Abstract
Objective
To compare the effect of two strategies (enhanced hand hygiene vs meticillin-resistant Staphylococcus aureus (MRSA) screening and decolonisation) alone and in combination on MRSA rates in surgical wards.
Design
Prospective, controlled, interventional cohort study, with 6-month baseline, 12-month intervention and 6-month washout phases.
Setting
33 surgical wards of 10 hospitals in nine countries in Europe and Israel.
Participants
All patients admitted to the enrolled wards for more than 24 h.
Interventions
The two strategies compared were (1) enhanced hand hygiene promotion and (2) universal MRSA screening with contact precautions and decolonisation (intranasal mupirocin and chlorhexidine bathing) of MRSA carriers. Four hospitals were assigned to each intervention and two hospitals combined both strategies, using targeted MRSA screening.
Outcome measures
Monthly rates of MRSA clinical cultures per 100 susceptible patients (primary outcome) and MRSA infections per 100 admissions (secondary outcome). Planned subgroup analysis for clean surgery wards was performed.
Results
After adjusting for clustering and potential confounders, neither strategy when used alone was associated with significant changes in MRSA rates. Combining both strategies was associated with a reduction in the rate of MRSA clinical cultures of 12% per month (adjusted incidence rate ratios (aIRR) 0.88, 95% CI 0.79 to 0.98). In clean surgery wards, strategy 2 (MRSA screening, contact precautions and decolonisation) was associated with decreasing rates of MRSA clinical cultures (15% monthly decrease, aIRR 0.85, 95% CI 0.74 to 0.97) and MRSA infections (17% monthly decrease, aIRR 0.83, 95% CI 0.69 to 0.99).
Conclusions
In surgical wards with relatively low MRSA prevalence, a combination of enhanced standard and MRSA-specific infection control approaches was required to reduce MRSA rates. Implementation of single interventions was not effective, except in clean surgery wards where MRSA screening coupled with contact precautions and decolonisation was associated with significant reductions in MRSA clinical culture and infection rates.
Trial registration
clinicaltrials.gov identifier: NCT00685867
Keywords: Infection Control < Infectious Diseases, Surgery
Article summary.
Strengths and limitations of this study
Unlike many previous studies, this was a large, controlled, prospective, multicentre, intervention study. The enrolled wards, from 10 hospitals in Europe and Israel, varied in terms of infection control infrastructure and meticillin-resistant Staphylococcus aureus prevalence, thus the results are likely to be generalisable to other settings.
Due to the nature of the quality improvement initiatives, investigators were not blinded to the allocated intervention. Interventions were not randomly allocated.
Introduction
Healthcare associated infections affect hundreds of millions of patients worldwide every year and represent an important cause of patient mortality and a major financial burden to health systems.1 Meticillin-resistant Staphylococcus aureus (MRSA), now endemic in many healthcare facilities, is a leading cause of healthcare associated infections2 and patients in surgical units are at increased risk due to factors such as invasive procedures, antibiotic exposure and prolonged healthcare contact. A number of countries mandate implementation of control measures, including MRSA screening.3 4 Not all mandated interventions, however, are supported by robust evidence.
Studies evaluating MRSA control strategies show conflicting results, particularly with regard to the use of active surveillance cultures.5–7 It is argued that broader infection control approaches, such as improving hand hygiene (HH) practices, may be as successful as MRSA-specific strategies.8 9 There are limitations, however, to current evidence with few prospective, controlled studies10 11 and many studies have assessed multiple interventions simultaneously.12 Quantifying the relative benefits of individual approaches is important, particularly as some strategies have significant cost implications, and will allow efficient use of limited resources.
Owing to the ongoing debate concerning optimal approaches to MRSA control,13 14 we performed a prospective, interventional, quality improvement study to compare the effect of an enhanced HH promotion strategy to an MRSA screening, isolation and decolonisation strategy when used alone and in combination on the incidence rates of MRSA clinical cultures and infections in surgical patients admitted to healthcare facilities across Europe and Israel. We also aimed to specifically assess these interventions in clean surgery wards where their benefits may be expected to be more pronounced.
Methods
Study design and population
This prospective, controlled, multicentre, interventional cohort study with a three phase interrupted time series design was conducted between March 2008 and July 2010. Thirty-three surgical wards of 10 hospitals in nine countries (Serbia, France, Spain (two hospitals), Italy, Greece, Scotland, Israel, Germany and Switzerland) were enrolled. Wards included orthopaedic (8), vascular (6), cardiothoracic/cardiovascular (5), general (4), abdominal (4), urology (3), neurosurgery (2) and plastic surgery (1) subspecialties. Characteristics of the enrolled wards varied (table 1).
Table 1.
Baseline phase characteristics of hospitals and wards enrolled in the study
| Hospital | Hospital characteristics |
Study ward characteristics |
Study arm | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Total beds (n) | Total number of single rooms (%) | Ratio of infection control nurses to beds | Surgical subspecialties | Total beds (n) | Total admissions during baseline phase (n) | Mean patient-to-nurse ratio (SD)* | Per cent hand hygiene compliance (95% CI) | Number of patients screened on admission (%) | Number identified MRSA positive on admission (%)† | ||
| 1 | 3611 | 45 (1.2) | 1:240 | Abdominal | 588 | 8018 | 6.4 (1.2) | 18.8 (15.1 to 22.9) | 0 (0) | 9 (0.1) | Enhanced hand hygiene |
| Cardiovascular | |||||||||||
| Orthopaedic | |||||||||||
| 2 | 317 | 235 (74.1) | 1:160 | Cardiothoracic | 72 | 1613 | 4.1 (1.8) | 75.4 (70.3 to 80.0) | 29 (1.8) | 20 (1.2) | Screening and decolonisation |
| Orthopaedic | |||||||||||
| Vascular | |||||||||||
| 3 | 850 | 135 (15.9) | 1:425 | Cardiovascular | 75 | 1841 | 5.6 (0.7) | 26.8 (24.4 to 29.4) | 14 (0.8) | 11 (0.6) | Screening and decolonisation |
| General | |||||||||||
| Orthopaedic | |||||||||||
| 4 | 822 | 0 (0) | 1:137 | Abdominal | 230 | 6574 | 3.7 (0.9) | 39.3 (34.6 to 44.1) | 182 (2.8) | 21 (0.3) | Combined |
| Orthopaedic | |||||||||||
| Urology | |||||||||||
| Vascular | |||||||||||
| 5 | 545 | 89 (16.3) | 1:272 | General | 121 | 1938 | 5.8 (1.5) | 14.3 (11.3 to 17.6) | 56 (2.9) | 4 (0.2) | Screening and decolonisation |
| Neurosurgery | |||||||||||
| Orthopaedic | |||||||||||
| Vascular | |||||||||||
| 6 | 547 | 4 (0.7) | 1:274 | General | 93 | 1300 | 16.8 (2.5) | 25.1 (20.7 to 30.1) | 0 (0) | 5 (0.4) | Screening and decolonisation |
| Orthopaedic | |||||||||||
| Vascular | |||||||||||
| 7 | 902 | 62 (6.9) | 1:180 | Abdominal | 84 | 1963 | 6.1 (1.5) | 76.5 (71.3 to 81.1) | 607 (30.9) | 41 (2.1) | Combined |
| General | |||||||||||
| Vascular | |||||||||||
| 8 | 850 | 202 (23.8) | 1:567 | Orthopaedic | 87 | 2434 | 5.5 (0.6) | 50.2 (44.6 to 55.8) | 0 (0) | 3 (0.1) | Enhanced hand hygiene |
| Urology | |||||||||||
| Vascular | |||||||||||
| 9 | 1350 | 150 (11.1) | 1:260 | Cardiothoracic | 164 | 1561 | 10.0 (2.2) | 67.0 (61.4 to 72.3) | 17 (1.1) | 15 (1.0) | Enhanced hand hygiene |
| Neurosurgery | |||||||||||
| Plastic surgery | |||||||||||
| 10 | 2044 | 402 (19.7) | 1:204 | Abdominal | 302 | 6366 | 4.8 (0.4) | 55.9 (51.2 to 60.5) | 1666 (26.2) | 140 (2.2) | Enhanced hand hygiene |
| Cardiovascular | |||||||||||
| Orthopaedic | |||||||||||
| Urology | |||||||||||
| Overall | 11 838 | 1324 (11.2) | 1816 | 33 608 | 6.6 (3.8) | 39.5 (38.1 to 40.9) | 2571 (7.6) | 269 (0.8) | |||
*Calculated by dividing the mean patient load by mean number of nurses working on the ward at a given time (averaged over day, evening and night shifts).
