Abstract
Immunization with a pandemic influenza A H1N1 2009 was recommended for HIV-infected patients. However, there is limited information concerning the impact of immunization with this vaccine on immune activation and HIV viral replication. In this study, 45 HIV-infected children and adolescents receiving antiretroviral therapy were immunized with a 2-dose series of nonadjuvated monovalent influenza A H1N1 2009 vaccine upon enrollment and approximately 1 month later. Immunogenicity was determined by haemagglutination inhibition assay. The level of immune activation was determined by identification of CD38 and HLA-DR on CD8+ T cells. Patients were divided into 2 groups which include patients who had an undetectable HIV viral load (HIV detectable group) and patients who show virological failure (HIV nondetectable group). The results showed seroconversion rate of 55.2% in HIV nondetectable group, whereas 31.3% was found in HIV detectable group. Both groups of patients showed no major increase in immune activation after immunization. Interestingly, a decrease in the frequency of CD8+ T cells that coexpressed CD38 and HLA-DR was observed after immunization in both groups of patients. We suggested that immunization with influenza A H1N1 2009 vaccine can induce immune response to the pandemic virus without major impact on HIV viral replication and immune activation.
1. Introduction
A recent pandemic of an influenza A H1N1 2009 caused a wide spread impact on global health [1]. It is becoming an important issue that an urgent production of influenza vaccine in response to a new pandemic is required in order to control the infection. In general, influenza vaccination is recommended for protection in a group of people at a high risk of susceptibility to infection. HIV-infected patients were normally recommended due to severe complications in this group of patients [2]. However, the potential benefit of vaccination in HIV-infected patients remains questionable due to an observation of suboptimal immune response [3] and concerns about an increasing HIV viral replication after vaccination [4].
A potential mechanism concerning an influence of immune activation on HIV viral replication suggested that the induction of immune activation during the course of vaccination might be able to induce an active HIV viral replication. Many studies demonstrated the alteration of CD4+ T cell count and the level of HIV viral load which are the markers for HIV disease progression. The studies in HIV-infected patients who immunized with influenza vaccine showed that there was a minor change in the level of HIV-1 proviral DNA and significantly increased in HIV-1 RNA when compared to prevaccination level [5]. Similar to this study, Staprans and his colleagues used the high sensitivity technique to detect HIV-1 RNA and found that plasma viremia was increased within 1-2 weeks after immunization and returned to the prevaccination level within 4 weeks after immunization. However, plasma viremia in a few patients did not return to baseline or even showed an increased level during the study [6]. In the study reported by Ramilo et al., which studied HIV-infected children, it showed that 5 of 15 HIV-1-infected children who received influenza vaccination showed an increase in HIV viral load up to 150% after immunization and 4 of 15 patients showed that the HIV viral load returns to baseline level within 6–8 weeks [7]. Furthermore, a study that determines the impact of influenza vaccine on HIV viral load and %CD4 in HIV-infected patients demonstrated that immunization with influenza vaccine has high impact on HIV viral load in HIV-infected patient who did not take antiretroviral therapy. Similarly, a decrease in %CD4 after immunization was also reported [8]. However, some studies indicated that immune activation from influenza vaccination did not significantly alter the HIV viral load and CD4+ T cell count [9].
Beside the detection of HIV viral load, there was a possibility of using immune activation as a marker of disease progression in HIV-infected patients. Previous observation showed that both CD8+ T lymphocyte number and percentage were increased, and their phenotype also changed after acute HIV infection [10]. Furthermore, an increased activation marker level including CD25, HLA-DR, CD38, and CD57 on CD8+ T lymphocytes was also observed [11–14]. More importantly, the increase of CD38 and HLA-DR on CD8+ T lymphocytes is found to correlate with HIV viral load and is inversely correlated with CD4+ T cell count [14–18].
In this study, the correlation between immunization, HIV viral replication, and immune activation was examined in order to give an understanding on a relationship between these parameters in HIV-infected children receiving antiretroviral therapy.
2. Materials and Methods
2.1. Study Population
Forty-five HIV-infected children at 6 months to 18 years of age who had been followed at Faculty of Medicine Siriraj Hospital, Mahidol University, Bangkok, Thailand, were recruited. Out of 45, 22 (48.9%) are male. All patients received anti-retroviral therapy at least 1 year before the beginning of the study and throughout the study period. Patients who received immunosuppressive therapy for the previous 6 months or blood products in the previous 3 months were excluded from this study. None of these patients received other licensed live vaccines within 4 weeks or inactivated vaccines within 2 weeks of study entry.
