Abstract
An electrophysiological analysis of canine single ventricular myocardial (VM) and Purkinje (P) cells was carried out by means of whole cell voltage clamp method. The following results in VM versus P cells were obtained. INa3 was present, had a threshold negative to the fast activating–inactivating INa1, its slow inactivation was cut off by INa1, and contributed to Na+ influx at INa1 threshold. INa1 was smaller and had a less negative threshold. There was no comparable slowly inactivating INa2, accounting for the shorter action potential. Slope conductance at resting potential was about double and decreased to a minimum value at the larger and less negative IK1 peak. The negative slope region of I-V relation was smaller during fast ramps and larger during slow ramps than in P cells, occurred in the voltage range of IK1 block by Mg2+, was not affected by a lower Vh and TTX and was eliminated by Ba2+, in contrast to P cells. ICa was larger, peaked at positive potentials and was eliminated by Ni2+. Ito was much smaller, began at more positive values, was abolished by less negative Vh and by 4-aminopyridine, included a sustained current that 4-aminopyridine decreased but did not eliminate. Steeper ramps increased IK1 peak as well as the fall in outward current during repolarization, consistent with a time-dependent block and unblock of IK1 by polyamines. During repolarization, the positive slope region was consistently present and was similar in amplitude to IK1 peak, whereas it was small or altogether missing in P cells. The total outward current at positive potentials comprised a larger IK1 component whereas it included a larger Ito and sustained current in P cells. These and other results provide a better understanding of the mechanisms underlying the action potential of VM and P cells under normal and some abnormal (arrhythmias) conditions.
Keywords: Cardiac electrophysiology, ionic currents, single ventricular myocardial and cardiac Purkinje cells, whole cell patch clamp method
Introduction
The different functions of Purkinje (P) and ventricular myocardial (VM) cells are associated with several electrophysiological and mechanical differences (e.g., see Lin and Vassalle 1978; Cordeiro et al. 1998). Thus, the action potential (AP) of canine P fibers is longer (+71%, Lin and Vassalle 1978), their plateau is more negative (e.g., Baláti et al. 1998) and their twitch is shorter (−40%) and smaller (−79%) (Lin and Vassalle 1978) than in ventricular myocardial fibers.
The longer AP of P cells appears related to a greater Na+ influx during the plateau through the slowly inactivating sodium current INa2 (Vassalle et al. 2007; Bocchi and Vassalle 2008). Indeed, the Purkinje fiber AP is markedly shortened by tetrodotoxin (TTX; Coraboeuf et al. 1979; Vassalle and Bhattacharyya 1980; Bhattacharyya and Vassalle 1982; Iacono and Vassalle 1990; Baláti et al. 1998) and by local anesthetics (Vassalle and Bhattacharyya 1980; Bhattacharyya and Vassalle 1981), whereas is prolonged by high [Na+]o and the Na+-channel agonist veratridine (Iacono and Vassalle 1990). In contrast, AP duration of ventricular myocytes is very little affected by TTX (Coraboeuf et al. 1979; Bhattacharyya and Vassalle 1982; Iacono and Vassalle 1990; Baláti et al. 1998), by local anesthetics (Vassalle and Bhattacharyya 1980), by veratridine and high [Na+]o (Iacono and Vassalle 1990).
These findings suggest that sodium influx during the action potential may be greater in Purkinje fibers because it also includes INa2, which slowly inactivates at plateau potentials (Vassalle et al. 2007; Bocchi and Vassalle 2008). In addition, in P cells the slowly inactivating sodium current INa3 is activated at potential negative to that of INa1 threshold (Rota and Vassalle 2003). Whether INa2 and INa3 are also present in VM cells or whether Na+ currents have identical features in P and VM cells have not been determined.
Furthermore, it is not known whether there are differences in negative slope (NS) and positive slope (PS) regions of the I-V relation between the two tissues. In P cells, INa3 and INa2 are involved in the NS region (Rota and Vassalle 2003), but the role of the block and unblock of inward rectifying IK1 channels (Ishihara 1997; Ishihara and Ehara 1998) in the NS and PS regions, respectively, is undefined. Furthermore, whether the mechanisms underlying NS and PS region are similar or differ in P and VM cells is unknown.
There are differences in electrophysiological features of other currents as rabbit P cells express smaller IK1, a larger transient outward current Ito than VM cells (Cordeiro et al. 1998) and a greater Ito sensitivity to TEA (Han et al. 2000). Whether voltage-and time-dependent features of IK1, Ito, sustained current, and ICa differ in P and VM cells have not been determined.
The general aim of the present experiments was to investigate several ionic currents by means of a whole cell patch clamp method in canine P and VM cells isolated with the same technique to determine their features (e.g., presence or absence, threshold potential, magnitude, time- and voltage-dependent characteristics).
The specific aims included the determination of differences in the following features in P and VM cells: (1) IK1 inward rectification and its characteristics; (2) slope conductance over the voltage range of the action potential; (3) presence of INa3 and INa2 and their characteristics; (4) INa1 amplitude; (5) threshold potential for different Na+ currents and their voltage- and time-dependent inactivation; (6) contribution of INa3 to peak INa1; (7) presence and magnitude of NS and PS regions and their underlying mechanisms; (8) Ito peak and sustained outward current; (9) magnitude and voltage range of the inward component related to ICa; and (10) identification of the various currents by different means including different Vh, different ramps slopes, and channel blockers.
It was found that the differences in ionic currents between P and VM cells are numerous and substantial and provide insights in the different mechanisms that shape the action potentials, in their modification by some physiological and pharmacological factors and in the mechanisms of induction of some ventricular arrhythmias.
Material and Methods
Institutional and national guide for the care and use of laboratory animals was followed. The protocols for the experiments were reviewed and approved by the local Animal Care and Use Committee.
The details of the methods have been published (Rota and Vassalle 2003; Vassalle et al. 2007; Bocchi and Vassalle 2008). In brief outline, adult dogs (beagle, n = 25) of either sex were euthanized by intravenous injection of sodium pentobarbital (60 mg kg−1). Once the respiration had stopped, the hearts were removed and rinsed in physiological saline solution. Purkinje fiber bundles and thin papillary muscles or trabeculae (diameter ≤ 1 mm) were cut from both ventricles and were driven at 60/min for 30 min while being superfused in a tissue bath at 37°C.
The composition of physiological saline solution in mmol L−1 was NaCl 140, KCl 5.4, CaCl2 1.8, MgCl2 1, HEPES 5.0, and glucose 5.5. The solution was gassed with 100% O2 and adjusted to pH 7.4 with NaOH. The P and VM fibers were then rinsed with Ca-free solution with added 25 mmol L−1 taurine, 5 mmol L−1 beta-hydroxybutyric acid and 5 mmol L−1 Na pyruvate for 5 min in the same tissue bath and washed in a test tube three times with the same Ca-free solution. Ca-free solution contained in mmol L−1: NaCl 140, KCl 5.4, KH2PO4 1.2, MgCl2 1.5, HEPES 5.0, and glucose 5.5 (pH adjusted to 7.2 with NaOH).
P and VM tissues were separately digested at 37.5°C in Ca-free physiological saline solution to which collagenase (1 mg/mL, type VIII, Sigma, St. Louis, MO), elastase (0.6 mg/mL, type II-A, Sigma), and essentially fat-free bovine serum albumin (2 mg/mL) had been added (“enzyme solution”). The cells were separated from the digested fibers by agitation by means of a mechanical “triturator” (Datyner et al. 1985). The cells were suspended in Kraftbrühe (KB) solution and samples of the cell suspension were perfused with physiological saline solution at 37°C in a chamber located on the stage of an inverted microscope (Nikon Diaphot, Nikon, Tokyo, Japan).
Whole cell patch clamp technique was employed using an Axopatch 1D amplifier. The pipettes were filled with the following solution (in mmol L−1): K-aspartate 100, KCl 30, MgCl2 2.0, EGTA 11.0, Na-HEPES 10.0, Na2-ATP 2.0, NaGTP 0.1, CaCl2 5.0 (pH 7.2) (resistance of filled pipettes 2–4 MΩ). The free Ca2+ in the pipette solution was 110 nmol L−1 as calculated using a computer program (WinMAXC 2.40; http://stanford.edu/cpatton/maxc.html). The electrical signals were digitized at 333 kHz 12-bit resolution using A/D converter (Digidata 1200, Axon Instruments, Foster City, CA) and recorded using Clampex software (pCLAMP 8.0, Axon Instruments) and low-pass filtering at 2 kHz.
We elected to study ionic current profiles under physiological conditions (intact intracellular and extracellular ionic concentration and absence of channel blockers). Although this approach does not allow to fully isolate single currents, it preserves ionic balances and electrochemical gradients during the acquisition. Therefore, the currents in P and VM cells were studied in the absence of any channel blocker (such as Ba2+, Ni2+, tetrodotoxin, 4-Aminopyridine, etc.) to compare and contrast the currents under physiological conditions and to avoid the multiple effects of channel blockers on currents and ionic gradients. Later on, we identified the current under study and their role on different parameters in different ways, including different Vh, different ramps slopes, and channel blockers.
Successive command steps of the same protocol were applied at intervals of at least 5 sec and different protocols were separated by intervals of 3–5 min to allow the effects of each procedure to fully subside.
The data were analyzed by means pCLAMP program (Axon Instruments Inc.). Steps from different holding potentials (Vh) were applied to activate voltage- and time-dependent currents and depolarizing and repolarizing ramps with different slopes were used to study the currents under different conditions. On step depolarization from Vh −80 mV, INa1 was often cut off at −10 nA by the saturation of the amplifier. As no differences were detected in the results obtained from male and female dog cells, the results were pooled together.
The amplitude of the slowly decaying component of INa2 was measured as the difference between the current at the beginning and the end of the step. The beginning was taken as the value at the intersection between the rapidly inactivating INa1 and the backward extrapolation of INa2, also checked by fitting the slowly inactivating INa2 with a double exponential function.
Unless otherwise specified, the current traces were fitted with two term standard exponential function using the Chebyshev technique with Clampfit software according to equation (1):
| (1) |
where A1 and A2 are the amplitudes, and τs and τf are the time constants and C is the offset constant.
Data were analyzed by mean of the Clampfit (pCLAMP 10.2) and Microsoft Excel programs. The results of tests carried out for each procedure are shown in the tables as means ± SEM (standard error of the mean) together with the number (n) of cells studied. Student's paired t test between two terms of comparison and one-way ANOVA (analyses of variance) between a data group were applied and a P < 0.05 was considered significant and was marked by an asterisk (*) in the tables and in text.
Results
INa3 and its relation to INa1
In P cells, INa3 appears at a potential (−57.8 mV) which is negative to INa1 threshold (−52 mV) (Rota and Vassalle 2003). INa1 suppresses the slow inactivation of INa3, as at its threshold INa1 is not followed by time-dependent current (Rota and Vassalle 2003; Vassalle et al. 2007; Bocchi and Vassalle 2008). Whether INa3 also is present in VM cells or whether its slow inactivation is suppressed by INa1 is not known.
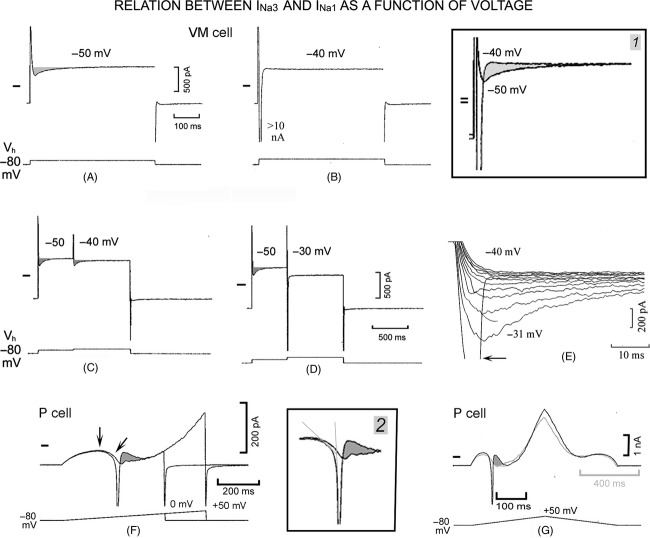
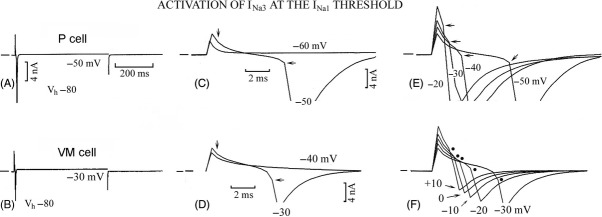
In Figure 1A, in a VM cell during the step from Vh −80 mV to −50 mV, an inward current appeared that decayed bi-exponentially. In Figure 1B, the step to −40 mV elicited INa1 (partially shown), which (as in P cells) was not followed by a slowly inactivating component. In Figure 1 inset 1, the shaded area emphasizes the fact that INa3 slow inactivation was present at −50 mV and absent at −40 mV.
Figure 1.
INa3 and its relation to INa1. In a VM cell, a depolarizing step from Vh −80 mV to −50 mV (lower trace in A) and to −40 mV (B) elicited the currents shown in the upper traces. The current traces have been superimposed in inset 1 and the shaded area emphasizes the suppression of the slow inactivation of INa3 by INa1. In C, a conditioning step was applied to −50 mV and a test step to −40 mV and in D to −50 and −30 mV, respectively. In E, depolarizing steps were applied from Vh −80 to −40 mV and increased by 1 mV to −30 mV: the horizontal arrow points to inactivating INa1. In F, in a P cell, ramps with the same slope and different duration were applied. Downward vertical arrow points to IK1 peak and downward oblique arrow to the negative slope region. In inset 2, part of the F traces are shown at greater gain. Dashed lines emphasize the different slopes of inward currents prior to and at beginning of INa1. In F and G, the shaded areas show that at the end of INa1 inactivation the magnitude of the outward current approached that of IK1 peak. The short dash in each panel indicates zero current in this and subsequent figures. In G, ramps with different slopes (260 mV sec−1, gray trace; 520 mV sec−1, black trace) were applied.
The absence of INa3 during the step at INa1 threshold potential could be due to either the suppression of INa3 slow inactivation by INa1 (as in P cells) or to the less negative voltage. To clarify this point, a two-step protocol was applied, as a suitable conditioning step may reduce INa1 channel availability just enough to shift its threshold to a less negative value. In Figure 1C, the conditioning step to −50 mV induced INa3 and the test step to −40 mV failed to activate INa1 and induced a smaller INa3. The finding suggests that in Figure 1B INa3 inactivation was not present because it was suppressed by INa1 and not because it could not occur at −40 mV.
In Figure 1D, the −30 mV test step initiated an inward transient (−3037 pA) which was followed by a small shallow tail (−93 pA), suggesting the induction of INa2 with a small slow inactivation component.
In n = 10, with the two steps protocol in VM cells during the step from −80 mV to −50 mV INa3 amplitude was −143.2 ± 54.9 pA and during the test step to −40 mV it was −72.4 ± 9.9 pA (not significantly different). During the test step to −30 mV, the inward transient was −3045 ± 576 pA and was followed by a decaying tail of 63.7 ± 14.8 pA. Therefore, in VM cells INa3 was present during the −40 mV test step in the absence of INa1. During the −30 mV test step, INa2 was followed by a small and quickly inactivating component (see below). Similarly, in P cells (n = 10) INa3 could be activated at the INa1 threshold if the activation of INa1 was prevented by the conditioning step. One difference with the VM cells was that in P cells the slowly inactivating INa2 was much larger (+673.1%*; see below).
INa3 was studied in VM and P cells by applying single steps (Fig. 1A) from Vh −80 mV (Table 1). With respect to P cells, in VM cells during depolarizing steps INa3 was consistently present, had a less negative threshold (*), and similar amplitude as well as time constants of inactivation.
Table 1.
INa3 in P and VM cells and its changes with lower Vh
| Vh (mV) | Param | VM cells | P cells |
|---|---|---|---|
| −80 | Th (mV) | −46.7 ± 1.1 | −53.3 ± 1.9* |
| INa3 (pA) | −168 ± 52 (18/18) | −217 ± 102 (13/18) | |
| τf (msec) | 15.3 ± 2.9 | 10.5 ± 2.8 | |
| τs (msec) | 82.7 ± 11.9 | 55.9 ± 12.9 | |
| −70 | Th (mV) | −43.3 ± 1.3 | −52.5 ± 1.8* |
| INa3 (pA) | −189 ± 66 (15/16) | −190 ± 63 (11/16) | |
| −60 | Th (mV) | −38.5 ± 1.0 | −46.6 ± 1.2* |
| INa3 (pA) | −120 ± 29 (12/15) | −235 ± 71 (14/15 | |
| −50 | Th (mV) | −33.6 ± 1.5 | −35.6 ± 2.0 |
| INa3 (pA) | −179 ± 76 (11/16) | −155 ± 42 (14/16) | |
| −40 | Th (mV) | −21.0 ± 1.0 | −27.1 ± 1.8* |
| INa3 (pA) | −29.9 ± 18.3 (3/10) | −84 ± 35 (6/9) |
Vh (mV), holding potential in mV; Param, parameters measured; VM cells, data from ventricular myocardial cells; P cells, data from Purkinje cells; Th (mV), threshold potential in mV of INa3; INa3 (pA), amplitude in pA of INa3 measured as the difference between its peak and the end of the step; τf (msec) and τs (msec), fast and slow time constants, respectively, of INa3 inactivation; Numbers in parenthesis (e.g., 18/18), number of cells in which INa3 was present over the total number of cells studied; *statistically significant difference between P and VM cells data.
The finding that the −50 mV conditioning step prevented the appearance of INa1 but not of INa3 during the −40 mV test step suggests that INa3 might be less sensitive to voltage-dependent inactivation than INa1. This was tested by applying depolarizing steps from gradually less negative Vh. As shown in Table 1, in P and VM cells with gradually less negative Vh, the amplitude of INa3 decreased very little until Vh was −40 mV and the threshold remained less negative (*) in VM cells. At all Vh, in both P and VM cells the inactivation of INa3 was slow in the absence of INa1. Even with Vh −40 mV, INa3 inactivated with τf 13.1 ± 7.5 msec and τs 41.3 ± 18.5 msec in VM cells and with τf 7.5 ± 1.4 msec and τs 82.1 ± 22.6 msec in P cells.
INa3 voltage-dependent increase and sudden suppression of its slow inactivation by INa1
Gradually increasing depolarizing steps might lead to a progressive increase in INa3. In Figure 1E, in a VM cell with depolarizing steps increasing by 1 mV between the INa3 and INa1 thresholds, INa3 magnitude increased progressively and inactivated relatively more quickly up to −29 mV. At −30 mV (INa1 threshold), the inactivation of INa1 (arrow) suppressed INa3 slow decay. Similar results with steps increasing at intervals of 1 mV were obtained in VM cells (n = 4) and in P cells (n = 16).
The above results suggest that during depolarizing ramps INa3 might precede INa1, as the continuous decline in voltage would initiate and increase INa3 at potentials negative to the INa1 threshold. Furthermore, the current at the end of INa1 inactivation would be expected to be more outward than in its absence due to the suppression of INa3 slow inactivation. In some cells, applying ramps of different duration with a borderline slope (150 mV sec−1) for INa1 activation led to a nonuniform induction of INa1, so that the events in the presence and absence of INa1 could be compared in the same cell as shown in Figure 1F.
In a P cell, during the ramps, the outward current gradually increased before peaking (IK1 peak, vertical arrow). The slope of the ramps being borderline for INa1 activation, during the shorter ramp, IK1 peak was followed by a negative slope (NS) region (oblique arrow). No INa1 was present and the current during the NS region (INS) was followed by a reincreasing outward current. During the longer ramp, INS more quickly turned inward and its steeper slope merged into that of the activating INa1, as emphasized by the gray lines in Figure 1 inset 2.
