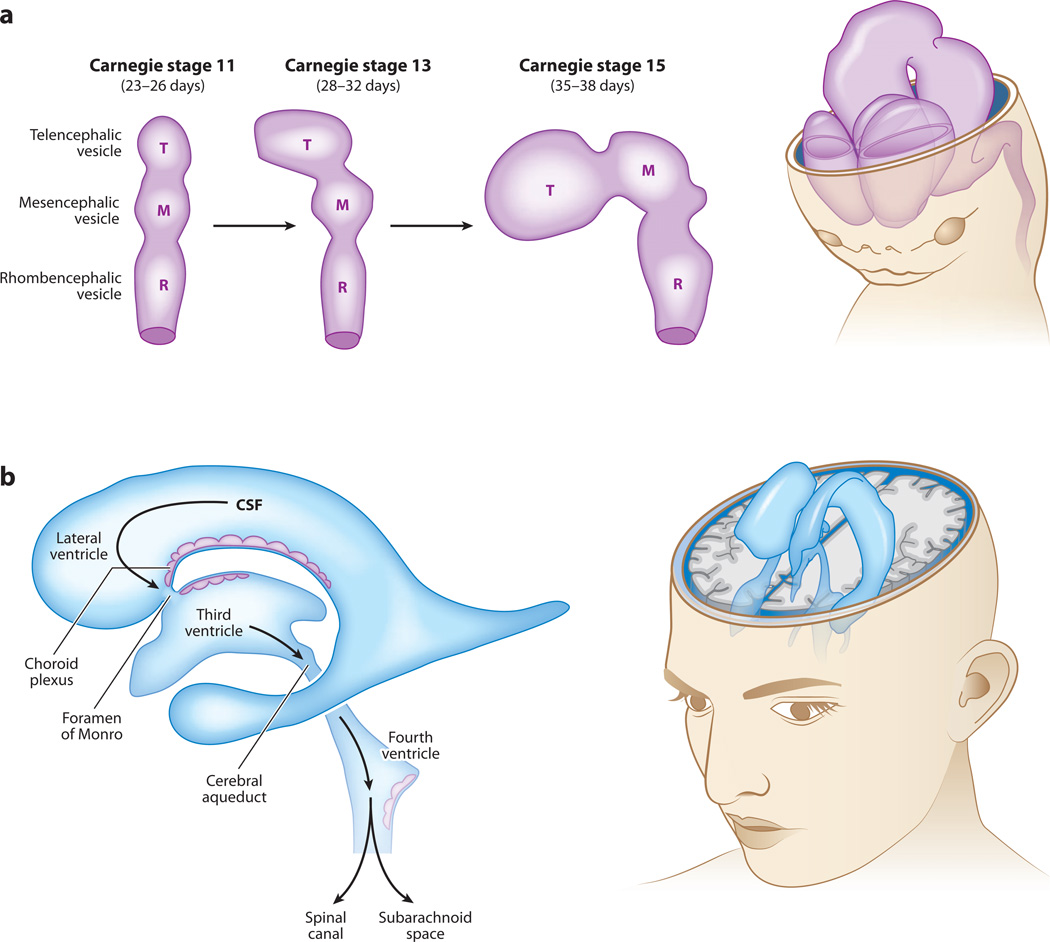
Figure 3.
Cerebrospinal fluid (CSF) flow during embryonic brain development and in adulthood. Schematics of the cerebroventricular system during early human brain development and in the mature adult brain. (a) Upon anterior neural tube closure, the three primary brain vesicles [telencephalic (T), mesencephalic (M), and rhombencephalic (R) vesicles] serve as the rudimentary cerebroventricular system for the developing central nervous system (CNS). Human Carnegie stage (CS) 11 corresponds to approximately embryonic day (E)8.5–9.75 during mouse embryogenesis, and CS13–15 corresponds to approximately E10–11.25. Drawings based on Lowery & Sive 2009. (b) In the mature CNS, CSF generated primarily by the choroid plexus tissues located in each ventricle in the brain fills the ventricles, subarachnoid space, and spinal canal. CSF flows from the lateral ventricles via the foramen of Monro/intraventricular foramen into the mesencephalic/third ventricle, and then via the aqueduct of Sylvius/cerebral aqueduct into the hindbrain/fourth ventricle. The CSF then continues through the foramina of Magendie/median apertures and Luschka/lateral apertures into the spinal canal and subarachnoid space, and is finally resorbed into the venous system via arachnoid villi. An adult human circulates approximately 150 ml of CSF within the cerebroventricular system. The CSF is estimated to turn over approximately three to four times per day, so a healthy CNS produces close to 500 ml of CSF daily.
Early interest in CSF regulation of brain development focused more on its role in transmitting pressure to the brain than on specific CSF constituent proteins (Desmond & Jacobson 1977, Lowery & Sive 2009, Pexieder & Jelinek 1970). For example, artificially disrupting the cerebroventricular system by draining CSF from chick ventricles by intubation decreases intraventricular pressure and globally affects brain size (Desmond & Jacobson 1977). Conversely, increased ventricular pressure may lead to increases in mitotic density (Desmond et al. 2005). Important, however, are observations from naturally occurring rodent models of hydrocephalus, a condition in which ventricles contain excess CSF (Mashayekhi et al. 2002). In these animals, early onset hydrocephalus leads to enlarged, dome-shaped heads and ataxia. Cell-based investigations with CSF obtained from the hydrocephalic Texas rat have found that hydrocephalus-associated CSF inhibits cell division in vitro (Mashayekhi et al. 2002, Owen-Lynch et al. 2003), which suggests that the CSF either accumulates signals that specifically interfere with normal cell proliferation or lacks the necessary proliferation-inducing signals.
The large literature regarding CSF is home to many hypotheses about potentially active roles for the CSF in the CNS (Zheng & Chodobski 2005), including relaying the body’s satiety levels (Martin et al. 1973) and maintaining circadian rhythms (Silver et al. 1996). However, the first compelling evidence that spatial gradients of CSF factors actively guide cell behavior came from an elegant study investigating the biological consequences of CSF flow generated by the beating cilia of the ependymal cells that line the adult ventricles (but are absent in the developing brain) (Sawamoto et al. 2006). The pattern of CSF flow and the migration of neuroblasts destined for the olfactory bulb were strikingly similar, an effect that was disrupted in mouse ciliary mutants with defective ciliary beating and CSF flow (Sawamoto et al. 2006). It had previously been suggested that the choroid plexus provides Slit chemorepellent activity in the developing and adult brain, possibly to induce neuronal migration from the ventricular zone to the cortical plate (Hu 1999, Nguyen-Ba-Charvet et al. 2004). Although SVZ and rostral migratory stream cells also express Slits (Nguyen-Ba-Charvet et al. 2004), gain-of-function experiments with intraventricularly injected Slit, loss-of-function experiments with Slit knockout mice, and choroid plexus grafts demonstrated that CSF-borne Slit1/2 play an important role in guiding neuroblast migration to the olfactory bulb (Sawamoto et al. 2006). Collectively, these studies inspired a renewed interest in understanding how the embryonic CSF proteome might also provide instructive cues for the developing brain.
SIGNALING FROM THE CEREBROSPINAL FLUID TO DEVELOPING CORTICAL TISSUES
Mass spectrometry has offered new approaches for characterizing the highly dynamic CSF proteome (Cavanagh et al. 1983, Dziegielewska et al. 1981, Parada et al. 2005a, Zappaterra et al. 2007, Zheng & Chodobski 2005). Unbiased analyses of human embryonic CSF from CS19–20 (approximately 48–51 days postovulation) together with CSF samples obtained from three distinct time points of rat cortical development—E12.5, E14.5, and E17.5— revealed developmental stage–specific similarities in the CSF proteomes of humans and rodents based on molecular function and biological process. Of the 188 proteins identified in the human embryonic CSF, 135 were identified in any one of the embryonic rat CSF samples, and 83 were present in all four samples of embryonic rat CSF (Zappaterra et al. 2007). The embryonic CSF of the chick and that of the rat also share many similarities, as both contain proteins of the extracellular matrix, regulators of osmotic pressure, ion carriers, hormone-binding proteins, regulators of lipid metabolism, and various enzymes and their regulators (Cavanagh et al. 1983, Parada et al. 2005a).
Distinct embryonic CSF protein signatures exist across ventricles, which raises the possibility of region-specific function (Cavanagh et al. 1983, Zappaterra et al. 2007). In a comparison between rat lateral and hindbrain ventricle CSF at E14.5, lateral ventricle CSF contained 61 distinct proteins not found in hindbrain ventricle CSF (Zappaterra et al. 2007). Because similar numbers of peptides were recovered from CSF samples in each ventricle, this difference between samples likely represents distinct ventricular proteomes. Consistent with this interpretation, evidence for the active secretion of distinct morphogens by the different choroid plexi has been documented. For example, Shh is highly expressed by the hindbrain ventricle choroid plexus (Awatramani et al. 2003, Huang et al. 2009). These findings raise interesting questions regarding local signaling gradients that may be generated in the CSF.
FIBROBLAST GROWTH FACTOR SIGNALING IN CEREBROSPINAL FLUID
Based on these clues, recent studies have investigated whether CSF plays an instructive role in CNS development. Primary CSF, removed from the ventricles of the mammalian brain and used as culture medium, is sufficient to maintain neural stem cells in neurosphere cultures and cortical explants with no other added factors (Lehtinen et al. 2011). This ability to support progenitor cell development was greatest for age-matched CSF samples and tissues. Although all of the factors responsible for the growth-promoting effects of CSF may not yet be known, several strong candidate molecules have been implicated (Figure 4). For example, FGF2 has been identified in embryonic chick CSF (Finch et al. 1995, Martin et al. 2006, Raballo et al. 2000). Immunodepletion of CSF-FGF2 reduces progenitor proliferation to the level of negative controls, which suggests that CSF-FGF2 plays a direct role in promoting the proliferation of chick midbrain progenitors (Martin et al. 2006, Tao et al. 1997). Intriguingly, intravascularly injected, FITC-conjugated FGF2 passes into the embryonic CSF (Martin et al. 2006), which suggests that in addition to choroid plexus–synthesized factors, somatic sources of CSF-distributed signaling cues potentially regulate neurogenesis as well.
