Abstract
Background
Few studies have compared different blood pressure (BP) indexes for end-stage renal disease (ESRD) risk among individuals with chronic kidney disease.
Methods
We examined the relationship between systolic BP (SBP), diastolic BP (DBP), pulse pressure (PP) and mean arterial pressure (MAP) and ESRD risk among 2,772 participants with an estimated glomerular filtration rate (eGFR) <60 ml/min/1.73 m2 calculated using the Chronic Kidney Disease Epidemiology Collaboration equation in the REasons for the Geographic And Racial Differences in Stroke (REGARDS) study. BP was measured during a baseline study visit between January 2003 and October 2007 with ESRD incidence through August 2009 ascertained via linkage with the United States Renal Data System (n = 138 ESRD cases).
Results
The mean age was 72.1(standard deviation: 8.7) years. After multivariable adjustment for socio-demographic and clinical risk factors including antihypertensive medication use, the hazard ratio (HR) for ESRD associated with one standard deviation higher SBP (18 mm Hg) was 1.67, (95% confidence intervals (CI) 1.43–1.96), DBP (11 mm Hg) was 1.38, (95% CI 1.16–1.63), PP (15 mm Hg) was 1.50, (95% CI 1.27–1.78) and MAP (11 mm Hg) was 1.54, (95% CI 1.32–1.79). Higher levels of SBP remained associated with an increased HR for ESRD after additional adjustment for DBP (1.65, 95% CI: 1.35–2.01), PP (1.73, 95% CI: 1.32–2.26), and MAP (1.61, 95% CI: 1.16–2.23). After adjustment for SBP, the other BP indexes were not significantly associated with incident ESRD.
Conclusions
These data suggest that of several blood pressure indexes including DBP, PP and MAP, SBP may have the strongest association with ESRD incidence among individuals with reduced eGFR.
Keywords: blood pressure, chronic kidney disease, end-stage renal disease, hypertension, pulse pressure, systolic blood pressure
Chronic kidney disease (CKD) is common with recent estimates indicating a prevalence of 13% among US adults.1 Individuals with CKD have a substantially increased risk for end-stage renal disease (ESRD).2–4 The vast majority of patients with CKD have hypertension5–8 and it is considered one of the most important risk factors for ESRD.9,10
Although hypertension is widely recognized as an independent risk factor for ESRD, few prospective studies have compared the risk for ESRD associated with different blood pressure (BP) indexes. Although two large studies11,12 have reported that systolic BP (SBP) was more strongly associated with incident ESRD as compared with diastolic BP (DBP) or pulse pressure (PP), neither of these studies had estimates of renal function at baseline. Data are limited as to the importance of SBP vs. other BP indexes in individuals with reduced estimated glomerular filtration rate (eGFR). In a small study of predominantly male veterans with CKD, SBP but not DBP was associated with ESRD; the association for PP was not reported.13 Understanding the relative strength of these associations could provide insight into mechanisms and optimal approaches for treating patients. Therefore, the goal of the current analysis was to assess the associations between SBP, DBP, PP and mean arterial pressure (MAP), and the incidence of ESRD among adults with moderate-to-severe CKD, defined as an eGFR <60 ml/min/1.73 m2. To accomplish this goal, we analyzed data from the REasons for the Geographic And Racial Differences in Stroke (REGARDS) study.
Methods
Study participants
The REGARDS study is a population-based investigation of stroke incidence among US adults ≥45 years of age.14 The study was designed to oversample African Americans and to provide approximate equal representation of men and women. By design, 56% (goal 50%) of the sample was recruited from the eight southern US states, commonly referred to as the “stroke buckle” (coastal North Carolina, South Carolina, and Georgia) and “stroke belt” (remainder of North Carolina, South Carolina, and Georgia as well as Alabama, Mississippi, Tennessee, Arkansas, and Louisiana), with the remaining 44% of the sample recruited from the rest of the continental US. Overall, 30,239 African-American and white US adults were enrolled between January 2003 and October 2007. We limited this analysis to the participants with an eGFR <60 ml/min/1.73m2 who were not receiving renal replacement therapy at baseline (n = 3,201). We excluded participants who were missing data on SBP or DBP (n = 11) or other covariates (n = 272). After these exclusions, 2,772 participants were included in all analyses. The REGARDS study protocol was approved by the Institutional Review Boards governing research in human subjects at the participating centers and all participants provided informed consent.
Data collection
Trained interviewers conducted computer assisted telephone interviews to obtain information on participants’ demographics, current cigarette smoking, physical activity, alcohol intake, annual household income, education, and self-report of prior diagnoses of major co-morbid conditions including diabetes and myocardial infarction.
Trained health professionals conducted in-home study visits that included a physical examination and the collection of blood and urine samples. Diabetes was defined as a serum glucose ≥126 mg/dl for participants who had fasted ≥8 h before their study visit, serum glucose ≥200 mg/dl for those who had not fasted, or self-report of a prior diagnosis of diabetes with current use of insulin or oral hypoglycemic medications. High-sensitivity C-reactive protein was measured using a high-sensitivity particle-enhanced immunonephelometric assay. High-density lipoprotein (HDL) cholesterol was measured using colorimetric reflectance spectrophotometry. Waist circumference was measured mid-way between the lowest rib and the iliac crest using a tape measure with participants standing. A history of cardiovascular disease was defined based on a self-reported history of myocardial infarction or coronary revascularization (coronary angioplasty or bypass surgery) or stroke.
