Abstract
To perform a systematic review of the utility of the Beck Depression Inventory for detecting depression in medical settings, this article focuses on the revised version of the scale (Beck Depression Inventory-II), which was reformulated according to the DSM-IV criteria for major depression. We examined relevant investigations with the Beck Depression Inventory-II for measuring depression in medical settings to provide guidelines for practicing clinicians. Considering the inclusion and exclusion criteria seventy articles were retained. Validation studies of the Beck Depression Inventory-II, in both primary care and hospital settings, were found for clinics of cardiology, neurology, obstetrics, brain injury, nephrology, chronic pain, chronic fatigue, oncology, and infectious disease. The Beck Depression Inventory-II showed high reliability and good correlation with measures of depression and anxiety. Its threshold for detecting depression varied according to the type of patients, suggesting the need for adjusted cut-off points. The somatic and cognitive-affective dimension described the latent structure of the instrument. The Beck Depression Inventory-II can be easily adapted in most clinical conditions for detecting major depression and recommending an appropriate intervention. Although this scale represents a sound path for detecting depression in patients with medical conditions, the clinician should seek evidence for how to interpret the score before using the Beck Depression Inventory-II to make clinical decisions.
Keywords: Beck Depression Inventory, Depression, Medical Illness, Psychometric Scale, Screening, Validation Study
INTRODUCTION
Patients with chronic medical illness have a high prevalence of major depressive illness (1). Depressive symptoms may co-occur with serious medical illnesses, such as heart disease, stroke, cancer, neurological disease, HIV infection, and diabetes (1-3). The functional impairment associated with medical illnesses often causes depression. Patients who present depression along with medical illness tend to have more severe symptoms, more difficulty adjusting to their health condition, and more medical costs than patients who do not have co-existing depression (2). While prompt treatment of depression can improve the outcome of the co-occurring physical illness, proper and early recognition of treatable depression can result in a faster recovery and can shorten the patient's hospital stay.
Formal assessment of depression by a liaison psychiatrist or clinician-administered instruments, such as the Hamilton Depression Rating Scale (4) and the Montgomery-Åsberg Depression Rating Scale (5), are onerous to implement in routine clinical settings. In contrast, self-report measures for depression can be cost-effective for use in busy specialty medical clinics. Throughout the second half of the 20th century, along with the discovery of effective antidepressant drugs and the development of cognitive-behavioral therapy, several patient-rated assessment scales for detecting depression were proposed. Popular instruments include the Beck Depression Inventory (BDI) (6), the Self-Rating Depression Scale (7), the Center for Epidemiologic Studies Depression Scale (8), the Patient Health Questionnaire-9 (9), the Inventory of Depressive Symptomatology (10), and the Depression in the Medically Ill (11). Alternative scales have been developed to measure depression in specific populations, such as postpartum women (12) and patients with schizophrenia (13). Other scales have been devoted to quantify depression in specific age groups, such as adolescents (14) and the elderly (15). The utility of these scales in the medically ill is challenging because the frequent presence of somatic symptoms in physical diseases can mislead their score interpretation. If the clinician is unable to decide which existing instrument to use and how to interpret the results, the advancement of self-rating scales can represent a step backward.
Among the investigations on using self-assessment measures to evaluate depression, the BDI outnumbers the other measures in the amount of published research: there are more than 7,000 studies so far using this scale. Aaron T. Beck and colleagues developed the 21-item BDI in 1961 to aid clinicians in the assessment of psychotherapy for depression (6). The easy applicability and psychometric soundness of this scale have popularized its use in a variety of samples (16-19) and in healthcare settings worldwide (20-22). This inventory has received two major revisions: in 1978 as BDI-IA (23) and in 1996 as BDI-II (24). This later reformulation covers psychological and somatic manifestations of a two-week major depressive episode, as operationalized in the DSM-IV (25). Four items of the BDI-IA (weight loss, distorted body image, somatic preoccupation, and inability to work) were replaced with agitation, worthlessness, difficulty concentrating, and energy loss to assess the intensity of depression. The items of appetite and sleep changes were amended to evaluate the increase and decrease in depression-related vegetative behaviors (24,26-28). Different from the original version, which intended to measure negative cognitions of depression, the BDI-II does not reflect any particular theory of depression. The English version of BDI-II has been translated and validated in 17 languages so far, and it is used among countries in Europe, the Middle East, Asia, and Latin America (29-32).
Investigations on depression and its instrumentation must be considered in view of the pressure for evidence-based decisions in clinical practice and the information explosion of the literature. Recently, the BDI-II has been ever-increasingly used in the medically ill to evaluate depressive states that occur at high prevalence in healthcare settings. The authors systematically reviewed the validity of the BDI-II to quantify the severity of depression among medical patients and discuss the interpretation of its metric conventions. The performance of the BDI-II (and its short version) among patients with medical illnesses who often present somatic complaints is contrasted with its performance among non-medical patients, among whom psychological symptoms are the most prominent features.
METHODS
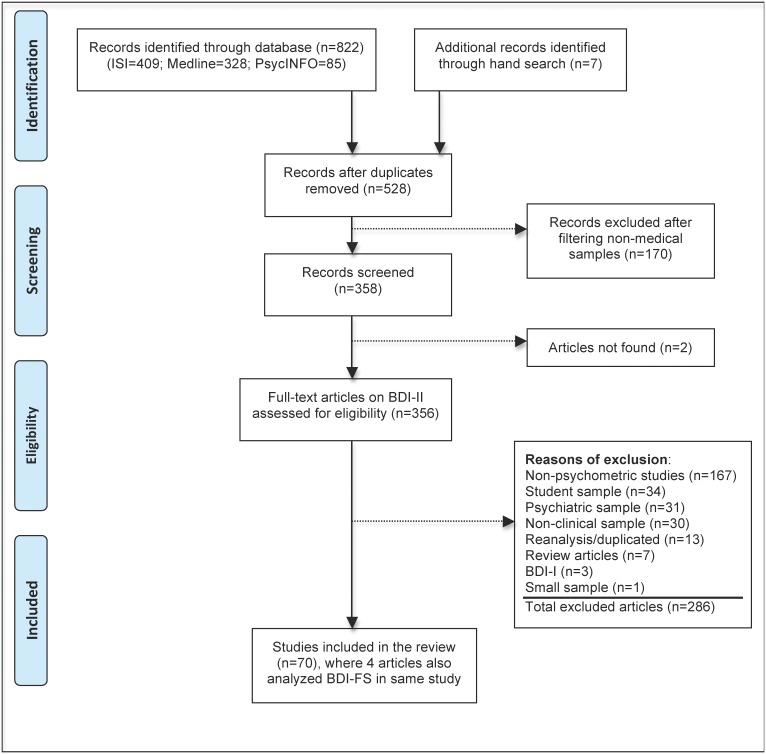
Both investigators, with previous experience on psychometric instruments, conducted this systematic review by searching the Web of Sciences (ISI), Medline, and PsycINFO databases. The following MeSH terms were used to scan studies through the search builder of each database: “valid*” OR “reliab*” OR “sensitiv*” OR “specific*” OR “concurrent” OR “divergent” OR “convergent” OR “factor analysis”. Following the search, we filtered articles containing the term “Beck Depression Inventory” published during the time period “1/1/1996 to 10/10/2012”. There was no language or age range restriction. The initial search resulted in 822 retrieved articles, with 409 from ISI, 328 from Medline, and 85 from PsycINFO. The reference sections of the review articles of the depression instruments (33-35) and book chapters (36-38) were examined to identify potential studies. Additional efforts to locate relevant studies by hand and to contact experts in the field identified seven psychometric articles on medical samples, totaling 829 articles.
After checking for duplication and overlap, 528 articles remained in the list. Filtering non-medical articles, we eliminated 170 articles in which “student,” “psychiatric,” or “community” was mentioned in the title or abstract. The retained 358 articles were screened for eligibility by reading the abstract. Two articles were not accessible, even upon request to the author, resulting in 356 full-text articles that were assessed for eligibility.
The exclusion criteria were as follows: (1) non-psychometric studies, such as clinical trials, editorials, letters, reviews, meta-analyses, practice guideline, randomized controlled trials, and case reports; (2) non-medical samples (student, psychiatric, or non-clinical); (3) small sample size (N<30); (4) BDI-I; and (5) reanalysis or duplicated analysis of an original dataset. The sample was considered “non-clinical” when study participants consisted of workers, caregivers, and community dwellers. Regardless of the nosological controversy of chronic fatigue syndrome and chronic pain as medical illnesses, these conditions were included due to their high occurrence in healthcare settings. Samples with less than 30 participants were only retained when the study addressed a very important problem, such as between-version comparison or content analysis. A summary analysis of the complete sample was preferable when multiple analyses were available (such as separate reports by gender, ethnicity, or depressed versus non-depressed groups).
The reasons for excluding 286 articles were as following: 174 studies did not contain the original data using the BDI-II (167 non-psychometric studies and seven reviews); 95 studies utilized non-medical samples (34 student samples, 31 psychiatric samples, and 30 non-clinical samples); 13 studies provided a reanalysis or secondary data analysis; three studies used BDI-I; and one study had a small sample size. The final list resulted in 70 articles that are dedicated to investigating the psychometric performance of the BDI-II in medical patients. The flowchart in Figure 1 displays each step of the search process.
Figure 1.
Flowchart of the search to scan for studies investigating psychometric properties of the Beck Depression Inventory-II among medical patients.
Studies on medical diseases were grouped according to the sample recruitment source as outpatients or primary care (k = 52) and hospital (k = 12) (Table 1). Studies investigating the short version BDI-FS (k = 10) are displayed separately. Four studies reported data on both BDI-II and BDI-FS. Several investigations did not provide a clear description of the healthcare setting or recruited participants from different levels of health service. Likewise, the heterogeneous selection of patients might reflect different groups of participants or stages of disease course. Sixteen studies reported a sample size with less than 100 respondents, but all of the studies had more than the minimum of 30 subjects.
Table 1.
Description of psychometric studies of the Beck Depression Inventory-II in medical samples by language version, sample size (N), sample description, gender distribution (%W), mean score (SD), and reliability (Cronbach's alpha).
