Abstract
Background
Binary toxin-producing Clostridium difficile infections (CDI) are known to be more severe and to cause higher case fatality rates than those by binary toxin-negative isolates. There has been few data of binary toxin-producing CDI in Korea. Objective of the study is to characterize clinical and microbiological trait of CDI cause by binary-toxin producing isolates in Korea.
Materials and Methods
From September 2008 through January 2010, clinical characteristics, medication history and treatment outcome of all the CDI patients were collected prospectively. Toxin characterization, PCR ribotyping and antibiotic susceptibility were performed with the stool isolates of C. difficile.
Results
During the period, CDI caused by 11binary toxin-producing isolates and 105 toxin A & toxin B-positive binary toxin-negative isolates were identified. Comparing the disease severity and clinical findings between two groups, leukocytosis and mucoid stool were more frequently observed in patients with binary toxin-positive isolates (OR: 5.2, 95% CI: 1.1 to 25.4, P = 0.043; OR: 7.6, 95% CI: 1.6 to 35.6, P = 0.010, respectively), but clinical outcome of 2 groups did not show any difference. For the risk factors for acquisition of binary toxin-positive isolates, previous use of glycopeptides was the significant risk factor (OR: 6.2, 95% CI: 1.4 to 28.6, P = 0.019), but use of probiotics worked as an inhibitory factor (OR: 0.1, 95% CI: 0.0 to 0.8; P = 0.026). PCR ribotypes of binary toxinproducing C. difficile showed variable patterns: ribotype 130, 4 isolates; 027, 3 isolates; 267 and 122, 1 each isolate and unidentified C1, 2 isolates. All 11 binary toxin-positive isolates were highly susceptible to clindamycin, moxifloxacin, metronidazole, vancomycin and piperacillin-tazobactam, however, 1 of 11 of the isolates was resistant to rifaximin.
Conclusions
Binary toxin-producing C. difficile infection was not common in Korea and those isolates showed diverse PCR ribotypes with high susceptibility to antimicrobial agents. Glycopeptide use was a risk factor for CDI by those isolates.
Keywords: Clostridium difficile, Binary toxin, Clinical characteristics, PCR ribotype, Susceptibility
Introduction
Some strain of Clostridium difficile isolates produce the binary toxin, C. difficile transferase (CDTa) in addition to pathogenic toxins like toxin A and toxin B. About 6-12.5% of isolates produce binary toxin. Since 2002 in North America and Europe, the proportion of binary toxin-producing strains has increased to 30-65% of all isolates [1, 2]. In Korea, the proportion of isolated binary toxin-producing strains is low with 4-7%, even after 2009 when these hypervirulent binary toxin strains were first reported [3, 4].
The pathogenicity of binary toxin is not clear yet. Binary toxin is an ADP-ribosylating toxin which disaggregates actin cytoskeleton, and induces effusion in intestinal epithelial cell cultures and changes the cells to a round shape which in turn causes apoptosis [5]. However, when binary toxin was given to hamsters, the colonization of the isolates occurred in 70-80% of the hamsters but they did not manifest diarrhea or expire [6]. From a recent report, binary toxin plays an important role in the immediate colonization of C. difficile by creating a thin microtubule in the surface of intestinal epithelial cells to assist in the easy adherence of C. difficile onto the surface of intestinal cells [7].
There are various ribotype strains which produce binary toxin. The hypervirulent strain, BI/NAP1/027 is a prevalent strain in North America and is known to significantly increase morbidity and mortality in infected patients compared to other stains [8]. In addition to the production of binary toxin, variations in tcdC, which is the regulatory gene of toxin A and toxin B, increases its virulence with an increase in the production and duration of the toxin [9]. This epidemic strain is resistant to fluoroquinolone; hence, the recent increase in the use of respiratory fluoroquinolone is expected to be an important selection pressure [10].
Recently, there was a case of the BI/NAP1/027 strain in Korea [11], but there are no data on the binary toxin-producing strain in general. Therefore, the authors conducted this study to find out the general current circumstances of binary toxin-producing C. difficile infection (CDI) in Korea. In this study, the clinical manifestations and risk factors for acquisition of binary toxin-producing CDI was compared to toxin A and B positive strain infections. Moreover, the distribution of PCR ribotypes in binary toxin-producing strains in Korea was identified along with the major results of antimicrobial susceptibility tests.
Materials and Methods
1. Study subjects
The study subjects were patients 18 years or older who were diagnosed as CDI with cultivated C. difficile in stool cultures and admitted to Hanyang University Seoul Hospital from September 2008 to January 2010. The toxin producing genes from the C. difficile isolates were confirmed and the patients were divided according to presence of binary toxin gene. The control group (A+B+CDT- group) included patients infected with a toxin A-positive, toxin B-positive, binary toxin-negative strain, and the patient group (A+B+CDT+ group) included patients infected with a toxin A-positive, toxin B-positive, binary toxin-positive strain. This study was approved by the institutional review board of Hanyang University Hospital (HYUH IRB 2010-R-12). Informed consent was waived by the board.
