Significance
Silicon (Si) is the second most abundant element in soil, and it can increase plant resistance against many abiotic and biotic stresses. The jasmonate (JA) signaling pathway plays a crucial role in mediating antiherbivore defense responses in plants. Our work shows that Si primes JA-mediated antiherbivore defense responses and increases rice resistance to the leaffolder caterpillar and that Si accumulation in rice leaves is mediated by the JA pathway, suggesting a strong interaction between Si and JA in rice defense against insect herbivores. This interaction may represent a widespread mechanism by which Si enhances plant resistance against biotic stresses in Si-accumulating plants.
Keywords: Oryza sativa, induced defense, jasmonic acid, mitogen-activated protein kinase
Abstract
Although the function of silicon (Si) in plant physiology has long been debated, its beneficial effects on plant resistance against abiotic and biotic stresses, including insect herbivory, have been well documented. In addition, the jasmonate (JA) signaling pathway plays a crucial role in mediating antiherbivore defense responses in plants. However, potential interactions between JA and Si in response to insect attack have not been examined directly. To explore the role JA may play in Si-enhanced resistance, we silenced the expression of allene oxide synthase (OsAOS; active in JA biosynthesis) and CORONATINE INSENSITIVE1 (OsCOI1; active in JA perception) genes in transgenic rice plants via RNAi and examined resulting changes in Si accumulation and defense responses against caterpillar Cnaphalocrocis medinalis (rice leaffolder, LF) infestation. Si pretreatment increased rice resistance against LF larvae in wild-type plants but not in OsAOS and OsCOI1 RNAi lines. Upon LF attack, wild-type plants subjected to Si pretreatment exhibited enhanced defense responses relative to untreated controls, including higher levels of JA accumulation; increased levels of transcripts encoding defense marker genes; and elevated activities of peroxidase, polyphenol oxidase, and trypsin protease inhibitor. Additionally, reduced Si deposition and Si cell expansion were observed in leaves of OsAOS and OsCOI1 RNAi plants in comparison with wild-type plants, and reduced steady-state transcript levels of the Si transporters OsLsi1, OsLsi2, and OsLsi6 were observed in Si-pretreated plants after LF attack. These results suggest a strong interaction between Si and JA in defense against insect herbivores involving priming of JA-mediated defense responses by Si and the promotion of Si accumulation by JA.
Silicon (Si) is the second most abundant element in soil (following oxygen) and comprises ∼28% (wt/wt) of Earth’s crust (1). Although Si is not considered an essential element for the majority of plants, the beneficial effects of this element on plant growth, development, stress resistance, and defense have been well documented in many plant species. Si treatments have been shown to be beneficial for plants exposed to an array of abiotic and biotic stresses (2). In the absence of stress, however, differences between Si-treated and untreated plants are not readily discernible (3, 4).
Although numerous studies have shown that Si application can enhance plant resistance to insect herbivores and other arthropods (5–7), the details surrounding the underlying mechanism(s) remain obscure. An increased physical barrier produced by the deposition of Si beneath the leaf cuticles has long been considered to represent a major component underlying Si-mediated plant resistance to insect pests (5, 8). Si deposition, occurring mainly as opaline phytoliths, is associated with increased rigidity and abrasiveness of plant tissues, thereby reducing their digestibility to herbivores. Recent studies also have shown that Si treatment induces the activities of plant defensive enzymes (9, 10) and leads to increased accumulation of defensive compounds such as phenolics, phytoalexins, and momilactones (11–13) and to increased release of plant volatiles that attract natural enemies of herbivorous insects (6). Furthermore, transcriptome analyses in Si-pretreated Arabidopsis thaliana plants inoculated with powdery mildew suggest that Si attenuates the decrease in primary metabolism associated with pathogen infection, leading to an overall more efficient defense response (4).
Plants use both constitutive and inducible defensive strategies against insect herbivores (14). Inducible defenses allow plants to manage energy reserves more efficiently by synthesizing defense compounds only when needed. For plant responses to chewing insects, the jasmonic acid (JA) signaling pathway plays an essential role in these processes (15, 16). Upon attack, plants initiate defense responses aided by the recognition of herbivore-specific molecular patterns (17), followed by the activation of a complex regulatory network involving mitogen-activated protein kinase (MAPK) cascades, jasmonate and ethylene signaling, the expression of defense-related genes, and the production of defense compounds (15, 18).
Although induced defense responses allow plants to conserve energy in the absence of herbivore challenge, plants may suffer considerable damage during the lag phase required to mount defenses once an attack occurs (19). To compensate for this vulnerability, some plants have evolved defense-priming mechanisms that facilitate more rapid and effective responses to herbivore attacks. Defense priming can lead to an enhanced state of readiness for the induction of defense responses following an initial stimulus (20). Primed plants thus are physiologically prepared to induce more potent defense responses upon subsequent attack, providing plants with a more effective means to respond to challenges, with minimal associated metabolic costs because metabolic requirements of the priming mechanism itself are relatively low (21). In addition to prior insect damage, priming also can be triggered by environmental cues such as insect oviposition, pathogen challenge, and volatile emissions from neighboring plants (e.g., refs. 22–24).
Rice (Oryza sativa L.) is an important crop and model system that accumulates high levels of Si constituting as much as 10% (wt/wt) of total plant dry mass (1, 25, 26). In this study, we investigated both the molecular and physiological aspects of Si amendment on resistance to the rice leaffolder (LF; Cnaphalocrocis medinalis) (Lepidoptera: Pyralidae), a chewing insect responsible for significant economic losses to farmers. Transgenic rice plants deficient in JA biosynthesis and perception were generated via RNAi and analyzed to determine the potential involvement of JA signaling in Si-mediated resistance. Si pretreatment of rice seedlings resulted in elevated JA-mediated transcriptional responses and defense-related enzymatic activities following LF attack and in increased levels of JA accumulation. Additionally, significant decreases in Si deposition and an apparent loss of Si-induced LF resistance were observed in transgenic events that silenced the expression of either allene oxide synthase (OsAOS), which is involved in JA biosynthesis or CORONATINE INSENSITIVE1 (OsCOI1), which is involved in JA perception. Taken together, these results strongly suggest a critical interplay between Si and JA in rice involving the priming of JA-mediated defense responses by Si and the regulation of Si-mediated herbivore resistance by JA via enhanced Si deposition and potentially other, as yet unidentified, mechanisms.
Results
Si-Enhanced Resistance to LF and Its Dependence on JA.