| Authors, year | Language | N | Sample description | %W | Mean Score (SD) | Alpha |
| Normative sample | ||||||
| Beck et al., 1996 (24) | English | 120 | College students | 44 | 12.6 (9.9) | 0.93 |
| 500 | Psychiatric outpatients | 62 | 22.5 (12.8) | 0.92 | ||
| Outpatients/Primary Care (k = 52) | ||||||
| Arnarson et al., 2008 (41) | Icelandic | 248 | Adult outpatients | 82 | 21.3 (12.2) | 0.93 |
| Arnau et al., 2001 (42) | English | 333 | Adult - primary care | 69 | 8.7 (9.4) | 0.94 |
| Brown et al., 2012 (43) | English | 111 | Chronic fatigue outpatients | 83 | 17.7 (9.1) | 0.89 |
| Beck & Gable, 2001 (44) | English | 150 | Postpartum outpatients | 100 | NR | 0.91 |
| Bunevicius et al., 2012 (45) | Lithuanian | 522 | Coronary outpatients | 28 | 11.0 (8.2) | 0.85 |
| Carney et al., 2009 (46) | English | 140 | Insomnia outpatients | 74 | 14.1 (10.2) | 0.91 |
| Carvalho Bos et al., 2009 (47) | Portuguese | 331 | Pregnancy outpatients | 100 | NR | 0.88 |
| 354 | Postpartum outpatients | 100 | NR | 0.89 | ||
| Chaudron et al., 2010 (48) | English | 198 | Postpartum outpatients | 100 | NR | NR |
| Chilcot et al., 2008 (49) | English | 40 | Renal hemodialysis outpatients | 40 | 11.1-12.9 (9.3-9.4) | NR |
| Chilcot et al., 2011 (50) | English | 460 | Renal disease outpatients | 35 | 11.9 (8.3) | NR |
| Chung et al., 2010 (51) | Chinese | 62 | Heart disease outpatients | 31 | 18.2 (7.9) | NR |
| Corbière et al., 2011 (29) | French | 206 | Chronic pain outpatients | 53 | 17.2 (11.5) | 0.84 |
| Dbouk et al., 2008 (52) | English | 129 | Hepatitis C outpatients | 50 | 17.1 (11.6) | NR |
| de Souza et al., 2010 (53) | English | 50 | Huntington's disease | 48 | 8.8 (8.9) ND 26.8 (6.9) D | NR |
| del Pino Pérez et al., 2012 (54) | Spanish | 205 | Coronary outpatients | 26 | 9.2 (7.6) | NR |
| Dutton et al., 2004; Grothe et al., 2005 (55,56) | English | 220 | Adult - primary care | 52 | 12.6 (10.4) | 0.90 |
| Findler et al., 2001 (57) | English | 98 | Traumatic brain injury (mild) | 55 | 12.2 (9.6) | NR |
| 228 | Traumatic brain injury (moderate to severe) | 33 | 9.7 (8.1) | NR | ||
| Frasure-Smith & Lespérance, 2008 (58) | English/French | 804 | Coronary outpatients | 19 | NR | 0.90 |
| Griffith et al., 2005 (59) | English | 132 | Epilepsy outpatients | 72 | 15.9 (11.1) | NR |
| Hamid et al., 2004 (60) | Arabic | 493 | Women - primary care | 100 | 13.0 (8.1) | NR |
| Harris & D'Eon, 2008 (61) | English | 481 | Chronic pain outpatients | 58 | 26.9 (11.7) | 0.92 |
| Hayden et al., 2012 (62) | English | 83 | Obese bariatric outpatients | 71 | 13.4 (9.1) | 0.89 |
| Jones et al., 2005 (63) | English | 174 | Epilepsy outpatients | 66 | NR | 0.94 |
| Kanner et al., 2010 (64) | English | 193 | Epilepsy outpatients | 68 | 10.6 (6.3) | NR |
| King et al., 2012 (65) | English | 489 | Traumatic brain injury | 10 | 19.7 (11.8) | NR |
| Kiropoulos et al., 2012 (66) | English | 152 | Coronary heart disease outpatients | 34 | 9.4 (8.9) ND 17.8 (8.7) D | NR |
| Kirsch-Darrow et al., 2011 (67) | English | 161 | Parkinson outpatients | 31 | 9.5 (7.2) | 0.89 |
| Ko et al., 2012 (68) | Korean | 121 | Epilepsy outpatients | 35 | 9.7 (6.3) ND 29.9 (11.7) D | NR |
| Lipps et al., 2010 (69) | English | 191 | HIV infection outpatients | 61 | 14.1 (11.0) W 10.2 (9.1) M) | 0,89 |
| Lopez et al., 2012 (70) | English | 345 | Chronic pain outpatients | 0 | 23.0 (12.2) | 0.93 |
| Masuda et al., 2012 (71) | Japanese | 327 | Myasthenia gravis outpatients | 67 | 11.3 (7.9) | NR |
| Neitzer et al., 2012 (72) | English | 150 | Renal hemodialysis outpatients | 48 | 12.3 (10.8) | NR |
| Ooms et al., 2011 (73) | Dutch | 136 | Tinnitus outpatients | 35 | 11.3 (9.5) | NR |
| Osada et al., 2011 (74) | Japanese | 56 | Fibromyalgia outpatients | 86 | NR | NR |
| Patterson et al., 2011 (75) | English | 671 | Hepatitis C outpatients | 3 | 16.2 (12.2) | 0.84-0.91 |
| Penley et al., 2003 (30) | English/Spanish | 122 | Chronic renal outpatients | 41 | 15.0 (12.5) | 0.92 |
| Pereira et al. 2011 (76) | Portuguese | 503 | Pregnant outpatients | 100 | NR | NR |
| Poole et al., 2009 (77) | English | 1227 | Chronic pain outpatients | 62 | 24.7 (11.6) | 0.92 |
| Rampling et al., 2012 (78) | English | 266 | Epilepsy outpatients | 59 | NR | 0.94 |
| Roebuck-Spencer, 2006 (79) | English | 60 | Systemic lupus erythematosus outpatients | 80 | NR | NR |
| Su et al., 2007 (80) | Chinese | 185 | Pregnant outpatients | 100 | 7.0 (5.0) ND 17.0 (10.2) D | NR |
| Suzuki et al., 2011 (81) | Japanese | 287 | Myasthenia gravis outpatients | 67 | 11.1 (8.1) | NR |
| Tandon et al., 2012 (82) | English | 95 | Perinatal women | 100 | NR | 0.9 |
| Teng et al., 2005 (83) | Chinese | 203 | Postpartum outpatients | 100 | 7.8 (6.3) ND 25.8 (10.4) D | NR |
| Turner et al., 2012 (84) | English | 72 | Stroke outpatients | 47 | 13.4 (12.9) | 0.94 |
| Turner-Stokes et al., 2005 (85) | English | 114 | Brain injury outpatients | 43 | Median 10 (IQR 5-19) | NR |
| Viljoen et al., 2003 (86) | English | 127 | Adult - primary care | 63 | NR | NR |
| Wan Mahmud et al., 2004 (87) | Malay | 61 | Postpartum I outpatients | 100 | 4.4 (5.5) | 0.89 |
| 354 | Postpartum II outpatients | 100 | 6.2 (6.4) | |||
| Warmenhoven et al., 2012 (88) | Dutch | 46 | Cancer outpatients | 43 | 14.7 (9.9) | NR |
| Williams et al., 2012 (89) | English | 229 | Parkinson disease outpatients | 33 | 6.5 (5.2) ND 14.7 (7.4) D | 0.90 |
| Young et al., 2007 (90) | English | 194 | Cardiac outpatients | 35 | 8.6-13.4 (7.7-12.3) | NR |
| Zahodne et al., 2009 (91) | English | 71 | Parkinson disease outpatients | 32 | 11.7 (7.9) | NR |
| Hospitalized (k = 12) | ||||||
| Di Benedetto et al., 2006 (92) | English | 81 | Acute cardiac syndrome | 19 | NR | > 0.90 |
| Gorenstein et al., 2011 (93) | Portuguese | 334 | Adult - hospitalized | 48 | 12.2 (11.6) | 0.91 |
| 170 physically disabled 164 intellectually disabled | 14.5 (11.2) 9.7 (11.4) | |||||
| Homaifar et al., 2009 (94) | English | 52 | Traumatic brain injury *) | 10 | 25 (14.6) | NR |
| Huffman et al., 2010 (95) | English | 131 | Myocardial infarction | 20 | 9.8 (9.4) | NR |
| Jamroz-Wisniewska et al., 2007 (96) | Polish | 104 | Multiple sclerosis | 74 | 14.4 (9.2) | NR |
| Low & Hubley, 2007 (97) | English | 119 | Coronary disease | 25 | 8.0 (7.1) | 0.89 |
| Pietsch et al., 2012 (40) | German | 314 | Adolescents patients*) (252 hospital inpatients) | 60 | 7.5 (6.5) ND 25.8 (10.1) D | 0.91 |
| Rowland et al., 2005 (98) | English | 51 | Traumatic brain injury | 28 | 5.6 ND 20.1 D | NR |
| Siegert et al., 2009 (99) | English | 353 | Neurological diseases | 40 | 13.6 (10.1) | 0.89 |
| Thomas et al., 2008 (100) | English | 50 | Stroke | 38 | 12.7 (8.9) | NR |
| Thombs et al., 2008 (101) | English/French | 477 | Acute myocardial infarction | 17 | 9.2 (7.9) | NR |
| Tully et al., 2011 (102) | English | 226 | Cardiac heart disease | 17 | 8.6 (6.2) a 9.1 (6.4) b | 0.850.87 |
| BDI Fast Screen version (k = 10) | ||||||
| Beck et al., 1997 (26) | English | 50 | Medical inpatients | 60 | 5.8 (4.5) | 0.86 |
| Brown et al., 2012 (43)†) | English | 111 | Chronic fatigue outpatients | 83 | 4.3 (3.2) | NR |
| Neitzer et al., 2012 (72)†) | English | 146 | Renal hemodialysis outpatients | 48 | 2.7 (3.4) | NR |
| Pietsch et al., 2012 (40)†) | German | 314 | Adolescents*) (252 hospital inpatients) | 60 | 1.9 (2.4) ND 8.1 (3.5) D | 0.82 |
| Poole et al., 2009 (103)†) | English | 1227 | Chronic pain outpatients | 62 | 7.1 (4.30) | 0.84 |
| Scheinthal et al., 2001 (104) | English | 75 | Geriatric outpatients | 56 | 2.3 (3.1) | 0.83 |
| Servaes et al., 2000 (105) | Dutch | 85 16 | Disease-free cancer outpatients Chronic fatigue outpatients | 43.5 50 | 0.4-2.3 (0.9-1.8) 2.6 (1.8) | NR |
| Servaes et al., 2002 (106) | Dutch | 57 57 | Disease-free breast cancer outpatients Chronic fatigue outpatients | 100 100 | 2.3-4.2 (2.2-3.9) 3.3 (2.6) | NR |
| Steer et al., 1999 (107) | English | 120 | Medical outpatients | 50 | 2.2 (3.0) | 0.85 |
| Winter et al., 1999 (39) | English | 100 | Adolescent outpatients | 50 | 1.9 (3.1) | 0.88 |
N: sample size;%W: percentage of women; SD: standard deviation; Alpha: Cronbach's alpha coefficient of internal consistency;
NR: not reported.
: men, W: women; ND: non-depressed; D: depressed; a: pre-surgery; b: post-surgery.
Mixed sample of in- and outpatients.
Separate analysis of the short version of the BDI-II in the same study.
IQR: interquartile range.
Among the 70 retained studies, the BDI-II was administered to adults in primary care (k = 4) and clinics of cardiology (k = 12), neurology (k = 12), obstetrics (k = 8), brain injury (k = 6), nephrology (k = 5), chronic pain (k = 4), chronic fatigue (k = 4), oncology (k = 3), and infectious disease (k = 3). Only two studies assessed adolescent medical patients (39,40).
Almost all of the identified studies were published after 2000, and the great majority (approximately 64%) of studies was published in the past five years, suggesting a recent trend for using the BDI-II in medical settings. Nearly 70% of the articles applied the English version of BDI-II, but 13 non-English versions of the scale were found.
Overview
The BDI-II performed well in adult patients with a wide array of medical diseases (Table 1). For the purpose of comparison, data from Beck's studies on non-medical and medical samples (24,26) are listed as normative references. Usually, non-patient samples reported the item scores in the lower part of the range of possible scores (from 0 to 3), with a skewed distribution of item scores. Based on scores of 500 psychiatric outpatients, Beck et al. (24) suggested the following ranges of BDI-II cut-off scores for depression: 0–13 (minimal), 14–19 (mild), 20–28 (moderate), and 29–63 (severe). As an example, the mean score of the BDI-II in samples with mood disorder was M = 26.6, and the mean scores for major depressive episode, recurrent depression, and dysthymia were 28.1, 29.4, and 24.0, respectively.
Confirming the expectation that medical patients would report more somatic symptoms, most of the investigations reported a slightly higher mean total score for medical patients than non-patients (Table 1), but scores were still around or below the threshold of 13/14 that is recommended by Beck to detect mild depression. Exceptions of this observation were studies on chronic pain (29,61,70,77), with mean total scores ranging from 17.2 to 26.9. The type of respondents might influence item endorsement and the scale total score.
In comparison with the previous version, the item characteristics of the BDI-II have been changed in terms of endorsement rate, homogeneity, and content coverage (34). The homogeneity of the scale was described for 17 of 21 items in the original study (24), showing acceptable item-total correlations of rit ≥0.5 (108). Different item endorsements and coverage are reported for different versions of the instrument: substantial item-total correlation was described for 15 items in the Brazilian-Portuguese version (93) and 10 items in the Arabic version (32). Direct comparison of the scores between different language versions should be avoided.
