Summary
A paracrine interaction between EGF-secreting tumor-associated macrophages (TAMs) and CSF-1-secreting breast carcinoma cells promotes invasion and metastasis. Here we show that mice deficient in the hematopoietic cell-specific Wiskott-Aldrich Syndrome protein (WASp), are unable to support TAM-dependent carcinoma cell invasion and metastasis in both orthotopic and transgenic models of mammary tumorigenesis. Motility and invasion defects of tumor cells were recapitulated ex vivo upon co-culture with WASp−/− macrophages. Mechanistically, WASp is required for macrophages to migrate towards CSF-1-producing carcinoma cells and for the release of EGF through metalloprotease-dependent shedding of EGF from the cell surface of macrophages. Our findings suggest WASp acts to support both migration of TAMs and production of EGF that, in concert, promote breast tumor metastasis.
Introduction
Cancer progression is a complex multi-step process consisting of neoplastic cell transformation, tumor growth, invasion and metastasis. While metastasis is the major cause of cancer deaths, it remains poorly characterized (Valastyan and Weinberg, 2011). Malignant dissemination is not an exclusively cancer cell-autonomous process; other constituents of the tumor microenvironment, such as inflammatory cells, heavily influence cancer progression (Hanahan and Coussens, 2012). Additionally, clinical studies on invasive breast cancer demonstrate tumor associated macrophage (TAM) density correlates with poor prognosis (Qian and Pollard, 2010). Indeed TAMs promote angiogenesis, extracellular matrix proteolysis, and enhance tumor cell invasion and dissemination (Joyce and Pollard, 2009).
Circulating levels of colony stimulating factor-1 (CSF-1), a pleiotropic macrophage growth factor and chemoattractant, correlate with poor prognosis in breast cancer patients and high levels of CSF-1 are found in invasive but not in in-situ breast cancer (Qian and Pollard, 2010). In agreement with these observations, CSF-1 deficiency in the polyoma middle-T antigen mouse model of mammary carcinogenesis (MMTV-PyMT), led to a reduction in TAM density, delayed development of tumors to invasive metastatic carcinoma and reduced lung metastasis with no change in primary tumor growth (Lin et al., 2001), demonstrating that CSF-1 played a critical role in breast tumor progression and metastasis.
Macrophages and breast carcinoma cells depend on an epidermal growth factor (EGF)/CSF-1 paracrine loop for invasion both in vitro (Goswami et al., 2005) and in vivo (Wyckoff et al., 2004). In vivo tumor cell invasion was blocked by inhibiting either the EGF receptor (EGFR) or the CSF-1 receptor (CSF-1R), expressed by breast carcinoma cells and macrophages, respectively (Wyckoff et al., 2004). Given that recruitment of higher numbers of TAMs has been reported in EGFR-positive breast tumors (Leek et al., 2000), blockade of the paracrine interaction may have therapeutic benefit in the treatment of breast cancer. Additionally targeting TAMs is attractive because, unlike cancer cells, they are genetically stable and less likely to develop drug resistance.
We demonstrate here that Wiskott-Aldrich syndrome protein (WASp) could be a potential therapeutic target. Wiskott-Aldrich syndrome (WAS) is a X-linked genetic disorder resulting from mutations in the WAS gene (Thrasher and Burns, 2010). WASp is exclusively expressed in non-erythroid hematopoietic cells unlike its ubiquitously expressed homologue, neural (N)-WASP. WASp is an actin nucleation promoting factor that regulates chemotaxis (Takenawa and Suetsugu, 2007). Since WAS patient macrophages are defective in chemotaxis towards CSF-1 (Zicha et al., 1998), we hypothesized that WASp-deficiency may block the paracrine interaction between TAMs and breast carcinoma cells, disabling their co-migration and attenuating metastatic dissemination.
