Abstract
Proper assessment of the physiologic impact of coronary artery stenosis on the LV myocardium can affect patient prognosis and treatment decisions. Cardiac magnetic resonance imaging (CMR) assesses myocardial perfusion by imaging the myocardium during a first-pass transit of an intravenous gadolinium bolus, with spatial and temporal resolution substantially higher than nuclear myocardial perfusion imaging. Coupled with late gadolinium enhancement (LGE) imaging for infarction during the same imaging session, CMR with vasodilating stress perfusion imaging can qualitatively and quantitatively assess the myocardial extent of hypoperfusion from coronary stenosis independent of infarcted myocardium. This approach has been validated experimentally, and multiple clinical trials have established its diagnostic robustness when compared to stress single-photon emission computed tomography. In specialized centers, dobutamine stress CMR has been shown to have incremental diagnostic value above stress echocardiography due to its high imaging quality and ability to image the heart with no restriction of imaging window. This paper reviews the technical aspects, diagnostic utility, prognostic values, challenges to clinical adaptation, and future developments of stress CMR imaging.
Keywords: cardiac magnetic resonance imaging, CMR, myocardial ischemia, coronary artery stenosis, dobutamine stress CMR, perfusion


Introduction
Myocardial ischemia is an imbalance between oxygen consumption and oxygen supply at the cellular level. It is accompanied by inadequate removal of metabolites because of reduced blood flow or perfusion—differentiating it from hypoxia, which is related to reduced oxygen supply with adequate perfusion as encountered in severe anemia, or carbon monoxide poisoning. Oxygen consumption is driven by metabolic demands, which are in turn affected by many factors including heart rate, contractility, systolic wall tension, direct metabolic effect of catecholamines, and fatty acid uptake. In the normal heart, coronary blood flow is controlled by a combination of endothelial function and myogenic control. In response to metabolic requirements, as occurs during physical exertion, coronary blood flow may be increased by a factor of between three- and five-fold.
In patients with coronary stenosis, autoregulatory dilation of the resistance vessels compensates for reductions in perfusion pressure distal to the stenosis. Thus tissue perfusion distal to the stenosis is usually adequate at rest; however, with increased demand and epicardial blood flow limited by the stensosis,1 distal perfusion is compromised and tissue ischemia occurs. Stenoses of intermediate severity by angiographic criteria have been shown to have widely disparate functional significance as assessed by fractional flow reserve or quantitative analysis of blood flow using positron emission tomography (PET).1 Therefore, in the clinical setting, functional studies may be necessary to ascertain the potential for myocardial ischemia.
Ischemia may also occur due to endothelial dysfunction without significant stenosis at the epicardial level, and endothelial dysfunction may occur throughout the coronary vasculature, from the epicardial level right down to the microvasculature. Because blood flows in the direction of subepicardium to subendocardium, and oxygen requirements are greater in the subendocardial layers, the subendocardium is more vulnerable to ischemia, creating a transmural gradient of ischemia.
Imaging Ischemia
The detection of ischemia in chronic stable coronary disease requires that a stimulus be applied to expose regional differences in coronary flow reserve (flow heterogeneity) and/or induce an imbalance between oxygen demand and supply (ischemia). Exercise is the simplest and most physiological stress, producing regional perfusion abnormalities and ischemia. Inotropic agents such as dobutamine increase the metabolic demand through increased heart rate (chronotropy) and contractility coupled with some vasodilator activity. Vasodilator stressors, such as dipyridamole, adenosine, or the selective A2a receptor agonist cause coronary vasodilation. Coronary blood flow increases three- to five-fold in normal arteries but cannot increase to the same extent in arteries with significant stenosis, therefore producing flow heterogeneity in hemodynamically significant coronary obstruction and ischemia when the obstruction is severe.2
CMR Detection of Regional Wall Motion Abnormalities
CMR offers several theoretical and practical advantages over both stress echo and nuclear perfusion. It provides substantially better temporal (≤45 msec as standard for assessment of ventricular function) and spatial resolution (1.5 mm x 1.5 mm in plane resolution) to nuclear imaging without the limitations associated with poor echo windows. Thus it is an ideal tool for investigating wall motion and/or wall thickening in response to stress. Early work that demonstrated the feasibility of dobutamine stress CMR and encouraging results for diagnostic accuracy has been confirmed.3 In the 15 years following, at least 14 further studies were published using regional wall motion abnormalities on stress CMR to detect angiographically significant coronary disease, reporting sensitivities of 78% to 91% and specificities of 75% to 100%. A recent meta-analysis reports summary estimates of sensitivity and specificity of 85% and 86%, respectively.4 During this time, there have been significant improvements in cine CMR imaging that have made an important contribution to the robustness of the technique, with fast gradient echo sequences largely superseded by cine steady-state free precession at the most common 1.5T environment, allowing for superior myocardial to blood pool contrast.
Further advances in stress CMR include a more quantitative approach to the analysis of wall motion through the use of tissue tagging. Tagging involves the placement of selective saturation bands at regular intervals through the slice of interest at the onset of image acquisition. Myocardial motion in circumferential, longitudinal, and radial dimensions throughout the cardiac cycle can then be tracked and quantified by analyzing the deformation of the saturation bands. Tagging correlates well with tissue Doppler measures of longitudinal and radial strain,5 and analysis of tagging improves the sensitivity of dobutamine CMR.6 Analysis of tagged images is time-consuming, but commercially available software allows semi-automated analysis of tagging and has improved significantly on the post-processing time.
