Abstract
OBJECTIVE
Prevalence of insulin resistance is high in the American Indian population, likely as a result of the high prevalence of obesity. This condition may be influential for clinical outcomes such as cardiovascular disease (CVD) and decreased kidney function.
RESEARCH DESIGN AND METHODS
Normal glucose tolerant (NGT) participants free of hypertension and CVD at the baseline examination (1989–1992) (N = 964) of the Strong Heart Study were selected to explore the cross-sectional association between insulin resistance quantified by homeostasis model assessment (HOMA-IR) and demographic, behavioral, and cardiometabolic variables. The longitudinal association between baseline HOMA-IR and the development of CVD was also explored. The longitudinal association between baseline HOMA-IR and the development of high urinary albumin-to-creatinine ratio was explored among nondiabetic participants (N = 1,401).
RESULTS
Cross-sectionally, HOMA-IR was associated with sex, residence location, smoking, and high-risk cardiometabolic profile. Prospectively, insulin resistance is associated with the development of CVD and decreased kidney function in this population.
CONCLUSIONS
Insulin resistance may have an important role in the pathogenesis of CVD and chronic kidney disease. Since obesity contributes to the development of insulin resistance, intervention focusing on modifiable factors such as physical activity and weight control may reduce the development of these diseases.
Insulin resistance plays a major role in the development of type 2 diabetes (1,2). It has been reported as an independent predictor of cardiovascular diseases (CVDs), hypertension, and dyslipidemia (3,4). It is also involved in the development of polycystic ovary syndrome (5), nonalcoholic fatty liver disease (6), and certain types of cancer (7,8). The list of clinical conditions associated with insulin resistance/compensatory hyperinsulinemia is expanding. With the increasing prevalence of insulin resistance in the American population (9), increased future burdens of associated clinical conditions are predicted.
The American Indian population has high prevalence of insulin resistance, especially among the younger age-groups compared with the general U.S. population (10) (at ages 45–49 years metabolic syndrome 44 and 57% among Strong Heart Study [SHS] men and women, respectively, vs. 20 and 23% among all men and women in the Third National Health and Nutrition Examination Survey [NHANES III]). Understanding the clinical outcomes of insulin resistance in this unique population may assist in reducing the burden of insulin resistance and related diseases in all Americans.
To the best of the authors’ knowledge, there are few reports about the prospective association of insulin resistance and the development of kidney dysfunction. Also, the role of insulin resistance per se in the development of CVD is less clear. Specifically, this analysis will study 1) prospective associations between insulin resistance quantified by homeostasis model assessment (HOMA-IR) and incident CVD events and 2) prospective association between insulin resistance and decreased kidney function measured by increased urinary albumin-to-creatinine ratio (UACR).
RESEARCH DESIGN AND METHODS
The SHS is a cohort study of CVD in 13 American Indian tribes/communities conducted in southwestern Oklahoma, central Arizona, and North and South Dakota. Participants (n = 4,549) aged 45–74 years underwent baseline examination from 1989 to 1992. The design, survey methods, and laboratory techniques were previously described (11,12). After excluding those who were hypertensive; who had diabetes, impaired glucose tolerance, or impaired fasting glucose; and who had previous CVD at the baseline examination, there was a total of 964 participants in the analyses for cross-sectional associations between HOMA-IR and the baseline characteristics and the prospective association between HOMA-IR and incident CVD. The prospective association between baseline HOMA-IR and the incidence rate of high UACR was studied among nondiabetic participants who were free of decreased kidney function defined by increased UACR at the baseline exam (n = 1,401). The Indian Health Service Institutional Review Board, participating institutions’ institutional review boards, and the participating tribes approved the study. Informed consent was obtained from all participants.