In contrast with patient samples, somatic items, such as “change in sleeping pattern” and “change in appetite,” presented low scores for non-clinical samples. However, “tiredness or fatigue,” might present special clinical significance in patients with chronic fatigue syndrome (43) or cardiac coronary disease (45,51). Regardless of the severity of depression, the item “loss of sexual interest” displayed the worst item-total correlation, although it was significantly related to the whole construct under consideration (23,24). Thombs et al. (101) suggested that the assessment of symptom severity with BDI–II would be substantially biased in medically ill patients compared with non-medically ill patients due to the misattribution of somatic symptoms from medical conditions to depression. The authors found that post-acute myocardial infarction patients did not have higher somatic symptom scores than psychiatry outpatients who were matched on cognitive/affective scores. Compared with undergraduate students, somatic symptom scores in cardiac patients were only approximately one point higher, indicating that somatic symptom variance is not necessarily related to depression in medically ill and non-medically ill respondents.
The item “suicidal thoughts” was the least reported item among non-medical settings; however, a substantial correlation still demonstrates its contribution to depression (23,24). Investigations on the ability of separate items, e.g., “pessimism” and “loss of energy,” to predict disease outcome or treatment response can help clinicians in the management of depression. The contribution of self-rated somatic vs. cognitive symptoms in medical samples should be clarified by item analysis to identify whether items are appropriately assigned to a scale.
BDI-Fast Screen
Experts view somatic symptoms among medical patient as the harbinger of depression and anxiety in the healthcare setting (3,109-111). Preferably, the assessment of depression in patients with medical illness should avoid confounding physical symptoms. The correct identification of comorbid depressive disorders in medical patients is crucial in understanding its origin and in controlling the physical symptom burden.
Two measures were designed with the objective of eliminating somatic items. The first proposed measure is the Hospital Anxiety Depression Scale (HADS) (112), which has a seven-item depression subscale. Despite the lack of comprehensive data on its psychometric properties (113) and challenges to its factorial validity (114), the HADS remained widely used as a research measure of depression in the medically ill.
The seven-item BDI for Primary Care (BDI-PC) (26) was developed in 1997 after removing somatic items, such as fatigue and sleep problems, from the BDI. This version was projected for evaluating depression in patients whose behavioral and somatic symptoms are attributable to biological, medical, alcohol, and/or substance abuse problems that may confound the diagnosis of depression. The BDI-PC was later renamed the BDI ® Fast Screen for Medical Patients (BDI-FS), and it consists of items 1 to 4 and 7 to 9 of the BDI-II (27).
The BDI-FS requires less than five minutes for completion, and scoring is similar to the BDI-II. For interpretation, the manual suggests that scores 0–3 indicate minimal depression; 4–6 indicate mild depression; 7–9 indicate moderate depression; and 10–21 indicate severe depression (27). Validation studies (k = 10) have demonstrated the ability of this non-somatic scale to discriminate depressed vs. non-depressed medical patients (39,26,104,107), chronic pain patients (103), and conditions where fatigue is a prominent feature (43,105,106). Less popular than its full version, more investigations are needed to establish the utility of this short version in medical settings before recommending its extensive use.
Reliability
Thirty-seven of 70 retrieved psychometric articles (52.9%) did not report reliability coefficients for the data. In comparison to the internal consistency of previous versions of the BDI (average Cronbach's alpha coefficient of approximately 0.85) (23), the reliability of the BDI-II among medical samples was satisfactory, with an alpha of approximately 0.9, ranging between 0.84 and 0.94 (Table 1). In addition, Beck (26) reported a coefficient of 0.86 for the BDI-FS, and further studies reported the coefficient ranging from 0.82-0.88 (39,40).
No information on the retest reliability is available for medical samples. However, the stability of the BDI-II, as expressed by retest coefficients of Pearson's r of 0.92 and 0.93, was reported by Beck and colleagues (24) for psychiatric and student samples, respectively. Further evidence of acceptable stability through re-application of the BDI-II was demonstrated for student samples (range: 0.73-0.96) (115,116).
The retest effect – that is, lower scores on the second application, even without intervention – may affect the reliability of BDI-II in healthcare settings. This effect could be unrelated to a true change in severity and could be purely the result of the measurement process. Although this fact would not preclude using this scale in follow-up or interventional studies among medical patients, nothing should be stated concerning the scale performance in this respect. Therefore, clinicians should be careful when making important treatment decisions based on non-empirical information assumed from non-clinical samples.
Item Response Theory
Most validation studies of BDI-II were analyzed in accordance with classic test theory, assuming a true score for each respondent's summed score and disregarding the measurement error. In other words, two individuals with the same total score may differ greatly in terms of relative severity and frequency of symptoms. This discrepancy might be particularly taxing in medical settings, where physical symptoms are common complaints and overlap with “true” depression-related somatic symptoms.
In the last decades, the item response theory (IRT) is an increasingly used method in psychometrics, in addition to the dominant classic test theory of true score paradigm. Briefly, the IRT distinguishes between moderate and severe cases of depression using item-level analysis to account for measurement error (117). The response of a respondent for a given ability should be modeled to each item in the test. For example, when a given depression scale is composed only of items that measure mild depression, this instrument would have great difficulty identifying severe depression because both levels of severity should be characterized by high scores on all items. In addition, if items assessing psychological and physical symptoms were only loosely related, a single score would not distinguish between two potentially different groups of depressed patients - with primarily psychological or with primarily vegetative symptoms. This scenario is particularly pressing in medical settings that are investigating clinical changes in depressive syndrome.
Seigert and colleagues (99) reported an illuminating study after examining each BDI-II item for differential item functioning in a neurological sample (n = 315). The authors identified misfits to model expectations for three items that seemed to measure different dimensions: changes in sleeping pattern, changes in appetite, and loss of interest in sex. These vegetative items were removed and re-scored in an iterative fashion to the scale. In the real world, the likelihood of receiving a rating of 1 on the insomnia item was essentially the same, regardless of the overall severity of depression, but the likelihood of receiving a rating of 3 on sad mood could be low, even when overall depression was severe.
Waller and colleagues (118) investigated the latent structure of the BDI-II through differential item functioning and item level factor analysis in samples of women with breast cancer and women with clinical depression. Items of negative cognitions about the self, e.g., worthlessness, self-dislike, and punishment feelings, were less likely to be reported by breast cancer patients than depressed patients. Negative cognitions about the self appear to be related to different factors in breast cancer. The analyses also found many differences at both the item and factor scale levels, suggesting caution when interpreting the BDI-II in breast cancer patients.
These studies advocate that the rating scheme is not ideal for many BDI-II items, thus affecting the scale's capacity to detect change in medical conditions. Systematic IRT analysis of the BDI-II items can strengthen the scale coverage in assessing heterogeneous depressive conditions among medical patients.
Convergent and Divergent Validity
Table 2 displays the studies that compared the BDI-II with scales measuring depression, anxiety, and miscellaneous constructs as criteria that were determined at essentially the same time to check for concurrent validity. The convergent validity between the BDI-II and the BDI-I was 0.93 (28). The shorter version, BDI-FS, also presented an acceptable correlation of 0.85 (72). In general, the overlap of the construct measured by BDI-II with other widely used scales to assess depression, e.g., the Center for Epidemiologic Studies of Depression, the Hamilton Depression Rating Scale, Edinburg Postnatal Depression Scale, and the Hospital Anxiety and Depression Scale-Depression, was adequate and ranged from 0.62 to 0.81 (Table 2).
Table 2.
Concurrent validity of the Beck Depression Inventory-II with measures of depression, anxiety, and other miscellaneous constructs in medical samples.*)
| Concurrent instrument | r | Study | |
| Depression measure | |||
| BDI-I | Beck Depression Inventory – I | 0.93 | 28 |
| BDI-FS | Beck Depression Inventory – Fast Screen | 0.85 | 72 |
| HADS-D | Hospital Anxiety and Depression Scale-Depression | 0.62 - 0.71 | 26†), 41 |
| CES-D | Centre for Epidemiologic Studies of Depression | 0.72 - 0.87 | 29, 41, 52, 63, 69 |
| HRSD | Hamilton Rating Scale for Depression - revised | 0.71 - 0.75 | 24, 87 |
| EPDS | Edinburgh Postnatal Depression Scale | 0.72 - 0.82 | 44, 83, 87 |
| GDS | Geriatric Depression Scale | 0.81 | 104 †) |
| PHQ | PRIME-MD Patient Health Questionnaire | 0.84 | 52 |
| CDS | Cardiac Depression Scale | 0.65; 0.69 | 66, 92 |
| POMS-D | Profile of Mood States Depression Scale | 0.77 | 59 |
| PDSS | Postpartum Depression Screening Scale | 0.68; 0.81 | 44, 76 |
| DISC | Depression Intensity Scale Circles | 0.66 | 85 |
| NGRS | Numbered Graphic Rating Scale | 0.65 | 85 |
| Anxiety measure | |||
| BAI | Beck Anxiety Inventory | 0.60 | 24, 41 |
| HARS | Hamilton Anxiety Rating Scale - revised | 0.47 | 24 |
| STAI | State-Trait Anxiety Inventory | 0.64; 0.83 | 66, 92 |
| PSWQ | Penn State Worry Questionnaire | 0.61 | 41 |
| HADS-A | Hospital Anxiety and Depression Scale-Anxiety | 0.65 | 41 |
| Miscellaneous | |||
| SSI | Scale for Suicide Ideation | 0.37 | 24 |
| BHS | Beck Hopelessness Scale | 0.68 | 24 |
| MPQ-PRI | McGill Pain Questionnaire (Pain Rating Index) | 0.32 | 61 |
| SF-36 MH | Short Form 36-Item Health Survey – Mental Health | 0.45 - 0.70 | 43†), 57 |
| SF-36 PH | Short Form 36-Item Health Survey – Physical Health | 0.12 - 0.29 | 43†), 57 |
| SPS | Social Provisions Scale | 0.39 - 0.42 | 69 |
| CIS-F | Checklist Individual Strength - Fatigue | 0.58 | 105 |
| NDDI-E | Neurologic Disorders Depressive Inventory in Epilepsy | 0.81 - 0.85 | 64, 68 |
| NSI | Neurobehavioral Symptom Inventory | 0.77 | 65 |
| MG-QOL | Myasthenia Gravis Quality of Life Scale | 0.52 | 71 |
| JFIQ | Fibromyalgia Impact Questionnaire | 0.58 | 74 |
| ANAM | Automated Neuropsychological Assessment Metrics-Mood | 0.67 | 79 |
| SCQR | Stroke Cognitions Questionnaire Revised | 0.54 - 0.80 | 100 |
| STOP-D | Screening Tool for Psychological Distress | 0.83 | 90 |
| LARS | Lille Apathy Rating Scale | 0.45 | 91 |
| AS | Apathy Scale | 0.58 | 91 |
| UPDRS-III | Unified Parkinson's Disease Rating Scale | 0.38 | 91 |
r: Pearson's product moment correlation. Negative correlation is omitted in the numerical value.
The concurrent validity refers to the BDI-FS version.
A complete list of retrieved studies can be obtained from the authors upon request.
Additionally, the convergent validity between the BDI-II and scales that assess anxiety was significant and differed across comparison instruments: Beck Anxiety Inventory (0.60) (24,41), Hamilton's Anxiety Rating Scale (0.47) (24), State-Trait Anxiety Inventory (0.83) (92), Penn State Worry Questionnaire (0.61) (41), and Hospital Anxiety and Depression Scale-Anxiety (0.65) (41). These results were expected due to the extent that anxiety symptoms were highly comorbid with depressive symptoms or that they could be attributed to the characteristics of the compared instruments. As a broad indicator of mental health, a high score on the BDI scale could also be explained by other disorders, physical illnesses, or social problems (69). Most likely, the construct covered by the BDI-II is beyond the “pure” depressive-type of psychopathology. As such, the convergent validity of the scale with hopelessness (24) and fatigue (105) was also substantial. In the medical setting, the clinician should not assume depression as a primary issue when BDI-II is used without a thorough clinical assessment.