To determine whether Si treatment increases rice resistance to LF, and, if so, whether resistance is JA dependent, rice seedlings of the three genotypes (WT and OsAOS and OsCOI1 RNAi lines) were treated with Si and methyl jasmonate (MeJA). Both Si and MeJA application significantly increased the resistance of WT plants to LF infestation (Fig. 1). LF larvae fed on control-treated WT plants increased in mass by 168.3% 3 d after insect infestation, whereas larvae fed on Si-treated and MeJA-treated WT plants increased in mass by only 84.2 and 69.4%, respectively. MeJA application on Si-treated WT plants further enhanced the resistance of WT plants to LF infestation, leading to a gain in larval mass of only 47.4% (Fig. 1). RNAi-mediated silencing of OsAOS and OsCOI1, two critical genes involved in JA biosynthesis and perception, respectively (15, 27), significantly reduced the resistance of WT plants to LF infestation (Fig. 1). LF larvae fed on non–Si-treated OsAOS and OsCOI1 RNAi lines increased in mass by 256.2 and 269.3%, respectively, demonstrating that JA plays an important role in rice resistance to LF infestation. Importantly, Si pretreatment had no significant effect on resistance to LF infestation in either the OsAOS or OsCOI1 RNAi lines, so the observed Si-mediated resistance to LF infestation in WT plants occurs in a JA-dependent manner. Exogenous MeJA applied to OsAOS RNAi plants increased resistance to LF infestation in both the Si-treated and untreated experimental groups; however, resistance levels were not fully restored to those observed in WT plants (Fig. 1). Not unexpectedly, the application of exogenous MeJA did not increase the resistance to LF infestation in Si-treated and untreated OsCOI1 RNAi plants thata were deficient in JA-signaling perception.
Fig. 1.
Gain in mass of LF larvae fed on WT rice plants and the OsCOI1 RNAi and OsAOS RNAi lines treated with Si and MeJA. Seven-day-old seedlings were transplanted to nutrient solution containing 2 mM K2SiO3. In the Si-deficient treatment, potassium chloride was used to replenish potassium. OsCOI1 and OsAOS RNAi lines and WT rice plants were sprayed with 1 mM MeJA or buffer (control) 20 d after transplanting. Two days later all plants were infected by third-instar LF larvae at leaf node 3. The individual larvae were measured 3 d later, and the mean percentage of gain in mass was calculated. Values are mean ± SE (n = 20). Letters above bars indicate significant differences among treatments (P < 0.05 according to Tukey’s multiple range test).
Si-Mediated Enhancement of JA Accumulation and OsAOS and OsCOI1 Transcriptional Responses.
Because JA is known to play a central role in mediating plant defense responses against insect herbivores, interactions between Si and JA in rice were examined further by monitoring the accumulation of JA and endogenous OsAOS and OsCOI1 transcript levels in Si-treated and untreated WT plants exposed to LF infestation or exogenous MeJA (Fig. 2). Si treatment alone did not significantly affect endogenous OsAOS and OsCOI1 steady-state transcript levels in leaves relative to untreated control plants during the 24-h time period (Fig. 2 A–D). In untreated plants, both LF infestation and MeJA application led to increased OsAOS and OsCOI1 transcript levels, and, significantly, the magnitude of these responses for both transcripts was higher in Si-treated than in untreated plants. This effect was particularly evident at 6–24 h after treatment; for example, in untreated plants at 6 h after LF exposure, OsAOS and OsCOI1 steady-state transcript levels increased by 1.75- and 1.84-fold, respectively, relative to controls not exposed to LF, whereas in Si-treated plants, OsAOS and OsCOI1 transcript levels increased by 3.28- and 2.80-fold, respectively (Fig. 2 A and B). Additionally, the data suggest that Si pretreatment also may facilitate more rapid responses to these stimuli. For example, in Si-treated plants, both OsAOS and OsCOI1 transcript levels were modestly induced within 1.5 h following LF infestation, whereas in untreated plants this induction was not clearly discernible until 3 and 6 h postinfestation for OsAOS and OsCOI1 transcripts, respectively (Fig. 2 A and B).
Fig. 2.
Steady-state levels of OsAOS (A and C) and OsCOI1 (B and D) transcripts and JA levels (E) in the leaves of WT rice plants not treated with Si or treated with Si with or without LF infestation (A and B) or treatment with MeJA (C and D). Si and MeJA treatments were performed as described for experiments shown in Fig. 1. (A–D) For gene-expression experiments the treatments included no Si added (Si−); no Si added followed by MeJA treatment (Si−MeJA+) or LF infestation (Si−LF+); 2 mM potassium silicate added (Si+); and 2 mM potassium silicate added followed by treatment with 1 mM MeJA (Si+MeJA+) or LF infestation (Si+LF+). Real-time qRT-PCR analysis was used to determine the relative steady-state transcript levels shown. Values shown are mean ± SE (n = 3). (E) For JA analysis the six treatments were Si−, Si+, Si−MeJA+, Si−LF+, Si+MeJA+, and Si+LF+; values are mean ± SE (n = 6). For each time point, letters above bars indicate significant difference among treatments (P < 0.05 according to Tukey’s multiple range test).
Similarly, Si pretreatment alone did not significantly alter the levels of JA accumulation relative to untreated control plants during the 24-h time course used (Fig. 2E). However, in response to LF infestation, JA levels in Si-treated WT plants were significantly higher than those of plants not receiving Si at 6 and 9 h following LF exposure (P = 0.0018 and 0.034, respectively) (Fig. 2E). Additionally, although exogenous MeJA applications increased JA levels in both Si-treated and untreated WT plants, Si-treated plants exhibited significantly higher JA levels than those observed in untreated plants at 3, 9, and 12 h after MeJA application (P = 0.036, 0.0022, and 0.015, respectively) (Fig. 2E). By 24 h after MeJA application, Si-treated and untreated plants exhibited similar JA levels that were approximately fivefold higher than those observed in the corresponding samples at 0 h.
Si-Mediated Enhancement of MAPK and WRKY Transcriptional Responses.
MAPK cascades and WRKY transcription factors are known to serve as components in plant signaling pathways involved in herbivore-induced defense responses (28, 29); therefore, steady-state accumulation levels for transcripts encoding selected MAPKs and WRKY factors also were examined in Si-treated and untreated WT plants exposed to LF infestation and exogenous MeJA (Fig. 3). As was observed for endogenous OsAOS and OsCOI1 transcripts (Fig. 2 A–D), real-time PCR analyses showed that Si treatment alone did not induce transcript levels of OsMPK3, OsMPK6, OsWRKY53, or OsWRKY70 in leaves of WT plants relative to untreated plants (Fig. 3). Overall, untreated plants exposed to LF infestation and MeJA application exhibited increased accumulation levels of all four transcripts relative to control plants, with the exception of OsWRKY53 transcripts following MeJA treatment (Fig. 3G), for which steady-state transcript levels did not change significantly. However, as was also the case for OsAOS and OsCOI1 transcripts in Si-pretreated plants, the magnitude of induction following LF infestation or MeJA application generally increased for all four transcripts in Si-treated versus untreated plants. For example, steady-state OsMPK3, OsMPK6, OsWRKY53, and OsWRKY70 transcript levels in untreated WT plants increased by 1.9-, 3.2-, 1.9-, and 2.3-fold, respectively, 1 h after LF infestation, whereas in Si-pretreated plants these induction levels increased by 2.5-, 4.2-, 2.6-, and 4.1-fold, respectively (Fig. 3 A–D). Additionally, in some instances the data suggested faster response times to MeJA or LF exposure; for example, increased accumulation levels of all four transcripts were observed in Si-treated plants at 0.5 h after LF infestation, but the induction of OsMPK3 and OsWRKY70 transcript levels was not observed at the same time point in plants not pretreated with Si (Fig. 3 A–D). Furthermore, only OsMPK3 transcript levels were increased at 0.5 h after MeJA treatment in untreated plants, but in Si-treated plants modest increases in OsMPK6 and OsWRKY70 transcript levels also were detected at 0.5 h (Fig. 3 E–H).