†By screening or clinical culture.
MRSA, meticillin-resistant Staphylococcus aureus.
The study consisted of baseline (6–7 months), intervention (12 months) and washout (6 months) phases. Initial baseline phase data collection started in one centre in March 2008 prior to the implementation of any interventions. All other centres started baseline phase data collection after May 2008. The intervention phase did not start for any study site until October 2008. During baseline and washout phases, wards employed their usual infection control practices. During the intervention phase, two strategies were investigated, with hospitals implementing one or both interventions in parallel (figure 1).
Figure 1.
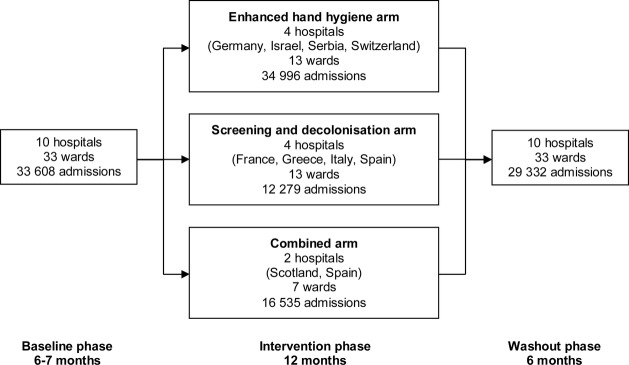
Flow of study wards through each phase of the study, 10 hospitals in nine countries were enrolled and were allocated to one of the three study arms during the intervention phase. The enhanced hand hygiene arm used hand hygiene promotion; the screening and decolonisation arm used universal meticillin-resistant Staphylococcus aureus (MRSA) screening coupled with contact precautions and decolonisation therapy with intranasal mupirocin and chlorhexidine body washes for identified MRSA carriers; the combined arm used a combination of hand hygiene promotion and targeted MRSA screening.
Interventions
The first intervention, the enhanced HH strategy, used the WHO multimodal HH promotion method consisting of (1) using alcohol-based handrub at the point of care, (2) training and education of healthcare workers, (3) observation and feedback of HH practices, (4) reminders in the workplace (eg, posters) and (5) improving the safety climate in the institution with management support for the initiative.15 Adherence to standard precautions (eg, gloves for body fluid contact) was encouraged. There was no attempt to change local practices regarding isolation of patients with MRSA as part of this intervention.
The second intervention, the screening and decolonisation strategy, used a universal MRSA screening approach. It consisted of screening patients admitted for more than 24 h for MRSA, on admission (within 48 h) then weekly. Patients were excluded from screening if they were undergoing ambulatory surgery or had already been screened within 5 days prior to admission to the surgical ward. The nares, perineum and wounds (if present) were swabbed. Chromogenic agar screening was used with the addition of PCR testing during the latter part of the intervention phase for patients who had risk factors for MRSA (eg, hospitalisation in the last year) whose chromogenic agar results were unlikely to be available before surgery. MRSA carriers were placed on contact precautions (gown and gloves during patient contact), administered decolonisation therapy with twice daily intranasal mupirocin and daily chlorhexidine washes for 5 days and perioperative prophylaxis was modified to reflect MRSA carriage. Chlorhexidine bathing was limited to the identified MRSA carriers and not used as a unit-wide intervention. Pre-emptive isolation was not used as part of this strategy.
The hospital was the unit for assignment of interventions due to practical reasons and the nature of the strategies. Four hospitals were assigned to each intervention and two hospitals used a combination of both strategies (the combined strategy) due to the introduction of national or local mandatory targeted MRSA screening policies during the study period which necessitated deviation from the original trial protocol (figure 1). The choice of allocation was influenced by the constraints on the study centres, such as cost and personnel (n=3), population size (n=1), capacity of the microbiology laboratories (n=3), prior exposure to specific interventions (n=1) and mandatory local or national interventions (n=2). Thus, this pragmatic approach took into account the institutions’ preferences, as participation in an entirely cluster-randomised trial would have meant that some of the hospitals could not have participated.
The targeted screening in the two hospitals in the combined strategy arm was based on risk factors for MRSA carriage (including patient characteristics or surgical subspecialty). One hospital using the combined strategy (hospital 4) introduced targeted screening of patients who were previously known to be MRSA positive, contacts of patients with MRSA and patients transferred from the intensive care unit or other healthcare facilities. The other hospital in the combined strategy arm (hospital 7) not only used targeted screening of patients with the same risk factors as hospital 4, but also screened nursing home residents, patients admitted to the hospital in the last 3 months, patients transferred from another ward within the same hospital and those admitted to vascular or abdominal surgery subspecialties. The assignment of hospitals to each study arm occurred prior to the start of the data collection. A summary of the nature of the interventions for each study arm is presented in table 2. The study protocol was registered with a public registry of clinical studies (available at: http://clinicaltrials.gov/ Identifier: NCT00685867).
Table 2.
Summary of the timing and nature of infection control interventions for each study arm
| Standard precautions | HH promotion | MRSA screening | MRSA isolation | MRSA decolonisation | |
|---|---|---|---|---|---|
| Baseline phase: 6–7 months (1 March 2008 to 31 January 2009)* | |||||
| Enhanced HH arm | –† | – | – | – | – |
| Screening and decolonisation arm | – | – | – | – | – |
| Combined arm | – | – | – | – | – |
| Intervention phase: 12 months (1 October 2008 to 31 January 2010)* | |||||
| Enhanced HH arm | Adherence to standard precautions (eg, gloves and other barriers as needed for contact with mucous membranes, wounds and body fluids) during care of all patients encouraged | HH promotion using the WHO multi-modal HH promotion method.15 Observation of 100 opportunities for HH per ward per month | – | – | – |
| Screening and decolonisation arm | – | – | Universal screening of patients admitted for more than 24 h, on admission then weekly (see ‘MRSA screening details’ in footnotes) | Patients MRSA colonised/infected placed on contact precautions (gown and gloves during contact). Patients with MRSA placed in single rooms or cohorted based on local capacity. Pre-emptive isolation of previously unknown MRSA carriers pending screening results not used | Patients MRSA colonised/infected given twice-daily intranasal mupirocin and daily chlorhexidine body washes (5 days) |
| Combined arm | Adherence to standard precautions (eg, gloves and other barriers as needed for contact with mucous membranes, wounds and body fluids) during care of all patients encouraged | HH promotion using the WHO multimodal HH promotion method.15 Observation of 100 opportunities for HH per ward per month | Targeted screening based on risk factors (see ‘MRSA screening details’ in footnotes) | Patients MRSA colonised/infected placed on contact precautions (gown and gloves during contact). Patients with MRSA placed in single rooms or cohorted based on local capacity. Pre-emptive isolation of previously unknown MRSA carriers pending screening results not used | Patients MRSA colonised/infected given topical decolonisation therapy at discretion of treating clinicians |
| Washout phase: 6 months (1 October 2009 to 31 July 2010)* | |||||
| Enhanced HH arm | – | – | – | – | – |
| Screening and decolonisation arm | – | – | – | – | – |
| Combined arm | – | – | Targeted screening based on risk factors (see ‘MRSA screening details’ in footnotes) | Patients MRSA colonised/infected placed on contact precautions (gown and gloves during contact). Patients with MRSA placed in single rooms or cohorted based on local capacity. Pre-emptive isolation of previously unknown MRSA carriers pending screening results not used | Patients MRSA colonised/infected given topical decolonisation therapy at discretion of treating clinicians |
*The start of the study period was staggered for hospitals. For each study phase, the start date is the date on which the first hospital entered the study phase and the end date indicates the date on which the last hospital completed the study phase.
†There were no specific interventions as part of the study. Hospitals employed their usual infection control practices during these study phases.
MRSA, meticillin-resistant Staphylococcus aureus; HH, hand hygiene.
MRSA screening details: Screening of nares, perineum and wounds (if present).
Universal screening (intervention phase) was used in the screening and decolonisation arm. It refers to screening patients admitted for more than 24 h and excluded patients undergoing ambulatory surgery and those screened within 5 days prior to admission to the surgical ward.
Targeted screening (intervention and washout phases) was used in the two centres in the combined arm due to introduction of local and national mandatory screening policies. One study centre (hospital 4) screened patients previously known to be MRSA positive, contacts of patients with MRSA and patients transferred from the intensive care unit or other healthcare facilities. The other centre (hospital 7) not only screened patients with the same risk factors as hospital 4, but also included nursing home residents, patients admitted to the hospital in the last 3 months, patients transferred from another ward within the same hospital and those admitted to vascular or abdominal surgery subspecialties.
