Abstract
Congenital Heart Disease (CHD) is the most frequent and deadly birth defect. Patients with CHD that survive the neonatal period often progress to develop advanced heart failure requiring specialized treatment including cardiac transplantation. A full understanding of the transcriptional networks that direct cardiac progenitors during heart development will enhance our understanding of both normal cardiac function and pathological states. These findings will also have important applications for emerging therapies and the treatment of congenital heart disease. Furthermore, a number of shared transcriptional pathways or networks have been proposed to regulate the development and regeneration of tissues such as the heart. We have utilized transgenic technology to isolate and characterize cardiac progenitor cells from the developing mouse heart and have begun to define specific transcriptional networks of cardiovascular development. Initial studies identified Tdgf1 as a potential target of Nkx2-5. To mechanistically dissect the regulation of this molecular program, we utilized an array of molecular biological techniques to confirm that Nkx2-5 is an upstream regulator of the Tdgf1 gene in early cardiac development. These studies further define Nkx2-5 mediated transcriptional networks and enhance our understanding of cardiac morphogenesis.
Keywords: Nkx2-5, Tdgf1, Cardiogenesis, Mouse
Introduction
Congenital Heart Disease (CHD) afflicts approximately 1% of live births and accounts for the largest incidence of birth defects [1,2]. Furthermore, the incidence of CHD may be more than 10 fold greater in nonviable embryos (i.e. spontaneously aborted fetuses or stillbirths) [1–3]. Cardiac development is a complex, well-orchestrated event that includes the specification, proliferation, migration and differentiation of cardiomyocytes that become electrically coupled and ultimately form a functional syncytium. Although recent advances have begun to unravel the anatomical defects associated with CHD, the molecular networks that accompany these perturbations are incompletely defined [4,5]. In addition, CHD contributes significantly to advanced heart failure in the pediatric and adult population and these patients require aggressive pharmacological and mechanical therapies and/or orthotopic heart transplantation. Developmental studies are paramount to the emerging field of regenerative medicine, as many of these embryonic transcriptional networks are also important during repair and regenerative responses in the injured adult heart. Elegant studies utilizing genetically modified mouse models have demonstrated the essential roles for transcription factors and signaling molecules at discrete stages of heart development. Nkx2-5 is one of the earliest markers of the cardiac lineage as it is abundantly expressed in the cardiac progenitors that form the cardiac crescent (E7.75) [6,7]. This homeodomain transcription factor (i.e. Nkx2-5) is expressed throughout development and in the adult. Embryos lacking Nkx2-5 are lethal (E9.5–E11.5) due to growth retardation and abnormalities of the left ventricular chamber of the developing heart [6,7]. The association of NKX2-5 mutations with human CHD further highlights the importance of Nkx2-5 in cardiac development [8–13]. These defects associated with NKX2-5 mutations appear to identify a role for Nkx2-5 early in cardiac morphogenesis as well as a later role in maintaining myocyte lineage identity.
Tdgf1 (Cripto), the prototypic member of the EGF-CFC (epidermal growth factor-like-cripto-FRL-1-cryptic) family of EGF-like molecules is also an important signaling factor during cardiogenesis. The earliest embryonic Tdgf1 expression is observed in the inner cell mass of the blastocyst [14]. As murine embryogenesis proceeds, Tdgf1 expression becomes limited to the mesodermal progenitors which include the cardiac progenitors. At E8.5, Tdgf1 expression is further restricted to the myocardium of the developing heart tube and by E9.5 expression is very localized and limited to the cells of the truncus arteriosus in the developing heart [15,16]. Tdgf1 continues to be expressed within the truncus arteriosus until E10.5, after which expression is relatively absent in the developing and adult heart [14]. Tdgf1 null embryos are lethal by E7.5 due to defects in axial organization and formation of mesodermal derivatives [17,18]. These Tdgf1 mutant embryos also exhibit defects in cardiac morphogenesis as they lack expression of myocardial differentiation genes Mlc2v and α-Mhc [18,19]. The critical role for Tdgf1 during cardiogenesis was further evident as the Tdgf1−/− mESCs lacked the ability to form contracting cardiomyocytes [19].