Concerning divergent validity, studies have indicated poor correlation (r<0.4) with instruments assessing chronic pain (61), physical health (43), and substance use disorders (119). Suicidal ideation, which is one of core features of depression and an item on the BDI-II, was only poorly correlated with the instrument (24).
Criterion-oriented Validity
Psychometric experts view the interpretation of the raw scores on tests, such as the BDI-II, as problematic, unless they are converted into standardized scores (e.g., T score or stanine method) (108,120). No known standardized norms have been reported for the BDI-II to date. As an alternative to the norm-referenced method, the criterion-referenced method is the most widespread practice for interpreting BDI-II scores. Usually, the total score is compared with a cut-off score established according to a gold-standard criterion (e.g., clinical assessment or structured interview).
When clinicians intend to screen probable cases of major depression in medical settings, the sensitivity should be viewed as the most important indicator to minimize the chance of false-negative cases (Table 3). Sometimes, the BDI-II can overestimate the prevalence of depression in particular conditions, e.g., medically ill patients would record more items that address physical complaints. According to the samples, medical studies have reported good performance with high sensitivity (from 72% to 100%). Occasionally, the researcher might want to improve the specificity to select a pure sample of depressed patients. For research purposes, Beck et al. (24) recommended raising the cut-off score to 17 to obtain homogeneous samples of depressed individuals.
Table 3.
Criterion validity and cut-off point of the Beck Depression Inventory-II for detecting major depressive episode in medical samples.
| Authors | Sample | Cut-off | Sensitivity | Specificity | PPV | NPV | AUC | % MDD | Criterion | ||
| Outpatients | |||||||||||
| Arnarson et al. (41) | Adult outpatients | 20 | 82 | 75 | NR | NR | 87 | 42.1 | MINI | ||
| Arnau et al. (42) | Adult - primary care | 18 | 94 | 92 | 54 | 99 | 96 | 23.2 | PHQ | ||
| Beck & Gable 2001 (44) | Postpartum outpatients | 20 | 56 | 100 | 100 | 93 | 95 | 12 | SCID-I | ||
| Bunevicius et al. (45) | Coronary outpatients | 14 | 89 | 74 | 29 | 98 | 90 | 11 | MINI | ||
| Carney et al. (46) | Insomnia outpatients | 17 | 81 | 79 | NR | NR | 83.8 | NR | SCID-I | ||
| Chaudron et al. (48) | Postpartum outpatients | 20 | 45.3 | 91.1 | NR | NR | 90 | 37 | SCID-I | ||
| Chilcot et al. (49) | Renal hemodialysis | 16 | 89 | 87 | 89 | 87 | 96 | 22.5 | MINI | ||
| de Souza et al. (53) | Huntington's disease | 11 | 100 | 66 | 48 | 100 | 85 | 50 | SCAN | ||
| Dutton et al. (55) | Adult - primary care | 14 | 87.7 | 83.9 | 69.5 | 94.2 | 91 | 29.5 | PRIME-MD | ||
| Frasure-Smith & Lespérance (58) | Coronary outpatients | 14 | 91.2 | 77.5 | NR | NR | 92 | 13.7 | SCID-I | ||
| Jones et al. (63) | Epilepsy outpatients | 11 | 96 | 80 | 48 | 99 | 94 | 17.2 | MINI | ||
| 15 | 84 | 87 | 55 | 97 | 92 | SCID-I | |||||
| 11 | 95.7 | 78.3 | 42 | 99 | 94 | MINI + SCID | |||||
| Hayden et al. (62) | Obese bariatric outpatients | 13 | 100 | 63.9 | 29.7 | 100 | 84.7 | 13.3 | SCID-I | ||
| Pereira et al. (76) | Pregnant outpatients | 16 | 83.3 | 93.1 | 14.3 | 99.7 | 95 | 1.3 | DIGS | ||
| Rampling et al. (78) | Epilepsy outpatients | 14 | 93.6 | 74 | 44 | 98 | 90 | 17.7 | MDI (ICD-10) | ||
| 15 | 93.8 | 78.9 | 49.5 | 98 | 93 | 18 | MDI (DSM-IV) | ||||
| Su et al. (80) | Pregnant outpatients | 12 | 72.7-75.0 | 82.7-82.9 | NR | NR | 81.9-86.6 | 12.4 | MINI | ||
| Tandon et al. (82) | Perinatal women | 12 | 84.4 | 81.0 | NR | NR | 91 | 33.7 | SCID-I | ||
| Teng et al. (83) | Postpartum outpatients | 14 | 92 | 83 | 42 | 99 | NR | 11.8 | MINI | ||
| 12 | 96 | 79 | |||||||||
| Turner et al. (84) | Stroke outpatients | 11 | 92 | 71 | NR | NR | 89 | 18 | SCID-I | ||
| Turner-Stokes et al. (85) | Brain injury outpatients | 14 | 74 | 80 | 69 | 84 | NR | 39.8 | DSM-IV | ||
| Wan Mahmud et al. (87) | Postpartum outpatients | 9 | 100 | 98 | 87.5 | 100 | 99.5 | 48 | CIS | ||
| Warmenhoven et al. (88) | Cancer outpatients | 16 | 90 | 69 | NR | NR | 82 | 22 | PRIME-MD | ||
| Williams et al. (89) | Parkinson outpatients | 7 | 95 | 60 | 62 | 94 | 85 | 34.1 | SCID-I | ||
| Hospital sample | |||||||||||
| Homaifar et al. (94) | Traumatic brain injury | 19 | 87 | 79 | NR | NR | NR | 44.2 | SCID-I | ||
| Huffman et al. (95) | Myocardial infarction | 16 | 88.2 | 92.1 | 62.5 | 98.1 | 96 | 13 | SCID-I | ||
| Low & Hubley (97) | Coronary disease | 10 | 100 | 75 | 21 | 100 | 92 | 11.8 | SCID-I | ||
| Pietsch et al. (40) | Adolescents | 19 | 86 | 93 | 47 | 99 | 93 | 6.7 | Kinder-DIPS | ||
| BDI-FS | |||||||||||
| Beck et al. (26) | Medical inpatients | 4 | 82 | 82 | NR | NR | 92 | 66 | PRIME-MD | ||
| Neitzer et al. (72) | Renal hemodialysis | 4 | 97.2 | 91.8 | 81.4 | 98.9 | 98 | 28.7 | BDI-II ≥ 16 | ||
| Pietsch et al. (40) | Adolescents | 6 | 81 | 90 | 37 | 99 | 92 | 6.7 | Kinder-DIPS | ||
| Poole et al. (103) | Chronic pain outpatients | 4 | 81 | 92 | NR | NR | 94 | 59.4 | BDI-II ≥ 19 | ||
| 5 | 75 | 93 | NR | NR | 94 | 47.8 | BDI-II ≥ 22 | ||||
| Scheinthal et al. (104) | Geriatric outpatients | 4 | 100 | 84 | NR | NR | 93 | 11 | Clinical assessment | ||
| Steer et al. (107) | Medical outpatients | 4 | 97 | 99 | NR | NR | 99 | 24.2 | PRIME-MD | ||
| Winters et al. (39) | Adolescent outpatients | 4 | 91 | 91 | NR | NR | 98 | 11 | PRIME-MD | ||
PPV: positive predictive value; NPV: negative predictive value; AUC: area under the curve;%MDD: proportion of major depression disorder; NR: not reported.
PHQ: PRIME-MD Patient Health Questionnaire; MINI: Mini International Neuropsychiatric Interview; PRIME-MD: Primary Care Evaluation of Mental Disorders; CIS: Clinical Interview Schedule; SCID-I: Structured Clinical Interview for DSM-IV Axis I Diagnosis; MDI: Major Depression Inventory; Kinder-DIPS: Diagnostisches Interview bei psychischen Störungen im Kindes und Jugendalter; DIGS: Diagnostic Interview for Genetic Studies; SCAN: Schedules for Clinical Assessment in Neuropsychiatry.
According to Table 3, the best cut-off to indicate cases of depressive syndrome in medical samples was established on the ground of the unique characteristics of the sample. The possible threshold ranged widely, from 7 to 22 (89,103). For example, Poole et al. (103) found that raising the BDI-II cut-off score to 22 could reduce the number of false-positives produced by the uneven item response of chronic pain patients. Consequently, the researcher can change the flexibility of the cut-off score by comparing different thresholds for a new sample or study purpose.
A significant diagnostic accuracy of 82% and higher, as expressed by the area under the receiver operating characteristics (ROC) curve, was calculated according to the tradeoff between sensitivity and specificity. However, the ability of a scale to differentiate between depressive vs. non-depressive groups depends not only on the sensitivity and specificity of its cut-off scores but also on the frequency of the disorder in the samples that are being studied. In addition, sources of threshold variation may depend on the type of the sample (outpatient or hospitalized), medical disease, and external gold-standard criterion for depression. Most investigators were unanimous in recommending the BDI-II as a screening tool in the first phase of two-stage studies to prevent excessive cases of false positives if the scale is used as a single tool (121). Caution is warranted when using the cut-off guidelines presented for criterion-referenced interpretation and when the BDI-II is misused as a diagnostic instrument.
The BDI-FS was projected to reduce the number of false-positives for depression in patients with medical problems. Similar to its full version, the BDI-FS has shown excellent performance to detect probable cases of depression with a cut-off of 4, as expressed by a large area under the ROC curve (Table 3). To reduce the number of false-positives in chronic pain patients, Poole et al. (103) suggested raising the cut-off value to 5. To detect depression in German adolescent medical patients, Pietsch et al. (40) recommended a threshold of 6. In comparison to the 21-item version, this non-somatic version of BDI has been less extensively investigated, which prevents a more conclusive recommendation for systematic use in medical conditions.
Using rating scales to identify patients for detailed assessment has been advocated to improve the search for depression through screening programs, but the detection rates, treatments, and outcomes are controversial. There is no agreement on the score interpretation of rating scales as screening tools, e.g., the Hamilton Rating Scale for Depression is viewed as a non-trustworthy judgment of the severity of a patient's depression (122,123). In addition, the four-option formulation of the BDI items is viewed as being more complicated than the yes-no alternative of a screening questionnaire, such as the Geriatric Depression Scale (15). Although existing literature supports the use of the BDI-II as a screening measure of depression, in-depth analysis of moderator factors that influence the performance of this scale should be conducted.
Content and Construct Validity
The acceptance of the content as a qualitative representation of the measured trait is critical for the content validity of a given scale (124). The BDI-I reflected six of the nine criteria for DSM-based depression (21,125), while the BDI-II encompassed all DSM-based depressive symptoms. As a consequence, the tests' ability to detect a broader concept of depression has been changed (28,126). The content covered by the BDI-II seems adequate but narrower than its former version (34).
Construct validation interprets a test measure through a specific attribute or quality that is not “operationally defined,” demonstrated as a latent structure or construct (127). Exploratory and confirmatory factor analyses determine which psychological events make up a test construct by reducing the item number to explain the structure of data covariance. This family of multivariate techniques demonstrates the dimensionality of a given scale and the pattern of item clustering on one, or more than one, factor (128). A robust measurement instrument for depression should establish the dimensions being measured and the types, categories, and behaviors that constitute an adequate representation of depression.
Table 4 lists 20 investigations that reported the factor structure of the BDI-II, which was used in 43% of the retained studies. These articles were grouped according to the healthcare setting and the factor extraction framework. Researchers have adopted both exploratory and confirmatory strategies with different purposes, e.g., to identify problems with items that have non-significant factor loadings or data cross-validation. The use of the state-of-art confirmatory approach is a trend in studies investigating the latent structure of BDI-II.