Outcome measures
The primary outcome measure was the monthly nosocomial MRSA isolation rate, defined as the number of MRSA clinical isolates (those from specimens collected other than for screening purposes, counting one isolate per patient per month), per 100 susceptible patients (not previously known to be MRSA colonised or infected). Isolates from specimens collected more than 48 h after admission or within 30 days after discharge from study wards were considered nosocomial.
Secondary outcomes were the monthly rate of nosocomial MRSA infections per 100 admissions and adherence to HH guidelines and contact precautions. Infections were defined using Centers for Disease Control and Prevention (CDC) criteria.16 Adherence to HH guidelines was measured as the percentage of opportunities for HH in which staff used alcohol-based handrub and/or washed their hands according to the WHO method.15 Adherence to contact precautions was measured as the percentage of randomly audited patients with MRSA for whom precautions with gown and gloves during patient contact had been implemented.
Microbiological methods
Standardised laboratory manuals were provided to centres. Samples were processed in local laboratories using standard culture-based identification of MRSA from clinical specimens. In hospitals assigned to the screening and decolonisation arm, nasal and perineal swabs were pooled in the laboratory then plated directly onto a chromogenic medium (BBL CHROMagar MRSA II, BD Diagnostics, Belgium) and also incubated overnight in an enrichment medium to increase test sensitivity.17 Positive results could be reported within 24–48 h.18 PCR testing directly from pooled screening swabs was performed with the BD GeneOhm MRSA (BD Diagnostics, Belgium) or GeneXpert MRSA (Cepheid, Belgium) tests, which have turnaround times of 2–3 h and 1.5 h, respectively (see online supplementary table A1).18 All laboratories participated in an external quality assurance programme to evaluate their ability to detect, identify and perform antibiotic sensitivity testing on staphylococci from a variety of different specimens.19 MRSA isolates were shipped to the central laboratory (University of Antwerp, Belgium) for confirmation of identification.
Data collection
Research personnel from each hospital collected data and implemented the interventions at their study site. These personnel were from departments that supervise infection control activities at the participating hospitals, including infection control, infectious diseases and hospital epidemiology departments. They were trained at the study coordinating centre with regard to the study protocol, the outcome definitions and the use of the data collection tools prior to the start of the study to ensure consistency of data collection across the hospitals. Local microbiology laboratory data were reviewed to obtain information regarding MRSA isolated from screening and clinical cultures. Infections were monitored by twice weekly ward visits to review medical records and interview staff. Surgical site infection surveillance occurred up to 30 days postprocedure (or 12 months after prosthetic device insertion).
HH adherence was monitored by the research personnel who had been trained and validated in the WHO method of direct observation at the study coordinating centre.15 A standardised observation form was used by all centres. All hospitals collected data for 100 HH opportunities per ward during baseline and washout phases.20 HH observers were specifically instructed not to provide feedback to healthcare workers concerning their HH practices during these study phases and the observers were independent of surgical ward staff, reducing the likelihood of the Hawthorne effect, in which staff improve their practices when they are aware that they are being observed.21 During the intervention phase, there was intensive monitoring of HH practices in wards using the enhanced HH and combined strategies. In these wards, 100 HH opportunities per ward per month were observed as part of the intervention. Implementation of contact precautions, decolonisation therapy and single room isolation for MRSA carriers was randomly audited each month. Signage of MRSA status and availability of gowns, gloves and alcohol-based handrub for contact with MRSA carriers was also audited.
Data regarding numbers of admissions, patient-days, surgical procedures and level of staffing were collected. Owing to variation in the availability and quality of electronic medical record and pharmacy data between the study sites, individual-level data (such as length of stay) and antibiotic utilisation data for the surgical wards was not collected as part of this study. Ward-level data were submitted monthly to a central data management centre through a password-protected secure online database which included range, consistency and missing data checks. Meetings, site visits and monthly teleconferences were held to review data, ensure adherence to study protocols and address queries. Data were reviewed monthly for completeness and 6 monthly for validity by teleconferences with individual study sites. Institutional review boards of all centres approved the study with a waiver of individual informed consent.
Statistical analysis
The study was designed to detect a 30% difference in nosocomial MRSA isolation rate assuming a baseline rate of 1.0 clinical isolate per 100 susceptible patients and an absolute difference of 10% between intervention arms. Sample size calculations assumed a two-sided test, a type I error rate of 0.05 and 80% power, taking the wards as the unit of analysis. A minimum of 15 wards were required per study arm.
Crude MRSA rates were calculated by study arm. Adjusted incidence rate ratios (aIRR) were calculated using multilevel Poisson segmented regression accounting for stepwise changes in MRSA level and changes in log-linear trends associated with the interventions.22 This analysis allowed for two levels of random-effects: hospital-level variation in intercepts and baseline trends and nested ward-level variation in intercepts. It was adjusted for exposure given by the monthly number of susceptible patients or admissions per ward and allowed for extra-Poisson variation. Surgical subspecialty, baseline HH compliance, seasonal effects (using calendar-month) and patient-to-nurse ratios were adjusted for. Autocorrelation was accounted for using a lagged dependent variable. A similar analysis was performed for HH compliance, but used segmented multilevel logistic regression, adjusting for ward-specific baseline levels and trends, professional category, HH indication, patient-to-nurse ratios and monthly MRSA colonisation pressure (number of days patients known to be MRSA colonised/infected were in the wards each month).
Planned subgroup analyses were performed by hospital and for clean surgery wards (cardiothoracic, neuro, orthopaedic, plastic and vascular surgery) as studies have shown that intranasal mupirocin, which is active against Gram-positive organisms, may be more effective for surgical site infection prevention in clean compared with clean-contaminated surgery (eg, general or gastrointestinal surgery) where Gram-negative and anaerobic organisms may play a larger role.23 As screening intensity varied in the combined arm, a planned exploratory analysis of MRSA outcome data was conducted to better quantify the intervention effects. It accounted for stepwise changes and log–linear trends in outcomes associated with the HH intervention, as well as the monthly proportion of patients screened and monthly cumulative screening rate on wards to account for changes in trends of outcomes associated with screening. Analyses were conducted with STATA V.11.0 (STATA Corp, USA).
Results
During the study period, there were a total of 126 750 admissions and 99 638 surgical procedures on the study wards. Baseline admission MRSA prevalence, without systematic screening of all admitted patients, was 0.8% (269 of 33 608), ranging from 0.1% to 2.2% across surgical wards of each hospital. Baseline HH adherence varied between hospitals (39.5% overall, 95% CI 38.1% to 40.9%) as did use of targeted MRSA screening (0–30.9% of admissions; table 1). Study characteristics are shown in table 3 and online supplementary table A2.
Table 3.
Study characteristics by study period
| Characteristic | Baseline phase | Intervention phase | Washout phase |
|---|---|---|---|
| Duration (months) | 6–7* | 12 | 6 |
| Total admissions (n) | 33 608 | 63 810 | 29 332 |
| Total patient-days (n) | 264 035 | 496 975 | 249 119 |
| Total surgical procedures (n) | 27 768 | 49 747 | 22 123 |
| Procedures in clean surgery wards (n)† | 12 916 | 21 463 | 8787 |
| Procedures in other types of surgery wards (n)† | 14 852 | 28 284 | 13 336 |
| Mean patient-to-nurse ratio (SD)‡ | 6.55 (3.78) | 6.67 (3.59) | 6.87 (4.18) |
| Total number of patients MRSA positive on admission (%)§ | 269 (0.8) | 724 (1.1) | 228 (0.8) |
| Number positive by clinical culture (%) | 65 (0.2) | 85 (0.1) | 41 (0.1) |
| Number positive by screening swab (%) | 204 (0.6) | 639 (1.0) | 187 (0.6) |
*Baseline phase was 6 months in six hospitals and 7 months in four hospitals (two in the screening and decolonisation arm and one hospital in each of the enhanced hand hygiene and combined arms).
†Clean surgery wards included cardiothoracic, neuro, orthopaedic, plastic and vascular surgery. Other types of surgery wards included abdominal, general and urological surgery.
‡Calculated by dividing the mean patient load by mean number of nurses working on the ward at a given time (averaged over day, evening and night shifts).
§By screening or clinical culture.
MRSA, meticillin-resistant Staphylococcus aureus.