2. Diagnosis related definitions and data collection
A diagnosis of CDI was made in the following cases: C. difficile toxin A&B assay (VIDAS® C. difficile toxin A&B; BioMerieux SA, Marcy I'Etoile, France) positive in patients who complained of unformed stools 3 times daily or for more than 2 days or longer, otherwise 6 times or more within 36 hours; the confirmation of toxin producing genes from the isolated C. difficile in stool cultures; or psuedomembranes on the endoscopy or biopsy [12].
Age, sex, length of stay in the hospital, underlying disease including Charlson's score, and catheter utilization were collected for the clinical data [13]. The admission history of the past 2 months, operation history, history of antibiotic use and the dose were collected and use of proton pump inhibitor (PPI), H2 blocker (H2B) and probiotics were checked. Amounts of antibiotics administered were calculated as the number of defined daily dose (DDD) [14]. The values for the white blood cell (WBC) level, albumin level, and C-reactive protein (CRP) at the time of diagnosis were recorded and the presence of the pseudomembrane was checked through endoscopy.
For severity assessment, a patient was over 60 years old, temperature over 38.3℃, WBC over 15,000 cells/mm3, and albumin less than 2.5 mg/dL, respectively, was given 1 point and added up. Then, severe CDI was defined as when the total points were 2 points or greater [15, 16]. For clinical outcome, success was defined as improvement in diarrhea and symptoms within 14 days of the initiation of treatment while other cases were regarded as failure [17]. Recurrence was defined as the manifestation of symptoms of CDI after the improvement of symptoms from day 10 after treatment initiation, and a global cure was defined when there was no recurrence in patients who received the initial treatment [18].
3. Microbiology tests
1) C. difficile culture
After alcohol shock treatment, stool specimen were cultivated on CDMN-TA agar (C. difficile Moxalactam-Norfloxacin-Taurocholate agar, Oxoid Ltd., Cambridge, UK) supplemented with 7% horse blood. Colonies of C. difficile were identified by Rapid ID 32A (BioMerieux SA, Marcy I'Etoile, France).
2) Multiplex PCR
To identify toxin genes, multiplex PCR was performed with template DNA. After electrophoresis of the amplified products, toxin genes (tcdA, tcdB, cdtA, and cdtB) were defined according to band size [19].
3) PCR ribotyping of C. difficile strains
With minor modifications, PCR ribotyping was performed as described elsewhere [3]. After electrophoresis of the PCR products, the pattern was checked visually. Ribotype 027 (BI/NAP1/027), ATCC 43598 (ribotype 017) and standard strains from the ECDC-Brazier collection were used for the reference [3].
4) Antimicrobial susceptibility test
Antimicrobial susceptibility tests for the subject strains were conducted with the following antibiotics: clindamycin, moxifloxacin, vancomycin, metronidazole, piperacillin-tazobactam and rifaximin [20]. Breakpoints for each antimicrobial agent were defined by CLSI guideline. C. difficile ATCC 700057 served as a quality control strain.
4. Statistical analysis
SPSS 18.0 version (SPSS, Chicago, IL, USA) was used for statistical analysis. Pearson's Chi-square test or Fisher's exact test was used for the analysis of categorical variables, and independent t-test or Mann-Whitney U-test was used for the analysis of continuous variables. Multiple logistic regressions were performed to identify risk factors for acquisition of binary toxin producing strains and clinical difference according to presence of binary toxin gene. A P-value of < 0.05 by two-tailed test was considered statistically significant.
Results
1. Clinical Characteristics
One hundred and five cases of A+B+CDT- groups and 11 cases of A+B+CDT+ groups were identified during the study, and the clinical characteristics were compared and analyzed between the 2 groups.
1) Comparisons of demographic characteristics and risk factors (Table 1)
Table 1.