Fig. 3.
Steady-state levels of OsMPK3 (A and E) OsMPK6 (B and F), OsWRKY53 (C and G), and OsWRKY70 (D and H) transcripts in leaves of WT rice plants treated with Si plus LF infestation (A–D) or MeJA (E–H) The treatments shown were performed as described in Fig. 2. Real-time qRT-PCR analysis was used to determine the relative steady-state transcript levels shown. Values are mean ± SE (n = 3). For each time point, letters above bars indicate significant difference among treatments (P < 0.05 according to Tukey’s multiple range test).
Si-Mediated Enhancement of Defense-Related Enzyme and Protease Inhibitor Induction.
Defense-related related enzymes such as polyphenol oxidase (PPO) and peroxidase (POD), as well as protease inhibitors such as trypsin protease inhibitor (TrypPI) and Bowman–Birk protease inhibitor (BBPI), represent important components of plant inducible defense responses; moreover, PPO, POD, TrypPI, and BBPI levels previously have been shown to increase in response to exogenous MeJA treatments in rice (e.g., refs. 30, 31). To examine further the potential effects of Si amendment on plant defense responses with respect to JA signaling, Si-treated and untreated WT, OsAOS, and OsCOI1 RNAi rice plants were subjected to LF attack, and activity levels for PPO, POD, and TrypPI as well as steady-state transcript levels for OsBBPI were monitored (Fig. 4). Consistent with the results shown in Figs. 2 and 3 in which steady-state transcript levels were monitored, Si amendment by itself did not significantly alter PPO, POD, TrypPI, or OsBBPI levels (Fig. 4) for any of the three rice genotypes tested. Importantly, levels of all four were induced in both Si-treated and untreated WT seedlings after LF attack, but in all cases the magnitude of induction was higher in Si-treated seedlings. For example, in response to LF exposure, POD, PPO, and TrypPI activity increased by 162.5, 95.0, and 108.4%, respectively, in untreated WT seedlings relative to control seedlings. In comparison, levels of POD, PPO, and TrypPI increased by 213.2, 264.9, and 163.9%, respectively, in WT seedlings pretreated with Si relative to Si-treated seedlings not exposed to LF attack (Fig. 4 A–C). Similarly, in the absence of Si amendment, OsBBPI steady-state transcript levels in WT plants increased ∼3.9-fold following LF infestation, whereas transcript levels were induced ∼5.2-fold in Si-treated seedlings (Fig. 4D). Consistent with prior results obtained for PPO, POD, TrypPI, or OsBBPI in rice (29, 30), the induction of all four in response to insect herbivore attack occurred in a JA-dependent manner, because levels of PPO, POD, TrypPI, or OsBBPI either were unchanged or increased only marginally in LF-exposed OsAOS and OsCOI1 RNAi seedlings relative to seedlings not exposed to LF attack (Fig. 4).
Fig. 4.
Activity levels of PPO (A), POD (B), and TrypPI (C) and steady-state transcript levels of OsBBPI (D) in leaves of WT, OsCOI1 RNAi, and OsAOS RNAi plants treated with Si (Si+) or with Si plus LF infestation or MeJA. Values are mean ± SE (n = 6). Letters above bars indicate significant differences among treatments (P < 0.05 according to Tukey’s multiple range test). Factorial ANOVA showed that the effects of genotype, LF infestation, Si treatment, and MeJA treatment and their interactions on POD, PPO, and TrypPI activities and OsBBPI transcript levels were significant.
Influence of JA on Leaf Silicification and Si Cell Development.
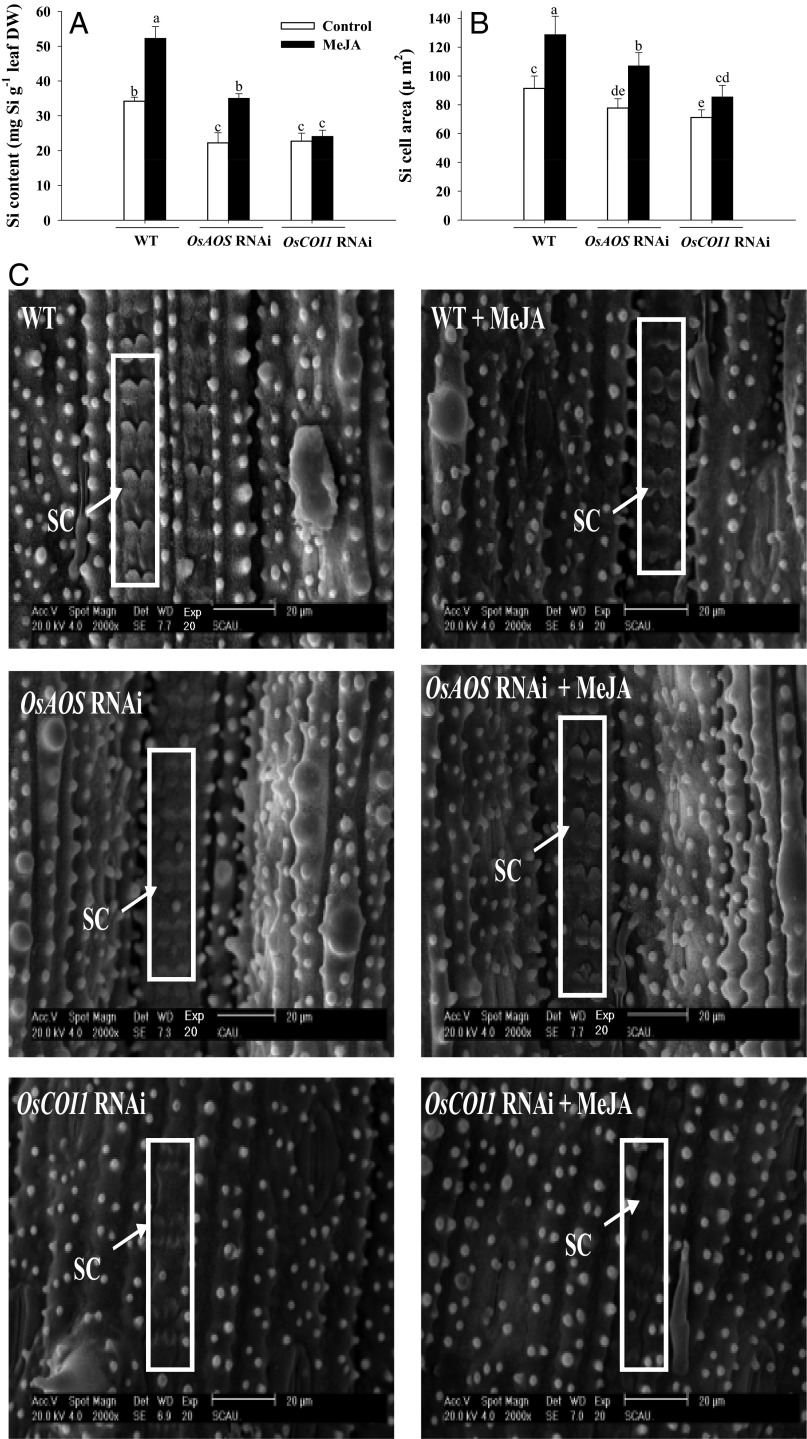
The experiments shown in Fig. 1 indicate that Si-mediated resistance against LF herbivory is JA dependent, so it is reasonable to speculate that the deposition of Si itself could be regulated by JA in rice. To address this question, leaf Si content and epidermal Si cell expansion were determined for rice plants of the three genotypes (WT and the OsAOS and OsCOI1 RNAi lines) treated and untreated with MeJA (Fig. 5). Importantly, MeJA treatment of WT plants resulted in increased Si content and Si cell area by 65.5 and 38.5%, respectively, relative to untreated plants (Fig. 5 A and B). Conversely, silencing of OsAOS and OsCOI1 led to a significant reduction in Si deposition and Si cell area. In plants not treated with MeJA, leaf Si contents in the OsAOS and OsCOI1 RNAi lines were 40.5 and 45.7% lower, respectively, than the levels observed in WT plants (Fig. 5A). Similarly, Si cell area was reduced by 15.9 and 19.3% in the leaves of OsAOS and OsCOI1 RNAi plants, respectively, relative to WT plants (Fig. 5B). MeJA treatment of OsAOS RNAi plants increased Si content and Si cell area in leaves by 36.2 and 54.3%, respectively, but these values were significantly lower than those observed in MeJA-treated WT plants (Fig. 5 A and B). Exogenous application of MeJA did not significantly alter Si content or epidermal Si cell area in leaves of OsCOI1 RNAi plants (Fig. 5 A and B). After LF infestation, WT plants exhibited an increase in Si content similar to that observed after MeJA treatment, but the Si contents of OsAOS and OsCOI1 RNAi plants were not altered significantly relative to untreated plants (Fig. S1).