Results
Macrophage WASp is required for breast carcinoma cell interaction in vitro
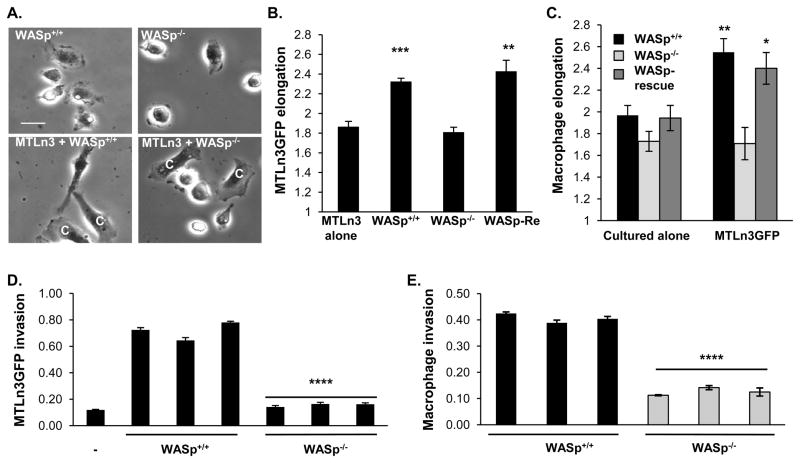
To test whether WASp plays a role in the EGF/CSF-1 paracrine interaction, bone marrow derived macrophages (BMMs) from WASp+/+ or WASp−/− mice were co-cultured with GFP-expressing MTLn3 cells (MTLn3GFP). Unlike WASp−/− BMMs, WASp+/+ BMMs induced an increase in MTLn3GFP elongation (Figure 1A, B), indicative of acquisition of a motile phenotype (Goswami et al., 2005). Concurrently, WASp−/− BMMs did not elongate in the presence of MTLn3GFP cells, unlike WASp+/+ BMMs (Figure 1A, C). Expression of human WASp in WASp−/− BMMs restored both MTLn3 and BMM elongation in co-cultures (Figure 1B, C). Live cell imaging of BMMs added to MTLn3GFP cultures showed that within the first few hours of co-culture WASp+/+ BMMs elongated robustly toward MTLn3GFP cells, while WASp−/− BMMs did not (Figure S1A-C; Movies S1, S2).
Figure 1. WASp is required for cell elongation and invasion of both breast carcinoma cells and macrophages.
MTLn3GFP cells cultured alone or with WASp+/+, WASp−/− or WASp−/− rescued (WASp-Re) BMMs for 16 hr. (A) Representative phase-contrast images of the indicated conditions. MTLn3 cells are denoted by a C in co-culture images. Scale bar, 10 μm. (B,C) Quantification of average elongation of (B) MTLn3GFP and (C) the indicated macrophage types. n ≥ 4 independent experiments. * p < 0.05, ** p < 0.01 compared to control cells. Quantification of the fraction of MTLn3 (D) or BMMs (E) invading above 20 μm in the absence (−) or presence of WASp+/+ or WASp−/− BMMs from 3 independent mice. Error bars signify SEM from 5 independent fields. **** p < 0.0001 compared to MTLn3 cultured conditions with WASp+/+ BMMs. See also Figure S1 and Movies S1 and S2.
To test whether WASp was required for macrophage-dependent MTLn3 cell invasion into 3D matrices, BMM and MTLn3GFP co-cultures were overlaid with a collagen I gel and the fraction of invading cells was determined (Goswami et al., 2005). WASp+/+ BMMs enhanced MTLn3GFP invasion into the matrix 7-fold, while MTLn3GFP invasion was not enhanced by WASp−/− BMMs (Figure 2A and 2B and Figure S1D). In the presence of MTLn3GFP cells the fraction of co-invading WASp+/+ BMMs was increased significantly compared to WASp−/− BMMs (Figure 2A and 2C and Figure S1D). WASp expression was therefore necessary for BMMs to adopt a migratory phenotype, to engage in and maintain the paracrine interaction with breast carcinoma cells, and for BMMs and breast carcinoma cells to co-invade a 3D collagen matrix. Importantly, WASp was not required for BMM invasion into a fibrous collagen matrix in the absence of carcinoma cells (Haein Park, A.D., I. Maridonneau-Parini and D.C., manuscript in preparation) and suggests that WASp is required for movement in response to a soluble guidance cue, in this case, CSF-1 provided by carcinoma cells. This is consistent with the well-established role of WASp in the chemotaxis of macrophages. These results prompted us to investigate whether WASp was required for macrophage dependent breast tumor invasiveness and metastasis in mouse models of breast cancer.
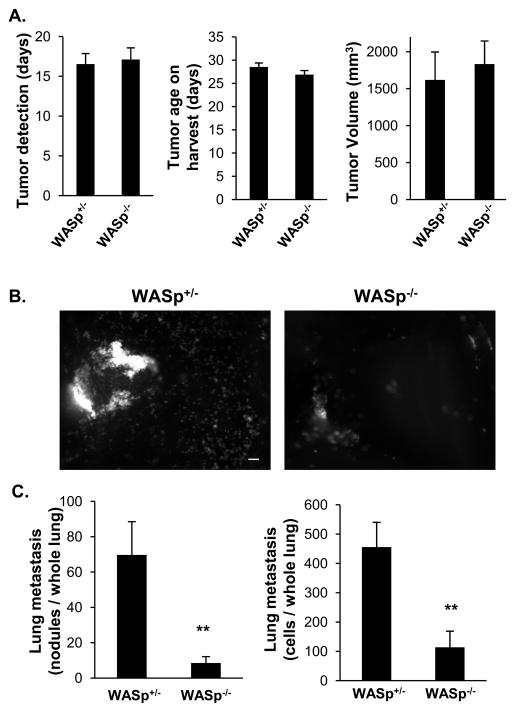
Figure 2. WASp is required for TAM-dependent breast carcinoma cell metastasis but not primary tumor growth in a rat orthotopic breast tumor model.