Table 4.
Construct validity of the latent structure of the Beck Depression Inventory-II in medical samples.
| Study | Sample | Method | Factor 1 | Factor 2 | Factor 3 | Factor 4 |
| Normative study | ||||||
| Beck et al. (24) | College students | EFA | Cognitive-affective | Somatic-vegetative | ||
| Psychiatric outpatients | EFA | Cognitive-affective | Somatic-vegetative | |||
| Outpatient/Primary Care | ||||||
| Arnau et al. (42) | Adult - primary care | PCA | Somatic-affective | Cognitive | (Depression) | |
| Brown et al. (43) | Chronic fatigue outpatients | EFA | Cognitive | Somatic-affective | ||
| Carvalho Bos et al. (47) | Pregnancy outpatients | PCA | Cognitive-affective | Anxiety | Fatigue | |
| Postpartum outpatients | PCA | Cognitive-affective | Somatic-anxiety | Guilt | ||
| Chilcot et al. (50) | Renal disease outpatients | EFA | Cognitive | Somatic | ||
| CFA | Cognitive | Somatic | General depression (G) | |||
| Corbière et al. (29) | Chronic pain outpatients | CFA | Cognitive | Affective | Somatic | |
| del Pino Pérez et al. (54) | Coronary outpatients | EFA | Somatic-affective | Cognitive | ||
| CFA | Somatic-affective | Cognitive | (Depression) | |||
| Grothe et al. (56) | Adult - primary care | CFA | Cognitive | Somatic | (Depression) | |
| Harris & D'Eon (61) | Chronic pain outpatients | CFA | Negative attitude | Performance difficulty | Somatic | (Depression) |
| Kirsch-Darrow et al. (67) | Parkinson outpatients | CFA | Dysphoric mood | Loss of interest/pleasure | Somatic | |
| Lipps et al. (69) | HIV infection outpatients | C-PCA | Cognitive | Affective | Somatic | |
| Lopez et al. (70) | Chronic pain outpatients | EFA | Negative rumination | Somatic Complaint | Mood | |
| Patterson et al. (75) | Hepatitis C outpatients | EFA | Cognitive-affective | Somatic | ||
| CFA | Cognitive-affective | Somatic | ||||
| Penley et al. (30) | Chronic renal outpatients | CFA | Cognitive | Somatic-affective | ||
| Poole et al. (77)*) | Chronic pain outpatients | EFA | Negative thoughts | Behavior and activities | ||
| CFA | Negative thoughts | Behavior and activities | ||||
| Viljoen et al. (86) | Adult - primary care | EFA | Somatic-affective | Cognitive | (Depression) | |
| Wan Mahmud et al. (87) | Postpartum outpatients | PCA | Affective | Somatic | Cognitive | |
| Hospital sample | ||||||
| Gorestein et al. (93) | Adult - hospitalized | EFA | Cognitive-affective | Somatic | ||
| Rowland et al. (98) | Traumatic brain injury | PCA | Negative self-evaluation | Symptoms of depression | Vegetative symptoms | |
| Siegert et al. (99) | Neurological disease | PCA | Cognitive-affective | Somatic | ||
| CFA | Cognitive-affective | Somatic | ||||
| Thombs et al. (101) | Acute myocardial infarction | CFA | Cognitive | Somatic | General depression (G) | |
| Tully et al. (102) | Cardiac heart disease | CFA | Cognitive | Affective | Somatic |
EFA: exploratory factor analysis; PCA: principal component analysis;
C-PCA: confirmatory principal component analysis; CFA: confirmatory factor analysis.
(G) General factor of depression for the bifactor model.
(Depression) Higher order depression dimension for the hierarchical model.
Only 18 items were used in the factorial model.
Using an exploratory strategy, Beck and colleagues reported a two-factor oblique structure for student and psychiatric samples (24), the cognitive-affective and somatic-vegetative dimensions. Although this bidimensional structure could be replicated among medical patients (30,42,43,50,54,56,75,77,86), several investigators reported different solutions (29,47,61,67,69,70,87). Somatic symptoms of depression have clustered as a dominant dimension, e.g., in primary care (42,86) and in coronary patients (54), or as an independent third dimension (29,61,67,69).
These alternative solutions could not be replicated by confirmatory strategy, but the somatic factor was observed as an ever-present factor among medical patients (Table 4). Summarizing the factor structure of the existing BDI investigations through meta-analysis (35), much of the data variability can be explained by the common dimension of "severity of depression" and by the other part, “somatic symptoms.” Due to the misattribution of somatic symptoms from medical conditions to depression, the assessment of depressive symptom severity with the BDI-II can be substantially biased in medically ill patients compared with non-medically ill patients. Among factor analytical investigations, the somatic dimension has emerged as being highly correlated with the cognitive dimension (>0.50, range 0.49-0.87).
The heterogeneous characteristics of depressive conditions could partially explain these proposed factor structures in medical patients. The alternative structural analysis of the BDI-II was strengthened by two model breakthroughs: the hierarchical model and the bifactor model. The hierarchical structure of higher-order depression to explain the variance of the lower-order cognitive and somatic dimensions was tested in several medical samples (42,54,56,61). Although scant, the bifactor model identified a scale solution with a general depression, in addition to the traditional bidimensional structure (50,101). The data variance of the BDI-II supported a higher order, or a parallel construct, of “general depression” and suggested caution when interpreting subscale scores.
DISCUSSION
The present systematic review is intended to aid practicing professionals and clinical researchers in several specialties in assessing depression in their patients and in interpreting the score through the BDI-II. Ideally, deciding which depression scale is optimal for use in medical settings should meet some desirable features from the patient's and the clinician's perspectives. Patients should find the measure user-friendly and the instructions easy to follow. The questions should be understandable and applicable to the patient's problem. The scale should be brief to allow routine administration at intake and follow-up visits. From the clinician's perspective, the instrument should provide clinically convenient information to increase the efficiency of medical evaluation. Clinicians should find the instrument user-friendly and easy to administer and score with minimal training. To be trustworthy, the information provided by any measure for depression should rely on sound psychometric characteristics and demonstrate good reliability, validity, and sensitivity to change.
The BDI-II is a brief scale that is acceptable to patients and clinicians, covers all DSM-IV diagnostic criteria for major depressive disorder, and stands as a reliable indicator of symptom severity and suicidal thoughts. Its validity and case-finding capability as a screening instrument is well established. Conversely, its use as an indicator of sensitivity to change, medical patient's remission status, psychosocial functioning, and quality of life deserve further investigation. The BDI-II is copyrighted and must be purchased from the publisher, which obstructs its wider use. Because direct comparisons demonstrating that the BDI-II is more reliable or valid than other depression scales are lacking, it is unwise to justify the cost of its systematic adoption.
Systematic reviews are susceptible to publication bias, that is the likelihood of over-representation of positive studies in contrast with non-significant results that frequently remain unpublished. In psychometric analyses due to its descriptive nature this kind of bias is minimized. Despite its reasonable psychometric characteristics, the BDI-II has some limitations. The spectrum bias refers to the differential performance of a test between different settings, thus affecting the generalizability of the results. For example, the somatic factor is a primary dimension among medical patients (42,54,86) instead of depressive cognition in non-clinical individuals. In addition, the work-up or verification bias occurs when respondents with positive (or negative) diagnostic procedure results are preferentially referred to receive verification by the gold-standard procedure, allowing considerable distortion in the accuracy of a given test. For example, medical patients with multiple somatic complaints might be routinely referred to psychiatric assessment and, thus, would be more likely labeled as depressed. To the extent that these types of bias may occur, the cut-off scores need to be checked psychometrically to convey the sample characteristics. Techniques assessing the item-level (e.g., item-total correlation and IRT analysis) and the scale-level (e.g., signal detection analysis and factor analysis) can improve the feasibility and strengthen the validity of using this scale to detect depressive symptoms in medical settings.
In the healthcare context, the perceived burden of scale completion by the clinician is the major obstacle to using standardized scales, such as the Hamilton Depression Rating Scale, which is unlikely to meet with success. As a self-report questionnaire to measure depression, the BDI-II holds the advantages of releasing the overburdened clinician from the paperwork of scale administration and of improving the efficiency of the clinical encounter by providing mental status assessment that correlates well with clinician-rated tools.
The stated purpose of the BDI-II is not to diagnose major depressive episode; thus, the investigators must grasp its appropriateness for detecting depressive symptoms and monitoring treatment efficacy and its comparability with observer-rated scales, such as the Hamilton Depression Rating Scale of Depression or the Montgomery-Åsberg Depression Rating Scale. Short scales that are less reliant on physical symptoms, such as the BDI-FS, should receive more investigation to demonstrate their usefulness in screening for depression in medically ill patients.
Finally, the BDI-II suffers from the intrinsic limitations of self-report questionnaires. Some individuals cannot complete the scale due to illiteracy, physical debility, or compromised cognitive functioning. The widespread use of the BDI-II among the elderly is not suggested. Reporting bias that minimizes or over-reports symptom severity is a possible hazard that reduces its validity in several patients.
As a tradeoff between the psychometric robustness and enumerated disadvantages of the BDI-II, this self-report scale can be viewed as a cost-effective option because it is inexpensive in terms of professional time needed for administration and because it correlates well with clinician's ratings. Therefore, the BDI-II stands as a valid DSM-based tool with broad applicability in routine screening for depression in specialized medical clinics.
ACKNOWLEDGMENTS
Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) sponsored this article, and Dr. Yuan-Pang Wang is the recipient of the Grant (Process# 2008/11415-9). Conselho Nacional de Pesquisa (CNPq) sponsors Prof. Clarice Gorenstein.
Footnotes
No potential conflict of interest was reported.