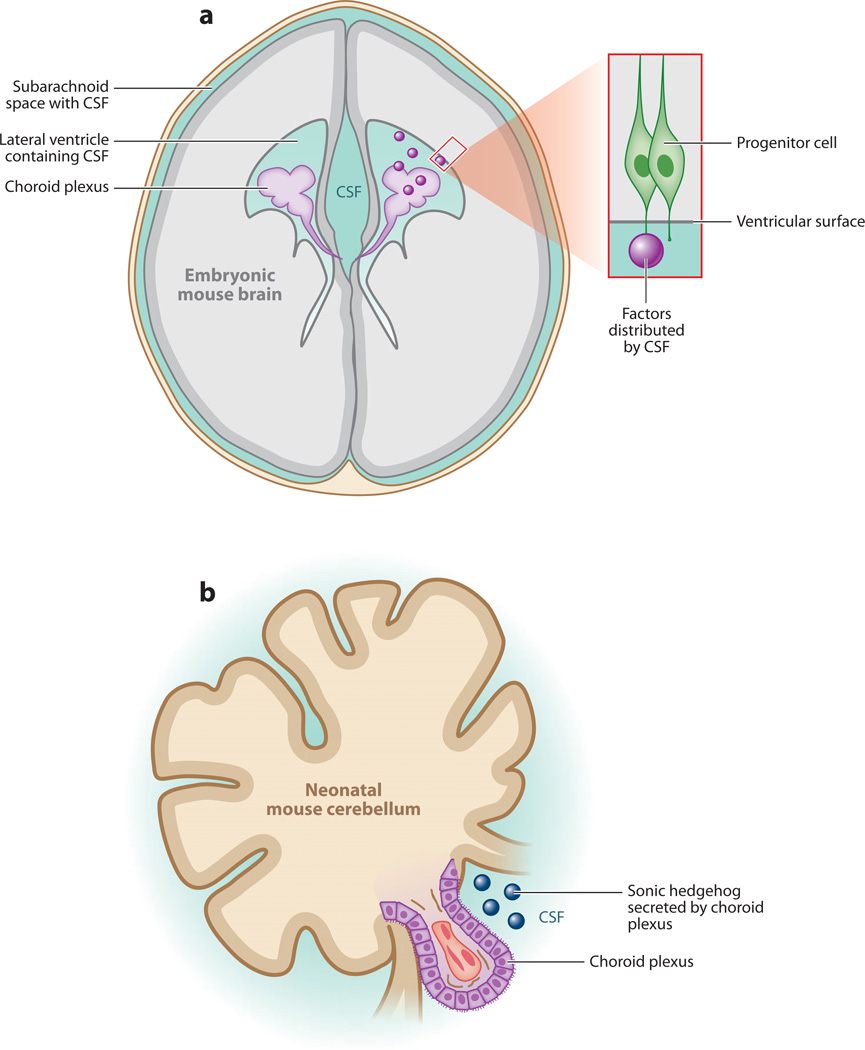
Figure 4.
The cerebrospinal fluid (CSF, blue) distributes diffusible factors during brain development. (a) IGF2 and other factors (purple spheres) are secreted by the choroid plexus and delivered by the CSF to targets on the apical ventricular surface of the developing cerebral cortex (Lehtinen et al. 2011). CSF and CSF-FGF2 have been shown to support proliferation of midbrain progenitor cells (Martin et al. 2006, 2009). Whether similar rules apply to CSF-borne retinoic acid, Wnt, and Bmp signaling, as well as other as yet uncharacterized signals, remains to be elucidated. (b) Sonic hedgehog (Shh) secreted by the hindbrain/fourth ventricle choroid plexus signals in an autocrine manner to instruct choroid plexus development. CSF-Shh also signals in a paracrine manner to stimulate the proliferation of cerebellar granule neuron precursors located in the external granule cell layer (Huang et al. 2009, 2010).
The identification of FGF2 in CSF is particularly intriguing because FGF family members play key roles in early CNS patterning, in stem cell proliferation, and as most recently shown, in the development of cilia (Iwata & Hevner 2009, Neugebauer et al. 2009). In the developing brain, signaling centers that secrete diffusible cues including FGF, Bmps, and Shh initially establish the dorsal/ventral and anterior/posterior axes (Grove & Fukuchi-Shimogori 2003). Remarkably, experimental manipulations that augment, diminish, or introduce entirely new sources of FGF8 to the developing brain accordingly affect area identity as assessed by the emergence of barrel columns in the postnatal somatosensory cortex (Fukuchi-Shimogori & Grove 2001). In the classic view of morphogens, the FGFs are generally believed to exert their effects by diffusing through tissues (Wolpert 1996), and this may be the case for FGF8 signaling (Toyoda et al. 2010). However, the apical enrichment and polarization of phosphotyrosine (Chenn et al. 1998) and phospho-ERK1/2 (Toyoda et al. 2010) activities in progenitors are highly suggestive that some growth factor signaling originates from the CSF as well.
INSULIN AND INSULIN-LIKE GROWTH FACTOR 1 AND 2 SIGNALING IN THE CEREBROSPINAL FLUID
The embryonic CSF also contains insulin and IGF1 and −2 (Holm et al. 1994, Lehtinen et al. 2011, Margolis & Altszuler 1967). IGF1 and IGF2 regulate prenatal growth and body size (Baker et al. 1993, DeChiara et al. 1991) mainly by binding to the IGF1R, which mediates the proliferative response (Weber et al. 1992). For example, consistent with a role for the IGF1R in the regulation of somatic size, all small dogs show a unique single nucleotide polymorphism in the IGF1R gene that is not found in the genomes of large dogs (Sutter et al. 2007). Furthermore, conditional deletion of IGF1R in neural precursors leads to microcephaly (Kappeler et al. 2008, Lehtinen et al. 2011, Liu et al. 2009). In contrast, IGF1 overexpression promotes S-phase commitment, accelerated cell cycle kinetics, and cell survival, leading to hyperplasia (Hodge et al. 2004, Liu et al. 2009, Mairet-Coello et al. 2009, Popken et al. 2004). IGF1 can also regulate neuronal differentiation, glial development, and cell size (reviewed in D’Ercole & Ye 2008). Downstream of IGF1R signaling, mutations of Irs2 (Schubert et al. 2003), Pdk1 (Chalhoub et al. 2009), and Pten (Groszer et al. 2001), among others, provide genetic tools for manipulating brain size.
The growth- and survival-promoting effects of embryonic CSF during neurogenesis depend in part on CSF-IGF2 (Lehtinen et al. 2011). A transient spike in CSF-IGF2 levels at mid-to late stages of neurogenesis (E16–19 in rat; E16–18 in mice) stimulates the proliferation of neural precursor cells in explant cultures of the developing cortex (Lehtinen et al. 2011) as well as the growth and maintenance of neuro-spheres, an in vitro model of neural stem cells (Vescovi et al. 1993). CSF-IGF2 binds directly to the apical membrane and primary cilia of cortical progenitors. IGF1R expression is also enriched along the apical ventricular surface of progenitors (Lehtinen et al. 2011). The proliferation-inducing effects of CSF appear IGF2 dependent, as gain-of-function and loss-of-function experiments in vitro in explants and neurospheres produce opposing effects on proliferation. Moreover, Igf2-deficient mice have a specific defect in the neurogenesis of the uppermost layers of the cortex (Lehtinen et al. 2011). The choroid plexus expresses Igf2 (McKelvie et al. 1992, Stylanopoulo et al. 1988) and is a source of IGF2 in the developing CSF. Other sources of IGF ligands exist, including the developing vasculature (Dugas et al. 2008), potentially neighboring cells, and perhaps even extraneural sources. Studies involving intra-cerebroventricular injections of IGF1, IGF1-neutralizing antibodies, and IGF1R inhibitors have confirmed that IGF signals delivered in the embryonic CSF trigger proliferative events in the cortical VZ (Mairet-Coello et al. 2009).
Although signaling from the CSF appears important in controlling apical progenitors that contact the ventricle, it is not yet known whether CSF or the apical complex regulates other progenitor populations in the cerebrum, such as the basal progenitors or OSVZ radial progenitors. Whereas the apical, radial glial progenitors maintain contact with the CSF as well as the pial surface, the OSVZ radial progenitors extend only a basal process toward the pia. The radically distinct morphologies of the several progenitor populations likely predispose them to distinct modes of regulation. For example, in contrast to radial glial progenitors, basal and OSVZ progenitors downreg-ulate apical polarity marker expression (Fietz et al. 2010). The degree to which IGF signaling contributes to OSVZ progenitor proliferation also remains to be explored. Although OSVZ cells do not contact the CSF and therefore are unlikely to receive CSF-IGF2 as a primary pro-liferative cue, other sources of IGF2 including the meninges or vasculature may play important roles, or IGF2 may conceivably diffuse into the SVZ at some rate.
The effects of CSF-IGF2 are also strikingly age dependent; they are maximal when CSF-IGF2 levels are highest, near the end of neu-rogenesis, and continue at a lower level postna-tally, but are also modest at the earliest stages of neurogenesis. Thus, IGF2 may specifically regulate one particular aspect of neurogenesis. Interestingly, insulin/IGF-like peptides secreted by glia were recently shown to stimulate quiescent neuroblasts to reenter the cell cycle in Drosophila (Chell & Brand 2010, Sousa-Nunes et al. 2011). An analogous role of IGF signaling in the mammalian brain is an interesting idea that has not been addressed.
SONIC HEDGEHOG SIGNALING IN THE CEREBROSPINAL FLUID
The transventricular delivery of secreted proteins with active roles in the developing brain extends to Shh as well. Recent evidence demonstrates that the hindbrain choroid plexus secretes Shh into the CSF (Huang et al. 2010). Shh stimulates the expansion of a distinct progenitor domain in the hindbrain choroid plexus adjoining the lower rhombic lip that does not itself express Shh but is responsive to it (Huang et al. 2009). In addition to this novel tissue-autonomous role for choroid plexus–secreted Shh, CSF-Shh appears to be a key mitogen for proliferating cerebellar granule precursors. Shh was previously shown to be secreted by Purkinje cells (Dahmane & Ruiz i Altaba 1999, Wallace 1999, Wechsler-Reya & Scott 1999). However, Wnt1-Cre-mediated deletion of Shh in the hindbrain choroid plexus also impairs proliferation of cerebellar granule neuron precursors (Huang et al. 2010), suggesting that choroid plexus–borne Shh acts in both an autocrine and a paracrine manner to instruct choroid plexus and cerebellar development as well.
IGF synergizes with Shh signaling to promote proliferation of healthy cerebellar granule neuron precursors (Fernandez et al. 2010, Ye et al. 1996) as well as tumorigenic cells that lead to medulloblastoma (Corcoran et al. 2008, Hartmann et al. 2005, Rao et al. 2004, Tanori et al. 2010), a childhood tumor that can originate from cerebellar granule neuron precursors (Gibson et al. 2010, Gilbertson & Ellison 2008, Pomeroy et al. 2002). Although the mechanisms by which IGF2 binding to primary cilia influence downstream signaling events remain to be elucidated, Shh is well established to signal via binding to primary cilia (Corbit et al. 2005, Han et al. 2008, Rohatgi et al. 2007). Mutations in genes encoding the basal body proteins Fantom and Ofd1 cause abnormal cerebellar development (Delous et al. 2007, Ferrante et al. 2006, Vierkotten et al. 2007). Primary cilia are also required for Shh-dependent expansion of the cerebellar progenitor cell pool (Spassky et al. 2008). Additionally, mice deficient in primary cilia as a result of Kif3a, Igft88, or Stumpy deletions have profound hypoplasia and foliation defects (Breunig et al. 2008, Chizhikov et al. 2007, Town et al. 2008). It will be interesting in future studies to elucidate the mechanisms by which IGF and Shh signaling may synergize at primary cilia. Shh-dependent signaling is also implicated in the formation and maintenance of adult neural stem cells (Breunig et al. 2008, Han et al. 2008), but the source of Shh and means of distributing it in the adult CSF remain to be fully elucidated.