In relation to the stress applied, exercise has been employed as a stressor,7 but the logistics of achieving a sufficient level of exercise to provoke ischemia are difficult within the confines of the magnet, and pharmacological stress is almost universally employed in clinical practice. As flow heterogeneity does not lead directly to ischemic wall motion abnormalities except in cases of more severe perfusion abnormality, where real ischemia is generated, it is not surprising that cine imaging during vasodilator stress tends to be associated with lower sensitivity8 and higher specificity for detecting coronary disease compared to inotropic drugs. In one study that directly compared adenosine and dobutamine, sensitivities and specificities for detection of coronary disease by cine CMR imaging were 40% and 96% for adenosine and 89% and 80% for dobutamine.9
The impact of detecting only severe stenoses with vasodilator stress when wall motion rather than perfusion is observed may partly explain the importance of dipyridamole-induced wall motion abnormalities in predicting prognosis. In a large study of the prognostic impact of stress MR, the presence (and extent) of wall motion abnormality with dipyridamole stress was shown to be the CMR variable most predictive of subsequent clinical events during follow-up of 420 days.10
Recently, two studies have highlighted the clinical utility of dobutamine cine CMR in cardiac stress testing. Nagel et al.11 compared ischemia-induced wall motion abnormalities between dobutamine cine CMR and dobutamine stress echocardiography with harmonic imaging in 208 consecutive patients with suspected CAD prior to cardiac catheterization. Dobutamine cine CMR provided better sensitivity (89% vs. 74%) and specificity (86% vs. 70%) than dobutamine stress echocardiography for detecting >50% coronary artery stenosis. Similarly, Hundley et al. performed dobutamine cine CMR on 163 patients with poor echocardiographic windows and demonstrated 83% sensitivity and specificity in detecting >50% coronary stenosis on quantitative coronary angiography.12
In addition, dobutamine cine CMR was associated with a negative predictive value of 97% for cardiac events over the subsequent 8-month period.13 Stress CMR can therefore be used as both a diagnostic tool and an aid to prognostic evaluation of patients with known or suspected coronary disease. In addition, as with stress echo, dobutamine CMR is useful not only for detecting, localizing, and quantifying ischemia but also for identifying the effects of ischemia on valvular function.
MR Myocardial Perfusion
MR myocardial perfusion, having evolved considerably in the past decade, is now a clinically useful tool rather than a research investigation, with significant advantages for assessing chronic ischemia and viability. In particular, the comprehensive nature of assessment, whereby function, perfusion, and viability can be optimally assessed in a single sitting, is attractive for patients and healthcare providers alike. The following sections outline the principles of MR myocardial perfusion imaging, the sequences and contrast agents employed in the clinical arena, the analysis of the results, the clinical utility of the test, and the challenges posed by the technique. Wider developments in the field of MR perfusion and imaging of ischemia will also be discussed.
The contrast between blood and myocardium with MR depends largely on the proton concentration and longitudinal (T1) and transverse (T2) relaxation times. This inherent contrast may be modulated through the use of MR contrast agents or different pulse sequences weighted to emphasize T1 or T2 effects. For myocardial perfusion, imaging efforts have concentrated on expanding the contrast between areas of normal and relative hypoperfusion by introducing contrast agents that increase or decrease signal intensity depending on coronary blood flow.
First-Pass Gadolinium Perfusion Imaging
While research is hoping to refine and develop alternative methodologies to assess myocardial perfusion, the technique that has achieved greatest clinical prominence is first-pass perfusion imaging using gadolinium-based extracellular contrast agents in combination with T1-sensitive pulse sequences. A major factor in the success of this technique has been the advent of extremely fast sequences capable of acquiring sequential images of each of several myocardial slices, each capturing the relative differences in signal intensity throughout the myocardium at multiple closely spaced time points. While T1 weighting may be introduced by a saturation, inversion recovery, or a notched saturation prepulse, a saturation prepulse is most commonly applied to null the myocardium. For readout, fast imaging techniques such as fast spoiled gradient-echo, echo planar imaging (EPI), and balanced steady-state free precession have been used in combination with parallel imaging techniques14 to further increase the speed of acquisition with the least compromise to spatial resolution. The relative lack of standardizing sequences for perfusion across vendors and centers has, until relatively recently, limited the comprehensive study of MR perfusion’s clinical utility.
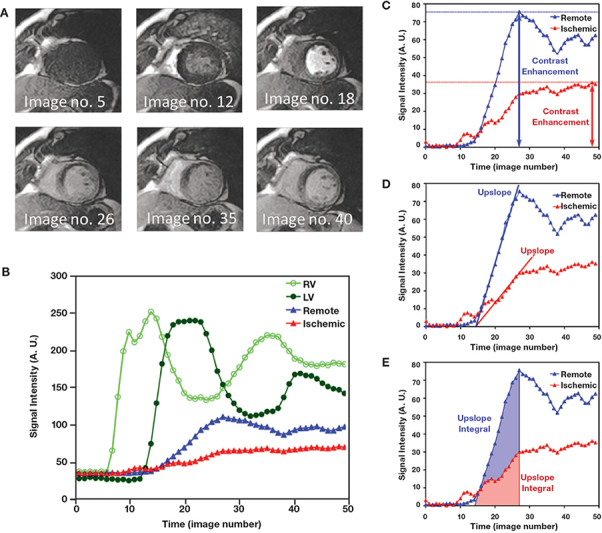
Gadolinium-based agents shorten both T1 and T2, but the predominant effect at low dose is T1 shortening, thus areas of contrast uptake are seen as hyperintense (bright) on T1 weighted images. For myocardial perfusion imaging, dynamic imaging is conducted as the contrast agent is injected through the circulation in a compact bolus. Up to 60 phases are acquired for each slice to capture the myocardium and blood pool pre-contrast, then as the contrast fills the right ventricle (RV), and then the left ventricle (LV), before perfusing the myocardium, thereby enhancing the myocardial walls. On the first pass through the capillary bed, up to 50% of circulating Gd-DTPA diffuses from the intravascular compartment to the extravascular compartment.15 Therefore, although recirculation is imaged, the most important portion of the acquisition for both visual and quantitative analysis is during the initial “first pass,” when the contrast enters and washes out of the myocardium for the first time. Differences in signal intensity between areas of myocardium reflecting areas of perfusion heterogeneity can be detected visually or analyzed semiquantitatively or quantitatively using the resultant time-intensity curves (Figure 1).
Figure 1.
