Abstract
OBJECTIVE
Acute hypoglycemia impairs cognitive function in several domains. Executive cognitive function governs organization of thoughts, prioritization of tasks, and time management. This study examined the effect of acute hypoglycemia on executive function in adults with and without diabetes.
RESEARCH DESIGN AND METHODS
Thirty-two adults with and without type 1 diabetes with no vascular complications or impaired awareness of hypoglycemia were studied. Two hyperinsulinemic glucose clamps were performed at least 2 weeks apart in a single-blind, counterbalanced order, maintaining blood glucose at 4.5 mmol/L (euglycemia) or 2.5 mmol/L (hypoglycemia). Executive functions were assessed with a validated test suite (Delis-Kaplan Executive Function). A general linear model (repeated-measures ANOVA) was used. Glycemic condition (euglycemia or hypoglycemia) was the within-participant factor. Between-participant factors were order of session (euglycemia-hypoglycemia or hypoglycemia-euglycemia), test battery used, and diabetes status (with or without diabetes).
RESULTS
Compared with euglycemia, executive functions (with one exception) were significantly impaired during hypoglycemia; lower test scores were recorded with more time required for completion. Large Cohen d values (>0.8) suggest that hypoglycemia induces decrements in aspects of executive function with large effect sizes. In some tests, the performance of participants with diabetes was more impaired than those without diabetes.
CONCLUSIONS
Executive cognitive function, which is necessary to carry out many everyday activities, is impaired during hypoglycemia in adults with and without type 1 diabetes. This important aspect of cognition has not received previous systematic study with respect to hypoglycemia. The effect size is large in terms of both accuracy and speed.
The human brain depends on glucose as its energy source; acute hypoglycemia results in neuroglycopenia with subsequent cognitive impairment. Individuals with type 1 diabetes are exposed to an average of two episodes of self-treated hypoglycemia per week (1). In general, performance on complex cognitive tasks deteriorates when blood glucose declines to <3.0 mmol/L (54 mg/dL) (2,3). Previous studies have demonstrated that for complex tasks, accuracy often is preserved at the expense of speed (4). The impairment of cognitive function is reversible, although full recovery requires between 20 and 75 min after the restoration of euglycemia (5,6). Acute hypoglycemia has been shown to impair various cognitive domains, including memory, attention, information processing, psychomotor function, and spatial ability (7–10). However, the effect of hypoglycemia on executive cognitive function, which is important for everyday functioning, has received little systematic study.
Executive function incorporates a number of complex, interdependent cognitive processes that allow an individual to plan, initiate, sequence, monitor, and inhibit complex behavior (11), allowing one to organize thoughts, prioritize tasks, manage time efficiently, and make decisions. Executive function, therefore, is vital for the performance of many everyday activities, and in children, inadequate executive functioning has been linked to poor adherence to treatment (12). Executive function is not localized to one particular area of the brain (13), although evidence from neuroimaging studies suggests that the frontal lobes of the brain (and their connections to other regions) are closely associated with this cognitive domain (14).
The current study examined the effects of acute hypoglycemia on executive function in adult humans with and without type 1 diabetes with use of a well-validated test battery (15–18). Performance was examined in a counterbalanced design under euglycemic and hypoglycemic conditions.
RESEARCH DESIGN AND METHODS
Sixteen adults with type 1 diabetes and 16 nondiabetic adults were studied. Baseline demographics are shown in Table 1. The groups were matched for age and BMI. Participants with diabetes were recruited from the diabetes clinic at the Royal Infirmary of Edinburgh and had no history of macrovascular or microvascular disease. Digital retinal screening was used to exclude diabetic eye disease, peripheral neuropathy was excluded by clinical examination, and nephropathy was excluded by the absence of microalbuminuria. Participants without diabetes were recruited by e-mail and paper advertisements within the local hospital and university. None of the participants had a history of seizures, head injury, psychiatric disorder, or alcohol or drug abuse. Participants were taking no medication other than insulin or oral contraceptives. Other exclusion criteria were impaired awareness of hypoglycemia (19), pregnancy, a coexisting systemic disease, or malignancy.
Table 1.