RETINOIC ACID IN THE CEREBROSPINAL FLUID
Retinoic acid (RA), a hormone signal derived from vitamin A, also acts on the developing forebrain far from its source (Chambon 1996, Haskell & LaMantia 2005, Ribes et al. 2006). The meninges are an important source of RA (Siegenthaler et al. 2009). Meningeal defects in Foxc1 mutant mice, including reduced intermediate progenitor production and diminished expansion of the neuroepithelium, are associated with impaired neurogenesis. Interestingly, these deficiencies are due to reduced RA signaling originating from the dorsal forebrain meninges (Siegenthaler et al. 2009). Because RA has been identified in CSF (Lehtinen et al. 2011, Parada et al. 2008, Redzic et al. 2005), and RA synthetic and catabolic enzymes are expressed in the choroid plexus as well as the meninges (Lehtinen et al. 2011, Siegenthaler et al. 2009), meningeally derived RA may reach the neuroepithelial cells via the lateral ventricular CSF as well as by crossing the cerebral mantle. However, the roles of meningeally derived RA suggest the remarkable range of mechanisms by which signals at both the basal process (Konno et al. 2008, Radakovits et al. 2009) and apical process may play key roles in the maintenance of the apical progenitors, as well as potentially the OSVZ, during neurogenesis (Fietz et al. 2010, Hansen et al. 2010, Reillo et al. 2011).
OTHER POTENTIAL SIGNALING ACTIVITIES IN THE CEREBROSPINAL FLUID
It remains an open question whether other secreted factors that act on the forebrain also act far from their source by diffusing through the CSF. The Wnt signaling pathway plays a fundamental role in regulating early development of the CNS (Freese et al. 2010, Wang & Wynshaw-Boris 2004, Zhou et al. 2006), and Wnt signaling activity has been identified in embryonic CSF (Lehtinen et al. 2011). Similarly, dynamic levels of Bmp signaling activity (Hebert et al. 2002, Shimogori et al. 2004) are also present in the CSF (Lehtinen et al. 2011). Consistent with the coordination of Bmp signaling by the choroid plexus-CSF system, growth and differentiation factors 3 and 8 (Gdf3 and Gdf8), both members of the TGF-β super-family of proteins that can modulate Bmp signaling (Levine & Brivanlou 2006), have been identified in CSF (Lehtinen et al. 2011).
POTENTIAL CLINICAL IMPLICATIONS
Dysregulated Shh signaling is central to medul-loblastoma progression (Gibson et al. 2010, Goodrich et al. 1997, Pomeroy et al. 2002). Shh and IGF signaling synergize to promote cerebellar granule precursor proliferation (Fernandez et al. 2010), for example through stabilization of N-Myc (Kenney et al. 2004). Pten deletion in mice with constitutively active Smoothened also enhances medulloblastoma tumorigenesis (Castellino et al. 2010). Because transventricular delivery of Shh supports the proliferation of cerebellar granule neuron progenitors (Huang et al. 2010) and CSF-IGF2 promotes proliferation of neural stem cells, CSF-borne Shh and/or IGF ligands, among other factors, may serve to exacerbate medulloblastomas.
IGF2 and other diffusible growth factors that drive PI3K signaling and neural progenitor proliferation during development are upregu-lated in some glioblastoma multiforme (GBM) patient tumors as well (Louis 2006, Soroceanu et al. 2007). Some GBM patients also have elevated CSF-IGF2 levels, and high lumbar CSF-IGF2 levels correlate with more advanced or lethal disease (Lehtinen et al. 2011). Moreover, CSF obtained from patients with GBM stimulates neural stem cell proliferation in an IGF2-dependent manner, suggesting that GBM CSF-IGF2 may act in a general autocrine as well as paracrine manner on putative tumor cells. The Shh and IGF2 data suggest the intriguing speculation that the protein content of the CSF may represent a brain tumor risk factor.
IGF signaling in the CNS has been implicated in a remarkable diversity of processes, including longevity and aging (Kenyon 2010) and axon outgrowth (Ozdinler & Macklis 2006), and IGF2 was recently shown to promote memory consolidation and enhancement (Chen et al. 2011). These diverse functions raise provocative questions about how the availability of IGF ligands in CSF may regulate postnatal neurogenesis as well as these other functions. Given the active role of CSF in instructing neurogenesis in the developing cortex, modulation of the proteomic composition of the CSF may provide new and unanticipated ways to regulate the CNS in health and disease.
CONCLUSION
Recent work from many labs suggests a reevaluation of the role of cell-intrinsic and cell-extrinsic cues during neurogenesis in the developing cerebral cortex. In addition to regulating progenitor proliferation, the apical complex appears to regulate cell survival and primary cilia. The cell polarity imparted by the apical complex allows for an enrichment of growth factor receptors along the apical surface of cortical progenitor cells, which strongly suggests that progenitors selectively sample the CSF for diffusible signals that instruct neurogenesis. Indeed, compelling evidence from several brain regions and different species supports a new model in which diffusible signals distributed by the CSF support the growth and survival of the developing brain (Figure 4). Collectively, these findings suggest that the embryonic CSF forms an integral component of the embryonic neural stem cell niche. Because the apical complex polarizes neural progenitor cells and helps anchor them to the ventricular surface, one possibility is that the apical complex may control neurogenesis by regulating progenitor access to the growth-promoting factors that circulate in the CSF. Although initial studies are exploring the active role of the CSF in the adult CNS, whether the CSF plays an essential, active role in maintaining adult neurogenesis and the health of the adult CNS, as well as whether it contributes to the pathogenesis of disease, remain open questions.
SUMMARY POINTS.
Apical complex proteins are necessary for progenitor cell proliferation.
The apical complex is also critical for cell survival.
The apical complex allows neural progenitor cells to integrate cell-intrinsic and cell-extrinsic signals.
The apical complex may regulate neuronal differentiation by controlling progenitor access to the CSF.
The CSF is a rich and dynamic source of proteins for the brain.
The enrichment of growth factor receptors along the ventricular surface of the developing brain allows progenitors to receive signals distributed in the CSF.
Embryonic CSF provides growth- and survival-promoting factors to the developing brain.
The active distribution of diffusible signals in the CNS may extend to the regulation of CNS diseases including brain tumors.
FUTURE ISSUES.
How can we better understand the sources and regulation of CSF-distributed factors?
How might local gradients and ventricle-specific CSF composition influence the development and functioning of distinct brain regions?
Do CSF-distributed factors play an active role in instructing adult neural stem cells as well?
Brain development is typically considered to take place independently of development in the rest of the body. Given the role of CSF in guiding development and the distribution of extraneural signals therein, to what extent do signals originating outside the CNS actually influence CNS development and maintenance?
ACKNOWLEDGMENTS
We thank our colleagues in the Walsh laboratory for helpful discussions. We apologize to investigators whose work we could not cite due to space limitations. We are grateful for support from the NIH [award numbers K99 NS072192, R01 NS032457, and R01 NS35129 from the National Institute of Neurological Disorders and Stroke (NINDS)], the Manton Center for Orphan Disease Research, and the Intellectual and Developmental Disabilities Research Centers (CHB DDRC, P30 HD18655). C.A.W. is an Investigator of the Howard Hughes Medical Institute. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NINDS or the NIH.
Glossary
- Neural progenitor cell
a neural stem cell that gives rise to future neurons in the brain
- Neurogenesis
the process of generating neurons from neural progenitor cells
- Adherens junctions
ring-like, cadherin-containing contacts between cells that regulate cell-cell adhesion
- VZ
ventricular zone
- SVZ
subventricular zone
- OSVZ
outer subventricular zone
- Apical complex
Par (partitioning defective) polarity proteins that in mammalian neural precursors assemble as three complexes: Crb/Pals1/Patj, Par3/Par6/aPKC, and Mals/Pals1
- Cerebrospinal fluid (CSF)
fluid that fills the ventricles, spinal canal, and subarachnoid space surrounding the central nervous system
- Par3/Par6/aPKC
a partitioning-defective “Par” protein complex known as the apical complex in mammalian cells
- Shh
Sonic hedgehog
- IGF
insulin-like growth factor
- FGF
fibroblast growth factor
- RA
retinoic acid
- GBM
glioblastoma multiforme
Footnotes
DISCLOSURE STATEMENT
The authors are not aware of any affiliations, memberships, funding, or financial holdings that might be perceived as affecting the objectivity of this review.
Contributor Information
Maria K. Lehtinen, Email: maria.lehtinen@childrens.harvard.edu.
Christopher A. Walsh, Email: christopher.walsh@childrens.harvard.edu.
LITERATURE CITED
- Aaku-Saraste E, Oback B, Hellwig A, Huttner WB. Neuroepithelial cells downregulate their plasma membrane polarity prior to neural tube closure and neurogenesis. Mech. Dev. 1997;69:71–81. doi: 10.1016/s0925-4773(97)00156-1. [DOI] [PubMed] [Google Scholar]
- Agnati LF, Zoli M, Stromberg I, Fuxe K. Intercellular communication in the brain: wiring versus volume transmission. Neuroscience. 1995;69:711–726. doi: 10.1016/0306-4522(95)00308-6. [DOI] [PubMed] [Google Scholar]
- Angevine JB, Jr., Bodian D, Coulombre AJ, Edds MVJr, Hamburger V, et al. Embryonic vertebrate central nervous system: revised terminology. Anat. Rec. 1970;166:257–261. doi: 10.1002/ar.1091660214. [DOI] [PubMed] [Google Scholar]
- Awatramani R, Soriano P, Rodriguez C, Mai JJ, Dymecki SM. Cryptic boundaries in roof plate and choroid plexus identified by intersectional gene activation. Nat. Genet. 2003;35:70–75. doi: 10.1038/ng1228. [DOI] [PubMed] [Google Scholar]
- Baker J, Liu JP, Robertson EJ, Efstratiadis A. Role of insulin-like growth factors in embryonic and postnatal growth. Cell. 1993;75:73–82. [PubMed] [Google Scholar]
- Berdnik D, Torok T, Gonzalez-Gaitan M, Knoblich JA. The endocytic protein α-Adaptin is required for Numb-mediated asymmetric cell division in Drosophila . Dev. Cell. 2002;3:221–231. doi: 10.1016/s1534-5807(02)00215-0. [DOI] [PubMed] [Google Scholar]
- Bilder D, Li M, Perrimon N. Cooperative regulation of cell polarity and growth by Drosophila tumor suppressors. Science. 2000;289:113–116. doi: 10.1126/science.289.5476.113. [DOI] [PubMed] [Google Scholar]
- Breunig JJ, Sarkisian MR, Arellano JI, Morozov YM, Ayoub AE, et al. Primary cilia regulate hippocampal neurogenesis by mediating sonic hedgehog signaling. Proc. Natl. Acad. Sci. USA. 2008;105:13127–13132. doi: 10.1073/pnas.0804558105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bultje RS, Castaneda-Castellanos DR, Jan LY, Jan YN, Kriegstein AR, Shi SH. Mammalian Par3 regulates progenitor cell asymmetric division via Notch signaling in the developing neocortex. Neuron. 2009;63:189–202. doi: 10.1016/j.neuron.2009.07.004. Showed that Par3 distributes unequally in cells and acts as a cell-autonomous regulator of Notch activity.