During a personal interview, information was collected on demographic factors, medical history, medication use, and personal health habits (physical activity, smoking, and alcohol consumption). A physical examination was conducted in the morning, which included collecting blood samples for laboratory tests and a 75-g oral glucose tolerance test. Anthropometric measurements were performed, and sitting blood pressure (first and fifth Korotkoff sounds) was measured three times consecutively using a standard mercury sphygmomanometer (WA Baum) after a 5-min rest (13). The average of the second and third systolic and diastolic blood pressure measurements was used in the analyses. A random urine sample was obtained on arrival to the clinic for measurement of creatinine and albumin content. Urine creatinine were measured by the picric acid method (14). Urine albumin content was measured by a sensitive, nephelometric technique (15).
Baseline insulin resistance was assessed by HOMA-IR using the equation [FI (μU/mL) × FG (mmol/L)]/22.5, where FG is fasting glucose (16). It was confirmed that in the SHS nondiabetic population, HOMA-IR correlates well with insulin sensitivity measured by the Minimal Model (17).
Information on leisure-time and occupation-related physical activities was collected using a questionnaire that has been validated in numerous populations, including Pima Indians (18). The questionnaire assesses the combination of reported leisure and occupational activity during the past year as an estimate of ‘‘usual’’ activity levels. Total physical activity was expressed as h/week or MET h/week. One MET represents the energy expenditure for an individual at rest, whereas a 10-MET activity requires 10 times the resting energy expenditure (19).
According to the Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC 7) criteria (20), hypertension was defined as systolic blood pressure ≥140 mmHg, diastolic blood pressure ≥90 mmHg, or use of antihypertensive medications.
According to the 1998 Provisional World Health Organization Report (21), diabetes was defined as use of an oral hypoglucose agent or insulin, fasting glucose ≥7.0 mmol/L, or postchallenge glucose ≥11.1 mmol/L (75-g oral glucose tolerance test). Impaired glucose tolerance was defined as fasting glucose <7.0 mmol/L with postchallenge glucose between 7.8 and 11.09 mmol/L. Impaired fasting glucose was defined as fasting glucose between 6.1 and 6.9 mmol/L with postchallenge glucose <7.8 mmol/L. Normal glucose tolerance was defined as fasting glucose <6.1 mmol/L with postchallenge glucose <7.8 mmol/L.
Outcome variables
Cardiovascular events.
Incident CVD events included fatal and nonfatal CVD events that occurred between the baseline examination and 31 December 2008. Fatal CVD events included fatal myocardial infarction, sudden death due to coronary heart disease, other fatal coronary heart disease, and fatal stroke. Deaths occurring during this period of time were confirmed through tribal and Indian Health Service hospital records and through direct contact with participants’ families or other informants by study personnel, as reported previously (11,12,22,23). Nonfatal CVD events included definite myocardial infarction, coronary heart disease, and stroke either identified by participant contact and medical record review or at the 2nd SHS examination in 1993–1995 and the 3rd SHS examination in 1997–1999 (11,22,23). During each examination, a 12-lead electrocardiogram and medical history including the Rose Questionnaire for angina pectoris were obtained. The average follow-up time is 14 years. Mortality follow-up data were available in 99.8% of the participants, and morbidity follow-up data were available in 99.2% of the participants.
High UACR.
High UACR was defined as UACR ≥13.3 mg/g. This represents the fourth quartile of UACR at the baseline examination (1989–1992). A previous SHS article indicated that high UACR within the normal range is related to increased CVD morbidity and mortality in this population (24). Specifically, participants with a UACR in the fourth quartile had 72% greater risk of incident CVD events and 199% greater risk of CVD mortality than those with a UACR in the lowest quartile. Nondiabetic participants who were free of CVD and not in the fourth quartile of UACR at the baseline visit were in this analysis (n = 1,401). The development of high UACR was recorded at the 2nd (1993–1995) and 3rd (1997–1999) visits.