In the present study, we further define the Nkx2-5 dependent molecular program in early cardiac development. We establish that Tdgf1 is a direct downstream target of Nkx2-5. Collectively, these studies enhance our understanding of molecular networks that govern discrete stages of early cardiogenesis.
Materials and Methods
FACS and transcriptome analyses
We utilized the previously generated Nkx2-5-EYFP transgenic mouse model and isolated cardiac progenitors from the E7.75 cardiac crescent following 0.25% Trypsin/EDTA (Invitrogen) digestion and FACS analysis [20]. Using a MoFlo Flow Cytometer (Beckman Coulter), EYFP labeled cells were collected directly into Tripure (Roche) and RNA was extracted and amplified as previously described [20]. Oligonucleotide array hybridizations were carried out according to the Affymetrix protocol as previously described and published [20,21].
Electrophoretic mobility shift and chromatin immunoprecipitation assays
C2C12 cells, grown in DMEM media supplemented with 20% FBS, were transfected with myc-tagged Nkx2-5 using Lipofectamine and Plus reagent (Invitrogen). After 24 hours, nuclear extracts were prepared and used for EMSA as previously described [22]. ChIP assays for evaluating occupancy of the Tdfg1 promoter binding by Nkx2-5 were performed as previously described [22], except that Myctagged Nkx2-5 was transfected into C2C12 cells. Chromatin solutions were prepared and immunoprecipitation reactions were performed using an anti-myc and control IgG sera. Promoter occupancy of Nkx2-5 was analyzed by amplifying the DNA fragment (314 bp) corresponding to the Tdgf1 promoter region containing three NKEs using the following set of primers (forward: 5′-caaaacacaaggtggggttggaaagc-3′ and reverse: 5′-ttgaaccctggactgtgcgtgc-3′). Co-Immunoprecipitation Assays were performed as previously described [21].
Transcriptional assays
Luciferase assays were performed as previously described [22]. Briefly, C2C12 myoblasts, in a six well plate, were transfected with a control (pGLT-Luc) or Tdgf1-Luc constructs with or without increasing amounts of Nkx2-5 overexpression plasmid as previously described [22].
Generation of an inducible ES/EB system for Nkx2-5 overexpression
Doxycycline-inducible Nkx2-5 overexpressing ES cells were generated from A2Lox ES cells as previously described [21]. Embryoid bodies (EBs) were prepared using the hanging drop technique and then cultured in suspension on a rotating plate beginning on day 2. Doxycycline (1 μg/ml) was added to the culture (48 hours) to induce protein expression and gene expression was analyzed using qRT-PCR as previously described [21].
Statistical analysis
All p-values were calculated using Student’s t-test analysis.