(A) Primary MTLn3-Cerulean tumor growth was determined by average day of tumor detection after injection, age of primary tumors and volume of primary tumors upon harvest. (B) Representative fluorescent images of spontaneous lung metastases. Scale bar, 10 μm. (C) Quantification of spontaneous lung metastases; number of metastatic nodules (< 5 cells in cluster; left graph) or total carcinoma cell number from the whole lung (right graph). n= 9 WASp+/− and 11 WASp−/− tumor bearing mice. * p < 0.05 ** p < 0.01 by two-tailed Student’s t-test and Mann-Whitney significance test. Error bars signify SEM. See also Figure S2.
WASp is required for efficient breast cancer metastasis
To specifically test the effect of myeloid WASp on tumor invasiveness and metastasis, WASp+/− or WASp−/− mice were crossed with Rag2−/− mice to generate xenograft-competent mice that lack mature and functional B- and T-lymphocytes, and cyan-fluorescent MTLn3 cells (MTLn3-Cerulean) were injected into the mammary glands. The time of initial tumor detection after injection, tumor age, and volume upon harvest were unaltered between WASp+/− and WASp−/− tumor-bearing mice (Figure 2A). However, lung metastasis, measured as metastatic nodules or total number of carcinoma cells per whole lung, was significantly reduced in WASp−/− mice compared to WASp+/− mice (Figure 2B and 2C). These data suggested that host WASp expression was required for efficient lung metastasis of breast tumor cells, but not for primary tumor growth.
The above data were confirmed using the triple negative human breast carcinoma cell line MDA-MB-231. In vitro MDA-MB-231 cell invasion was significantly reduced in the presence of WASp−/− BMMs (Figure S2A), whereas WASp+/− and WASp+/+ BMMs promoted MDA-MB-231 cell invasion equally. Similar results were obtained using the highly invasive subline MDA-MB-231-4173, characterized for its lung tropism in metastasis (Figure S2B). Likewise, orthotopic tumors of MDA-MB-231-4173 Cerulean cells in Rag2−/−WASp+/− or Rag2−/−WASp−/− mice resulted in fewer lung metastases in WASp−/− mice compared to WASp+/− mice despite equal growth parameters (Figure S2C, D).
We also used the immune competent MMTV-PyMT transgenic mouse breast cancer model that resembles human breast cancer progression (Lin et al., 2003). No significant differences in primary tumor growth were detected between PyMT+WASp+/− or PyMT+WASp−/− mice (Figure S2E). Semi-quantitative RT-PCR of PyMT mRNA was performed to determine the extent of metastasis in the lung (Lin et al., 2001), revealed significantly more PyMT mRNA levels in lungs of WASp+/− mice compared to WASp−/− mice (Figure S2F). Histologic evaluations of H&E stained lungs of PyMT tumors showed more metastatic nodules in PyMT+WASp+/− mice compared PyMT+WASp−/− mice (Figure S2G).
Crucially, none of the tumor cells examined expressed WASp (Figure S2H). We have thus shown in three different breast tumor models, a rat, a human xenograft, and a transgenic model of mammary carcinogenesis that leukocyte-restricted WASp was required for efficient metastasis to the lung.
WASp is required for TAM-mediated breast carcinoma cell motility, invasion and intravasation in vivo
Because WASp is required for macrophage chemotaxis, it was possible that WASp deficiency affected TAM recruitment. To address this concern the TAM content of primary tumors was examined by intravital imaging of MTLn3-Cerulean tumors from Rag2−/−WASp+/− or WASp−/− mice where TAMs were identified by the uptake of Texas red-dextran (Wyckoff et al., 2007). MTLn3-Cerulean tumors from WASp−/− mice contained slightly lower numbers of TAMs (Figure 3A), especially at the tumor cortex, however these differences were not statistically significant. These results suggested that WASp did not play a major role in the recruitment of macrophages to late-stage carcinomas and that this could not account for the dramatic reduction in tumor metastasis.
Figure 3. WASp is required for TAM-dependent carcinoma cell motility, invasion and intravasation in vivo.