(A) Qualitative interpretation of a first-pass myocardial perfusion study as a bolus of gadolinium contrast transits through the heart. Note that an anterior subendocardial perfusion defect appears starting at image no. 18 and persists at peak contrast enhancement (image no. 26) and later. (B) Time-intensity curves measured from a first-pass CMR perfusion study. The open green circles were measured in the right ventricular cavity. The dark green circles were measured in the left ventricular cavity. The blue triangles were measured in a sector representing normal vasodilated myocardium, while the red triangles came from a region of the myocardium with severely reduced perfusion (anterior subendocardium in this case). (C, D, E) Methods for semiquantitative analysis of perfusion. Common semiquantitative parameters in measuring myocardial perfusion include peak contrast enhancements, upslopes of the time-intensity curves, and upslope integrals. Courtesy of Andrew Arai, M.D., National Heart, Lung, and Blood Institute, National Institutes of Health, Bethesda, Maryland
Requirements and assumptions that must be met for MR myocardial perfusion to produce an accurate and clinically meaningful result are outlined below.
Image parameters:
Adequate temporal resolution; i.e., at least one image per slice location per heart beat during hyperemic stress state
Acquisition window as short as possible to reduce blurring from cardiac motion
Acceptable spatial resolution (approximately 2 mm in-plane, to allow discrimination of transmural perfusion gradient)
Constant magnetization to ensure similar T1-weighting across slice locations
Coverage of the left ventricle maximized as far as possible
Contrast-to-noise and signal-to-noise ratios should be sufficient to discriminate between normal and hypoperfused myocardium
Contrast dosage and administration:
Direct linear relationship between contrast dose and SI; true at low dose contrast but not high dose (>.12 mmol/kg)
Compact bolus of contrast facilitated by saline “chaser”
Contrast injection rate of (5-7 mL/sec)
Experimental models of first-pass perfusion employ left atrial catheter placement for contrast injection,1 but it is neither possible nor necessary to reproduce such conditions for clinical scans. Peripheral intravenous cannulation, preferably in the antecubital fossa, is sufficient for delivery of contrast. More important is the rate of contrast delivery, >3 mL/sec, preferably >5 mL/sec, and the coadministration of a saline bolus immediately after the contrast to ensure a compact bolus to facilitate both visual and quantitative analysis. Perfusion imaging to quantify ischemia is generally performed at rest and during stress for comparative purposes and to allow calculation of the myocardial perfusion reserve, although stress-only protocols have been validated.16 A sample protocol is shown in Table 1. Vasodilator stress using adenosine or dipyridamole is most commonly used due to the excellent safety profile and tolerability of these agents,17 but dobutamine may be used as an alternative. However, dobutamine perfusion imaging with MR is complicated by the increase in heart rate that may limit adequate coverage of the left ventricle during stress perfusion, as the number of slices that can be prescribed is inversely related to the patient heart rate. For patients undergoing vasodilator stress, depending on the sequence used, 3 to 6 short-axis slices may be combined with one or more long-axis views of the left ventricle to facilitate assessment of apical perfusion. If a notched pulse preparatory sequence is employed, the combination of long- and short-axis planes is not possible. However, this disadvantage may be somewhat offset if coverage of the left ventricle is maximized by prescribing as many short-axis slices as can be accommodated in the available time within the cardiac cycle.
Table 1.
Sample stress MR perfusion study protocol.
| Sample stress MR perfusion study protocol |
| 1) Localizers (2 minutes) |
2) Pharmacological stress infusion (5 minutes)
|
3) Stress perfusion imaging (2 minutes)
|
| 4) Top-off contrast to make up a total of 0.1 mmol/kg if necessary (1 minute) |
5) Rest cine function (5 minutes)
|
6) LGE imaging
|
Consideration should be given from the outset to the type of analysis to be undertaken as the dose administered will affect quantification of perfusion. At low doses, the relationship between contrast dose and signal intensity is approximately linear, but at higher doses (>0.12 mmol/kg) this relationship no longer holds true. A potential conflict exists between the requirements for quantitative or visual analysis. Greater contrast-to-noise ratio is available with the use of higher doses (0.1-0.15 mmol/kg), thus facilitating detection of perfusion defects by visual analysis, but signal saturation may render quantitative analyses less reliable at higher doses. For clinical purposes, qualitative visual analysis yields very acceptable diagnostic accuracy. In the Non-ST Elevation Myocardial Infarction (NSTEMI) study, the sensitivity and specificity of visual assessment of perfusion alone (using a dose of 0.05 mmol/kg) was 88% and 83%, respectively.18 In the same study, comprehensive assessment of MR information including late enhancement increased the sensitivity to 96%. In another study in which fewer than 50% of the population had prior MI, the sensitivity and specificity of visual assessment of perfusion (using a dose of 0.1 mmol/kg) for detecting coronary disease was 93% and 85%, respectively.19 However, quantitative or at least semiquantitative techniques are desirable for both clinical and research purposes.
Quantification of Perfusion
Most semiquantitative techniques involve regional analysis of the time-intensity curve. Using an accepted model of LV segmentation (such as the 17-segment AHA model), the LV wall can be divided into regions corresponding to those used to analyze wall motion abnormality or late enhancement. A region of interest is placed within the myocardial segment (or within the different layers of the myocardium, subepicardium to subendocardium, if so desired) during sequential phases; this should be done with care, to track a consistent portion of the LV wall notwithstanding the presence of ectopic beats or respiratory movement and to avoid blood pool contamination and/or other potential causes of error. The signal intensity (SI) within the region of interest is depicted as a function of time, and from this a number of parameters may be measured that can be related to perfusion, including: (1) the rate of increase in signal intensity or upslope after the bolus of contrast; (2) the maximum signal intensity increase; (3) the time to maximum increase in signal intensity; (4) the time from contrast appearance in the LV cavity to contrast appearance in myocardium; (5) time to 50% maximal signal intensity; (6) the rate of decrease in signal intensity after peak; and (7) the mean transit time using an exponential fit.
Of these, the upslope or rate of increase in signal intensity is emerging as the most suited semiquantitative analysis to clinical application due to the depth of data validating its robustness and diagnostic accuracy.20 A recent dose-ranging study using myocardial upstroke as the semiquantitative measurement parameter found that doses of 0.1 and 0.15 mmol/kg were superior to 0.05 mmol/kg for detection of coronary disease, with pooled sensitivity and specificity of 93% and 75%.16 Using the upslope of the curve renders calculation of the myocardial perfusion reserve index (MPRI) relatively straightforward: MPRI = upslope of SI curve during hyperemia/upslope of SI curve at rest.