Adherence to HH guidelines
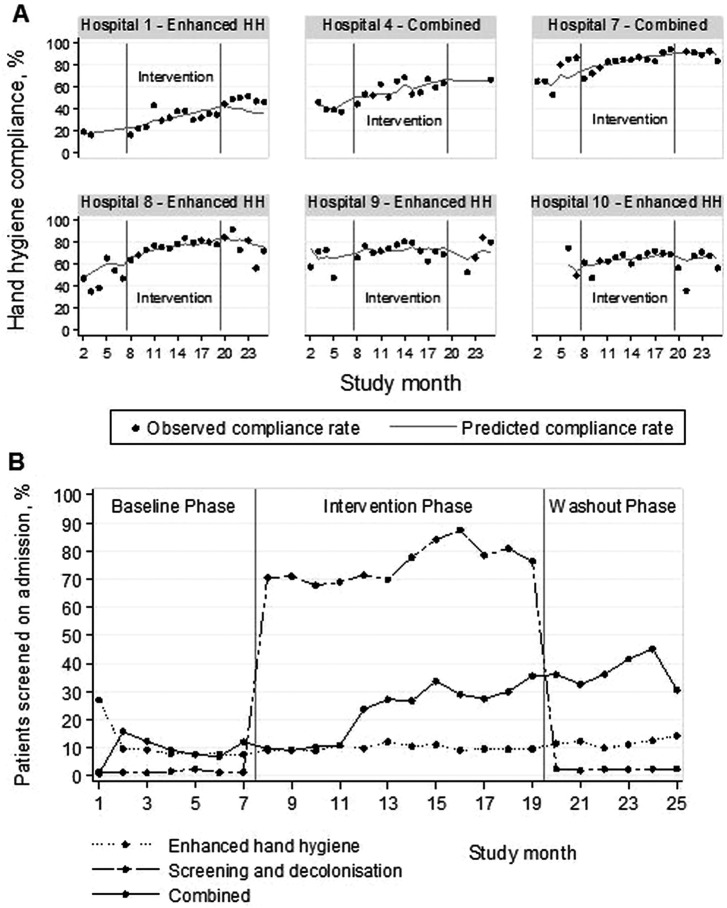
In the enhanced HH and combined arms, HH compliance improved in all centres with overall compliance increasing from 49.3% (95% CI 47.2% to 51.4%) to 63.8% (95% CI 63.2% to 64.4%) from baseline to intervention phases (figure 2A). After multivariable analysis, starting HH promotion was associated with a significant immediate increase in HH compliance (adjusted OR (aOR) 1.19, 95% CI 1.01 to 1.42; see online supplementary table A3). However, this benefit was not sustained after cessation of the HH campaign with a significant decreasing trend in HH adherence of 9% per month (aOR for month postintervention 0.91, 95% CI 0.85 to 0.97) during the washout phase. In wards in the screening and decolonisation arm, where no HH promotion occurred, compliance remained low at 30.5% (95% CI 28.7% to 32.4%) at baseline and 23.9% (95% CI 22.0% to 25.9%) during the washout phase.
Figure 2.
Implementation of the interventions, the top panel (A) shows the monthly hand hygiene (HH) compliance rates for hospitals in the enhanced HH and combined arms that used HH promotion campaigns. The solid dots represent the observed compliance rates while the lines represent the predicted compliance rates based on the regression model. The bottom panel (B) shows the proportion of patients screened on admission to the study wards by study arm.
Screening, contact precautions and decolonisation of MRSA carriers
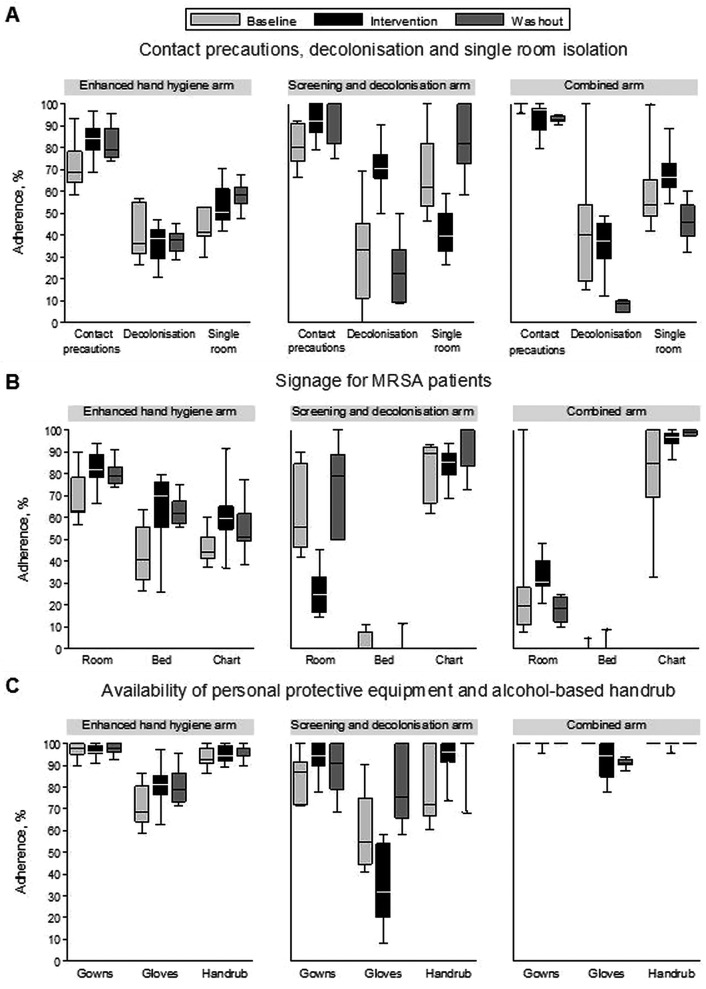
During the intervention phase, 9250 (75.3%) of 12 279 patients were screened on admission to wards in the screening and decolonisation arm. Admission MRSA prevalence was 2.1% (259 of 12 279), consisting of 27 patients (10.4%) with MRSA-positive clinical cultures and 232 patients (89.6%) identified by screening alone. PCR screening was used in addition to chromogenic agar cultures in 1047 (11.3%) of 9250 patients. Between baseline and intervention phases in screening and decolonisation wards, the proportion of audited MRSA carriers placed on contact precautions increased (from 81.1% to 90.7%), as did administration of decolonisation therapy (from 34.4% to 69.8%; figure 3). However, the proportion of audited MRSA carriers in single rooms decreased (from 67.8% to 40.1%), possibly due to a shortage of rooms for the higher number of identified MRSA carriers. Reasons for non-adherence to decolonisation therapy included discharge prior to an MRSA-positive result, discharge prior to commencement of decolonisation therapy or the patient declining the intervention.
Figure 3.
Adherence to contact precautions, decolonisation and isolation measures for meticillin-resistant Staphylococcus aureus (MRSA) carriers, this figure shows the distribution of monthly adherence to infection control measures for randomly audited patients known to be colonised or infected with MRSA for each study arm. The top panel (A) shows adherence to implementation of contact precautions, decolonisation therapy and isolation in single rooms. The middle panel (B) shows the presence of signage of MRSA status on the patients’ room, bed or nursing chart. The bottom panel (C) shows the availability of gowns, gloves and alcohol-based handrub in or at the entrance of the room. The horizontal line in each box represents the median, the box represents the interquartile range and the vertical lines represent the minimum and maximum values.
Screening occurred to a lesser extent in the other study arms (figure 2B). About 10% of admissions to wards in the enhanced HH arm were screened throughout the study. In wards in the combined arm, screening increased from 9.2% to 22.3%, then 36.9% during baseline, intervention and washout phases, respectively. In this arm, adherence to contact precautions was high throughout the study (93.0–99.6%), but only 32.9% of patients with MRSA at baseline and 35.9% of patients during the intervention phase received decolonisation therapy (figure 3).