2.2. Vaccine
An influenza A H1N1 2009 vaccine was the monovalent, nonadjuvated influenza A H1N1 2009 vaccine (Panenza, Sanofi Pasteur, Val de Reuil, France). Each dose (0.5 mL) contained 15 μg of haemagglutinin antigen of influenza A/California/07/2009 (H1N1) v-like virus.
2.3. Study Procedure
The vaccine was provided for 2 doses, approximately 1 month apart. The children 6 months to 3 years of age received half dose (0.25 mL) of the vaccine. The first dose of vaccine was given intramuscularly on enrollment day and the second dose was given 28 days after the first dose. Blood samples were drawn from the patients at enrollment (baseline), 28 days after the first dose of immunization, and 28 day after the second dose of immunization.
2.4. Evaluation of Immunogenicity
Immunogenicity was evaluated by measuring haemagglutination inhibition (HAI) antibody titer at baseline and 28 days after each dose of immunization. The HAI assay against the pandemic H1N1 strain (A/Thailand/104/09) and the seasonal H1N1 (A/Brisbane/59/07-like) strains was performed at the Department of Microbiology, Faculty of Medicine Siriraj Hospital, Mahidol University using standard assay. Protective antibody titers were defined as HAI antibody titers ≥1 : 40. Seroconversion was defined as a four-fold increase from baseline HAI antibody titers in the subjects with a prevaccination HAI antibody titer >1 : 10, or postvaccination HAI antibody titer ≥1 : 40 in the subjects with prevaccination HAI antibody titer ≤1 : 10.
2.5. CD4 Count and HIV Viral Load Detection
Routine CBC-based absolute lymphocyte counts and CD4 T cell subset analysis by flow cytometry was determined on aliquots of a blood sample from each patient. Plasmas were also collected by centrifugation of an aliquot of each blood sample and kept at −70°C and used in batches for HIV viral load determination. Abbott RealTime HIV-1 was used to determine HIV-1 viral load level for each sample. The limit of detection is 40 copies/mL.
2.6. Immunofluorescence Staining and Flow Cytometric Analysis for Immune Activation
The following anti-human monoclonal antibodies (mAbs) and their conjugated fluorochromes were commercially obtained from Becton Dickinson Biosciences (BDB, San Jose, CA) and utilized at the concentration recommended by the manufacturer: anti-human CD3, clone UCHT1, conjugated with fluorescein isothiocyanate (FITC); anti-human CD38, clone HB7, conjugated with phycoerythrin (PE); anti-human CD8, clone SK1, conjugated with peridinin chlorophyll protein (PerCP); anti-human HLA-DR, clone L243, conjugated with allophycocyanin (APC). The whole blood procedure was utilized for flow cytometric studies. Briefly, an aliquot of blood was incubated with a combination of mAbs (CD3-FITC/CD38-PE/CD8-PerCP/HLA DR-APC) followed by the addition of FACS lysing solution (BDB) in order to lyse the red blood cells. Following centrifugation and washing with phosphate buffered saline (PBS), the cell pellet was resuspended in freshly prepared 1% paraformaldehyde in PBS pH 7.4 and subjected to flow cytometric analysis. Six-parameter analysis was performed on a FACSCalibur flow cytometer (BDB) using Cellquest software. Cells were collected using lymphogate, and at least 30,000 cells were analyzed per sample. CD8+ T cells were identified based on CD3+ CD8+ population. The frequency and density (mean fluorescence intensity, MFI) of CD38 or HLA-DR expressing subsets was determined by a histogram plot whereas the percentage of the CD38+ HLA DR+ cells were determined by two-color dot plot (FITC versus PE). Isotype controls were used to define the positive and negative population.
2.7. Statistical Analysis
Data for each assay are presented as mean ± S.D. of samples. The Mann-Whitney U test was used to determine the statistical significance of the difference observed between groups. The Wilcoxon signed-rank test was used to determine the statistical significance of the difference observed between each time point. P values <0.05 were considered significant.