Demographic and clinical characteristics of patients infected by binary toxin-positive Clostridium difficile compared with those by binary toxin-negative toxin A and toxin B-positive isolates
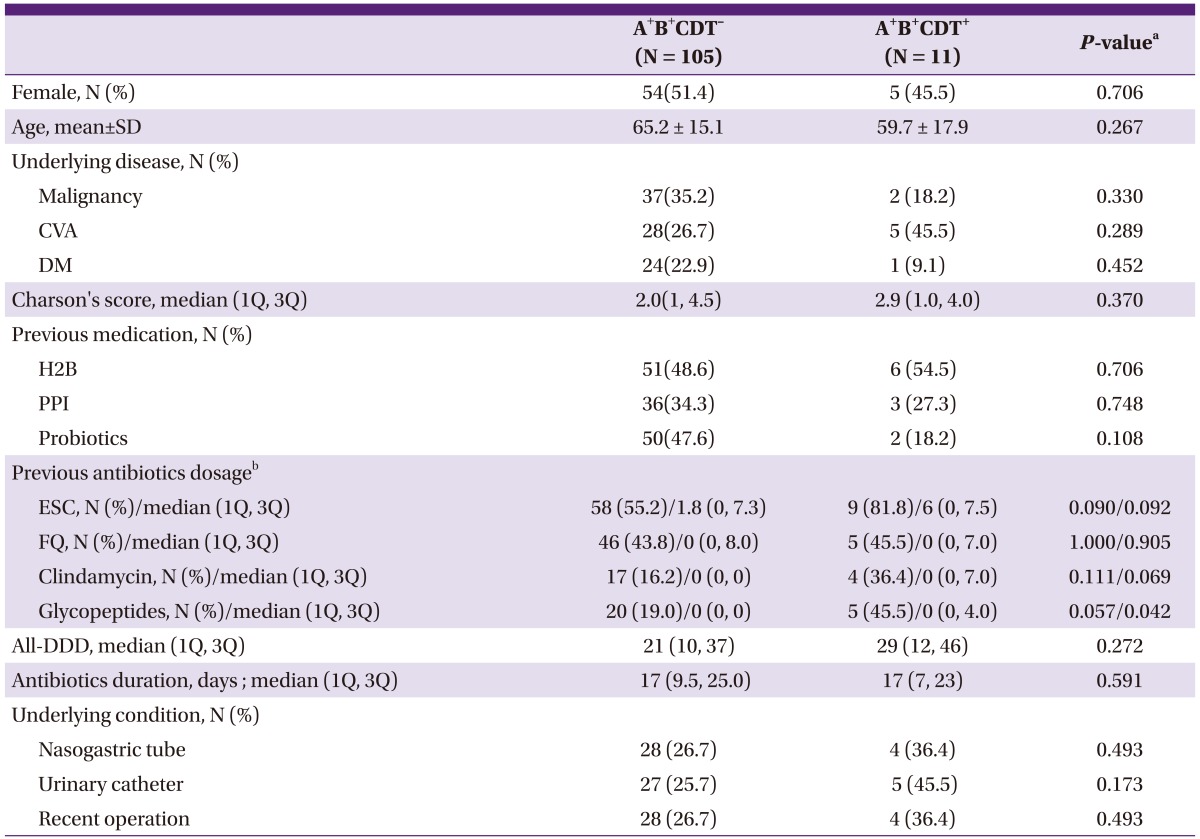
A+B+CDT-, toxin A and toxin B-positive binary toxin-negative; A+B+CDT+, toxin A and toxin B-positive binary toxin-positive; CVA, cerebrovascular accident; DM, diabete mellitus; H2B, H 2 receptor blocker; PPI, proton pump inhibitor; ESC, extended spectrum cephalosporins; FQ, fluoroquinolones; DDD, defined daily dose.
aBy Pearson's Chi-square test or Fisher's exact test for categorical variables and by Student t-test or Mann-Whitney U-test for continuous variables.
bPrevious antibiotics within 2 months prior to Clostridium difficile infection development.
Patient age, sex, underlying disease and operation history and the length of stay in hospital were similar between two groups. From the previous medication history, PPI and probiotics were administered more frequently in the A+B+CDT- group, but there was no statistically significant difference. According to classes of antimicrobial agents, the glycopeptide class was administered more frequently (19.0% vs 45.5%, P = 0.057) and in higher doses (P = 0.042) in the A+B+CDT+ group. Clindamycin was administered more frequently in the A+B+CDT+ group, but there was no statically significant difference (P = 0.069).
2) Comparisons of clinical features (Table 2)
Table 2.
Comparison of clinical findings of C. difficile infections of A+B+CDT- isolates and A+B+CDT+ isolates
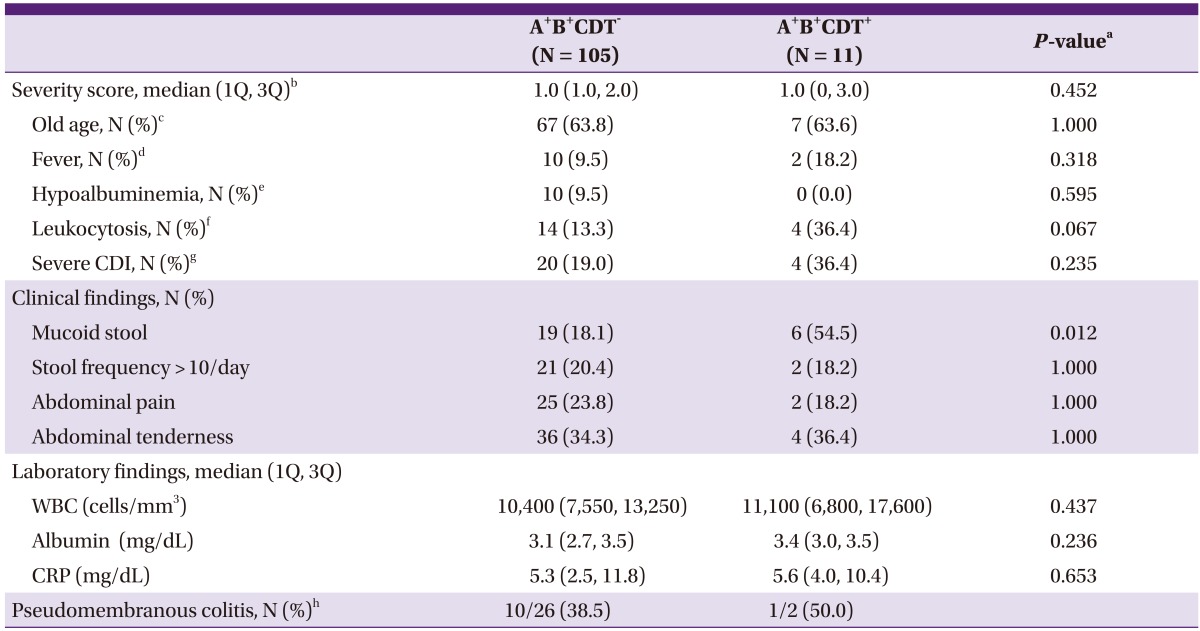
A+B+CDT-, toxin A and toxin B-positive binary toxin-negative; A+B+CDT+, toxin A and toxin B-positive binary toxin-positive; CDI, Clostridium difficile infection.