- Cappello S, Attardo A, Wu X, Iwasato T, Itohara S, et al. The Rho-GTPase cdc42 regulates neural progenitor fate at the apical surface. Nat. Neurosci. 2006;9:1099–1107. doi: 10.1038/nn1744. [DOI] [PubMed] [Google Scholar]
- Castellino RC, Barwick BG, Schniederjan M, Buss MC, Becher O, et al. Heterozygosity for Pten promotes tumorigenesis in a mouse model of medulloblastoma. PLoS ONE. 2010;5:e10849. doi: 10.1371/journal.pone.0010849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cavanagh ME, Cornelis ME, Dziegielewska KM, Evans CA, Lorscheider FL, et al. Comparison of proteins in CSF of lateral and IVth ventricles during early development of fetal sheep. Dev. Brain Res. 1983;11:159–167. doi: 10.1016/0165-3806(83)90213-4. [DOI] [PubMed] [Google Scholar]
- Chae TH, Kim S, Marz KE, Hanson PI, Walsh CA. The hyh mutation uncovers roles for αSnap in apical protein localization and control of neural cell fate. Nat. Genet. 2004;36:264–270. doi: 10.1038/ng1302. [DOI] [PubMed] [Google Scholar]
- Chalhoub N, Zhu G, Zhu X, Baker SJ. Cell type specificity of PI3K signaling in Pdk1- and Pten-deficient brains. Genes Dev. 2009;23:1619–1624. doi: 10.1101/gad.1799609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chambon P. A decade of molecular biology of retinoic acid receptors. FASEB J. 1996;10:940–954. [PubMed] [Google Scholar]
- Chell JM, Brand AH. Nutrition-responsive glia control exit of neural stem cells from quiescence. Cell. 2010;143:1161–1173. doi: 10.1016/j.cell.2010.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen DY, Stern SA, Garcia-Osta A, Saunier-Rebori B, Pollonini G, et al. A critical role for IGF-II in memory consolidation and enhancement. Nature. 2011;469:491–497. doi: 10.1038/nature09667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chenn A, McConnell SK. Cleavage orientation and the asymmetric inheritance of Notch1 immunore-activity in mammalian neurogenesis. Cell. 1995;82:631–641. doi: 10.1016/0092-8674(95)90035-7. [DOI] [PubMed] [Google Scholar]
- Chenn A, Walsh CA. Regulation of cerebral cortical size by control of cell cycle exit in neural precursors. Science. 2002;297:365–369. doi: 10.1126/science.1074192. [DOI] [PubMed] [Google Scholar]
- Chenn A, Zhang YA, Chang BT, McConnell SK. Intrinsic polarity of mammalian neuroepithelial cells. Mol. Cell. Neurosci. 1998;11:183–193. doi: 10.1006/mcne.1998.0680. [DOI] [PubMed] [Google Scholar]
- Chizhikov VV, Davenport J, Zhang Q, Shih EK, Cabello OA, et al. Cilia proteins control cerebellar morphogenesis by promoting expansion of the granule progenitor pool. J. Neurosci. 2007;27:9780–9789. doi: 10.1523/JNEUROSCI.5586-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clevers H. Wnt/β-catenin signaling in development and disease. Cell. 2006;127:469–480. doi: 10.1016/j.cell.2006.10.018. [DOI] [PubMed] [Google Scholar]
- Cohen E, Binet S, Meininger V. Ciliogenesis and centriole formation in the mouse embryonic nervous system. An ultrastructural analysis. Biol. Cell. 1988;62:165–169. [PubMed] [Google Scholar]
- Corbit KC, Aanstad P, Singla V, Norman AR, Stainier DY, Reiter JF. Vertebrate Smoothened functions at the primary cilium. Nature. 2005;437:1018–1021. doi: 10.1038/nature04117. [DOI] [PubMed] [Google Scholar]
- Corcoran RB, Bachar Raveh T, Barakat MT, Lee EY, Scott MP. Insulin-like growth factor 2 is required for progression to advanced medulloblastoma in patched1 heterozygous mice. Cancer Res. 2008;68:8788–8795. doi: 10.1158/0008-5472.CAN-08-2135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Costa MR, Wen G, Lepier A, Schroeder T, Gotz M. Par-complex proteins promote proliferative progenitor divisions in the developing mouse cerebral cortex. Development. 2008;135:11–22. doi: 10.1242/dev.009951. Demonstrated that Par3 and Par6 are key players in promoting progenitor polarity and proliferation in the developing cerebral cortex.
- Currle DS, Cheng X, Hsu C-m, Monuki ES. Direct and indirect roles of CNS dorsal midline cells in choroid plexus epithelial formation. Development. 2005;132:3549–3559. doi: 10.1242/dev.01915. [DOI] [PubMed] [Google Scholar]
- Cushing H. Studies on the cerebro-spinal fluid. J. Med. Res. 1914;31:1–19. [PMC free article] [PubMed] [Google Scholar]
- Dahmane N, Ruiz i Altaba A. Sonic hedgehog regulates the growth and patterning of the cerebellum. Development. 1999;126:3089–3100. doi: 10.1242/dev.126.14.3089. [DOI] [PubMed] [Google Scholar]
- DeChiara TM, Robertson EJ, Efstratiadis A. Parental imprinting of the mouse insulin-like growth factor II gene. Cell. 1991;64:849–859. doi: 10.1016/0092-8674(91)90513-x. [DOI] [PubMed] [Google Scholar]
- Delous M, Baala L, Salomon R, Laclef C, Vierkotten J, et al. The ciliary gene RPGRIP1L is mutated in cerebello-oculo-renal syndrome (Joubert syndrome type B) and Meckel syndrome. Nat. Genet. 2007;39:875–881. doi: 10.1038/ng2039. [DOI] [PubMed] [Google Scholar]
- D’Ercole AJ, Ye P. Expanding the mind: IGF-I and brain development. Endocrinology. 2008;149:5958–5962. doi: 10.1210/en.2008-0920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desmond ME, Jacobson AG. Embryonic brain enlargement requires cerebrospinal fluid pressure. Dev. Biol. 1977;57:188–198. doi: 10.1016/0012-1606(77)90364-5. [DOI] [PubMed] [Google Scholar]
- Desmond ME, Levitan ML, Haas AR. Internal luminal pressure during early chick embryonic brain growth: descriptive and empirical observations. Anat. Rec. A. 2005;285:737–747. doi: 10.1002/ar.a.20211. [DOI] [PubMed] [Google Scholar]
- Dubreuil V, Marzesco AM, Corbeil D, Huttner WB, Wilsch-Brauninger M. Midbody and primary cilium of neural progenitors release extracellular membrane particles enriched in the stem cell marker prominin-1. J. Cell Biol. 2007;176:483–495. doi: 10.1083/jcb.200608137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dugas JC, Mandemakers W, Rogers M, Ibrahim A, Daneman R, Barres BA. A novel purification method for CNS projection neurons leads to the identification of brain vascular cells as a source of trophic support for corticospinal motor neurons. J. Neurosci. 2008;28:8294–8305. doi: 10.1523/JNEUROSCI.2010-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dziegielewska K, Evans C, Lai P, Lorscheider F, Malinowska D, et al. Proteins in cerebrospinal fluid and plasma of fetal rats during development. Dev. Biol. 1981;83:193–200. doi: 10.1016/s0012-1606(81)80024-3. [DOI] [PubMed] [Google Scholar]
- Englund C, Fink A, Lau C, Pham D, Daza RA, et al. Pax6, Tbr2, and Tbr1 are expressed sequentially by radial glia, intermediate progenitor cells, and postmitotic neurons in developing neocortex. J. Neurosci. 2005;25:247–251. doi: 10.1523/JNEUROSCI.2899-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng W, Wu H, Chan LN, Zhang M. Par-3-mediated junctional localization of the lipid phosphatase PTEN is required for cell polarity establishment. J. Biol. Chem. 2008;283:23440–23449. doi: 10.1074/jbc.M802482200. [DOI] [PubMed] [Google Scholar]
- Fernandez C, Tatard VM, Bertrand N, Dahmane N. Differential modulation of Sonic-hedgehog-induced cerebellar granule cell precursor proliferation by the IGF signaling network. Dev. Neurosci. 2010;32:59–70. doi: 10.1159/000274458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferrante MI, Zullo A, Barra A, Bimonte S, Messaddeq N, et al. Oral-facial-digital type I protein is required for primary cilia formation and left-right axis specification. Nat. Genet. 2006;38:112–117. doi: 10.1038/ng1684. [DOI] [PubMed] [Google Scholar]
- Fietz SA, Huttner WB. Cortical progenitor expansion, self-renewal and neurogenesis—a polarized perspective. Curr. Opin. Neurobiol. 2011;21:23–35. doi: 10.1016/j.conb.2010.10.002. [DOI] [PubMed] [Google Scholar]
- Fietz SA, Kelava I, Vogt J, Wilsch-Brauninger M, Stenzel D, et al. OSVZ progenitors of human and ferret neocortex are epithelial-like and expand by integrin signaling. Nat. Neurosci. 2010;13:690–699. doi: 10.1038/nn.2553. [DOI] [PubMed] [Google Scholar]
- Finch PW, Cunha GR, Rubin JS, Wong J, Ron D. Pattern of keratinocyte growth factor and ker-atinocyte growth factor receptor expression during mouse fetal development suggests a role in mediating morphogenetic mesenchymal-epithelial interactions. Dev. Dyn. 1995;203:223–240. doi: 10.1002/aja.1002030210. [DOI] [PubMed] [Google Scholar]
- Freese JL, Pino D, Pleasure SJ. Wnt signaling in development and disease. Neurobiol. Dis. 2010;38:148–153. doi: 10.1016/j.nbd.2009.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fukuchi-Shimogori T, Grove EA. Neocortex patterning by the secreted signaling molecule FGF8. Science. 2001;294:1071–1074. doi: 10.1126/science.1064252. [DOI] [PubMed] [Google Scholar]
- Gaiano N, Nye JS, Fishell G. Radial glial identity is promoted by Notch1 signaling in the murine forebrain. Neuron. 2000;26:395–404. doi: 10.1016/s0896-6273(00)81172-1. [DOI] [PubMed] [Google Scholar]
- Gal JS, Morozov YM, Ayoub AE, Chatterjee M, Rakic P, Haydar TF. Molecular and morphological heterogeneity of neural precursors in the mouse neocortical proliferative zones. J. Neurosci. 2006;26:1045–1056. doi: 10.1523/JNEUROSCI.4499-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghosh S, Marquardt T, Thaler JP, Carter N, Andrews SE, et al. Instructive role of aPKCζ subcellular localization in the assembly of adherens junctions in neural progenitors. Proc. Natl. Acad. Sci. USA. 2008;105:335–340. doi: 10.1073/pnas.0705713105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibson P, Tong Y, Robinson G, Thompson MC, Currle DS, et al. Subtypes of medulloblastoma have distinct developmental origins. Nature. 2010;468:1095–1099. doi: 10.1038/nature09587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilbertson RJ, Ellison DW. The origins of medulloblastoma subtypes. Annu. Rev. Pathol. 2008;3:341–365. doi: 10.1146/annurev.pathmechdis.3.121806.151518. [DOI] [PubMed] [Google Scholar]
- Goodrich LV, Milenkovic L, Higgins KM, Scott MP. Altered neural cell fates and medulloblastoma in mouse patched mutants. Science. 1997;277:1109–1113. doi: 10.1126/science.277.5329.1109. [DOI] [PubMed] [Google Scholar]