In Figure 1F and inset 2, the shaded area shows that the current at the end of INa1 inactivation was more outward than in the INa1 absence, as expected from the suppression of the slow inactivation of INa3 by INa1. The inactivation of INa1 was still followed by NS region with smaller amplitude and less steep slope, consistent with the slow inactivation of INa2 (see below). The patterns illustrated in Figure 1F were present in n = 3, the consistent presence of INa1 with its inactivation approaching IK1 peak in n = 4 and the absence of INa1 with the consistent presence of INS in n = 19.
In another approach, ramps with different slopes (Fig. 1G) were applied, as in several instances in P cells no INa1 was initiated during slower ramps. In Figure 1G, the 260 mV sec−1 ramp (lighter trace) did not activate INa1, whereas the superimposed 520 mV sec−1 ramp (darker trace) did. During the slower ramp, IK1 peak was followed by INS but not by INa1. Instead, with the steeper ramp, the end of INa1 inactivation approached the IK1 peak and was followed by INS.
The asymmetry between slower activation and faster inactivation of the overall Na+ current (Fig. 1 F and G) was a consistent finding that might be expected from INa3 preceding INa1 and its slow inactivation being cut off by it.
In VM cells, INa1 was less frequently absent with slower ramps. With 260 mV sec−1 ramps, INa1 was absent in 9/17 P cells and in 3/17 VM cells whereas with the 520 mV sec−1 ramp, INa1 was absent only in 3/17 P cells and in none of 17 VM cells (the NS region being present with or without INa1).
Therefore, in VM cells INa3: (1) was present with a similar magnitude and rate constants of inactivation; (2) had a threshold less negative than in P cells and negative to that of INa1; (3) increased progressively at potentials between its threshold and that of INa1; (4) its slow inactivation was consistently eliminated by INa1; (5) could appear and inactivate slowly at voltages less negative than INa1 threshold if INa1 activation was prevented by a conditioning step; (6) contributed to the beginning of INS during depolarizing ramps, and (7) was less sensitive than INa1 to voltage- and time-dependent inactivation.
Currents during larger depolarizing steps in Purkinje and myocardial cells
Ionic currents in P and VM cells were investigated also by applying 500 msec depolarizing steps from Vh −80 mV to +40 mV in increments of 10 mV (Fig. 2, protocol in c).
Figure 2.
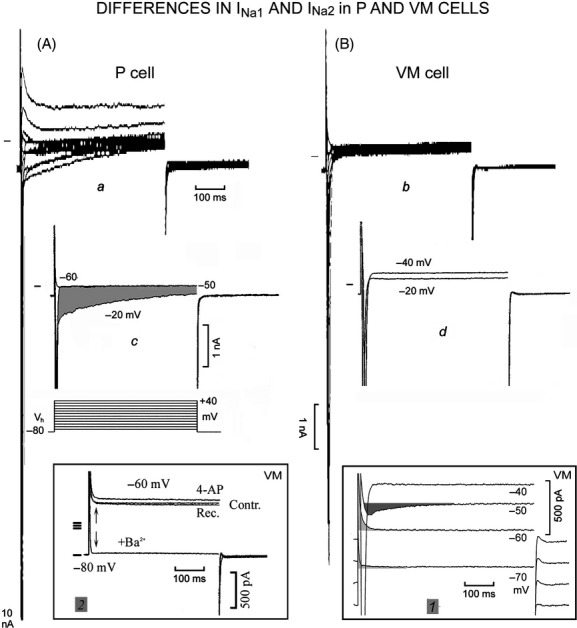
Larger range of inward and outward currents in P than in VM cells. The protocol is shown at the bottom of c. In the P cell, the currents flowing during the steps from Vh −80 to −60, −50, and −20 mV are shown in c, where the shaded area emphasizes the slow decay of INa2. In the VM cell, the currents recorded with steps from Vh −80 to −40 and −20 mV are shown in d. In inset 1, the current traces were displaced by 200 pA for a better visualization. The light-shaded areas emphasize the time-dependent component of IK1 and the dark-shaded area INa3. In inset 2, the current traces during the −60 mV step are shown in control (Contr.) in the presence of 4-aminopyridine (4-AP), of 4-AP plus Ba2+ (+Ba2+), and during recovery (Rec.). The upward arrow indicates the initial decline in the outward current to a steady value and the downward arrow its suppression by Ba2+.
In Figure 2, in the P cell (A) the range of inward and outward currents was larger than in the VM cell (Fig. 2B). In P cell, INa1 was truncated by the saturation of the amplifier at −10,000 pA whereas in VM cell the largest INa1 was −8428 pA. In the P cell (Fig. 2c), the −50 mV step initiated INa1 whose inactivation was followed by a steady current that overlapped the current trace at −60 mV (where no INa1 was present; see also Vassalle et al. 2007; Bocchi and Vassalle 2008), thus suggesting no loss of voltage control. The slowly inactivating INa2 appeared at −40 mV and reached its largest value during −20 mV step (shaded area in Fig. 2c). Typically, INa2 was still decreasing by the end of the 500 msec step (Vassalle et al. 2007; Bocchi and Vassalle 2008).
In the VM cell, the threshold for INa1 activation was less negative (−40 mV, Fig. 2d) than in the P cell (−50 mV). During the depolarizing −20 mV step, the inactivation of INa1 was not followed by a decaying INa2, in contrast to the P cell. The comparison of Figure 2a and b indicates that in VM cell the large and slowly decaying INa2 was absent at other potentials as well. The sustained current at the end of −50 mV step (measured as the difference from the holding current Ih) was larger in the VM cell than in the P cell. In the VM cell, the sustained current at −40 mV (INa1 threshold) was similar to that at −50 mV (not shown). In both cells, the sustained current at −20 mV was less outward than at −50 mV (P cell) and at −40 mV (VM cell), as expected from the onset of INS. With more positive steps, the outward currents were far larger in P cell than in VM cell (cf. Fig. 2a and b).
In Table 2, with Vh −80 mV, in VM cells INa1 threshold was less negative in P cells (*), INa1 was smaller (*) in VM cells, as it was truncated in all P cells, but only in 13/18 VM cells. In both VM and P cells, INa1 inactivated exponentially with τ ∼1.5 msec. In VM cells, INa1 was smaller (*) than in P cells also with Vh −70 mV when INa1 was less often truncated.
Table 2.
INa1 in P and VM cells and its changes with lower Vh
| Vh (mV) | Param | VM cells | P cells |
|---|---|---|---|
| −80 | Th (mV) | −36.7 ± 1.1 | −48.3 ± 1.5* |
| INa1 (pA) | −8857 ± 461 (18/18) | −10, 000 ± 0.0* (18/18) | |
| τ (msec) | 1.3 ± 0.2 | 1.6 ± 0.2 | |
| −70 | Th (mV) | −33.7 ± 1.2 | −43.1 ± 1.8* |
| INa1 (pA) | −8551 ± 445 (16/16) | −9622 ± 263.0* (16/16) | |
| τ (msec) | 1.5 ± 0.2 | 1.6 ± 0.2 | |
| −60 | Th (mV) | −30.7 ± 1.5 | −34.7 ± 1.7 |
| INa1 (pA) | −6167 ± 959 (15/15) | −7984 ± 844 (15/15) | |
| τ (msec) | 1.4 ± 0.2 | 1.6 ± 0.2 | |
| −50 | Th (mV) | −23.3 ± 1.4 | −26.0 ± 1.3 |
| INa1 (pA) | −2153 ± 618 (11/16) | −3404 ± 820 (14/16) | |
| τ (msec) | 2.4 ± 0.7 | 1.6 ± 0.1 | |
| −40 | Th (mV) | −13.3 ± 3.3 | −18.75 ± 1.3 |
| INa1 (pA) | −58.1 ± 33 (3/10) | −642 ± 325 (5/8) | |
| τ (msec) | 3.7 ± 0.6 | 2.9 ± 0.4 |
INa1 (pA), amplitude of INa1 from its beginning to its peak in pA; τ (msec), time constant of the exponential inactivation in ms of INa1; VM cells, data from ventricular myocardial cells; P cells, data from Purkinje cells; Th (mV), threshold potential in mV of INa1; Numbers in parenthesis (e.g., 18/18), number of cells in which INa1 was present over the total number of cells studied; *statistically significant difference between P and VM cells data.
In Table 3, at −20 mV in VM cells an inactivating INa2 tail was present in 6/18 cells, was small and decayed quickly. In contrast, in P cells the slowly inactivating INa2 was much larger (*) and inactivated more slowly (*). With gradually less negative Vh, INa1 (Table 2) and INa2 (Table 3) gradually decreased.
Table 3.
INa2 in P and inward tail in VM cells and their changes with lower Vh
| Vh (mV) | Param | VM cells | P cells |
|---|---|---|---|
| −80 | Peak (mV) | −20.0 ± 0.0 | −21.2 ± 0.8 |
| INa2 slow (pA) | −110.2 ± 39.3 (6/18) | −1212 ± 208* (15/18) | |
| τf (msec) | 4.0 ± 1.8 | 8.1 ± 1.0* | |
| τs (msec) | 60.8 ± 22.1 | 233.9 ± 24.7* | |
| −70 | Peak (mV) | −20.0 ± 0.0 | −20.6 ± 0.6 |
| INa2 slow (pA) | −66.1 ± 25.8 (9/16) | −1985 ± 263* (15/16) | |
| −60 | Peak (mV) | −20.0 ± 0.0 | −22.1 ± 1.1 |
| INa2 slow (pA) | −46.1 ± 20.0 (5/15) | −854 ± 140* (13/15) | |
| −50 | Peak (mV) | −13.3 ± 2.1 | −15.3 ± 1.6 |
| INa2 slow (pA) | −35.7 ± 18.4 (5/16) | −382 ± 195 (9/16) |
Peak (mV), voltage at which the largest slowly inactivating INa2 in P cells or in VM cell was measured; INa2 slow (pA), amplitude of slowly inactivating INa2 in pA, measured from its beginning to the end of the step; VM cells, data from ventricular myocardial cells; P cells, data from Purkinje cells; τf (msec) and τs (msec), fast and slow time constants, respectively, of INa2 inactivation; Numbers in parenthesis (e.g., 6/18), number of cells in which INa2 was present over the total number of cells studied; *statistically significant difference between P and VM cells data.
The slope conductance was measured by superimposing small hyperpolarizing pulses on the parent steps in VM cells (n = 9). At the INa1 threshold, after the INa1 inactivation, the slope conductance was minimal and did not vary with time, as shown in P cells by Bocchi and Vassalle (2008). This finding also is consistent with no loss of voltage control.
Time-dependent decay of IK1 on depolarization in Purkinje and myocardial cells
In Figure 2, in the same VM cell, the currents during the −70, −60, −50, and −40 mV steps are shown in inset 1. At the beginning of the −70 and −60 mV steps, the outward current quickly declined (light-shaded areas) to a steady value. At −50 mV, INa3 appeared and declined slowly (dark-shaded area), and at −40 mV INa1 quickly activated and inactivated to a steady value.
One possible explanation for the initial decline of the outward current at −60 and −70 mV might be a noninstantaneous block of IK1 by polyamines during the depolarizing steps (Ishihara 1997; Ishihara and Ehara 1998), a block which is eliminated by Ba2+ (Ishihara and Ehara 1998). In Figure 2 inset 2, the traces during the step to −60 mV were recorded in control (Contr.), in the presence of 4-aminopyridine (4-AP), of Ba2+ (+Ba2+), and during recovery (Rec.). The upward arrow indicates the initial decline in the outward current to a steady value, a decline that was little affected by 4-AP. Instead, Ba2+ suppressed both the initial decay and the steady current during the step (downward arrow).
In Table 4, with Vh −80 mV, at the voltages indicated in VM cells the outward current decreased more (*) in VM cells than in P cells. The time constant of the exponential decline was similar (∼6 msec).
Table 4.
IK1 time-dependent decay during depolarizing steps from different Vh
| Vh (mV) | Param | VM cells | P cells |
|---|---|---|---|
| −80 | Measured at (mV) | −58.8 ± 0.7 | −59.6 ± 6.4 |
| IK1 decay (pA) | 317 ± 29 (18/18) | 237 ± 24* (18/18) | |
| τ (msec) | 6.3 ± 0.9 | 5.9 ± 1.2 | |
| −70 | Measured at (mV) | −55.6 ± 1.2 | −59.3 ± 0.6* |
| IK1 decay (pA) | 226 ± 3.7 (16/16) | 62.9 ± 16.5* (11/16) | |
| −60 | Measured at (mV) | −50.0 ± 0.0 | −50.0 ± 0.0 |
| IK1 decay (pA) | 78.4 ± 26.2 (6/15) | 21.1 ± 18.2 (2/15) | |
| −50 | Measured at (mV) | −40.0 ± 0.0 | −40.0 ± 0.0 |
| IK1 decay (pA) | 24.1 ± 15.6 (4/16) | 12.8 ± 8.8 (2/16) |
Measured at (mV), voltage in mV at which the decay of IK1 was measured; IK1 decay (pA), amplitude of IK1 time-dependent decay at beginning of step; τ (msec), time constant of IK1 exponential decay; Vh (mV), holding potential in mV; VM cells, data from ventricular myocardial cells; P cells, data from Purkinje cells; Numbers in parenthesis (e.g., 18/18), number of cells in which IK1 decay was present over the total number of cells studied; *statistically significant difference between P and VM cells data.
If the decline of the current during the step is indeed due to a time-dependent block of IK1 on depolarization, then decreasing Vh should reduce the declining current, as increasing degrees of IK1 block would occur during the less negative Vh, prior to the depolarizing step. To test this point, Vh was reduced in 10 mV increment to −40 mV. As shown in Table 4, in both P and VM cells with gradually less negative Vh, the initial decay of the current became gradually smaller and less frequent, and disappeared altogether with Vh −40 mV. These findings indicate a time- and voltage-dependent block of IK1, which was significantly larger in VM cells at Vh −70 and −60 mV.
Contribution of INa3 to Na+ inflow due to INa1
As INa3 occurs also at the INa1 threshold, at that potential INa3 would be expected to precede INa1 and therefore contribute to the peak Na+ current. To verify such a possibility, the current traces at the beginning of depolarizing steps were displayed at suitably greater time base.
In Figure 3, in a P cell (A) and in a VM cell (B), at the usual time base only INa1 was visible. However, when the traces were displayed at much greater time base, a slower inward component (comprised between the downward and horizontal arrows) preceded INa1 both in P (Fig. 3C) and in VM cell (Fig. 3D). In Figure 3C (P cell), the trace recorded at −60 mV show the initial decay of the outward current to a steady value (time-dependent block of IK1). Instead, the trace recorded at −50 mV departed from the exponential decay (vertical arrow), crossed in an inward direction the −60 mV trace (as expected from the activation of INa3). After a delay, it was followed by the fast initiation of INa1 (sudden beginning of the steeper slope, horizontal arrow). Similar events occurred in the VM cell (Fig. 3D).
Figure 3.
The activation of INa3 precedes that of INa1 at the latter's threshold. In A (P cell) and B (VM cell), the traces are shown at the usual time base whereas in C, D, E, and F the traces are shown at the greater time base indicated. In C and D, the downward vertical arrows point to the beginning of INa3 and the leftward horizontal arrows point to the beginning of INa1. The traces recorded at the beginning of the −50, −40, −30, and −20 mV steps have been superimposed in E (P cell) and those at the beginning of the steps to −30, −20, −10, 0, and +10 mV have been superimposed in F (VM cell). The arrows in E and the dots in F indicate the beginning of INa1. The numbers next to the traces indicate the voltage of the respective steps.
In Figure 3E (P cell), the current traces at the beginning of −50, −40, −30, and −20 mV steps were superimposed and show the progressively earlier onset of INa1 with larger depolarizing steps until no distinct INa3 component was apparent. In Figure 3F, similar events occurred in the VM cell in that larger depolarizing steps elicited an earlier INa1 with the eventual disappearance of INa3.
In n = 18, in VM cells, INa3 (measured between the departure of the trace from exponential decay and the sudden onset of INa1) had a magnitude of −2677 ± 201 pA and a duration of 2.2 ± 0.3 msec; INa1 (measured from the sudden increase in steepness to its peak) had a threshold of –36.6 ± 1.1 mV) and an amplitude of −8857 ± 461 pA. In P cells, INa3 had a amplitude of −2330 ± 235* pA and a duration of 2.6 ± 0.8 msec; INa1 had a more negative threshold (−48.3 ± 1.4* mV) and an amplitude >−10,000 ± 0.0* pA.
Thus, INa3 was a substantial fraction of the inward current flowing at the threshold for INa1. With steps to less negative values, INa3 consistently decreased and INa1 was activated sooner.
Differences in I-V relation of the sustained current in Purkinje and myocardial cells
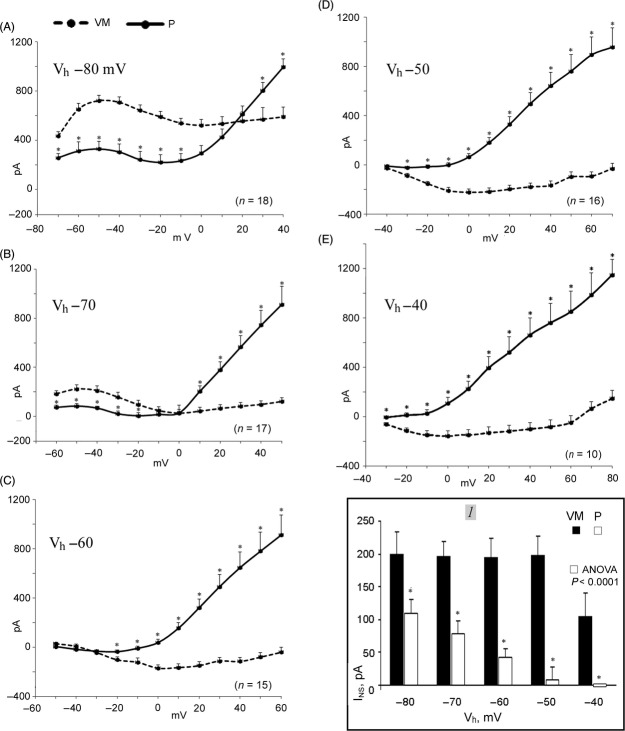
To investigate the quasi-steady state I-V relations in VM and P cells, the sustained current at the end of 500 msec depolarizing steps from Vh −80 mV to +40 mV was measured as the difference from Ih. The same procedure was applied with less negative Vh to determine how the I-V relation would be affected in P versus VM cells.
In Figure 4, in A, with Vh −80 mV, in VM cells the sustained current was more outward at negative potentials and less outward at positive potential than in P cells. In both types of cells, past IK1 peak, the NS region was present, but in VM cells INS was larger and peaked at a less negative value (∼0 mV), whereas in P cells the smaller INS peaked at ∼−20 mV. The outward current began to increase at +10 mV in VM cells and at −10 mV in P cells, suggesting a different Ito threshold.
Figure 4.
The I-V relation as a function of Vh in P and VM cells. The number of P and VM cells studied is indicated in parenthesis in each panel. The ordinates show the magnitude of the sustained current in pA at the end of 500 ms depolarizing steps applied from the Vh indicated in each panel to the voltage in mV indicated on the abscissae. The VM cells mean data are connected by dashed line and those of P cells by continuous line. The vertical bars indicate the standard error of the mean. In inset 1, the mean values of INS are the difference between the most outward sustained current and the subsequent least outward sustained (or the largest inward) current in pA at voltage indicated on abscissa. The asterisks (*) indicate a statistical difference between the data in VM and P cells. The difference between the values in P cells at the various Vh was statistically significant (ANOVA P < 0.0001).
INS was differently affected by lower Vh in P and VM cells. With Vh −70 (Fig. 4B), overall the sustained current was much less outward in both VM and P cells, as expected from the inward rectification of IK1 channel at less negative Vh. However, INS was still large in VM cells whereas it was diminished in P cells, suggesting that in P cells the decrease in INS might be related to a partial inactivation of INa2.