Results
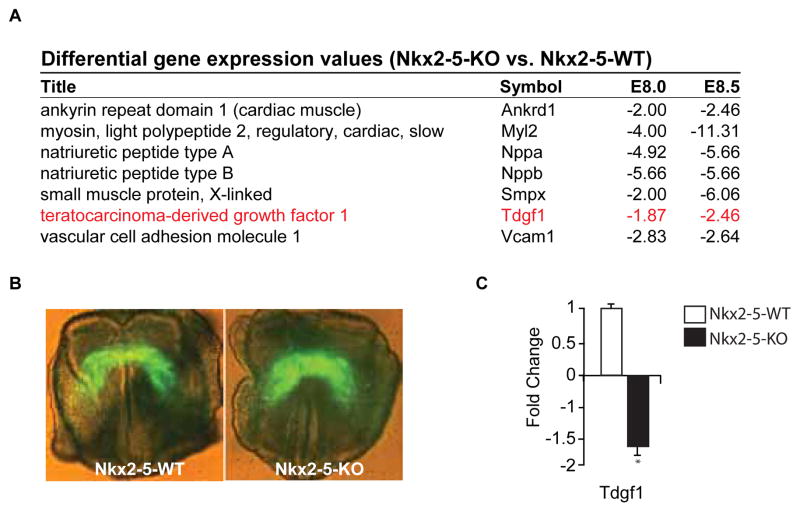
We have previously utilized the Nkx2-5-WT-EYFP and Nkx2-5- KO-EYFP transgenic mouse models to identify the Nkx2-5 dependent transcriptome of cardiac progenitors [20,21]. Selected representative transcripts found to be enriched in the Nkx2-5-WT-EYFP population compared to the Nkx2-5-KO-EYFP population at two stages of early cardiac development, E8.0 and E8.5, are listed in Figure 1A. Note that the EGF-CFC family member, Tdgf1, was one of such transcripts enriched at both time points. Utilizing these same transgenic mouse models, we collected the EYFP positive cardiac progenitors from the crescent of the developing Nkx2-5 WT and KO embryos (Figure 1B). Using qRT-PCR we confirmed that Tdgf1 was also enriched (1.6 fold +/− 0.2, p<0.05, n=3) in these earliest EYFP positive cardiac progenitors from the Nkx2-5-WT embryo compared to the Nkx2-5-KO embryo (Figure 1C). This Nkx2-5 dependent expression of Tdgf1 appeared to be limited to the early stages of cardiac development as at E9.0 there was no significant difference of Tdgf1 expression in the Nkx2-5-WT-EYFP population compared to the Nkx2-5-KO-EYFP population and at E9.5 and E10.5 there was no longer expression of Tdgf1 at the transcript level in either the Nkx2-5-WT-EYFP or Nkx2-5-KO-EYP positive cells (data not shown). These data support the hypothesis that Nkx2-5 acts as an important regulator of Tdgf1 early during cardiac development.
Figure 1. Tdgf1 transcript expression in WT and Nkx2-5 null cardiac progenitor cells.
(A) Selected transcripts that were significantly dysregulated at defined developmental stages in the Nkx2-5 null background compared to the age-matched WT cardiac progenitor cells. Note the statistically significant downregulation of Tdgf1. (B) EYFP is directed to the cardiac progenitor cells in the 6kbNkx2-5-EYFP:Nkx2-5-WT and 6kbNkx2-5-EYFP:Nkx2-5- KO littermates at E7.75. Note that the WT and Nkx2-5 mutant embryos are indistinguishable at E7.75. Individual EYFP positive embryos are dissociated and sorted using flow cytometry to collect EYFP positive and negative cells, respectively. RNA is isolated, amplified and qRT-PCR performed from EYFP positive cells of age matched Nkx2-5-WT and Nkx2-5-KO littermates. (C) qRT-PCR of Tdgf1 in WT (open bar) and null (black bar) cardiac progenitors of the cardiac crescent confirming decreased Tdgf1 transcript expression in the Nkx2-5-KO cardiac progenitors vs. the Nkx2-5-WT controls.