aBy Fisher's exact test for categorical variables and by Mann-Whitney U-test for continuous variables.
bSeverity score is sum of 4 point; old age, fever, hypoalbuminemia, and leukocytosis got 1 point each.
cDefined as an age of > 60 years.
dDefined as a temperature of > 38.3℃.
eDefined as an albumin level of < 2.5 mg/dL.
fDefined as a WBC count of > 15,000 cells/mm3.
gMore than 2 points of severity score regarded as severe CDI.
hThe denominator comprised the patients who were performed endoscopy.
The severity of CDI between the 2 groups was compared (Table 2). The incidence of leukocytosis was higher in the A+B+CDT+ group (36.4% vs. 13.3%)but there was no statistically significant difference in WBC counts (P = 0.067). Percentage (%) of old age, fever, and hypoalbuminemia also were not different between groups. The incidence of severe CDI was 36.4% and 19.0% in the A+B+CDT+ group and A+B+CDT- group, respectively, but there was no statically significant difference. Propertional pattients with mucoid stool was greater in the A+B+CDT+ group with statistical significance (18.1% vs. 54.5%, P = 0.012). However, there was no significant difference between the 2 groups in stool frequency, abdominal pain, and tenderness. There were no significant differences between the 2 groups in albumin values and CRP. The confirmed cases of pseudomembranous colitis among the patients who had endoscopy done were 38.5% (10/26) and 50.0% (1/2) in the A+B+CDT- group and A+B+CDT+ group, respectively, in which the A+B+CDT+ group had an odds ratio of 1.6 relative to the A+B+CDT- group (95% Confidence Interval (CI: 0.1 to 28. 6).
3) Clinical outcome (Table 3)
Table 3.
Comparison of clinical outcome between Clostridium difficile infected patients with binary toxin-positive, toxin A & toxin B-positive strains and those with binary toxin-negative, toxin A & toxin B-positive strains
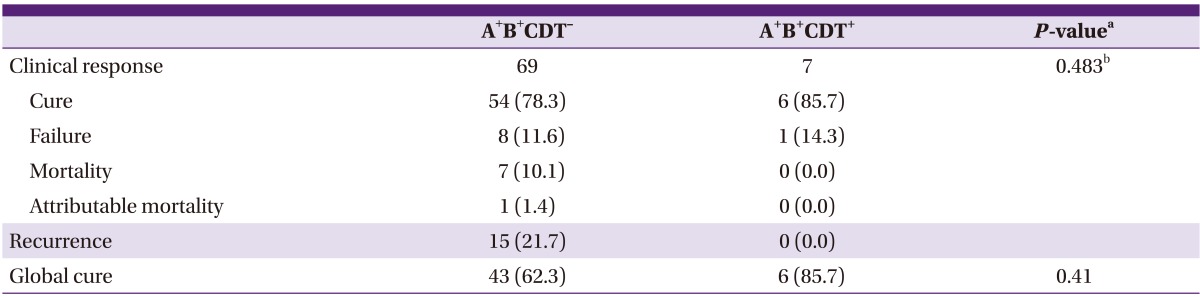
A+B+CDT-, toxin A and toxin B-positive binary toxin-negative; A+B+CDT+, toxin A and toxin B-positive binary toxin-positive.
aBy Fisher's exact test for categorical variables and by Mann-Whitney U-test for continuous variables.
bP for trend.
There was no difference in the proportion of treated patients (65.7% in the A+B+CDT- group vs. 63.6% in the A+B+CDT+ group, P > 0.99). As the initial drug for treatment, 86.8% of CDI patients were received metronidazole. There was no difference between the 2 groups for the initial drug used in the treatment (87.0% in the A+B+CDT- group vs. 85.7% in the A+B+CDT+ group, P > 0.99). From a comparison of the mortality rate and attributable mortality rate, the mortality rate was 10.1% and the attributable mortality rate was 1.4% in the A+B+CDT- group, and there were no cases of death in the A+B+CDT+ group; however, there was no statistical difference. Recurrence was lower in the A+B+CDT+ group (0% vs. 21.7%), and global cure was higher in the A+B+CDT+ group (85.7% vs. 62.3%), but both results did not have any statistically significant differences.
4) Analysis of risk factors and predictable factors in the acquisition of binary toxin producing strains (Table 4)
Table 4.