This interpretation is supported by the findings with still lower Vh. In VM cells, the current became inward and INS persisted unaltered up to Vh −50 mV (Fig. 4B–D). Only with Vh −40 mV did INS decrease (Fig. 4E) as apparently the channel contributing to INS was partially blocked prior to the depolarizing step. Instead, in P cells INS markedly decreased with Vh −60 to disappear altogether with Vh −40 mV. In Figure 4 inset 1, the graph shows the difference between the outward current peak prior to the NS region (corresponding to IK1 peak) and the smallest current value of I-V relation prior to the reincrease in outward current (a measure of INS peak). The graph shows how differently INS amplitude varied in VM and P cells as a function of Vh, the decrease of INS in P cells being statistically significant (ANOVA < 0.0001).
The increase in outward current with the larger depolarizing steps was much greater in P than in VM cells (helped in this by the inward shift of the current in VM cells), and it was little affected by less negative Vh. At each Vh, the outward current in P cells increased past ∼−20 mV, as expected for Ito. As the protocol applied at different Vh was the same, with the gradually less negative Vh the depolarizing steps attained gradually more positive values. Hence, the sustained current with the largest depolarizations increased to similar values in spite of the decreasing Vh.
These results raise the possibility that in P cells Na+ currents might mainly contribute to INS (with this protocol, INa2) whereas the voltage-dependent block of IK1 channel may predominantly determine INS in VM cells.
INa2 and the ICa component in Purkinje and myocardial cells
As at plateau voltages the slowly inactivating INa2 prevails in P cells and presumably ICa prevails in VM cells, the different amplitude, voltage range, voltage- and time-dependent inactivation of INa2 in P cells and of the ICa component in VM cells were investigated as shown in Figure 5.
Figure 5.
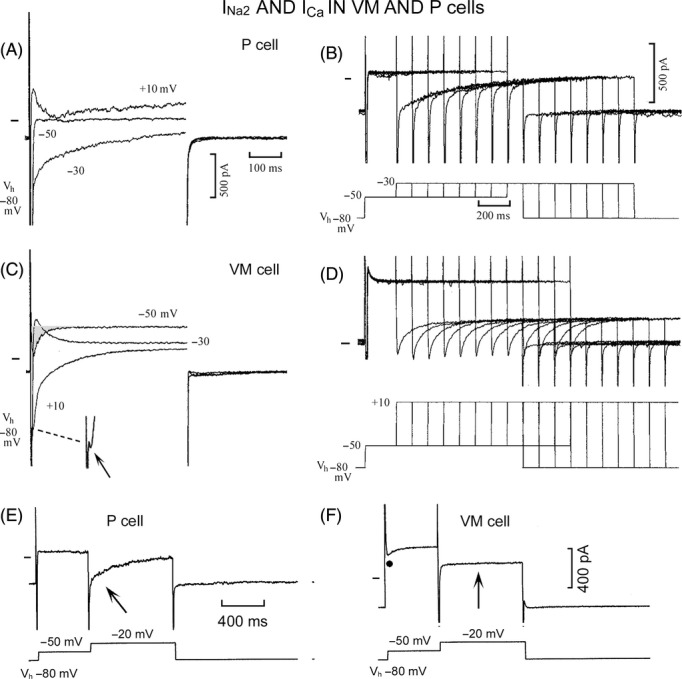
Different amplitude, voltage range, kinetics, and time-dependent inactivation of INa2 in P cells and of ICa in VM cells. Steps were applied from Vh −80 to −50, −30 mV +10 mV in the P cell (A) and in the VM cell (C) where the shaded area emphasizes INa3 and the small arrow points to the beginning of a large ICa. In the P cell (B), progressively longer conditioning steps to −50 mV were followed by test steps to −30 mV. In the VM cell (D), conditioning steps to −50 mV were followed by test steps to +10 mV. In E, the oblique upward arrow points to inactivating INa2 during the test step to −20 mV in the P cell. In F, the filled circle labels INa3 and the vertical upward arrow points to the absence of comparable slowly inactivating INa2 at −20 mV in the VM cell.
In the P cell (Fig. 5A), a step from Vh −80 to −50 mV elicited INa1 which was not followed by time-dependent currents, as usual. During the step to −30 mV, INa1 was followed by the slowly decaying INa2 (τs 297 msec). During the +10 mV step, a small inward component was superimposed on a small outward current. In the VM cell (Fig. 5C), the step from Vh −80 to −50 mV elicited INa3 (shaded area). During the step to −30 mV, INa1 was followed by a small outward component, but not by decaying INa2. During the +10 mV step, INa1 was followed by a large ICa component (−1699 pA) which inactivated with a τs of 106 msec and whose beginning during INa1 inactivation is indicated by the arrow in the magnified trace.
In the P cell (Fig. 5B), after progressively longer conditioning steps at −50 mV, the test steps to −30 mV elicited a gradually smaller inactivating INa2 (with the last test step, −47%) (see Bocchi and Vassalle 2008). In the VM cell (Fig. 5D), the conditioning step was the same, but (as there was no INa2 at −30 mV) test steps were applied to the voltage where ICa component was largest (+10 mV, protocol in Fig. 5D). The test step elicited an inward current, whose amplitude was not decreased by progressively longer conditioning steps (with the last test step, +3.7%). In the VM cell, ICa decayed more quickly than INa2 did in the P cell. The findings point to a different voltage range, kinetics, and voltage- and time-dependent inactivation of INa2 in P cells and of ICa in VM cells.
In order to separate INa1 from INa2, a double step protocol was applied from Vh −80 mV to −50 and to −20 mV in a P cell (Fig. 5E) and in a VM cell (Fig. 5F). In the P cell, at −50 mV INa1 was followed by a steady current, and at −20 mV INa2 activated rapidly and decayed slowly (oblique arrow). In the VM cell, at −50 INa3 was present as usual at that voltage (filled circle; see Fig. 1) and at −20 mV an inward transient was followed by a faint and brief tail at a potential where the slowly decaying INa2 was large in the P cell.
With Vh −80 mV, in VM cells at +20 mV the ICa component was −436.7 ± 105.2 pA (17/17 cells) with τf 19.1 ± 8.4 msec and τs 112 ± 15.4 msec, whereas in P cells at +18.8 mV the ICa component was −96.8 ± 44.5* pA (present in 6/17 cells) with τf 11.4 ± 6.3 msec and τs 157.6 ± 61.2* msec.
I-V relation during slow depolarizing and repolarizing ramps in myocardial and Purkinje cells
Because in VM cells the sustained current at the end of depolarizing steps was larger at potentials negative to IK1 peak and smaller at positive potentials than in P cells (Fig. 4), the steady state I-V relation was studied during slowly depolarizing and repolarizing ramps in the two tissues.
In Figure 6A, in the P cell during 6.5 mV sec−1 depolarizing ramp, the outward current increased gradually less to stop increasing altogether between point 1 (−58 mV, IK1 peak) and point 2 (−30.7 mV). On further depolarization between points 2 and 3, the outward current increased markedly and before the ramp peak, underwent an enhancement (shaded area, the “bulge”; Du and Vassalle 1999). During the repolarizing ramp, the outward current initially decreased more rapidly, but 16 sec after the ramp peak it was similar to 16 sec before (at IK1 peak) (292 and 275 pA, respectively).
Figure 6.
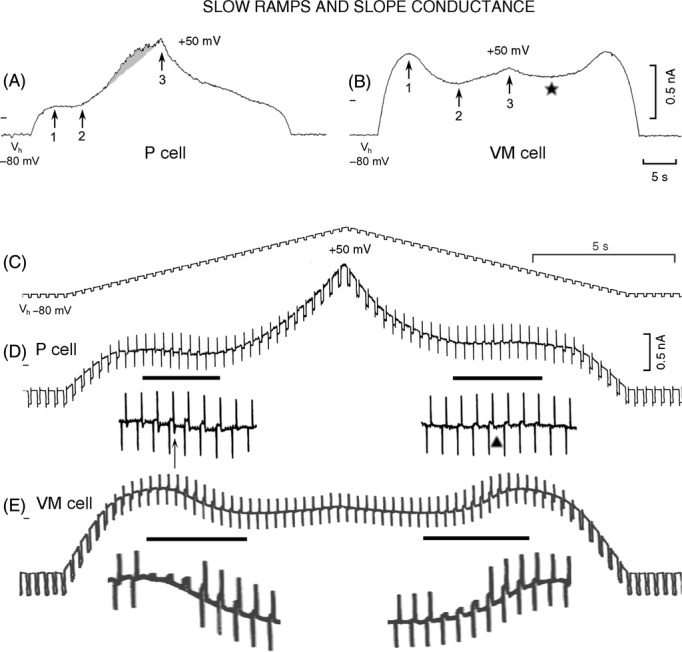
I-V relation and changes in slope conductance during slow ramps in P and VM cells. A 6.5 mV sec−1 depolarizing and repolarizing ramp was applied to a P cell (A) and to a VM cell (B). Point 1 indicates the IK1 peak, point 2 the beginning of increasing outward current, and point 3 the current at ramp peak. In A, the shaded area indicates the enhancement of the outward current (“the bulge”). In B, the asterisk indicates the transition between the decreasing outward current and the beginning of IPS. Hyperpolarizing voltage pulses (amplitude 7 mV, duration 200 msec, rate 90 min−1; C) were superimposed on the parent ramp to measure slope conductance. In D (P cell) and E (VM cell) (same heart but different from that for A and B), the horizontal lines indicate the sections of current records shown at higher gain underneath. In D, the upward vertical arrow points to an inward transient after the reversed pulse current and the triangle points to its absence. In the enlarged sections of VM trace, some of the capacity spikes have been deleted for a better visualization of the increase and reversal of pulse current in the NS and PS regions.
In Figure 6B, in the VM cell, the outward current also increased gradually less as a function of depolarization, but at point 1 (−48 mV, IK1 peak) the outward current was 179% larger than in the P cell. Also, a distinct NS region began at point 1 and was followed (change in slope) by an ICa component that peaked at +2 mV (point 2). Between points 2 and 3, the current reincreased in an outward direction, but it was much smaller than in the P cell. Furthermore, there was no enhancement of the outward current (no “bulge”) and at the ramp peak (point 3) the current was less outward than at point 1, in sharp contrast with the P cell.
During the repolarizing ramp, the outward current decreased much less than in the P cell and it was less inward (star) than at point 2. The outward current reincreased in the positive slope (PS) region to a peak value (786 pA) similar to that of IK1 peak (769 pA). Past the peak of IPS, the outward current underwent a progressively quicker decrease as a function of repolarization.
In Table 5, with respect to P cells, in VM cells IK1 was larger (*) and peaked at a less negative potential (*). INS was larger (*), was more frequently present and peaked at less negative value (*). The ICa component was larger when measured from its beginning to its peak and when the ICa peak was compared to the symmetrical peak during repolarizing ramp. The outward current began to reincrease (“Ito start”) at less negative potential (*) to reach a value at the ramp peak (“Ito”) which was smaller (*) (although it was similar when measured with respect to Ih).
Table 5.
Currents during 6.5 mV sec−1 ramp in VM and P cells
| n 15, Param | VM cells | P cells | Δ (mV or %) |
|---|---|---|---|
| Vh (mV) | −80.6 ± 0.6 | −81.3 ± 0.9 | −0.7 mV |
| IK1 peak (pA) | 965 ± 124 | 465 ± 57* | +107.5% |
| IK1 peak (mV) | −44.2 ± 1.7 | −50.1 ± 1.2* | 5.9 mV |
| INS (pA) | −218 ± 31 (14/15) | −45.6 ± 12.0* (10/15) | +379.3% |
| INS peak (mV) | −3.9 ± 3.1 | −23.2 ± 5.1* | 19.3 mV |
| ICa start (mV) | −11.6 ± 5.2 | −4.4 ± 3.0 | −7.2 mV |
| ICa (pA) | −39.9 ± 17.5 (6/15) | −20.1 ± 9.6 (4/15) | +98.5% |
| ICa peak (mV) | −5.1 ± 4.7 | −10.5 ± 7.2 | 5.4 mV |
| Δ (pA) | −51.9 ± 6.9 (15/15) | −29.8 ± 9.3 (7/15) | +74.1% |
| Ito start (mV) | 0.64 ± 2.1 | −19.2 ± 3.7* | 19.8 mV |
| Ito (pA) | 109 ± 14 | 436 ± 62* | −75% |
| Iramp peak−Ih (pA) | 812 ± 114 | 863 ± 71 | −5.9% |
| Irepol (pA) | 64.7 ± 10.8 | 396.4 ± 62.2* | −83.6% |
| Irepol peak (mV) | 8.8 ± 2.6 | −15.9 ± 4.6* | 24.7 mV |
| IPS start (mV) | 8.9 ± 2.6 | −5.6 ± 2.7* | 14.5 mV |
| IPS peak (pA) | 197.0 ± 28.4 (15/15) | 36.0 ± 19.0* (5/15) | +447.2% |
| IPS peak (mV) | −44.4 ± 1.6 | −52.1 ± 1.8 | 7.7 mV |
| IPS peak−Ih (pA) | 981 ± 124 | 428 ± 68* | +53.3% |
n 15, number of cells studied; Δ (mV or %), difference in mV or percent of VM cells data with respect to P cells data; IK1 peak (pA), amplitude of IK1 peak in pA, measured as the difference from Ih; IK1 peak (mV), voltage in mV at which IK1 peaked; INS (pA), current amplitude in pA during the negative slope region; INS, peak (mV), voltage in mV of INS peak; ICa start (mV), beginning of ICa component in mV determined as the departure of current trace from INS peak; ICa (pA), amplitude of ICa component in pA as the difference between its beginning and its peak; ICa peak (mV), peak in mV of ICa; Δ (pA), difference in pA between ICa peak during depolarization and minimum outward current preceding the beginning of IPS on repolarization; Ito, start (mV), voltage in mV at which the increasing outward current started at INS or ICa peaks; Ito (pA), amplitude of outward current in pA measured between its beginning and ramp peak; Iramp peak−Ih (pA), outward current at ramp peak measured as difference from Ih; Irepol (pA), amplitude in pA of the outward current between ramp peak and its smallest value prior to the beginning of IPS; Irepol peak (mV), voltage in mV at which the outward current was smallest prior to INS beginning; IPS start (mV), voltage in mV at which IPS began; IPS peak (pA), current in pA at IPS peak, measured as difference between its beginning and its peak; IPS peak (mV), voltage in mV at which PS region peaked; IPS peak−Ih (pA), current in pA measured as difference between INS peak and Ih; Vh (mV), holding potential in mV; Param, parameters measured; VM cells, data from ventricular myocardial cells; P cells, data from Purkinje cells; *statistically significant difference between P and VM cells data.
During the repolarizing ramp, the decrease of outward current (Irepol) was smaller (*) and peaked at less negative potential (*). IPS (consistently present in VM but not in P cells) began at a more positive potential (*) and it was much larger (*) as it was (*) when measured with respect to Ih although less so. In VM cells, IK1 peak was similar to IPS peak as it was in P cells at IPS peak (or at the value at which the outward current began to decrease rapidly). This suggests that in neither tissue the Na+ currents contributed to IK1 or IPS peaks.
Thus, with slow ramps, with respect to P cells, in VM cells: (1) IK1 peak was much larger and peaked at a less negative potential; (2) INS was larger and peaked at less negative potentials; (3) ICa component was larger; (4) the outward current enhancement prior to ramp peak (the “bulge”) was absent; (5) the outward current at ramp peak was much smaller (but less so when compared to Ih); (6) during repolarizing ramps, the smaller Irepol declined much less; (7) IPS was present more frequently, was larger and with a more positive beginning, and (8) IPS and IK1 peaks were similar, both being larger than the ramp peak current.
Slope conductance changes during slow ramps in Purkinje and myocardial cells
If, during depolarization, the gradually smaller increase in outward current is due to the inward rectification of IK1 channel, the slope conductance should decrease accordingly. To find out, the slope conductance was measured by superimposing small hyperpolarizing voltage pulses on the parent ramp (protocol in Fig. 6C).
In the P cell (Fig. 6D), the amplitude of the pulse current at Vh −80 mV decreased gradually on depolarization to reach a minimal value at −44 mV, just prior the beginning of a small NS region. During INS, the pulse current reversed polarity and reincreased. With further depolarization, the pulse current decreased again, became once more negative and reincreased in amplitude. During the repolarizing ramp, similar events occurred in reverse order, including a smaller increase in slope conductance during a rather small IPS.
The sections of the traces labeled with a horizontal line are shown underneath at higher gain for a better visualization of pulse current changes. The arrow under the magnified trace points to a small inward component that followed the outward pulse current: such an inward component was not present during the repolarizing ramp (triangle) or in the VM cells (see below), suggesting that the brief hyperpolarizing step allowed an increased availability of sodium channels.
In the VM cell (Fig. 6E), at Vh the amplitude of the pulse current was larger (+72%) than in the P cell. The pulse current decreased on depolarization to become minimal at −31 mV, reversed polarity during INS and increased to a maximum at −14 mV, decreased again and then underwent a much smaller increase than in the P cell during the remainder of the ramp. During the repolarizing ramp, similar events occurred in reverse order, including an increase in slope conductance during IPS.
In VM cells (n = 11, of which 3 from the same hearts as P cells) the pulse current amplitude varied as follows: −381.7 ± 34.6 pA at Vh −81.8 mV, 0 pA at IK1 peak (−39.2 mV), +75.6 ± 19.8 pA at the −14.3 mV reversal peak during INS (which was −310.3 ± 80.3 pA, 11/11 cells), and −16.7 ± 4.9 pA at ramp peak. With repolarizing ramps, the pulse current amplitude was −107.1 ± 18.5 pA at +13.7 mV, +84.6 ± 22.0 pA at −16.1 mV during IPS, and 3.9 ± 3.9 pA (10/11 cells) at IPS peak (which was at −42.5 ± 1.7 mV). IK1 and IPS peaks were 1208 ± 207 and 1224 ± 210 pA, respectively.
In P cells (n = 21), the pulse current amplitude varied as follows. It was −190.8 ± 20.1* pA at Vh −88.5 mV and 0 pA at IK1 peak (−47.1 ± 1.5* mV). INS was 7.2 ± 4.4* pA and was present only in 3/21 cells. The pulse current amplitude was −110.0 ± 11.7* pA at the ramp peak. During the repolarizing ramp, the pulse current amplitude was −71.4 ± 6.8 pA at +17.7 mV (* with respect to −119 pA at +20.1 mV during depolarization) and 2.5 ± 2.2 pA at the potential where the final faster depolarization began (IPS was present in 2/21 cells).
In the cells from the same three hearts with the same −83.3 mV Vh, similar results were obtained in that in VM cells the pulse current amplitude at Vh was larger by +88.8%, the pulse current fell to 0 pA at a less negative potential, INS was +923% larger, the pulse current at ramp peak smaller by −87.5%, the reversed pulse current during IPS was 73 pA (there was no IPS in P cells).
Thus, with respect to P cells, in VM cells the pulse current: (1) was larger at Vh (+100.0%*); (2) fell to a minimum at the 7.9 mV* less negative IK1 peak; (3) consistently reversed and reincreased during INS; (4) fell again by INS end and reincreased but much less (−84.8%* than P cells at the ramp peak; and (5) during the repolarizing ramp, the smaller VM conductance underwent the converse changes, reincreasing during IPS.
Current during fast ramps in myocardial and Purkinje cells
As with the 6.5 mV sec−1 ramps, the Na+ channels would be inactivated, depolarizing and repolarizing ramps with progressively steeper slopes were applied to VM and P cells.
In Figure 7, 260 mV sec−1 ramp, with respect to the P cell (Fig. 7A), in the VM cell (Fig. 7B) at point 1 IK1 peak was much larger and less negative. In both P and VM cells, immediately after IK1 peak, a slowly increasing INa3 (shaded areas labeled by downward arrows) preceded the activation of INa1, as expected from the more negative threshold of INa3 (see Figs. 1 and 3). In the P cell, INS (empty circle) peaked at point 2 and was followed by an increasing outward current. A very shallow inward component, peaking at +12 mV, appeared as an “indentation” on the increasing outward current (empty square and small shaded area). In the VM cell, INS (filled circle) was smaller and was followed by a large inward component (filled square) which peaked at +17 mV, as expected from a larger ICa component.