Using isotope-dilution mass spectrometry-traceable serum creatinine, eGFR was calculated using the Chronic Kidney Disease Epidemiology Collaboration equation.15,16 Reduced eGFR was defined as levels <60 ml/min/1.73m2. Using a random spot urine collection, albuminuria was calculated as the ratio of urinary albumin concentration to urinary creatinine concentration.17
BP
During the in-home examination, BP was measured two times by a trained technician following a standardized protocol and regularly tested aneroid sphygmomanometer (American Diagnostic, Hauppauge, NY). Participants were asked to sit for 3 min with both feet on the floor before the BP measurement. Where possible, blood pressures were taken in the left arm and a large size cuff was used if the arm circumference was >13 inches. Both the cuff bladder width and pulse obliteration level were recorded. The cuff was inflated to 20 mm Hg above the pulse obliteration level and slowly deflated (~2 mm Hg/s). This process was repeated to obtain the second blood pressure on the same arm. A 30-s rest occurred between BP measurements. BP quality control was monitored by central examination of digit preference and retraining of technicians took place as necessary. The two BP measurements were averaged for the current analyses.18
Study outcome
ESRD subsequent to the REGARDS study inhome examination and through 31 August 2009 was assessed via linkage with the United States Renal Data System (USRDS). The USRDS is a registry of ESRD and records over 95% of incident ESRD cases in the United States. Time to ESRD was recorded as the number of days between a participant’s baseline examination and the date of initiation of renal replacement therapy recorded in the USRDS. Participants who did not develop ESRD were censored on their date of death or the end of follow-up (31 August 2009), whichever occurred first.
Statistical analyses
Baseline characteristics were calculated for REGARDS study participants with an eGFR <60 ml/min/1.73m2. The incidence rate of ESRD was calculated by a priori selected levels of SBP (<120, 120–129, 130–139, and ≥140 mm Hg), DBP (<70, 70–79, 80–89, and ≥90 mm Hg), PP (<45, 45–54, 55–64, and ≥65 mm Hg) and MAP (<86, 86–93, 93–100, and ≥100 mm Hg). Hazard ratios (HR) for ESRD associated with these BP levels were calculated using Cox proportional hazard regression models after initial adjustment for age, race, sex, and geographic region. Subsequent models included additional adjustment for current smoking, physical activity, annual household income, waist circumference, high-sensitivity C-reactive protein, history of cardiovascular disease, diabetes, HDL cholesterol, antihypertensive medicine use, and eGFR. To explore further, the association of each BP index with ESRD, we calculated the multivariable adjusted HR for ESRD associated with SBP, DBP, PP and MAP, modeled as continuous variables. First, these associations were assessed using multivariable adjusted restricted quadratic splines. Next, each of the BP indexes were modeled to reflect one standard deviation higher level (18 mm Hg for SBP, 11 mm Hg for DBP, 15 mm Hg for PP, and 11 mm Hg for MAP). Single BP indexes were included in multivariable adjusted regression models, separately. Also, combinations of two BP indexes were included in multivariable adjusted regression models simultaneously (i.e., SBP and DBP, SBP and PP, SBP and MAP, and DBP and PP, DBP and MAP, PP and MAP). To determine the consistency of the relationships detected in the multivariable adjusted models, the HR for ESRD associated with SBP, modeled as a continuous variable, was calculated in models stratified by age (<65 and ≥65 years), sex, race, diabetes, antihypertensive medication use, eGFR (30 to <60 and <30 ml/min/1.73 m2), and level of albuminuria (<300 and ≥300 mg/g). Multiplicative interaction was tested between SBP and age, sex, race, diabetes, antihypertensive medication use, level of ACR, and eGFR. The multivariable analysis did not include adjustment for albuminuria because it may be an intermediate in the pathway between SBP and ESRD. However, to determine the independent association of SBP on ESRD, we adjusted for albuminuria (<30, 30 to <300 and ≥ 300 mg/g) in a sensitivity analysis. All analyses were conducted using SAS 9.2 (SAS Institute, Cary, NC).
Results
Baseline characteristics of the 2,772 study participants with an eGFR <60 ml/min/1.73m2 are presented in Table 1. Overall, 2,542 (91.7%) participants had an eGFR of 30 to <60 and 230 (8.3%) participants had an eGFR <30 ml/min/1.73 m2. The mean ± standard deviation age of REGARDS study participants included in this analysis was 72.1 ± 8.7 years, 41.2% were African-American and 46.4% were men. The mean eGFR was 47.3 ± 10.7 ml/min/1.73 m2. The mean SBP was 130.7 ± 17.8 mm Hg, the mean DBP was 74.7 ± 10.5 mm Hg, the mean PP was 56.0 ± 15.0 mm Hg and the mean MAP was 93.3 ± 11.3 mm Hg.
Table 1.