Absolute quantification of myocardial blood flow is possible, but requires considerably more complex modeling and is subject to several important confounders. These include nonlinearity in the relationship between SI and contrast most pronounced at higher contrast dose, temporal differences between images in the saturation pulse that creates T1 weighting, uncertainty that the rapid exchange condition is met, and regional variation in the main magnetic field (B0) or radiofrequency field (B1). Measurements of absolute regional myocardial blood flow (in mL/g/min) can be made by mathematical deconvolution of the time-intensity curve using one of several models, including the Fermi21 or modified Kety techniques.22 Deconvolution modeling requires a measured arterial input function (AIF), usually from the left ventricular cavity or ascending aorta (Figures 2, 3). Measurement of the true AIF is affected by short T1s and T2* effects that occur with high-contrast agent concentrations during bolus contrast agent passage in the blood pool. Two different strategies have been proposed to overcome this problem: the “dual-bolus” method,20, 23 or the “dual-sequence” method.24-26
Figure 2.
Absolute myocardial blood flow quantitation has been well validated in experimental models.21
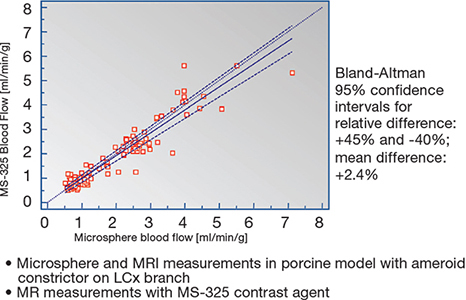
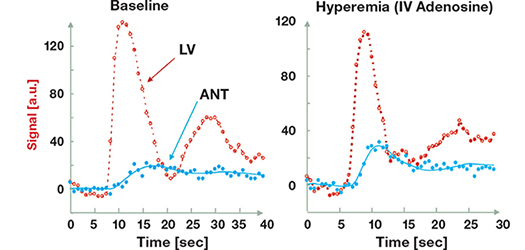
Figure 3.
MRI perfusion measured by absolute myocardial blood flow quantitation during rest and hyperemia. This patient has a normal LAD coronary flow reserve of 1:3.4
Clinical Utility of MR Myocardial Perfusion
Preliminary reports of the clinical feasibility of the first-pass technique in patients1 have been followed by multiple studies confirming its feasibility, documenting technical advances in terms of sequence design, and demonstrating highly satisfactory diagnostic performance indices. In 2000, Al Saadi et al. reported coronary sensitivity and specificity of 90% and 83% for the detection of coronary stenosis >75% in a population with suspected coronary disease (n=40) using an inversion recovery single-shot turbo gradient echo technique with dipyridamole stress.27 Ishida et al. reported per-patient sensitivity of 90% and specificity of 85% in a similar but larger population (n=104) using qualitative analysis, dipyridamole stress, and fast echoplanar readout with a notched interleaved saturation preparatory pulse.28 In the same year, Nagel et al. reported sensitivity and specificity of 88% and 90% in a cohort of 90 patients using adenosine stress.29 Similar excellent results have been reported more recently for adenosine perfusion imaging in a larger patient cohort (n=176), showing 96% sensitivity and 83% specificity for the detection of stenosis >70% using hybrid fast gradient echo and echo-planar imaging.30
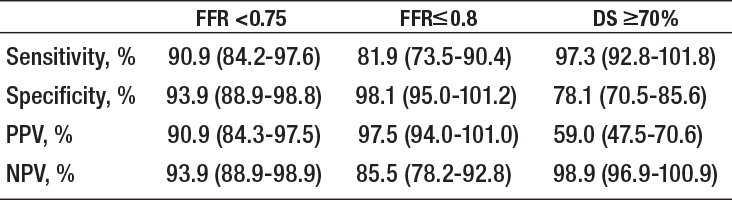
In a meta-analysis that pooled data from 14 studies of perfusion tested against coronary angiography as the gold standard, the summary estimates for sensitivity and specificity were 91% and 81% respectively.4 The studies included in this meta-analysis were relatively small single-center studies, and diagnostic accuracy was assessed only in terms of detection of angiographic stenosis, thus the widespread applicability of the technique in a clinical context based solely on these results would be questionable. However, there is substantially more evidence supporting the utility of myocardial perfusion. Single-center results have been reproduced in multicenter settings,1, 31, 32 and MR perfusion compares well with other available noninvasive methods of assessing perfusion. Comparative studies with existing nuclear perfusion techniques suggest MR perfusion imaging could realistically be used as an alternative, with superior performance relative to SPECT and excellent agreement with PET perfusion data reported.33, 34 In the recent single-center CE-MARC study that directly compared stress MR perfusion with SPECT, stress MR was found to have higher sensitivity but similar specificity as SPECT, using angiographic stenosis as a study endpoint (Figure 4).35 Comparisons with invasive physiological measurements such as fractional flow reserve (FFR) have also yielded favorable results. Two studies have shown that calculation of the myocardial perfusion reserve index or visual assessment of myocardial perfusion data enabled detection of hemodynamically important stenoses, as determined by the usual FFR cutoffs of >0.75 or 0.8 (Figure 5).36, 37 With the cardiology community increasingly focused on physiological data to aid appropriate selection of lesions for percutaneous intervention, and evidence that FFR calculation may improve selection,38, 39 noninvasive preprocedural assessment of perfusion by MR is likely to prove beneficial.
Figure 4.
Results of the CE-MARC study. Stress MR perfusion had higher diagnostic sensitivities for both single and multivessel coronary stenosis than SPECT.35 MR: magnetic resonance; CMR: cardiac magnetic resonance; SPECT: single-photon emission computed tomography; LAD: left anterior descending; LCx: left circumflex; RCA: right anterior descending; CE-MARC: clinical evaluation of magnetic resonance imaging in coronary heart disease.
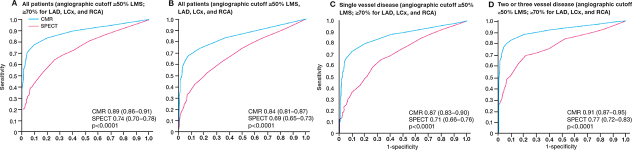
Figure 5.