REFERENCES
- 1.Katon W, Ciechanowski P. Impact of major depression on chronic medical illness. J Psychosomat Res. 2002;53(4):859–63. doi: 10.1016/s0022-3999(02)00313-6. [DOI] [PubMed] [Google Scholar]
- 2.Katon WJ. Clinical and health services relationships between major depression, depressive symptoms, and general medical illness. Biol Psychiatry. 2003;54(3):216–26. doi: 10.1016/s0006-3223(03)00273-7. [DOI] [PubMed] [Google Scholar]
- 3.Katon W, Lin EHB, Kroenke K. The association of depression and anxiety with medical symptom burden in patients with chronic medical illness. Gen Hosp Psychiatry. 2007;29(2):147–55. doi: 10.1016/j.genhosppsych.2006.11.005. [DOI] [PubMed] [Google Scholar]
- 4.Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23:56–62. doi: 10.1136/jnnp.23.1.56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Montgomery SA, Asberg M. A new depression scale designed to be sensitive to change. Br J Psychiatry. 1979;134:382–9. doi: 10.1192/bjp.134.4.382. [DOI] [PubMed] [Google Scholar]
- 6.Beck AT, Ward CH, Mendelson M, Mock J, Erbaugh J. An inventory for measuring depression. Arch Gen Psychiatry. 1961;4:561–71. doi: 10.1001/archpsyc.1961.01710120031004. [DOI] [PubMed] [Google Scholar]
- 7.Zung WW. A self-rating depression scale. Arch Gen Psychiatry. 1965;12:63–70. doi: 10.1001/archpsyc.1965.01720310065008. [DOI] [PubMed] [Google Scholar]
- 8.Radloff L. The CES-D Scale: A self-report depression scale for research in the general population. Appl Psychol Meas. 1977;1:385–401. [Google Scholar]
- 9.Spitzer RL, Kroenke K, Williams JBW. Validation and utility of a self-report version of PRIME-MD: the PHQ primary care study. JAMA. 1999;282(18):1737–44. doi: 10.1001/jama.282.18.1737. [DOI] [PubMed] [Google Scholar]
- 10.Rush AJ, Gullion CM, Basco MR, Jarrett RB, Trivedi MH. The Inventory of Depressive Symptomatology (IDS): psychometric properties. Psychol Med. 1996;26(3):477–86. doi: 10.1017/s0033291700035558. [DOI] [PubMed] [Google Scholar]
- 11.Parker G, Hilton T, Bains J, Hadzi-pavlovic D. Cognitive-based measures screening for depression in the medically ill: the DMI-10 and the DMI-18. Acta Psychiatr Scand. 2002;105(6):419–26. doi: 10.1034/j.1600-0447.2002.01248.x. [DOI] [PubMed] [Google Scholar]
- 12.Cox JL, Holden JM, Sagovsky R. Detection of postnatal depression. Development of the 10-item Edinburgh Postnatal Depression Scale. Br J Psychiatry. 1987;150:782–6. doi: 10.1192/bjp.150.6.782. [DOI] [PubMed] [Google Scholar]
- 13.Addington D, Addington J, Schissel B. A depression rating scale for schizophrenics. Schizophr Res. 1990;3(4):247–51. doi: 10.1016/0920-9964(90)90005-r. [DOI] [PubMed] [Google Scholar]
- 14.Fendrich M, Weissman MM, Warner V. Screening for depressive disorder in children and adolescents: validating the Center for Epidemiologic Studies Depression Scale for Children. Am J Epidemiol. 1990;131(3):538–51. doi: 10.1093/oxfordjournals.aje.a115529. [DOI] [PubMed] [Google Scholar]
- 15.Yesavage JA. Geriatric Depression Scale. Psychopharmacol Bull. 1988;24(4):709–11. [PubMed] [Google Scholar]
- 16.Andrade L, Gorenstein C, Vieira Filho Ah, Tung Tc, Artes R. Psychometric properties of the Portuguese version of the State-Trait Anxiety Inventory applied to college students: factor analysis and relation to the Beck Depression Inventory. Braz J Med Biol Res. 2001;34(3):367–74. doi: 10.1590/s0100-879x2001000300011. [DOI] [PubMed] [Google Scholar]
- 17.Gorenstein C, Andrade L, Zanolo E, Artes R. Expression of Depressive Symptoms in a Nonclinical Brazilian Adolescent Sample. Can J Psychiatry. 2005;50(3):129–36. doi: 10.1177/070674370505000301. [DOI] [PubMed] [Google Scholar]
- 18.Wang YP, Andrade LH, Gorenstein C. Validation of the Beck depression inventory for a Portuguese-speaking Chinese community in Brazil. Braz J Med Biol Res. 2005;38(3):399–408. doi: 10.1590/s0100-879x2005000300011. [DOI] [PubMed] [Google Scholar]
- 19.Wang YP, Lederman LP, Andrade LH, Gorenstein C. Symptomatic expression of depression among Jewish adolescents: effects of gender and age. Soc Psychiatry Psychiatr Epidemiol. 2008;43(1):79–86. doi: 10.1007/s00127-007-0270-4. [DOI] [PubMed] [Google Scholar]
- 20.Beck AT, Steer RA, Carbin MG. Psychometric properties of the Beck Depression Inventory: Twenty-five years of evaluation. Clin Psychol Rev. 1988;8(1):77–100. [Google Scholar]
- 21.Richter P, Werner J, Heerlein A, Kraus A, Sauer H. On the validity of the Beck Depression Inventory. A review. Psychopathology. 1998;31(3):160–8. doi: 10.1159/000066239. [DOI] [PubMed] [Google Scholar]
- 22.Wesley AL, Gatchel RJ, Garofalo JP, Polatin PB. Toward more accurate use of the Beck Depression Inventory with chronic back pain patients. Clin J Pain. 1999;15(2):117–21. doi: 10.1097/00002508-199906000-00008. [DOI] [PubMed] [Google Scholar]
- 23.Beck A, Rush A, Shaw B, Emery G. New York: Guilford Press; 1979. Cognitive therapy of depression. [Google Scholar]
- 24.Beck AT, Steer RA, Brown GK. 2nd ed San Antonio, TX: Psychological Corporation; 1996. BDI-II: Beck Depression Inventory Manual. [Google Scholar]
- 25.American Psychiatric Association. 4th ed Washington DC: American Psychiatric Association Press; 1994. Diagnostic and Statistical Manual of Mental Disorders. [Google Scholar]
- 26.Beck AT, Guth D, Steer RA, Ball R. Screening for major depression disorders in medical inpatients with the Beck Depression Inventory for Primary Care. Behav Res Ther. 1997;35(8):785–91. doi: 10.1016/s0005-7967(97)00025-9. [DOI] [PubMed] [Google Scholar]
- 27.Beck AT, Steer RA, Brown GK. San Antonio, TX: Psychological Corporation; 2000. Manual for the Beck Depression Inventory - Fast Screen for Medical Patients. [Google Scholar]
- 28.Beck AT, Steer RA, Ball R, Ranieri WF. Comparison of Beck Depression Inventories-IA and -II in psychiatric outpatients. J Pers Assess. 1996;67(3):588–97. doi: 10.1207/s15327752jpa6703_13. [DOI] [PubMed] [Google Scholar]
- 29.Corbière M, Bonneville-Roussy A, Franche RL, Coutu MF, Choiniere M, Durand MJ, et al. Further validation of the BDI-II among people with chronic pain originating from musculoskeletal disorders. Clin J Pain. 2011;27(1):62–9. doi: 10.1097/AJP.0b013e3181eef903. [DOI] [PubMed] [Google Scholar]
- 30.Penley JA, Wiebe JS, Nwosu A. Psychometric properties of the Spanish Beck Depression inventory II in a medical sample. Psychol Assess. 2003;15(4):569–77. doi: 10.1037/1040-3590.15.4.569. [DOI] [PubMed] [Google Scholar]
- 31.Gomes-Oliveira MH, Gorenstein C, Neto FL, Andrade LH, Wang YP. Validation of the Brazilian Portuguese version of the Beck Depression Inventory-II in a community sample. Rev Bras Psiquiatr. 2012;34(4):389–94. doi: 10.1016/j.rbp.2012.03.005. [DOI] [PubMed] [Google Scholar]
- 32.Alansari BM. Beck Depression Inventory (BDI-II) items characteristics among undergraduate students of nineteen Islamic countries. Soc Behav Pers. 2005;33(7):675–84. [Google Scholar]
- 33.McPherson A, Martin CR. A narrative review of the Beck Depression Inventory (BDI) and implications for its use in an alcohol-dependent population. J Psychiatr Ment Health Nurs. 2010;17(1):19–30. doi: 10.1111/j.1365-2850.2009.01469.x. [DOI] [PubMed] [Google Scholar]
- 34.Furukawa TA. Assessment of mood: guides for clinicians. J Psychosom Res. 2010;68(6):581–9. doi: 10.1016/j.jpsychores.2009.05.003. [DOI] [PubMed] [Google Scholar]
- 35.Shafer AB. Meta-analysis of the factor structures of four depression questionnaires: Beck, CES-D, Hamilton, and Zung. J Clin Psychol. 2006;62(1):123–46. doi: 10.1002/jclp.20213. [DOI] [PubMed] [Google Scholar]
- 36.Kazdin A. Oxford: American Psychological Association; 2000. Encyclopedia of Psychology. [Google Scholar]
- 37.McDowell I. New York: Oxford University Press; 2006. Measuring health: a guide to rating scales and questionnaires. 3rd ed. [Google Scholar]
- 38.Dozois D. Beck Depression Inventory-II. In: Weiner I, Craighead W, editors. The Corsini Encyclopedia of Psychology. New York: John Wiley & Sons; 2010. pp. 210–1. 4th ed. p. [Google Scholar]
- 39.Winter LB, Steer RA, Jones-Hicks L, Beck AT. Screening for major depression disorders in adolescent medical outpatients with the Beck Depression Inventory for Primary Care. J Adolesc Health. 1999;24(6):389–94. doi: 10.1016/s1054-139x(98)00135-9. [DOI] [PubMed] [Google Scholar]
- 40.Pietsch K, Hoyler A, Fruhe B, Kruse J, Schulte-Korne G, Allgaier AK. [Early detection of major depression in paediatric care: validity of the beck depression inventory-second edition (BDI-II) and the beck depression inventory-fast screen for medical patients (BDI-FS)] Psychother Psychosom Med Psychol. 2012;62(11):418–24. doi: 10.1055/s-0032-1314869. [DOI] [PubMed] [Google Scholar]
- 41.Arnarson TO, Olason DT, Smari J, Sigurethsson JF. Nord J Psychiatry. Norway: 2008. The Beck Depression Inventory Second Edition (BDI-II): psychometric properties in Icelandic student and patient populations; pp. 360–5. 62. p. [DOI] [PubMed] [Google Scholar]
- 42.Arnau RC, Meagher MW, Norris MP, Bramson R. Psychometric evaluation of the Beck Depression Inventory-II with primary care medical patients. Health Psychol. 2001;20(2):112–9. doi: 10.1037//0278-6133.20.2.112. [DOI] [PubMed] [Google Scholar]
- 43.Brown M, Kaplan C, Jason L. Factor analysis of the Beck Depression Inventory-II with patients with chronic fatigue syndrome. J Health Psychol. 2012;17(6):799–808. doi: 10.1177/1359105311424470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Beck CT, Gable RK. Comparative analysis of the performance of the postpartum depression screening scale with two other depression instruments. Nurs Res. 2001;50(4):242–50. doi: 10.1097/00006199-200107000-00008. [DOI] [PubMed] [Google Scholar]
- 45.Bunevicius A, Staniute M, Brozaitiene J, Bunevicius R. Diagnostic accuracy of self-rating scales for screening of depression in coronary artery disease patients. J Psychosom Res. 2012;72(1):22–5. doi: 10.1016/j.jpsychores.2011.10.006. [DOI] [PubMed] [Google Scholar]
- 46.Carney CE, Ulmer C, Edinger JD, Krystal AD, Knauss F. Assessing depression symptoms in those with insomnia: an examination of the beck depression inventory second edition (BDI-II) J Psychiatr Res. 2009;43(5):576–82. doi: 10.1016/j.jpsychires.2008.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Carvalho Bos S, Pereira AT, Marques M, Maia B, Soares MJ, Valente J, et al. The BDI-II factor structure in pregnancy and postpartum: Two or three factors. Eur Psychiatry. 2009;24(5):334–40. doi: 10.1016/j.eurpsy.2008.10.003. [DOI] [PubMed] [Google Scholar]