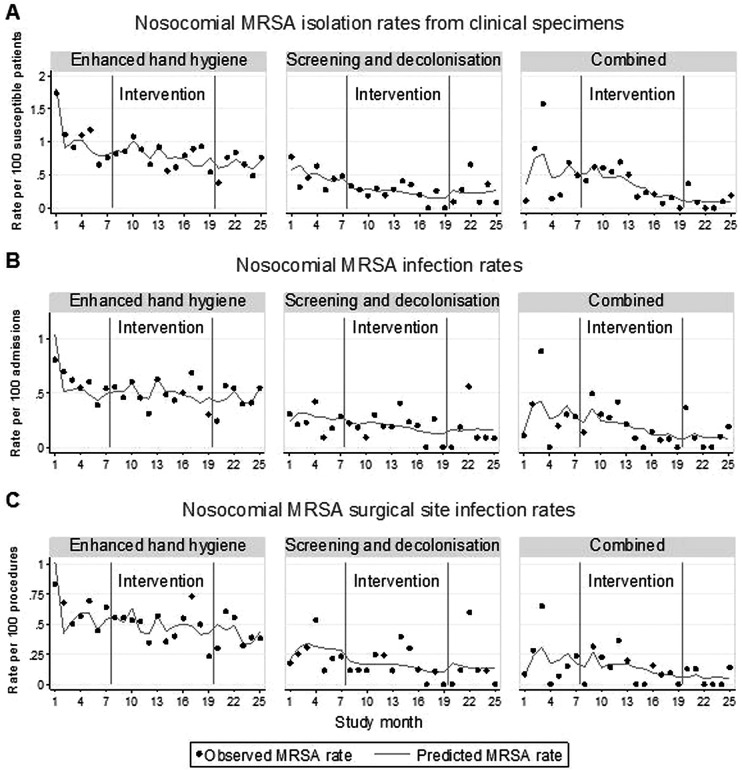
Nosocomial MRSA isolation rate from clinical cultures
Crude MRSA isolation rates from clinical cultures decreased in all study arms during the intervention phase (enhanced HH arm: from 0.99 to 0.80; screening and decolonisation arm: from 0.47 to 0.23; combined arm: from 0.55 to 0.36; p=0.04; per 100 susceptible patients; table 4). After adjusting for clustering and potential confounders with multilevel segmented Poisson regression (table 5 and online supplementary table A4 for full model), the start of HH promotion in the enhanced HH arm was associated with an immediate non-significant increase in nosocomial MRSA isolation rate (aIRR 1.44, 95% CI 0.96 to 2.15) with no change in the trend in rates over time. In clean surgery wards, HH promotion was associated with a non-significant decreasing monthly MRSA isolation rate (aIRR 0.89, 95% CI 0.78 to 1.01; table 6 and see online supplementary table A5 for full model).
Table 4.
Crude nosocomial MRSA incidence rates and incidence rate ratios by study arm for each study period*
| Outcome | Baseline phase | Intervention phase | Washout phase | Crude IRR (95% CI) for intervention vs baseline phases | Crude IRR (95% CI) for washout vs intervention phases |
|---|---|---|---|---|---|
| MRSA isolation rate from clinical cultures (number per 100 susceptible patients) | |||||
| Enhanced hand hygiene | 0.99 (181/183.47) | 0.80 (279/349.50) | 0.65 (106/163.83) | 0.81 (0.67 to 0.98) | 0.81 (0.65 to 1.01) |
| Screening and decolonisation | 0.47 (31/66.61) | 0.23 (28/122.56) | 0.26 (17/66.04) | 0.49 (0.29 to 0.82) | 1.13 (0.62 to 2.06) |
| Combined | 0.55 (47/85.35) | 0.36 (60/165.23) | 0.13 (8/63.04) | 0.66 (0.45 to 0.97) | 0.35 (0.17 to 0.73) |
| MRSA infection rate (number per 100 admissions) | |||||
| Enhanced hand hygiene | 0.58 (106/183.79) | 0.50 (175/349.96) | 0.45 (74/164.13) | 0.87 (0.68 to 1.10) | 0.90 (0.69 to 1.18) |
| Screening and decolonisation | 0.24 (16/66.92) | 0.19 (23/122.79) | 0.17 (11/66.15) | 0.78 (0.41 to 1.48) | 0.89 (0.43 to 1.82) |
| Combined | 0.29 (25/85.37) | 0.19 (32/165.35) | 0.13 (8/63.04) | 0.66 (0.39 to 1.12) | 0.66 (0.30 to 1.42) |
| MRSA surgical site infection rate (number per 100 surgical procedures) | |||||
| Enhanced hand hygiene | 0.60 (79/132.27) | 0.49 (123/250.03) | 0.42 (54/127.06) | 0.82 (0.62 to 1.09) | 0.86 (0.63 to 1.19) |
| Screening and decolonisation | 0.26 (14/54.00) | 0.15 (15/99.63) | 0.16 (8/50.74) | 0.58 (0.28 to 1.20) | 1.05 (0.44 to 2.47) |
| Combined | 0.20 (18/91.41) | 0.14 (21/147.81) | 0.07 (3/43.43) | 0.72 (0.38 to 1.35) | 0.49 (0.15 to 1.63) |
| MRSA bloodstream infection rate (number per 10 000 patient-days) | |||||
| Enhanced hand hygiene | 0.93 (14/15.0757) | 0.56 (16/28.6667) | 0.44 (6/13.5745) | 0.60 (0.29 to 1.23) | 0.79 (0.31 to 2.02) |
| Screening and decolonisation | 0.17 (1/5.7754) | 0.18 (2/11.2971) | 0.17 (1/5.8473) | 1.02 (0.09 to 11.28) | 0.97 (0.09 to 10.65) |
| Combined | 0.18 (1/5.5524) | 0.00 (0/9.7337) | 0.00 (0/5.4901) | – | – |
*MRSA was defined as nosocomial if it was isolated from specimens collected more than 48 h after admission or within 30 days (or 12 months for infections of prosthetic devices) after discharge from the surgical ward.
IRR, incidence rate ratio; MRSA, meticillin-resistant Staphylococcus aureus.
Table 5.
Multiple segmented multilevel Poisson regression models showing adjusted incidence rate ratios for changes in level and trend of nosocomial MRSA rates*
| Variable | MRSA clinical isolates (per 100 susceptible patients) | Total MRSA infections (per 100 admissions) | MRSA surgical site infections (per 100 procedures) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| aIRR | 95% CI | p Value | aIRR | 95% CI | p Value | aIRR | 95% CI | p Value | |
| Baseline phase | |||||||||
| Trend | 0.97 | 0.89 to 1.06 | 0.55 | 1.00 | 0.90 to 1.11 | 0.98 | 1.02 | 0.90 to 1.16 | 0.75 |
| Intervention phase | |||||||||
| Change in level | |||||||||
| Enhanced hand hygiene | 1.44 | 0.96 to 2.15 | 0.076 | 1.28 | 0.79 to 2.06 | 0.31 | 1.25 | 0.70 to 2.23 | 0.45 |
| Screening and decolonisation | 0.87 | 0.49 to 1.57 | 0.65 | 0.97 | 0.49 to 1.92 | 0.94 | 0.79 | 0.35 to 1.79 | 0.58 |
| Combined | 1.63 | 0.96 to 2.75 | 0.070 | 1.17 | 0.62 to 2.20 | 0.63 | 1.33 | 0.59 to 3.00 | 0.49 |
| Change in trend | |||||||||
| Enhanced hand hygiene | 0.99 | 0.91 to 1.09 | 0.88 | 0.99 | 0.89 to 1.10 | 0.84 | 0.98 | 0.86 to 1.12 | 0.75 |
| Screening and decolonisation | 0.94 | 0.85 to 1.05 | 0.26 | 0.93 | 0.82 to 1.05 | 0.27 | 0.90 | 0.78 to 1.04 | 0.162 |
| Combined | 0.88 | 0.79 to 0.98 | 0.016 | 0.90 | 0.80 to 1.02 | 0.096 | 0.86 | 0.74 to 1.01 | 0.059 |
| Washout phase | |||||||||
| Change in level | 1.90 | 0.91 to 3.95 | 0.087 | 1.52 | 0.66 to 3.51 | 0.32 | 1.90 | 0.69 to 5.27 | 0.21 |
| Change in trend | 1.02 | 0.91 to 1.15 | 0.74 | 1.00 | 0.88 to 1.15 | 0.95 | 0.95 | 0.80 to 1.12 | 0.53 |
*MRSA was defined as nosocomial if it was isolated from specimens collected more than 48 h after admission or within 30 days (or 12 months for infections of prosthetic devices) after discharge from the surgical ward. The model used a lagged dependent variable to account for autocorrelation and adjusted for staffing (patient-to-nurse ratios), seasonal effects, type of surgical ward and baseline hand hygiene compliance rates. The model also accounted for overdispersion. Random effects for intercepts at the hospital and ward levels and random baseline trends at the hospital level were all significant and baseline trends were negatively correlated with intercepts (ie, hospitals with higher baseline MRSA rates tended to have larger decreases in baseline rates).
aIRR, adjusted incidence rate ratio; MRSA, meticillin-resistant Staphylococcus aureus.
Table 6.