Statistical methods
Baseline behavioral, demographic, and cardiometabolic variables including age, sex, study location, physical activity duration per week, physical activity energy cost per week measured by MET h, smoking, waist circumference, systolic blood pressure, diastolic blood pressure, LDL cholesterol, HDL cholesterol, triglyceride, and 2-h glucose were presented by the quartiles of baseline HOMA-IR. Generalized linear model was used to test trends for continuous variables, and Mantel-Haenszel χ2 test was used to do trend analyses for categorical variables. If the distribution of a continuous variable was skewed (median and lower and upper quartiles are presented), natural log-transformed values were used in the generalized linear model. Two-tailed P < 0.05 was considered to be statistically significant.
The incidence of CVD was calculated by dividing the number of participants who had CVD events by the end of 2008 by the total number of person-years of follow-up for all eligible participants. Incidence of high UACR was calculated by dividing the number of participants who had high UACR by the end of the third exam by the total number of person-years of follow-up for all eligible participants who were free of high UACR at the first examination. The Cox proportional hazards model was used to compute the hazard ratios of respective events for participants with different HOMA-IR levels. All possible risk factors of CVD or decreased kidney function reported from previous studies were included in initial models of these two outcomes. Then, a stepwise elimination method with significant entry and stay level of 0.05 was used to select factors that were significantly associated with incident CVD or decreased kidney function. The results of the initial models are described in the text. The results of stepwise selected Cox proportional hazards models are reported in the Tables. For the association between HOMA-IR and incident CVD events, baseline age, sex, systolic and diastolic blood pressure, LDL cholesterol, HDL cholesterol, smoking, and micro- and macroalbuminuria were included in the initial Cox model as covariates (25,26). For the association between HOMA-IR and the development of decreased kidney function, baseline UACR, age, sex, BMI, smoking, and systolic and diastolic blood pressure were included in the initial Cox model as covariates (27,28).