Fig. 5.
Total leaf Si content (A), Si cell area (B), and scanning electron micrographs of Si cells (C) in leaves of WT, OsCOI1 RNAi, and OsAOS RNAi plants treated or not treated with MeJA. MeJA treatments were performed as described in Fig. 1. Values are mean ± SE (n = 10). (A and B) Letters above bars indicate significant differences among all treatments and rice lines (P < 0.05 according to Tukey’s multiple range test). (C) Scanning electron micrographs of rice leaf cross-sections. Bilobed (“dumbbell”-shaped) SI cells from node 3 leaves of WT, OsAOS RNAi, and OsCOI1 RNAi plants are shown. (Scale bar: 20 μm.) Silica cells were quantified at a magnification of 2,000×. The length and width of the Si cell were measured; then the Si area was calculated. SC, silica cell.
Furthermore, environmental scanning electron microscopy (ESEM) analyses revealed significant morphological differences in the leaf epidermal Si cells from WT and OsAOS and OsCOI1 RNAi plants (Fig. 5C). Si cells in the leaves of WT plants were more clearly defined and more expanded than the Si cells in the leaves of the OsAOS and OsCOI1 RNAi lines. Additionally, for both WT and OsAOS RNAi plants, MeJA application appeared to increase differentiation and expansion of Si cells relative to untreated plants, but this effect was not readily discernible in the Si cells of OsCOI1 RNAi plants (Fig. 5C).
Si-Mediated Enhancement of Transcriptional Responses of Si Transporter Genes OsLsi1, OsLsi2, and OsLsi6.
OsLsi1, OsLsi2, and OsLsi6 are Si transporters that mediate the uptake and translocation of Si in rice plants (25, 26, 32). Steady-state transcript levels of OsLsi1, OsLsi2, and OsLsi6 were examined in Si-treated and untreated rice plants of WT and OsAOS and OsCOI1 RNAi lines treated with MeJA or exposed to LF infestation (Fig. 6).
Fig. 6.
Steady-state levels of OsLsi1 and OsLsi2 transcripts in roots and of OsLsi6 in the leaf sheath of WT rice plants and OsCOI1 RNAi and OsAOS RNAi plants treated with Si plus MeJA or LF infestation. The treatments shown were performed as described in Fig. 2. Transcripts levels of OsLsi1 (A), OsLsi2 (B), and OsLsi6 (C) in OsAOS RNAi, OsCOI1 RNAi, and WT plants were analyzed 24 h after MeJA application or LF infestation. Real-time qRT-PCR analysis was used to determine the relative steady-state transcript levels shown. Values are mean ± SE (n = 3). For each time point, letters above bars indicate significant difference among treatments (P < 0.05 according to Tukey’s multiple range test).
Real-time PCR analyses revealed that, in the absence of Si amendment, exogenous MeJA application or LF infestation alone either did not induce or only marginally induced transcript levels of OsLsi1, OsLsi2, and OsLsi6 relative to untreated plants (Fig. 6). Si application to WT plants significantly increased transcript levels of the three Si transporter genes (Fig. 6), and MeJA application or LF infestation enhanced the induction of all three genes in Si-treated plants. Although Si application alone induced OsLsi1, OsLsi2, and OsLsi6 transcript levels by 2.0-, 4.2-, and 3.3-fold, respectively, relative to untreated controls, LF infestation on Si-treated plants induced OsLsi1, OsLsi2, and OsLsi6 transcript levels by 4.2-, 10.1-, and 7.9-fold, respectively, and MeJA application induced transcript levels by 4.8-, 15.8-, and 10.2-fold, respectively (Fig. 6). However, silencing of OsAOS and OsCOI1 led to no or only marginal induction of the three Si transporter transcript levels after Si application, and this also was the case for Si-treated plants in response to LF infestation. This impaired induction could not be rescued by exogenous MeJA in the OsCOI1 RNAi line and was only partially rescued by MeJA in the OsAOS RNAi line.
Discussion
The high levels of Si accumulated by rice plants likely indicate that Si plays a particularly prominent role in Oryza spp. in mediating resistance to various biotic and abiotic stresses (2, 10, 12, 25, 26). In the present work, we found that Si amendment significantly increases rice resistance to infestation by LF, an economically important insect herbivore. Exogenous MeJA application alone increases resistance to LF attack (Fig. 1), but Si amendment appeared to amplify the JA-mediated induced defense responses, serving as an apparent priming agent for JA. Importantly, because Si amendment had no discernible effect on LF resistance in JA-deficient OsAOS and OsCOI1 RNAi lines, Si-mediated LF resistance likely occurs in a JA-dependent manner in rice. Furthermore, Si previously has been demonstrated to play a role in constitutive plant defenses against herbivore insects (7), and our study, along with other recent work (4, 6, 12), now indicates its involvement in induced defenses, particularly those mediated by JA.