Baseline characteristics of REGARDS study participants with an estimated glomerular filtration rate <60 ml/min/1.73m2
| Characteristic | Mean (standard deviation)a or percentage (n = 2,772) |
|---|---|
| Age (years) | 72.1 (8.7) |
| African American (%) | 41.2 |
| Men (%) | 46.4 |
| Geographic region of residence (%) | |
| Stroke belt | 32.9 |
| Stroke buckle | 21.0 |
| Other | 46.1 |
| Current smoking (%) | 10.5 |
| Physically inactive (%) | 54.7 |
| Household income <$20,000 per year (%) | 24.2 |
| Waist circumference (cm) | 98.6 (15.4) |
| HDL-cholesterol (mg/dl) | 48.5 (16.0) |
| High-sensitivity C-reactive protein (mg/l)a | 2.8 (1.2–6.4) |
| History of cardiovascular disease (%) | 32.1 |
| History of diabetes (%) | 33.9 |
| Albumin-to-creatinine ratio (mg/g)a | 14.3 (6.2–63.6) |
| Estimated GFR (ml/min/1.73m2) | 47.3 (10.7) |
| Antihypertensive medication use | 79.8 |
| Systolic blood pressure (mm Hg) | 130.7 (17.8) |
| Diastolic blood pressure (mm Hg) | 74.7 (10.5) |
| Pulse pressure (mm Hg) | 56.0 (15.0) |
| Mean arterial pressure (mm Hg) | 93.3 (11.3) |
HDL, high-density lipoprotein; GFR, glomerular filtration rate; REGARDS, REasons for the Geographic and Racial Differences in Stroke.
Median (25th–75th percentiles) are presented for C-reactive protein and albumin-tocreatinine ratio.
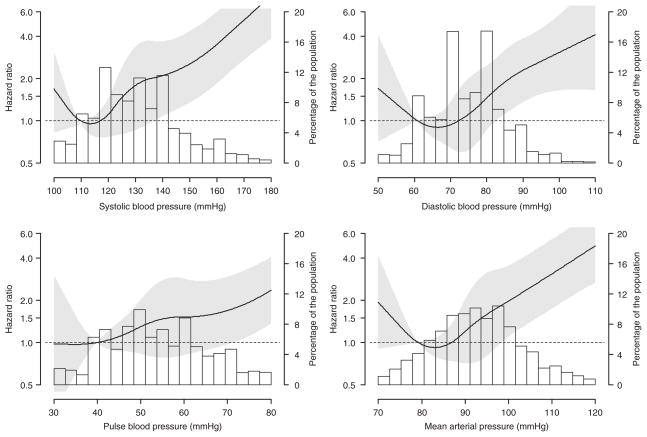
Over a median follow-up of 4 years, 138 participants developed ESRD. The incidence rate of ESRD was increased at higher levels of SBP, DBP, PP, and MAP (Table 2). These associations remained present after adjustment for age, race, sex, and geographic region of residence and after further adjustment for current smoking, physical activity, income, waist circumference, high sensitivity C-reactive protein, history of cardiovascular disease, diabetes, HDL-cholesterol, antihypertensive medication use, and eGFR. Higher levels of each BP index were associated with increased HRs when modeled as continuous variables using restricted quadratic splines (Figure 1).
Table 2.
Hazard ratios for incident ESRD associated with systolic blood pressure, diastolic blood pressure, pulse pressure and mean arterial pressure in REGARDS study participants with an estimated glomerular filtration rate <60 ml/min/1.73 m2
| ESRD | Hazard ratio (95% CI) for ESRD | ||||
|---|---|---|---|---|---|
| N (at risk) | Cases (n; %) | Incidence rate | Adjusted model 1a | Adjusted model 2b | |
| SBP (mm Hg) | |||||
| <120 | 707 | 17 | 6.1 | 1.0 (ref) | 1.0 (ref) |
| 120–129 | 670 | 22 | 8.3 | 1.27 (0.68–2.40) | 1.19 (0.63–2.26) |
| 130–139 | 631 | 28 | 10.8 | 1.67 (0.91–3.06) | 1.57 (0.84–2.93) |
| ≥140 | 764 | 71 | 23.3 | 3.39 (1.99–5.77) | 2.32 (1.33–4.03) |
| DBP (mm Hg) | |||||
| <70 | 829 | 28 | 8.7 | 1.0 (ref) | 1.0 (ref) |
| 70–79 | 1018 | 40 | 9.8 | 0.97 (0.60–1.58) | 1.19 (0.73–1.96) |
| 80–89 | 718 | 44 | 14.9 | 1.26 (0.78–2.04) | 1.90 (1.15–3.14) |
| ≥90 | 207 | 26 | 33.3 | 2.30 (1.33–3.98) | 2.81 (1.58–4.98) |
| PP (mm Hg) | |||||
| <45 | 626 | 17 | 6.9 | 1.0 (ref) | 1.0 (ref) |
| 45–54 | 792 | 30 | 9.1 | 1.43 (0.79–2.60) | 0.99 (0.54–1.81) |
| 55–64 | 665 | 35 | 13.2 | 2.15 (1.20–3.85) | 1.54 (0.85–2.79) |
| ≥65 | 689 | 56 | 20.4 | 3.69 (2.12–6.42) | 1.74 (0.99–3.07) |
| MAP (mm Hg) | |||||
| <86 | 684 | 19 | 7.1 | 1.0 (ref) | 1.0 (ref) |
| 86–93 | 685 | 21 | 7.8 | 1.01 (0.54–1.88) | 1.18 (0.62–2.22) |
| 93–100 | 734 | 29 | 9.5 | 1.08 (0.60–1.93) | 1.38 (0.76–2.49) |
| ≥100 | 669 | 69 | 25.8 | 2.72 (1.63–4.55) | 2.92 (1.71–4.97) |
Incidence rates are per 10,000 person years.