- 48.Chaudron LH, Szilagyi PG, Tang W, Anson E, Talbot NL, Wadkins HI, et al. Accuracy of depression screening tools for identifying postpartum depression among urban mothers. Pediatrics. 2010;125(3):e609–17. doi: 10.1542/peds.2008-3261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Chilcot J, Wellsted D, Farrington K. Screening for depression while patients dialyse: an evaluation. Nephrol Dial Transplant. 2008;23(8):2653–9. doi: 10.1093/ndt/gfn105. [DOI] [PubMed] [Google Scholar]
- 50.Chilcot J, Norton S, Wellsted D, Almond M, Davenport A, Farrington K. A confirmatory factor analysis of the Beck Depression Inventory-II in end-stage renal disease patients. J Psychosom Res. 2011;71(3):148–53. doi: 10.1016/j.jpsychores.2011.02.006. [DOI] [PubMed] [Google Scholar]
- 51.Chung L-J, Tsai P-S, Liu B-Y, Chou K-R, Lin W-H, Shyu Y-K, et al. Home-based deep breathing for depression in patients with coronary heart disease: A randomised controlled trial. Int J Nurs Stud. 2010;47(11):1346–53. doi: 10.1016/j.ijnurstu.2010.03.007. [DOI] [PubMed] [Google Scholar]
- 52.Dbouk N, Arguedas MR, Sheikh A. Assessment of the PHQ-9 as a screening tool for depression in patients with chronic hepatitis C. . Dig Dis Sci. 2008;53(4):1100–6. doi: 10.1007/s10620-007-9985-z. [DOI] [PubMed] [Google Scholar]
- 53.de Souza J, Jones LA, Rickards H. Validation of Self-Report Depression Rating Scales in Huntigton's Disease. Mov Disord. 2010;25(1):91–6. doi: 10.1002/mds.22837. [DOI] [PubMed] [Google Scholar]
- 54.del Piño Perez A, Ibanez Fernandez I, Bosa Ojeda F, Dorta Gonzalez R, Gaos Miezoso MT. [Factor models of the Beck Depression Inventory-II. Validation with coronary patients and a critique of Ward's model]. . Psicothema. 2012;24(1):127–32. [PubMed] [Google Scholar]
- 55.Dutton GR, Grothe KB, Jones GN, Whitehead D, Kendra K, Brantley PJ. Use of the Beck Depression Inventory-II with African American primary care patients. Gen Hosp Psychiatry. 2004;26(6):437–42. doi: 10.1016/j.genhosppsych.2004.06.002. [DOI] [PubMed] [Google Scholar]
- 56.Grothe KB, Dutton GR, Jones GN, Bodenlos J, Ancona M, Brantley PJ. Validation of the Beck Depression Inventory-II in a low-income African American sample of medical outpatients. Psychol Assess. 2005;17(1):110–4. doi: 10.1037/1040-3590.17.1.110. [DOI] [PubMed] [Google Scholar]
- 57.Findler M, Cantor J, Haddad L, Gordon W, Ashman T. The reliability and validity of the SF-36 health survey questionnaire for use with individuals with traumatic brain injury. Brain Inj. 2001;15(8):715–23. doi: 10.1080/02699050010013941. [DOI] [PubMed] [Google Scholar]
- 58.Frasure-Smith N, Lespérance F. Depression and anxiety as predictors of 2-year cardiac events in patients with stable coronary artery disease. Arch Gen Psychiatry. 2008;65(1):62–71. doi: 10.1001/archgenpsychiatry.2007.4. [DOI] [PubMed] [Google Scholar]
- 59.Griffith NM, Szaflarski JP, Szaflarski M, Kent GP, Schefft BK, Howe SR, et al. Measuring depressive symptoms among treatment-resistant seizure disorder patients: POMS Depression scale as an alternative to the BDI-II. Epilepsy Behav. 2005;7(2):266–72. doi: 10.1016/j.yebeh.2005.05.004. [DOI] [PubMed] [Google Scholar]
- 60.Hamid H, Abu-Hijleh NS, Sharif SL, Raqab ZM, Mas, ad D, et al. A primary care study of the correlates of depressive symptoms among Jordanian women. Transcult Psychiatry. 2004;41(4):487–96. doi: 10.1177/1363461504047931. [DOI] [PubMed] [Google Scholar]
- 61.Harris CA, D'Eon JL. Psychometric properties of the Beck Depression Inventory-Second Edition (BDI-II) in individuals with chronic pain. Pain. 2008;137(3):609–22. doi: 10.1016/j.pain.2007.10.022. [DOI] [PubMed] [Google Scholar]
- 62.Hayden MJ, Brown WA, Brennan L, Brien PE. Validity of the Beck Depression Inventory as a Screening Tool for a Clinical Mood Disorder in Bariatric Surgery Candidates. Obes Surg. 2012;22(11):1666–75. doi: 10.1007/s11695-012-0682-4. [DOI] [PubMed] [Google Scholar]
- 63.Jones JE, Hermann BP, Woodard JL, Barry JJ, Gilliam F, Kanner AM, et al. Screening for major depression in epilepsy with common self-report depression inventories. Epilepsia. 2005;46(5):731–5. doi: 10.1111/j.1528-1167.2005.49704.x. [DOI] [PubMed] [Google Scholar]
- 64.Kanner AM, Barry JJ, Gilliam F, Hermann B, Meador KJ. Anxiety disorders, subsyndromic depressive episodes, and major depressive episodes: Do they differ on their impact on the quality of life of patients with epilepsy. Epilepsia. 2010;51(7):1152–8. doi: 10.1111/j.1528-1167.2010.02582.x. [DOI] [PubMed] [Google Scholar]
- 65.King PR, Donnelly KT, Donnelly JP, Dunnam M, Warner G, Kittleson CJ, et al. Psychometric study of the Neurobehavioral Symptom Inventory. J Rehabil Res Dev. 2012;49(6):879–88. doi: 10.1682/jrrd.2011.03.0051. [DOI] [PubMed] [Google Scholar]
- 66.Kiropoulos LA, Meredith I, Tonkin A, Clarke D, Antonis P, Plunkett J. Psychometric properties of the cardiac depression scale in patients with coronary heart disease. BMC Psychiatry. 2012;12:216. doi: 10.1186/1471-244X-12-216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Kirsch-Darrow L, Marsiske M, Okun MS, Bauer R, Bowers D. Apathy and depression: separate factors in Parkinson's disease. J Int Neuropsychol Soc. 2011;17(6):1058–66. doi: 10.1017/S1355617711001068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Ko P-W, Hwang J, Lim H-W, Park S-P. Reliability and validity of the Korean version of the Neurological Disorders Depression Inventory for Epilepsy (K-NDDI-E) Epilepsy Behav. 2012;25(4):539–42. doi: 10.1016/j.yebeh.2012.09.010. [DOI] [PubMed] [Google Scholar]
- 69.Lipps GE, Lowe GA, De La Haye W, Longman-Mills S, Clarke TR, Barton EN, et al. Validation of the Beck Depression Inventory II in HIV-positive Patients. West Indian Med J. 2010;59(4):374–9. [PubMed] [Google Scholar]
- 70.Lopez MN, Pierce RS, Gardner RD, Hanson RW. Standardized Beck Depression Inventory-II Scores for Male Veterans Coping With Chronic Pain. Psychol Serv. 2013;10(2):257–63. doi: 10.1037/a0027920. [DOI] [PubMed] [Google Scholar]
- 71.Masuda M, Utsugisawa K, Suzuki S, Nagane Y, Kabasawa C, Suzuki Y, et al. The MG-QOL15 Japanese version: validation and associations with clinical factors. Muscle Nerve. 2012;46(2):166–73. doi: 10.1002/mus.23398. [DOI] [PubMed] [Google Scholar]
- 72.Neitzer A, Sun S, Doss S, Moran J, Schiller B. Beck Depression Inventory-Fast Screen (BDI-FS): an efficient tool for depression screening in patients with end-stage renal disease. Hemodial Int. 2012;16(2):207–13. doi: 10.1111/j.1542-4758.2012.00663.x. [DOI] [PubMed] [Google Scholar]
- 73.Ooms E, Meganck R, Vanheule S, Vinck B, Watelet JB, Dhooge I. Tinnitus severity and the relation to depressive symptoms: a critical study. Otolaryngol Head Neck Surg. 2011;145(2):276–81. doi: 10.1177/0194599811403381. [DOI] [PubMed] [Google Scholar]
- 74.Osada K, Oka H, Isomura T, Nakamura I, Tominaga K, Takahashi S, et al. Development of the Japanese version of the Fibromyalgia Impact Questionnaire (JFIQ): psychometric assessments of reliability and validity. Int J Rheum Dis. 2011;14(1):74–80. doi: 10.1111/j.1756-185X.2010.01585.x. [DOI] [PubMed] [Google Scholar]
- 75.Patterson AL, Morasco BJ, Fuller BE, Indest DW, Loftis JM, Hauser P. Screening for depression in patients with hepatitis C using the Beck Depression Inventory-II: do somatic symptoms compromise validity. Gen Hosp Psychiatry. 2011;33(4):354–62. doi: 10.1016/j.genhosppsych.2011.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Pereira AT, Bos SC, Marques M, Maia BR, Soares MJ, Valente J, et al. The postpartum depression screening scale: is it valid to screen for antenatal depression. Arch Womens Ment Health. 2011;14(3):227–38. doi: 10.1007/s00737-010-0178-y. [DOI] [PubMed] [Google Scholar]
- 77.Poole H, White S, Blake C, Murphy P, Bramwell R. Depression in chronic pain patients: prevalence and measurement. Pain Pract. 2009;9(3):173–80. doi: 10.1111/j.1533-2500.2009.00274.x. [DOI] [PubMed] [Google Scholar]
- 78.Rampling J, Mitchell AJ, Von Oertzen T, Docker J, Jackson J, Cock H, et al. Screening for depression in epilepsy clinics. A comparison of conventional and visual-analog methods. Epilepsia. 2012;53(10):1713–21. doi: 10.1111/j.1528-1167.2012.03571.x. [DOI] [PubMed] [Google Scholar]
- 79.Roebuck-Spencer TM, Yarboro C, Nowak M, Takada K, Jacobs G, Lapteva L, et al. Use of computerized assessment to predict neuropsychological functioning and emotional distress in patients with systemic lupus erythematosus. Arthritis Rheum. 2006;55(3):434–41. doi: 10.1002/art.21992. [DOI] [PubMed] [Google Scholar]
- 80.Su KP, Chiu TH, Huang CL, Ho M, Lee CC, Wu PL, et al. Different cutoff points for different trimesters. The use of Edinburgh Postnatal Depression Scale and Beck Depression Inventory to screen for depression in pregnant Taiwanese women. . Gen Hosp Psychiatry. 2007;29(5):436–41. doi: 10.1016/j.genhosppsych.2007.05.005. [DOI] [PubMed] [Google Scholar]
- 81.Suzuki Y, Utsugisawa K, Suzuki S, Nagane Y, Masuda M, Kabasawa C, et al. Factors associated with depressive state in patients with myasthenia gravis: a multicentre cross-sectional study. BMJ Open. 2011;1(2):e000313. doi: 10.1136/bmjopen-2011-000313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Tandon SD, Cluxton-Keller F, Leis J, Le HN, Perry DF. A comparison of three screening tools to identify perinatal depression among low-income African American women. J Affect Disord. 2012;136(1-2):155–62. doi: 10.1016/j.jad.2011.07.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Teng HW, Hsu CS, Shih SM, Lu ML, Pan JJ, Shen WW. Screening postpartum depression with the Taiwanese version of the Edinburgh Postnatal Depression Scale. Compr Psychiatry. 2005;46(4):261–5. doi: 10.1016/j.comppsych.2004.10.003. [DOI] [PubMed] [Google Scholar]
- 84.Turner A, Hambridge J, White J, Carter G, Clover K, Nelson L, et al. Depression Screening in Stroke A Comparison of Alternative Measures With the Structured Diagnostic Interview for the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (Major Depressive Episode) as Criterion Standard. Stroke. 2012;43(4):1000–5. doi: 10.1161/STROKEAHA.111.643296. [DOI] [PubMed] [Google Scholar]
- 85.Turner-Stokes L, Kalmus M, Hirani D, Clegg F. The Depression Intensity Scale Circles (DISCs): a first evaluation of a simple assessment tool for depression in the context of brain injury. J Neurol Neurosurg Psychiatry. 2005;76(9):1273–8. doi: 10.1136/jnnp.2004.050096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Viljoen JL, Iverson GL, Griffiths S, Woodward TS. Factor structure of the Beck Depression Inventory-II in a medical outpatient sample. J Clin Psychol Med Settings. 2003;10(4):289–91. [Google Scholar]