Furthermore, JA-deficient OsAOS and OsCOI1 RNAi plants exhibited reduced total leaf Si content (Fig. 5A) and reduced development of Si cells and Si body formation, as indicated by ESEM (Fig. 5B). Conversely, exogenous MeJA applications increased leaf Si content, leaf Si cell development, and Si transporters OsLsi1, OsLsi2, and OsLsi6 transcript levels in WT and OsAOS RNAi plants, demonstrating an interplay between JA and Si in rice during defense against insect herbivores. Exogenous MeJA applications were not observed to increase either Si content or Si cell area in OsCOI1 RNAi plants (Fig. 5 A and B); this result was not unexpected, given that OsCOI1 deficiency would render plants unable to respond to JA. To date, no study has demonstrated a role for JA in regulating Si uptake and transport, although specific Si transporters associated with the root vascular stele have been identified in rice (26).
The role of JA in mediating responses to abiotic and biotic stresses has been investigated extensively, but JA and other oxylipins also are known to influence specific developmental processes, including male reproductive development in A. thaliana and embryogenesis in tomato (33). It was shown previously by Li et al. (34) that leaves of COI-deficient (JA-insensitive) tomato have significantly reduced densities of glandular (type VI) trichomes, which are multicellular epidermal structures associated with the storage and synthesis of defense-related secondary metabolites. Nonglandular trichome development is unaffected in COI-deficient tomato, and leaf development occurs normally otherwise. Thus, in this case JA appears to influence specifically the formation of glandular trichomes (33). Silica cells such as those found on rice leaf epidermal surfaces (Fig. 5C) are anucleate, lack cytoplasm, and function primarily as deposition sites for silica bodies (phytoliths) at maturity (35). Given that both silica bodies and glandular trichomes are highly specialized cellular structures associated with defense responses, it is reasonable to speculate that the hormone JA plays a general role in promoting the development of such structures within plant epidermal tissues.
The results obtained in this study also strongly suggest a role for Si in the priming of JA-mediated inducible defense responses, in addition to the potential of Si to act as an insect antifeedant or as a component of physical barriers inhibitory to feeding (2, 5, 8). The analysis of JA-dependant defense-associated enzymes and genes (Figs. 2–4) revealed that Si amendment consistently increased the magnitude of induction observed in response to insect herbivory and exogenous MeJA and also indicated decreased response times to these stimuli in specific instances (Figs. 2 and 3). Defense priming often is associated with enhanced or more rapid defense responses occurring subsequent to initial exposures to biotic or abiotic stresses (20). One frequently invoked mechanism for this phenomenon in plants involves the accumulation of inactive cellular proteins involved in signal transduction, such as MAPKs, following initial exposure to stress. Subsequent exposure to stress could lead to the rapid activation of these inert signaling components, thereby increasing the magnitude of host defensive processes and/or the speed with which they are activated (20, 36, 37). In eukaryotes, MAPK cascades are involved in the transduction of diverse stimuli, including abiotic and biotic stresses, and act downstream of various receptors (28). MAPK cascades serve to amplify the initial transducing signals, ultimately leading to the activation of batteries of defensive genes, which frequently are targets of WRKY-type transcription factors (38). Our results revealed that Si amendment alone did not induce expression of the MAPKs OsMPK3 and OsMPK6 or the WRKY transcription factors OsWRKY53 and OsWRKY70; however, upon LF infestation or MeJA application, Si-pretreated plants exhibited enhanced transcriptional responses for these genes as well as more rapid induction in specific instances (Figs. 2 and 3). The lack of induction of OsMPK3, OsMPK6, OsWRKY53, or OsWRKY70 transcripts by Si in the absence of LF exposure or exogenous MeJA suggests that these specific signaling components do not accumulate in inactive form as components of a Si-based defense-priming mechanism, but this possibility cannot be discounted based on monitoring the levels of steady-state transcript accumulation alone (Fig. 3).
Although Si-mediated priming ostensibly provides a means for mounting a more efficient defense response against insect herbivory in rice, it is also somewhat puzzling, in that it appears unlikely that increased soil-soluble Si levels or growth in more highly silicified soils would be predictive for increased likelihood of future encounters with insect herbivores. However, priming of plant emissions of antiherbivore volatile organic compounds by abiotic stimuli such as copper toxicity and water deficit previously has been shown to occur in Zea mays (39, 40). Although the mechanistic details have not been elucidated, it was hypothesized that in the case of copper toxicity overlapping abiotic and biotic stress-signaling mechanisms involving reactive oxygen species could be involved (40). Si is taken up from soil solutions in the form of monomeric silicic acid (H4SiO4) and becomes deposited as amorphous SiO2·nH2O (opal) within silica bodies, intercellular spaces, external surfaces, and cell walls (41, 42). Thus, the possibility cannot be discounted that the interaction of Si, for example with specific cell-wall components, is perceived as a mild stress stimulus in Si-accumulating plants, leading to the potentiation of antiherbivore defenses. Perturbations in cell-wall status previously have been shown to increase both JA and ethylene accumulation significantly in Arabidopsis (43), and such regulatory interactions are likely required to contend with cell-wall damage inflicted by insect herbivore attack (44).
Although the involvement of JA and Si in plant resistance to insect herbivores is well established, details concerning the relationship between JA and Si remain largely obscure. Collectively, our results suggest an interplay between JA and Si in which Si enhances or primes JA-inducible responses to herbivory, including the enhanced induction of defense-related enzymes and proteins, enhanced induction of transcripts encoding proteins involved in JA signaling (Figs. 2–4), and increased accumulation of JA itself following insect attack (Fig. 2E). Leaf bioassays clearly demonstrated enhanced resistance to LF herbivory in Si-treated versus untreated plants and the loss of resistance in OsAOS and OsCOI1 RNAi strains, regardless of Si treatment, indicating that Si effects are JA dependent. As mentioned, a significant body of evidence suggests that Si itself serves as an insect antifeedant, so Si likely plays a role in passive or constitutive defenses as well as a role in promoting JA-mediated inducible defenses. Our results further indicate that the effects of Si are, in turn, modulated by JA, which promotes overall leaf silicification and the maturation of phytolith-bearing silica cells (Fig. 5). Thus, rice, and likely other plants accumulating high levels of Si, may have evolved JA-mediated antiherbivore defense mechanisms in which Si is an integrated component.
Materials and Methods
Generation of Transgenic Plants.