CI, confidence interval; DBP, diastolic blood pressure; ESRD, end-stage renal disease; HDL, high-density lipoprotein; MAP, mean arterial pressure; PP, pulse pressure; REGARDS, REasons for the Geographic and Racial Differences in Stroke; SBP, systolic blood pressure.
Model 1, adjusted for age, race, sex, and geographic region of residence.
Model 2, adjusted for age, race, sex, geographic region of residence, current smoking, physical activity, income, waist circumference, high-sensitivity C-reactive protein, history of cardiovascular disease, diabetes, HDL-cholesterol, antihypertensive medication use, and estimated glomerular filtration rate.
Figure 1.
Multivariable adjusted hazard ratios for end-stage renal disease associated with systolic blood pressure, diastolic blood pressure, pulse pressure, and mean arterial pressure. Black line represents the hazard ratio, gray area is the 95% confidence interval. Bars represent the percentage of the population. Hazard ratios are adjusted for age, race, sex, geographic region of residence, current smoking, physical activity, income, waist circumference, highsensitivity C-reactive protein, history of cardiovascular disease, diabetes, high-density lipoprotein-cholesterol, antihypertensive medication use, and estimated glomerular filtration rate.
After full multivariable adjustment, each standard deviation higher SBP was associated with a HR for ESRD of 1.67 (95% confidence intervals (CI): 1.43–1.96; Table 3). The analogous HRs for a one standard deviation higher DBP, PP, and MAP were 1.38 (95% CI 1.16–1.63), 1.50 (95% CI: 1.27–1.78), and 1.54 (95% CI 1.32–1.79), respectively. In models that included SBP and DBP, SBP and PP, and SBP and MAP simultaneously, SBP but not the other BP indexes was significantly associated with an increased HR for ESRD. Data from models including DBP and PP, DBP and MAP, and PP and MAP, simultaneously, are shown in Supplementary Table S1 online.
Table 3.
Hazard ratios for incident ESRD associated with one standard deviation higher systolic blood pressure, diastolic blood pressure, pulse pressure and mean arterial pressure in REGARDS study participants with an estimated glomerular filtration rate <60 ml/min/1.73 m2
| Blood pressure component | Model 1a | Model 2b |
|---|---|---|
| Single components | Hazard ratio (95% CI) | Hazard ratio (95% CI) |
| SBP (18 mm Hg) | 1.70 (1.48–1.96) | 1.67 (1.43–1.96) |
| DBP (11 mm Hg) | 1.26 (1.07–1.49) | 1.38 (1.16–1.63) |
| PP (15 mm Hg) | 1.67 (1.45–1.94) | 1.50 (1.27–1.78) |
| MAP (11 mm Hg) | 1.47 (1.28–1.69) | 1.54 (1.32–1.79) |
| Two components | Hazard ratio (95% CI) | Hazard ratio (95% CI) |
| SBP and DBP | ||
| SBP (18 mm Hg) | 1.86 (1.56–2.21) | 1.65 (1.35–2.01) |
| DBP (11 mm Hg) | 0.85 (0.70–1.04) | 1.03 (0.84–1.26) |
| SBP and PP | ||
| SBP (18 mm Hg) | 1.43 (1.11–1.85) | 1.73 (1.32–2.26) |
| PP (15 mm Hg) | 1.24 (0.95–1.62) | 0.96 (0.73–1.27) |
| SBP and MAP | ||
| SBP (18 mm Hg) | 2.12 (1.57–2.85) | 1.61 (1.16–2.23) |
| MAP (11 mm Hg) | 0.79 (0.59–1.05) | 1.04 (0.77–1.42) |
ESRD, end-stage renal disease; DBP, diastolic blood pressure; HDL, high-density lipoprotein; MAP, mean arterial pressure; PP, pulse pressure; REGARDS, REasons for the Geographic and Racial Differences in Stroke; SBP, systolic blood pressure.
Model 1, adjusted for age, race, sex, and geographic region of residence.
Model 2, adjusted for age, race, sex, geographic region of residence, current smoking, physical activity, income, waist circumference, high-sensitivity C-reactive protein, history of cardiovascular disease, diabetes, HDL-cholesterol, antihypertensive medication use and estimated glomerular filtration rate.