- 87.Wan Mahmud WM, Awang A, Herman I, Mohamed MN. Analysis of the psychometric properties of the Malay version of Beck Depression Inventory II (BDI-II) among postpartum women in Kedah, North West of Peninsular Malaysia. Malays J Med Sci. 2004;11(2):19–25. [PMC free article] [PubMed] [Google Scholar]
- 88.Warmenhoven F, van Rijswijk E, Engels Y, Kan C, Prins J, van Weel C, et al. The Beck Depression Inventory (BDI-II) and a single screening question as screening tools for depressive disorder in Dutch advanced cancer patients. Support Care Cancer. 2012;20(2):319–24. doi: 10.1007/s00520-010-1082-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Williams JR, Hirsch ES, Anderson K, Bush AL, Goldstein SR, Grill S, et al. A comparison of nine scales to detect depression in Parkinson disease: which scale to use. Neurology. 2012;78(13):998–1006. doi: 10.1212/WNL.0b013e31824d587f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Young QR, Ignaszewski A, Fofonoff D, Kaan A. Brief screen to identify 5 of the most common forms of psychosocial distress in cardiac patients: validation of the screening tool for psychological distress. J Cardiovasc Nurs. 2007;22(6):525–34. doi: 10.1097/01.JCN.0000297383.29250.14. [DOI] [PubMed] [Google Scholar]
- 91.Zahodne LB, Young S, Kirsch-Darrow L, Nisenzon A, Fernandez HH, Okun MS, et al. Examination of the Lille Apathy Rating Scale in Parkinson disease. Mov Disord. 2009;24(5):677–83. doi: 10.1002/mds.22441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Di Benedetto M, Lindner H, Hare DL, Kent S. Depression following acute coronary syndromes: a comparison between the Cardiac Depression Scale and the Beck Depression Inventory II. J Psychosom Res. 2006;60(1):13–20. doi: 10.1016/j.jpsychores.2005.06.003. [DOI] [PubMed] [Google Scholar]
- 93.Gorenstein C, Wang Y, Argimon I, Werlang B. São Paulo: Editora Casa do Psicólogo; 2011. Manual do Inventário de Depressão de Beck - BDI-II. [Google Scholar]
- 94.Homaifar BY, Brenner LA, Gutierrez PM, Harwood JF, Thompson C, Filley CM, et al. Sensitivity and specificity of the Beck Depression Inventory-II in persons with traumatic brain injury. Arch Phys Med Rehabil. 2009;90(4):652–6. doi: 10.1016/j.apmr.2008.10.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Huffman JC, Doughty CT, Januzzi JL, Pirl WF, Smith FA, Fricchione GL. Screening for major depression in post-myocardial infarction patients: operating characteristics of the Beck Depression Inventory-II. Int J Psychiatry Med. 2010;40(2):187–97. doi: 10.2190/PM.40.2.e. [DOI] [PubMed] [Google Scholar]
- 96.Jamroz-Wisniewska A, Papuc E, Bartosik-Psujek H, Belniak E, Mitosek-Szewczyk K, Stelmasiak Z. [Validation of selected aspects of psychometry of the Polish version of the Multiple Sclerosis Impact Scale 29 (MSIS-29)] Neurol Neurochir Pol. 2007;41(3):215–22. [PubMed] [Google Scholar]
- 97.Low GD, Hubley AM. Screening for depression after cardiac events using the Beck Depression Inventory-II and the Geriatric Depression Scale. Social Indicators Research. 2007;82(3):527–48. [Google Scholar]
- 98.Rowland SM, Lam CS, Leahy B. Use of the Beck Depression Inventory-II (BDI-II) with persons with traumatic brain injury: Analysis of factorial structure. Brain Inj. 2005;19(2):77–83. doi: 10.1080/02699050410001719988. [DOI] [PubMed] [Google Scholar]
- 99.Siegert RJ, Walkey FH, Turner-Stokes L. An examination of the factor structure of the Beck Depression Inventory-II in a neurorehabilitation inpatient sample. J Int Neuropsychol Soc. 2009;15(1):142–7. doi: 10.1017/S1355617708090048. [DOI] [PubMed] [Google Scholar]
- 100.Thomas SA, Lincoln NB. Depression and cognitions after stroke: validation of the Stroke Cognitions Questionnaire Revised (SCQR) Disabil Rehabil. 2008;30(23):1779–85. doi: 10.1080/09638280701661430. [DOI] [PubMed] [Google Scholar]
- 101.Thombs BD, Ziegelstein RC, Beck CA, Pilote L. A general factor model for the Beck Depression Inventory-II: validation in a sample of patients hospitalized with acute myocardial infarction. J Psychosom Res. 2008;65(2):115–21. doi: 10.1016/j.jpsychores.2008.02.027. [DOI] [PubMed] [Google Scholar]
- 102.Tully PJ, Winefield HR, Baker RA, Turnbull DA, de Jonge P. Confirmatory factor analysis of the Beck Depression Inventory-II and the association with cardiac morbidity and mortality after coronary revascularization. J Health Psychol. 2011;16(4):584–95. doi: 10.1177/1359105310383604. [DOI] [PubMed] [Google Scholar]
- 103.Poole H, Bramwell R, Murphy P. The utility of the Beck Depression Inventory Fast Screen (BDI-FS) in a pain clinic population. Eur J Pain. 2009;13(8):865–9. doi: 10.1016/j.ejpain.2008.09.017. [DOI] [PubMed] [Google Scholar]
- 104.Scheinthal SM, Steer RA, Giffin L, Beck AT. Evaluating geriatric medical outpatients with the Beck depression Inventory-Fast Screen for medical patients. Aging Ment Health. 2001;5(2):143–8. doi: 10.1080/13607860120038320. [DOI] [PubMed] [Google Scholar]
- 105.Servaes P, van der Werf S, Prins J, Verhagen S, Bleijenberg G. Fatigue in disease-free cancer patients compared with fatigue in patients with Chronic Fatigue Syndrome. Support Care Cancer. 2001;9(1):11–7. doi: 10.1007/s005200000165. [DOI] [PubMed] [Google Scholar]
- 106.Servaes P, Prins J, Verhagen S, Bleijenberg G. Fatigue after breast cancer and in chronic fatigue syndrome - Similarities and differences. J Psychosomat Res. 2002;52(6):453–9. doi: 10.1016/s0022-3999(02)00300-8. [DOI] [PubMed] [Google Scholar]
- 107.Steer RA, Cavalieri TA, Leonard DM, Beck AT. Use of the Beck Depression Inventory for Primary Care to screen for major depression disorders. Gen Hosp Psychiatry. 1999;21(2):106–11. doi: 10.1016/s0163-8343(98)00070-x. [DOI] [PubMed] [Google Scholar]
- 108.Nunnally J, Bernstein I. 3rd ed New York: McGraw-Hill; 1994. Psychometric theory. [Google Scholar]
- 109.Hanel G, Henningsen P, Herzog W, Sauer N, Schaefert R, Szecsenyi J, et al. Depression, anxiety, and somatoform disorders: vague or distinct categories in primary care. Results from a large cross-sectional study. J Psychosomat Res. 2009;67(3):189–97. doi: 10.1016/j.jpsychores.2009.04.013. [DOI] [PubMed] [Google Scholar]
- 110.Voigt K, Nagel A, Meyer B, Langs G, Braukhaus C, Löwe B. Towards positive diagnostic criteria: A systematic review of somatoform disorder diagnoses and suggestions for future classification. J Psychosomat Res. 2010;68(5):403–14. doi: 10.1016/j.jpsychores.2010.01.015. [DOI] [PubMed] [Google Scholar]
- 111.Ramasubbu R, Beaulieu S, Taylor VH, Schaffer A, McIntyre RS. The CANMAT task force recommendations for the management of patients with mood disorders and comorbid medical conditions: diagnostic, assessment, and treatment principles. Ann Clin Psychiatry. 2012;24(1):82–90. [PubMed] [Google Scholar]
- 112.Zigmond AS, Snaith RP. The hospital anxiety and depression scale. Acta Psychiatr Scand. 1983;67(6):361–70. doi: 10.1111/j.1600-0447.1983.tb09716.x. [DOI] [PubMed] [Google Scholar]
- 113.Bjelland I, Dahl AA, Haug TT, Neckelmann D. The validity of the Hospital Anxiety and Depression Scale - An updated literature review. J Psychosomat Res. 2002;52(2):69–77. doi: 10.1016/s0022-3999(01)00296-3. [DOI] [PubMed] [Google Scholar]
- 114.Cosco TD, Doyle F, Ward M, McGee H. Latent structure of the Hospital Anxiety And Depression Scale: a 10-year systematic review. J Psychosomat Res. 2012;72(3):180–4. doi: 10.1016/j.jpsychores.2011.06.008. [DOI] [PubMed] [Google Scholar]
- 115.Ghassemzadeh H, Mojtabai R, Karamghadiri N, Ebrahimkhani N. Psychometric properties of a Persian-language version of the Beck Depression Inventory second edition: BDI-II-Persian. Depress Anxiety. 2005;21(4):185–92. doi: 10.1002/da.20070. [DOI] [PubMed] [Google Scholar]
- 116.Sprinkle SD, Lurie D, Insko SL, Atkinson G, Jones GL, Logan AR, et al. Criterion Validity, Severity Cut Scores, and Test-Retest Reliability of the Beck Depression Inventory-II in a University Counseling Center Sample. J Couns Psychol. 2002;49(3):381–5. [Google Scholar]
- 117.Hambleton R, Swaminathan H, HJ R. Newbury Park, CA: Sage; 1991. Fundamentals of item response theory. [Google Scholar]
- 118.Waller NG, Compas BE, Hollon SD, Beckjord E. Measurement of depressive symptoms in women with breast cancer and women with clinical depression: A differential item functioning analysis. J Clin Psychol Med Settings. 2005;12(2):127–41. [Google Scholar]
- 119.Leonardson GR, Kemper E, Ness FK, Koplin BA, Daniels MC, Leonardson GA. Validity and reliability of the audit and CAGE-AID in Northern Plains American Indians. Psychol Rep. 2005;97(1):161–6. doi: 10.2466/pr0.97.1.161-166. [DOI] [PubMed] [Google Scholar]
- 120.Anastasi A, Urbina S. 7th ed London: Prentice-Hall International; 1997. Psychological Testing. [Google Scholar]
- 121.Shean G, Baldwin G. Sensitivity and specificity of depression questionnaires in a college-age sample. J Genet Psychol. 2008;169(3):281–8. doi: 10.3200/GNTP.169.3.281-292. [DOI] [PubMed] [Google Scholar]
- 122.Bagby RM, Ryder AG, Schuller DR, Marshall MB. The Hamilton depression rating scale: Has the gold standard become a lead weight. Am J Psychiatry. 2004;161(12):2163–77. doi: 10.1176/appi.ajp.161.12.2163. [DOI] [PubMed] [Google Scholar]
- 123.Kriston L, von Wolff A. Not as golden as standards should be: Interpretation of the Hamilton Rating Scale for Depression. J Affect Disord. 2011;128(1):175–7. doi: 10.1016/j.jad.2010.07.011. [DOI] [PubMed] [Google Scholar]
- 124.Cicchetti DV. Guidelines, Criteria, and Rules of Thumb for Evaluating Normed and Standardized Assessment Instruments in Psychology. Psychol Assess. 1994;6(4):284–90. [Google Scholar]
- 125.Moran P, Lambert M. A review of current assessment tools for monitoring changes in depression. In: Lambert M, Christensen E, DeJulio S, editors. New York: The Assessment of Psychotherapy Outcome; Wiley; 1983. [Google Scholar]
- 126.Osman A, Barrios FX, Gutierrez PM, Williams JE, Bailey J. Psychometric properties of the Beck Depression Inventory-II in nonclinical adolescent samples. J Clin Psychol. 2008;64(1):83–102. doi: 10.1002/jclp.20433. [DOI] [PubMed] [Google Scholar]
- 127.Byrne BM. Factor analytic models: viewing the structure of an assessment instrument from three perspectives. J Pers Assess. 2005;85(1):17–32. doi: 10.1207/s15327752jpa8501_02. [DOI] [PubMed] [Google Scholar]
- 128.Byrne B. Mahwah, New Jersey: Lawrence Erlbaum Associates; 1998. Structural equation modeling with LISREL, PRELIS and SIMPLIS: Basic concepts, applications and programming. [Google Scholar]