The sensitivity, specificity, and positive and negative predictive values of stress MR perfusion imaging for the detection of significant coronary artery disease by fractional flow reserve measurements using various thresholds. Values in parentheses are 95% confidence intervals.36,37 MR: magnetic resonance; FFR: fractional flow reserve; DS: angiographic stenosis; PPV: positive predictive value; NPV: negative predictive value.
Beyond diagnosis, and prediction of the hemodynamic significance of stenoses, MR myocardial perfusion has been shown to be an important method to determine prognosis. In a population with chest pain presenting to the emergency department, MR perfusion has been shown to have a sensitivity of 100% and a specificity of 93% in predicting subsequent death from myocardial infarction or detection of coronary stenosis over a 1-year follow-up.40
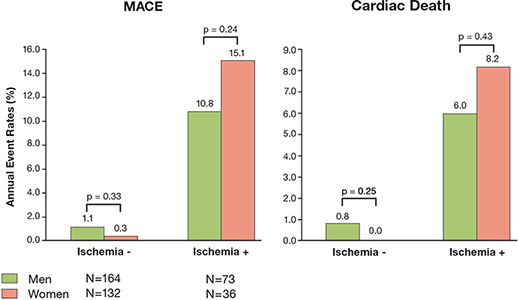
In another study of 218 patients followed for 2 years after having negative perfusion studies, there were no deaths or infarction, one percutaneous intervention, and one coronary artery bypass operation.41 Jahnke et al. used both adenosine myocardial perfusion and dobutamine MR for wall motion abnormalities to study 493 patients who were subsequently followed up for 3 years.42 In this study, stress MR, either perfusion or wall motion, contributed incremental value over traditional factors such as age, gender, tobacco smoking, and diabetes in risk stratification, although combining both techniques did not further increase the yield in a statistically significant manner. A normal perfusion study was associated with an extremely low event rate—0.7% at 2 years, 2.3% at 3 years—while an abnormal perfusion study had corresponding event rates of 12.2% and 16.3%. In multivariate analysis, adjusting for other risks, detection of a perfusion abnormality was associated with a 10-fold increase in risk of hard events (death or myocardial infarction). In a study of 243 patients, Steel demonstrated that reversible perfusion abnormality as a marker of ischemia and presence of infarction, especially when infarction was clinically unrecognized, provided complementary prognostic value in patients with a clinical suspicion of ischemia. Stress MR has substantially higher spatial resolution and potentially less artifacts in imaging women, and it does not involve ionizing radiation.43 It has also been shown to have strong prognostic value for imaging of CAD in women (Figure 6).44
Figure 6.
Stress MR for assessment of coronary artery disease across the genders. In this study of 424 patients, a negative stress MR was associated with very low event rates in both genders.44 Ischemia -: evidence of ischemia absent; Ischemia +: evidence of ischemia present; R: magnetic resonance.
In light of the convincing evidence supporting the diagnostic and prognostic value of stress MR in the detection if ischemia, it can no longer be considered an investigative technique but recognized as a reasonable investigative tool in the appropriate clinical setting.
Future Directions in Perfusion MR
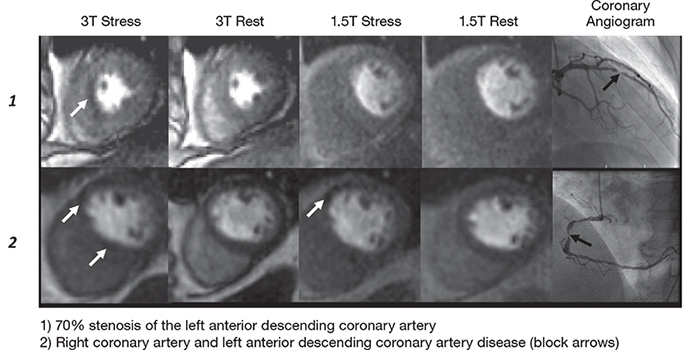
Developments in MR perfusion can be broadly categorized as occurring in the imaging sequences or contrast agents. Goals in sequence development are to further improve spatial resolution, increase ventricular coverage, and limit artifacts. The potential for increased signal-to-noise ratio and contrast enhancement used to improve spatial resolution and image quality has already been realized with the advent of clinical 3T systems, and preliminary reports demonstrate promise for improved clinical diagnostic accuracy (Figure 7).45
Figure 7.
Cardiac magnetic resonance perfusion imaging at 3T for the detection of coronary artery disease: a comparison with 1.5T. 3T provides higher signal-to-noise and contrast-to-noise ratios to allow a higher diagnostic accuracy.45
Other investigators are pursuing methods of speeding image acquisition, such as k space and time-sensitive encoding with parallel imaging (kt SENSE), to improve spatial resolution to the level achievable with cine or late-enhancement imaging (<1.5 mm x 1.5 mm in-plane).46 Clinical benefits would include the potential to image RV perfusion and to facilitate greater integration of cine, perfusion, and late enhancement images.
Although, as discussed above, extracellular gadolinium-based agents are the mainstay of myocardial perfusion imaging in the clinical setting, assessment of myocardial perfusion may be performed with both endogenous and exogenous contrast. Exogenous contrast agents may be further classified according to cellular distribution (intravascular, intracellular, or extracellular), the effect on signal intensity (increase in signal intensity with paramagnetic agents or decrease in signal intensity with supermagnetic agents), or specificity for target tissue (e.g., ischemia or necrotic tissue).47
One example of endogenous contrast in CMR is blood oxygen level dependency (BOLD) imaging, which operates on the principle that deoxygenated hemoglobin is paramagnetic and causes signal loss in T2*- or T2-weighted images. BOLD has been used to assess changes in myocardial venous blood oxygenation secondary to perfusion changes in human as well as animal studies, and it has demonstrated characteristics that capture the abundance of deoxygenated hemoglobin downstream from a severe stenotic coronary lesion. While BOLD is currently not at the stage of clinical application, it has the advantage of characterizing a unique physiologic consequence of coronary artery disease without any need for endogenous contrast. Arterial spin-labeling is another experimental method that uses endogenous contrast to assess myocardial perfusion. Both BOLD and arterial spin-labeling may be advantageous in a 3T environment given a higher signal-to-noise level compared to 1.5T.