(A) The number of Texas-red dextran positive TAMs from WASp+/− and WASp−/− mice was quantified 24 hours after Texas-red dextran injection. Images were acquired at 5 μm steps starting from the tumor cortex to a final depth of 100 μm using intravital two-photon microscopy. (B) Quantification of the average number of motile MTLn3 cells per field determined by intravital imaging. n > 3 mice per genotype. * p < 0.05 using Mann-Whitney’s significance test. See also Movies S3 and S4. (C) Total number of invasive cells collected into microneedles containing either 25 nM EGF or buffer. n=4 mice per genotype. ** p < 0.01 Student’s T-test compared to buffer containing needles in WASp+/− mice. (D) Tumor cell blood burden in WASp+/− or WASp−/− mice bearing MTLn3-derived breast tumor following 3 weeks of tumor growth. Graphs show average number of cells (left) and colonies (right) per mL of blood. n > 4 mice per genotype. Error bars signify SEM. * p < 0.05.
It has been shown that TAMs promote the motility of tumor cells that are in close proximity (Roussos et al., 2011; Wyckoff et al., 2007). Multiphoton intravital imaging showed decreased TAM-proximal tumor cell motility throughout the tumors of WASp−/− mice compared to those of WASp+/− mice (Figure 3B; Movies S3, S4). Therefore, despite the slightly lower numbers of TAMs in WASp−/− mice at the tumor cortex, carcinoma cell motility throughout the tumor, even in areas of similar TAM density, was significantly reduced. To directly test whether WASp in TAMs was required for the paracrine interaction with breast tumor cells in vivo we employed an in vivo invasion assay (Ojalvo et al., 2010; Patsialou et al., 2012; Wyckoff et al., 2000) in which collection needles containing Matrigel and either EGF or buffer were inserted into MTLn3-Cerulean tumors of Rag2−/−WASp+/− and WASp−/− mice. EGF-filled needles collected significantly more MTLn3-Cerulean cells compared to buffer-filled needles from tumors of WASp+/− mice (Figure 3C). In contrast, there was no EGF-induced in vivo invasion from tumors of WASp−/− mice even though TAM levels were only reduced by a maximum of 30%.
TAMs also assist tumor cell intravasation (Wyckoff et al., 2007). Consistently, blood from WASp−/− mice contained fewer viable circulating tumor cells compared to WASp+/− mice (Figure 3D), suggesting breast tumor cell intravasation was reduced in WASp−/− mice. Overall, these data support a role of host WASp in tumor cell motility, invasion and intravasation at the primary tumor site.
WASp is required for EGF shedding
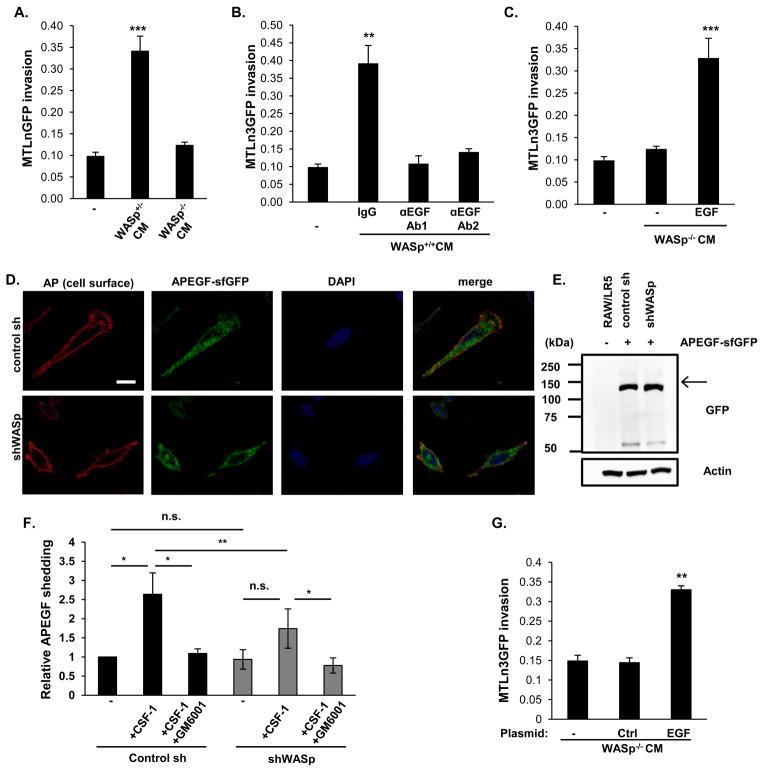
The inability of WASp−/− mice to support carcinoma cell invasion prompted us to further examine the mechanisms by which WASp contributed to the paracrine interaction with carcinoma cells. Apart from motility, WASp regulates secretory processes (Thrasher and Burns, 2010). Given the central role of TAM-secreted EGF in the paracrine loop, we explored EGF secretion in BMMs. Conditioned media from WASp+/+ BMMs promoted MTLn3GFP 3D invasion, whereas conditioned media from WASp−/− BMM did not (Figure 4A). Neutralizing antibodies against EGF attenuated MTLn3GFP cell invasion by the conditioned media (Figure 4B). Consistently, addition of purified EGF to conditioned media from WASp−/− BMMs rescued MTLn3 cell invasion (Figure 4C). Collectively, these results demonstrated that EGF was necessary and sufficient to promote MTLn3 invasion in vitro, and suggested that WASp was required for EGF secretion.