Figure 7.
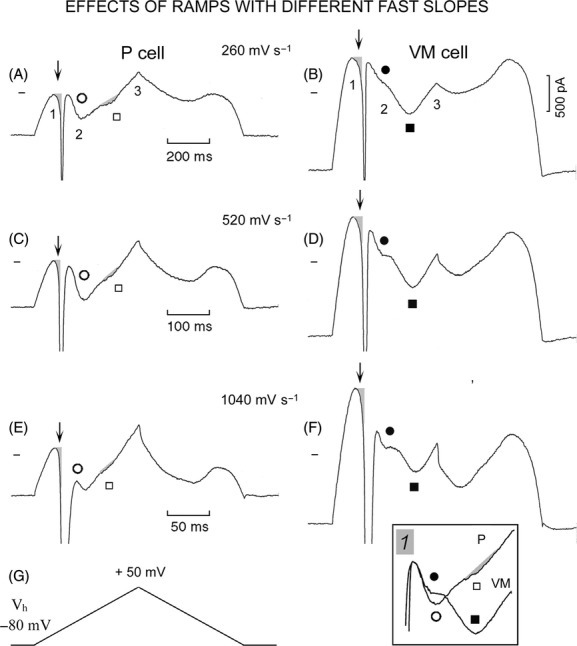
I-V relation during steep ramps in P and VM cells. The 260, 520, and 1040 mV sec−1 ramps were applied from Vh −80 to +50 mV (G) to a P cell (A, C, and E, respectively) and a VM cell (B, D, and F, respectively). The numbers 1, 2, and 3 label the peaks of IK1, of INS, and of the ramp, respectively. INS is labeled by empty circles in the P cell and by filled circles in the VM cell. The inward component attributable to ICa is labeled by empty squares and small shaded areas in the P cell, and by filled squares in the VM cell. In VM cells, ICa was measured from its beginning (taken as the point at which the slope of INS met the backward extrapolation of ICa) and ICa peak. The downward vertical arrows point to the slowly increasing inward current (shaded areas) preceding the activation of INa1. In inset 1, the traces from C and D were superimposed by the end of INa1 inactivation. In both cells, INa1 was cut off by the saturation of the amplifier at −10 nA.
Similar but not identical results were obtained during the 520 mV sec−1 (Fig. 7C and D) and 1040 mV sec−1 (Fig. 7E and F) ramps. IK1 peak increased in magnitude with the steeper ramps both in P and VM cell, still being much smaller in the P cell. In both cells, the steeper ramp slope caused INa1 to inactivate closer to the end of INS. As usual, the outward current at ramp peak (point 3) was larger than IK1 peak (point 1) in the P cell (Fig. 7C and E) whereas it was smaller in the VM cell (Fig. 7D and F). INS of the P cell (Fig. 7C) and that of the VM cell (Fig. 7D) were superimposed by the end of INa1 inactivation in Figure 7 inset 1. While INS was larger in the P cell, the subsequent ICa component was much larger (filled square) in the VM cell.
On repolarization, in the P cell the outward current decreased more and peaked at more negative values than in the VM cell. In both cells, on repolarization with steeper ramps the decrease in outward current was faster initially and it was larger. Also, IPS started from a less outward value and, on that account, IPS peak became smaller than the IK1 peak.
As shown in Table 6, during the 260 mV sec−1 ramp with respect to P cells, in VM cells IK1 peak was larger (*) and less negative (*), INS was smaller and its peak was less negative (*). INa3 amplitude was somewhat greater, INa1 threshold was less negative (*), INa1 amplitude was similar (there was INa1 in 14/17 VM cells and in 8/17 P cells).
Table 6.
Currents during the 260 mV sec−1 ramp in VM and P cells
| n 17, Param | VM cells | P cells | Δ (mV or %) |
|---|---|---|---|
| Vh (mV) | −80.6 ± 0.6 | −81.8 ± 1.0 | −1.2 mV |
| IK1 peak (pA) | 913 ± 101 | 477 ± 56.8* | +91.4% |
| IK1 peak (mV) | −52.1 ± 1.0 | −57.3 ± 1.0* | 5.2 mV |
| INS (pA) | −216.6 ± 26.3 | −340.5 ± 44.3* | −36.3 % |
| INS peak (mV) | −14.0 ± 1.6 | −26.5 ± 1.5* | 12.5 mV |
| INa3 (pA) | −941 ± 119 (14/17) | −733 ± 89 (8/17) | +28.3% |
| INa1Th (mV) | −40 ± 1.5 | −47.3 ± 1.8* | −7.2 mV |
| INa1 (pA) | −7298 ± 842 (14/17) | −6978 ± 850 (8/17) | +4.5% |
| End INa1 (pA) | 910 ± 109 (14/17) | 324 ± 64* (8/17) | +180.8% |
| End INa1 (mV) | −34.2 ± 1.5 | −39.5 ± 1.5* | 5.3 mV |
| ICa start (mV) | −9.2 ± 2.0 | −15 ± 1.0* | 5.8 mV |
| ICa (pA) | −108.3 ± 24.2 (15/17) | −19.6 ± 11.6* (3/17) | +452.5% |
| ICa peak (mV) | 14.0 ± 0.9 | −3.3 ± 3.7* | 17.3 mV |
| Ito start (mV) | 13.7 ± 0.9 | −21.3 ± 2.8* | 35 mV |
| Ito (pA) | 206.0 ± 25.3 (17/17) | 1270 ± 166* (17/17) | −83.7% |
| Iramp peak−Ih (pA) | 783 ± 78 (17/17) | 1373 ± 166* (17/17) | −42.9% |
| Irepol (pA) | 123.5 ± 22 | 967 ± 139* | −87.2% |
| Irepol peak (mV) | 7.2 ± 3.2 | −15.9 ± 1.6* | 23.1 mV |
| IPS start (mV) | 3.3 ± 3.2 | −17.2 ± 1.7* | 20.4 mV |
| IPS (pA) | 214 ± 29.8 (17/17) | 64.6 ± 13.2* (15/17) | +231.2% |
| IPS peak (mV) | −45.2 ± 1.6 | −46.7 ± 2.9 | 1.5 mV |
| IPS peak−Ih (pA) | 895 ± 103 (17/17) | 395 ± 58* (15/17) | +126.5% |
End INa1 (pA), Amplitude in pA of the current at the end of INa1 inactivation; End INa1 (mV), voltage in mV of the current at the end of INa1 inactivation; Vh (mV), holding potential in mV; Param, parameters measured; VM cells, data from ventricular myocardial cells; P cells, data from Purkinje cells; INa1 Th (mV), threshold potential in mV of INa1; INa3 (pA), amplitude in pA of INa3 measured as the difference between its peak and the end of the step; INa1 (pA), amplitude in pA of INa1; Numbers in parenthesis (e.g., 14/17), number of cells in which the parameter was present over the total number of cells studied; *statistically significant difference between P and VM cells data. Other explanations as in the legend of Table 5.
The ICa component was larger (*) (there was a measurable ICa component in15/17 VM cells and in 3/17 P cells). The outward current between ICa and ramp peaks (“Ito”) began at a more positive potential (*) and was smaller (*). When the ramp peak current was measured as the difference from Ih, Ito was smaller (*) but less so, due to the larger IK1 upon which the ramp peak current was superimposed.
The IK1 peak was similar to the current at the end of INa1 inactivation both in the VM cells (0.003%) and in the eight P cells in which INa1 was present (IK1 peak 389.1 pA and at the end of INa1 324.6 pA, difference not statistically significant). These results are consistent with the elimination of the slow inactivation of INa3 by INa1 in both tissues. At the end of INa1, the voltage was less negative in VM cells (*), reflecting the less negative INa1 threshold (*).
In VM cells, with respect to the 260 mV sec−1 ramp, the 520 (Table 7) and 1040 mV sec−1 (Table 8) depolarizing ramps induced the following changes, respectively: peak IK1 +5.4% and +18.5%, INa3 +75.0%* and +132%*, INa1 +39.3% and +53.0%*, INS +28.3% and +86.9%,* ICa component +10.0% and +3.0%, “Ito” +8.2% and +7.8%, and Iramp peak−Ih, +5.1% and +12.8%.
Table 7.
Currents during the 520 mV sec−1 ramp in VM and P cells
| n 17, Param | VM cells | P cells | Δ (mV or %) |
|---|---|---|---|
| Vh (mV) | −80.6 ± 0.5 | −81.8 ± 0.9 | −0.7 mV |
| IK1 peak (pA) | 963 ± 108 | 518 ± 57* | +46.2% |
| IK1 peak (mV) | −52.3 ± 0.7 | −58.3 ± 0.7* | 6 mV |
| INS (pA) | −278 ± 36 | −546 ± 48* | −49.0% |
| INS, peak | −18.1 ± 2.6 | −22.4 ± 1.6 | 4.3 mV |
| INa3 (pA) | −1458 ± 170 (17/17) | −1352 ± 175 (14/17) | +7.8% |
| INa1Th (mV) | −39.2 ± 1.2 | −46.8 ± 1.2* | 7.6 mV |
| INa1 (pA) | −8373 ± 553 (17/17) | −9439 ± 352 (14/17) | −11.9% |
| End INa1 (pA) | 837 ± 113 | 381 ± 69* | +119.6% |
| End INa1 (mV) | −32.6 ± 1.2 | −38.5 ± 1.0* | 5.9 mV |
| ICa, start (mV) | −4.9 ± 1.9 | −11.5 ± 0.5* | 6.6 mV |
| ICa (pA) | −119.2 ± 31.7 (16/17) | −16.4 ± 14.1* (2/17) | +626.8% |
| ICa peak (mV) | 17.4 ± 0.9 | −3.5 ± 7.5 | 20.9 mV |
| Ito, start (mV) | 10.3 ± 3.4 | −17.1 ± 2.0* (17/17) | 27.4 mV |
| Ito (pA) | 223 ± 27.0 | 1478 ± 173* (17/17) | −84.9% |
| Iramp peak−Ih (pA) | 823 ± 88 | 1405 ± 167 | −41.4% |
| Irepol (pA) | 198 ± 32 | 1169 ± 144* | −83.0% |
| Irepol peak (mV) | 3.1 ± 3.6 | −16.6 ± 2.0* | 19.4 mV |
| IPS, start (mV) | 0.2 ± 3.7 | −17.1 ± 1.8* | 17.3 mV |
| IPS (pA) | 219 ± 34 | 122 ± 20 | 79.5% |
| IPS peak (mV) | −45.3 ± 1.7 | −49.8 ± 1.0* | 4.5 mV |
| IPS peak−Ih (pA) | 850 ± 105 | 349 ± 55* | +143.5% |
n, number of cells studied; Δ (mV or%), difference in mV or percent of VM cells data with respect to P cells data; IK1 peak (pA), amplitude of IK1 peak in pA, measured as the difference from Ih; IK1 peak (mV), voltage in mV at which IK1 peaked; INS (pA), current amplitude in pA during the negative slope region; INS, peak (mV), voltage in mV of INS peak; INa1 Th (mV), voltage at which INa1 began; INa1 (pA), amplitude in pA of INa1; ICa start (mV), beginning of ICa component in mV determined as the departure of current trace from INS peak; ICa (pA), amplitude of ICa component in pA as the difference between its beginning and its peak; ICa peak (mV), peak in mV of ICa; Ito, start (mV), voltage in mV at which the increasing outward current started at INS or ICa peaks; Ito (pA), amplitude of outward current in pA measured between its beginning and ramp peak; Iramp peak−Ih (pA), outward current at ramp peak measured as difference from Ih; Irepol (pA), amplitude in pA of the outward current between ramp peak and its smallest value prior to the beginning of IPS; Irepol peak (mV), voltage in mV at which the outward current was smallest prior to INS beginning; IPS start (mV), voltage in mV at which IPS began; IPS (pA), current in pA at IPS peak, measured as difference between its beginning and its peak; IPS peak (mV), voltage in mV at which PS region peaked; IPS peak−Ih (pA), current in pA measured as difference between INS peak and Ih; End INa1 (pA), amplitude in pA of the current at the end of INa1 inactivation; End INa1 (mV), voltage in mV of the current at the end of INa1 inactivation; Vh (mV), holding potential in mV; Param, parameters measured; VM cells, data from ventricular myocardial cells; P cells, data from Purkinje cells; INa3 (pA), amplitude in pA of INa3 measured as the difference between its peak and the end of the step; Numbers in parenthesis (e.g., 17/17), number of cells with the parameter present over the total number of cells studied; *statistically significant difference between P and VM cells data.
Table 8.
Currents during the 1040 mV sec−1 ramp in VM and P cells
| n 17, Param | VM cells | P cells | Δ (mV or %) |
|---|---|---|---|
| Vh (mV) | −80.6 ± 0.5 | −81.8 ± 0.9 | 1.2 mV |
| IK1 peak (pA) | 1082 ± 113 | 614 ± 60* | +76.2% |
| IK1 peak (mV) | −52.2 ± 0.7 | −58.0 ± 0.7* | 5.8 mV |
| INS (pA) | −405 ± 57 | −710 ± 74* | −42.9% |
| INS peak (mV) | −16.2 ± 2.6 | −19.8 ± 1.4 | 3.6 mV |
| INa3 (pA) | −1986 ± 132 | −1645 ± 199 | +20.7% |
| INa1 Th (mV) | −38.3 ± 1.1 | −47.1 ± 0.9* | 8.8 mV |
| INa1 (pA) | −9197 ± 393 (17/17) | −9491 ± 280 (16/17) | −3.09% |
| End INa1 (pA) | 863 ± 105 (17/17) | 351 ± 69* (16/17) | +145.8% |
| End INa1 (mV) | −27.9 ± 1.5 | −30.8 ± 4.4 | 2.9 mV |
| ICa start (mV) | −0.6 ± 1.7 | −8.0 ± 0.0 | 7.2 mV |
| ICa (pA) | −111.6 ± 28.6 (14/17) | −19.5 ± 18.0* (2/17) | +472% |
| ICa peak (mV) | 20.9 ± 1.1 | −4.0 ± 1* | 24.9 mV |
| Ito start (mV) | 14.0 ± 4.0 | −14.1 ± 3.25* | 28.1 mV |
| Ito (pA) | 222 ± 28 | 1776 ± 187* | −87.5% |
| Iramp peak−Ih (pA) | 884 ± 85 | 1658 ± 172 | −46.6% |
| Irepol (pA) | 396 ± 50 | 1522 ± 158* | −73.9% |
| Irepol peak (mV) | 10 ± 3.3 | −16.7 ± 2.0* | 26.7 mV |
| IPS start (mV) | 5.4 ± 3.6 | −18.9 ± 1.9* | 24.3 mV |
| IPS (pA) | 223 ± 40 | 163 ± 24 (15/17) | +36.8% |
| IPS peak (mV) | −47.2 ± 1.7 | −50.4 ± 1.0 | 3.2 mV |
| IPS peak−Ih (pA) | 733 ± 100 | 244 ± 47* | +200.4% |
n, number of cells studied; Δ (mV or%), difference in mV or percent of VM cells data with respect to P cells data; IK1 peak (pA), amplitude of IK1 peak in pA, measured as the difference from Ih; IK1 peak (mV), voltage in mV at which IK1 peaked; INS (pA), current amplitude in pA during the negative slope region; INS, peak (mV), voltage in mV of INS peak; INa1 Th (mV), voltage at which INa1 began; INa1 (pA), amplitude in pA of INa1; ICa start (mV), beginning of ICa component in mV determined as the departure of current trace from INS peak; ICa (pA), amplitude of ICa component in pA as the difference between its beginning and its peak; ICa peak (mV), peak in mV of ICa; Ito, start (mV), voltage in mV at which the increasing outward current started at INS or ICa peaks; Ito (pA), amplitude of outward current in pA measured between its beginning and ramp peak; Iramp peak−Ih (pA), outward current at ramp peak measured as difference from Ih; Irepol (pA), amplitude in pA of the outward current between ramp peak and its smallest value prior to the beginning of IPS; Irepol peak (mV), voltage in mV at which the outward current was smallest prior to INS beginning; IPS start (mV), voltage in mV at which IPS began; IPS (pA), current in pA at IPS peak, measured as difference between its beginning and its peak; IPS peak (mV), voltage in mV at which PS region peaked; IPS peak−Ih (pA), current in pA measured as difference between IPS peak and Ih; End INa1 (pA), Amplitude in pA of the current at the end of INa1 inactivation; End INa1 (mV), voltage in mV of the current at the end of INa1 inactivation; Vh (mV), holding potential in mV; Param, parameters measured; VM cells, data from ventricular myocardial cells; P cells, data from Purkinje cells; INa3 (pA), amplitude in pA of INa3 measured as the difference between its peak and the end of the step; Numbers in parenthesis (e.g., 17/17), number of cells in which the parameter was present over the total number of cells studied; *statistically significant difference between P and VM cells data. Other explanations as in the legend of Table 7.
In P cells, with respect to the 260 mV sec−1 ramp, the 520 (Table 7) and 1040 mV sec−1 (Table 8) ramps induced the following changes, respectively: peak IK1 +8.5% and +28.7%, INa3 +291.8%* and +376.8%*, INa1 +187.5%* and +189.0%*, INS +60.5%* and +108.8%*, “Ito” +16.3% and +39.8%*, and Iramp peak−Ih, +2.3% and +20.7%*.
Thus, in both VM and P cells the faster ramps increased IK1 peak, INa3, INa1, INS, and Iramp peak when measured either from its beginning during the ramp or from Ih. The increase in INa3 and in INS was larger in P cells, consistent with INa3 role in INS.
As for the repolarizing ramps, with respect to P cells, in VM cells during the 260 mV sec−1 ramp (Table 6), the decreasing outward current was smaller (*) and peaked at a more positive potential (*). IPS began at a less negative potential (*) and was larger (*). It was larger also when measured as the difference from Ih (*).
In VM cells, with respect to the 260 mV sec−1 repolarizing ramp, the 520 (Table 7) and 1040 mV sec−1 (Table 8) repolarizing ramps induced the following changes, respectively: Irepol larger by +60.3% and by +220%*, the difference in initiation of IPS 3.1 and 2.1 mV, difference in voltage of IPS peak 0.1 and 2 mV, amplitude of IPS peak +0.2% and +4.2% and, when compared to Ih, −5.0% and −18.1%. Therefore, on repolarization the decrease of Irepol (but not IPS) was sensitive to the repolarizing ramp slope.
In P cells, with respect to the 260 mV sec−1 repolarizing ramp, the 520 (Table 7) and 1040 mV sec−1 (Table 8) repolarizing ramps induced the following changes, respectively: Irepol larger by 20.8% and by 57.3%*, difference in IPS initiation 0.1 and 1.8 mV, difference in voltage of IPS peak −3.1 and −3.7 mV, amplitude of IPS peak +88.8%* mV and +152.3%* and, when measured from Ih, −11.6% and −38.2%*.
Therefore, in P cells INa3 and INS became greater with faster depolarizing ramps, suggesting that sodium currents play a larger role in NS region of P cells than in that of VM cells. Also, during repolarizing ramp, the outward current decreased more with faster ramps in both P and VM cells. With faster ramps, starting from a lower value, IPS increased in P cells, but decreased when measured as the difference from Ih.
Differences in INS in Purkinje versus myocardial cells
A decrease of Vh from −90 mV to −60 mV markedly decreased the amplitude of INS in P cells (Rota and Vassalle 2003; Vassalle et al. 2007; present results), but not in VM cells as measured using the sustained current at the end of depolarizing steps (Fig. 4). The finding suggests that the Na+ current may play a predominant role in the mechanisms underlying INS in P but not in VM cells. This was tested by applying ramps from different Vh.