Multiple segmented multilevel Poisson regression models showing changes in nosocomial MRSA rates for the subgroup analysis of clean surgery only*
| Variable | MRSA clinical isolates (per 100 susceptible patients) | Total MRSA infections (per 100 admissions) | MRSA surgical site infections (per 100 procedures) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| aIRR | 95% CI | p Value | aIRR | 95% CI | p Value | aIRR | 95% CI | p Value | |
| Baseline phase | |||||||||
| Trend | 1.05 | 0.93 to 1.18 | 0.41 | 1.10 | 0.94 to 1.28 | 0.23 | 1.11 | 0.93 to 1.33 | 0.26 |
| Intervention phase | |||||||||
| Change in level | |||||||||
| Enhanced hand hygiene | 1.31 | 0.75 to 2.30 | 0.34 | 1.06 | 0.52 to 2.16 | 0.88 | 1.09 | 0.47 to 2.53 | 0.83 |
| Screening and decolonisation | 0.87 | 0.41 to 1.85 | 0.71 | 1.03 | 0.39 to 2.69 | 0.96 | 0.92 | 0.29 to 2.92 | 0.89 |
| Combined | 1.79 | 0.86 to 3.74 | 0.121 | 1.15 | 0.44 to 2.96 | 0.78 | 1.21 | 0.39 to 3.73 | 0.75 |
| Change in trend | |||||||||
| Enhanced hand hygiene | 0.89 | 0.78 to 1.01 | 0.063 | 0.88 | 0.75 to 1.04 | 0.127 | 0.89 | 0.73 to 1.07 | 0.21 |
| Screening and decolonisation | 0.85 | 0.74 to 0.97 | 0.019 | 0.83 | 0.69 to 0.99 | 0.041 | 0.81 | 0.66 to 1.00 | 0.054 |
| Combined | 0.82 | 0.71 to 0.95 | 0.007 | 0.84 | 0.70 to 1.00 | 0.055 | 0.84 | 0.68 to 1.03 | 0.095 |
| Washout phase | |||||||||
| Change in level | 3.01 | 1.05 to 8.63 | 0.041 | 2.21 | 0.61 to 8.04 | 0.23 | 2.59 | 0.59 to 11.46 | 0.21 |
| Change in trend | 0.96 | 0.81 to 1.15 | 0.67 | 0.91 | 0.73 to 1.12 | 0.37 | 0.86 | 0.67 to 1.09 | 0.21 |
aIRR, adjusted incidence rate ratio; MRSA, meticillin-resistant Staphylococcus aureus.
*MRSA was defined as nosocomial if it was isolated from specimens collected more than 48 h after admission or within 30 days (or 12 months for infections of prosthetic devices) after discharge from the surgical ward. Clean surgery included cardiothoracic, neuro, orthopaedic, plastic and vascular surgery subspecialties. The model used a lagged dependent variable to account for autocorrelation and adjusted for staffing (patient-to-nurse ratios), seasonal effects, type of surgical ward and baseline hand hygiene compliance rates. The model also accounted for overdispersion. Random effects for intercepts at the hospital and ward levels and random baseline trends at the hospital level were all significant and baseline trends were negatively correlated with intercepts (ie, hospitals with higher baseline MRSA rates tended to have larger decreases in baseline rates).
In the screening and decolonisation arm, there were no significant changes in MRSA isolation rates. However, in clean surgery, this intervention was associated with a reduction in MRSA clinical cultures of 15% per month (aIRR 0.85, 95% CI 0.74 to 0.97).
In the combined arm (wards that used a combination of HH promotion with targeted screening), there was a significant decreasing trend in MRSA isolation rate of 12% per month overall (aIRR 0.88, 95% CI 0.79 to 0.98) and 18% per month in clean surgery (aIRR 0.82, 95% CI 0.71 to 0.95). Observed and model-predicted MRSA isolation rates from clinical cultures are illustrated in figure 4A and online supplementary figure A1.
Figure 4.
Nosocomial meticillin-resistant Staphylococcus aureus (MRSA) rates by study arm, the top panel (A) shows the nosocomial MRSA isolaton rates from clinical specimens. The middle panel (B) shows the nosocomial MRSA infection rates. The bottom panel (C) shows the nosocomial MRSA surgical site infection rates. The solid dots represent the observed MRSA rates while the lines represent the predicted MRSA rates based on the regression models.
During the washout phase, MRSA clinical culture isolation rates increased. A post hoc analysis of the washout phase results by study arm showed that the increase in MRSA rates was due to an abrupt increase in the level of MRSA clinical cultures on cessation of the intervention phase in all study arms, but particularly with the conclusion of the intensive HH promotion campaign in the combined arm (see online supplementary table A6).
Nosocomial MRSA infection rates
There were 470 nosocomial MRSA infections in total (335 (71.3%) surgical site, 41 (8.7%) bloodstream and 94 (20.0%) other infections). Crude infection rates decreased over time in all study arms (table 4). After multivariable analysis (table 5, figure 4B and online supplementary table A4), enhanced HH promotion alone was not associated with changes in MRSA infection rates. The screening/decolonisation and combined interventions resulted in non-significant decreasing trends in total MRSA infection (screening and decolonisation arm: aIRR 0.93, 95% CI 0.82 to 1.05; combined arm: aIRR 0.90, 95% CI 0.80 to 1.02) and surgical site infection rates (table 5, figure 4C and online supplementary table A4).
In clean surgery, the screening and decolonisation strategy was associated with significant reductions in total MRSA infection rate of 17% per month (aIRR 0.83, 95% CI 0.69 to 0.99) and MRSA surgical site infection rate of 19% per month (aIRR 0.81, 95% CI 0.66 to 1.00; table 6 and online supplementary table A5).
Exploratory analysis to directly assess implemented interventions
The exploratory analysis did not show any significant effects of HH promotion on nosocomial MRSA isolation rates (see online supplementary table A7). The intensity of admission screening was associated with a decreasing trend in monthly MRSA isolation rate from clinical cultures (aIRR 0.91/month with 100% compliance with screening, 95% CI 0.85 to 0.98). A similar effect was seen in the trend in MRSA infection rate (aIRR 0.92, 95% CI 0.85 to 0.99).
Discussion
We found that implementation of individual interventions in surgical wards, with either an enhanced HH promotion strategy or universal MRSA screening with contact precautions and decolonisation of MRSA carriers, was not effective in reducing MRSA rates. However, using a combination of HH promotion and targeted screening was associated with a reduction in MRSA isolation rate from clinical cultures of 12% per month. When the interventions were specifically evaluated in the subgroup of clean surgery wards, the screening and decolonisation strategy was most effective. In these wards, this intervention was associated with significant reductions in both MRSA clinical culture isolation rate of 15% per month and MRSA infection rate of 17% per month.
This study is unique in that it directly compared strategies individually and in combination using a large, prospective, controlled design.10 In addition, we used a planned exploratory analysis to separate out the individual effects of the HH and MRSA screening strategies. Interventions were implemented and assessed under operational conditions in 10 heterogeneous hospitals across Europe and Israel with widely varying infection control practices, staffing, infrastructure and MRSA epidemiology, increasing the generalisability of our findings. This study has been reported using standard reporting guidelines that are designed to maximise transparency and scientific rigour of intervention studies of healthcare associated infection.24
Our analysis, which adjusted for confounders, seasonal effects and baseline MRSA trends, found no evidence that enhanced HH promotion was effective. MRSA rates are declining in many countries.25 Failing to account for this would overestimate intervention effects. Overall baseline HH compliance was 49% in study wards that used the HH intervention. In settings where compliance is already above about 50%, modelling studies suggest that further increases in compliance will have rapidly diminishing returns for reducing MRSA transmission.26 In facilities with lower HH compliance or higher MRSA rates, this intervention may be more effective than we were able to demonstrate. In addition, HH campaigns involve education and behavioural change and are therefore unlikely to have a short-term effect. Other studies have shown that they may be beneficial if activity is sustained over years.27 28 Although we did not detect any intervention effects of the HH promotion strategy, cessation of this intervention was associated with an increase in MRSA rates in our study, suggesting that discontinuing activities to optimise HH practices may be detrimental.