RESULTS
Cross-sectional associations
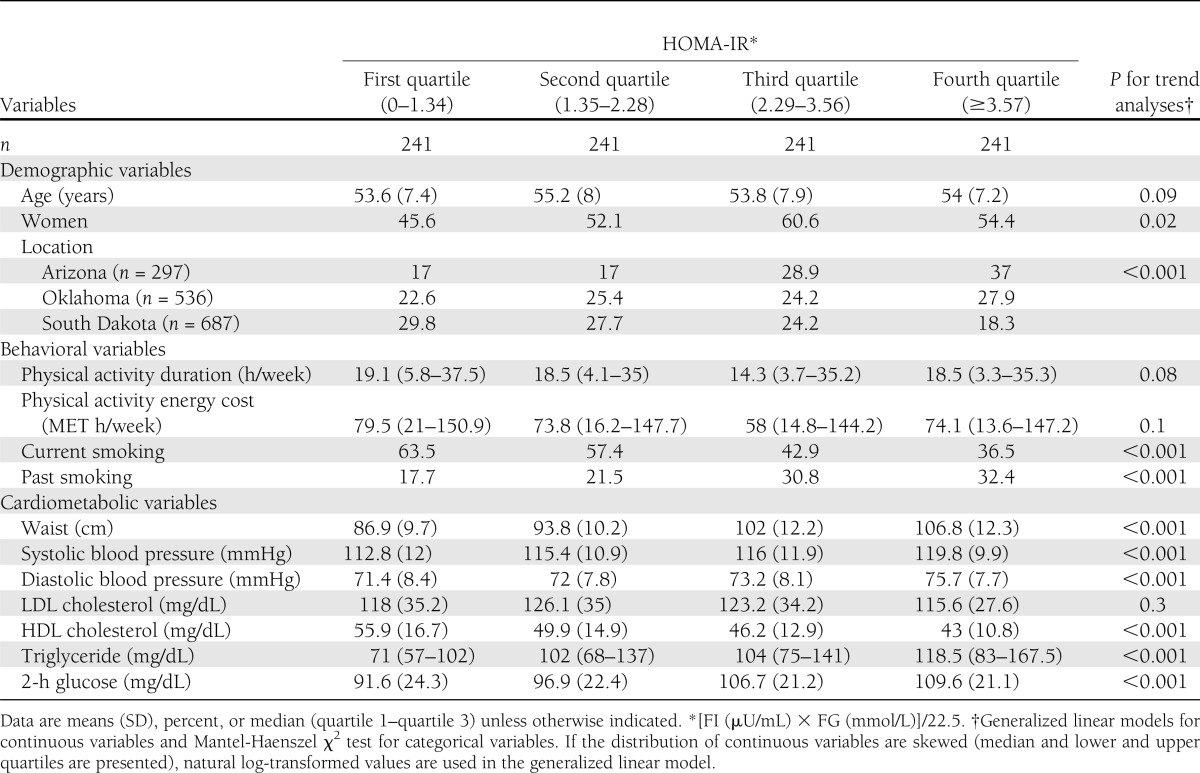
At baseline exam, more women than men had high HOMA-IR. HOMA-IR was higher among participants residing in Arizona than participants in Oklahoma or the Dakotas. There are more past smokers but fewer current smokers who had high HOMA-IR. Physical activity levels measured by duration or MET h were not significantly different among participants in different quartiles of HOMA-IR. Except LDL cholesterol, all cardiometabolic variables including waist circumference, systolic and diastolic blood pressure, HDL cholesterol, triglyceride, and 2-h glucose became unfavorable with increases of HOMA-IR (Table 1).
Table 1.
Baseline demographic, behavioral, and cardiometabolic variables according to insulin resistance level
Incident CVD
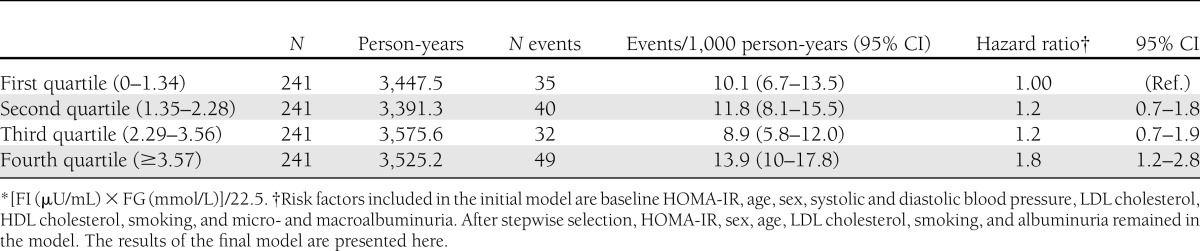
In the Cox proportional hazards model that included all possible risk factors, only age, sex, LDL cholesterol, and smoking were significantly associated with incident CVD events. After stepwise selection of covariates, HOMA-IR level, sex, age, LDL cholesterol, smoking, and albuminuria remained in the Cox proportional hazards model. The hazard ratio of incident CVD increased as baseline HOMA-IR increased and reached statistical significance at the fourth quartile of HOMA-IR (1.8 [95% CI 1.2–2.8]) (Table 2).
Table 2.
Cox proportional hazards model for incident CVD according to baseline quartiles of HOMA-IR*
Incident high UACR
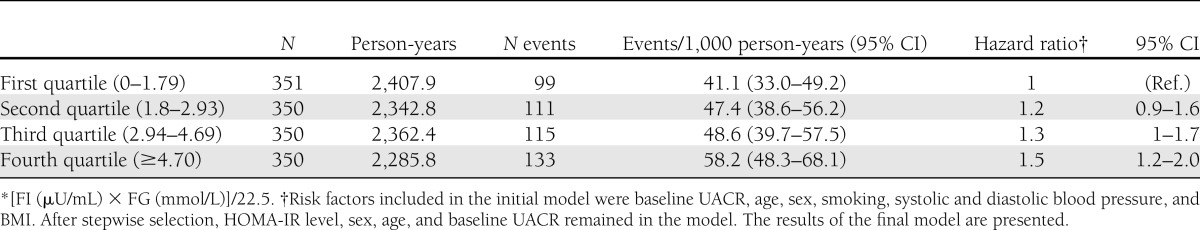
In the initial model that included all possible risk factors of decreased kidney function, compared with participants in the first quartile of HOMA-IR, those in the fourth quartile of HOMA-IR had 1.4 times higher risk of decreased kidney function (hazard ratio 1.43 [95% CI 1.03–1.98], P = 0.031). The other significant factors are age, sex, and baseline UACR levels. Baseline blood pressure levels, BMI, and smoking status were not significantly related to the development of decreased kidney function in this initial model. After stepwise selection, baseline HOMA-IR, UACR, age, and sex remained significant, and no other factors were selected in the model. Compared with participants in the first quartile of HOMA-IR, those in the fourth quartile of HOMA-IR have 1.5 times higher risk of decreased kidney function (1.5 [1.19–2.02], P = 0.001) (Table 3).