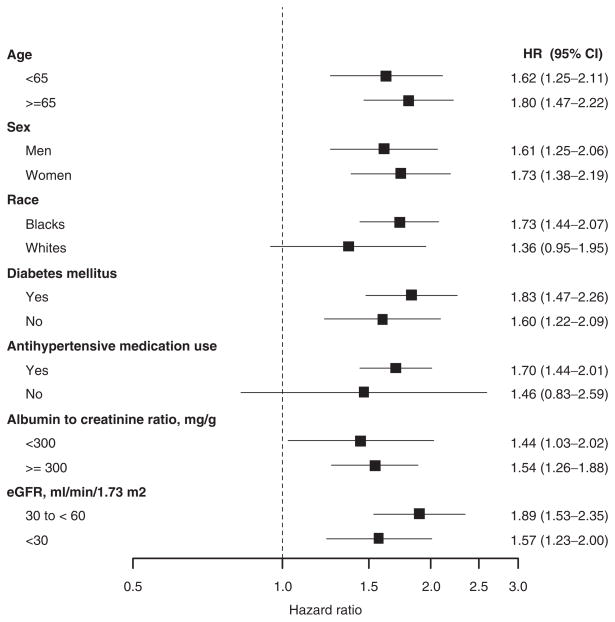
After multivariable adjustment, higher SBP was associated with an increased multivariable adjusted HR for ESRD for individuals <65 and ≥65 years of age, men and women, African Americans, individuals with and without diabetes, taking antihypertensive medication, with an ACR <300 mg/g and ACR ≥300 mg/g, and with an eGFR of 30 to <60 ml/min/1.73 m2 and an eGFR <30 ml/min/1.73 m2 (Figure 2). Although increased HRs for ESRD were present for whites and individuals not taking antihypertensive medications, the 95% CI in these subgroups included one. Multiplicative interaction was not present between SBP and age, sex, race, diabetes, antihypertensive medication use, level of ACR, or eGFR (each P-interaction > 0.1).
Figure 2.
Hazard ratios for developing end-stage renal disease associated with one standard deviation higher systolic blood pressure (18 mm Hg) in subgroups of REasons for the Geographic and Racial Differences in Stroke (REGARDS) study participants with an estimated glomerular filtration rate (eGFR) <60 ml/min/1.73 m2. Hazard ratios (HRs) are adjusted for age, race, sex, geographic region of residence, current smoking, physical activity, income, waist circumference, high-sensitivity C-reactive protein, history of cardiovascular disease, diabetes, high-density lipoprotein-cholesterol, antihypertensive medication use, and estimated glomerular filtration rate.
Sensitivity analysis
After adding albuminuria to the full multivariable adjusted model, each standard deviation higher SBP (18 mm Hg) was associated with a HR for ESRD of 1.44 (95% CI: 1.21–1.70). After further adjustment for DBP, PP, or MAP, each in separate models, the HR for ESRD associated with one standard deviation higher SBP was 1.42 (95% CI: 1.16–1.74), 1.47 (95% CI: 1.11–1.96), and 1.39 (95% CI: 1.00–1.94), respectively.
Discussion
In the present study, a strong and graded association between SBP and risk for ESRD was present among individuals with an eGFR <60 ml/min/1.73m2. This association remained present after multivariable adjustment including antihypertensive medication use as well as DBP, PP, or MAP. Higher levels of DBP, PP, and MAP were associated with increased HRs for ESRD in multivariable adjusted models. However, these associations were attenuated and no longer statistically significant after adjustment for SBP.
Hypertension is very common among individuals with CKD. The prevalence of hypertension was 86% among individuals with an eGFR between 20 and 70 ml/min/1.73 m2 in the Chronic Renal Insufficiency Cohort study.19 In Chronic Renal Insufficiency Cohort study, 41% of participants had a SBP ≥140 mm Hg compared with only 9% with DBP ≥90 mm Hg. In addition, among 3,213 participants of the National Health and Nutrition Survey 1999–2004 with eGFR <60 ml/min/1.73 m2 or an albumin-to-creatinine ratio ≥30 mg/g, Peralta et al.20 identified a high rate of uncontrolled BP, primarily as a result of elevated SBP levels.
Elevated SBP has been associated with an increased risk of incident ESRD in previous studies. For example, Klag et al.21 found a strong, graded and continuous association between SBP and ESRD risk over an average of 16 years among 332,544 men ages 35–57 years screened for the Multiple Risk Factor Intervention Trial. However, serum creatinine was not available in individuals screened for Multiple Risk Factor Intervention Trial and it is unclear whether those who developed ESRD had CKD at baseline. Hsu et al.22 observed a graded and continuous association between SBP and ESRD risk among a cohort of 316,675 members of Kaiser Permanente of Northern California, a health care delivery system, with eGFR ≥60 ml/min/1.73 m2 who participated in health check-ups from 1964 to 1985. The association between SBP and ESRD risk among individuals with an eGFR <60 ml/min/1.73 m2 was not reported. Agarwal et al.23 identified an association between SBP and risk for ESRD among 220 veterans with reduced renal function followed over 6 years. This study extends these prior results to a contemporary cohort and highlights the importance of SBP on ESRD risk among a population-based sample of individuals with CKD.
Previous studies have investigated the association between different BP indexes, with renal function decline and incident ESRD.24–27 Young et al.28 found that SBP maintained the strongest association with a decline in kidney function among 2,181 men and women over age 65 with isolated systolic hypertension participating in the placebo arm of the Systolic Hypertension in the Elderly Program. Reynolds et al.29 compared the association of SBP, DBP, and PP with the risk of ESRD in a study of 158,365 Chinese men and women ≥40 years. Similar to our findings, SBP remained a risk factor for ESRD after adjustment for DBP and PP. However, information on baseline kidney disease status, eGFR or albuminuria, was not available.