Active MRSA surveillance identifies the reservoir of asymptomatic carriers, enabling early implementation of contact precautions and decolonisation, which can reduce transmission.29 30 With universal screening, we found that 90% of patients with MRSA would have been missed using clinical cultures alone. However, our results suggest that rather than universal screening of all surgical patients admitted for more than 24 h, selective screening in clean surgery wards or a combination of HH promotion and targeted screening of high-risk patients may be more effective strategies. The relative burden of Gram-positive infections is greater in clean compared to clean-contaminated surgery where other pathogens, including bowel flora, may be more important.23 31 Thus, it is biologically plausible that MRSA-specific interventions would potentially have a greater impact in clean surgery. Indeed, intranasal mupirocin has been shown to reduce surgical site infections in cardiothoracic and orthopaedic surgery, but is less effective in general surgery.23 The commencement of such decolonisation regimens prior to surgical procedures, which can be facilitated by rapid detection of S aureus carriage with molecular tests, is likely a key factor in the success of this approach.32 The use of molecular tests in the latter part of the intervention phase in our study could have significantly contributed to the reduction in MRSA rates seen over the period of the intervention phase, particularly in clean surgery wards.
The exploratory analysis suggests that the screening intensity, rather than HH promotion, explained the intervention effects. It is curious, then, that universal screening did not perform better than HH promotion combined with targeted screening. A significant reduction in MRSA clinical cultures was seen with the combined strategy despite the enrolment of only two hospitals in this study arm. This suggests that the effect of the combined intervention was robust. It is certainly biologically plausible that using two interventions that aim to control MRSA in different ways would be more effective than use of single interventions. Although the universal screening arm enrolled four hospitals, low baseline MRSA rates in this arm and shortage of isolation rooms for the larger number of identified MRSA carriers may have reduced our ability to detect significant effects. In addition, targeted screening may have been more effective if it had identified ‘superspreaders’,33 facilitating more efficient use of resources including limited single rooms. Modelling studies also demonstrate that targeted screening has the advantage of increased cost-effectiveness compared to universal screening for reducing healthcare associated MRSA infections.34 35
This study adds to the conflicting literature regarding active surveillance cultures. Our results apply to surgical settings. The risk of MRSA infection in other wards, such as intensive care units or general medical wards, would differ due to variation in patient comorbidities and exposure to invasive procedures or antibiotics. It is also important to note that previous studies have used a variety of interventions in combination with screening. In some cases, the use of pre-emptive isolation in both study arms36 or lack of decolonisation strategies,6 may have led to effect sizes that studies had insufficient power to detect. Comparison of rapid screening to conventional rather than no screening,36 differences in screening methods,10 variation in MRSA strains37 or limitations in study design and analyses10 11 are other potential explanations for the conflicting results of screening studies.
There are some limitations to this study. Research personnel assessing HH, screening, decolonisation, contact precautions and isolation practices were not blinded to study assignment as they were responsible for implementing the interventions. Decisions to take culture samples were initiated by treating physicians, not research personnel and standardised definitions for infections were used, reducing the likelihood of bias in the measurement of the study outcomes by unblinded assessors. Although allocation of interventions was not randomised, we accounted for differences in hospitals by adjusting for potential confounders and comparing outcomes between baseline and intervention phases within the same study arm. We used MRSA-positive clinical cultures as our primary outcome. Although this measure does not distinguish between colonisation and infection, it can be a more sensitive marker for changes in MRSA disease rates.38 We found the results for MRSA clinical cultures similar to those for infections, suggesting that this measure was clinically relevant. Patient level data, such as age, comorbidities and length of stay and antibiotic use were not measured for this study. However, results were similar when each centre was excluded in turn from the analysis (data not shown) so changes in factors in individual centres are unlikely to have had a major effect on study outcomes.
Conclusion
In surgical wards with relatively low MRSA prevalence, a combination of enhanced standard infection control measures emphasising HH promotion and MRSA-specific (targeted screening of high-risk patients) approaches was required to reduce MRSA rates. Implementation of single interventions was not effective, except in clean surgery wards where MRSA screening coupled with contact precautions and decolonisation of identified MRSA carriers was associated with significant reductions in MRSA clinical culture and infection rates. These findings are likely generalisable to other settings with varying infection control practices. In addition, the WHO multimodal HH promotion strategy15 implemented in this study is already being used in many parts of the world. Therefore our study, which provides evidence that this intervention alone is insufficient to reduce MRSA rates, potentially has widespread implications for best clinical practice recommendations and policy change. Further research regarding the cost-effectiveness of these interventions will allow better utilisation of limited healthcare resources.
Supplementary Material
Acknowledgments
The authors wish to thank Christine Lammens from the Central Laboratory, Antwerp, Belgium for assistance with screening implementation; and BD Diagnostics, Belgium and Cepheid, Belgium for supplying MRSA screening assays at a reduced price as well as logistic support. In addition, the authors would also like to thank other contributors to the study as follows: Microbiology Departments at the participating centres: John Adam, Francesco Bernieri, Jina Bouzala, Ivana Ćirković, María Ángeles Dominguez Luzón, Paolo Mangoni, Jean Claude Nguyen, Nick Parsons, Gesuele Renzi, Zmira Samra, Jacques Schrenzel, Jordi Vila, Neil Young; Surgical Departments at the participating centres: M Isabel Baños, Vittorio Baratta, Giuseppe Galli, Sebastián García, Alessandro Luzzati, Mario Martinotti, Carlos Mestres, Teresa Pascual, Montse Venturas; University of Geneva Hospitals and WHO, World Alliance for Patient Safety, Geneva, Switzerland: Didier Pittet, Marie-Noelle Chraiti, Hugo Sax, Benedetta Allegranzi; University Medical Center, Utrecht, the Netherlands: Frank Leus, Joost Schotsman, Jildou Zwerver; National Medicines Institute, Warsaw, Poland: Waleria Hryniewicz, Joanna Empel; University Val-de-Marne, Créteil, France: Isabelle Durand-Zaleski, Stéphane Bahrami, Michael Padget. The MOSAR WP4 trial investigators: the authors would like to thank the following investigators and research staff from the MOSAR WP4 group who contributed data to the clinical trial. University of Geneva Hospitals, Geneva, Switzerland: Américo Agostinho; Hospital Universitari de Bellvitge, Barcelona, Spain: Marta Banque Navarro, Josep Maria Ramon-Torrell; Groupe Hospitalier Paris Saint-Joseph, Paris, France: Julien Fournier; Istituti Ospitalieri di Cremona, Cremona, Italy: Silvia Garilli; Rabin Medical Center, Beilinson Hospital, Petah-Tikva, Israel: Rita Hollinger, Hefziba Madar; Clinical Center of Serbia, Belgrade, Serbia: Natasa Mazic, Vesna Mioljevic; Ninewells Hospital, Dundee, Scotland: Joanne McEwen, Gilian Stevenson; Hospital Clínic de Barcelona, Barcelona, Spain: Encarna Moreno, Raquel Piñer; Laiko General Hospital, Athens, Greece: Mina Psichogiou; Universitätsklinikum Aachen, Aachen, Germany: Thomas Schwanz, Birgit Waitschies.
Footnotes
Contributors: SH and CBB conceived and designed the experiments. ASL analysed the data, enrolled patients and wrote the first draft of the manuscript. BSC provided statistical support and analysed the data. SMK and HG contributed in arranging reagents/materials. AC, GLD, CF, BC, SL, JAM, CMA, AP, GP and BR enrolled patients. All the authors read and met the ICMJE criteria for authorship, contributed to the writing of the manuscript and agreed with the manuscript results and conclusions.
Funding: This work was supported by the European Commission under the Life Science Health Priority of the 6th Framework Program (MOSAR network contract LSHP-CT-2007-037941).
Competing interests: SH is a member of the speakers’ bureau for bioMérieux and Pfizer and the scientific advisory board of Destiny Pharma, DaVolterra and bioMérieux. He has received financial support for MRSA research activities from Geneva University Hospitals, B. Braun and Pfizer. AP is a member of the speakers’ bureau for Cubist and has received financial support for MRSA research activities from BD. There were no other financial or non-financial relationships or interests that may be relevant to the submitted work.
Ethics approval: Institutional Review Boards at each participating study centre.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data sharing statement: The dataset is available from the corresponding author at stephan.harbarth@hcuge.ch.
References
- 1.WHO Report on the burden of endemic health care-associated infection worldwide. http://whqlibdoc.who.int/publications/2011/9789241501507_eng.pdf (accessed 24 Apr 2013).
- 2.Cosgrove SE, Qi Y, Kaye KS, et al. The impact of methicillin resistance in Staphylococcus aureus bacteremia on patient outcomes: mortality, length of stay, and hospital charges. Infect Control Hosp Epidemiol 2005;26:166–74 [DOI] [PubMed] [Google Scholar]
- 3.Weber SG, Huang SS, Oriola S, et al. Legislative mandates for use of active surveillance cultures to screen for methicillin-resistant Staphylococcus aureus and vancomycin-resistant enterococci: position statement from the joint SHEA and APIC task force. Am J Infect Control 2007;35:73–85 [DOI] [PubMed] [Google Scholar]
- 4.UK Department of Health MRSA Screening—Operational Guidance 2. http://www.dh.gov.uk/en/Publicationsandstatistics/Lettersandcirculars/Dearcolleagueletters/DH_092844 (accessed 24 Apr 2013).