Table 3.
Cox proportional hazards model for incident high UACR (≥13.3 mg/g) according to baseline quartiles of HOMA-IR*
CONCLUSIONS
This study demonstrated that insulin resistance is related to the development of decreased kidney function measured by high UACR among nondiabetic participants. To the best of our knowledge, this is the first large population-based prospective study to report this association. In this cohort study, insulin resistance is also associated with the development of incident CVD events.
All possible CVD risk factors were included in the initial Cox proportional hazards model to explore the association between HOMA-IR and incident CVD events. While HOMA-IR remained in the model after stepwise selection, HDL cholesterol and blood pressure levels were not. The prospective association between insulin resistance and CVD was only shown when HDL and blood pressure were not included in the model. HDL cholesterol and blood pressure levels are closely related to insulin resistance in this and other populations (29). Insulin resistance may increase the risk of CVD through dyslipidemia and high blood pressure. Similar mechanisms have been proposed by other studies (30–32). The success of Stop Atherosclerosis in Native Patients with diabetes Study (33) provides some support for this point of view. Further study in the SHS population will give more insight into this topic.
Our findings of longitudinal associations between HOMA-IR and markers of renal dysfunction are unique. There is a small prospective study in 116 nondiabetic Japanese Americans living in Hawaii demonstrating that insulin resistance preceded the development of albuminuria (34). Another study conducted in a Chinese population (n = 652) indicated that insulin resistance is related to rapid decline in renal function in the nondiabetic elderly (35). Reports using NHANES III data showed a cross-sectional association between insulin resistance and the presence of chronic kidney disease (36). The mechanisms linking insulin resistance to decreased kidney function are not fully understood. However, oxidative stress and endothelial dysfunction related to insulin resistance may cause glomerulosclerosis or atherosclerosis-related kidney damage (35,37). Hypertriglyceridemia caused by insulin resistance may be a risk factor for developing proteinuria (31,38). Our findings from a large prospective population study suggest that a more aggressive approach to reduce insulin resistance in high-risk individuals may substantially lower the risk of chronic kidney disease.
It is interesting to see that more participants with high HOMA-IR levels had stopped smoking compared with those with low HOMA-IR levels. A similar situation has been previously reported in participants with diabetes in this population (39). In the previous report (39), more diabetic participants stopped smoking compared with those who did not have diabetes. It is likely that participants with high HOMA-IR had stopped smoking because of their high-risk lipid profile or other related medical conditions.
The strength of the study is the complete collection of information related to insulin resistance syndrome, the long follow-up time, and systematic collection of clinical outcomes. Limitations include the lack of a direct measure of insulin resistance and the fact that participants were of middle age and older and thus the earlier effects of insulin resistance could not be analyzed.