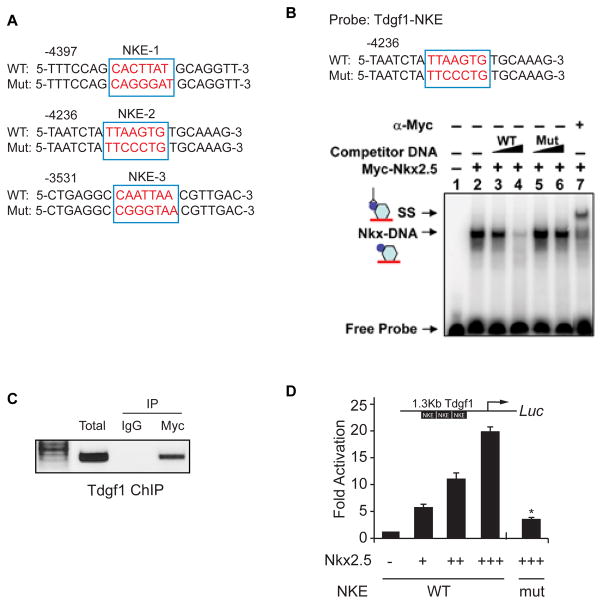
The 10kb upstream region of the murine Tdgf1 gene was evaluated for the presence of Nkx2-5 binding elements (NKE). Three potential NKEs with greater than 90% prediction value as determined by ConSite, were identified in the putative promoter region and are highlighted in Figure 2A. EMSA revealed that Nkx2-5 binds the Tdgf1 promoter, the binding could be competed with cold competitor and the complex could be supershifted with a myc antibody that recognizes the Nkx2-5-myc fusion construct (Figure 2B). Using ChIP assays, we confirmed that Nkx2-5 binds the Tdgf1 promoter in vivo (Figure 2C). We next undertook transcriptional assays to determine whether Nkx2-5 regulates Tdgf1 gene expression. We fused a 1.3 kb (−3 kb to −4 kb) region of the Tdgf1 upstream fragment harboring the three previously identified NKEs to the luciferase reporter. As shown in Figure 2D, cotransfection of the wild type Tdgf1 reporter plasmid and an Nkx2-5- expression vector resulted in a dose dependent activation of luciferase activity, which increased 6.5 +/− 1.1 fold, 10.2 +/− 1.3 fold and 19.4+/− 1.3 fold (p<0.05, n=3) with increasing amounts of Nkx2-5 compared to the control (empty reporter plasmid). Moreover, mutation of the three NKEs significantly reduced the Nkx2-5-dependent activation of the Tdgf1 promoter (4.2 +/− 0.3 fold with maximum Nkx2-5 dose; p<0.05, n=3; Figure 2D). These results support the notion that Nkx2-5 is a direct transcriptional activator of the Tdgf1 gene.
Figure 2. Nkx2-5 binds to and activates Tdgf1 expression.
(A) Identification of three potential NKEs in the 10kb upstream region of the Tdgf1 gene. (B) Sequence of oligonucleotides used for the EMSA is shown where wild type (WT) and the corresponding mutated (Mut) NKE are in red. Competition studies included increasing amounts of unlabeled WT probe (containing the NKE) or the same unlabeled probe with the NKE mutated (Mut). 32P-labeled WT (NKE) oligonucleotide probe was used for the EMSA and revealed a stable Nkx2-5-DNA (NKE) (lane 2) that could be competed with WT cold competitor (lanes 3–4) but not with Mut cold competitor (lanes 5–6). The complex could be supershifted with anti-myc serum (lane 7). (C) ChIP assay for binding of Nkx2-5 to the Tdgf1 promoter. Anti-myc but not the control antibody is capable of IP of the Tdgf1 promoter from C2C12 cells expressing myc-tagged Nkx2-5. (D) A 1.3 Kb Tdgf1 promoter harboring wild type (WT) or mutated (Mut) NKEs was fused to luciferase (luc) reporter gene and was transfected into C2C12 myoblast cells with or without increased amounts of the Nkx2-5 expressing plasmid. Fold change represents luciferase activity normalized to renilla expression; *, p<0.05, n=3.