- Gorivodsky M, Mukhopadhyay M, Wilsch-Braeuninger M, Phillips M, Teufel A, et al. Intraflagellar transport protein 172 is essential for primary cilia formation and plays a vital role in patterning the mammalian brain. Dev. Biol. 2009;325:24–32. doi: 10.1016/j.ydbio.2008.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gotz M, Huttner WB. The cell biology of neurogenesis. Nat. Rev. Mol. Cell Biol. 2005;6:777–788. doi: 10.1038/nrm1739. [DOI] [PubMed] [Google Scholar]
- Groszer M, Erickson R, Scripture-Adams DD, Lesche R, Trumpp A, et al. Negative regulation of neural stem/progenitor cell proliferation by the Pten tumor suppressor gene in vivo. Science. 2001;294:2186–2189. doi: 10.1126/science.1065518. [DOI] [PubMed] [Google Scholar]
- Grove EA, Fukuchi-Shimogori T. Generating the cerebral cortical area map. Annu. Rev. Neurosci. 2003;26:355–380. doi: 10.1146/annurev.neuro.26.041002.131137. [DOI] [PubMed] [Google Scholar]
- Han YG, Alvarez-Buylla A. Role of primary cilia in brain development and cancer. Curr. Opin. Neurobiol. 2010;20:58–67. doi: 10.1016/j.conb.2009.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han YG, Spassky N, Romaguera-Ros M, Garcia-Verdugo JM, Aguilar A, et al. Hedgehog signaling and primary cilia are required for the formation of adult neural stem cells. Nat. Neurosci. 2008;11:277–284. doi: 10.1038/nn2059. [DOI] [PubMed] [Google Scholar]
- Hansen DV, Lui JH, Parker PR, Kriegstein AR. Neurogenic radial glia in the outer subventricular zone of human neocortex. Nature. 2010;464:554–561. doi: 10.1038/nature08845. [DOI] [PubMed] [Google Scholar]
- Harris TJ, Tepass U. Adherens junctions: from molecules to morphogenesis. Nat. Rev. Mol. Cell Biol. 2010;11:502–514. doi: 10.1038/nrm2927. [DOI] [PubMed] [Google Scholar]
- Hartmann W, Koch A, Brune H, Waha A, Schuller U, et al. Insulin-like growth factor II is involved in the proliferation control of medulloblastoma and its cerebellar precursor cells. Am. J. Pathol. 2005;166:1153–1162. doi: 10.1016/S0002-9440(10)62335-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haskell GT, LaMantia AS. Retinoic acid signaling identifies a distinct precursor population in the developing and adult forebrain. J. Neurosci. 2005;25:7636–7647. doi: 10.1523/JNEUROSCI.0485-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haubensak W, Attardo A, Denk W, Huttner WB. Neurons arise in the basal neuroepithelium of the early mammalian telencephalon: a major site of neurogenesis. Proc. Natl. Acad. Sci. USA. 2004;101:3196–3201. doi: 10.1073/pnas.0308600100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hebert JM, Mishina Y, McConnell SK. BMP signaling is required locally to pattern the dorsal telen-cephalic midline. Neuron. 2002;35:1029–1041. doi: 10.1016/s0896-6273(02)00900-5. [DOI] [PubMed] [Google Scholar]
- Hinds JW, Ruffett TL. Cell proliferation in the neural tube: an electron microscopic and Golgi analysis in the mouse cerebral vesicle. Z. Zellforsch. Mikrosk. Anat. 1971;115:226–264. doi: 10.1007/BF00391127. [DOI] [PubMed] [Google Scholar]
- Hirabayashi Y, Itoh Y, Tabata H, Nakajima K, Akiyama T, et al. The Wnt/β-catenin pathway directs neuronal differentiation of cortical neural precursor cells. Development. 2004;131:2791–2801. doi: 10.1242/dev.01165. [DOI] [PubMed] [Google Scholar]
- Hirata H, Yoshiura S, Ohtsuka T, Bessho Y, Harada T, et al. Oscillatory expression of the bHLH factor Hes1 regulated by a negative feedback loop. Science. 2002;298:840–843. doi: 10.1126/science.1074560. [DOI] [PubMed] [Google Scholar]
- Hodge RD, D’Ercole AJ, O’Kusky JR. Insulin-like growth factor-I accelerates the cell cycle by decreasing G1 phase length and increases cell cycle reentry in the embryonic cerebral cortex. J. Neurosci. 2004;24:10201–10210. doi: 10.1523/JNEUROSCI.3246-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holm NR, Hansen LB, Nilsson C, Gammeltoft S. Gene expression and secretion of insulin-like growth factor-II and insulin-like growth factor binding protein-2 from cultured sheep choroid plexus epithelial cells. Brain Res. Mol. Brain Res. 1994;21:67–74. doi: 10.1016/0169-328x(94)90379-4. [DOI] [PubMed] [Google Scholar]
- Hoskins R, Hajnal AF, Harp SA, Kim SK. The C. elegans vulval induction gene lin-2 encodes a member of the MAGUK family of cell junction proteins. Development. 1996;122:97–111. doi: 10.1242/dev.122.1.97. [DOI] [PubMed] [Google Scholar]
- Hu H. Chemorepulsion of neuronal migration by Slit2 in the developing mammalian forebrain. Neuron. 1999;23:703–711. doi: 10.1016/s0896-6273(01)80029-5. [DOI] [PubMed] [Google Scholar]
- Huang X, Ketova T, Fleming JT, Wang H, Dey SK, et al. Sonic hedgehog signaling regulates a novel epithelial progenitor domain of the hindbrain choroid plexus. Development. 2009;136:2535–2543. doi: 10.1242/dev.033795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang X, Liu J, Ketova T, Fleming JT, Grover VK, et al. Transventricular delivery of Sonic hedgehog is essential to cerebellar ventricular zone development. Proc. Natl. Acad. Sci. USA. 2010;107:8422–8427. doi: 10.1073/pnas.0911838107. Demonstrated that hindbrain Shh signaling, distributed in the CSF, regulates choroid plexus and cerebellar development.
- Imai F, Hirai S, Akimoto K, Koyama H, Miyata T, et al. Inactivation of aPKC.results in the loss of adherens junctions in neuroepithelial cells without affecting neurogenesis in mouse neocortex. Development. 2006;133:1735–1744. doi: 10.1242/dev.02330. [DOI] [PubMed] [Google Scholar]
- Imayoshi I, Sakamoto M, Yamaguchi M, Mori K, Kageyama R. Essential roles of Notch signaling in maintenance of neural stem cells in developing and adult brains. J. Neurosci. 2010;30:3489–3498. doi: 10.1523/JNEUROSCI.4987-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iwata T, Hevner RF. Fibroblast growth factor signaling in development of the cerebral cortex. Dev. Growth Differ. 2009;51:299–323. doi: 10.1111/j.1440-169X.2009.01104.x. [DOI] [PubMed] [Google Scholar]
- Jan YN, Jan LY. Asymmetric cell division in the Drosophila nervous system. Nat. Rev. Neurosci. 2001;2:772–779. doi: 10.1038/35097516. [DOI] [PubMed] [Google Scholar]
- Junghans D, Hack I, Frotscher M, Taylor V, Kemler R. p-catenin-mediated cell-adhesion is vital for embryonic forebrain development. Dev. Dyn. 2005;233:528–539. doi: 10.1002/dvdy.20365. [DOI] [PubMed] [Google Scholar]
- Kamberov E, Makarova O, Roh M, Liu A, Karnak D, et al. Molecular cloning and characterization of Pals, proteins associated with mLin-7. J. Biol. Chem. 2000;275:11425–11431. doi: 10.1074/jbc.275.15.11425. [DOI] [PubMed] [Google Scholar]
- Kappeler L, De Magalhaes Filho C, Dupont J, Leneuve P, Cervera P, et al. Brain IGF-1 receptors control mammalian growth and lifespan through a neuroendocrine mechanism. PLoS Biol. 2008;6:e254. doi: 10.1371/journal.pbio.0060254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawaguchi A, Ikawa T, Kasukawa T, Ueda HR, Kurimoto K, et al. Single-cell gene profiling defines differential progenitor subclasses in mammalian neurogenesis. Development. 2008;135:3113–3124. doi: 10.1242/dev.022616. [DOI] [PubMed] [Google Scholar]
- Kemphues KJ, Priess JR, Morton DG, Cheng NS. Identification of genes required for cytoplasmic localization in early C. elegans embryos. Cell. 1988;52:311–320. doi: 10.1016/s0092-8674(88)80024-2. [DOI] [PubMed] [Google Scholar]
- Kenney AM, Widlund HR, Rowitch DH. Hedgehog and PI-3 kinase signaling converge on Nmyc1 to promote cell cycle progression in cerebellar neuronal precursors. Development. 2004;131:217–228. doi: 10.1242/dev.00891. [DOI] [PubMed] [Google Scholar]
- Kenyon CJ. The genetics of aging. Nature. 2010;464:504–512. doi: 10.1038/nature08980. [DOI] [PubMed] [Google Scholar]
- Kim S, Lehtinen MK, Sessa A, Zappaterra MW, Cho SH, et al. The apical complex couples cell fate and cell survival to cerebral cortical development. Neuron. 2010;66:69–84. doi: 10.1016/j.neuron.2010.03.019. Showed that Pals1 and the apical complex are indispensable for progenitor proliferation, survival of newly generated neurons, and cerebral cortex development.
- Klezovitch O, Fernandez TE, Tapscott SJ, Vasioukhin V. Loss of cell polarity causes severe brain dysplasia in Lgl1 knockout mice. Genes Dev. 2004;18:559–571. doi: 10.1101/gad.1178004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knoblich JA. Mechanisms of asymmetric stem cell division. Cell. 2008;132:583–597. doi: 10.1016/j.cell.2008.02.007. [DOI] [PubMed] [Google Scholar]
- Konno D, Shioi G, Shitamukai A, Mori A, Kiyonari H, et al. Neuroepithelial progenitors undergo LGN-dependent planar divisions to maintain self-renewability during mammalian neurogenesis. Nat. Cell Biol. 2008;10:93–101. doi: 10.1038/ncb1673. [DOI] [PubMed] [Google Scholar]
- Kosodo Y, Roper K, Haubensak W, Marzesco AM, Corbeil D, Huttner WB. Asymmetric distribution of the apical plasma membrane during neurogenic divisions of mammalian neuroepithelial cells. EMBO J. 2004;23:2314–2324. doi: 10.1038/sj.emboj.7600223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kriegstein A, Alvarez-Buylla A. The glial nature of embryonic and adult neural stem cells. Annu. Rev. Neurosci. 2009;32:149–184. doi: 10.1146/annurev.neuro.051508.135600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kriegstein A, Noctor S, Martinez-Cerdeno V. Patterns of neural stem and progenitor cell division may underlie evolutionary cortical expansion. Nat. Rev. Neurosci. 2006;7:883–890. doi: 10.1038/nrn2008. [DOI] [PubMed] [Google Scholar]
- Kuwahara A, Hirabayashi Y, Knoepfler PS, Taketo MM, Sakai J, et al. Wnt signaling and its downstream target N-myc regulate basal progenitors in the developing neocortex. Development. 2010;137:1035–1044. doi: 10.1242/dev.046417. [DOI] [PubMed] [Google Scholar]
- Lehtinen MK, Zappaterra MW, Chen X, Yang Y, Hill A, et al. The cerebrospinal fluid provides a proliferative niche for neural progenitor cells. Neuron. 2011;69:893–905. doi: 10.1016/j.neuron.2011.01.023. Showed that the CSF, and in particular CSF-IGF2, provides essential growth- and survival-promoting cues for neural stem cells.