3. Results
3.1. HIV Viral Replication and CD4+ T Cell Level
Out of 45 patients, 37 (82.2%) patients had undetectable HIV viral load at baseline. Eight patients who previously had undetectable HIV viral load showed a transient increase in HIV viral load after immunization (7 patients after the first dose and 1 patient after the second dose). In contrast, 7 of 8 HIV viremic patients maintained detectable HIV viral load throughout the study period. Thus, patients were divided into 2 groups based on the presence of HIV viral replication during immunization period. Sixteen (35.6%) patients who had detectable HIV viral load were identified as HIV detectable group whereas 29 patients who had undetectable HIV viral load were identified as HIV nondetectable group. The values of CD4 count at baseline and after immunization are shown in Table 1. The result showed no significant difference in the CD4 count at the baseline between 2 groups. No significant change in CD4 count was observed in both groups after immunization.
Table 1.
Values of CD4+ T cells.
| Group | Absolute CD4 Count (cells/μL) | %CD4 | ||||
|---|---|---|---|---|---|---|
| Baseline | One month after 1st immunization | One month after 2nd immunization | Baseline | One month after 1st immunization | One month after 2nd immunization | |
| Controller group (n = 29) | 966 ± 521 | 985 ± 549 | 967 ± 549 | 30.15 ± 8.15 | 31.47 ± 7.70 | 31.85 ± 8.03 |
| Noncontroller group (n = 16) | 1176 ± 940 | 1192 ± 1007 | 1210 ± 896 | 25.35 ± 12.25 | 25.63 ± 12.11 | 26.64 ± 11.57 |
3.2. Immunogenicity Evaluation
The serological responses were showed in Table 2. At baseline, seroprotection to H1N1 2009 was found in 14 of 29 patients in HIV nondetectable group (48.3%), whereas 5 of 16 patients were found in HIV detectable group (31.3%). Furthermore, seroprotection to seasonal influenza A was found in 14 of 29 patients in HIV nondetectable group (48.3%), whereas 4 of 16 patients in HIV detectable group (25%) showed seroprotection to seasonal influenza A.
Table 2.
Seroprotection and seroconversion rates against pandemic H1N1 2009.
| Immunogenicity | Controller | Noncontroller | ||||
|---|---|---|---|---|---|---|
| Baseline proportion (%) | After 1st dose proportion (%) | After 2nd dose proportion (%) | Baseline proportion (%) | After 1st dose proportion (%) | After 2nd dose proportion (%) | |
| Seroprotection | 14/29 (48.3) | 19/29 (65.5) | 21/29 (72.4) | 5/16 (31.3) | 7/16 (43.8) | 8/16 (50.0) |
| Seroconversion | N/A | 13/29 (44.8) | 16/29 (55.2) | N/A | 5/16 (31.3) | 5/16 (31.3) |
Note: N/A: non applicable.
When seroconversion was observed, the results from HIV nondetectable group showed that seroconversion to H1N1 2009 was found in 13 of 29 patients after the first dose of immunization (44.8%) and increased to 16 of 29 patients after the second dose of immunization (55.2%). More importantly, seroprotection has increased in 19 of 29 patients after the first dose of immunization (65.5%) and 21 of 29 patients after the second dose of immunization (72.4%). However, no seroconversion to seasonal influenza A was observed in this group of patients except 2 patients who showed an increase in the antibody titer that reaches seroprotection level after the first and second doses of immunization, respectively. In HIV detectable group, seroconversion to H1N1 2009 was found in 5 of 16 patients after the first and second doses of immunization (31.3%). Furthermore, seroprotection was found in 2 patients after the first and second dose of immunization even though the antibody titer did not reach seroconversion level. Thus, seroprotection has increased in 7 of 16 patients after the first dose of immunization (43.8%) and 8 of 16 patients after the second dose of immunization (50.0%). However, no seroconversion to seasonal influenza A was observed in this group of patients.