To determine the role of the JA pathway in Si-mediated resistance to insects, RNAi was used to silence the expression of rice OsAOS (GenBank accession no. AY062258) and OsCOI1 (GenBank accession no. DQ028826) genes. Allene oxide synthase (AOS), a cytochrome P450 enzyme of the CYP74A family, converts 13(S)-hydroperoxylinolenic acid to 12,13-epoxylinolenic acid in the first specific reaction leading to JA synthesis. CORONATINE INSENSITIVE 1 (COI1) is an F-box protein essential for jasmonate perception (27, 30). The OsCOI1 RNAi line was generated as described by Ye et al. (30), and the OsAOS RNAi line was generated in a similar way: A 580-bp cDNA fragment of OsAOS first was amplified by RT-PCR using the primers 5′-cgaggatccaccgcctcgatttctactac-3′ and 5′-tctaagcttatcatgggcaggccgagcgt-3′ derived from conserved sequence regions (Fig. S2). RT-PCR conditions were as follows: 95 °C for 4 min, 30 cycles of 95 °C for 30 s, 52 °C for 30 s, 75 °C for 1.5 min, and then extension for 8 min at 72 °C. The OsAOS fragment then was inserted into the intermediate vector pRNAi.5 (kindly provided by Yao-Guang Liu, South China Agricultural University, Wushan, Guangzhou, People's Republic of China) (Fig. S3) between the unique BamHI and HindIII restriction sites. DNA sequencing confirmed both the correct orientation and the cDNA identities (Fig. S4).
Agrobacterium tumefaciens (strain EHA105) harboring binary vectors was used for transformation of rice (Oryza sativa L. cv. Shishoubaimao) according to the method described by Hiei et al. (45). Southern blot analysis was used to detect the copy number of the inserted fragment as described in Ye et al. (30). For all experiments, one T2 homozygous OsAOS and OsCOI1 RNAi line harboring a single insertion (Fig. S5A) was used. Real-time quantitativePCR (qRT-PCR) analysis showed efficient silencing of OsAOS and OsCOI1 in leaves, stems, and roots of the OsAOS and OsCOI1 RNAi lines, respectively (Fig. S5B).
Plant Growth.
Three different genotypes, including WT and OsAOS and OsCOI1 RNAi lines, were used in this work. For all experiments, seeds first were surface-sterilized with 10% H2O2 (vol/vol) for 10 min and were rinsed with distilled water. The seeds were soaked in sterile distilled water for 1 d and then were germinated on moist filter paper for 3 d in Petri dishes at 25 °C. After germination, the seeds were transferred to nets floated on 0.5 mM CaCl2 in plastic containers. At 7 d, six seedlings of uniform size were transferred to polyethylene pots containing sterile vermiculite and 500 mL 0.5× Kimura B nutrient solution containing 2 mM potassium silicate (K2SiO3). For Si-negative treatments, potassium chloride was used to replenish potassium. The plants were maintained in a growth chamber with a day:night temperature regime of 25 °C (12 h):22 °C (12 h) and a light intensity of 300 μmol⋅m−2⋅s−1. Rice plants of different genotypes were used for experiments 20 d after transplanting.
Plant Treatments.
MeJA treatment.
In this study, we used MeJA, which is converted to JA and then to bioactive jasmonoyl-isoleucine in plants (46), to treat rice plants. The OsCOI1/OsAOS RNAi lines and WT rice plants were sprayed with 1 mM MeJA solution prepared in 50 mM sodium phosphate buffer with 0.01% Tween 20, pH adjusted to 8.0 using 1 M citric acid. Control plants were sprayed with buffer lacking MeJA.
LF treatment.
Plants with or without Si amendment were individually infested with third-instar LF larvae that had been starved for 2 h before placement on leaves at node 3 (the youngest fully expanded leaf was defined as leaf 1).
Insects.
LF larvae were collected originally from rice fields of the Dafeng Experimental Base of Guangdong Academy of Agricultural Sciences, Guangzhou, China, and were maintained on WT rice plants in a climate-controlled room at 26 ± 2 °C, 12 h:12 h light:dark regimen, and 80% relative humidity. Larvae at the third-instar stage were used for all bioassays described.
JA Analyses.
WT plants with or without Si amendment were left untreated, were individually infested by LF larvae, or were sprayed with 1 mM MeJA (Fig. 2). Third leaves were harvested from LF- and MeJA-treated plants and untreated plants after 0, 3, 6, 9, 12, and 24 h. Samples were collected, immersed immediately in liquid nitrogen, and then were stored at −80 °C before analysis. Six plants were sampled for each treatment at each time point. Total JA levels, representing the summation of both cis and trans isomers, were determined by gas chromatography as described by Ye et al. (30).
Real-Time qRT-PCR Analysis.
Differential expression of selected genes of WT rice plants in response to Si and MeJA treatments and LF infestation was determined by real-time qRT-PCR. Total RNA was extracted using the RNAisoTN Plus Reagent (TaKaRa) according to the manufacturer’s instructions. First-strand cDNA was synthesized from 1 µg of total RNA using MMLV-reverse transcriptase (Promega) according to the manufacturer’s instructions, and qRT-PCR was performed using SYBR Premix EX TaqTM (TaKaRa) with a DNA Engine Opticon 2 Continuous Fluorescence Detection System (MJ Research Inc.). The rice housekeeping gene OsActin (GenBank accession no. X15865) was used as an endogenous control for sample normalization. The gene-specific primer sequences used are listed in Table S1. Reaction conditions for thermal cycling were 95 °C for 1 min, 40 cycles of 95 °C for 20 s, 58–60 °C for 15 s, and 72 °C for 30 s. Fluorescence data were collected during the cycle at 72 °C. Amplicon specificity was verified by melting curve analysis and agarose gel electrophoresis. Transcripts of targeted genes were calculated by the double-standard curves method. Three independent biological replicates for each treatment were used for qRT-PCR analyses.
LF Bioassays.
OsAOS and OsCOI1 RNAi lines and WT rice plants treated with or without Si as described above were sprayed with 1 mM MeJA or buffer (control). Two days after treatment, node 3 leaves on each plant were individually infested with third-instar LF larvae that had been weighed and starved for 2 h. All larvae were weighed after 3 d of infestation. The percentages of mass gain of larvae on each plant were calculated. Twenty plants of each genotype were used for each treatment.
Enzyme Activity Assays and TrypPI Analyses.
OsAOS and OsCOI1 RNAi lines and WT rice plants with or without Si amendment were assigned to LF infestation or MeJA (1 mM) treatment or were left untreated (control). The node 3 leaves (0.2 g) were harvested 24 h after treatment, immersed immediately in liquid nitrogen, and stored at −80 °C for analysis of enzyme activity, TrypPI levels, and relative expression of OsLsi1, OsLsi12, and OsLsi16. POD activities were determined as described by Kraus and Fletcher (47). PPO activity was assayed with 0.05 M catechol as substrate, as described by Zauberman et al. (48). TrypPI levels were measured using a colorimetric assay, as described by Ye et al. (30).
Determination of Si Concentration in Leaves.