It has been suggested that insulin resistance is the link between obesity and clinical outcomes (40). Our study provides further evidence for this point of view. Insulin resistance coexists or precedes many important clinical outcomes in modern society. Though these clinical outcomes could develop for reasons not related to insulin resistance and those with insulin resistance do not invariably develop those abnormalities, we cannot ignore the major contribution of insulin resistance/compensatory hyperinsulinemia to the development of many chronic diseases. It is reasonable to hypothesize that insulin resistance/compensatory hyperinsulinemia is a common pathophysiological pathway leading to the development of many chronic diseases. Interventions that can ameliorate the cycle of obesity and insulin resistance deserve serious effort to prevent CVD and chronic kidney disease. Physical activity and weight control, known to reduce insulin resistance, are likely to bring significant health benefits to populations with rising prevalence of insulin resistance.
Acknowledgments
This study was supported by cooperative agreement grants U01-HL41642, U01-HL41652, UL01-HL41654, U01-HL65520, and U01-HL65521 from the National Heart, Lung, and Blood Institute, Bethesda, MD.
No potential conflicts of interest relevant to this article were reported.
Y.Z. conceived the idea, conducted statistical analyses, and wrote the manuscript. E.T.L., B.V.H., L.G.B., J.G.U., J.Y., W.W., F.Y., T.A., R.B.D., and G.d.S. contributed to discussion and reviewed and edited the manuscript. Y.Z. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Parts of this study were presented in poster form at the Scientific Sessions of the American Heart Association, Atlanta, Georgia, 22–25 March 2011, and as an oral presentation at the Harold Hamm Oklahoma Diabetes Center 7th Diabetes Research Symposium, Oklahoma City, Oklahoma, 12 November 2011.
The authors acknowledge the assistance and cooperation of the Ak-Chin Tohono O'Odham (Papago)/Pima, Apache, Caddo, Cheyenne River Sioux, Comanche, Delaware, Spirit Lake Sioux, Fort Sill Apache, Gila River Pima Maricopa, Kiowa, Oglala Sioux, Salt River Pima/Maricopa, and Wichita Indian communities. It would have been impossible to conduct this study without their support. The authors also thank the Indian Health Service hospitals and clinics at each center and the SHS investigators and staff.
Footnotes
The opinions expressed in this article are those of the authors and do not necessarily reflect the views of the Indian Health Service.
References
- 1.Lillioja S, Mott DM, Spraul M, et al. Insulin resistance and insulin secretory dysfunction as precursors of non-insulin-dependent diabetes mellitus. Prospective studies of Pima Indians. N Engl J Med 1993;329:1988–1992 [DOI] [PubMed] [Google Scholar]
- 2.Resnick HE, Jones K, Ruotolo G, et al. Strong Heart Study Insulin resistance, the metabolic syndrome, and risk of incident cardiovascular disease in nondiabetic american indians: the Strong Heart Study. Diabetes Care 2003;26:861–867 [DOI] [PubMed] [Google Scholar]
- 3.Després JP, Lamarche B, Mauriège P, et al. Hyperinsulinemia as an independent risk factor for ischemic heart disease. N Engl J Med 1996;334:952–957 [DOI] [PubMed] [Google Scholar]
- 4.Salonen JT, Lakka TA, Lakka HM, Valkonen VP, Everson SA, Kaplan GA. Hyperinsulinemia is associated with the incidence of hypertension and dyslipidemia in middle-aged men. Diabetes 1998;47:270–275 [DOI] [PubMed] [Google Scholar]
- 5.Dunaif A. Insulin resistance and the polycystic ovary syndrome: mechanism and implications for pathogenesis. Endocr Rev 1997;18:774–800 [DOI] [PubMed] [Google Scholar]