3.3. Immune Activation
Immune activation level was observed by the detection of CD38 and HLA-DR that expressed on CD8+ T cells. The flow cytometric data analysis of a representative sample is illustrated in Figure 1. When immune activation levels at baseline were examined, a significantly higher frequency of CD8+ T cells that expressed HLA-DR was observed in HIV detectable group when compared to HIV nondetectable group (P = 0.0156). Thus, the mean frequency of CD8+ T cells that expressed HLA-DR was 37.0 ± 15.3 in HIV detectable group as compared with 25.5 ± 14.3 in HIV nondetectable group. Moreover, a significantly higher frequency of CD8+ T cells that coexpressed CD38 and HLA-DR was observed in HIV detectable group when compared to HIV nondetectable group (P = 0.0057). Thus, the mean frequency of CD8+ T cells that coexpressed CD38 and HLA-DR was 31.4 ± 14.6 in HIV detectable group as compared with 19.9 ± 13.0 in HIV nondetectable group.
Figure 1.
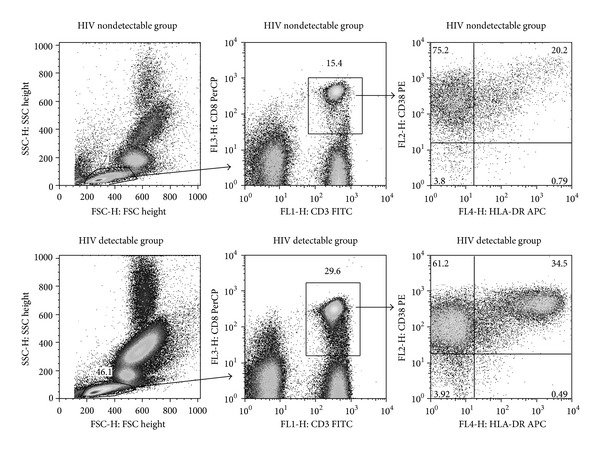
Representative gating and analysis of the frequency of CD8+ T cells that coexpressed CD38 and HLA-DR from HIV nondetectable group and HIV detectable group. Lymphocyte population is identified as a cell population with small forward scatter (FSC) and side scatter (SSC). Then, CD8+ T cells are identified by the expression of CD3 and CD8. The frequency of CD8+ T cells that expressed CD38 and HLA-DR is determined by the number in the upper right quadrant.
In order to determine the alteration in immune activation after immunization with an influenza A H1N1 2009 vaccine, the level of immune activation was compared between each time point. As shown in Figure 2, both groups of patients showed no significant change in the frequency of CD8+ T cells that expressed CD38 as well as the density of CD38 on CD8+ T cell population. In contrast, significant decreases in both frequency and density of HLA-DR expression were observed in both groups of patients.
Figure 2.
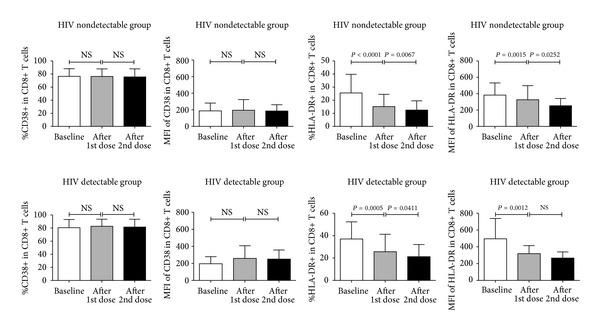
Comparison of mean frequency and mean fluorescence intensity of CD38+ or HLA-DR within CD8+ T cell population. Three different time points including baseline (open columns), 28 days after the first dose of immunization (gray columns), and 28 days after the second dose of immunization (black columns) are demonstrated for HIV nondetectable group and HIV detectable group. NS indicates no significance.
When coexpression of CD38 and HLA-DR was examined, a decrease in the frequency of CD8+ T cells that co-expressed CD38 and HLA-DR was observed after immunization when compared with each time point in both groups of patients (Figure 3). Similarly, a decrease in the frequency of CD8+ T cells that expressed HLA-DR without CD38 was observed after immunization when compared with each time point in both groups of patients. In contrast to those 2 populations, an increase in the frequency of CD8+ T cells that expressed CD38 without HLA-DR was observed after immunization when compared with each time point in both groups of patients.
Figure 3.