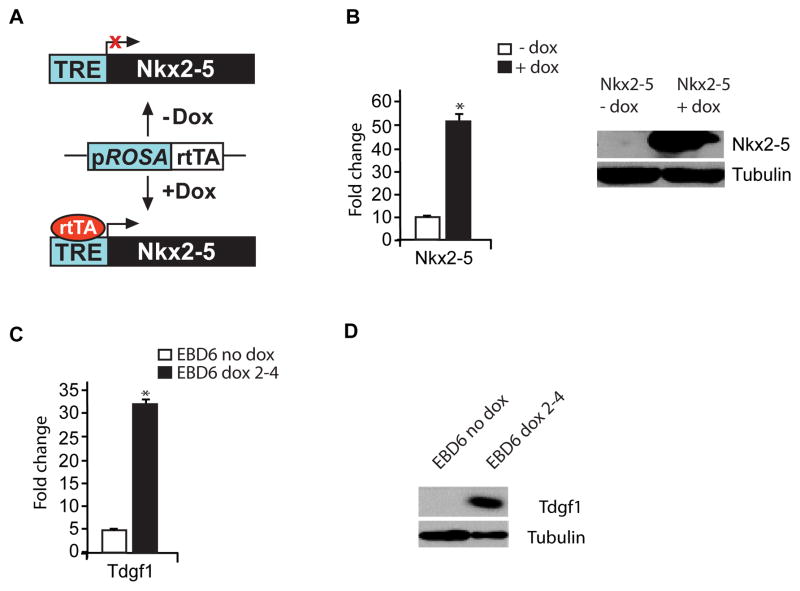
To further examine our hypothesis, we complemented our in vivo studies utilizing the Nkx2-5 null embryos with the generation of a doxycycline-inducible Nkx2-5 overexpressing embryonic stem (ES) cell line. Our strategy is schematized in Figure 3A and highlights that the reverse tetracycline transactivator (rtTA) binds to the tetracycline responsive element (TRE) to induce Nkx2-5 expression only after the addition of doxycycline. Using these engineered ES cells, embryoid bodies (EBs) were formed using the hanging drop technique. Overexpression of Nkx2-5 after induction with doxycycline was confirmed at both the RNA and protein level (Figure 3B). Using these engineered ES cells, embryoid bodies (EBs) were formed using the hanging drop technique. Nkx2-5 expression was induced for 48 hours at days 2-4 of EB formation, EBs were harvested on day 6 and RNA and protein were extracted from induced and control EBs. qRT-PCR analyses revealed that the induction of Nkx2-5 expression results in 31.1 +/− 1.9 fold increase in Tdgf1 expression (p<0.5, n=3; Figure 3C). Western blot analysis also confirmed increased protein expression of Tdgf1 after induction of Nkx2-5 expression (Figure 3D). These overexpression studies further support the hypothesis that Tdgf1 is a direct downstream target gene of Nkx2-5.
Figure 3. Nkx2-5 overexpression in ES/EB system results in increased levels of Tdgf1.
(A) Integration of Nkx2-5-expressing vector, pLoxNkx2-5, into the X chromosome of the Ainv15 ES cells to place the cDNA of Nkx2-5 under the control of tetracycline responsive element (TRE). Upon addition of doxycycline (+ Dox), reverse tetracycline transactivator (rtTA) binds to the TRE and induces Nkx2-5 expression. (B) Increased expression of Nkx2-5 RNA and protein in EB cells following the induction with Dox for 48hrs is confirmed; *, p<0.05, n=3. (C) Analyses of fold change in the expression of the Tdgf1 transcript using qRT-PCR following the induction with doxycycline (Dox) for 48hrs (day 2–4) of EB formation; *, p<0.05, n=3. (E) Western blot analysis confirms increased expression of Tdgf1 in EBs overexpressing Nkx2-5 following the induction with Dox for 2 days (day 2–4) vs. control EBs.
Discussion
We have previously identified the transcriptome of cardiac progenitors utilizing the Nkx2-5-WT-EYFP and Nkx2-5-KO-EYFP transgenic mouse models [20,21]. Employing these same transgenic mouse models, we confirmed that at the early stages of cardiac development, prior to E9.0, Tdgf1 expression is reduced in cardiac progenitors in the absence of Nkx2-5. Previous in-vitro studies of cardiac myocyte differentiation in P19 cells have also identified Tdgf1 as a downstream target of Nkx2-5 [23].