- 6.Ardigò D, Numeroso F, Valtueña S, et al. Hyperinsulinemia predicts hepatic fat content in healthy individuals with normal transaminase concentrations. Metabolism 2005;54:1566–1570 [DOI] [PubMed] [Google Scholar]
- 7.Argilés JM, López-Soriano FJ. Insulin and cancer (Review). Int J Oncol 2001;18:683–687 [Review] [PubMed] [Google Scholar]
- 8.Facchini FS, Hua N, Abbasi F, Reaven GM. Insulin resistance as a predictor of age-related diseases. J Clin Endocrinol Metab 2001;86:3574–3578 [DOI] [PubMed] [Google Scholar]
- 9.Li C, Ford ES, McGuire LC, Mokdad AH, Little RR, Reaven GM. Trends in hyperinsulinemia among nondiabetic adults in the U.S. Diabetes Care 2006;29:2396–2402 [DOI] [PubMed] [Google Scholar]
- 10.Resnick HE, Strong Heart Study Investigators Metabolic syndrome in American Indians. Diabetes Care 2002;25:1246–1247 [DOI] [PubMed] [Google Scholar]
- 11.Lee ET, Welty TK, Fabsitz R, et al. The Strong Heart Study. A study of cardiovascular disease in American Indians: design and methods. Am J Epidemiol 1990;132:1141–1155 [DOI] [PubMed] [Google Scholar]
- 12.Howard BV, Welty TK, Fabsitz RR, et al. Risk factors for coronary heart disease in diabetic and nondiabetic Native Americans. The Strong Heart Study. Diabetes 1992;41(Suppl 2):4–11 [DOI] [PubMed] [Google Scholar]
- 13.Howard BV, Lee ET, Yeh JL, et al. Hypertension in adult American Indians. The Strong Heart Study. Hypertension 1996;28:256–264 [DOI] [PubMed] [Google Scholar]
- 14.Chasson AL, Grady HJ, Stanley MA. Determination of creatinine by means of automatic chemical analysis. Tech Bull Regist Med Technol 1960;30:207–212 [PubMed] [Google Scholar]
- 15.Vasquez B, Flock EV, Savage PJ, et al. Sustained reduction of proteinuria in type 2 (non-insulin-dependent) diabetes following diet-induced reduction of hyperglycaemia. Diabetologia 1984;26:127–133 [DOI] [PubMed] [Google Scholar]
- 16.Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985;28:412–419 [DOI] [PubMed] [Google Scholar]
- 17.Resnick HE, Bergman RN, Henderson JA, Nez-Henderson P, Howard BV. Utility of a surrogate measure of insulin resistance in American Indians: the Strong Heart Study. Ethn Dis 2002;12:523–529 [PubMed] [Google Scholar]
- 18.Kriska AM, Brach JS, Jarvis BJ, et al. Physical activity and gallbladder disease determined by ultrasonography. Med Sci Sports Exerc 2007;39:1927–1932 [DOI] [PubMed] [Google Scholar]
- 19.American College of Sports Medicine Guidelines for Graded Exercise Testing and Exercise Prescription. Philadelphia, PA, Lippincott Williams & Wilkins, 2000 [Google Scholar]
- 20.Chobanian AV, Bakris GL, Black HR, et al. Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. National Heart, Lung, and Blood Institute. National High Blood Pressure Education Program Coordinating Committee Seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. Hypertension 2003;42:1206–1252 [DOI] [PubMed] [Google Scholar]
- 21.Alberti KG, Zimmet PZ. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet Med 1998;15:539–553 [see comment] [DOI] [PubMed] [Google Scholar]
- 22.Lee ET, Cowan LD, Welty TK, et al. All-cause mortality and cardiovascular disease mortality in three American Indian populations, aged 45-74 years, 1984-1988. The Strong Heart Study. Am J Epidemiol 1998;147:995–1008 [DOI] [PubMed] [Google Scholar]
- 23.Howard BV, Robbins DC, Sievers ML, et al. LDL cholesterol as a strong predictor of coronary heart disease in diabetic individuals with insulin resistance and low LDL: The Strong Heart Study. Arterioscler Thromb Vasc Biol 2000;20:830–835 [DOI] [PubMed] [Google Scholar]