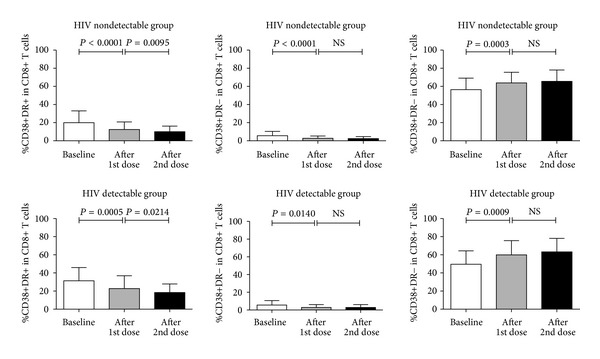
Comparison of mean frequency of CD8+ T cell population that coexpressed CD38 and HLA-DR. Three different time points including baseline (open columns), 28 days after the first dose of immunization (gray columns), and 28 days after the second dose of immunization (black columns) are demonstrated for HIV nondetectable group and HIV detectable group. NS indicates no significance.
4. Discussion
The results reported in this study showed that the use of a monovalent, nonadjuvated influenza A H1N1 2009 vaccine is moderately immunogenic in HIV-infected children receiving antiretroviral therapy. More importantly, the presence of HIV viral replication in the patients does not have an influence on the induction of the immune response by the vaccine as demonstrated by a similar seroconversion rate that was observed in both HIV nondetectable and HIV detectable group.
Studies using similar vaccine in healthy adults showed higher seroprotection and seroconversion rates than that observed in this study [19–21]. The results reported in this study demonstrated low seroprotection and seroconversion rates similar to that observed in the previous studies which showed a low immunogenicity to a pandemic influenza A H1N1 vaccine [22, 23]. Apart from a similar immunogenicity after immunization with a pandemic influenza A H1N1 vaccine when compared to other studies, the results reported in this study focused on the effect of a pandemic influenza A H1N1 immunization on the level of immune activation which has never been observed in other studies. This study answered a question that is necessary and will be relevant to clinical practice, since immunization alone might induce a disease progression in HIV infected patients. If vaccination induces a strong generalized immune activation in the patients, it might cause a serious side effect to the patients such as an induction of HIV viral replication. An extremes caution needs to be considered when starting an immunization program in HIV infected patients.
Furthermore, in an attempt to examine the induction of HIV viral replication and generalized immune activation after immunization with a pandemic influenza A H1N1 vaccine, the result showed that immunization with this vaccine did not induce a marked increase in HIV viral replication except a transient increase in some patients. Furthermore, no marked increase in the level of immune activation, as determined by a combination of CD38 and HLA-DR expression, since decrease levels of either HLA-DR alone or in combination with CD38 were observed in patients during baseline and after immunization. The reason for a decrease in HLA-DR expression after immunization with a pandemic influenza A H1N1 vaccine as observed in this study remains unclear since a previous study showed that an immunization with DNA-based HIV vaccine did not induce any alteration in the expression of activation markers [24]. While another study showed that different cytokines had different effects on the expression of HLA-DR on specific T cell population [25]. It might be possible that an induction of specific pattern of cytokine response during immunization might induce a downregulation of HLA-DR expression on CD8+ T cells.
While previous studies showed an increases HIV viral replication after immunization [5–7], the finding that immunization by an influenza A H1N1 vaccine in this study did not induce a marked increase in HIV viral replication or immune activation suggested that the immune stimulation induced by the vaccine is safe in HIV-infected children. It might be possible that the antiretroviral therapy which was taken by these HIV-infected children has enough HIV viral suppression effect during the time of antigen stimulation by the vaccine. These results are in agreement with a previous study that shows a stable HIV viral load and CD4 T cell count after immunization with an influenza vaccine [9].
5. Conclusion
In conclusion, the results in this study demonstrated that a monovalent, nonadjuvated influenza A H1N1 2009 vaccine is immunogenic in HIV-infected children receiving antiretroviral therapy regardless of neither the patients had active HIV viral replication nor they had a complete suppression. Furthermore, immunization did not significantly induce either HIV viral replication, alteration of CD4+ T cell level, or immune activation in HIV-infected children receiving antiretroviral therapy.
Conflict of Interests
The authors certify that there is no conflict of interests with any financial organizations regarding the material discussed in this study.