Figure 4. WASp is required for EGF release.
(A) Quantification of in vitro 3D invasion using MTLn3GFP cells cultured alone (−) or with conditioned media (CM) from WASp+/+ or WASp−/− BMMs. (B) Quantification of 3D invasion of MTLn3GFP cells in the absence (−) or presence of WASp+/+ conditioned media with either a non-specific IgG or two different EGF neutralizing antibodies (αEGF Ab1 or Ab2). (C) Addition of exogenous EGF (5nM) to WASp−/− BMM conditioned media restores invasion of MTLn3GFP cells. n≥3 experiments per condition. (D) Confocal sections of control sh and shWASp RAW/LR5 cells expressing APEGF-sfGFP. Unpermeabilized fixed cells were stained with an antibody against extracellular AP. Scale bar, 10 μm. (E) Equal expression levels of APEGF-sfGFP (arrow) expressed by the control sh and shWASp cells, detected with an antibody against GFP. (F) Conditioned media from the indicated APEGF-sfGFP expressing cells under the conditions shown were collected and assayed for shed AP activity. (G) Quantification of MTLn3GFP invasion with CM collected from WASp−/− BMM infected with either a control vector or a vector expressing recombinant soluble EGF. Error bars signify SEM. * p < 0.05, ** p<0.01 and ***p<0.001. See also Figures S3 and S4.
However, EGF mRNA and proEGF protein levels were equal between WASp+/+ and WASp−/− BMMs, under basal conditions and were upregulated to the same extent following CSF-1 treatment (Figure S3). This prompted us to investigate macrophage EGF trafficking and shedding. We used a well-established reporter of EGF shedding in which alkaline phosphatase (AP) replaces part of the extracellular portion of proEGF (APEGF) (Sahin et al., 2006). Infection of primary BMMs with this reported proved inefficient for a consistent quantitative assay (data not shown), so we assayed previously characterized macrophage cell lines (RAW/LR5) infected with a non-targeting, control shRNA (control sh) or one that targets WASp (shWASp) (Dovas et al., 2009) (Figure S4A). Similar to BMMs, these cells engaged in WASp-dependent paracrine interactions with carcinoma cells (Figure S4B-D). Control sh or shWASp cells were infected with a retrovirus expressing APEGF fused to super-folder GFP in its cytoplasmic tail (APEGF-sfGFP). Both control sh and shWASp cells were able to traffic APEGF-sfGFP to the cell surface, with no observable differences in subcellular distribution (Figure 4D). Despite equal expression levels of APEGF-sfGFP (Figure 4E) and basal shedding of AP activity, shWASp cells shed significantly lower amounts of the APEGF-sfGFP than control sh cells following CSF-1 stimulation (Figure 4F). Treatment of cells with the broad-spectrum inhibitor GM6001 reduced shedding to its basal levels (Figure 7F), indicating that WASp regulates CSF-1-mediated EGF shedding in a metalloprotease-dependent manner. Consistently, media from WASp−/− BMMs infected with a construct expressing a secretable form of the mature, soluble human EGF (Yao et al., 1998) bypassed the requirement for EGF shedding and promoted invasion of MTLn3 cells, (Figure 4G).
Discussion
We have demonstrated that WASp regulated macrophage-mediated tumor cell invasion in vitro and tumor cell motility, invasion, intravasation and lung metastasis, but not tumor growth, in vivo. These results corroborate previous studies implicating CSF-1 and TAMs in metastasis and suggest inhibition of effectors downstream of CSF-1R signaling as targets of anti-metastatic therapy.
Additionally, we uncovered a novel role for WASp in the regulation of CSF-1R-mediated EGF shedding, while EGF mRNA, protein levels and cell surface expression were WASp-independent. Our data suggest that WASp may bridge CSF-1R signaling to a cell surface metalloprotease activity. Importantly, dendritic cells deficient in WASp interacting protein (WIP), which binds and regulates WASp levels in vivo, showed reduced secretion of proMMP2 and proMMP9 (Banon-Rodriguez et al., 2011). Interference with the WASp pathway in myeloid cells may thus lead to systemic defects in pericellular proteolysis.