Participant baseline demographics
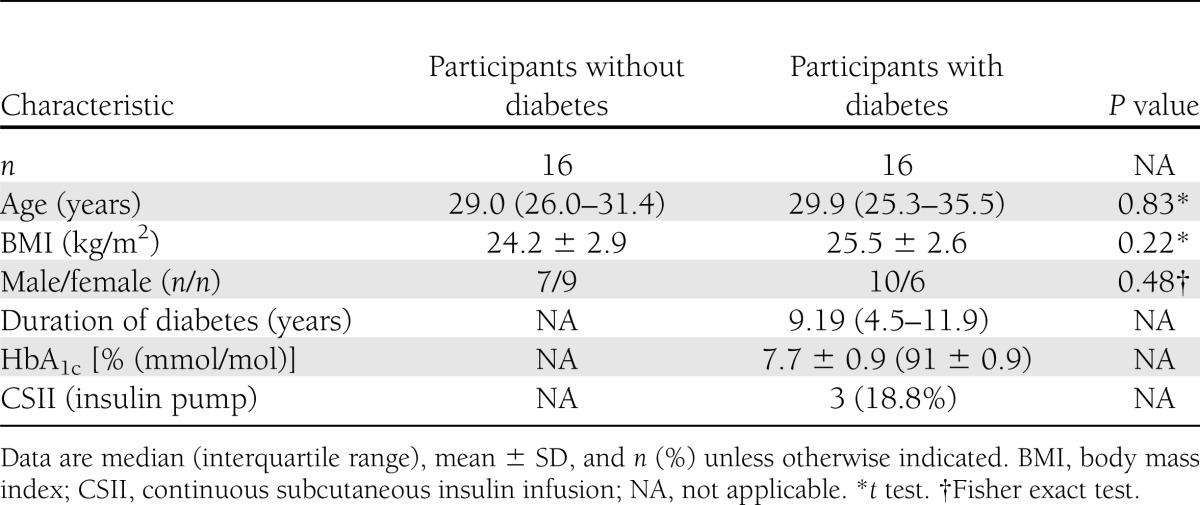
HbA1c was measured by high-performance liquid chromatography (nondiabetic reference range 5.0–6.05% [31–43 mmol/mol]; Bio-Rad Laboratories, Munich, Germany) and was aligned with the Diabetes Control and Complications Trial. All participants gave written consent before participating in the study, which had been approved by the local medical ethics advisory committee.
Experimental sessions were performed in the Wellcome Trust Clinical Research Facility at the Royal Infirmary of Edinburgh. Participants attended for two experimental sessions at least 2 weeks apart. Participants with type 1 diabetes were required to monitor their blood glucose frequently for the 48 h preceding each experimental session, which was postponed if they had recorded a blood glucose level <3.5 mmol/L (<63 mg/dL) or had experienced symptoms suggestive of hypoglycemia. Sessions commenced at 0800 h, and all patients fasted overnight. Participants with type 1 diabetes omitted their morning fast-acting insulin or switched off their insulin pump.
During each session, a modified hyperinsulinemic glucose clamp was performed (20). To arterialize blood samples, the nondominant arm was wrapped in a warmed blanket with a retrograde intravenous cannula inserted into the forearm. An additional cannula was inserted into the nondominant antecubital fossa to infuse insulin (human Actrapid; Novo Nordisk, Crawley, U.K.) and 20% dextrose. Insulin was infused at a constant rate of 1.5 mU/kg/min with a Gemini PCI pump (Alaris Medical Systems, San Diego, CA). Blood samples were taken at 5-min intervals and analyzed by a glucose oxidase method (2300 STAT; YSI, Yellow Springs, OH). The dextrose infusion rate was adjusted to maintain the appropriate arterialized blood glucose concentration.
Two experimental conditions (hypoglycemia and euglycemia) were studied in a single-blind, random, counterbalanced order. During each experimental session, arterialized blood glucose was maintained at 4.5 mmol/L (81 mg/dL) for 30 min. It was then either maintained at 4.5 mmol/L throughout (the euglycemia condition) or lowered over 20 min to 2.5 mmol/L (45 mg/dL) (the hypoglycemia condition). The experimental condition lasted for 60 min, after which time euglycemia was restored. Participants consumed a meal on completion of the study.
Hypoglycemia scores
Participants scored their symptoms at baseline and during the experimental period with a subjective, validated questionnaire, the Edinburgh Hypoglycemia Symptom Scale, that measures the intensity of commonly experienced hypoglycemic symptoms on a 7-point Likert scale (1 = not present, 7 = very intense). These symptoms have been grouped previously into autonomic, neuroglycopenic, and malaise subgroupings (21).
Baseline intelligence and educational achievement
The National Adult Reading Test (NART) assesses the pronunciation of 50 phonologically irregular English words and is widely used as an estimate of peak intellectual ability (22). Educational achievement was determined by whether a participant had high school, degree, or doctoral-level qualifications.