- 5.Robicsek A, Beaumont JL, Paule SM, et al. Universal surveillance for methicillin-resistant Staphylococcus aureus in 3 affiliated hospitals. Ann Intern Med 2008;148:409–18 [DOI] [PubMed] [Google Scholar]
- 6.Huskins WC, Huckabee CM, O'Grady NP, et al. Intervention to reduce transmission of resistant bacteria in intensive care. N Engl J Med 2011;364:1407–18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Harbarth S, Fankhauser C, Schrenzel J, et al. Universal screening for methicillin-resistant Staphylococcus aureus at hospital admission and nosocomial infection in surgical patients. JAMA 2008;299:1149–57 [DOI] [PubMed] [Google Scholar]
- 8.Edmond MB, Ober JF, Bearman G. Active surveillance cultures are not required to control MRSA infections in the critical care setting. Am J Infect Control 2008;36:461–3 [DOI] [PubMed] [Google Scholar]
- 9.Wenzel RP, Bearman G, Edmond MB. Screening for MRSA: a flawed hospital infection control intervention. Infect Control Hosp Epidemiol 2008;29:1012–18 [DOI] [PubMed] [Google Scholar]
- 10.Tacconelli E, De Angelis G, De Waure C, et al. Rapid screening tests for meticillin-resistant Staphylococcus aureus at hospital admission: systematic review and meta-analysis. Lancet Infect Dis 2009;9:546–54 [DOI] [PubMed] [Google Scholar]
- 11.Loveday HP, Pellowe CM, Jones SR, et al. A systematic review of the evidence for interventions for the prevention and control of meticillin-resistant Staphylococcus aureus (1996–2004): report to the Joint MRSA Working Party (subgroup A). J Hosp Infect 2006;63(Suppl 1):S45–70 [DOI] [PubMed] [Google Scholar]
- 12.Jain R, Kralovic SM, Evans ME, et al. Veterans affairs initiative to prevent methicillin-resistant Staphylococcus aureus infections. N Engl J Med 2011;364:1419–30 [DOI] [PubMed] [Google Scholar]
- 13.Farr BM, Jarvis WR. Searching many guidelines for how best to control methicillin-resistant Staphylococcus aureus healthcare-associated spread and infection. Infect Control Hosp Epidemiol 2009;30:808–9 [DOI] [PubMed] [Google Scholar]
- 14.Nijssen S, Bonten MJ, Weinstein RA. Are active microbiological surveillance and subsequent isolation needed to prevent the spread of methicillin-resistant Staphylococcus aureus? Clin Infect Dis 2005;40:405–9 [DOI] [PubMed] [Google Scholar]
- 15.WHO WHO Guidelines on hand hygiene in health care. World alliance for patient safety. Geneva: WHO Press Geneva, 2009 [Google Scholar]
- 16.Horan TC, Andrus M, Dudeck MA. CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control 2008;36:309–32 [DOI] [PubMed] [Google Scholar]
- 17.Van Heirstraeten L, Cortinas Abrahantes J, Lammens C, et al. Impact of a short period of pre-enrichment on detection and bacterial loads of methicillin-resistant Staphylococcus aureus from screening specimens. J Clin Microbiol 2009;47:3326–8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Malhotra-Kumar S, Haccuria K, Michiels M, et al. Current trends in rapid diagnostics for methicillin-resistant Staphylococcus aureus and glycopeptide-resistant enterococcus species. J Clin Microbiol 2008;46:1577–87 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gazin M, Lee A, Derde L, et al. Culture-based detection of methicillin-resistant Staphylococcus aureus by a network of European laboratories: an external quality assessment study. Eur J Clin Microbiol Infect Dis 2012;31:1765–70 [DOI] [PubMed] [Google Scholar]
- 20.Lee A, Chalfine A, Daikos GL, et al. Hand hygiene practices and adherence determinants in surgical wards across Europe and Israel: a multicenter observational study. Am J Infect Control 2011;39:517–20 [DOI] [PubMed] [Google Scholar]
- 21.Harbarth S, Pittet D, Grady L, et al. Interventional study to evaluate the impact of an alcohol-based hand gel in improving hand hygiene compliance. Pediatr Infect Dis J 2002;21:489–95 [DOI] [PubMed] [Google Scholar]
- 22.Shardell M, Harris AD, El-Kamary SS, et al. Statistical analysis and application of quasi experiments to antimicrobial resistance intervention studies. Clin Infect Dis 2007;45:901–7 [DOI] [PubMed] [Google Scholar]
- 23.Kallen AJ, Wilson CT, Larson RJ. Perioperative intranasal mupirocin for the prevention of surgical-site infections: systematic review of the literature and meta-analysis. Infect Control Hosp Epidemiol 2005;26:916–22 [DOI] [PubMed] [Google Scholar]
- 24.Stone SP, Cooper BS, Kibbler CC, et al. The ORION statement: guidelines for transparent reporting of outbreak reports and intervention studies of nosocomial infection. Lancet Infect Dis 2007;7:282–8 [DOI] [PubMed] [Google Scholar]
- 25.Struelens MJ, Monnet DL. Prevention of methicillin-resistant Staphylococcus aureus infection: is Europe winning the fight? Infect Control Hosp Epidemiol 2010;31(Suppl 1):S42–4 [DOI] [PubMed] [Google Scholar]
- 26.Cooper BS, Medley GF, Scott GM. Preliminary analysis of the transmission dynamics of nosocomial infections: stochastic and management effects. J Hosp Infect 1999;43:131–47 [DOI] [PubMed] [Google Scholar]
- 27.Pittet D, Hugonnet S, Harbarth S, et al. Effectiveness of a hospital-wide programme to improve compliance with hand hygiene. Infection Control Programme. Lancet 2000;356:1307–12 [DOI] [PubMed] [Google Scholar]
- 28.Stone SP, Fuller C, Savage J, et al. Evaluation of the national Cleanyourhands campaign to reduce Staphylococcus aureus bacteraemia and Clostridium difficile infection in hospitals in England and Wales by improved hand hygiene: four year, prospective, ecological, interrupted time series study. BMJ 2012;344:e3005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lee AS, Huttner B, Harbarth S. Control of methicillin-resistant Staphylococcus aureus. Infect Dis Clin North Am 2011;25:155–79 [DOI] [PubMed] [Google Scholar]
- 30.Ammerlaan HS, Kluytmans JA, Wertheim HF, et al. Eradication of methicillin-resistant Staphylococcus aureus carriage: a systematic review. Clin Infect Dis 2009;48:922–30 [DOI] [PubMed] [Google Scholar]
- 31.Huttner B, Robicsek AA, Gervaz P, et al. Epidemiology of methicillin-resistant Staphylococcus aureus carriage and MRSA surgical site infections in patients undergoing colorectal surgery: a cohort study in two centers. Surg Infect (Larchmt) 2012;13:401–5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bode LG, Kluytmans JA, Wertheim HF, et al. Preventing surgical-site infections in nasal carriers of Staphylococcus aureus. N Engl J Med 2010;362:9–17 [DOI] [PubMed] [Google Scholar]
- 33.Lloyd-Smith JO, Schreiber SJ, Kopp PE, et al. Superspreading and the effect of individual variation on disease emergence. Nature 2005;438:355–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hubben G, Bootsma M, Luteijn M, et al. Modelling the costs and effects of selective and universal hospital admission screening for methicillin-resistant Staphylococcus aureus. PLoS ONE 2011;6:e14783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Infectious Disease Research Network (IDRN) Report on MRSA Screening Audit. http://idrn.org/audit.php (accessed 28 Jul 2013).
- 36.Jeyaratnam D, Whitty CJ, Phillips K, et al. Impact of rapid screening tests on acquisition of meticillin resistant Staphylococcus aureus: cluster randomised crossover trial. BMJ 2008;336:927–30 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Cooper BS, Kypraios T, Batra R, et al. Quantifying type-specific reproduction numbers for nosocomial pathogens: evidence for heightened transmission of an Asian sequence type 239 MRSA clone. PLoS Comput Biol 2012;8:e1002454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Walker S, Peto TE, O'Connor L, et al. Are there better methods of monitoring MRSA control than bacteraemia surveillance? An observational database study. PLoS ONE 2008;3:e2378. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.