WASp may regulate EGF shedding from the cell surface via several, perhaps interdependent, mechanisms: direct delivery of vesicles containing the sheddase(s) via F-actin–mediated propulsion; indirectly via the establishment of cell polarity and the formation of F-actin conduits for sheddase delivery; via the formation of podosomes, F-actin rich structures that concentrate proteolytic activities. Such mechanisms have been proposed to act in the delivery of proteases to the plasma membrane and to invadopodia of invasive tumor cells (Poincloux et al., 2009). Further experiments will be needed to determine the exact mechanism by which WASp regulates EGF shedding.
The decreased in vivo cell invasion observed in WASp−/− mice despite the presence of exogenous EGF supplied through the microneedle (Figure 3C) may appear to contradict the in vitro data where EGF alone restored breast carcinoma cell invasion (Figure 4C). However, efficient chemotaxis in the tumor requires propagation of chemotactic signals from cell to cell in a streaming fashion (Roussos et al., 2011; Sharma et al., 2012) so as to maintain the migration of cells. Consistent with this, blockade of CSF-1R in the presence of EGF was equally effective as EGFR blockade in reducing the number of invasive cells collected (Wyckoff et al., 2004). Given that WASp was required for the elongation of BMMs towards tumor cells in vitro (Figure 1), our data suggest that WASp in macrophages contributes to carcinoma cell invasion by supporting both macrophage chemotaxis and EGF release.
Our findings suggest that WASp inhibition may have therapeutic benefit in breast tumors. We argue that inhibition of a protein downstream of CSF-1R could bypass several issues associated with CSF-1R blockade such as the non-specificity of kinase inhibitors, or elevated levels of circulating CSF-1 in the case of anti-CSF-1R antibodies (Hume and MacDonald, 2012). Additionally, WASp inhibition could affect the functions of other hematopoietic cells that play a role in metastasis, such as IL-4 secreting CD4+ T cells that stimulate EGF production by TAMs (DeNardo et al., 2009) and platelets, important in tumor cell seeding at the metastatic site (Gay and Felding-Habermann, 2011). While we cannot completely rule out the contributions of other hematopoietic cells in our system, we argue that TAMs play the most critical roles in the functions tested. Indeed, specific ablation of TAMs by genetic deletion of CSF-1 (Lin et al., 2001), administration of liposome-encapsulated clodronate or inhibition of CSF-1R (Hernandez et al., 2009; Patsialou et al., 2009; Wyckoff et al., 2004) recapitulates all of our observed long and short-term phenotypes. In addition, WASp deficiency caused defects in tumor cell invasion in the primary tumor prior to entry of tumor cells into the circulation and seeding at secondary sites, steps where other leukocytes may contribute. Therefore, the defects observed in the absence of WASp can be largely attributed to defects in TAMs. Importantly, TAM-specific delivery of pharmacological agents is feasible (Leuschner et al., 2011). In this respect, WASp could be selectively targeted in TAMs and not other leukocytes.
WASp may be a viable candidate as we have shown that WASp is required for TAMs to promote breast carcinoma cell invasion and metastasis, yet it is expressed exclusively in genetically stable hematopoietic cells. In support of our results, Oncomine™ analysis demonstrated that WASp was significantly overexpressed in high-grade (p<1.1×10−6) and triple negative tumors (p<1.6×10−8) while significantly underexpressed in ER positive tumors (p<5.3×10−14). This finding agrees with a study showing that inflammatory leukocyte infiltration correlates with the aggressiveness of estrogen receptor negative breast carcinomas (Chavey et al., 2007). The identification and characterization of host factors that play an essential role in tumor progression may ultimately lead to new therapeutic targets and expand the available options for cancer therapy.
EXPERIMENTAL PROCEDURES
Mice
All procedures involving mice were conducted in accordance with National Institutes of Health regulations concerning the use and care of experimental animals. The Albert Einstein College of Medicine animal use committee approved the use of mice in this study.
In vivo experiments
In vivo experiments including intravital microscopy, in vivo invasion, intravasation and lung metastasis are described in Supplemental Experimental Procedures.
In vitro experiments
In vitro experiments including 2D and 3D co-culture assays, AP assay and confocal microscopy are described in Supplemental Experimental Procedures.
Data analysis
Results were analyzed by two-tailed Student’s t-test. Non-parametric statistical analysis using two-tailed Mann-Whitney significance test was performed for analysis of lung metastasis from MTLn3-Cerulean injected mice and intravital imaging analysis; a p value of < 0.05 was considered significant.
Supplementary Material
Highlights.
WASp is required for paracrine signaling between TAMs and breast carcinoma cells.
WASp is required for EGF secretion, aiding breast carcinoma cell invasion in vitro.
WASp is required for breast carcinoma cell motility and invasion in vivo.
WASp is not required for tumor growth but for efficient lung metastasis.