Multiple logistic regressions of risk factors associated with previous medication for binary toxin producing Clostridium difficile infection
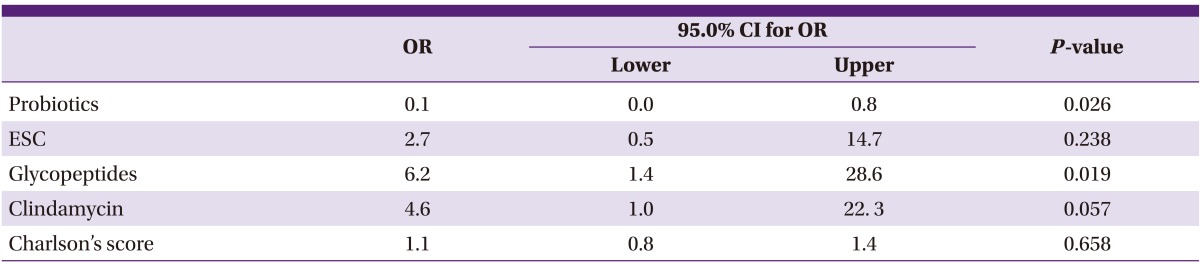
OR, odds ratio; CI, confidence interval; ESC, extended spectrum cephalosporins.
To evaluate the risk factor associated with previous medication within 2 months for binary toxin producing CDI, multiple logistic regressions were performed. In this model, Charlson's score was adjusted for the risk of medication use due to severe underlying disease. The use of glycopeptides and the risk of A+B+CDT+ strain acquisition were statistically significant (OR: 6.2, 95% CI: 1.4 to 28.6, P = 0.019). There was a correlation with the use of clindamycin and A+B+CDT+ strain acquisition, but it was not statistically significant (OR: 4.6, 95% CI: 1.0 to 22. 3, P = 0.057), and the use of probiotics showed a statistically significant inhibitory effect on the acquisition of the A+B+CDT+ strain (OR: 0.1, 95% CI: 0.0 to 0.8, P = 0.026).
To determine whether the clinical findings or laboratory findings at the time of diagnosis can predict CDI caused by binary toxin-producing strains, multiple logistic regressions were performed. Age, fever, leukocytosis and albumin level, as well as cases with mucoid stool which showed a significant difference in cross tabulation analysis were included. The odds ratio of leukocytosis and mucoid stool in binary toxin-producing CDI was 5.2 times (95% CI: 1.1 to 25.4, P = 0.043) and 7.6 times (95% CI: 1.6 to 35.6, P = 0.010), respectively (Table 5).
Table 5.
Multiple logistic regressions of risk factors associated with clinical and laboratory findings for binary toxin producing Clostridium difficile infection
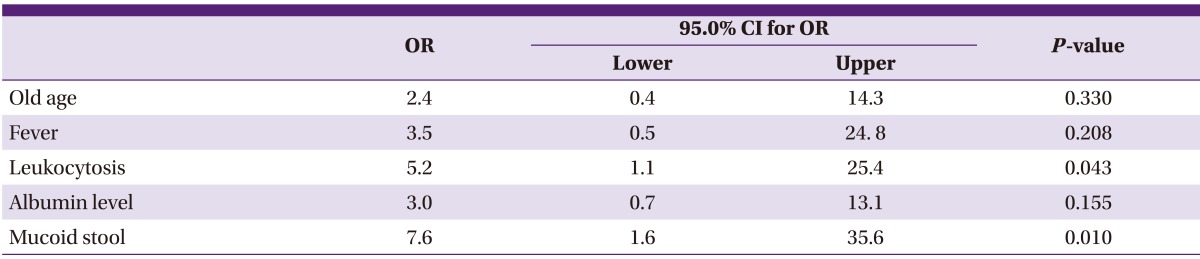
OR, odds ratio; CI, confidence interval.
2. Microbiological characteristics
1) PCR ribotype distribution of binary toxin-producing C. difficile strains
The PCR ribotypes of a total of 11 binary toxin-producing strains were identified. A total of 5 types of PCR ribotypes were identified with 4 strains of ribotype 130, 3 strains of ribotype 027, 1 strain of ribotype 267, and 1 strain of ribotype 122 were identified. Two strains had a new PCR ribotype which is not listed on the ECDC-Brazier collection, and they were classified as C1 according to the classification by Kim et al. [4].
2) Antimicrobial susceptibility of binary toxin-producing strains
The antimicrobial susceptibility of the 11 A+B+CDT+ and A+B+CDT- strains was compared (Table 6) [20]. The A+B+CDT- strains had a high resistance rate for clindamycin and moxifloxacin, 69% and 63%, respectively, but the A+B+CDT+ strains were susceptible to both clindamycin and moxifloxacin. One case out of the 11 A+B+CDT+ strains manifested resistance to rifaximin with the patient from which the isolate was from having no history of rifaximin use; hence, the resistance rate was 9.1%.
Table 6.