In a secondary analysis of the Reduction of Endpoint in NIDDM with Angiotensin II Antagonist Losartan (RENAAL) study, Bakris and colleagues investigated the association between SBP, DBP and PP, with renal function decline, incident ESRD and composite outcome of ESRD and death.30 In this study of patients with type 2 diabetes associated nephropathy, SBP and PP had a strong and graded association with renal function decline and were better predictors for each outcome than DBP. After adjustment for SBP, PP remained associated with the composite outcome of ESRD and death while SBP was not associated with this outcome after adjustment for PP. In this study, PP was not associated with ESRD risk after adjustment for SBP. The discrepant findings may be a result of several differences between the populations enrolled in Reduction of Endpoint in NIDDM with Angiotensin II Antagonist Losartan study and REGARDS. The Reduction of Endpoint in NIDDM with Angiotensin II Antagonist Losartan study population was younger, limited to patients with diabetes and albuminuria, and had higher mean SBP, DBP, and PP levels. The REGARDS study includes a population-based sample of individuals with reduced eGFR and may better reflect the association between BP indexes and ESRD risk in the general population.
A few randomized clinical trials have compared different BP targets on the progression of renal disease in individuals with CKD. Wright et al.31 found participants randomized to MAP goals of 102 to 107 or to 92 mm Hg or less had similar rates of renal function decline among 1094 African Americans with hypertensive renal disease and eGFR levels between 20 and 65 ml/min/1.73 m2 in the African-American Study of Kidney Disease trial. Among 850 participants in the Modification of Diet in Renal Disease study, the rate of decline in renal function was similar among participants randomized to a MAP <92 mm Hg vs. <107 mm Hg.32 The National Institutes of Health recently launched the Systolic Blood Pressure Intervention Trial which will compare the benefits of a SBP target <120 mm Hg (intervention) vs. <140 mm Hg (usual care) on outcomes including renal disease progression and ESRD incidence. As designed, approximately 30% of Systolic Blood Pressure Intervention Trial participants will have an eGFR <60 ml/min/1.73 m2. Although the present study identified a greater than threefold higher ESRD risk associated with SBP ≥140 mm Hg vs. <120 mm Hg, Systolic Blood Pressure Intervention Trial will provide important data on the benefits of lowering SBP to <120 mm Hg on outcomes among individuals with reduced eGFR.
The results of the present study need to be considered in the context of potential and known limitations. Serum creatinine was only available from a single baseline visit and we were unable to evaluate the association between BP indexes and the progression of CKD. BP was measured two times on a single occasion. Also, the present study is observational and the causality of the relationships detected could not be determined. Another limitation is a relatively low number of ESRD cases. Therefore, some results in subgroup analysis were not statistically significant. This study also has several strengths. These include its recruitment of a population-based sample from across the United States with oversampling of African-Americans, a population with a high burden of ESRD. The REGARDS study included a stringent quality control process, standardized criteria and protocols for assessing BP and eGFR which should limit potential bias. In addition, ESRD cases were identified through linkage with the USRDS which has been shown to maintain high-sensitivity and specificity.33 In the present study of individuals with reduced eGFR, SBP maintained a strong, graded association with risk for ESRD. This association was independent of DBP, PP, or MAP and use of antihypertensive medication. In contrast, DBP, PP, and MAP were not significantly associated with ESRD after adjustment for SBP. These data support further studies of interventional strategies that focus on the control of elevated SBP to reduce ESRD incidence among individuals with reduced eGFR.
Supplementary Material
Acknowledgments
This research project is supported by a cooperative agreement U01 NS041588 from the National Institute of Neurological Disorders and Stroke, National Institutes of Health, Department of Health and Human Service. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Neurological Disorders and Stroke or the National Institutes of Health. Representatives of the funding agency have been involved in the review of the manuscript but not directly involved in the collection, management, analysis or interpretation of the data. The authors thank the other investigators, the staff, and the participants of the REGARDS study for their valuable contributions. A full list of participating REGARDS investigators and institutions can be found at http://www.regardsstudy.org. Additional funding was provided by an investigator-initiated grant-in-aid from Amgen Corporation to D.G.W. Amgen did not have any role in the design and conduct of the study, the collection, management, data analysis, or interpretation of the data, or the preparation or approval of the manuscript L.G. had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Amgen did not have any role in the design and conduct of the study, the collection, management, data analysis, or interpretation of the data, or the preparation or approval of the manuscript.
Footnotes
Supplementary material is linked to the online version of the paper at http://www.nature.com/ajh
Disclosure: The authors declared no conflict of interest.