- Levine AJ, Brivanlou AH. GDF3 at the crossroads of TGF-|3 signaling. Cell Cycle. 2006;5:1069–1073. doi: 10.4161/cc.5.10.2771. [DOI] [PubMed] [Google Scholar]
- Li HS, Wang D, Shen Q, Schonemann MD, Gorski JA, et al. Inactivation of Numb and Numblike in embryonic dorsal forebrain impairs neurogenesis and disrupts cortical morphogenesis. Neuron. 2003;40:1105–1118. doi: 10.1016/s0896-6273(03)00755-4. [DOI] [PubMed] [Google Scholar]
- Lien WH, Klezovitch O, Fernandez TE, Delrow J, Vasioukhin V. αE-catenin controls cerebral cortical size by regulating the hedgehog signaling pathway. Science. 2006;311:1609–1612. doi: 10.1126/science.1121449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu W, Ye P, O’Kusky JR, D’Ercole AJ. Type 1 insulin-like growth factor receptor signaling is essential for the development of the hippocampal formation and dentate gyrus. J. Neurosci. Res. 2009;87:2821–2832. doi: 10.1002/jnr.22129. [DOI] [PubMed] [Google Scholar]
- Louis DN. Molecular pathology of malignant gliomas. Annu. Rev. Pathol. 2006;1:97–117. doi: 10.1146/annurev.pathol.1.110304.100043. [DOI] [PubMed] [Google Scholar]
- Louvi A, Grove EA. Cilia in the CNS: the quiet organelle claims center stage. Neuron. 2011;69:1046–1060. doi: 10.1016/j.neuron.2011.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lowery LA, Sive H. Totally tubular: the mystery behind function and origin of the brain ventricular system. BioEssays. 2009;31:446–458. doi: 10.1002/bies.200800207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Machon O, Backman M, Machonova O, Kozmik Z, Vacik T, et al. A dynamic gradient of Wnt signaling controls initiation of neurogenesis in the mammalian cortex and cellular specification in the hippocampus. Dev. Biol. 2007;311:223–237. doi: 10.1016/j.ydbio.2007.08.038. [DOI] [PubMed] [Google Scholar]
- Machon O, van den Bout CJ, Backman M, Kemler R, Krauss S. Role of β-catenin in the developing cortical and hippocampal neuroepithelium. NeuroScience. 2003;122:129–143. doi: 10.1016/s0306-4522(03)00519-0. [DOI] [PubMed] [Google Scholar]
- Mairet-Coello G, Tury A, DiCicco-Bloom E. Insulin-like growth factor-1 promotes G1/S cell cycle progression through bidirectional regulation of cyclins and cyclin-dependent kinase inhibitors via the phosphatidylinositol 3-kinase/Akt pathway in developing rat cerebral cortex. J. Neurosci. 2009;29:775–788. doi: 10.1523/JNEUROSCI.1700-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manabe N, Hirai S, Imai F, Nakanishi H, Takai Y, Ohno S. Association of ASIP/mPAR-3 with adherens junctions of mouse neuroepithelial cells. Dev. Dyn. 2002;225:61–69. doi: 10.1002/dvdy.10139. [DOI] [PubMed] [Google Scholar]
- Manzini MC, Walsh CA. What disorders of cortical development tell us about the cortex: one plus one does not always make two. Curr. Opin. Genet. Dev. 2011;21:333–339. doi: 10.1016/j.gde.2011.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Margolis B, Borg JP. Apicobasal polarity complexes. J. Cell Sci. 2005;118:5157–5159. doi: 10.1242/jcs.02597. [DOI] [PubMed] [Google Scholar]
- Margolis RU, Altszuler N. Insulin in the cerebrospinal fluid. Nature. 1967;215:1375–1376. doi: 10.1038/2151375a0. [DOI] [PubMed] [Google Scholar]
- Marthiens V, ffrench-Constant C. Adherens junction domains are split by asymmetric division of embryonic neural stem cells. EMBO Rep. 2009;10:515–520. doi: 10.1038/embor.2009.36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin C, Alonso MI, Santiago C, Moro JA, De la Mano A, et al. Early embryonic brain development in rats requires the trophic influence of cerebrospinal fluid. Int. J. Dev. Neurosci. 2009;27:733–740. doi: 10.1016/j.ijdevneu.2009.06.002. [DOI] [PubMed] [Google Scholar]
- Martin C, Bueno D, Alonso MI, Moro JA, Callejo S, et al. FGF2 plays a key role in embryonic cerebrospinal fluid trophic properties over chick embryo neuroepithelial stem cells. Dev. Biol. 2006;297:402–416. doi: 10.1016/j.ydbio.2006.05.010. Showed that CSF-FGF2 stimulates proliferation of chick midbrain progenitor cells.
- Martin FH, Seoane JR, Baile CA. Feeding in satiated sheep elicited by intraventricular injections of CSF from fasted sheep. Life Sci. 1973;13:177–184. doi: 10.1016/0024-3205(73)90193-8. [DOI] [PubMed] [Google Scholar]
- Martinez-Cerdeno V, Noctor SC, Kriegstein AR. The role of intermediate progenitor cells in the evolutionary expansion of the cerebral cortex. Cereb. Cortex. 2006;16(Suppl. 1):152–161. doi: 10.1093/cercor/bhk017. [DOI] [PubMed] [Google Scholar]
- Mashayekhi F, Draper CE, Bannister CM, Pourghasem M, Owen-Lynch PJ, Miyan JA. Deficient cortical development in the hydrocephalic Texas (H-Tx) rat: a role for CSF. Brain. 2002;125:1859–1874. doi: 10.1093/brain/awf182. [DOI] [PubMed] [Google Scholar]
- Mason HA, Rakowiecki SM, Gridley T, Fishell G. Loss of Notch activity in the developing central nervous system leads to increased cell death. Dev. Neurosci. 2006;28:49–57. doi: 10.1159/000090752. [DOI] [PubMed] [Google Scholar]
- McKelvie PA, Rosen KM, Kinney HC, Villa-Komaroff L. Insulin-like growth factor II expression in the developing human brain. Neuropathol. Exp. Neurol. 1992;51:464–471. doi: 10.1097/00005072-199207000-00009. [DOI] [PubMed] [Google Scholar]
- Mission JP, Takahashi T, Caviness VS., Jr. Ontogeny of radial and other astroglial cells in murine cerebral cortex. Glia. 1991;4:138–148. doi: 10.1002/glia.440040205. [DOI] [PubMed] [Google Scholar]
- Mizutani K, Saito T. Progenitors resume generating neurons after temporary inhibition of neurogenesis by Notch activation in the mammalian cerebral cortex. Development. 2005;132:1295–1304. doi: 10.1242/dev.01693. [DOI] [PubMed] [Google Scholar]
- Mizutani K, Yoon K, Dang L, Tokunaga A, Gaiano N. Differential Notch signalling distinguishes neural stem cells from intermediate progenitors. Nature. 2007;449:351–355. doi: 10.1038/nature06090. [DOI] [PubMed] [Google Scholar]
- Munji RN, Choe Y, Li G, Siegenthaler JA, Pleasure SJ. Wnt signaling regulates neuronal differentiation of cortical intermediate progenitors. J. Neurosci. 2011;31:1676–1687. doi: 10.1523/JNEUROSCI.5404-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mutch CA, Funatsu N, Monuki ES, Chenn A. β-catenin signaling levels in progenitors influence the laminar cell fates of projection neurons. J. Neurosci. 2009;29:13710–13719. doi: 10.1523/JNEUROSCI.3022-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mutch CA, Schulte JD, Olson E, Chenn A. β-catenin signaling negatively regulates intermediate progenitor population numbers in the developing cortex. PLoS ONE. 2010;5:e12376. doi: 10.1371/journal.pone.0012376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neugebauer JM, Amack JD, Peterson AG, Bisgrove BW, Yost HJ. FGF signalling during embryo development regulates cilia length in diverse epithelia. Nature. 2009;458:651–654. doi: 10.1038/nature07753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nguyen-Ba-Charvet KT, Picard-Riera N, Tessier-Lavigne M, Baron-Van Evercooren A, Sotelo C, Chedotal A. Multiple roles for slits in the control of cell migration in the rostral migratory stream. J. Neurosci. 2004;24:1497–1506. doi: 10.1523/JNEUROSCI.4729-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nishimura T, Kaibuchi K. Numb controls integrin endocytosis for directional cell migration with aPKC and PAR-3. Dev. Cell. 2007;13:15–28. doi: 10.1016/j.devcel.2007.05.003. [DOI] [PubMed] [Google Scholar]
- Noctor SC, Martinez-Cerdeno V, Ivic L, Kriegstein AR. Cortical neurons arise in symmetric and asymmetric division zones and migrate through specific phases. Nat. Neurosci. 2004;7:136–144. doi: 10.1038/nn1172. [DOI] [PubMed] [Google Scholar]
- Noctor SC, Martinez-Cerdeno V, Kriegstein AR. Contribution of intermediate progenitor cells to cortical histogenesis. Arch. Neurol. 2007;64:639–642. doi: 10.1001/archneur.64.5.639. [DOI] [PubMed] [Google Scholar]
- Noctor SC, Martinez-Cerdeno V, Kriegstein AR. Distinct behaviors of neural stem and progenitor cells underlie cortical neurogenesis. J. Comp. Neurol. 2008;508:28–44. doi: 10.1002/cne.21669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohata S, Aoki R, Kinoshita S, Yamaguchi M, Tsuruoka-Kinoshita S, et al. Dual roles of Notch in regulation of apically restricted mitosis and apicobasal polarity of neuroepithelial cells. Neuron. 2011;69:215–230. doi: 10.1016/j.neuron.2010.12.026. [DOI] [PubMed] [Google Scholar]