Funding Statement
Funding/Support: Dr. Daly receives research funding from Abbott Vascular and speaker’s fees on behalf of Sanofi, Menarini Group, and Servier pharmaceuticals.
Footnotes
Conflict of Interest Disclosure: The authors have completed and submitted the Methodist DeBakey Cardiovascular Journal Conflict of Interest Statement and none were reported.
References
- 1.Di Carli M, Czernin J, Hoh CK, Gerbaudo VH, Brunken RC, Huang SC, et al. Relation among stenosis severity, myocardial blood flow, and flow reserve in patients with coronary artery disease. Circulation. 1995 Apr 1;91(7):1944–51. doi: 10.1161/01.cir.91.7.1944. [DOI] [PubMed] [Google Scholar]
- 2.Daly CA, Coelho-Filho OR, Kwong RY. Chronic myocardial ischemia and viability. In: Kramer CM editor. Multimodality imaging in cardiovascular medicine. New York: Demos Medical Publishing, LLC.; 2011. p. 138-57. [Google Scholar]
- 3.Pennell DJ, Underwood SR, Manzara CC, Swanton RH, Walker JM, Ell PJ, et al. Magnetic resonance imaging during dobutamine stress in coronary artery disease. Am J Cardiol. 1992 Jul 1;70(1):34–40. doi: 10.1016/0002-9149(92)91386-i. [DOI] [PubMed] [Google Scholar]
- 4.Nandalur KR, Dwamena BA, Choudhri AF, Nandalur MR, Carlos RC. Diagnostic performance of stress cardiac magnetic resonance imaging in the detection of coronary artery disease: a meta-analysis. J Am Coll Cardiol. 2007 Oct 1;50(14):1343–53. doi: 10.1016/j.jacc.2007.06.030. [DOI] [PubMed] [Google Scholar]
- 5.Edvardsen T, Gerber BL, Garot J, Bluemke DA, Lima JAC, Smiseth OA. Quantitative assessment of intrinsic regional myocardial deformation by Doppler strain rate echocardiography in humans: validation against three-dimensional tagged magnetic resonance imaging.[see comment]. Circulation. 2002 Jul 2;106(1):50–6. doi: 10.1161/01.cir.0000019907.77526.75. [DOI] [PubMed] [Google Scholar]
- 6.Kuijpers D, Ho KY, van Dijkman PR, Vliegenthart R, Oudkerk M. Dobutamine cardiovascular magnetic resonance for the detection of myocardial ischemia with the use of myocardial tagging. Circulation. 2003 Apr 1;107(12):1592–7. doi: 10.1161/01.CIR.0000060544.41744.7C. [DOI] [PubMed] [Google Scholar]
- 7.Rerkpattanapipat P, Gandhi SK, Darty SN, Williams RT, Davis AD, Mazur W, et al. Feasibility to detect severe coronary artery stenoses with upright treadmill exercise magnetic resonance imaging. Am J Cardiol. 2003 Aug 31;92(5):603–6. doi: 10.1016/s0002-9149(03)00734-3. [DOI] [PubMed] [Google Scholar]
- 8.Pennell DJ, Underwood SR, Ell PJ, Swanton RH, Walker JM, Longmore DB. Dipyridamole magnetic resonance imaging: a comparison with thallium-201 emission tomography. Br Heart J. 1990 Dec 1;64(6):362–9. doi: 10.1136/hrt.64.6.362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Paetsch I, Jahnke C, Wahl A, Gebker R, Neuss M, Fleck E, et al. Comparison of dobutamine stress magnetic resonance, adenosine stress magnetic resonance, and adenosine stress magnetic resonance perfusion. Circulation. 2004 Aug 17;110(7):835–42. doi: 10.1161/01.CIR.0000138927.00357.FB. [DOI] [PubMed] [Google Scholar]
- 10.Bodi V, Sanchis J, Lopez-Lereu MP, Nunez J, Mainar L, Monmeneu JV, et al. Prognostic value of dipyridamole stress cardiovascular magnetic resonance imaging in patients with known or suspected coronary artery disease. J Am Coll Cardiol. 2007 Sep 17;50(12):1174–9. doi: 10.1016/j.jacc.2007.06.016. [DOI] [PubMed] [Google Scholar]
- 11.Nagel E, Lehmkuhl HB, Bocksch W, Klein C, Vogel U, Frantz E, et al. Noninvasive diagnosis of ischemia-induced wall motion abnormalities with the use of high-dose dobutamine stress MRI: comparison with dobutamine stress echocardiography. Circulation. 1999 Feb 16;99(6):763–70. doi: 10.1161/01.cir.99.6.763. [DOI] [PubMed] [Google Scholar]
- 12.Hundley WG, Hamilton CA, Thomas MS, Herrington DM, Salido TB, Kitzman DW, et al. Utility of fast cine magnetic resonance imaging and display for the detection of myocardial ischemia in patients not well suited for second harmonic stress echocardiography.[see comment]. Circulation. 1999 Oct 18;100(16):1697–702. doi: 10.1161/01.cir.100.16.1697. [DOI] [PubMed] [Google Scholar]
- 13.Hundley WG, Morgan TM, Neagle CM, Hamilton CA, Rerkpattanapipat P, Link KM. Magnetic resonance imaging determination of cardiac prognosis. Circulation. 2002 Oct 29;106(18):2328–33. doi: 10.1161/01.cir.0000036017.46437.02. [DOI] [PubMed] [Google Scholar]
- 14.Heidemann RM, Ozsarlak O, Parizel PM, Michiels J, Kiefer B, Jellus V, et al. A brief review of parallel magnetic resonance imaging. Eur Radiol. 2003 Sep 30;13(10):2323–37. doi: 10.1007/s00330-003-1992-7. [DOI] [PubMed] [Google Scholar]
- 15.Tong CY, Prato FS, Wisenberg G, Lee TY, Carroll E, Sandler D, et al. Measurement of the extraction efficiency and distribution volume for Gd-DTPA in normal and diseased canine myocardium. Magn Reson Med. 1993 Sep 1;30(3):337–46. doi: 10.1002/mrm.1910300310. [DOI] [PubMed] [Google Scholar]