References
- 1.Coresh J, Selvin E, Stevens LA, Manzi J, Kusek JW, Eggers P, Van Lente F, Levey AS. Prevalence of chronic kidney disease in the United States. JAMA. 2007;298:2038–2047. doi: 10.1001/jama.298.17.2038. [DOI] [PubMed] [Google Scholar]
- 2.Meguid El Nahas A, Bello AK. Chronic kidney disease: the global challenge. Lancet. 2005;365:331–340. doi: 10.1016/S0140-6736(05)17789-7. [DOI] [PubMed] [Google Scholar]
- 3.Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med. 2004;351:1296–1305. doi: 10.1056/NEJMoa041031. [DOI] [PubMed] [Google Scholar]
- 4.Hallan SI, Coresh J, Astor BC, Asberg A, Powe NR, Romundstad S, Hallan HA, Lydersen S, Holmen J. International comparison of the relationship of chronic kidney disease prevalence and ESRD risk. J Am Soc Nephrol. 2006;17:2275–2284. doi: 10.1681/ASN.2005121273. [DOI] [PubMed] [Google Scholar]
- 5.Muntner P, Anderson A, Charleston J, Chen Z, Ford V, Makos G, O’Connor A, Perumal K, Rahman M, Steigerwalt S, Teal V, Townsend R, Weir M, Wright JT, Jr Chronic Renal Insufficiency Cohort (CRIC) Study Investigators. Hypertension awareness, treatment, and control in adults with CKD: results from the Chronic Renal Insufficiency Cohort (CRIC) Study. Am J Kidney Dis. 2010;55:441–451. doi: 10.1053/j.ajkd.2009.09.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Peralta CA, Hicks LS, Chertow GM, Ayanian JZ, Vittinghoff E, Lin F, Shlipak MG. Control of hypertension in adults with chronic kidney disease in the United States. Hypertension. 2005;45:1119–1124. doi: 10.1161/01.HYP.0000164577.81087.70. [DOI] [PubMed] [Google Scholar]
- 7.Coresh J, Wei GL, McQuillan G, Brancati FL, Levey AS, Jones C, Klag MJ. Prevalence of high blood pressure and elevated serum creatinine level in the United States: findings from the third National Health and Nutrition Examination Survey (1988–1994) Arch Intern Med. 2001;161:1207–1216. doi: 10.1001/archinte.161.9.1207. [DOI] [PubMed] [Google Scholar]
- 8.Andersen MJ, Khawandi W, Agarwal R. Home blood pressure monitoring in CKD. Am J Kidney Dis. 2005;45:994–1001. doi: 10.1053/j.ajkd.2005.02.015. [DOI] [PubMed] [Google Scholar]
- 9.Kannel WB. Hypertension: reflections on risks and prognostication. Med Clin North Am. 2009;93:541–58. doi: 10.1016/j.mcna.2009.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Khosla N, Kalaitzidis R, Bakris GL. The kidney, hypertension, and remaining challenges. Med Clin North Am. 2009;93:697–715. doi: 10.1016/j.mcna.2009.02.001. [DOI] [PubMed] [Google Scholar]
- 11.Reynolds K, Gu D, Muntner P, Kusek JW, Chen J, Wu X, Duan X, Chen CS, Klag MJ, Whelton PK, He J. A population-based, prospective study of blood pressure and risk for end-stage renal disease in China. J Am Soc Nephrol. 2007;18:1928–1935. doi: 10.1681/ASN.2006111199. [DOI] [PubMed] [Google Scholar]
- 12.Bakris GL, Weir MR, Shanifar S, Zhang Z, Douglas J, van Dijk DJ, Brenner BM RENAAL Study Group. Effects of blood pressure level on progression of diabetic nephropathy: results from the RENAAL study. Arch Intern Med. 2003;163:1555–1565. doi: 10.1001/archinte.163.13.1555. [DOI] [PubMed] [Google Scholar]
- 13.Agarwal R. Blood pressure components and the risk for end-stage renal disease and death in chronic kidney disease. Clin J Am Soc Nephrol. 2009;4:830–837. doi: 10.2215/CJN.06201208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Howard VJ, Cushman M, Pulley L, Gomez CR, Go RC, Prineas RJ, Graham A, Moy CS, Howard G. The reasons for geographic and racial differences in stroke study: objectives and design. Neuroepidemiology. 2005;25:135–143. doi: 10.1159/000086678. [DOI] [PubMed] [Google Scholar]
- 15.Kurella Tamura M, Wadley V, Yaffe K, McClure LA, Howard G, Go R, Allman RM, Warnock DG, McClellan W. Kidney function and cognitive impairment in US adults: the Reasons for Geographic and Racial Differences in Stroke (REGARDS) Study. Am J Kidney Dis. 2008;52:227–234. doi: 10.1053/j.ajkd.2008.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF, 3rd, Feldman HI, Kusek JW, Eggers P, Van Lente F, Greene T, Coresh J CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration) A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150:604–612. doi: 10.7326/0003-4819-150-9-200905050-00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.KDOQI Working Group. Definition and classification of stages of chronic kidney disease. Am J Kidney Dis. 2002;39:S46–S75. [Google Scholar]
- 18.Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL, Jr, Jones DW, Materson BJ, Oparil S, Wright JT, Jr, Roccella EJ Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. National Heart, Lung, and Blood Institute; National High Blood Pressure Education Program Coordinating Committee. Seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. Hypertension. 2003;42:1206–1252. doi: 10.1161/01.HYP.0000107251.49515.c2. [DOI] [PubMed] [Google Scholar]