- O’Rahilly R, Muller F. The Embryonic Human Brain: An Atlas of Developmental Stages. New York: Wiley-Liss; 1994. [Google Scholar]
- Owen-Lynch PJ, Draper CE, Mashayekhi F, Bannister CM, Miyan JA. Defective cell cycle control underlies abnormal cortical development in the hydrocephalic Texas rat. Brain. 2003;126:623–631. doi: 10.1093/brain/awg058. [DOI] [PubMed] [Google Scholar]
- Ozdinler PH, Macklis JD. IGF-1 specifically enhances axon outgrowth of corticospinal motor neurons. Nat. Neurosci. 2006;9:1371–1381. doi: 10.1038/nn1789. [DOI] [PubMed] [Google Scholar]
- Palmer TD, Willhoite AR, Gage FH. Vascular niche for adult hippocampal neurogenesis. J. Comp. Neurol. 2000;425:479–494. doi: 10.1002/1096-9861(20001002)425:4<479::aid-cne2>3.0.co;2-3. [DOI] [PubMed] [Google Scholar]
- Parada C, Gato A, Bueno D. Mammalian embryonic cerebrospinal fluid proteome has greater apolipoprotein and enzyme pattern complexity than the avian proteome. J. Proteome Res. 2005a;4:2420–2428. doi: 10.1021/pr050213t. [DOI] [PubMed] [Google Scholar]
- Parada C, Gato A, Bueno D. All-trans retinol and retinol-binding protein from embryonic cerebrospinal fluid exhibit dynamic behaviour during early central nervous system development. Neuroreport. 2008;19:945–950. doi: 10.1097/WNR.0b013e3283021c94. [DOI] [PubMed] [Google Scholar]
- Parada C, Martin C, Alonso MI, Moro JA, Bueno D, Gato A. Embryonic cerebrospinal fluid collaborates with the isthmic organizer to regulate mesencephalic gene expression. J. Neurosci. Res. 2005b;82:333–345. doi: 10.1002/jnr.20618. [DOI] [PubMed] [Google Scholar]
- Petersen PH, Zou K, Hwang JK, Jan YN, Zhong W. Progenitor cell maintenance requires numb and numblike during mouse neurogenesis. Nature. 2002;419:929–934. doi: 10.1038/nature01124. [DOI] [PubMed] [Google Scholar]
- Petersen PH, Zou K, Krauss S, Zhong W. Continuing role for mouse Numb and Numbl in maintaining progenitor cells during cortical neurogenesis. Nat. Neurosci. 2004;7:803–811. doi: 10.1038/nn1289. [DOI] [PubMed] [Google Scholar]
- Pexieder T, Jelinek R. Pressure of the CSF and the morphogenesis of the CNS. II. Pressure necessary for normal development of brain vesicles. Folia Morphol. (Praha) 1970;18:181–192. [PubMed] [Google Scholar]
- Pinal N, Goberdhan DC, Collinson L, Fujita Y, Cox IM, et al. Regulated and polarized PtdIns(3,4,5)P3 accumulation is essential for apical membrane morphogenesis in photoreceptor epithelial cells. Curr. Biol. 2006;16:140–149. doi: 10.1016/j.cub.2005.11.068. [DOI] [PubMed] [Google Scholar]
- Pomeroy SL, Tamayo P, Gaasenbeek M, Sturla LM, Angelo M, et al. Prediction of central nervous system embryonal tumour outcome based on gene expression. Nature. 2002;415:436–442. doi: 10.1038/415436a. [DOI] [PubMed] [Google Scholar]
- Pontious A, Kowalczyk T, Englund C, Hevner RF. Role of intermediate progenitor cells in cerebral cortex development. Dev. Neurosci. 2008;30:24–32. doi: 10.1159/000109848. [DOI] [PubMed] [Google Scholar]
- Popken GJ, Hodge RD, Ye P, Zhang J, Ng W, et al. In vivo effects of insulin-like growth factor-I (IGF-I) on prenatal and early postnatal development of the central nervous system. Eur. J. Neurosci. 2004;19:2056–2068. doi: 10.1111/j.0953-816X.2004.03320.x. [DOI] [PubMed] [Google Scholar]
- Raballo R, Rhee J, Lyn-Cook R, Leckman JF, Schwartz ML, Vaccarino FM. Basic fibroblast growth factor (Fgf2) is necessary for cell proliferation and neurogenesis in the developing cerebral cortex. J. Neurosci. 2000;20:5012–5023. doi: 10.1523/JNEUROSCI.20-13-05012.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Radakovits R, Barros CS, Belvindrah R, Patton B, Muller U. Regulation of radial glial survival by signals from the meninges. J. Neurosci. 2009;29:7694–7705. doi: 10.1523/JNEUROSCI.5537-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rakic P. Evolution of the neocortex: a perspective from developmental biology. Nat. Rev. Neurosci. 2009;10:724–735. doi: 10.1038/nrn2719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Randhawa R, Cohen P. The role of the insulin-like growth factor system in prenatal growth. Mol. Genet. Metab. 2005;86:84–90. doi: 10.1016/j.ymgme.2005.07.028. [DOI] [PubMed] [Google Scholar]
- Rao G, Pedone CA, Del Valle L, Reiss K, Holland EC, Fults DW. Sonic hedgehog and insulin-like growth factor signaling synergize to induce medulloblastoma formation from nestin-expressing neural progenitors in mice. Oncogene. 2004;23:6156–6162. doi: 10.1038/sj.onc.1207818. [DOI] [PubMed] [Google Scholar]
- Rasin MR, Gazula VR, Breunig JJ, Kwan KY, Johnson MB, et al. Numb and Numbl are required for maintenance of cadherin-based adhesion and polarity of neural progenitors. Nat. Neurosci. 2007;10:819–827. doi: 10.1038/nn1924. [DOI] [PubMed] [Google Scholar]
- Redzic ZB, Preston JE, Duncan JA, Chodobski A, Szmydynger-Chodobska J. The choroid plexus-cerebrospinal fluid system: from development to aging. Curr. Top. Dev. Biol. 2005;71:1–52. doi: 10.1016/S0070-2153(05)71001-2. [DOI] [PubMed] [Google Scholar]
- Reillo I, de Juan Romero C, Garc´ıa-Cabezas MA, Borrell V. A role for intermediate radial glia in the tangential expansion of the mammalian cerebral cortex. Cereb. Cortex. 2011;21:1674–1694. doi: 10.1093/cercor/bhq238. [DOI] [PubMed] [Google Scholar]
- Rhyu MS, Jan LY, Jan YN. Asymmetric distribution of numb protein during division of the sensory organ precursor cell confers distinct fates to daughter cells. Cell. 1994;76:477–491. doi: 10.1016/0092-8674(94)90112-0. [DOI] [PubMed] [Google Scholar]
- Ribes V, Wang Z, Dolle P, Niederreither K. Retinaldehyde dehydrogenase 2 (RALDH2)-mediated retinoic acid synthesis regulates early mouse embryonic forebrain development by controlling FGF and sonic hedgehog signaling. Development. 2006;133:351–361. doi: 10.1242/dev.02204. [DOI] [PubMed] [Google Scholar]
- Rohatgi R, Milenkovic L, Scott MP. Patched1 regulates Hedgehog signaling at the primary cilium. Science. 2007;317:372–376. doi: 10.1126/science.1139740. [DOI] [PubMed] [Google Scholar]
- Sawamoto K, Wichterle H, Gonzalez-Perez O, Cholfin JA, Yamada M, et al. New neurons follow the flow of cerebrospinal fluid in the adult brain. Science. 2006;311:629–632. doi: 10.1126/science.1119133. Provided the first compelling evidence that gradients of CSF-distributed factors actively influence the brain.
- Schubert M, Brazil DP, Burks DJ, Kushner JA, Ye J, et al. Insulin receptor substrate-2 deficiency impairs brain growth and promotes tau phosphorylation. J. Neurosci. 2003;23:7084–7092. doi: 10.1523/JNEUROSCI.23-18-07084.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen Q, Zhong W, Jan YN, Temple S. Asymmetric Numb distribution is critical for asymmetric cell division of mouse cerebral cortical stem cells and neuroblasts. Development. 2002;129:4843–4853. doi: 10.1242/dev.129.20.4843. [DOI] [PubMed] [Google Scholar]
- Shen Q, Goderie SK, Jin L, Karanth N, Sun Y, et al. Endothelial cells stimulate self-renewal and expand neurogenesis of neural stem cells. Science. 2004;304:1338–1340. doi: 10.1126/science.1095505. [DOI] [PubMed] [Google Scholar]
- Shen Q, Wang Y, Kokovay E, Lin G, Chuang SM, et al. Adult SVZ stem cells lie in a vascular niche: a quantitative analysis of niche cell-cell interactions. Cell Stem Cell. 2008;3:289–300. doi: 10.1016/j.stem.2008.07.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimogori T, Banuchi V, Ng HY, Strauss JB, Grove EA. Embryonic signaling centers expressing BMP, WNT and FGF proteins interact to pattern the cerebral cortex. Development. 2004;131:5639–5647. doi: 10.1242/dev.01428. [DOI] [PubMed] [Google Scholar]
- Shimojo H, Ohtsuka T, Kageyama R. Oscillations in Notch signaling regulate maintenance of neural progenitors. Neuron. 2008;58:52–64. doi: 10.1016/j.neuron.2008.02.014. [DOI] [PubMed] [Google Scholar]
- Shitamukai A, Konno D, Matsuzaki F. Oblique radial glial divisions in the developing mouse neocortex induce self-renewing progenitors outside the germinal zone that resemble primate outer subventricular zone progenitors. J. Neurosci. 2011;31:3683–3695. doi: 10.1523/JNEUROSCI.4773-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siegenthaler JA, Ashique AM, Zarbalis K, Patterson KP, Hecht JH, et al. Retinoic acid from the meninges regulates cortical neuron generation. Cell. 2009;139:597–609. doi: 10.1016/j.cell.2009.10.004. Demonstrated that meningeally derived retinoic acid regulates neurogenesis.