Acknowledgments
We thank researchers for providing reagents as indicated. We thank members of the Condeelis, Segall, Hogdson and Stanley laboratories for helpful comments and discussions. We thank Einstein core facilities (Analytical Imaging Facility, Animal Housing Facility) for technical assistance. This work was supported by National Institute of Health grants GM071828 (DC, AD, DI), CA100324 (AD, ARB, JS, JC, DC) and CA110269 (LH).
Footnotes
The authors declare no conflict of interest.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Banon-Rodriguez I, Monypenny J, Ragazzini C, Franco A, Calle Y, Jones GE, Anton IM. The cortactin-binding domain of WIP is essential for podosome formation and extracellular matrix degradation by murine dendritic cells. European journal of cell biology. 2011;90:213–223. doi: 10.1016/j.ejcb.2010.09.001. [DOI] [PubMed] [Google Scholar]
- Chavey C, Bibeau F, Gourgou-Bourgade S, Burlinchon S, Boissiere F, Laune D, Roques S, Lazennec G. Oestrogen receptor negative breast cancers exhibit high cytokine content. Breast Cancer Res. 2007;9:R15. doi: 10.1186/bcr1648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeNardo DG, Barreto JB, Andreu P, Vasquez L, Tawfik D, Kolhatkar N, Coussens LM. CD4(+) T cells regulate pulmonary metastasis of mammary carcinomas by enhancing protumor properties of macrophages. Cancer cell. 2009;16:91–102. doi: 10.1016/j.ccr.2009.06.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dovas A, Gevrey JC, Grossi A, Park H, Abou-Kheir W, Cox D. Regulation of podosome dynamics by WASp phosphorylation: implication in matrix degradation and chemotaxis in macrophages. Journal of cell science. 2009;122:3873–3882. doi: 10.1242/jcs.051755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gay LJ, Felding-Habermann B. Contribution of platelets to tumour metastasis. Nat Rev Cancer. 2011;11:123–134. doi: 10.1038/nrc3004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goswami S, Sahai E, Wyckoff JB, Cammer M, Cox D, Pixley FJ, Stanley ER, Segall JE, Condeelis JS. Macrophages promote the invasion of breast carcinoma cells via a colony-stimulating factor-1/epidermal growth factor paracrine loop. Cancer Res. 2005;65:5278–5283. doi: 10.1158/0008-5472.CAN-04-1853. [DOI] [PubMed] [Google Scholar]
- Hanahan D, Coussens LM. Accessories to the crime: functions of cells recruited to the tumor microenvironment. Cancer cell. 2012;21:309–322. doi: 10.1016/j.ccr.2012.02.022. [DOI] [PubMed] [Google Scholar]
- Hernandez L, Smirnova T, Kedrin D, Wyckoff J, Zhu L, Stanley ER, Cox D, Muller WJ, Pollard JW, Van Rooijen N, Segall JE. The EGF/CSF-1 paracrine invasion loop can be triggered by heregulin beta1 and CXCL12. Cancer Res. 2009;69:3221–3227. doi: 10.1158/0008-5472.CAN-08-2871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hume DA, MacDonald KP. Therapeutic applications of macrophage colony-stimulating factor-1 (CSF-1) and antagonists of CSF-1 receptor (CSF-1R) signaling. Blood. 2012;119:1810–1820. doi: 10.1182/blood-2011-09-379214. [DOI] [PubMed] [Google Scholar]
- Joyce JA, Pollard JW. Microenvironmental regulation of metastasis. Nat Rev Cancer. 2009;9:239–252. doi: 10.1038/nrc2618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leek RD, Hunt NC, Landers RJ, Lewis CE, Royds JA, Harris AL. Macrophage infiltration is associated with VEGF and EGFR expression in breast cancer. J Pathol. 2000;190:430–436. doi: 10.1002/(SICI)1096-9896(200003)190:4<430::AID-PATH538>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- Leuschner F, Dutta P, Gorbatov R, Novobrantseva TI, Donahoe JS, Courties G, Lee KM, Kim JI, Markmann JF, Marinelli B, et al. Therapeutic siRNA silencing in inflammatory monocytes in mice. Nature biotechnology. 2011;29:1005–1010. doi: 10.1038/nbt.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin EY, Jones JG, Li P, Zhu L, Whitney KD, Muller WJ, Pollard JW. Progression to malignancy in the polyoma middle T oncoprotein mouse breast cancer model provides a reliable model for human diseases. Am J Pathol. 