- 24.Xu J, Knowler WC, Devereux RB, et al. Albuminuria within the “normal” range and risk of cardiovascular disease and death in American Indians: the Strong Heart Study. Am J Kidney Dis 2007;49:208–216 [DOI] [PubMed] [Google Scholar]
- 25.Lee ET, Howard BV, Wang W, et al. Prediction of coronary heart disease in a population with high prevalence of diabetes and albuminuria: the Strong Heart Study. Circulation 2006;113:2897–2905 [DOI] [PubMed] [Google Scholar]
- 26.Zhang Y, Galloway JM, Welty TK, et al. Incidence and risk factors for stroke in American Indians: the Strong Heart Study. Circulation 2008;118:1577–1584 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lucove J, Vupputuri S, Heiss G, North K, Russell M. Metabolic syndrome and the development of CKD in American Indians: the Strong Heart Study. Am J Kidney Dis 2008;51:21–28 [DOI] [PubMed] [Google Scholar]
- 28.Xu J, Lee ET, Devereux RB, et al. A longitudinal study of risk factors for incident albuminuria in diabetic American Indians: the Strong Heart Study. Am J Kidney Dis 2008;51:415–424 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Reaven GM. The individual components of the metabolic syndrome: is there a raison d’etre? J Am Coll Nutr 2007;26:191–195 [DOI] [PubMed] [Google Scholar]
- 30.DeFronzo RA. Insulin resistance, lipotoxicity, type 2 diabetes and atherosclerosis: the missing links. The Claude Bernard Lecture 2009. Diabetologia 2010;53:1270–1287 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Brown MS, Goldstein JL. Selective versus total insulin resistance: a pathogenic paradox. Cell Metab 2008;7:95–96 [DOI] [PubMed] [Google Scholar]
- 32.Einhorn D, Reaven GM, Cobin RH, et al. American College of Endocrinology position statement on the insulin resistance syndrome. Endocr Pract 2003;9:237–252 [PubMed] [Google Scholar]
- 33.Howard BV, Roman MJ, Devereux RB, et al. Effect of lower targets for blood pressure and LDL cholesterol on atherosclerosis in diabetes: the SANDS randomized trial. JAMA 2008;299:1678–1689 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Fujikawa R, Okubo M, Egusa G, Kohno N. Insulin resistance precedes the appearance of albuminuria in non-diabetic subjects: 6 years follow up study. Diabetes Res Clin Pract 2001;53:99–106 [DOI] [PubMed] [Google Scholar]
- 35.Cheng HT, Huang JW, Chiang CK, Yen CJ, Hung KY, Wu KD. Metabolic syndrome and insulin resistance as risk factors for development of chronic kidney disease and rapid decline in renal function in elderly. J Clin Endocrinol Metab 2012;97:1268–1276 [DOI] [PubMed] [Google Scholar]
- 36.Chen J, Muntner P, Hamm LL, et al. Insulin resistance and risk of chronic kidney disease in nondiabetic US adults. J Am Soc Nephrol 2003;14:469–477 [DOI] [PubMed] [Google Scholar]
- 37.Hostetter TH, Rennke HG, Brenner BM. The case for intrarenal hypertension in the initiation and progression of diabetic and other glomerulopathies. Am J Med 1982;72:375–380 [DOI] [PubMed] [Google Scholar]
- 38.Tozawa M, Iseki K, Iseki C, Oshiro S, Ikemiya Y, Takishita S. Triglyceride, but not total cholesterol or low-density lipoprotein cholesterol levels, predict development of proteinuria. Kidney Int 2002;62:1743–1749 [DOI] [PubMed] [Google Scholar]
- 39.Zhang Y, Lee ET, Devereux RB, et al. Prehypertension, diabetes, and cardiovascular disease risk in a population-based sample: the Strong Heart Study. Hypertension 2006;47:410–414 [DOI] [PubMed] [Google Scholar]
- 40.Reaven GM. Insulin resistance: the link between obesity and cardiovascular disease. Med Clin North Am 2011;95:875–892 [DOI] [PubMed] [Google Scholar]