- 19.Muntner P, Anderson A, Charleston J, Chen Z, Ford V, Makos G, O’Connor A, Perumal K, Rahman M, Steigerwalt S, Teal V, Townsend R, Weir M, Wright JT, Jr Chronic Renal Insufficiency Cohort (CRIC) Study Investigators. Hypertension awareness treatment and control in adults with CKD: results from the Chronic Renal Insufficiency Cohort (CRIC) Study. Am J Kidney Dis. 2010;55:441–451. doi: 10.1053/j.ajkd.2009.09.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Peralta CA, Hicks LS, Chertow GM, Avanian JZ, Vittinghoff E, Lin F, Shlipak MG. Control of hypertension in adults with chronic kidney disease in the United States. Hypertension. 2005;45:1119–1124. doi: 10.1161/01.HYP.0000164577.81087.70. [DOI] [PubMed] [Google Scholar]
- 21.Klag MJ, Whelton PK, Randall BL, Neaton JD, Brancati FL, Ford CE, Shulman NB, Stamler J. Blood pressure and end-stage renal disease in men. N Engl J Med. 1996;334:13–18. doi: 10.1056/NEJM199601043340103. [DOI] [PubMed] [Google Scholar]
- 22.Hsu CY, McCulloch CE, Darbinian J, Go AS, Iribarren C. Elevated blood pressure and risk of end-stage renal disease in subjects without baseline kidney disease. Arch Intern Med. 2005;165:923–928. doi: 10.1001/archinte.165.8.923. [DOI] [PubMed] [Google Scholar]
- 23.Agarwal R, Bunaye Z, Bekele DM, Light RP. Competing risk factor analysis of endstage renal disease and mortality in chronic kidney disease. Am J Nephrol. 2008;28:569–575. doi: 10.1159/000115291. [DOI] [PubMed] [Google Scholar]
- 24.MacMahon S, Peto R, Cutler J, Collins R, Sorlie P, Neaton J, Abbott R, Godwin J, Dyer A, Stamler J. Blood pressure, stroke, and coronary heart disease. Part 1, Prolonged differences in blood pressure: prospective observational studies corrected for the regression dilution bias. Lancet. 1990;335:765–774. doi: 10.1016/0140-6736(90)90878-9. [DOI] [PubMed] [Google Scholar]
- 25.Psaty BM, Furberg CD, Kuller LH, Cushman M, Savage PJ, Levine D, O’Leary DH, Bryan RN, Anderson M, Lumley T. Association between blood pressure level and the risk of myocardial infarction, stroke, and total mortality: the cardiovascular health study. Arch Intern Med. 2001;161:1183–1192. doi: 10.1001/archinte.161.9.1183. [DOI] [PubMed] [Google Scholar]
- 26.Kannel WB, Gordon T, Schwartz MJ. Systolic versus diastolic blood pressure and risk of coronary heart disease. The Framingham study. Am J Cardiol. 1971;27:335–346. doi: 10.1016/0002-9149(71)90428-0. [DOI] [PubMed] [Google Scholar]
- 27.Franklin SS, Larson MG, Khan SA, Wong ND, Leip EP, Kannel WB, Levy D. Does the relation of blood pressure to coronary heart disease risk change with aging? The Framingham Heart Study. Circulation. 2001;103:1245–1249. doi: 10.1161/01.cir.103.9.1245. [DOI] [PubMed] [Google Scholar]
- 28.Young JH, Klag MJ, Muntner P, Whyte JL, Pahor M, Coresh J. Blood pressure and decline in kidney function: findings from the Systolic Hypertension in the Elderly Program (SHEP) J Am Soc Nephrol. 2002;13:2776–2782. doi: 10.1097/01.asn.0000031805.09178.37. [DOI] [PubMed] [Google Scholar]
- 29.Reynolds K, Gu D, Muntner P, Kusek JW, Chen J, Wu X, Duan X, Chen CS, Klag MJ, Whelton PK, He J. A population-based, prospective study of blood pressure and risk for end-stage renal disease in China. J Am Soc Nephrol. 2007;18:1928–1935. doi: 10.1681/ASN.2006111199. [DOI] [PubMed] [Google Scholar]
- 30.Bakris GL, Weir MR, Shanifar S, Zhang Z, Douglas J, van Dijk DJ, Brenner BM RENAAL Study Group. Effects of blood pressure level on progression of diabetic nephropathy: results from the RENAAL study. Arch Int Med. 2003;163:1555–1565. doi: 10.1001/archinte.163.13.1555. [DOI] [PubMed] [Google Scholar]
- 31.Wright JT, Jr, Bakris G, Greene T, Agodoa LY, Appel LJ, Charleston J, Cheek D, Douglas-Baltimore JG, Gassman J, Glassock R, Hebert L, Jamerson K, Lewis J, Phillips RA, Toto RD, Middleton JP, Rostand SG African American Study of Kidney Disease and Hypertension Study Group. Effect of blood pressure lowering and antihypertensive drug class on progression of hypertensive kidney disease: results from the AASK trial. JAMA. 2002;288:2421–2431. doi: 10.1001/jama.288.19.2421. [DOI] [PubMed] [Google Scholar]
- 32.Klahr S, Levey AS, Beck GJ, Caggiula AW, Hunsicker L, Kusek JW, Striker G. The effects of dietary protein restriction and blood-pressure control on the progression of chronic renal disease. Modification of Diet in Renal Disease Study Group . N Engl J Med. 1994;330:877–884. doi: 10.1056/NEJM199403313301301. [DOI] [PubMed] [Google Scholar]
- 33.US Renal Data System. USRDS 2010 Annual Data Report: Atlas of End-Stage Renal Disease in the United States. National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Disease; Bethesda: 2010. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.