2003;163:2113–2126. doi: 10.1016/S0002-9440(10)63568-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin EY, Nguyen AV, Russell RG, Pollard JW. Colony-stimulating factor 1 promotes progression of mammary tumors to malignancy. J Exp Med. 2001;193:727–740. doi: 10.1084/jem.193.6.727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ojalvo LS, Whittaker CA, Condeelis JS, Pollard JW. Gene expression analysis of macrophages that facilitate tumor invasion supports a role for Wnt-signaling in mediating their activity in primary mammary tumors. J Immunol. 2010;184:702–712. doi: 10.4049/jimmunol.0902360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patsialou A, Wang Y, Lin J, Whitney K, Goswami S, Kenny PA, Condeelis JS. Selective gene-expression profiling of migratory tumor cells in vivo predicts clinical outcome in breast cancer patients. Breast Cancer Res. 2012;14:R139. doi: 10.1186/bcr3344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patsialou A, Wyckoff J, Wang Y, Goswami S, Stanley ER, Condeelis JS. Invasion of human breast cancer cells in vivo requires both paracrine and autocrine loops involving the colony-stimulating factor-1 receptor. Cancer Res. 2009;69:9498–9506. doi: 10.1158/0008-5472.CAN-09-1868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poincloux R, Lizarraga F, Chavrier P. Matrix invasion by tumour cells: a focus on MT1-MMP trafficking to invadopodia. J Cell Sci. 2009;122:3015–3024. doi: 10.1242/jcs.034561. [DOI] [PubMed] [Google Scholar]
- Qian BZ, Pollard JW. Macrophage diversity enhances tumor progression and metastasis. Cell. 2010;141:39–51. doi: 10.1016/j.cell.2010.03.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roussos ET, Balsamo M, Alford SK, Wyckoff JB, Gligorijevic B, Wang Y, Pozzuto M, Stobezki R, Goswami S, Segall JE, et al. Mena invasive (MenaINV) promotes multicellular streaming motility and transendothelial migration in a mouse model of breast cancer. Journal of cell science. 2011;124:2120–2131. doi: 10.1242/jcs.086231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sahin U, Weskamp G, Zheng Y, Chesneau V, Horiuchi K, Blobel CP. A sensitive method to monitor ectodomain shedding of ligands of the epidermal growth factor receptor. Methods in molecular biology. 2006;327:99–113. doi: 10.1385/1-59745-012-X:99. [DOI] [PubMed] [Google Scholar]
- Sharma VP, Beaty BT, Patsialou A, Liu H, Clarke M, Cox D, Condeelis JS, Eddy RJ. Reconstitution of in vivo macrophage-tumor cell pairing and streaming motility on one-dimensional micro-patterned substrates. IntraVital. 2012;1:77–85. doi: 10.4161/intv.22054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takenawa T, Suetsugu S. The WASP-WAVE protein network: connecting the membrane to the cytoskeleton. Nat Rev Mol Cell Biol. 2007;8:37–48. doi: 10.1038/nrm2069. [DOI] [PubMed] [Google Scholar]
- Thrasher AJ, Burns SO. WASP: a key immunological multitasker. Nat Rev Immunol. 2010;10:182–192. doi: 10.1038/nri2724. [DOI] [PubMed] [Google Scholar]
- Valastyan S, Weinberg RA. Tumor metastasis: molecular insights and evolving paradigms. Cell. 2011;147:275–292. doi: 10.1016/j.cell.2011.09.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wyckoff J, Wang W, Lin EY, Wang Y, Pixley F, Stanley ER, Graf T, Pollard JW, Segall J, Condeelis J. A paracrine loop between tumor cells and macrophages is required for tumor cell migration in mammary tumors. Cancer Res. 2004;64:7022–7029. doi: 10.1158/0008-5472.CAN-04-1449. [DOI] [PubMed] [Google Scholar]
- Wyckoff JB, Segall JE, Condeelis JS. The collection of the motile population of cells from a living tumor. Cancer Res. 2000;60:5401–5404. [PubMed] [Google Scholar]
- Wyckoff JB, Wang Y, Lin EY, Li JF, Goswami S, Stanley ER, Segall JE, Pollard JW, Condeelis J. Direct visualization of macrophage-assisted tumor cell intravasation in mammary tumors. Cancer Res. 2007;67:2649–2656. doi: 10.1158/0008-5472.CAN-06-1823. [DOI] [PubMed] [Google Scholar]
- Yao F, Svensjo T, Winkler T, Lu M, Eriksson C, Eriksson E. Tetracycline repressor, tetR, rather than the tetR-mammalian cell transcription factor fusion derivatives, regulates inducible gene expression in mammalian cells. Hum Gene Ther. 1998;9:1939–1950. doi: 10.1089/hum.1998.9.13-1939. [DOI] [PubMed] [Google Scholar]
- Zicha D, Allen WE, Brickell PM, Kinnon C, Dunn GA, Jones GE, Thrasher AJ. Chemotaxis of macrophages is abolished in the Wiskott-Aldrich syndrome. Br J Haematol. 1998;101:659–665. doi: 10.1046/j.1365-2141.1998.00767.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.