Cognitive function tests
Tests of executive function were performed during both experimental conditions. All tests were from the Delis-Kaplan Executive Function (D-KEFS) test suite. The D-KEFS is a well-validated series of tests that comprehensively assesses the domain of executive functions and is suitable for adults with a range of ability levels (15,16). Practice effect was controlled for by counterbalancing the order of the experimental conditions (euglycemia before hypoglycemia and vice versa) and counterbalancing the parallel forms of the tests (battery A before battery B and vice versa) with use of a Latin square. This study design minimizes many of the previous criticisms of cognitive function testing during hypoglycemia (23). Order effects were sought, but from prior experience in cognitive testing in hypoglycemic clamp studies, none are usually found.
The Digit Symbol Substitution Test (DSST) (from the Wechsler Adult Intelligence Scale III-UK) and Trail Making B test (from the D-KEFS) were used as marker cognitive tests that are reliably affected by moderate hypoglycemia (18,24). The DSST tests coding performed at speed, and for each mistake, 5 points were subtracted from the score achieved to give an overall score. Trail Making B also tests a wide range of cognitive processes, including complex attention, visual scanning, psychomotor speed, and mental flexibility (17). The test requires the participant to switch back and forth between connecting numbers and letters in sequence (e.g., 1-A-2-B-3-C); it covers two pages, which increases spatial scanning demands. Time taken (in seconds) was subtracted from 200; for each mistake, 5 additional points were subtracted to give a total score.
Executive function tests
Verbal fluency (category switching).
This test requires both rapid retrieval from semantic knowledge and cognitive flexibility to allow switching between categories. Participants were required to generate words, alternating between two different semantic categories (e.g., fruit and furniture). The outcome variable was the total score, with 1 point awarded for each correct pair named during the time limit.
Sorting test.
Based on the Wisconsin Card Sorting Test (25), this test isolates and measures multiple components of concept formation and problem-solving abilities. Each participant was shown two groups of three cards each and was then required to state how the two groups were sorted (e.g., one card set shows singular words, and the other displays plural words). Outcome variables were the total score, with 4 points awarded for each correct description, and time taken.
Twenty questions test.
The participant is presented with a stimulus page depicting pictures of 30 common objects (e.g., banana, airplane, bowl). The participant must ask the fewest number of yes/no questions possible to identify the unknown target object. The ideal response would eliminate one-half of the remaining objects. This test assesses the respondent’s ability to perceive the various subcategories (e.g., land-based objects, flying objects). By incorporating feedback from previous answers, participants can formulate a yes/no question to eliminate the maximum number of objects. One point was awarded for each correctly phrased question, with a lower score denoting better performance. Participants performed this test twice, and outcome variables were combined score and total time taken. A quantitative analysis of the quality of responses was then made to give the third outcome variable. Participants scored 5 points for an answer that eliminated one-half of the remaining objects, 4 points for a response that eliminated ±1 of one-half of the remaining objects, 3 points for ±2, 2 points for ±3, 1 point for ±4, and 0 points for ±5. So, if there were eight remaining objects and a response eliminated three objects, then that question would score 4 points. This score was then divided by the number of questions asked to give the third outcome variable.
Tower test.
The tower test assesses several key executive functions, including spatial planning and maintaining an instructional set (26). Participants must build the designated tower in the fewest number of moves possible by moving differently sized disks across three pegs. Four towers of increasing difficulty were constructed during each experimental session. To avoid a practice effect, a parallel version of equivalent difficulty was created by shifting each piece to the right. One point was awarded for each move taken; therefore, a lower score denoted better performance. Outcome variables were the combined score for all four towers and the total time taken.
Color–word interference test (Stroop).