The OsAOS and OsCOI1 RNAi lines and WT plants treated with Si were sprayed with 1 mM MeJA or buffer (control), as described. After 2 d, node 3 leaves were harvested for analysis of Si content and morphological observations of silica bodies on leaf surfaces. The Si content of rice leaves was determined by the colorimetric molybdenum blue method described by van der Vorm (49). Leaves (0.3 g) were ashed in porcelain crucibles for 3 h at 550 °C and then were dissolved in 1.3% (vol/vol) hydrogen fluoride. Si concentrations in the resulting solutions were measured at 811 nm using a spectrophotometer.
Microscopy.
Fresh leaf samples were prepared as described by Cai et al. (10). The morphology of silica bodies on leaf surfaces of rice was observed by a XL 30 ESEM (Philips) equipped with advanced image analysis software for detecting silica bodies. Ten pictures for each replicate were analyzed randomly. Magnifications of 2,000× were used to quantify silica cells; the size (length and width) of the Si cell was measured, and then the Si area (in square micrometers) was calculated.
Data Analysis.
The SPSS 14.0 (SPSS) package for Windows was used for statistical analyses. Differences in JA content between Si-treated and untreated WT plants were determined using Student t tests. Other data were evaluated by factorial ANOVA with treatment differences among means tested at P = 0.05 using a Tukey post hoc test. For both statistical tests, differences among means were compared across all treatments included within a given experiment.
Supplementary Material
Acknowledgments
We thank Professor Yao-Guang Liu for providing the pRNAi.5 vector and Dr. May Berenbaum and Dr. Ron A. Salzman for comments on the manuscript. This research was supported financially by the National 973 Project of China Grant 2011CB100400, National Natural Science Foundation of China Grants 31070388, 31028018, and 31100286, the Guangdong Province Universities and Colleges Pearl River Scholar Funded Scheme (2010), and the Ph.D. Programs Foundation of the Ministry of Education of China Grant 20104404110004.
Footnotes
The authors declare no conflict of interest.
This article is a PNAS Direct Submission.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1305848110/-/DCSupplemental.
References
- 1.Epstein E. Silicon. Annu Rev Plant Physiol Plant Mol Biol. 1999;50:641–664. doi: 10.1146/annurev.arplant.50.1.641. [DOI] [PubMed] [Google Scholar]
- 2.Ma JF. Role of silicon in enhancing the resistance of plants to biotic and abiotic stresses. Soil Sci Plant Nutr. 2004;50(1):11–18. [Google Scholar]
- 3.Watanabe S, et al. Identification of several rice genes regulated by Si nutrition. Soil Sci Plant Nutr. 2004;50(8):1273–1276. [Google Scholar]
- 4.Fauteux F, Chain F, Belzile F, Menzies JG, Bélanger RR. The protective role of silicon in the Arabidopsis-powdery mildew pathosystem. Proc Natl Acad Sci USA. 2006;103(46):17554–17559. doi: 10.1073/pnas.0606330103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kvedaras OL, Keeping MG. Silicon impedes stalk penetration by the borer Eldana saccharina in sugarcane. Entomol Exp Appl. 2007;125(1):103–110. [Google Scholar]
- 6.Kvedaras OL, An M, Choi YS, Gurr GM. Silicon enhances natural enemy attraction and biological control through induced plant defences. Bull Entomol Res. 2010;100(3):367–371. doi: 10.1017/S0007485309990265. [DOI] [PubMed] [Google Scholar]
- 7.Reynolds OL, Keeping MG, Meyer JH. Silicon-augmented resistance of plants to herbivorous insects: A review. Ann Appl Biol. 2009;155(2):171–186. [Google Scholar]
- 8.Keeping MG, Meyer JH. Silicon-mediated resistance of sugarcane to Eldana saccharina Walker (Lepidoptera: Pyralidae): Effects of silicon source and cultivar. J Appl Entomol. 2006;130(8):410–420. [Google Scholar]
- 9.Liang Y, Chen Q, Liu Q, Zhang W, Ding R. Exogenous silicon (Si) increases antioxidant enzyme activity and reduces lipid peroxidation in roots of salt-stressed barley (Hordeum vulgare L.) J Plant Physiol. 2003;160(10):1157–1164. doi: 10.1078/0176-1617-01065. [DOI] [PubMed] [Google Scholar]
- 10.Cai KZ, et al. Physiological and cytological mechanisms of silicon-induced resistance in rice against blast disease. Physiol Plant. 2008;134(2):324–333. doi: 10.1111/j.1399-3054.2008.01140.x. [DOI] [PubMed] [Google Scholar]
- 11.Fawe A, Abou-Zaid M, Menzies JG, Bélanger RR. Silicon-mediated accumulation of flavonoid phytoalexins in cucumber. Phytopathology. 1998;88(5):396–401. doi: 10.1094/PHYTO.1998.88.5.396. [DOI] [PubMed] [Google Scholar]
- 12.Rodrigues FA, et al. Silicon enhances the accumulation of diterpenoid phytoalexins in rice: A potential mechanism for blast resistance. Phytopathology. 2004;94(2):177–183. doi: 10.1094/PHYTO.2004.94.2.177. [DOI] [PubMed] [Google Scholar]
- 13.Rémus-Borel W, Menzies JG, Bélanger RR. Silicon induces antifungal compounds in powdery mildew-infected wheat. Physiol Mol Plant Pathol. 2005;66(3):108–115. [Google Scholar]
- 14.Mithöfer A, Boland W. Plant defense against herbivores: Chemical aspects. Annu Rev Plant Biol. 2012;63:431–450. doi: 10.1146/annurev-arplant-042110-103854. [DOI] [PubMed] [Google Scholar]
- 15.Browse J, Howe GA. New weapons and a rapid response against insect attack. Plant Physiol. 2008;146(3):832–838. doi: 10.1104/pp.107.115683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Howe GA, Jander G. Plant immunity to insect herbivores. Annu Rev Plant Biol. 2008;59:41–66. doi: 10.1146/annurev.arplant.59.032607.092825. [DOI] [PubMed] [Google Scholar]
- 17.Schmelz EA, Engelberth J, Alborn HT, Tumlinson JH, 3rd, Teal PEA. Phytohormone-based activity mapping of insect herbivore-produced elicitors. Proc Natl Acad Sci USA. 2009;106(2):653–657. doi: 10.1073/pnas.0811861106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wu JQ, Baldwin IT. New insights into plant responses to the attack from insect herbivores. Annu Rev Genet. 2010;44:1–24. doi: 10.1146/annurev-genet-102209-163500. [DOI] [PubMed] [Google Scholar]
- 19.Frost CJ, Mescher MC, Carlson JE, De Moraes CM. Plant defense priming against herbivores: Getting ready for a different battle. Plant Physiol. 2008;146(3):818–824. doi: 10.1104/pp.107.113027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Conrath U. Molecular aspects of defence priming. Trends Plant Sci. 2011;16(10):524–531. doi: 10.1016/j.tplants.2011.06.004. [DOI] [PubMed] [Google Scholar]