Using Embryoid Body (EB) differentiation assays, Embryonic Stem (ES) cells have the capacity to differentiate to all mesodermal-derived lineages, including a hematopoietic, endothelial, cardiac and skeletal muscle cell which faithfully recapitulates development [24–26]. EB developmental transcript profiles of Tdgf1 reveal expression of Tdgf1 in ES cells. The Tdgf1 expression continues in early EBs peaking at EB day 4, and then is rapidly downregulated [16]. Nkx2-5 expression is first noted at EB day 4 with the emergence of the cardiac progenitors and maintains its expression throughout cardiac development [23,27]. Thus, although there is overlap in expression, initial Tdgf1 expression is significantly earlier than that of Nkx2-5 leading to the hypothesis that although initiation of Tdgf1 expression is Nkx2-5 independent, the maintenance of Tdgf1 in developing cardiomyocytes is dependent upon Nkx2-5. Several examples of this type of transcriptional regulation have previously been described in cardiogenesis; most notably, the Nodal dependent initiation of Pitx2c expression, with Nkx2-5 dependent maintenance of Pitx2c expression at later stages of development in the absence of Nodal signaling [28].
Our underlying hypothesis is that Nkx2-5 transcriptionally activates the Tdgf1 gene and the Tdgf1 gene plays an essential role in not only specification but also maintenance and differentiation of early cardiac progenitors. Although Tdfg1 null ES cells fail to develop beating cardiomyocytes, the cells do express early cardiac transcription factors such as Nkx2-5, Mef2 and Gata4 [19]. Consistent with the early transient expression of Tdfg1 during differentiation of wild-type mESCs, it was shown that the time point of adding recombinant soluble Tdgf1 protein, its concentration and the duration of exposure to this signaling factor are critical parameters that determine the efficiency of obtaining beating cardiomyocytes [16]. These findings suggest that Tdgf1 is involved in the transition from myocardial specification to differentiation. Tdgf1 has also been shown to be highly expressed in many cancers including breast, pancreatic, ovarian and colon carcinomas [29]. Further studies exploring Tdgf1’s oncogenic potential has shown that in vivo, Tdgf1 induces cell proliferation, reduces apoptosis, and increases cell migration [30]. These properties however also may be important in its function during embryonic development and cardiogenesis.
CHD significantly contributes to embryonic lethality and advanced heart failure in the pediatric and adult population. An enhanced understanding of the regulatory mechanisms of the cardiac progenitor cells and the signaling pathways that promote cellular proliferation will serve as a platform for therapies directed towards advanced heart failure. In our current study we utilized multiple techniques to confirm that Tdgf1 is a direct downstream target of Nkx2-5. These studies suggest new insights into molecular mechanisms by which Nkx2-5 and Tdgf1 cooperatively regulate cardiac development.
Acknowledgments
We thank Richard Harvey for providing the Nkx2-5 null mice and Michael Kyba for providing the Teton inducible A2Lox ES cell line. We also acknowledge Michelina Iacovino and Xiaozhong Shi for critical discussions and technical assistance.