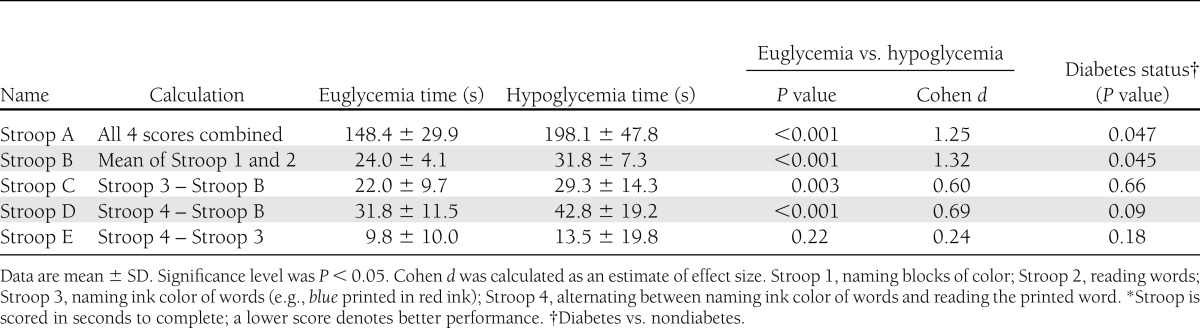
Based on the classic Stroop procedure, these four tests assess the participant’s ability to inhibit an overlearned verbal response (i.e., naming the ink color of the word instead of reading the printed word) (27). The first two (baseline) tasks require the participant to name blocks of color (Stroop 1) and read color words printed in black ink (Stroop 2). The third task prints color words in a different color (Stroop 3), for example, the word red printed in blue ink (inhibiting). The fourth task involves asking the participant to switch back and forth between naming ink colors and inhibiting (Stroop 4), testing both inhibition and cognitive flexibility. Time required for completion became the score for that task; 4 s were added for an uncorrected error and 2 s for a corrected error. Five combined task scores were calculated (Table 2). Stroop A is the sum of all four tasks; Stroop B is the sum of the first two tasks and an indication of performance in key lower-level skills (i.e., reading, naming). Stroop C and D demonstrate any effect on executive functioning while attenuating the impact of any deficit in more basic cognitive functioning (i.e., reading printed words). The Stroop Interference Effect (Stroop C) indicates the increase in time taken to perform the task requiring an inhibited response compared with simply reading or naming the ink color (28). Stroop E demonstrates the additional time required to switch between tasks (cognitive flexibility) while attenuating the effect of inhibition. Many of the Stroop tests rely on color discrimination, which has been shown previously to be unaffected by moderate hypoglycemia although may become impaired with more profound hypoglycemia (29).
Table 2.
Stroop test results*
Statistical analysis
A general linear model (repeated-measures ANOVA) was used. Glycemic condition (euglycemia or hypoglycemia) was the within-participant factor (repeated measure). Order of session (euglycemia followed by hypoglycemia or vice versa), order of test battery (test battery A followed by battery B or vice versa), and diabetes status (participants with or without diabetes) were between-participant factors, with the NART score as a covariate. Effect sizes were calculated using Cohen d to assess the extent of any cognitive decrement caused by hypoglycemia (d = 0.2 [small], d = 0.5 [medium], d = 0.8 [large]) (30). All analyses were performed with Microsoft Excel 2010 (Redmond, WA) and SPSS version 18.0 (IBM Corp., Chicago, IL) software, both for Windows. Unless otherwise stated, data are mean ± SD, and P < 0.05 was considered significant.
RESULTS
Blood glucose
Target blood glucose levels were achieved for both the hypoglycemic (2.45 ± 0.11 mmol/L) and the euglycemic (4.54 ± 0.09 mmol/L) conditions. Glucose levels were similar in participants with and without diabetes (P = 0.23 and 0.11, respectively, t test).
Symptom scores
Autonomic symptom scores increased from 10.6 ± 4.1 during euglycemia to 22.6 ± 9.3 during hypoglycemia (P < 0.001, Cohen d = 1.68). Neuroglycopenic symptom scores increased from 10.8 ± 5.5 to 19.6 ± 8.7 (P < 0.001, Cohen d = 1.22). Malaise symptom scores increased from 2.5 ± 1.3 to 3.4 ± 1.8 (P = 0.004, Cohen d = 0.63).
Educational achievement and baseline intelligence
Nine (56.3%) participants without diabetes had doctoral-level qualifications compared with four (25.0%) with diabetes (P = 0.20, Pearson χ2). Mean NART scores were significantly lower in participants with diabetes than in those without (36.3 ± 3.8 and 40.5 ± 5.4, respectively, P < 0.02, t test). As expected, NART scores (taken at baseline) did not differ significantly between the two experimental sessions (P = 0.18, t test).