Antimicrobial susceptibility of 11 binary toxin producing Clostridium difficile strainsa
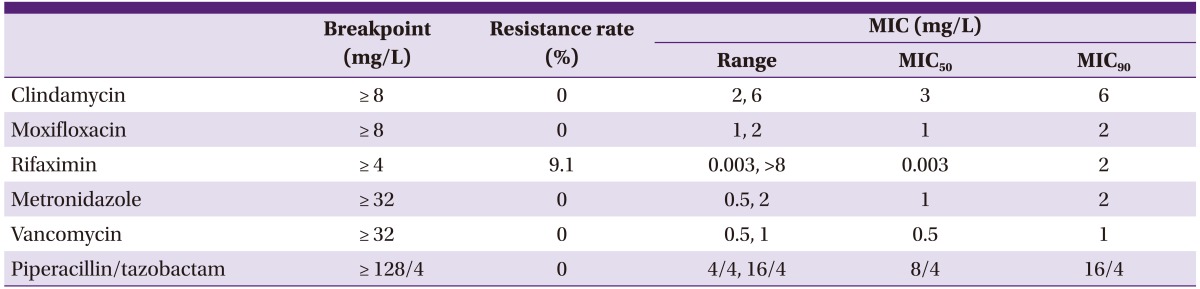
MIC, minimal inhibitory concentration.
aResults of antimicrobial susceptibility test was reproduced with a different description and perspective from References [20].
Discussion
There has been a remarkable increase in the binary toxin-producing BI/NAP1/027 strain in North America since 2002. On the other hand, binary toxin-producing strains have been identified sporadically in Korea. Data on the features of infections, distribution of PCR ribotypes, and antimicrobial susceptibility tests for binary toxin-producing CDI other than 027 strains are not available [21]. Therefore, the authors investigated the clinical impact and microbiologic characteristics of binary toxin-producing strains isolated from a hospital. Binary toxin-producing strains in Korea showed various ribotypes including the 027 strain, and the proportion of the BI/NAP1/027 strain among the entire binary toxin-producing strains was 15-27% and 5% among the entire C. difficile strains.
Generally in North America and Europe, the frequency of the BI/NAP1/027 strain among binary toxin-producing strains varies from 20-80%, and includes 30-60% of all C. difficile strains [22, 23]. The increase in those binary toxin-producing strains was determined as the major cause of the increase in the CDI incidence since 2002 [9].
The epidemic BI/NAP1/027 strain shows a resistant pattern against respiratory fluoroquinolones such as moxifloxacin, and recently, the resistant 027 strain is increasing due to selective pressure with the increase in use of those medications [10]. In this respect, there was a report on the epidemics of ribotype 018 and 017 in Korea correlated with the high resistant rate to moxifloxacin and clindamycin [3]. However, binary toxin-producing strains in this study were highly susceptible to antimicrobial agents and could not have survival benefits in a hospital environment. This would be the reason for the low incidence of binary toxin-producing strains in hospitals in Korea. Ribotype 027 in Korea, which is highly susceptible to antimicrobials, was presumed to be a historical strain that is different to the recent epidemic strains. However, from a recent study, resistance to moxifloxacin was observed in the 027 strain in Korea (6/7, 85%); hence, the possibility of an increase in CDI incidence or outbreak in the future should be considered [21].
Infection due to binary toxin-producing strains is known to have higher mortality relative to binary toxin-negative strain infections, and increases the mortality and attributable mortality [24, 25]. In infections due to binary toxin-producing strains, abdominal pain and diarrhea were more severe [24], the incidence of severe CDI more frequent, and mortality 2.5 times higher [25]. Some contradicting reports have been published [26], but mortality from infection due to binary toxin-producing strains would be markedly high because the mortality rate of CDI due to the historical strains was 3.0-3.5% and the CDI mortality rate in Canada and the United States where binary toxin-producing strains are endemic is 13-14% [27-29]. Bacci et al. investigated the 30-day mortality by comparing 193 cases of PCR ribotype 027 and 72 cases of other ribotypes among the binary toxin-producing strains to 212 infection cases of the A+B+CDT- strain, and as a result, the additional mortality from the infection due to the binary toxin-producing strains was up to 60%, regardless of the ribotype (OR: 1.60, 95% CI: 1.0 to 2.4) [23]. In this study, the frequency of leukocytosis and mucoid stool were high in binary toxin-producing CDI. However, the overall incidence of severe CDI was not different between the 2 groups and there was no difference in mortality.
The use of glycopeptides was a significant risk factors for infection by binary toxin-producing strains. However, the strains in both of the groups showed sensitivity to vancomycin and the vancomycin MIC50 of binary toxin-producing strains was 0.5 mg/L which was lower than the MIC50 of the A+B+CDT- strain with 1 mg/L. From amicrobiological perspective, vancomycin is not considered to be a medication which causes selective pressure in binary toxin-producing strains; however, more cases are warranted to further verify this in the future.
There were limitations in this study. It was difficult to obtain statistical significance because only 11 cases of infection due to binary toxin-producing strains occurred during 17 months. In addition, the data were from a single center limiting its applicability to the general population in Korea. Therefore, further studies are warranted in which data are collected from a number of centers.