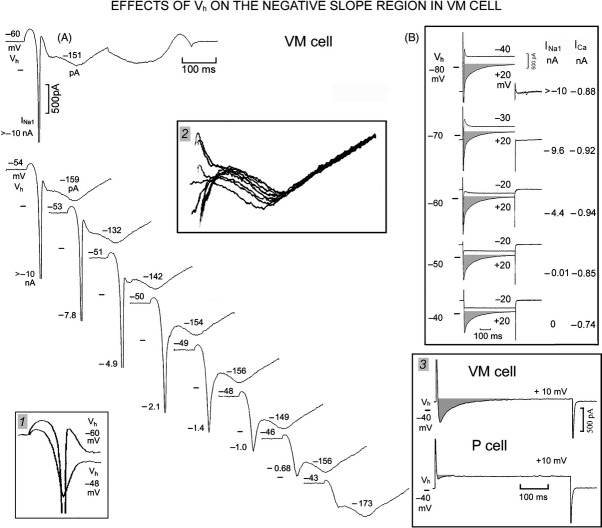
In Figure 8A, in a VM cells, Vh was progressively decreased by 1 mV and 520 mV sec−1 ramps were applied from Vh indicated above the Ih trace (−60 to −43 mV). At Vh −60 mV (top A trace), INa1 was truncated at −10 nA and INS was followed by ICa component (−151 pA, as indicated above the trace) peaking at +22 mV. With Vh −54 mV, INa1 was still larger than −10 nA, but beginning with Vh –53 mV, INa1 gradually decreased as indicated in nA next to the tip of INa1 traces. Also, as INa1 became much smaller, the activation and inactivation of the inward transient became slower, as illustrated in Figure 8 inset 1 by the superimposed Vh −60 and −48 mV traces. At Vh −43 mV (bottom trace), there was no apparent INa1 and INS had an amplitude of −462 pA, a duration of 36 msec and a voltage range between −40 and −12 mV. Over the range of Vh tested, the ICa component peaked at ∼+20 mV and its amplitude remained at ∼−150 pA (see numbers above the ICa component traces and the superimposed traces in Figure 8 inset 2).
Figure 8.
Persistence of NS region and of ICa with ramps from lower holding potentials in VM cells. In A, 520 mV sec−1 ramps were applied from the Vh indicated above Ih (−60 to −43 mV in 1 mV decrements; not all traces shown). INa1 amplitude in nA is indicated next to the tip of INa1 traces. In inset 1, the traces with Vh −60 and −48 mV have been superimposed. The amplitude in pA of ICa component (measured from its beginning to its peak) is indicated by the number above the traces. In inset 2, the superimposed traces show that ICa component was not affected by gradually smaller Vh. In B (from a different heart), depolarizing steps were applied from gradually lower Vh and the currents were superimposed at the indicated voltages. The amplitudes of INa1 and of ICa (shaded area) are indicated in nA at the right hand of the traces. In inset 3, the voltage step was applied from Vh −40 to +10 mV in a VM and P cell.
That the inward component positive to INS was due to ICa is supported by the results obtained with depolarizing steps applied from Vh −80, −70, −60, −50, and −40 mV (Fig. 8B). The amplitudes of INa1 and of that of ICa (shaded areas) are indicated, respectively, in nA at the right hand of traces. INa1 decreased gradually with the less negative Vh and disappeared with Vh −50 and −40 mV. Instead, the slowly inactivating current persisted even with Vh of −40 mV (albeit somewhat decreased) as expected for ICa (Isenberg and Klöckner 1982). In Figure 8 inset 3, depolarizing steps were applied from Vh −40 to +10 mV and show that ICa was far larger in the VM than in the P cell, as usual.
The results are consistent with Na+ currents playing little role in INS of VM cells and also with ICa underlying the inward component that peaked at ∼+20 mV (as well as the small indentation over a similar voltage range in P cells).
In Table 9, in VM cells fast ramps were applied from Vh −80 and −50 mV with the following changes. IK1 peak decreased (*) as the K1 channel rectified inwardly at lower Vh before the ramp was applied. INS was not affected whereas INa1 markedly decreased (*). The ICa component was similar. The ramp peak current (Ito) increased (the ramp peak voltage being more positive), although it was smaller when measured as Iramp peak−Ih (*) due to the inward shift of the current with less negative Vh. IPS and its peak voltage were similar. However, IPS peak measured relative to Ih was 820 pA in control and −5.0 pA* with the less negative Vh, reflecting the inward shift of IK1 with Vh −50 mV. In 6 of 7 of these experiments, Vh was also decreased by 1–2 mV with the results similar to those illustrated in Figure 8A.
Table 9.
Less negative Vh of fast ramps markedly decreases INS and IPS in P but not in VM cells
| Param | VM cells n 7 | VM cells | P cells n 4 | P cells |
|---|---|---|---|---|
| Vh (mV) | −80 ± 0 | −50 ± 0 | −82.5 ± 2.5 | −55.0 ± 2.8 |
| IK1 peak (pA) | 999 ± 223 | 92 ± 19* | 270 ± 57 | 53.0 ± 15.5* |
| IK1 peak (mV) | −47.7 ± 4.4 | −41.5 ± 2.8 | −60 ± 3.4 | −47.0 ± 2.8* |
| INS (pA) | −388 ± 75 | −246 ± 46 | −261 ± 91 | −10.5 ± 6.5 (2/4) * |
| INS peak (mV) | −9.3 ± 4.7 | −22.7 ± 3.4 | −26.3 ± 3.0 | −35.8 ± 6.3 (2/4) |
| INa1 (pA) | −7988 ± 1170 | −298 ± 87* | −9334 ± 665 | −123 ± 123 (1/4) * |
| ICa start (mV) | −7.5 ± 4.7 | −7.1 ± 5.8 | 0 ± 0 | 0 ± 0 |
| ICa (pA) | −184 ± 52 | −183 ± 77 | 0 ± 0 | 0 ± 0 |
| ICa peak (mV) | 15.5 ± 10.3 | 13.6 ± 5.4 | 0 ± 0 | 0 ± 0 |
| Ito start (mV) | 5.6 ± 7.8 | 14.2 ± 4.6 | −15.4 ± 12 | −6.8 ± 17.7 |
| Ito peak (pA) | 341 ± 87 | 542 ± 132 | 1586 ± 579 | 1789 ± 667 |
| Iramp peak−Ih (pA) | 842 ± 154 | 284 ± 98* | 1597 ± 565 | 1826 ± 677 |
| Irepol (pA) | 192 ± 53 | 511 ± 104* | 1420 ± 582 | 1831 ± 679 |
| Irepol peak (mV) | 8.6 ± 7.5 | −5.6 ± 5.2 | −31.1 ± 5.3 | −30.9 ± 9.2 |
| IPS start (mV) | 5.7 ± 8.0 | 4.4 ± 4.5 | −28.8 ± 6.8 | 0 ± 0* |
| IPS peak (pA) | 214 ± 61 | 218 ± 63 | 36.0 ± 20 (3/4) | 0 ± 0 |
| IPS peak (mV) | −39.2 ± 6.0 | −41 ± 5.0 | −50.5 ± 5.0 | 0 ± 0* |
| IPS−Ih (pA) | 820 ± 198 | −5.0 ± 15.2* | 213 ± 81 (3/4) | 24.6 ± 15* |
n, number of cells studied; IK1 peak (pA), amplitude of IK1 peak in pA, measured as the difference from Ih; IK1 peak (mV), voltage in mV at which IK1 peaked; INS (pA), current amplitude in pA during the negative slope region; INS, peak (mV), voltage in mV of INS peak; INa1 (pA), amplitude in pA of INa1; ICa start (mV), beginning of ICa component in mV determined as the departure of current trace from INS peak; ICa (pA), amplitude of ICa component in pA as the difference between its beginning and its peak; ICa peak (mV), voltage in mV at which ICa peaked; Ito, start (mV), voltage in mV at which the increasing outward current started at INS or ICa peaks; Ito peak (pA), amplitude of outward current in pA measured between its beginning and ramp peak; Iramp peak−Ih (pA), outward current at ramp peak measured as difference from Ih; Irepol (pA), amplitude in pA of the outward current between ramp peak and its smallest value prior to the beginning of IPS; Irepol peak (mV), voltage in mV at which the outward current was smallest prior to INS beginning; IPS start (mV), voltage in mV at which IPS began; IPS peak (pA), current in pA at IPS peak, measured as difference between its beginning and its peak; IPS peak (mV), voltage in mV at which PS region peaked; IPS peak−Ih (pA), current in pA measured as difference between IPS peak and Ih; Vh (mV), holding potential in mV; Param, parameters measured; VM cells, data from ventricular myocardial cells; P cells, data from Purkinje cells; *statistically significant difference between the data at the two Vh values either in VM or P cells.
In Table 9, in P cells, the same procedure decreased IK1 peak (*), INS (*), and INa1 (*). ICa was not measurable (only small indentations) either in control or at less negative Vh. “Ito” and Iramp peak–Ih increased but not significantly. Furthermore, IPS was smaller than in VM cells by 83.1% with Vh −80 mV and was not present with Vh −50 mV.
In Table 10, with Vh −80 mV during the step to ∼+20 mV ICa was larger in VM cells (328%*) and did not decrease with gradually lower Vh in either VM cells (−24.8%, −6.5%, +17.5% and −29.6%, respectively) nor in P cells. However, in the latter tissue the ICa values varied irregularly, possibly due to the far fewer of P cells displaying it. Instead, as reported in Table 2, during depolarizing steps from Vh −70, −60, −50, and −40 mV, in VM cells INa1 decreased by −3.4%, −30.3%, −75.6%, and −99.3%, and in P cells by −3.7%, −20.1%, −65.9%, −93.5%, respectively.
Table 10.
ICa during depolarizing steps from different Vh
| Vh (mV) | Param | VM cells | P cells |
|---|---|---|---|
| −80 | ICa peak (mV) | +19.4 ± 0.5 | +18.8 ± 1.2 |
| ICa (pA) | −415 ± 101 (18/18) | −96.8 ± 44.6* (6/18) | |
| τf (msec) | 17.9 ± 7.9 | 11.4 ± 6.3 | |
| τs (msec) | 107.8 ± 15.1 | 157 ± 61.2* | |
| −70 | ICa peak (mV) | 19.4 ± 0.6 | 19.4 ± 0.6 |
| ICa (pA) | −312 ± 71 (15/16) | −18.9 ± 10.7* (3/16) | |
| −60 | ICa peak (mV) | 19.3 ± 0.7 | 20.0 ± 0.0 |
| ICa (pA) | −388 ± 73 (15/15) | −8.8 ± 6.1* (2/15) | |
| −50 | ICa peak (mV) | 20.0 ± 0.9 | 16.9 ± 1.5 |
| ICa (pA) | −488 ± 76 (16/16) | −9.1 ± 34.7* (5/16) | |
| −40 | ICa peak (mV) | 20.0 ± 0.0 | 18.3 ± 1.6 |
| ICa (pA) | −292 ± 67 (10/10) | −87.3 ± 41.3* (3/6) |
ICa peak (mV), voltage in mV at which the ICa was largest; ICa (pA), amplitude of ICa in pA measured from its peak to the end of the step; Vh (mV), holding potential in mV; Param, parameters measured; VM cells, data from ventricular myocardial cells; P cells, data from Purkinje cells; τf (msec) and τs (msec), fast and slow time constants, respectively, of ICa inactivation; Numbers in parenthesis (e.g., 18/18), number of cells in which ICa was present over the total number of cells studied; *statistically significant difference between P and VM cells data.
Thus, with less negative Vh, INa1 markedly decreased in both tissues during both ramps and depolarizing steps. In VM cells, INS and IPS regions persisted even at Vh −50 mV. Instead, in P cells with Vh −80 mV, IPS was much smaller than INS and, with Vh −50 mV, INS was markedly diminished and IPS abolished. Furthermore, in VM cells ICa was consistently present with Vh −80 mV and did not decrease with Vh −50 mV. In P cells, ICa was either not apparent or it was a small inward indentation on the outward current.
The transient outward current Ito in Purkinje and myocardial cells
Some of differences between currents in P and VM cells in the −20 to +40 mV range (see Fig. 2) were analyzed by applying the depolarizing steps from Vh −80 and Vh −40 mV in the absence and presence of Ito blocker 4-aminopyridine.
In Figure 9A, in P cell, during the step from Vh −80 to −20 mV, INa2 slowly decayed (arrow). A small outward peak appeared at the beginning of the step to 0 mV and progressively increased in size with more positive steps. In Figure 9B, in VM cell from a different heart, no decaying inward INa2 was apparent and, instead, a smaller outward current grew in amplitude over the range −20 to +40 mV. The sustained current at the end of the steps was smaller than in P cell (note the different current calibration).
Figure 9.
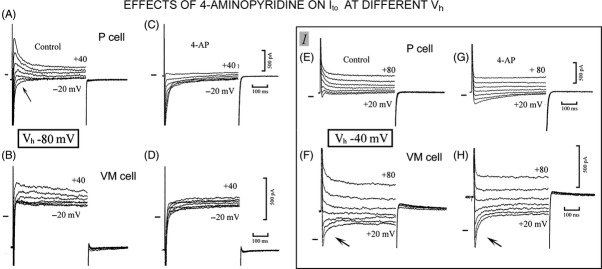
Ito in P and VM cells. In A–D, steps were applied from Vh −80 to the −20/+40 mV range. In P cell, the arrow points to slowly decaying INa2 during the −20 mV step (A). The currents in a VM cell taken from a different heart are shown in B. The procedure was repeated in the presence of 4-AP in P (C) and VM cell (D). In inset 1, steps were applied from Vh −40 to +80 mV in P (E) and VM cell (F), where the arrow points to the current recorded at +20 mV. The procedure was repeated in the presence of 4-AP in P (G) and VM cell (H), where the arrow points the current recorded at +20 mV. VM cells from the same heart as in Fig. 2.
In the presence of 4-AP in the P cell (Fig. 9C) the peak Ito was abolished, the decaying INa2 was larger at negative values, and the sustained current was reduced. In the VM cell (Fig. 9D), 4-AP abolished the peak Ito and reduced the sustained current. An inward transient was present at −20 mV which increased at +20 mV. Thus, at −20 mV in P cells the inward current was much larger, inactivated more slowly than the current in VM cell, and decreased markedly at + 20 mV.
In Figure 9 inset 1, Vh was decreased to −40 mV in order to inactivate INa2 (Vassalle et al. 2007; Bocchi and Vassalle 2008; present results) but not ICa (Isenberg and Klöckner 1982). At Vh −40 mV, Ito channel is partially inactivated, mid-inactivation voltage being ∼−35 mV (Dumaine and Cordeiro 2007). In the P cell (Fig. 9E), the inward transients were markedly reduced and peak Ito was decreased. In the VM cell (Fig. 9F), inward transients were present during the +20 (arrow), +30, and +40 mV steps and Ito was present during the +60, +70, and +80 mV steps.
In the presence of 4-AP, in the P cell (Fig. 9G), Ito was eliminated and the small slowly decaying inward component was largest at 0 mV. In the VM cell (Fig. 9H), in the presence of 4-AP, Ito peak was eliminated and the sustained current was smaller. ICa (arrow) was largest at +30 mV and reversed between +60 and +70 mV.
Thus, Ito patterns were distinctly different in that in the P cell Ito peak was larger and declined more by the end of the step. Also, Ito block by 4-AP unmasked inward currents with voltage range, magnitude, and speed of inactivation consistent with INa2 in P cell and with ICa in the VM cell. With Vh −40 mV, in the P cell INa2 was small and in the presence of 4-AP the current decreased slowly. In the VM cell, with Vh −40 mV, ICa was larger in the presence of 4-AP and inactivated quickly.
In Table 11, with Vh −80 mV and test steps to +40 mV, in P cells Ito when measured as the difference between its peak and sustained current at end of 500 msec steps was larger (*) as it was when measured as the difference between Ito peak and Ih (*). Ito decreased from its peak to the end of the step by −27.1% in VM and by −45.1% in P cells, the slow time constant of inactivation being smaller in VM cells (*).
Table 11.
Ito in VM and P cells during depolarizing steps from different Vh
| Vh to test step (mV) | Param | VM cells | P cells |
|---|---|---|---|
| −80 to +40 | Ito (pA) | 394 ± 136 (12/18) | 914 ± 168* (18/18) |
| τf (msec) | 8.8 ± 1.6 | 13.4 ± 2.2 | |
| τs (msec) | 59.4 ± 9.9 | 147.1 ± 34.9* | |
| Ito−Ih (pA) | 780 ± 189 | 1817 ± 248* | |
| −70 to +40 | Ito (pA) | 235 ± 84 (10/16) | 825 ± 157* (16/16) |
| Ito−Ih (pA) | 300 ± 82 | 1557 ± 239* | |
| −60 to +40 | Ito (pA) | 26.7 ± 19 (2/15) | 663 ± 164* (15/16) |
| Ito−Ih (pA) | 50.7 ± 34.8 | 1412 ± 249* | |
| −50 to +40 | Ito (pA) | 00 ± 0 (0/16) | 532 ± 88* (16/16) |
| Ito−Ih (pA) | 00 ± 0 | 1163 ± 177* | |
| −40 to +40 | Ito (pA) | 00 ± 0 (0/10) | 439 ± 111* (9/9) |
| Ito−Ih (pA) | 00 ± 0 | 948 ± 249* | |
| −40 to +80 | Ito (pA) | 182 ± 44 (10/10) | 1093 ± 220* (9/9) |
| Ito−Ih (pA) | 370 ± 52 | 2176 ± 327* |
Vh to test step (mV), depolarizing steps from holding potential to voltage indicated; Ito (pA), amplitude of Ito in pA as the difference between Ito peak and the end of 500 msec steps; Ito−Ih (pA), amplitude of the current measured as the difference between Ito peak and Ih; Param, parameters measured; VM cells, data from ventricular myocardial cells; P cells, data from Purkinje cells; τf (msec) and τs (msec), fast and slow time constants, respectively, of Ito inactivation; Numbers in parenthesis (e.g., 18/18), number of cells in which Ito was present over the total number of cells studied; *statistically significant difference between P and VM cells data.
In VM cells, with Vh −70 and −60, Ito decreased by −40.3% and −93.2%, respectively. With Vh −50 and −40 mV there was no apparent Ito on depolarization to +40 mV. In P cells, with Vh −70, −60, −50, and −40 mV, at +40 mV Ito decreased by −9.7%, −27.4%, −41.7%, and −51.9%, respectively. With Vh −40 mV, at +80 mV (past the reversal potential of ICa) in VM cells Ito was smaller (*) than in P cells. The difference between peak Ito and Ih (Ito−Ih) was larger than Ito in both tissues and was much larger (*) in P cells at all Vh.
Thus, with respect to P cells, in VM cells Ito peak: (1) was much smaller; (2) decreased less to a smaller sustained current; (3) was reduced more or altogether eliminated by less negative Vh; and (4) could be made to reappear with steps from −40 to +80 mV, still being much smaller than in P cells.
Effect of tetrodotoxin on INS and of nickel on ICa component
In P cells, the sodium channel blocker tetrodotoxin (TTX) markedly reduces INS (Vassalle et al. 2007), including the fraction caused by INa3 (Rota and Vassalle 2003). As in VM cells, sodium currents do not seem to play a dominant role in INS, TTX would not be expected to suppress the NS region. Also, if ICa is responsible for the inward component positive to INS, the Ca2+ channel blocker Ni2+ should eliminated it.
In Figure 10A, control, in the VM cell during the 260 mV sec−1 ramp INa1 was superimposed on NS region and the ICa component was emphasized by shaded area. In Figure 10B, TTX (30 μmol L−1) eliminated INa1 (short downward arrow) and only INS was left. That indeed INS was not abolished by TTX is confirmed by the fact during the repolarizing ramp a distinct IPS was present as in control.
Figure 10.