Acknowledgments
The authors gratefully acknowledge the kind cooperation of the HIV-infected patients for the studies reported herein. They also thank the primary care physicians and nurses who worked hard to provide with them the needed samples. This project is supported by Thailand Research Fund and Faculty of Medicine Siriraj Hospital, Mahidol University, under TRF Master Research Grant (TRF-MRG-WINDOW II), the Office of the Higher Education Commission and Mahidol University under the National Research Universities Initiative, as well as the Senior Research Scholar grant from the Thailand Research Fund (to Kovit Pattanpanyasat). Kovit Pattanpanyasat and Nattawat Onlamoon are supported by Chalermphrakiat Grant from Faculty of Medicine Siriraj Hospital. The opinions and assertions contained herein are private views of the authors and are not to be construed as official opinions or as reflecting the views of the Thailand Research Fund and Faculty of Medicine Siriraj Hospital, Mahidol University.
References
- 1.Sullivan SJ, Jacobson RM, Dowdle WR, Poland GA. 2009 H1N1 influenza. Mayo Clinic Proceedings. 2010;85(1):64–76. doi: 10.4065/mcp.2009.0588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lin JC, Nichol KL. Excess mortality due to pneumonia or influenza during influenza seasons among persons with acquired immunodeficiency syndrome. Archives of Internal Medicine. 2001;161(3):441–446. doi: 10.1001/archinte.161.3.441. [DOI] [PubMed] [Google Scholar]
- 3.Zanetti AR, Amendola A, Besana S, Boschini A, Tanzi E. Safety and immunogenicity of influenza vaccination in individuals infected with HIV. Vaccine. 2002;20(supplement 5):B29–B32. doi: 10.1016/s0264-410x(02)00511-x. [DOI] [PubMed] [Google Scholar]
- 4.Pinto LA, Blazevic V, Anderson SA, et al. Influenza virus-stimulated generation of anti-human immunodeficiency virus (HIV) activity after influenza vaccination in HIV-infected individuals and healthy control subjects. Journal of Infectious Diseases. 2001;183(7):1000–1008. doi: 10.1086/319277. [DOI] [PubMed] [Google Scholar]
- 5.O’Brien WA, Grovit-Ferbas K, Namazi A, et al. Human immunodeficiency virus-type 1 replication can be increased in peripheral blood of seropositive patients after influenza vaccination. Blood. 1995;86(3):1082–1089. [PubMed] [Google Scholar]
- 6.Staprans SI, Hamilton BL, Follansbee SE, et al. Activation of virus replication after vaccination of HIV-1-infected individuals. Journal of Experimental Medicine. 1995;182(6):1727–1737. doi: 10.1084/jem.182.6.1727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ramilo O, Hicks PJ, Borvak J, et al. T cell activation and human immunodeficiency virus replication after influenza immunization of infected children. Pediatric Infectious Disease Journal. 1996;15(3):197–203. doi: 10.1097/00006454-199603000-00004. [DOI] [PubMed] [Google Scholar]
- 8.Tasker SA, O’Brien WA, Treanor JJ, et al. Effects of influenza vaccination in HIV-infected adults: a double-blind, placebo-controlled trial. Vaccine. 1998;16(9-10):1039–1042. doi: 10.1016/s0264-410x(97)00275-2. [DOI] [PubMed] [Google Scholar]
- 9.Fowke KR, D’Amico R, Chernoff DN, et al. Immunologic and virologic evaluation after influenza vaccination of HIV-1-infected patients. AIDS. 1997;11(8):1013–1021. doi: 10.1097/00002030-199708000-00010. [DOI] [PubMed] [Google Scholar]
- 10.McMichael AJ, Rowland-Jones SL. Cellular immune responses to HIV. Nature. 2001;410(6831):980–987. doi: 10.1038/35073658. [DOI] [PubMed] [Google Scholar]