In conclusion, binary toxin-producing strains in Korea are not common, presenting various PCR ribotypes and are susceptible to various antimicrobial agents. Clinically, the incidence of leukocytosis and mucoid stool were more frequent in binary toxin-producing CDI, but there was no remarkable difference in the mortality rate. The risk factor for binary toxin-producing CDI in Korea was the use of glycopeptides.
Acknowledgement
This work was supported by a grant from the National Research Foundation of Korea (KRF-2011-0014685).
References
- 1.Hubert B, Loo VG, Bourgault AM, Poirier L, Dascal A, Fortin E, Dionne M, Lorange M. A portrait of the geographic dissemination of the Clostridium difficile North American pulsed-field type 1 strain and the epidemiology of C. difficile-associated disease in Québec. Clin Infect Dis. 2007;44:238–244. doi: 10.1086/510391. [DOI] [PubMed] [Google Scholar]
- 2.Cheknis AK, Sambol SP, Davidson DM, Nagaro KJ, Mancini MC, Hidalgo-Arroyo GA, Brazier JS, Johnson S, Gerding DN. Distribution of Clostridium difficile strains from a North American, European and Australian trial of treatment for C. difficile infections: 2005-2007. Anaerobe. 2009;15:230–233. doi: 10.1016/j.anaerobe.2009.09.001. [DOI] [PubMed] [Google Scholar]
- 3.Kim J, Kang JO, Kim H, Seo MR, Choi TY, Pai H, Kuijper EJ, Sanders I, Fawley W. Epidemiology of Clostridium difficile infections in a tertiary-care hospital in Korea. Clin Microbiol Infect. 2012;19:521–527. doi: 10.1111/j.1469-0691.2012.03910.x. [DOI] [PubMed] [Google Scholar]
- 4.Kim H, Jeong SH, Roh KH, Hong SG, Kim JW, Shin MG, Kim MN, Shin HB, Uh Y, Lee H, Lee K. Investigation of toxin gene diversity, molecular epidemiology, and antimicrobial resistance of Clostridium difficile isolated from 12 hospitals in South Korea. Korean J Lab Med. 2010;30:491–497. doi: 10.3343/kjlm.2010.30.5.491. [DOI] [PubMed] [Google Scholar]
- 5.Carroll KC, Bartlett JG. Biology of Clostridium difficile: implications for epidemiology and diagnosis. Annu Rev Microbiol. 2011;65:501–521. doi: 10.1146/annurev-micro-090110-102824. [DOI] [PubMed] [Google Scholar]
- 6.Geric B, Carman RJ, Rupnik M, Genheimer CW, Sambol SP, Lyerly DM, Gerding DN, Johnson S. Binary toxin-producing, large clostridial toxin-negative Clostridium difficile strains are enterotoxic but do not cause disease in hamsters. J Infect Dis. 2006;193:1143–1150. doi: 10.1086/501368. [DOI] [PubMed] [Google Scholar]
- 7.Schwan C, Stecher B, Tzivelekidis T, van Ham M, Rohde M, Hardt WD, Wehland J, Aktories K. Clostridium difficile toxin CDT induces formation of microtubule-based protrusions and increases adherence of bacteria. PLoS Pathog. 2009;5:e1000626. doi: 10.1371/journal.ppat.1000626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kelly CP, LaMont JT. Clostridium difficile--more difficult than ever. N Engl J Med. 2008;359:1932–1940. doi: 10.1056/NEJMra0707500. [DOI] [PubMed] [Google Scholar]
- 9.Warny M, Pepin J, Fang A, Killgore G, Thompson A, Brazier J, Frost E, McDonald LC. Toxin production by an emerging strain of Clostridium difficile associated with outbreaks of severe disease in North America and Europe. Lancet. 2005;366:1079–1084. doi: 10.1016/S0140-6736(05)67420-X. [DOI] [PubMed] [Google Scholar]
- 10.McDonald LC, Killgore GE, Thompson A, Owens RC, Jr, Kazakova SV, Sambol SP, Johnson S, Gerding DN. An epidemic, toxin gene-variant strain of Clostridium difficile. N Engl J Med. 2005;353:2433–2441. doi: 10.1056/NEJMoa051590. [DOI] [PubMed] [Google Scholar]
- 11.Tae CH, Jung SA, Song HJ, Kim SE, Choi HJ, Lee M, Hwang Y, Kim H, Lee K. The first case of antibiotic-associated colitis by Clostridium difficile PCR ribotype 027 in Korea. J Korean Med Sci. 2009;24:520–524. doi: 10.3346/jkms.2009.24.3.520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kim J, Pai H, Seo MR, Kang JO. Epidemiology and clinical characteristics of Clostridium difficile infection in a Korean tertiary hospital. J Korean Med Sci. 2011;26:1258–1264. doi: 10.3346/jkms.2011.26.10.1258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ferguson MK. The rationale for developing scoring systems for clinical practice. Thorac Surg Clin. 2007;17:343–351. doi: 10.1016/j.thorsurg.2007.07.004. [DOI] [PubMed] [Google Scholar]