Footnotes
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
References
- 1.Hoffman JI. Incidence of congenital heart disease: II. Prenatal incidence. Pediatr Cardiol. 1995;16:155–165. doi: 10.1007/BF00794186. [DOI] [PubMed] [Google Scholar]
- 2.Olson EN, Srivastava D. Molecular pathways controlling heart development. Science. 1996;272:671–676. doi: 10.1126/science.272.5262.671. [DOI] [PubMed] [Google Scholar]
- 3.Fishman MC, Olson EN. Parsing the heart: genetic modules for organ assembly. Cell. 1997;91:153–156. doi: 10.1016/s0092-8674(00)80397-9. [DOI] [PubMed] [Google Scholar]
- 4.Harvey RP. Patterning the vertebrate heart. Nat Rev Genet. 2002;3:544–556. doi: 10.1038/nrg843. [DOI] [PubMed] [Google Scholar]
- 5.Olson EN, Schneider MD. Sizing up the heart: development redux in disease. Genes Dev. 2003;17:1937–1956. doi: 10.1101/gad.1110103. [DOI] [PubMed] [Google Scholar]
- 6.Tanaka M, Chen Z, Bartunkova S, Yamasaki N, Izumo S. The cardiac homeobox gene Csx/Nkx2.5 lies genetically upstream of multiple genes essential for heart development. Development. 1999;126:1269–1280. doi: 10.1242/dev.126.6.1269. [DOI] [PubMed] [Google Scholar]
- 7.Lyons I, Parsons LM, Hartley L, Li R, Andrews JE, et al. Myogenic and morphogenetic defects in the heart tubes of murine embryos lacking the homeo box gene Nkx25. Genes Dev. 1995;9:1654–1666. doi: 10.1101/gad.9.13.1654. [DOI] [PubMed] [Google Scholar]
- 8.Benson DW, Silberbach GM, Kavanaugh-McHugh A, Cottrill C, Zhang Y, et al. Mutations in the cardiac transcription factor NKX2.5 affect diverse cardiac developmental pathways. J Clin Invest. 1999;104:1567–1573. doi: 10.1172/JCI8154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gutierrez-Roelens I, Sluysmans T, Gewillig M, Devriendt K, Vikkula M. Progressive AVblock and anomalous venous return among cardiac anomalies associated with two novel missense mutations in the CSX/NKX25 gene. Hum Mutat. 2002;20:75–76. doi: 10.1002/humu.9041. [DOI] [PubMed] [Google Scholar]
- 10.Ikeda Y, Hiroi Y, Hosoda T, Utsunomiya T, Matsuo S, et al. Novel point mutation in the cardiac transcription factor CSX/NKX2.5 associated with congenital heart disease. Circ J. 2002;66:561–563. doi: 10.1253/circj.66.561. [DOI] [PubMed] [Google Scholar]
- 11.McElhinney DB, Geiger E, Blinder J, Benson DW, Goldmuntz E. NKX2.5 mutations in patients with congenital heart disease. J Am Coll Cardiol. 2003;42:1650–1655. doi: 10.1016/j.jacc.2003.05.004. [DOI] [PubMed] [Google Scholar]
- 12.Schott JJ, Benson DW, Basson CT, Pease W, Silberbach GM, et al. Congenital heart disease caused by mutations in the transcription factor NKX2-5. Science. 1998;281:108–111. doi: 10.1126/science.281.5373.108. [DOI] [PubMed] [Google Scholar]
- 13.Watanabe Y, Benson DW, Yano S, Akagi T, Yoshino M, et al. Two novel frameshift mutations in NKX2.5 result in novel features including visceral inversus and sinus venosus type ASD. J Med Genet. 2002;39:807–811. doi: 10.1136/jmg.39.11.807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Johnson SE, Rothstein JL, Knowles BB. Expression of epidermal growth factor family gene members in early mouse development. Dev Dyn. 1994;201:216–226. doi: 10.1002/aja.1002010305. [DOI] [PubMed] [Google Scholar]
- 15.Dono R, Scalera L, Pacifico F, Acampora D, Persico MG, et al. The murine cripto gene: expression during mesoderm induction and early heart morphogenesis. Development. 1993;118:1157–1168. doi: 10.1242/dev.118.4.1157. [DOI] [PubMed] [Google Scholar]