- 16.Giang TH, Nanz D, Coulden R, Friedrich MG, Graves MJ, Al-Saadi N, et al. Detection of coronary artery disease by magnetic resonance myocardial perfusion imaging with various contrast medium doses: first European multi-centre experience. Eur Heart J. 2004 Sep;25(18):1657–65. doi: 10.1016/j.ehj.2004.06.037. [DOI] [PubMed] [Google Scholar]
- 17.Cerqueira M, Verani MS, Schwaiger M, Heo J, Iskandrian AS. Safety profile of adenosine stress perfusion imaging: results from the Adenoscan Multicenter Trial Registry. J Am Coll Cardiol. 1994 Feb;23(2):384–89. doi: 10.1016/0735-1097(94)90424-3. [DOI] [PubMed] [Google Scholar]
- 18.Plein S, Greenwood JP, Ridgway JP, Cranny G, Ball SG, Sivananthan MU. Assessment of non-ST-segment elevation acute coronary syndromes with cardiac magnetic resonance imaging. J Am Coll Cardiol. 2004 Dec 7;44(11):2173–81. doi: 10.1016/j.jacc.2004.08.056. [DOI] [PubMed] [Google Scholar]
- 19.Takase B, Nagata M, Kihara T, Kameyawa A, Noya K, Matsui T, et al. Whole-heart dipyridamole stress first-pass myocardial perfusion MRI for the detection of coronary artery disease. Jpn Heart J. 2004 May;45(3):475–86. doi: 10.1536/jhj.45.475. [DOI] [PubMed] [Google Scholar]
- 20.Hsu LY, Ingkanisorn WP, Kellman P, Aletras AH, Arai AE. Quantitative myocardial infarction on delayed enhancement MRI. Part II: Clinical application of an automated feature analysis and combined thresholding infarct sizing algorithm. J Magn Reson Imaging. 2006 Mar 1;23(3):309–14. doi: 10.1002/jmri.20495. [DOI] [PubMed] [Google Scholar]
- 21.Jerosch-Herold M, Wilke N, Stillman AE. Magnetic resonance quantification of the myocardial perfusion reserve with a Fermi function model for constrained deconvolution. Med Phys. 1998;25(1):73–84. doi: 10.1118/1.598163. [DOI] [PubMed] [Google Scholar]
- 22.Larsson HB, Stubgaard M, Sondergaard L, Henriksen O. In vivo quantification of the unidirectional influx constant for Gd-DTPA diffusion across the myocardial capillaries with MR imaging. J Magn Reson Imaging. 1994 May-Jun;4(3):433–40. doi: 10.1002/jmri.1880040332. [DOI] [PubMed] [Google Scholar]
- 23.Christian TF, Rettmann DW, Aletras AH, Liao SL, Taylor JL, Balaban RS, et al. Absolute myocardial perfusion in canines measured by using dual-bolus first-pass MR imaging. Radiology. 2004 Sep;232(3):677–84. doi: 10.1148/radiol.2323030573. [DOI] [PubMed] [Google Scholar]
- 24.Gatehouse PD, Elkington AG, Ablitt NA, Yang GZ, Pennell DJ, Firmin DN. Accurate assessment of the arterial input function during high-dose myocardial perfusion cardiovascular magnetic resonance. J Magn Reson Imaging. 2004 Jul;20(1):39–45. doi: 10.1002/jmri.20054. [DOI] [PubMed] [Google Scholar]
- 25.Elkington AG, He T, Gatehouse PD, Prasad SK, Firmin DN, Pennell DJ. Optimization of the arterial input function for myocardial perfusion cardiovascular magnetic resonance. J Magn Reson Imaging. 2005 Apr 1;21(4):354–9. doi: 10.1002/jmri.20294. [DOI] [PubMed] [Google Scholar]
- 26.Kim D, Axel L. Multislice dual-imaging sequence for increasing the dynamic range of the contrast-enhanced blood signal and CNR of myocardial enhancement at 3T. J Magn Reson Imaging. 2006 Jan;23(1):81–6. doi: 10.1002/jmri.20471. [DOI] [PubMed] [Google Scholar]
- 27.Al-Saadi N, Nagel E, Gross M, Bornstedt A, Schnackenburg B, Klein C, et al. Noninvasive detection of myocardial ischemia from perfusion reserve based on cardiovascular magnetic resonance. Circulation. 2000 Mar 28;101(12):1379–83. doi: 10.1161/01.cir.101.12.1379. [DOI] [PubMed] [Google Scholar]
- 28.Ishida N, Sakuma H, Motoyasu M, Okinaka T, Isaka N, Nakano T, et al. Noninfarcted myocardium: correlation between dynamic first-pass contrast-enhanced myocardial MR imaging and quantitative coronary angiography. Radiology. 2003 Sep 30;229(1):209–16. doi: 10.1148/radiol.2291021118. [DOI] [PubMed] [Google Scholar]
- 29.Nagel E, Klein C, Paetsch I, Hettwer S, Schnackenburg B, Wegscheider K, et al. Magnetic resonance perfusion measurements for the noninvasive detection of coronary artery disease. Circulation. 2003 Jul 29;108(4):432–7. doi: 10.1161/01.CIR.0000080915.35024.A9. [DOI] [PubMed] [Google Scholar]
- 30.Pilz S, Scharnagl H, Tiran B, Seelhorst U, Wellnitz B, Boehm BO, et al. Free fatty acids are independently associated with all-cause and cardiovascular mortality in subjects with coronary artery disease. J Clin Endocrinol Metab. 2006 Jul;91(7):2542–7. doi: 10.1210/jc.2006-0195. [DOI] [PubMed] [Google Scholar]
- 31.Schwitter J, Wacker CM, van Rossum AC, Lombardi M, Al-Saadi N, Ahlstrom H, et al. MR-IMPACT: comparison of perfusion-cardiac magnetic resonance with single-photon emission computed tomography for the detection of coronary artery disease in a multicentre, multivendor, randomized trial. Eur Heart J. 2008 Jan 31;29(4):480–9. doi: 10.1093/eurheartj/ehm617. [DOI] [PubMed] [Google Scholar]