- 11.Mahalingam M, Peakman M, Davies ET, et al. T cell activation and disease severity in HIV infection. Clinical & Experimental Immunology. 1993;93(3):337–343. doi: 10.1111/j.1365-2249.1993.tb08182.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Mahalingam M, Pozniak A, McManus TJ, Vergani D, Peakman M. Cell cycling in HIV infection: analysis of in vivo activated lymphocytes. Clinical and Experimental Immunology. 1995;102(3):481–486. doi: 10.1111/j.1365-2249.1995.tb03841.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Levacher M, Hulstaert F, Tallet S, Ullery S, Pocidalo JJ, Bach BA. The significance of activation markers on CD8 lymphocytes in human immunodeficiency syndrome: staging and prognostic value. Clinical and Experimental Immunology. 1992;90(3):376–382. doi: 10.1111/j.1365-2249.1992.tb05854.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Orendi JM, Bloem AC, Borleffs JCC, et al. Activation and cell cycle antigens in CD4+ and CD8+ T cells correlate with plasma human immunodeficiency virus (HIV-1) RNA level in HIV-1 infection. Journal of Infectious Diseases. 1998;178(5):1279–1287. doi: 10.1086/314451. [DOI] [PubMed] [Google Scholar]
- 15.Bouscarat F, Levacher-Clergeot M, Dazza M-C, et al. Correlation of CD8 lymphocyte activation with cellular viremia and plasma HIV RNA levels in asymptomatic patients infected by human immunodeficiency virus type 1. AIDS Research and Human Retroviruses. 1996;12(1):17–24. doi: 10.1089/aid.1996.12.17. [DOI] [PubMed] [Google Scholar]
- 16.Ondoa P, Koblavi-Dème S, Borget M-Y, Nolan ML, Nkengasong JN, Kestens L. Assessment of CD8+ T cell immune activation markers to monitor response to antiretroviral therapy among HIV-1 infected patients in Cote d’Ivoire. Clinical and Experimental Immunology. 2005;140(1):138–148. doi: 10.1111/j.1365-2249.2005.02734.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhang Z, Shang H, Jiang Y, et al. Activation and coreceptor expression of T lymphocytes induced by highly active antiretroviral therapy in Chinese HIV/AIDS patients. Chinese Medical Journal. 2006;119(23):1966–1971. [PubMed] [Google Scholar]
- 18.Onlamoon N, Tabprasit S, Suwanagool S, Louisirirotchanakul S, Ansari AA, Pattanapanyasat K. Studies on the potential use of CD38 expression as a marker for the efficacy of anti-retroviral therapy in HIV-1-infected patients in Thailand. Virology. 2005;341(2):238–247. doi: 10.1016/j.virol.2005.07.018. [DOI] [PubMed] [Google Scholar]
- 19.Greenberg ME, Lai MH, Hartel GF, et al. Response to a monovalent 2009 influenza A (H1N1) vaccine. The New England Journal of Medicine. 2009;361(25):2405–2413. doi: 10.1056/NEJMoa0907413. [DOI] [PubMed] [Google Scholar]
- 20.Zhu FC, Wang H, Fang HH, et al. A novel influenza A, (H1N1) vaccine in various age groups. The New England Journal of Medicine. 2009;361(25):2414–2423. doi: 10.1056/NEJMoa0908535. [DOI] [PubMed] [Google Scholar]
- 21.Liang X, Wang H, Wang J, et al. Safety and immunogenicity of 2009 pandemic influenza A H1N1 vaccines in China: a multicentre, double-blind, randomised, placebo-controlled trial. The Lancet. 2010;375(9708):56–66. doi: 10.1016/S0140-6736(09)62003-1. [DOI] [PubMed] [Google Scholar]
- 22.Crum-Cianflone NF, Eberly LE, Duplessis C, et al. Immunogenicity of a monovalent 2009 influenza a (H1N1) vaccine in an immunocompromised population: a prospective study comparing HIV-infected adults with HIV-uninfected adults. Clinical Infectious Diseases. 2011;52(1):138–146. doi: 10.1093/cid/ciq019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Tebas P, Frank I, Lewis M, et al. Poor immunogenicity of the H1N1 2009 vaccine in well controlled HIV-infected individuals. AIDS. 2010;24(14):2187–2192. doi: 10.1097/QAD.0b013e32833c6d5c. [DOI] [PubMed] [Google Scholar]
- 24.Reuter MA, Yuan S, Marx PA, et al. DNA-based HIV vaccines do not induce generalized activation in mucosal tissue T cells. Human Vaccines & Immunotherapeutics. 2012;8(11):1648–1653. doi: 10.4161/hv.22247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Salgado FJ, Lojo J, Fernández-Alonso CM, et al. Interleukin-dependent modulation of HLA-DR expression on CD4and CD8 activated T cells. Immunology and Cell Biology. 2002;80(2):138–147. doi: 10.1046/j.1440-1711.2002.01055.x. [DOI] [PubMed] [Google Scholar]