- 16.Parisi S, D’Andrea D, Lago CT, Adamson ED, Persico MG, et al. Nodal-dependent Cripto signaling promotes cardiomyogenesis and redirects the neural fate of embryonic stem cells. J Cell Biol. 2003;163:303–314. doi: 10.1083/jcb.200303010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Liguori GL, Echevarría D, Improta R, Signore M, Adamson E, et al. Anterior neural plate regionalization in cripto null mutant mouse embryos in the absence of node and primitive streak. Dev Biol. 2003;264:537–549. doi: 10.1016/j.ydbio.2003.08.023. [DOI] [PubMed] [Google Scholar]
- 18.Ding J, Yang L, Yan YT, Chen A, Desai N, et al. Cripto is required for correct orientation of the anteriorposterior axis in the mouse embryo. Nature. 1998;395:702–707. doi: 10.1038/27215. [DOI] [PubMed] [Google Scholar]
- 19.Xu C, Liguori G, Adamson ED, Persico MG. Specific arrest of cardiogenesis in cultured embryonic stem cells lacking Cripto1. Dev Biol. 1998;196:237–247. doi: 10.1006/dbio.1998.8862. [DOI] [PubMed] [Google Scholar]
- 20.Masino AM, Gallardo TD, Wilcox CA, Olson EN, Williams RS, et al. Transcriptional regulation of cardiac progenitor cell populations. Circ Res. 2004;95:389–397. doi: 10.1161/01.RES.0000138302.02691.be. [DOI] [PubMed] [Google Scholar]
- 21.Ferdous A, Caprioli A, Iacovino M, Martin CM, Morris J, et al. Nkx25 transactivates the Ets-related protein 71 gene and specifies an endothelial/endocardial fate in the developing embryo. Proc Natl Acad Sci U S A. 2009;106:814–819. doi: 10.1073/pnas.0807583106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Martin CM, Ferdous A, Gallardo T, Humphries C, Sadek H, et al. Hypoxia-inducible factor-2alpha transactivates Abcg2 and promotes cytoprotection in cardiac side population cells. Circ Res. 2008;102:1075–1081. doi: 10.1161/CIRCRESAHA.107.161729. [DOI] [PubMed] [Google Scholar]
- 23.Liu H, Harris TM, Kim HH, Childs G. Cardiac myocyte differentiation: the Nkx2.5 and Cripto target genes in P19 clone 6 cells. Funct Integr Genomics. 2005;5:218–239. doi: 10.1007/s10142-005-0140-7. [DOI] [PubMed] [Google Scholar]
- 24.Chung YS, Zhang WJ, Arentson E, Kingsley PD, Palis J, et al. Lineage analysis of the hemangioblast as defined by FLK1 and SCL expression. Development. 2002;129:5511–5520. doi: 10.1242/dev.00149. [DOI] [PubMed] [Google Scholar]
- 25.Keller G, Lacaud G, Robertson S. Development of the hematopoietic system in the mouse. Exp Hematol. 1999;27:777–787. doi: 10.1016/s0301-472x(99)00024-7. [DOI] [PubMed] [Google Scholar]
- 26.Lacaud G, Gore L, Kennedy M, Kouskoff V, Kingsley P, et al. Runx1 is essential for hematopoietic commitment at the hemangioblast stage of development in vitro. Blood. 2002;100:458–466. doi: 10.1182/blood-2001-12-0321. [DOI] [PubMed] [Google Scholar]
- 27.Hsiao EC, Yoshinaga Y, Nguyen TD, Musone SL, Kim JE, et al. Marking embryonic stem cells with a 2A self-cleaving peptide: a NKX25 emerald GFP BAC reporter. PLoS One. 2008;3:e2532. doi: 10.1371/journal.pone.0002532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Shiratori H, Sakuma R, Watanabe M, Hashiguchi H, Mochida K, et al. Two-step regulation of left-right asymmetric expression of Pitx2: initiation by nodal signaling and maintenance by Nkx2. Mol Cell. 2001;7:137–149. doi: 10.1016/s1097-2765(01)00162-9. [DOI] [PubMed] [Google Scholar]
- 29.Saloman DS, Bianco C, Ebert AD, Khan NI, De Santis M, et al. The EGF-CFC family: novel epidermal growth factor-related proteins in development and cancer. Endocr Relat Cancer. 2000;7:199–226. doi: 10.1677/erc.0.0070199. [DOI] [PubMed] [Google Scholar]
- 30.Adamson ED, Minchiotti G, Salomon DS. Cripto: a tumor growth factor and more. J Cell Physiol. 2002;190:267–278. doi: 10.1002/jcp.10072. [DOI] [PubMed] [Google Scholar]