- 32.Schwitter J, Wacker CM, Wilke N, Al-Saadi N, Sauer E, Huettle K, et al. MR-IMPACT II: Magnetic Resonance Imaging for Myocardial Perfusion Assessment in Coronary artery disease Trial: perfusion-cardiac magnetic resonance vs. single-photon emission computed tomography for the detection of coronary artery disease: a comparative multicentre, multivendor trial. Eur Heart J. 2013 Mar;34(10):775–81. doi: 10.1093/eurheartj/ehs022. [DOI] [PubMed] [Google Scholar]
- 33.Schwitter J. Myocardial perfusion imaging by cardiac magnetic resonance. J Nucl Cardiol. 2006 Nov;13(6):841–54. doi: 10.1016/j.nuclcard.2006.09.008. [DOI] [PubMed] [Google Scholar]
- 34.Schwitter J, Nanz D, Kneifel S, Bertschinger KM, Büchi M, Knüsel PR, et al. Assessment of myocardial perfusion in coronary artery disease by magnetic resonance: a comparison with positron emission tomography and coronary angiography. Circulation. 2001 May 8;103(18):2230–5. doi: 10.1161/01.cir.103.18.2230. [DOI] [PubMed] [Google Scholar]
- 35.Greenwood JP, Maredia N, Younger JF, Brown JM, Nixon J, Everett CC, et al. Cardiovascular magnetic resonance and single-photon emission computed tomography for diagnosis of coronary heart disease (CE-MARC): a prospective trial. Lancet. 2012 Feb 4;379(9814):453–60. doi: 10.1016/S0140-6736(11)61335-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rieber J, Huber A, Erhard I, Mueller S, Schweyer M, Koenig A, et al. Cardiac magnetic resonance perfusion imaging for the functional assessment of coronary artery disease: a comparison with coronary angiography and fractional flow reserve. Eur Heart J. 2006 Jun;27(12):1465–71. doi: 10.1093/eurheartj/ehl039. [DOI] [PubMed] [Google Scholar]
- 37.Watkins S, McGeoch R, Lyne J, Steedman T, Good R, McLaughlin MJ, et al. Validation of magnetic resonance myocardial perfusion imaging with fractional flow reserve for the detection of significant coronary heart disease. Circulation. 2009 Dec 1;20(22):2207–13. doi: 10.1161/CIRCULATIONAHA.109.872358. [DOI] [PubMed] [Google Scholar]
- 38.Fearon WF, Tonino PA, De Bruyne B, Siebert U, Pijls NH. Rationale and design of the Fractional Flow Reserve versus Angiography for Multivessel Evaluation (FAME) study. Am Heart J. 2007 Sep 30;154(4):632–6. doi: 10.1016/j.ahj.2007.06.012. [DOI] [PubMed] [Google Scholar]
- 39.Pijls NH, van Schaardenburgh P, Manoharan G, Boersma E, Bech JW, van’t Veer M, et al. Percutaneous coronary intervention of functionally nonsignificant stenosis: 5-year follow-up of the DEFER Study. J Am Coll Cardiol. 2007 May 29;49(21):2105–11. doi: 10.1016/j.jacc.2007.01.087. [DOI] [PubMed] [Google Scholar]
- 40.Ingkanisorn WP, Kwong RY, Bohme NS, Geller NL, Rhoads KL, Dyke CK, et al. Prognosis of negative adenosine stress magnetic resonance in patients presenting to an emergency department with chest pain. J Am Coll Cardiol. 2006 Apr 4;47(7):1427–32. doi: 10.1016/j.jacc.2005.11.059. [DOI] [PubMed] [Google Scholar]
- 41.Pilz G, Jeske A, Klos M, Ali E, Hoefling B, Scheck R, et al. Prognostic value of normal adenosine-stress cardiac magnetic resonance imaging. Am J Cardiol. 2008 May 15;101(10):1408–12. doi: 10.1016/j.amjcard.2008.01.019. [DOI] [PubMed] [Google Scholar]
- 42.Jahnke C, Nagel E, Gebker R, Kokocinski T, Kelle S, Manka R, et al. Prognostic value of cardiac magnetic resonance stress tests: adenosine stress perfusion and dobutamine stress wall motion imaging. Circulation. 2007 Apr 2;115(13):1769–76. doi: 10.1161/CIRCULATIONAHA.106.652016. [DOI] [PubMed] [Google Scholar]
- 43.Steel K, Broderick R, Gandla V, Larose E, Resnic F, Jerosch-Herold M, et al. Complementary prognostic values of stress myocardial perfusion and late gadolinium enhancement imaging by cardiac magnetic resonance in patients with known or suspected coronary artery disease. Circulation. 2009 Oct 6;120(14):1390–400. doi: 10.1161/CIRCULATIONAHA.108.812503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Coelho-Filho OR, Seabra LF, Mongeon FP, Abdullah SM, Francis SA, Blankstein R, et al. Stress myocardial perfusion imaging by CMR provides strong prognostic value to cardiac events regardless of patient’s sex. JACC Cardiovasc Imaging. 2011 Aug;4(8):850–61. doi: 10.1016/j.jcmg.2011.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cheng AS, Pegg TJ, Karamitsos TD, Searle N, Jerosch-Herold M, Choudhury RP, et al. Cardiovascular magnetic resonance perfusion imaging at 3-tesla for the detection of coronary artery disease: a comparison with 1.5-tesla. J Am Coll Cardiol. 2007 Jun 26;49(25):2440–9. doi: 10.1016/j.jacc.2007.03.028. [DOI] [PubMed] [Google Scholar]
- 46.Plein S, Schwitter J, Suerder D, Greenwood JP, Boesiger P, Kozerke S. k-Space and time sensitivity encoding-accelerated myocardial perfusion MR imaging at 3.0 T: comparison with 1.5T. Radiology. 2008 Nov;249(2):493–500. doi: 10.1148/radiol.2492080017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Croisille P, Revel D, Saeed M. Contrast agents and cardiac MR imaging of myocardial ischemia: from bench to bedside. Eur Radiol. 2006 Sep;16(9):1951–63. doi: 10.1007/s00330-006-0244-z. [DOI] [PubMed] [Google Scholar]