- Siller KH, Doe CQ. Spindle orientation during asymmetric cell division. Nat. Cell Biol. 2009;1l:365–374. doi: 10.1038/ncb0409-365. [DOI] [PubMed] [Google Scholar]
- Silver R, LeSauter J, Tresco PA, Lehman MN. A diffusible coupling signal from the transplanted suprachiasmatic nucleus controlling circadian locomotor rhythms. Nature. 1996;382:810–813. doi: 10.1038/382810a0. [DOI] [PubMed] [Google Scholar]
- Simske JS, Kaech SM, Harp SA, Kim SK. LET-23 receptor localization by the cell junction protein LIN-7 during C. elegans vulval induction. Cell. 1996;85:195–204. doi: 10.1016/s0092-8674(00)81096-x. [DOI] [PubMed] [Google Scholar]
- Smart IH, Dehay C, Giroud P, Berland M, Kennedy H. Unique morphological features of the prolifer-ative zones and postmitotic compartments of the neural epithelium giving rise to striate and extrastriate cortex in the monkey. Cereb. Cortex. 2002;12:37–53. doi: 10.1093/cercor/12.1.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith CA, Lau KM, Rahmani Z, Dho SE, Brothers G, et al. aPKC-mediated phosphorylation regulates asymmetric membrane localization of the cell fate determinant Numb. EMBO J. 2007;26:468–480. doi: 10.1038/sj.emboj.7601495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soroceanu L, Kharbanda S, Chen R, Soriano RH, Aldape K, et al. Identification of IGF2 signaling through phosphoinositide-3-kinase regulatory subunit 3 as a growth-promoting axis in glioblastoma. Proc. Natl. Acad. Sci. USA. 2007;104:3466–3471. doi: 10.1073/pnas.0611271104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sousa-Nunes R, Yee LL, Gould AP. Fat cells reactivate quiescent neuroblasts via TOR and glial insulin relays in Drosophila . Nature. 2011;471:508–512. doi: 10.1038/nature09867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spassky N, Han YG, Aguilar A, Strehl L, Besse L, et al. Primary cilia are required for cerebellar development and Shh-dependent expansion of progenitor pool. Dev. Biol. 2008;317:246–259. doi: 10.1016/j.ydbio.2008.02.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Srinivasan K, Roosa J, Olsen O, Lee SH, Bredt DS, McConnell SK. MALS-3 regulates polarity and early neurogenesis in the developing cerebral cortex. Development. 2008;135:1781–1790. doi: 10.1242/dev.013847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stancik EK, Navarro-Quiroga I, Sellke R, Haydar TF. Heterogeneity in ventricular zone neural pre cursors contributes to neuronal fate diversity in the postnatal neocortex. J. Neurosci. 2010;30:7028–7036. doi: 10.1523/JNEUROSCI.6131-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stocker AM, Chenn A. Focal reduction of αE-catenin causes premature differentiation and reduction of beta-catenin signaling during cortical development. Dev. Biol. 2009;328:66–77. doi: 10.1016/j.ydbio.2009.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stottmann RW, Tran PV, Turbe-Doan A, Beier DR. Ttc21b is required to restrict sonic hedgehog activity in the developing mouse forebrain. Dev. Biol. 2009;335:166–178. doi: 10.1016/j.ydbio.2009.08.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stoykova A, Gotz M, Gruss P, Price J. Pax6-dependent regulation of adhesive patterning, R-cadherin expression and boundary formation in developing forebrain. Development. 1997;124:3765–3777. doi: 10.1242/dev.124.19.3765. [DOI] [PubMed] [Google Scholar]
- Stylianopoulou F, Efstratiadis A, Herbert J, Pintar J. Pattern of the insulin-like growth factor II gene expression during rat embryogenesis. Development. 1988;103:497–506. doi: 10.1242/dev.103.3.497. [DOI] [PubMed] [Google Scholar]
- Sun Y, Goderie SK, Temple S. Asymmetric distribution of EGFR receptor during mitosis generates diverse CNS progenitor cells. Neuron. 2005;45:873–886. doi: 10.1016/j.neuron.2005.01.045. [DOI] [PubMed] [Google Scholar]
- Sutter NB, Bustamante CD, Chase K, Gray MM, Zhao K, et al. A single IGF1 allele is a major determinant of small size in dogs. Science. 2007;316:112–115. doi: 10.1126/science.1137045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tajbakhsh S, Rocheteau P, Le Roux I. Asymmetric cell divisions and asymmetric cell fates. Annu. Rev. Cell Dev. Biol. 2009;25:671–699. doi: 10.1146/annurev.cellbio.24.110707.175415. [DOI] [PubMed] [Google Scholar]
- Takahashi T, Misson JP, Caviness VS., Jr. Glial process elongation and branching in the developing murine neocortex: a qualitative and quantitative immunohistochemical analysis. J. Comp. Neurol. 1990;302:15–28. doi: 10.1002/cne.903020103. [DOI] [PubMed] [Google Scholar]
- Tanori M, Santone M, Mancuso M, Pasquali E, Leonardi S, et al. Developmental and oncogenic effects of insulin-like growth factor-I in Ptc1+/− mouse cerebellum. Mol. Cancer. 2010;9:53. doi: 10.1186/1476-4598-9-53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tao Y, Black IB, DiCicco-Bloom E. In vivo neurogenesis is inhibited by neutralizing antibodies to basic fibroblast growth factor. J. Neurobiol. 1997;33:289–296. [PubMed] [Google Scholar]
- Tascioglu AO, Tasciolgu AB. Ventricular anatomy: illustrations and concepts from antiquity to Renaissance. Neuroanatomy. 2005;4:57–63. [Google Scholar]
- Tavazoie M, Van der Veken L, Silva-Vargas V, Louissaint M, Colonna L, et al. A specialized vascular niche for adult neural stem cells. Cell Stem Cell. 2008;3:279–288. doi: 10.1016/j.stem.2008.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taverna E, Huttner WB. Neural progenitor nuclei IN motion. Neuron. 2010;67:906–914. doi: 10.1016/j.neuron.2010.08.027. [DOI] [PubMed] [Google Scholar]
- Thornton GK, Woods CG. Primary microcephaly: do all roads lead to Rome? Trends Genet. 2009;25:501–510. doi: 10.1016/j.tig.2009.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Town T, Breunig JJ, Sarkisian MR, Spilianakis C, Ayoub AE, et al. The stumpy gene is required for mammalian ciliogenesis. Proc. Natl. Acad. Sci. USA. 2008;105:2853–2858. doi: 10.1073/pnas.0712385105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toyoda R, Assimacopoulos S, Wilcoxon J, Taylor A, Feldman P, et al. FGF8 acts as a classic diffusible morphogen to pattern the neocortex. Development. 2010;137:3439–3448. doi: 10.1242/dev.055392. Showed that FGF8 generates classical diffusion gradients in the developing brain.
- Vescovi AL, Reynolds BA, Fraser DD, Weiss S. bFGF regulates the proliferative fate of unipotent (neuronal) and bipotent (neuronal/astroglial) EGF-generated CNS progenitor cells. Neuron. 1993;11:951–966. doi: 10.1016/0896-6273(93)90124-a. [DOI] [PubMed] [Google Scholar]
- Vierkotten J, Dildrop R, Peters T, Wang B, Ruther U. Ftm is a novel basal body protein of cilia involved in Shh signaling. Development. 2007;134:2569–2577. doi: 10.1242/dev.003715. [DOI] [PubMed] [Google Scholar]
- von Stein W, Ramrath A, Grimm A, Muller-Borg M, Wodarz A. Direct association of Bazooka/PAR-3 with the lipid phosphatase PTEN reveals a link between the PAR/aPKC complex and phosphoinositide signaling. Development. 2005;132:1675–1686. doi: 10.1242/dev.01720. [DOI] [PubMed] [Google Scholar]
- Wallace VA. Purkinje-cell-derived Sonic hedgehog regulates granule neuron precursor cell proliferation in the developing mouse cerebellum. Curr. Biol. 1999;9:445–448. doi: 10.1016/s0960-9822(99)80195-x. [DOI] [PubMed] [Google Scholar]
- Wang J, Wynshaw-Boris A. The canonical Wnt pathway in early mammalian embryogenesis and stem cell maintenance/differentiation. Curr. Opin. Genet. Dev. 2004;14:533–539. doi: 10.1016/j.gde.2004.07.013. [DOI] [PubMed] [Google Scholar]
- Wang X, Tsai JW, Lamonica B, Kriegstein AR. A new subtype of progenitor cells in the mouse embryonic neocortex. Nat. Neurosci. 2011;14:555–561. doi: 10.1038/nn.2807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber MM, Melmed S, Rosenbloom J, Yamasaki H, Prager D. Rat somatotroph insulin-like growth factor-II (IGF-II) signaling: role of the IGF-I receptor. Endocrinology. 1992;131:2147–2153. doi: 10.1210/endo.131.5.1425415. [DOI] [PubMed] [Google Scholar]
- Wechsler-Reya RJ, Scott MP. Control of neuronal precursor proliferation in the cerebellum by Sonic Hedgehog. Neuron. 1999;22:103–114. doi: 10.1016/s0896-6273(00)80682-0. [DOI] [PubMed] [Google Scholar]
- Willaredt MA, Hasenpusch-Theil K, Gardner HA, Kitanovic I, Hirschfeld-Warneken VC, et al. A crucial role for primary cilia in cortical morphogenesis. J. Neurosci. 2008;28:12887–12900. doi: 10.1523/JNEUROSCI.2084-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wirtz-Peitz F, Nishimura T, Knoblich JA. Linking cell cycle to asymmetric division: Aurora-A phos-phorylates the Par complex to regulate Numb localization. Cell. 2008;135:161–173. doi: 10.1016/j.cell.2008.07.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wodarz A. Molecular control of cell polarity and asymmetric cell division in Drosophila neuroblasts. Curr. Opin. Cell Biol. 2005;17:475–481. doi: 10.1016/j.ceb.2005.08.005. [DOI] [PubMed] [Google Scholar]
- Wolpert L. One hundred years of positional information. Trends Genet. 1996;12:359–364. doi: 10.1016/s0168-9525(96)80019-9. [DOI] [PubMed] [Google Scholar]
- Woodhead GJ, Mutch CA, Olson EC, Chenn A. Cell-autonomous β-catenin signaling regulates cortical precursor proliferation. J. Neurosci. 2006;26:12620–12630. doi: 10.1523/JNEUROSCI.3180-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wrobel CN, Mutch CA, Swaminathan S, Taketo MM, Chenn A. Persistent expression of stabilized β-catenin delays maturation of radial glial cells into intermediate progenitors. Dev. Biol. 2007;309:285–297. doi: 10.1016/j.ydbio.2007.07.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu H, Feng W, Chen J, Chan LN, Huang S, Zhang M. PDZ domains of Par-3 as potential phospho-inositide signaling integrators. Mol. Cell. 2007;28:886–898. doi: 10.1016/j.molcel.2007.10.028. [DOI] [PubMed] [Google Scholar]
- Ye P, Xing Y, Dai Z, D’Ercole AJ. In vivo actions of insulin-like growth factor-I (IGF-I) on cerebellum development in transgenic mice: evidence that IGF-I increases proliferation of granule cell progenitors. Brain Res. Dev. Brain Res. 1996;95:44–54. doi: 10.1016/0165-3806(96)00492-0. [DOI] [PubMed] [Google Scholar]
- Zappaterra MD, Lisgo SN, Lindsay S, Gygi SP, Walsh CA, Ballif BA. A comparative proteomic analysis of human and rat embryonic cerebrospinal fluid. J. Proteome Res. 2007;6:3537–3548. doi: 10.1021/pr070247w. [DOI] [PubMed] [Google Scholar]
- Zhang J, Woodhead GJ, Swaminathan SK, Noles SR, McQuinn ER, et al. Cortical neural precursors inhibit their own differentiation via N-cadherin maintenance of β-catenin signaling. Dev. Cell. 2010;18:472–479. doi: 10.1016/j.devcel.2009.12.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng W, Chodobski A, editors. The Blood-Cerebrospinal Fluid Barrier. Boca Raton: Taylor & Francis; 2005. [Google Scholar]
- Zhou CJ, Borello U, Rubenstein JL, Pleasure SJ. Neuronal production and precursor proliferation defects in the neocortex of mice with loss of function in the canonical Wnt signaling pathway. NeuroScience. 2006;142:1119–1131. doi: 10.1016/j.neuroscience.2006.07.007. [DOI] [PubMed] [Google Scholar]
- Zhou Y, Atkins JB, Rompani SB, Bancescu DL, Petersen PH, et al. The mammalian Golgi regulates numb signaling in asymmetric cell division by releasing ACBD3 during mitosis. Cell. 2007;129:163–178. doi: 10.1016/j.cell.2007.02.037. [DOI] [PubMed] [Google Scholar]
RELATED RESOURCES
- He S, Nahada D, Morrison SJ. Mechanisms of stem cell self-renewal. Annu. Rev. Cell Dev. Biol. 2009;25:377–406. doi: 10.1146/annurev.cellbio.042308.113248. [DOI] [PubMed] [Google Scholar]
- Jackson Lab. Hydrocephalus in laboratory mice. JAX® NOTES. 2003:490. http://jaxmice.jax.org/jaxnotes/archive/490f.html.
- Nachury MV, Seeley ES, Jin H. Trafficking to the ciliary membrane: how to get across the periciliary diffusion barrier? Annu. Rev. Cell Dev. Biol. 2010;26:59–87. doi: 10.1146/annurev.cellbio.042308.113337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sundberg JP, Woolcott BL, Cunlifee-Beamer T, Brown KS, Bronson R. Spontaneous hydrocephalus in inbred strains of mice. JAX® NOTES. 1991:445. http://jaxmice.jax.org/jaxnotes/archive/445a.html. [Google Scholar]