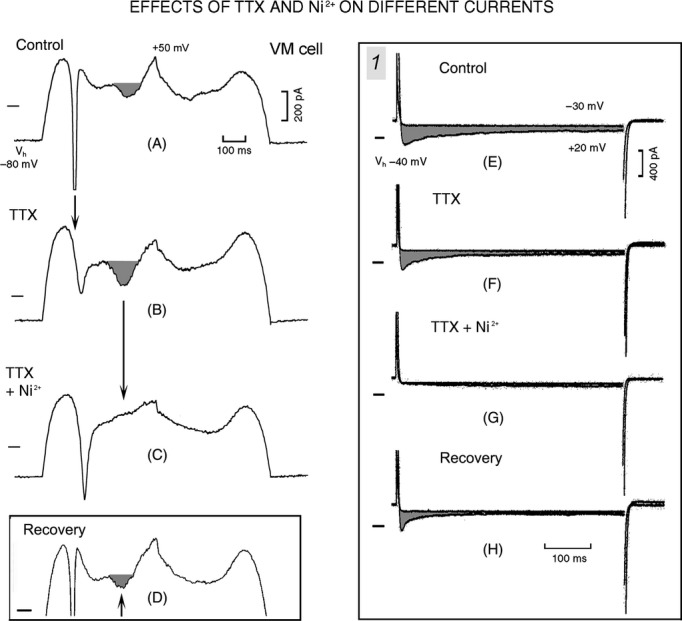
TTX does not eliminate INS and Ni2+ abolishes ICa component. In A, during the 260 mV sec−1 ramp, INa1 was superimposed on INS which was followed by the ICa component (shaded area). TTX (30 μmol L−1, B) eliminated INa1 (short arrow) but not INS as confirmed by the presence of IPS. Ni2+ (2 mmol L−1, C) abolished the ICa component (long arrow). Recovery is shown in D. In inset 1, depolarizing steps from Vh −40 mV to +20 mV elicited ICa (control, E) which was not suppressed by TTX (TTX, F) but was eliminated by Ni2+ (TTX + Ni2+, G). During the recovery from TTX and Ni2+, ICa returned to control value (recovery, H).
In the presence of TTX, ICa (shaded area) was greater than in control. A possible reason for this increase might be that TTX decreases intracellular Na+ activity (Abete and Vassalle 1988; Iacono and Vassalle1990), thereby increasing the transmembrane Na+ gradient. In turn, a larger Na+ gradient increases the extrusion of Ca2+ through an enhanced Na+-Ca2+ exchange: this would increase the transmembrane Ca2+ gradient and therefore ICa.
In Figure 10C, Ni2+ (2 mmol L−1) altogether abolished ICa, as emphasized by the long downward arrow. Although 2 mmol L−1 Ni2+ blocks also the Na+-Ca2+ exchange, such a block seems unlikely to account for the observed phenomenon, as in the voltage range of ICa, the Na+-Ca2+ exchange would be operating in the reverse mode generating an outward current.
During the recovery in physiological saline solution (Fig. 10D), INa1 and ICa reappeared.
In another approach (Fig. 10, inset 1), depolarizing steps were applied from a Vh of −40 mV so that Na+ channels (but not ICa channel) were inactivated. In Figure 10E, control, a step from Vh −40 mV to −30 mV did not elicit time-dependent currents, whereas depolarization to +20 mV elicited ICa. In Figure 10F, TTX (30 μmol L−1) did not suppress ICa at +20 mV. In Figure 10G, adding Ni2+ (2 mmol L−1) to TTX solution eliminated ICa and allowed a small decaying current to appear (presumably an unmasked small Ito). In Figure 10H, during the recovery from TTX and Ni2+, ICa returned to the control value. Similar results were obtained in n = 2.
Effects of 4-AP and Ba2+ on fast and slow ramps in myocardial cells
In Purkinje cells, Cs+ and Ba2+ (blockers of IK1) markedly decreased IK1 as well as the slope conductance, but only slightly reduced the outward current at positive potentials (Du and Vassalle 1999). Furthermore, Ba2+ eliminated IK1 peak but not INS, whereas low Vh and TTX eliminated INS (Rota and Vassalle 2003; Vassalle et al. 2007). As neither a low Vh nor TTX abolished INS in VM cells (present results), in VM cells INS might be mostly related to Mg2+ block of IK1. To verify this point, in one experiment fast and slow ramps were applied to test whether Ba2+ (by blocking IK1 and therefore preventing the block and unblock by Mg2+ in the NS and PS regions, respectively) abolished INS and IPS.
In Figure 11A, in a VM cell, during a 260 mV sec−1 ramp (bottom trace), in control the current exhibited the usual features. In the presence of 4-AP, INS was little changed whereas ICa component became larger (+129%, rhombus). While overall the current was less outward, IK1 and IPS peaks were somewhat larger. The current between ICa and ramp peaks was unchanged (+1.6%) as in P cells (Du and Vassalle 1999). In the presence of Ba2+, not only the IK1 peak, but also INS and IPS were abolished (as pointed out by double headed arrows). Neither the outward current between ICa and ramp peaks nor the initial fast decrease in current at beginning of repolarization was suppressed. INS and IPS reappeared during recovery (bottom current trace).
Figure 11.
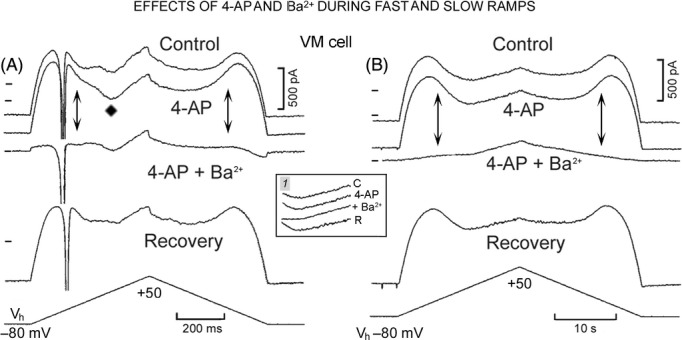
NS and PS regions are abolished by Ba2+. In A, 260 mV sec−1 ramps were applied in control, in the presence of 4-AP, of 4-AP plus Ba2+, and during recovery. In B, the same procedures were applied during 6.5 mV sec−1 ramp. The larger ICa component in the presence of 4-AP is labeled with a rhombus. In the two panels, the left hand double headed arrows emphasize the disappearance of INS and the right hand double headed arrows that of IPS. In inset 1, the current traces recorded between ICa and ramp peaks in B have been superimposed: C, control; 4-AP, in the presence of 4-aminopyridine; +Ba2+, in the presence of 4−AP and Ba2+; R, recovery in physiological saline solution.
In Figure 11B, during a 6.5 mV sec−1 ramp (bottom trace), in the presence of 4-AP, IK1 peak was also somewhat larger and so was ICa. Instead, the current between ICa and ramp peaks was not affected (−0.6%, see 4-AP trace in Figure 11 inset 1). Ba2+ abolished IK1 peak and INS, as pointed out by the left hand double headed arrow, leaving only the slowly increasing outward current. As in P cells (Du and Vassalle 1999), the current during the last part of the depolarizing ramp was unaffected (see Ba2+ trace in Figure 11 inset 1). During the repolarizing ramp, the current decreased continuously (no IPS, as indicated by the right hand double headed arrow). The recovery from the procedures is shown by the bottom current trace and by trace R in Figure 11 inset 1).
Thus, while in P cells Ba2+ abolished IK1 peak, but not INS (Rota and Vassalle 2003), in VM cells it abolished IK1 peak, INS and IPS. However, neither 4-AP nor Ba2+ abolished the increase in outward current between ICa and ramp peaks.
Discussion
The present results show numerous dissimilarities between VM and P cell currents. The features involved concern: (1) the slope conductance at the resting potential and its changes during depolarization and repolarization; (2) the sodium currents INa1, INa2, and INa3 (presence or absence, amplitude, threshold, voltage and time dependence, contributions to peak sodium current); (3) outward and inward current during ramps with different slopes; (4) time and voltage dependence of IK1 blocking and unblocking; (5) NS and PS regions (voltage range, voltage and time dependence, slope conductance, depolarization vs. repolarization); (6) prevalent role of slowly inactivating sodium currents in the mechanisms underlying NS region in P cells; (7) prevalence of block and unblock of IK1 in NS and PS regions, respectively, in VM cells; (8) characteristics of ICa as well as Ito and sustained current; (9) different contributions of IK1 and Ito to the I-V relation; (10) voltage and time dependence of currents during repolarizing ramps, and (11) response to channels blockers.
We conclude that the differences between VM and P cells involved all the ionic currents studied and account for several electrophysiological differences in resting and action potentials.
Membrane conductance as a function of voltage and time in VM and P cells
As at negative potentials IK1 predominates in determining the I-V relation (Shah et al. 1987), at potentials negative to IK1 peak the I-V relation was taken to essentially reflect that of IK1. Near the resting potential, the larger slope conductance in VM cells (+100%) is consistent with the findings that IK1 in rabbit myocardial ventricular cells is larger than in P cells (Cordeiro et al. 1998). Indeed, the expression of transcripts underlying IK1 channel (Kir2.1, Kir2.2, Kir2.3) in human Purkinje fibers is about half that in ventricular myocardium (Gaborit et al. 2007).
The larger slope conductance in VM cells at resting potential could be related to the fact that IK1 channels are located also in T-tubules (e.g., Lopatin and Nichols 2001), which are absent in P cells (see Vassalle et al. 1995 for references). In VM cells, the larger IK1 peak could possibly be due to the larger resting conductance and/or to lower polyamines level in myocardial cells.
The smaller conductance in P cells would seem at odds with the fact that in 2.7 mmol L−1 [K+]o their maximum diastolic potential (∼−95 mV; e.g., Vassalle 1965) is more negative than the resting potential in VM cells (∼−80 mV). This discrepancy is due to the presence of the pacemaker current IKdd, (the potassium current underlying diastolic depolarization; Vassalle 1966; Vassalle et al. 1995; Vassalle 2007, 2013), which is present in P cells, but not in VM cells.
Thus, in quiescent P cells in 5.4 mmol L−1 [K+]o the resting potential is ∼−80 mV and does not increase when [K+]o is lowered to 2.7 mmol L−1 (Vassalle 1965, 1966) due to K+-dependent fall in K1 channel conductance. However, in cells active in 2.7 mmol L−1 [K+]o, the activation of IKdd during the AP (Vassalle 1966; Vassalle et al. 1995) is responsible for the voltage undershoot to the maximum diastolic potential. As IKdd deactivates as a function of time in the diastolic potential range, the pacemaker potential and slope conductance decrease toward the resting potential value (Weidmann 1951; Vassalle 1965, 1966). Therefore, in P cells diastolic conductance at resting potential is mostly a function of IK1 (as in VM cells) and at the maximum diastolic potential of both IK1 and IKdd.
During ramp depolarization, the progressive decline of slope conductance to a minimum at IK1 peak shows that the gradually smaller increase in outward current is due to IK1 inward rectification, rather than a progressive increase of an inward current. At IK1 peak, IK1 is matched by the inward current, as the outward current stopped increasing and the slope conductance became minimal (Fig. 6). Soon after, in both tissues, the activation of INa3 (together with the decreasing IK1) accounts for the initiation of the NS region.
During INS, the reversal and reincrease of pulse current indicate that IK1 became smaller than the inward current. In fact, IK1 reaches its minimum at the peak of INS (−20 mV in P cells) as shown by IK1 I-V relation (control minus IK1 blockers) (Shah et al. 1987; Cordeiro et al. 1998). In P cells, the reversal of pulse current occurred both during the part of INS due to INa3 (Rota and Vassalle 2003) and that due to the inactivation of INa2 (Bocchi and Vassalle 2008). The relation of the reversed pulse current to the activation and decay of INa2 is demonstrated by the findings that during depolarizing steps the amplitude of pulse current became larger when INa2 appeared, decreased gradually during INa2 slow inactivation, and was markedly reduced by lidocaine (Bocchi and Vassalle 2008).
However, as pulse current reversal in NS range occurred also during the 6.5 mV sec−1 ramp (Fig. 6E) when the Na+ currents are inactivated (and in VM cells the inactivating INa2 would play little role), the reversal appears to involve also the block of IK1 channel. In both cases (increase in Na+ currents and decrease in IK1) the net current would become inward (INS) and therefore the pulse current would reverse. The role of IK1 change in pulse current reversal is supported by the occurrence of the reversal also during IPS (Fig. 6D and E), when any possible residual Na+ currents would be inactivated.
Contribution of INa3, INa2, and rectification of IK1 to NS region in P and VM cells
Our findings show that the mechanisms underlying INS in P cells differ qualitatively and quantitatively from those in VM cells and account for some previously reported results.
In the presence of TTX and of Ca2+ blocker nicardipine in rabbit, the NS region was present in VM cells but not in P cells (Cordeiro et al. 1998). This difference is accounted by the finding that INa3 (Rota and Vassalle 2003) and INa2 (Vassalle et al. 2007; Bocchi and Vassalle 2008) are blocked by TTX. However, when measured as the difference current (control minus Ba2+), a small NS region was present also in P cells (Cordeiro et al. 1998), suggesting that, in the absence of Na+ and Ca2+ currents, the residual INS was due to a block of IK1.
Similarly, in P cells with steps from a Vh of −50 mV, the NS region was not always found, but it was present in IK1 I-V relation (control minus current in Ba2+ or Cs+) (Shah et al. 1987). In retrospect, at Vh −50 mV the Na+ currents would have been inactivated or markedly reduced (Rota and Vassalle 2003; Vassalle et al. 2007; present results). Therefore, the findings of Shah et al. (1987) can also be accounted for by the inactivation of Na+ currents at −50 mV and a small contribution of IK1 block to INS in P cells. That in P cells, IK1 rectification contributes to INS is also indicated by a net decrease of radioactive K+ efflux in the −60 to −40 mV range (e.g., Vereecke et al. 1980).
In our experiments, in the absence of blockers, with respect to VM cells, in P cells the increase in INS with the steeper ramps and its decrease or absence with 6.5 mV sec−1 ramp are consistent with time-dependent inactivation of INa3 and INa2. In P cells, with Vh −80 mV, the smaller INS in I-V relation of the sustained current is consistent with inactivated INa3 and substantially reduced INa2 by the end of 500 msec steps. In addition, the gradual decrease of INS with less negative Vh in P but not in VM cells (Fig. 4, inset 1) indicates a voltage-dependent inactivation of Na+ currents.
Conversely, with 6.5 mV sec−1 ramps the much smaller INS in P cells is explained by INa3 and INa2 being reduced or absent, in agreement with the little effects of TTX on INS and by the similar values of INS and IPS. Furthermore, with Vh −50 mV, during fast ramps the near abolition of INS in P cells but not in VM cells also points to a greater role of Na+ currents.
In P cells, TTX, lidocaine, and low Vh markedly reduced INa3 (Rota and Vassalle 2003) as well as INa2 (Vassalle et al. 2007) and so did low [Na+]o (Bocchi and Vassalle 2008). Yet, INS persisted in VM cells with some of these procedures (present results). Furthermore, in P cells, Ba2+ abolished IK1 peak but not INS (Rota and Vassalle 2003) whereas it abolished IK1 peak, INS, and IPS in VM cells.
These results indicate a predominant role of Na+ currents in P cell INS and of IK1 inward rectification in VM cell INS. Indeed, in guinea pig ventricular myocytes, the K+ channel opener cromakalim abolished INS and markedly shortened the action potential (Liu et al. 1990).
As for voltage ranges of Na+ currents and of INS, the beginning of INS during the ramps (∼−58 mV) indicates the participation of INa3 as in P cells this current started at ∼−58 mV (Table 8; Rota and Vassalle 2003) and peaked before or by the end of ramps to −42 mV (Rota and Vassalle 2003). In both P and VM cells, INa3 contributed to INS prior to INa1, but (its slow inactivation being cut off by INa1) presumably little after INa1.
In P cells, the slowly inactivating INa2 can contribute to INS after the inactivation of INa1, as INa2 has a −40 mV threshold and is largest at a voltage (−30/−20 mV, Vassalle et al. 2007) which is near to the peak of INS (−26.5 mV with 260 mV sec−1 ramp). INa2 activates also in the absence of INa1 (Fig. 5B and E), in agreement with the findings of Bocchi and Vassalle (2008). Indeed, in those P cells in which INa1 was not present with 260 mV sec−1 ramps, INS peaked at a potential (−29.2 mV, present results) near the INa2 peak.
Therefore, with respect to VM cells, in P cells INS: (1) had a smaller voltage range; (2) was larger with faster ramps; and (3) was smaller with slow ramps, with lower Vh and in the presence of TTX. These finding indicate a predominant role of the sodium currents in INS of P cells and of IK1 block in VM cells.
Dual mechanism of IK1 inward rectification and the NS and PS regions in P and VM cells
As for the inward rectification of IK1 channel, two mechanisms have been demonstrated: block by intracellular polyamines (channel gating by spermine and spermidine; Lopatin et al. 1994; see Lopatin and Nichols 2001) and block by Mg2+ (Matsuda et al. 1987; Vandenberg 1987). The IK1 block by spermine and spermidine is time dependent (Ishihara 1997), whereas the block by Mg2+ and putrescine is voltage dependent and virtually instantaneous (Ishihara and Ehara 1998).
In guinea pig VM cells, in the absence of internal Mg2+, the block by polyamines occurs between −80 and ∼−40 mV whereas, with in the presence of internal Mg2+, IK1 block is present also between −40 and 0 mV (Ishihara 1997). Furthermore, after a depolarization larger than 0 mV (which would cause Mg2+ block), on repolarization to −50 mV there was a sudden transient increase in outward current (see also Shimoni et al. 1992). The amplitude of the outward current was correlated to the degree of Mg2+ block during the previous depolarization, indicating that the increase in outward current was due to the removal of Mg2+ block. During a repolarizing ramp, the outward current at −50 mV was substantially larger in the presence than in the absence of internal Mg2+, indicating the importance of the removal of Mg2+ block of IK1 for the repolarization of the action potential (Ishihara 1997; Ishihara and Ehara 1998).
Because IK1 block by polyamines begins at more negative voltage than that by Mg2+ (Ishihara et al. 1989; Ishihara 1997; Ishihara and Ehara 1998), the block of IK1 ought to be solely due to polyamines up to IK1 peak which with the slowest ramp occurred at −44.2 mV in VM cells (Table 5). Instead, the voltage range of the IK1 block by Mg2+ (−40 to 0 mV) overlaps the NS region in VM cells.
Time dependence of IK1 block by polyamines at the beginning of depolarizing steps would account for the initial decline of the outward current. On step repolarization from positive potentials toward EK the sudden increase in outward current due to instantaneous relief of Mg2+ block gradually declined due to time-dependent block by spermine and spermidine (Ishihara 1997). The time constant of the exponential decay of the outward current was ∼5 msec at −50 mV which is close to the τ of ∼6 msec for the current decline on depolarization from Vh −80 to ∼−60 mV (Table 4).
As for the symmetrical voltage ranges of NS and PS regions, if the voltage-dependent block of IK1 by Mg2+ initiates near the beginning of INS, its complete removal at the peak of IPS (ceteris paribus) should occur at a similar potential. Indeed, in VM cells, with the slowest ramp, INS initiated at −44.2 mV and IPS peaked at −44.4 mV and their amplitudes were similar (218 and 197 nA, respectively). The peak of INS (full block) was at −3.9 mV and the beginning of IPS was 8.9 mV (initiation of block removal) (Table 5).
With faster ramps, other factors modified IK1 and IPS peaks. In P cells, the larger INS with faster ramps implicates additional time-dependent factors such as a greater activation of INa3 and of INa2. In keeping with this conclusion, in P cells with 6.5 mV sec−1 ramp, INS was much smaller and so was IPS, as the Na+ currents would be inactivated during the depolarizing and repolarizing ramp.
That IK1 undergoes a progressively greater inward rectification on depolarization from the Vh is also shown by the progressively smaller increase in sustained outward current at the end of 500 msec depolarizing steps (when most of the other currents would be completely or partially inactivated). Actual decline of the sustained current at potentials positive to −50 mV in P and to −40 mV on VM cells reflects the INS seen during the ramps. The less negative potential at which the sustained current decreased with respect to the beginning of INS during the ramps might be ascribed to lack of INa3 contribution to the sustained current.
At more negative potentials, block and unblock of IK1 might contribute to several changes in the I-V relation during ramps of different steepness. Thus, voltage- and time-dependent block by spermine and spermidine on depolarization from Vh −80 mV to IK1 peak would account for the gradually smaller increase of outward current during a ramp and for the decreasing slope conductance both in P (Rota and Vassalle 2003; Bocchi and Vassalle 2008; present results) and in VM cells (Fig. 6).