- 21.van Hulten M, Pelser M, van Loon LC, Pieterse CM, Ton J. Costs and benefits of priming for defense in Arabidopsis. Proc Natl Acad Sci USA. 2006;103(14):5602–5607. doi: 10.1073/pnas.0510213103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Peng J, van Loon JJA, Zheng S, Dicke M. Herbivore-induced volatiles of cabbage (Brassica oleracea) prime defence responses in neighbouring intact plants. Plant Biol (Stuttg) 2011;13(2):276–284. doi: 10.1111/j.1438-8677.2010.00364.x. [DOI] [PubMed] [Google Scholar]
- 23.Kim J, Tooker JF, Luthe DS, De Moraes CM, Felton GW. Insect eggs can enhance wound response in plants: A study system of tomato Solanum lycopersicum L. and Helicoverpa zea Boddie. PLoS ONE. 2012;7(5):e37420. doi: 10.1371/journal.pone.0037420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rasmann S, et al. Herbivory in the previous generation primes plants for enhanced insect resistance. Plant Physiol. 2012;158(2):854–863. doi: 10.1104/pp.111.187831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ma JF, Tamai K, Ichii M, Wu GF. A rice mutant defective in Si uptake. Plant Physiol. 2002;130(4):2111–2117. doi: 10.1104/pp.010348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ma JF, et al. A silicon transporter in rice. Nature. 2006;440(7084):688–691. doi: 10.1038/nature04590. [DOI] [PubMed] [Google Scholar]
- 27.Yan JB, et al. The Arabidopsis CORONATINE INSENSITIVE1 protein is a jasmonate receptor. Plant Cell. 2009;21(8):2220–2236. doi: 10.1105/tpc.109.065730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Skibbe M, Qu N, Galis I, Baldwin IT. Induced plant defenses in the natural environment: Nicotiana attenuata WRKY3 and WRKY6 coordinate responses to herbivory. Plant Cell. 2008;20(7):1984–2000. doi: 10.1105/tpc.108.058594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rodriguez MCS, Petersen M, Mundy J. Mitogen-activated protein kinase signaling in plants. Annu Rev Plant Biol. 2010;61:621–649. doi: 10.1146/annurev-arplant-042809-112252. [DOI] [PubMed] [Google Scholar]
- 30.Ye M, et al. silencing COI1 in rice increases susceptibility to chewing insects and impairs inducible defense. PLoS ONE. 2012;7(4):e36214. doi: 10.1371/journal.pone.0036214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rakwal R, Kumar Agrawal G, Jwa NS. Characterization of a rice (Oryza sativa L.) Bowman-Birk proteinase inhibitor: Tightly light regulated induction in response to cut, jasmonic acid, ethylene and protein phosphatase 2A inhibitors. Gene. 2001;263(1-2):189–198. doi: 10.1016/s0378-1119(00)00573-4. [DOI] [PubMed] [Google Scholar]
- 32.Yamaji N, Mitatni N, Ma JF. A transporter regulating silicon distribution in rice shoots. Plant Cell. 2008;20(5):1381–1389. doi: 10.1105/tpc.108.059311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wasternack C, Forner S, Strnad M, Hause B. Jasmonates in flower and seed development. Biochimie. 2013;95(1):79–85. doi: 10.1016/j.biochi.2012.06.005. [DOI] [PubMed] [Google Scholar]
- 34.Li L, et al. The tomato homolog of CORONATINE-INSENSITIVE1 is required for the maternal control of seed maturation, jasmonate-signaled defense responses, and glandular trichome development. Plant Cell. 2004;16(1):126–143. doi: 10.1105/tpc.017954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Prychid CJ, Rudall PJ, Gregory M. Systematics and biology of silica bodies in monocotyledons. Bot Rev. 2004;69(4):377–440. [Google Scholar]
- 36.Beckers GJ, et al. Mitogen-activated protein kinases 3 and 6 are required for full priming of stress responses in Arabidopsis thaliana. Plant Cell. 2009;21(3):944–953. doi: 10.1105/tpc.108.062158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Conrath U, et al. Prime-A-Plant Group Priming: Getting ready for battle. Mol Plant Microbe Interact. 2006;19(10):1062–1071. doi: 10.1094/MPMI-19-1062. [DOI] [PubMed] [Google Scholar]
- 38.Eulgem T. Regulation of the Arabidopsis defense transcriptome. Trends Plant Sci. 2005;10(2):71–78. doi: 10.1016/j.tplants.2004.12.006. [DOI] [PubMed] [Google Scholar]
- 39.Gouinguené SP, Turlings TCJ. The effects of abiotic factors on induced volatile emissions in corn plants. Plant Physiol. 2002;129(3):1296–1307. doi: 10.1104/pp.001941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Winter TR, Borkowski L, Zeier J, Rostás M. Heavy metal stress can prime for herbivore-induced plant volatile emission. Plant Cell Environ. 2012;35(7):1287–1298. doi: 10.1111/j.1365-3040.2012.02489.x. [DOI] [PubMed] [Google Scholar]
- 41.Epstein E. The anomaly of silicon in plant biology. Proc Natl Acad Sci USA. 1994;91(1):11–17. doi: 10.1073/pnas.91.1.11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Cooke J, Leishman MR. Is plant ecology more siliceous than we realise? Trends Plant Sci. 2011;16(2):61–68. doi: 10.1016/j.tplants.2010.10.003. [DOI] [PubMed] [Google Scholar]
- 43.Ellis C, Karafyllidis I, Wasternack C, Turner JG. The Arabidopsis mutant cev1 links cell wall signaling to jasmonate and ethylene responses. Plant Cell. 2002;14(7):1557–1566. doi: 10.1105/tpc.002022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wolf S, Hématy K, Höfte H. Growth control and cell wall signaling in plants. Annu Rev Plant Biol. 2012;63:381–407. doi: 10.1146/annurev-arplant-042811-105449. [DOI] [PubMed] [Google Scholar]
- 45.Hiei Y, Ohta S, Komari T, Kumashiro T. Efficient transformation of rice (Oryza sativa L.) mediated by Agrobacterium and sequence analysis of the boundaries of the T-DNA. Plant J. 1994;6(2):271–282. doi: 10.1046/j.1365-313x.1994.6020271.x. [DOI] [PubMed] [Google Scholar]
- 46.Browse J. Jasmonate passes muster: A receptor and targets for the defense hormone. Annu Rev Plant Biol. 2009;60:183–205. doi: 10.1146/annurev.arplant.043008.092007. [DOI] [PubMed] [Google Scholar]
- 47.Kraus TE, Fletcher RA. Paclobutrazol protects wheat seedlings from heat and 16 paraquat injury Is detoxification of active oxygen involved? Plant Cell Physiol. 1994;35(1):45–52. [Google Scholar]
- 48.Zauberman G, et al. Post-harvest retention of the red colour of litchi fruit pericarp. Sci Hortic (Amsterdam) 1991;47(1-2):89–97. [Google Scholar]
- 49.Van der Vorm PDJ. Dry ashing of plant material and dissolution of the ash in HF for the colorimetric determination of silicon. Commun Soil Sci Plant Anal. 1987;18(11):1181–1189. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.