General cognitive function tests
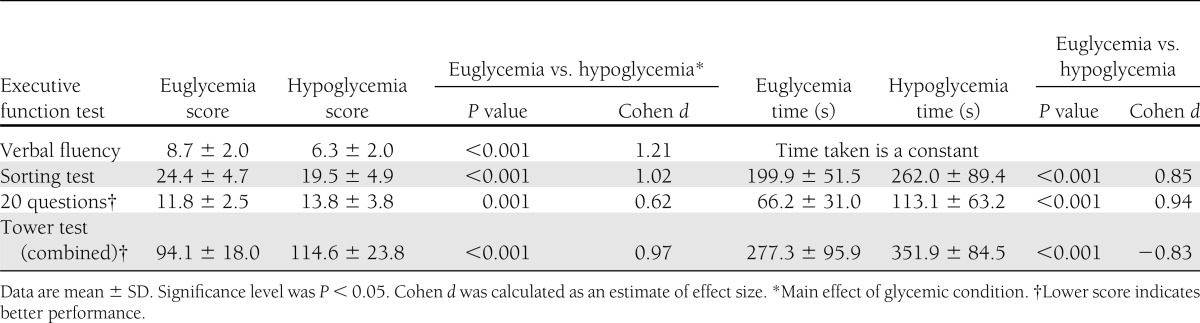
Performance during hypoglycemia was significantly impaired in both the Trail Making B test and DSST (P < 0.001 for both, Cohen’s d = 1.16 and 0.84 for Trail Making B and DSST, respectively). No significant difference for these tests was observed between participants with and without diabetes (data not shown).
Executive function tests
No significant effects were found for order of session (hypoglycemia followed by euglycemia or vice versa). The only significant effects for diabetes status were for the Stroop A (P < 0.05), Stroop B (P < 0.05), and verbal fluency (P < 0.03) tasks; performance during both euglycemia and hypoglycemia of participants with diabetes was worse than for those without diabetes.
Performance during hypoglycemia was significantly impaired on every measure of executive function examined other than Stroop E in terms of both scores achieved and time taken (Tables 2 and 3). Hypoglycemia significantly prolonged the time to completion in all tests where time was not fixed. The high Cohen d values (>0.8) indicated that hypoglycemia induced decrements with large effect sizes.
Table 3.
Executive function test scores and times
The lower performance scores achieved during hypoglycemia in the Stroop test were not simply a result of slower information processing; participants made a significantly greater number of errors during hypoglycemia than during euglycemia (5.81 ± 3.93 and 1.91 ± 1.96, respectively, P < 0.001, unpaired t test). Both self-corrected and uncorrected errors were significantly increased during hypoglycemia compared with euglycemia (data not shown, P < 0.001 and P = 0.002, respectively).
In the 20 questions test, responses given during hypoglycemia were often of poor quality; for example, participants asked very specific questions that would only eliminate one or two of the remaining objects (i.e., Does it fly? Is it a bowl? Is it orange?). The quality of responses showed that scores during hypoglycemia were significantly lower than during euglycemia (see research design and methods for scoring system) (5.11 ± 1.40 and 6.24 ± 1.48, respectively, P = 0.003, unpaired t test).
Significant glycemic condition by diabetes status interactions were observed for Stroop A (P = 0.02), Stroop B (P = 0.002), and the sorting test (time taken) (P = 0.004), suggesting that the effects of hypoglycemia differ significantly between participants with and without diabetes. Figure 1 shows that in all three tests, participants with diabetes were affected by hypoglycemia to a greater degree than those without diabetes. When the NART scores were entered as a covariate, participants with diabetes continued to experience a greater detrimental effect of hypoglycemia than those without diabetes in the sorting test (P = 0.02) and Stroop B (P = 0.01) but not in Stroop A (P = 0.12).
Figure 1.
A: Time taken to complete the sorting test during hypoglycemia and euglycemia according to diabetes status (P = 0.004); a lower score denotes better performance. B: Comparison of performance during Stroop A and B during hypoglycemia and euglycemia according to diabetes status (P < 0.05 for both); a lower score denotes better performance. Data are mean ± SD. ND, participants without diabetes.
CONCLUSIONS
The current study demonstrates that acute hypoglycemia markedly impairs performance in almost all the aspects of executive function that were tested in adults with and without type 1 diabetes. All domains of executive function were significantly impaired except for Stroop E. Where the time taken was a variable rather than a constant, the time to completion was significantly longer in all tests. The generally large Cohen d values (>0.8) indicate that hypoglycemia accounted for a large part of the variation in results.
It is debatable about whether the decrement in processing speed is solely responsible for the observed impairment in executive function (31). However, the current study demonstrates more-specific decrements. The quality of the responses was poorer during hypoglycemia in the 20 questions test, with subjects more likely to ask closed questions that failed to eliminate many of the remaining objects. In the Stroop test, the number of uncorrected and self-corrected errors was greater during hypoglycemia, suggesting that participants were less aware of an error being made so were less likely to correct the error; this has been shown previously in driving simulator studies in which participants were less likely to correct driving errors while hypoglycemic (32).