As at less negative potentials the channel block by polyamines is much slower (Ishihara et al. 1989; Ishihara and Ehara 1998), such a block may also contribute to the current changes that occur during the ramps with steeper slopes. A greater lag between faster voltage change and block of IK1 channel might be responsible for the increase in magnitude of IK1 peak with faster ramps. Also, the larger decrease in outward current during faster repolarizations might include a delay in the removal of IK1 block by polyamines.
The fact that during repolarizing 260 mV sec−1 ramps, in P cells IPS was much smaller (−81%) than INS (Table 6) suggests that INa3 and INa2 have a major role in INS and that the remainder (∼19%) is due to IK1 block by Mg2+. The removal of IK1 block would account for the IPS beginning being more outward (no Na+ currents contribution) than the INS peak and IPS peak being similar to INS beginning. In contrast, in VM cells the similarity of INS and IPS (Table 6) suggests that block and unblock of IK1 channel by Mg2+ were the major mechanisms underlying INS and IPS, respectively.
This is consistent with Mg2+ block and unblock being only voltage dependent and with the slowly inactivating INa2 not contributing to either INS or IPS in VM cells. During the slowest ramps, there would be little or no contribution by Na+ currents to either INS or IPS, as supported by the similarity of INS and IPS in VM as well as in P cells (Table 5).
In VM cells, with 260 mV sec−1 ramp the larger difference between IPS peak and Ih (Table 6) reflected the almost double IK1 upon which IPS was superimposed. In contrast, the similarity of the IPS peak voltage in VM and in P cells is consistent with the voltage dependence of the removal of IK1 block by Mg2+. With the 6.5 mV sec−1 ramp, the much larger INS and IPS in VM than in P cells support the role of Mg2+ block and unblock, respectively, as in both tissues the Na+ currents would be largely inactivated during both depolarizing and repolarizing ramps.
Therefore, the time- and voltage-dependent block of IK1 by polyamines appears to prevail at potentials negative to the IK1 peak whereas the inward rectification of IK1 during INS is attributable to Mg2+ block. This conclusion is supported by the findings that in P cells: (1) when INa3 and INa2 were present, INS was larger than in VM cells and more so the faster the ramp; (2) when INa3 and INa2 were inactivated (repolarizing ramps) IPS was smaller in P cells; and (3) with the slowest ramp (INa3 and INa2 being inactivated during the depolarizing and repolarizing phases), both INS and IPS were smaller in P cells.
INa3 in Purkinje and myocardial cells
With the approach of Rota and Vassalle (2003), in P cells INa3 was mostly studied at potentials negative to INa1 threshold. With depolarizing ramps (in the absence of INa1), INS began at −57.7 mV and peaked before or by the end of the ramp at −42 mV. INS was attributed to the activation of INa3, as shown by its threshold, the marked reduction by TTX and lidocaine and its little sensitivity of Cs+ and Ba2+. In contrast to INa3, TTX or lidocaine did not abolish ICaT (Tytgat et al. 1990). In addition, INa3 was markedly reduced by 70 mmol L−1 [Na+]o and it was not abolished by 100 μmol L−1 Ni2+ (M. Rota and M. Vassalle, unpubl. experiments). These findings contribute to rule out that INa3 might in actuality be ICaT.
Suitably large and slow ramps that did not activate INa1 initiated INa3 at about −60 mV followed at about −40 mV by the activation of INa2 (Rota and Vassalle 2003). In retrospect, these results in P cells separated for the first time the contributions of INa3 and INa2 to INS. That INa3 is a sodium current also in VM cells is shown by its decrease with less negative Vh (depolarizing steps, Table 1) or ramps (Table 9).
A population of slowly inactivating Na+ channels has been also reported in giant squid action. These channels are much fewer than the normal Na+ channels, activate on depolarization to ∼−65 mV whereas INa threshold is −50 mV, activate maximally at −40 mV and undergo a very slow inactivation (Gilly and Armstrong 1984), similar to INa3. As for the mechanism by which INa1 block INa3 slow inactivation, it appears that fast INa1 inactivation blocks slow inactivation of Na+ channels by charge immobilization (Richmond et al. 1998).
As to Na+ channel isoforms involved in INa3, among several Na+ channels isoforms cloned (NaV1 to NaV9; cardiac, neuronal, and skeletal), cardiac NaV1.5 isoform has a low TTX-sensitivity, whereas the neuronal (NaV1.1, NaV1.2, NaV1.3, NaV1.6) and skeletal muscle (NaV1.4) isoforms have a high TTX-sensitivity (Zimmer et al. 2002; Haufe et al. 2005a). The neuronal Nav1.2, Nav1.3, and Nav1.6 isoforms are expressed in VM cells and Nav1.1 and Nav1.2 (Haufe et al. 2005a) as well as skeletal NaV1.4 isoform (Qu et al. 2007) have been identified in P cells. Although INa3 and INa2 are more sensitive to TTX block than INa1, the noncardiac channels isoform involved is not known, as the skeletal muscle Na+ channel isoform Nav1.4 expressed in P cells is also blocked by low concentrations of TTX (Qu et al. 2007).
The neuronal TTX-sensitive Na+ channels were found to contribute to peak sodium current by 22% in P cells and by 10% in VM cells (Haufe et al. 2005b) and therefore to the sodium current responsible for the AP upstroke. Our results show that at the INa1 threshold, INa3 was a sizeable fraction of the total INa current. However, because INa1 was truncated by the saturation of the amplifier consistently in P cells and often in VM, the INa3 contribution to the total INa is bound to be overestimated. In addition (as shown by means of double steps), the fast inward component of INa2 would be expected to contribute to the total Na+ influx during the upstroke.
INa3 appears to be the link between DD and upstroke of AP in P cells by being responsible (Rota and Vassalle 2003) for the depolarizing phase of ThVos (the oscillatory potentials near the threshold for the upstroke; Vassalle 1965; Spiegler and Vassalle 1995; Berg and Vassalle 2000). Successive ThVos increase progressively in size during diastole until the depolarizing phase becomes large enough to attain the threshold for INa1 (Vassalle 1965; Spiegler and Vassalle 1995; Berg and Vassalle 2000). In P fibers, TTX suppressed the spontaneous discharge by abolishing the upward swing of the late diastolic depolarization, although the fibers could still be electrically driven due to smaller sensitivity of INa1 to TTX (see Fig. 3 by Vassalle and Scidà shown in Vassalle 1980). ThVos are present also in sino-atrial node (Kim et al. 1997) and are essential for both the initiation and maintenance of spontaneous activity of cardiac pacemakers (Vassalle 2007, 2013).
In VM cells, the voltage gap between the resting potential and the threshold for INa3 is closed by the depolarization induced by the conducted AP, as of necessity INa3 threshold is less negative than the resting potential. INa3 would then speed up the attainment of INa1 threshold and the induction of the upstroke.
While a 5 mmol L−1 [Na+]o has been successfully used to compare Na+ currents in normal and diseased VM cells (Pu and Boyden 1997; Valdivia et al. 2005), the low [Na+]o solution markedly reduced INa1. This apparently makes it unsuited to study the much smaller INa3, as no slowly inactivating INa3 was apparent in those studies. In our experiments, INa3 was demonstrated under physiological conditions at potentials negative to INa1 threshold. Also, less negative Vh markedly reduced INa1 (Table 2) leaving a separately identifiable slowly inactivating INa3 (Table 1).
Thus, the present results show that INa3: (1) was also present in VM cells; (2) had threshold that was negative to that of INa1 and was less negative than that in P cells; (3) inactivated slowly; (4) could occur at potentials less negative than INa1 threshold (as shown by preventing INa1 activation); (5) was less inactivated at a lower Vh than INa1; (6) increased in amplitude during depolarizing steps past ∼−60 mV due to its voltage-dependent activation and during faster depolarizing ramps (greater Na+ channel availability); (7) contributed to magnitude of total Na+ influx associated with INa1; (8) was absent during repolarizing ramps; (9) was associated with a reversal of pulse currents and with an increase in slope conductance during INS; and (10) its slow inactivation was suppressed by INa1.
A limitation of our study is represented by the lack of measurements of membrane capacitance (Cm) in the two cell populations analyzed, posing concerns related to the comparison of ionic current amplitudes. In this regard, values of Cm of 121.9 ± 4.8 pF (Rota and Vassalle 2003) and of 125 ± 6 pF (Han et al. 2000) have been reported for canine P cells, whereas Cm values of 113 ± 6 pF (Han et al. 2000) and of 133.4 ± 6 pF (Pu and Boyden 1997) have been reported for canine VM cells. Thus, the average value of the above capacitances for the P cells is 123.4 pF and that for the VM cells is 123.2 pF.
While P cells have no T tubule system, which is present in VM cells, P cells are larger in size than VM cells. Thus, these two features (cell size and T tubule system) tend to offset each other in determining the surface area in the cell populations.
The fast sodium current INa1 in P and VM cells
The measurement of INa1 magnitude in P and VM cells was hindered by its being truncated by the saturation of the amplifier. However, the finding that with Vh −80 mV INa1 was consistently cut off at −10 nA in P cells but only in 13/18 in VM cells indicates that INa1 was smaller in VM than in P cells. Furthermore, with less negative Vh (Na+ channels being partially inactivated), INa1 was less often truncated and yet in VM cells INa1 was still smaller than in P cells. Also, the less frequent INa1 activation during the 260 mV sec−1 ramp in P cells suggests that during this slower ramp INa1 channels are more susceptible to time-dependent inactivation in P than in VM cells.
Several differences regarding INa1 in P and VM cells have been reported. Thus, total amplitude and maximal rate of rise of AP upstroke were much larger in Purkinje than in ventricular fibers (Baláti et al. 1998). Also, neuronal sodium channels contribute less to the peak sodium current in dog ventricular than in Purkinje fibers (Haufe et al. 2005b). A larger INa1 with a more negative threshold in P cells (present results) would contribute to the faster conduction of P cells with respect to VM cells. Both in nonspontaneous P and VM cells, conducted action potentials would depolarize the membrane to the threshold of INa3, which in turn would allow the attainment of INa1 threshold, as seen during the ramps.
The slowly inactivating INa2
In VM cells, the absence of a large slowly decaying component of INa2 comparable to that of P cells accounts for the fact that (see Introduction), in contrast to P cells, the AP duration of ventricular myocytes was very little affected by TTX, local anesthetics, veratridine, and high [Na+]o, suggesting that there is little or no slow decaying INa2 in VM cells under physiological conditions. INa2 is more sensitive to TTX than INa1 (Vassalle et al. 2007) and therefore is possibly due to the activation of neuronal or skeletal muscle isoforms. In P cells, the larger contribution of the noncardiac Na+ channels (Haufe et al. 2005b) might be due to both INa3 and INa2. In P cells, the fast activation of INa2 (Bocchi and Vassalle 2008) would contribute to the influx of Na+ during the upstroke and its slow inactivation contributes to the longer plateau.
A late INa was found in canine myocardial cells perfused in a K+-free and very low Cl− medium by applying a 2000-msec pulse to −130 mV (to remove steady-state inactivation) prior to depolarizing steps applied at intervals of 30 sec (Zygmunt et al. 2001). Our results show that INa2 is small or absent under physiological conditions in VM cells, in contrast to P cells.
However, the findings of Zygmunt et al. (2001) could suggest that under abnormal situations remodeling may shift the voltage range of the late INa activation to less negative values. In this regard, it is of interest that INaL was increased in cardiac failure and yet no differences were found in isoform NaV1.1, 1.3, 1.5 subunits and in the subunit b1 and b2, leading to the conclusion that INaL increase was not due to a subunit isoform switching or to an altered b subunit expression (Valdivia et al. 2005).
In normal human ventricular myocytes, there is little or no INa2 but in myocytes from patient with hypertrophic cardiomyopathy remodeling leads to the appearance of a late Na+ current. Ranolazine, a blocker of the late Na+ current, had negligible effect on the action potential duration of normal ventricular myocytes, but shortened the longer AP of the myopathic myocytes and reduced the related arrhythmias (Coppini et al. 2013). Therefore, under some pathological conditions remodeling-related INa2 may induce arrhythmias also in VM cells.
As for role of P cell slowly inactivating INa2 in cardiac arrhythmias, an increase in INa2 (but not of INa1) by neurotoxins anthopleurin or ATX II in Purkinje fibers led to the onset of Torsades de pointes. This electrophysiological mechanism appears responsible for congenital and acquired long QT syndromes (LQTS), as abnormally prolonged repolarizing phase of AP leads to early after-depolarizations (EADs) in Purkinje fibers. In vitro and in vivo, in Purkinje fibers the neurotoxins induced EADs were abolished by concentrations of mexiletene that had little effect on INa1 (El-Sherif and Turitto 2003). The present characterization of differences between P and VM cells add insights relevant to mechanisms underlying some LQTS syndromes, especially the drug-induced acquired syndromes.
The calcium current in P and VM cells
That ICa was larger in VM cells was shown by the appearance of a large inward component, which peaked in the +10 to +20 mV range and became more conspicuous after the block of Ito by 4-AP. Instead, in P cells, the smaller ICa appeared as an indentation on Ito trace or it was not altogether apparent, even in the presence of Ito block by 4-AP.
That the inward component peaking at positive values was due to ICa was shown by its persistence at low Vh and by the abolition by Ni2+ of both the inward component during the ramps and ICa during depolarizing steps. In VM cells, ICa-induced positive shift of the onset of the smaller outward current would contribute to the more positive plateau and the larger ICa to the greater force of contraction (Lin and Vassalle 1978) with respect to P cells.
As for the identity of Ca2+ currents, the L-type (ICaL, isoform Cav1.2 containing the pore forming α1C subunit) and T-type (ICaT, isoforms Cav3.1, Cav3.2, and Cav3.3) are expressed both in P and M cells, ICaT being more abundantly expressed in P cells and ICaL predominating in VM cells (see Dun and Boyden 2008). These differences suggest that calcium current recorded in VM cells is of the ICaL type, in accordance with fact that it was recorded with less negative Vh and at less negative voltage.
The transient outward current Ito in P and VM cells
With respect to VM cells, in P cells a larger Ito contributed to the much larger outward current between INS and ramp peaks, consistent with the shift of the plateau to more positive values by 4-AP (Kenyon and Gibbons 1979; Dumaine and Cordeiro 2007).
However, our results show that the total outward current included different components in P and VM cells, as in VM cells a smaller Ito was superimposed on a larger IK1. In VM cells, the smaller Ito would induce a smaller phase 1 repolarization and (together with the larger ICa) would contribute to the more positive plateau than in P cells (e.g., see Fig. 1 in Baláti et al. 1998).
The finding that 4-AP abolished the peak Ito, but only reduced the sustained current at the end of depolarizing steps and the outward current during latter part of depolarizing ramps agrees with the results shown in other reports (no elimination of sustained current by 4-AP; Kenyon and Gibbons 1979; Cordeiro et al. 1998; Han et al. 2000). Therefore, Ito may have a time-independent background component or it may not inactivate completely (Kenyon and Gibbons 1979).
Also, there might be the contribution of an unidentified outward current present at positive potentials which is not eliminated by 0 mmol L−1 [K+]o (Ishihara et al. 1989). Even IK1 block by Cs+ and by Ba2+ did not suppress the increase in outward current at potentials positive to −40 mV, as shown by figures in Shah et al. (1987) and reported by Du and Vassalle (1999). This is consistent with the finding that 42K efflux at potential positive to −30 mV was not blocked by Cs+ (Vereecke et al. 1980).
This could be due either to the voltage dependence of Cs+ induced block as it is for Ba2+ (Hirano and Hiraoka 1986) or to a Cs+ insensitive outward rectifier. In the experiments of Cordeiro et al. (1998), 0.1 mmol L−1 Ba2+ reduced the inward IK1, but did not decrease the large outward current at positive potentials. In P cells (Du and Vassalle 1999) and in VM cells, 2 mmol L−1 Ba2+ eliminated IK1 peak, but did not eliminate the outward current at positive potentials.
However, this per se does not rule out a contribution of IK1 to the outward current, as Ba2+ block of IK1 is removed on depolarization and slowly reestablished on repolarization (Hirano and Hiraoka 1986; Imoto et al. 1987; Valenzuela and Vassalle 1991). Therefore, in P cells after INS peak, the increase in outward current includes Ito (which is not blocked by Ba2+, e.g., Cordeiro et al. 1998; Du and Vassalle 1999) and a current which is not suppressed by 4-AP, Ba2+, Cs+, or 0 mmol L−1 [K+]o. The delayed rectifier IK could contribute to the outward current, but is blocked by Ba2+, albeit to a lesser extent at positive values (Osterrieder et al. 1982). In addition, in P cells IK is rather small (<20 pA, Cordeiro et al. 1998) or undetectable (Kenyon and Gibbons 1979). Indeed, in the presence of 4-AP the outward current did not increase with depolarizing steps from Vh −40 mV (Fig. 9).
The increase in peak outward current with steeper ramps in P cells could be due to a larger Ito and/or to a lag in IK1 block by polyamines which is much slower at positive potentials (Ishihara et al. 1989). In VM cells, with steeper ramps the failure of the ramp peak current to appreciably increase suggests little change in Ito whereas the increase in current between ramp peak and Ih indicates a role of IK1.
On repolarization, with respect to VM cells, in P cells the greater fall in current is bound to reflect the much greater Ito activated on depolarization, as 4-AP reduced both Ito and the decrease in current on repolarization. The finding that on repolarization the decrease in outward current reached a more negative peak in P cells (∼−16 mV) than in VM cells (∼+7 mV) suggests that on depolarization the less negative reincrease of the outward current in M cells does not entirely depend on being masked by a larger ICa.
In P cell, Ito induces the large phase 1 repolarization of AP, and keeps the plateau at more negative level (Kenyon and Gibbons 1979). In rabbit P cells, 4-AP slowed phase 1 repolarization and shifted the plateau to positive potentials, while having in VM cells a much smaller effect on phase 1 repolarization and no effect on the plateau (Cordeiro et al. 1998).
In P cells, the persistence of the bulge current during the slow ramps and its elimination by 4-AP (which only reduced the sustained current) suggest that in P cells the bulge current is related to the activation of Ito whereas the 4-AP resistant increase in outward current toward the ramp peak might reflect a voltage-dependent increase in the sustained current. Another difference is that in VM cells Ito markedly decrease with lower Vh whereas in P cells a smaller Ito persisted even with Vh −40 (Table 11), consistent with the persistence of the bulge current with Vh −50 mV (Du and Vassalle 1999).
The greater Ito in canine P cells has been also found rabbit P cells versus VM cells (Cordeiro et al. 1998). However, in P cells the larger Ito and the smaller IK1 contributions to the total outward current at the plateau may be important in different situations. For example, an increase rate of discharge (tachycardia) may modify Ito (due to its slow recovery; e.g., Cordeiro et al. 1998) more than IK1.
In agreement with the larger Ito in P cells, Kv3.4 is more abundant at both the mRNA and protein level in Purkinje fibers than in ventricular myocardium. As the Kv3.4 subunit carries a TEA-sensitive Ito outward current, Kv3.4 current may be responsible for the large TEA-sensitive component of Ito in canine Purkinje cells (see Schram et al. 2002 for review). Among the differences between Ito in P and in VM cells is the different sensitivity to various blockers (e.g., TEA) and a smaller time constant of inactivation in VM cells (Han et al. 2000).
Thus, with respect to the P cells, in VM cells Ito: (1) became apparent at more positive potentials during the ramps; (2) did not undergo an enhancement (the “bulge”) during depolarizing ramps; (3) was smaller as measured from Ito peak either to the end of the step or with respect to Ih; (4) was abolished by 4-AP with a reduced sustained current (as in P cells); (5) had a smaller time constant of inactivation (in agreement with the finding of Han et al. 2000); (6) was associated with smaller slope conductance; (7) during repolarizing ramps, was smaller and had a smaller voltage range; and (8) was inactivated by lower Vh.