- 14.Anonymous. World Health Organisation. The anatomical therapeutic chemical (ATC) and defined daily dosing (DDD) system index 2010. [Accessed 28 October 2010]. Available at: http://www.whocc.no/
- 15.Zar FA, Bakkanagari SR, Moorthi KM, Davis MB. A comparison of vancomycin and metronidazole for the treatment of Clostridium difficile-associated diarrhea, stratified by disease severity. Clin Infect Dis. 2007;45:302–307. doi: 10.1086/519265. [DOI] [PubMed] [Google Scholar]
- 16.Gerding DN, Muto CA, Owens RC., Jr Treatment of Clostridium difficile infection. Clin Infect Dis. 2008;46(Suppl 1):S32–S42. doi: 10.1086/521860. [DOI] [PubMed] [Google Scholar]
- 17.Cheong HS, Kim JK, Lim TK, Kwon KT, Ryu SY, Heo ST, Ko KS, Oh WS, Peck KR, Lee NY, Song JH. Therapeutic efficacy of metronidazole for patients with Clostridium difficile-associated diarrhea. Korean J Med. 2007;72:639–646. [Google Scholar]
- 18.Barbut F, Richard A, Hamadi K, Chomette V, Burghoffer B, Petit JC. Epidemiology of recurrences or reinfections of Clostridium difficile-associated diarrhea. J Clin Microbiol. 2000;38:2386–2388. doi: 10.1093/gao/9781884446054.article.t031141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Seo MR, Kim J, Kang JO, Choi TY, Pai H. Multiplex PCR method for detection of Clostridium difficile tcdA, tcdB and binary toxin genes; 2012 Annual Meeting of the Korean Society of Infectious Disease and the Korean Society for Chemotherapy; 2012 Sep 1-2; Jeju, Korea. p. 179. [Google Scholar]
- 20.Kim J, Kang JO, Pai H, Choi TY. Association between PCR ribotypes and antimicrobial susceptibility among Clostridium difficile isolates from healthcare-associated infections in South Korea. Int J Antimicrob Agents. 2012;40:24–29. doi: 10.1016/j.ijantimicag.2012.03.015. [DOI] [PubMed] [Google Scholar]
- 21.Kim H, Lee Y, Moon HW, Lim CS, Lee K, Chong Y. Emergence of Clostridium difficile Ribotype 027 in Korea. Korean J Lab Med. 2011;31:191–196. doi: 10.3343/kjlm.2011.31.3.191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bauer MP, Notermans DW, van Benthem BH, Brazier JS, Wilcox MH, Rupnik M, Monnet DL, van Dissel JT, Kuijper EJ ECDIS Study Group. Clostridium difficile infection in Europe: a hospital-based survey. Lancet. 2011;377:63–73. doi: 10.1016/S0140-6736(10)61266-4. [DOI] [PubMed] [Google Scholar]
- 23.Bacci S, Mølbak K, Kjeldsen MK, Olsen KE. Binary toxin and death after Clostridium difficile infection. Emerg Infect Dis. 2011;17:976–982. doi: 10.3201/eid1706.101483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Barbut F, Decré D, Lalande V, Burghoffer B, Noussair L, Gigandon A, Espinasse F, Raskine L, Robert J, Mangeol A, Branger C, Petit JC. Clinical features of Clostridium difficile-associated diarrhoea due to binary toxin (actin-specific ADP-ribosyltransferase)-producing strains. J Med Microbiol. 2005;54:181–185. doi: 10.1099/jmm.0.45804-0. [DOI] [PubMed] [Google Scholar]
- 25.Barbut F, Gariazzo B, Bonné L, Lalande V, Burghoffer B, Luiuz R, Petit JC. Clinical features of Clostridium difficile-associated infections and molecular characterization of strains: results of a retrospective study, 2000-2004. Infect Control Hosp Epidemiol. 2007;28:131–139. doi: 10.1086/511794. [DOI] [PubMed] [Google Scholar]
- 26.Walk ST, Micic D, Jain R, Lo ES, Trivedi I, Liu EW, Almassalha LM, Ewing SA, Ring C, Galecki AT, Rogers MA, Washer L, Newton DW, Malani PN, Young VB, Aronoff DM. Clostridium difficile ribotype does not predict severe infection. Clin Infect Dis. 2012;55:1661–1668. doi: 10.1093/cid/cis786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jobe BA, Grasley A, Deveney KE, Deveney CW, Sheppard BC. Clostridium difficile colitis: an increasing hospital-acquired illness. Am J Surg. 1995;169:480–483. doi: 10.1016/S0002-9610(99)80199-8. [DOI] [PubMed] [Google Scholar]
- 28.Lyytikäinen O, Turunen H, Sund R, Rasinperä M, Könönen E, Ruutu P, Keskimäki I. Hospitalizations and deaths associated with Clostridium difficile infection, Finland, 1996-2004. Emerg Infect Dis. 2009;15:761–765. doi: 10.3201/eid1505.081154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rubin MS, Bodenstein LE, Kent KC. Severe Clostridium difficile colitis. Dis Colon Rectum. 1995;38:350–354. doi: 10.1007/BF02054220. [DOI] [PubMed] [Google Scholar]