During euglycemia, glucose is the main source of energy for the brain. During hypoglycemia, alternative substrates may be used as energy sources, although in general, they ameliorate but do not reverse the effects of neuroglycopenia (33). Administration of some of these alternative fuels (e.g., amino acids, lactate) has been shown to reduce the decrement in cognitive performance observed during hypoglycemia (34,35). In these studies, insufficient information was given to determine whether there had been a significant improvement in the quality of the answers given.
Glycemic targets were maintained and symptom scores incremented appropriately during hypoglycemia. The expected decrement in performance during hypoglycemia in the cognitive marker tests was consistent with the results of similar studies (4,7,10). Previous studies have shown that several domains of cognitive function are impaired during acute hypoglycemia, including memory and spatial awareness (9,36). Previous research also demonstrated that performance of complex cognitive tasks is preferentially impaired by hypoglycemia, whereas simpler tasks, such as finger tapping or reaction time, are less affected (4,10).
The executive function tests used in the current study examined a series of diverse yet interdependent complex cognitive processes. The category switching part of the verbal fluency test examined cognitive switching abilities similar to those examined by the Trail Making B test. The sorting test examined concept formation and problem-solving abilities. The 20 questions test assessed the ability to perceive subcategories within the list of objects presented. The tower test assessed spatial planning and the ability to maintain an instruction set. All of these cognitive tests showed impairment during hypoglycemia.
The Stroop test uses a series of combined scores to give an indication of performance in both lower- and higher-level cognitive functioning (Table 2); all but Stroop E showed significant impairment during hypoglycemia. Stroop E is the additional time taken to switch between tasks and, thus, is a measure of cognitive flexibility; it is noteworthy that other measures of cognitive flexibility, such as the verbal fluency test and Trail Making B test were impaired by hypoglycemia.
The cognitive processes affected will impinge on performance of everyday activities. Impaired ability to switch between semantic categories and retrieval from semantic knowledge would interfere with planning and make it difficult to construct a list of items suitable for a specific purpose, such as for a camping trip). In such a scenario, impaired spatial planning and failure to maintain an instructional set would make loading an automobile or erecting a tent more difficult to undertake.
Compared with their performance during euglycemia, participants with diabetes experienced a greater detrimental effect of hypoglycemia than those without diabetes in the sorting test (time taken), Stroop A, and Stroop B. Higher baseline intelligence is known to improve performance during the Stroop test; the effect of higher intelligence scores (in adults) on performance during the sorting test is less clear cut (28,37). Whereas the groups with and without diabetes were otherwise well matched (Table 1), the significantly lower NART scores (a measure of crystallized intelligence) in the participants with diabetes may have been a confounding factor. However, NART scores entered as a covariate indicate that participants with diabetes experienced a greater detrimental effect of hypoglycemia compared with those without diabetes in the sorting test (time taken) and Stroop B but not in Stroop A. Differential effects of hypoglycemia between participants with and without diabetes were found in a study of a different cognitive domain, psychomotor function (10). However, in that study, the detrimental effect of hypoglycemia was greater in participants without diabetes. The mechanism underlying this differing effect of diabetes status of performance during hypoglycemia is unknown.
A possible weakness of the current study is that time constraints allowed each test of cognitive function to be performed only once during each experimental session. Tests to assess several cognitive domains take time to perform, and it has been suggested that cognitive adaptation may occur when exposure to hypoglycemia is relatively prolonged (5,38), although this premise has been disputed (39). Cerebral adaptation to hypoglycemia, which results in a lesser magnitude of impairment of cognitive function, is a feature of persons with type 1 diabetes who have an impaired awareness of hypoglycemia (6); individuals with this acquired syndrome were excluded from the study.
The current study provides further evidence of a global impairment of most high-level cognitive functions during hypoglycemia. Both the speed of information processing and the quality of answers given were adversely affected by hypoglycemia. Executive function is essential for many everyday activities (e.g., driving, analyzing data, planning events); its disruption during hypoglycemia will have a profound effect on the functioning of a person with diabetes.
Acknowledgments
I.J.D. and B.M.F. are members of the University of Edinburgh Centre for Cognitive Ageing and Cognitive Epidemiology, part of the cross council Lifelong Health and Wellbeing Initiative (G0700704/84698). The study received funding from the Biotechnology and Biological Sciences Research Council, Engineering and Physical Sciences Research Council, Economic and Social Research Council, and Medical Research Council.
No potential conflicts of interest relevant to this article were reported.
A.J.G. researched data, performed the statistical analysis in discussion with I.J.D., and wrote the manuscript. I.J.D. and B.M.F. helped with the study design, contributed to the discussion, and reviewed and edited the manuscript. A.J.G. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
The authors thank the staff of the Wellcome Trust, Clinical Research Facility, Royal Infirmary of Edinburgh for assistance.