General conclusions
The present results indicate substantial differences between P and VM cells that bear on the different functions of P (conduction and pacemaker activity) and VM cells (contraction). In both tissues, IK1 is important for a normal resting potential and its inward rectification is important in more than one way. In P cells, the smaller IK1 conductance at the resting potential is essential for pacemaker function in two respects: (1) it keeps the resting potential less negative than EK and therefore it allows the undershoot to the maximum diastolic potential by the pacemaker current IKdd; and (2) IK1 inward rectification by polyamines contributes to diastolic depolarization caused by time-dependent decay of IKdd and therefore to the attainment of threshold for INa3. INa3 initiates INS and the associated depolarization allows the attainment of INa1 threshold.
Especially in VM cells, IK1 inward rectification caused by Mg2+ block in the voltage range −40 to 0 mV facilitates the attainment of threshold for Na+ currents and contributes (see Shimoni et al. 1992) to maintain the plateau. The large and fast INa1 cuts short the no longer needed INa3 and (being large) rapidly depolarizes the membrane and causes the overshoot to positive potentials (fast and large depolarization for rapid conduction). The depolarization induced by INa1 allows the attainment of the thresholds for other currents in an orderly fashion. INa2 (by inactivating slowly in P cells) contributes to their long plateau, thereby preventing the reentry of excitation from myocardium. Once the thresholds for Ito and ICa have been reached, at the end of the upstroke INa1 is rapidly inactivated (thereby preventing a useless Na+ influx).
The fast activation of Ito eliminates the overshoot which is no longer needed for fast conduction. In the meanwhile, ICa initiates the events leading to contraction and its inactivation balances the inactivating Ito at the plateau over a background of IK1 inward rectification. In VM cells, ICa shifted the outward current reincrease by 35 mV and therefore would contribute to the more positive plateau. In P cells, the inwardly rectifying IKdd becomes activated and whatever slowly decaying INa2 is not inactivated by the end of the plateau becomes deactivated as a function of voltage (Bocchi and Vassalle 2008). During the repolarization, the removal of Mg2+ block of the K1 channels leads to IPS and speeds up phase 3 repolarization, which, in turn, removes the polyamines block. In P cells, IKdd begins to increase at about IPS peak (∼−50 mV, Vassalle et al. 1995) leading to undershoot of resting potential, followed diastolic depolarization.
Among the differences between the two tissues, in P cells the more negative thresholds for the Na+ currents, the larger INa1 and the larger overshoot contribute to earlier activation and faster conduction. The larger Ito contributes to less negative plateau and the slowly inactivating INa2 to the longer AP. The larger sodium influx may involve a higher Na+-K+ pump activity, which is the major mechanisms underlying overdrive suppression (Vassalle 1977). Thus, the outward current created by Na+-K+ pump activity maintains the diastolic depolarization negative to INa3 threshold, so that P cell spontaneous discharge is suppressed when not needed during sino-atrial rhythm.
In VM cells, the larger resting conductance contributes to set the resting potential near the K+ equilibrium potential, as a resting potential lower than EK is needed only for pacemaker activity (see Vassalle 2007, 2013). In VM cells, the smaller INa1 is consistent with the smaller rate of rise of the smaller upstroke (e.g., Baláti et al. 1998), as the conduction path is shorter (from Purkinje network to epicardium). In VM cells, the larger ICa contributes to stronger contraction (Lin and Vassalle 1978); the smaller Ito together with the positive range of the larger ICa contributes to the more positive plateau. The lack of slowly inactivating INa2 contributes to a shorter AP (which in turn regulates the duration of twitch and of diastole) and the greater IK1 and removal of its block during the larger IPS contributes to an earlier and faster phase 3 repolarization.
The definition of these differences is a precondition also for the understanding of deranged function under pathological conditions.
Author Contributions
The experiments were carried out in Dr. Vassalle's lab in the Department of Physiology at SUNY, Downstate Medical Center, Brooklyn, NY. Dr. Vassalle conceived and designed the experiments, participated in part of the experiments, analyzed the data, drew the conclusions and wrote the manuscript. Dr. Bocchi participated in the collection, analysis, and interpretation of the data, their statistical evaluation, and supported various aspects of the writing of the manuscript. All authors approved the final version of the manuscript and qualify for authorship, and all those who qualify for authorship are listed.
Acknowledgments
This work was supported in part by a grant from the National Institutes of Health (HL56092). We thank Dr. Marcello Rota for reading the manuscript and offering helpful criticism.
Conflict of Interest
None declared
References
- Abete P, Vassalle M. Relation among Na+–K+ pump, Na+ activity and force in strophanthidin inotropy in sheep cardiac Purkinje fibres. J. Physiol. 1988;404:275–299. doi: 10.1113/jphysiol.1988.sp017290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baláti B, Varro A, Papp JG. Comparison of the cellular electrophysiological characteristics of canine left ventricular epicardium, M cells, endocardium and Purkinje fibers. Acta Physiol. Scand. 1998;164:181–190. doi: 10.1046/j.1365-201X.1998.00416.x. [DOI] [PubMed] [Google Scholar]
- Berg DE, Vassalle M. Oscillatory zones and their role in normal and abnormal sheep Purkinje fiber automaticity. J. Biomed. Sci. 2000;7:364–379. doi: 10.1007/BF02255812. [DOI] [PubMed] [Google Scholar]
- Bhattacharyya ML, Vassalle M. The effect of local anaesthetics on strophanthidin toxicity in canine cardiac Purkinje fibres. J. Physiol. 1981;312:125–142. doi: 10.1113/jphysiol.1981.sp013620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhattacharyya ML, Vassalle M. Effects of tetrodotoxin on electrical and mechanical activity of cardiac Purkinje fibers. J. Electrocardiol. 1982;15:351–360. doi: 10.1016/s0022-0736(82)81008-x. [DOI] [PubMed] [Google Scholar]
- Bocchi L, Vassalle M. Characterization of the slowly inactivating sodium current INa2 in canine cardiac single Purkinje cells. Exp. Physiol. 2008;93:347–361. doi: 10.1113/expphysiol.2007.040881. [DOI] [PubMed] [Google Scholar]
- Coppini R, Ferrantini C, Yao L, Fan P, Stillitano M, Del Lungo F, et al. Late sodium current inhibition reverses electro-mechanical dysfunction in human hypertrophic cardiomyopathy. Circulation. 2013;127:575–584. doi: 10.1161/CIRCULATIONAHA.112.134932. [DOI] [PubMed] [Google Scholar]
- Coraboeuf E, Deroubaix E, Coulombe A. Effect of tetrodotoxin on action potentials of the conducting system in the dog heart. Am. J. Physiol. 1979;236:561–567. doi: 10.1152/ajpheart.1979.236.4.H561. [DOI] [PubMed] [Google Scholar]
- Cordeiro JM, Spitzer KW, Giles WR. Repolarizing K+ currents in rabbit heart Purkinje cells. J. Physiol. 1998;508:811–823. doi: 10.1111/j.1469-7793.1998.811bp.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Datyner N, Gintant G, Cohen IS. Microprocessor controlled trituration device for the dissociation of cardiac and other tissues. Pflügers Arch. 1985;403:105–108. doi: 10.1007/BF00583289. [DOI] [PubMed] [Google Scholar]
- Du F, Vassalle M. A 4-aminopyridine sensitive current activating during slow ramps in cardiac single Purkinje cells. FASEB J. 1999;13:A97. (Abstract) [Google Scholar]
- Dumaine R, Cordeiro JM. Comparison of K+ currents in cardiac Purkinje cells isolated from rabbit and dog. J. Mol. Cell. Cardiol. 2007;42:378–389. doi: 10.1016/j.yjmcc.2006.10.019. [DOI] [PubMed] [Google Scholar]
- Dun W, Boyden PA. The Purkinje cell. J. Mol. Cell. Cardiol. 2008;45:617–624. doi: 10.1016/j.yjmcc.2008.08.001. 2008 style. [DOI] [PMC free article] [PubMed] [Google Scholar]
- El-Sherif N, Turitto G. Torsade de pointes. Curr. Opin. Cardiol. 2003;18:6–13. doi: 10.1097/00001573-200301000-00002. [DOI] [PubMed] [Google Scholar]
- Gaborit N, Szuts S, Le Bouter V, Varro A, Escande D, Nattel S, et al. Regional and tissue specific transcript signatures of ion channel genes in the non-diseased human heart. J. Physiol. 2007;582:675–693. doi: 10.1113/jphysiol.2006.126714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilly WF, Armstrong CM. Threshold channels – a novel type of sodium channel in squid giant axon. Nature. 1984;309:448–450. doi: 10.1038/309448a0. [DOI] [PubMed] [Google Scholar]
- Han W, Wang Z, Nattel S. A comparison of transient outward currents in canine cardiac Purkinje cells and ventricular myocytes. Am. J. Physiol. Heart Circ. Physiol. 2000;279:466–474. doi: 10.1152/ajpheart.2000.279.2.H466. [DOI] [PubMed] [Google Scholar]
- Haufe V, Camacho JA, Dumaine R, Günther B, Bollensdorff C, von Banchet GS, et al. Expression pattern of neuronal and skeletal muscle voltage-gated Na+ channels in the developing mouse heart. J. Physiol. 2005a;564:683–696. doi: 10.1113/jphysiol.2004.079681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haufe V, Cordeiro JM, Zimmer T, Wu YS, Schiccitano S, Benndorf K, et al. Contribution of neuronal sodium channels to the cardiac fast sodium current INa is greater in dog heart Purkinje fibers than in ventricles. Cardiovasc. Res. 2005b;65:117–127. doi: 10.1016/j.cardiores.2004.08.017. [DOI] [PubMed] [Google Scholar]
- Hirano Y, Hiraoka M. Changes in K+ currents induced by Ba2+ in guinea pig ventricular muscles. Am. J. Physiol. Heart Circ. Physiol. 1986;251:H24–H33. doi: 10.1152/ajpheart.1986.251.1.H24. [DOI] [PubMed] [Google Scholar]
- Iacono G, Vassalle M. On the mechanism of the different sensitivity of Purkinje and myocardial fibers to strophanthidin. J. Pharmacol. Exp. Ther. 1990;253:1–12. [PubMed] [Google Scholar]
- Imoto Y, Ehara T, Matsuura H. Voltage- and time-dependent block of iK1 underlying Ba2+ -induced ventricular automaticity. Am. J. Physiol. Heart Circ. Physiol. 1987;252:H325–H333. doi: 10.1152/ajpheart.1987.252.2.H325. [DOI] [PubMed] [Google Scholar]
- Isenberg G, Klöckner U. Calcium currents of isolated bovine ventricular myocytes are fast and of large amplitude. Pflügers Arch. 1982;395:30–41. doi: 10.1007/BF00584965. [DOI] [PubMed] [Google Scholar]
- Ishihara K. Time-dependent outward currents through the inward rectifier potassium channel IRK1. The role of weak blocking molecules. J. Gen. Physiol. 1997;109:229–243. doi: 10.1085/jgp.109.2.229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishihara K, Ehara T. A repolarization-induced transient increase in the outward current of the inward rectifier K+ channel in guinea-pig cardiac myocytes. J. Physiol. 1998;510:755–771. doi: 10.1111/j.1469-7793.1998.755bj.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishihara K, Mitsuiye T, Noma A, Takano M. The Mg2+ block and intrinsic gating underlying inward rectification of the K1 current in guinea-pig cardiac myocytes. J. Physiol. 1989;419:297–320. doi: 10.1113/jphysiol.1989.sp017874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kenyon JL, Gibbons WR. 4-Aminopyridine and the early outward current of sheep cardiac Purkinje fibers. J. Gen. Physiol. 1979;73:139–157. doi: 10.1085/jgp.73.2.139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim EM, Choy Y, Vassalle M. Mechanisms of suppression and initiation of pacemaker activity in guinea pig sino-atrial node superfused in high [K+]o. J. Mol. Cell. Cardiol. 1997;29:1433–1445. doi: 10.1006/jmcc.1997.0382. [DOI] [PubMed] [Google Scholar]
- Lin C-I, Vassalle M. Role of sodium in strophanthidin toxicity of Purkinje fibers. Am. J. Physiol. 1978;234:H477–H486. doi: 10.1152/ajpheart.1978.234.4.H477. [DOI] [PubMed] [Google Scholar]
- Liu B, McCullough JR, Vassalle M. On the mechanism of increased potassium conductance by the potassium channel opener BRL 34915 in isolated ventricular myocytes. Drug Dev. Res. 1990;19:409–423. [Google Scholar]
- Lopatin AN, Nichols CG. Inward rectifiers in the heart: an update on IK1. J. Mol. Cell. Cardiol. 2001;33:625–638. doi: 10.1006/jmcc.2001.1344. [DOI] [PubMed] [Google Scholar]
- Lopatin AN, Makhina EN, Nichols CG. Potassium channel block by cytoplasmic polyamines as the mechanism of intrinsic rectification. Nature. 1994;372:366–369. doi: 10.1038/372366a0. [DOI] [PubMed] [Google Scholar]
- Matsuda H, Saigusa A, Irisawa H. Ohmic conductance through the inwardly rectifying K channel and blocking by internal Mg2+ Nature. 1987;325:156–159. doi: 10.1038/325156a0. [DOI] [PubMed] [Google Scholar]
- Osterrieder W, Yang QF, Trautwein W. Effects of barium on the membrane currents in the rabbit S-A node. Pflügers Arch. 1982;394:78–84. doi: 10.1007/BF01108311. [DOI] [PubMed] [Google Scholar]
- Pu J, Boyden PA. Alterations of Na+ currents in myocytes from epicardial border zone of the infarcted heart. A possible ionic mechanism for reduced excitability and postrepolarization refractoriness. Circ. Res. 1997;81:110–119. doi: 10.1161/01.res.81.1.110. [DOI] [PubMed] [Google Scholar]
- Qu Y, Karnabi E, Chahine M, Vassalle M, Boutjdir M. Expression of skeletal muscle NaV1.4 Na channel isoform in canine cardiac Purkinje myocytes. Biochem. Biophys. Res. Commun. 2007;355:28–33. doi: 10.1016/j.bbrc.2007.01.101. Epub. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richmond JE, Featherstone DE, Hartmann HA, Ruben PC. Slow inactivation in human cardiac sodium channels. Biophys. J. 1998;74:2945–2952. doi: 10.1016/S0006-3495(98)78001-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rota M, Vassalle M. Patch-clamp analysis in canine cardiac Purkinje cells of a novel sodium component in the pacemaker range. J. Physiol. 2003;548:147–165. doi: 10.1113/jphysiol.2003.039263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schram G, Pourrier M, Melnyk P, Nattel S. Differential distribution of cardiac ion channel expression as a basis for regional specialization in electrical function. Circ. Res. 2002;90:939–950. doi: 10.1161/01.res.0000018627.89528.6f. [DOI] [PubMed] [Google Scholar]
- Shah AK, Cohen IS, Datyner NB. Background K+ current in isolated canine cardiac Purkinje myocytes. Biophys. J. 1987;52:519–525. doi: 10.1016/S0006-3495(87)83241-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimoni Y, Clark RB, Giles WR. Role of an inwardly rectifying potassium current in rabbit ventricular action potential. J. Physiol. 1992;448:709–727. doi: 10.1113/jphysiol.1992.sp019066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spiegler PA, Vassalle M. Role of voltage oscillations in the automaticity of sheep cardiac Purkinje fibers. Can. J. Physiol. Pharmacol. 1995;73:1165–1180. doi: 10.1139/y95-166. [DOI] [PubMed] [Google Scholar]
- Tytgat J, Vereecke J, Carmeliet E. A combined study of sodium current and T-type calcium current in isolated cardiac cells. Pflügers Arch. 1990;417:142–148. doi: 10.1007/BF00370691. [DOI] [PubMed] [Google Scholar]
- Valdivia CR, Chu WW, Pu J, Foell JD, Haworth RA, Wolff MR, et al. Increased late sodium current in myocytes from a canine heart failure model and from failing human heart. J. Mol. Cell. Cardiol. 2005;38:475–483. doi: 10.1016/j.yjmcc.2004.12.012. [DOI] [PubMed] [Google Scholar]
- Valenzuela F, Vassalle M. Role of the membrane potential on Ba2+ -induced automaticity in guinea pig cardiac myocytes. Cardiovasc. Res. 1991;25:421–430. doi: 10.1093/cvr/25.5.421. [DOI] [PubMed] [Google Scholar]
- Vandenberg CA. Inward rectification of a potassium channel in cardiac ventricular cells depends on internal magnesium ions. Proc. Natl Acad. Sci. USA. 1987;84:2560–2564. doi: 10.1073/pnas.84.8.2560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vassalle M. Cardiac pacemaker potentials at different extra- and intracellular K concentrations. Am. J. Physiol. 1965;208:770–775. doi: 10.1152/ajplegacy.1965.208.4.770. [DOI] [PubMed] [Google Scholar]
- Vassalle M. An analysis of cardiac pacemaker potential by means of a “voltage clamp” technique. Am. J. Physiol. 1966;210:1335–1341. doi: 10.1152/ajplegacy.1966.210.6.1335. [DOI] [PubMed] [Google Scholar]
- Vassalle M. The relationship among cardiac pacemakers: overdrive suppression. Circ. Res. 1977;41:269–277. doi: 10.1161/01.res.41.3.269. [DOI] [PubMed] [Google Scholar]
- Vassalle M. The role of the slow inward current in impulse formation. In: Zipes DP, Bailey JC, Elharrar V, editors. The slow inward current and cardiac arrhythmias. The Hague: Martinus Nijhoff Publishers; 1980. pp. 127–148. [Google Scholar]
- Vassalle M. The vicissitudes of the pacemaker current IKdd of cardiac Purkinje fibers. J. Biomed. Sci. 2007;14:699–716. doi: 10.1007/s11373-007-9182-2. [DOI] [PubMed] [Google Scholar]
- Vassalle M. Cardiac Purkinje fibers: normal function and its derangements. In: Parveen M, editor. Cardiac functioning disorders, challenges and therapies. New Delhi & London: Japee Medical Publishers; 2013. pp. 48–86. [Google Scholar]
- Vassalle M, Bhattacharyya M. Local anesthetics and the role of sodium in the force development by canine ventricular muscle and Purkinje fibers. Circ. Res. 1980;47:666–674. doi: 10.1161/01.res.47.5.666. [DOI] [PubMed] [Google Scholar]
- Vassalle M, Yu H, Cohen IS. The pacemaker current in cardiac Purkinje myocytes. J. Gen. Physiol. 1995;106:559–578. doi: 10.1085/jgp.106.3.559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vassalle M, Bocchi L, Du F. A slowly inactivating sodium current (INa2) in the plateau range in canine cardiac Purkinje single cells. Exp. Physiol. 2007;92:161–173. doi: 10.1113/expphysiol.2006.035279. [DOI] [PubMed] [Google Scholar]
- Vereecke J, Isenberg G, Carmeliet E. K efflux through inward rectifying K channels in voltage clamped Purkinje fibers. Pflügers Arch. 1980;384:207–217. doi: 10.1007/BF00584555. [DOI] [PubMed] [Google Scholar]
- Weidmann S. Effect of current flow on the membrane potential of cardiac muscle. J. Physiol. 1951;115:227–236. doi: 10.1113/jphysiol.1951.sp004667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zimmer T, Bollensdorff C, Haufe V, Birch-Hirschfeld E, Benndorf K. Mouse heart Na+ channels: primary structure and function of two isoforms and alternatively spliced variants. Am. J. Physiol. Heart Circ. Physiol. 2002;282:H1007–H1017. doi: 10.1152/ajpheart.00644.2001. [DOI] [PubMed] [Google Scholar]
- Zygmunt AC, Eddlestone GT, Thomas GP, Nesterenko VV, Antzelevitch C. Larger late sodium conductance in M cells contributes to electrical heterogeneity in canine ventricle. Am. J. Physiol. 2001;281:H689–H697. doi: 10.1152/ajpheart.2001.281.2.H689. [DOI] [PubMed] [Google Scholar]