References
- 1.Pedersen-Bjergaard U, Pramming S, Heller SR, et al. Severe hypoglycaemia in 1076 adult patients with type 1 diabetes: influence of risk markers and selection. Diabetes Metab Res Rev 2004;20:479–486 [DOI] [PubMed] [Google Scholar]
- 2.Mitrakou A, Ryan C, Veneman T, et al. Hierarchy of glycemic thresholds for counterregulatory hormone secretion, symptoms, and cerebral dysfunction. Am J Physiol 1991;260:E67–E74 [DOI] [PubMed] [Google Scholar]
- 3.Maran A, Lomas J, Macdonald IA, Amiel SA. Lack of preservation of higher brain function during hypoglycaemia in patients with intensively-treated IDDM. Diabetologia 1995;38:1412–1418 [DOI] [PubMed] [Google Scholar]
- 4.Warren RE, Frier BM. Hypoglycaemia and cognitive function. Diabetes Obes Metab 2005;7:493–503 [DOI] [PubMed] [Google Scholar]
- 5.Evans ML, Pernet A, Lomas J, Jones J, Amiel SA. Delay in onset of awareness of acute hypoglycemia and of restoration of cognitive performance during recovery. Diabetes Care 2000;23:893–897 [DOI] [PubMed] [Google Scholar]
- 6.Zammitt NN, Warren RE, Deary IJ, Frier BM. Delayed recovery of cognitive function following hypoglycemia in adults with type 1 diabetes: effect of impaired awareness of hypoglycemia. Diabetes 2008;57:732–736 [DOI] [PubMed] [Google Scholar]
- 7.Sommerfield AJ, Deary IJ, McAulay V, Frier BM. Short-term, delayed, and working memory are impaired during hypoglycemia in individuals with type 1 diabetes. Diabetes Care 2003;26:390–396 [DOI] [PubMed] [Google Scholar]
- 8.Warren RE, Zammitt NN, Deary IJ, Frier BM. The effects of acute hypoglycaemia on memory acquisition and recall and prospective memory in type 1 diabetes. Diabetologia 2007;50:178–185 [DOI] [PubMed] [Google Scholar]
- 9.Wright RJ, Frier BM, Deary IJ. Effects of acute insulin-induced hypoglycemia on spatial abilities in adults with type 1 diabetes. Diabetes Care 2009;32:1503–1506 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Geddes J, Deary IJ, Frier BM. Effects of acute insulin-induced hypoglycaemia on psychomotor function: people with type 1 diabetes are less affected than non-diabetic adults. Diabetologia 2008;51:1814–1821 [DOI] [PubMed] [Google Scholar]
- 11.American Psychiatric Association Diagnostic and statistical manual of mental disorders. Washington, American Psychiatric Association, 1994 [Google Scholar]
- 12.McNally K, Rohan J, Pendley JS, Delamater A, Drotar D. Executive functioning, treatment adherence, and glycemic control in children with type 1 diabetes. Diabetes Care 2010;33:1159–1162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Miller E. Verbal fluency as a function of a measure of verbal intelligence and in relation to different types of cerebral pathology. Br J Clin Psychol 1984;23:53–57 [DOI] [PubMed] [Google Scholar]
- 14.Elliott R. Executive functions and their disorders. Br Med Bull 2003;65:49–59 [DOI] [PubMed] [Google Scholar]
- 15.Delis D, Kaplan E, Kramer J. Delis Kaplan Executive Function System (Examiner’s Manuel). San Antonio, The Psychological Corporation, 2001 [Google Scholar]
- 16.Homack S, Lee D, Riccio CA. Test review: Delis-Kaplan executive function system. J Clin Exp Neuropsychol 2005;27:599–609 [DOI] [PubMed] [Google Scholar]
- 17.Salthouse TA. What cognitive abilities are involved in trail-making performance? Intelligence 2011;39:222–232 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wechsler D. Wechsler Adult Intelligence Scale III Manual. New York, The Psychological Corporation, 1981 [Google Scholar]
- 19.Gold AE, MacLeod KM, Frier BM. Frequency of severe hypoglycemia in patients with type I diabetes with impaired awareness of hypoglycemia. Diabetes Care 1994;17:697–703 [DOI] [PubMed] [Google Scholar]
- 20.DeFronzo RA, Tobin JD, Andres R. Glucose clamp technique: a method for quantifying insulin secretion and resistance. Am J Physiol 1979;237:E214–E223 [DOI] [PubMed] [Google Scholar]
- 21.Deary IJ, Hepburn DA, MacLeod KM, Frier BM. Partitioning the symptoms of hypoglycaemia using multi-sample confirmatory factor analysis. Diabetologia 1993;36:771–777 [DOI] [PubMed] [Google Scholar]
- 22.Deary I, Whalley L, Crawford J. An ‘instantaneous’ estimate of a lifetime's cognitive change. Intelligence 2004;32:113–119 [Google Scholar]
- 23.Heller SR, Macdonald IA. The measurement of cognitive function during acute hypoglycaemia: experimental limitations and their effect on the study of hypoglycaemia unawareness. Diabet Med 1996;13:607–615 [DOI] [PubMed] [Google Scholar]
- 24.Reitan R. Validity of the trail making test as an indicator of organic brain damage. Percept Mot Skills 1958;8:271–276 [Google Scholar]
- 25.Berg EA. A simple objective technique for measuring flexibility in thinking. J Gen Psychol 1948;39:15–22 [DOI] [PubMed] [Google Scholar]
- 26.Goel V, Grafman J. Are the frontal lobes implicated in “planning” functions? Interpreting data from the Tower of Hanoi. Neuropsychologia 1995;33:623–642 [DOI] [PubMed] [Google Scholar]
- 27.Stroop JR. Studies of interference in serial verbal reactions. J Exp Psychol 1935;18:643–662 [Google Scholar]
- 28.Van der Elst W, Van Boxtel MP, Van Breukelen GJ, Jolles J. The Stroop color-word test: influence of age, sex, and education; and normative data for a large sample across the adult age range. Assessment 2006;13:62–79 [DOI] [PubMed] [Google Scholar]
- 29.Hardy KJ, Scase MO, Foster DH, Scarpello JH. Effect of short term changes in blood glucose on visual pathway function in insulin dependent diabetes. Br J Ophthalmol 1995;79:38–41 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cohen J. The analysis of variance. In Statistical Power Analysis for the Behavioral Sciences. 2nd ed. Hillsdale, Lawrence Erlbaum Associates, 1988, p. 273–406 [Google Scholar]
- 31.Albinet CT, Boucard G, Bouquet CA, Audiffren M. Processing speed and executive functions in cognitive aging: how to disentangle their mutual relationship? Brain Cogn 2012;79:1–11 [DOI] [PubMed] [Google Scholar]
- 32.Cox DJ, Gonder-Frederick LA, Kovatchev BP, Julian DM, Clarke WL. Progressive hypoglycemia’s impact on driving simulation performance. Occurrence, awareness and correction. Diabetes Care 2000;23:163–170 [DOI] [PubMed] [Google Scholar]
- 33.Beall C, Ashford ML, McCrimmon RJ. The physiology and pathophysiology of the neural control of the counterregulatory response. Am J Physiol Regul Integr Comp Physiol 2012;302:R215–R223 [DOI] [PubMed] [Google Scholar]
- 34.Rossetti P, Porcellati F, Busciantella Ricci N, et al. Effect of oral amino acids on counterregulatory responses and cognitive function during insulin-induced hypoglycemia in nondiabetic and type 1 diabetic people. Diabetes 2008;57:1905–1917 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Page KA, Williamson A, Yu N, et al. Medium-chain fatty acids improve cognitive function in intensively treated type 1 diabetic patients and support in vitro synaptic transmission during acute hypoglycemia. Diabetes 2009;58:1237–1244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sommerfield AJ, Ewing FM, Strachan MW, Deary IJ, Aitken G, Frier BM. Self-treatment of mild symptomatic hypoglycaemia by people with insulin-treated diabetes. Diabet Med 2003;20:686–687 [DOI] [PubMed] [Google Scholar]
- 37.Arffa S. The relationship of intelligence to executive function and non-executive function measures in a sample of average, above average, and gifted youth. Arch Clin Neuropsychol 2007;22:969–978 [DOI] [PubMed] [Google Scholar]
- 38.Kerr D, Macdonald IA, Tattersall RB. Patients with type 1 diabetes adapt acutely to sustained mild hypoglycaemia. Diabet Med 1991;8:123–128 [DOI] [PubMed] [Google Scholar]
- 39.Gold AE, Deary IJ, MacLeod KM, Thomson KJ, Frier BM. Cognitive function during insulin-induced hypoglycemia in humans: short-term cerebral adaptation does not occur. Psychopharmacology (Berl) 1995;119:325–333 [DOI] [PubMed] [Google Scholar]



